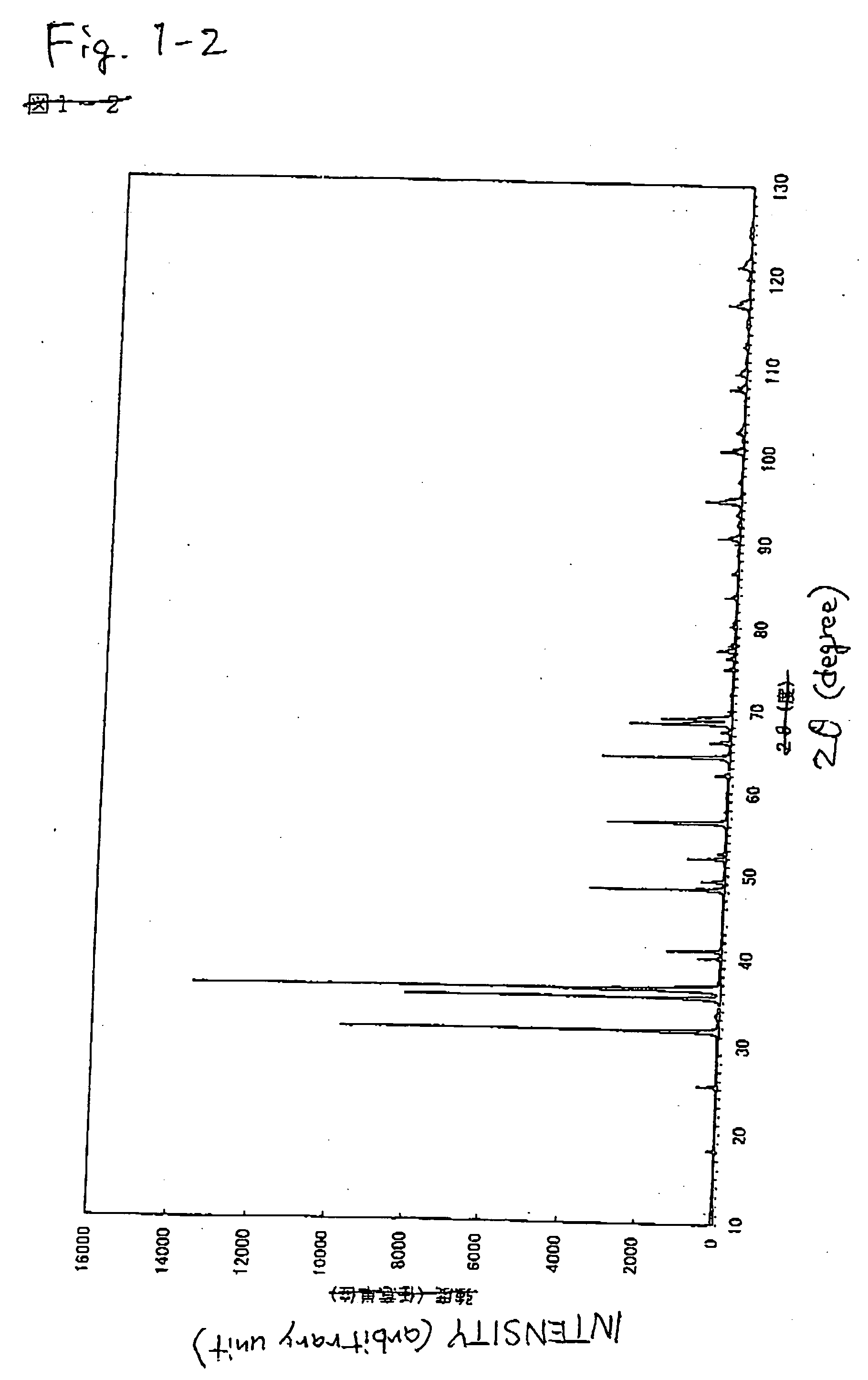
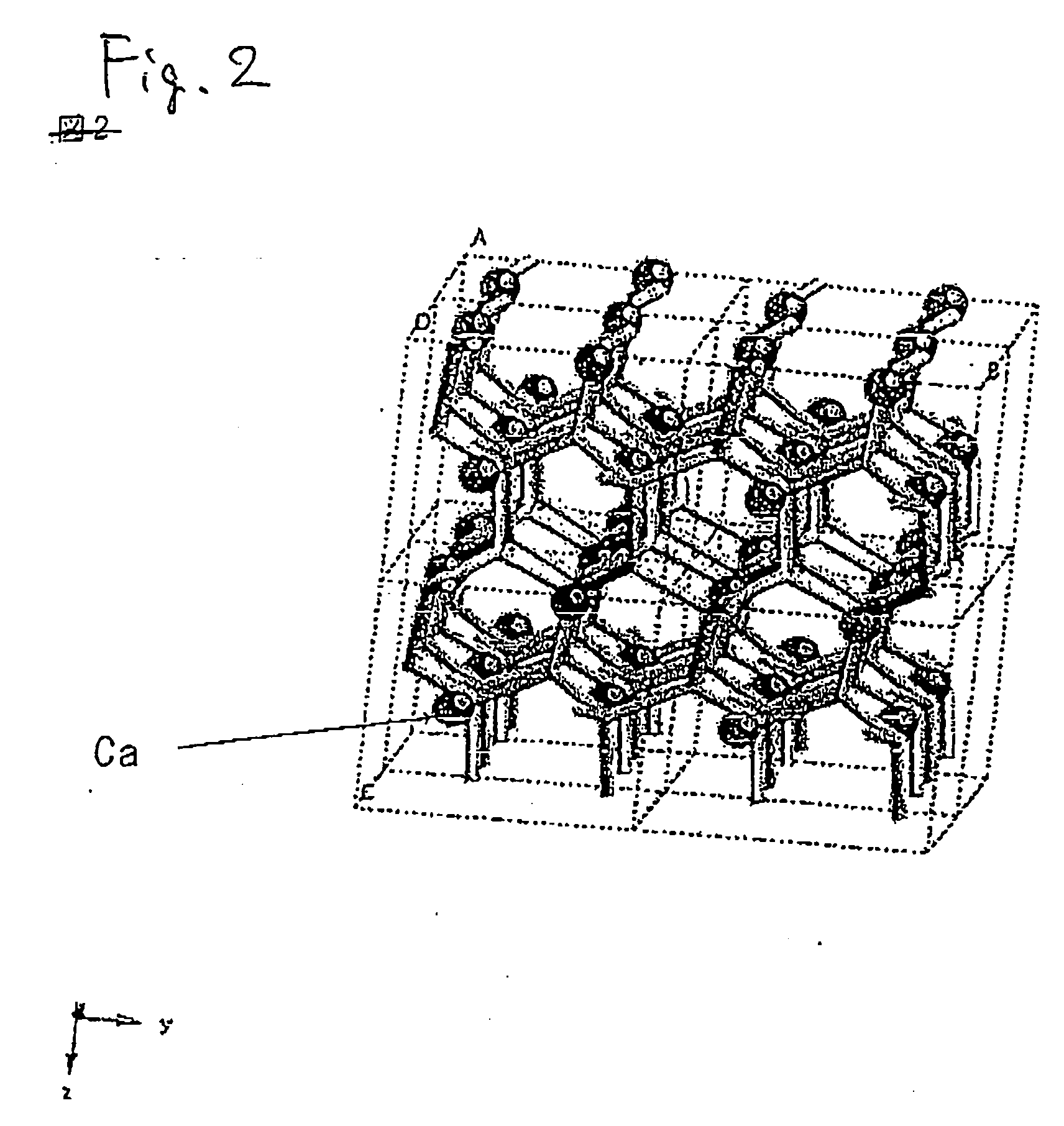
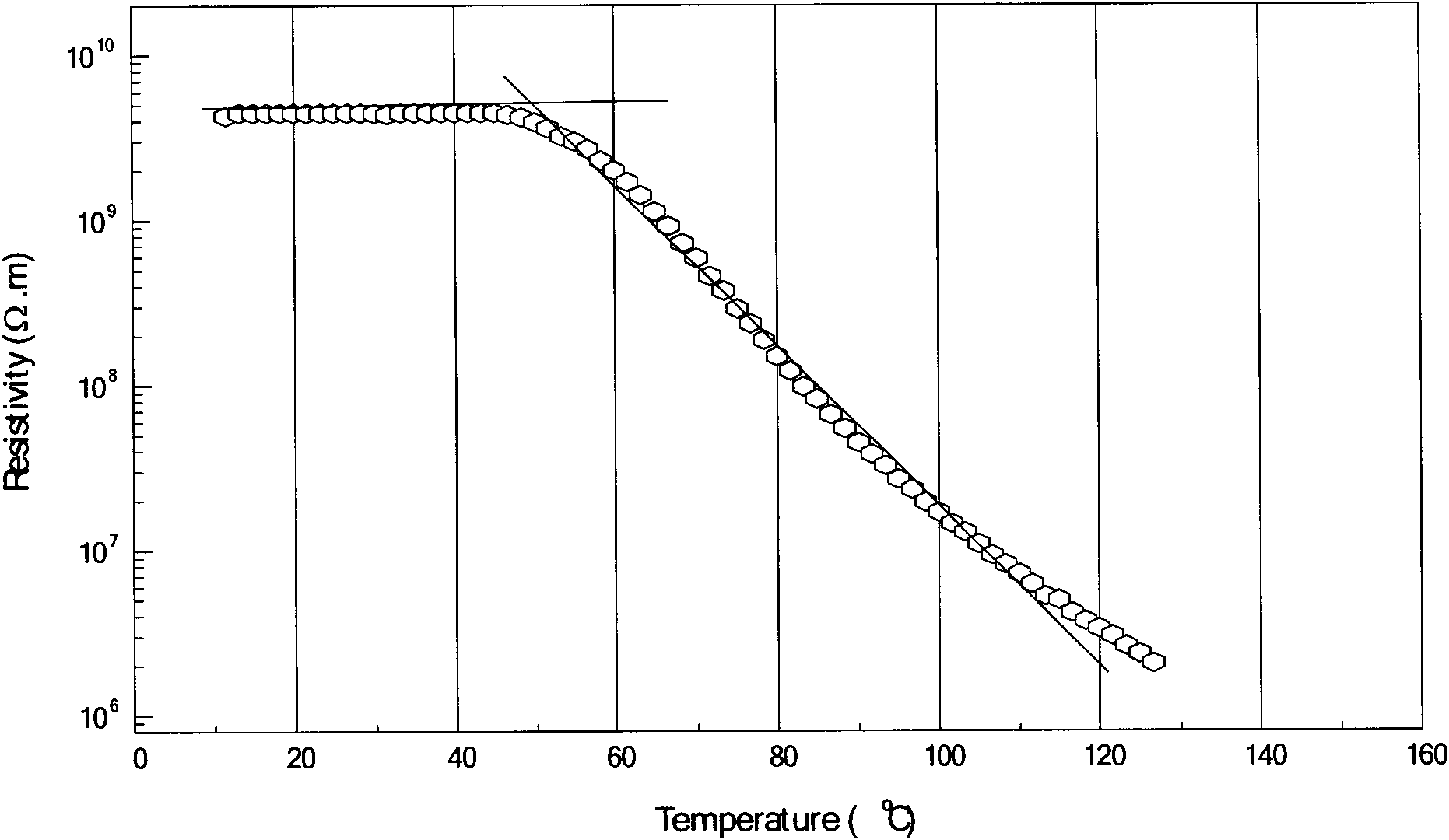
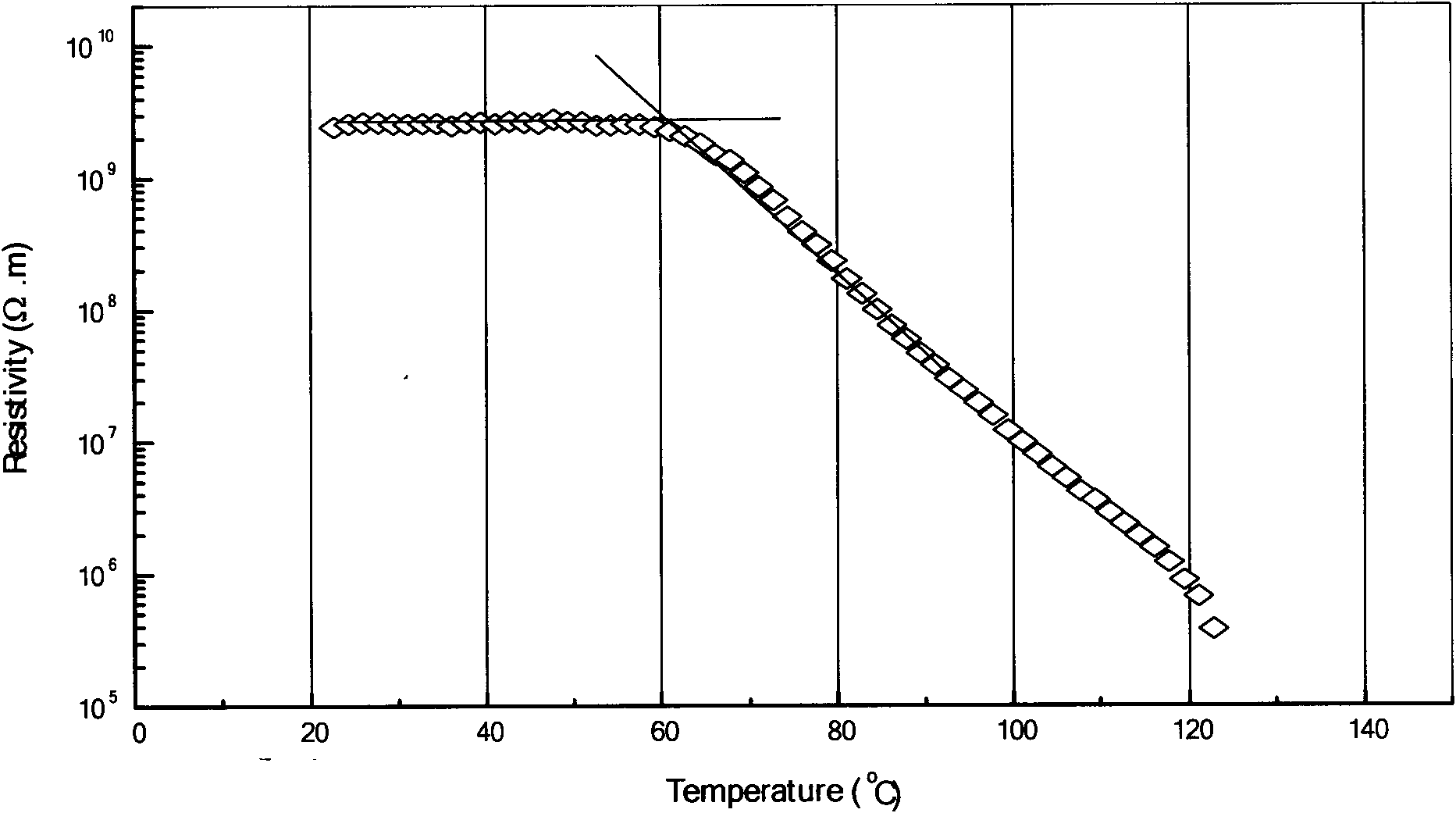
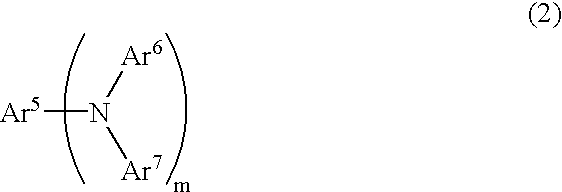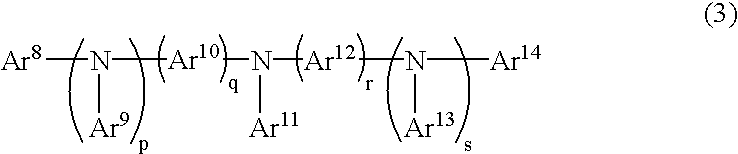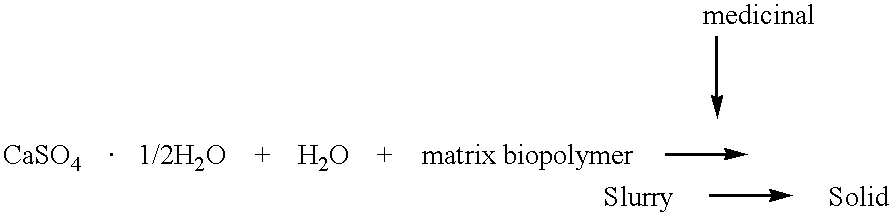Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
2511 results about "Inorganic compound" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
An inorganic compound is typically a chemical compound that lacks C-H bonds, that is, a compound that is not an organic compound, but the distinction is not defined or even of particular interest. Inorganic compounds comprise most of the Earth's crust, although the compositions of the deep mantle remain active areas of investigation.
