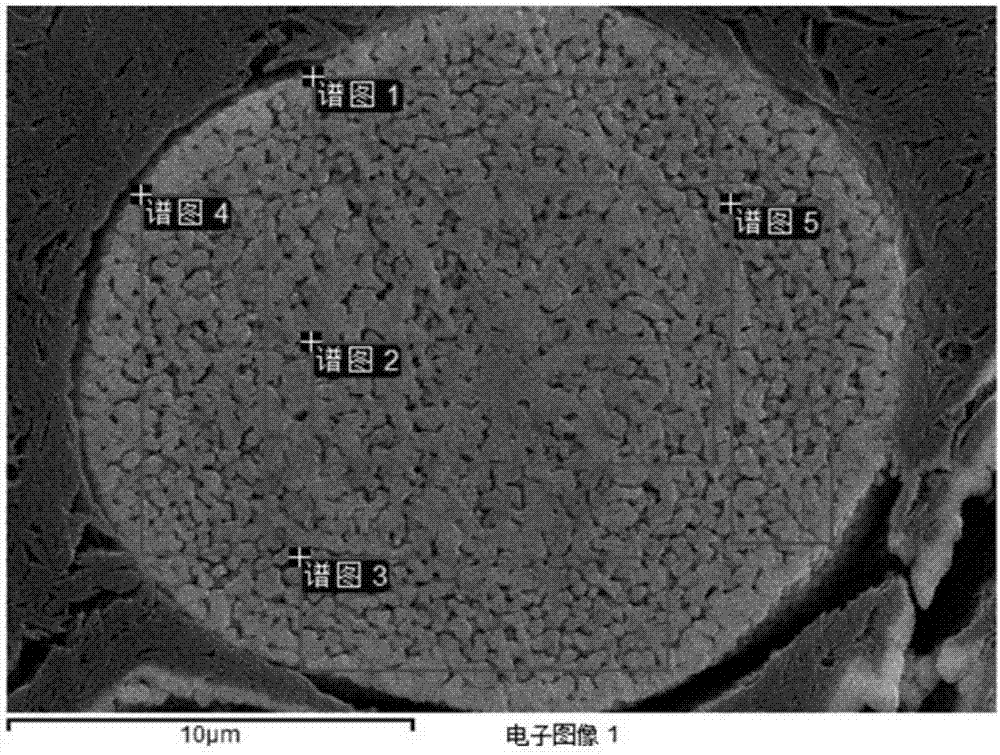
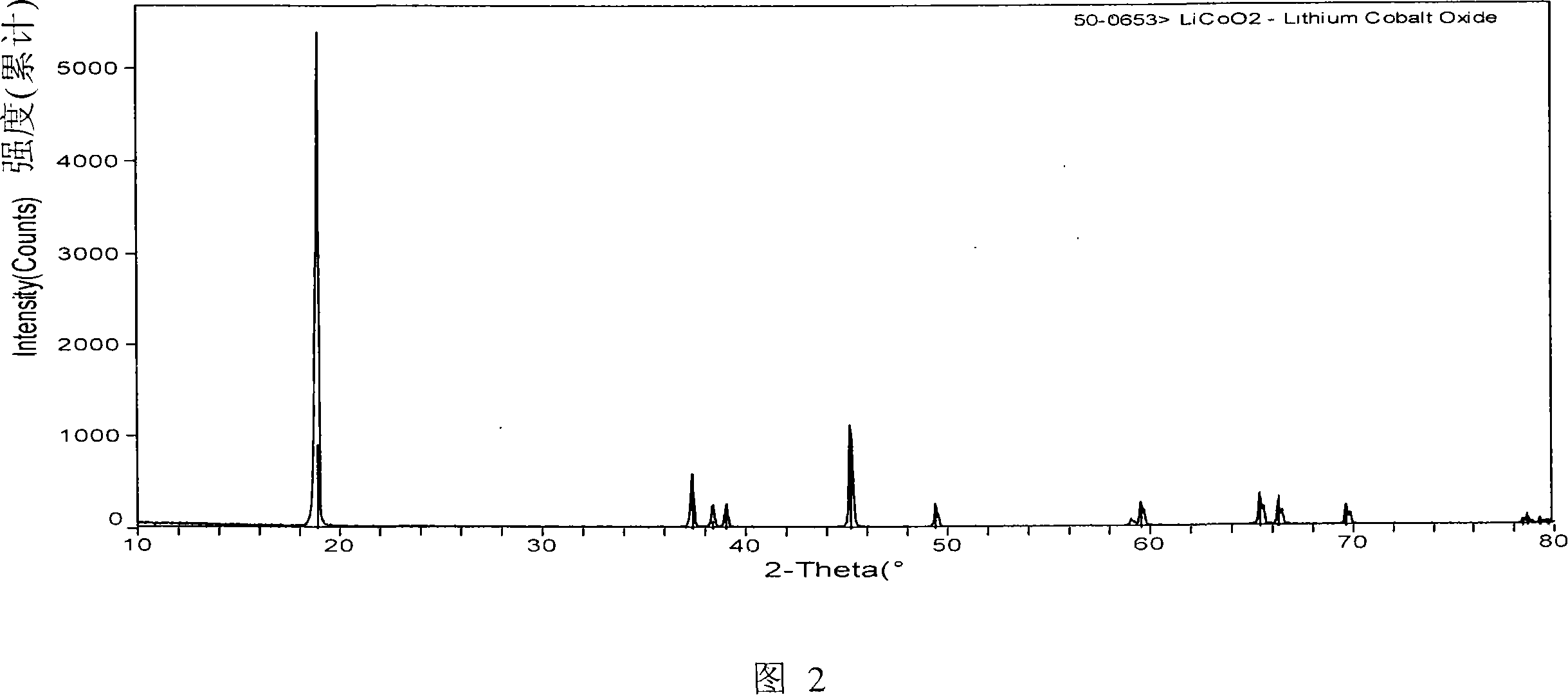
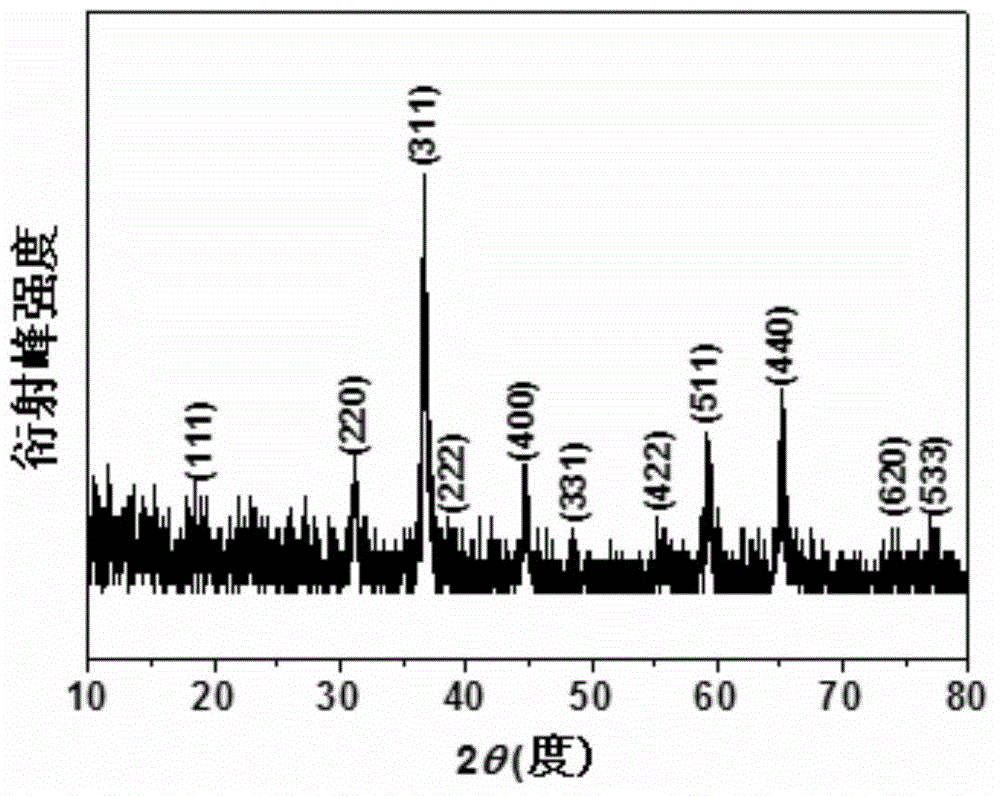
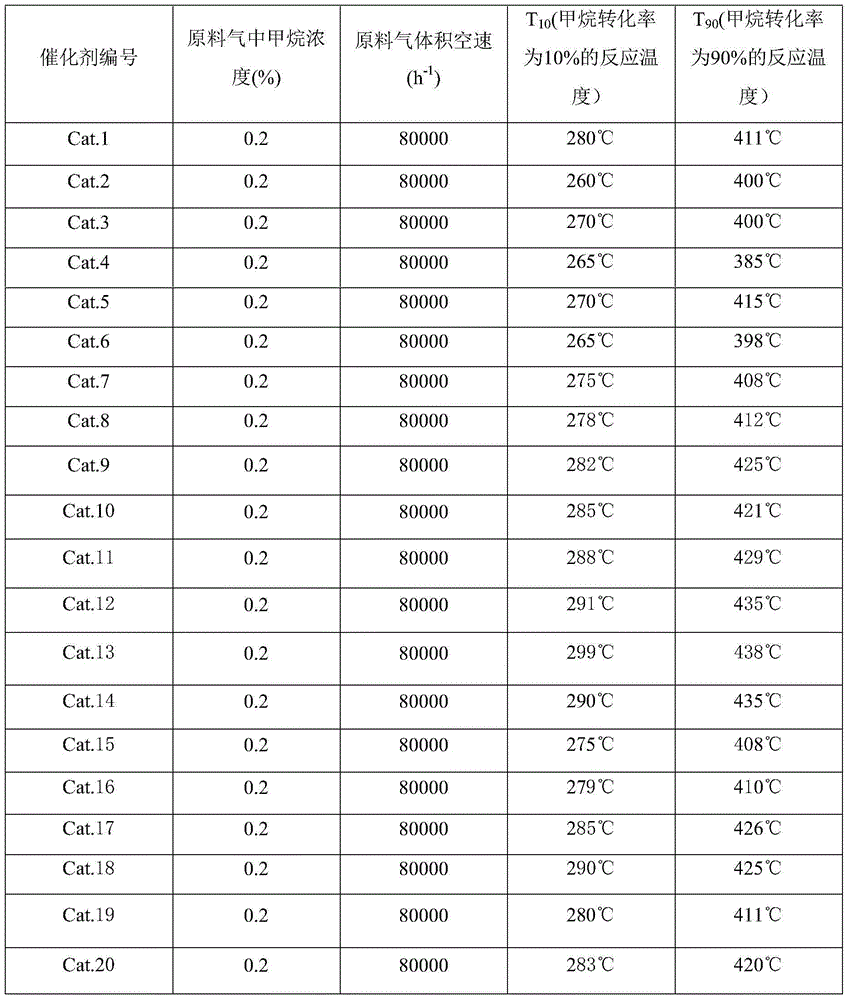
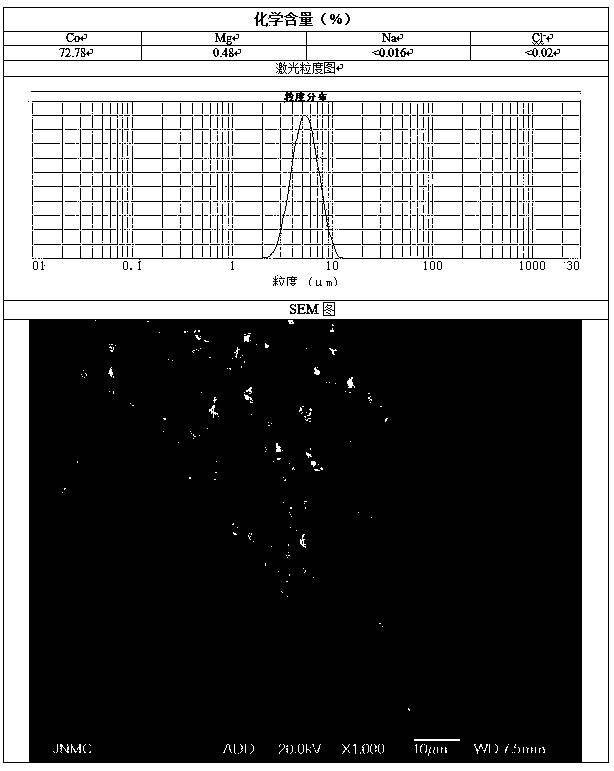
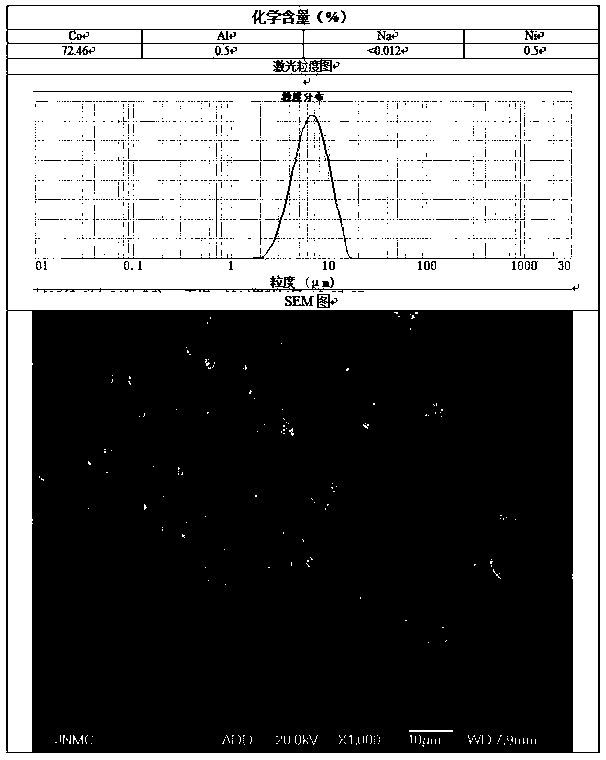
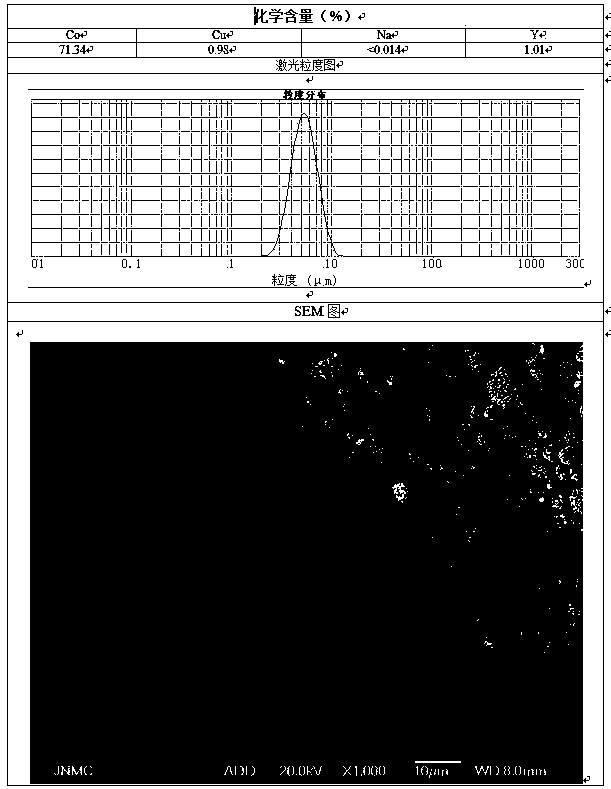
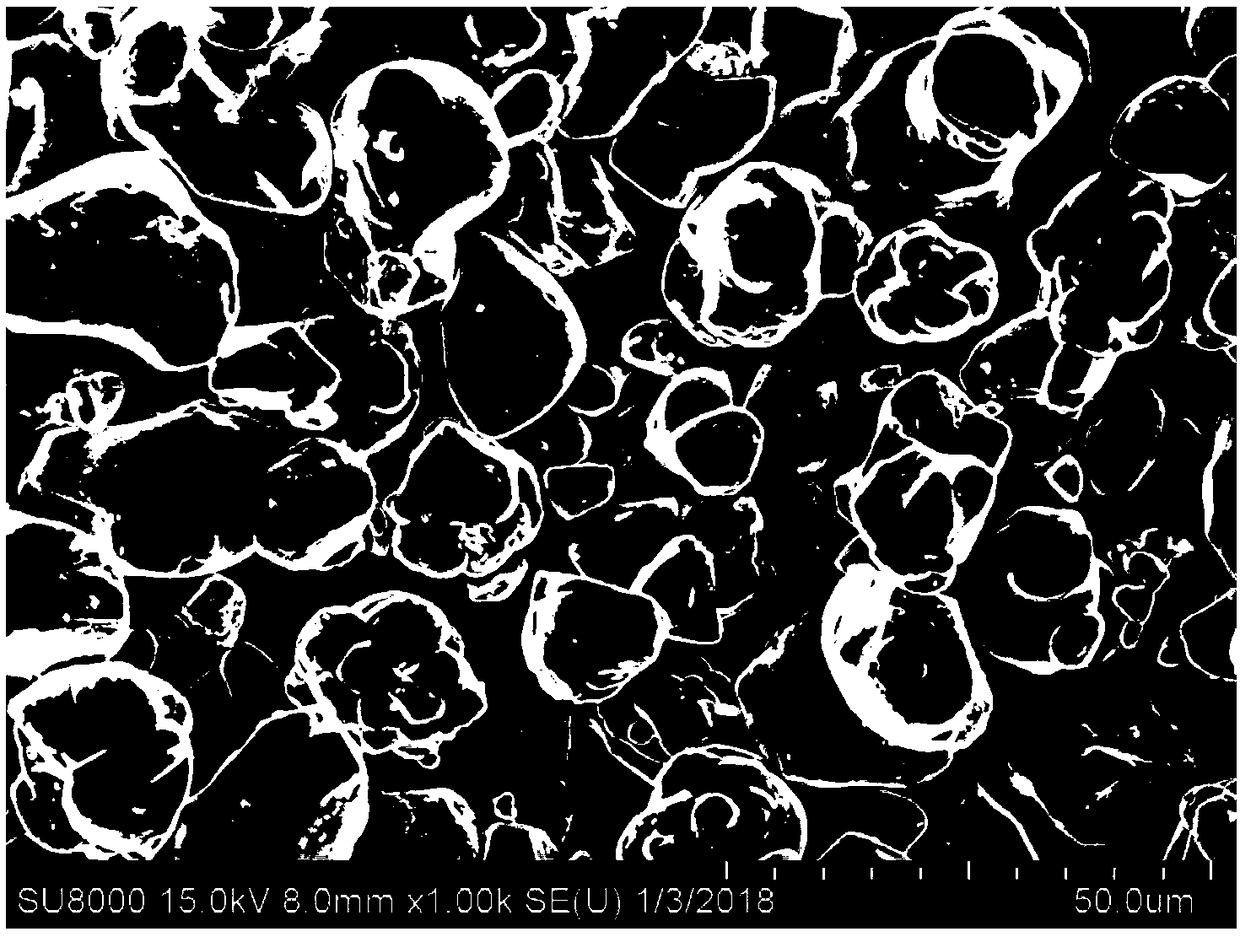
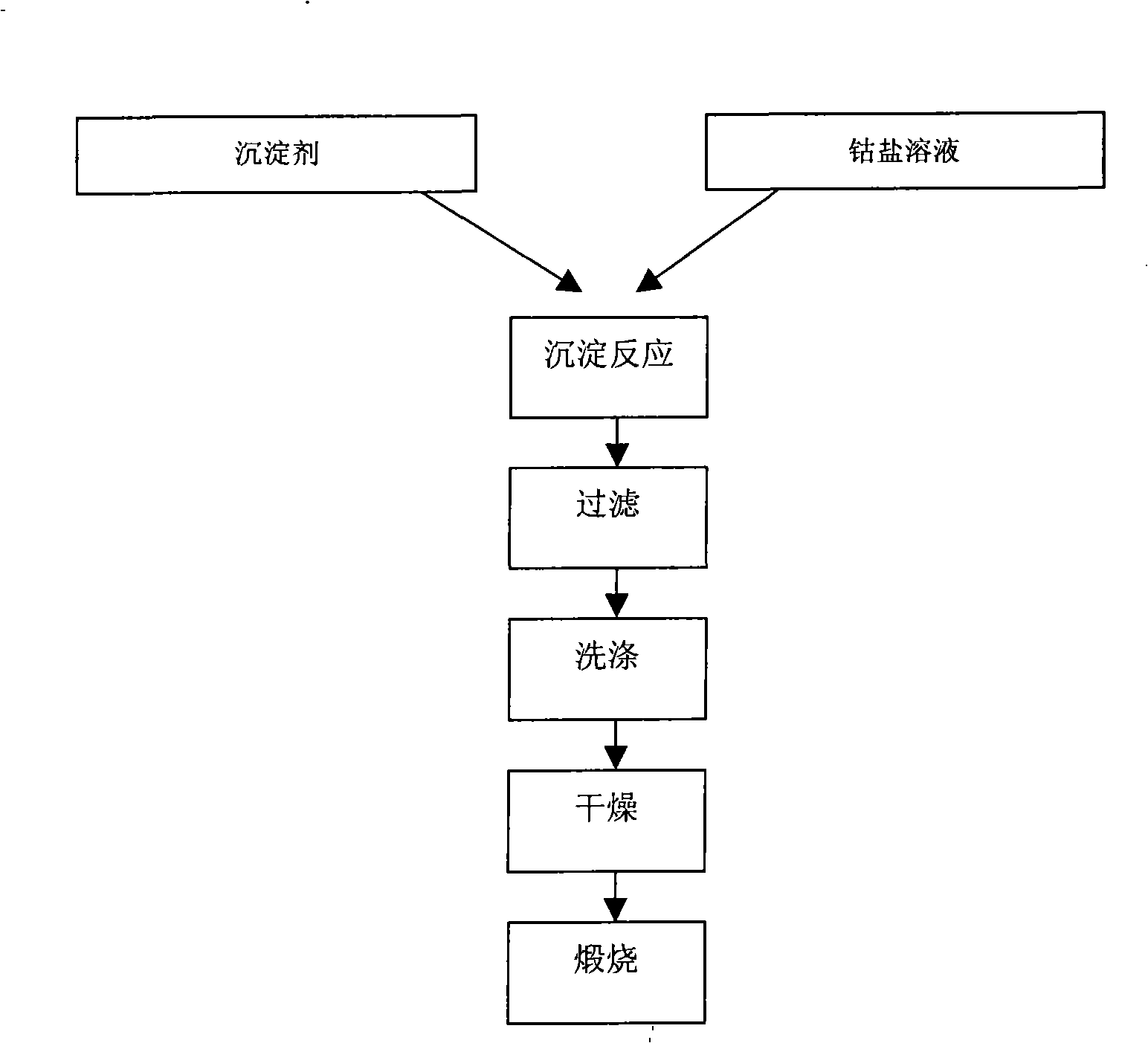
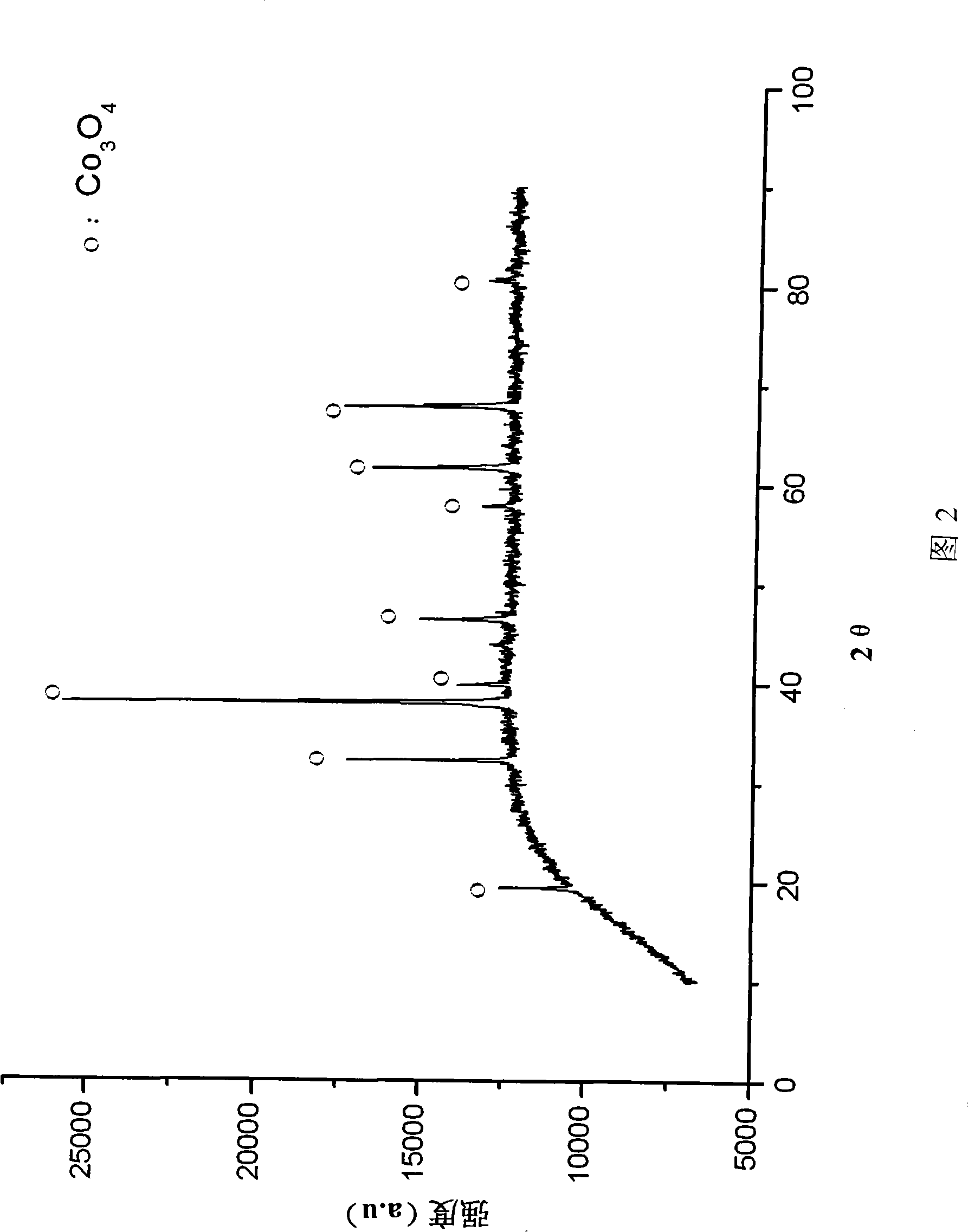
