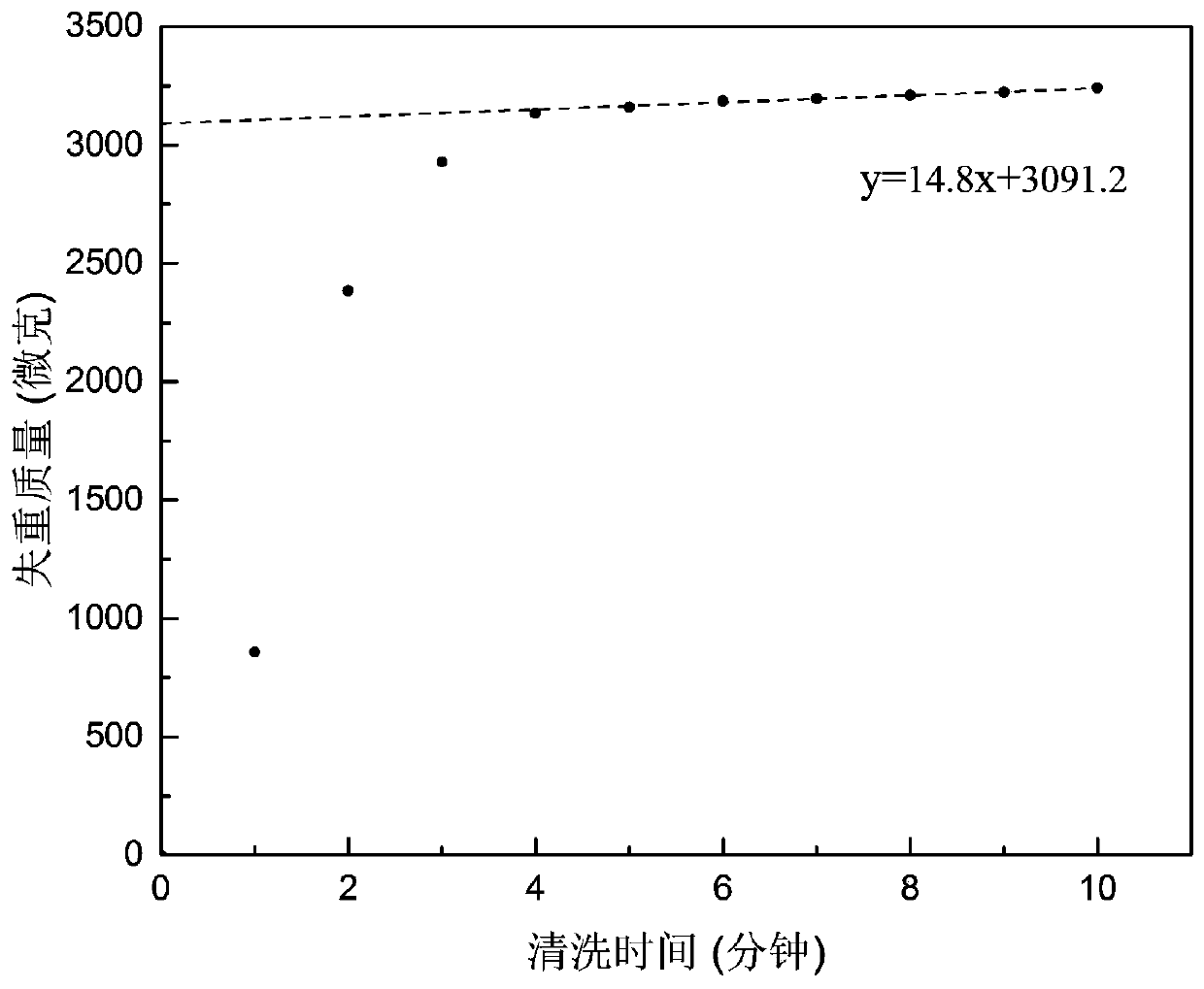
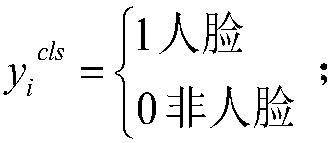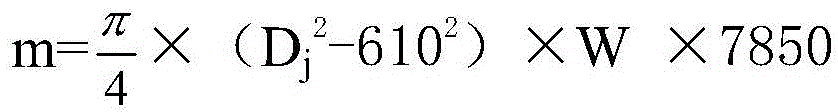Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
31 results about "Loss weight" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Face attribute recognition method of deep neural network based on cascaded multi-task learning
ActiveCN108564029APromote resultsThe result of face attribute recognition is improvedCharacter and pattern recognitionNeural architecturesVisual technologyCrucial point
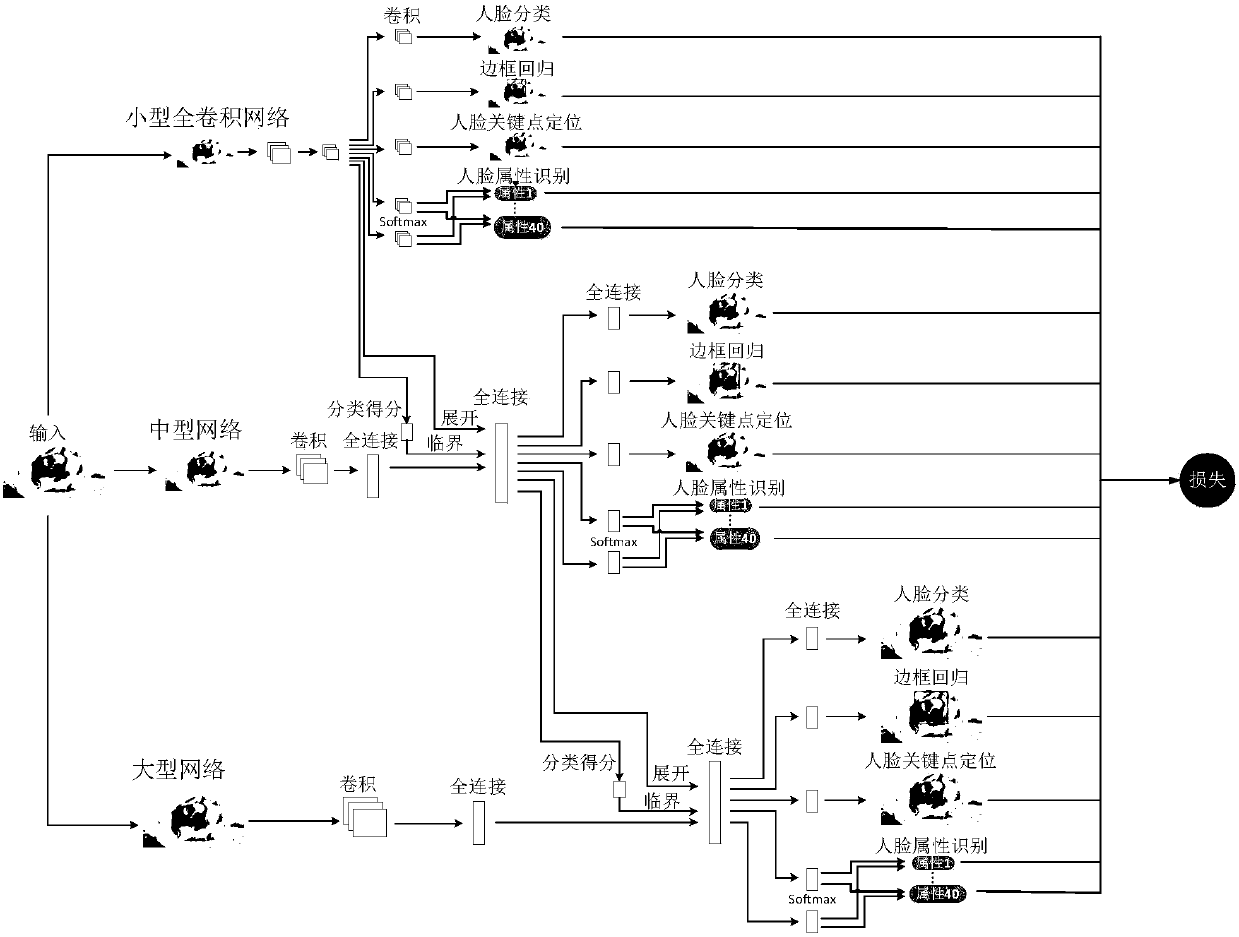
The invention provides a face attribute recognition method of a deep neural network based on cascaded multi-task learning and relates to the computer vision technology. Firstly, a cascaded deep convolutional neural network is designed, then multi-task learning is used for each cascaded sub-network in the cascaded deep convolutional neural network, four tasks of face classification, border regression, face key point detection and face attribute analysis are learned simultaneously, then a dynamic loss weighting mechanism is used in the deep convolutional neural network based on the cascaded multi-task learning to calculate the loss weights of face attributes, finally a face attribute recognition result of a last cascaded sub-network is used as the final face attribute recognition result based on a trained network model. A cascading method is used to jointly train three different sub-networks, end-to-end training is achieved, the result of face attribute recognition is optimized, different from the use of fixed loss weights in a loss function, a difference between the face attributes present is considered in the invention.
