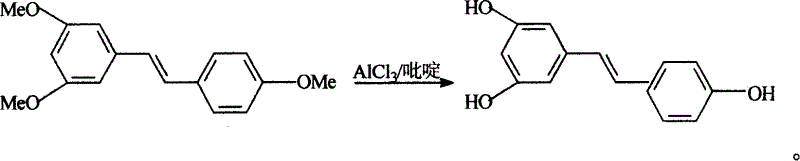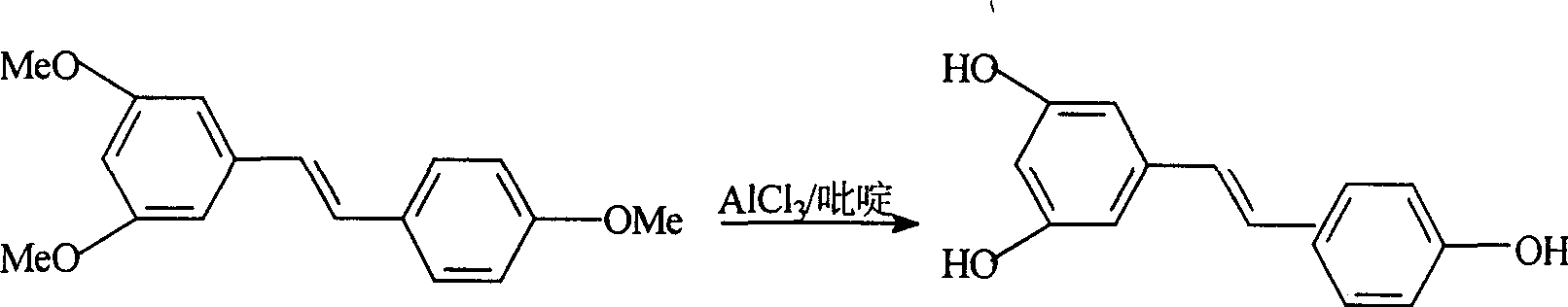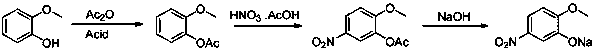Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
41results about How to "Excellent yield" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Multifunctional seedling cultivation substrate
ActiveCN103392575AControl leggyImprove root vitalityCultivating equipmentsSoilless cultivationBiotechnologyGrowth retardant
The invention discloses a multifunctional seedling cultivation substrate which comprises the components of EM fermented maize straws, perlite, vermiculite and prohexadione calcium, wherein the volume ratio of the EM fermented maize straws, the perlite and the vermiculite is 6: 3: 1, and the amount of the prohexadione calcium is 10-20g / m<3>. The multifunctional seedling cultivation substrate can control excessive growth of seedlings and can effectively prevent side effects of sprayed conventional growth inhibitors, and cultivated seedlings are superior to existing conventional seedling cultivation substrates in root activity, strong seedling index, yield and other aspects. Meanwhile, using amount of grass carbon is reduced, grass carbon resource saving is facilitated, and the purposes of resource utilization of agricultural wastes and environment pollution reduction are achieved.
Owner:SHANDONG SHOUGUANG VEGETABLE IND GRP
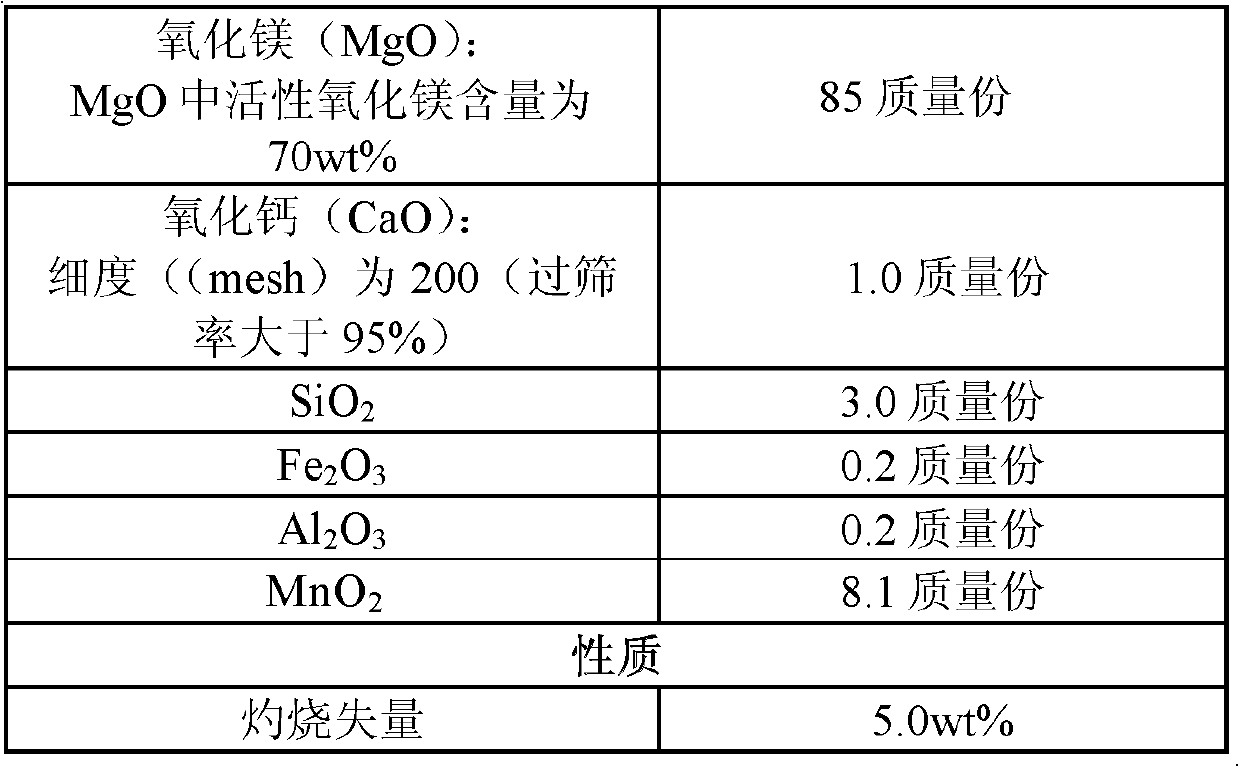
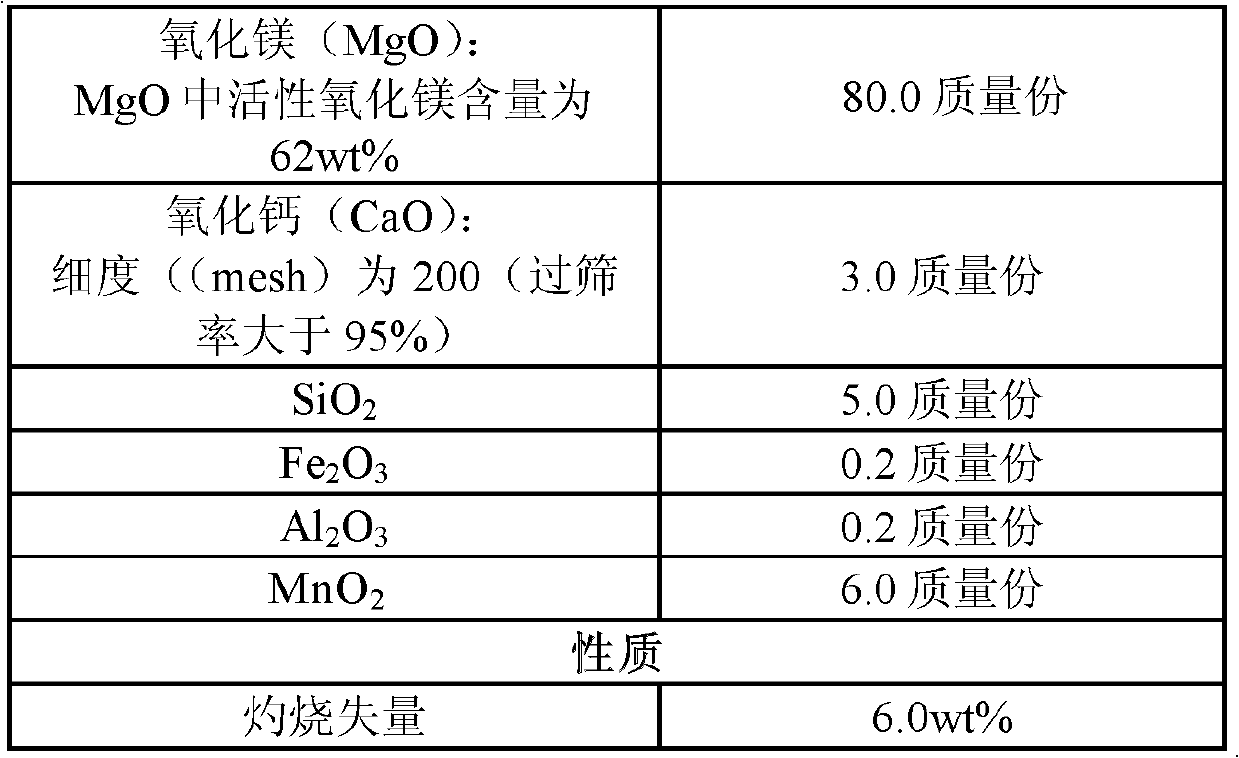
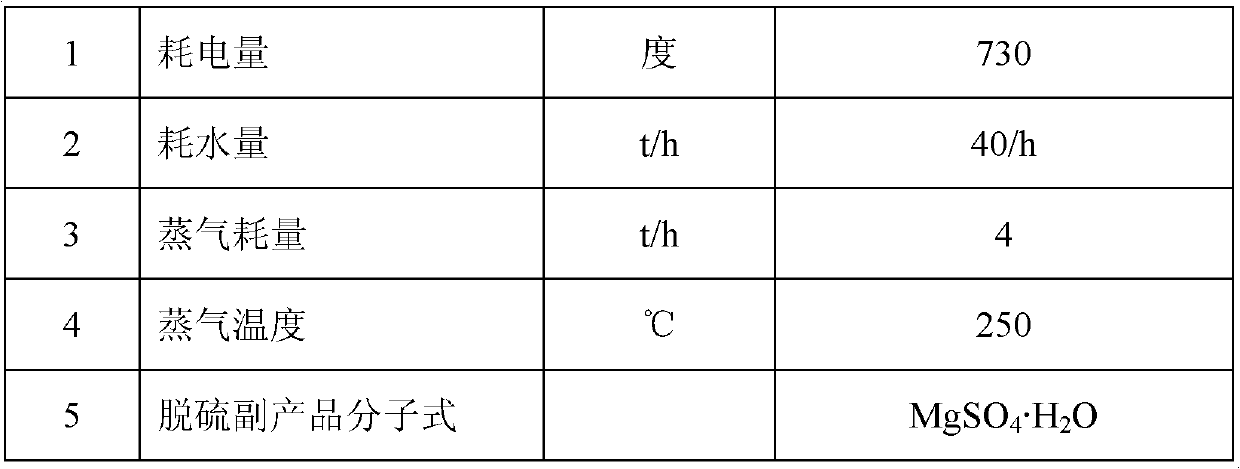
Method for producing magnesium sulfate monohydrate by using desulfurization wastewater
ActiveCN102745725AQuality up to standardGuaranteed uptimeMagnesium sulfatesWastewaterCrystallization
The present invention discloses a method for producing magnesium sulfate monohydrate by using desulfurization wastewater. The method comprises the following steps: (1) pulping, (2) carrying out desulfurization, (3) discharging the pulp, (4) filtering, (5) carrying out concentration, and (6) carrying out crystallization. With the method of the present invention, high value byproducts can be synchronously produced during a flue gas desulfurization process by using a magnesium oxide method. In addition, with the method of the present invention, a stable running characteristic is provided, and quality of the produced magnesium sulfate monohydrate after desulfurization reaches standards.
