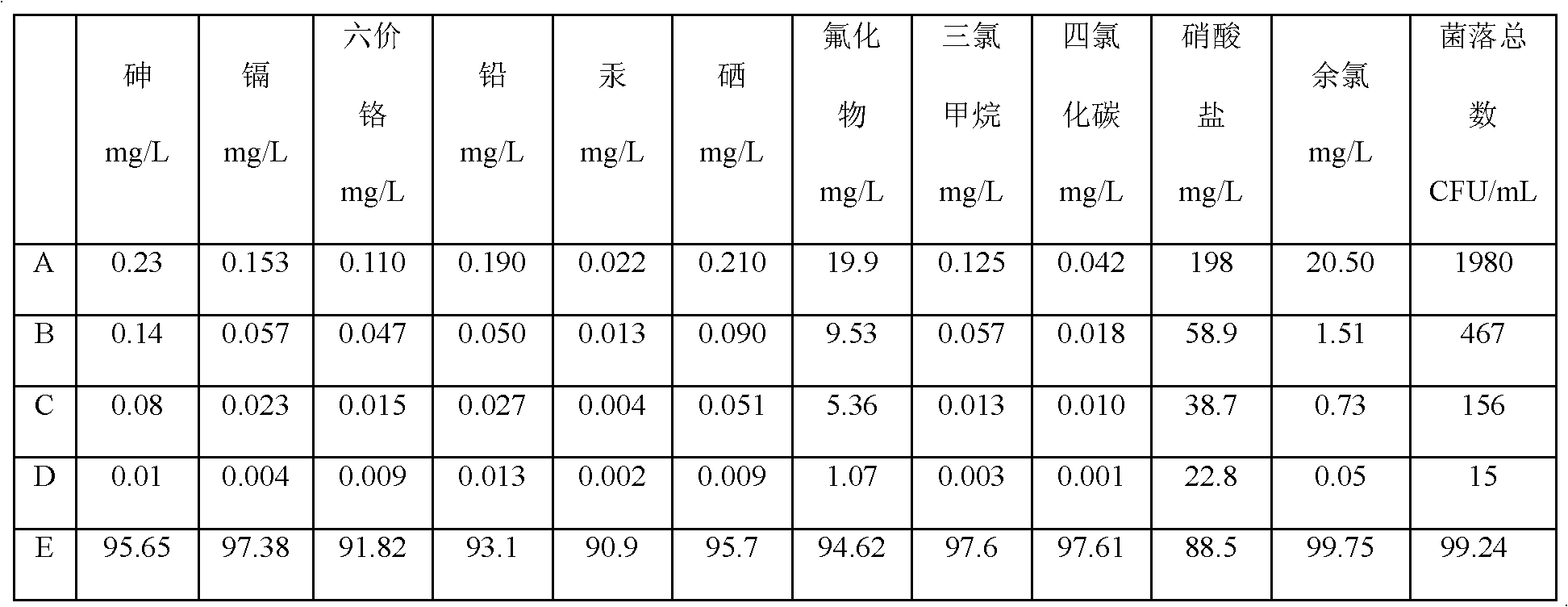
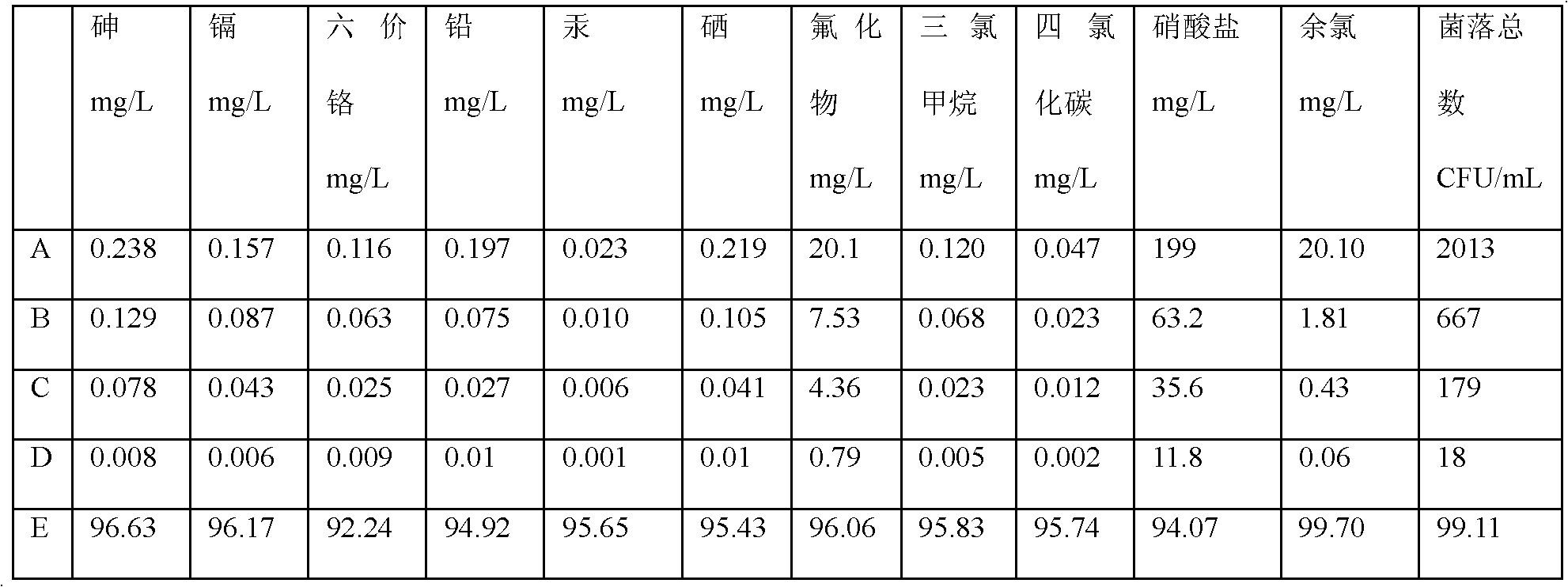
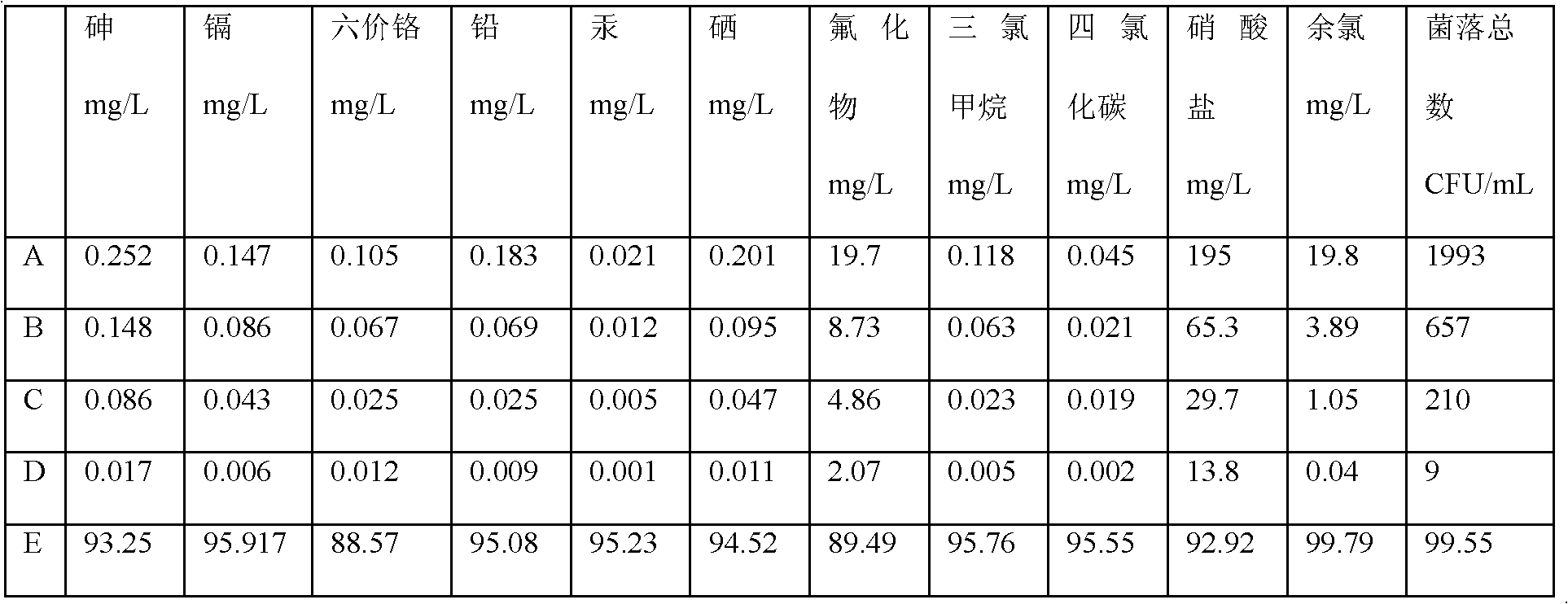
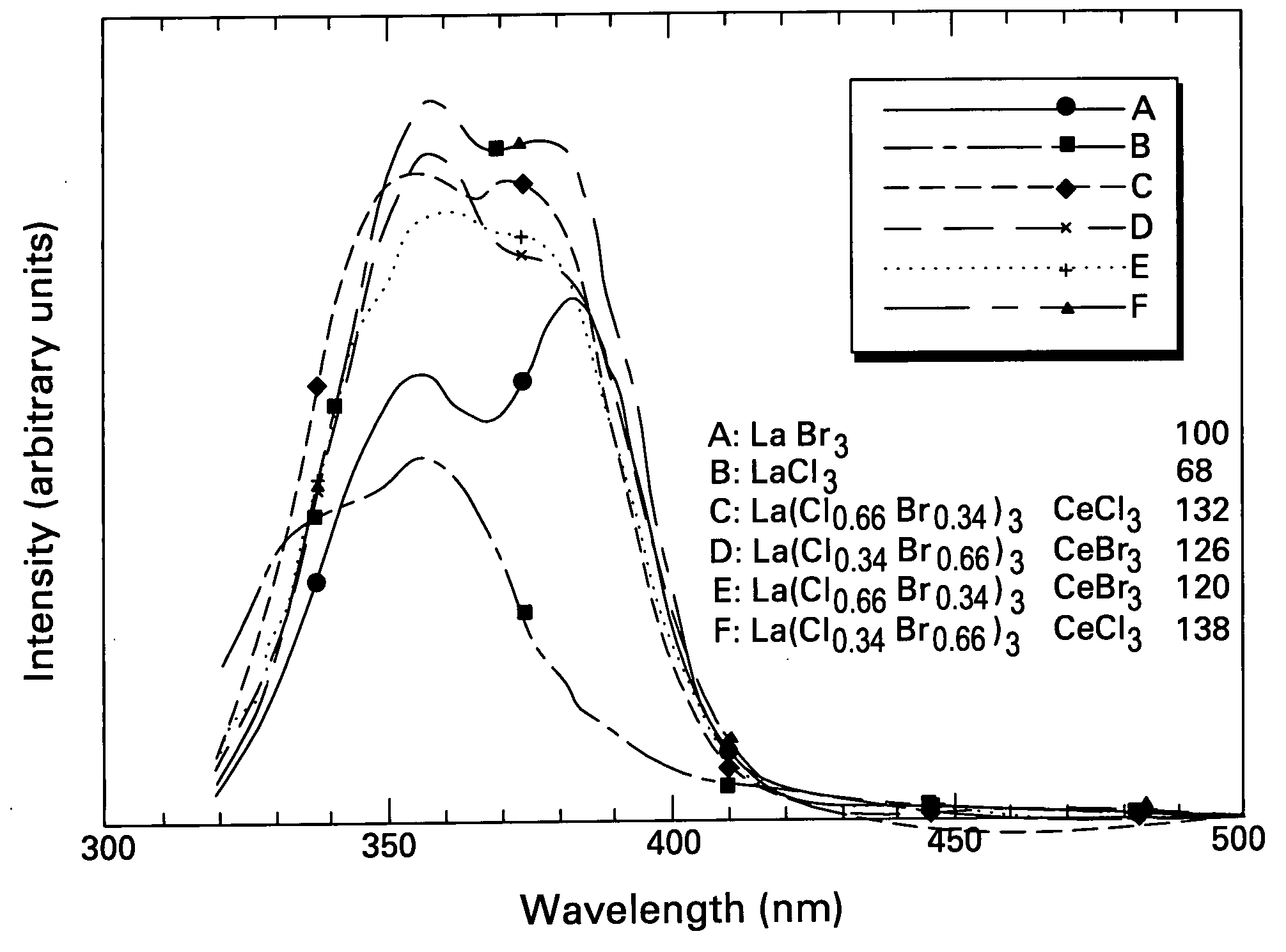
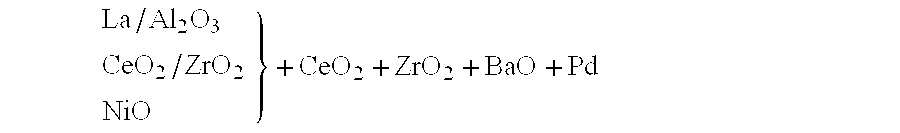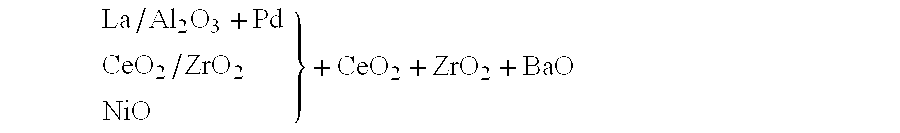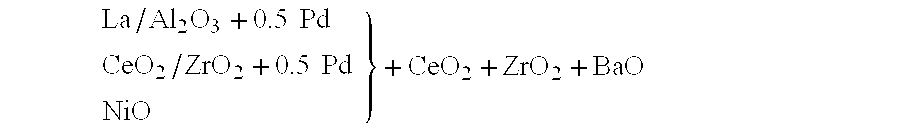Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
9621 results about "Cerium" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Cerium is a chemical element with the symbol Ce and atomic number 58. Cerium is a soft, ductile and silvery-white metal that tarnishes when exposed to air, and it is soft enough to be cut with a knife. Cerium is the second element in the lanthanide series, and while it often shows the +3 oxidation state characteristic of the series, it also exceptionally has a stable +4 state that does not oxidize water. It is also considered one of the rare-earth elements. Cerium has no biological role in humans and is not very toxic.
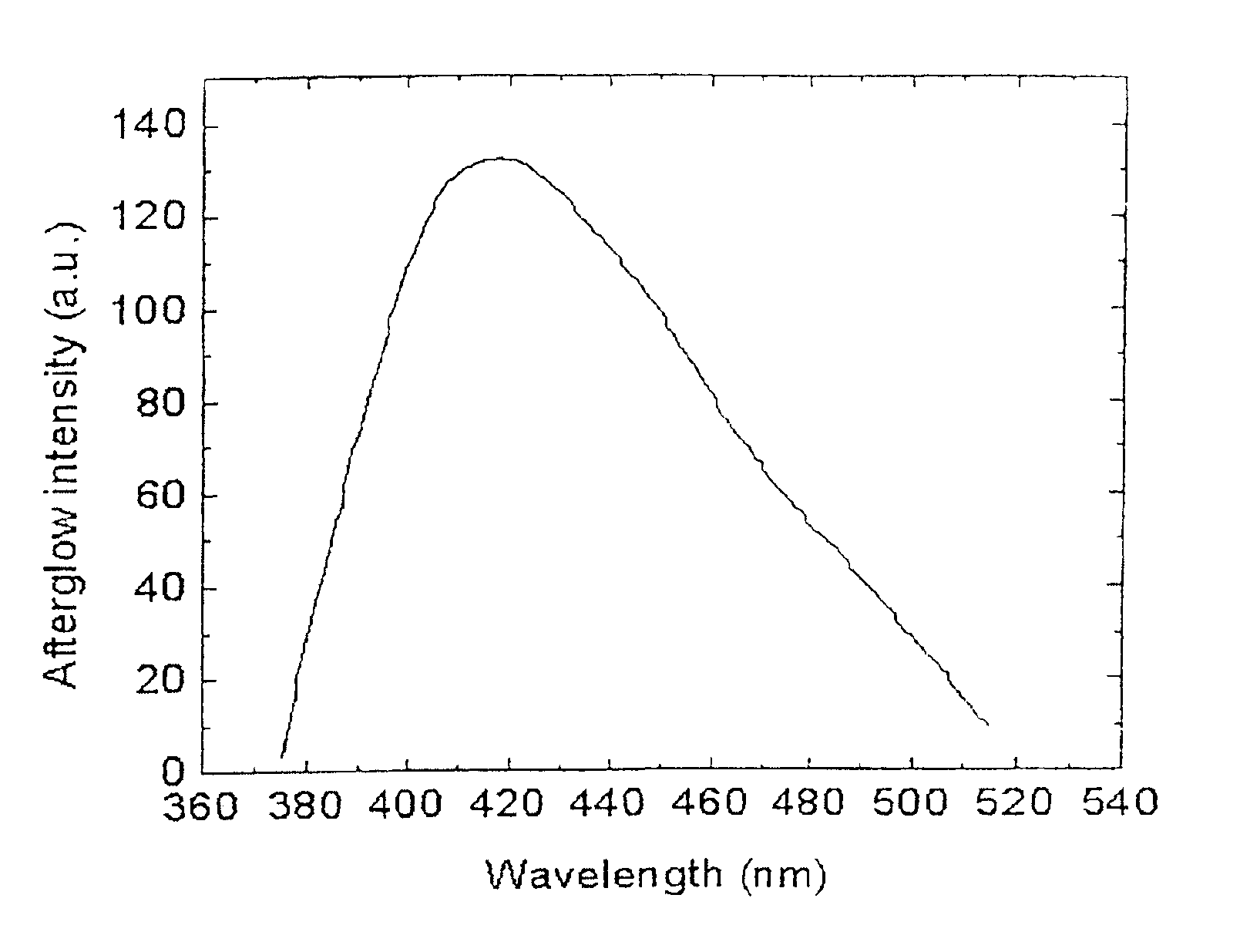
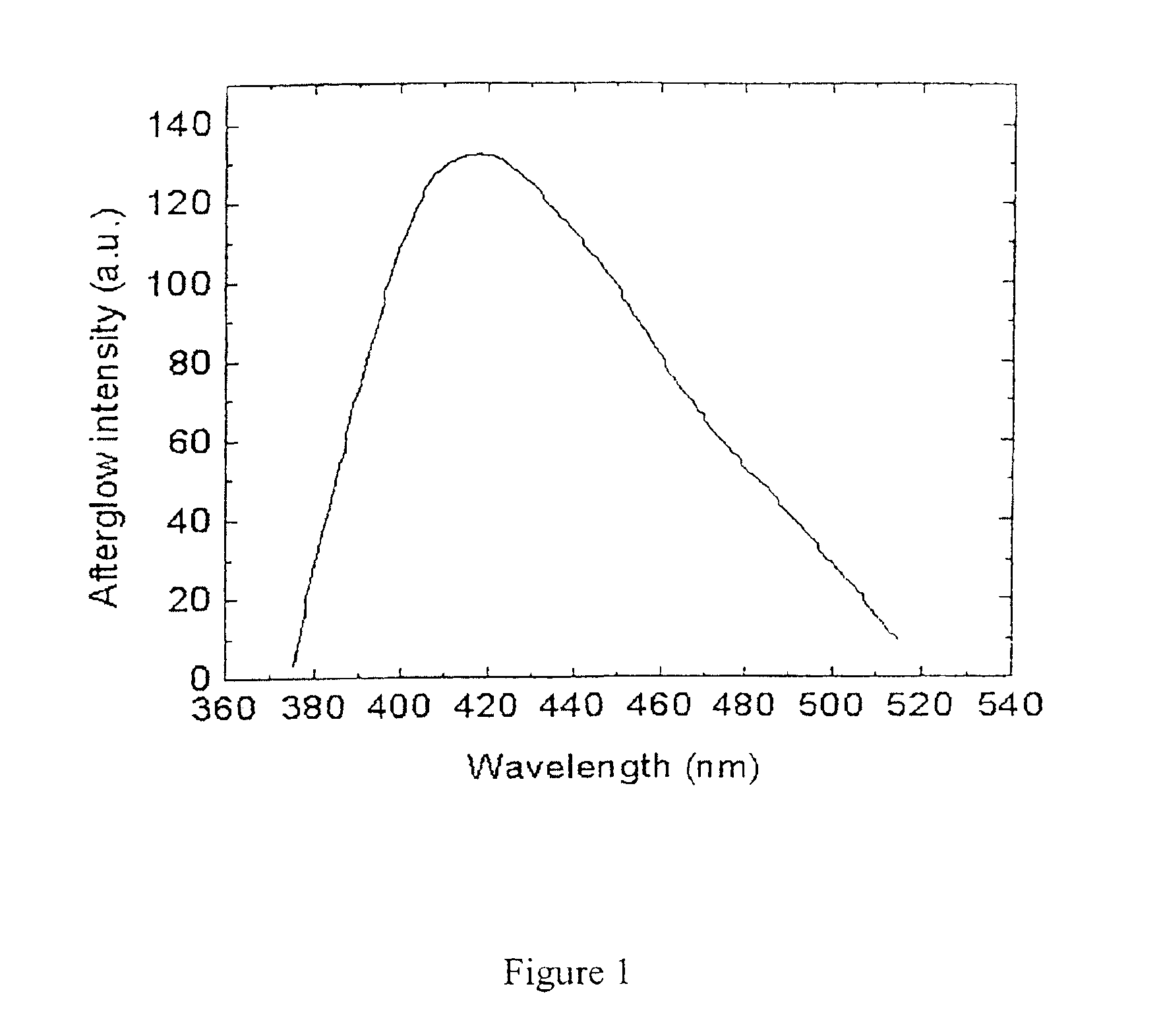
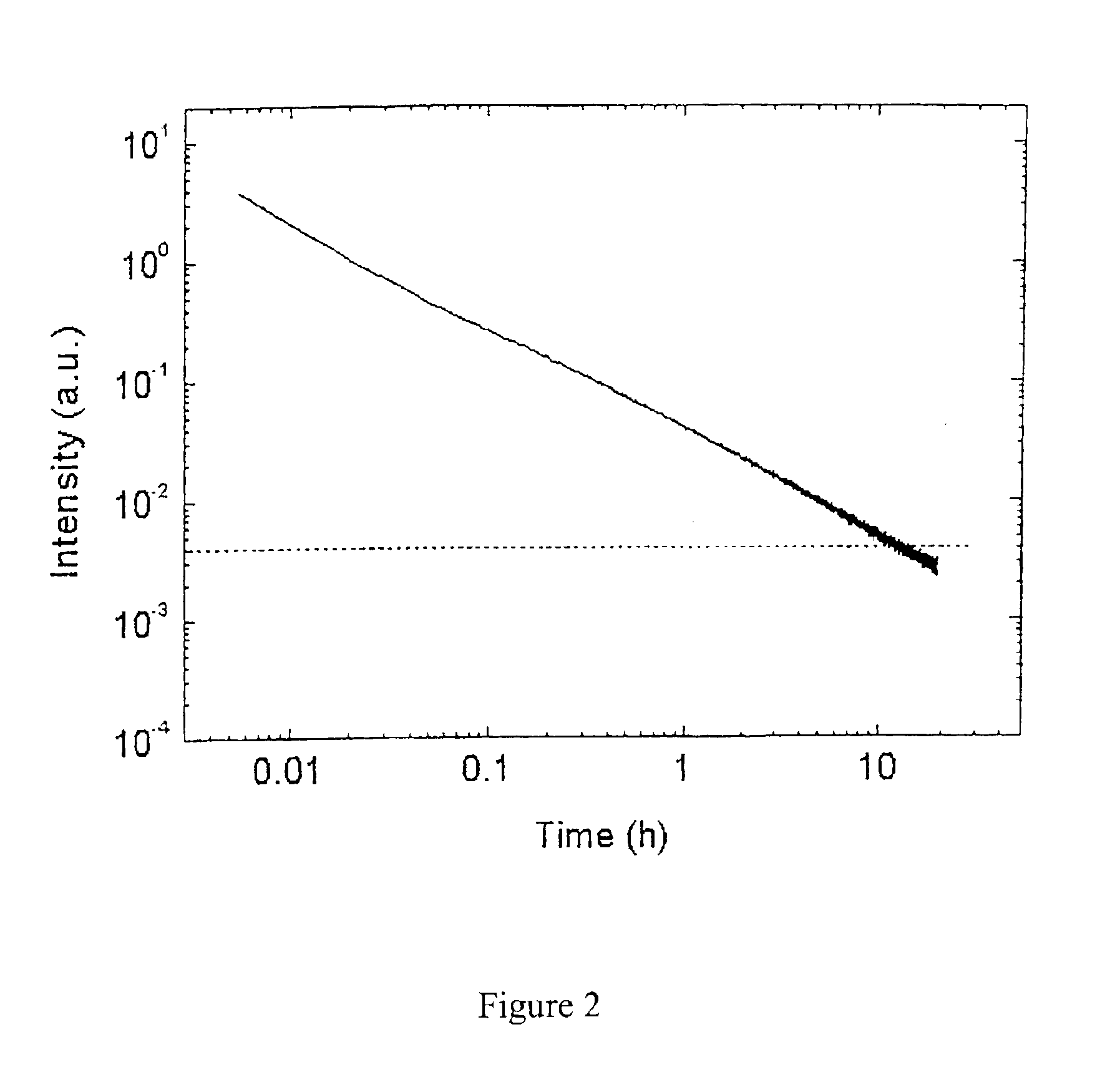
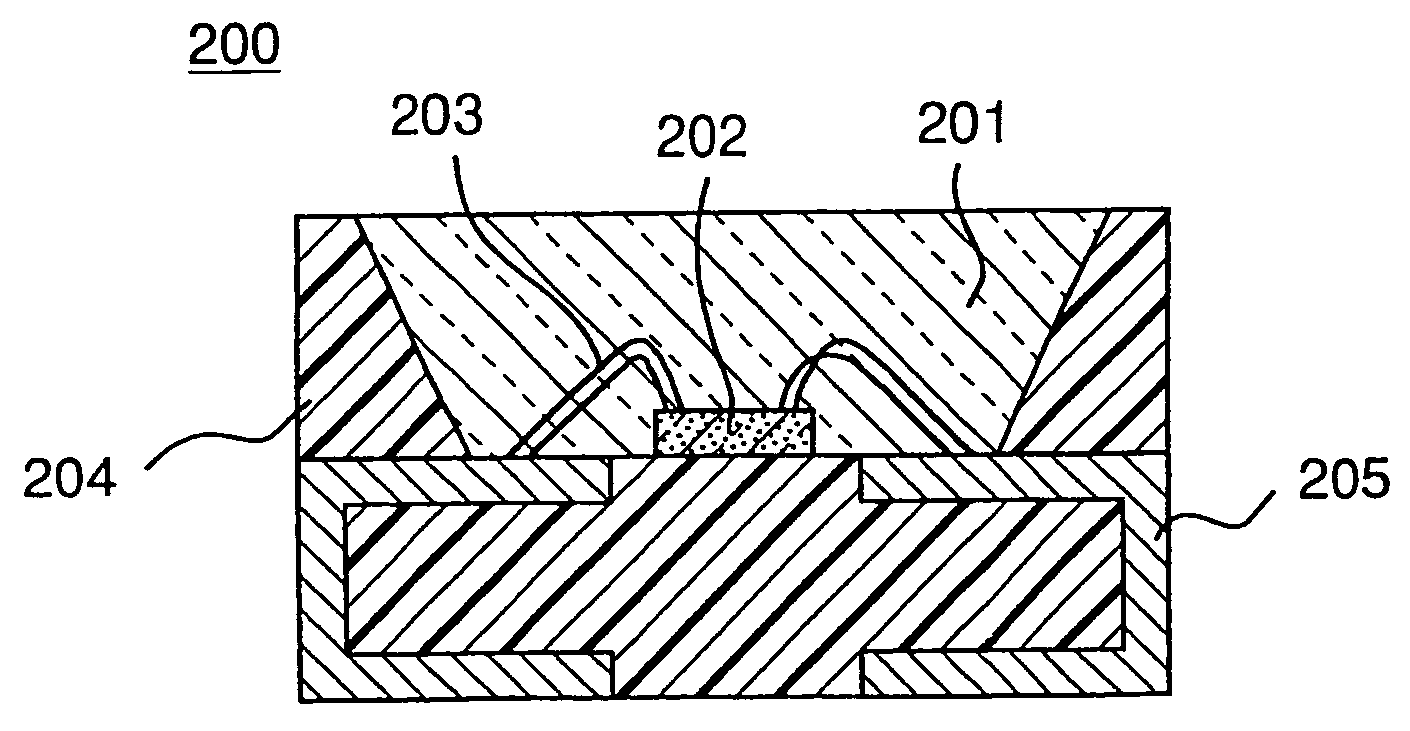
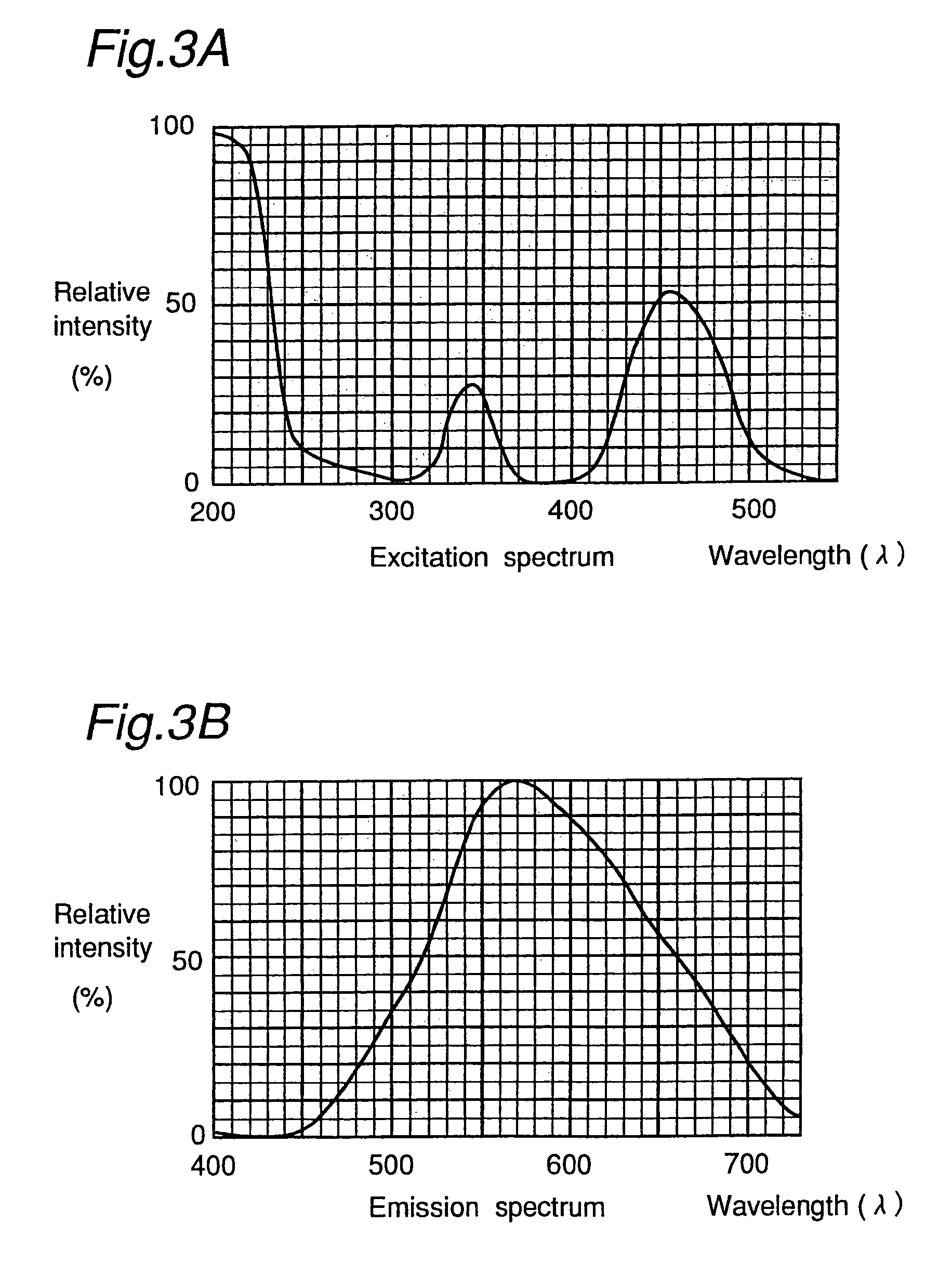
Long persistent phosphors and persistent energy transfer technique
The invention provides long-persistent phosphors, methods for their manufacture and phosphorescent articles. The invention also provides a method for generating a long-persistent phosphorescence at a selected color. The phosphors of the invention may be alkaline earth