Owner:XIAMEN UNIV
Facial expression identification method based on multi-task convolutional neural network
ActiveCN108764207AImprove the difference between classesImprove discrimination abilityCharacter and pattern recognitionNeural architecturesData setFeature extraction
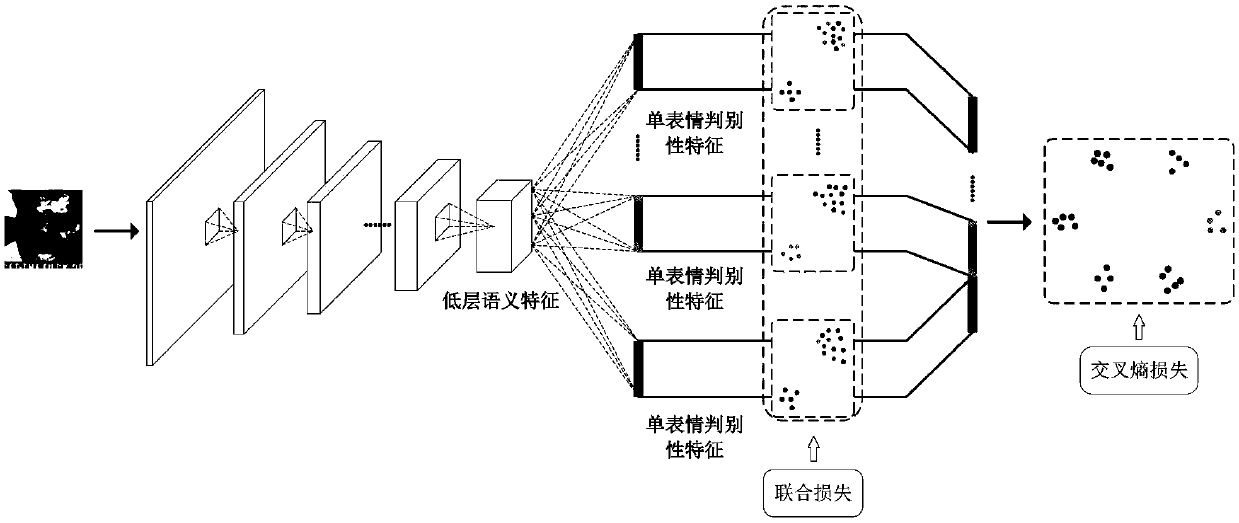
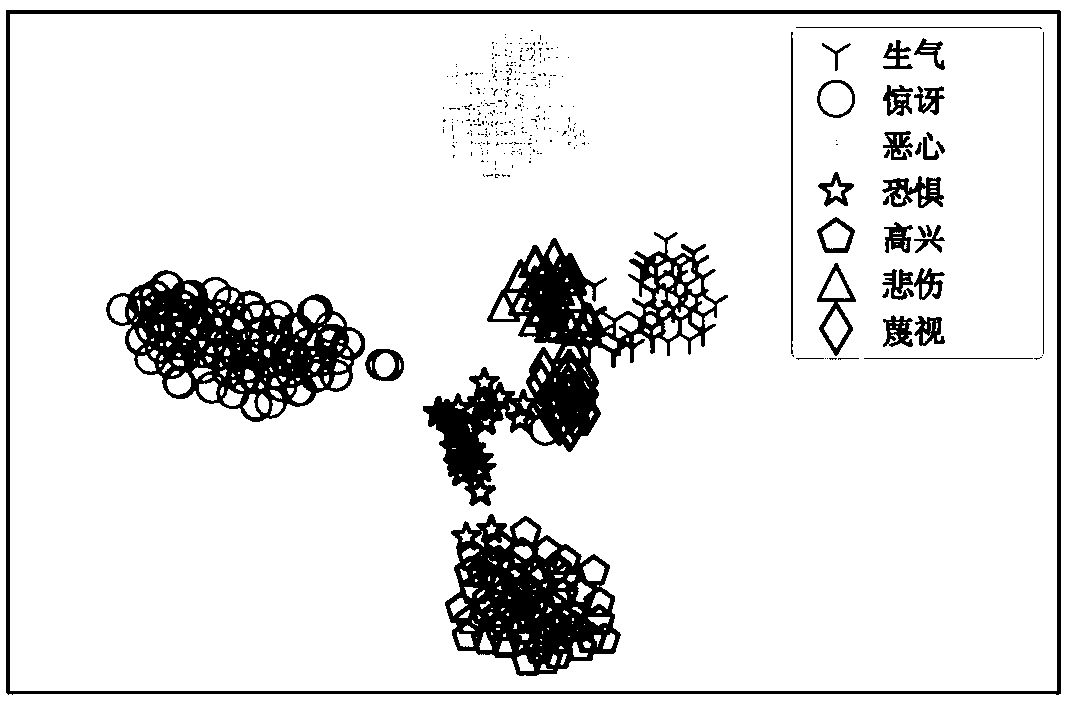
The invention discloses a facial expression identification method based on multi-task convolutional neural network. The expression identification method comprises the following steps: firstly, designing a multi-task convolutional neural network structure, and sequentially extracting low-level semantic features shared by all expressions and a plurality of single-expression distinguishing characteristics in the network; then adopting multi-task learning and simultaneously learning learning tasks of the plurality of single-expression distinguishing characteristics and multi-expression identification tasks; monitoring the all tasks of the network by using combined loss, and balancing the loss of the network by using the two loss weights; finally, acquiring a final facial expression identification result from a maximum flexible classification layer arranged at the last of a model according to the trained network model. Characteristic extraction and expression classification are put in an end-to-end framework to be learned, the distinguishing characteristics are extracted from input images, and expression identification on the input images are reliably carried out. Experimental analysisshows that the algorithm is excellent in performance, complicated facial expressions can be effectively distinguished, and good identification performance on a plurality of published data sets can beachieved.
Owner:XIAMEN UNIV
Gastrodia elata medicinal materials discrimination method
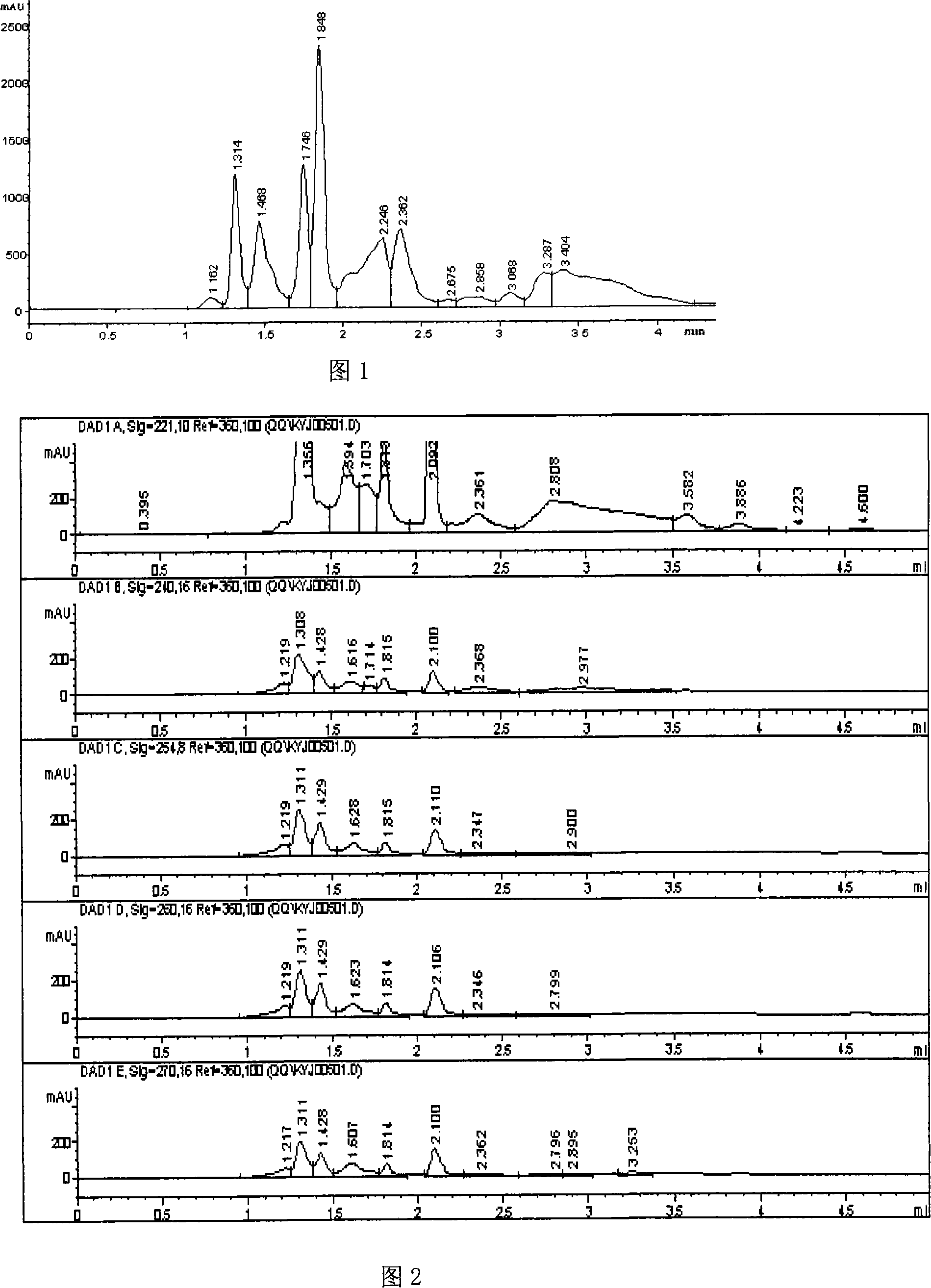
An identification method of the rhizome gastrodiae includes the below processes: (1) setting the standard chemical fingerprint atlas of rhizome gastrodiae: a. putting the rhizome gastrodiae standard sample into the sorbitic extractor; b. adding the ethanol and putting in the water bath for hot reflux, treating it by the ultrasonic cell knapper and weight, then to loss weight by the corresponding solvent; repeating it again; after filtration, concentration and drying, it uses the 10% acetonitrile to constant volume; c. using the HPLC, Eclipse, XD13-C8 chromatographic column, the 10% acetonitrile solution is as the mobile phase, the light of 221+-1nm wavelength is as the detection light source to determine the chemical fingerprint atlas of rhizome gastrodiae of rhizome gastrodiae standard sample; which has seven evidence peaks, the peak with 1.920min holding time is the rhizome gastrodiae element peak, the rhizome gastrodiae element peak of the other rhizome gastrodiae has the big warp of 1.9% with the standard sample. (2) Determining the chemical fingerprint of the detected sample and the dummy sample according to the progress (1) and comparing to identify.
Owner:CHANGSHA UNIVERSITY OF SCIENCE AND TECHNOLOGY
Test method for corrosion- resistance performance contrast of lead-acid battery grid
InactiveCN106323852AImprove accuracyAvoid test batch errorsWeather/light/corrosion resistanceSurface oxidationEngineering
The invention discloses a test method for corrosion-resistance performance contrast of a lead-acid battery grid, comprising the steps of (1) taking several kinds of grids, each for two pieces, soaked in the sugar alkali solution to remove surface oxide, weighing after cleaning & drying, records the weight of the monolithic grid under test, remembers as w1; (2) placing the grid under test respectively in the corresponding corrosion device containing sulfuric acid solution; (3) connecting all the corrosion device in series, and charged with constant current; (4) after the charge, takes out the grid under test which represents the anode, weighs after processing according to the method of step (1) , remembers as w2; (5) adopts the formula w = w1-W2 to calculate the corrosion loss weight for each grid under test, and compares the corrosion resistance performance of several kind of grids under test based on the quantities of w. The invention takes the grid weight of removing surface oxide layer as the initial weight, makes the last calculated corrosion loss weight more accurately to reflect the corrosion resistance performance of the grid under test.