Organic devices, organic electroluminescent devices, organic solar cells, organic FET structures and production method of organic devices
ActiveUS20050098207A1TransistorDischarge tube luminescnet screensElectronic transmissionOrganic solar cell
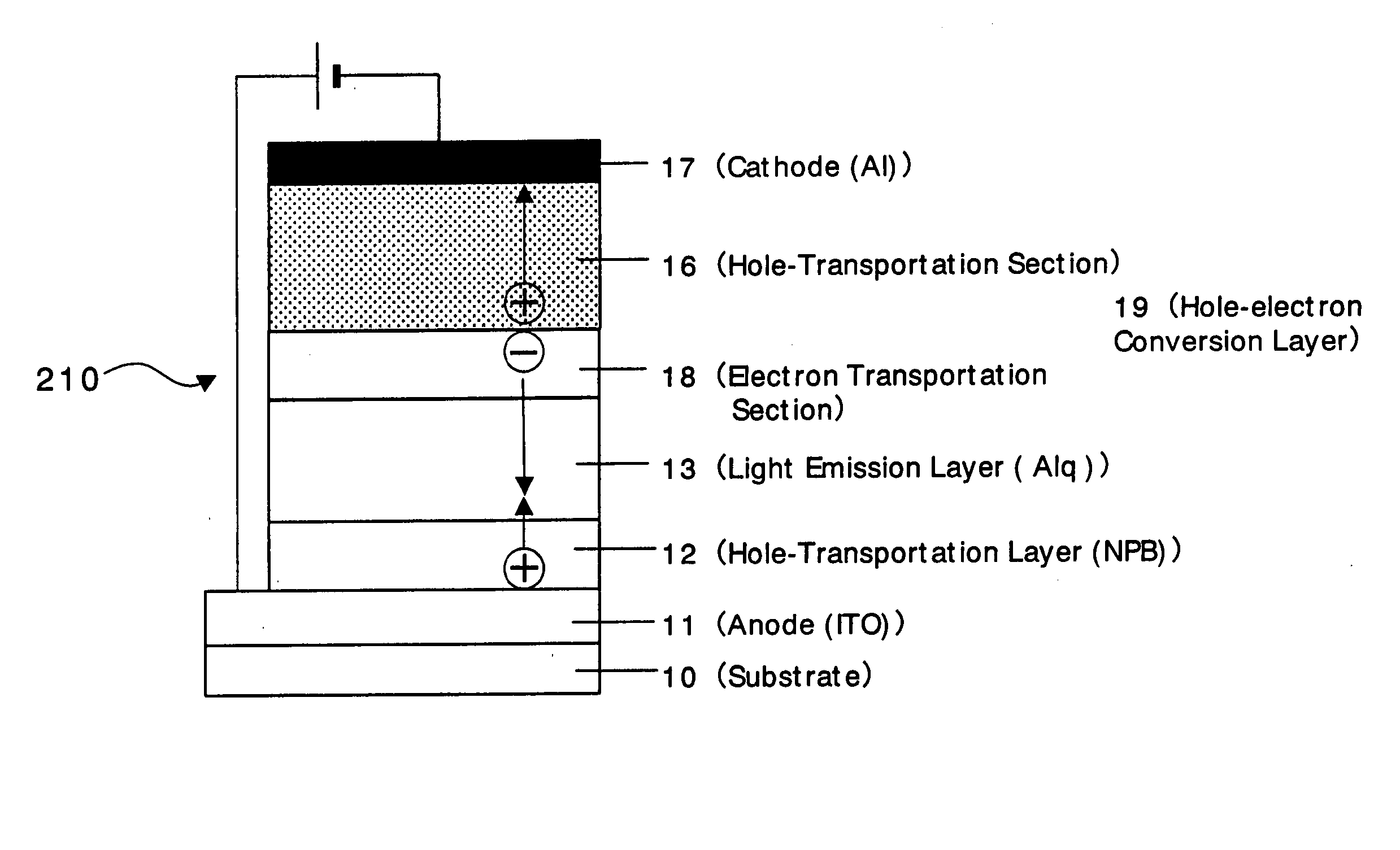
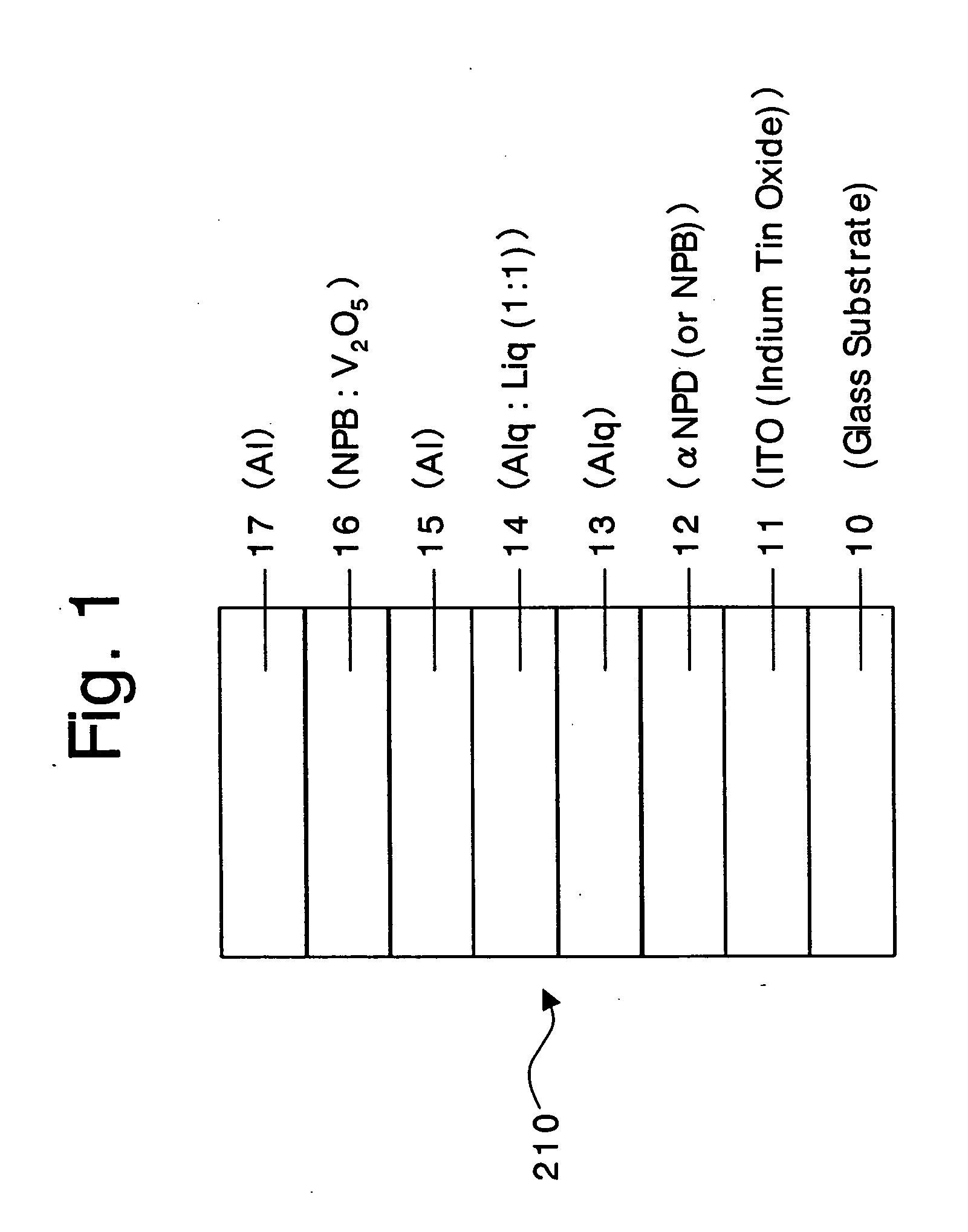
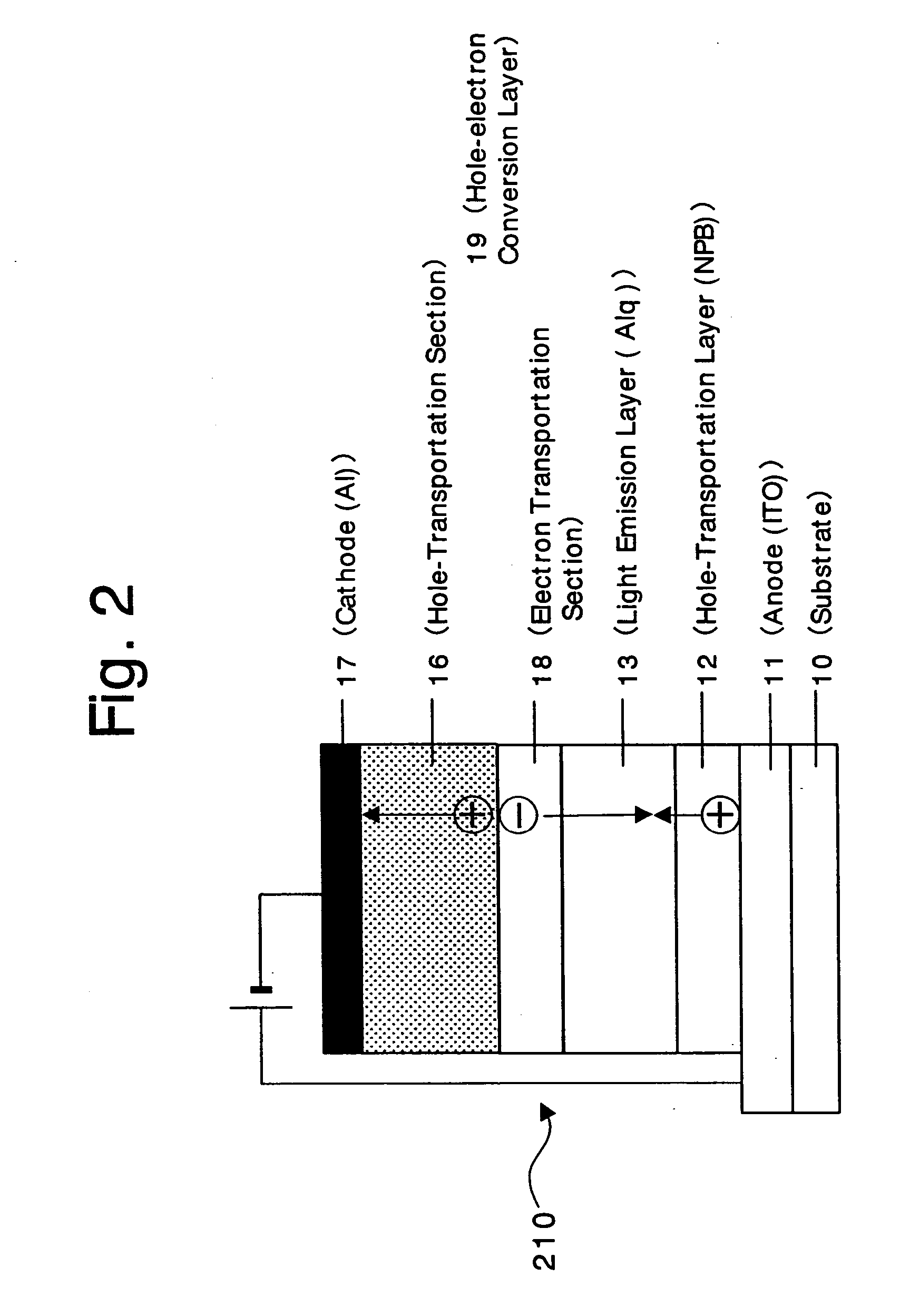
An organic device has a hole current-electron current conversion layer which comprises a laminate of an electron transportation section and a hole transportation section. The electron transportation section includes a charge transfer complex formed upon an oxidation-reduction reaction between a reduced low work function metal and an electron-accepting organic compound, the reduced metal being produced upon an in-situ thermal reduction reaction caused upon contact, through lamination or mixing by co-deposition, of an organic metal complex compound or an inorganic compound containing at least one metal ion selected from ions of low work function metals having a work function of not more than 4.0 eV, and a thermally reducible metal capable of reducing a metal ion contained in the organic metal complex compound or the inorganic compound in vacuum to the corresponding metal state, and the electron transportation section having the electron-accepting organic compound in the state of radical anions. The hole transportation section includes an organic compound having an ionization potential of less than 5.7 eV and an electron-donating property and an inorganic or organic substance capable of forming a charge transfer complex upon its oxidation-reduction reaction with the organic compound, the organic compound and the inorganic or organic substance being contacted through lamination or mixing, and the electron-donating organic compound is in the state of radical cations.
Owner:MITSUBISHI HEAVY IND LTD +1
Phosphor and light-emitting equipment using phosphor
ActiveUS20070007494A1Increase brightnessGood colorOther chemical processesNitrogen-metal/silicon/boron binary compoundsFluorescenceRare earth
An object of the present invention is to provide an inorganic phosphor having fluorescence properties emitting an orange or red light which has a longer wavelength as compared with the cases of conventional sialon phosphors activated with a rare earth. The invention relates to a design of white light-emitting diode rich in a red component and having good color-rendering properties by employing a solid solution crystal phase phosphor which uses as a host crystal an inorganic compound having the same crystal structure as that of a CaSiAlN3 crystal phase and to which M Element (wherein M Element is one or two or more elements selected from the group consisting of Mn, Ce, Pr, Nd, Sm, Eu, Tb, Dy, Ho, Er, Tm, and Yb) is added as an emission center.
Owner:NICHIA CORP +1
Temperature sensing cable material with CRT characteristics and preparation method thereof
ActiveCN101613504AWith CTR featureFlexibleRubber insulatorsPlastic/resin/waxes insulatorsProcedure AgentsAntioxidant
The invention relates to technical fields of materials, chemical engineering and electronics, in particular to a cable material with a polymer substrate and CTR effect and a preparation method thereof. The temperature sensing cable material with CRT characteristics consists of thermoplastic polymer, a filling agent, a plasticizer, functional additives, a stabilizing agent, a flame retardant, a lubricant, an antioxidant and a processing agent, has the use temperature range between -30 and 150 DEG C, and can be applied on occasions such as recoverable line-type temperature sensing cables, fire prewarning and alarm devices, temperature measurement, overheating alarm and protection and the like. The temperature sensing cable material with the CRT characteristics takes a polymer material as a substrate, and is added with multiple organic and inorganic compounds with special CTR characteristics taken as the functional additives, and has the characteristics of flexibility, and capacities of being continuously processed and being used like a common cable material.
Owner:QINGDAO SUNYFIRE TECHNOLOGE
Stably tethered structures of defined compositions with multiple functions or binding specificities
ActiveUS20060228300A1Reduce exposureReduce deliveryAntibacterial agentsSenses disorderAntibody fragmentsBinding peptide
The present invention concerns methods and compositions for stably tethered structures of defined compositions with multiple functionalities and / or binding specificities. Particular embodiments concern stably tethered structures comprising a homodimer of a first monomer, comprising a dimerization and docking domain attached to a first precursor, and a second monomer comprising an anchoring domain attached to a second precursor. The first and second precursors may be virtually any molecule or structure, such as antibodies, antibody fragments, antibody analogs or mimetics, aptamers, binding peptides, fragments of binding proteins, known ligands for proteins or other molecules, enzymes, detectable labels or tags, therapeutic agents, toxins, pharmaceuticals, cytokines, interleukins, interferons, radioisotopes, proteins, peptides, peptide mimetics, polynucleotides, RNAi, oligosaccharides, natural or synthetic polymeric substances, nanoparticles, quantum dots, organic or inorganic compounds, etc. The disclosed methods and compositions provide a simple, easy to purify way to obtain any binary compound attached to any monomeric compound, or any trinary compound.
Owner:IBC PHARMACEUTICALS INC
Chemical manufacture of nanostructured materials
InactiveUS6872330B2High strengthIncrease volumeMaterial nanotechnologyOxide/hydroxide preparationInorganic compoundTe element
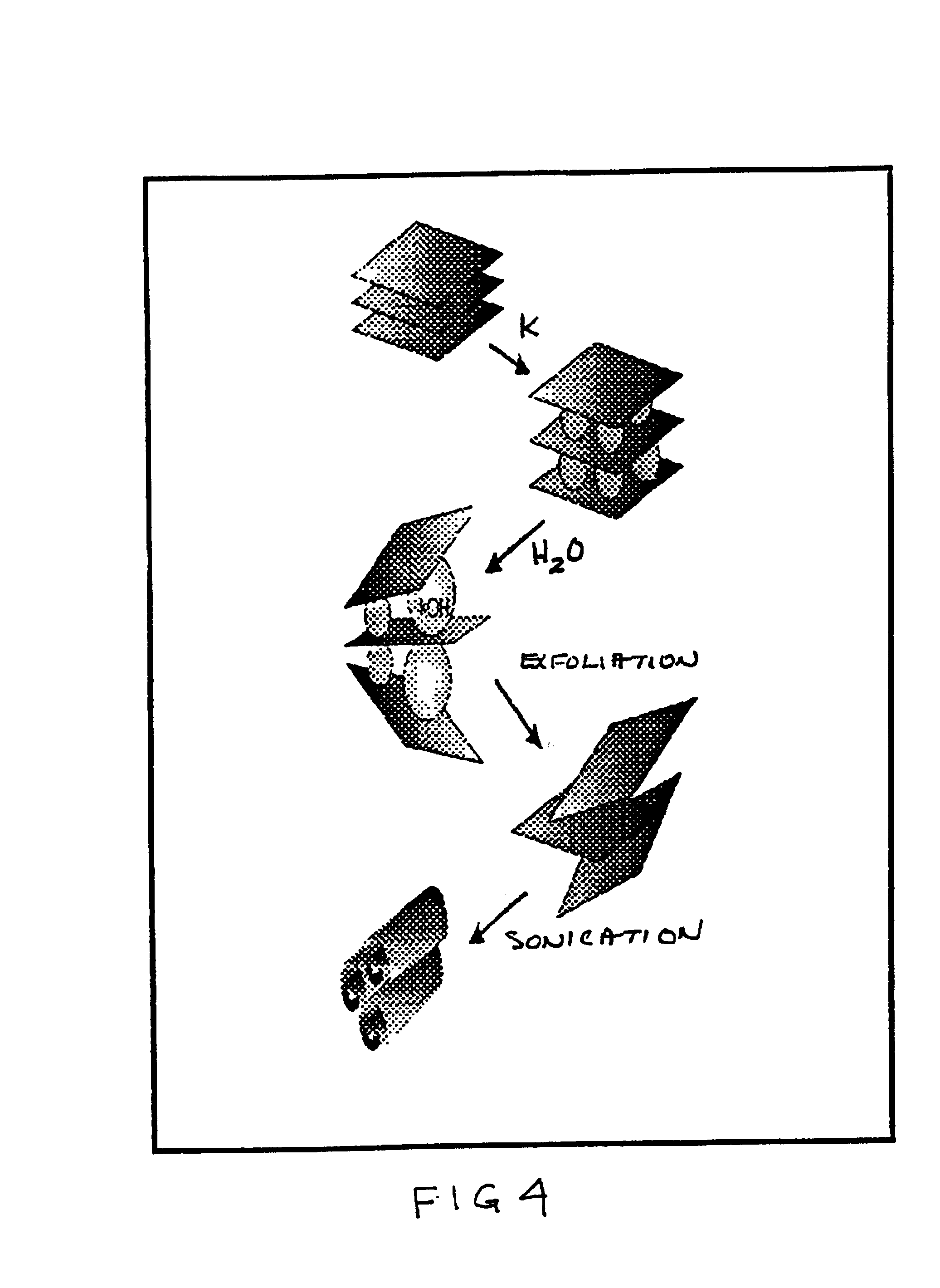
A low temperature chemical route to efficiently produce nanomaterials is described. The nanomaterials are synthesized by intercalating ions into layered compounds, exfoliating to create individual layers and then sonicating to produce nanotubes, nanorods, nanoscrolls and / or nanosheets. It is applicable to various different layered inorganic compounds (for example, bismuth selenides / tellurides, graphite, and other metal complexes, particularly transition metal dichalcogenides compounds including oxygen, sulfur, tellurium or selenium).
Owner:RGT UNIV OF CALIFORNIA
Organic electroluminescent device and method of manufacture thereof
InactiveUS6416888B1Increased durabilityReduce the driving voltageDischarge tube luminescnet screensElectroluminescent light sourcesInorganic compoundThin layer
An organic EL device having a low driving voltage and exhibiting high luminous brightness and superior durability, and a method of manufacturing the same. The organic EL device has an anode layer, an organic light-emitting layer, and a cathode layer. An inorganic thin layer, comprising an inorganic compound of Ge, Sn, Zn, Cd, etc. and an inorganic compound of an element of Group 5A to Group 8 in the periodic table in combination, is provided between the anode layer and the organic light-emitting layer and between the cathode layer and the organic light-emitting layer, or the anode layer or the cathode layer comprises a chalcogenide of Si, Ge, Sn, Pb, Ga, In, Zn, Cd, Mg, etc. and an inorganic compound of an element of Group 5A to Group 8 in the periodic table in combination.