aluminates, alkaline earth silicates, and alkaline earth aluminosilicates. The phosphors include those activated by cerium. The phosphors also include those in which persistent energy transfer occurs from a donor ion to an acceptor ion, producing persistent emission largely characteristic of the acceptor ion.
Owner:UNIV OF GEORGIA RES FOUND INC +1
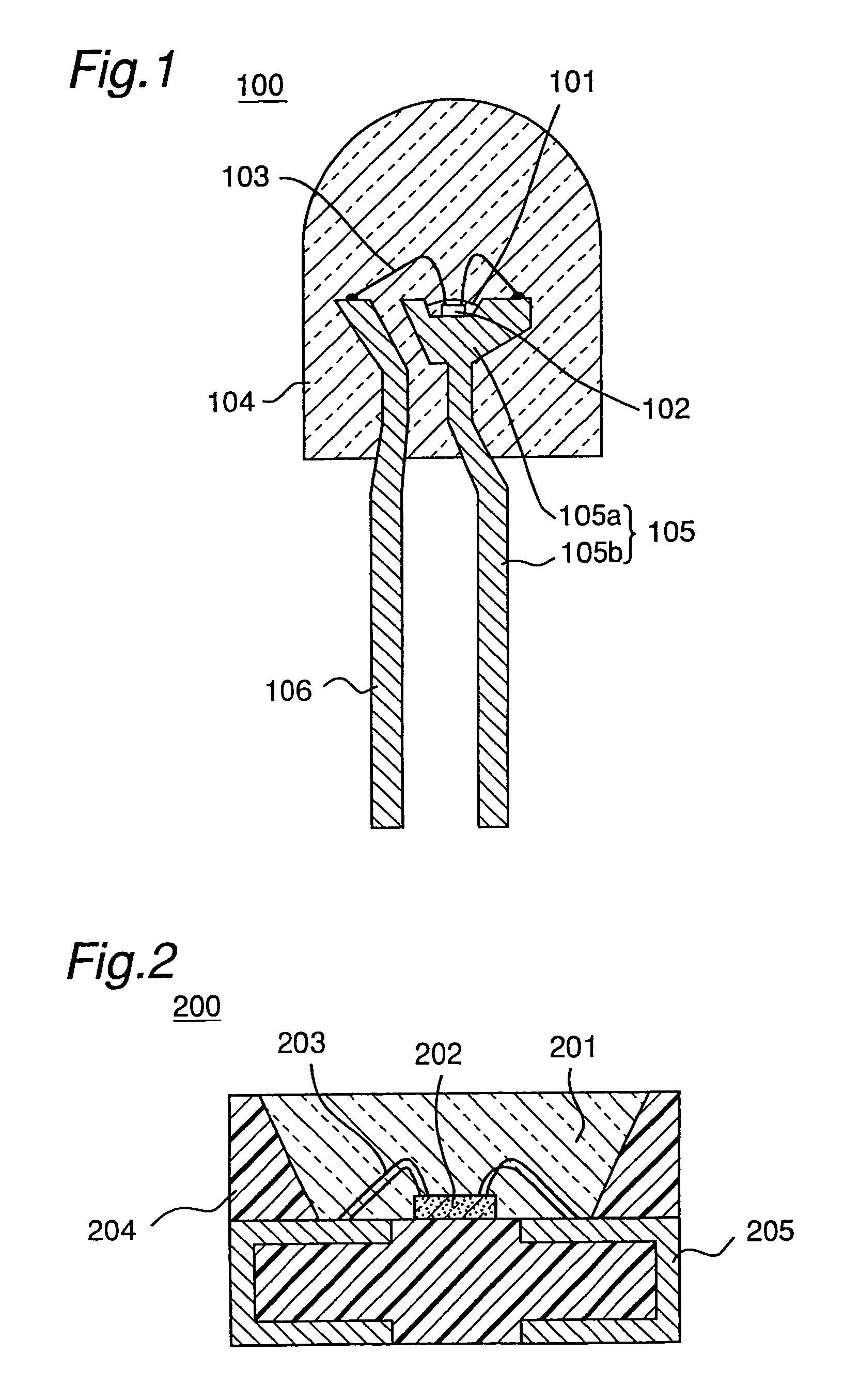
Light emitting device with blue light LED and phosphor components
InactiveUS7026756B2Low degree of deterioration in emission light intensityIncrease brightnessMechanical apparatusDischarge tube luminescnet screensIndiumPhosphor
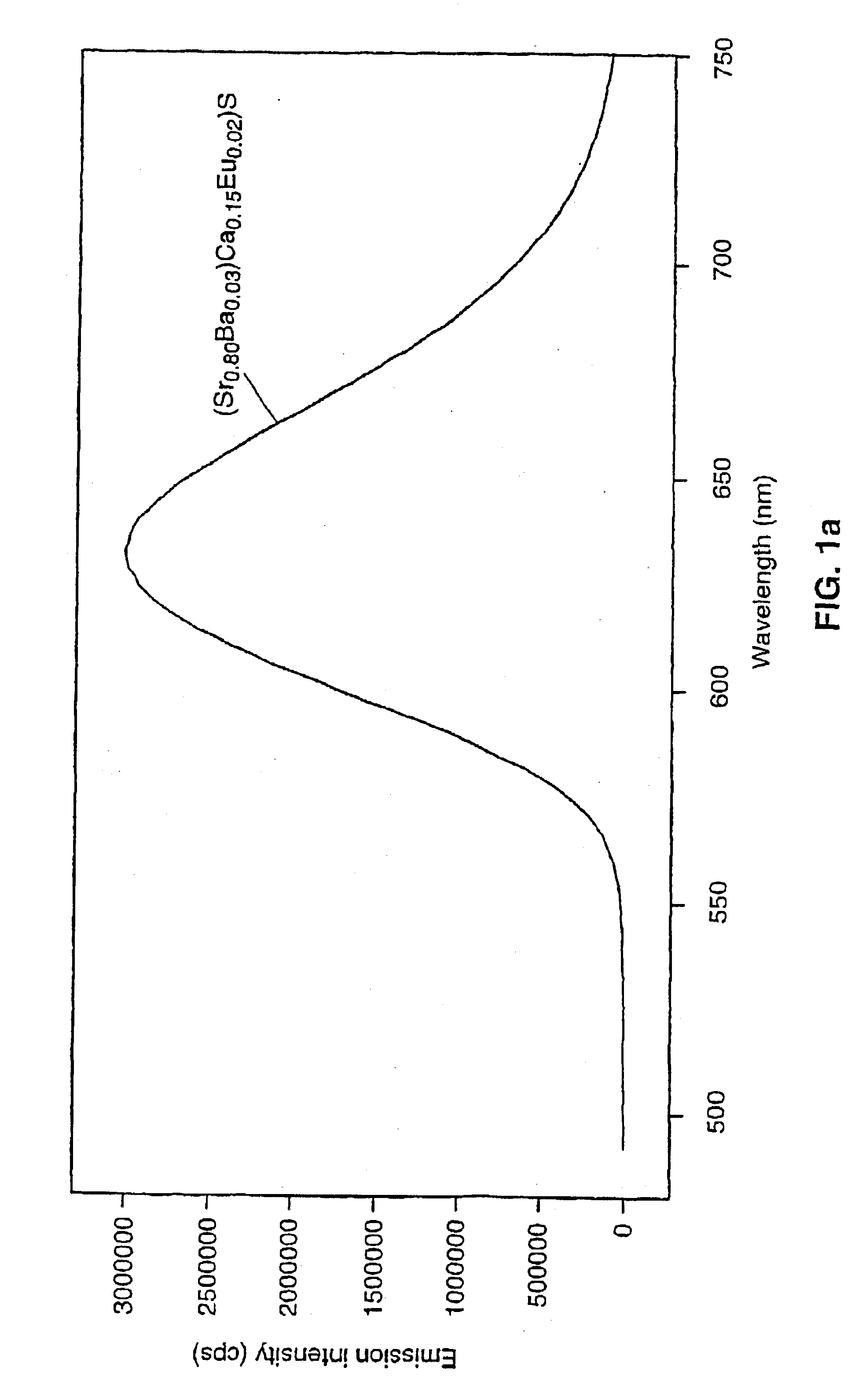
A light emitting device includes a light emitting component having an active layer of a semiconductor and a phosphor capable of absorbing a part of light emitted from the light emitting component and emitting light of wavelength different from that of the absorbed light, wherein the light emitting component is a LED which has an active layer constituting a gallium nitride based semiconductor containing Indium and is capable of emitting a blue color light with a peak wavelength within the range from 420 to 490 nm. The phosphor is a garnet fluorescent material activated with cerium which is capable of absorbing a part of the blue color light and thereby emitting light having a broad emission spectrum with a peak wavelength existing around the range from 510 to 600 nm and a tail continuing into the region from 700 to 750 nm.