Owner:TIANNENG BATTERY GROUP
Landslide debris flow area detection method based on sparse representation classification
ActiveCN104615999AEfficient detectionImprove accuracyScene recognitionLandslideClassification methods
The invention provides a landslide debris flow area detection method based on sparse representation classification. The method includes: dividing a target area image to be detected into multiple image blocks to be detected with the same sizes, analyzing and judging the brightness of three channels of red, green and blue through a sparse representation classification method, obtaining a suspected landslide debris flow image block collection, presetting the mean value of the difference between the first threshold value and the sparse representation errors; obtaining the landslide debris flow severely damaged area and the area boundaries in the suspected landslide debris flow image block collection through a connected area detection method; obtaining the land type of the image of each point before a disaster according to the position of each point of the area boundaries, obtaining the expansion degree influence coefficient of each point corresponds to the disaster loss weight according to the preset land type, obtaining the extended distance of the landslide debris flow area according to the mean value of the difference between the preset first threshold value and the sparse representation errors, and obtaining the landslide debris flow general damaged area through the extension of the landslide debris flow seriously damaged area. The landslide debris flow detection method is high in detection efficiency and accuracy and low in false alarm rate.
Owner:BEIJING NORMAL UNIVERSITY
Beautifing and health care tea and its making method
A face-beautifying health-care tea is prepared from honeysuckile flower, yellow chrysanthemum flower, white chrysanthemum flower, rose, cassia seed and lotus through respectively treating, and proportionally mixing. Its advantages are high effect on beautifying face, loss weight, improving immunity and delaying senility, and no toxic by effect.
Owner:韩伟
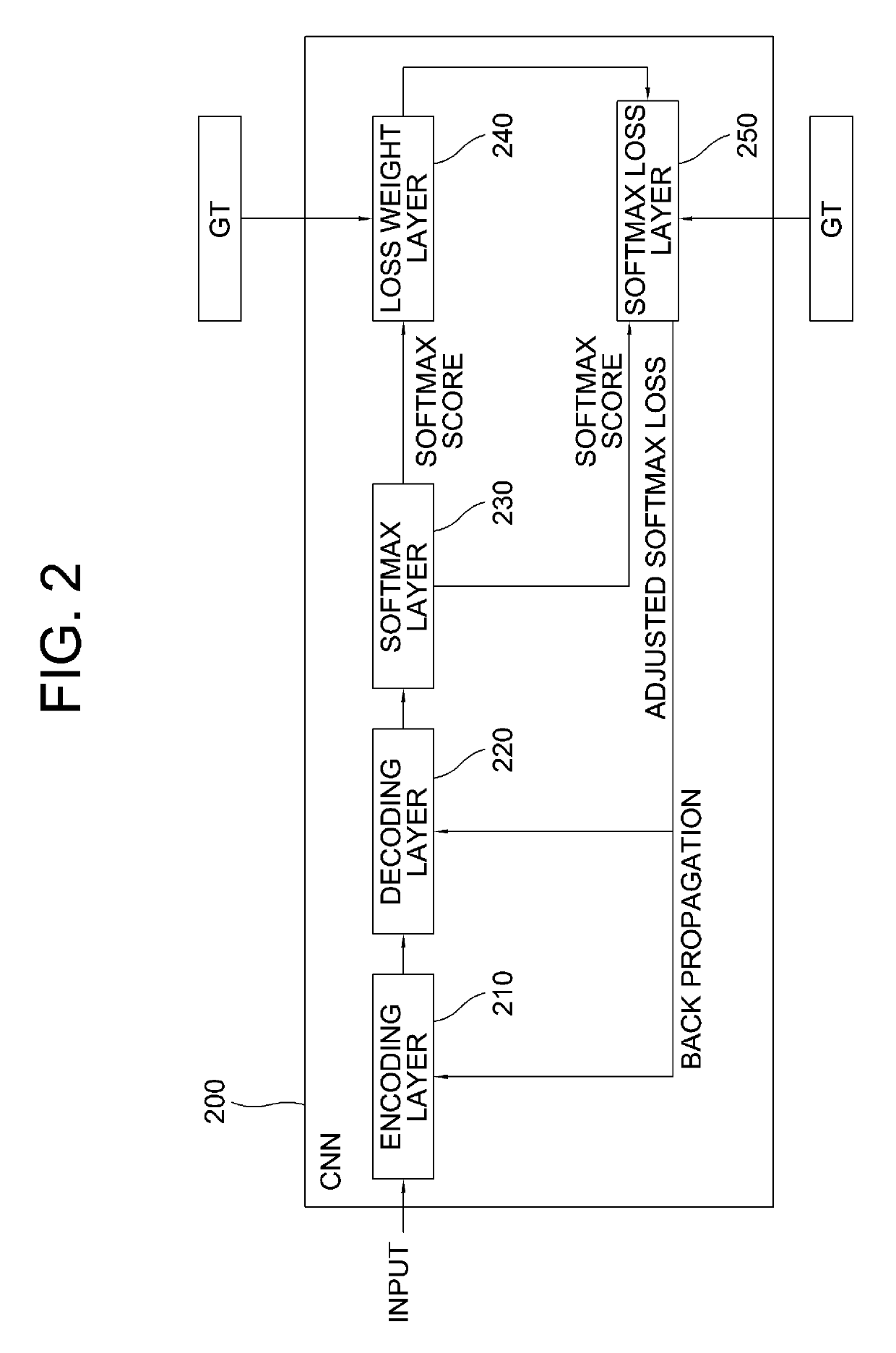
Learning method and learning device for attention-driven image segmentation by using at least one adaptive loss weight map to be used for updating HD maps required to satisfy level 4 of autonomous vehicles and testing method and testing device using the same
A method for an attention-driven image segmentation by using at least one adaptive loss weight map is provided to be used for updating HD maps required to satisfy level 4 of autonomous vehicles. By this method, vague objects such as lanes and road markers at distance may be detected more accurately. Also, this method can be usefully performed in military, where identification of friend or foe is important, by distinguishing aircraft marks or military uniforms at distance. The method includes steps of: a learning device instructing a softmax layer to generate softmax scores; instructing a loss weight layer to generate loss weight values by applying loss weight operations to predicted error values generated therefrom; and instructing a softmax loss layer to generate adjusted softmax loss values by referring to initial softmax loss values, generated by referring to the softmax scores and their corresponding GTs, and the loss weight values.
Owner:STRADVISION
Aurea helianthus slimming paste and preparation method thereof
InactiveCN105010924AQuick and Unique Diet Therapy and Health BenefitsFood ingredient functionsFood preparationPreservativeAdditive ingredient
The present invention relates to an aurea helianthus slimming paste which includes main ingredients and accessory ingredients, and the mass ratio of the main ingredients and the accessory ingredients is 3:7-4:6; the main ingredients comprise the following components in mass percentages: aurea helianthus dry pollen 33%-50%, aurea helianthus straw powder 12.5%-17%, aurea helianthus root powder 12.5%-17%, aurea helianthus seed powder after oil extraction 12.5%-17%, and aurea helianthus leaf powder 0-17%. The whole plant of aurea helianthus roots, stems, leaves, flowers, and fruits at a certain ratio are matched with brown rice, oats, red dates, soybeans, etc., and all the ingredients are blended to prepare the nutritious aurea helianthus slimming paste by a special process and an exclusive formula. The aurea helianthus slimming paste is free of preservatives and additives, and is a pure green botanical slimming food. The slimming paste (aurea helianthus health preserving powder) show a rapid and unique therapeutic health-care effect, and the experiencing crowd can get rid of constipation in one day, loss weight in one week, and have lowered high blood pressure, high blood sugar and high blood lipids in one month. The slimming paste enables the medicinal and edible concept not just to be an idea, but verifies the concept through the products and the experience; and the medicinal nourishment is not as good as food nourishment, and the medicinal slimming is not as good as food slimming.
Owner:马宝明
Glutinous rice cake chilli seasoning and production process thereof
The invention discloses a glutinous rice cake chilli seasoning and a production process thereof. The formula and contents of glutinous rice cake chilli seasoning include 100 kg of dried chili, 8-12 kg of garlic, 7-10 kg of ginger, an appropriate amount of salt, an appropriate amount of high quality Daqu liquor, an appropriate amount of tsaoko. The production processes of the glutinous rice cake chilli include: cleaning the dried chili after removing stems and impurities, immersing in warm water for 3-5 hours, adding the peeled garlic, the peeled ginger, the Daqu liquor, the salt and the tsaoko, triturating to 10-20 meshes, sterilizing, testing, bagging and sealing. The product is fragrant in smell, has the nature of acridity and warmth, is spicy but not fierce, is rich in flavor, invigorates spleen and warms middle, is anti-inflammatory and diuretic, and helps to improve the body resistance, while can sterilize, fend off cold, lower lipid, help to loss weight and supply vitamin C.
Owner:岳杰
Intestine-opening and lung-moistening ferment and preparation method thereof
InactiveCN106174520APromote growth and developmentPrevent anemiaFood ingredient functionsBiotechnologyToxicant
The invention discloses an intestine-opening and lung-moistening ferment and a preparation method thereof. The intestine-opening and lung-moistening ferment comprises the following raw materials in parts by weight: 10-15 parts of pumpkins, 15-20 parts of unpolished rice, 6-8 parts of white fungi, 5-10 parts of edible amaranth, 5-8 parts of chenopodium album, 8-10 parts of watermelon rind, 5-8 parts of tangerine peel, 3-5 parts of walnut kernels, 5-8 parts of bananas and 5-8 parts of honey. By adopting scientific matches of the raw materials, the prepared ferment is capable of decomposing and eliminating the toxins in the intestinal tract and in the body so as to achieve the effects of beautifying and keeping youth; the ferment is also capable of readjusting the balance performance of the human body so as to promote massive decomposition of the deposited fat to achieve the effect of reducing weight; moreover, the ferment further has the functions of eliminating dryness and moistening the lung. Thus, the intestine-opening and lung-moistening ferment is especially suitable for people with obesity or constipation. Furthermore, the intestine-opening and lung-moistening ferment is also rich in nutrient contents, and capable of satisfying demands of the body. The consumers of the ferment can loss weight without malnutrition.
