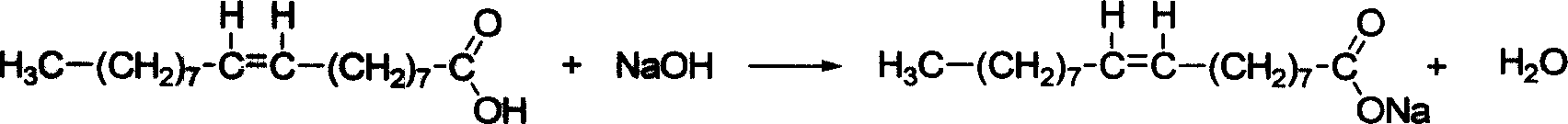Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
6633 results about "Oleic Acid Triglyceride" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Instead fatty acids like oleic acid occur as their esters, commonly triglycerides, which are the greasy materials in many natural oils. Fatty acids can be obtained by the saponification of triglycerides. Triglycerides of oleic acid comprise the majority of olive oil. Free oleic acid render olive oil inedible.
Methods for producing plants with elevated oleic acid content
By this invention, methods to produce oleic fatty acids in plant seed oils are provided. The methods of the present invention generally involve the suppression of a host plant cells endogenous beta-ketoacyl-ACP synthase I protein. Also described in the instant invention are the plants, cells and oils obtained therefrom.
Owner:CALGENE LLC +1
Oil of Brassica napus
InactiveUS6169190AOrganic compound preparationFatty substance preservation using additivesBrassicaAntioxidant
Oil produced from the seed of Brassica napus plant has an oleic acid content of 71.4-77.4% and a linoleic acid content of no more than 3%. The oil is useful in food and industrial applications. The oil has substantially improved oxidative stability relative to normal rapeseed oil, when both are identically treated with antioxidant.
Owner:LANUZA JUAN ENRIQUE ROMERO +1
Delta12 desaturases suitable for altering levels of polyunsaturated fatty acids in oleaginous yeast
The present invention relates to fungal Δ12 fatty acid desaturases that are able to catalyze the conversion of oleic acid to linoleic acid (LA; 18:2). Nucleic acid sequences encoding the desaturases, nucleic acid sequences which hybridize thereto, DNA constructs comprising the desaturase genes, and recombinant host microorganisms expressing increased levels of the desaturases are described. Methods of increasing production of specific ω-3 and ω-6 fatty acids by over-expression of the Δ12 fatty acid desaturases are also described herein.
Owner:EI DU PONT DE NEMOURS & CO
Corn plants and products with improved oil composition
InactiveUS6248939B1Increase contentHigh oleic acid producing characteristicsMutant preparationAnimal feeding stuffChemical compositionComponents of crude oil
This invention relates to corn (Zea mays L.) seed and grain having a significantly higher oleic acid content than conventional corn by virtue of heritable genes for increased oil and oleic acid content and / or lowered levels of linoleic acid. The present invention also relates to the production of high oil, high oleic grain, its oil, its progeny and its use.
Owner:EI DU PONT DE NEMOURS & CO
Nutritional intervention composition containing a source of proteinase inhibitor extending post meal satiety
InactiveUS6838431B2Effective quantityBiocidePeptide/protein ingredientsSolanum tuberosumOleic Acid Triglyceride
The invention relates to a nutritional intervention composition in powder form that is taken before a meal and that extends post meal satiety. The compositions of the invention are comprised of at least one protein, C12-18 fatty acids, preferably oleic acid, all of which stimulate CCK release in the body, and a source of proteinase inhibitor which acts to prevent the deactivation of CCK. The subject compositions preferably additionally contain a source of calcium and are advantageous in that they utilize a source of proteinase inhibitor as opposed to the more costly extracts. The source of proteinase inhibitor is advantageously an extract of potato, soy, or beans containing about 10 weight percent of proteinase inhibitor. The powder compositions are mixed with a suitable liquid, preferably water, prior to ingestion.
Owner:ANAYA PERSONAL CARE PROD LLC +1
Methods for increasing oleic acid content in seeds from transgenic plants containing a mutant delta 12 desaturase
InactiveUS7109392B1Sugar derivativesOther foreign material introduction processesLipid compositionOleic Acid Triglyceride
The preparation and use of nucleic acid fragments encoding fatty acid desaturase enzymes are described. The invention permits alteration of plant lipid composition. Chimeric genes incorporating mutant delta-12 fatty acid desaturase nucleic acid fragments with suitable regulatory sequences may be used to create transgenic plants with altered levels of unsaturated fatty acids.
Owner:CARGILL INC
Fatty acid desaturases from fungi
ActiveUS20060156435A1High nutritional valueIncrease SDA contentSugar derivativesOther foreign material introduction processesBiotechnologyFungal microorganisms
The invention relates generally to methods and compositions concerning fungal desaturase enzymes that modulate the number and location of double bonds in long chain poly-unsaturated fatty acids (LC-PUFA's). In Particular, the invention relates to methods and compositions for improving omega-3 fatty acid profiles in plant products and parts using desaturase enzymes and nucleic acids encoding for such enzymes. In particular embodiments, the desaturase enzymes are fungal −15 desaturases. Also provided are improved canola oil compositions having SDA and maintaining beneficial oleic acid content.
Owner:ADEKA CORP +1
Mixed micellar delivery system and method of preparation
InactiveUS6221378B1Antibacterial agentsOrganic active ingredientsOctylphenoxy PolyethoxyethanolChamomile extract
A mixed micellar pharmaceutical formulation includes a micellar proteinic pharmaceutical agent, an lkali metal C8 to C22 alkyl sulphate, alkali metal salicylate, a pharmaceutically acceptable edetate and at least one absorption enhancing compounds. The absorption enhancing compounds are selected from the group consisting of lecithin, hyaluronic acid, pharmaceutically acceptable salts of hyaluronic acid, octylphenoxypolyethoxyethanol, glycolic acid, lactic acid, chamomile extract, cucumber extract, oleic acid, linolenic acid, borage oil, evening of primrose oil, trihydroxy oxo cholanylglycine, glycerin, polyglycerin, lysine, polylysine, triolein and mixtures thereof. The amount of each absorption enhancing compound is present in a concentration of from 1 to 10 wt. / wt. % of the total formulation, and the total concentration of absorption enhancing compounds are less than 50 wt. / wt. % of the formulation. Preferably, the formulation is administered, in combination with a propellant, to the buccal cavity, using a metered dose dispenser.
Owner:GENEREX PHARMA INC
Edible nourishing oil with balanced fatty acid rate
The invention relates to edible nutritious oil which is characterized in that the contents of saturated fatty acid, oleic acid, linoleic acid and linolenic acid are respectively 8 to 16 percent, 42 to 56 percent, 13 to 23 percent and 13 to 30 percent of total fatty acid. With the proposal of eating less saturated fatty acid and linoleic acid and more oleic acid and linolenic acid from the international alimentology community, the edible oil is designed based on the particular reference that the ratio of the linoleic acid to the linolenic acid is close to 1 to 1 in the meal of human beings and people in Crete, Greece 100 years ago. The edible oil which has more balanced ratio between the fatty acid is quite suitable for human beings and has very important effects on reducing human beings fatness, hypertension, high blood fat, cardiovascular and cranial vascular diseases and cancers and reinforcing intelligence development. In addition, the edible oil has high quality and low price.
Owner:罗红宇
Method for synthesizing amphoteric slow-breaking quick-setting asphalt emulsifier
InactiveCN101712625AWide selectionWide adaptabilityOrganic compound preparationTransportation and packagingChemical synthesisSolubility
The invention discloses a method for synthesizing a novel amphoteric slow-breaking quick-setting asphalt emulsifier, belonging to the technical field of organic chemical synthesis. The method comprises the following steps of: reacting oleic acid with polyamine to generate acid amide polyamine, and then adding chloroactic acid to generate halogenating reaction to prepare the asphalt emulsifier, wherein the polyamine is the mixture of diethylenetriamine and triethylene tetramine. The water solubility of the hydrophilic radical of the emulsifier synthesized by the process is stronger than those of like emulsifiers, thereby obviously lowering the critical micelle concentration (CMC) value, and the particles of the produced asphalt emulsion are finer, smoother and evener. The emulsifier can effectively prevent asphalt from settling, increases the storage stability of the asphalt emulsion, reasonably regulates the charge ratio, widens the selectivity on stone materials in the application of slurry seal construction, enables the mixing time to be sufficient, and has obvious quick-setting effect and improved project quality.