Owner:NICHIA CORP
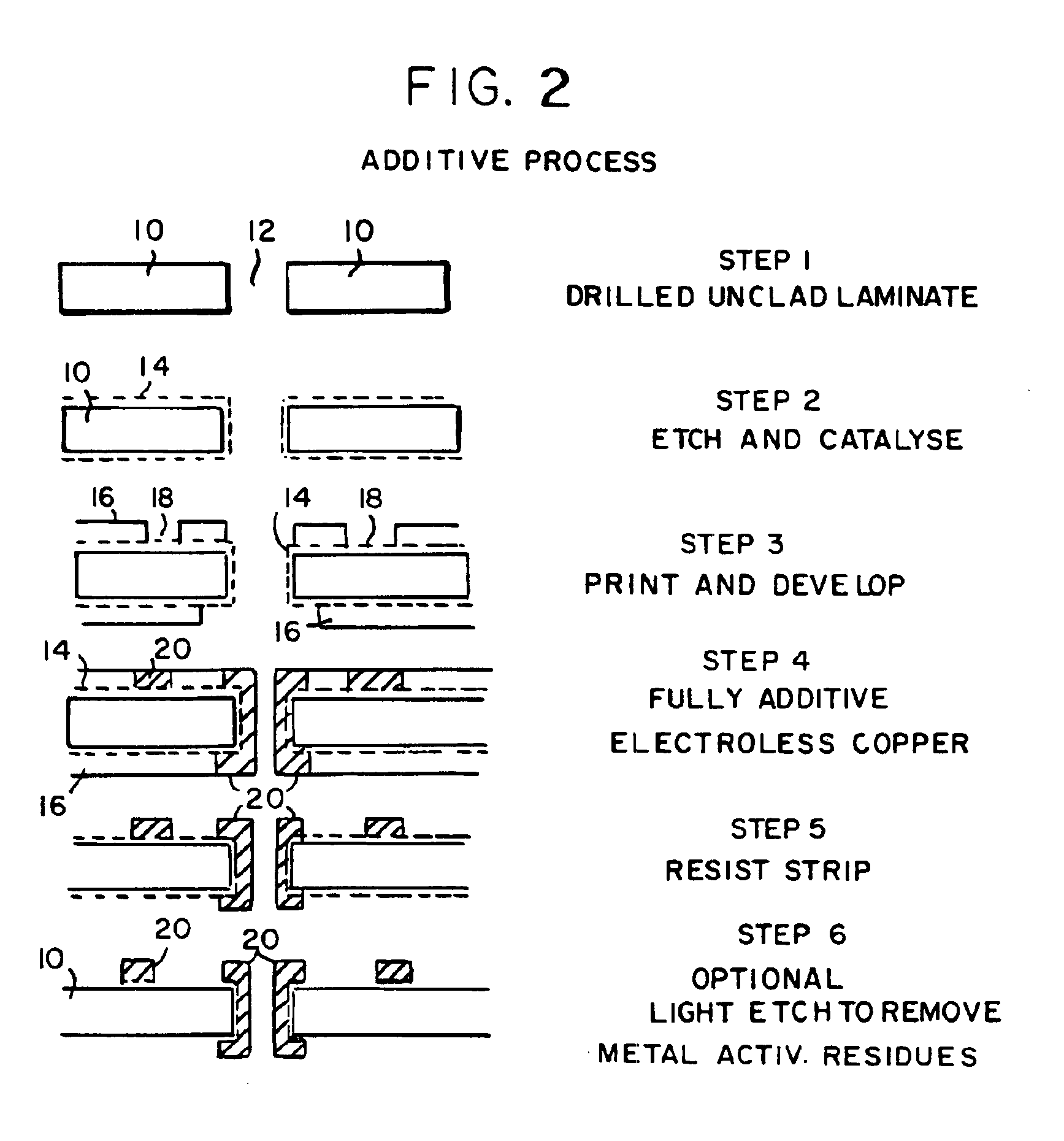
Electroless plating processes
InactiveUS6861097B1Reducing problem encounteredSimple methodPaper/cardboard articlesDecorative surface effectsPolymeric surfaceOxidation state
The invention includes processes for combined polymer surface treatment and metal deposition. Processes of the invention include forming an aqueous solution containing a metal activator, such as an oxidized species of silver, cobalt, ruthenium, cerium, iron, manganese, nickel, rhodium, or vanadium. The activator can be suitably oxidized to a higher oxidation state electrochemically. Exposing a part to be plated (such as an organic resin, e.g. a printed circuit board substrate) to the solution enables reactive hydroxyl species (e.g. hydroxyl radicals) to be generated and to texture the polymer surface. Such texturing facilitates good plated metal adhesion. As part of this contacting process sufficient time is allowed for both surface texturing to take place and for the oxidized metal activator to adsorb onto said part. The part is then contacted with a reducing agent capable of reducing the metal activator to a lower ionic form, or a lower oxidation state. That reduction can result in the formation of metallic catalytic material over the surface of the part. The reduced metal activator can then function to catalyze the electroless deposition of metal such as copper from solution by contacting the part with the plating solution.
Owner:SHIPLEY CO LLC
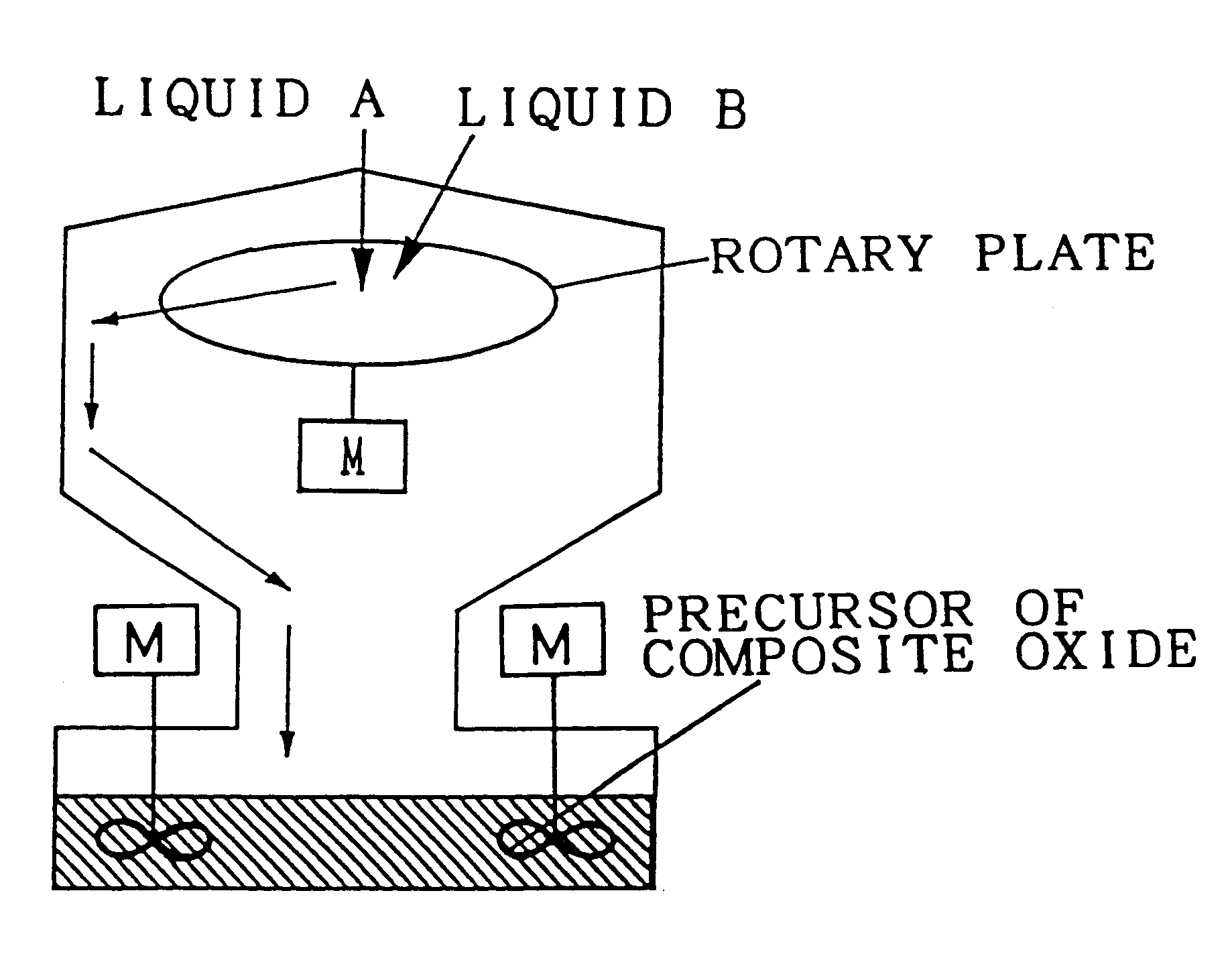
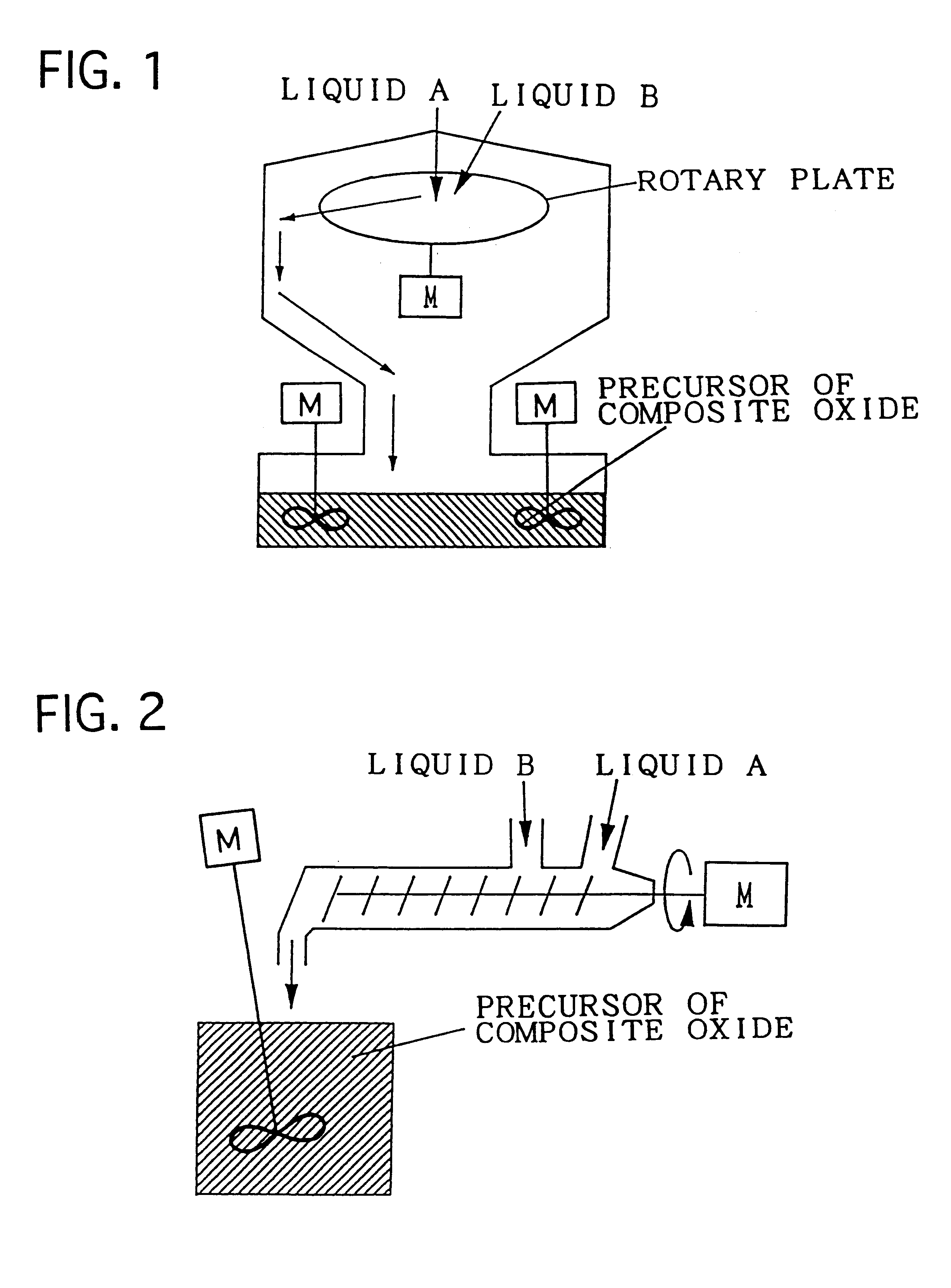
Composite oxide, composite oxide carrier and catalyst
InactiveUS6306794B1Improve heat resistanceImprove homogeneityInternal combustion piston enginesDispersed particle separationHeat resistanceCerium
The composite oxide and the composite oxide carrier are manufactured by the precursor forming step and firing step. The composite oxide catalyst is obtained by preparing a composite of catalytic components simultaneously with the formation of the precursor of composite oxide in the step of forming the precursor of composite oxide. The composite oxide and the composite oxide carrier are composed of a composite oxide in which at least one of cerium and zirconium, and aluminium disperse with extremely high homogeneity. With this structure, the heat resistance of the carrier is improved and consequently, enlargement of particles of the composite oxide defining the carrier, and sintering of adjacent particles of the composite oxide can be restrained, whereby the catalyst using the composite oxide carrier in accordance with the present invention is excellent in heat resistance.
Owner:TOYOTA CENT RES & DEV LAB INC
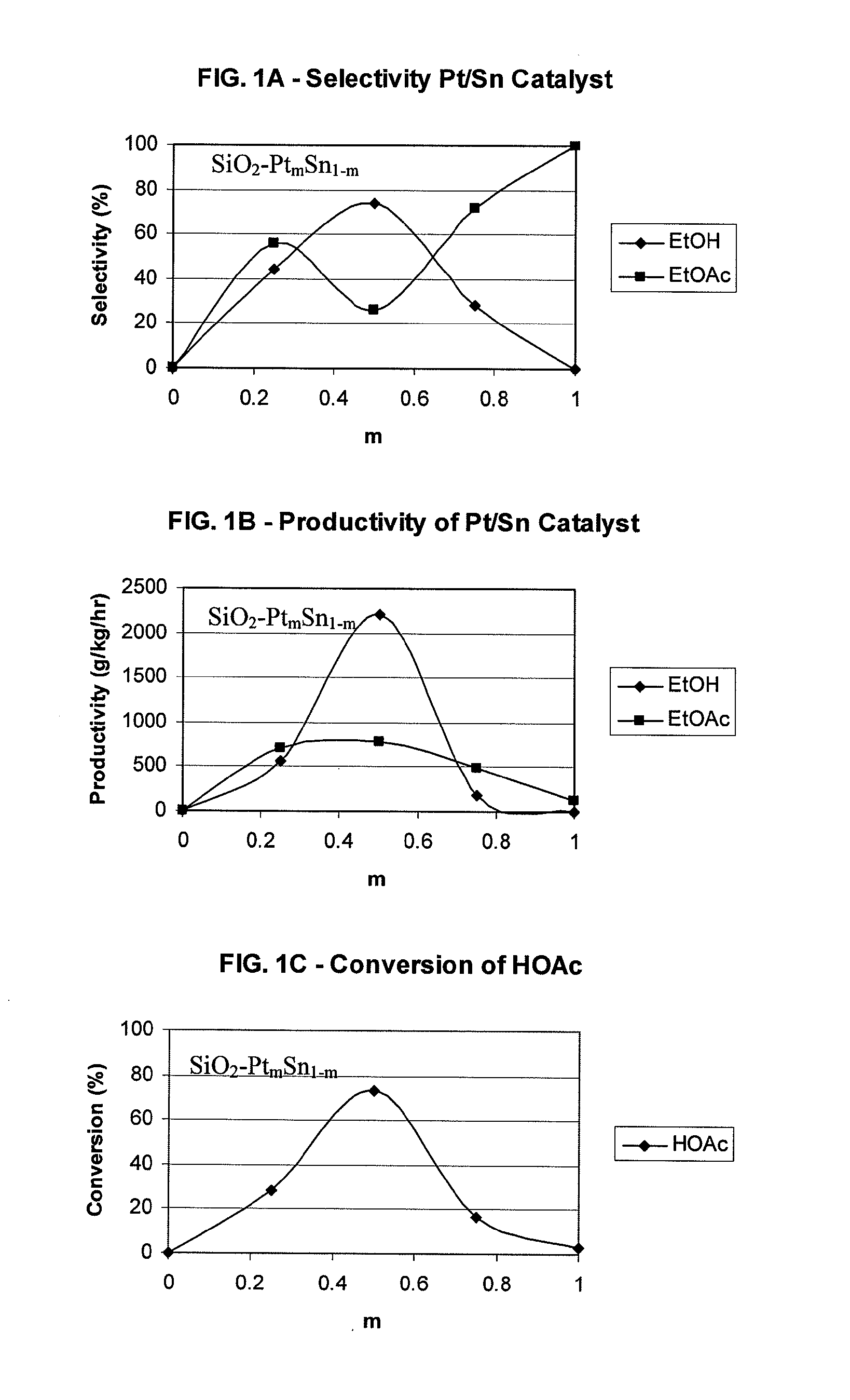
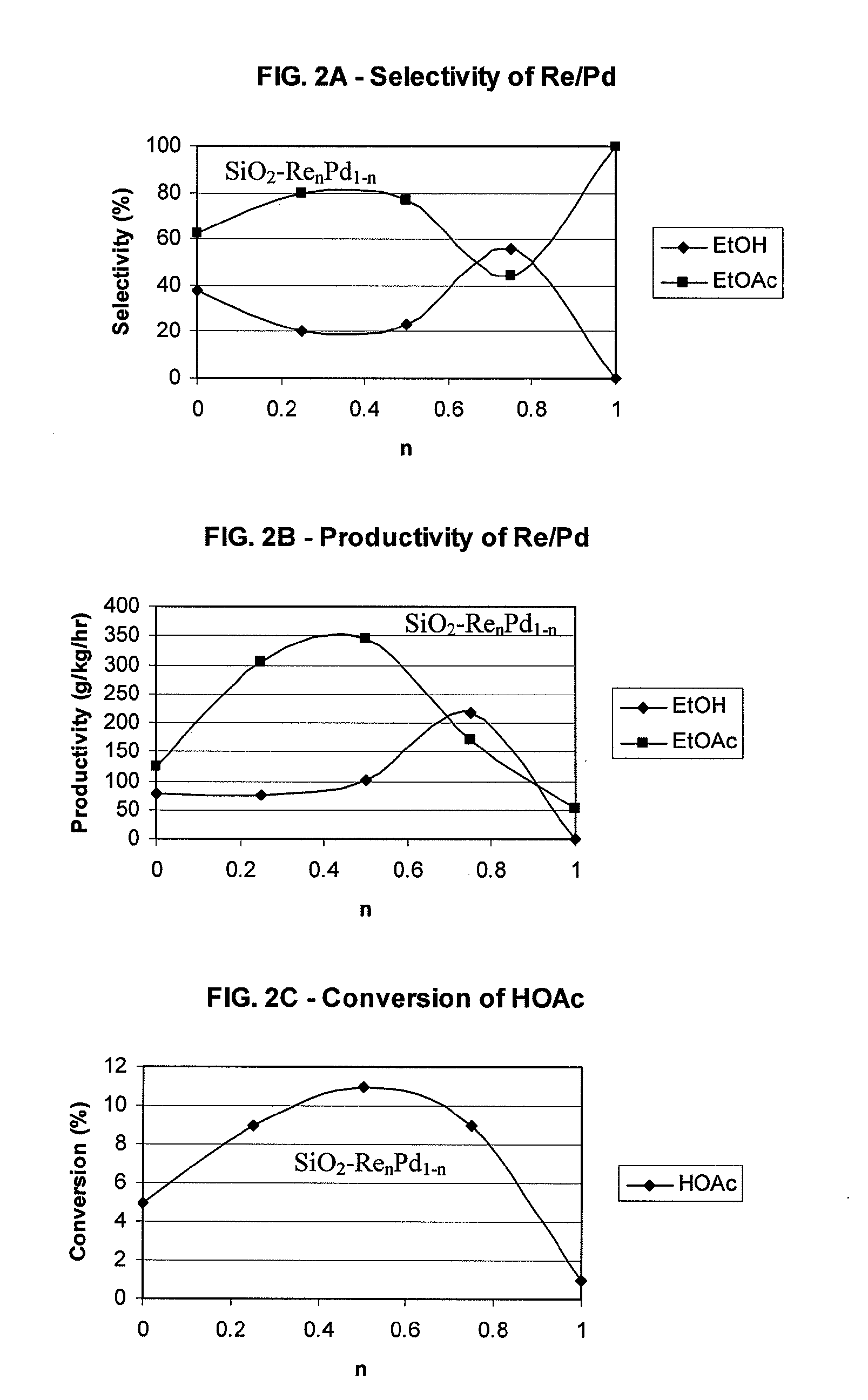
Processes for making ethanol from acetic acid
InactiveUS20100197985A1High selectivityPreparation by oxo-reaction and reductionEthylene productionCeriumCobalt
A process for selective formation of ethanol from acetic acid by hydrogenating acetic acid in the presence of first metal, a silicaceous support, and at least one support modifier. Preferably, the first metal is selected from the group consisting of copper, iron, cobalt, nickel, ruthenium, rhodium, palladium, osmium, iridium, platinum, titanium, zinc, chromium, rhenium, molybdenum, and tungsten. In addition the catalyst may comprise a second metal preferably selected from the group consisting of copper, molybdenum, tin, chromium, iron, cobalt, vanadium, tungsten, palladium, platinum, lanthanum, cerium, manganese, ruthenium, rhenium, gold, and nickel.
Owner:CELANESE INT CORP
Exhaust gas treatment catalyst for internal combustion engines with two catalytically active layers on a carrier structure
InactiveUS6348430B1Improve heat resistanceHigh activityInorganic chemistryInternal combustion piston enginesParticulatesPartial oxidation
A catalyst for treating the exhaust gas from internal combustion engines is provided, wherein the catalyst contains two catalytically active layers supported on a support. The first catalytically active layer contains a platinum group metal in close contact with all of the constituents of the first catalytically active layer, wherein the constituents of the first catalytically active layer include particulate aluminum oxide; particulate oxygen storage material, such as cerium oxide, cerium / zirconium and zirconium / cerium mixed oxides, and alkaline earth metal oxides. The second catalytically active layer, which is in direct contact with the exhaust gas, contains particulate aluminum oxide and at least one particulate oxygen storage material, such as cerium oxide, cerium / zirconium and zirconium / cerium mixed oxides. Rhodium is supported on part of the aluminum oxides in the second catalytically active layer or on the particulate oxygen storage material in the second catalytically active layer. By providing the platinum group metal in close contact with all of the constituents of the first catalytically active layer, improved conversion efficiency of the impurities in the exhaust gas can be achieved.
