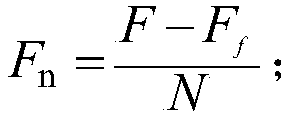Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
279results about How to "The implementation method is simple" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
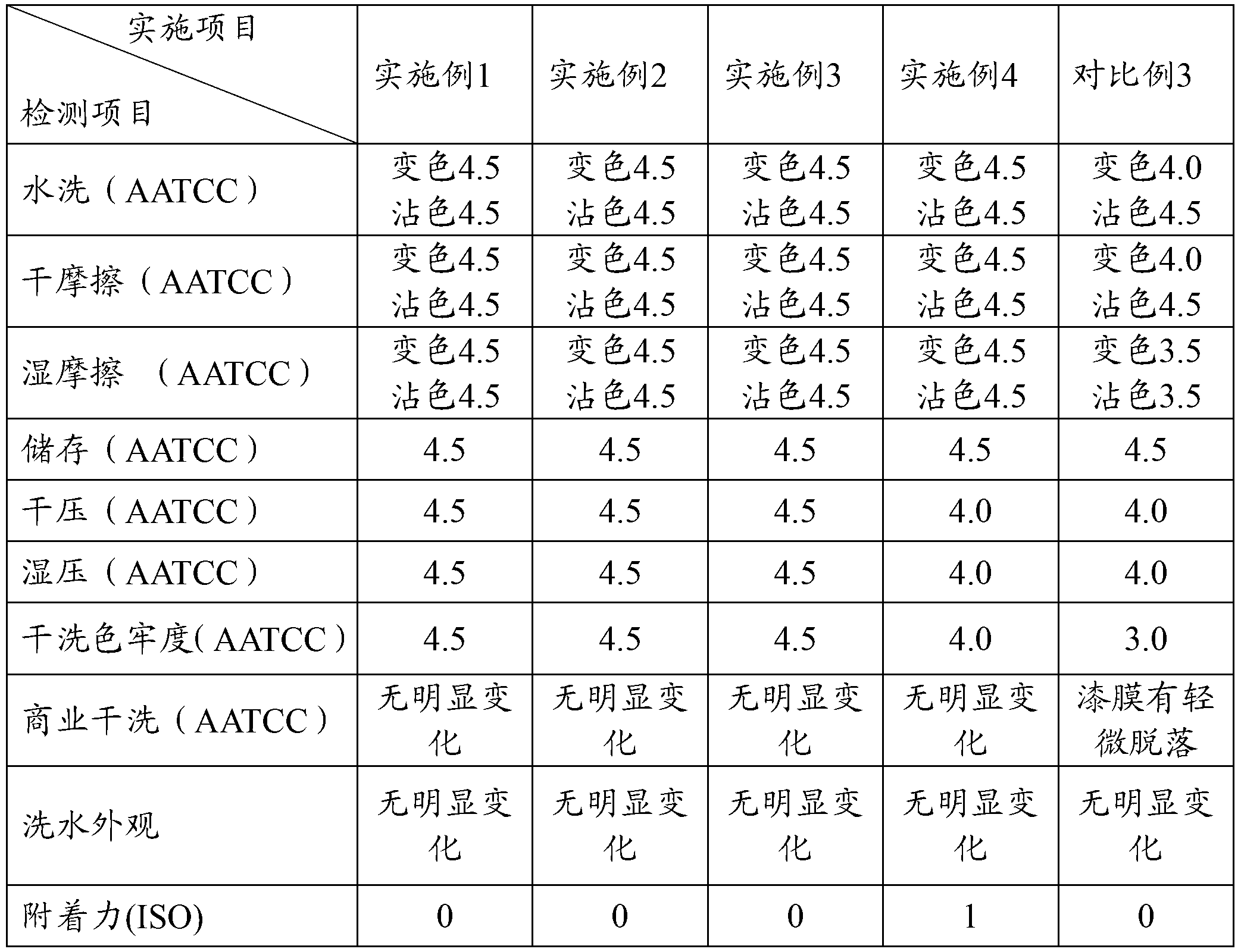
Terahertz radiation detector
InactiveCN103575407AThe implementation method is simpleLow costSolid-state devicesFluid speed measurementTerahertz radiationAbsorbed energy
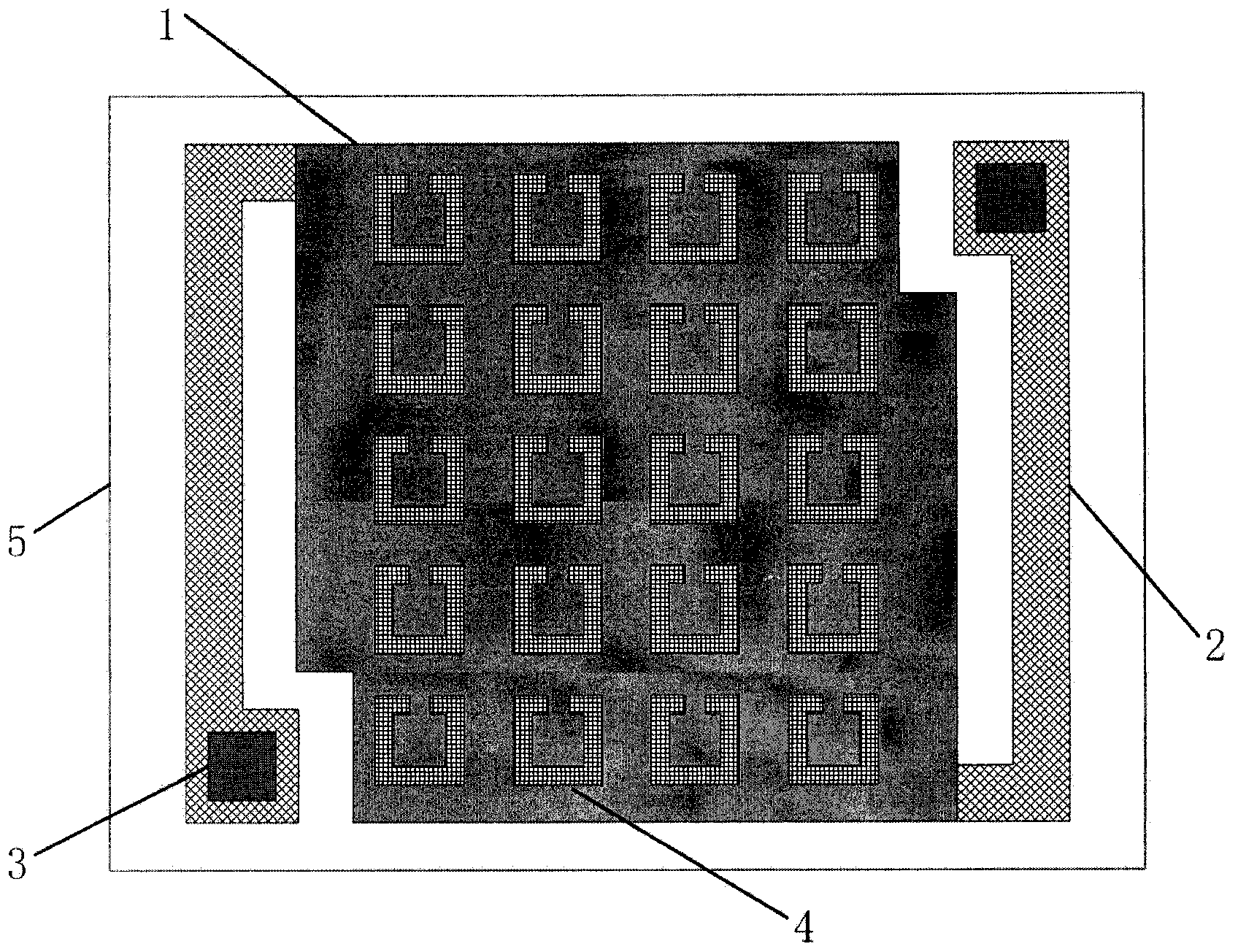
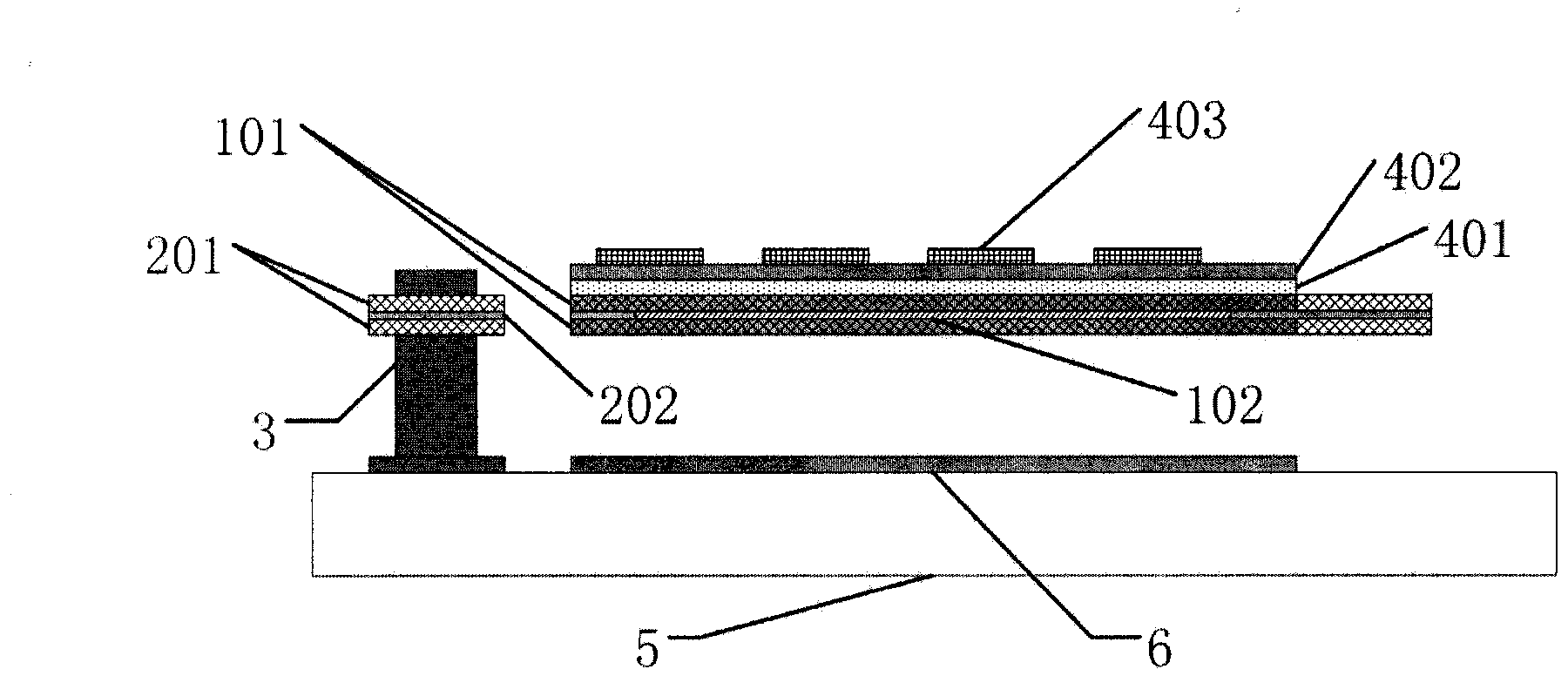
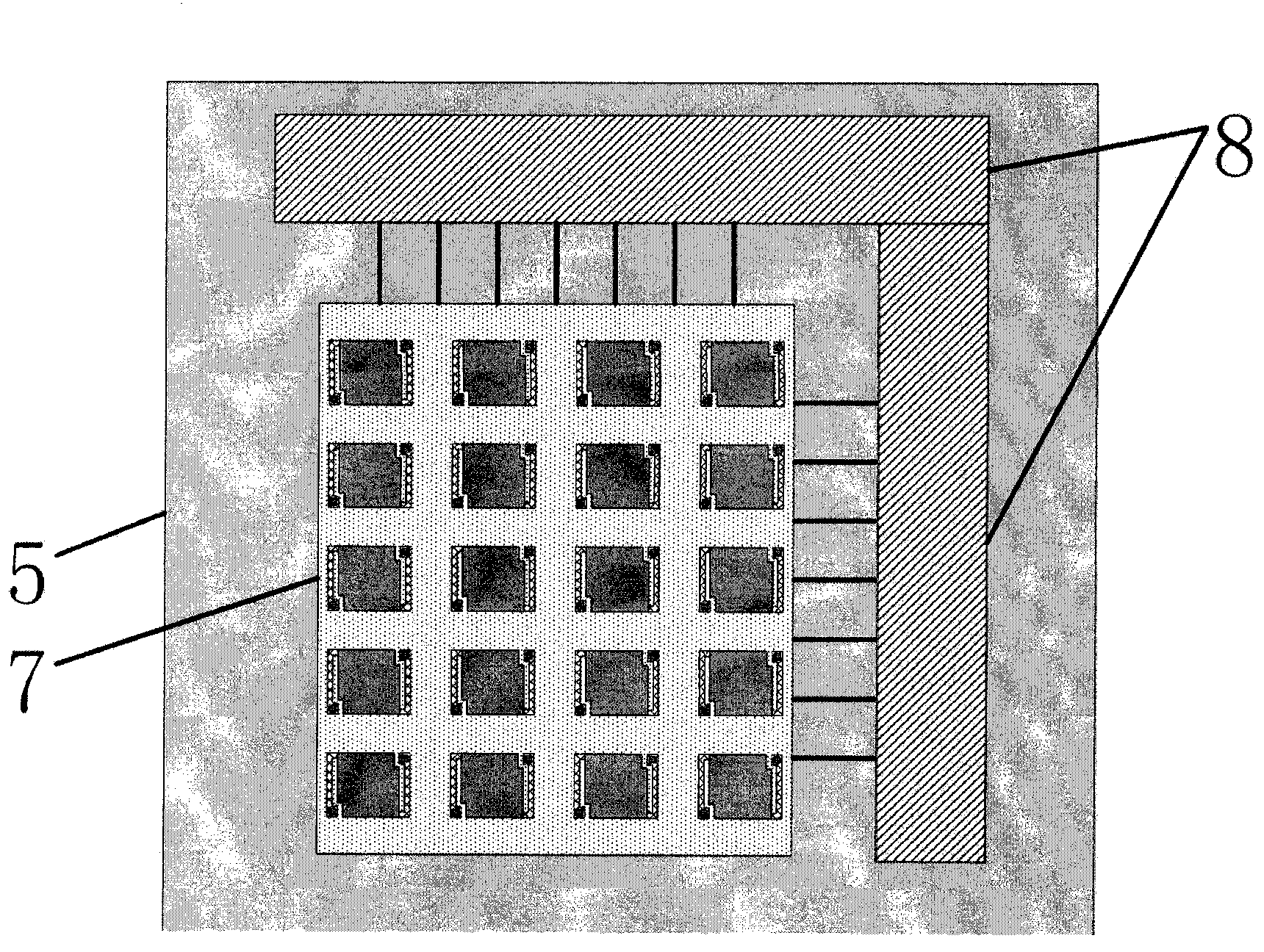
The invention provides a terahertz radiation detector. The terahertz radiation detector comprises a terahertz sensitive structure and a substrate chip including a reading circuit, wherein the terahertz sensitive structure comprises a terahertz absorption structure, a heat conversion layer and a protection layer, when passive / active terahertz waves are focused on the terahertz sensitive structure through a terahertz object lens, absorbed energy is converted into heat energy through the terahertz absorption structure, the heat energy is converted into an electric signal through the heat conversion layer, and the electric signal is read through the reading circuit on the substrate chip. According to the terahertz radiation detector, a terahertz micro-bolometer micro-bolometer operates under a non-refrigeration environment, single point detection can be carried out, terahertz imaging can further be realized through a focal plane array.