Owner:IDEMITSU KOSAN CO LTD
Organic electroluminescent device and production method of the device, and display apparatus
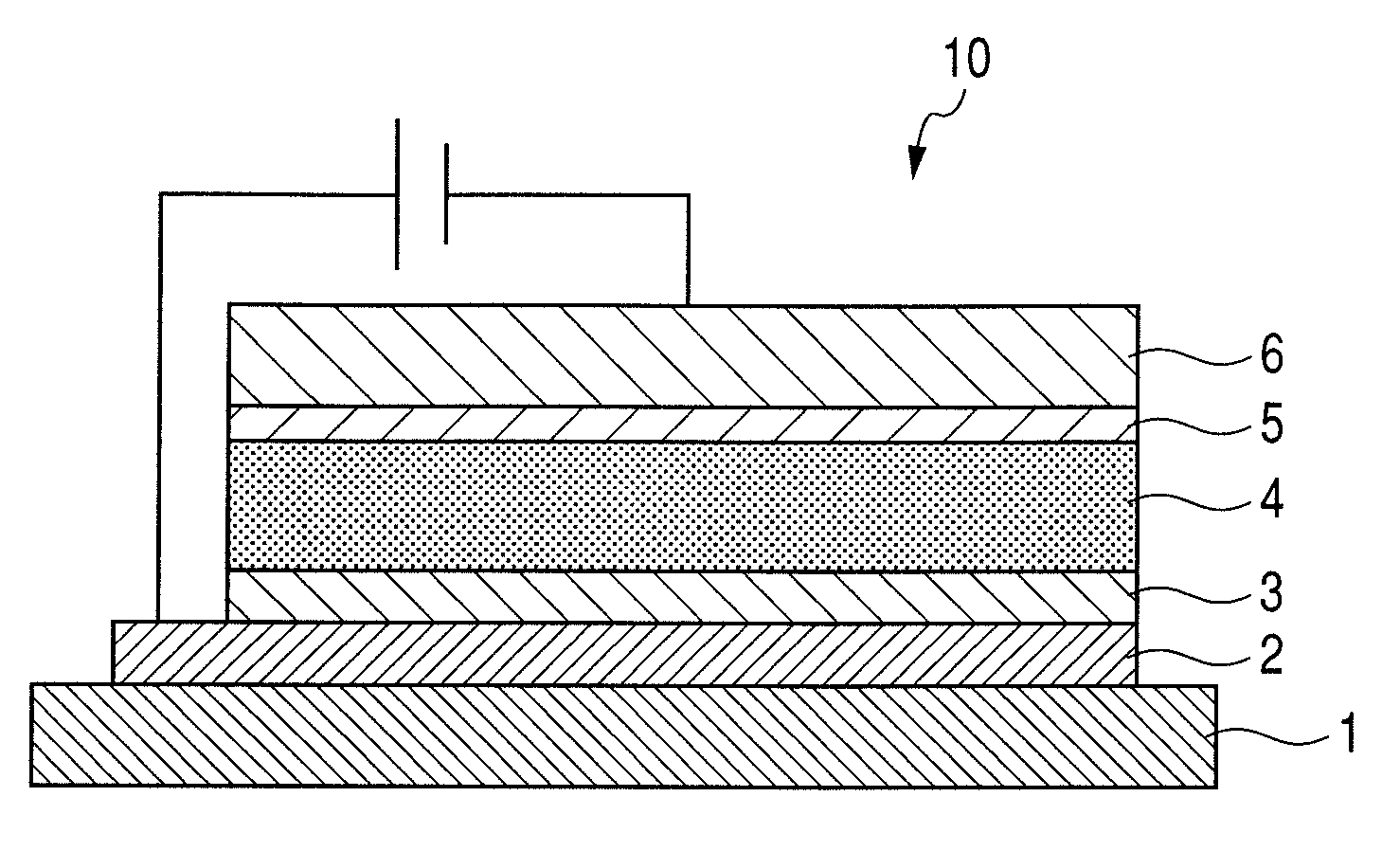
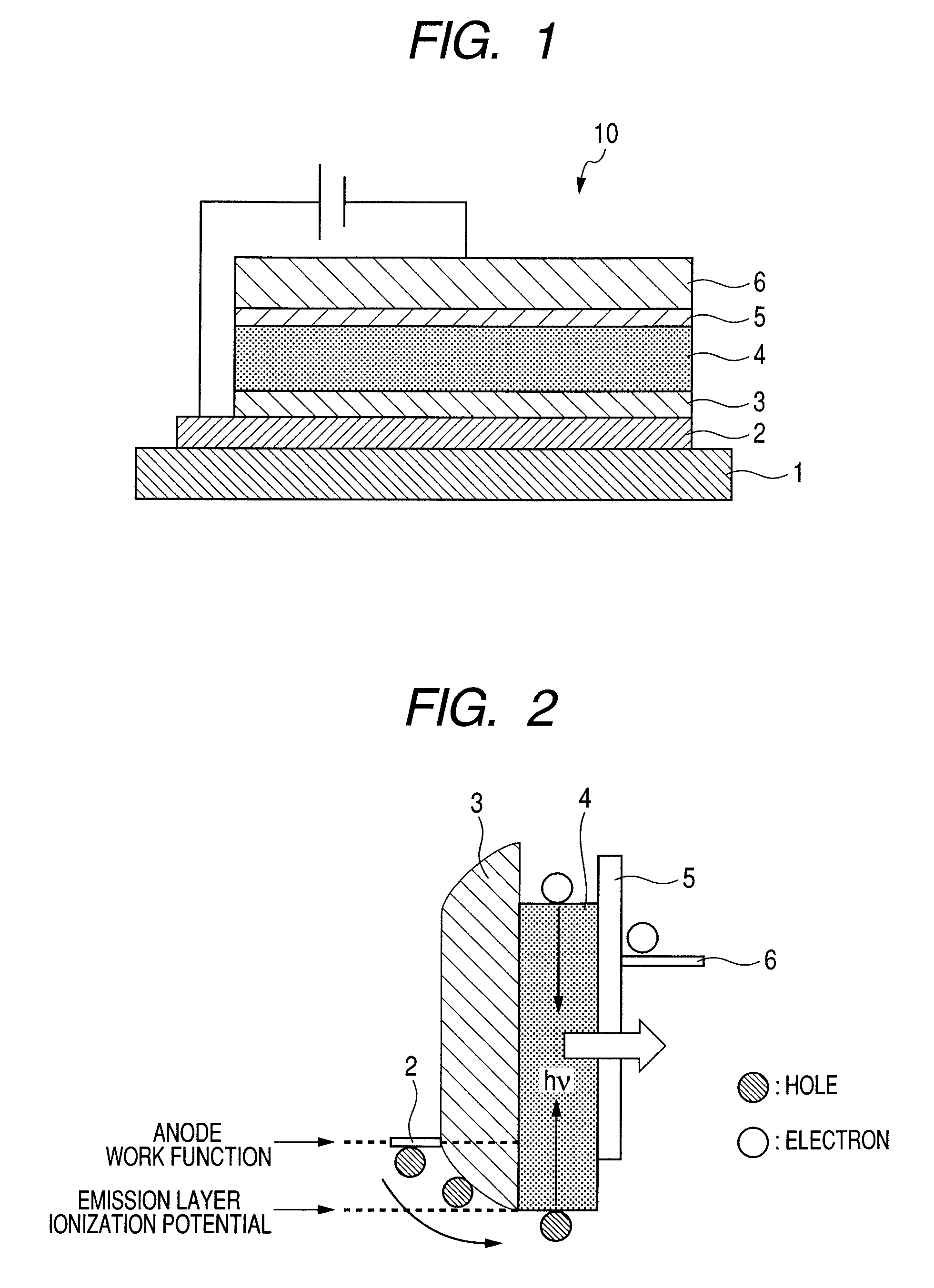
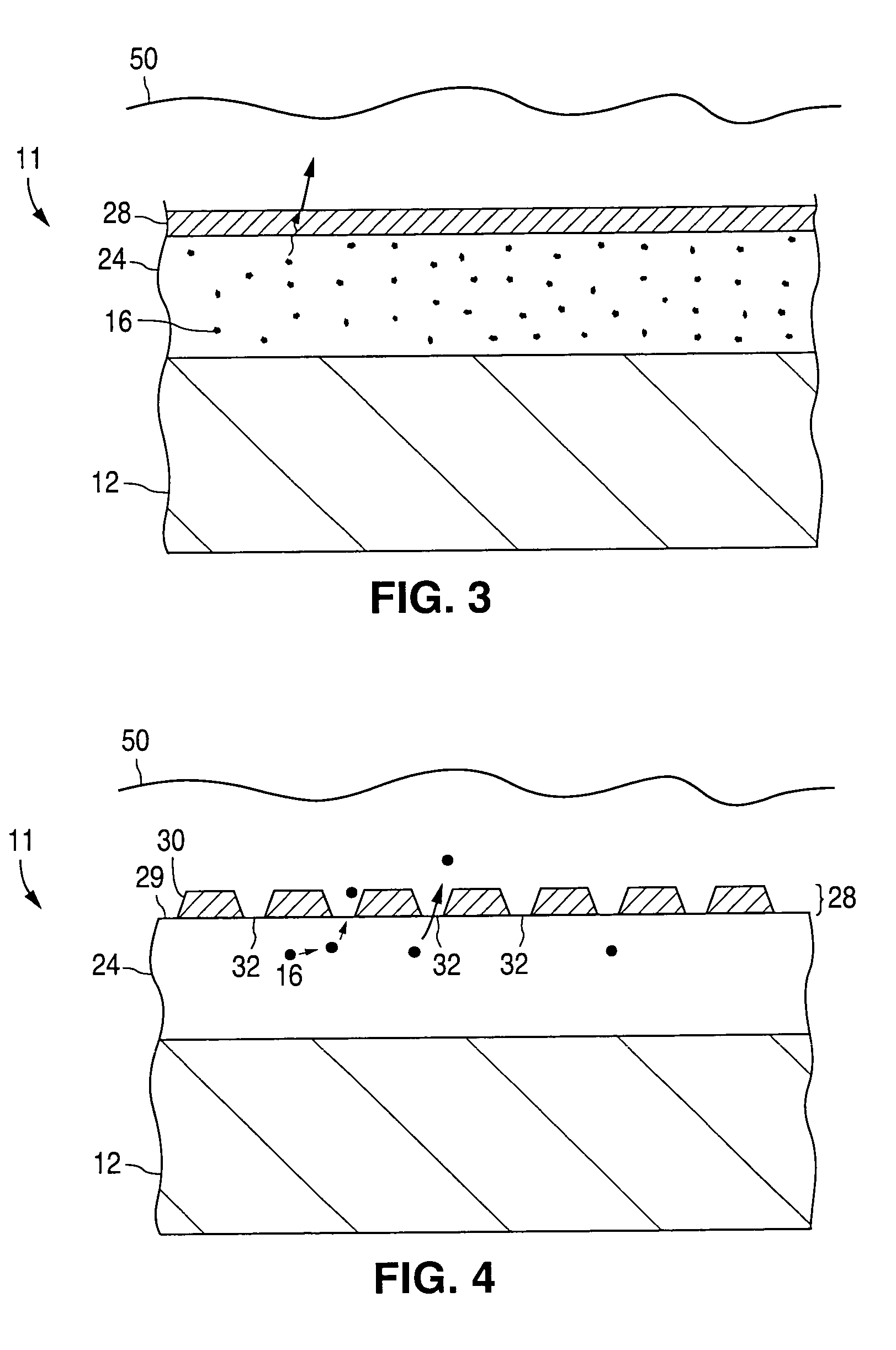
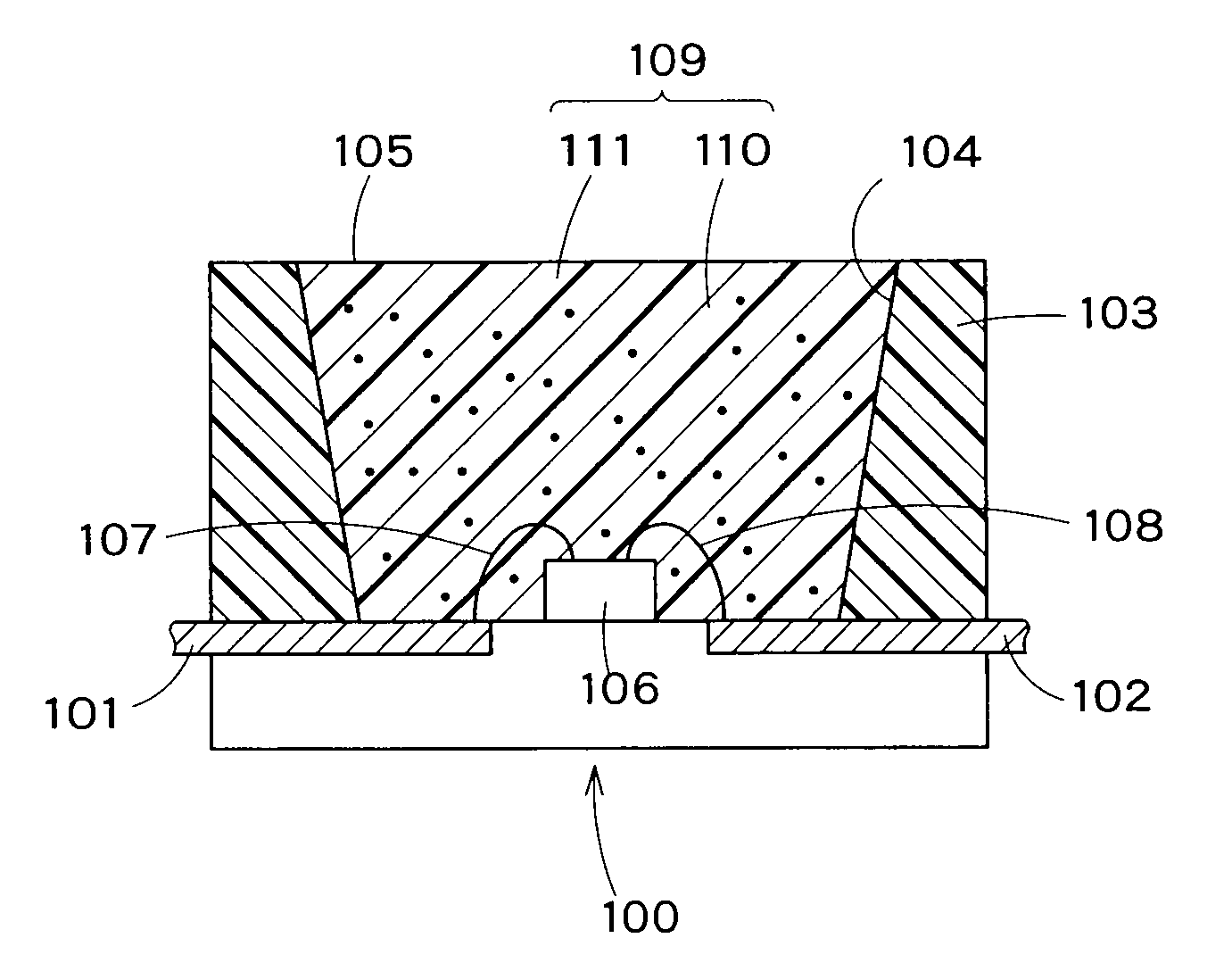
InactiveUS7990049B2Improve efficiencyLong lastingDischarge tube luminescnet screensLamp detailsWork functionInorganic compound
Owner:CANON KK
Ultraviolet water purification system
ActiveUS20090084734A1Inhibits ultraviolet disinfectionImprove efficiencySamplingExhaust apparatusWireless mesh networkClosed loop feedback
An Ultraviolet-C (UVC) based portable water purification system employing a novel array of baffles increases the efficiency per unit energy of irradiating UVC light in the eradication of pathogens in the water. Closed loop feedback allows monitoring the application of UVC light power to ensure high levels of pathogen eradication. This system is capable of eradicating a wide range of waterborne bacteria, viruses, protozoa, helminthes, yeast, and mold found in natural freshwater sources worldwide. By adding pre- or post-filters, the system can remove harmful organic compounds, pesticides, inorganic compounds and heavy metals from the water. The system can also be used to eradicate pathogens in fluids other than water. As a feature of this invention, a communications systems that can reach geographically dispersed populations at low cost without the need to install costly wired communications infrastructure is combined with and powered by the water purification system. In one embodiment, a packet radio system is provided to create nodes in a wireless mesh communications system to provide voice, data, video and internet communications using an array of the water purifiers to create a wireless mesh network.
Owner:WATER OF LIFE
Silicon composite particles, preparation thereof, and negative electrode material for non-aqueous electrolyte secondary cell
ActiveUS20050214644A1Improve cycle performanceMinimize changesSilicaNitrogen compoundsSilicon alloyInorganic compound
Silicon composite particles are prepared by sintering primary fine particles of silicon, silicon alloy or silicon oxide together with an organosilicon compound. Sintering of the organosilicon compound results in a silicon-base inorganic compound which serves as a binder. Each particle has the structure that silicon or silicon alloy fine particles are dispersed in the silicon-base inorganic compound binder, and voids are present within the particle.
Owner:SHIN ETSU CHEM IND CO LTD
Electrochemical methods, devices, and structures
ActiveUS20060102455A1Increase strainDeferred-action cellsCell seperators/membranes/diaphragms/spacersInorganic compoundElectrochemistry
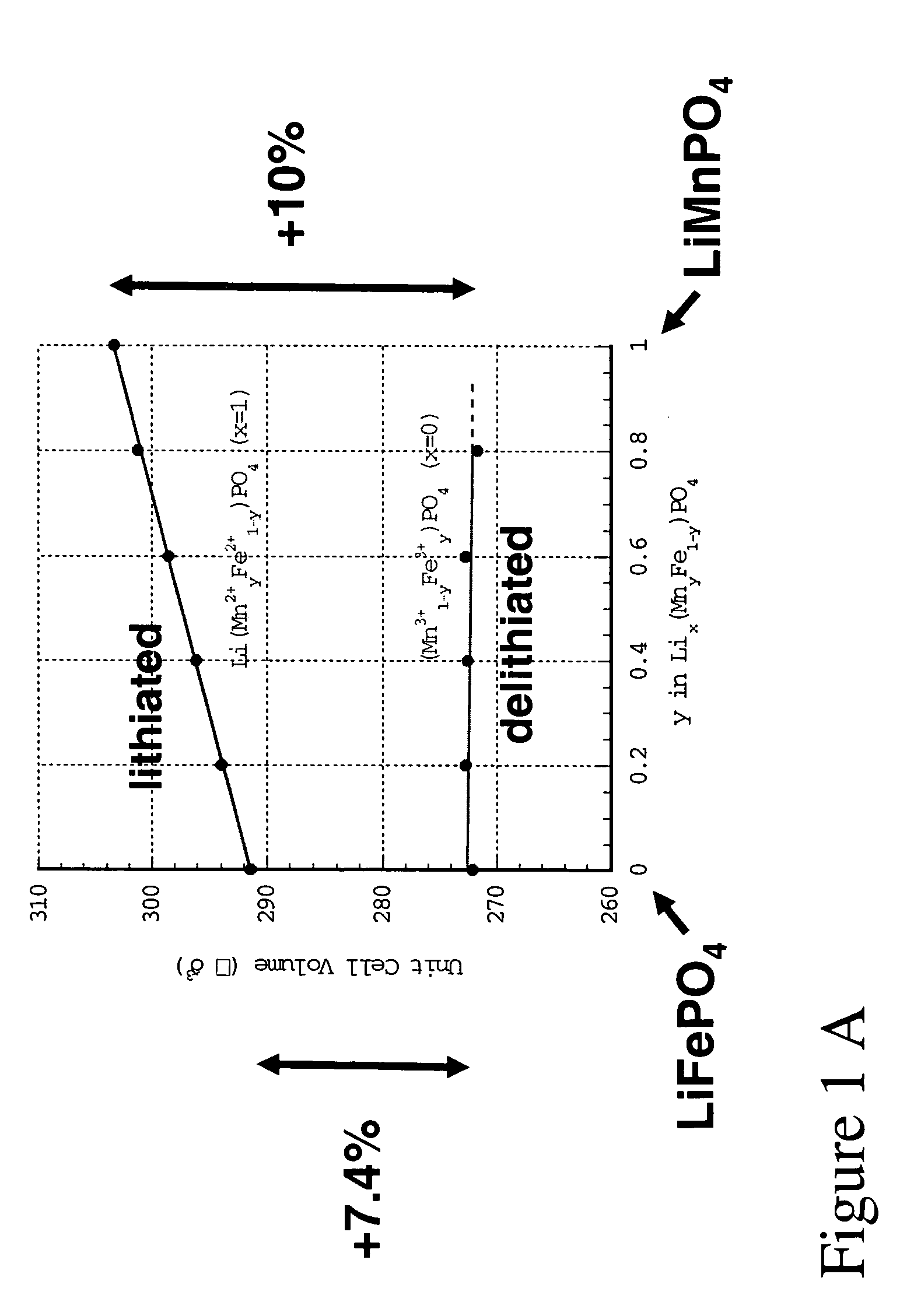
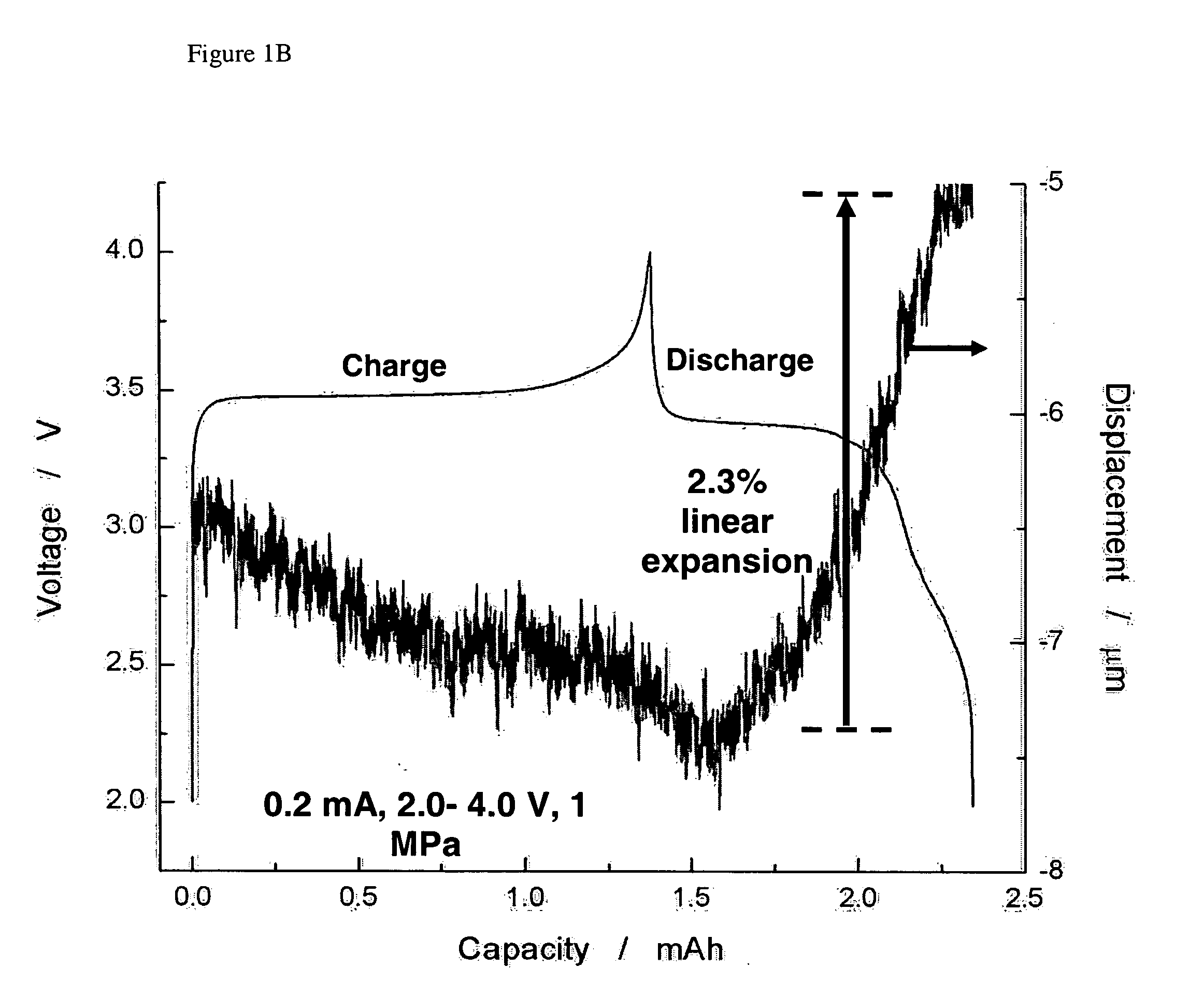
The present invention provides devices and structures and methods of use thereof in electrochemical actuation. This invention provides electrochemical actuators, which are based, inter-alia, on an electric field-driven intercalation or alloying of high-modulus inorganic compounds, which can produce large and reversible volume changes, providing high actuation energy density, high actuation authority and large free strain.