Owner:UMICORE AG & CO KG +1
Polymer nanocomposite implants with enhanced transparency and mechanical properties for administration within humans or animals
Polymer nanocomposite implants with nanofillers and additives are described. The nanofillers described can be any composition with the preferred composition being those composing barium, bismuth, cerium, dysprosium, europium, gadolinium, hafnium, indium, lanthanum, neodymium, niobium, praseodymium, strontium, tantalum, tin, tungsten, ytterbium, yttrium, zinc, and zirconium. The additives can be of any composition with the preferred form being inorganic nanopowders comprising aluminum, calcium, gallium, iron, lithium, magnesium, silicon, sodium, strontium, titanium. Such nanocomposites are particularly useful as materials for biological use in applications such as drug delivery, biomed devices, bone or dental implants.
Owner:PPG IND OHIO INC
Inorganic dopants, inks and related nanotechnology
InactiveUS6849109B2Facilitated DiffusionLower transition temperatureSelenium/tellurium compundsCell electrodesIndiumCerium
Ink compositions with modified properties result from using a powder size below 100 nanometers. Colored inks are illustrated. Nanoscale coated, uncoated, whisker inorganic fillers are included. The pigment nanopowders taught comprise one or more elements from the group actinium, aluminum, antimony, arsenic, barium, beryllium, bismuth, cadmuim, calcium, cerium, cesium, chalcogenide, cobalt, copper, dysprosium, erbium, europium, gadolinium, gallium, gold, hafnium, hydrogen, indium, iridium, iron, lanthanum, lithium, magnesium, manganese, mendelevium, mercury, molybdenum, neodymium, neptunium, nickel, niobium, nitrogen, oxygen, osmium, palladium, platinum, potassium, praseodymium, promethium, protactinium, rhenium, rubidium, scandium, silver, sodium, strontium, tantalum, terbium, thallium, thorium, tin, titanium, tungsten, vanadium, ytterbium, yttrium, zinc, and zirconium.
Owner:PPG IND OHIO INC
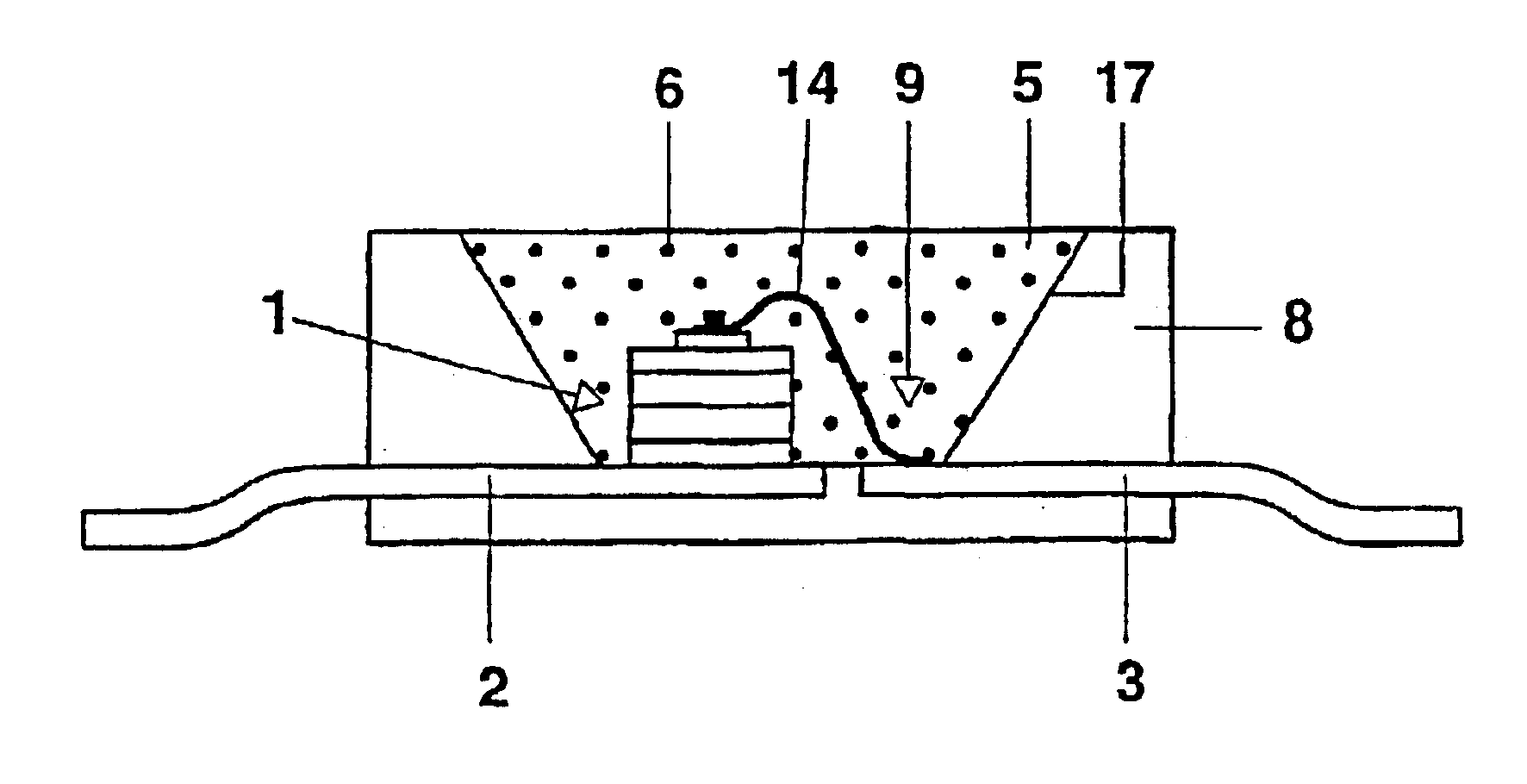
Scintillating substance and scintillating wave-guide element
InactiveUS6278832B1Improve light outputShorten the timePolycrystalline material growthOptical fibre with graded refractive index core/claddingFiberAdditive ingredient
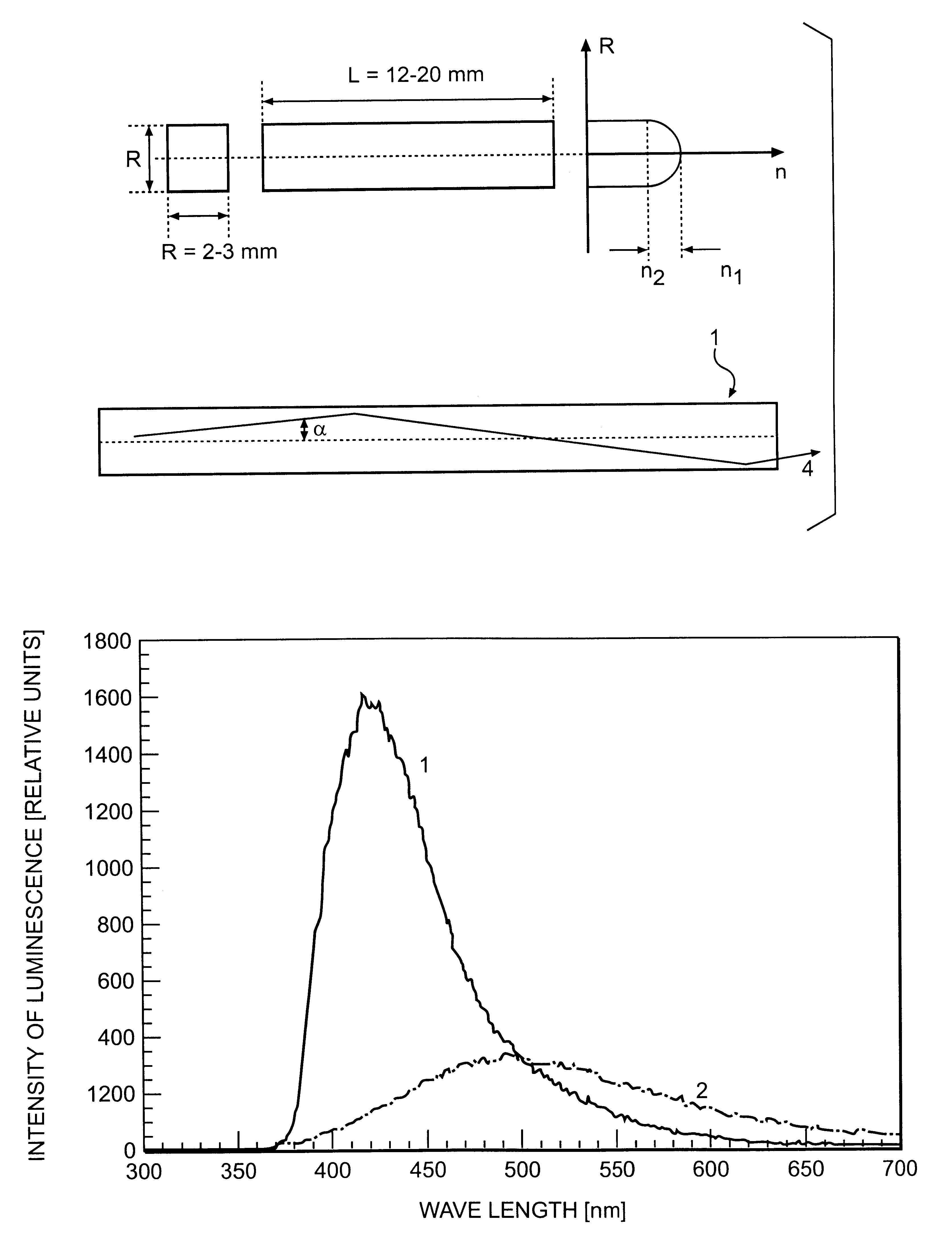
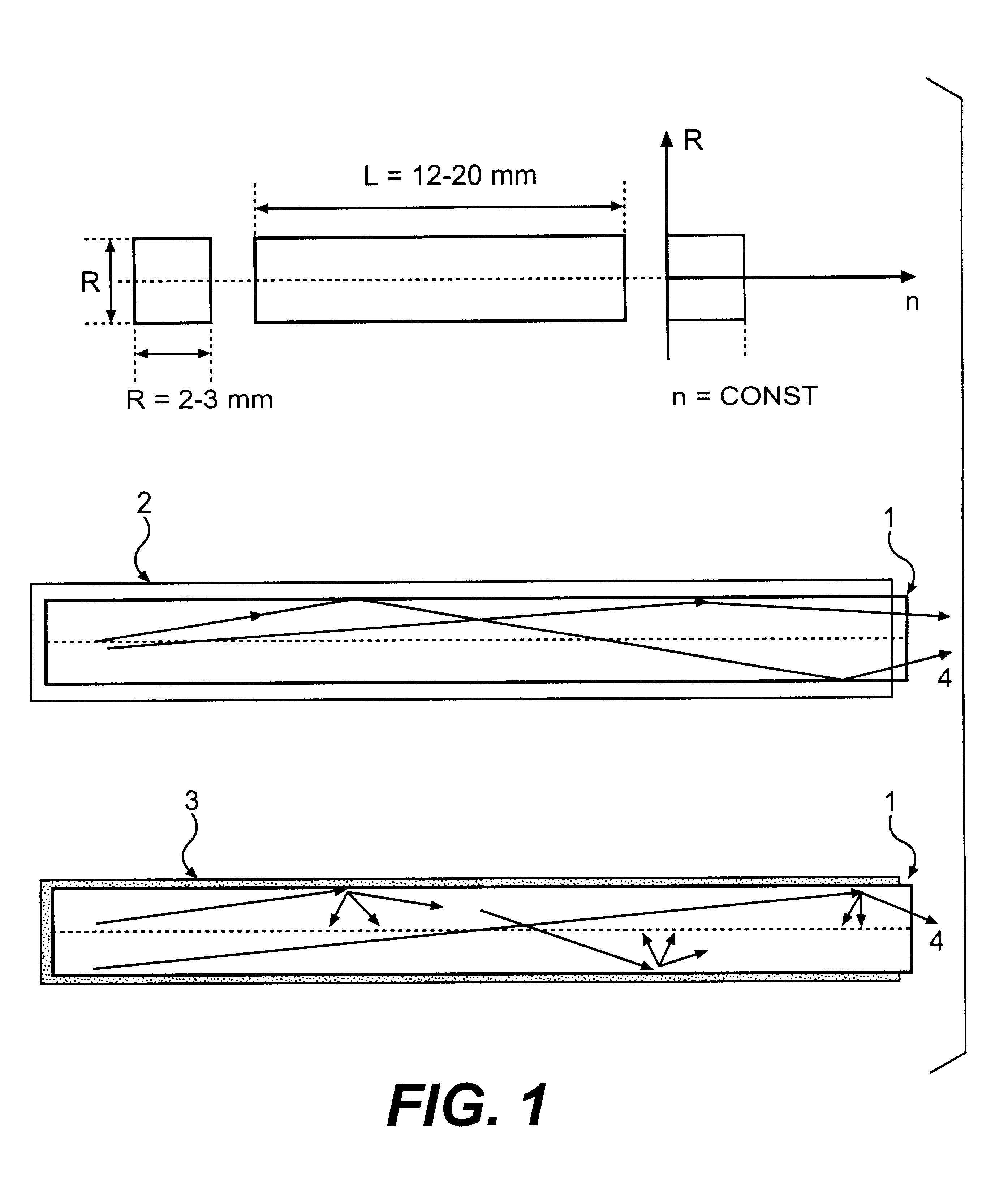
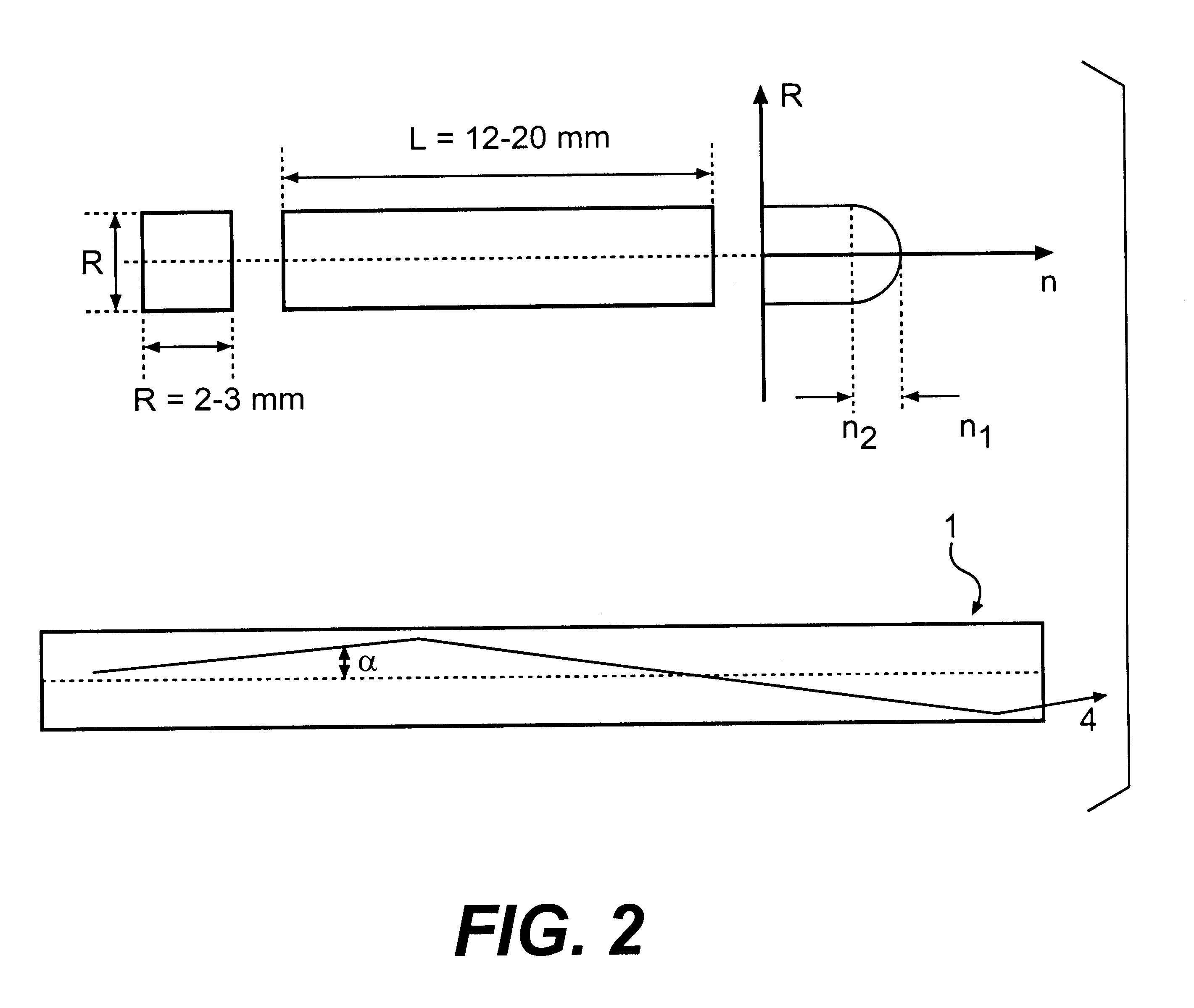
The invention is related to nuclear physics, medicine and oil industry, namely to the measurement of x-ray, gamma and alpha radiation; control for trans uranium nuclides in the habitat of a man; non destructive control for the structure of heavy bodies; three dimensional positron-electron computer tomography, etc.The essence of the invention is in additional ingredients in a chemical composition of a scintillating material based on crystals of oxyorthosilicates, including cerium Ce and crystallized in a structural type Lu2SiO5.The result of the invention is the increase of the light output of the luminescence, decrease of the time of luminescence of the ions Ce3+, increase of the reproducibility of grown crystals properties, decrease of the cost of the source melting stock for growing scintillator crystals, containing a large amount of Lu2O3, the raise of the effectiveness of the introduction of SCintillating crystal luminescent radiation into a glass waveguide fibre, prevention of cracking of crystals during the production of elements, creation of waveguide properties in scintillating elements, exclusion of expensive mechanical polishing of their lateral surface.
Owner:SOUTHBOURNE INVESTMENTS
Multifunctional composite absorbing material for purifying water and preparation method thereof
ActiveCN102350298AReduce wasteEfficient removalOther chemical processesWater/sewage treatment bu osmosis/dialysisAcrylic resinCerium
The invention provides a multifunctional composite absorbing material for purifying water and a preparation method thereof, relating to an absorbing material. The invention provides the multifunctional composite absorbing material for purifying water, which can be used for effectively removing a plurality of harmful substances in the water and has higher removal efficiency and lower production cost, and the preparation method thereof. The absorbing material is selected from at least one of absorbing materials A, B and C; the absorbing material A takes a mesoporous adsorption ceramic material as a carrier to load nano metals including nano silver, nano zinc, nano iron and nano cerium; the absorbing material B takes the mesoporous adsorption ceramic material as the carrier to load nano metal oxides including nano titanium dioxide, nano zinc oxide, nano ferric oxide and nano cerium dioxide; and the absorbing material C is prepared from the following raw materials according to the mass ratio: 100-200 parts of active carbon powder, 20-30 parts of polyethylene powder, 10-30 parts of calcium sulfite powder, 10-30 parts of natural zeolite powder, 20-40 parts of macroporous acrylic resin and 10-20 parts of attapulgite powder.