Owner:ZHONGJING ENVIRONMENTAL TECH CO LTD
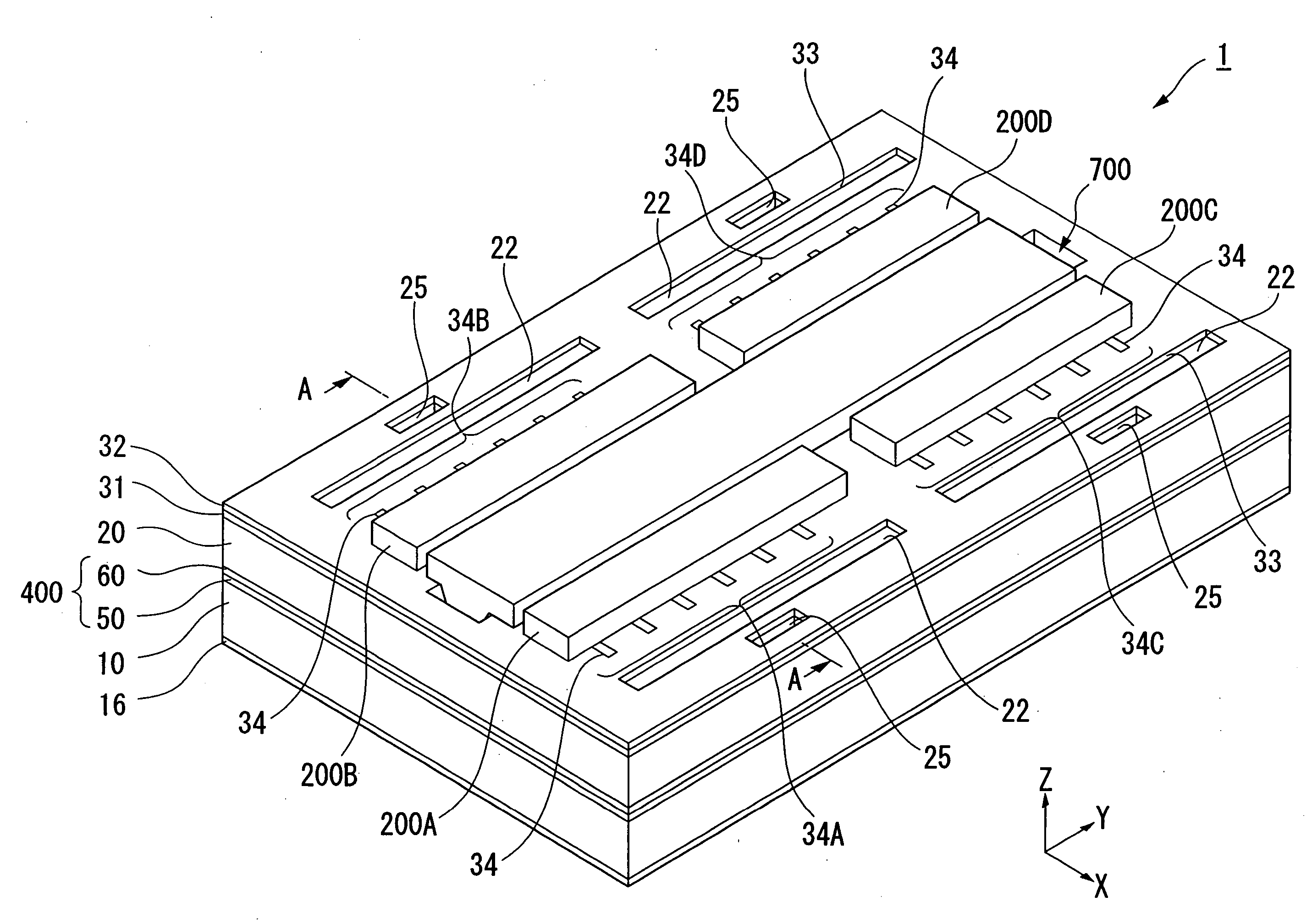
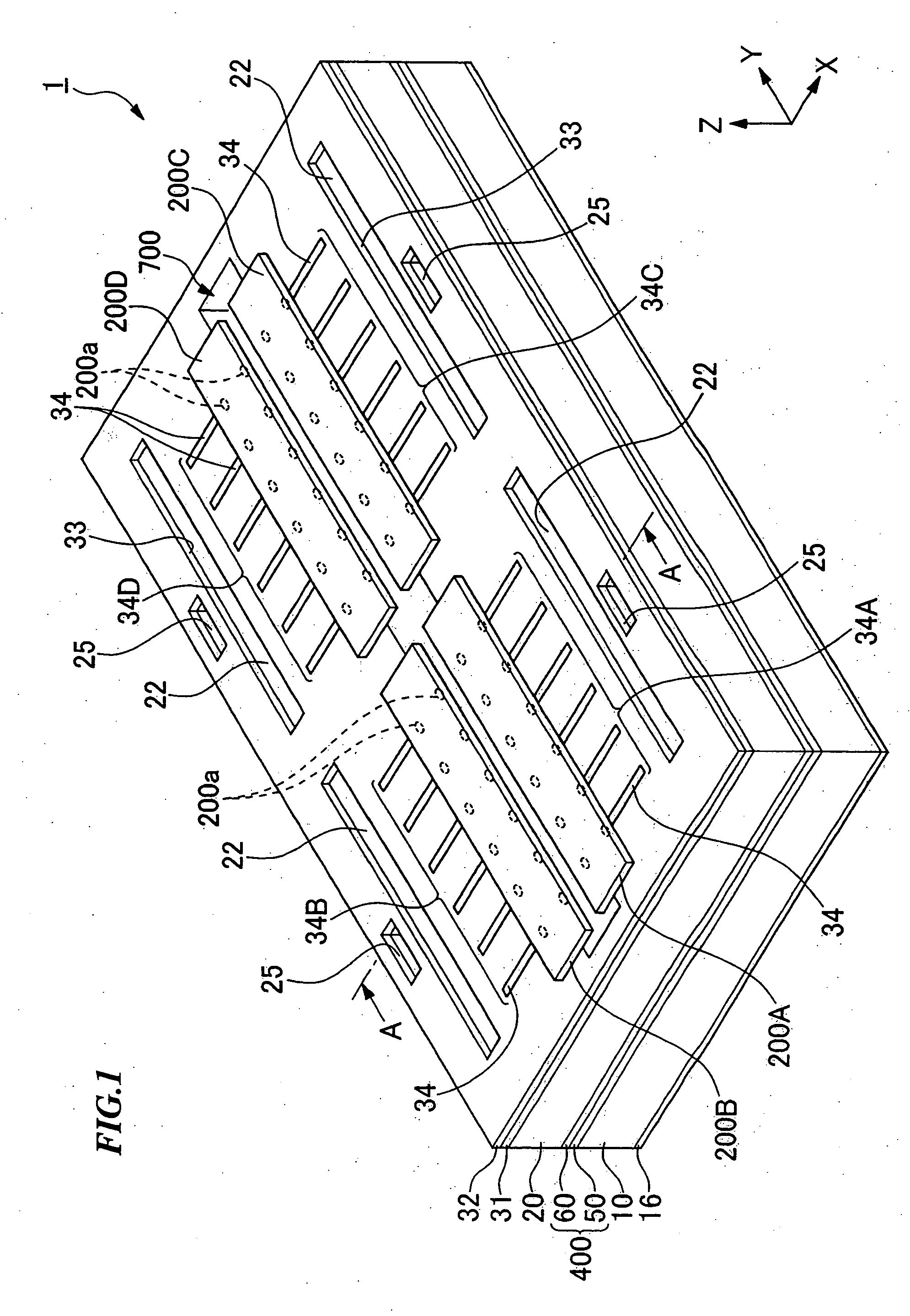
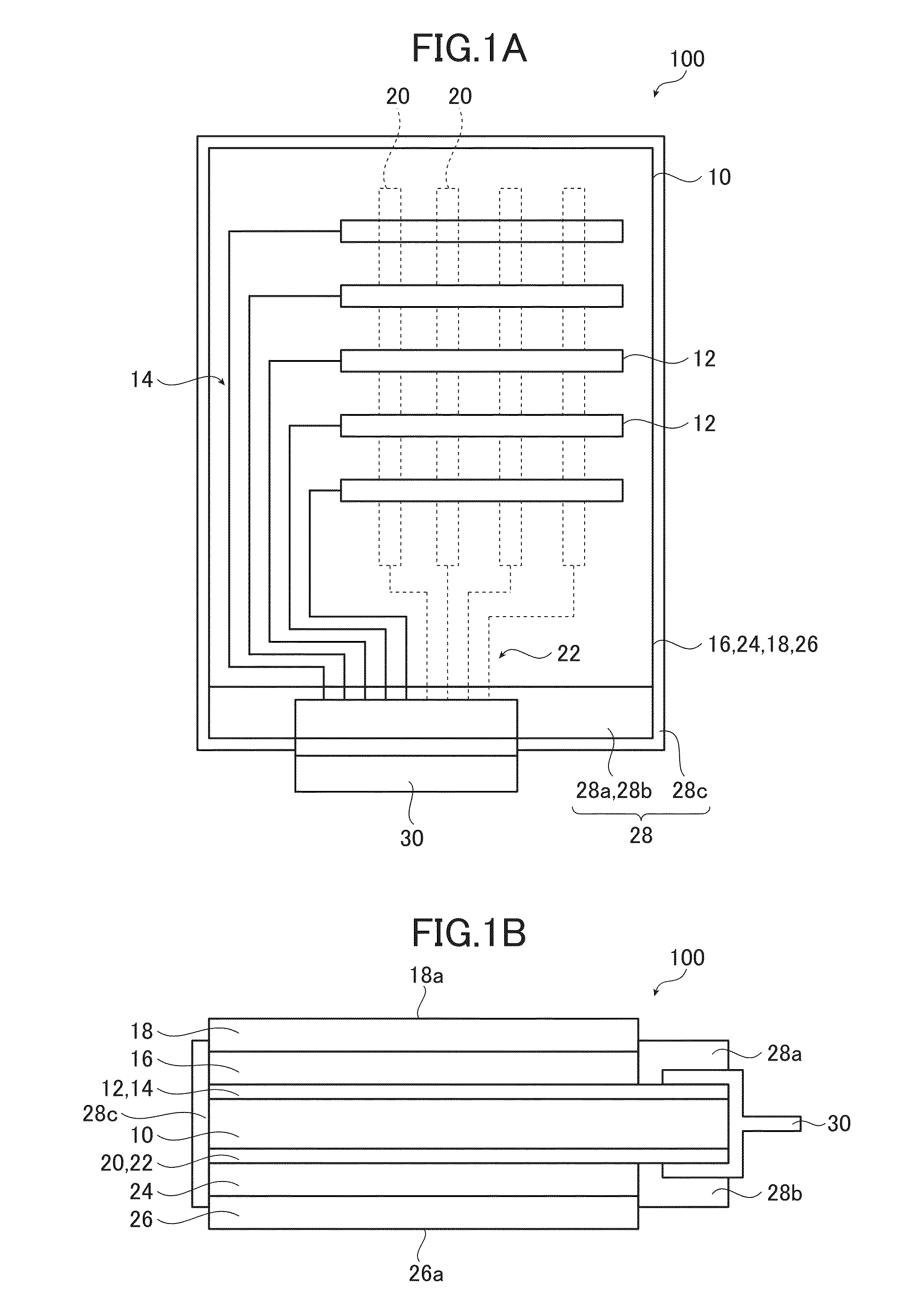
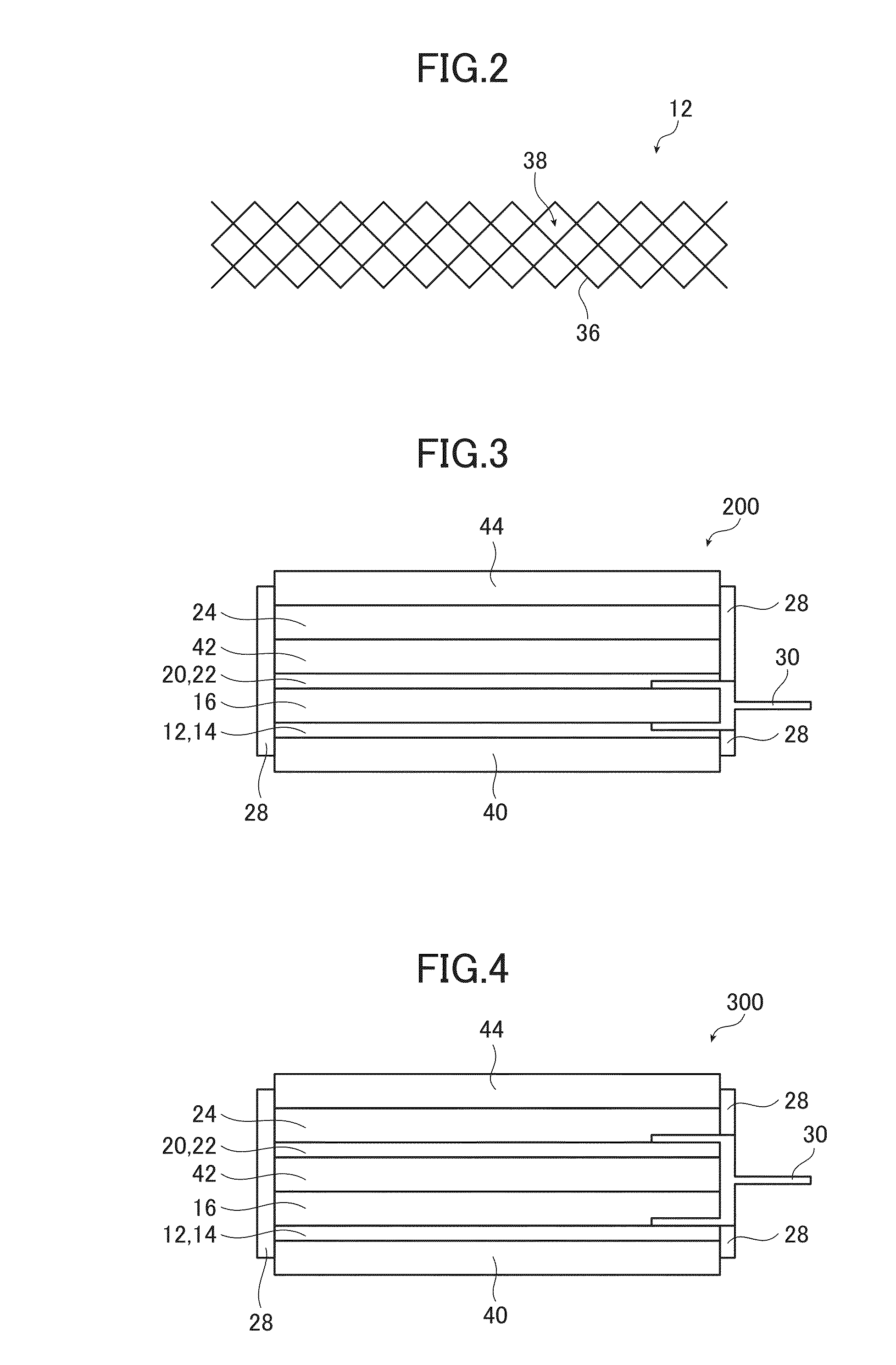
Device package structure, device packaging method, droplet ejection head, connector, and semiconductor device
ActiveUS20060164466A1Superior reliabilityExcellent yieldInking apparatusSemiconductor/solid-state device detailsEngineeringHead parts
A device package structure includes: a base body having a conductive connection portion and a level difference portion; a device arranged on the base body, having a connection terminal electrically connected to the conductive connection portion via the level difference portion on the base body; and a connector electrically connecting the connection terminal and the conductive connection portion, having substantially the same height as a height of the level difference portion.