Owner:PEKING UNIV
Super high molecular mass polythylene/carbon nano tube composite fiber used in jelly glue spinning and its preparation
InactiveCN1431342AImprove performanceAvoid damageMonocomponent polyolefin artificial filamentFiberRefractory
A gel-spinning ultrahigh-molecular-weight composite polyethene (UHMWPE) / carbon nanotube fibre is composed of UHMWPE and carbon nanotubes (0.01-5 wt.%) as modifier. It is prepared through purifying and organizing carbon nanotubes, preparing gel, and spinning. Its advantages are simple preparing process and high refractory and anticreep performance.
Owner:DONGHUA UNIV
Temperature-resistant salt-tolerant viscous-oil biological viscosity reducer and preparation method thereof
ActiveCN105154050AAvoid scaling and corrosionThe implementation method is simpleDrilling compositionLiquid mediumTemperature resistance
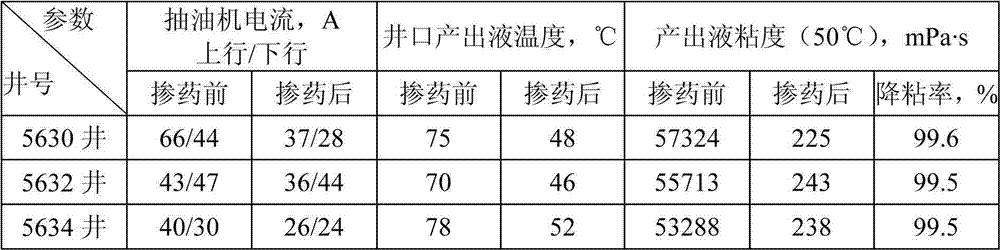
The invention discloses a temperature-resistant salt-tolerant viscous-oil biological viscosity reducer and a preparation method thereof, and belongs to the technical field of petroleum exploitation. The temperature-resistant salt-tolerant viscous-oil biological viscosity reducer is composed of microzyme, a seed liquid medium, a fermentation liquor medium and water. The microzyme is torulopsis candida. The preparation method of the viscosity reducer comprises (1) preparation of a microzyme seed liquid; (2) early-stage preparation of the fermentation liquor medium; (3) fermentation production of microzyme; (4) collection of a fermentation product; and (5) preparation of the viscous-oil biological viscosity reducer. The viscous-oil biological viscosity reducer is green and free of pollution by employing agricultural and sideline products as raw materials. The provided viscous-oil biological viscosity reducer possesses the temperature resistance up to 150 DEG C, the salt tolerance up to 2*10<5> mg / L, and the viscous-oil viscosity reduction rate up to 99% or more. The enforcement method of the viscous-oil biological viscosity reducer is simple. The viscous-oil biological viscosity reducer is substantial in effect, is capable of saving energy, reducing consumption and preventing pipelines from fouling and being corroded, and the subsequent crude-oil demulsification dewatering processing is easy. Therefore, the viscous-oil biological viscosity reducer is widely applicable to shaft elevation of a viscous oil well and ground pipeline conveying technology.
Owner:CHINA PETROLEUM & CHEM CORP +1
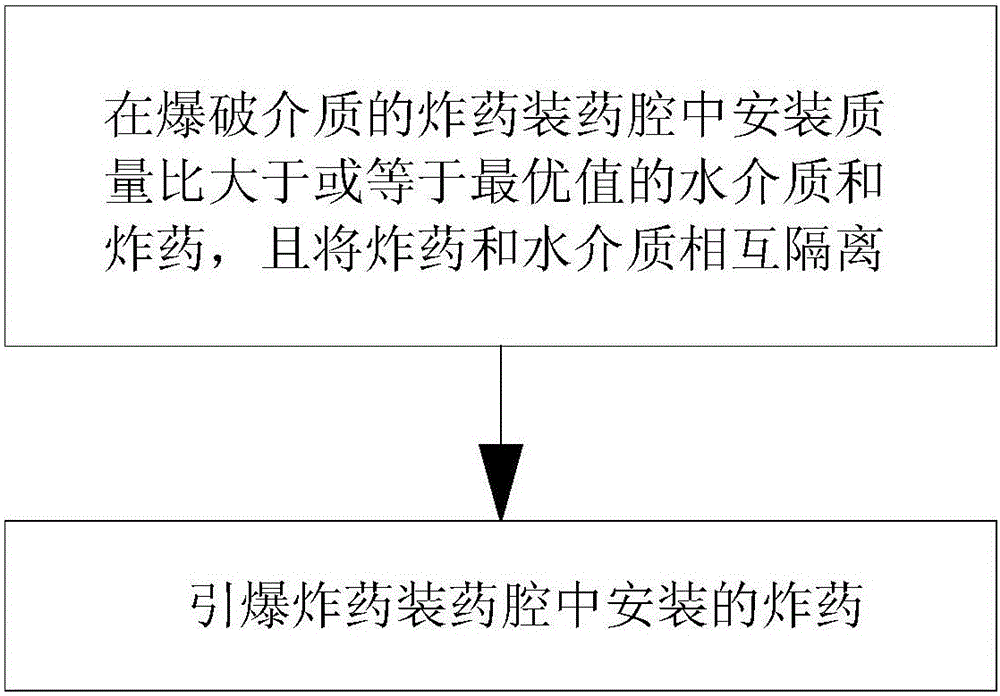
Aqueous medium energy transition blasting method and charging cavity
ActiveCN105674819ASolve the problem of insufficient effective utilization of energyAir Shock MitigationBlastingHydrogenMass ratio
The invention discloses an aqueous medium energy transition blasting method and charging cavity. The aqueous medium energy transition blasting method comprises the steps that firstly, an aqueous medium and explosives with the mass ratio larger than or equal to an optimal value are mounted in the explosive charging cavity of a blasting medium, and the explosives and the aqueous medium are isolated from each other; and then the explosives mounted in the explosive charging cavity are detonated. The aqueous medium and the explosives with the mass ratio larger than or equal to the optimal value M are mounted in the charging cavity, the explosives and the aqueous medium are isolated from each other, and the optimal value M is the ratio of explosion heat of the explosives adopted in the blasting process to heat energy released when water is synthesized by hydrogen and oxygen. The aqueous medium energy transition blasting method and charging cavity have the beneficial effects that the effective utilization rate of energy of the explosives can be increased, the blasting effect of the explosives on the blasting medium can be improved, blasting damage can be reduced, and blasting smoke is less.