Owner:XIAMEN BAILIN WATER PURIFICATION TECH CO LTD
Scintillator compositions, and related processes and articles of manufacture
ActiveUS20050082484A1Improve performanceExcellent and reproducible scintillation responseMaterial analysis by optical meansLuminescent compositionsIodideHigh energy
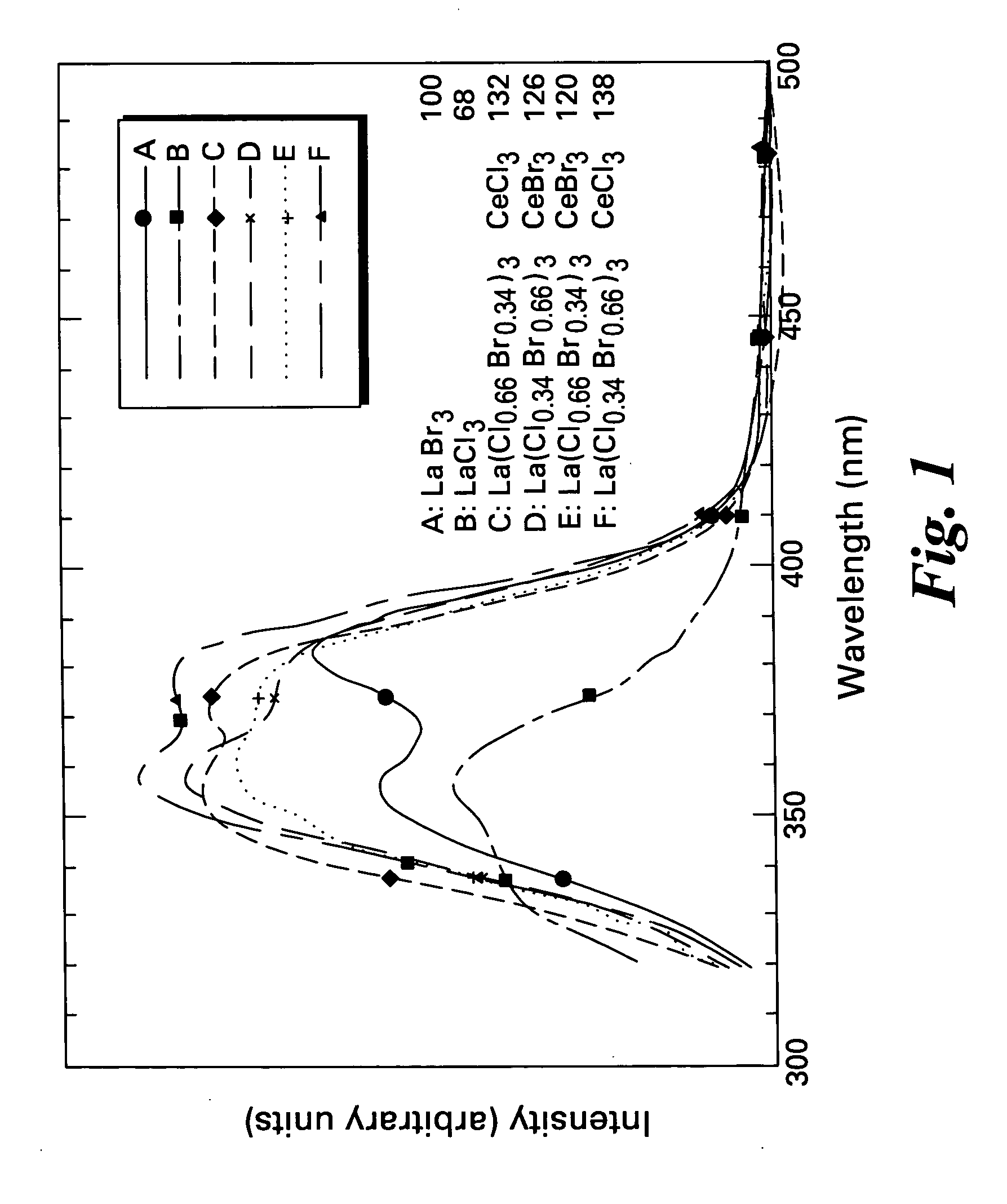
Scintillator materials based on certain types of halide-lanthanide matrix materials are described. In one embodiment, the matrix material contains a mixture of lanthanide halides, i.e., a solid solution of at least two of the halides, such as lanthanum chloride and lanthanum bromide. In another embodiment, the matrix material is based on lanthanum iodide alone, which must be substantially free of lanthanum oxyiodide. The scintillator materials, which can be in monocrystalline or polycrystalline form, also include an activator for the matrix material, e.g., cerium. Radiation detectors that use the scintillators are also described, as are related methods for detecting high-energy radiation.
Owner:BAKER HUGHES OILFIELD OPERATIONS LLC
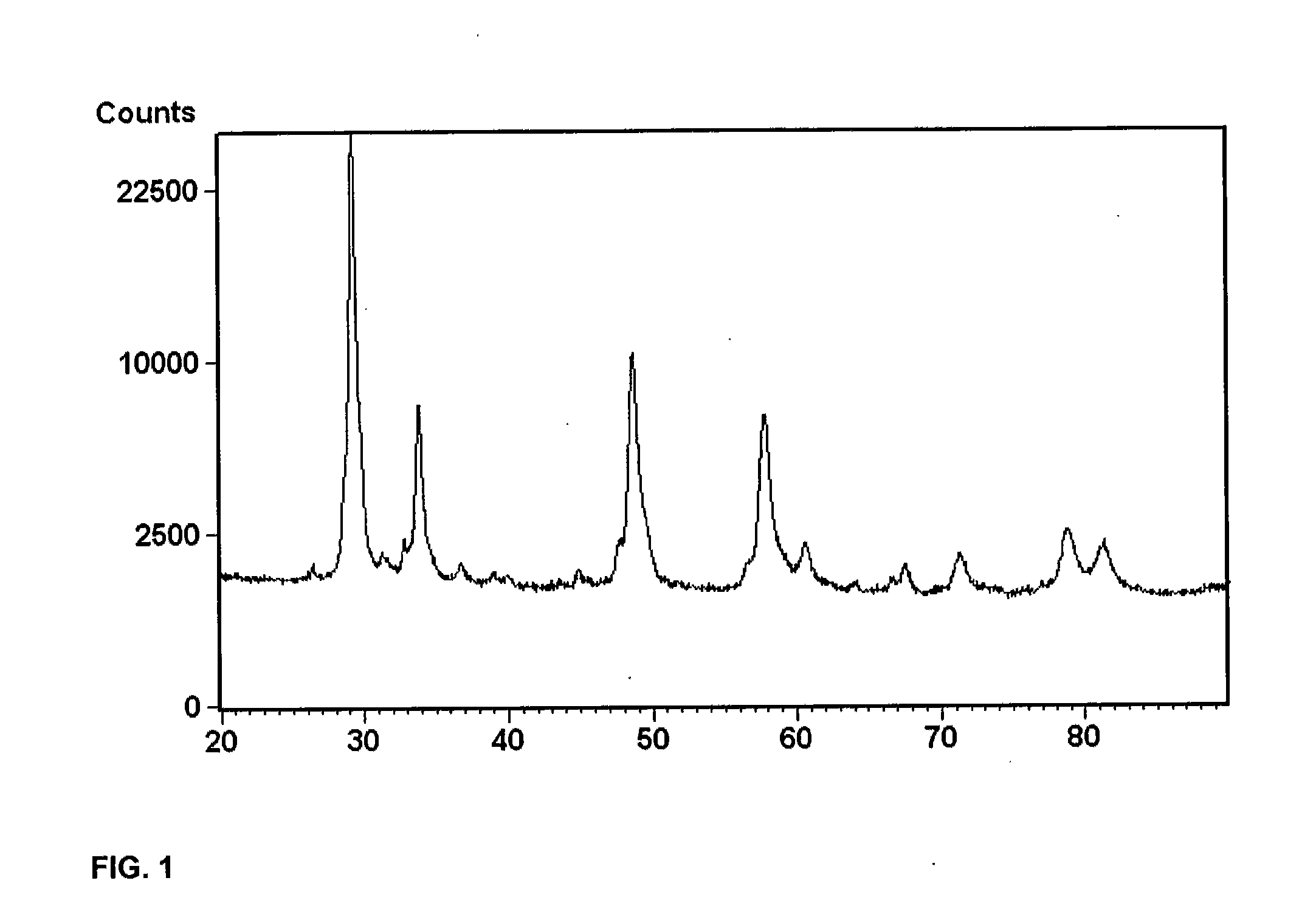
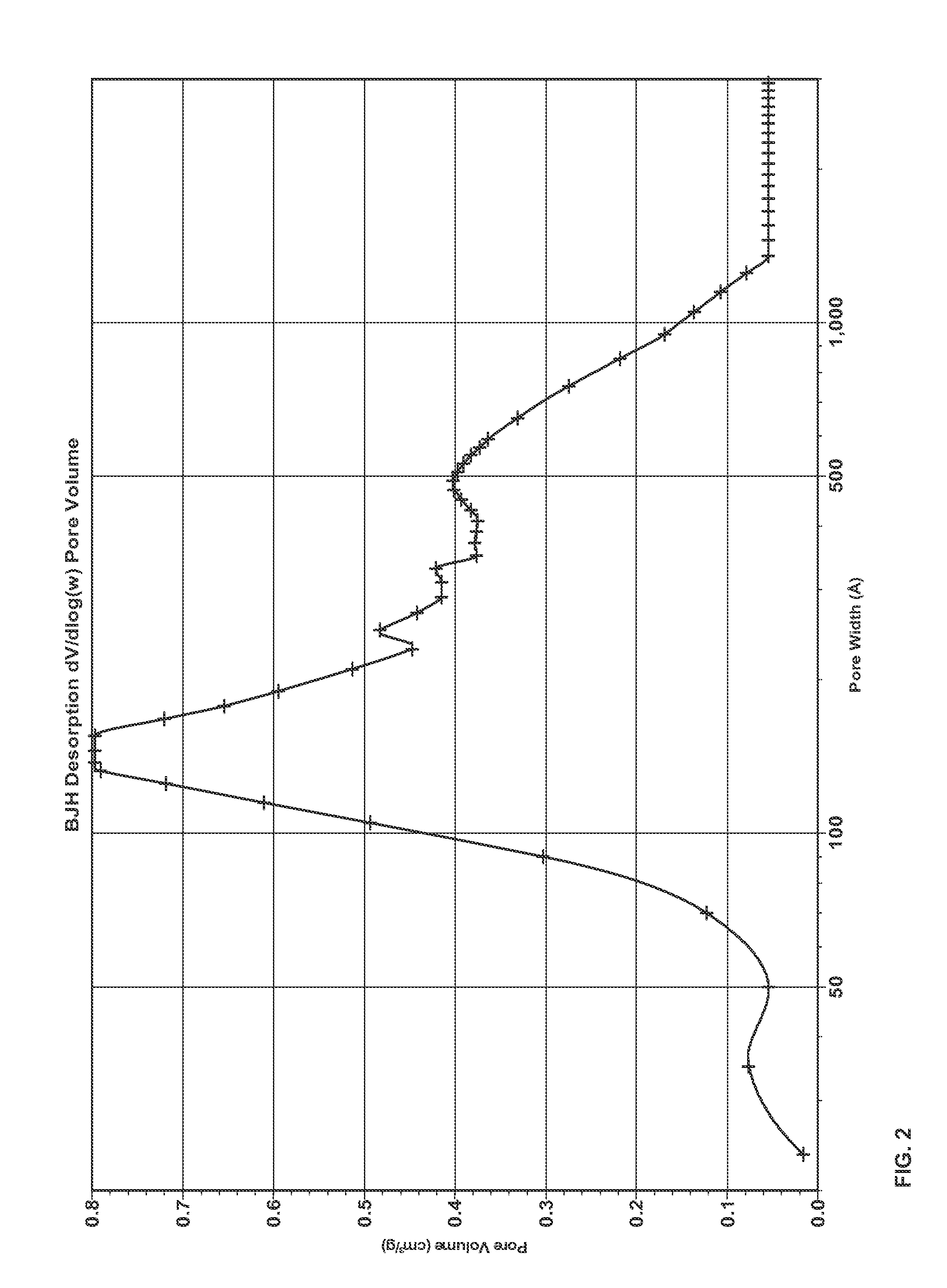
Porous inorganic composite oxide
ActiveUS20120129690A1Improve thermal stabilityHigh pore volumeHeat treatmentsInternal combustion piston enginesPtru catalystCerium
A porous inorganic composite oxide containing oxides of aluminum and of cerium and / or zirconium, and, optionally, oxides of one or more dopants selected from transition metals, rare earths, and mixtures thereof, and having a specific surface area, in m2 / g, after calcining at 1100° C. for 5 hours, of ≧0.8235[Al]+11.157 and a total pore volume, in cm3 / g, after calcining at 900° C. for 2 hours, of ≧0.0097[Al]+0.0647, wherein [Al] is the amount of oxides of aluminum, expressed as pbw Al2O3 per 100 pbw of the composite oxide; a catalyst containing one or more noble metals dispersed on the porous inorganic composite oxide; and a method for making the porous inorganic composite oxide.
Owner:RHODIA OPERATIONS SAS
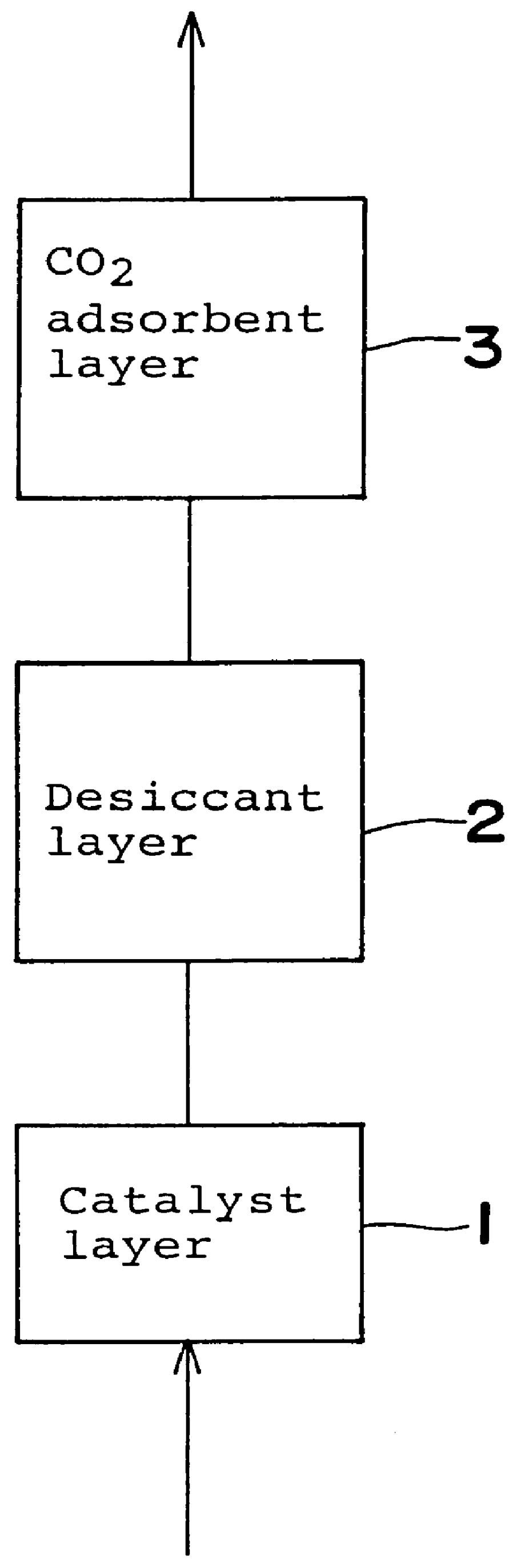
Gas purifying process and gas purifying apparatus
A method is provided for removing water, carbon monoxide and carbon dioxide out of a gas, such as air, by passing the gas through a packed column so that the gas sequentially contacts a catalyst consisting of platinum or palladium and at least one member selected from the group consisting of iron, cobalt, nickel, manganese, copper, chromium, tin, lead and cerium wherein the catalyst is supported on alumina containing substantially no pores having pore diameters of 110 Angstroms or less under conditions which oxidize the carbon monoxide in the gas into carbon dioxide; an adsorbent selected from the group consisting of silica gel, activated alumina, zeolite and combinations thereof under conditions in which water is adsorbed and removed from the gas and an adsorbent selected from the group consisting of calcium ion exchanged A zeolite; calcium ion exchanged X zeolite; sodium ion exchanged X zeolite and mixtures thereof under conditions which carbon dioxide is adsorbed and removed from the gas. The gas may also be subjected to a catalyst / adsorbent in the packed column to effect oxidation and removal of hydrogen in the gas.
Owner:NIPPON SANSO CORP
Vanadium-titanium oxide catalyst, and preparation method and application thereof
InactiveCN102764643ANo effect on activityDo not change the production processDispersed particle separationMetal/metal-oxides/metal-hydroxide catalystsMetal poisoningAlkaline earth metal
The invention relates to a vanadium-titanium oxide catalyst which can resist alkaline metal and alkaline earth metal poisoning. The catalyst is characterized in that the vanadium-titanium oxide catalyst is doped with an element Ce. The catalyst has fine resistance to alkaline metal poisoning, and above all, the doped cerium component has no influence on the activity of the SCR (selective catalytic reduction) catalyst while improving the resistance of the V2O5 / (MoO3)x(WO3)1-x-TiO2 catalyst to alkaline metal poisoning. The preparation method of the catalyst is simple and easy to implement and has very excellent N2 generation selectivity; and meanwhile, the Ce is a non-poisonous component and can not do harm to human health and ecological environment.