Owner:河南新友公路技术有限公司 +1
Drug-oligomer conjugates with polyethylene glycol components
InactiveUS7030084B2Reduce deliveryExtended durationPeptide/protein ingredientsDepsipeptidesOligomerPolyethylene glycol
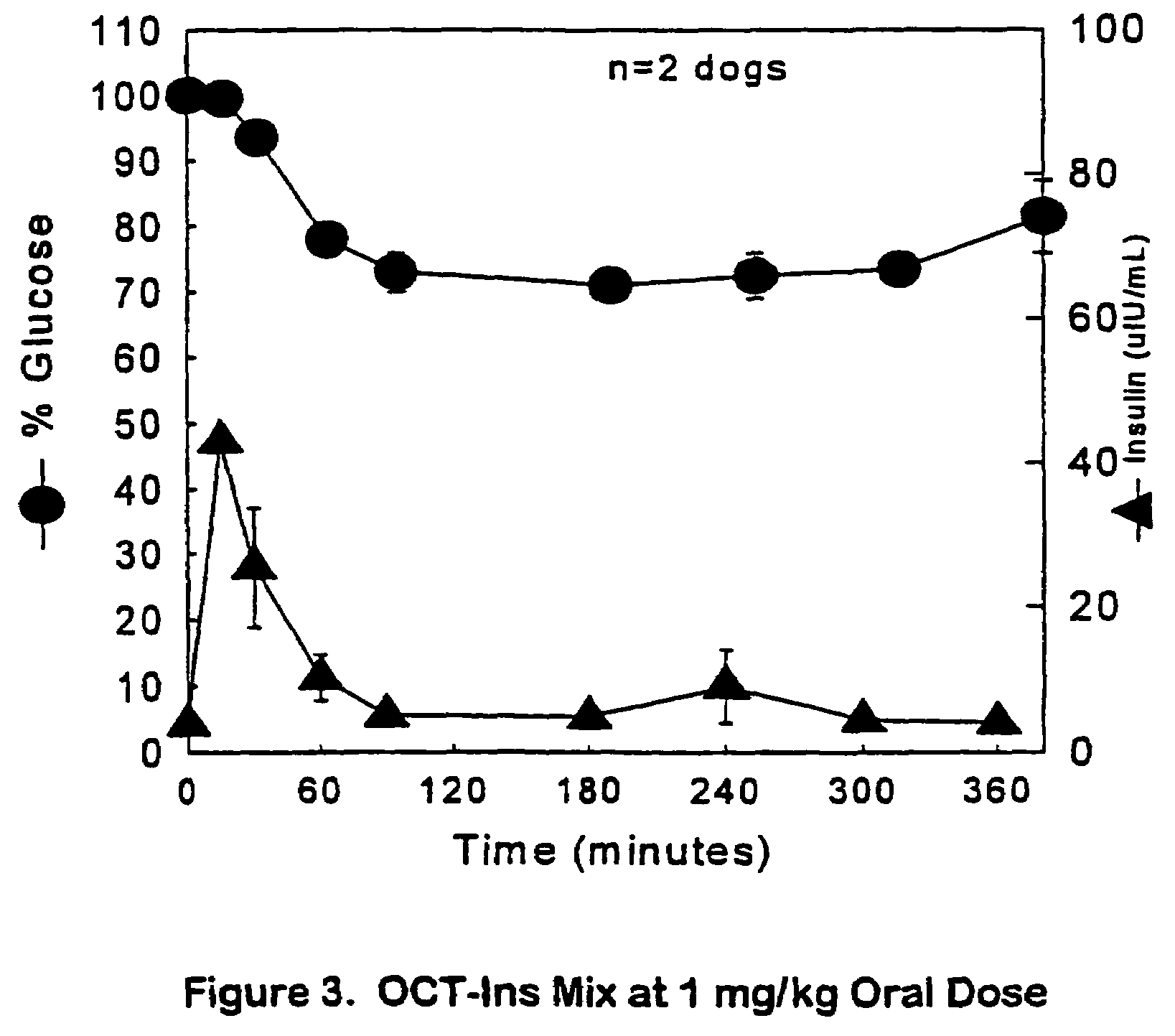
The present invention relates generally to hydrolyzable drug-oligomer conjugates, pharmaceutical compositions comprising such conjugates, and to methods for making and using such conjugates and pharmaceutical compositions. For example, a conjugate of insulin, PEG, and oleic acid can be orally administered.
Owner:BIOCON LTD
High-yield preparing method for inorganic halogen perovskite fluorescent quantum dots at room temperature
ActiveCN105331362AImprove performanceScalable productionMaterial nanotechnologyNanoopticsFluorescenceShielding gas
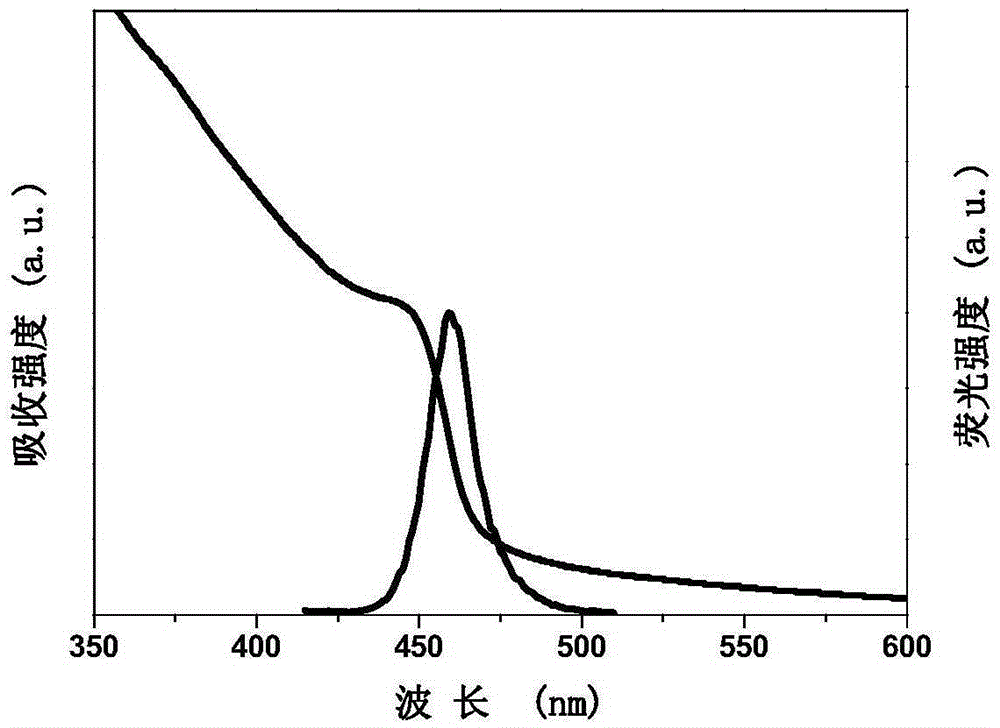
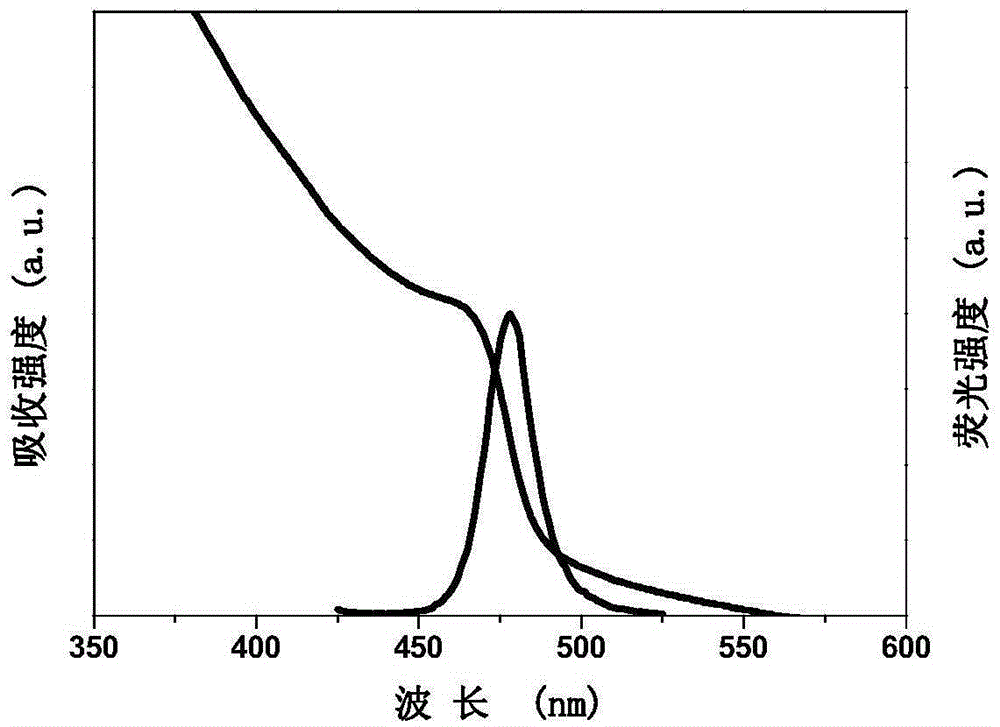
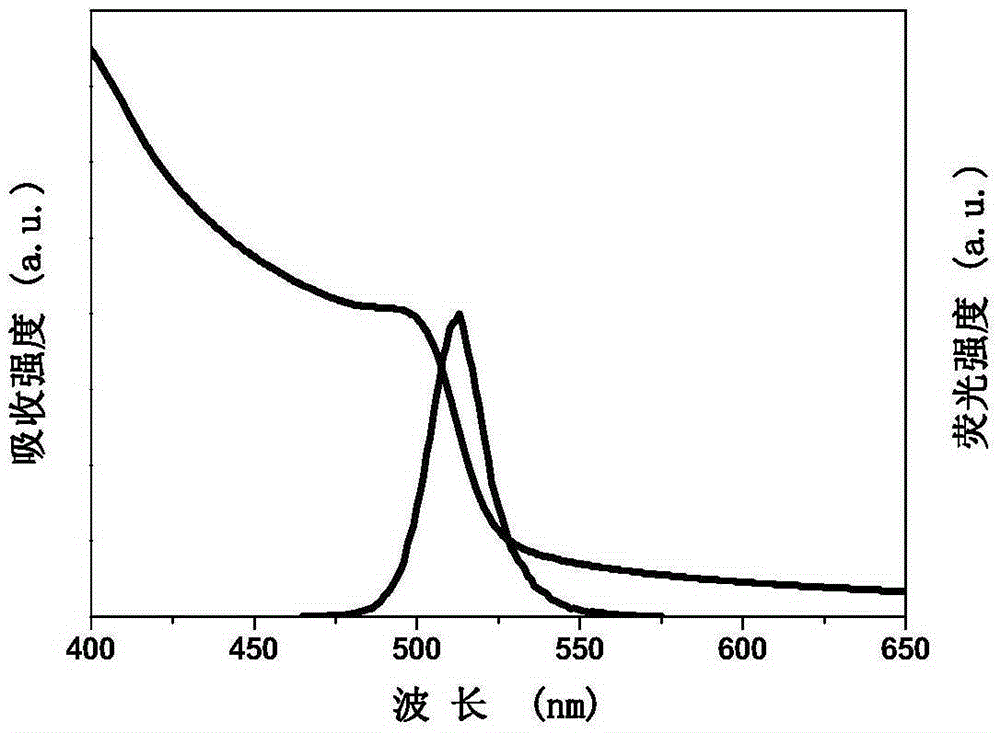
The invention discloses a high-yield preparing method for inorganic halogen perovskite fluorescent quantum dots at the room temperature. The fluorescent quantum dots are CsPbX3, wherein X is equal to AxB1-x and is larger than or equal to 0 and smaller than or equal to 1, and A and B are any one of Cl, Br and I. The method comprises the following steps that firstly, lead halide and cesium halide are dissolved into dimethyl formamide, surfactant oleylamine and oleic acid are added, the mixture is stirred until complete dissolution, and a precursor solution is obtained; secondly, the precursor solution is dripped into a poor solvent at the speed of 0.08-0.13 mL / s and stirred evenly at the uniform speed, and the inorganic halogen perovskite fluorescent quantum dots CsPbX3 are obtained. The preparing method is implemented at the room temperature, protection gas is not needed, equipment is simple, mass production can be achieved, and full visible light band shining can be achieved by selecting halogen and adjusting the proportion of halogen. The full width at half maximum of the inorganic halogen perovskite fluorescent quantum dots prepared through the preparing method ranges from 16 nm to 39 nm, the fluorescence quantum efficiency is close to 90%, and the inorganic halogen perovskite fluorescent quantum dots can be stably stored for more than three months, and can be used in the field of solar cells, lasers, light detectors, light-emitting diodes and the like.