Owner:MASSACHUSETTS INST OF TECH
Barriers for polymer-coated implantable medical devices and methods for making the same
InactiveUS6953560B1Reduce and prevent and inflammationReduce and prevent proliferationStentsSurgeryHafniumPt element
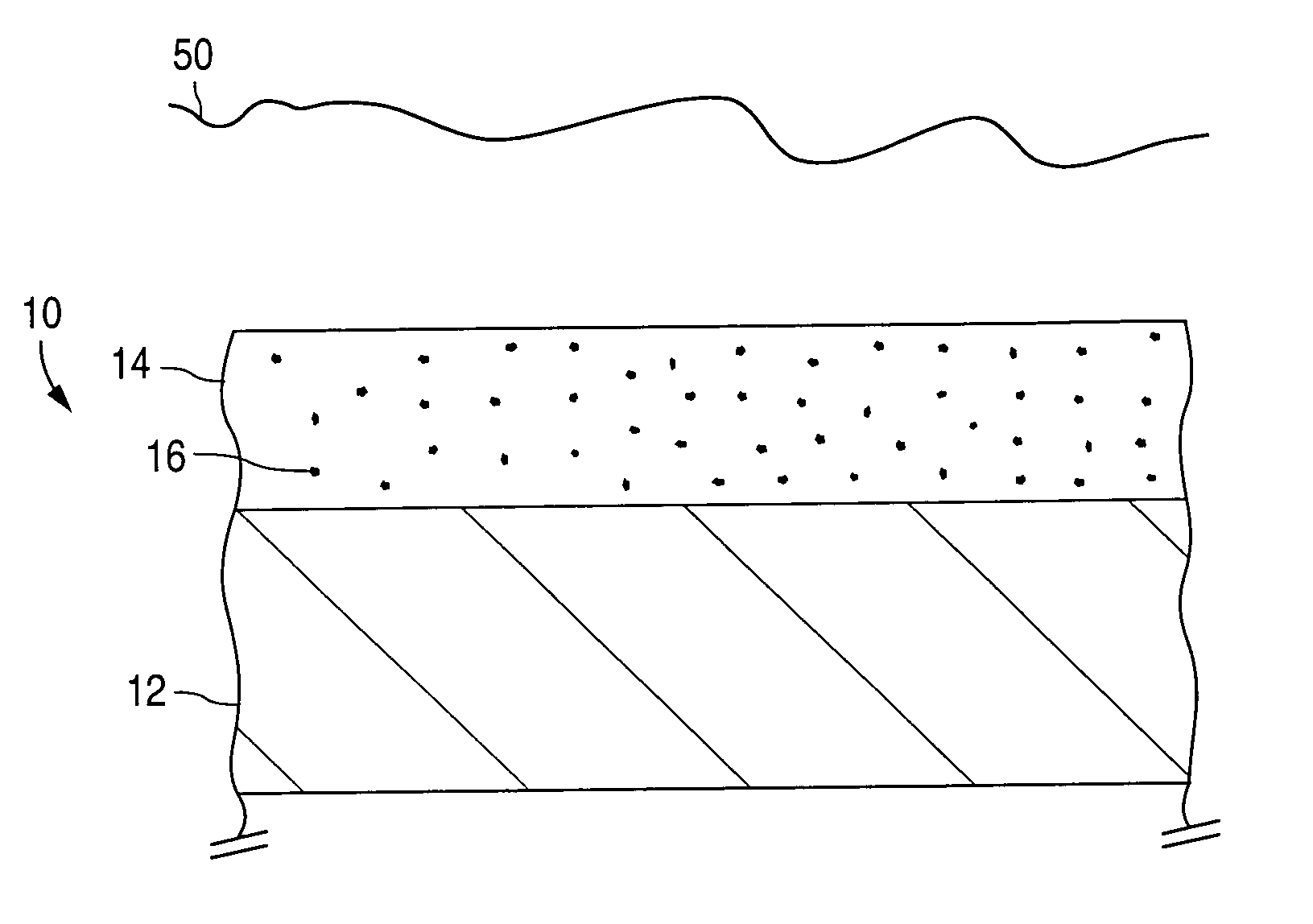
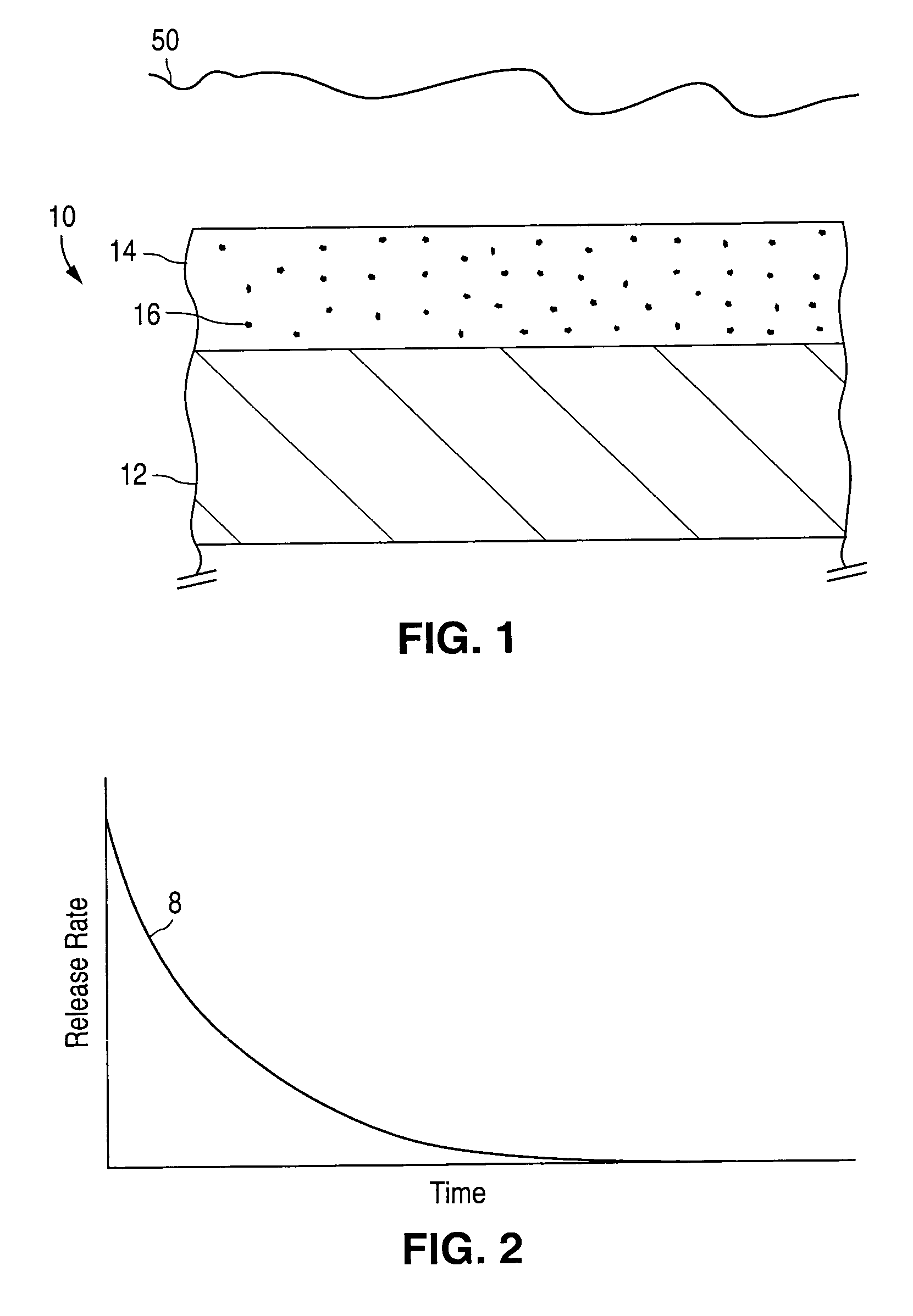
An implantable medical device and methods for making the implantable medical device are disclosed. The implantable medical device includes a substrate. At least a portion of the substrate is coated with a first layer including a polymer containing a drug. A barrier overlies the first layer. The barrier significantly reduces the rate of release of the drug from the polymer, thereby sustaining release of the drug from the medical device for a longer time.The barrier may be a homogeneous layer overlying the first layer, or a number of discrete deposits over the first layer. Alternatively, the barrier may be intermixed with an outer portion of the first layer. The barrier material is biocompatible, and typically has a thickness ranging from about 50 angstroms to about 20,000 microns. Suitable materials for the barrier include, but are not limited to, inorganic compounds, such as inorganic silicides, oxides, nitrides, carbides, as well as pure metals such as aluminum, chromium, gold, hafnium, iridium, niobium, palladium, platinum, tantalum, titanium, tungsten, zirconium, and alloys of these metals. The barriers disclosed may be applied to the first layer by several techniques, depending on the material being applied. Exemplary deposition techniques include physical vapor deposition, alkoxide hydrolysis, and electroless plating.The implantable device may be a stent or a graft, among other possibilities.
Owner:ABBOTT CARDIOVASCULAR
Red fluorescent substance and light-emitting device employing the same
InactiveUS20100102707A1Reduce colorSufficient changeDischarge tube luminescnet screensLamp detailsFluorescenceInorganic compound
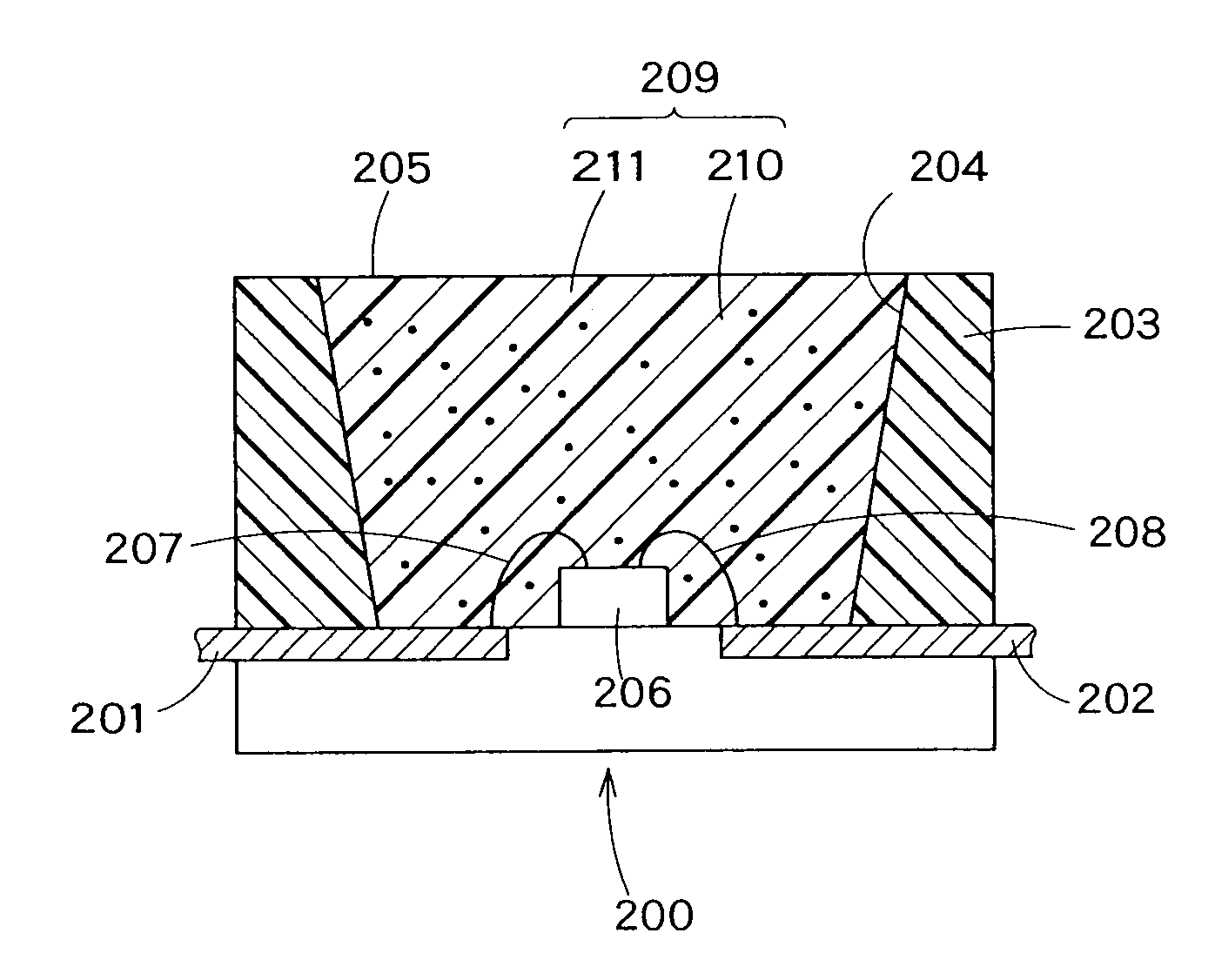
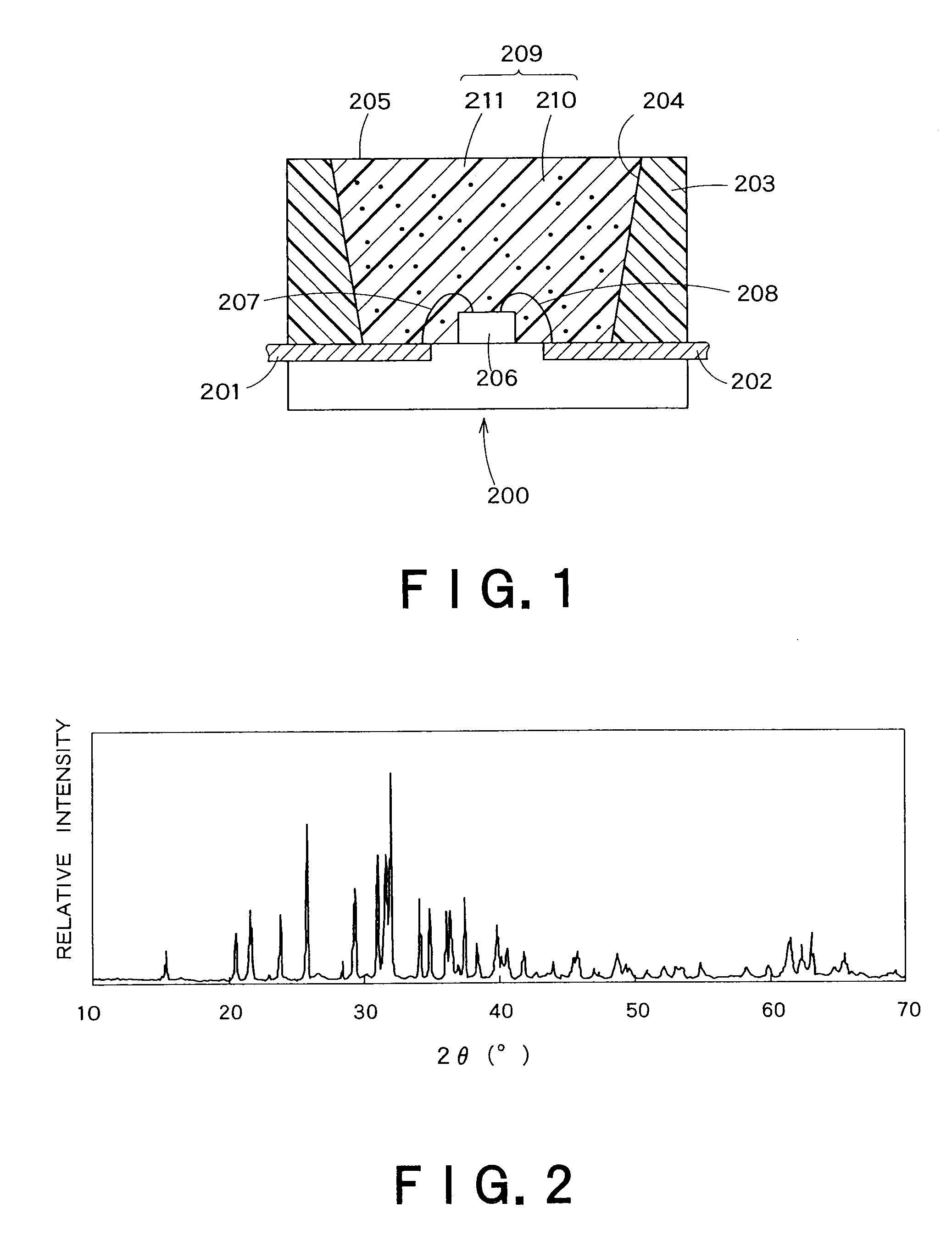
The present invention provides a red fluorescent substance and a light-emitting device utilizing the substance. This device less suffers from color drift even when working with high power. The light-emitting device comprises a blue LED, a green fluorescent substance of Sr3Al3Si13O2N21 activated with a luminescent center, and the red fluorescent substance. The red fluorescent substance contains an inorganic compound comprising a metal element M, a trivalent element M1 other than the M, a tetravalent element M2 other than the M, and nitrogen element. In the inorganic compound, the M is partly replaced with a luminescent center element EC. The red fluorescent substance has basically the same crystal structure as Sr2Al3Si7ON13. The red fluorescent substance emits luminescence having a peak in the range of 580 to 650 nm when excited with light of 250 to 500 nm.
Owner:KK TOSHIBA
Method for measuring substance and testing piece
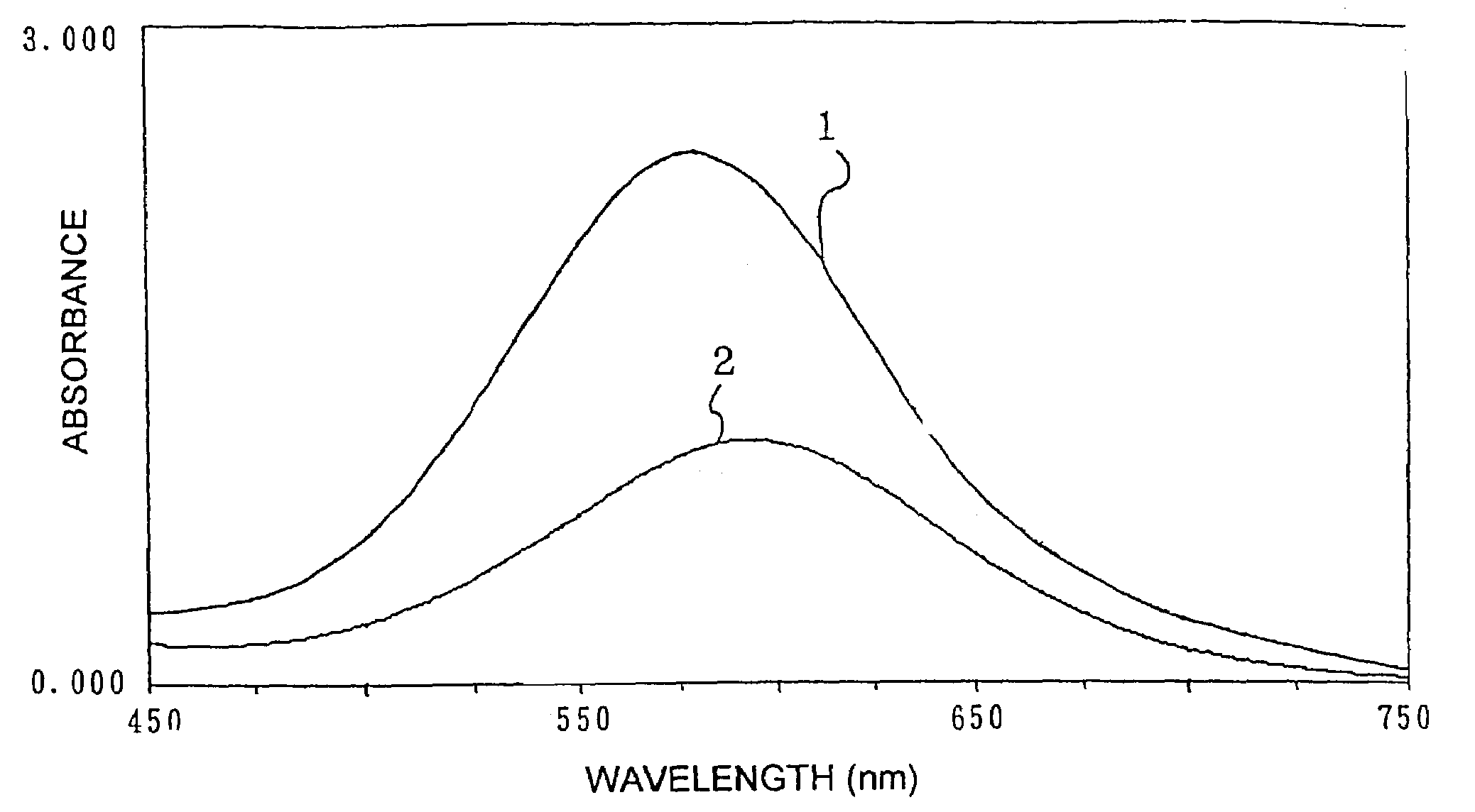
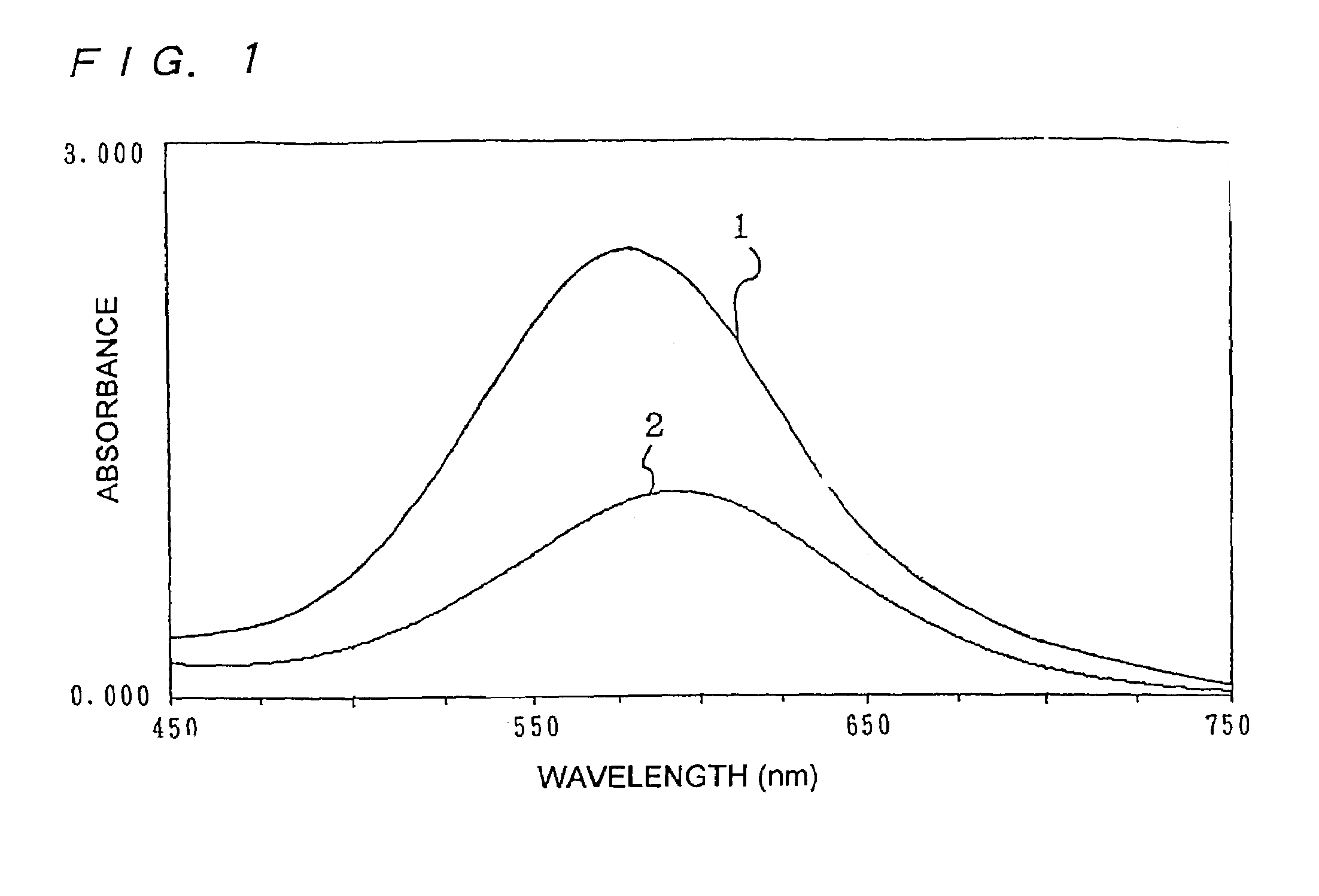
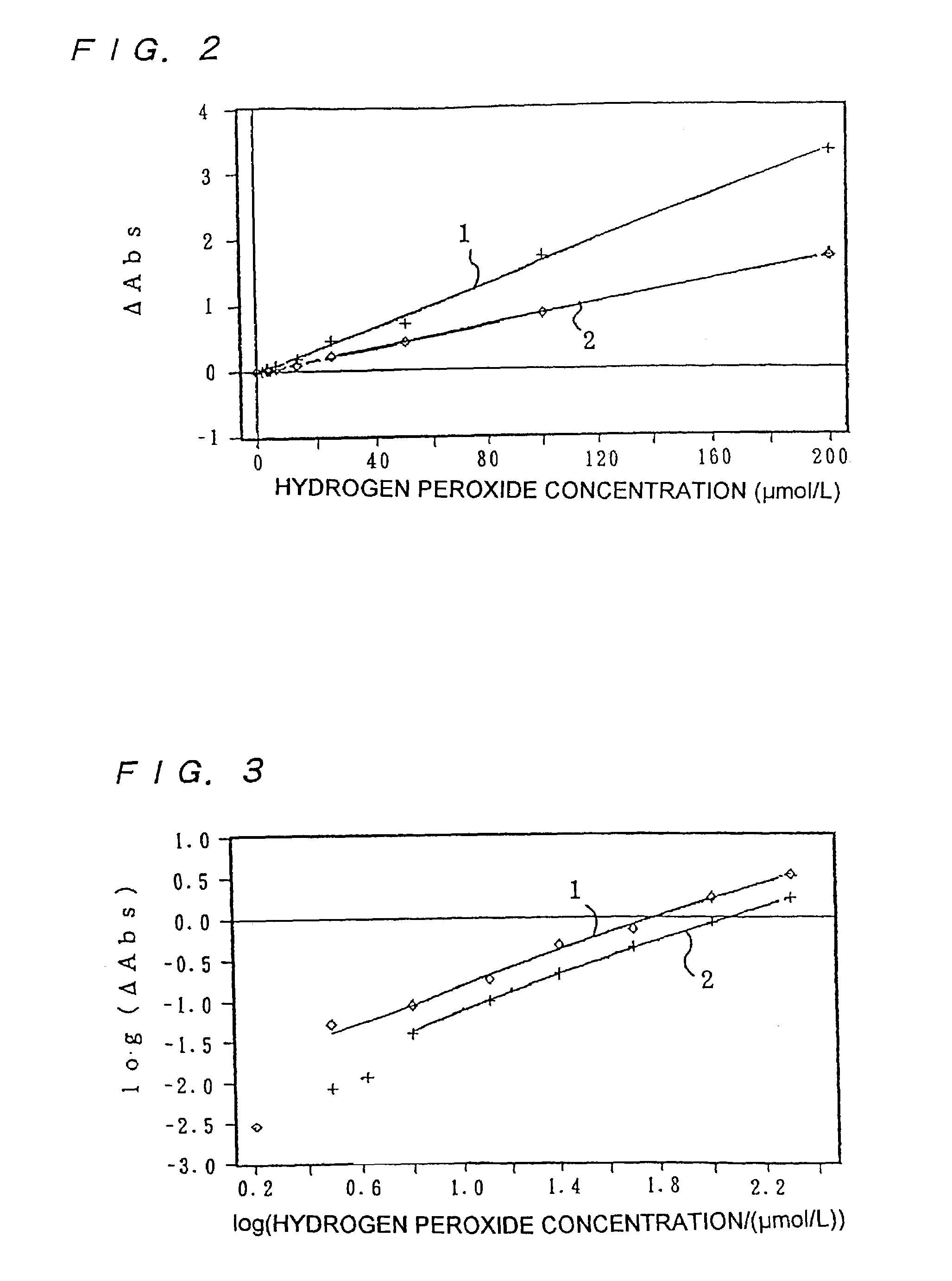
InactiveUS7153696B2High measurement accuracyHigh measurement sensitivityAnalysis using chemical indicatorsMaterial analysis by observing effect on chemical indicatorDiffusionChemical reaction
A method of measuring an analyte, comprising a step of measuring a detectable substance by using a reaction system including a formation reaction of the detectable substance based on a chemical reaction of the analyte contained in a sample, wherein a layered inorganic compound is caused to exist in the reaction system including the formation reaction of the detectable substance, whereby high-sensitivity measurement is made possible, the detectable substance can be stabilized to improve accuracy of the measurement, a rate of a chemical reaction is increased to enable quick measurement, and high-sensitivity measurement is made possible even in a reaction system which forms an insoluble substance. Also, it can be provided an analytical testing piece for measuring an analyte, by measuring a detectable substance by using a reaction system including a formation reaction of the detectable substance based on a chemical reaction of the analyte contained in a sample, wherein the testing piece comprises at least one test portion having a detection portion for detecting the detectable substance and contains a layered inorganic compound at least in the test portion, whereby diffusion and elution of a dyestuff or the like is prevented, more sensitive and accurate simple analysis is made possible, and easy handling is possible.