Owner:RES CENT FOR ECO ENVIRONMENTAL SCI THE CHINESE ACAD OF SCI
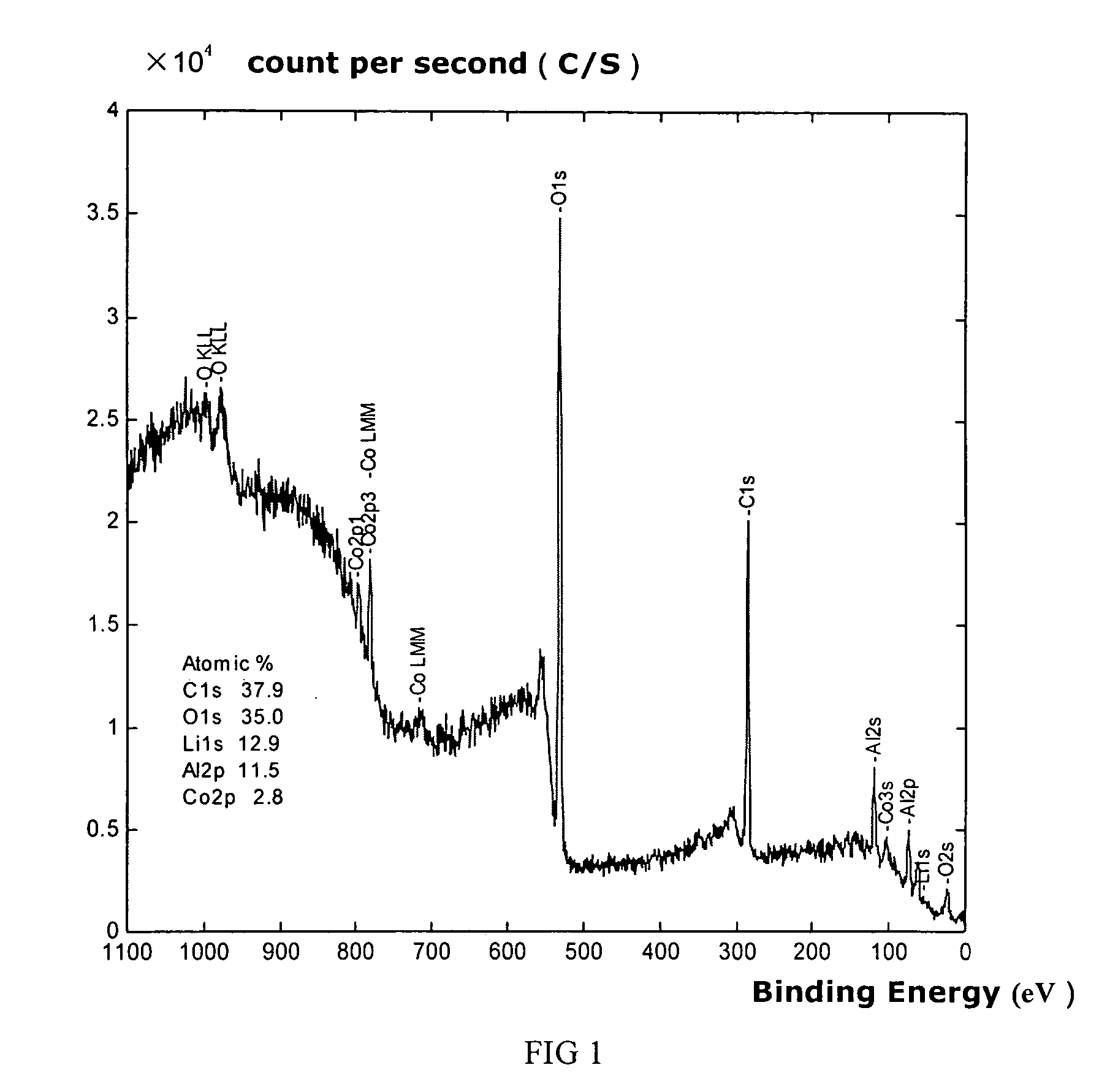
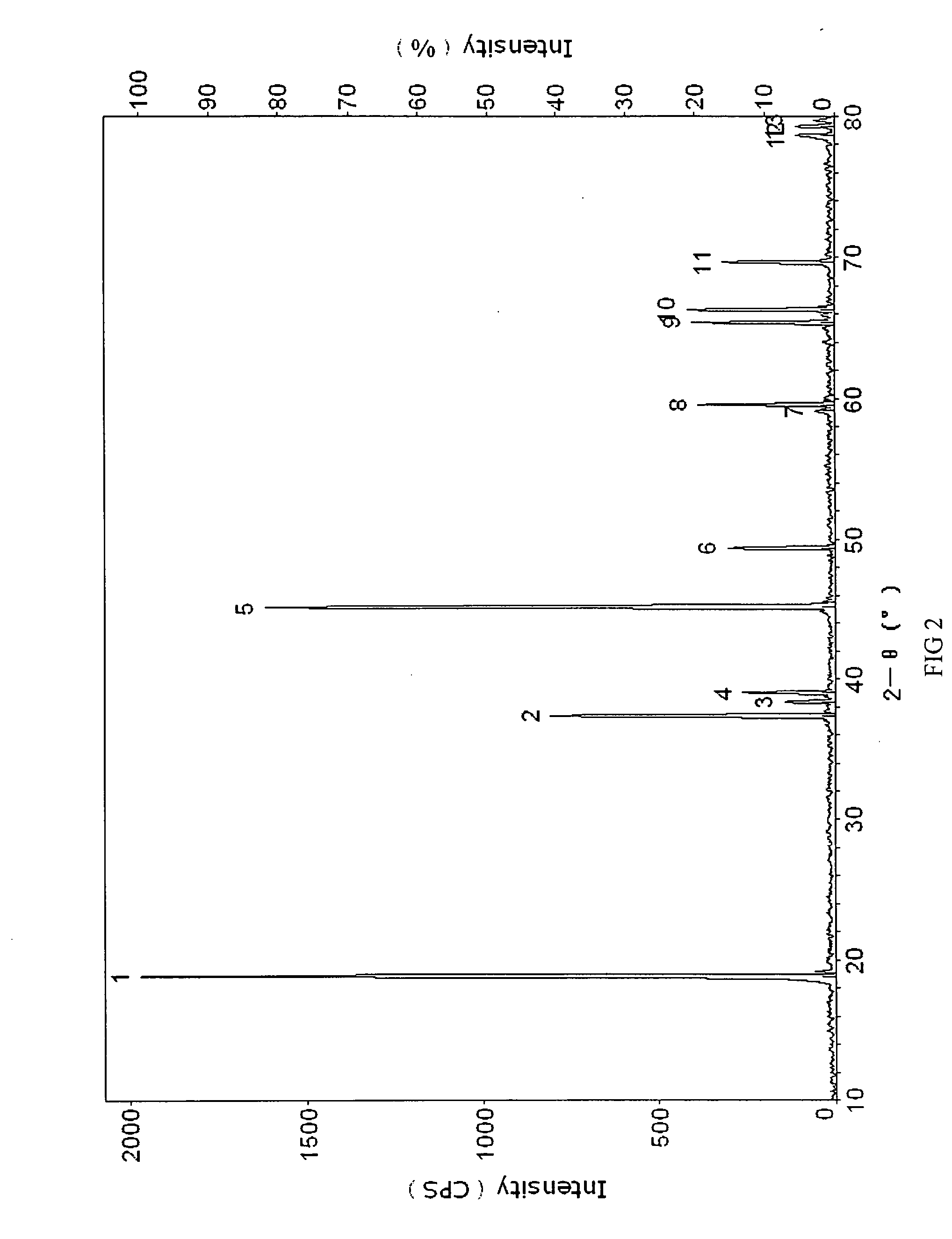
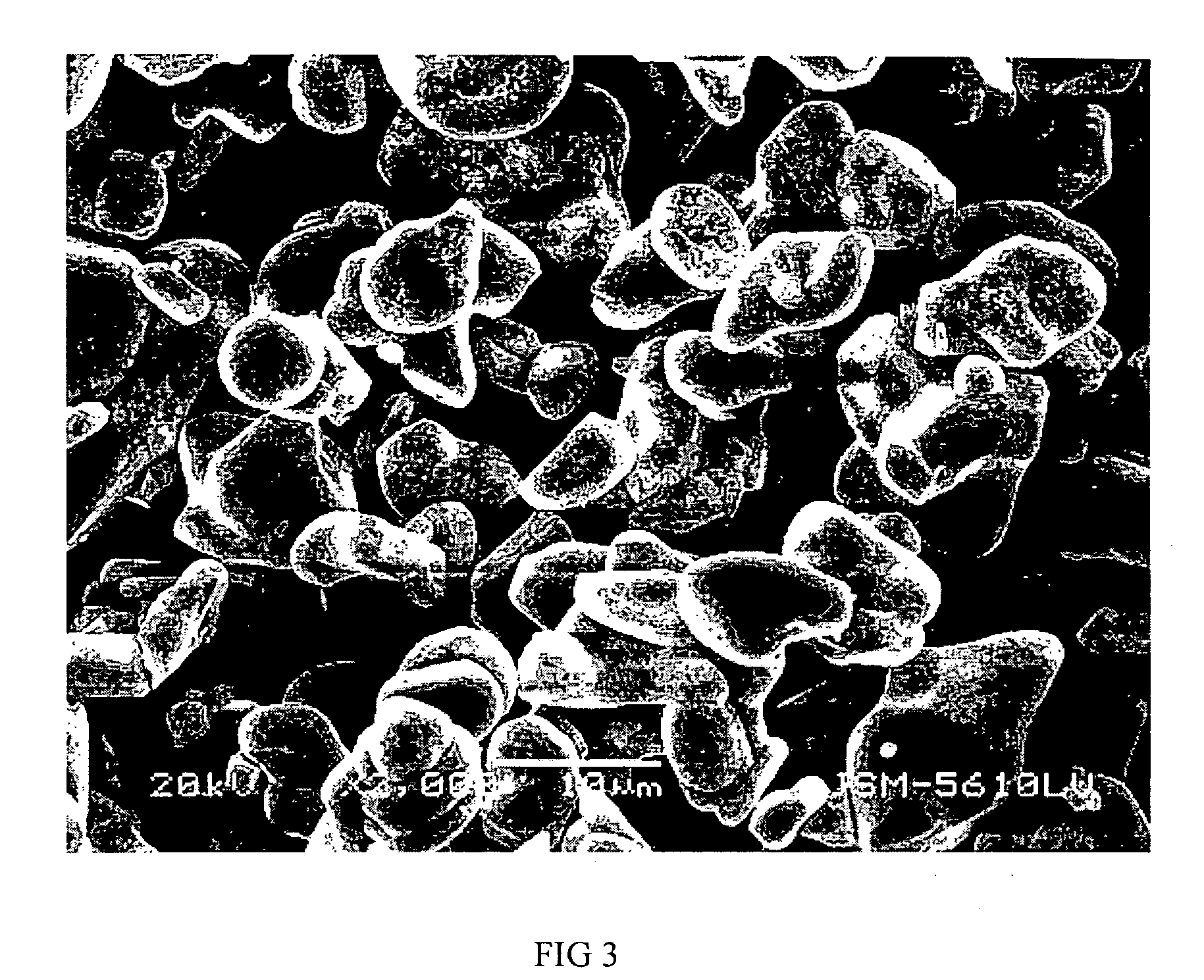
Materials for positive electrodes of lithium ion batteries and their methods of fabrication
InactiveUS20050130042A1Cycle wellEasy dischargeElectrode thermal treatmentSecondary cellsCeriumSolvent
This invention discloses materials for positive electrodes of secondary batteries and their methods of fabrication. Said materials comprise of granules of an active material for positive electrodes coated with an oxide layer. The active material is one or more of the following: oxides of lithium cobalt, oxides of lithium nickel cobalt, oxides of lithium nickel cobalt manganese, oxides of lithium manganese, LiCoO2, LiNi1-xCoxO2, LiNi1 / 3Co1 / 3Mn1 / 3O2, and LiMn2O4. The non-oxygen component in the oxide layer is one or more of the following: aluminum, magnesium, zinc, calcium, barium, strontium, lanthanum, cerium, vanadium, titanium, tin, silicon, boron, Al, Mg, Zn, Ca, Ba, Sr, La, Ce, V, Ti, Sn, Si, and B. Said non-oxygen component of the granules is between 0.01 wt. % to 10 wt. % of said granules of active material. The methods of fabrication for said materials includes the steps of mixing an additive and an active material for positive electrodes uniformly in water or solvent, evaporating said solvent or water, and heat treating the remaining mixture at 300° C. to 900° C. for between 1 hour to 20 hours. The additive is a compound of one or more of the following elements: aluminum, magnesium, zinc, calcium, barium, strontium, lanthanum, cerium, vanadium, titanium, tin, silicon, boron, Al, Mg, Zn, Ca, Ba, Sr, La, Ce, V, Ti, Sn, Si, and B where the element is between 0.01 wt. % to 10 wt. % of said active material. Using the materials of positive electrodes disclosed above or materials for positive electrodes fabricated in the methods disclosed above in batteries produces batteries with excellent cycling and high temperature properties.
Owner:BYD AMERICA CORP
Light emitting device for generating specific colored light, including white light
InactiveUS6850002B2Improve efficiencyDischarge tube luminescnet screensElectric discharge tubesCeriumMetallic sulfide
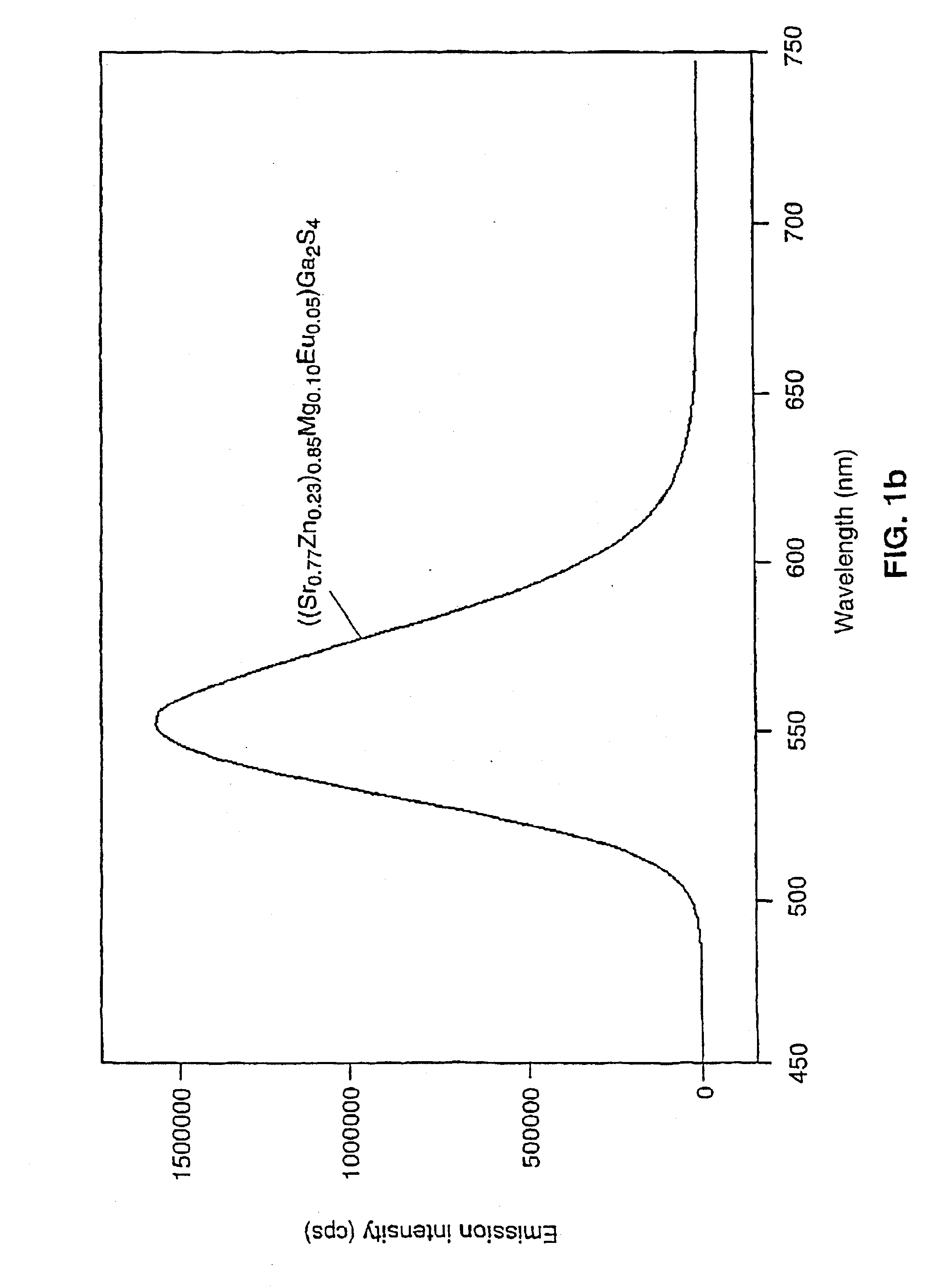
A device for the generation of specific colored light including white light by luminescent down conversion and additive color mixing based on a light-emitting diode (LED) comprising a semiconductor light-emitting layer emitting near UV light about 370-420 nm or blue light about 420-480 nm and phosphors which absorb completely or partly the light emitted by the light-emitting component and emit light of wavelengths longer than that of the absorbed primary light, wherein the light emitting layer of the light emitting component is preferably a Ga(In)N-based semiconductor; and at least one of the phosphors contains a metal sulfide fluorescent material activated with europium containing at least one element selected from the group consisting of Ba, Sr, Ca, Mg and Zn; and / or at least another phosphor which contains a complex thiometalate fluorescent material activated with either europium, cerium or both europium and cerium containing 1) at least one element selected from the group consisting of Ba, Sr, Ca, Mg and Zn and 2) at least one element selected from the group consisting of Al, Ga, In, Y, La and Gd.
Owner:OSRAM OPTO SEMICONDUCTORS GMBH +1
Shift converter having an improved catalyst composition, and method for its use
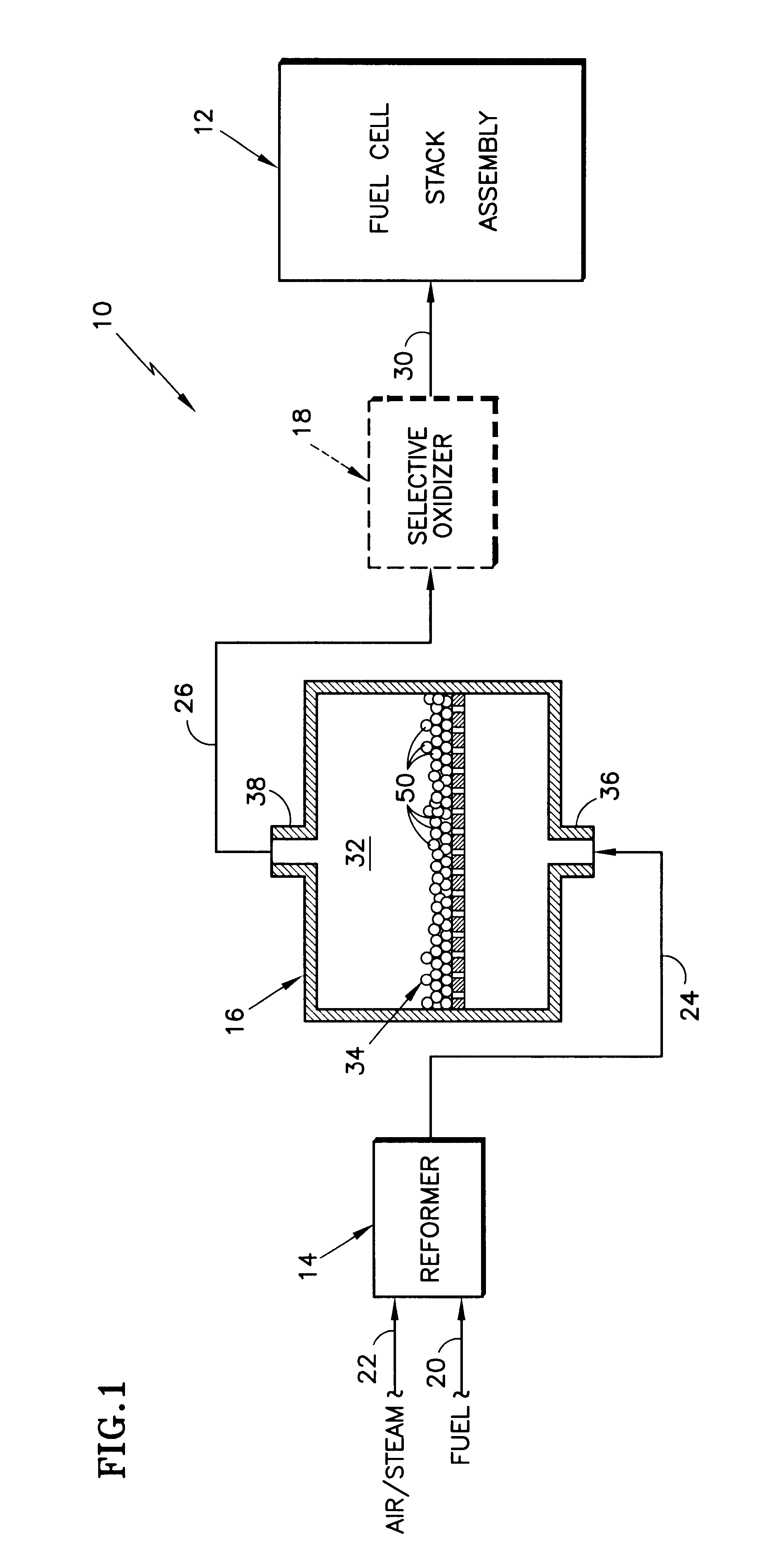
InactiveUS6455182B1Reduce amountImproved catalyst compositionHydrogenCatalyst carriersFuel cellsCerium
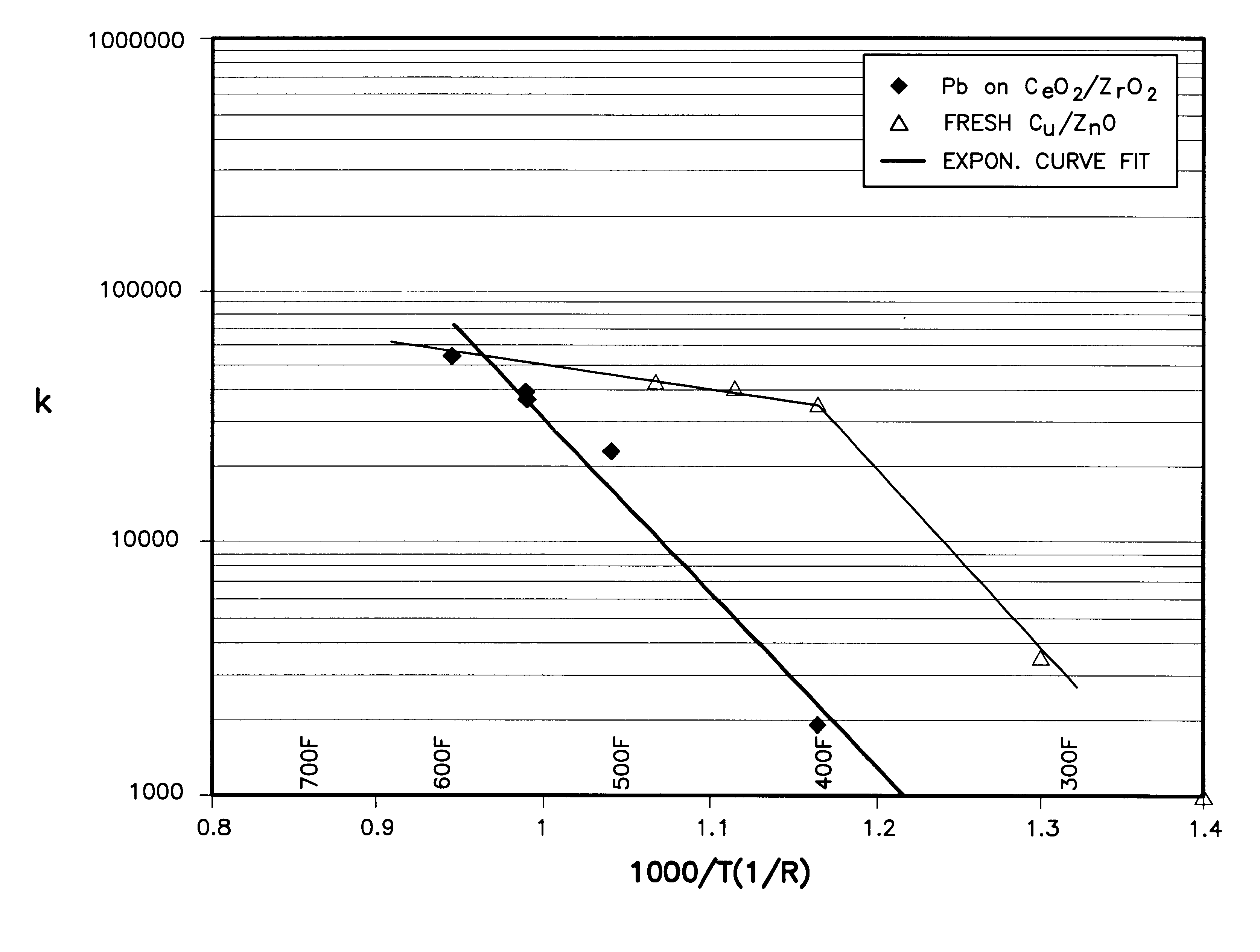
A shift converter (16) in a fuel processing subsystem (14, 16, 18) for a fuel cell (12) uses an improved catalyst composition (50) to reduce the amount of carbon monoxide in a process gas for the fuel cell (12). The catalyst composition (50) is a noble metal catalyst having a promoted support of mixed metal oxide, including at least both ceria and zirconia. Cerium is present in the range of 30 to 50 mole %, and zirconium is present in the range of 70 to 50 mole %. Additional metal oxides may also be present. Use of the catalyst composition (50) obviates the requirement for prior reducing of catalysts, and minimizes the need to protect the catalyst from oxygen during operation and / or shutdown.
Owner:HYAXIOM INC
Layered thermal barrier coatings containing lanthanide series oxides for improved resistance to CMAS degradation
InactiveUS20070160859A1Avoid damageElimination of expensiveBlade accessoriesEfficient propulsion technologiesReaction layerCerium
A coating applied as a two layer system. The outer layer is an oxide of a group IV metal selected from the group consisting of zirconium oxide, hafnium oxide and combinations thereof, which are doped with an effective amount of a lanthanum series oxide. These metal oxides doped with a lanthanum series addition comprises a high weight percentage of the outer coating. As used herein, lanthanum series means an element selected from the group consisting of lanthanum (La), cerium (Ce), praseodymium (Pr), neodymium (Nd), promethium (Pm), samarium (Sm), europium (Eu), gadolinium (Gd), terbium (Tb), dysprosium (Dy), holmium (Ho), erbium (Er), thulium (Tm), ytterbium (Yb), lutetium (Lu) and combinations thereof, and lanthanum series oxides are oxides of these elements. When the zirconium oxide is doped with an effective amount of a lanthanum series oxide, a dense reaction layer is formed at the interface of the outer layer of TBC and the CMAS. This dense reaction layer prevents CMAS infiltration below it. The second layer, or inner layer underlying the outer layer, comprises a layer of partially stabilized zirconium oxide.