Owner:NANJING UNIV OF SCI & TECH
Aerosol formulations for buccal and pulmonary application
InactiveUS6436367B1Antibacterial agentsOrganic active ingredientsChamomile extractOleic Acid Triglyceride
A mixed micellar aerosol pharmaceutical formulation includes a micellar proteinic pharmaceutical agent, an alkali metal lauryl sulphate, at least three micelle forming compounds, a phenol and a propellant. The micelle forming compounds are selected from the group consisting of lecithin, hyaluronic acid, pharmaceutically acceptable salts of hyaluronic acid, glycolic acid, lactic acid, chamomile extract, cucumber extract, oleic acid, linoleic acid, linolenic acid, monoolein, monooleates, monolaurates, borage oil, evening of primrose oil, menthol, trihydroxy oxo cholanyl glycine and pharmaceutically acceptable salts thereof, glycerin, polyglycerin, lysine, polylysine, triolein, polyoxyethylene ethers and analogues thereof, polidocanol alkyl ethers and analogues thereof. The amount of each micelle forming compound is present in a concentration of from 1 to 20 wt. / wt. % of the total formulation, and the total concentration of micelle forming compounds are less than 50 wt. / wt. % of the formulation. The propellant, e.g. a fluorocarbon propellant, provides enhanced absorption of the pharmaceutical agent.
Owner:GENEREX PHARMA
Delta-12 desaturase gene suitable for altering levels of polyunsaturated fatty acids in oleaginous yeasts
The present invention relates to a Δ12 fatty acid desaturase able to catalyze the conversion of oleic acid to linoleic acid (LA; 18:2). Nucleic acid sequences encoding the desaturase, nucleic acid sequences that hybridize thereto, DNA constructs comprising the desaturase gene, and recombinant host microorganisms expressing increased levels of the desaturase are described. Methods of increasing production of specific ω-3 and / or ω-6 fatty acids are described by overexpression of the Δ12 fatty acid desaturase or by disruption of the native gene.