Owner:SEIKO EPSON CORP
Capacitance type touch panel, manufacturing method of the same, and input device
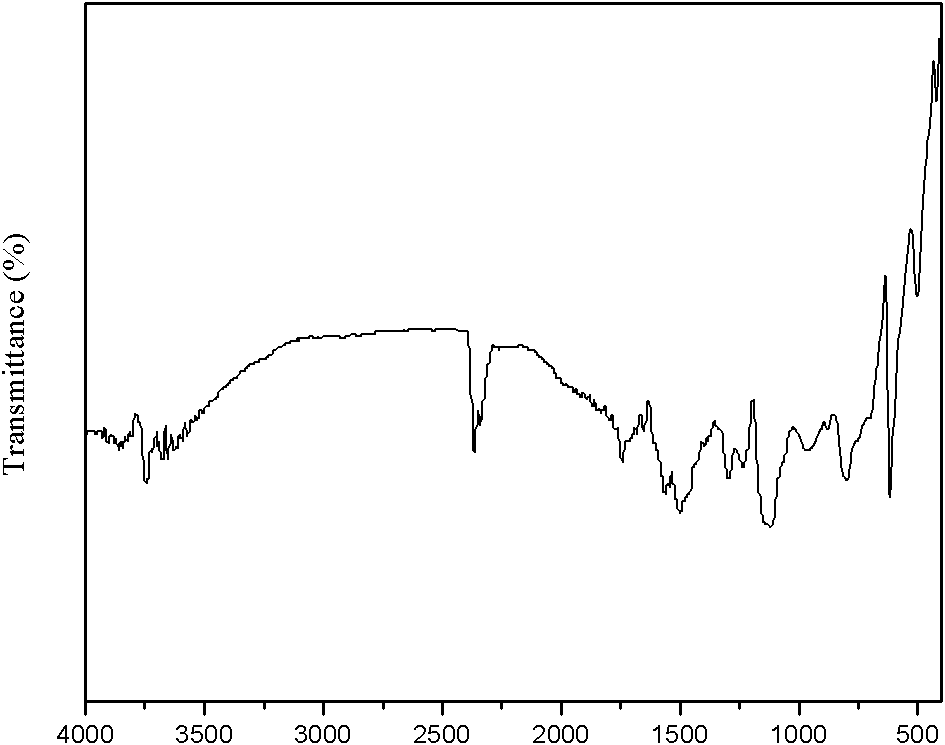
ActiveUS20150169111A1Excellent yieldGood yieldStatic indicating devicesInput/output processes for data processingTouch panelTransmittance
A capacitance type touch panel includes: an insulating layer; a plurality of electrode portions; a plurality of lead-out wiring portions; a transparent resin layer; and a substrate disposed on the transparent resin layer, wherein at least on the surface of the peripheral edge of the transparent resin layer exposed between the insulating layer and the substrate and on the exposed surface of the lead-out wiring portions, a sealing layer is disposed, and the sealing layer has a moisture vapor transmittance equal to or less than 20 g / m2 / 24 h / atm (25° C., 90% RH, 25 μm), and has a thickness equal to or greater than 1.0 μm.
Owner:FUJIFILM CORP
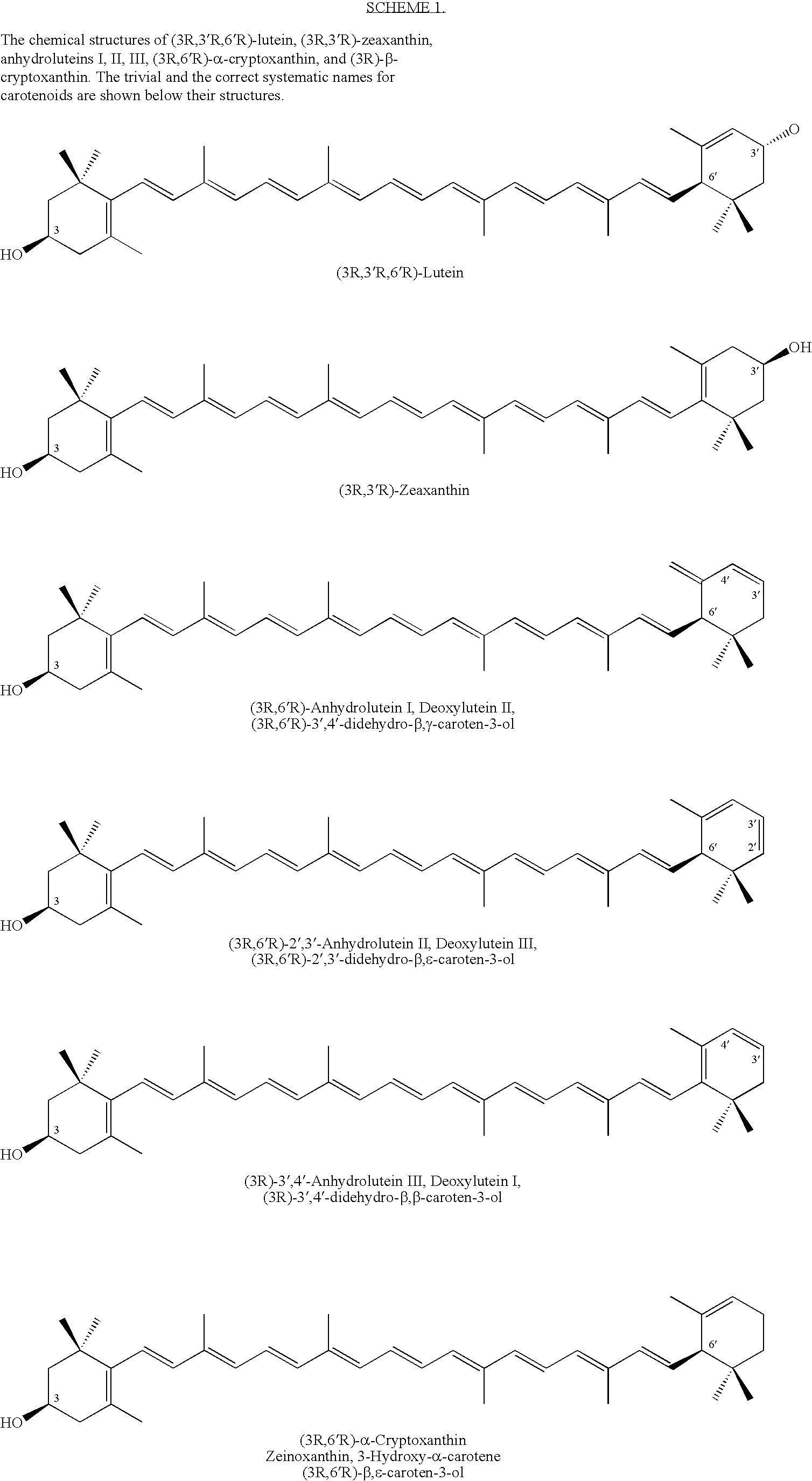
Method for production of rare carotenoids from commercially available lutein
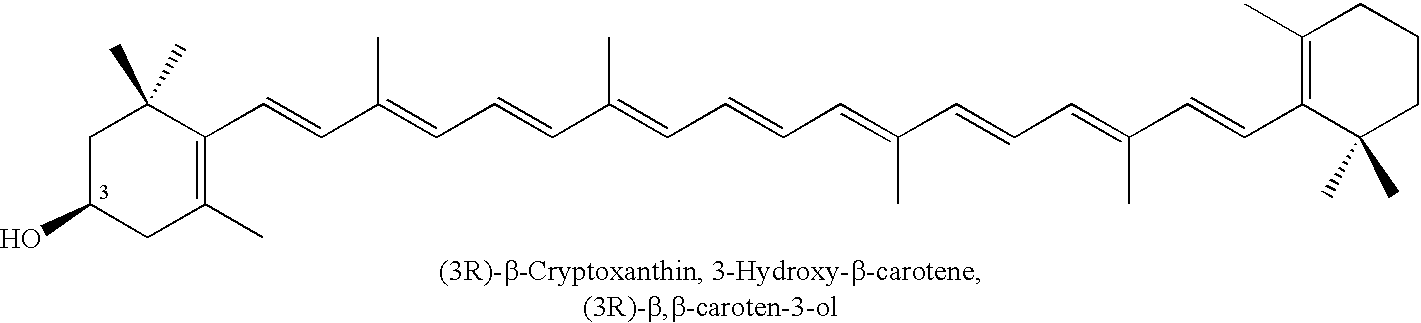
InactiveUS6911564B2Excellent yieldSuperior yieldOrganic compound preparationCarbonyl compound preparation by oxidationChemistryBeta-cryptoxanthin
Disclosed are processes for conversion of (3R,3′R,6′R)-lutein to (3R,6′R)-α-cryptoxanthin, (3R)-β-cryptoxanthin, anhydroluteins I, II, and III (dehydration products of lutein), and a method for separating and purifying the individual carotenoids including the unreacted (3R,3′R)-zeaxanthin. The invention also includes two methods that transform (3R,3′R,6′R)-lutein into (3R,6′R)-α-cryptoxanthin in excellent yields.
Owner:MARYLAND COLLEGE PARK UNIV OF
Process for synthesizing resveratrol by using de-methylation technology
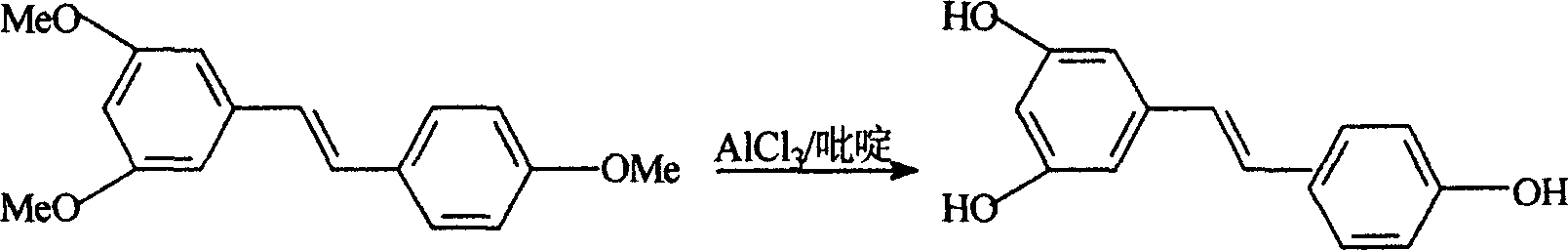
InactiveCN1663939AExcellent qualityExcellent yieldOrganic chemistryOrganic compound preparationAluminium chlorideWittig reaction
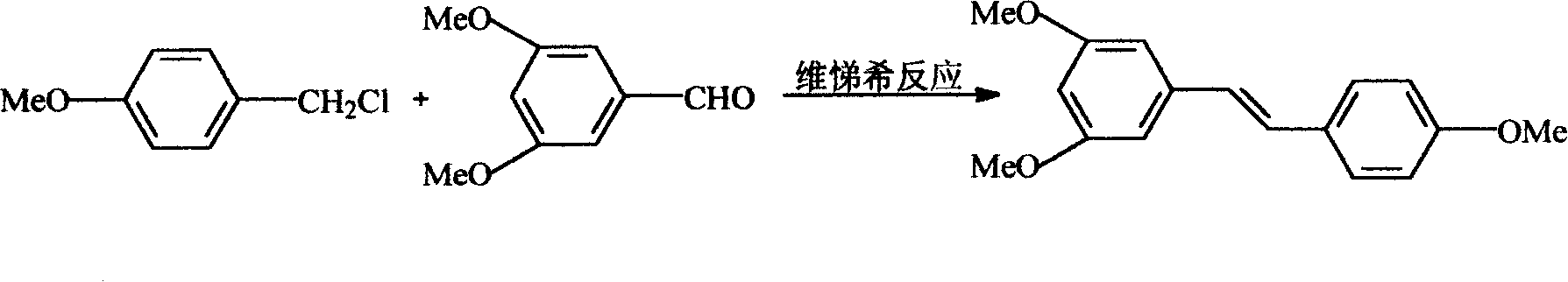
The invention relates to a process for synthesizing resveratrol by using de-methylation technology, which comprises using 3,5,4'-trimethoxyl diphenyl ethylene as raw material, using aluminium chloride / pyridine as catalyst for synthesizing Resveratrol through demethyl process, and employing market available methoxyl benzyl chloride for Wittig reaction with 3,5- dimethoxybenzaldehyde, thus preparing intermediate 3,5,4'-trimethoxyl diphenyl ethylene.