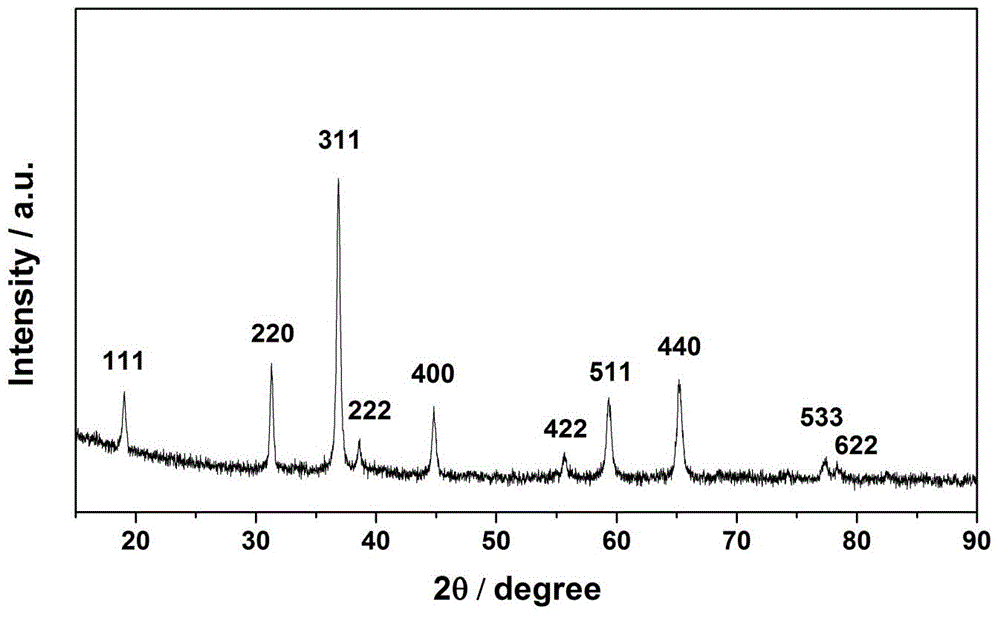
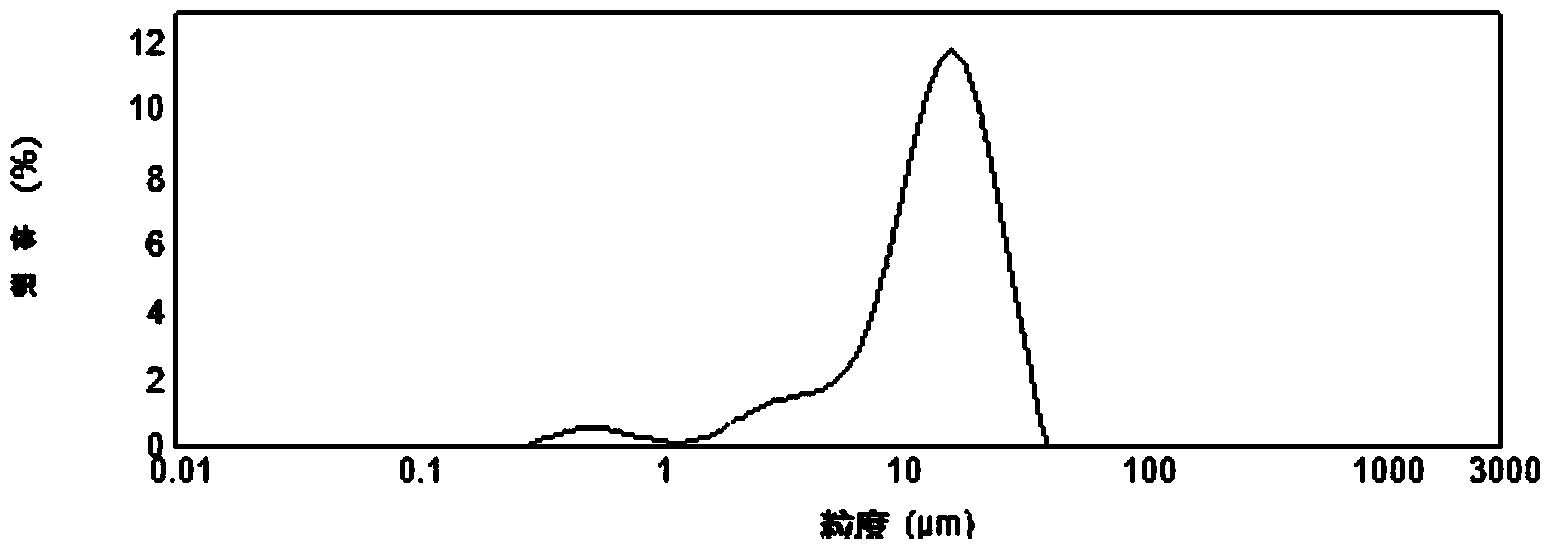
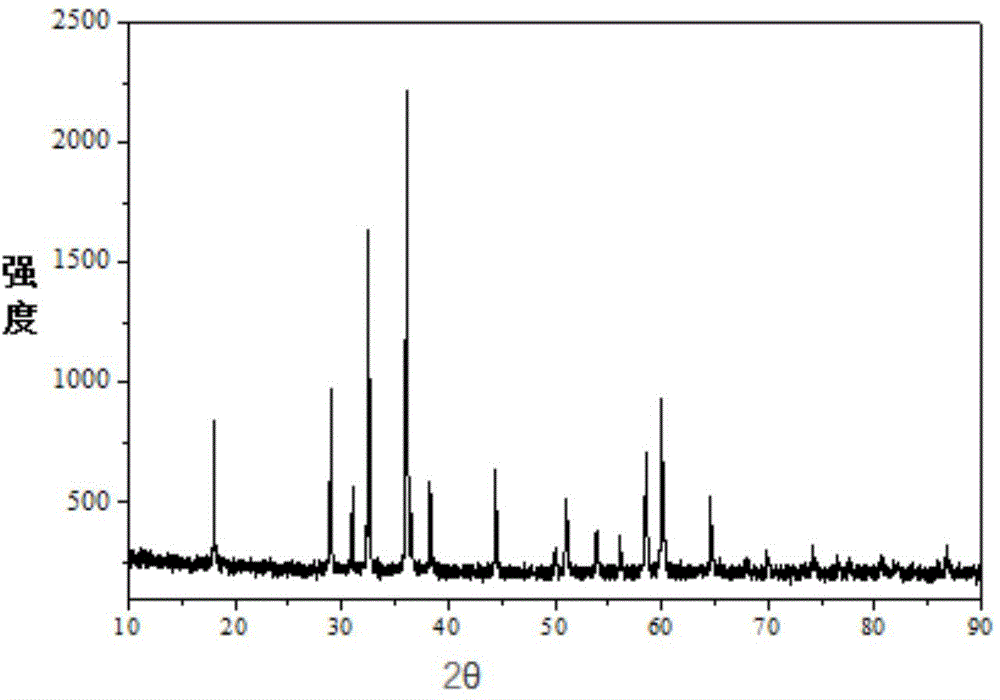
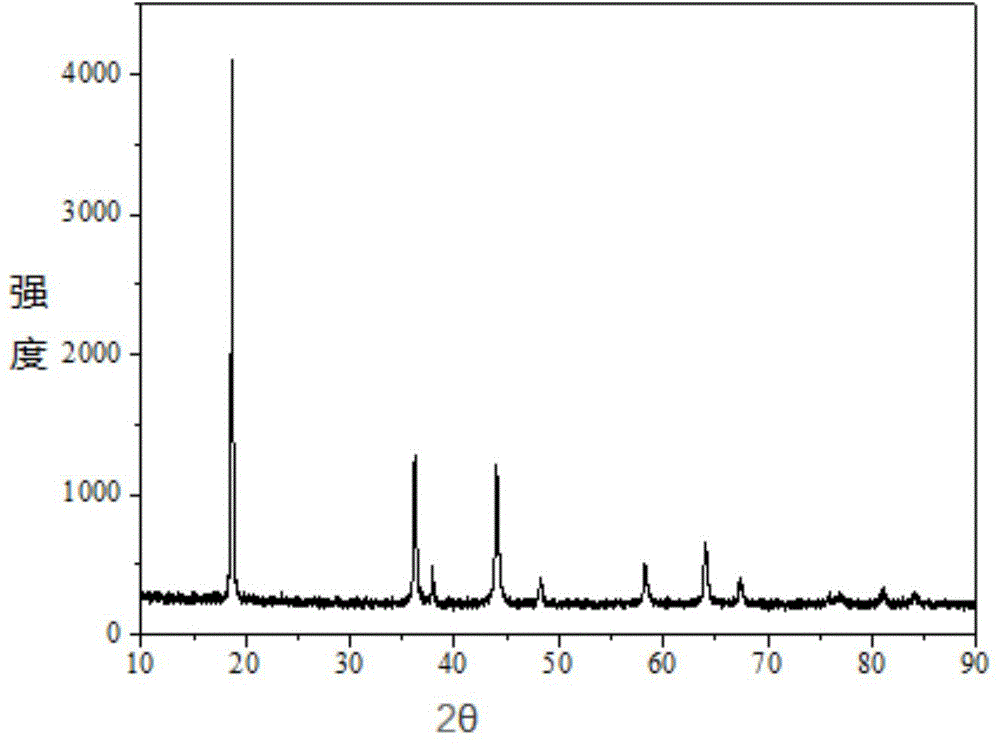
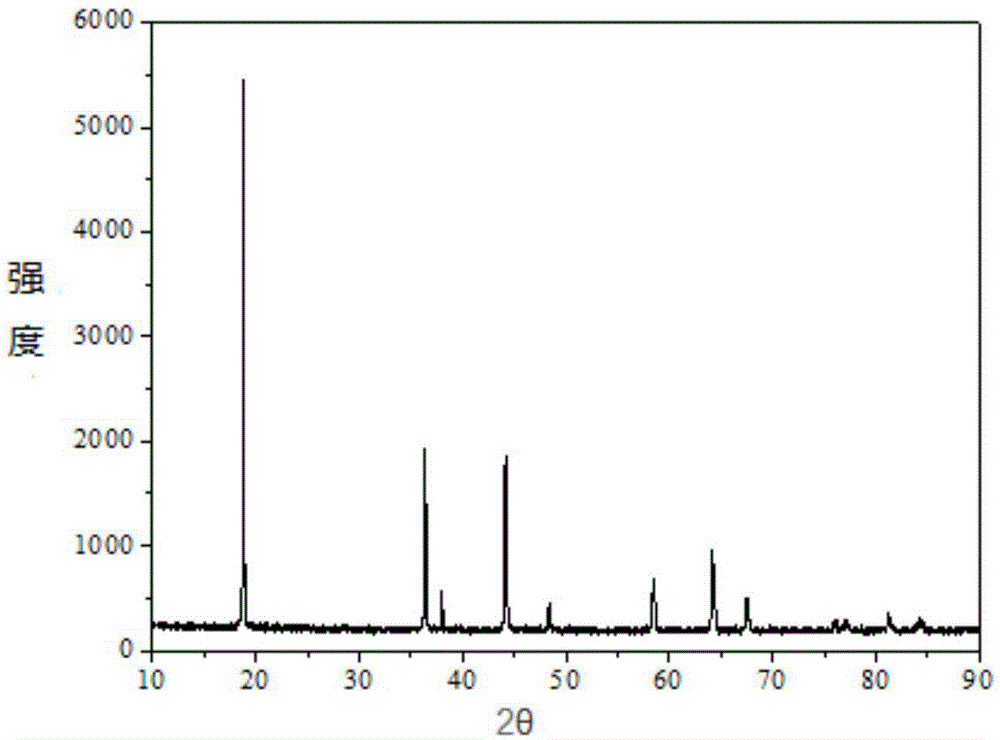
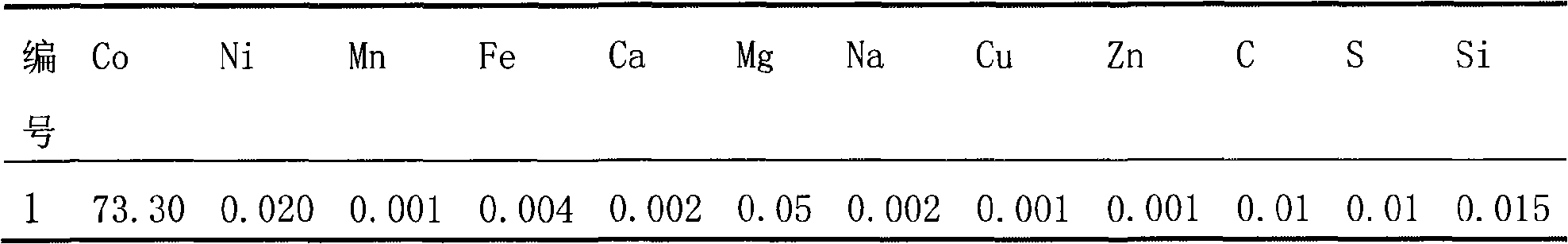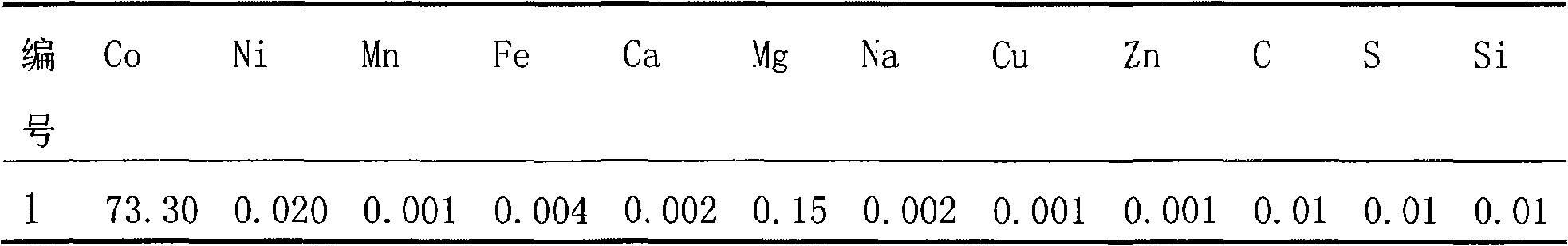Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
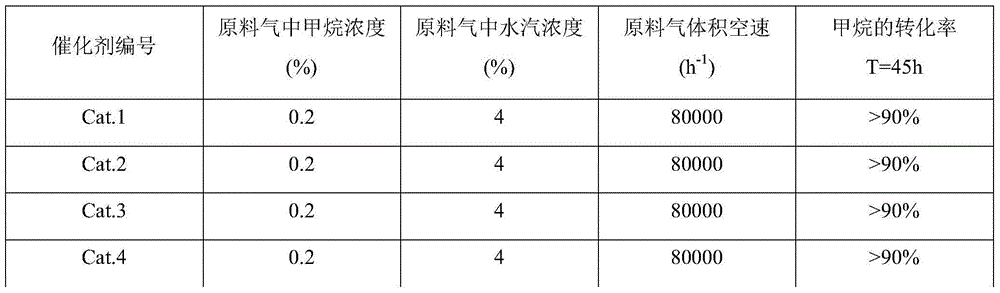
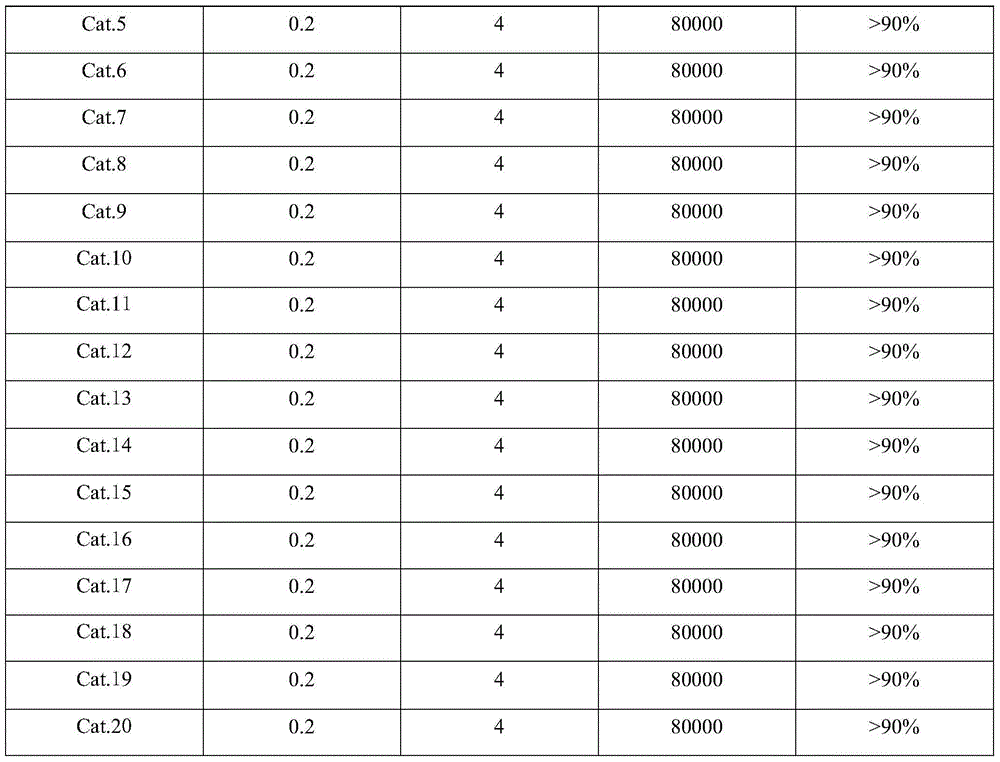