Owner:秦健飞
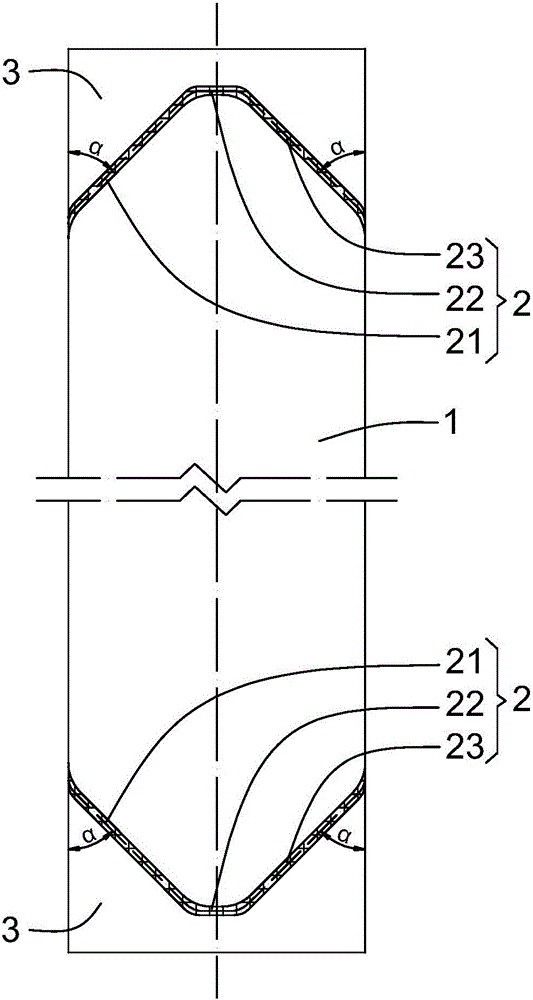
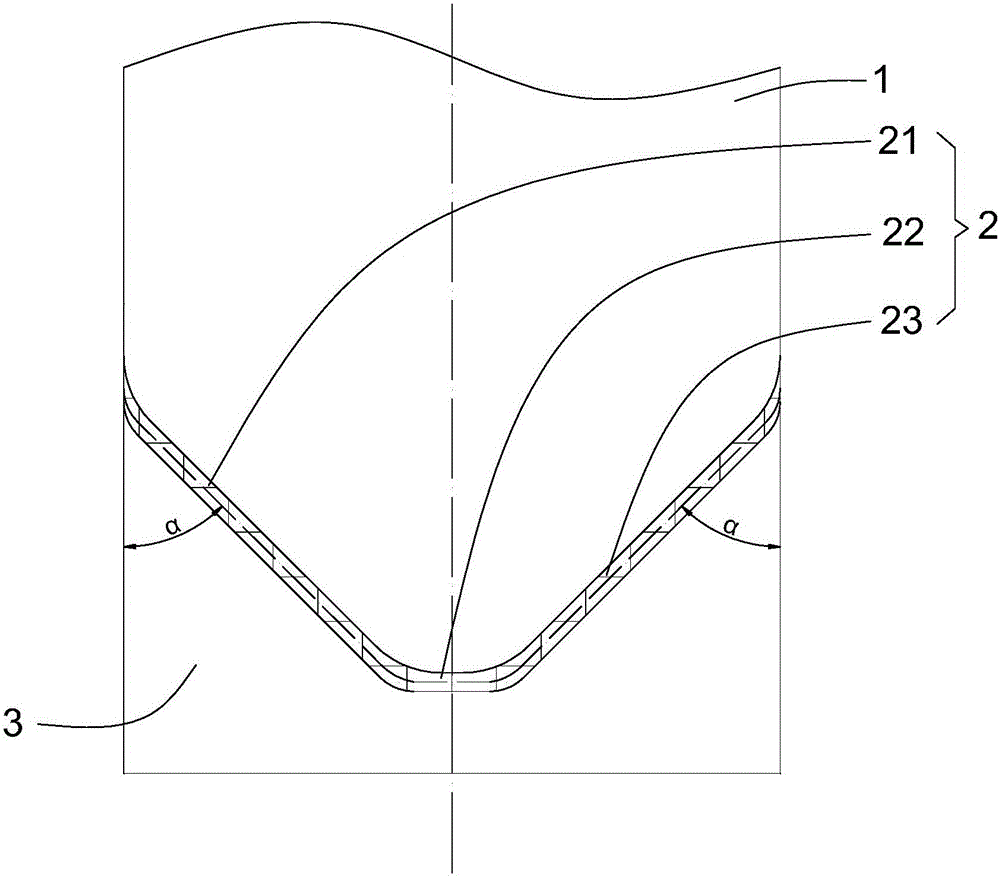
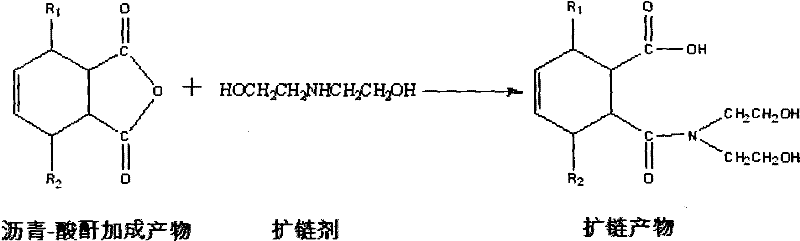
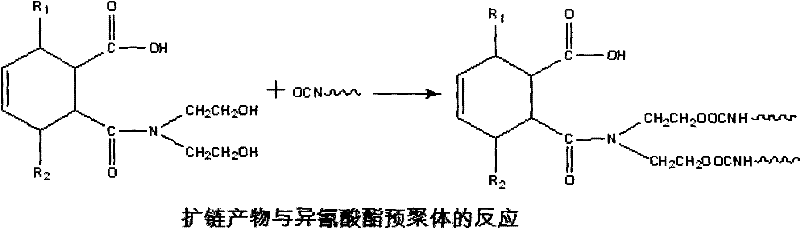
Emulsified asphalt with strong compatibility, and preparation method thereof
ActiveCN102190895AThe implementation method is simpleBroad application spaceBuilding insulationsPrepolymerIsocyanate
The invention relates to emulsified asphalt with strong compatibility, and a preparation method thereof. The preparation method comprises: heating base asphalt to a temperature of 100-160 DEG C to remove moisture, then sequential adding unsaturated anhydride and a catalyst, a hydrophilic chain extender, isocyanate prepolymer, wherein the isocyanate prepolymer is added in 2-3 hours through controlling feeding speed, followed by reacting for 2 hours to obtain a modified asphalt; preparing soap liquid by a emulsifier, hydrochloric acid and water, and heating the soap liquid to a temperature of 60-70 DEG C; heating the modified asphalt to a temperature of 130-150 DEG C, and emulsifying the soap liquid and the modified asphalt to obtain the modified emulsified asphalt. The modified emulsified asphalt comprises, by weight, 60-65% of the modified asphalt, 2-4.0% of an emulsifier, 0-1.0% of a stabilizer, 29.0-36.8% of water. The modified asphalt provided by the present invention has special structure. According to the present invention, emulsified asphalt after emulsifying is compatible with stone of different activities, such that a purpose of the strong compatibility is achieved.
