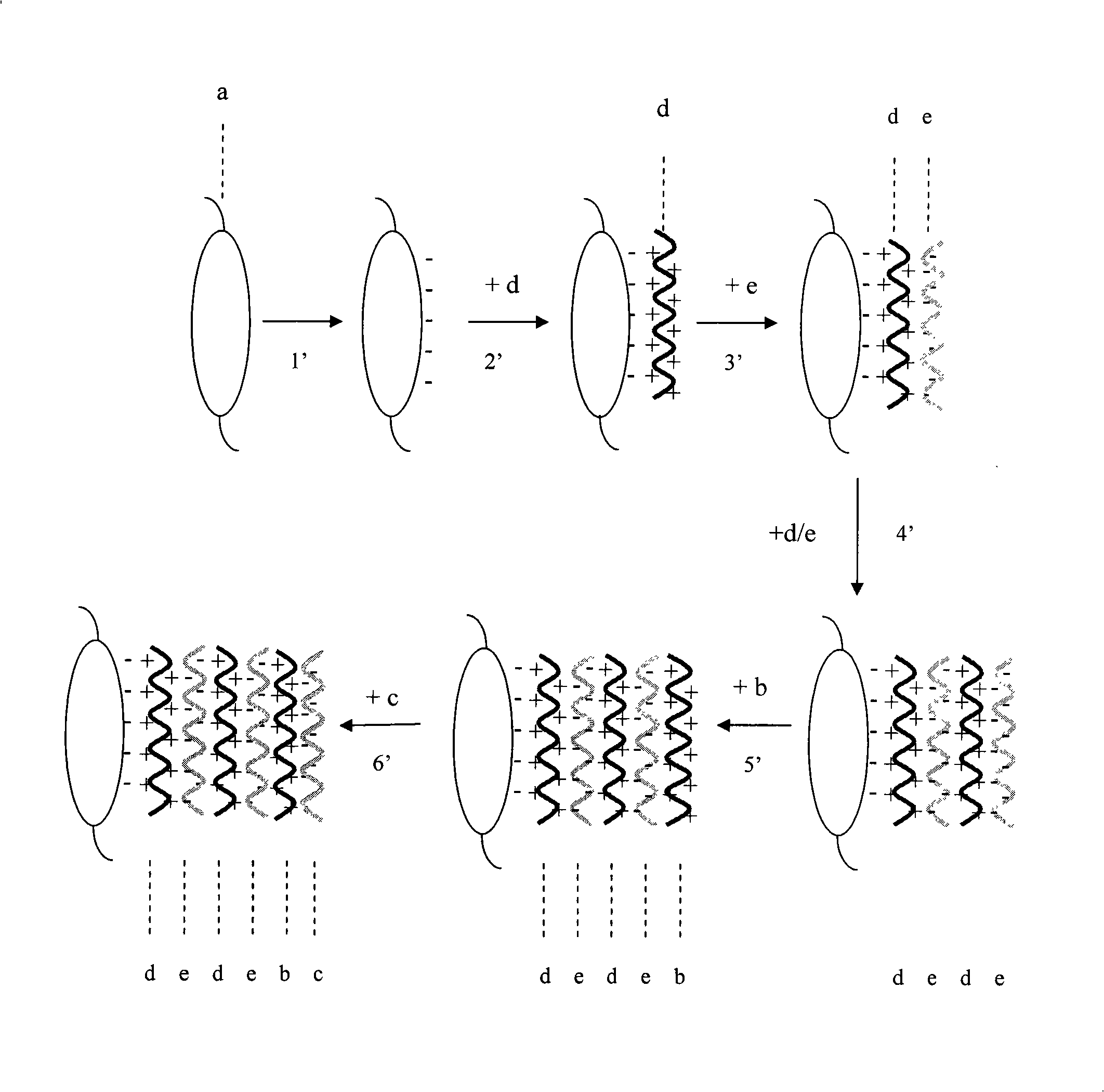
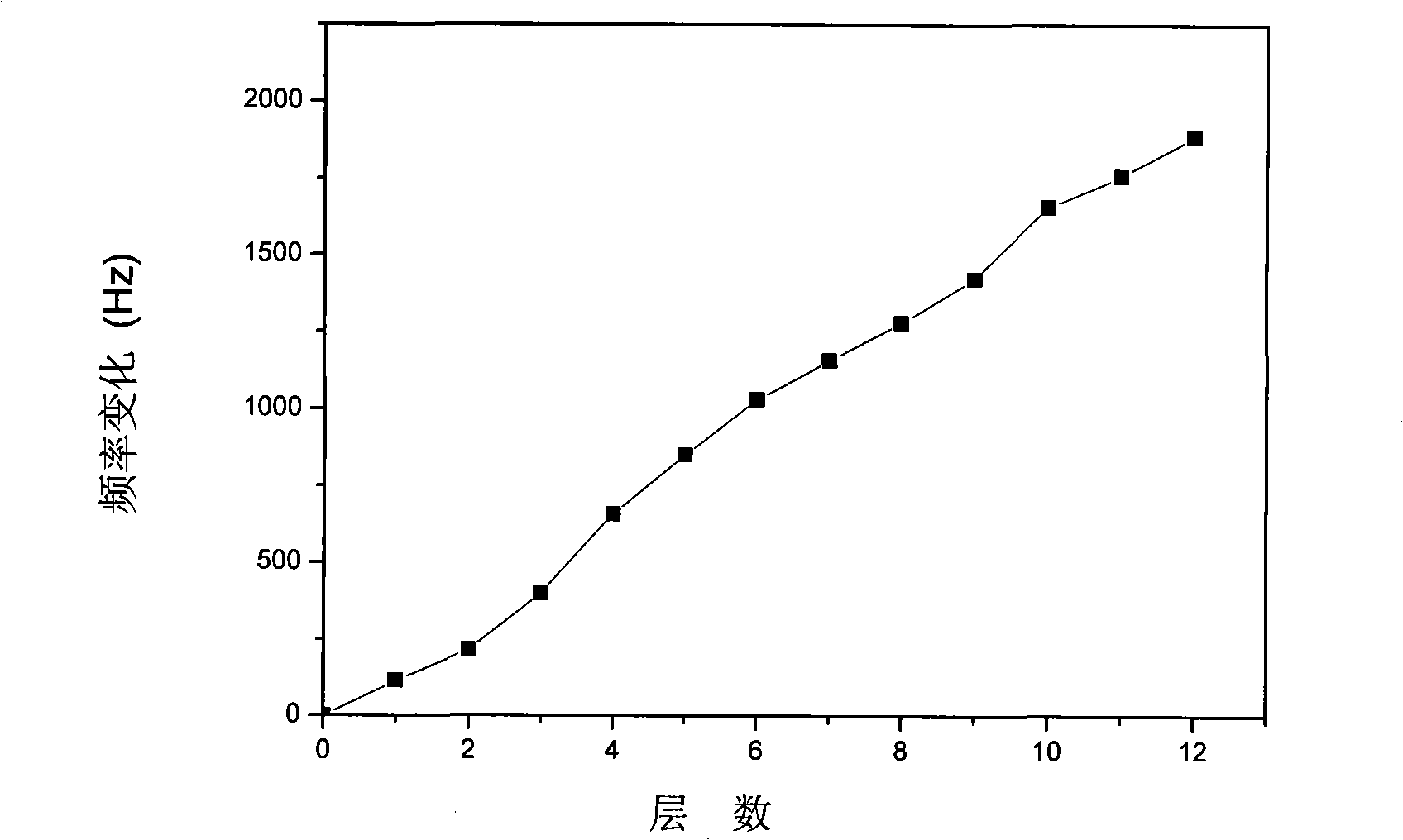
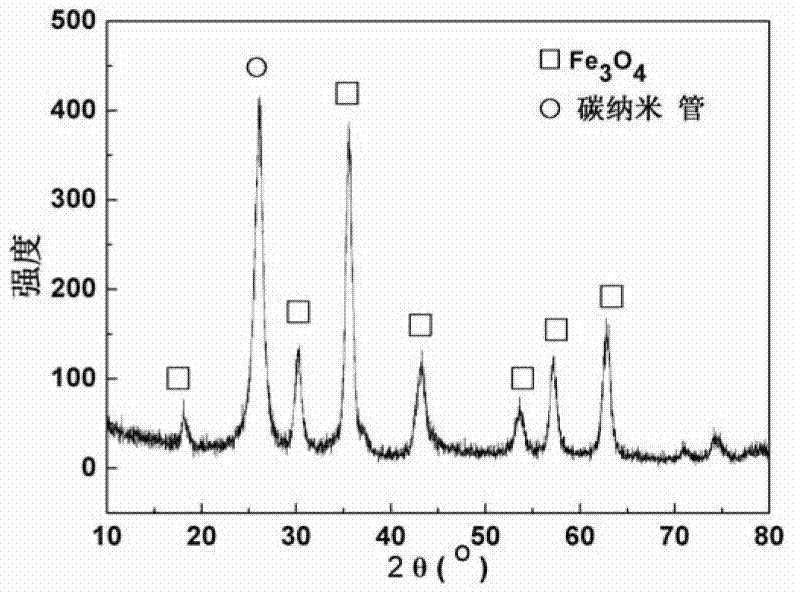
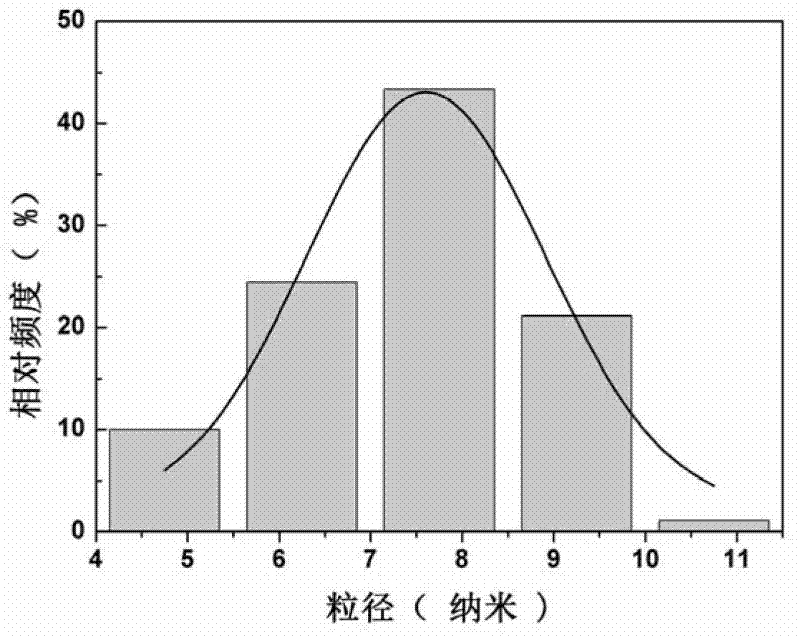
Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
185results about How to "Change pH" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
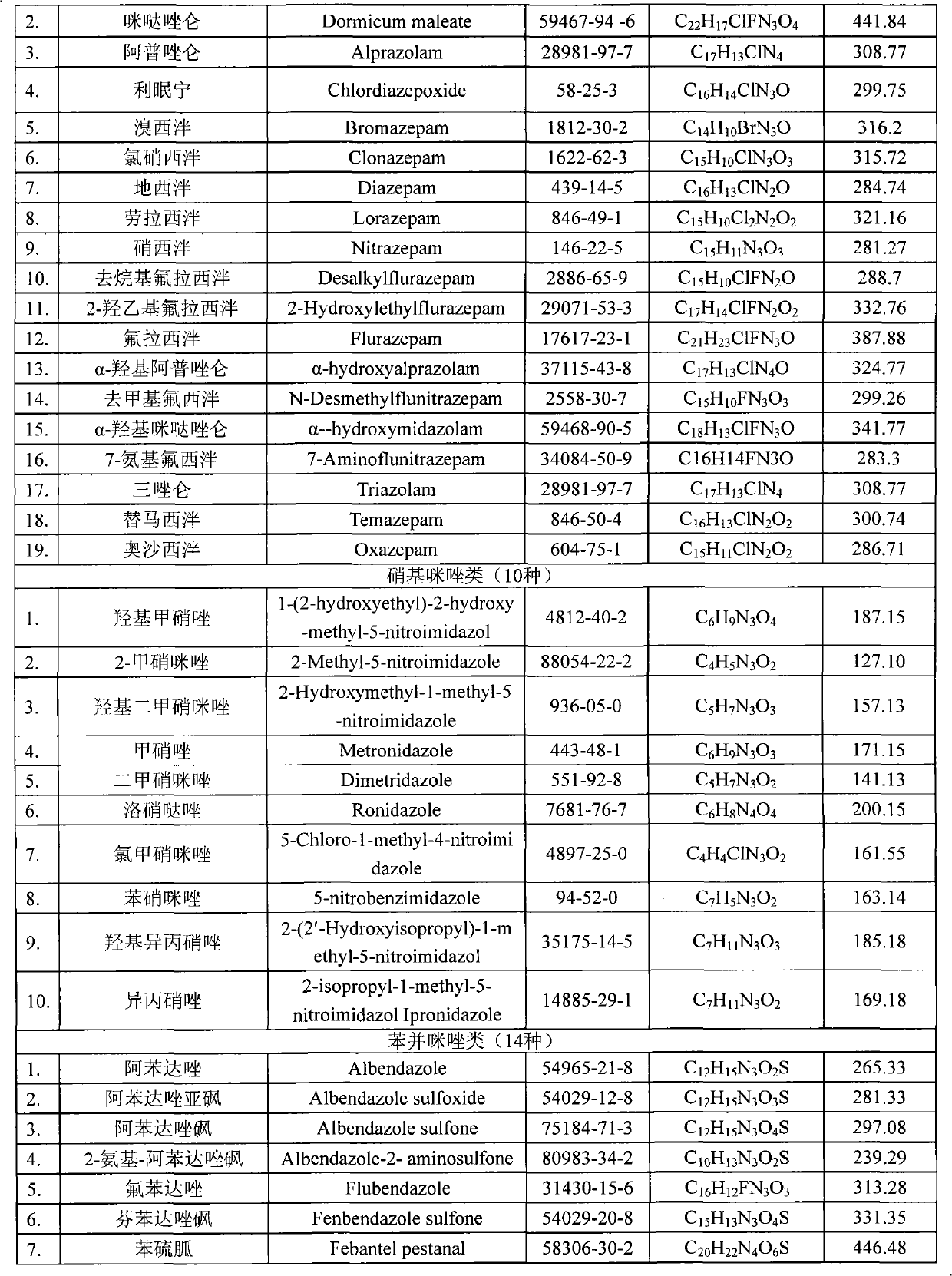
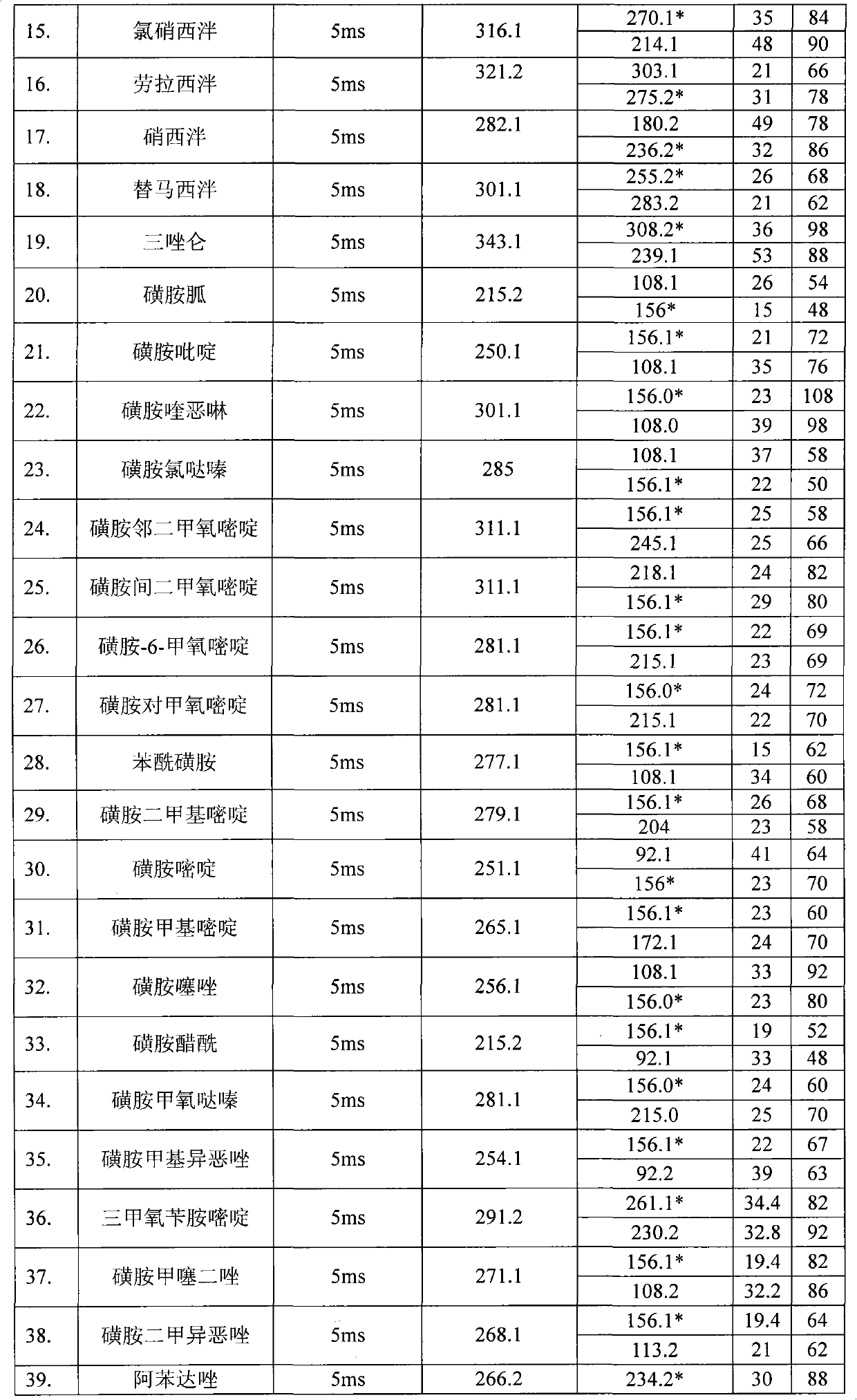
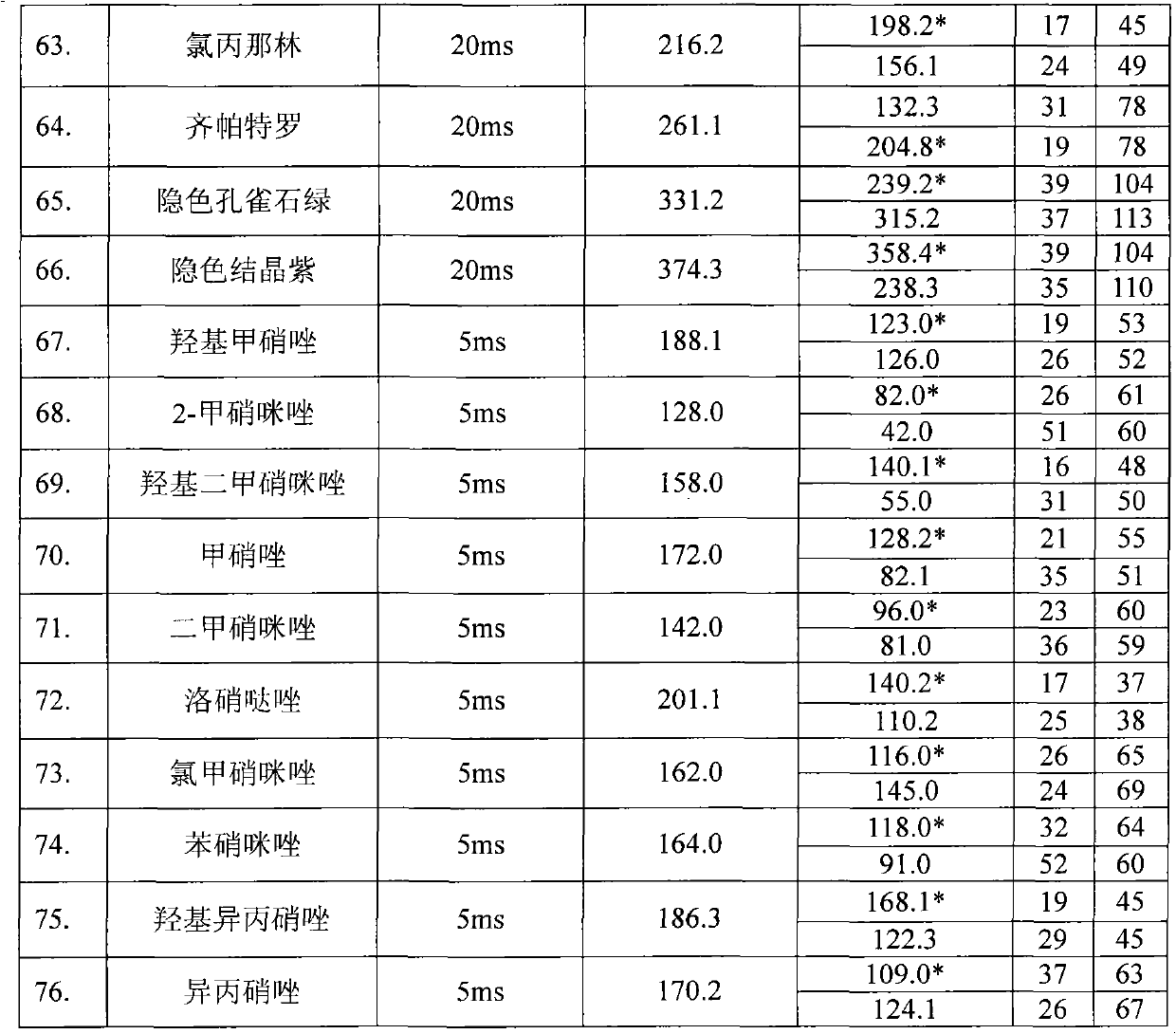
Method for detecting residual quantity of multiple alkaline drugs in animal derived food
The invention relates to the fields of analytical chemistry and food safety, in particular to a method for detecting the residual quantity of multiple alkaline drugs in animal derived food. Based on the vortex mixed extracting of acetonitrile, isopropanol and citric acid buffer solutions, the purification of a hydrophilic polystyrene-divinylbenzene solid phase extraction column and a cation exchange solid phase extraction column and the liquid phase chromatography-mass spectra determination, the method can detect the residual quantity of multiple alkaline drugs in pork, pork liver, eggs, shrimps and milk, such as beta-receptor agonists,sulfonamides, benzodiazepines, nitroimidazoles, benzimidazoles and triphenylmethanes. The method has the advantages of simple operation, fast and accurate detection and high efficiency.