1149 results about "Cobalt(II,III) oxide" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
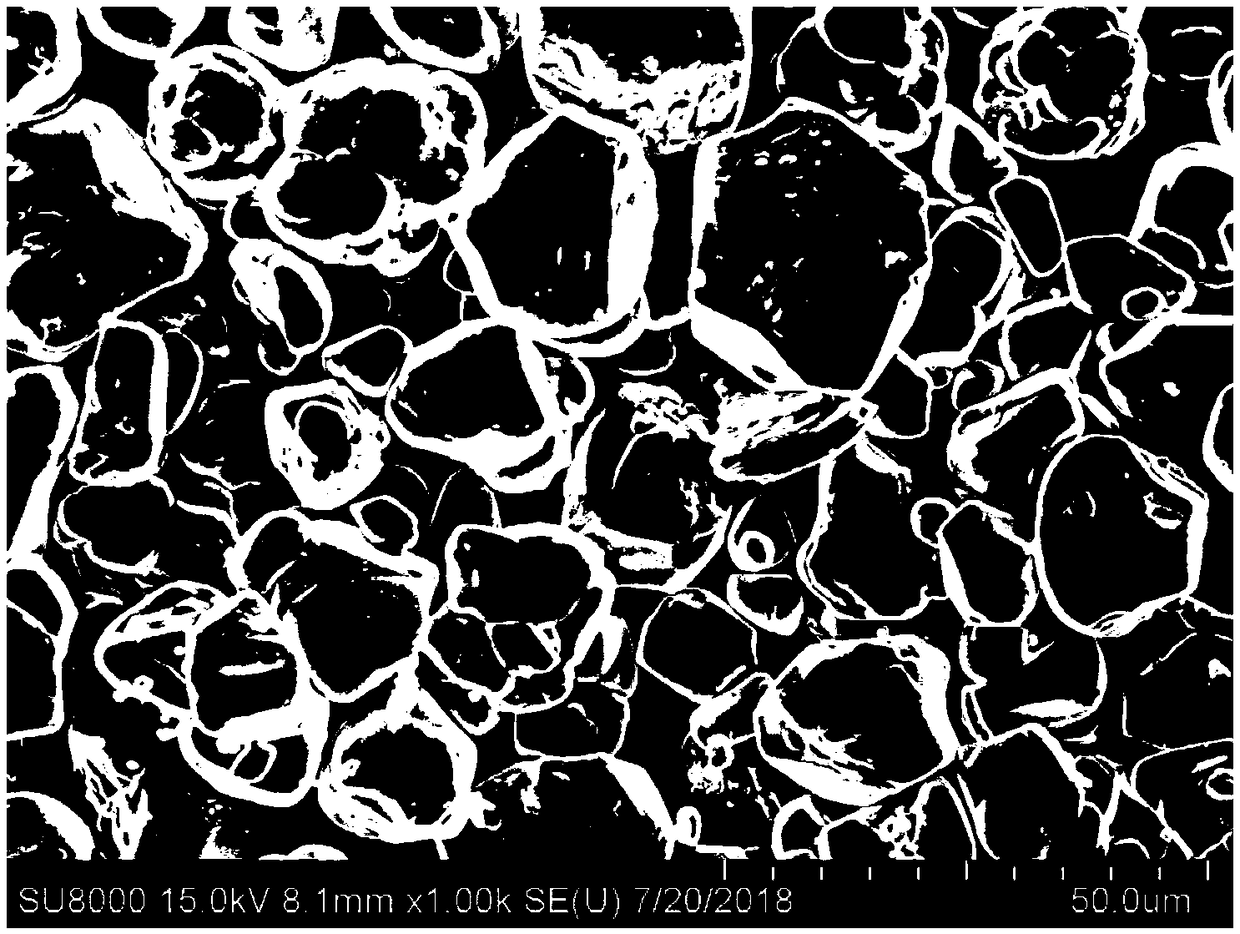
Cobalt(II,III) oxide is an inorganic compound with the formula Co₃O₄. It is one of two well characterized cobalt oxides. It is a black antiferromagnetic solid. As a mixed valence compound, its formula is sometimes written as CoᴵᴵCoᴵᴵᴵ₂O₄ and sometimes as CoO•Co₂O₃.