Owner:DUPONT US HLDG LLC
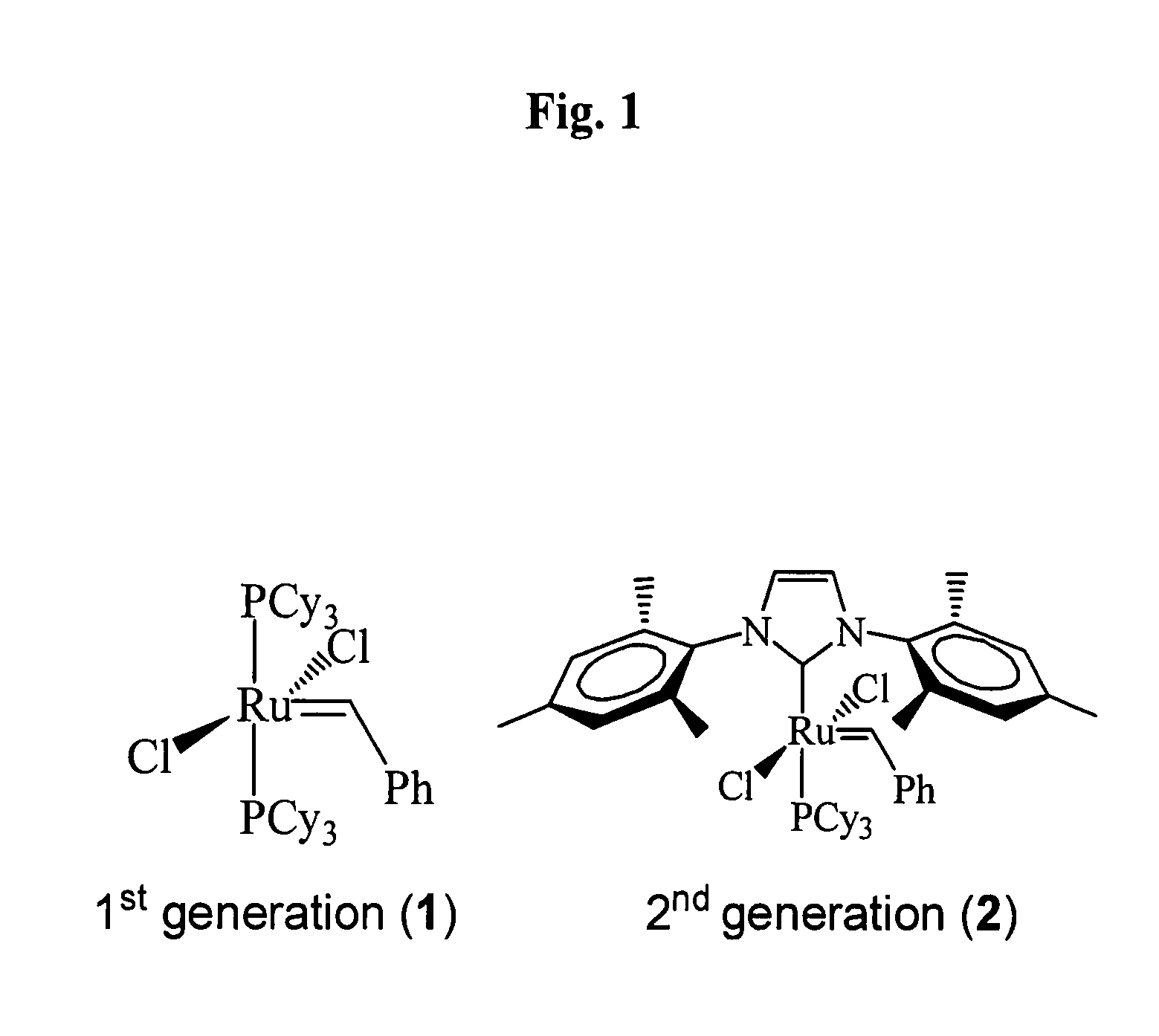
Catalytic compositions for the metathesis of unsaturated fatty bodies with olefins and metathesis methods using catalytic compositions
InactiveUS20100022789A1High conversion levelSignificant selectivityFatty acid chemical modificationOrganic compound preparationPtru catalystUnsaturated fat
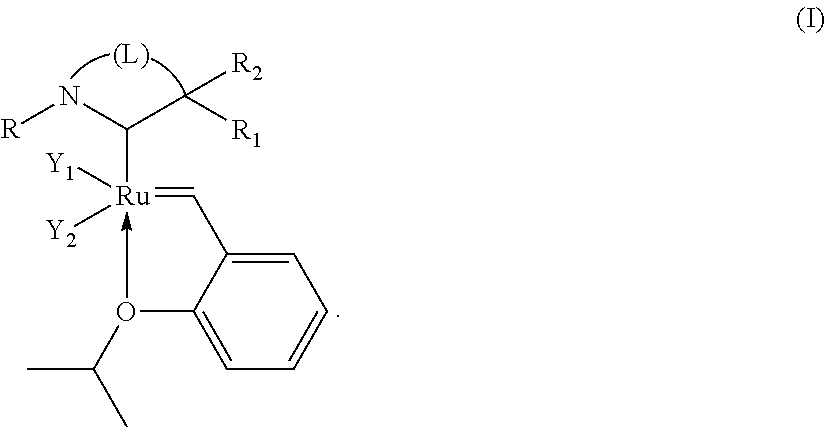
Catalytic compositions useful for carrying out the metathesis of unsaturated fatty bodies, in particular unsaturated fatty bodies comprising oleic acid or oleic acid esters, contain a ruthenium-based catalyst which can be isolated and which has the formula (I) below:
Owner:RHODIA OPERATIONS SAS +1
Rubber composition for tire tread
The present invention relates to a cross-linkable or cross-linked rubber composition usable in a tread of a tire, and to a tire incorporating this tread. A rubber composition according to the invention is based on at least one diene elastomer, and a plasticizer comprising a glycerol oleic acid triester, said plasticizer comprises:one or more synthetic and / or natural compounds not extracted from petroleum in a mass fraction of from 45% to 100%, said compounds comprising at least one glycerol fatty acid triester, and the fatty acid(s) as a whole comprise oleic acid in a mass fraction equal to or greater than 60%, andone or more paraffinic, aromatic or naphthenic type plasticizing oils extracted from petroleum in a mass fraction of from 55% to 0%. The invention applies in particular to tires of passenger-vehicle type.
Owner:MICHELIN & CO CIE GEN DES ESTAB MICHELIN
Surface activating agent for oil-field thick-oil well
InactiveCN101024764AGood viscosity reduction effectImprove performanceTransportation and packagingMixingPhenolOleic Acid Triglyceride
The invention discloses a surface activator used in oil field thick oil well that is made up from water 1500kg, sheet alkali 330kg, alcohol 450kg, oleic acid 275kg, alkyl phenol polyoxyethylene (15) ether 200kg, sodium dodecyl benzene sulfonate 150kg. The invention could decrease interface tension, and under the emulsion effecting, the o / w or w / o would form flow state in normal state to be convenient to convey and pump. The invention has low cost and high effect.
Owner:图们市科兴石化技术开发有限公司
Solar cell
InactiveUS20090120490A1Improve conversion efficiencyReduce warpageNon-conductive material with dispersed conductive materialPhotovoltaic energy generationStearic acidSolar cell
A solar cell, comprising: a substrate, including p-n doping structure formed within said substrate; material attached to the back of said substrate, where said material includes glass mixture, aluminum material, organic medium and additive. Wherein said glass needs to be formed by combining two or more glasses: the main composition for post-mixed glass should include Al2O3, Bi2O5, B2O3, SiO2, PbO, Tl2O3 and other metal and non-metal oxides. For the material composition, aluminum powder content is 60˜80 mass %, with approximately 90 to 99.99% purity. Additives include C10˜C24 stearic acid, with the content of less than 5 mass %. The C10˜C24 stearic acid may include oleic acid. Organic medium content is roughly 20˜35 mass %. Wherein said organic medium includes 60˜90 mass % ether class organic solvent, 10˜20 mass % cellulosic resin and 1˜5 mass % of leveling agent, rheological agent or thixotropic agent.
Owner:GIGASTORAGE CORP
Use of nano structured lipid carrier drug feeding system
InactiveCN101129335AIncrease intakeGood curative effectPowder deliveryOrganic active ingredientsLipid formationMonoglyceride
The invention discloses an application of nanometer structure lipid carrier administration system in the antineoplastic drug to reverse the multiple-drug tolerance of tumour cell, which comprises the following parts: solid lipid material, liquid lipid material and antineoplastic drug, wherein the rate of the liquid lipid (such as oleic acid) is 0-30wt%; the solid lipid is selected from monoglyceride; the liquid lipid is oleic acid; the antineoplastic drug is Paclitaxel or adriablastina. The invention has highly effective cell uptaking and cytolymph condensing function with packing molecular target in the antineoplastic drug of cell, which avoids P-glucoprotein in the drug tolerant cytolymph from identifying the antineoplastic drug to reduce exclusion.