Owner:SHANGHAI ANPEL SCI INSTR +1
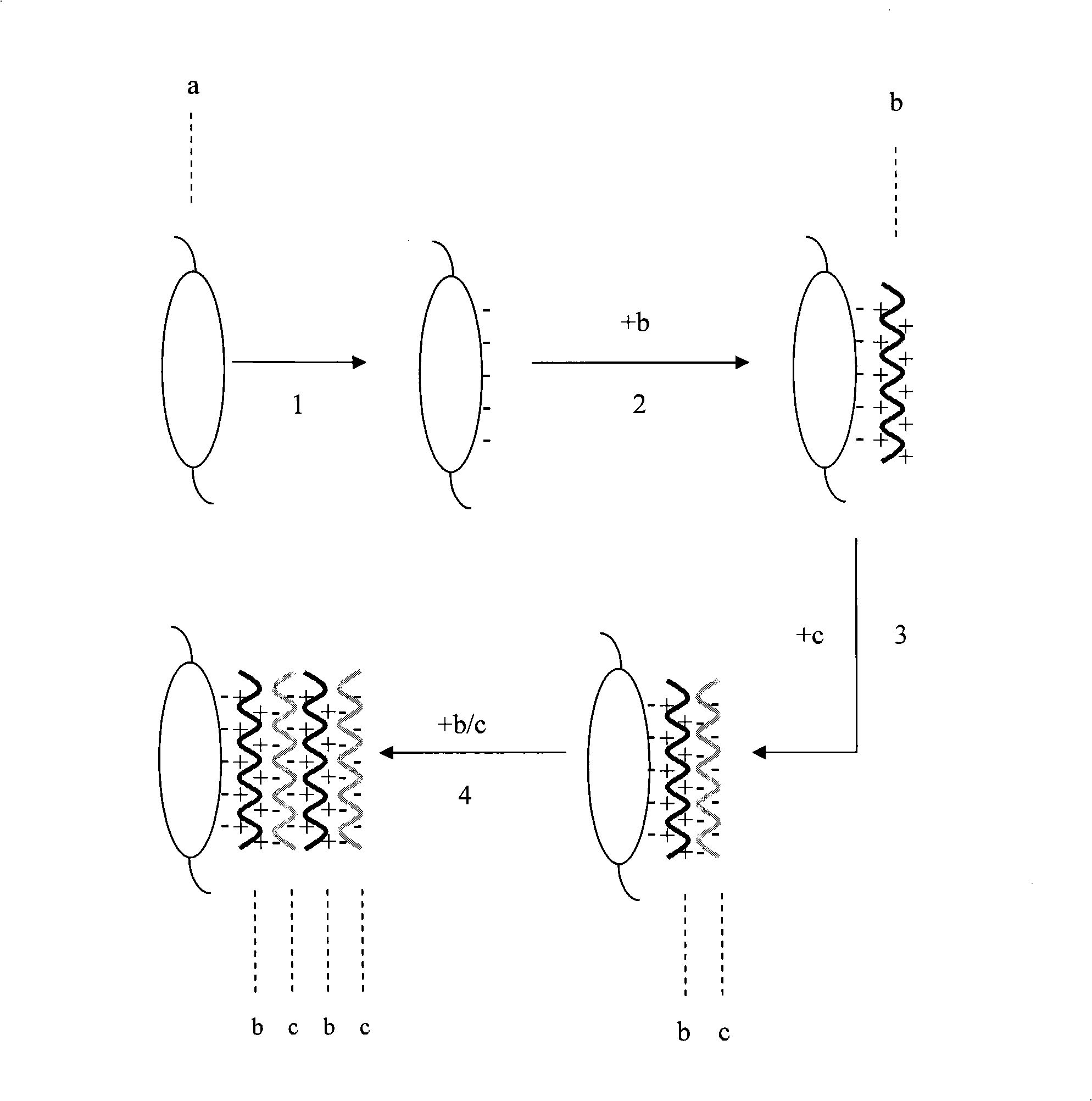
Artificial crystalline len with transforming growth factor resistant beta2 antibody membrane on surface and manufacturing method thereof
The invention provides an artificial lentis, which contains anti-transforming growth factor beta 2 antibody membrane on the surface and can inhibit intercurrent post-cataract after cataract surgery, and also provides a the production method thereof. The production method includes the steps that: the artificial lentis is charged with positive electricity or negative electricity after the artificial lentis is cleaned, dried, and pretreated on the surface; the artificial lentis is soaked in a polyelectrolyte solution the charge of which is opposite to the surface charge of the artificial lentis for adsorbing, and rinsing the artificial lentis by deionized water, and drying the artificial lentis by nitrogen gas; the artificial lentis is soaked in a phosphate buffering solution of anti-transforming growth factor beta 2 antibody, the pH value of which is 4-10, and the carried charge of which is opposite to that of the polyelectrolyte, for adsorption; finally, the artificial lentis is rinsed by phosphate buffering solution, and the artificial lentis is dried by nitrogen gas; the alternating assembly steps are repeated. The artificial lentis of the invention can inhibit the transformation and differentiation as well as cyst membrane shrinkage of the lentis epithelial cells in a target way, and then interdicts the occurrence of the post-cataract, and has excellent biocompatibility. The production method of the invention is scientific and simple, and can ensure the activity under a dry state and the safety and reliability during medical transplantation of the anti-transforming growth factor beta 2.
Owner:SECOND AFFILIATED HOSPITAL ZHEJIANG UNIV COLLEGE OF MEDICINE
Multi-walled carbon nanotube-loaded nano ferroferric oxide catalyst and preparation method and application thereof
ActiveCN102671661AOvercoming technical difficultiesHigh activityMetal/metal-oxides/metal-hydroxide catalystsOrganic synthesisCarbon nanotube
The invention discloses a multi-walled carbon nanotube-loaded nano ferrofferic oxide (Fe3O4) catalyst and a preparation method and an application thereof. In the multi-walled carbon nanotube-loaded nano ferrofferic oxide catalyst, ferroferric oxide is distributed on the surface of the outer wall of the multi-walled carbon nanotube. The preparation process is simple, the cost is low, and easiness in industrialized popularization is realized. In the synthesized catalyst, the crystallinity of the Fe3O4 nano particles is high, the particle size is controllable, and the particles distribution is narrow. The catalytic activity of the multi-walled carbon nanotube-loaded nano ferrofferic oxide (Fe3O4) catalyst multi-walled carbon nanotube-loaded nano ferrofferic oxide (Fe3O4) catalyst (Fe3O4-MWCNTs) is high and the Fe3O4-MWCNTs is easy for being repeatedly used through magnetic separation. The Fe3O4-MWCNTs which is prepared by the method can be used as peroxide mimic enzyme and can be used as catalyst for organic pollutants which are difficult to degrade and for organic synthesis.
Owner:TSINGHUA UNIV
Preparation method and using method of human mesenchymal stem cell exosome freeze-dried powder
ActiveCN107245472AEasy recyclingImprove cortexPowder deliveryCulture processUltrafiltrationPlastic surgery
The invention provides a preparation method and a using method of human mesenchymal stem cell exosome freeze-dried powder, used for solving the problems of exosome such as low separation and recovery quantity, poor quality and low efficiency. The preparation method of the human mesenchymal stem cell exosome freeze-dried powder comprises the following steps: 1) performing culture, purification and passage on human mesenchymal stem cells; 2) inducing large-scale synthesis of the human mesenchymal stem cells and secreting functional exosome; 3) performing ultrafiltration and concentration on the exosome of the human mesenchymal stem cells, preparing a concentrated culture solution, and refrigerating; 4) preparing the freeze-dried powder from the refrigerated concentrated culture solution, and adding complex liquid into the freeze-dried powder for preserving. According to the method disclosed by the invention, synthesis of the human mesenchymal stem cells and secretion of the exosome can be effectively induced to the greatest degree, and the active ingredients are obtained on a large scale and are conveniently and directly used for various aspects of massive health, such as diagnosis and treatment of diseases and cosmetic plastic surgery of the human body.