Owner:XIAMEN YILIKANG BIOTECH CO LTD
Vehicle weighing state detection system and method
ActiveCN109855710ARapid positioningProvide in timeWeighing apparatus testing/calibrationSpecial purpose weighing apparatusComputer moduleFlow diverter
The invention discloses a vehicle weighing state detection system, which comprises weight counting equipment, a weight counting charging system, a signal shunt, a front-end collecting module and a rear-end early warning module. The signal shunt collects analog signals from the weight counting equipment, and the analog signals are imaged in the signal shunt by an emitter follower to form two signals equal to the original signals in magnitude, wherein one way of the signals is returned to the weight counting charging system, and the other way of signal is returned to the front-end collecting module to be subjected to data analysis; the front-end collecting module conducts data analysis on each signal, and returns the calculation results to the rear-end early warning module; and the rear-endearly warning module can directly calculate an equipment failure module, sends out a reminding repairing signal, and automatically compensates the loss total weight to the system. An owner monitoringplatform is effectively combined, the failure position can be quickly determined, meanwhile, the weight counting result after data repair of damaged equipment is given, and owners are assisted in counting the loss weight.
Owner:耿建航
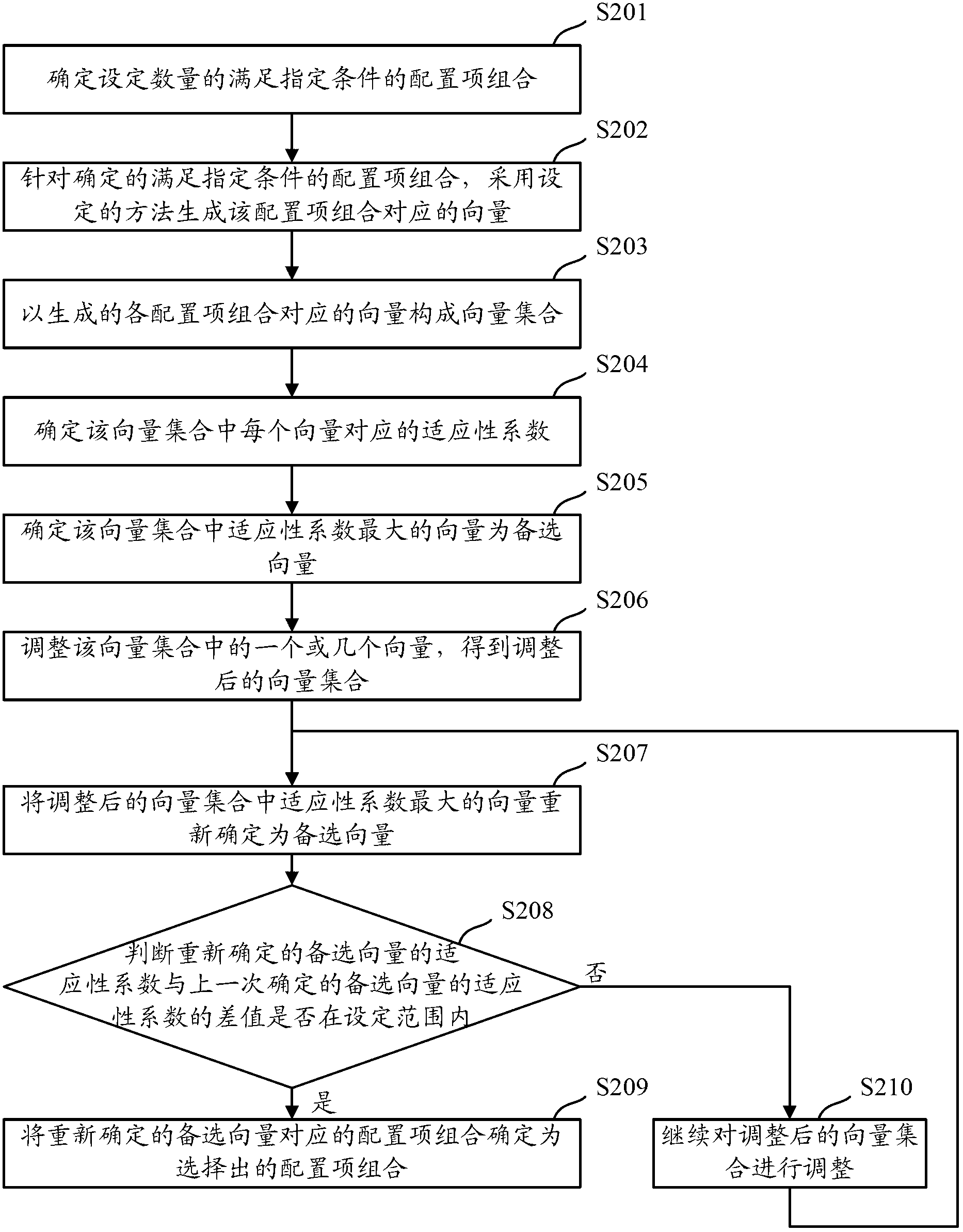
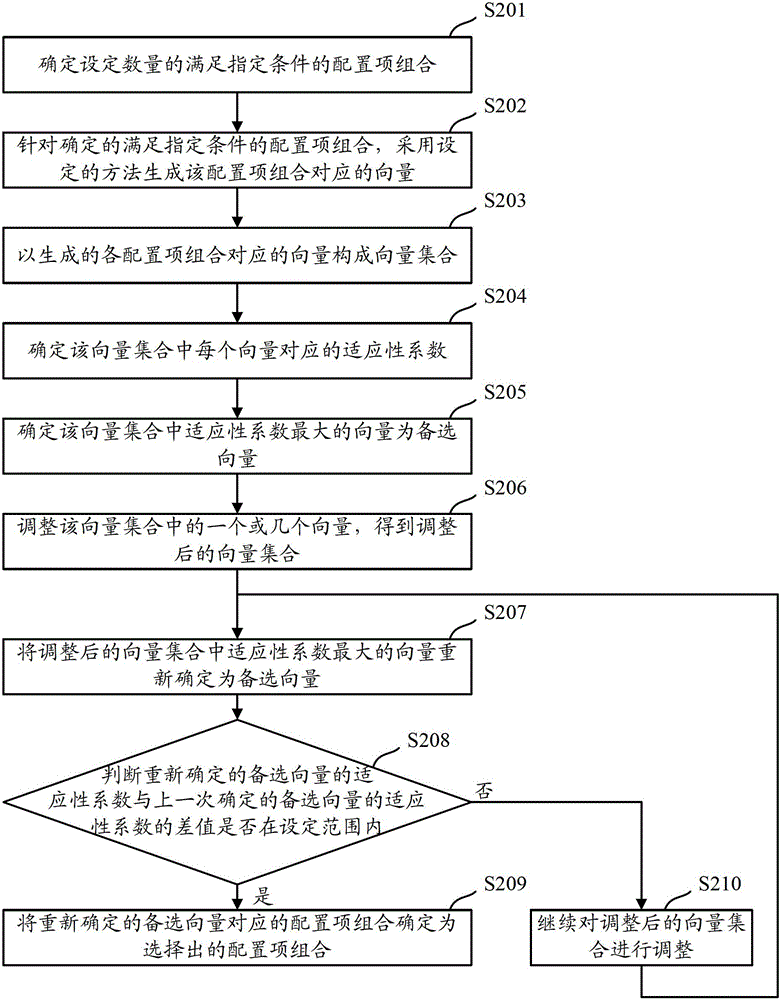
Method and device for security configuration optimization
ActiveCN102710641AMeet safety requirementsLow usability impactTransmissionConfiguration optimizationConfiguration item
The invention discloses a method and a device for security configuration optimization, which are used for solving the problems that the usability of a configured system is low in the prior art. The method comprises the steps of determining configuration item assemblies with the sum of the security weights corresponding to contained configuration items larger than a set security threshold according to the security weights corresponding to the configuration items contained in the system, selecting a configuration item assembly with the minimum sum of usability loss weights corresponding to the contained configuration terms in the determined configuration item assemblies according to the usability loss weights corresponding to the configuration terms, and configuring the system according to the contained configuration terms in the selected configuration item assembly. According to the method, the selected configuration item assembly not only meets the security requirement of the system, but also least affects the usability of the system, so that as being configured according to the selected configuration item assembly, the configured system meets the security requirement and simultaneously improves the usability.