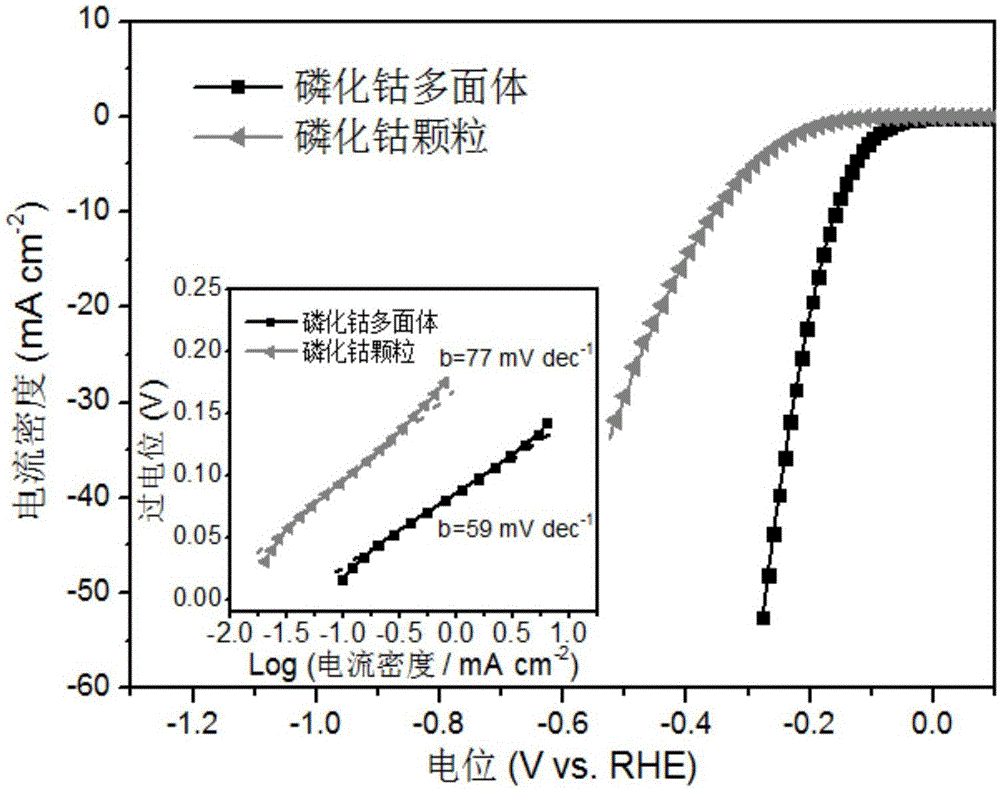
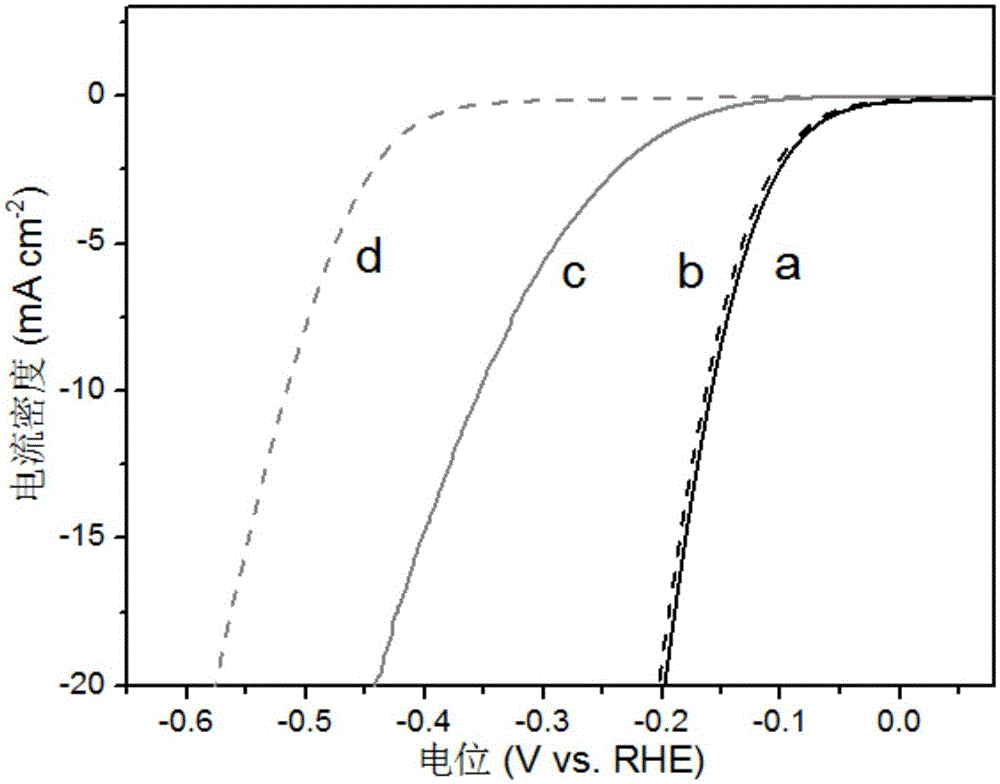
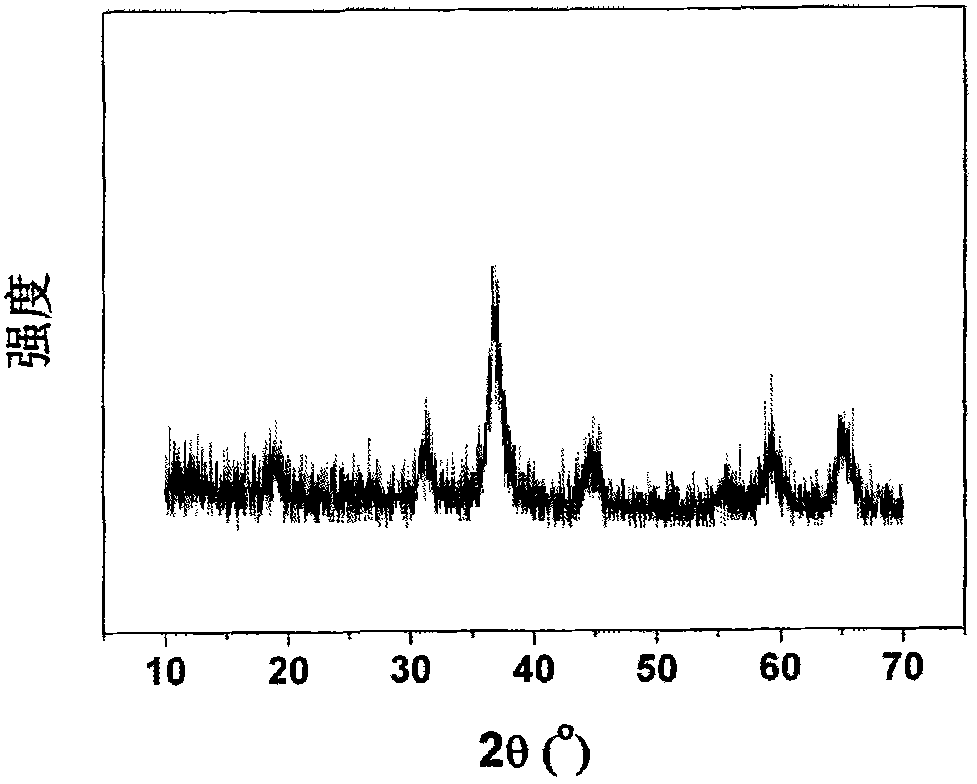
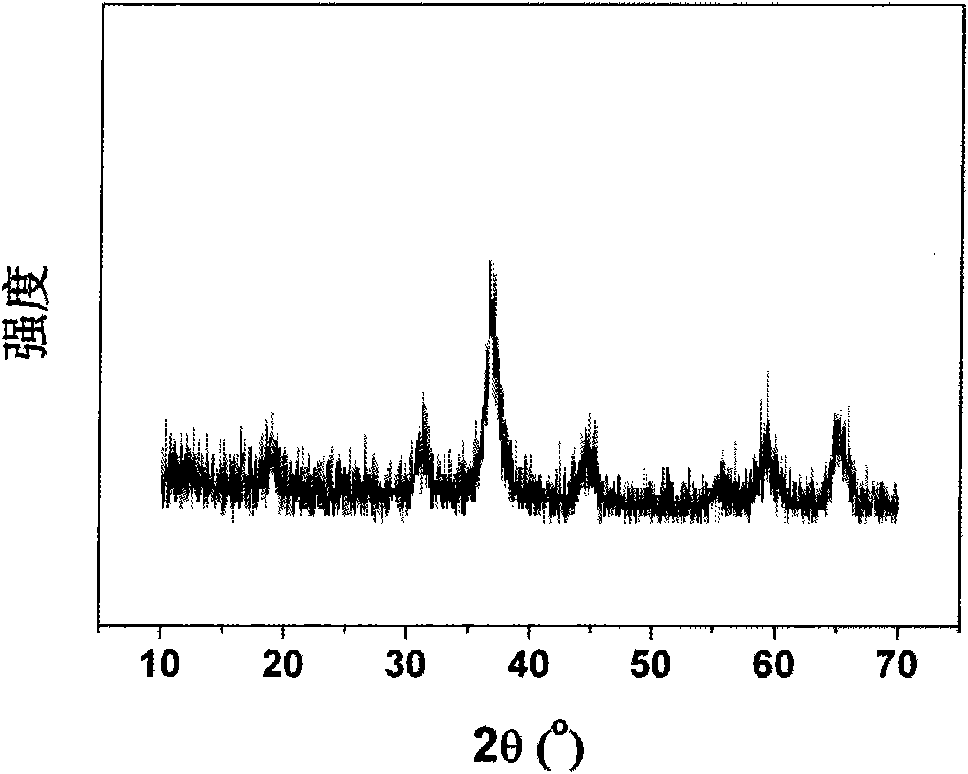
Preparation method of polyhedral cobalt phosphide catalyst for hydrogen production through water electrolysis
ActiveCN105107536AHigh crystallinityIncrease the areaElectrolysis componentsPhysical/chemical process catalystsAir atmosphereElectrolysis
A preparation method of a polyhedral cobalt phosphide catalyst for hydrogen production through water electrolysis comprises steps as follows: Co(NO3)2*6H2O and 2-methylimidazole are dissolved in methanol respectively, a 2-methylimidazole solution is poured into a Co(NO3)2 solution, the mixture is stirred and then aged at the room temperature, a product is centrifugally separated, vacuum drying is performed after washing with methanol, and a polyhedral metal organic frame ZIF-67 is obtained; then the polyhedral metal organic frame ZIF-67 is placed in a tube furnace, cobaltosic oxide is obtained through calcination in the air atmosphere, then the cobaltosic oxide and NaH2PO2*H2O are placed at two ends of a porcelain boat respectively, the NaH2PO2*H2O is located in the windward position of the tube furnace, and the polyhedral cobalt phosphide catalyst for hydrogen production through water electrolysis is obtained through calcination in the inert atmosphere. The crystallinity of the prepared cobalt phosphide catalyst material is high, the polyhedral morphology of a metal organic frame template is kept, the catalyst shows excellent properties in an electrocatalytic hydrogen evolution reaction, and the preparation technology is simple in process.
Owner:TSINGHUA UNIV
Graphene-supported cobaltosic oxide nano composite material and preparation method thereof
InactiveCN101811696ASmall sizeLarge specific surface areaCobalt oxides/hydroxidesCobalt(II,III) oxideCobalt salt
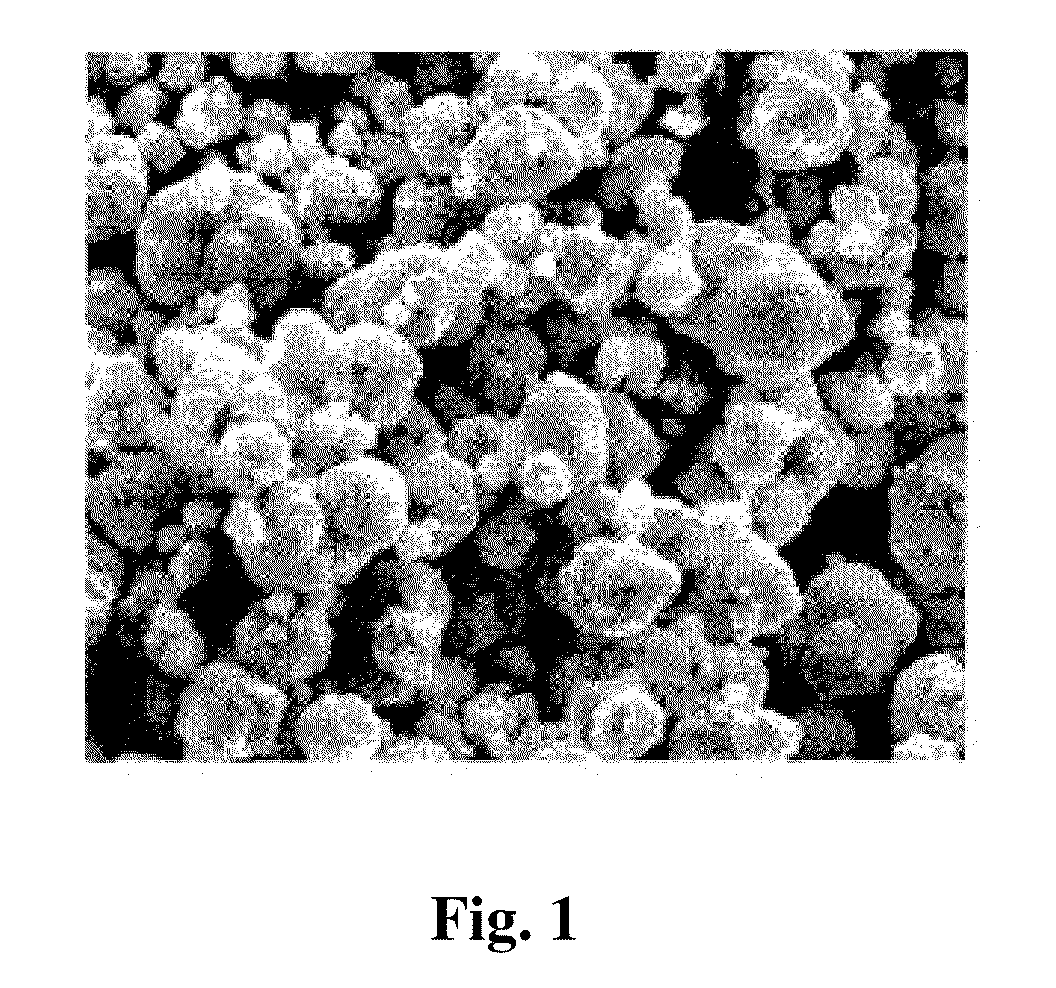
The invention discloses a graphene-supported cobaltosic oxide nano composite material and a preparation method thereof. The graphene-supported cobaltosic oxide nano composite material consists of graphene and cobaltosic oxide, wherein the cobaltosic oxide is loaded on graphene nano sheets; the content of the graphene nano sheets is 2 to 95 weight percent, and the thickness of the graphene nano sheets is 0.3 to 50 nanometers; and the particle size of the cobaltosic oxide is 1 to 200 nanometers and the cobaltosic oxide is ball-shaped or flaky. The preparation method comprises: firstly, mixing solution of graphene oxide, a bivalent cobalt salt and a polymer surfactant; and secondly, mixing the solution obtained by the first step with alkaline solution added with an oxidant, stirring the mixed solution or stirring the mixed solution by ultrasonic waves for 0.2 to 5 hours, transferring the mixed solution to a high-temperature reaction kettle, annealing the reaction product at 100 to 250 DEG C for 3 to 30 hours to obtain a product and washing and drying the product to obtain the graphene-supported cobaltosic oxide nano composite material. The size of the cobaltosic oxide is controllable. The reduction of the graphene oxide and the generation of the cobaltosic oxide are accomplished at the same time.