Owner:北京智能宝生物科技有限公司
Microfluidic chip electrophoretic-electrochemical detecting device with adjustable pH after separation and use thereof
InactiveCN102788831AChange pHDoes not affect electrophoretic separationMaterial analysis by electric/magnetic meansLaboratory glasswaresElectrode placementAntibiotic Y
The invention discloses a microfluidic chip electrophoretic-electrochemical detecting device with adjustable pH after separation and use of the device in aminoglycoside antibiotics analysis. The analysis device comprises a double-T shaped sampling channel, a separating channel, two auxiliary channels, a W-shaped channel and an electrode placement channel. Through adding the two auxiliary channels and the W-shaped channel at the tail end of the separating channel in a microfluidic chip, an alkaline solution is added through the auxiliary channels after being electrophoretically separated, and the solution from the separating channel and the auxiliary channels is mixed evenly through the W-shaped channel, a pH value of the solution in a working electrode determining region is improved, so that the solution meets a strong alkaline condition needed by the detection of carbohydrates, simultaneously, electrophoretic separation under an acidic separation buffer solution is not influenced. By the device disclosed by the invention, a transition metal nano material is decorated on the surface of the electrode by using an electrodeposition method and by means of the relatively large specific surface area and the special catalytic property of the metal nano material, so that the working electrode with high performance is obtained, and aminoglycoside antibiotics are determined through the amperometry.
Owner:GRADUATE SCHOOL OF THE CHINESE ACAD OF SCI GSCAS +1
Process for synthesizing benzaldehyde by selective oxidation of toluene
InactiveCN1663941AChange distributionChange pHOrganic compound preparationCarbonyl compound preparationBenzaldehydeReaction temperature
Disclosed is a process for synthesizing benzaldehyde by selective oxidation of toluene, wherein the catalyst mainly comprises manganese, copper, cobalt, ferrum, nickel, zirconium, zinc, Argentine or / and halides of sodium, calcium, potassium, nitrates, sulfates, acetates, benzoates. The preparing process comprises letting in oxygen at the presence of the catalyst. When the conversion rate of toluene is 10%, the selectivity of benzaldehyde is 62%, the selectivity of benzoic alcohol is 14%, the selectivity of benzene carbonic acid is 22%, when the conversion rate of toluene is 39%, the selectivity of benzaldehyde is 20%, the selectivity of benzoic alcohol is 5%, the selectivity of benzene carbonic acid is 73%.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
Active component extraction method of plant polyphenol kind substance
ActiveCN1824084AAvoid oxidation reactionsHigh activityMagnoliophyta medical ingredientsPlant ingredientsMicrowaveActive component
A process for extracting the active polyphenol substances from the natural plants includes such steps as washing, drying, pulverizing, loading it along with the deoxidizing purified water in an extractor, heating at 50-90 deg.C under the protection of inertial gas while stirring for 30-60 min in order to extract the active components or ultrasonic or microwave extracting for 10-40 min 1-5 times, collecting liquid extract, filtering, cooling concentrating and drying.
Owner:谢君
Soil conditioner for reducing cadmium and lead pollution to crop
InactiveCN101724405AChange pHImprove adsorption capacitySolid waste managementOrganic fertilisersLeafy vegetablesPb contaminated soil
The invention relates to a soil conditioner for reducing the cadmium and lead pollution to crops, comprising inorganic components and organic components, wherein the effective components of the inorganic components are Ca(OH)2 and Na2SiO7 or mixtures of the Ca(OH)2 and the Na2SiO7; and the organic components include rice straws and tephrosia caudida casts or mixtures of the rice straws and the tephrosia caudida casts. The soil conditioner is used before the crops are cultivated; the inorganic components are prepared into agricultural-grade powder and 0.5-4 g of the inorganic components is used for one kilogram of wind drying soil; and the grain diameter of the crushed organic components is less than 1 cm and 5-40 g of the organic components is used for one kilogram of wind drying soil. The soil conditioner effectively reduces the biological effectiveness of heavy metal, the absorption of cadmium and lead by the crops and the heavy metal pollution to the crops and especially leafy vegetables without reducing the production property of the corps.
Owner:SUN YAT SEN UNIV
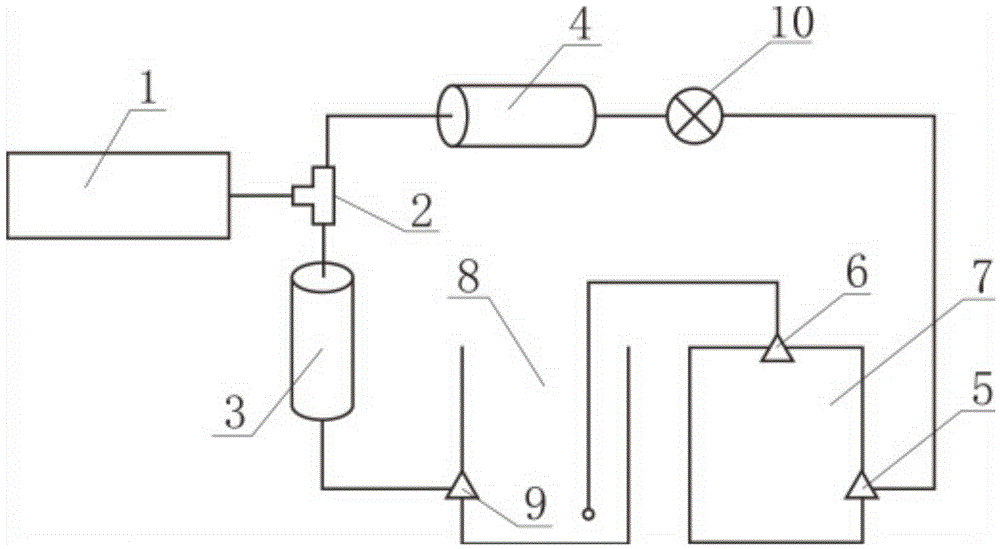
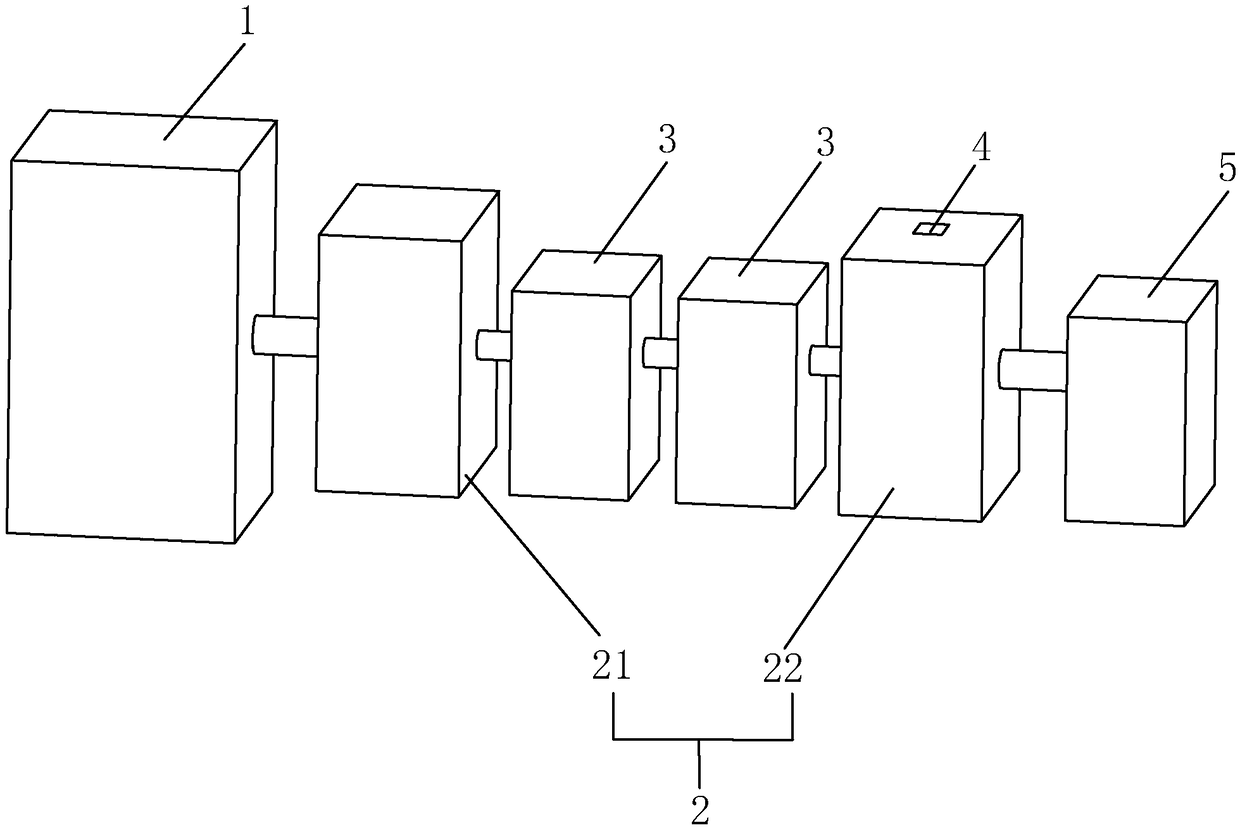
Device and method for filling medical infusion bag with hydrogen in non-contact manner
ActiveCN104398475AImprove solubilityShorten the timePharmaceutical delivery mechanismOpen waterSolubility
The invention relates to a technique of filling an infusion bag with hydrogen and discloses a device for filling a medical infusion bag with hydrogen in a non-contact manner. The device comprises a hydrogen generator (1), a gas and liquid mixer (2), a positive displacement pump (3), a vane pump (4), an open water tank (8), a pressure regulation valve (10), and a sealed water tank (7). By means of the hydrogen generator, the solubility of the hydrogen is improved while any characteristic of water is not changed. The positive displacement pump and the vane pump are used, so the hydrogen is processed into micro-nanometer bubbles, the solubility of the hydrogen in water is greatly improved, and the dissolving time of the hydrogen is shortened. The invention further discloses a method for filling the medical infusion bag with the hydrogen in a non-contact manner. The method comprises the steps of mixing gas and liquid, dissolving the gas under high pressure and releasing the micro-nanometer hydrogen bubbles under low pressure. The solubility of the hydrogen is high, the consumed time is short, the production cost is saved, the purity of the product is improved, the content of impurities is reduced, it is guaranteed that bacteria and heat sources are avoided in the operation process, and accordingly clinical requirements for injection are met.