Owner:PETROCHINA CO LTD +1
Multi-frequency base station antenna for eliminating coupling resonance
ActiveCN107546489AEliminate coupling resonanceReduce mutual couplingAntenna couplingsSeparate antenna unit combinationsCouplingResonance
The invention provides a multi-frequency base station antenna for eliminating coupling resonance. The multi-frequency base station antenna comprises a reflecting plate, a first low-frequency radiationarray, a second low-frequency radiation array and a first high-frequency radiation array, wherein the first low-frequency radiation array and the second low-frequency radiation array are arranged onthe reflecting plate, are parallel to each other and are arranged on the same radiation surface, the first high-frequency radiation array is arranged between the first low-frequency radiation array and the second low-frequency radiation array, the first low-frequency radiation array and the second low-frequency radiation array respectively comprises a plurality of low-frequency radiation units, the first high-frequency radiation array comprises a plurality of first high-frequency radiation arrays, the multi-frequency base station antenna also comprises isolation strips, and the isolation strips are parallel to the reflecting plate, are insulated with the reflecting plate, extend along central axes of the first low-frequency radiation array and the second low-frequency radiation array and are arranged between radiation surfaces of the first high-frequency radiation units and radiation surfaces of the first low-frequency radiation units and the second low-frequency radiation units. By the multi-frequency base station antenna, coupling among the arrays is reduced, and the technical effects of improving the isolation among the arrays and reducing resonances among the arrays are achieved.
Owner:COMBA TELECOM TECH (GUANGZHOU) CO LTD