Owner:GENERAL ELECTRIC CO
Nitrogen oxide storage material and nitrogen oxide storing catalyst prepared therefrom
InactiveUS6350421B1Determine efficiencyNitrogen compoundsExhaust apparatusAlkaline earth metalCuprate
A nitrogen oxide storage material is disclosed which contains at least one storage component for nitrogen oxides in the form of an oxide, mixed oxide, carbonate or hydroxide of the alkaline earth metals magnesium, calcium, strontium and barium and the alkali metals potassium and caesium on a high surface area support material. The support material can be doped cerium oxide, cerium / zirconium mixed oxide, calcium titanate, strontium titanate, barium titanate, barium stannate, barium zirconate, magnesium oxide, lanthanum oxide, praseodymium oxide, samarium oxide, neodymium oxide, yttrium oxide, zirconium silicate, yttrium barium cuprate, lead titanate, tin titanate, bismuth titanate, lanthanum cobaltate, lanthanum manganate and barium cuprate or mixtures thereof.
Owner:DMC2 DEGUSSA METALS +1
Raspberry-type metal oxide nanostructures coated with ceo2 nanoparticles for chemical mechanical planarization (CMP)
InactiveUS20120077419A1Efficient processCost-effective and convenient processMaterial nanotechnologySilicaCeo2 nanoparticlesWater soluble
Raspberry-type coated particles comprising a core selected from the group consisting of metal oxides of Si, Ti, Zr, Al, Zn and mixtures thereof with a core size of from 20 to 100 nm wherein the core is coated with CeCO2 particles having a particle size below 10 nm; process for preparing raspberry type coated particles comprising the steps of i) providing a mixture containing: a) core particles selected from the group of metal oxides of Si, Ti, Zr, Al, Zn and mixtures thereof, with a particle size of from 20 to 100 nm; b) a water soluble Ce-salt and c) water; ii) adding an organic or inorganic base to the mixture of step i) at temperatures of from 10 to 90° C. and iii) aging the mixture at temperatures of from 10 to 90° C.; and polishing agents containing the particles and their use for polishing surfaces.
Owner:BASF AG
Radiation-curable metal particles and curable resin compositions comprising these particles
InactiveUS6521677B2High refractive indexHigh hardnessMaterial nanotechnologyConductive materialPolymer scienceMeth-
Provided is a liquid curable resin composition which can produce transparent cured products with a high refractive index, high hardness, and superior abrasion resistance, and which can be suitably used as a coating material. The liquid curable resin composition comprises: (A) a poly-functional (meth)acrylic compound having at least three (meth) acryloyl groups in the molecule; (B) a reaction product obtained by the reaction of a compound having a polymerizable unsaturated group and alkoxysilyl group in the molecule and particles metal oxide, the metals being preferably selected from the group consisting of zirconium, antimony, zinc, tin, cerium, and titanium; and (C) a radiation polymerization initiator.
Owner:JSR CORPORATIOON
Catalyst for treatment of waste water, and method for treatment of waste water using the catalyst
InactiveCN102686521AStabilizationImprove purification effectWater contaminantsCatalyst activation/preparationIridiumWater use
Disclosed are: a catalyst which can exhibit an excellent catalytic activity and excellent durability for a long period in the wet oxidation treatment of waste water; a wet oxidation treatment method for waste water using the catalyst; and a novel method for treating waste water containing a nitrogenated compound, in which a catalyst to be used has a lower catalytic cost, the waste water containing the nitrogenated compound can be treated at high purification performance, and the high purification performance can be maintained. The catalyst for use in the treatment of waste water comprises an oxide of at least one element selected from the group consisting of iron, titanium, silicon, aluminum, zirconium and cerium as a component (A) and at least one element selected from the group consisting of silver, gold, platinum, palladium, rhodium, ruthenium and iridium as a component (B), wherein at least 70 mass% of the component (B) is present in a region positioned within 1000 [mu]m from the outer surface of the component (A) (i.e., the oxide), the component (B) has an average particle diameter of 0.5 to 20 nm, and the solid acid content in the component (A) (i.e., the oxide) is 0.20 mmol / g or more. The waste water treatment method uses a catalyst (a pre-catalyst) which is placed on an upstream side of the direction of the flow of the waste water and can convert the nitrogenated compound contained in the waste water into ammoniacal nitrogen in the presence of an oxidizing agent at a temperature of not lower than 100 DEG C and lower than 370 DEG C under a pressure at which the waste water can remain in a liquid state and a downstream-side catalyst (a post-catalyst) which is placed downstream of the direction of the flow of the waste water and can treat the waste water containing ammoniacal nitrogen.
Owner:NIPPON SHOKUBAI CO LTD
Platinum group metal-free catalysts for reducing the ignition temperature of particulates on a diesel particulate filter
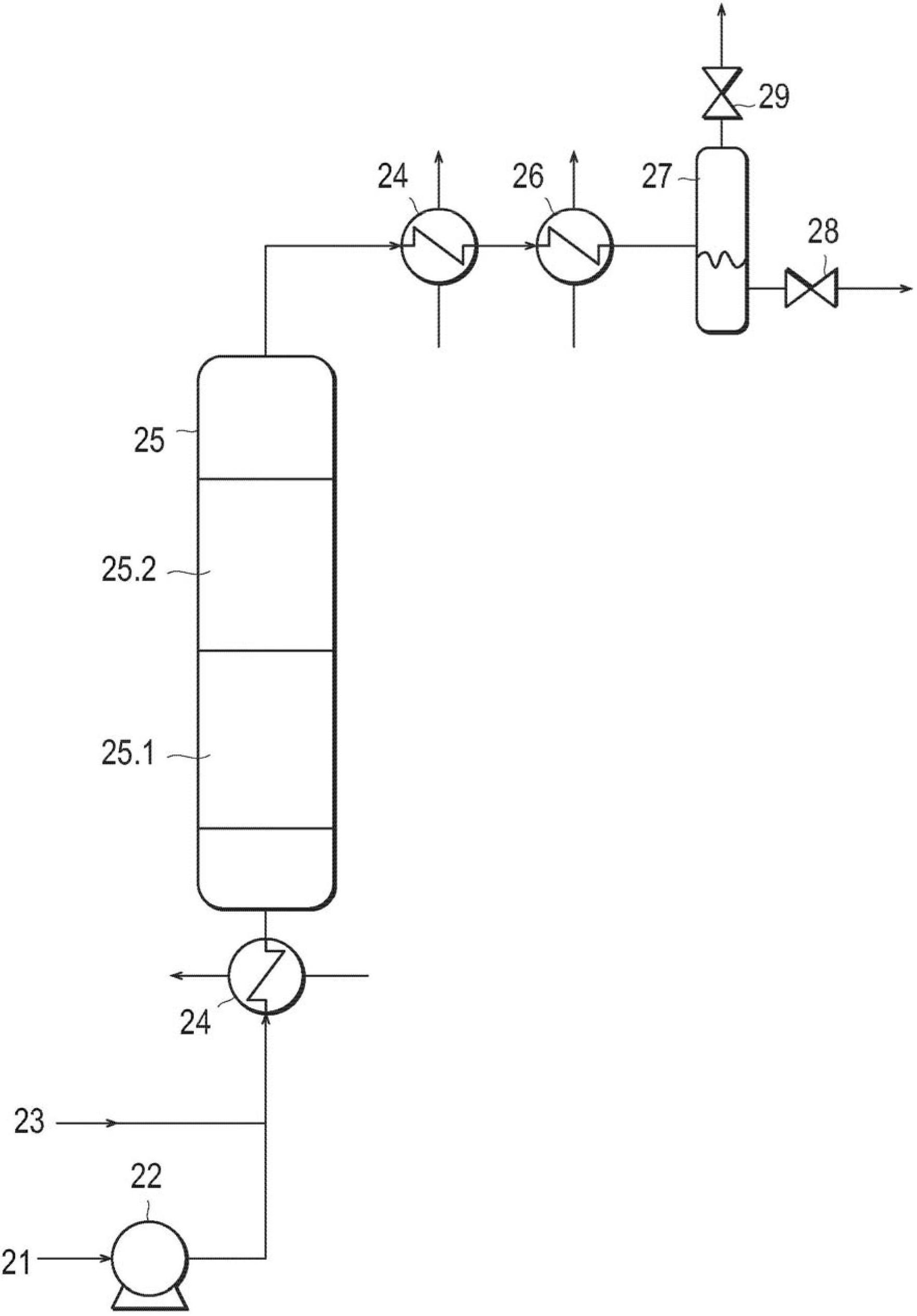
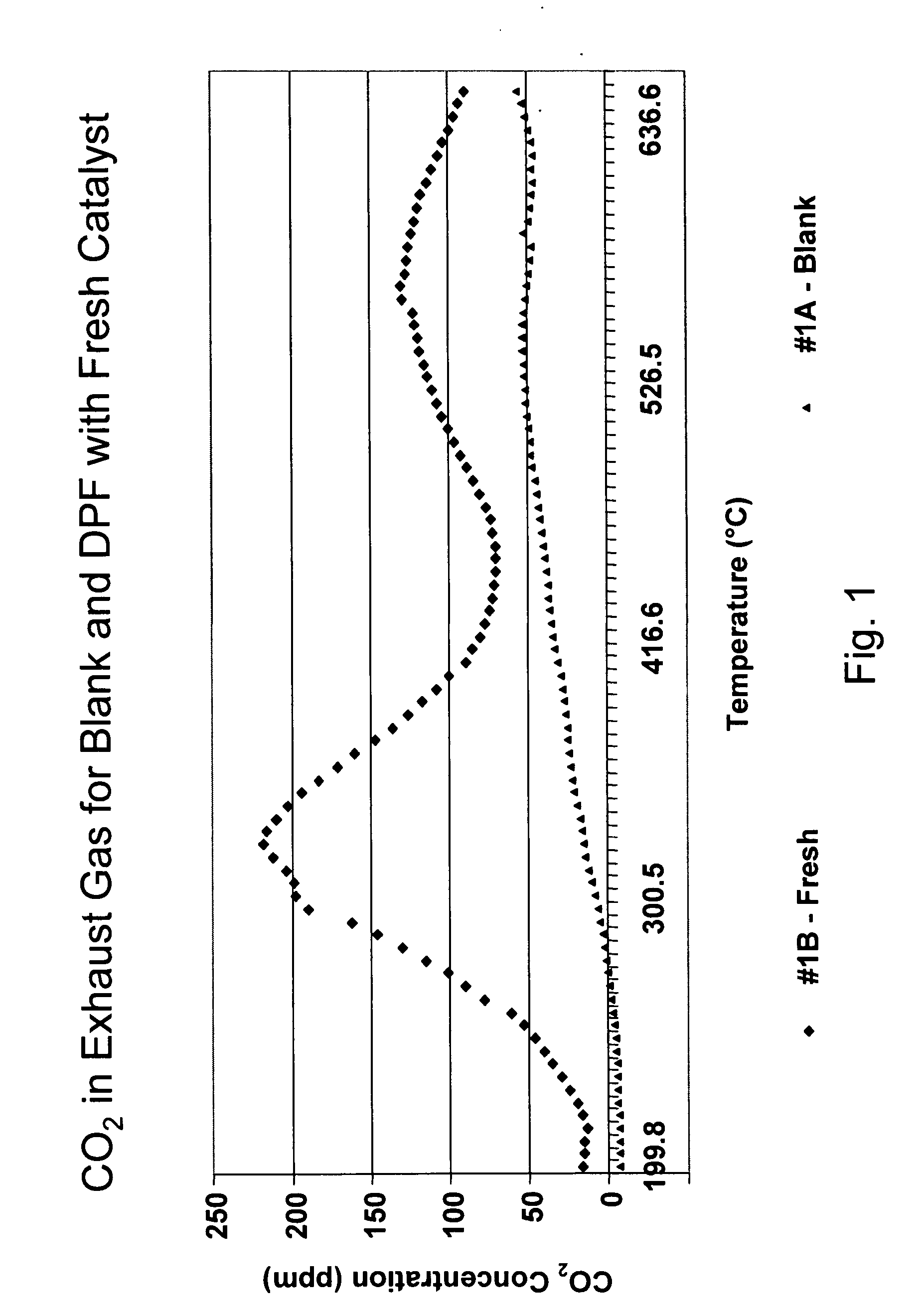
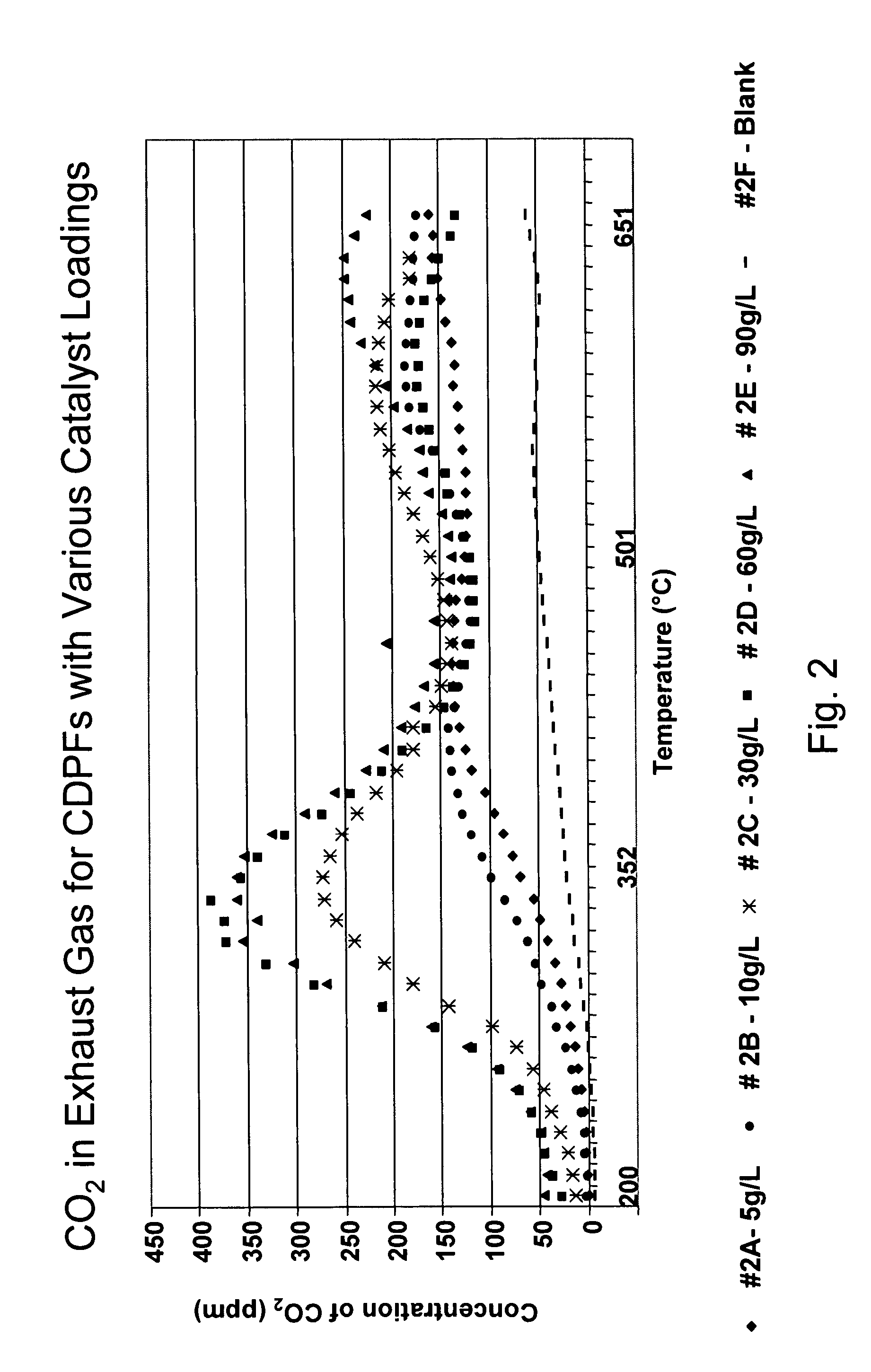
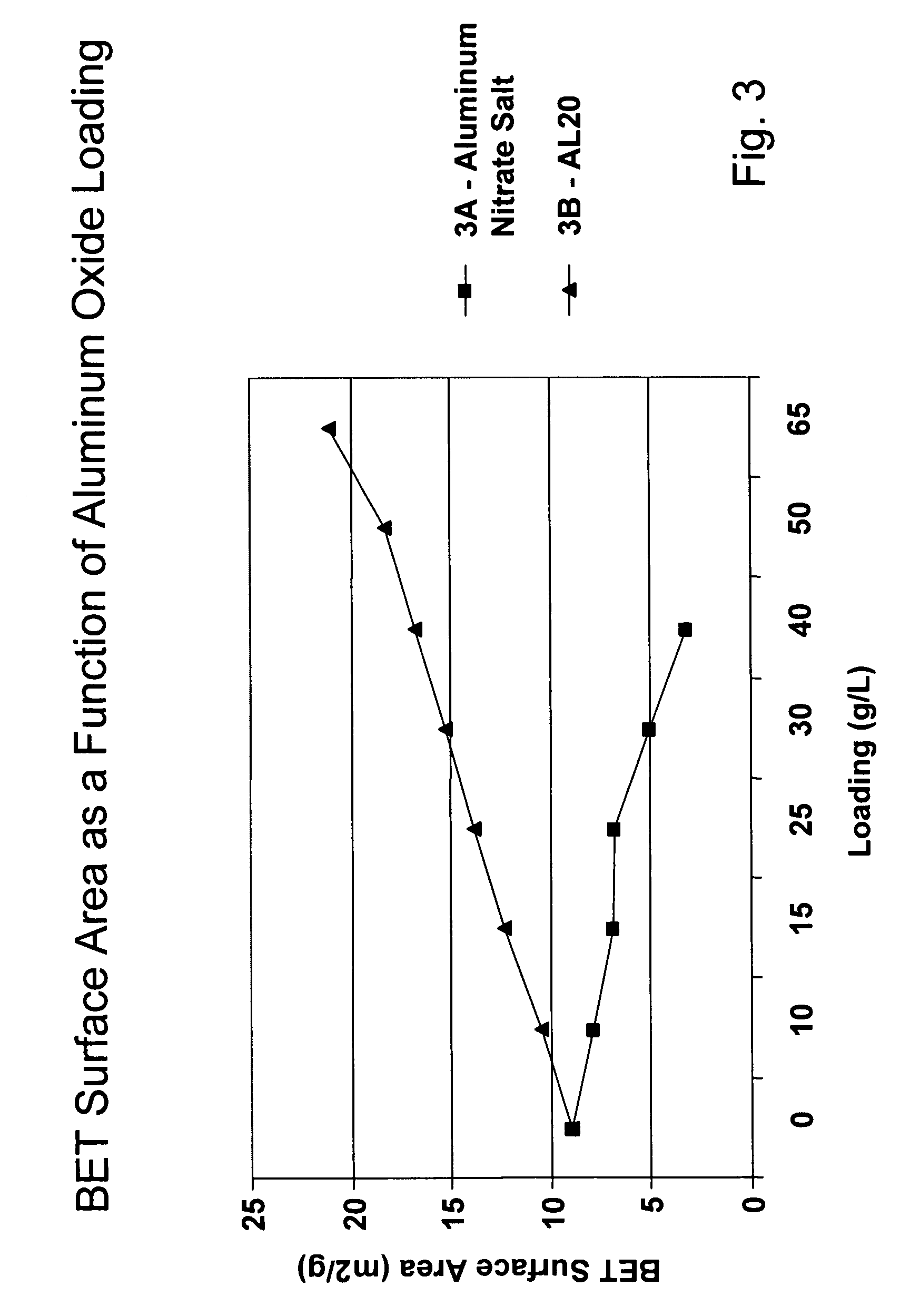
A catalyzed diesel particulate filter (CDPF) and a method for filtering particulates from diesel engine exhaust are provided, where the catalyzed diesel particulate filter includes a substrate and a catalyst composition, where the catalyst composition contains at least one first component, at least one second component, and at least one third component, where the first component is at least one first component selected from the group consisting of cerium and a lanthanide and mixtures thereof, the at least one second component is selected from the group consisting of cobalt, copper, manganese and mixtures thereof; and the third component comprises strontium, where the first component, the second component, and the third component are in an oxide form after calcination. The catalyst on the catalyzed diesel particulate filter lowers the temperature at which particulates are removed from the CDPF by oxidizing the particulates on the filter. The catalyzed diesel particulate filter may also include a washcoat. Washcoats prepared from colloidal aluminum oxide may have higher surface areas and pore volumes loadings than washcoats containing aluminum oxide prepared from aluminum nitrate.