Owner:SHANGHAI NANOBUBBLE TECH
SCR (Selective Catalytic Reduction) de-nitration catalyst based on natural manganese iron ore as well as preparation method and application method thereof
InactiveCN107282066AImprove denitrification effectExtended service lifeHeterogenous catalyst chemical elementsDispersed particle separationNitrationManganese oxide
The invention discloses an SCR (Selective Catalytic Reduction) de-nitration catalyst based on natural manganese iron ore as well as a preparation method and application method thereof, and belongs to the technical field of atmospheric pollution. The SCR de-nitration catalyst is characterized in that the natural manganese iron ore containing ferric oxide, manganese oxide and aluminum oxide is taken as a raw material, and is smashed, ground, screened, and calcined to obtain powder-like particles; during de-nitration, SCR de-nitration catalyst powder is covered with heat preservation cotton and is fixedly arranged at the axis of an entrance of a flue gas pipe behind an ESP (Electrostatic Precipitator). The SCR de-nitration catalyst is convenient to produce, is easy to use, has a relatively good de-nitration effect at low temperature, capability of solving the problems of relatively low de-nitration efficiency and slightly-high application temperature section existing in an conventional de-nitration catalyst, and contribution to prolonging the service life of the catalyst, and the cost is reduced.
Owner:ANHUI UNIVERSITY OF TECHNOLOGY
Magnetic field controllable sustained-release magnetic substance thickening liquid flow polishing pad and polishing method
ActiveCN110788743AExtended service lifeShorten the timeLapping toolsAqueous solutionMagnetic field magnitude
The invention provides a magnetic field controllable sustained-release magnetic substance thickening liquid flow polishing pad. The polishing pad is a multilayer superimposed porous wear-resistant polishing pad. Pores are reserved in the polishing pad; abrasive capsules are embedded in the holes and contain a magnetic abrasive, a thickening phase and an acid-base agent. The invention discloses a polishing method applying the magnetic field controllable sustained-release magnetic substance thickening liquid flow polishing pad. After the magnetic field is applied, the abrasive capsules in the polishing pad are subjected to the action of the magnetic field; the magnetic abrasive in the abrasive capsules impacts shells of the capsules; when the magnetic field intensity is sufficient, the capsules are broken by impact, the polishing substance is released along the pores, and is gradually dispersed and dissolved in an aqueous solution to form sustained-release magnetic substance thickening liquid flow polishing liquid. After the magnetic field is removed, the abrasive capsules stop releasing the polishing substance, and the concentration of the polishing liquid no longer changes. Duringpolishing, the magnetic field acts on the magnetic abrasive. On the basis of the thickening phase thickening liquid flow, the thickening liquid flow characteristics of the polishing liquid are furtherenhanced, and the polishing efficiency is improved. The polishing pad has long service life and high polishing efficiency.
Owner:HUNAN UNIV OF SCI & TECH
Special biological organic fertilizer for iron-rich green vegetables as well as preparation method and application of fertilizer
The invention provides a special biological organic fertilizer for iron-rich green vegetables as well as a preparation method and an application of the fertilizer. The organic fertilizer is prepared from the following components in parts by weight: 60-80 parts of edible mushroom residues, 15-35 parts of humic acid, 5-15 parts of rapeseed dregs, 5-10 parts of iron in a chelating state and ferric citrate and 0.2-2 parts of a compound microbial agent. By using edible mushroom residues, humic acid and rapeseed dregs as raw materials, adding trace element iron and taking bacillus amyloliquefaciens and trichoderma aureoviride as special effect microbial bacteria, the biological organic fertilizer provided by the invention has high fertilizer efficiency and has multiple functions of improving physical and chemical properties of soil, effectively reducing soil-borne disease, improving the crop output and nutritional quality and the like.
Owner:SICHUAN HUAZHI BIOLOGICAL ENG
Synthetic system for killing soil root nematode by heat injection in root position
InactiveCN103340193AChange pHHigh degree of automationInsect catchers and killersNematodeProgrammable logic controller
The invention discloses a synthetic system for killing soil root nematode by heat injection in a root position, and the system comprises a carrier, a lifting rocker arm, a programmable logic controller, a steam generator, a steam distribution cabin, a heat injection mechanism and a bearing plate, wherein the lifting rocker arm, the controller and the steam generator are both installed on the carrier; the steam distribution cabin is connected to the lifting rocker arm and connected with the steam generator; the heat injection mechanism is connected between the steam distribution cabin and the bearing plate; the steam distribution cabin is connected with the bearing plate by a spring; the lifting rocker arm and the steam generator are both connected with the programmable logic controller; the steam distribution cabin is provided with a pressure sensor and a steam temperature sensor; a limiting sensor is arranged at the bottom of the steam distribution cabin; a soil temperature sensor is arranged at the bottom of the bearing plate; the limiting sensor, the pressure sensor, the steam temperature sensor and the soil temperature sensor are connected with the controller. The system can be used for effectively killing the root nematodes in the soil at the root position of the soil by means of steam heat injection, and realizing high automation degree and good effect.
Owner:孙腾腾
Method of producing hepatic targeting drug microcapsule
InactiveCN101129342AHigh selectivityLess by-productsPharmaceutical non-active ingredientsMicroballoon preparationDrug capsuleVinyl ester
The invention discloses a method for preparing a medicine with liver target taxis for slow-releasing the nanometer microcapsule. The method comprises the following steps: proceeding with enzymatic reaction of liver target taxis gene with sugar monomer of cerebrose residue and ethylated carboxylate; copolymerizing vinyl ester with glycosyl and unsaturated cationic or anion; getting liver target taxis polyelectrolyte; getting the medicinal microcapsule with liver target taxis by multilayer packaging polyelectrolyte on the surface of medicine by electrostatic action, wherein the deactivation speed of microcapsule medicine can be controlled. The polymerization method is simple, high effective and non-toxicity. The coating method has the high efficient, the simple operation and the mild technology, which can cycle several times. The preparing medicine can save for a long time, which can release step by step, can accumulate in the liver, improves the medicinal effect of disease portion, reduces the toxic effect of the other healthy organ, and has the good application prospect.
Owner:ZHEJIANG UNIV
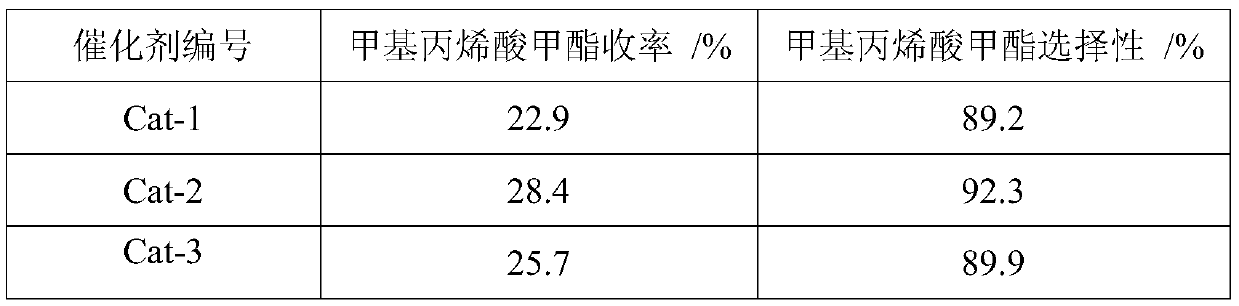
Acid-base bifunctional catalyst for synthesis of methyl methacrylate by methyl propionate and formaldehyde
InactiveCN109999922AEffective regulation of activityEffective regulation of intensityOrganic compound preparationCarboxylic acid esters preparationActive componentPhosphoric acid
The invention discloses an acid-base bifunctional catalyst for synthesis of methyl methacrylate by methyl propionate and formaldehyde. Active components of the catalyst are Cs and P, an auxiliary agent of the catalyst is one selected from the group consisting of Zr, Fe, Cu, La, Ce, Zn, Co, W and Mo, and a carrier of the catalyst is Al2O3 or Al2O3 treated by phosphoric acid, wherein Al2O3 is one selected from alpha-Al2O3, beta-Al2O3 and gamma-Al2O3. Based on the carrier, the loading capacity of the active components Cs and P is 5-20 wt% based on oxides, the loading capacity of the auxiliary agent is 0.1-1 wt% based on an oxide, and the concentration of a phosphoric acid treatment solution of the carrier is 1-5 wt%. The catalyst provided by the invention has better activity, selectivity andstability, and a simple preparation process, and is suitable for large-scale industrial application.
Owner:INST OF PROCESS ENG CHINESE ACAD OF SCI
Matrix special for roof greening
InactiveCN103766200AGood physical and chemical propertiesChange pHAgriculture gas emission reductionCultivating equipmentsNutritionAgricultural engineering
The invention discloses a matrix special for roof greening and belongs to the technical field of soilless culture. The matrix consists of vinegar residue, manioc waste, river sands, ceramsite, vermiculite and perlite and is characterized in that a formula of the matrix special for roof greening comprises, by volume, vinegar residue 20-30%, manioc waste 20-30%, river sands 10-20%, ceramsite 5-10%, vermiculite 15-20% and pumice 5-10%. The matrix water content is 25-35%, the organic content is larger than or equal to 25%, the total nutrient content is 1-3%, an EC value is 0.5-2.0mS / cm, and pH is 6-7.5. The soilless culture matrix special for roof greening mainly utilizes the vinegar residue and other organic wastes as raw materials, is produced through high-temperature fermentation treatment, turns waste into wealth and turns harm into good. In addition, the matrix is low in cost, light in texture, full in nutrition and capable of fixing plants well, does not influence the roof safety, is very suitable for roof greening and is a good matrix for horticultural crop soilless culture.