Owner:NANJING RALLY BIOCHEM
Method for modifying synthetic veratric alcohol
InactiveCN101033172AExcellent qualityExcellent yieldOrganic chemistryOrganic compound preparationAlcoholWittig reaction
This invention relates to an improved method for synthesizing resveratrol, which takes 3, 5, 4'-methylate toluylene as the raw material, 1, 3, 5-trimethylbenzene as the solvent and hydroquinone as the antioxidant to synthesize resveratrol with alchlor / pyridine as the catalyst and applies commercial cheap anisyl chlorben and 3, 5-dimethoxy benzene formaldehyde for wittig reaction to prepare an intermediate 3, 5, 4'-methylate toluylene.
Owner:NANJING RALLY BIOCHEM
Process for the preparation of acyl heteroaromatic compounds from heteroaromatic compounds by metal ion exchanged clays
The present invention relates to a process for the preparation of the acyl heteroaromatic compounds useful as important intermediates for drugs, pharmaceuticals and flavouring agents, said process comprising reacting an heteroaromatic compound selected from furan, thiophene and pyrrole with a C2-C5 acid anhydride as an acylating agent employing metal ion exchanged clays as catalysts at temperatures in the range of 0-130° C. for a period of 1-24 h, and separating the acyl heteroaromatic compound by a conventional method to obtain a product of high purity.
Owner:COUNCIL OF SCI & IND RES
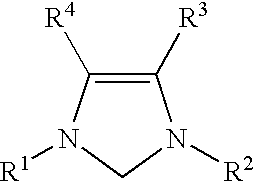
Catalyzed coupling reactions of aryl halides with silanes
InactiveUS6586599B1Excellent yieldEliminate needOrganic compound preparationOrganic-compounds/hydrides/coordination-complexes catalystsBromineIodine atom
A process for conducting coupling reactions of aryl halides with unsaturated silanes is described. The processes use N-heterocyclic carbenes as ancillary ligands in these coupling reactions. A coupling of an aryl halide with an unsaturated silane can be carried out by mixing, in a liquid medium, at least one strong base; at least one aryl halide or aryl pseudohalide in which all substituents are other than silyl groups, wherein the aryl halide has, directly bonded to the aromatic ring(s), at least one chlorine atom, bromine atom, or iodine atom; at least one silane wherein the silicon atom is quaternary, wherein one group bound to the silicon atom is unsaturated at the alpha or beta position, and wherein each of the remaining groups bound to the silicon atom is a saturated hydrocarbyl or a saturated hydrocarbyloxy group; at least one nickel, palladium, or platinum compound, wherein the formal oxidation state of the metal is zero or two; and at least one N-heterocyclic carbene. One preferred type of N-heterocyclic carbene is an imidazoline-2-ylidene of the formulawherein R1 and R2 are each, independently, alkyl or aryl groups having at least 3 carbon atoms, R3 and R4 are each, independently, a hydrogen atom, a halogen atom, or a hydrocarbyl group.
Owner:UNIV OF NEW ORLEANS RES TECH FOUND
Preparation method for 4-chlorophenylhydrazine hydrochloride
InactiveCN103910650AEliminates the problem of easy agglomeration and difficult operation of feedingEasy dischargeHydrazine preparationSolubilityHydrogen Sulfate
A provided preparation method for 4-chlorophenylhydrazine hydrochloride comprises the following steps: 1) under an acidic condition, at 5-10 DEG C, dropwise adding a sodium nitrite aqueous solution with a concentration of 20% into 4-chloroaniline to perform a diazo reaction and to generate a diazo salt; 2) under a room temperature condition, dropwise adding an ammonium sulfite aqueous solution into a solution of the diazo salt obtained in the step 1, heating to 50-60 DEG C and keeping the temperature for 3-4 h; and 3) at 50-70 DEG C, dropwise adding a hydrochloric acid solution with a concentration of 20% into the solution obtained in the step 2, keeping the temperature for 1-2 h, reducing the temperature, filtering, washing, discharging the material, and drying to obtain a finished product. The beneficial effects comprise that by using ammonium sulfite aqueous solution as a reducing agent in the reduction reaction, the problems are solved that sodium sulfite solid is easy to cake during material charging in a conventional technology and material charging is not easy to operate; ammonium chloride and ammonium hydrogen sulfate which are generated in the acidification reaction have high solubility in water, and crystallization precipitation is late, thereby facilitating performing of the acidification reaction in the solution and reducing side reactions; and the product crystallizes loosely, is good in fluidity, easy for material discharging and washing, and product quality and yield are both better than conventional production levels.
Owner:TIANJIN QIDONG CHEM PLANT
Ceramic tile decorated by dry lamination of colorful particles and micro powder and preparation method of ceramic tile
ActiveCN107116675AEasy to shapeExcellent color richnessMixing operation control apparatusCeramic shaping plantsPrillBrick
The invention provides a ceramic tile decorated by dry lamination of colorful particles and micro powder and a preparation method of the ceramic tile which are improved on floral designs, hues, and production technologies. The preparation method of the ceramic tile includes the steps that basic powder materials and colored materials are dry-mixed to prepare toner; the toner is prilled to form colored particles in different shapes; the colored particles and the toner with different colors are conveyed to and stirred in a plurality of stirrers correspondingly according to set proportions; the powder materials which are stirred in the multiple stirrers are conveyed through a plurality of feeding belts correspondingly and blanked into a vertical material loading machine according to a preset material loading procedure, and the powder materials naturally stack to form a flower shape; and the formed flower-shaped powder is conveyed to a press mold frame, pressed and formed.
Owner:广东清远蒙娜丽莎建陶有限公司 +1
Method for preparing polyaniline/silver nanocomposite material
InactiveCN101838461AImprove conductivityGood dispersionElectrolytic capacitorsActive material electrodesMicroemulsionElectrochemistry
The invention provides a method for preparing a polyaniline / silver nanocomposite material, which belongs to the technical field of composite materials. The method comprises the following steps of: preparing nano-silver particles in microemulsion; and compounding polyaniline in situ to form a nanocomposite material. By using the method, the problem of the agglomeration of nano-silver particles in a composite material can be solved; an organic-inorganic nanocomposite material of a core-shell coating structure of which the nano-silver particles are inside and the polyaniline is outside is formed; and the organic-inorganic nanocomposite material has the characteristics of high electric conductivity, thermal property and electrochemical performance, and good application prospect. The method has the characteristics of simple process, low production cost, high production efficiency and good industrial production prospect.
Owner:CENT SOUTH UNIV
Chiral polyacid-based metal organic framework material and preparing method and application thereof
ActiveCN109772459AAccurate understanding of structural featuresUnderstanding Structural FeaturesOrganic chemistryOrganic-compounds/hydrides/coordination-complexes catalystsSpace groupElectron donor
The invention provides a chiral polyacid-based metal organic framework material. The chemical formula of the polyacid-based metal organic framework material is C74H101B2Zn2N22O84W24, and is recorded as ZnW-DPNDI-PYI1 and ZnW-DPNDI-PYI2 according to the chirality, wherein the DPNDI is N,N'-bi(4-picoline) naphthalimide and serves as a bridging ligand and a photosensitizer, and the PYI is L-pyrrolidine-2-imidazole and serves as a chiral ligand and an electron donor; crystals belong to a monoclinic system, and both the space group of the ZnW-DPNDI-PYI1 and the space group of the ZnW-DPNDI-PYI2 areP2(1). The chiral polyacid-based metal organic framework material is a multifunctional inorganic porous material, and has an asymmetric photocatalytic oxidation technology, and in the technology, a photocatalyst is driven by visible light to be an excited state, then the excited-state photocatalyst and substrates are subjected to an oxidation-reduction reaction, and therefore reactions of relatedfree radical types are completed.
Owner:HENAN UNIVERSITY
Enantioselective 1,4-addition of aromatic nucleophiles to alpha,beta-unsaturated aldehydes using chiral organic catalysts
InactiveUS7173139B2Excellent yieldHigh enantioselectivityOrganic compound preparationOrganic chemistry methodsAniline CompoundsAldehyde
Nonmetallic, chiral organic catalysts are used to catalyze the 1,4-addition of an aromatic nucleophile to an α,β-unsaturated aldehyde. The aromatic nucleophile may be an N,N-disubstituted aniline compound, or an analog thereof. The reaction is efficient and enantioselective, and proceeds with a variety of substituted and unsubstituted aromatic nucleophiles and aldehydes. The invention also provides a method for the deamination of aromatic N,N-disubstituted amines such as those resulting from the 1,4-addition of an aromatic nucleophile to an α,β-unsaturated aldehyde.