Owner:NSFOCUS INFORMATION TECHNOLOGY CO LTD
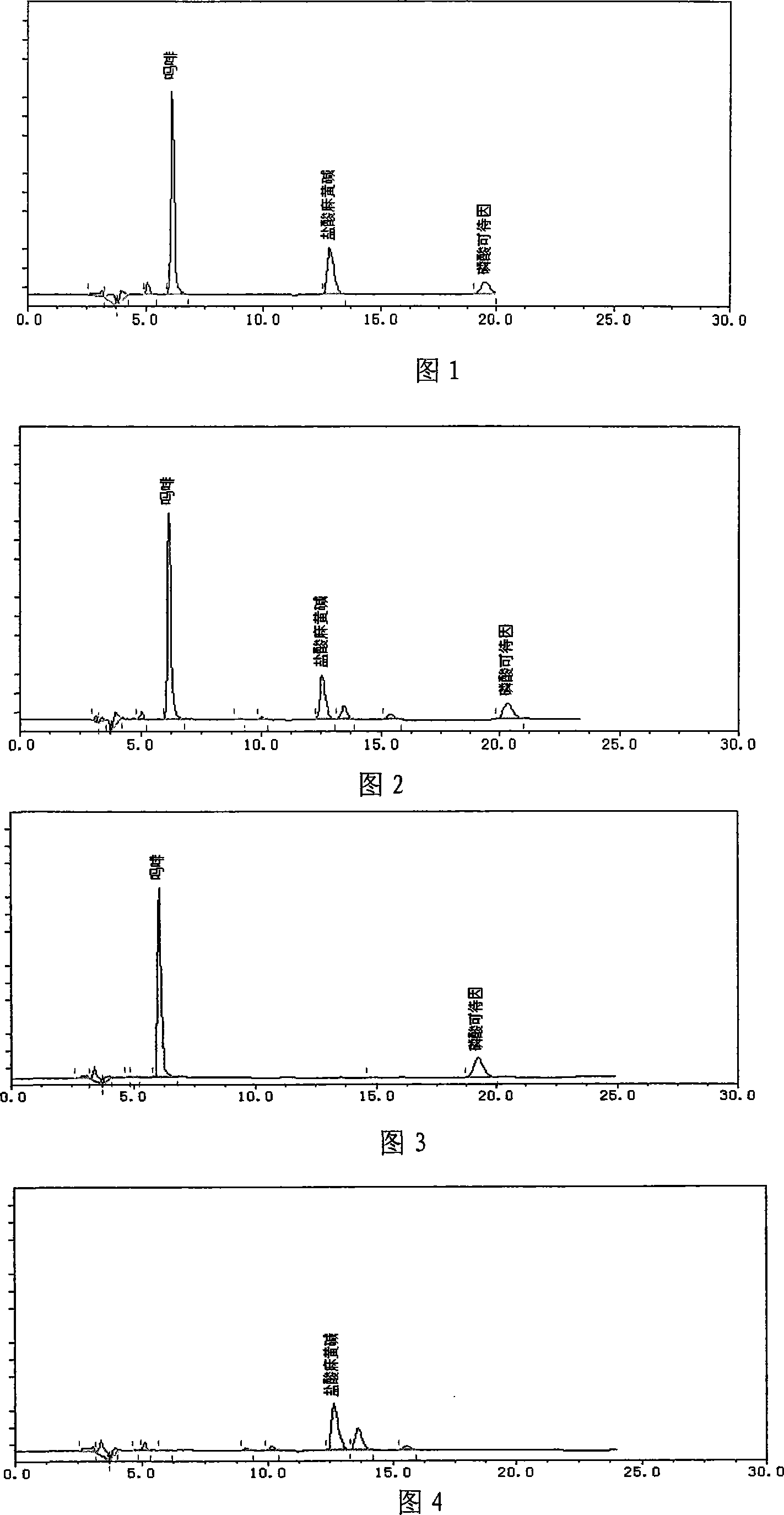
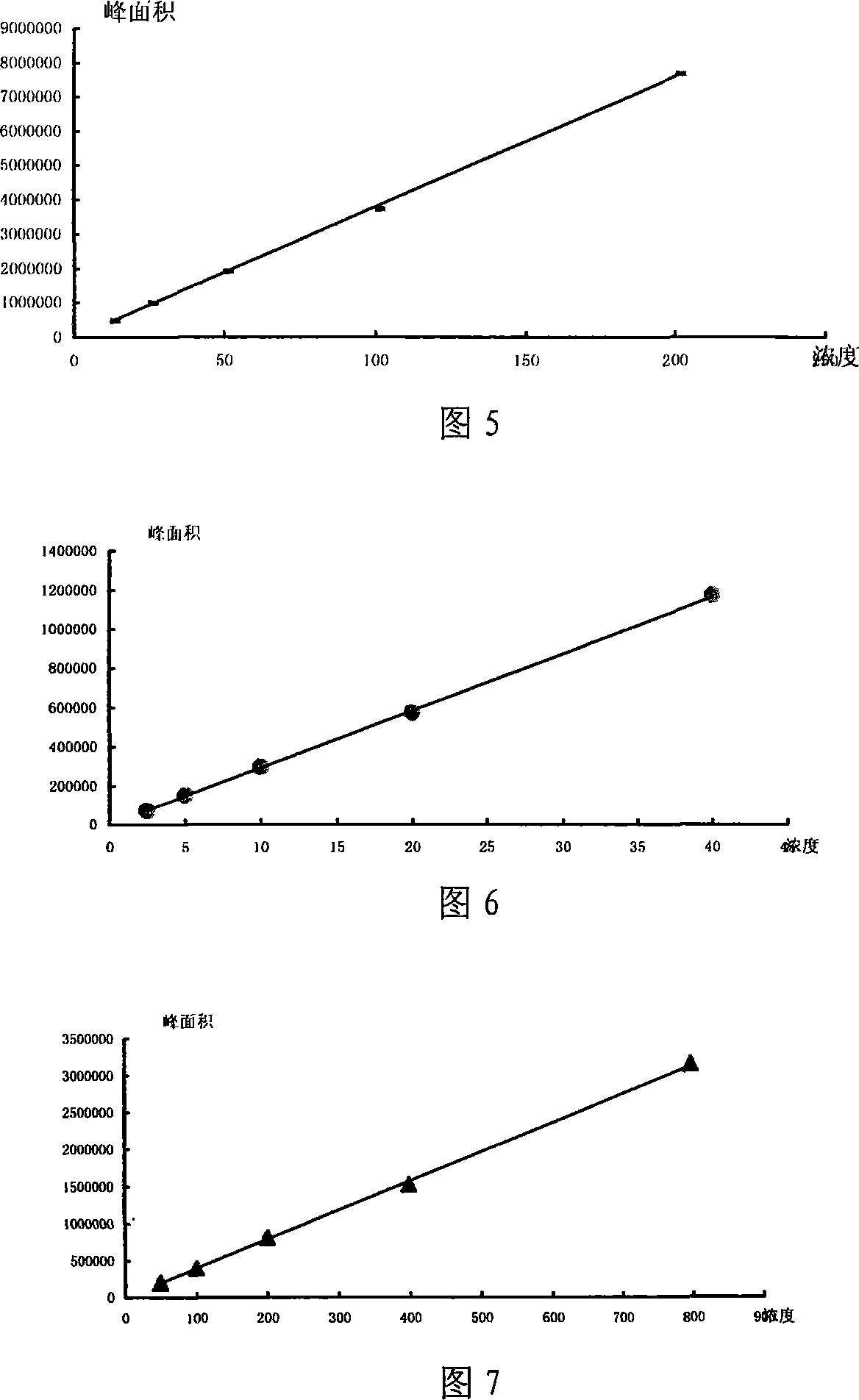
Quality control method of asthma tablets
ActiveCN101112424AGuaranteed reproducibilityGuaranteed efficacyRespiratory disorderPlant ingredientsWater bathsSlag
The present invention discloses a quality control method of asthma tablets, which includes the flowing steps: (1) chromatography conditions and system suitability experiment; (2) preparation of control solution; (3) preparation of test solution is as following: taking the product, removing the coating, porphyrizing, taking about 1g, placing into a 50ml volumetric flask, adding 1ml of ammonia test solution and 25ml of chloroform precisely, weighing the weight, carrying out the ultrasonic treatment with the power of 260w and the frequency of 40KHz for 30 minutes, cooling, weighing the weight, using the chloroform to supplement the loss weight, shaking evenly, filtering, sucking 5ml of filtrate precisely, adding 2ml of 2 percent hydrochloric ethanol anhydrous, shaking evenly, drying up by evaporation in a water bath, adding 10ml of 0.1percent hydrochloric acid in the residual slag, full dissolving by ultrasound, filtering, and taking the subsequent filtrate; (4) weigh and suck the control solution and the test solution precisely and respectively to inject to an HPLC instrument for determination. The method adopts the HPLC to carry out the content determination of morphine, codeine phosphate and ephedrine hydrochloride in the prescription under a same system, which can not only ensure the efficacy of the products, but can also enable the quality of the products to be stable.