Owner:江苏培蕾基质科技发展有限公司
Ceramic glaze with water activating function, preparation method thereof, ceramic prepared by ceramic glaze and preparation method of ceramic
The invention belongs to the technical field of ceramic and particularly relates to ceramic glaze with a water activating function, a preparation method thereof, ceramic prepared by the ceramic glaze and a preparation method of the ceramic. The ceramic glaze is prepared by, by mass percentage, 60-80% of ceramic glaze base material, 5-20% of ceramic additives with the water activating function, 5-8% of nano-zirconia, 0.5-5% of phosphate, 1-5% of nano cesium oxide, 2-8% of yttrium oxide, 1-5% of nano titanium dioxide and 3-8% of tourmaline. The preparation method of the ceramic glaze includes: ball milling the ceramic glaze base material until all the ceramic glaze base material can pass a 250-mesh sieve, adding the rest of components, adding water and ceramic diluent to prepare slurry, ball milling, and discharging. The ceramic glaze has the advantages that the ceramic glaze can acts on water molecular cluster structures to allow the hydrogen bonds to be destroyed and chemical bonds to break, large molecular clusters become small molecular clusters only containing 5-6 water molecules, toxic gas dissolved in water is released at the same time, the water become slightly alkaline, and the activity of the water is increased.
Owner:ZIBO BAIKANG ECONOMIC & TRADE
Fertilizer for preventing bacterial wilt and promoting growth special for tomatoes
InactiveCN106242843AReduce the severity of the diseaseImprove disease resistanceSuperphosphatesMagnesium fertilisersPhosphorous acidMetasilicate
The invention discloses a fertilizer for preventing bacterial wilt and promoting growth special for tomatoes. The fertilizer is prepared from pig manure, chicken manure, earthworm manure, bagasse, crucifer, urea, superphosphate, potassium sulphate, manganese sulfate, magnesium sulfate, ferrous sulfate, ammonium molybdate, borax, prohexadione-calcium, calcium metasilicate, calcium carbonate, phosphorous acid, heteroauxin, humic acid, attapulgite, bentonite, microbial flora, alkyl glycoside, oligosaccharins and polyaspartic acid. The fertilizer for preventing bacterial wilt and promoting growth special for the tomatoes is reasonable in formula, comprehensive in nutrition and capable of promoting growth of the tomatoes, the disease resistance of tomato plants is improved, and bacterial wilt can be effectively prevented and controlled.
Owner:ANHUI SIERTE FERTILIZER IND
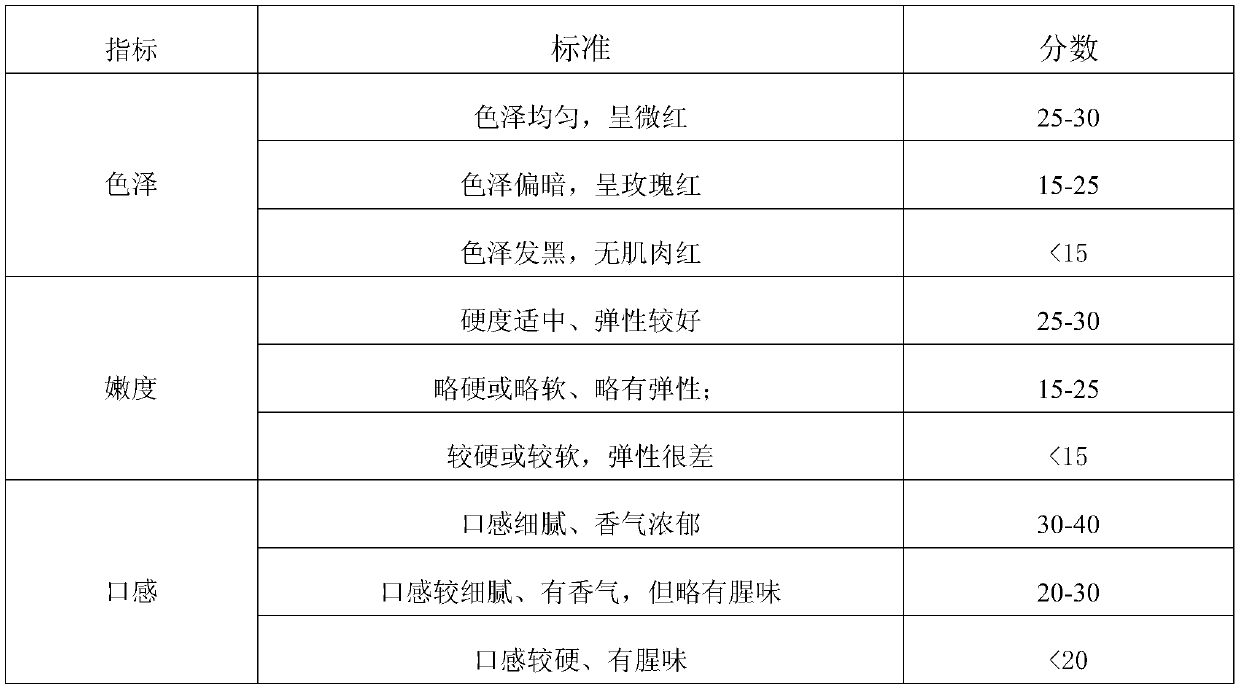
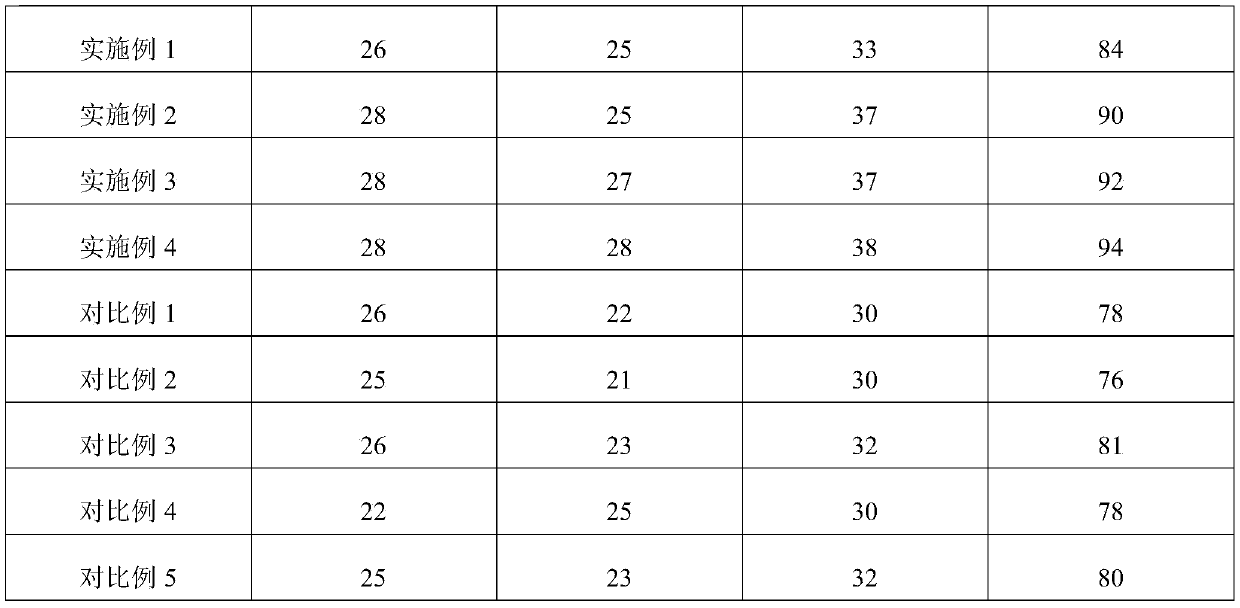
Preparation method of instant pickled beef
ActiveCN109730238AIncrease elasticityImprove tendernessClimate change adaptationFood scienceCarrageenanPhosphate
Owner:GUANGXI ZHUANG AUTONOMOUS REGION ACAD OF AGRI SCI
Fire coal desulfurization and denitrification decoking additive and preparation process thereof
The invention provides a fire coal desulfurization and denitrification decoking additive and a preparation process thereof. The fire coal desulfurization and denitrification decoking additive comprises a coal dust additive, which is added to the fire coal, and a catalytic activator, which is added to a slurry tank of a desulfurization system; and the preparation process comprises the following steps of: first, adding sodium carbonate, magnesium oxide, silicon dioxide, zinc oxide, manganese dioxide, copper oxide and barium oxide in sequence and stirring the mixture for 20 minutes; and then adding active calcium oxide and ammonium bicarbonate, and stirring the mixture for 10 minutes; uniformly mixing the mixture to obtain the coal dust additive; first stirring the active calcium oxide, active aluminum oxide and caustic calcined magnesite for 15 minutes; and then adding active carbon and calcium ammonium nitrate, and uniformly stirring the mixture to obtain the catalytic activator. According to the invention, without changing the traditional boiler and desulfurization equipment as well as the process thereof, the user selects corresponding additive and catalytic activator provided by the invention according to physical and chemical indexes of the coal so as to produce coal-saving, desulfurization, denitrification and decoking effects. During using high sulfur coals, the desulfurization efficiency is obviously increased and the SO2 emission reduction reaches the standard.
Owner:窦崇庆 +2
Production method for dried mutton
InactiveCN102919864AQuality improvementPreserve meaty flavorFood preparationMicrowaveProcess engineering
The invention discloses a production method for dried mutton. The production method comprises four steps of selecting and preprocessing a raw material, salting the raw material, curing and cutting, and drying by hot wind and microwave. Compared with the conventional drying method for producing dried mutton, a hot wind and microwave combined drying technology is adopted, so that the shortage of a single drying technology can be overcome, the advantages of hot wind drying and microwave drying are given into full play, the production cycle is shortened, the energy consumption is reduced, and the quality of a dried mutton product is improved. By adopting a salting way of uniformly applying salting materials and stacking material layers and mutton layers, the contact areas of the salting materials and mutton cubes can be greatly increased, the material liquid permeation speed is increased, and the produced dried mutton is fragrant and delicious in taste, proper in softness and hardness, chewy, scented, unique and long in aftertaste.