Owner:ARKRAY INC
Mxene nanosheet and manufacturing method thereof
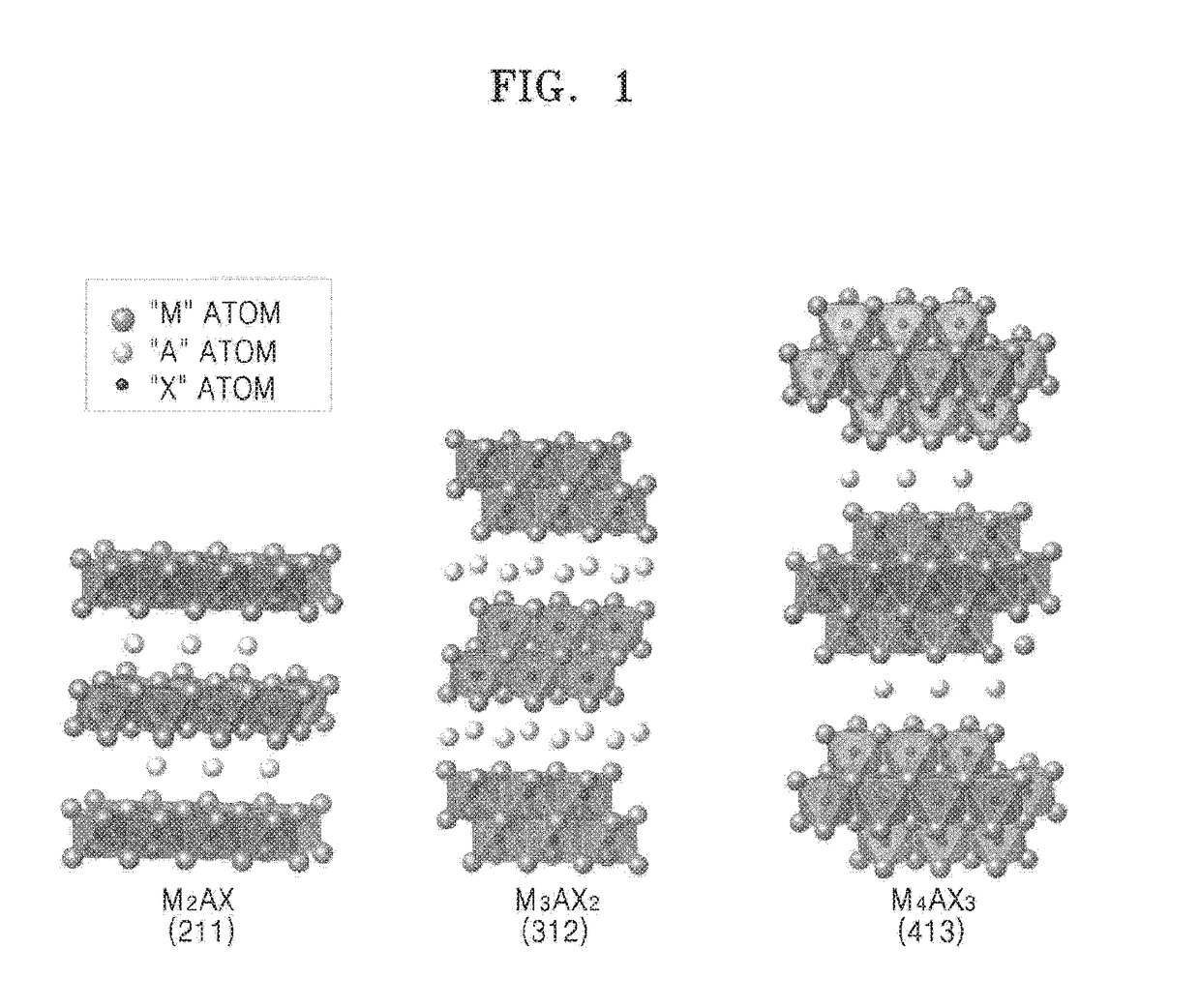
ActiveUS20170088429A1Material nanotechnologyNitrogen-metal/silicon/boron binary compoundsIodideInorganic compound
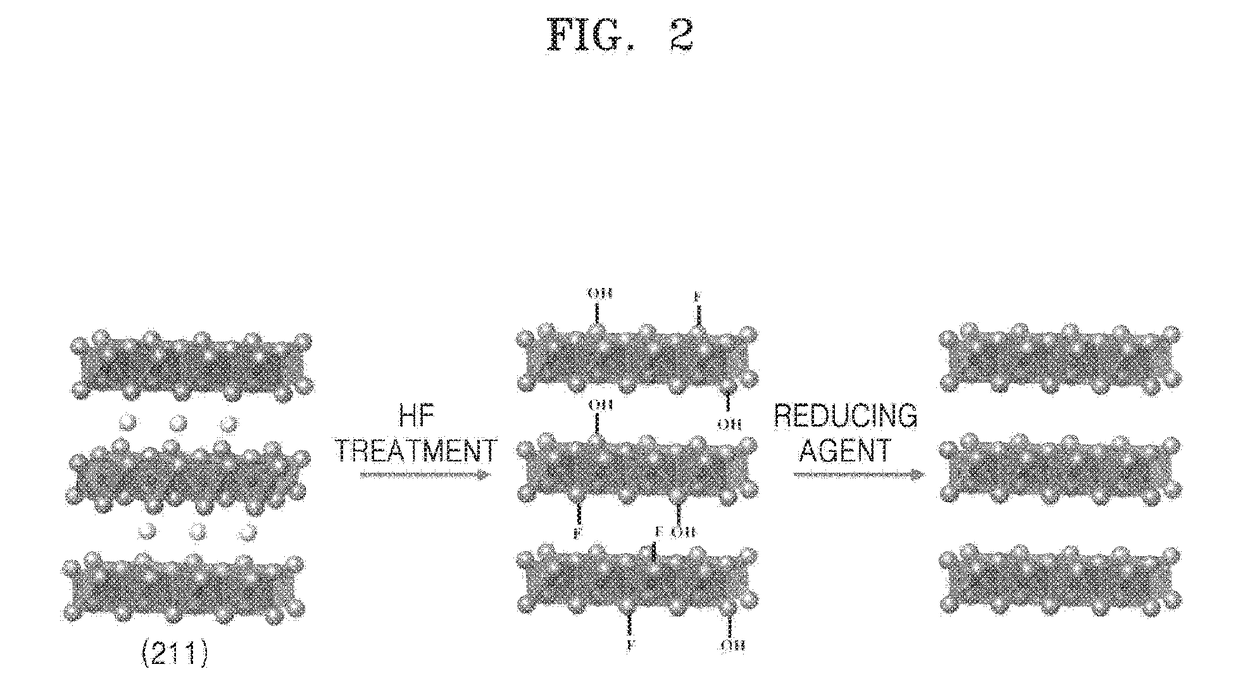
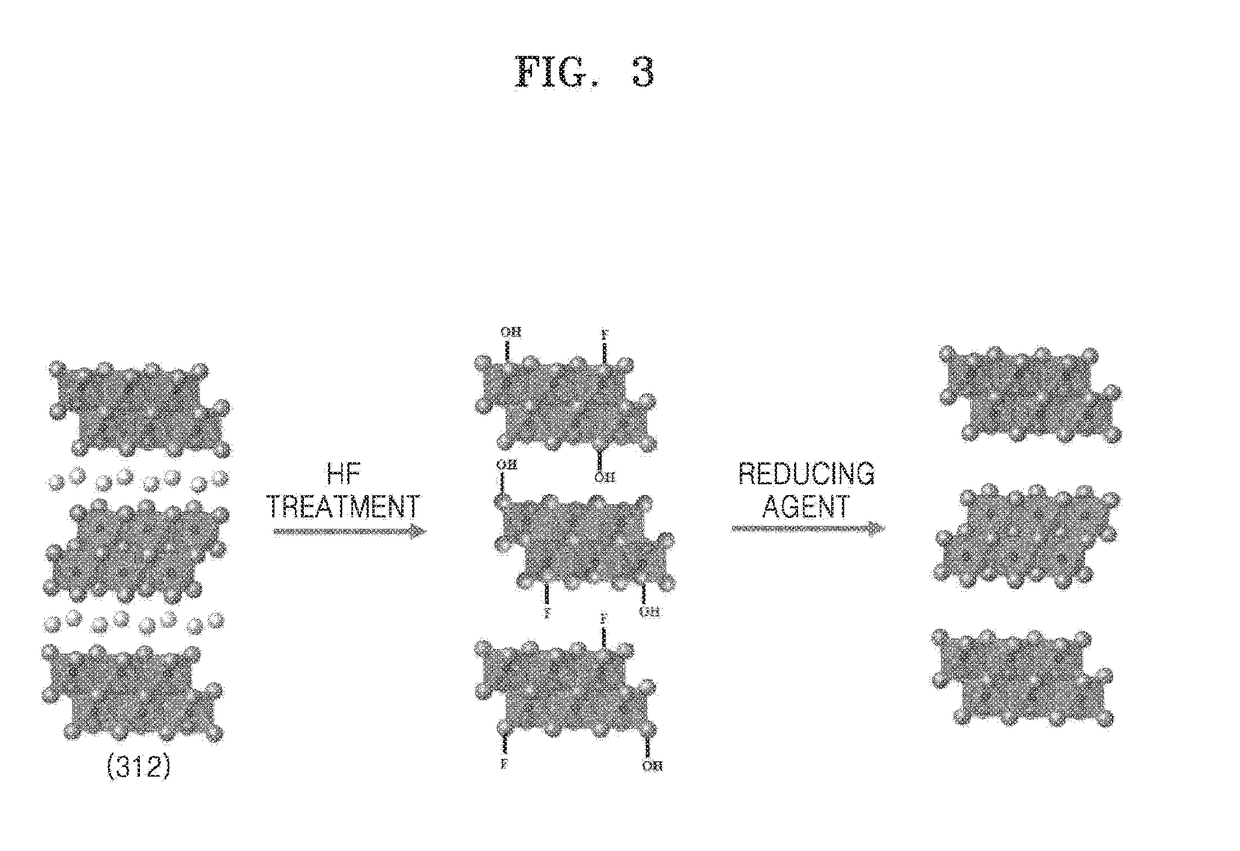
A method of manufacturing a MXene nanosheet includes removing an A atomic layer from an inorganic compound having a formula of Mn+1AXn to form a nanosheet, the nanosheet having a formula of Mn+1XnTs, and reducing the nanosheet having a formula of Mn+1XnTsto form an MXene nanosheet, the MXene nanosheet having a formula of Mn+1Xn, wherein M is at least one of Group 3 transition metal, Group 4 transition metal, Group 5 transition metal, and Group 6 transition metal, A is at least one of a Group 12 element, Group 13 element, Group 14 element, Group 15 element and Group 16 element, X is one of carbon (C), nitrogen (N) and a combination thereof, Ts is one of oxide (O), epoxide, hydroxide (OH), alkoxide having 1-5 carbon atoms, fluoride (F), chloride (Cl), bromide (Br), iodide (I), and a combination thereof, and n is one of 1, 2 and 3.
Owner:RES & BUSINESS FOUND SUNGKYUNKWAN UNIV +1
Organic electroluminescence device and method for fabricating the same
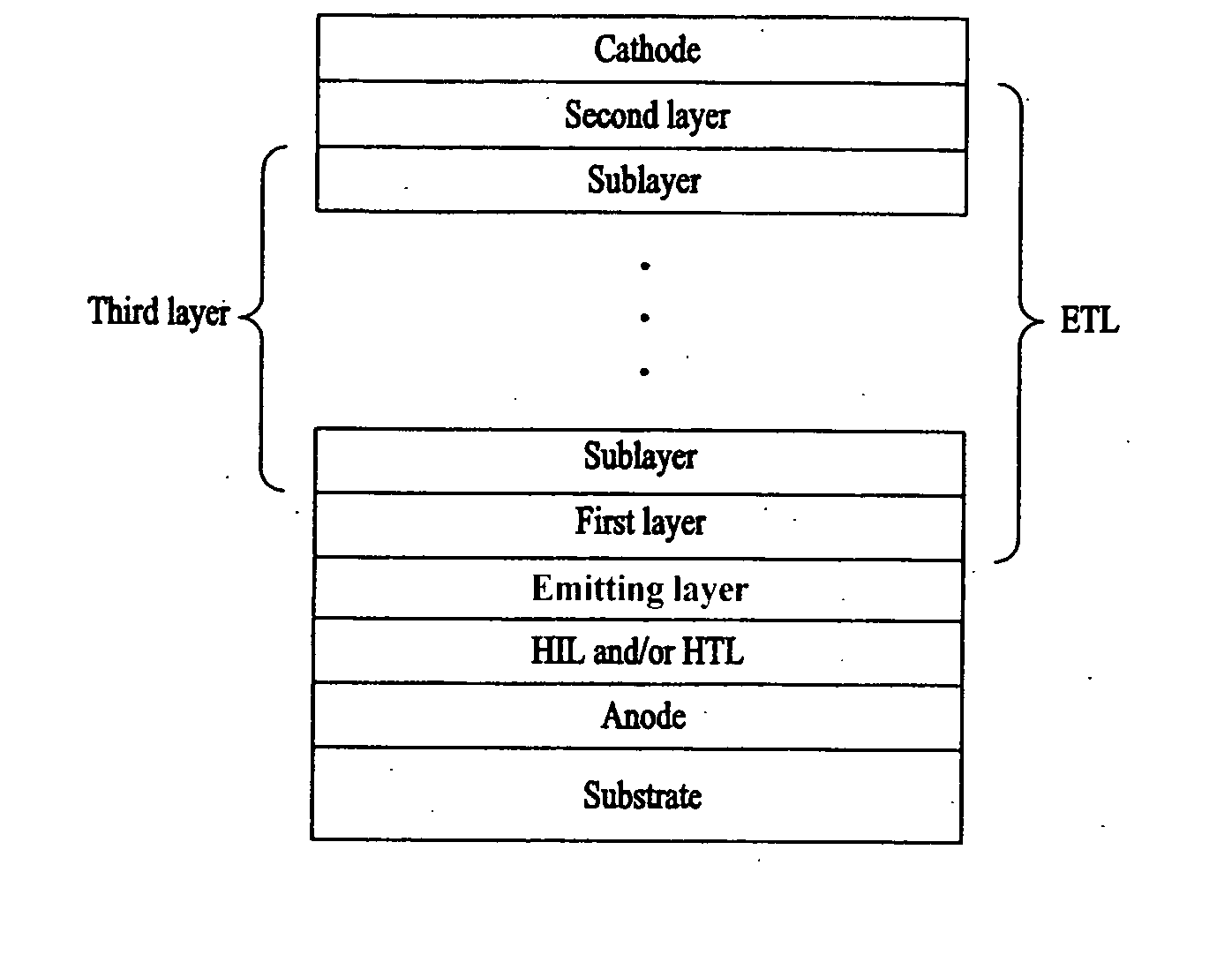
ActiveUS20070020483A1Improve efficiencyProlong lifeDischarge tube luminescnet screensElectroluminescent light sourcesSimple Organic CompoundsInorganic compound
An organic electroluminescent (EL) device having improved efficiency and service life is provided. The organic electroluminescent device has a stack structure including an emitting layer and an electron-transport layer positioned between an anode and a cathode. The electron-transport layer includes a first layer adjacent to the emitting layer which may be a mixture of at least two materials, and a second layer adjacent to the cathode which may be a mixture of at least two materials. The mixture of at least two materials may be a mixture of an organic compound and one or more other organic compounds, or may be a mixture of a metal or inorganic compound and one or more other metal or inorganic compounds, or may be a mixture of one or more organic compounds and one or more metal or inorganic compounds.