Owner:贵州省科晖制药有限公司
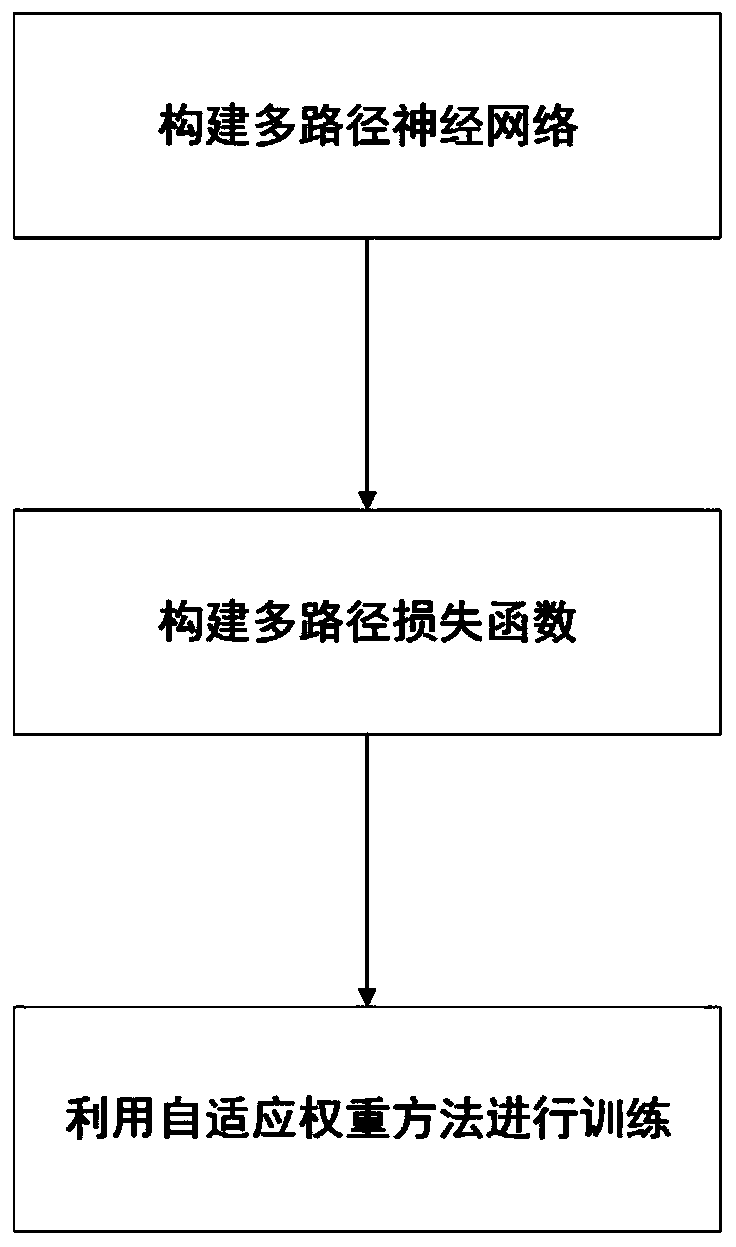
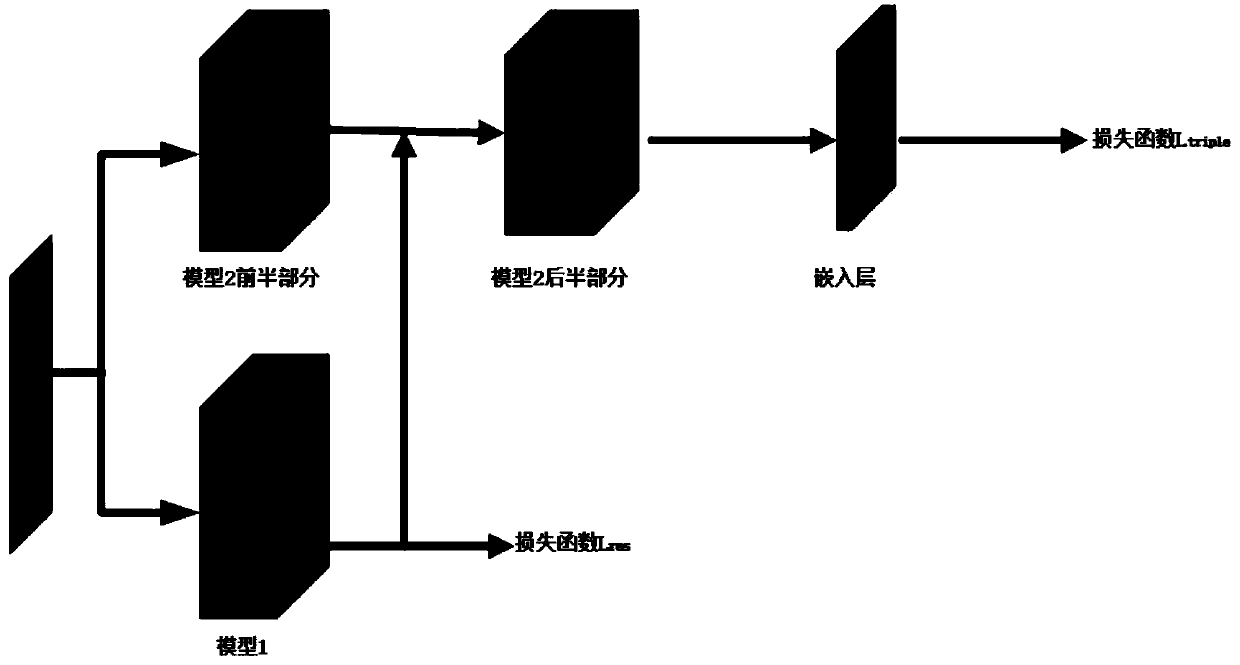
Face recognition multi-path deep neural network method based on self-adaptive weight
InactiveCN110472570AImprove training effectImprove performanceCharacter and pattern recognitionNeural architecturesPattern recognitionPath network
The invention belongs to the technical field of image recognition, and particularly relates to a face recognition multi-path deep neural network method based on adaptive weight, and the technical scheme comprises the following steps: firstly, constructing a multi-path neural network suitable for face recognition; secondly, establishing a corresponding multi-path loss function based on a task of face recognition; finally, using an adaptive weight algorithm for training, wherein the algorithm adaptively adjusts loss weights of different path networks according to corresponding thresholds in thetraining process, and a final model is obtained. According to the invention, the multi-path neural network is established; according to the invention, an end-to-end face recognition process is completed, training of a deep network is carried out through an adaptive weight algorithm, the training effect is significantly improved, the method can also be suitable for faces of different distances, theperformance of practical application is greatly improved, the market demand can be well met, and large-area popularization and application of the method in the market are facilitated.
Owner:旭辉卓越健康信息科技有限公司
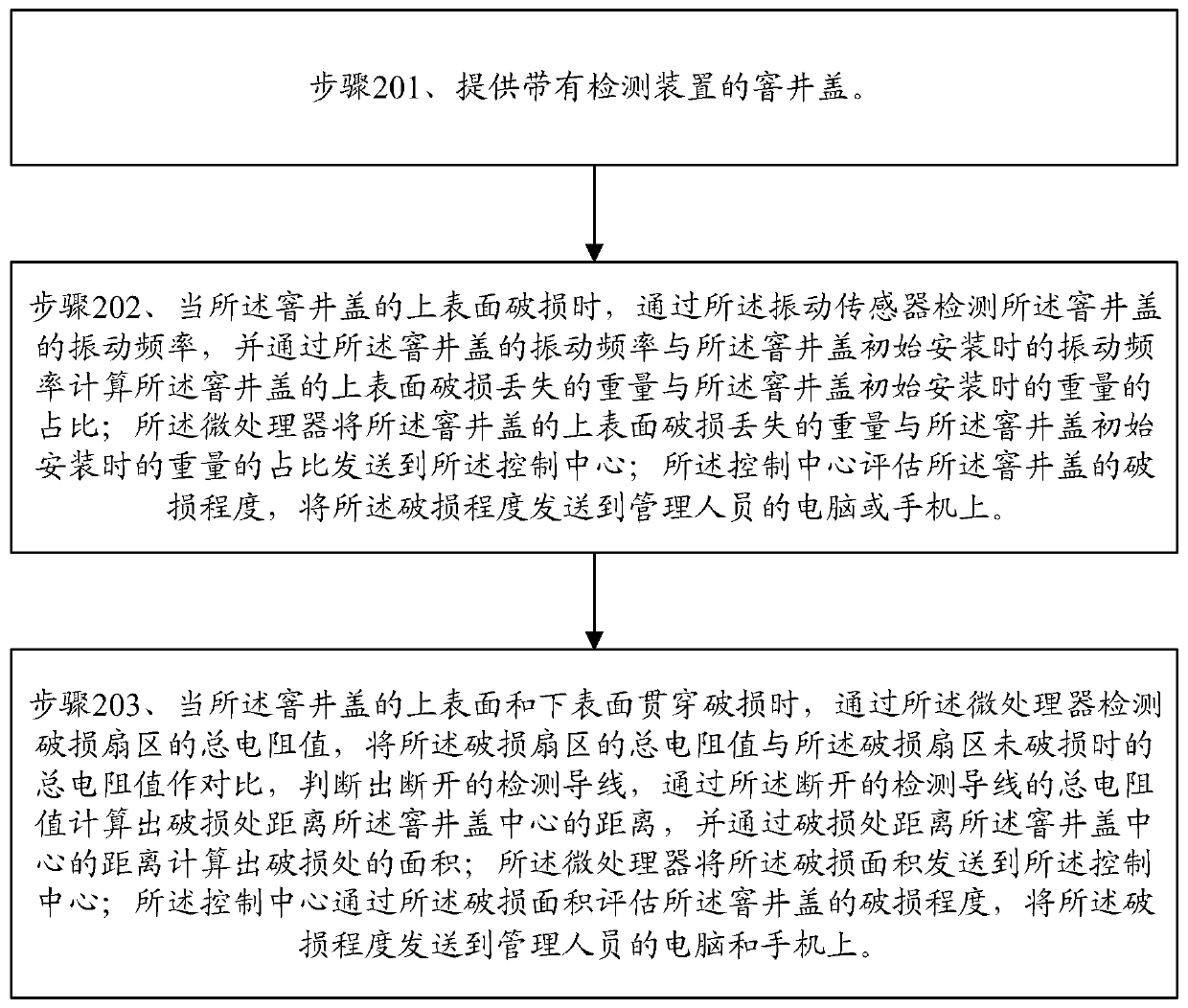
Method for detecting breakage of road surface manhole cover
ActiveCN109811798ARealize quantitative analysisComprehensive detectionArtificial islandsUnderwater structuresEngineeringRoad surface
The application discloses a method for detecting breakage of a road surface manhole cover. The method comprises the following steps: providing a manhole cover with a detecting device; when the upper surface of the manhole cover is broken, calculating the ratio of the breakage and loss weight of the upper surface of the manhole cover to the weight when the manhole cover is initially installed; whenthe upper surface and the lower surface of the manhole cover are broken in a penetrating manner, calculating the distance from the broken position to the centre of the manhole cover and the area of the broken position. According to the method for detecting the breakage of the road surface manhole cover, quantitative analysis of the breakage of the manhole cover is realized by adopting the detecting method of a multi-sector detection network; the area, the position and the broken area of the manhole cover can be accurately detected; comprehensive detection of the breakage of the manhole coveris facilitated, so that the level of the potential safety hazard of the manhole cover is evaluated, and priority processing of managerial personnel is facilitated; the treatment efficiency is improved. The penetrating breakage of the manhole cover can be detected; the breakage and the fragmentation of the upper surface of the manhole cover can also be detected and evaluated in time.
Owner:TAIHUA WISDOM IND GRP CO LTD
Nutritious breakfast and preparation method thereof
ActiveCN104116074APromote absorptionGreat tasteLactobacillusNatural extract food ingredientsAdditive ingredientSterculia nobilis
The invention belongs to the technical field of food and in particular relates to a nutritious breakfast and a preparation method thereof. The nutritious breakfast comprises the following ingredients in parts by weight: 4-8 parts of black soy beans, 2-4 parts of red dates, 2-4 parts of peanuts, 4-8 parts of soya beans, 1-2 parts of walnuts, 1-2 parts of eggs, 1-1.8 parts of kelp, 0.5-1 part of abalone, 0.4-0.8 part of corn starch, 0.5-0.9 part of gelatin, 1-2 parts of sterculia nobilis, 1-2 parts of indochina dragonplum fruit, 0.2-0.7 part of perilla leaf, 0.3-0.6 part of fresh ginger, 0.5-2 parts of lemon, 0.2-0.3 part of lactic acid bacteria and 0.1-0.2 part of beta-cyclodextrin. The nutritious breakfast contains various nutrient elements and is simple and convenient to eat and convenient to carry. The nutritious breakfast has rich and balanced nutrition and meets the physiological needs of a human body in the morning. Meanwhile, the nutritious breakfast also has the effect of reducing fat so as to help women to loss weight. The nutritious breakfast has varied edible methods and is unique in taste and can greatly improve appetite.