Owner:洛宁农本畜牧科技开发有限公司
Method and system for detecting SF6 gas in high-voltage insulation equipment
ActiveCN108731880AChange pHEasy Color ContrastDetection of fluid at leakage pointDecompositionElectrical devices
The invention provides a method and a system for detecting SF6 gas in high-voltage insulation equipment, which relates to the technical field of electrical equipment fault gas detection. The key points of the technical scheme comprise the steps that S1, aqueous solution is added in a first detection box, pH test paper is fixedly installed in the first detection box, and the pH test paper is soakedin the aqueous solution; S2, the first detection box is communicated to a sealing connection port of the high-voltage insulation equipment, and a pipeline is in sealing connection with the high-voltage insulation equipment; S3, a camera is installed in the first detection box, photographing is carried out on the pH test paper, and a photo is transmitted to a display terminal; S4, the pH test paper in the photo is compared with a standard colorimetric card. The gas flows into the first detection box, the SF6 gas and the decomposition product of the SF6 gas are partially dissolved in the aqueous solution, so that the pH value of the aqueous solution is changed. The pH test paper is compared with a standard colorimetric card, so that whether SF6 gas leaks or not is judged.
Owner:北京华电中试电力工程有限责任公司
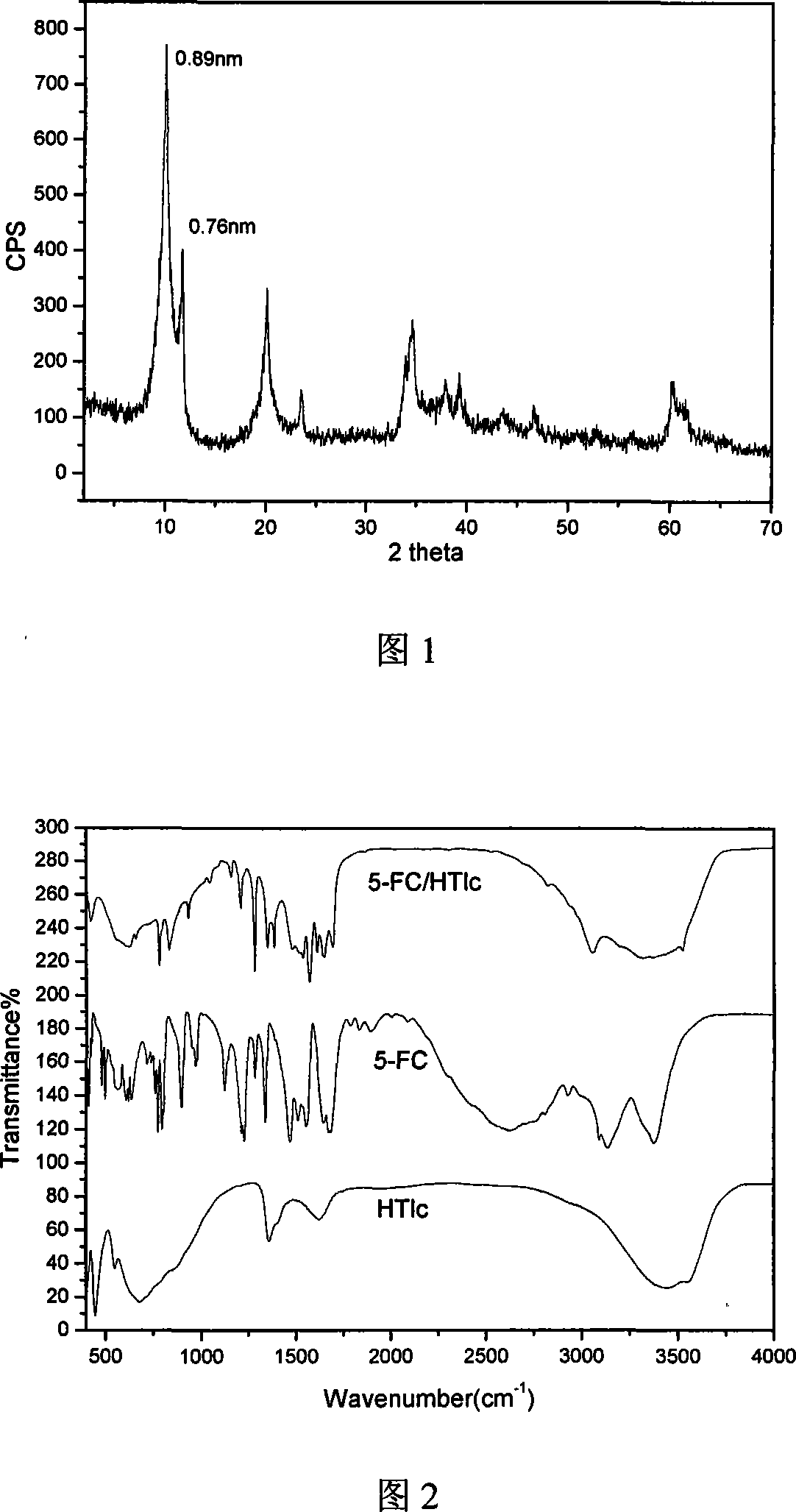
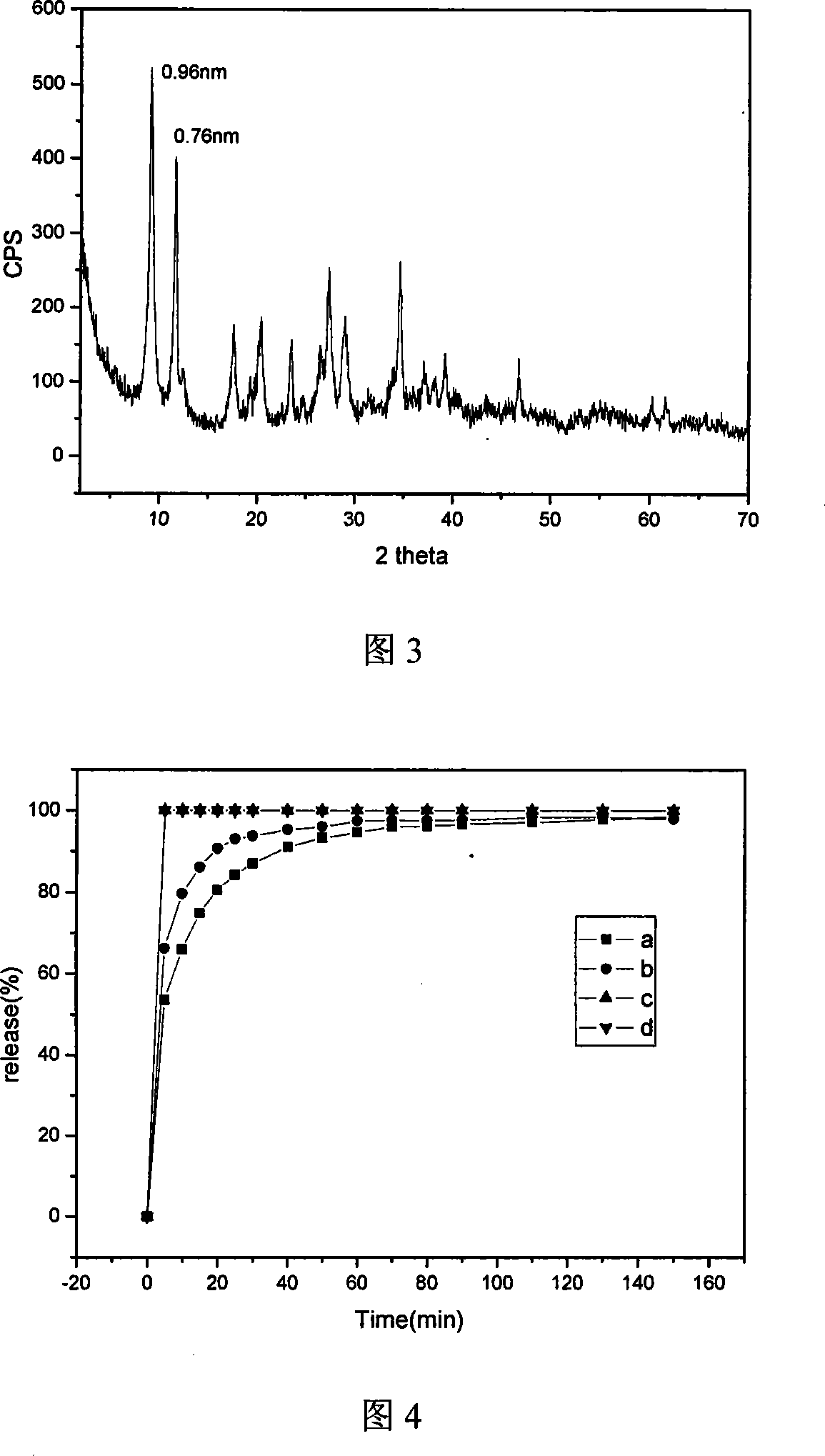
5-flucytosine/hydrotalcite-like NANO hybrid compound and preparation method thereof
InactiveCN101085355AGood slow releaseSimple processOrganic active ingredientsAntimycoticsFlucytosineSynthesis methods
A nano-hybrid of 5-fluctyosine (5-FC) and hydrotalcite-like compounds (HTlc) belongs to technology field of new material and pharmaceutical preparation.5-FC / HTlc nanohybrid is synthesized by mixed salt solution of divalent metal ion trivalent metal ion and 5-fluctyosine alkali solution throug coprecipitation method, and has a chemical constitution general formula of [MII(1-x)MIIIx(OH)2](An-)a(5-FC-)b.nH2O.The inventive 5-FC / HTlc nanohybrid has simple and easy synthetic method, large drug loading dosage, certain sustained release effect compared with raw material of 5-FC, and important significance for clinical application of 5-FC.