Owner:LG DISPLAY CO LTD
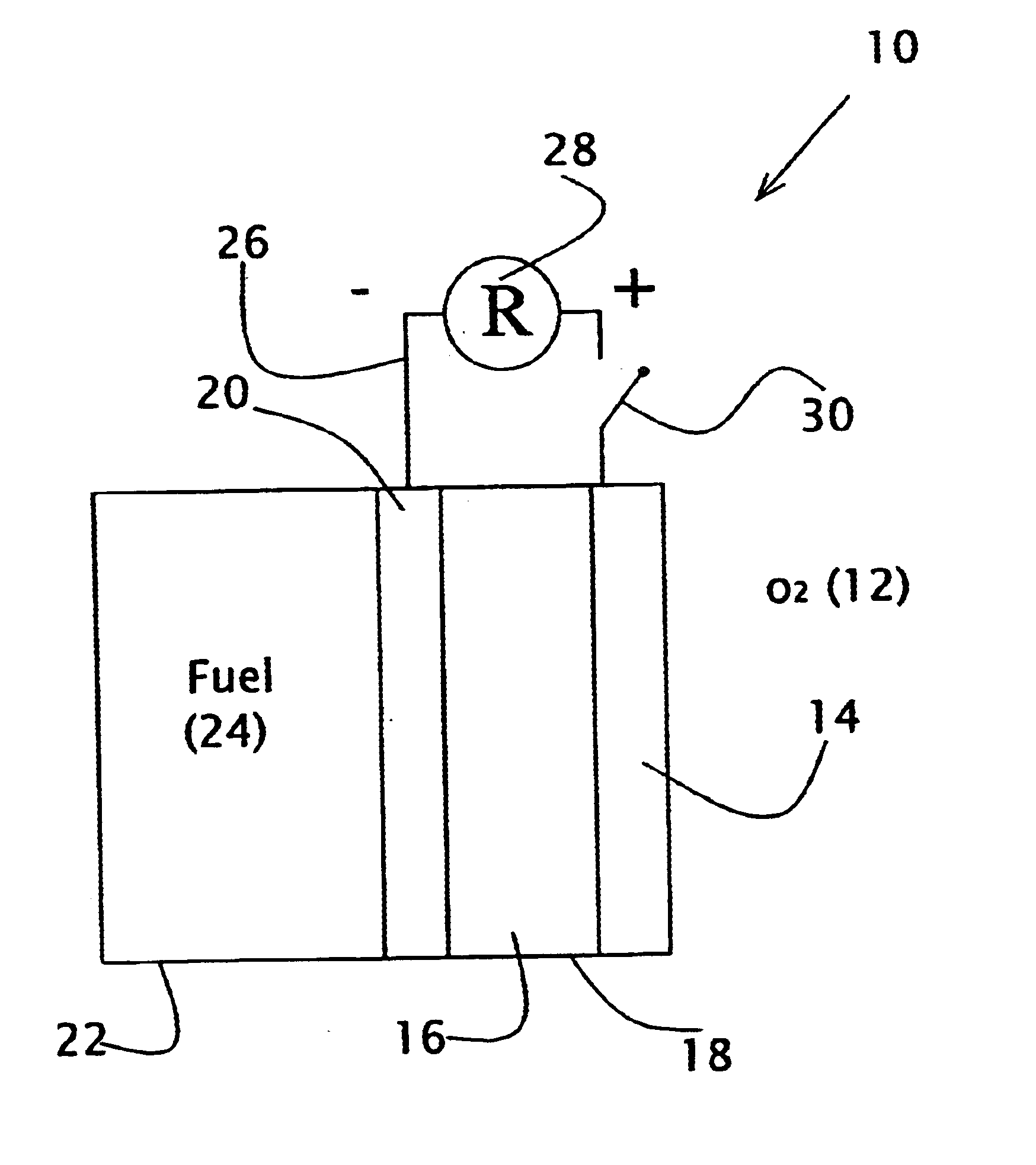
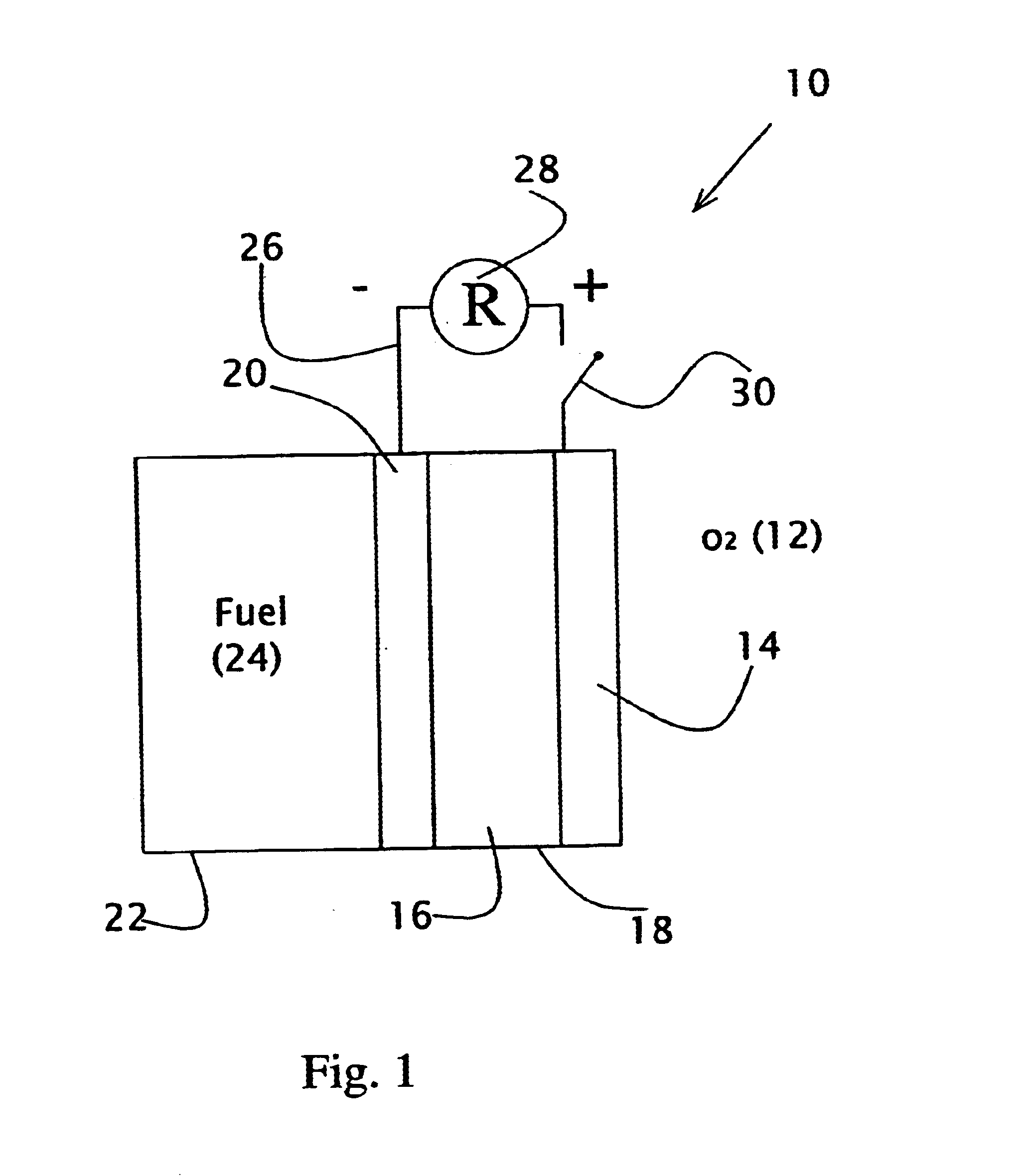
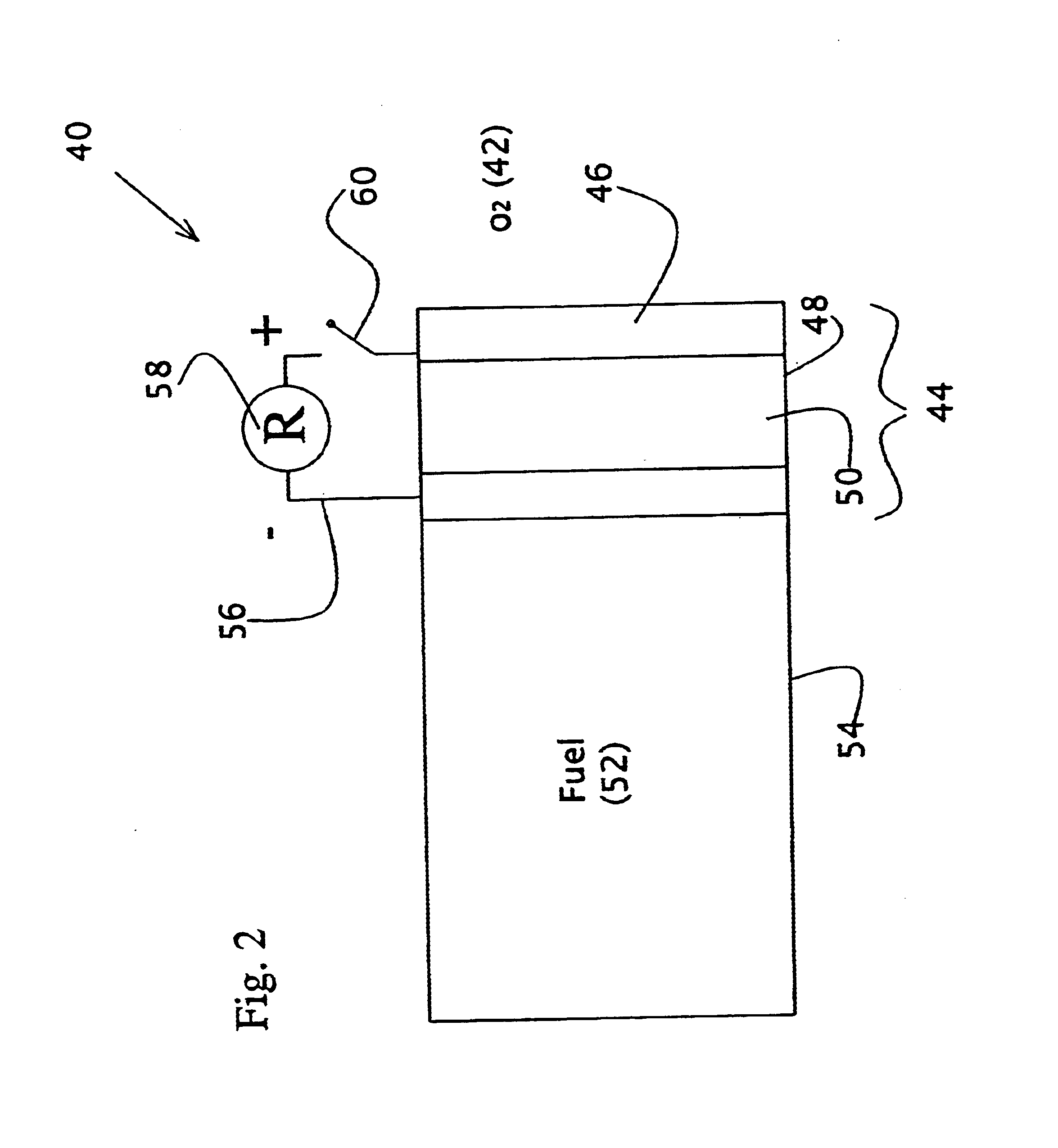
Liquid fuel compositions for electrochemical fuel cells
A new fuel composition useful for catalytic fuel cells is made up of at least two components. The primary fuel component is a surface active compound, such as methanol, that is a source of and acts to prevent unwanted decomposition of the auxiliary fuel. The auxiliary fuel is a hydrogen-containing inorganic compound with a high reduction potential, such as NaBH4, which acts as a highly reactive source of energy and serves to catalyze the catalytic oxidation of the primary fuel.
Owner:MORE ENERGY
Golf balls comprising glass ionomers, ormocers, or other hybrid organic/inorganic compositions
A golf ball comprising a core and a cover layer, wherein at least one of the core or cover layer comprises a hybrid material that contains fatty acid-modified glass ionomers, an ormocer or other inorganic-organic materials.
Owner:ACUSHNET CO
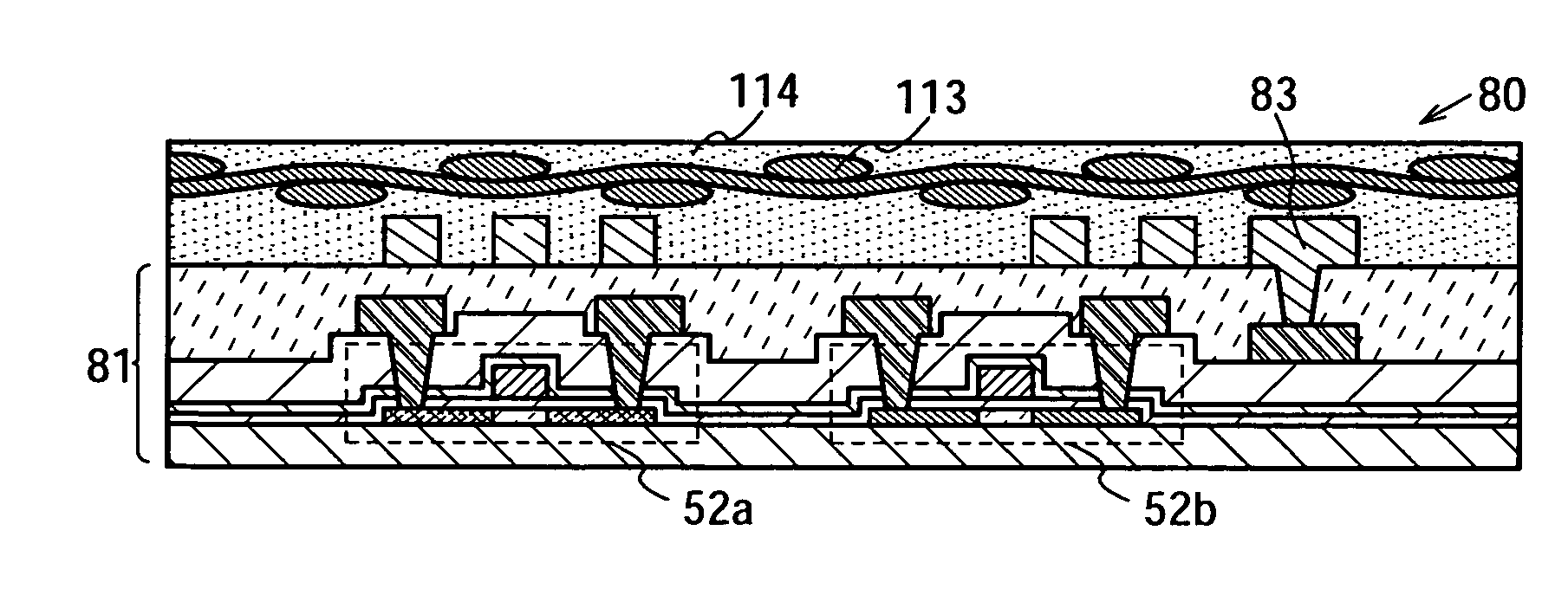
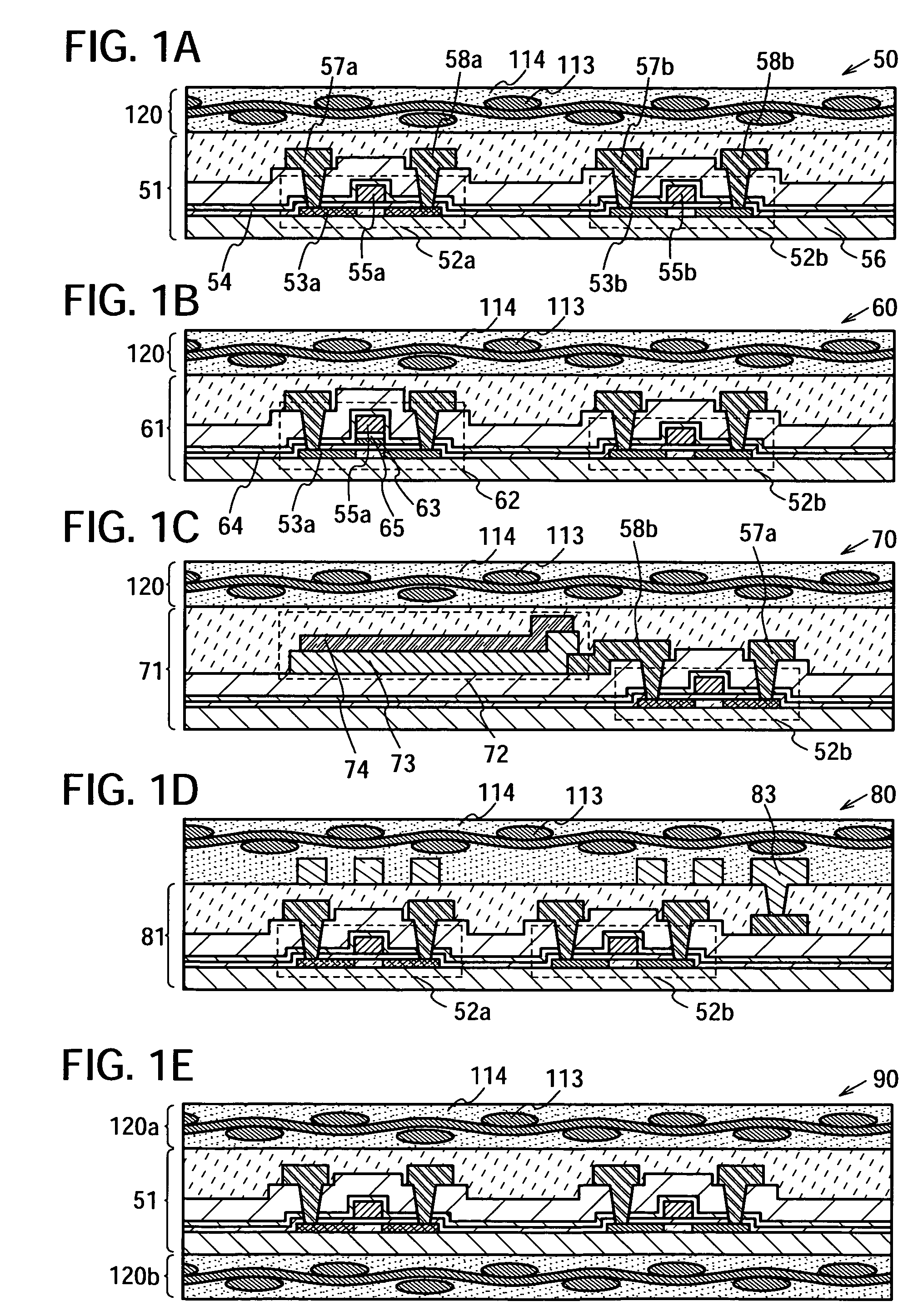
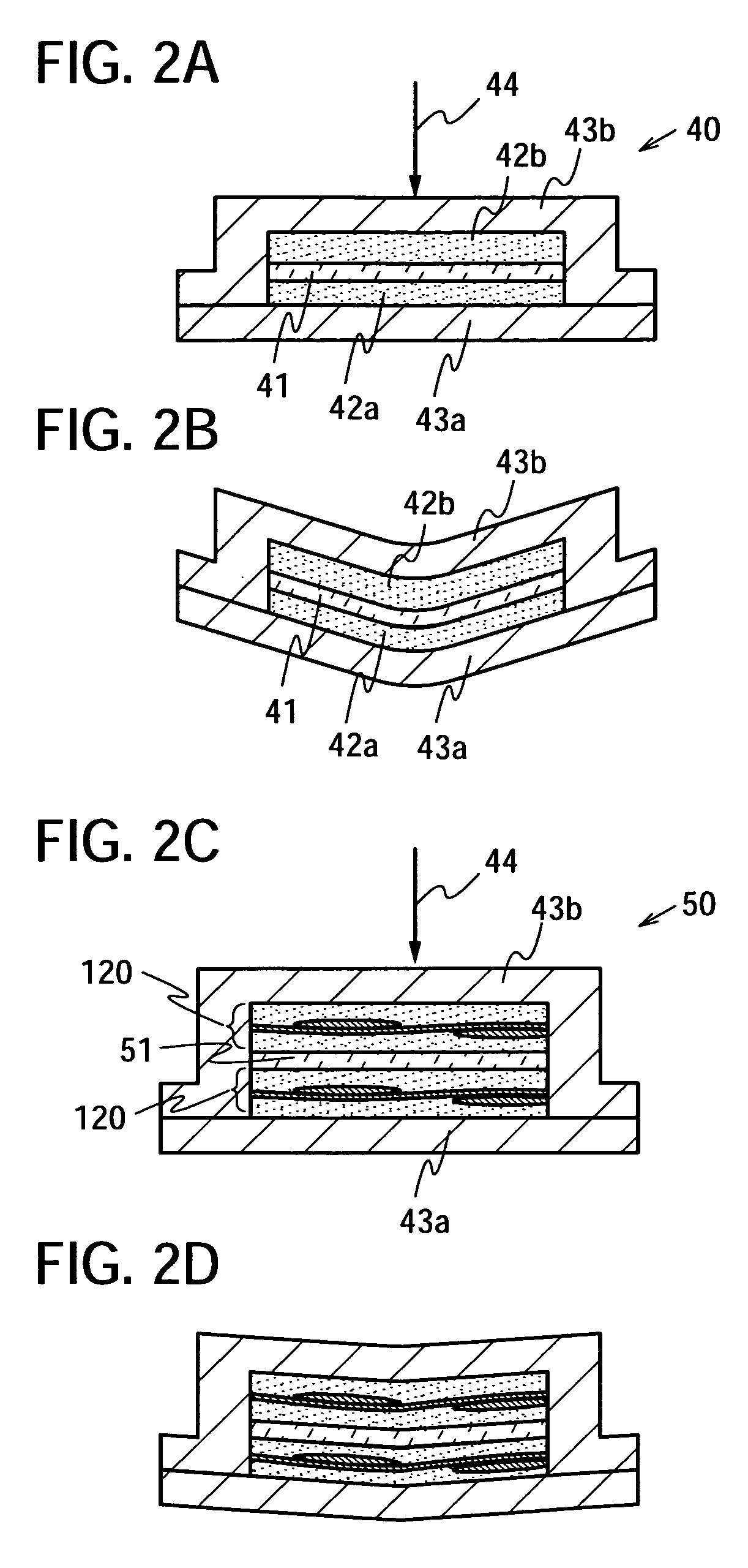
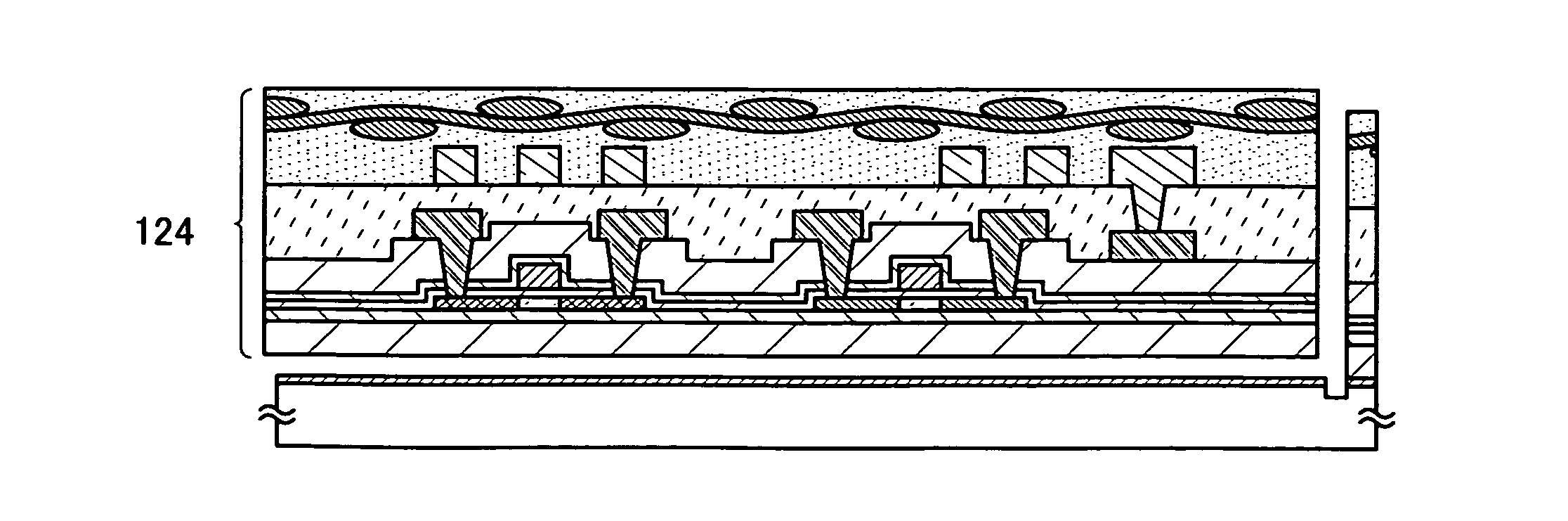
Semiconductor device and manufacturing method thereof
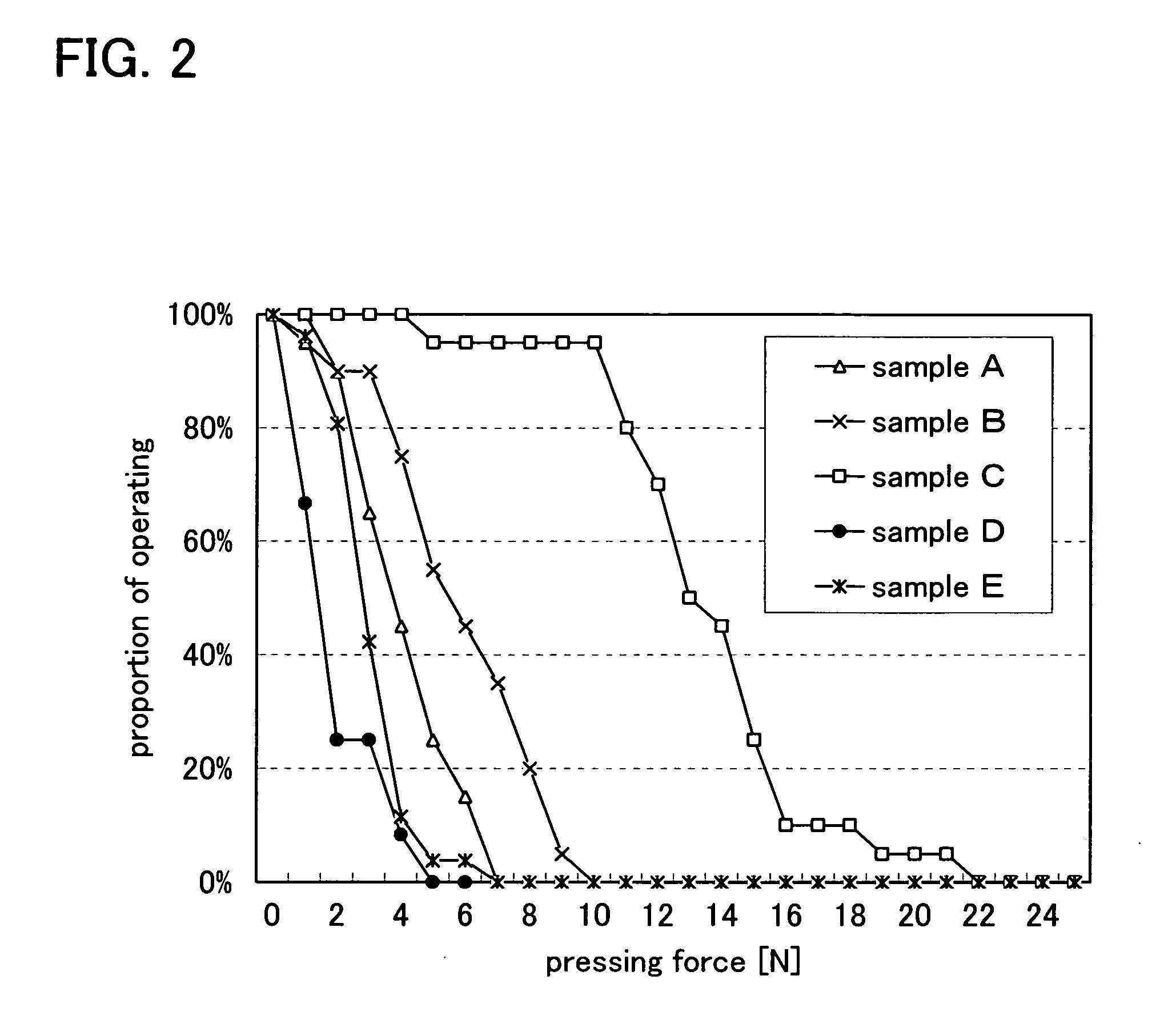
InactiveUS20080224941A1Avoid partialAvoid destructionSemiconductor/solid-state device detailsSolid-state devicesFiberInorganic compound
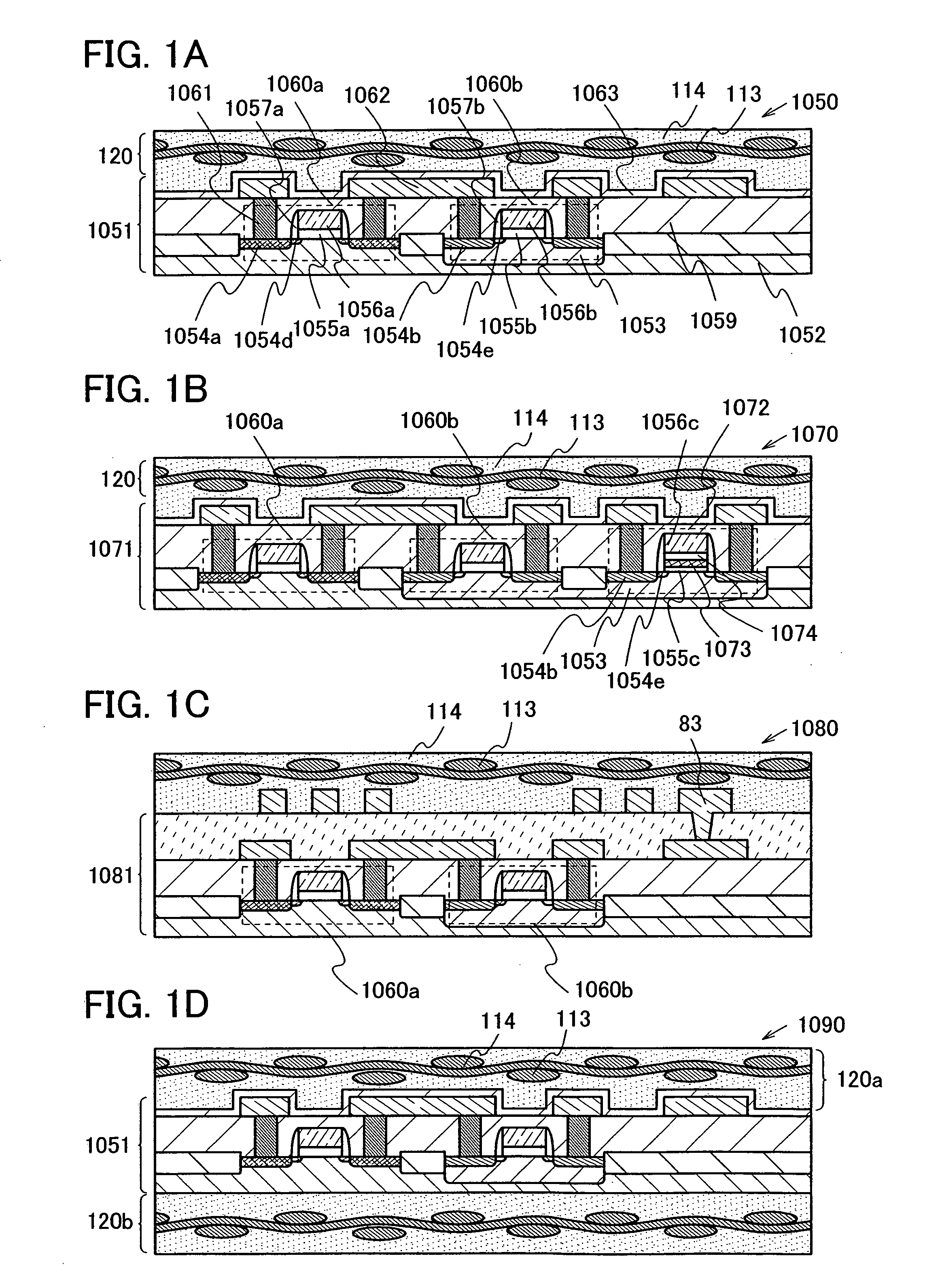
The present invention provides a semiconductor device which is not easily damaged by external local pressure. The present invention further provides a method for manufacturing a highly-reliable semiconductor device, which is not destructed by external local pressure, with a high yield. A structure body, in which high-strength fiber of an organic compound or an inorganic compound is impregnated with an organic resin, is provided over an element layer having a semiconductor element formed using a non-single crystal semiconductor layer, and heating and pressure bonding are performed, whereby a semiconductor device is manufactured, to which the element layer and the structure body in which the high-strength fiber of an organic compound or an inorganic compound is impregnated with the organic resin are firmly fixed together.