Owner:SHANDONG AGRI SUSTAINABLE DEV INST
Fat losing beauty device
InactiveCN103479504ABurn fullyLose weight easily and completelyElectrotherapyVibration massageBody shapePower flow
The invention relates to a shaping apparatus, in particular to a fat losing beauty device, which comprises a rectangular main body and a long binding belt. A control switch, a storage battery, an infrared heating unit or a low-current heating unit and a plurality spherical magnets are arranged in the main body, wherein the magnets are provided with quiet motor and embedded uniformly on the main body, the binding belt is fixed on and connected with one end of the main body, one surface of the binding belt is provided with a paste sub button and a paste snap button, and the end of the main body is connected with buckle via connecting band. The invention allows the user to loss weight safely and thoroughly by the simplest sliming way, moreover, the fat losing beauty device has the good and stable body shaping effects without rebound.
Owner:宋波
Build-in telescopic arrest spontoon
The invention discloses a build-in telescopic arrest spontoon. The build-in telescopic arrest spontoon comprises a fixed tube with one end provided with a handle and a telescopic tube arranged in the fixed tube in a sleeving mode; oppositely-arranged arrest forks are arranged in the telescopic tube, the other end of each arrest fork is connected with an arrest rod, and shrapnel used for opening the arrest forks are arranged on the connection portion between the arrest forks and the arrest rods. The build-in telescopic arrest spontoon is small in volume and convenient to carry on, and in the normal state, the build-in telescopic arrest spontoon is a spontoon and capable of performing deterring and hitting on a criminal offender and knocking on objects. The criminal offender can be arrested after an arrest device hidden in the spontoon is opened; the neck or the legs of the criminal offender are caught by the area formed by the arrest forks and the arrest rods and then pulled backwards, the criminal offender losses weight to fall down, so that the goal of subduing the criminal offender is achieved, and good initiative offense and passive defense functions are achieved.
Owner:天津苇江安防装备科技发展有限公司
New method for measuring alloy boiling point-method of loss of weight
InactiveCN1444039AThe method is simpleSolve the problem that the boiling point of the alloy cannot be determinedMaterial weighingInvestigating phase/state changeBoiling pointAlloy
The method for measuring alloy property boiling point-weight has method adopted the following steps: using precision balance to weight small quantity of tested alloy powder, placing the alloy powder into a porcelain boat, weighing total weight of alloy powder and porcelain boat, placing them into a porcelain pipe in high-temp. furnace, under the condition of argon protection and constant pressurequickly-heating to predefined temp., heat-insualting for 10-30 min., then quickly-cooling to room temp., then weight the total weight of porcelain boat and residual alloy powder and resolving evaporated quantity, according to the evaporated loss weight quantity measured at several temp. points calcualting evaporated loss weight percentage content and drawing temp.-evaporated percentage content curve.
Owner:DALIAN UNIV OF TECH
Traffic illuminating system
PendingCN107990250ALess hitPrevent sprayingMechanical apparatusLighting support devicesEngineeringLighting system
The invention discloses a traffic illuminating system which comprises a lamp pole and a lamp holder which are fixedly connected. A watering device, a water pump and an infrared induction device used for detecting the human body are arranged on the lamp pole, the end, close to the lamp holder, of the lamp pole is rotationally connected with the watering device with the lamp pole as the axis, the water pump is communicated with the watering device, the infrared induction device is coupled with the water pump, when the human body is detected by the infrared induction device, an induction signal is transmitted to the water pump, watering is stopped after the induction signal is received by the water pump, the watering device is rotationally connected to the lamp pole, and therefore the watering device rotates to achieve large-area afforesting through spraying; and as the watering device is arranged at the end, close to the lamp holder, of the lamp pole, water can be sprayed out from the high position, water drops loss weight when falling from the high position, water can be further scattered, the weight of water can be scattered, and impact to afforesting is reduced.
Owner:三石建工集团有限公司
Method for manufacturing light superfine fiber base cloth
InactiveCN1294302CFilament/thread formingMonocomponent polyolefin artificial filamentPolyolefinPolymer science
The invention associates to a method preparing supercritical fiber, which includes island component polymer and sea component polymer spinning supercritical fiber. The key point is that island component is polyolefin polymer and sea component is a polymer which is different in solution. The method offers a way to produce a kind of superfine fiber base cloth. Using superfine fiber produced in that way we can get a sort of backing material, and then let sea component of backing material solute to get the superfine fiber base cloth. Because of the feature that island component made from polyolefin polymer is of high bending elasticity and low density, the the superfine fiber base cloth produced in this way can loss weight very much and be up to the thickness when sea component solute.
Owner:SAN FANG CHEM IND
Dynamic weight measuring method for steel ball mill
InactiveCN104236685AGood milling stateImprove milling efficiencyWeighing apparatus using fluid action balancingMetallurgySteel ball
The invention provides a dynamic weight measuring method for a steel ball mill, wherein a B-P function characteristic curve among a cylinder body total weight B (comprising milled object weight and steel ball weight), a high-pressure lubricant pressure P and a contact area S of the steel ball mill can be obtained through establishing a basic model and a true model. According to the high-pressure lubricant pressure P which is measured in real time in operation, the real-time cylinder body weight B can be obtained through reverse computing. The dynamic weight measuring method can conveniently and quickly detect the real-time dynamic weight of the steel ball mill, thereby calculating steel loss weight, obtaining a steel loss curve, and correctly guiding personnel to add steel balls. Simultaneously through comparing the steel loss of steel balls and liner plates which are in different brands or are made of different materials, the dynamic weight measuring method can easily determine which material or brand of the liner plates or steel balls is better. Furthermore a real-time ball to powder ratio can be detected, so that the steel ball mill is always kept in an optimal pulverizing state, thereby greatly improving powder the pulverizing efficiency of the steel ball mill, greatly reducing power consumption of the steel ball mill, and truly realizing energy reduction and production increase.
Owner:SHANGHAI YIFENG ELECTRICAL & MECHANICAL TECH DEV
Method and device for security configuration optimization
ActiveCN102710641BMeet safety requirementsLow usability impactTransmissionConfiguration optimizationConfiguration item
The invention discloses a method and a device for security configuration optimization, which are used for solving the problems that the usability of a configured system is low in the prior art. The method comprises the steps of determining configuration item assemblies with the sum of the security weights corresponding to contained configuration items larger than a set security threshold according to the security weights corresponding to the configuration items contained in the system, selecting a configuration item assembly with the minimum sum of usability loss weights corresponding to the contained configuration terms in the determined configuration item assemblies according to the usability loss weights corresponding to the configuration terms, and configuring the system according to the contained configuration terms in the selected configuration item assembly. According to the method, the selected configuration item assembly not only meets the security requirement of the system, but also least affects the usability of the system, so that as being configured according to the selected configuration item assembly, the configured system meets the security requirement and simultaneously improves the usability.
Owner:NSFOCUS INFORMATION TECHNOLOGY CO LTD
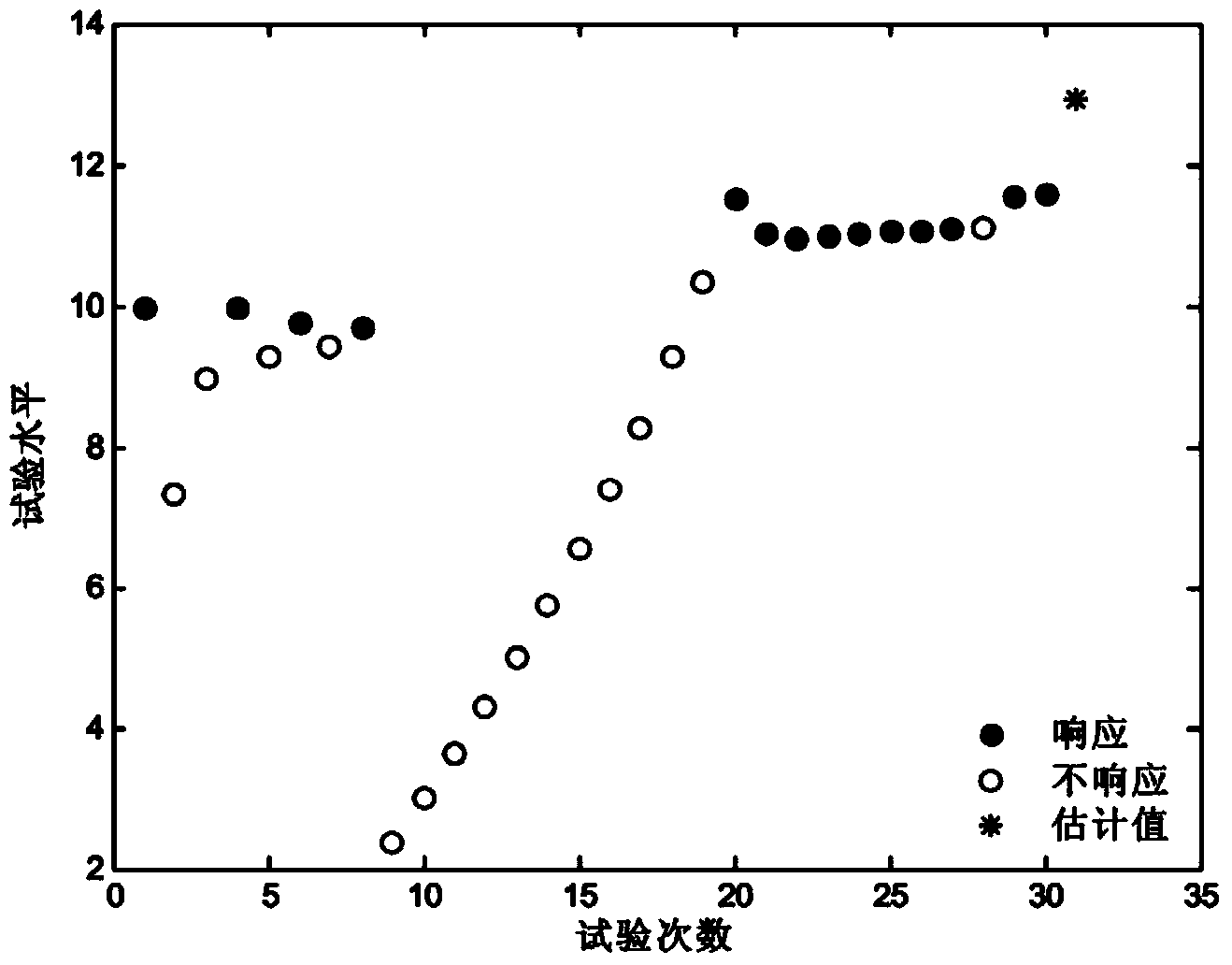
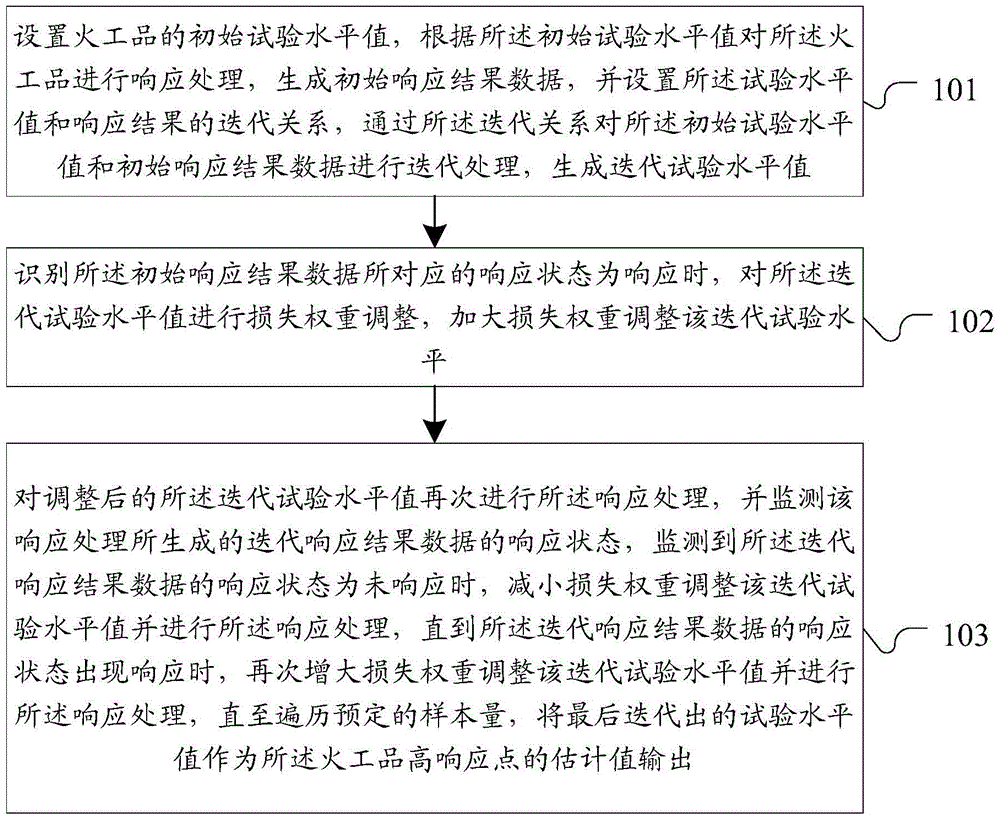