Owner:SHANDONG UNIV
Chemical solution preparation method for tungsten molybdate solid solution luminescent microcrystal
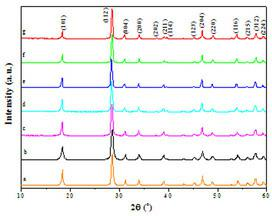
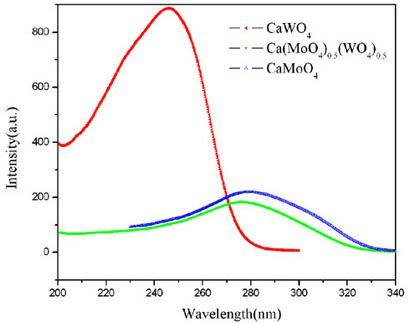
InactiveCN102433117ABroaden the field of applicationChange pHLuminescent compositionsBeakerPolytetrafluoroethylene
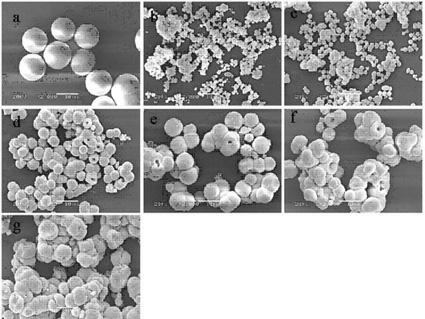
A chemical solution preparation method for a tungsten molybdate solid solution microcrystal is disclosed, which belongs to the field of luminescent material with a scheelite structure, and relates to a research about a novel chemical solution method preparation for a tungsten dimolybdate solid solution luminescent microcrystal host material. The solid solution microcrystal provided by the invention is expressed by a chemical general formula A(WO4)1-x(MoO4)x, wherein A is Ca, Sr or Ba; and x is not less than 0 and not greater than 1. The solid solution luminescent microcrystal of A(MoO4)x(WO4)(1-x) is obtained by adding A(NO3)2 solution, Na2MoO4 solution and Na2WO4 solution in corresponding amounts in a polytetrafluoroethylene beaker respectively according to the molar ratio of each raw material needed to prepare the microcrystal of a target system A(MoO4)x(WO4)(1-x) in a certain amount, stirring for 5 to 20 minutes at a normal temperature, then placing the polytetrafluoroethylene beaker in a high-pressure reactor; performing a hydrothermal reaction for 15 to 30 hours at 60 to 180 DEG C, and naturally cooling to a room temperature; centrifugally separating a precipitate, cleaning the precipitate by deionized water, then drying at 100 to 150 DEG C. The preparation technique disclosed by the invention is simple in equipment and low in reaction temperature; and the prepared luminescent microcrystal is pure in phase, uniform in granularity, controllable in grain size, regular in shape, excellent in luminescent performance, and good in practicability.
Owner:SICHUAN NORMAL UNIVERSITY
Power battery housing cleaning agent
The invention provides a power battery housing cleaning agent. The power battery housing cleaning agent comprises, by weight, 2-4 parts of sodium carbonate, 10-20 parts of ethanol, 2-3 parts of fatty alcohol polyoxyethylene ether, 1 part of sodium citrate and the balance water. A part of components in the power battery housing cleaning agent can be dissolved to form other substances and the substances and impurities undergo a chemical reaction so that the substances change pH of the cleaning agent, impurities outside a steel housing can be easily removed and work efficiency is high. The power battery housing cleaning agent has a low manufacturing cost, can be prepared easily and is convenient for promotion and use.
Owner:OPTIMUM BATTERY CO LTD
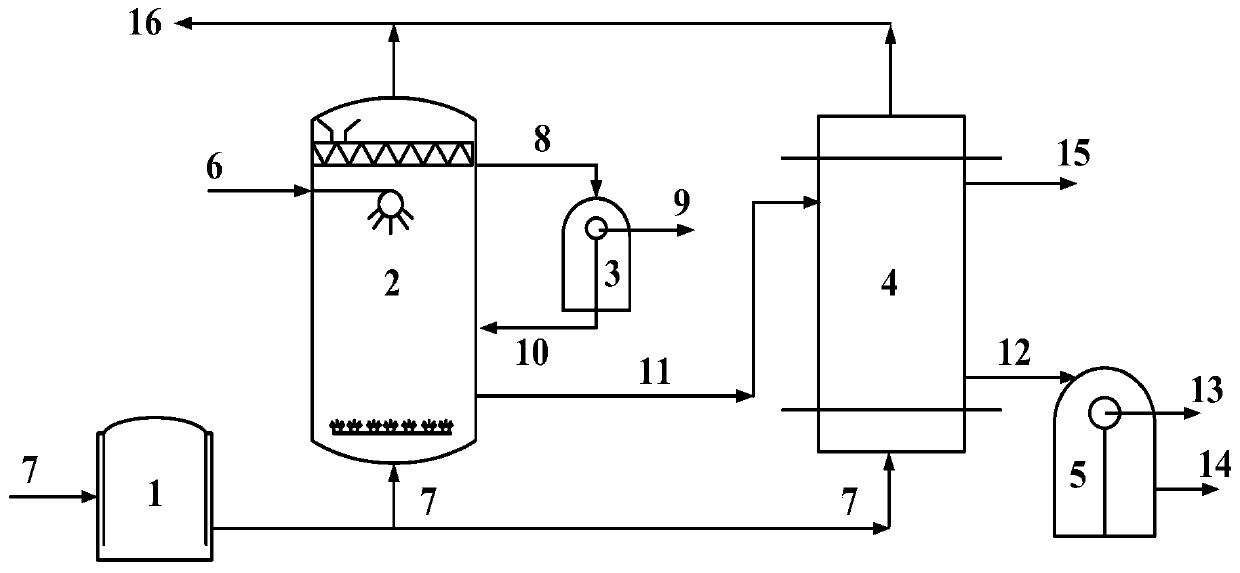
Deoiling-dephenolization combined treatment method for high-concentration phenol ammonia wastewater and system
ActiveCN110002629AHigh oil removal efficiencyImprove processing efficiencyFatty/oily/floating substances removal devicesWater contaminantsHigh concentrationRisk stroke
The invention relates to a deoiling-dephenolization combined treatment method for high-concentration phenol ammonia wastewater and a system. The treatment method comprises the following steps: (1) carrying out gas floating deoiling on a coke oven flue gas; (2) carrying out oil-water separation; (3) carrying out pressurized extraction dephenolization. A treatment system comprises a gas floating tank, an oil-water separator, an extraction device, a recycling tank and a flue gas buffer tank. As the coke oven flue gas is adopted as a foaming gas, the deoiling efficiency can be improved by virtue of nonpolar characteristics of carbon dioxide in the coke oven flue gas and the waste heat of the coke oven flue gas, and the acid gas carbon dioxide can be subjected to saturated absorption while deoiling is carried out; by changing the pH value of a solution, phenolate in wastewater can be induced to release phenolic substances of rich molecule modes, the allocation coefficient of the phenolic substances in an extraction agent can be increased, and the extraction efficiency can be improved; finally, through synergetic effects of gas floating deoiling and acidification and extraction procedures, the wastewater treatment efficiency is greatly improved, and three purposes can be achieved with one stroke.
Owner:SINOSTEEL ANSHAN RES INST OF THERMO ENERGY CO LTD +1
Heavy metal curing agent for repairing vanadium ore contaminated soil and repairing method
ActiveCN104772331AHigh organic contentImprove fertilityContaminated soil reclamationNatural mineralGoethite
The invention discloses a heavy metal curing agent for repairing vanadium ore contaminated soil and a repairing method. The heavy metal curing agent consists of the following components in percentage by weight: 65-75% of goethite and 25-35% of protonated chitosan, and the goethite which is ground to be 50 meshes and the ground protonated chitosan are mixed uniformly; the method for repairing the vanadium ore contaminated soil with the heavy metal curing agent comprises the following steps: deeply ploughing the contaminated soil, applying the curing agent and uniformly raking, watering in the process of deep ploughing and uniformly raking, wherein watering is performed once every other day in a maintenance period; and curing for 1-2 weeks. According to the heavy metal curing agent disclosed by the invention, the goethite, which is taken as a natural mineral, is free from toxicity and pollution, and the protonated chitosan is also conducive to the improvement of organic matter content in the soil and the increase of soil fertility; therefore after the mixture is applied, the pH value of the soil is not significantly changed, and the toxicity leaching rate of vanadium and other heavy metals in the repaired soil can be effectively reduced.
Owner:TSINGHUA UNIV
Clean pulping process for dissociated modified plant fibers
The invention discloses a clean pulping process for dissociated modified plant fibers. The clean pulping process comprises the following steps: (1) pre-processing raw materials; (2) preparing coarse pulp; (3) pulping; (4) removing sand; (5) screening; and (6) concentrating and extruding pulp. According to the clean pulping process disclosed by the invention, no sodium hydroxide is added, pulp-washing and bleaching processes can be reduced, and the wastewater in the pulping process can be sufficiently reutilized through simple treatment, so that pulping black liquor which is severely polluted is not produced. Meanwhile, plant fibers can be sufficiently dissociated, and the pulping quality is greatly improved.
Owner:山东省壮丰环境工程有限公司
Method for preparing oil soluble citrus coloring matter
A preparation method for oil-soluble orange flavochrome takes orange peel dry powder as the raw material and the oil-soluble orange flavochrome is obtained by the processes of acidolysis, zymohydrolysis and extraction. The preparation method adopts the zymohydrolysis method to complete the zymohydrolysis process to obtain the orange pectine and then the flavochrome is extracted, thus greatly saving the consumption of extractant and obviously lowering the cost. The product is natural pigment and is nontoxic and non-polluted, thus being applicable to be the colorant of oleaginous foods to replace synthetic food color.
Owner:FUJIAN AGRI & FORESTRY UNIV
Novel special liquid fertilizer for rice capable of comprehensively controlling plant diseases and insect pests, and preparation method thereof
InactiveCN106748147ADoes not affect listing and salesReduce the amount of applicationAlkali orthophosphate fertiliserAmmonium orthophosphate fertilisersRed CloverInsect pest
The invention provides a novel special liquid fertilizer for rice capable of comprehensively controlling plant diseases and insect pests, and a preparation method thereof. The novel special liquid fertilizer is prepared from the following components: 5 to 15 parts of urea-ammonium nitrate solution, 5 to 15 parts of ammonium polyphosphate solution, 5 to 10 parts of potash fertilizer, 0.1 to 2 parts of microelement, 20 to 50 parts of water, 1 to 10 parts of homemade plant pesticide extracting solution, 1 to 2 parts of soil conditioner, 1 to 5 parts of natural activity organic materials, and 1 to 5 parts of natural surfactant, wherein the homemade plant pesticide extracting solution is prepared from mixing trifolium pratense, folium artemisiae argyi, herba houttuyniae, semen persicae and black false hellebore in proportion, and extracting with water. The preparation method comprises the steps of adding the prepared urea-ammonium nitrate solution and the ammonium polyphosphate solution in proportion according to a formula into a mixing tank; then adding the other metered materials into the mixing tank; stirring, dissolving and filtering to prepare the liquid fertilizer. The liquid fertilizer provided by the invention is convenient to use and good in effect, and is a special multi-functional environment-friendly liquid fertilizer for the rice.
Owner:GUIZHOU UNIV
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com