Owner:SOUTHEAST UNIV
Preparation of doped cobaltic-cobaltous oxide
InactiveCN101279771AImprove stabilityImprove electrochemical performanceCobalt oxides/hydroxidesSal ammoniacNitrate
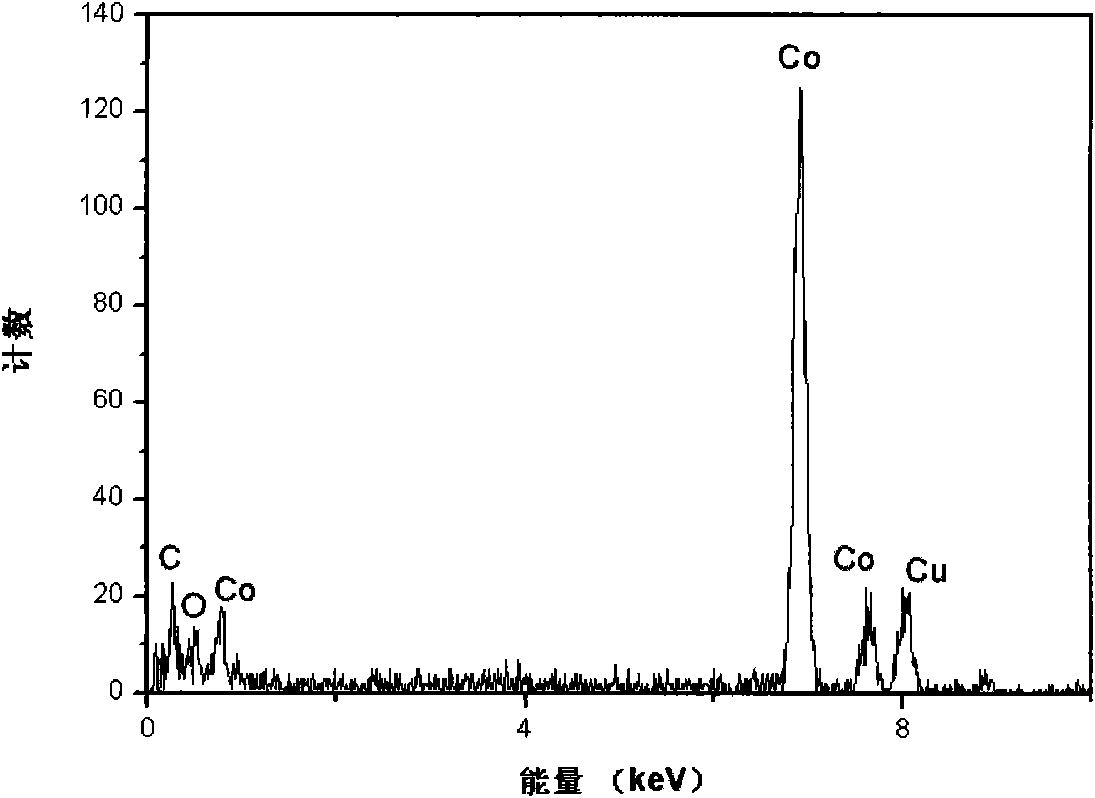
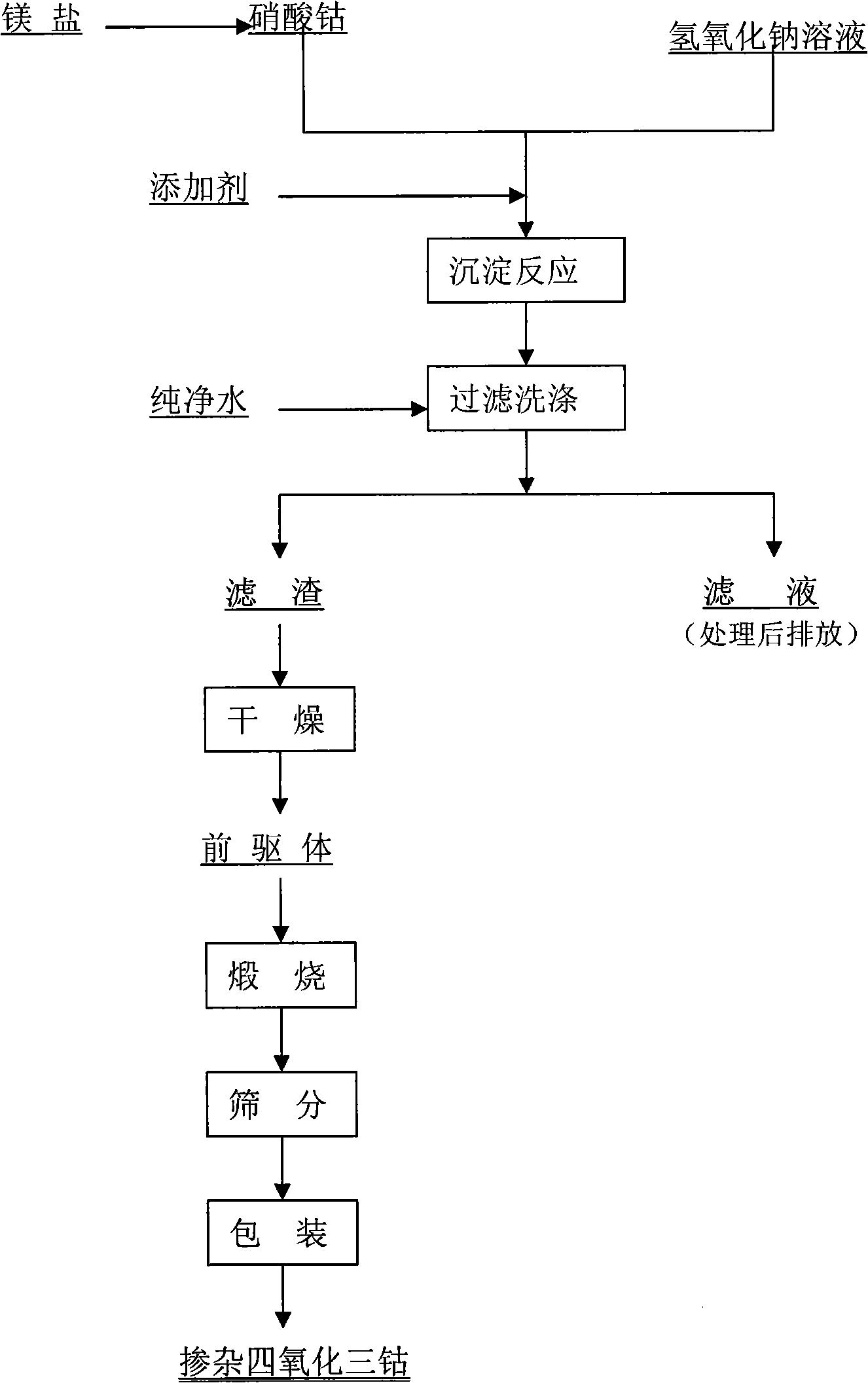
Disclosed is a process for preparing a doping cobaltosic oxide, which relates to a method for the production of a modified cobaltosic oxide used for a Lithium-ion battery anode material. The method is characterized in that the preparation process comprises: 1) mixing a cobalt nitrate solution containing doped chemical ions with a mixed precipitator solution containing ammonia and sodium hydroxide and making the mixture react for eight to twenty hours at a pH value of between 8.4 to 10 and a temperature of between 40 and 80 DEG C so as to prepare a cobalt hydroxide precipitation containing doped chemical; and 2) washing and drying the cobalt hydroxide precipitation containing the doped chemical and then burning the precipitation for two to six hours at a temperature of between 500 and 800 DEG C so as to obtain the doping cobaltosic oxide. The method of the invention can get even particles with regular shapes after the reaction, the particle sizes of the doping cobalt hydroxide are controllable in a certain range, and the doping cobaltosic oxide can be obtained by calcinations. The method of preparation is characterized in that a magnesium source, an aluminum source, a titanium source, etc. are induced to the cobalt nitrate solution directly, and the process and operation are simple and easy.
Owner:JINCHUAN GROUP LIMITED
Preparation method of Co3O4 with large grain size and uniformly doped with aluminum
ActiveCN108011101AEvenly distributedUniform particle size distributionCell electrodesCobalt oxides/hydroxidesCobalt(II,III) oxideMicrometer
The invention relates to a preparation method of Co3O4 with large grain size and uniformly doped with aluminum. The invention provides the preparation method of the Co3O4 which is uniformly doped withaluminum and is large in grain size and uniform in particle distribution, and the obtained Co3O4 with the large grain size and uniformly doped with aluminum can completely conform to the requirementof preparation of 4.45V high-voltage lithium cobalt oxide. According to the method, the large-grain size and aluminum-doped cobalt carbonate is synthesized by a wet method, and the problems of difficulty in enlargement of cobalt hydroxide (or hydroxyl cobalt) system grain size and non-uniform particle distribution are solved; with the regard to the problem of uniform aluminum doping of a cobalt carbonate system, a parameter is set from principle, an aluminum compound is prevented from being independently separated out and gathered, the doped Al element can be uniformly distributed in the Co3O4, the grain size reaches 15 micrometers or above, and the particle distribution is uniform; and the lithium cobalt oxide prepared from the aluminum-doped Co3O4 has high specific capacity and excellentcycle property under 4.45V.
Owner:취저우화여우코발트뉴머터리얼컴퍼니리미티드
Ultrathin nanosheet array electro-catalytic material with nano-porous structure and oxygen vacancies
The invention relates to an ultrathin nanosheet array electro-catalytic material with a nano-porous structure and oxygen vacancies. The material is a cobaltosic oxide primary nanosheet array which grows vertically on a conductive substrate and is doped with a metal; an ultrathin nanosheet with oxygen vacancies and nanopores is obtained on each primary nanosheet; the conductive substrate is a titanium sheet or a foamed nickel sheet, and the doped metal is zinc, nickel or manganese; and the thickness of each cobaltosic oxide ultrathin nanosheet doped with the metal is 1.22 nm, nanosheets are in a three-dimensional porous structure, and the nano-pore diameter is 3-6 nm. The ultrathin nanosheet array electro-catalytic material with the nano-porous structure and oxygen vacancies has the following advantages: the material can effectively reduce the overpotential and the spike potential of an oxygen evolution reaction, increase the conversion rate of a single cobalt atom and work continuously and stably in an alkali environment; the steps of a preparation method of the material are simple, the operation is convenient, the cost is low, and the material is environmental-friendly; and new ideas and strategies are provided for the function-oriented design and the performance optimization of an oxygen evolution catalyst of a water electrolysis system.
Owner:TIANJIN UNIVERSITY OF TECHNOLOGY
Anode material zirconium and phosphor adulterated lithium cobaltate of lithium ion secondary battery and its making method
ActiveCN101150190AImprove cycle performanceImprove securityCell electrodesCobalt compoundsLithium hydroxidePhosphate
This invention relates to LiCoO2 doped with Zr and P, positive material of Li ionic secondary cells and its preparation method characterizing that the chemical formula is: LiZrxCo(1-x-y)PyO2, in which, it is a laminated structure, and x=0.001-0.003, y=0.02-06, the preparation steps are as folow: 1, mixing Co3O4, CoCO3 or CoC2O4 with Li2CO3 or LiOH in the atomic ratio of Li and Co(0.9801.05) :1.00, 2, baking it for 6-24h under 600-1000deg.C, 3, crushing LiCoO2 to particles of 6-15mum, 4, mixing the LiCoO2 and water 1-3times weight into pulp, 5, adding ZrNO3 and phosphate at the same time, 6, spraying and drying it, 7, baking it for 4-12h under 600-1000deg.C, 8, crushing the doped LiCoO2 to particles of 6-15mum.
Owner:TIANJIN B&M SCI & TECH
Nitrogen-doped graphene and Co3O4 hollow nanosphere composite material as well as preparation method and application of composite material
ActiveCN104681823AImprove conductivityLarge specific surface areaMaterial nanotechnologyPhysical/chemical process catalystsDoped grapheneNitrogen doped graphene
The invention relates to a novel composite material, and particularly relates to a preparation method and application of a nitrogen-doped graphene and cobaltosic oxide hollow nanosphere composite material. The novel composite material comprises a doped graphene substrate and cobaltosic oxide hollow nanospheres which is attached to the surface of the doped graphene substrate. Melamine resin is taken as a cross-linking reagent for integrating graphite oxide with Co<2+> into a single coordination precursor. The preparation method comprises the following steps: in a pyrolysis process of the precursor, taking the melamine resin as a new nitrogen source to uniformly dope the graphene with nitrogen, fixing cobalt oxide which is generated in situ, and finally preparing the nitrogen-doped graphene / Co3O4 hollow nanosphere composite material with a sandwich structure. The composite material has a graded porous structure, is high in specific surface area, more in active sties, good in electron conductivity and ion conductivity, and good in application prospect in the field of new energy resources and catalysis.
Owner:CHINA WEST NORMAL UNIVERSITY
Preparation method of large-particle-size and high-density spherical cobalt oxide
The invention discloses a preparation method of large-particle-size and high-density spherical cobalt oxide. The preparation method aims at achieving the purposes that the median particle size D50 of the cobalt oxide is larger than or equal to 15 microns, particle size distribution is uniform, compacting density (TD) is larger than or equal to 2.2 g / cm<3>, and the large-particle-size and high-density spherical cobalt oxide can meet the development requirement of high-voltage lithium cobaltate. The preparation method of the cobalt oxide includes the cobalt carbonate synthesis stage and the spherical cobalt carbonate thermal decomposition stage. In the cobalt carbonate synthesis stage, a cobalt carbonate precursor prepared through many times of cyclic crystallization is compact and uniform in particle size distribution; due to the segmented thermal decomposition, cobalt carbonate is locally decomposed in the low-temperature pre-decomposing stage, a micro hole channel is formed, release of CO2 gas during subsequent decomposition is facilitated, and particles are prevented from crazing or being broken; in the high-temperature thermal decomposition stage, the particle surface is compact, and therefore the large-particle-size and high-density spherical cobalt oxide is prepared.
Owner:취저우화여우코발트뉴머터리얼컴퍼니리미티드 +1
Preparation technology for spherical lithium cobalt oxide doped with Ti, Mg and Al
InactiveCN102583585ASimple processReaction is easy to controlCell electrodesCobalt carbonatesSlurryCarbonate
The invention relates to the field of lithium ion battery materials and particularly relates to a preparation technology for spherical lithium cobalt oxide doped with Ti, Mg and Al. According to the preparation technology, pure water serves as base solution under the conditions that temperature ranges from 40 DEG C to 80 DEG C and a stirring speed is controlled to 60-180 r / min, ammonium bicarbonate is added into the base solution to adjust the alkalinity of the base solution to range from 5 to 30, cobalt-salt mixed solution and ammonium bicarbonate solution are continuously pumped into the base solution in parallel flow and are subjected to sedimentation reaction under the temperature of 40-80 DEG C, the solid content and the alkalinity of slurry in a reaction system are controlled, reaction feed liquid flows out continuously from an overflow gate at the upper part of a reaction kettle to enter into an ageing kettle and then is washed by a washing press and dried by a vacuum drier, spherical cobalt carbonate is subjected to primary calcination to obtain cobaltosic oxide, and the cobaltosic oxide and a lithium source are subjected to secondary calcination to obtain the spherical lithium cobalt oxide doped with Ti, Mg and Al. According to the invention, the process is simple, the reaction is easy to control, the production cost is low, the product application range is wide, and thus, the method is relatively large in production value; and the spherical lithium cobalt oxide is stable in performance and better in overcharging prevention performance.
Owner:ANHUI ALAND NEW ENERGY MATERIALS
Permanent ferrite magnetic tile and preparation method thereof
InactiveCN101202138AIncrease solid solutionImprove performancePermanent magnetsInorganic material magnetismFiberSilicon oxide
The invention relates to a permanent ferrite magnetic arc and the manufacturing method thereof, belonging to a manufacturing field of a motor permanent ferrite. Weight proportions of main material components and additive components are as the following: main materials: 8.7 to 9.0 percent of strontium oxide; 86 to 87 percent of iron oxide red; additives: 0.6 to 1.0 percent of calcium carbonate; 0.6 to 1.0 percent of aluminum oxide; 0.4 to 0.8 percent of chrome oxide; 0.3 to 0.6 percent of boric acid; 0.3 to 0.5 percent of silicon oxide; 0.2 to 0.4 percent of cobalt oxide; 0.2 to 0.4 percent of lanthanum oxide and 0.1 to 0.3 percent of silicate fiber. The invention is provided with high surplus magnetic induction strength and high intrinsic coercivity so as to enhance a mechanical strength of the magnetic arc.