Owner:ZHEJIANG UNIV
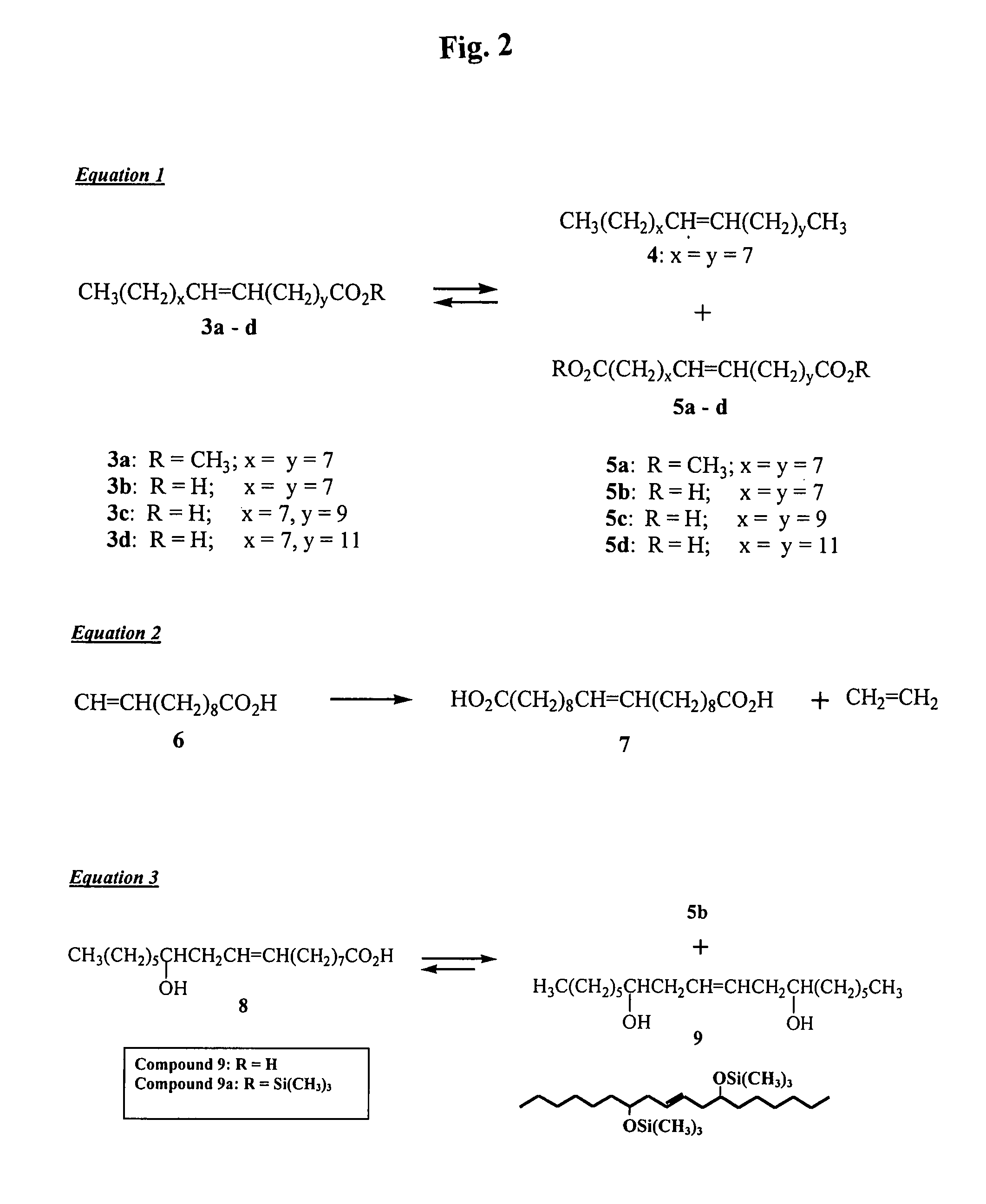
Method of producing dicarboxylic acids
InactiveUS7534917B1Organic compound preparationCarboxylic acid esters preparationCarboxylic acidDouble bond
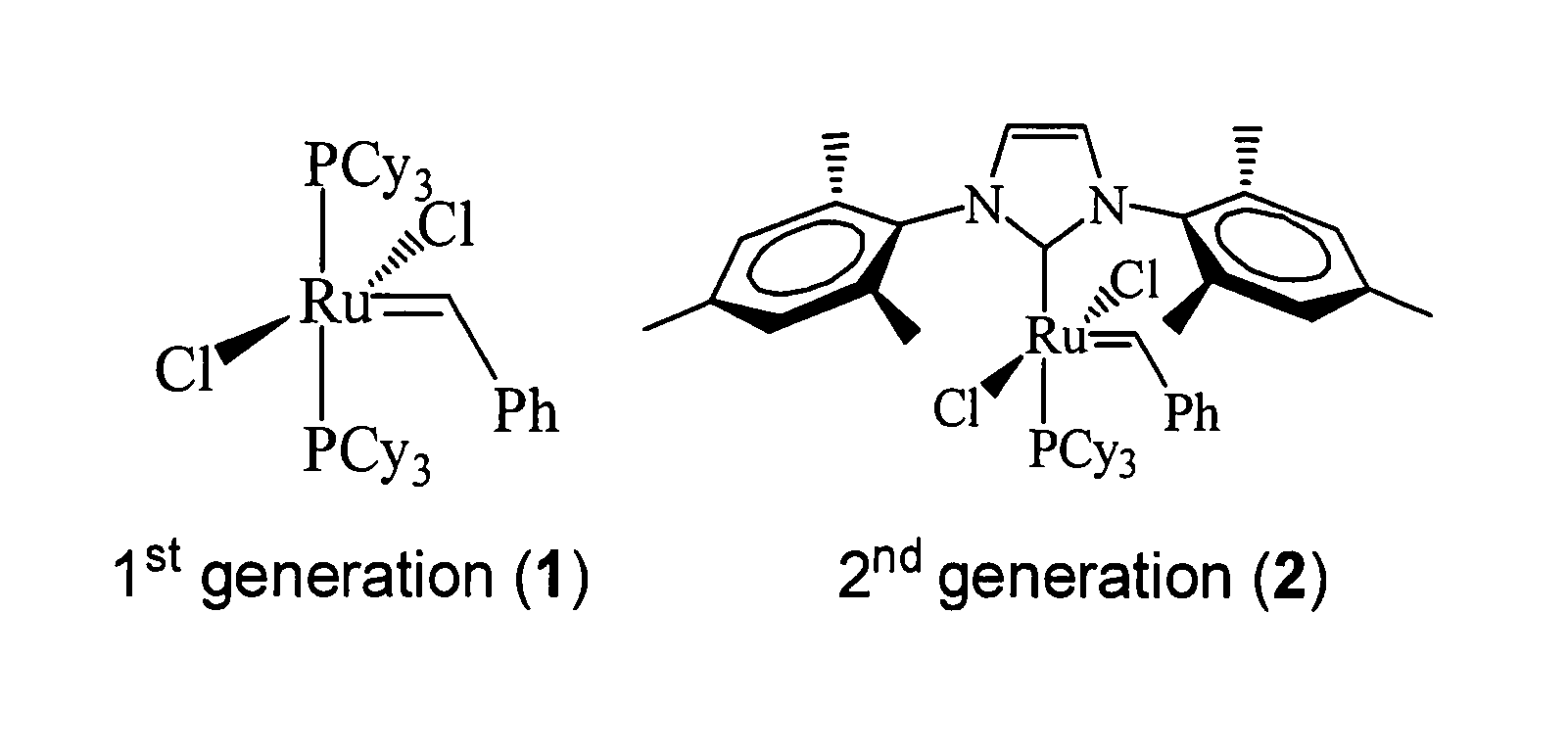
A method of producing dicarboxylic acids (e.g., α,ω dicarboxylic acids) by reacting a compound having a terminal COOH (e.g., unsaturated fatty acid such as oleic acid) and containing at least one carbon-carbon double bond with a second generation Grubbs catalyst in the absence of solvent to produce dicarboxylic acids. The method is conducted in an inert atmosphere (e.g., argon, nitrogen). The process also works well with mixed unsaturated fatty acids obtained from soybean, rapeseed, tall, and linseed oils.