Electricity-saving control device for interaction type intelligent drinking machine
InactiveCN101424923AAchieve intelligent power saving effectThe implementation method is simpleBeverage vesselsProgramme control in sequence/logic controllersMicrocontrollerElectricity
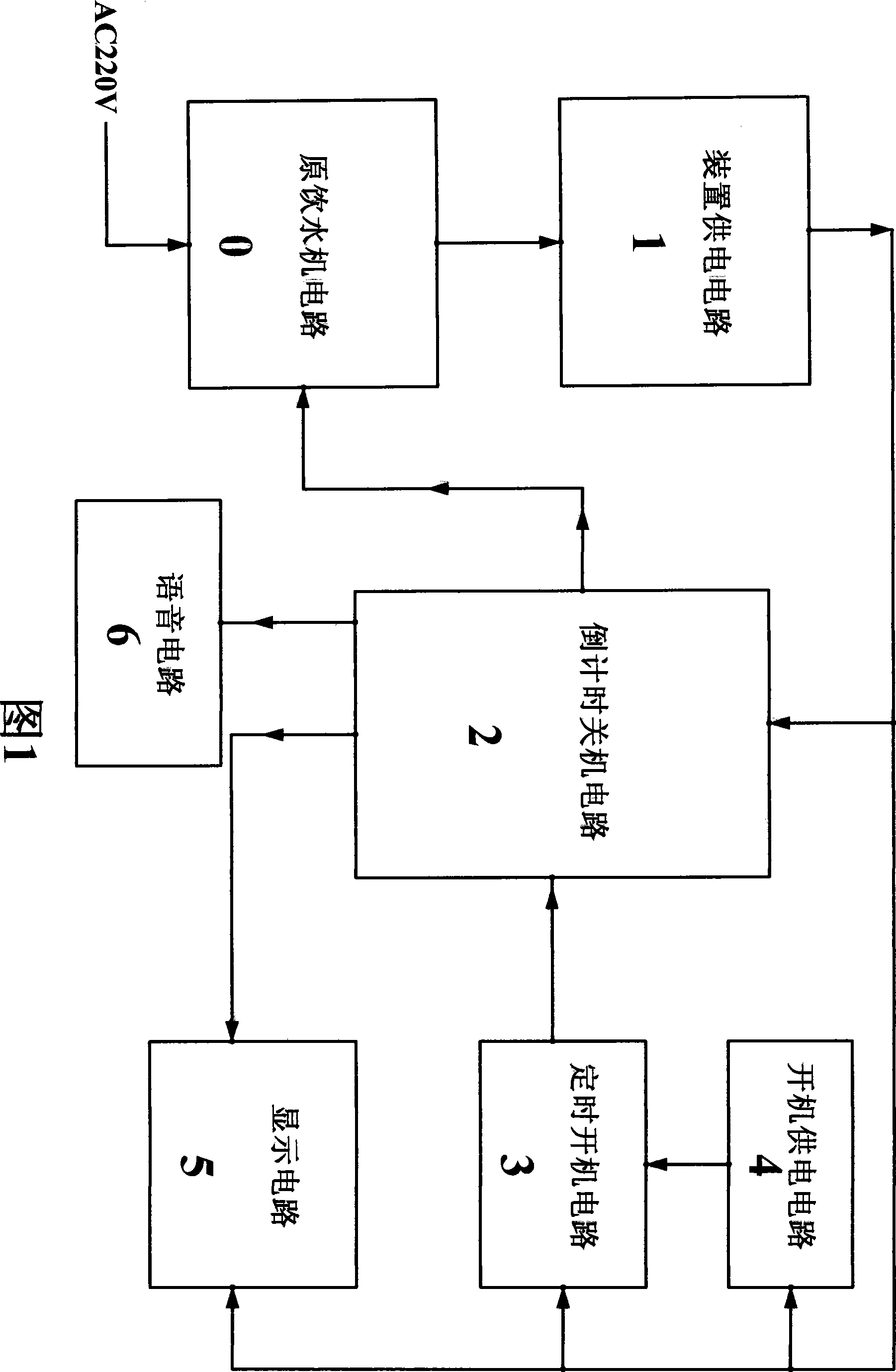
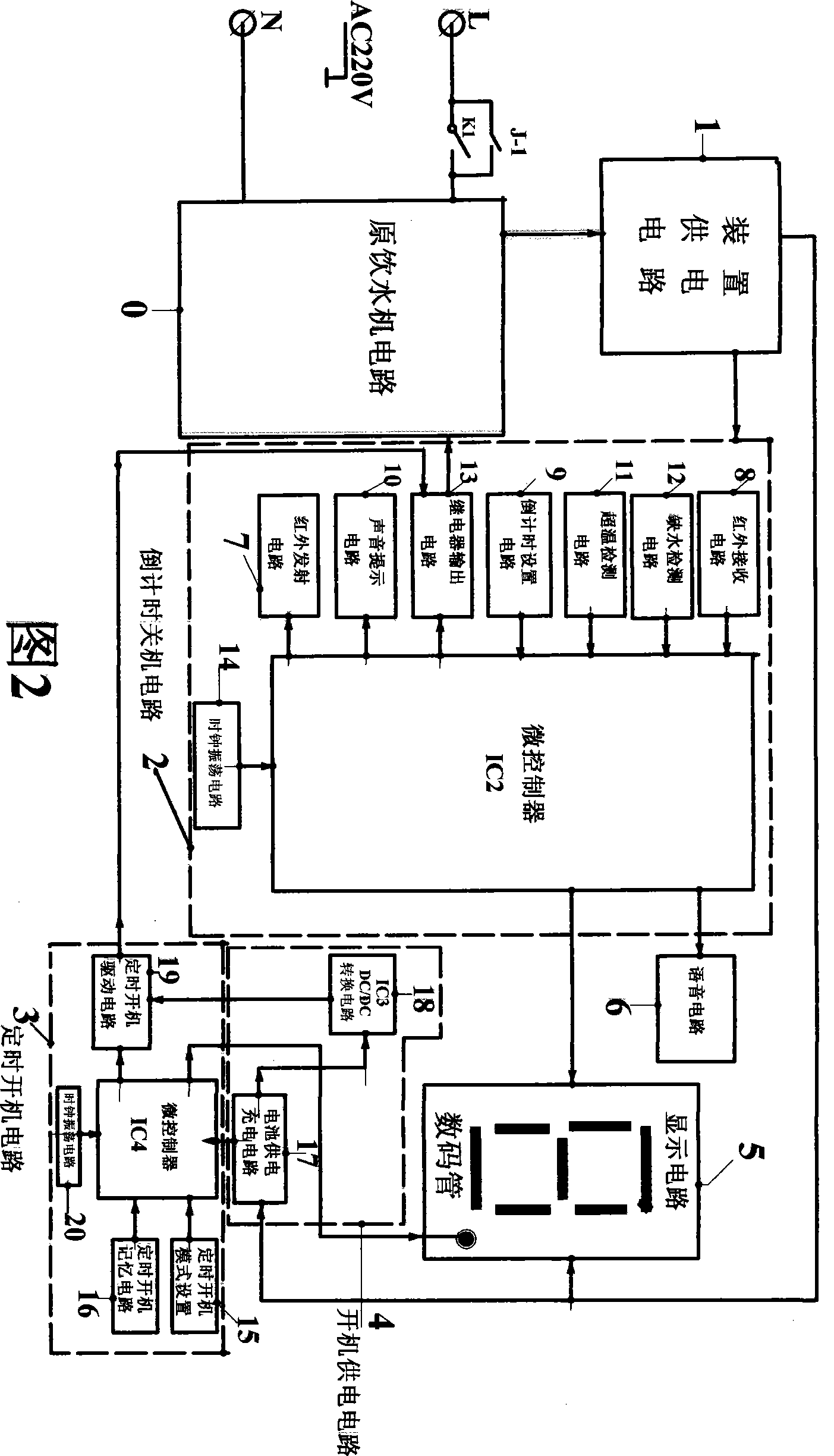
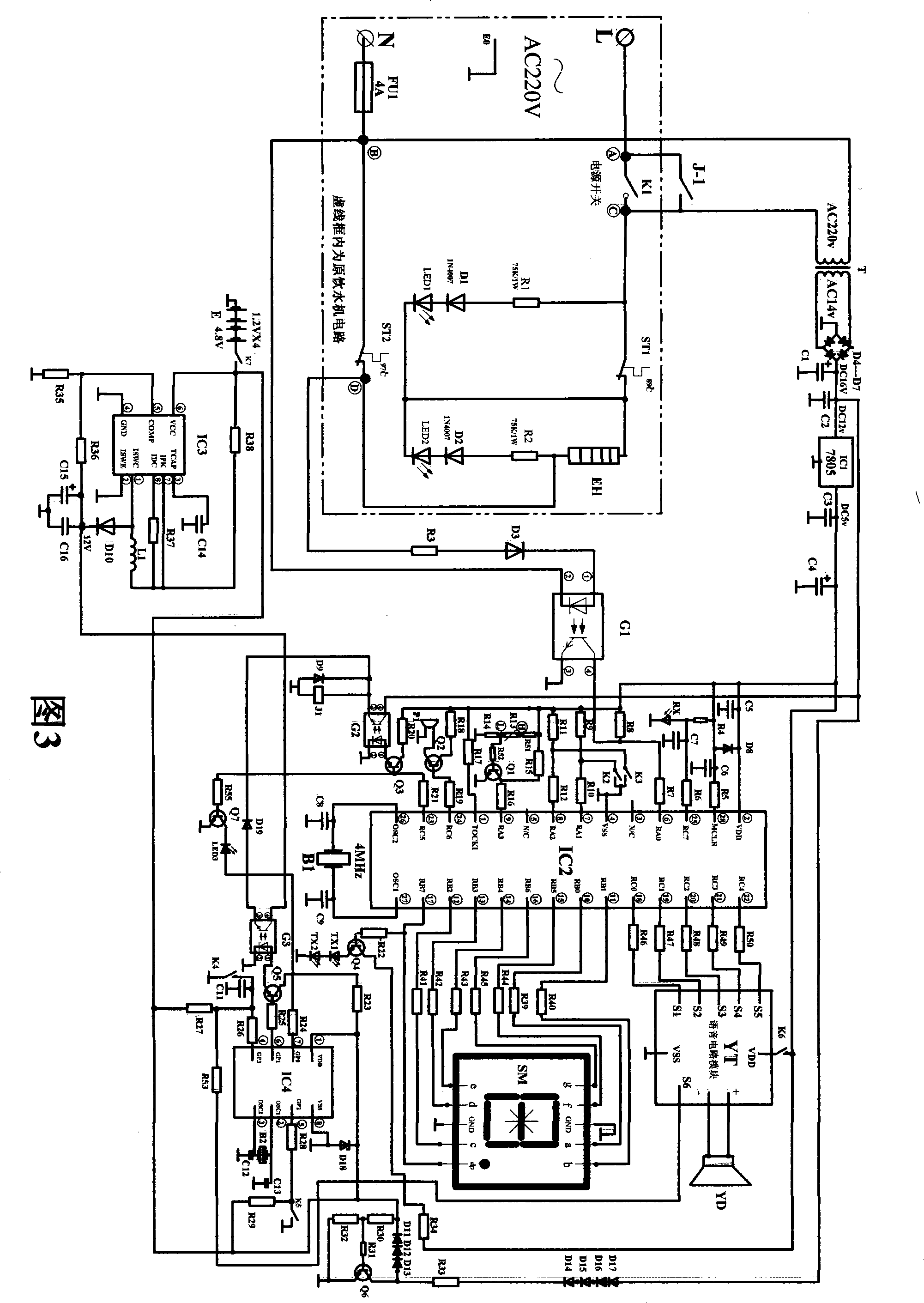
The invention discloses an electricity-saving controlling device of an interactive intelligent drinking machine, which relates to the application of a microcontroller on the aspects of saving electricity and controlling the drinking machine. The electricity-saving controlling device comprises a device feed circuit (1), a countdown shutdown circuit (2), a display circuit (5), a sound circuit (6), a timing starting circuit (3) and a starting feed circuit (4), wherein the input end of the device feed circuit (1) is connected with the input end of the commercial power of an original drinking machine circuit (0), and the output end of the device feed circuit (1) is connected with the circuits of the device. The countdown shutdown circuit (2) completes the functions of sending and the receiving of infrared signals, the overtemperature detection, water deficiency detection, the countdown, the control of the drinking machine power supply and the sound prompt. The display circuit (5) is connected with the display output end of the countdown shutdown circuit (2) so as to have countdown display. The timing starting circuit (3) completes the timing starting function through the starting feed circuit (4). The electricity-saving controlling device determines the shutdown time according to the specific using condition of a user and has corresponding sound and voice prompt, has no stand-by power consumption because the shutdown is in full shutdown state, and also has the function of timing starting in advance.