Owner:CHANGZHOU DIER MAGIC MATERIALS
Non-noble metal oxide combustion catalyst, and preparation method and use thereof
InactiveCN105381800AEasy to synthesizeLow costDispersed particle separationCatalyst activation/preparationChemical synthesisCobalt(II,III) oxide
The present invention relates to a non-noble metal oxide combustion catalyst and a preparation method and use thereof, and belongs to the technical field of energy utilization and environment protection. The catalyst is iron oxide, cobaltosic oxide, nickel oxide, cupric oxide, vanadium oxide, chrome oxide, manganese dioxide or cerium dioxide prepared according to the following steps: (1) using hydrothermal chemical synthesis to form a precursor of a catalyst; and (b) washing, filtering, shaping, drying and calcinating the obtained precursor of the catalyst, and finally forming a combustion catalyst or coating the precursor of the catalyst on a carrier to form a combustion catalyst. The catalyst prepared by the method can be used in catalytic combustion of methane and other VOC gas, and has the advantages that the synthesis process is simple, the cost is low, the catalytic activity and hydrothermal stability is high, and the light-off temperature and complete combustion temperature of methane are low.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
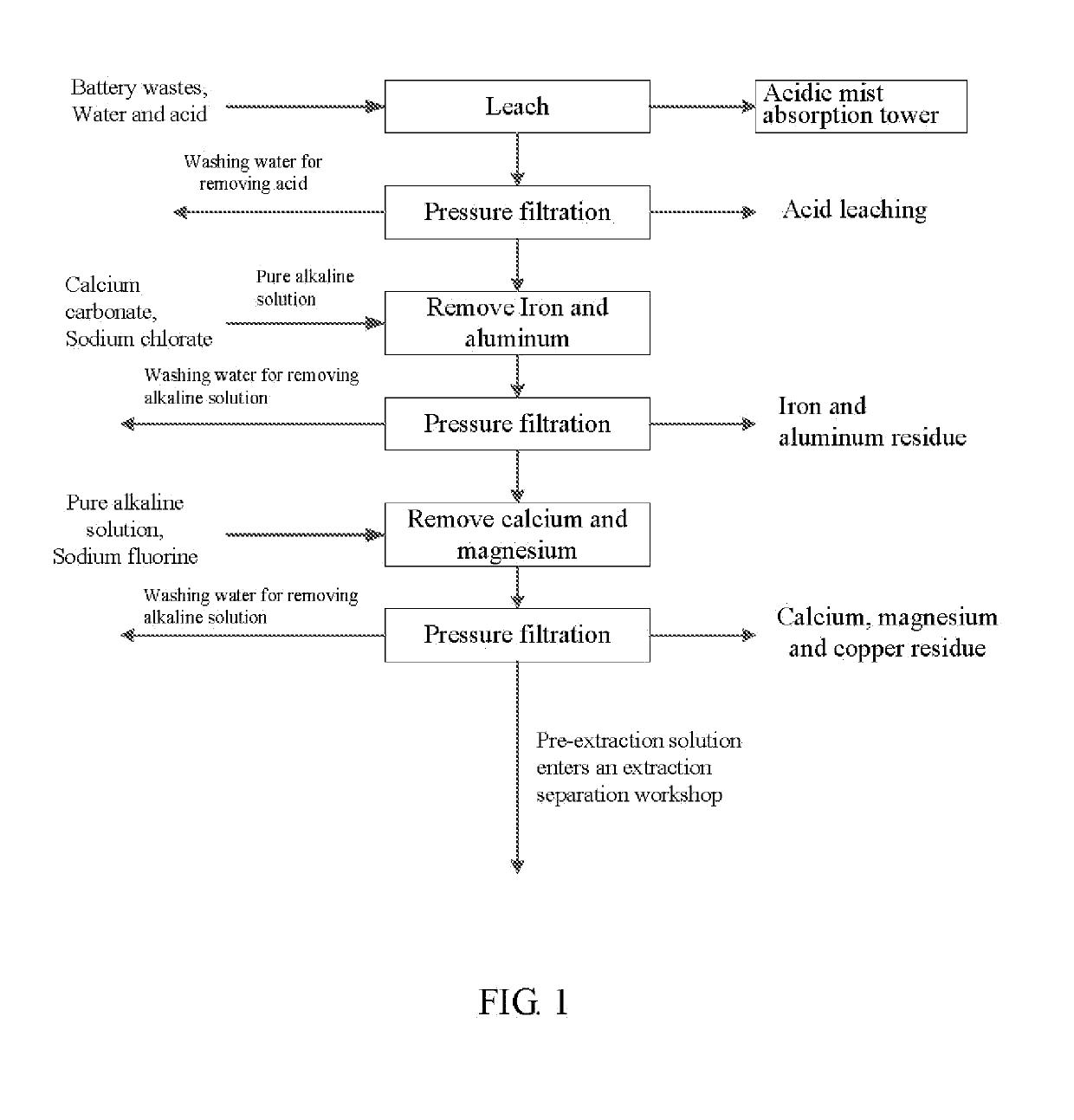
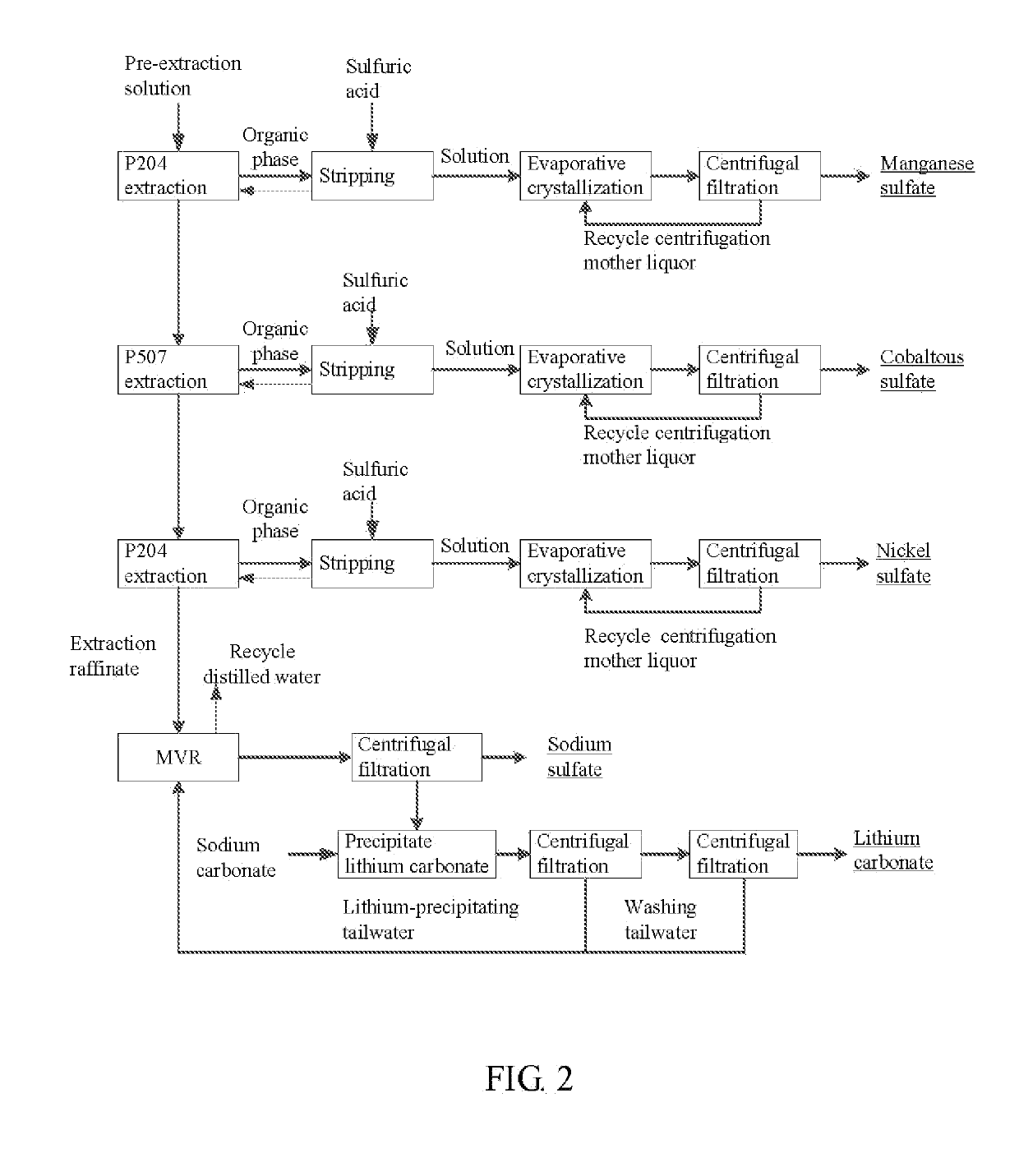
Method for preparing nickel/manganese/lithium/cobalt sulfate and tricobalt tetraoxide from battery wastes
ActiveUS20190152797A1Reduce productionHigh puritySolvent extractionCobalt sulfatesManganeseCobalt Sulfate
A method for preparing nickel / manganese / lithium / cobalt sulfate and tricobalt tetraoxide from battery wastes adopts the following process: dissolving battery wastes with acid, removing iron and aluminum, removing calcium, magnesium and copper, carrying extraction separation, and carrying out evaporative crystallization to prepare nickel sulfate, manganese sulfate, lithium sulfate, cobalt sulfate or / and tricobalt tetraoxide. By using the method, multiple metal elements, such as nickel, manganese, lithium and cobalt, can be simultaneously recovered from the battery wastes, the recovered products are high in purity and can reach battery grade, battery-grade tricobalt tetraoxide can also be directly produced. The method is simple in process, low in, energy consumption and free in exhaust gas pollution, and can realize zero release of wastewater.
Owner:HUNAN JINYUAN NEW MATERIALS CO LTD
Preparation method of cobaltosic oxide for electric battery
ActiveCN101200308AImprove liquidityUniform particle sizeCobalt oxides/hydroxidesGranularityCobalt(II,III) oxide
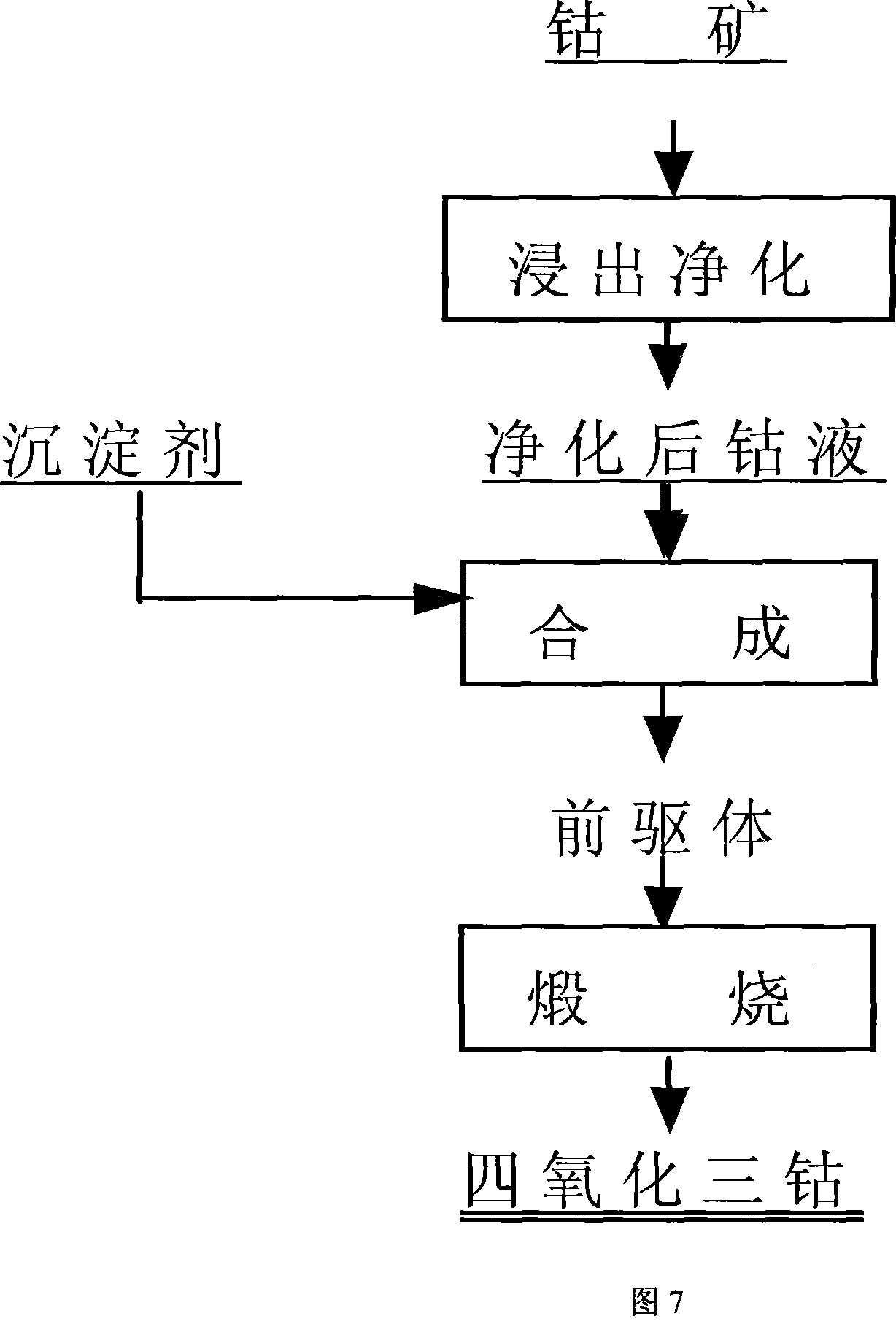
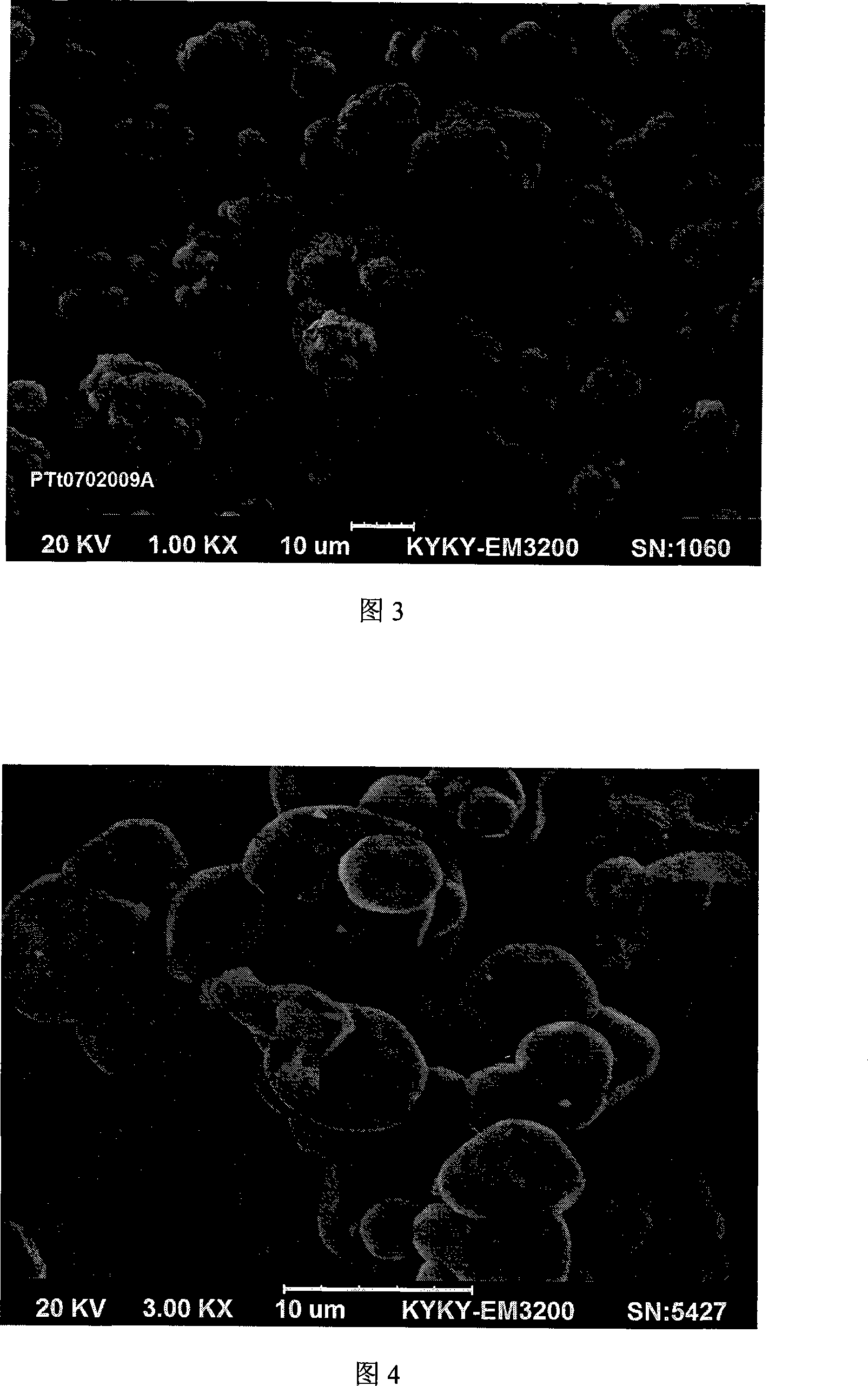
The invention discloses a preparation method of Co3O4 for batteries, Co mineral is used as Co raw material, after purified the Co liquid is obtained, complexing-homogeneous sedimentation method is adopted to prepare precursor, i.e the Co liquid is prepared into the complexing Co salt solution, and then added with precipitator solution, the synthesis of the precursor is controlled to obtain single-dispersion sedimentation precursor so as to make the production and growth of the nucleation be separated, and then sintered under high temperature, the precursor is thermally decomposed to obtain the Co3O4 for batteries finally. The prepared Co3O4 powder has good flow property, even granularity and can utilize DCS system to adjust process parameters accurately to realize the control (D50=3um-6um,6um-9um,9um-12um), the tap density can reach 2.5g per cubic centimeter to 3.5g per cubic centimeter, microcosmic shape is single-crystal or like spherical, moreover the invention has the advantages of high chemical purity, good electrochemical performance, and no environmental pollution. The invention has simple process, stable product quality, low cost, and can adapt for the requirements of material performance and cost of various lithium cobalt oxide manufacturing factory.