Owner:SEMICON ENERGY LAB CO LTD
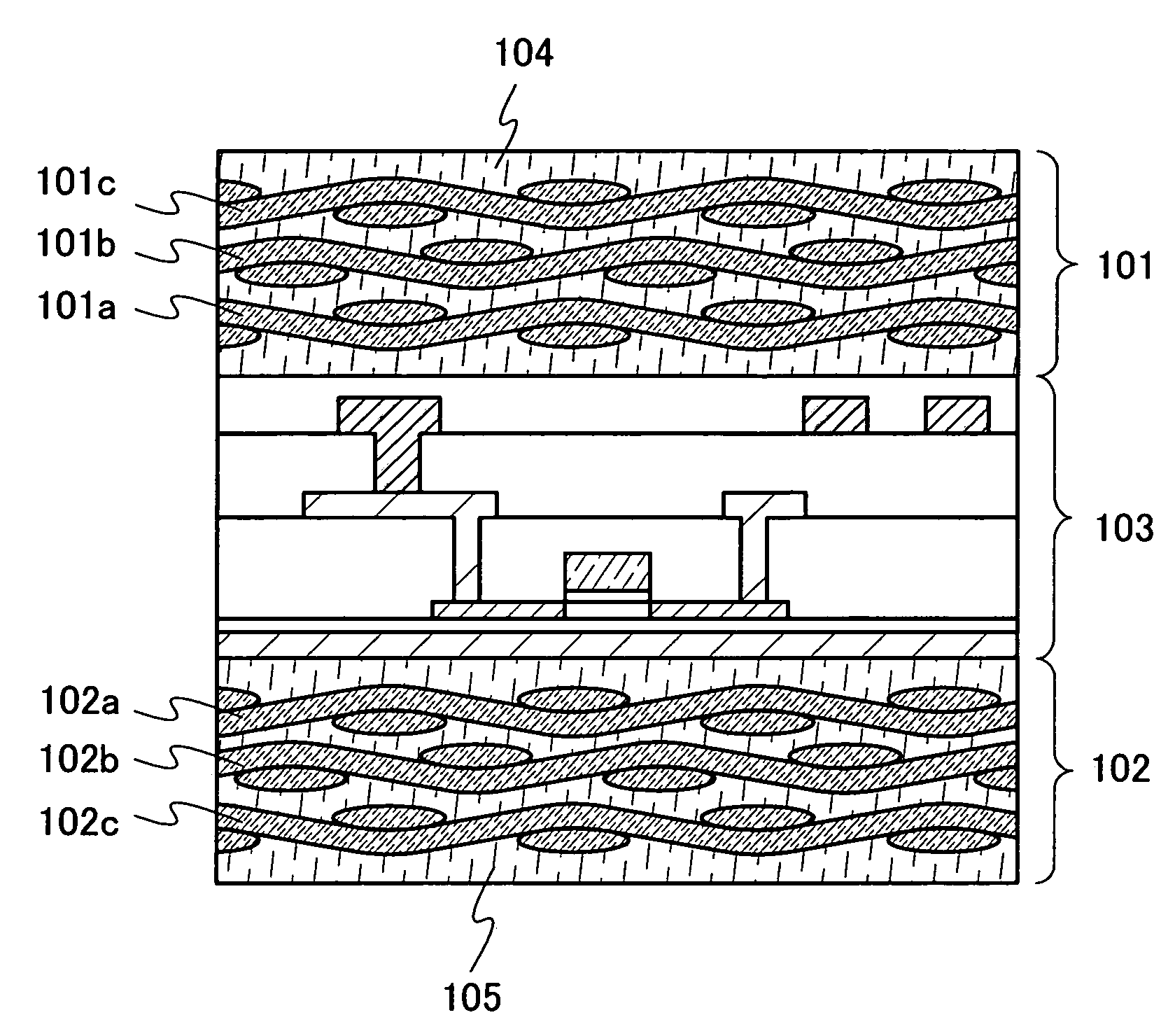
Semiconductor device
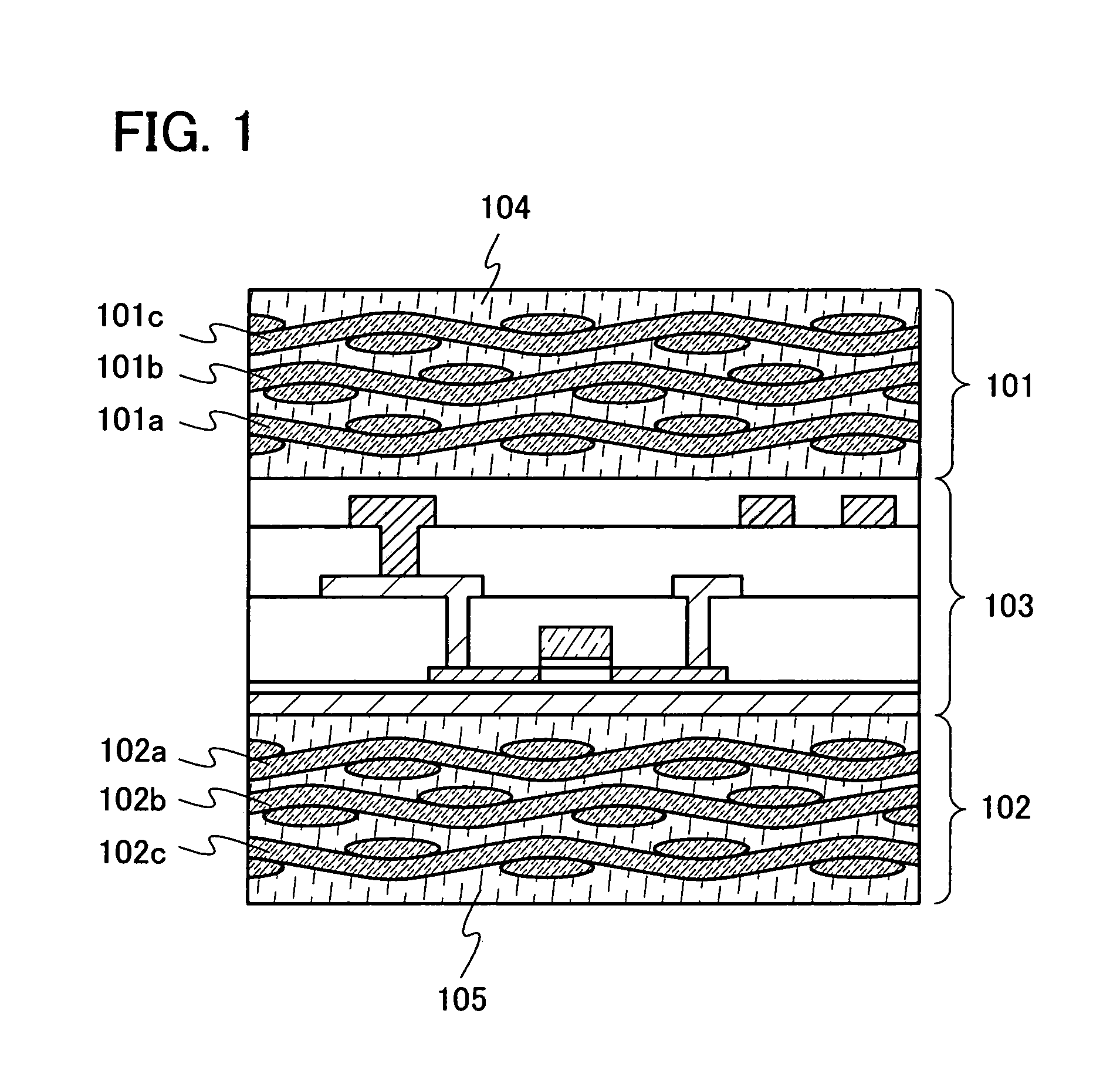
InactiveUS20080303140A1Improve reliabilityHigh modulusSemiconductor/solid-state device detailsSolid-state devicesInorganic compoundFibrous body
To provide a semiconductor device which can increase reliability with respect to external force, especially pressing force, while the circuit size or the capacity of memory is maintained. A pair of structure bodies each having a stack of fibrous bodies of an organic compound or an inorganic compound, which includes a plurality of layers, especially three or more layers, is impregnated with an organic resin, and an element layer provided between the pair of structure bodies are included. The element layer and the structure body can be fixed to each other by heating and pressure bonding. Further, a layer for fixing the element layer and the structure body may be provided. Alternatively, the structure body fixed to an element layer can be formed in such a way that after a plurality of fibrous bodies is stacked over the element layer, the fibrous bodies are impregnated with an organic resin.
Owner:SEMICON ENERGY LAB CO LTD
Method for preparing cross-linked ceramic-coated separator containing ionic polymer, ceramic-coated separator prepared by the method, and lithium secondary battery using the same
ActiveUS20110027658A1High bonding strengthImprove battery performanceLi-accumulatorsCell component detailsCross-linkHigh rate
The present invention provides method for preparing a cross-linked ceramic-coated separator containing an ionic polymer, a ceramic-coated separator prepared by the method, and a lithium secondary battery using the same. According to preferred methods for preparing a cross-linked ceramic-coated separator, a coating material containing ceramic particles for improving thermal and mechanical characteristics, a functional inorganic compound for improving cycle characteristics and high rate characteristics of a battery, and an ionic polymer for bonding the ceramic particles and the functional inorganic compound on a porous membrane substrate is coated on the porous membrane substrate and subjected to chemical cross-linking.