Method and system for estimating initiating explosive device high-response points
ActiveCN103823987AAvoid futile trialsImprove estimation accuracySpecial data processing applicationsValue setComputer science
The invention discloses a method and a system for estimating initiating explosive device high-response points. According to the method, an initiating explosive device initial test level value is set, the response processing is carried out on initiating explosive devices, initial response result data is generated, the iteration relationship between the test level value and the response result is set, and the iteration processing is carried out for generating the iteration test level value; when the identification result shows that the response state corresponding to the initial response result data is response, the loss weight is increased, the iteration test level value is regulated, the processing response is carried out again, when no response is monitored, the loss weight is decreased, the iteration test level value is regulated for realizing the processing response until the response occurs, the weight loss is increased again, in addition, the response processing is carried out until the preset sample quantity is traversed, and finally, the test level value obtained through iteration is used as the estimation value of the initiating explosive device high-response points to be output. The method and the system solve the problems that in the estimation test method of the initiating explosive device high-response points, the estimation precision is poor, the invalid test occurrence rate is higher, and the estimation precision is seriously reduced due too the initial value setting deviation.
Owner:BEIJING INSTITUTE OF TECHNOLOGYGY
Method and system for estimating pyrotechnic high response point
ActiveCN103823987BAvoid futile trialsImprove estimation accuracySpecial data processing applicationsValue setSimulation
The invention discloses a method and a system for estimating initiating explosive device high-response points. According to the method, an initiating explosive device initial test level value is set, the response processing is carried out on initiating explosive devices, initial response result data is generated, the iteration relationship between the test level value and the response result is set, and the iteration processing is carried out for generating the iteration test level value; when the identification result shows that the response state corresponding to the initial response result data is response, the loss weight is increased, the iteration test level value is regulated, the processing response is carried out again, when no response is monitored, the loss weight is decreased, the iteration test level value is regulated for realizing the processing response until the response occurs, the weight loss is increased again, in addition, the response processing is carried out until the preset sample quantity is traversed, and finally, the test level value obtained through iteration is used as the estimation value of the initiating explosive device high-response points to be output. The method and the system solve the problems that in the estimation test method of the initiating explosive device high-response points, the estimation precision is poor, the invalid test occurrence rate is higher, and the estimation precision is seriously reduced due too the initial value setting deviation.
Owner:BEIJING INSTITUTE OF TECHNOLOGYGY
Fresh-keeping storage method of garlic
InactiveCN102204585ALong validity periodThere will be no loss of weightFruits/vegetable preservation by freezing/coolingAllium sativumBiology
The invention belongs to the field of fruit and vegetable fresh-keeping storage technologies, and discloses a fresh-keeping storage method of garlic. The method comprises the following steps: placing garlic in a sealing bag; sealing the sealing bag, and placing the bag of garlic in a cold storage for refrigerating and preserving. Advantages of the present invention are that: the expiration date of the garlic stored with the method provided by the present invention can be prolonged to about two years, the garlic does not loss weight during the preservation period, original mouthfeel of the garlic can be effectively maintained, and the operation of the storage processes is simple.
Owner:周再兴
Quality control method of asthma tablets
ActiveCN101112424BGuaranteed reproducibilityGuaranteed efficacyComponent separationRespiratory disorderWater bathsSlag
Owner:贵州省科晖制药有限公司
Pickling solution and application of weight loss analysis to detect corrosion of metal materials
ActiveCN106894025BEasy to prepareGuaranteed stabilityWeighing by removing componentTetramineAcid corrosion
The invention belongs to the field of application of a weightlessness method in the field of metal material corrosion, in particular to a pickling solution for detecting corrosive substances in a metal material through weightlessness analysis and an application. The pickling solution contains concentrated hydrochloric acid, deionized water and hexamethylene tetramine in a volume-mass (ml / g) ratio being 5:100:9-80, wherein pH of the pickling solution is controlled to be 4.15-6.15. With the adoption of the method, accurate weightlessness data of copper corrosion products can be obtained, and loss of base metal is reduced. Meanwhile, a specified experimental process can guarantee comparability of data of different experiments.
Owner:INST OF OCEANOLOGY - CHINESE ACAD OF SCI
A Method of Improving the Accuracy of Steel Coil Weight Calculation
ActiveCN103530510BEliminate slippageGuaranteed accuracySpecial data processing applicationsMedicineRepair material
The invention provides a method for improving the calculation accuracy of the steel coil weight, adding a coil diameter calculation program with the outlet steering roller as the reference speed, when the strip steel thickness is ≥ 0.4mm, press D j =1.89×400×Pzl / Pj to calculate the coil diameter value; when the thickness of the strip steel is less than 0.4mm, press D j =K×1.64×600×Pzx / Pj to calculate the volume diameter value; according to m=π×(D j 2 -610 2 )×W×7850 to calculate the coil weight. K is the steering roller compensation factor, Pzl is the tension roller power value displayed by the code disk pulse counter, Pj is the coiler power value displayed by the code disk pulse counter, Pzx is the steering roller power value displayed by the code disk pulse counter, W is the belt steel width. The invention can eliminate the skidding phenomenon of the tension roller when the steel strip is thin or is repairing material, improve the calculation accuracy of the coil weight, narrow the gap between the actual coil weight and the calculated coil weight, and solve the problem of overweight or insufficient steel coils that has plagued enterprises for a long time. Heavy problems, reduce customer objections and processing workload.
Owner:ANGANG STEEL CO LTD
A construction method of millennium health fingerprints and its standard fingerprints
ActiveCN107037157BGuaranteed stabilityEffectively Characterize QualityComponent separationFiltrationSilanes
The invention discloses a method for constructing fingerprint spectra of rhizoma homalomenae and a standard fingerprint spectrum of the rhizoma homalomenae. The method includes steps of S1, precisely weighing rhizoma homalomenae powder, adding methanol into the rhizoma homalomenae powder, carrying out ultrasonic extraction, allowing extract to stand still, cooling the extract, complementing loss weights by the aid of methanol, centrifuging the extract, drying supernatant by evaporation, adding methanol into residues, re-dissolving the residues and carrying out vortex treatment and filtration to obtain subsequent filtrate which is test article solution; S2, sucking the test article solution into a liquid chromatograph and measuring the test article solution by means of gradient elution by the aid of high-performance liquid chromatography to obtain the fingerprint spectra of the rhizoma homalomenae. High-performance liquid chromatographic conditions include that octadecyl silane chemically bonded silica is used as a filler for each chromatographic column; acetonitrile is used as a mobile phase A, phosphoric acid aqueous solution is used as a mobile phase B, and a volume ratio of the mobile phase A to the mobile phase B is gradually increased along with the time; detection wavelengths are 285-305 nm. The method and the standard fingerprint spectrum have the advantages that the method is simple, convenient and stable and is high in precision and repeatability, and the method and the standard fingerprint spectrum can be used for controlling the comprehensive quality of rhizoma homalomenae medicinal materials.
Owner:SICHUAN PROVINCIAL PEOPLES HOSPITAL
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com