Owner:ZHEJIANG HUAYOU COBALT
Cobalt-covered lithium ion cell anode material precursor as well as preparation method and application
InactiveCN103359795AImprove performanceImproved magnification performanceCell electrodesManganese oxides/hydroxidesPower batteryLithium
The invention relates to a cobalt-covered compound polybasic lithium ion cell anode material precursor as well a as preparation method and application. The precursor has the following formula: NixCoy+zMn1-x-y(OH)2, wherein x is more than 0 and less than 0.8, y is more than 0 and less than 0.5 and z is more than 0 and less than 0.05; the precursor is formed by a core part and a nano cobaltosic oxide layer covering the surface of the core; the molecular formula of the core part is as follows: NikConMn1-k-n(OH)2, wherein k is more than 0 and less than 0.8 ad n is more than 0 and less than 0.5. According to the precursor disclosed by the invention, the outer surface of the core part is covered with one layer of nano cobalt hydroxide through a nano technology; and a covering layer is formed by uniformly growing in a liquid phase so that the very good and dense covering layer is formed on a spherical surface. Then, a strong oxidant is added under a strong alkali environment so that the cobalt hydroxide is oxidized into cobalt hydroxyl cobalt oxide and a cobaltosic oxide covering layer is formed on the surface of the material in a following sintering process. The cobaltosic oxide has the very good electronic conduction capability so that the heavy load discharge performance of the material is greatly improved and the material can be more suitable for the requirements of a power battery.
Owner:协鑫动力新材料(盐城)有限公司
Graphite-phase nitrogen carbide nanosheet/cobaltosic oxide nanosheet composite nanomaterial of scale-shaped structure and preparation method and application thereof
ActiveCN106622324ALarge specific surface areaMany sitesPhysical/chemical process catalystsHigh heatNanomaterials
The invention aims at providing a method for synthesizing a scale-shaped graphite-phase nitrogen carbide nanosheet / cobaltosic oxide nanosheet composite material and belongs to the technical field of material preparation and catalysis. The composite material is prepared from the raw materials of functionalized graphite-phase nitrogen carbide and cobalt salt through a hydrothermal method at high temperature. The prepared scale-shaped two-dimensional nitrogen carbide nanosheet / cobaltosic oxide nanosheet composite material is in morphology that a scale-shaped cobaltosic oxide nanosheet nanocomposite material evenly grows on the surface of a two-dimensional nitrogen carbide nanosheet. Compared with a traditional pure two-dimensional nitrogen carbide nanomaterial, the specific surface area of the material and the screening capacity of the material to a catalytic substrate are effectively improved, and the material has more efficient photocatalytic performance. The preparation method has the advantages of simple technology, low cost and high catalytic efficiency; the prepared material has efficient carbon dioxide reducing capacity, and thus has good application prospect in the photocatalysis field of environmental modification, greenhouse gas elimination and the like.
Owner:FUJIAN MEDICAL UNIV
Process for preparing Co3O4
InactiveCN1376638ASimple processThe process is simple and easy to controlCobalt oxides/hydroxidesNitrateCobalt(II,III) oxide
A process for preparing Co3O4 includes such steps as dissolving Co in nitric acid, hydrochloric acid or sulfuric acid to generate cobolt nitrate (or sulfate, or chloride), dissolving precipitant and complexing agent in deionized water to prepare saturated solution, adding the prepared to solution to reactor, regulating pH=4-5.5, adding complexing precipitant at 35-100 deg.c while stirring, reacting, filtering, washing with deionized water at temp higher than 90 deg.C, calcining at 300-1000 deg.C for 5-25 hr, pulverizing and classifying.
Owner:戴振华
Cobalt oxide anode material, amorphous carbon coated cobalt oxide anode material and preparation method and application of cobalt oxide anode material and amorphous carbon coated cobalt oxide anode material
InactiveCN102659192ASimple preparation processPromote circulationCell electrodesCobalt oxides/hydroxidesHigh rateRetention ratio
The invention discloses a preparation method of a cobalt oxide anode material or an amorphous carbon coated cobalt oxide anode material, which comprises the following steps of: placing a conductive metal substrate in aqueous solution containing soluble cobalt salt and hexamethylene tetramine and carrying out heat preservation for 3h to 12h at a temperature of 80 DEG C to 150 DEG C to obtain the conductive metal substrate on which a cobalt hydroxide thin film is deposited; calcining the conductive metal substrate for 1h to 3h at a temperature of 200 DEG C to 400 DEG C to obtain the cobalt oxide anode material; and soaking the cobalt oxide anode material into aqueous solution of glucose, drying and calcining for 1h to 8h at a temperature of 300 DEG C to 500 DEG C to obtain the amorphous carbon coated cobalt oxide anode material. The preparation method has a simple preparation process and good reproducibility, is easy to implement, has an environmental-friendly production process and low cost and is beneficial to industrial production. The invention also provides the cobalt oxide anode material and the amorphous carbon coated cobalt oxide anode material. The cobalt oxide anode material and the amorphous carbon coated cobalt oxide anode material have high capacity retention ratios and good high-rate capabilities and are particularly suitable to use as a cathode electrode of a lithium ion battery.
Owner:ZHEJIANG UNIV
Preparation method of cobaltosic oxide with graded distribution of doping elements
InactiveCN107768646AAvoid the risk of excessive impuritiesWon't happenCell electrodesSecondary cellsCobalt(II,III) oxideCobalt salt
The invention discloses a preparation method of cobaltosic oxide with graded distribution of doping elements. The method comprises the steps of adopting a cobalt salt solution as a raw material, passing through a sodium hydroxide solution, adding a certain proportion of ammonium hydroxide, calculating the amount of a doping element soluble salt solution by volume and a prepared hydrogen peroxide solution, carrying out synthesis reaction, filtering, washing, and drying to obtain a final product. According to the preparation method of the cobaltosic oxide with graded distribution of the doping elements provided by the invention, along with the extending of synthesis reaction time and the step-increasing of the amounts of a doping agent in a reaction still, the doping elements are in graded distribution in a cobaltosic oxide product, and the cobaltosic oxide product with graded distribution of the doping elements is prepared for the first time.
Owner:LANZHOU JINCHUAN NEW MATERIAL SCI & TECH
Preparation method of cobalt oxyhydroxide
The invention discloses a preparation method of cobalt oxyhydroxide. In the method, a cobalt salt solution is used as a raw material; precipitation and oxidation reaction are performed at the same time in an alkaline solution in the presence of an oxidant; and the precipitate is subjected to washing, impurity removal, drying, granulation, mixing and screening to obtain cobalt oxyhydroxide of which D50 is 10-20 microns. Compared with lithium cobaltate prepared by taking cobaltosic oxide as the raw material traditionally, the compaction density of the lithium cobaltate prepared from cobalt oxyhydroxide disclosed by the invention can be obviously improved by 0.2-0.3g / cm<3>, and the capacity and cycling performance are also obviously improved. Meanwhile, compared with the traditional preparation technology of the battery positive material raw material cobaltosic oxide, the technology disclosed by the invention is low in cost and low in energy consumption, is more environment-friendly and realizes economic and social benefits.
Owner:GUANGDONG BRUNP RECYCLING TECH
Cobaltosic oxide hierarchical structure nano array materials and preparation method thereof, and application of cobaltosic oxide hierarchical structure nano array materials
ActiveCN108346522AHigh purityGood dispersionMaterial nanotechnologyHybrid capacitor electrodesNanowireDecomposition
The present invention provides cobaltosic oxide hierarchical structure nano array materials and a preparation method thereof, and an application of cobaltosic oxide hierarchical structure nano array materials. In a closed high temperature and high pressure reactor, redistilled water is taken as reaction solvent, cobalt salt, ammonium fluoride and urea are added into the reaction solvent for uniform mixing, and a reaction system is heated to generate a high-pressure environment to prepare cobaltous hydroxide precursor nanowire materials; then redistilled water is taken as reaction solvent, 2-methylimidazole is added into the reaction solvent, the cobaltous hydroxide precursor loaded on carbon cloth is immersed in the solution, the cobaltous hydroxide is taken as a cobalt source, the reaction system is heated and the reaction time is controlled to control a ZIF67 nucleation rate so as to prepare a cobalt-based metal organic framework (ZIF67) array; and finally, calcining decomposition ofthe cobalt-based metal organic framework (ZIF67) array is performed to obtain a cobaltosic oxide hierarchical structure. The product purity is high, the dispersibility is good, the controllability isgood, the production cost is low, the reproducibility is good, the cobaltosic oxide hierarchical structure nano array materials have cycling stability and large active surface area, and have a potential application value at the aspect of supercapacitors.
Owner:ANHUI NORMAL UNIV
Preparation method of high-voltage lithium cobalt oxide cathode material
ActiveCN109326781AEvenly distributedWell mixedSecondary cellsPositive electrodesDual effectCobalt(II,III) oxide
The invention discloses a preparation method of a high-voltage lithium cobalt oxide cathode material which comprises the steps: (1) mixing cobaltosic oxide containing doped element M, a lithium source, oxide containing doped element M', a grain refiner and a fluxing agent to obtain a primary mixture; (2) sintering and smashing the primary mixture to obtain primary sintered powder; (3) mixing the primary sintered powder with a coating material to obtain a secondary mixture; (4) sintering and smashing the secondary mixture to obtain the lithium cobalt oxide cathode material. According to the preparation method disclosed by the invention, the problem of microscale segregation of distribution of doped elements in the product is solved by two modes of doped element premixing and dry-method mixing; a coulter type mixing technology is utilized to solve the problem of segregation of different matters in material mixing; the grain refiner and the fluxing agent are combined for use to synthesizea material in a mixed morphology; technologies such as dual effects of doped element surface crystallization catalysis and the coating material and the like are utilized; thus, performance of the high-voltage lithium cobalt oxide is obviously improved.
Owner:HUNAN CHANGYUAN LICO CO LTD
Method for preparing cobaltic-cobaltous oxide powder with octahedron shape
ActiveCN101293677ACrystallization intactMeet the requirements of lithium cobalt oxide for high-performance lithium-ion batteriesCobalt oxides/hydroxidesApparent densityOctahedron
The invention provides a preparation method of an electronic grade cobaltosic oxide powder which is shaped like an octahedron.The cobaltosic oxide powder has the complete appearance of the octahedron, the middle diameter (D50) of the powder is 0.5 to 5 Mum (the value can be adjusted by changing process parameters), the apparent density is more than 0.7g / cm<3>, and the tap density is more than 2.0g / cm<3>. The technological process is characterized in that the soluble cobalt salt solution, such as cobalt chloride and cobalt acetate, and ammonium bicarbonate solution are strongly stirred when being added to a reactor in the form of cocurrent flow at a definite speed, and the reaction temperature and PH value are strictly controlled, so the crystallization process is controlled. The obtained cobalt carbonate sediment is dried at a certain temperature after washing and filtration, and finally calcined at a certain temperature to get the ultimate cobaltosic oxide powder in the shape of an octahedron. The preparation method has the advantages that the cobaltosic oxide powder with the complete crystal form and in the form of the octahedron is prepared at atmospheric pressure, the specific surface area is high and the activity is high, etc., and besides the technique is simple, reliable and easy to industrialize.
Owner:GRIPM ADVANCED MATERIALS CO LTD
In-situ grown three-dimensional multi-structural cobaltosic oxide/carbon composite micro-nanomaterial and controllable preparation method thereof
The invention relates to an in-situ grown three-dimensional multi-structural cobaltosic oxide / carbon composite micro-nanomaterial and its controllable preparation method. A cobalt material, ammonium fluoride and urea are used as raw materials to prepare a cobaltosic oxide micro-nanomaterial with multiple specific-morphologies three-dimensional multi-structures in situ; and the cobaltosic oxide micro-nanomaterial is processed with a carbon-rich solution to obtain a corresponding three-dimensional multi-structural carbon-coated cobaltosic oxide composite structure. The preparation method provided by the invention is simple, has strong operationality, is low-cost and is green and environmentally friendly. Problems of tedious steps, poor combinability between a nano-powder and a substrate, poor repeatability, poor electron-transporting property and the like when the traditional nano-powder is used in a device are solved. The three-dimensional multi-structures and specific morphology of cobaltosic oxide can be effectively maintained. The obtained product has diversified morphologies, is uniform in size, is not easy to agglomerate and has high purity. The three-dimensional pores and multi-structures of the product are beneficial to electron-transport. The product will be hopefully and widely applied in fields of supercapacitor, lithium ion battery, catalysis, magnetic material, sensor, photoelectricity and the like.
Owner:INST OF PROCESS ENG CHINESE ACAD OF SCI
Spherical tricobalt tetraoxide and method of preparing the same
ActiveUS20100135897A1Stable structureMaintain good propertiesIron oxides/hydroxidesCell electrodesCobalt(II,III) oxideHigh activity
A method of preparation of spherical tricobalt tetraoxide, including at least oxidizing a bivalent cobalt salt in a wet environment and in the presence of a precipitant, a complexing agent, and an oxidant to yield spherical cobalt oxyhydroxide.cobalt hydroxide according to the following equation Co2++3OH−+O→CoOOH.Co(OH)2; oxidizing the spherical hydroxy cobalt oxyhydroxide.cobalt hydroxide to yield spherical tricobalt tetraoxide according to the following equation 6 CoOOH.Co(OH)2+O→4 Co3O4+9 H2O; and roasting the spherical tricobalt tetraoxide at low or intermediate temperature to yield a black powder. The method is easily practiced and suitable for mass production, and the resultant spherical tricobalt tetraoxide has stable structure, reliable properties, and high activity.