Owner:US SEC AGRI
Novel canola cultivars having high yield and stabilized fatty acid profiles
ActiveUS20080260930A1Improve nutritional qualityFood preparationAngiosperms/flowering plantsBiotechnologyΑ-linolenic acid
Owner:CORTEVA AGRISCIENCE LLC
Low grade fluorite and barite flotation separation method
A low grade fluorite and barite flotation separation method uses waterglass as the inhibitor for silicious mineral and calcium carbonate mineral; uses oleic acid as the collector for fluorite and barite; and uses starch, sodium sulphate, hydrochloric acid and sodium hexametaphosphate as the inhibitor for barite mineral. The invention can perform flotation separation of low grade fluorite and barite effectively; under the condition that the given fluorite grade is 33.92%-40.41%, and the barite grade is 34.67%-35.33%, by mixing, flotation and separation, can achieves the fluorite concentrate with grade of above 95% and recovery rate of 75.5%-78.5%, and the barite concentrate with grade of 92.3%-93.27% and recovery rate of 75.2%-79.73%.
Owner:GUANGXI UNIV
Method of stabilizing imiquimod
InactiveUS7902242B2Short lifeImpaired stabilityBiocideOrganic chemistryPharmaceutical formulationOleic Acid Triglyceride
Pharmaceutical formulations and methods including an immune response modifier (IRM) compound and an oleic acid component are provided where stability is improved by using oleic acid have low polar impurities such as peroxides.
Owner:MEDICIS PHARMA CORP
Superparamagnetism mesoporous silicon dioxide composite ball and preparing method thereof
InactiveCN101256864ALarge specific surface areaHigh content of magnetic substancesSilicaInorganic material magnetismParticulatesMesoporous silica
A superparamagnetism mesoporous silicon dioxide composite ball of the present invention and manufacture method thereof belong to nucleocapsid type technology field of magnetic nano particle. The shape of the composite ball is global; inner core being made of magnetic ferrite nano particle cluster, shell coating layer being made of mesoporous silicon dioxide; quality ratio of magnetic ferrite nano particle in particulate being 40-80%. Method of manufacturing has that producing magnetic ferrite nano particle is by using coprecipitation method; executing surface modification by adding oleic acid stirring; ultrasonic forming oil-in-water emulsion in the mix solution of ethyl orthosilicate and cyclohexane, cetyl trimethyl ammonium bromide as surfactant; ethyl orthosilicate hydrolytic condensation forming mesoporous silicon dioxide coating layer by adding ammonia spirit; finally taking off molding plate and getting products. Product of the invention has bigger specific surface area and stronger magnetic separation capacity, and has good dispersancy in water, and is further functionalization after being decorated at surface. The method of the present invention has simple process, and lower equipment requirement.
Owner:JILIN UNIV
Fat compositions for infant formula and methods therefor
InactiveUS20040013787A1Improve the level ofLower levelVitamin food ingredientsFood preparationPhysiologyLinoleic acid
Fat compositions and infant formulas containing oleic acid in an amount of at least about 50% by weight of total fatty acids, lauric acid in an amount of at least about 10% by weight, palmitic acid in an amount of not more than about 10% by weight and, in certain embodiments, linoleic acid in an amount of not more than about 16% by weight. The fat compositions can be prepared to contain one or more oleic acid oils in a total amount of at least about 50% by weight, one or more linoleic acid oils in a total amount of not more than about 18% by weight and, in certain embodiments, one or more lauric acid oils in an amount of at least about 20% by weight. Also disclosed are methods of making the fat compositions and infant formulas and methods for providing a fat component to infant diets.
Owner:THEUER RICHARD C
Composition based on fish oil
InactiveUS6020020AEfficient dosingFatty oils/acids recovery from wasteDough treatmentFish oilOleic Acid Triglyceride
PCT No. PCT / EP96 / 05025 Sec. 371 Date May 26, 1998 Sec. 102(e) Date May 26, 1998 PCT Filed Nov. 12, 1996 PCT Pub. No. WO97 / 19601 PCT Pub. Date Jun. 5, 1997Fish oil concentrates, having: < / =40% of long chain poly unsaturated fatty acids <20% of saturated fatty acids (C14-C18) <15% oleic acid <12% C16:1-fatty acid and with a weight-ratio: can be obtained by a process involving the following steps subjecting a fish oil to an enzymic conversion removing free fatty acids or esters from conversion-product subjecting product of above to enzymic hydrolysis, using specific lipase or partial glyceride-specific lipase wash and dry product re-esterify dried product.
Owner:CONOPCO INC D B A UNILEVER
Preparation method of grease with humanized structure
ActiveCN102776077AHuge market potentialHigh number of reusesMilk preparationFatty acid esterificationVegetable oilMolecular rearrangement
The invention discloses a preparation method of grease with a humanized structure. The preparation method comprises the following steps: carrying out intra-molecular rearrangement reaction to palm oil with palmitic acid content of higher than 50% under the action of catalyst to obtain Sn-2 locus triglyceride with palmitic acid content of higher than 50%; and carrying out ester exchange reaction to the Sn-2 locus triglyceride with palmitic acid content of higher than 50% with mixed fatty acid or non-glyceride under the action of 1,3 locus Specificity lipase, and then subjecting the reaction product to after treatment, so as to obtain the grease with humanized structure, wherein the mixed fatty acid is mixture of at least two selected from the group consisting of decanoic acid, lauric acid,myristic acid, palmitoleic acid, stearic acid, oleic acid, linoleic acid and linolenic acid. The vegetable oils are used as the raw material according to the method, so the safety is high; the preparation process is simple, the reaction time is short, the enzyme dosage is low, and the cost is low; and the prepared grease with the humanized structure is very similar to the beast milk fat in the composition of fatty acid and the structural distribution of triglyceride, so the grease can be added in infant formula or formula foods and used as breast milk fat substitute.