Owner:刘建国
Multi-polarized radiation oscillator and antenna
ActiveCN107808998AImprove performanceCompact structureAntenna supports/mountingsRadiating elements structural formsMagnetic dipoleElectric field
The invention provides a multi-polarized radiation oscillator and an antenna. The multi-polarized radiation oscillator comprises a radiation unit, a first group of polarization feed units and a secondgroup of polarization feed units. The radiation unit comprises a first group of polarization radiation units and a second group of polarization radiation units, which are mutually connected. Each group of polarization radiation units comprise two mutually-orthogonal polarization directions. Polarization electric field vector directions of two adjacent polarizations of the first group of polarization feed units and the second group of polarization feed units form a 45-degree included angle. The first group of polarization feed units are electric dipole units; and the second group of polarization radiation units are magnetic dipole units. The first group of polarization feed units feed the first group of polarization radiation units; and the second group of polarization feed units feed thesecond group of polarization radiation units. Since four polarizations share one radiation unit, and four linear polarizations are realized without combination of multiple oscillators, so that the multi-polarized radiation oscillator can enable antenna structure to be more compact, and size of the antenna is reduced.
Owner:COMBA TELECOM TECH (GUANGZHOU) CO LTD
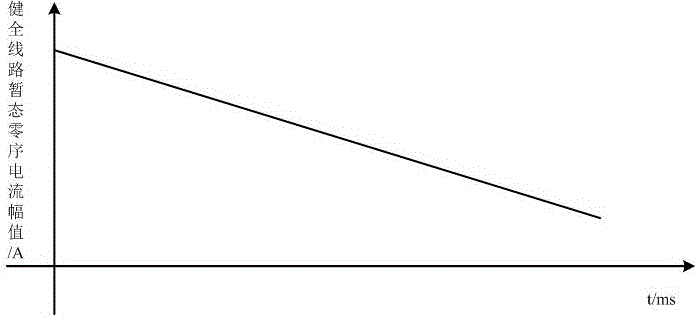
Small-current grounding fault positioning method employing transient zero sequence current
InactiveCN106443343AFound in timeRealize online monitoringFault location by conductor typesTransient stateCurrent amplitude
The invention relates to a small-current grounding fault positioning method employing a transient zero sequence current, and the method is especially used for a system of an instation no-selection-line device. The method comprises the steps: selecting at least three lines with the bigger transient zero sequence current through employing the transient zero sequence current amplitudes of all detection points at an outlet of a bus; determining that a jump point is a fault point through employing the characteristics that the monotone decreasing of amplitudes of the transient zero sequence currents of healthy lines and the monotone increasing of the amplitudes of the transient zero sequence currents of fault lines at first and then the monotone decreasing of the amplitudes of the transient zero sequence currents of fault lines, and determining that fault segment is a segment where the jump point is located. The method irons out the defects of the instation no-selection-line device, just employs the transient current signals, is wide in application range, is simple, and is easy to implement.
Owner:STATE GRID FUJIAN ELECTRIC POWER CO LTD +4
Hot rolling bar plate scythes bend and bias-running online detection method
InactiveCN101182984ASimple methodEasy to operateFurnace typesMechanical measuring arrangementsContinuous measurementEngineering
The invention discloses an online detection method for a hot rolling plate-blank camber and running deviation. The method is that: on the condition of the normal production of a continuous casting machine and a heating furnace, the centerline of the whole heating furnace, namely, the centerline is found out according to the width size of the surrounding of the heating furnace and the width size of the produced plate-blank, and then the horizontal distance from the plate-blank to a fixed point can be respectively measured and compared at the inlet and the outlet of the heating furnace, and continuous measurement is conducted for many times along the length direction of the plate-blank, at last, the graph of the plate-blank camber can be achieved and the intuitive description about the plate-blank camber and the shape of the camber can be realized; therefore, the origin of the problem can be analyzed and the good condition for later workstage rolling can be created.
Owner:GUANGZHOU PEARL RIVER STEEL & IRON
Field rock mass poisson ratio test method
InactiveCN104483199AFully reflect the real stateLow costMaterial strength using tensile/compressive forcesElectrical/magnetic solid deformation measurementElectrical resistance and conductanceBedrock
The invention discloses a field rock mass poisson ratio test method. The method comprises the steps of firstly digging a test flat hole in a mountain, drilling a cylindrical test piece on bedrock on the bottom surface of the test flat hole by utilizing a hollow drill bit, and symmetrically perforating test holes in the bottom of an annular groove at the periphery of the test piece; sleeving the test piece with an elastic steel lantern ring provided with a steel contact and a strain plate, connecting a strain plate test line and a resistance strain gauge, and calculating transverse deformation of the test piece by virtue of test; fixing the top surface of the test piece together with a test flat hole top plate sequentially through a pressure plate, a jack, a reducing connector steel plate, a combined force transfer rod and a hinge anchor, installing measuring points in bolt holes in two sides of the pressure plate and in the test holes in the bottom of the annular groove, and installing a dial indicator above the measuring point; calculating vertical deformation of the test piece after the load test; then calculating a corresponding transverse strain value and a vertical strain value, and calculating the poisson ratio of the test piece according to the transverse strain value and the vertical strain value. The field rock mass poisson ratio test method has advantages that the implementation method is simple and rapid, the measurement data is accurate and reliable, and the real condition of the rock mass can be comprehensively reflected.