Owner:HYUNDAI MOTOR CO LTD +1
Microfluidic apparatus and method for synthesis of molecular imaging probes
InactiveUS20050232387A1Fast synthesis timeHigh synthetic yieldIn-vivo radioactive preparationsConversion outside reactor/acceleratorsMicroreactorMolecular imaging
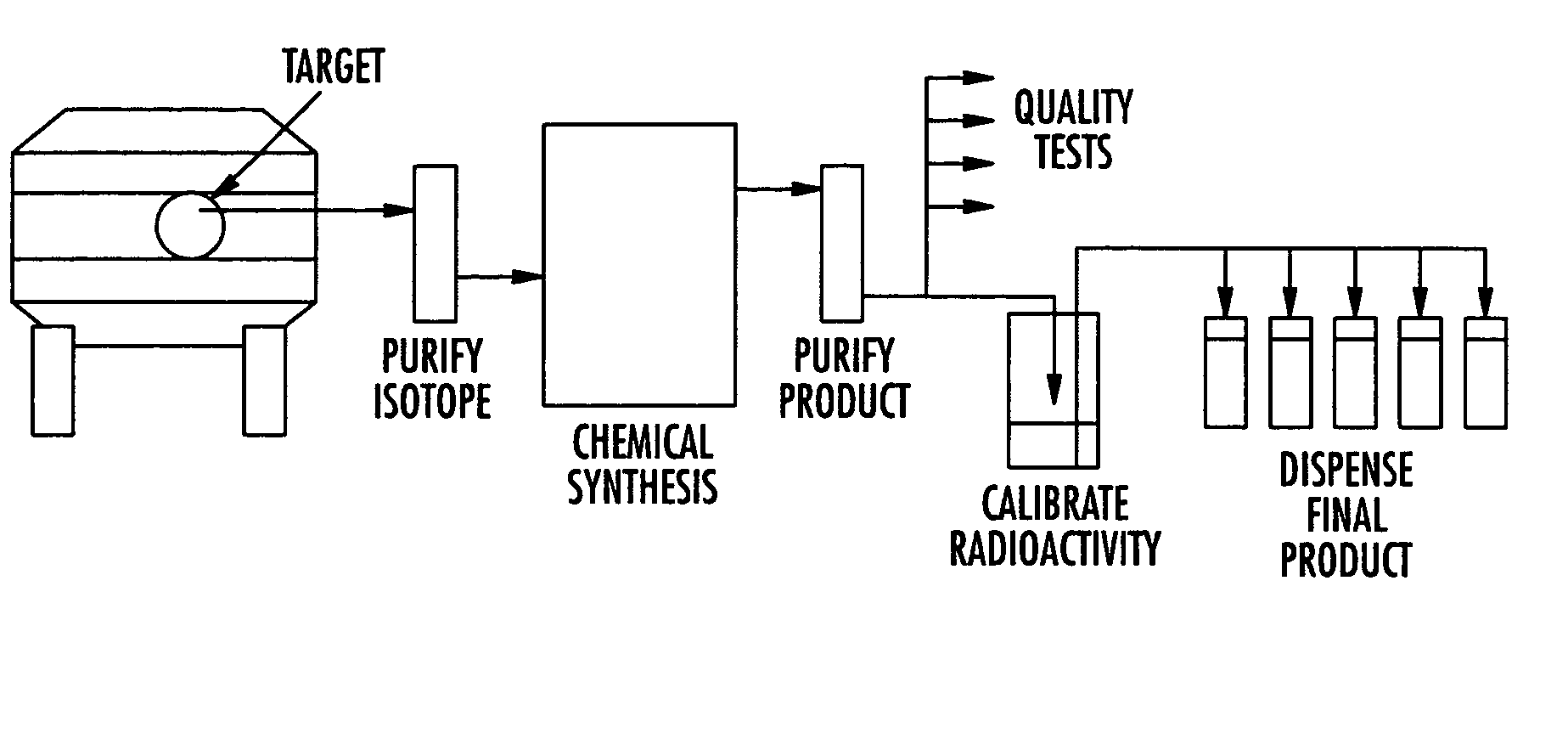
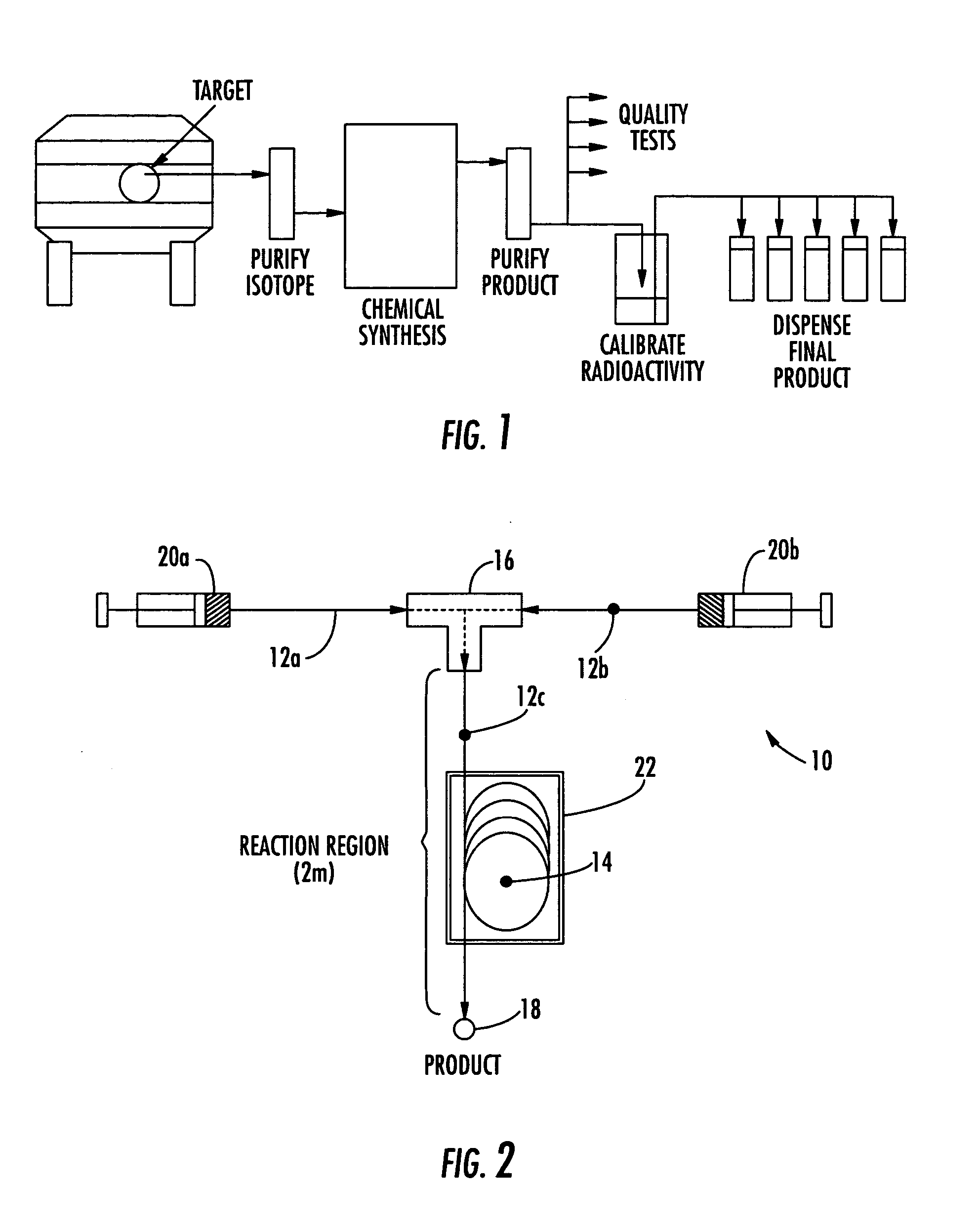
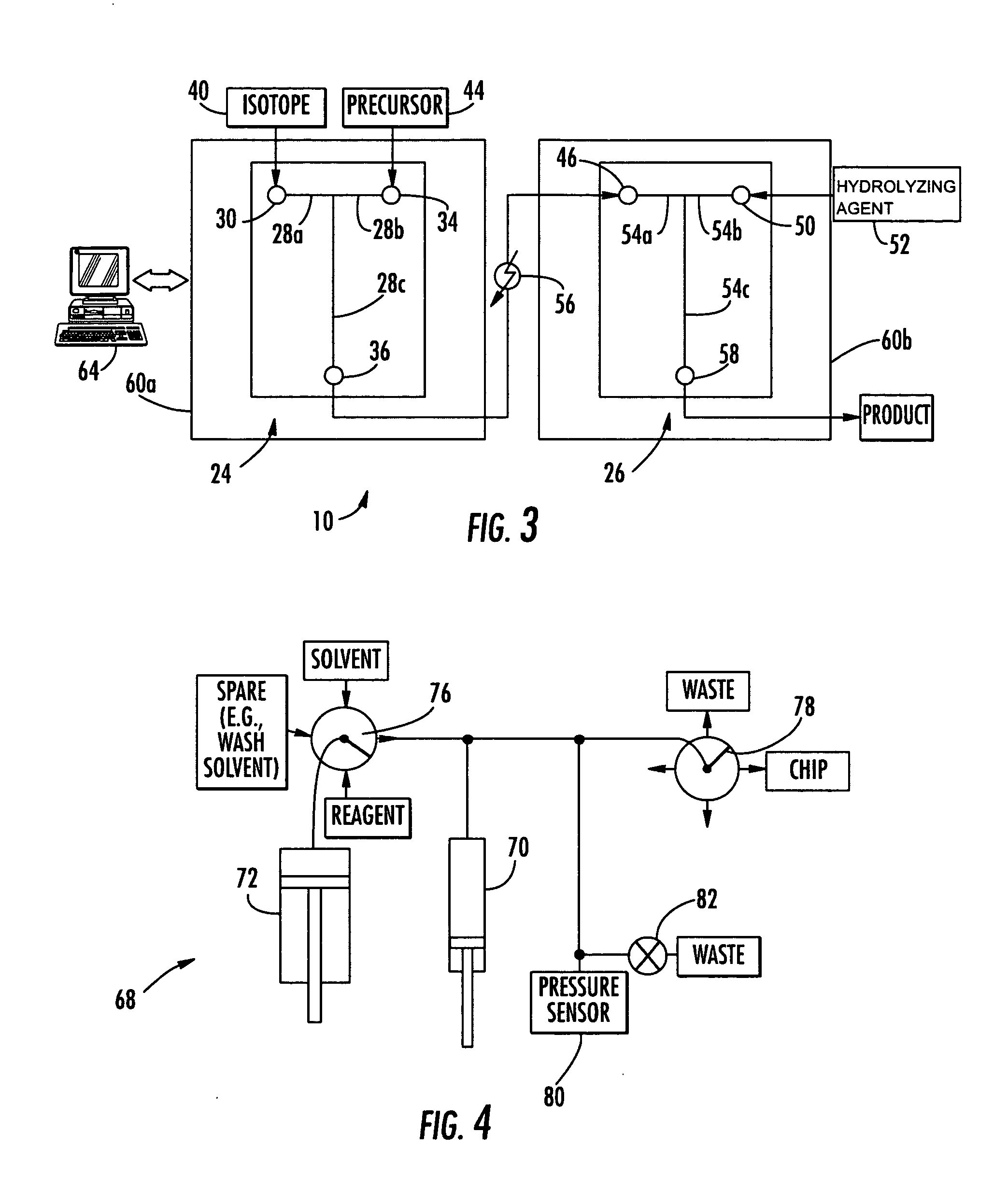
The invention provides a method and apparatus for preparation of radiochemicals, such as PET molecular imaging probes, wherein the reaction step or steps that couple the radioactive isotope to an organic or inorganic compound to form a positron-emitting molecular imaging probe are performed in a microfluidic environment. The method for synthesizing a radiochemical in a microfluidic environment comprises: i) providing a micro reactor comprising a first inlet port, a second inlet port, an outlet port, and at least one microchannel in fluid communication with the first and second inlet ports and the outlet port; ii) introducing a reactive precursor into the first inlet port of the micro reactor, the reactive precursor adapted for reaction with a radioactive isotope to form a radiochemical; iii) introducing a solution comprising a radioactive isotope into the second inlet port of the micro reactor; iv) contacting the reactive precursor with the isotope-containing solution in the microchannel of the micro reactor; v) reacting the reactive precursor with the isotope-containing solution as the reactive precursor and isotope-containing solution flow through the microchannel of the micro reactor, the reacting step resulting in formation of a radiochemical; and vi) collecting the radiochemical from the outlet port of the micro reactor.
Owner:MOLECULAR TECH
Semiconductor device and manufacturing method thereof
InactiveUS20080224940A1Avoid partialAvoid destructionSemiconductor/solid-state device detailsSolid-state devicesFiberInorganic compound
The present invention provides a semiconductor device which is not easily damaged by external local pressure. The present invention further provides a manufacturing method of a highly-reliable semiconductor device, which is not destroyed by external local pressure, with a high yield. A structure body, in which high-strength fiber of an organic compound or an inorganic compound is impregnated with an organic resin, is provided over an element substrate having a semiconductor element formed using a single crystal semiconductor region, and heating and pressure bonding are performed, whereby a semiconductor device is manufactured, to which the element substrate and the structure body in which the high-strength fiber of an organic compound or an inorganic compound is impregnated with the organic resin are fixed together.
Owner:SEMICON ENERGY LAB CO LTD
Fluorescent substance and light-emitting device employing the same
ActiveUS20100025632A1Improve quantum efficiencyHigh emission intensitySolid-state devicesLuminescent compositionsQuantum efficiencyFluorescence
The present invention provides a fluorescent substance excellent both in quantum efficiency and in temperature characteristics, and also provides a light-emitting device utilizing the fluorescent substance. This fluorescent substance contains an inorganic compound comprising a metal element M, a trivalent element M1 other than the metal element M, a tetravalent element M2 other than the metal element M, and either or both of O and N. In the inorganic compound, the metal element M is partly replaced with a luminescence center element R. The crystal structure of the fluorescent substance is basically the same as Sr3Al3Si13O2N21, but the chemical bond lengths of M1-N and M2-N are within the range of ±15% based on those of Al—N and Si—N calculated from the lattice constants and atomic coordinates of Sr3Al3Si13O2N21, respectively. The fluorescent substance emits luminescence having a peak in the range of 490 to 580 nm when excited with light of 250 to 500 nm.
Owner:KK TOSHIBA
Inorganic-polymer complexes for the controlled release of compounds including medicinals
InactiveUS6391336B1Control releasePowder deliveryPeptide/protein ingredientsControlled releaseActive agent
This invention relates generally to the production and use of inorganic-polymer complexes for the controlled release of compounds including medicinals. The inorganic compound used is advantageously calcium sulfate-hemihydrate. The invention includes a composition for the controlled release of an active agent comprising: a) a hydrated or crystallized inorganic compound, and b) a matrix polymer which slows the release of the active agent, wherein the composition is a solid matrix due to the hydration or crystallization of the inorganic compound. Further included is a composition for the controlled release of an active agent comprising: a) a hydrated or crystallized inorganic compound, and b) a complexing agent which forms a salt or conjugate with the active agent, wherein the composition is a solid matrix due to the hydration or crystallization of the inorganic compound.
Owner:ROYER BIOMEDICAL INC
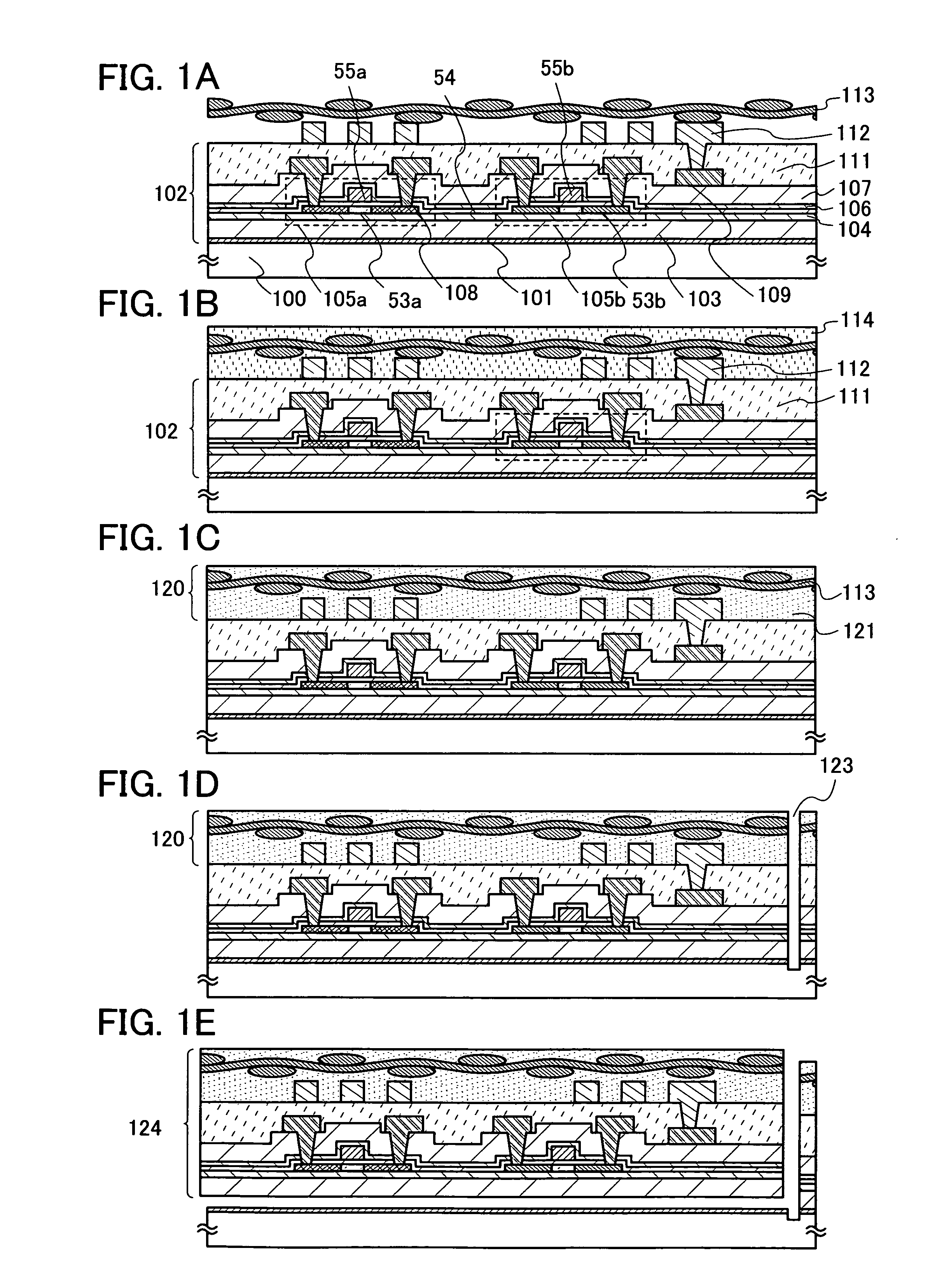
Display device and method for manufacturing the same
InactiveUS20060091397A1Improve reliabilityHigh definitionElectroluminescent light sourcesSolid-state devicesSimple Organic CompoundsDisplay device
It is an object of the invention to manufacture a highly reliable display device at a low cost with high yield. A display device of the invention includes: a first reflective electrode layer; and a second transparent electrode layer with an electroluminescent layer interposed therebetween, wherein the electroluminescent layer has a layer containing an organic compound and an inorganic compound, and the first electrode layer contains an aluminum alloy containing at least one or more selected from the group consisting of molybdenum, titanium, and carbon.
Owner:SEMICON ENERGY LAB CO LTD
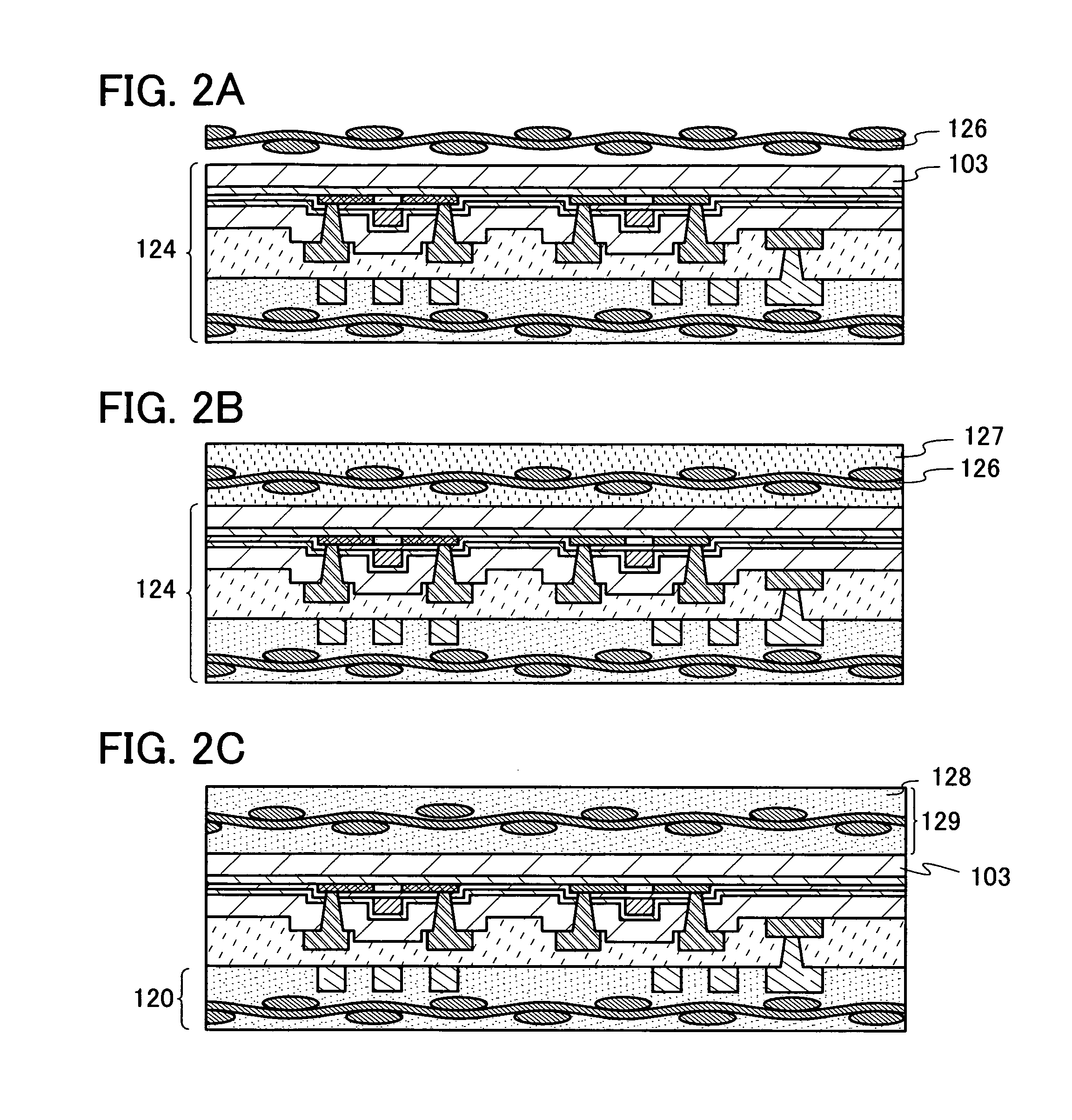
Method for manufacturing semiconductor device
InactiveUS20080242005A1Reduce communicationReduce connectionsSolid-state devicesSemiconductor/solid-state device manufacturingInorganic compoundSingle crystal
In the present application, is disclosed a method of manufacturing a flexible semiconductor device having an excellent reliability and tolerance to the loading of external pressure. The method includes the steps of: forming a separation layer over a substrate having an insulating surface; forming an element layer including a semiconductor element comprising a non-single crystal semiconductor layer, over the separation layer; forming an organic resin layer over the element layer; providing a fibrous body formed of an organic compound or an inorganic compound on the organic resin layer; heating the organic resin layer; and separating the element layer from the separation layer. This method allows the formation of a flexible semiconductor device having a sealing layer in which the fibrous body is impregnated with the organic resin.
Owner:SEMICON ENERGY LAB CO LTD
Non-alkaline micronized wood preservative formulations
Provided is a non-alkaline composition for the preservation of wood and other cellulosic materials. The composition comprises an aqueous dispersion of micronized inorganic compounds and / or organic biocides. Also provided is a method for making the composition.
Owner:OSMOSE
Semiconductor device and manufacturing method thereof
ActiveUS20070045621A1Reduce adhesionImprove adhesionElectroluminescent light sourcesSolid-state devicesSimple Organic CompoundsDevice material
It is an object of the present invention to manufacture, with high yield, semiconductor devices in each of which an element which has a layer containing an organic compound is provided over a flexible substrate. A method for manufacturing a semiconductor device includes: forming a separation layer over a substrate; forming an element-forming layer by forming an inorganic compound layer, a first conductive layer, and a layer containing an organic compound over the separation layer, and forming a second conductive layer which is in contact with the layer containing an organic compound and the inorganic compound layer; and after attaching a first flexible substrate over the second conductive layer, separating the separation layer and the element-forming layer at the separation layer.
Owner:SEMICON ENERGY LAB CO LTD
Composite particles and method for production thereof and use thereof
InactiveUS20060116279A1Improve photocatalytic activityImprove efficiencyPigmenting treatmentMaterial nanotechnologyFiberUltraviolet
A composite particle comprised of a larger particle and, supported thereon, smaller particles wherein the smaller particles are photocatalyst-containing fine particles with an average particle diameter of 0.005-0.5 μm as calculated from a BET specific surface area, and the larger particle has an average particle diameter of 2-200 μm as measured by the laser diffraction-scattering particle size measuring method. The smaller particle is preferably a composite particle of titanium dioxide with an inorganic compound exhibiting no catalytic activity, such as silica, or a particle containing a Brφonsted acid salt, especially on the surface thereof; and an advantageous method for producing the above composite particles wherein the above larger particles and smaller particles are dry mixed by a ball-mill or mixed by rotation of blades or by shaking, with an energy constant controlled within a specific range. A composition comprising an organic polymer and the above composite particles can give a shaped article, such as fiber, film or a molding, exhibiting ultraviolet ray-screening function.
Owner:SHOWA DENKO KK
Enhanced boron nitride composition and compositions made therewith
A boron nitride composition having its surface treated with a coating layer comprising at least one of a silane, a siloxane, a carboxylic derivative, and mixtures thereof, wherein the coating layer adheres to at least 10% of the surface of the boron nitride. The boron nitride powder surface is first treated by either a calcination process, or by coating with at least an inorganic compound for the surface to have a plurality of reactive sites containing at least a functional group that is reactive to at least one functional group of the final coating layer.
Owner:MOMENTIVE PERFORMANCE MATERIALS QUARTZ INC
Features
- Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com