Owner:ZANYU TECH GRP CO LTD
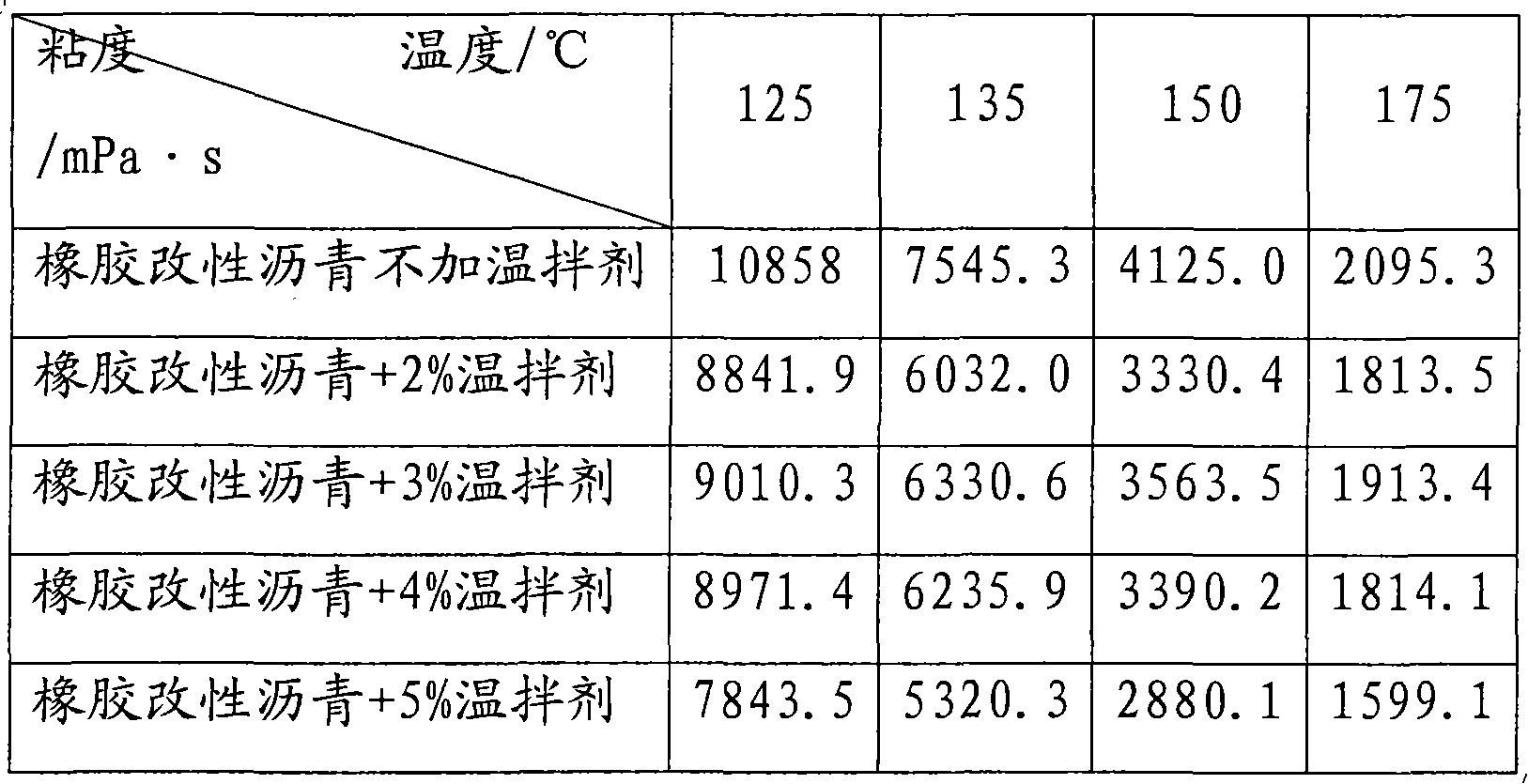
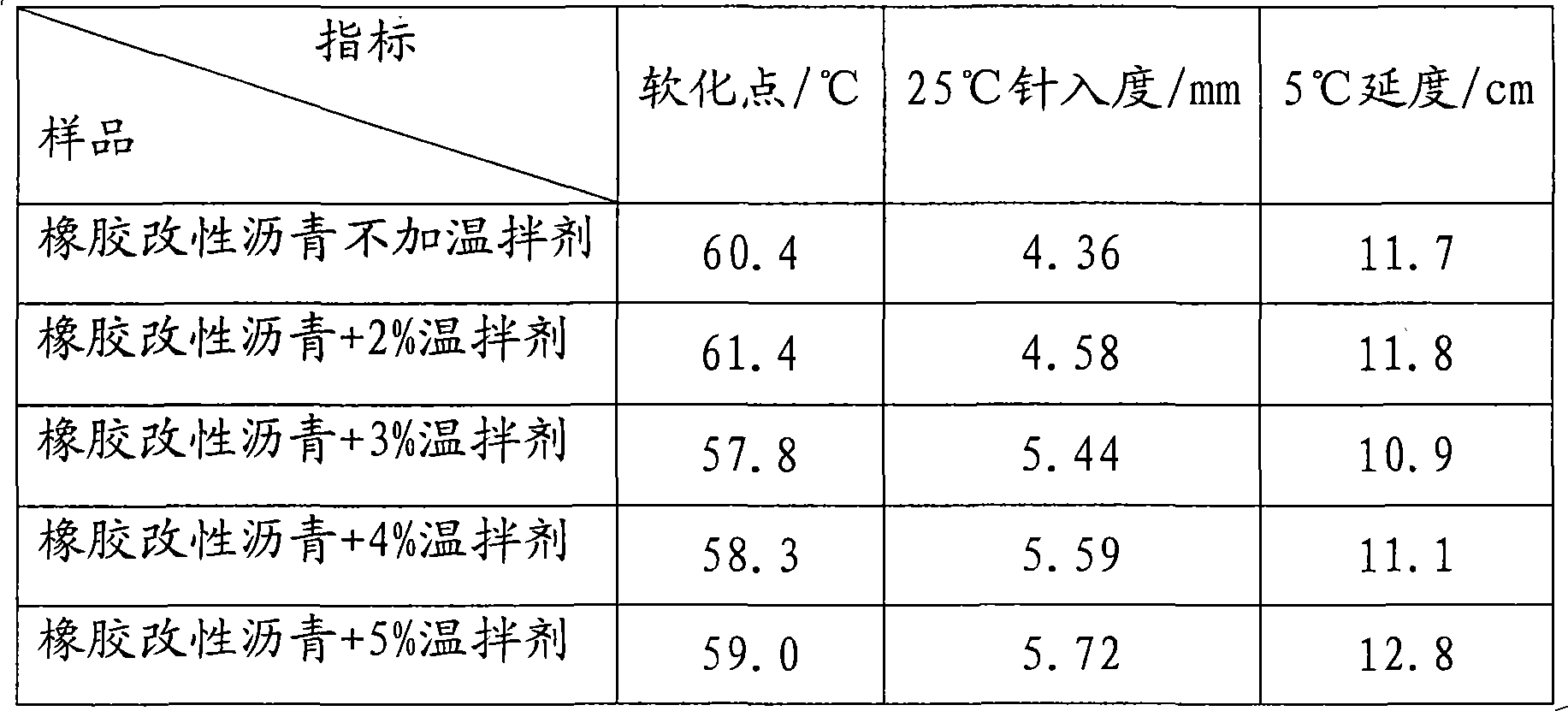
Rubber modified asphalt warm-mixing agent, preparation method and application
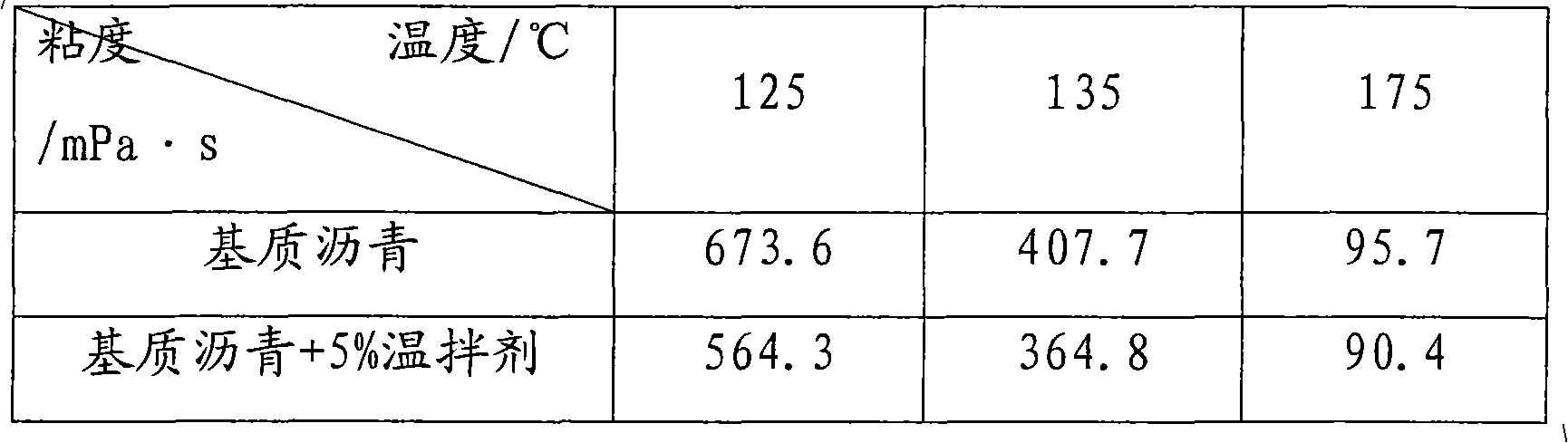
InactiveCN101831187ALow viscosityLow softening pointClimate change adaptationBuilding insulationsSurface-active agentsFatty alcohol
The invention discloses a rubber modified asphalt warm-mixing agent, a preparation method and application thereof. The warm-mixing agent comprises the following components in parts by weight: 100 parts of water, 5-30 parts of fatty alcohol sodium sulfate, 15-30 parts of palmitic acid, 5-30 parts of oleic acid, 1-20 parts of sodium dodecyl benzene sulfonate, 1-15 parts of cetyltrimethylammonium chloride and 10-30 parts of imidazoline-type cationic surface active agent ODD. The preparation method comprises the following steps of: adding the water in a container at the temperature of 65 DEG C, and then adding the following substances under the condition of stirring: the fatty alcohol sodium sulfate, the palmitic acid, the oleic acid, the sodium dodecyl benzene sulfonate, the cetyltrimethylammonium chloride and the imidazoline-type cationic surface active agent ODD, and stirring for 15 min to obtain the warm-mixing agent. The warm-mixing agent is directly applied to rubber asphalt or matrix asphalt to reduce the viscosity of the asphalt. The kinematic viscosity of the rubber powder asphalt added with the warm-mixing agent at the temperature of 150 DEG C can reach less than 3.0Pa.s of the kinematic viscosity at the temperature of 180 DEG C, and the production temperature can be reduced by about 30 DEG C, thereby not only preventing the rubber asphalt from being aged but also saving energy and reducing equipment loss.