Owner:CALIFORNIA INST OF TECH
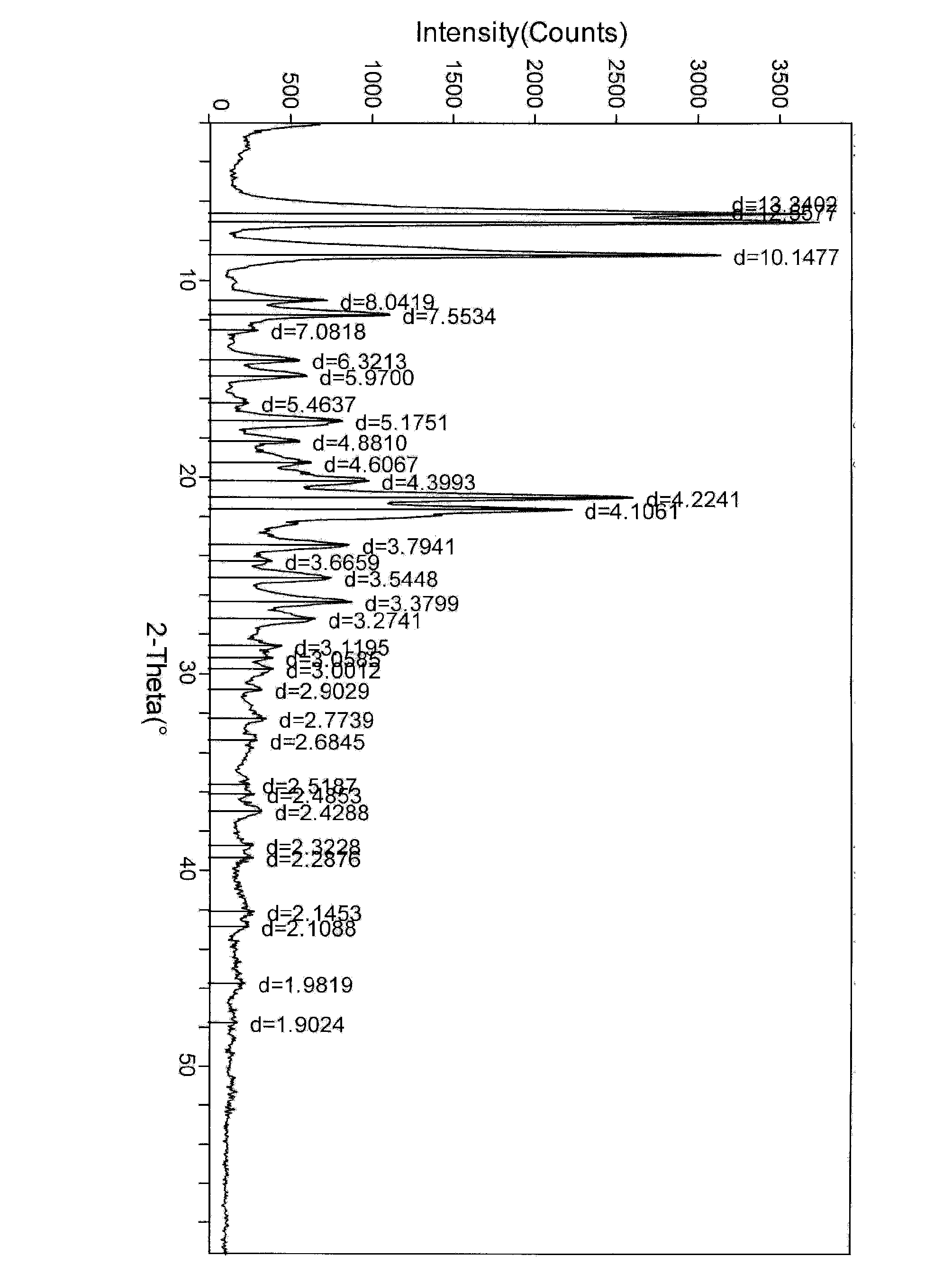
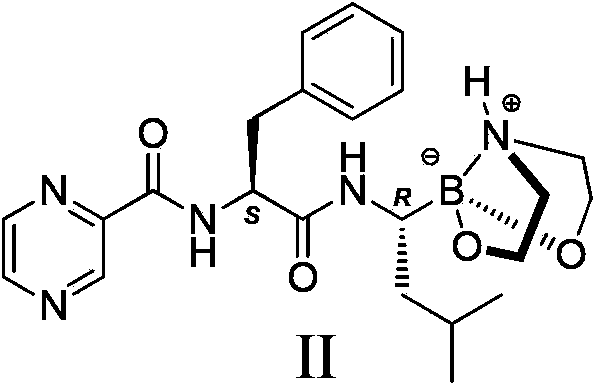
Crystal form of bortezomib key intermediate, and preparation method and application of crystal form
The invention discloses a novel crystal form of a bortezomib key intermediate II for the first time, provides a novel crystal form of a bortezomib key intermediate, namely (2S)-N-[(1R)-1-(1,2,6,2-dioxo-aza-boron-octane-2-yl)-3-methyl butyl]-3-phenyl-2-pyrazine-2-yl-formamido) propanamide (compound II), namely an alpha crystal form, and discloses a preparation of the compound II, a preparation method of the alpha crystal form, and a method for preparing high-purity bortezomib by using the alpha crystal form intermediate II. The alpha crystal form of the compound II is stable in property, and is conductive to purification of the bortezomib, long-term storage under conventional storage and transportation conditions and especially, development of industrial purification of the bortezomib.
Owner:朱继东
Method for efficiently preparing lithium carbonate
InactiveCN108892157AExcellent purityExcellent yieldMagnesium carbonatesLithium carbonates/bicarbonatesLithium carbonateChloride
The invention discloses a method for efficiently preparing lithium carbonate. The method specifically comprises the following steps: 1, performing filtering pretreatment on carbonic acid type salt lake brine, and regulating the pH value to be 3.4-4.8 so as to obtain first liquid; 2, adding magnesium chloride into the first liquid, uniformly mixing and stirring, performing solid-liquid separation,evaporating and concentrating the liquid part, and treating in an electric field so as to prepare second liquid; 3, adding an alkaline solution into the second liquid, uniformly mixing and stirring, and performing solid-liquid separation to obtain third liquid; 4, fully stirring the third liquid, an aqueous dispersant and loofah extract in a hydrothermal synthesis reactor, sealing the reactor, heating the reactor and reacting, and filtering the slurry in the reactor so as to obtain a lithium carbonate filter cake; and 5, washing the lithium carbonate filter cake with deionized water, and drying, thereby obtaining the finished product. According to the method disclosed by the invention, different interfering ions are removed by different steps, and the purity and yield of the obtained lithium carbonate are better than those of the conventional method.
Owner:江西金辉锂业有限公司
Process for Producing Coagulated Latex Particles
InactiveUS20090124778A1Excellent yieldWithout deteriorate original qualityCosmetic preparationsOther chemical processesInorganic saltsSoftening point
An object of the present invention is to provide a new granulating process which can recover desired coagulated latex particles with a significantly excellent yield, and can suppress secondary coagulation under the condition having a higher temperature than a softening point of a polymer, without deteriorating the original quality of the polymer itself. A process for producing coagulated latex particles which can suppress the secondary coagulation under the condition having the broad temperature range, without deteriorating the original quality of the polymer itself by spraying or dropping a polymer latex into a gas-phase containing an inorganic salt and / or an acid, and a dispersant in an aerosol form, and dropping or feeding the droplets of the polymer latex into an aqueous phase containing a dispersant.
Owner:KANEKA CORP
Method for producing disodium uridine-5'-monophosphate by a continuous flow reaction device
PendingCN110981928AShort reaction timeExcellent selectivitySugar derivativesChemical/physical/physico-chemical microreactorsReagentAnalytical chemistry
The invention discloses a method for producing disodium uridine-5'-monophosphate by a continuous flow reaction device. The method includes the following steps: A, preparing a mixed solution by simultaneously pumping solutions for preparing the mixed solution into a mixer, performing uniform mixing, pumping the mixed solution into a mixed solution storage tank to obtain the mixed solution; B, preparing a uridylic acid reaction solution by mixing a matched reagent and the mixed solution to prepare the uridylic acid reaction solution; and C, preparing the disodium uridine-5'-monophosphate: performing salt formation on the uridylic acid reaction solution, performing crystallization, performing filtration, and performing drying to obtain the disodium uridine-5'-monophosphate. In the preparationmethod for the disodium uridine-5'-monophosphate provided by the invention, the reaction time is shorter, in the reaction process, the mass transfer effect of a microchannel reactor is good, so thatthe selectivity and yield of the reactor are better than those of a previous preparation method, and the yield of the uridylic acid is about 97%.
Owner:美亚药业海安有限公司
Hybrid maize 37F73
ActiveUS20050120438A1Excellent yieldExcellent drought tolerancePlant genotype modificationPlant cellsMutantGenetic Materials
A novel hybrid maize variety designated 37F73 and seed, plants and plant parts thereof, produced by crossing two Pioneer Hi-Bred International, Inc. proprietary inbred maize lines. Methods for producing a maize plant that comprises crossing hybrid maize variety 37F73 with another maize plant. Methods for producing a maize plant containing in its genetic material one or more traits introgressed into 37F73 through backcross conversion and / or transformation, and to the maize seed, plant and plant part produced thereby. This invention relates to the hybrid seed 37F73, the hybrid plant produced from the seed, and variants, mutants, and trivial modifications of hybrid 37F73. This invention further relates to methods for producing maize lines derived from hybrid maize variety 37F73 and to the maize lines derived by the use of those methods.