Owner:NINGBO RONBAY LITHIUM BATTERY MATERIAL CO LTD
Method for preparing metal oxide-graphene nanocomposite and method for preparing electrode using metal oxide-graphene nanocomposite
InactiveUS20160218353A1Reduce processing stepsReduce processing costsSpecific nanostructure formationHybrid capacitor electrodesGraphene nanocompositesGraphene flake
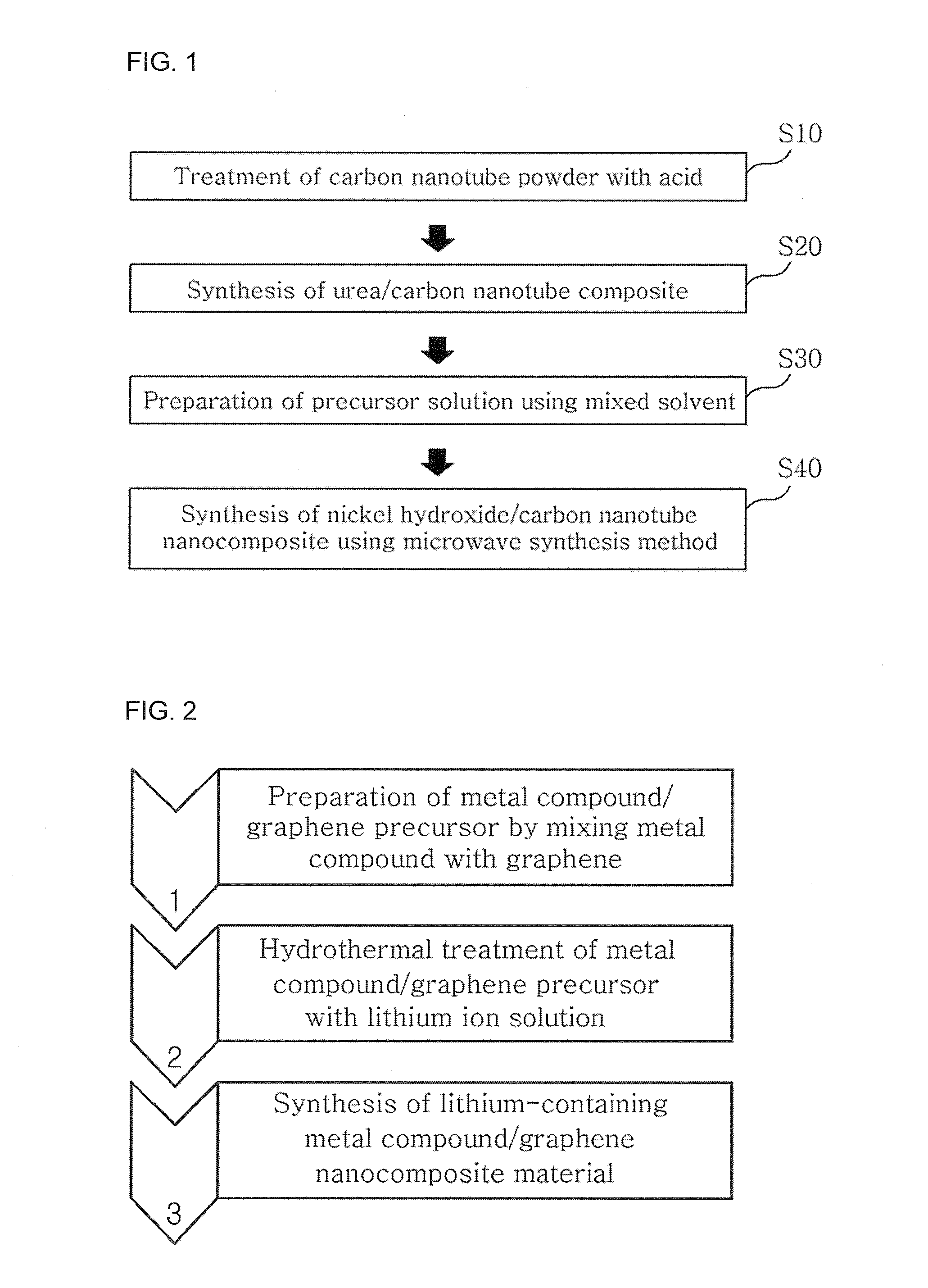
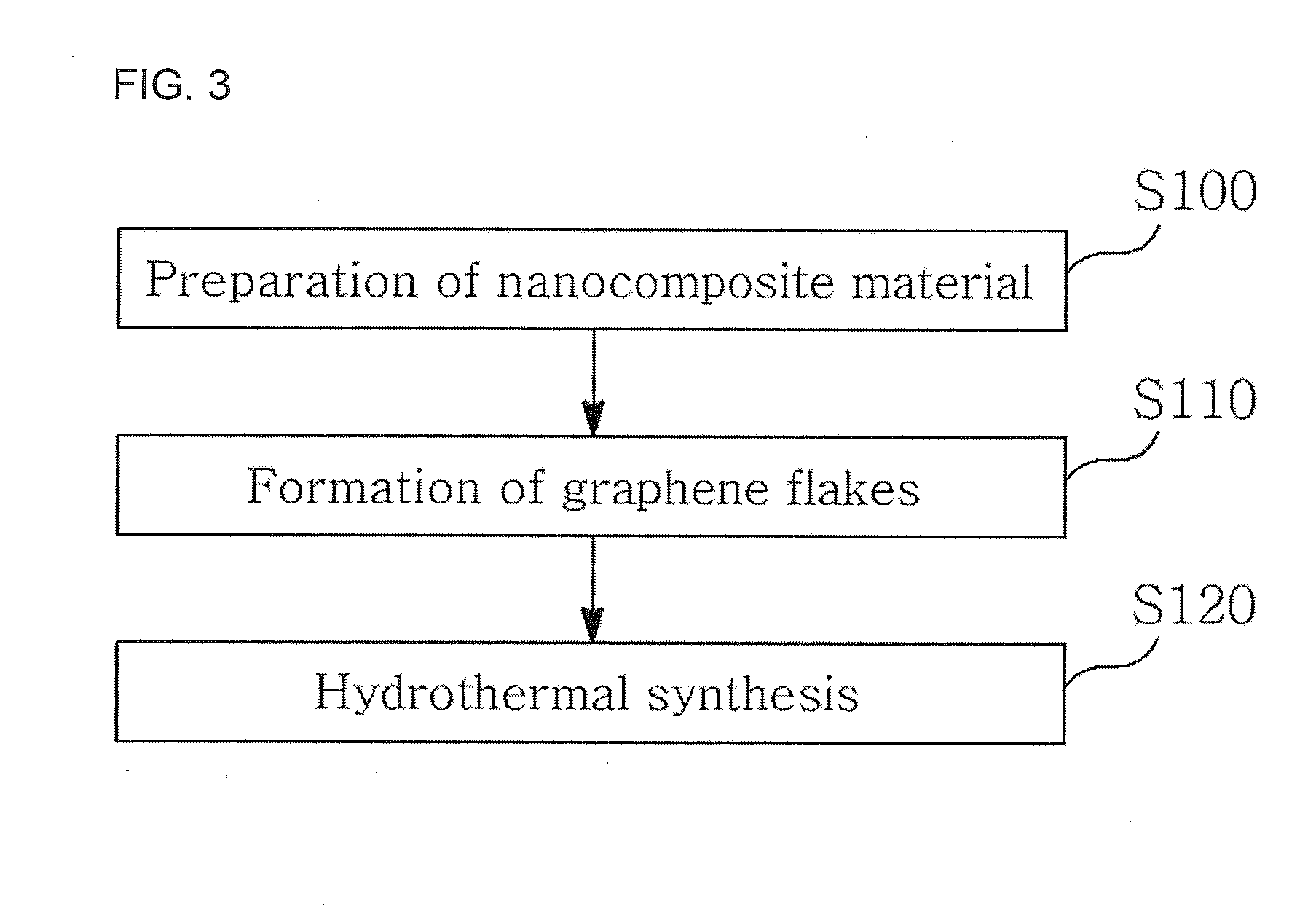
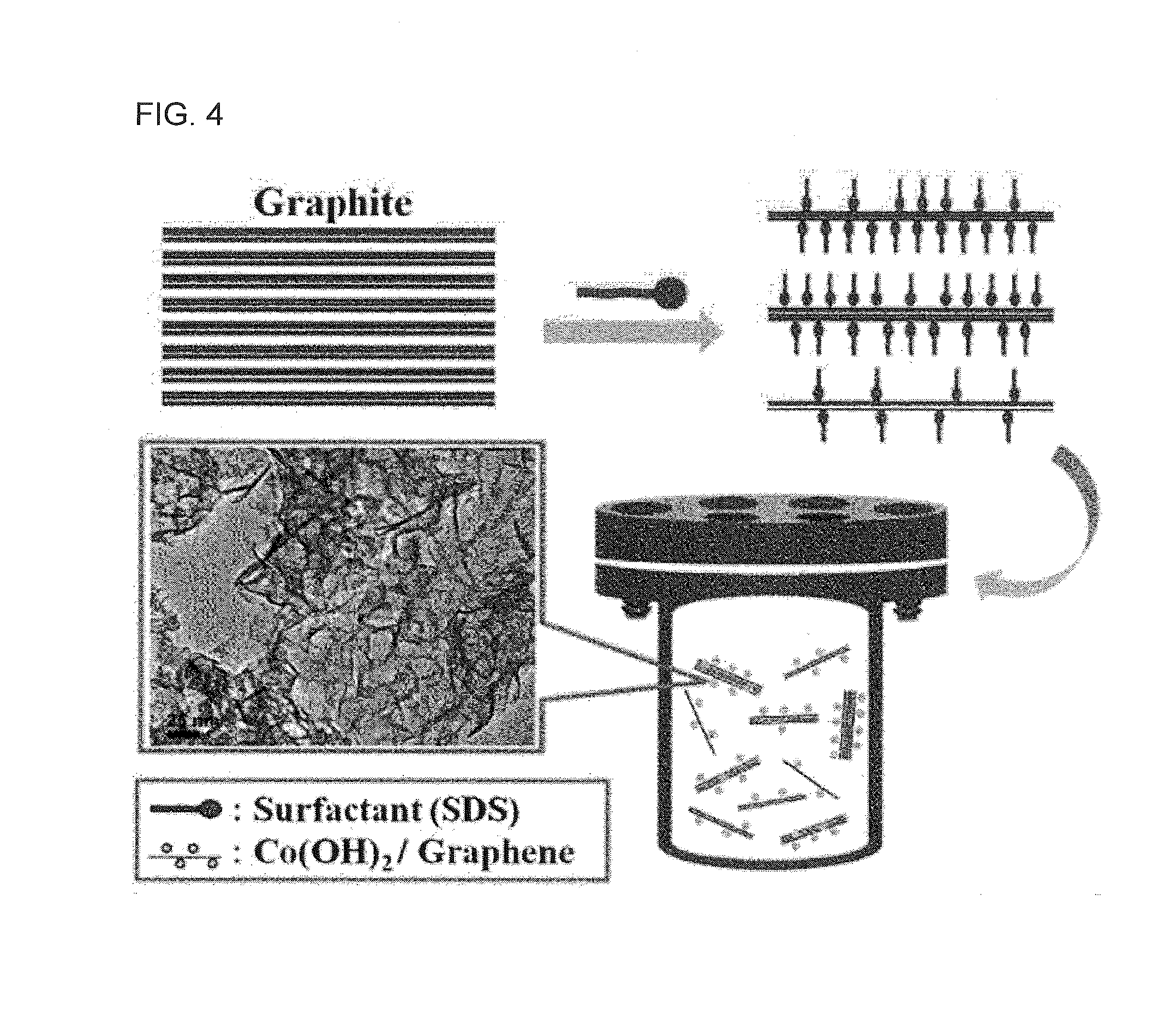
Disclosed is a method of preparing a metal oxide-graphene nanocomposite, including preparing a nanocomposite material, forming graphene flakes by pretreating the nanocomposite material, and hydrothermally synthesizing the pretreated nanocomposite material. A method of manufacturing an electrode using the metal oxide-graphene nanocomposite is also provided. According to this invention, the metal oxide-graphene nanocomposite is synthesized from inexpensive graphite through one-step processing using only a surfactant, in place of conventional methods using oxidants, reductants and high-temperature heat, thereby lowering the number of processing steps and processing costs. Also, in the fabrication of the electrode, low electrical resistance characteristic of graphene is applied as it is, in place of the conventional use of active material, conductive material and binder, thereby exhibiting desired processing efficiency without the addition of the conductive material. Furthermore, highly pure graphene is prepared in a short time and various metal oxide active materials suitable for use in energy storage devices, for example, unary, binary, and multicomponent metal oxides, is formed through one-step processing, and necessary oxides having desired weight ratios {cobalt oxide (CoO), tricobalt tetraoxide (Co3O4), and cobalt hydroxide [Co(OH)2]} can be easily prepared, and thus very wide application ranges (secondary batteries, gas sensors, etc.) are expected.
Owner:AJOU UNIV IND ACADEMIC COOP FOUND
Preparation of metal organic framework membrane and application in gas separation
InactiveCN105126642AImprove mechanical propertiesSemi-permeable membranesHydrogen separation using solid contactNanowireNickel substrate
A preparation of metal organic framework membrane and an application in gas separation. The invention belongs to the technical field of membrane separation. The preparation method includes the steps of (A) adding a treated porous metal nickel substrate into a precursor solution of Co3O4 and heating the solution at a high temperature; (B) rinsing and drying the nickel substrate and heating the nickel substrate under protection of inert gas at a high temperature to obtain the nickel substrate modified with Co3O4 nano wires; and (C) preparing a MOF mother liquid and vertically placing the modified nickel substrate thereinto, so that an organic metal framework compound (MOF), which is generated through coordination effect between an organic ligand and metal ions, is grown on the nickel substrate modified with the Co3O4 nano wires uniformly, thereby preparing a continuous compact MOF membrane. The invention provides a new preparation method of the metal organic framework membrane, wherein the method is simple in process. The MOF membrane is available in gas separation.
Owner:BEIJING UNIV OF TECH
Preparation method for lithium cobalt oxide anode material
ActiveCN103746114ALarge powder particlesEvenly distributedCell electrodesCobalt compoundsCobalt(II,III) oxideCobalt salt
A preparation method for a lithium cobalt oxide anode material relates to ananode material for lithium ion secondary batteries. The method comprises thefollowing steps: preparing a cobalt salt solution and a precipitator solution, precipitating a cobalt carbonate precursor by adopting a controlled crystallization process, and sintering cobalt carbonate to obtain cobaltosic oxide, wherein the cobalt carbonate precursor requires that the median diameter D50 ranges from 13-20 mum and the consistency is greater than or equal to 0.40 and smaller than or equal 0.50; mixing and then sintering the cobaltosic oxide, an additive and a lithium compound, milling and screening the product of sintering to obtain a lithium cobalt oxide semi-finished product; mixing thelithium compound and a coating element M, adding the mixture of the lithium compound and the coating element M and the lithium cobalt oxide semi-finished product into a ball mill for mixing, sintering the mixed materials, and milling and screening the product of sintering to obtain the lithium cobalt oxide anode material. The prepared lithium cobalt oxide product has the advantages that the size distribution of particles is uniform, and the product compaction densitycan be remarkably improved.
Owner:XTC NEW ENERGY MATERIALS(XIAMEN) LTD +1
High-temperature type lithium manganate anode material for power lithium ion battery and preparation method of high-temperature type lithium manganate anode material
ActiveCN104485452AImprove high temperature resistanceExcellent discharge specific capacityCell electrodesCobalt(II,III) oxideManganate
The invention discloses a high-temperature type lithium manganate anode material for a power lithium ion battery. The high-temperature type lithium manganate anode material consists of a core material as shown in a formula Li1+xMn2-y-zAyQzO4, and a coating layer on the surface of the core material, wherein the coating layer is one or more of cobaltosic oxide, aluminum oxide and nickel protoxide. The anode material is excellent in high-temperature property, and is good in specific discharge capacity, capacity retention ratio and electrochemical circulation property at high temperature. In addition, the preparation method of the anode material disclosed by the invention is simple in production process, easy to achieve and low in cost, and can be applied to large-scale industrialization production.
Owner:北京盟固利新材料科技有限公司
Cobaltosic oxide multi-shell hollow sphere cathode material for lithium ion battery and preparation method thereof
ActiveCN103247777AHigh specific surface areaImprove cycle performanceCell electrodesCell component detailsCapacitanceSodium-ion battery
The invention provides a cobaltosic oxide multi-shell hollow sphere cathode material for a lithium ion battery and a preparation method thereof. The method comprises the steps of: taking a carbon sphere prepared by a hydrothermal method as a template; controlling the quantity of cobalt ions in the carbon sphere and the entering depth thereof by controlling the ratio of water to ethanol in a cobalt salt solution, the temperature of the solution and the adsorption capacity of the carbon sphere; and preparing single-shell, dual-shell, tri-shell and four-shell cobaltosic oxide hollow spheres. The lithium ion battery of taking the cobaltosic oxide multi-shell hollow sphere as a cathode has large specific surface area and a plurality of lithium ion storage sites; the specific capacitance is improved; meanwhile, the appropriate multi-shell cavity structure not only can adjust the electrode structure and volume change, but also can effectively reduce the transmission distance between the lithium ion and electrons. Thus, the cycling performance and rapid charge and discharge capacity are obviously improved. The method disclosed by the invention is convenient and concise to operate, and high in controllability, and has a wide application prospect; and the performance of the lithium ion battery can be obviously improved.
Owner:INST OF PROCESS ENG CHINESE ACAD OF SCI
Single-cell-thickness nano porous cobalt oxide nanosheet array electrocatalytic material
InactiveCN106025302AEnsure continuous and stable workExcellent hydrogen evolution reaction performanceMaterial nanotechnologySpecific nanostructure formationDecompositionOxygen
A single-cell-thickness nano porous cobalt oxide nanosheet array electrocatalytic material is characterized in that a metal-doped cobalt oxide primary nanosheet array is perpendicularly grown on a conductive substrate, a porous nanosheet is obtained from each primary nanosheet, and the nanosheets are of porous structure; the material is used as an electrocatalyst for oxygen evolution reaction; the material also has excellent hydrogen evolution performance and may function as a bifunctional catalyst for an alkaline full-decomposition water system. The invention has the advantages that the material can effectively reduce overpotential and peaking potential of oxygen evolution reaction, increase conversion rate of single cobalt atoms and operate stably and continuously in a strong alkali environment; the material has excellent oxygen evolution reaction performance and can be applied as an anode and cathode of a full-decomposition water system, effectively reducing trough voltage; the material is simple to prepare, convenient to operate, low in cost and environment-friendly, and new idea and strategy are provided for the guide design and performance optimization of the bifunctional catalyst for the full-decomposition water system.
Owner:TIANJIN UNIVERSITY OF TECHNOLOGY
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com