Owner:BEIJING UNIV OF CHEM TECH
Chlorofluorocarbon-free mometasone furoate aerosol formulations
The invention relates to suspension aerosol formulations which exhibit stable particle sizes, containing mometasone furoate, about 1 to about 10 weight percent ethanol and 1,1,1,2,3,3,3-Heptafluoropropane as the propellant. A surfactant, such as oleic acid, can also be included. These formulations are suitable for use in metered dose inhalers.
Owner:MERCK SHARP & DOHME CORP
Plastic fat composition
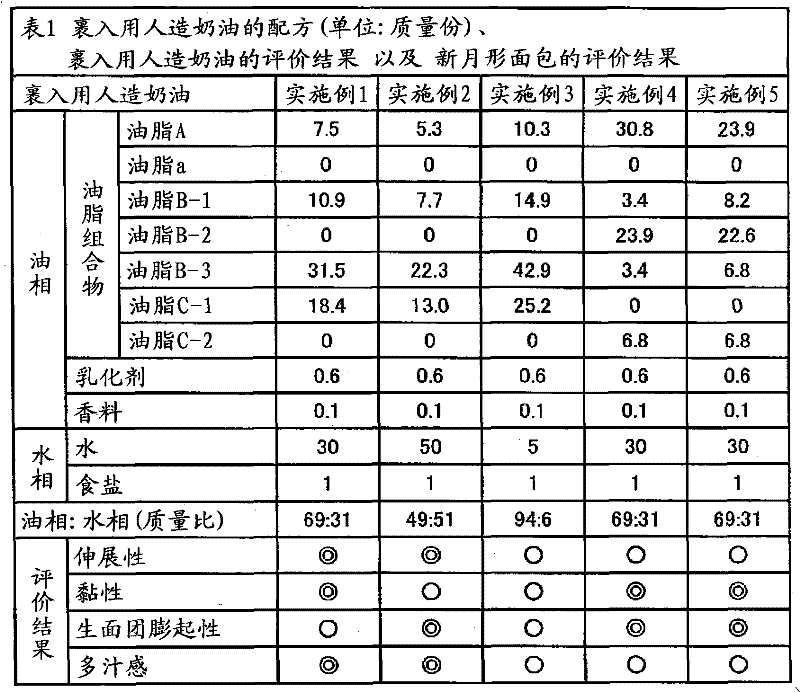
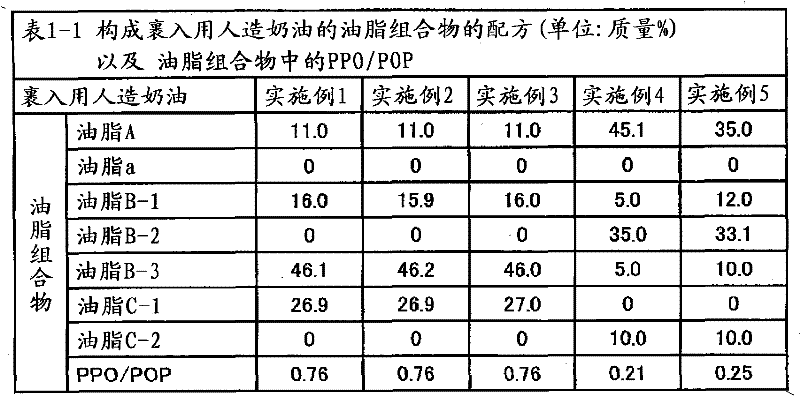
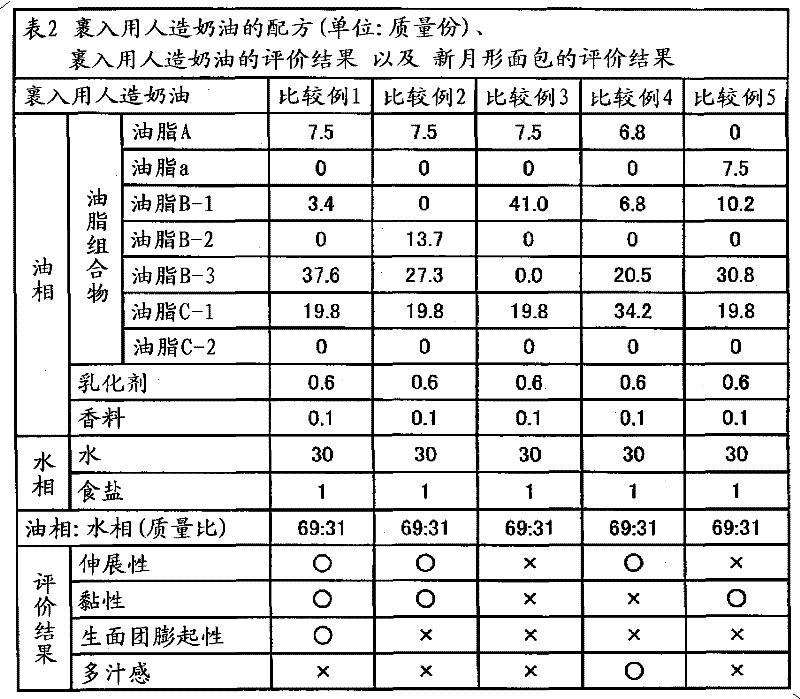
ActiveCN102056493AEasy to stretchEasy to operateSpread compositionsFatty acid esterificationStearic acidOleic Acid Triglyceride
Provided are a plastic fat composition having a small trans fatty acid content, which is suitable for roll-in, spreading, kneed-in and similar purposes, and a fat composition to be used in the preceding plastic fat composition. A fat composition which contains the following fats A, B and C and satisfies the following requirements (a) to (c): fat A: a fat which contains 20 to 60% by mass of a saturated fatty acid having 12 to 14 carbon atoms and 40 to 80% by mass of a saturated fatty acid having 16 to 18 carbon atoms in the total constituting fatty acids thereof, and in which the fats have been obtained by ester exchange; fat B: a fat which contains 20 to 60% by mass of palmitic acid, 0.5 to 6% by mass of stearic acid and 30 to 60% by mass of oleic acid in the total constituting fatty acids thereof; and fat C: a liquid oil; (a) the content of fat A in the fat composition being 0.1 to 50% by mass; (b) the content of fat B in the fat composition being 0.1% by mass or more but less than 40% by mass; and (c) the PPO / POP of the fat composition being 0.15 to 1.00.
Owner:THE NISSHIN OILLIO GRP LTD
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com