Owner:CATALYTIC SOLUTIONS INC
High performance lithium ion battery anode material lithium manganate and preparation method thereof
The invention provides a high performance lithium ion battery anode material lithium manganate and a preparation method of the material. The lithium manganate is a doped lithium manganate LiMn2-yXy04 which is doped with one kind or a plurality of other metal elements X, wherein X element is at least one kind selected form the group of aluminium, lithium, fluorine, silver, copper, chromium, zinc, titanium, bismuth, germanium, gallium, zirconium, stannum, silicon, cobalt, nickel, vanadium, magnesium, calcium, strontium, barium and rare earth elements lanthanum, cerium, praseodymium, neodymium, promethium, samarium, europium, gadolinium, terbium, dysprosium, holmium, erbium, thulium, ytterbium and lutetium, and y is larger than 0 but less than or equal to 0.11. The lithium ion battery anode material lithium manganate provided in the invention has extraordinary charge and discharge cycle performance both in the environments of normal temperature and high temperature. According to the invention, the preparation method of the material is a solid phase method, the operation is simple and controllable and the cost is low so that it is easy to realize large-scale productions.
Owner:INST OF PROCESS ENG CHINESE ACAD OF SCI
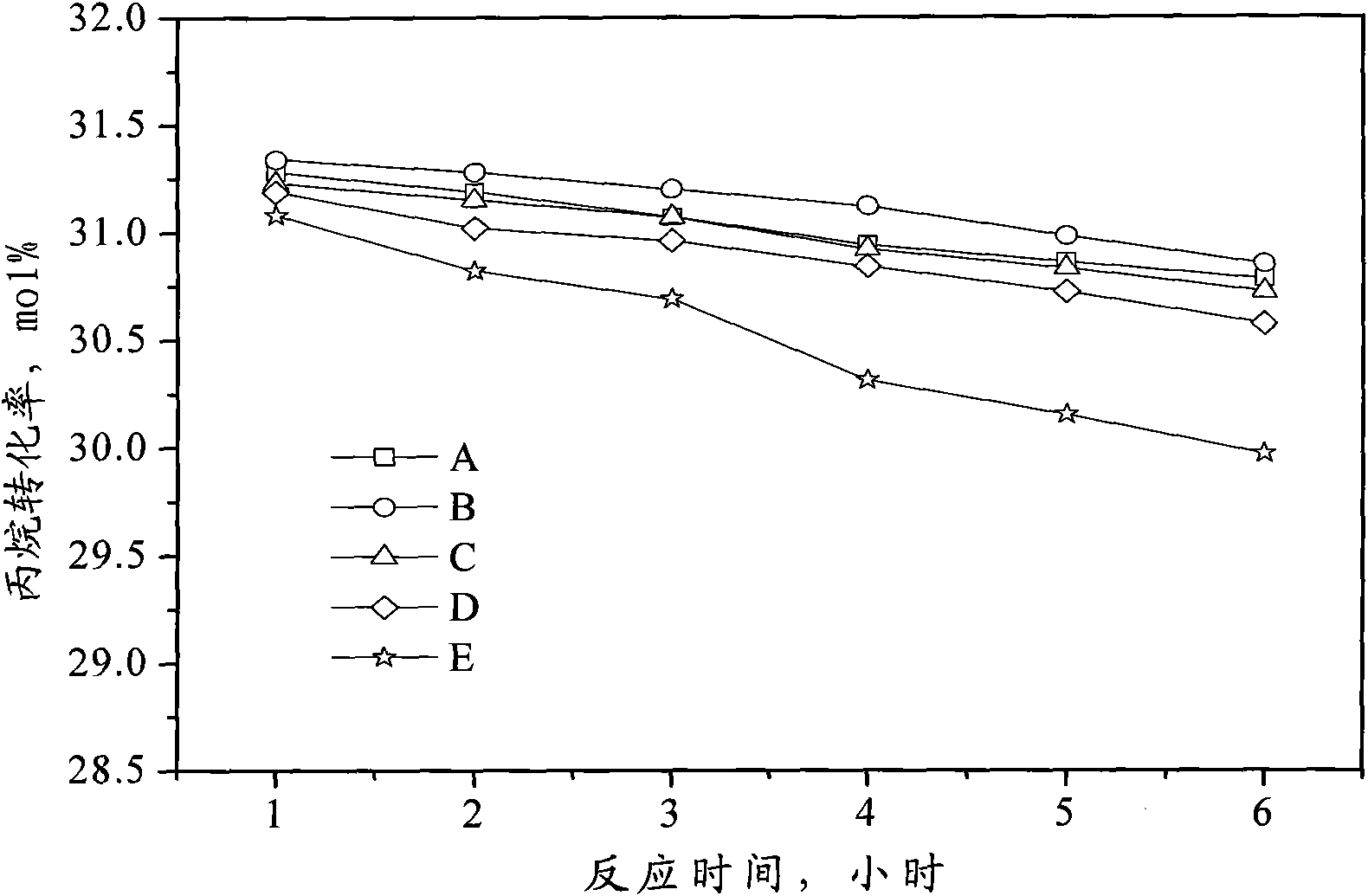
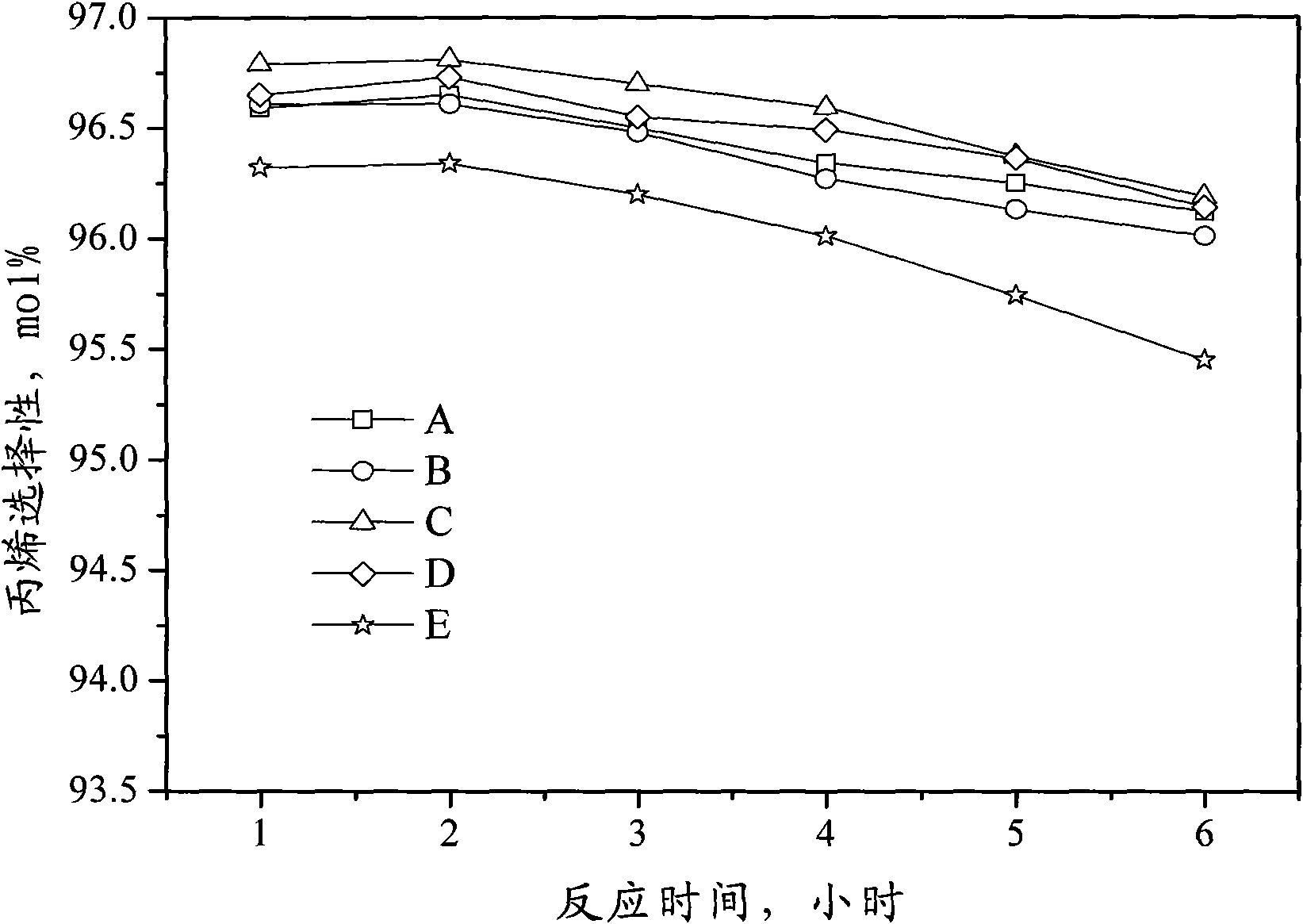
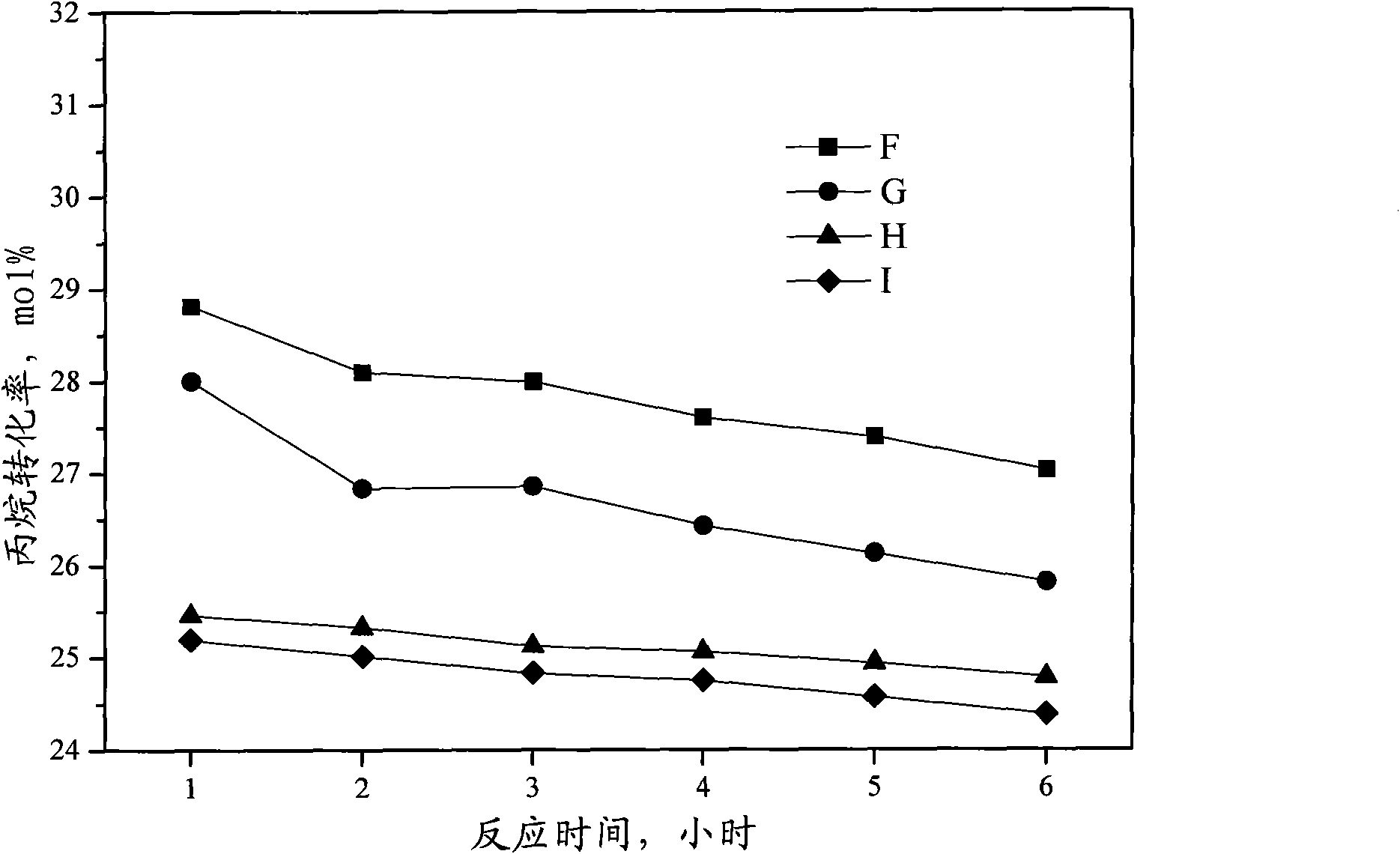
Catalyst for preparing propylene by dehydrogenating propane as well as preparation method and applications thereof
ActiveCN102049267AHigh activityReduce carbon depositionPhysical/chemical process catalystsHydrocarbonsHalogenActive component
The invention relates to a catalyst for preparing propylene by dehydrogenating propane, which comprises an alumina supporter and active components in the following contents by using the alumina supporter as a calculation basis: 0.1-2.0 percent by mass of platinum group metals, 0.1-2.0 percent by mass of IVA group metals, 0.5-5.0 percent by mass of potassium, 0.2-5.0 percent by mass of cerium or samarium, and 0.3-10 percent by mass of halogen. The catalyst is used in the reaction for preparing propylene by dehydrogenating propane. The catalyst has higher conversion rate of propane, higher selectivity of propylene and good regeneration performance.
Owner:CHINA PETROLEUM & CHEM CORP +1
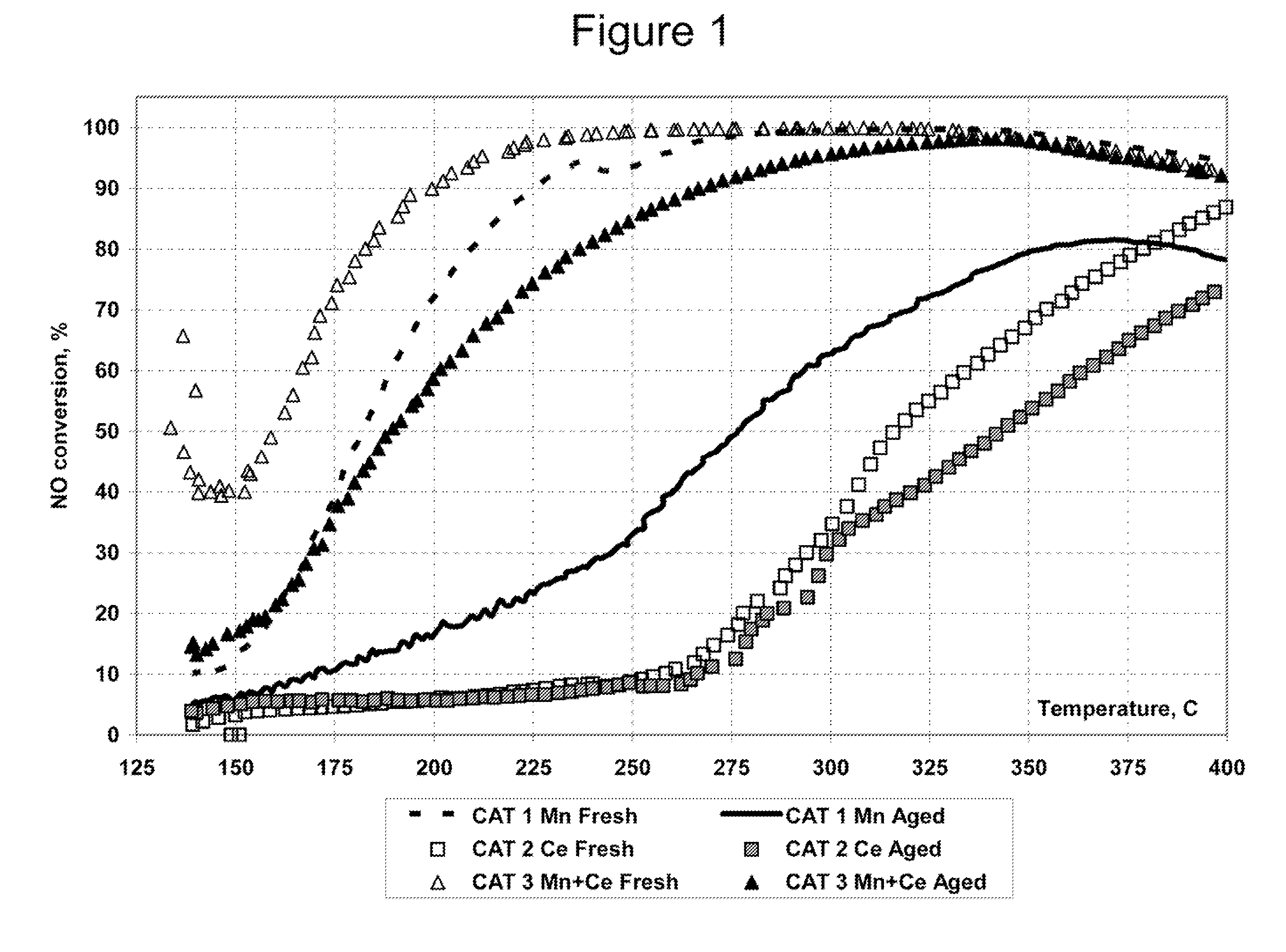
Ammonia scr catalyst and method of using the catalyst
ActiveUS20090304566A1Enhanced Surface AcidityCombination devicesOrganic chemistryParticulatesNiobium
A DPF with an SCR catalyst and a method for selectively reducing nitrogen oxides with ammonia, filtering particulates, and reducing the ignition temperature of soot on a DPF are provided. The catalyst includes a first component of copper, chromium, cobalt, nickel, manganese, iron, niobium, or mixtures thereof, a second component of cerium, a lanthanide, a mixture of lanthanides, or mixtures thereof, and a component characterized by increased surface acidity. The catalyst may also include strontium as an additional second component. The catalyst selectively reduces nitrogen oxides to nitrogen with ammonia and oxidizes soot at low temperatures. The catalyst has high hydrothermal stability.
Owner:CATALYTIC SOLUTIONS INC
Dehydrogenation catalyst for feed gas containing carbon monoxide, preparation method and application method thereof
ActiveCN101543776AWide range of CO content requirementsGood choiceCarbon monoxideMetal/metal-oxides/metal-hydroxide catalystsPlatinum saltsCerium
The invention discloses a dehydrogenation catalyst for feed gas containing carbon monoxide, a preparation method and an application method thereof. The catalyst takes alumina as a carrier, palladium and / or platinum as an active component and 2 to 4 MOxes as an additive, M is sodium, potassium, magnesium, titanium, zirconium, vanadium, manganese, iron, nickel, cobalt, copper, molybdenum, tungsten or cerium, and components of the catalyst (calculated by carrier mass) are: 0.01 to 2 percent of the palladium and / or 0.01 to 1 percent of the platinum, and 1 to 2 percent of MOxes. The preparation method for the catalyst comprises the following steps that: firstly, an additive metal salt solution is used to impregnate the carrier; a palladium and / or platinum salt solution is used to impregnate the carrier after the carrier is dried and roasted; and the impregnated carrier is roasted at a temperature of between 450 and 850 DEG C to obtain the catalyst. Before the use, H2-N2 mixed gas containing more than 10 percent of hydrogen or pure hydrogen is activated by the catalyst at a temperature of between 450 and 650 DEG C. The catalyst can perform deep removal on less than 5 percent of hydrogen in the feed gas with the CO content of between 10 and 99 percent, the using temperature is between 100 and 300 DEG C, the space velocity is between 500 and 9,000h<-1>, the dehydrogenation rate is more than 99 percent, the content of outlet hydrogen is less than 100 ppm, and the loss of the carbon monoxide is less than 0.5 percent.
Owner:HAISO TECH
Catalytic converter for cleaning exhaust gas
InactiveUS6261989B1Reduce and preventEasy to cleanNitrogen compoundsInternal combustion piston enginesCeriumCe element
A catalytic converter for cleaning exhaust gas includes a heat-resistant support, and a catalytic coating formed on the heat-resistant support. The catalytic coating contains Pd-carrying particles of a cerium complex oxide, Pt & Rh-carrying particles of zirconium complex oxide, and particles of a heat-resistant inorganic oxide.
Owner:DAIHATSU MOTOR CO LTD +1
Inorganic material that has metal nanoparticles that are trapped in a mesostructured matrix
An inorganic material that consists of at least two elementary spherical particles, each of said spherical particles comprising metal nanoparticles that are between 1 and 300 nm in size and a mesostructured matrix with an oxide base of at least one element X that is selected from the group that consists of aluminum, titanium, tungsten, zirconium, gallium, germanium, tin, antimony, lead, vanadium, iron, manganese, hafnium, niobium, tantalum, yttrium, cerium, gadolinium, europium and neodymium is described, whereby said matrix has a pore size of between 1.5 and 30 nm and has amorphous walls with a thickness of between 1 and 30 nm, said elementary spherical particles having a maximum diameter of 10 μm. Said material can also contain zeolitic nanocrystals that are trapped within said mesostructured matrix.
Owner:INST FR DU PETROLE
Liquid composition, process for its production and process for producing membrane-electrode assembly for polymer electrolyte fuel cells
ActiveUS20060019140A1Improve the immunityIncreased durabilityIon-exchanger regenerationFinal product manufacturePolymer scienceHigh energy
An electrolyte membrane is prepared from a liquid composition comprising at least one member selected from the group consisting of trivalent cerium, tetravalent cerium, bivalent manganese and trivalent manganese; and a polymer with a cation-exchange group. The liquid composition is preferably one containing water, a carbonate of cerium or manganese, and a polymer with a cation-exchange group, and a cast film thereof is used as an electrolyte membrane to prepare a membrane-electrode assembly. The present invention successfully provides a membrane-electrode assembly for polymer electrolyte fuel cells being capable of generating the electric power in high energy efficiency, having high power generation performance regardless of the dew point of the feed gas, and being capable of stably generating the electric power over a long period of time.
Owner:ASAHI GLASS CO LTD
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com