Owner:PIONEER HI BRED INT INC
Method and apparatus for circular knitting with elastomeric yarn that compensate for yarn package relaxation
ActiveUS20060242999A1Excellent yieldConstant tensionWarp knittingCircular knitting machinesYarnUniform property
When elastomeric yarns are unwound and fed to the needles of a circular knitting apparatus, a yarn package relaxation profile is set, or determined, with corresponding adjustments made to the unwinding speed based on such package relaxation profile. Variably compensating for a varying package relaxation while knitting results in more uniform properties in a circular knit fabric and improved elastomeric yarn yield.
Owner:THE LYCRA CO LLC
Burner
InactiveCN105090960AReduce the temperatureIncrease the cross-sectional areaGaseous fuel burnerCombustorNitrogen oxides
The invention discloses a burner. The burner comprises a shell body, a burning torch and a center wind passage. The center wind passage is arranged in the shell body. The burning torch is arranged in the center wind passage. The shell body is provided with an air inlet. The center wind passage is connected with the air inlet. The periphery of the center wind passage is provided with at least one smoke protection channel. The smoke protection channel / channels is / are connected with a smoke inlet. A fuel torch / fuel torches is / are arranged in the smoke protection channel / channels. The fuel torch / fuel torches is / are connected with a fuel inlet. A fuel medium passage / passages is / are arranged in the smoke protection channel / channels, so that the fuel density on fire is reduced; the interval on fire is extended; the sectional area of the initial flame is enlarged; the temperature of the flame high-temperature region is effectively decreased. Therefore, the generation conditions of nitrogen oxide are effectively restrained, combustion is full, and the generation of the nitrogen oxide is reduced. Meanwhile, the service life of a temperature-resistant component / components on the head portion / portions of the fuel torch / fuel torches can be greatly prolonged due to the cooling protection of smoke.
Owner:SHANGHAI QIYAO THERMAL ENERGY ENG CO LTD +1
Method for synthesizing penciclovir analogue
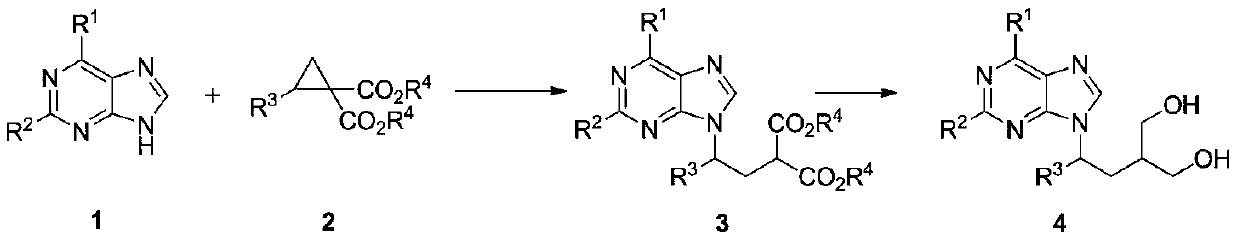
The invention discloses a method for synthesizing a penciclovir analogue and belongs to the technical field of medicine intermediate synthesis. The method comprises the following steps: by taking purine 1 and amino cyclopropane 2 as raw materials, and lewis acid as a catalyst, carrying out a molecular sieve reaction, thereby obtaining non-cyclic purine nucleoside 3. The reaction is single in areaselectivity and has a medium or excellent yield. The non-cyclic purine nucleoside 3 can be further reduced by using a reducing agent to obtain the penciclovir analogue. The method is easy in raw material obtaining and simple to operate, and a novel way is provided for synthesis of penciclovir analogues.
Owner:HENAN NORMAL UNIV
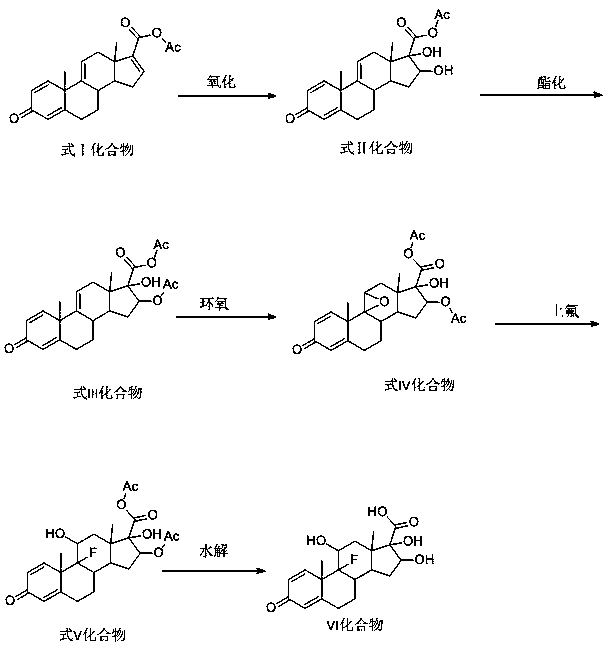
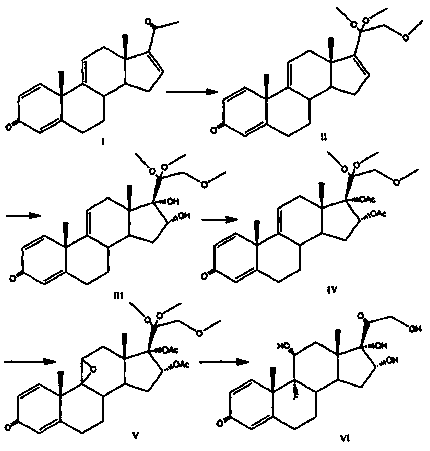
Triamcinolone preparation method
The invention discloses a triamcinolone preparation method, and belongs to the technical field of pharmacy. The triamcinolone preparation method comprises: selecting a tetraenyl acetate material as araw material, carrying out oxidation, esterification at 16 site, epoxidation and fluorination, and hydrolyzing after the fluorination refining to obtain triamcinolone. According to the present invention, the yield and the cost of the method of the present invention are obviously superior to the synthesis method sequentially comprising oxidation and direct epoxidation, wherein the impurity is removed after the fluorination process, such that the problem that the impurity is difficultly removed in the final refining can be avoided, and the impurity can be controlled at the intermediate stage soas to reduce the difficulty in the separation and purification.
Owner:SHANDONG TAIHUA BIO & TECH
Method for producing 5-nitroguaiacol sodium salt by using micro-channel reaction device
PendingCN111333515AShort reaction timeThe reaction temperature is low and controllableOrganic compound preparationCarboxylic acid esters preparationAcetic acidAcetic anhydride
The invention discloses a method for continuously producing 5-nitroguaiacol sodium salt by using a micro-channel reaction device. The method comprises the following steps of: (1) pumping a guaiacol-acetic anhydride solution containing an acidic catalyst into a micro-channel reactor I for reaction to obtain an acetyl guaiacol solution; (2) respectively pumping the cooled acetyl guaiacol solution and a nitric acid-acetic acid mixed solution into a micro-channel reactor II simultaneously for reaction to obtain a 5-nitroacetyl guaiacol solution; (3) cooling the 5-nitroacetyl guaiacol solution, pouring the 5-nitroacetyl guaiacol solution into water for crystallization, performing filtering, and collecting filter residue to obtain a 5-nitroacetyl guaiacol wet product; and (4) adding the 5-nitroacetyl guaiacol wet product into a sodium hydroxide solution, carrying out heating reaction, at the end of the reaction, conducting cooling, crystallizing and filtering, collecting the filter residue,and performing drying to obtain the 5-nitroguaiacol sodium salt. According to the method, the utilization rate of acetic anhydride reaches 95% or above, the nitration reaction yield is increased to 90% or above, and the three-step yield of 5-nitroguaiacol sodium salt is 80% or above.
Owner:郑州郑氏化工产品有限公司
Process for producing porous polymer particles
According to the invention, there is provided a process for producing porous polymer particles, which includes: dissolving in an organic solvent a monomer mixture containing an aromatic vinyl monomer and an aromatic divinyl monomer as major components together with a polymerization initiator to obtain a monomer solution; dispersing the monomer solution in water containing a dispersant and sodium nitrite to obtain a suspension polymerization dispersion; suspension-polymerizing the monomers in the suspension polymerization dispersion to yield porous polymer particles; and separating the porous polymer particles by filtration through a filter medium, in which the sodium nitrite is incorporated into the water in an amount in the range of 0.005 to 0.1 mg per 1 g of the water.
Owner:NITTO DENKO CORP
Double-working-media natural circulating bin type solar dryer
ActiveCN102818431AGood value for moneyExcellent yieldSolar heat devicesDrying machines with local agitationNatural ventilationEngineering
A double-working-media natural circulating bin type solar dryer can realize high-efficient heating and dewatering of things to be dried by controllable indirect heating and convection drying manner on the premise of no consumption of auxiliary energy and no power drive during dewatering, drying and long-term safety storage. Further, by temperature regulating and dehumidifying, the inside environment of a storage device can be kept dry by natural ventilation, and damage caused by insects, birds and rats and dust pollution are prevented; energy saving, emission reduction and low-carbon effect are achieved, production management cost is reduced, and safety, sanitation and long-term storage of storage things are guaranteed. The double-working-media natural circulating bin type solar dryer is widely applicable to rural households and massive production and application in the fields of the industry and the agriculture.
Owner:涂济民 +1
H9 subtype avian influenza virus Vero cell adapted strain and application thereof
ActiveCN102586194AExcellent growth rateExcellent yieldMicroorganism based processesAntiviralsEmbryoFlu immunization
The invention discloses a H9 subtype avian influenza virus Vero cell adapted strain and application of the Vero cell adapted strain in preparing of H9 subtype avian influenza vaccines. When being applied to Vero cells, viral multiplication speed and yield of the adapted strain are all superior to the existing avian influenza virus cultured through chicken embryos and superior to viral strains of non-naturalized H9 subtype avian influenza virus. The H9 subtype avian influenza virus Vero cell adapted strain can replace the chicken embryos to massively produce influenza viruses fast and has wideapplication prospect.
Owner:哈药集团生物疫苗有限公司
Method of preparing high purity zinc phosphate by mechanical activation continuous reaction
ActiveCN108946693ASemi-continuous processSimple production processPhosphorus compoundsSulfate radicalsZinc Phosphate Cement
The invention discloses a method of preparing high purity zinc phosphate by a mechanical activation continuous reaction. The method comprises the following steps: S1, dissolving soluble carbonate or hydrogen carbonate and soluble zinc salt in water, separately to prepare a carbonate or hydrogen carbonate solution and a zinc salt solution; S2, mechanically activating the zinc salt solution and thecarbonate or hydrogen carbonate solution in a first reaction kettle synergically in a wet process for a reaction to prepare a precipitate; S3, conveying the precipitate and a solution to a second reaction kettle, adding a phosphoric acid solution, and carrying out a reaction in a mechanical activating condition in a synergic wet process to prepare zinc phosphate. The yield of the obtained productreaches 97.89-98.34%, the content of zinc phosphate dehydrate is 99.27-99.56%, the content of sulfate radicals is 0.032-0.041%, the content of chlorine ions is 0.028-0.042% and the content of lead is0.0012-0.0019%. Compared with the prior art, the output and quality of the product are superior.
Owner:广西科立方新材料有限公司 +2
Stator and resolving method and device thereof
InactiveUS20050172477A1Easily resolveExcellent yieldWindings insulation shape/form/constructionMagnetic circuit stationary partsStatorEngineering
The method for resolving the stator having a coil disposed in the slots provided around an inner circumferential face of a cylindrical iron core and having an opening portion includes a step of enlarging the opening portion of the slot, and a step of extracting the coil from the slot.
Owner:MITSUBISHI ELECTRIC CORP
Method for reducing early bolting of Chinese angelica
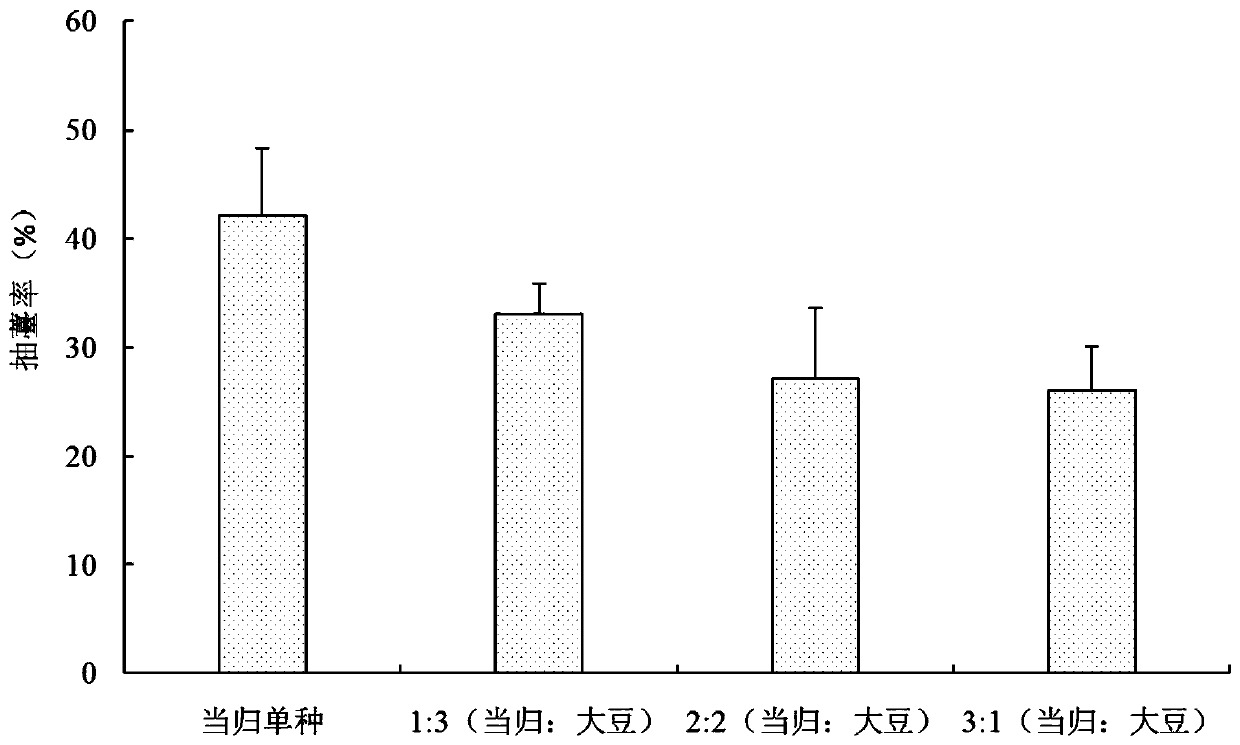
PendingCN111133969APromote hydrationAdequate nutritionClimate change adaptationFabaceae cultivationDiammonium phosphateBolting
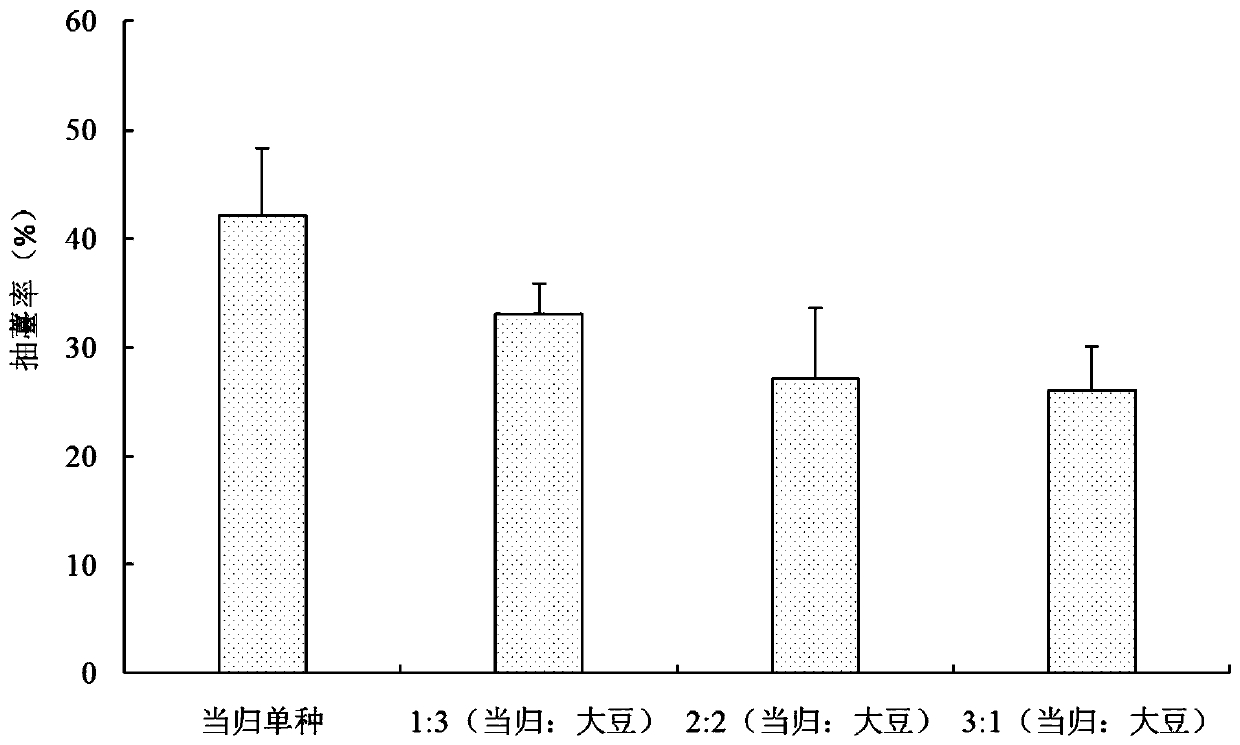
The invention discloses a method for reducing the early bolting of Chinese angelica. The method includes selecting black soil and heilu soil with excellent conditions to perform sowing, the period ofcrop rotation being more than 3 years, performing shallow ploughing for one time before sowing, applying well-rotted farmyard manure, urea, diammonium phosphate and potassium sulfate before the soil is turned up, and killing pests; then transplanting seedlings, and selecting soybean seeds to perform dibbling; performing sterilization on the seedlings before transplanting, and performing air-dryingwhile performing soaking; performing transplanting in mid-to-late March, performing covering with wide films to perform dibbling, and planting one row of soybeans and three rows of Chinese angelica after mulching; during the transplanting of the Chinese angelica, respectively planting one seedling on the two sides of each hole, the distances of the terminal bud on the heads of the seedlings of the Chinese angelica from the mulching films being 2-3 cm, performing compacting with fine soil, and tightly sealing broken gaps; and during the dibbling of the soybeans, putting 2 soybean seeds in eachhole, performing compacting with the fine soil, and tightly sealing the broken gaps. The bolting rates after the intercropping of the Chinese angelica and the soybeans are lower than that of the monoculture of the Chinese angelica; the influence of different intercropping ratios on the bolting rates is different; when the Chinese angelica is intercropped with the soybeans in a ratio of 3:1, the bolting rate of the Chinese angelica is the lowest, which is only 26.06%; and the annual growth rate of the Chinese angelica can reach up to 66%.
Owner:CHINA ACAD OF SCI NORTHWEST HIGHLAND BIOLOGY INST
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com