Owner:YELLOW RIVER ENG CONSULTING
Highly-steep rock slope ecological restoration structure and implementation method
ActiveCN104314086AThe implementation method is simpleReduce construction costsExcavationsVegetative propogationVegetationSoil science
The invention is applicable to the field of rock slope ecological restoration and provides a highly-steep rock slope ecological restoration structure. The highly-steep rock slope ecological restoration structure comprises a soil fixing cage fixed on a rock slope, a soil dressing layer which is sprayed into the soil fixing cage and attaches to the surface of the rock slope, a plant sprout layer sowing on the soil dressing layer in a sprayed manner, and non-woven fabric covering the soil fixing cage and the plant sprout layer. The soil fixing cage comprises a plurality layers of wood plates mounted on the surface of the rock slope, a plurality of reinforced steel bars supporting the wood plates and a steel wire mesh mounted on the surface of the rock slope. The invention further discloses an implement method of the highly-steep rock slope ecological restoration structure. The highly-steep rock slope ecological restoration structure has the advantages that the soil dressing layer is fixed and falling off of the soil dressing layer is prevented by disposing the soil fixing cage on the highly-steep rock slope, rain wash resistance of the soil dressing layer is increased, a stable growing platform and environment is provided for plants, vegetation degradation is prevented, a good and appropriate growing environment is provided for the plants by the plate sprout layer, the plants can spout and grow fast, and ecological restoration speed is increased.
Owner:广东腾景养老产业运营管理有限公司
Button with hammer effect and preparation method of button
ActiveCN103194144AHas a hammer effectStrong visual impactClothes buttonsPolyurea/polyurethane coatingsDiluentShock resistance
The invention discloses a button with a hammer effect and a preparation method of the button. The button comprises a base material, and a coating with a hammer effect is sprayed onto the base material and is a mixture of hammer paint, a curing agent and a diluent with the ratio in parts by weight of 1: (0.2-0.3): 0.5. The preparation method comprises the following steps of: processing the base material: carrying out water-grinding treatment on the base material to be sprayed to guarantee that the surface of the base material is clean without pollution; spraying, wherein the viscosity of the coating is controlled for 14s-20s, and then the coating is sprayed onto the base material; and molding, namely naturally drying or stoving the base material with the coating. The button provided by the invention is strong in visual impact and decoration function, simple in preparation method and beneficial to waste reduction, and also has good performance of scratch resistance, friction resistance, shock resistance, washing durability and general chemical corrosion resistance; and the raw materials are easily available, and suitable materials are more.
Owner:广东康派环创科技有限公司
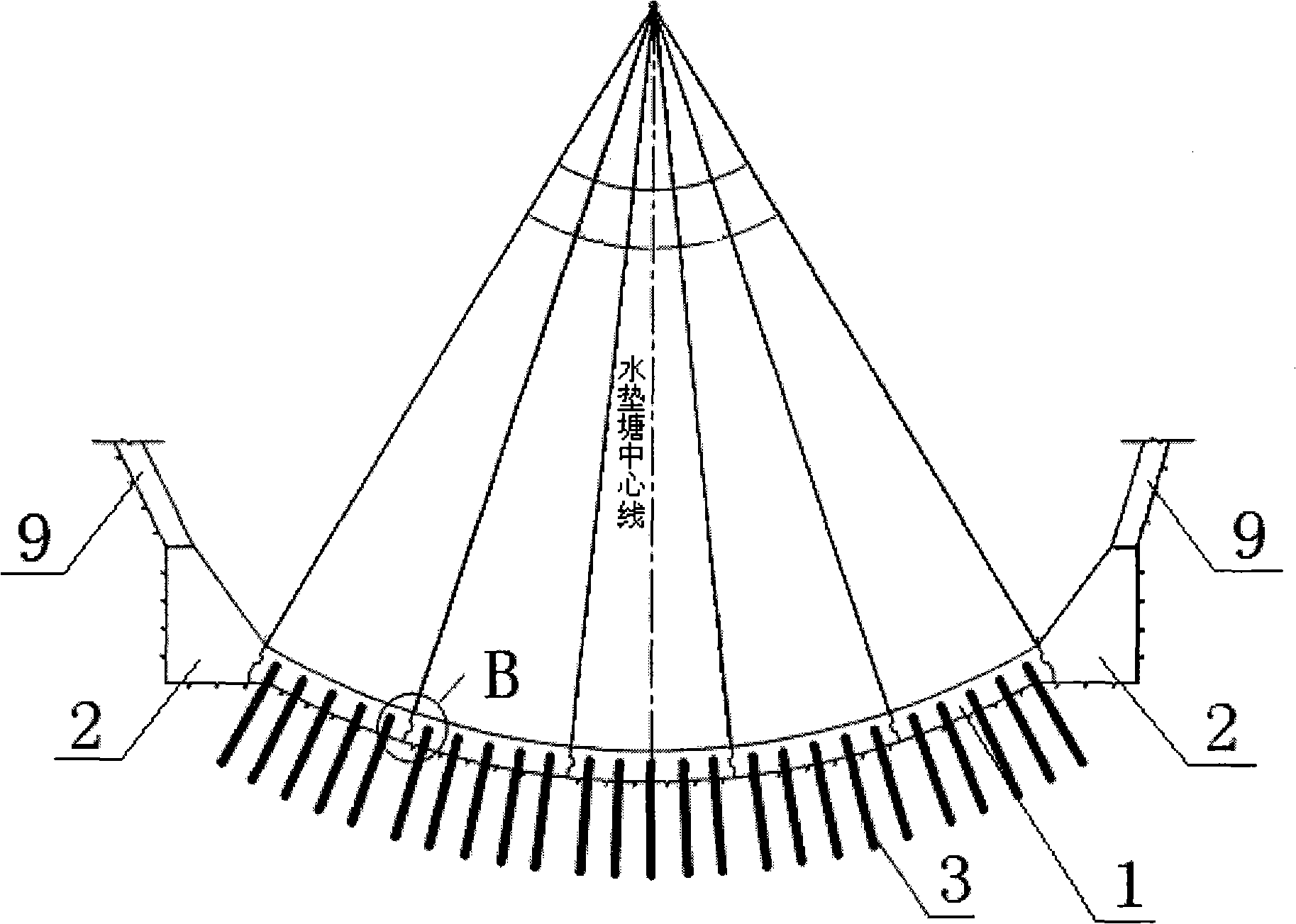
High-geostress narrow-valley inverted arch plunge pool and design method thereof
ActiveCN101555687AAvoid touchingImprove stress concentrationBarrages/weirsStress concentrationRiver bed
The invention belongs to a concentrated flood discharge and energy dissipation structure behind a hydroelectric dam, and more particularly relates to a high-geostress narrow-valley inverted arch plunge pool and a design method thereof. The plunge pool is structurally characterized by comprising a bottom plate (1), arch supports (2) and anchor bar piles (3), wherein the bottom plate (1) is an arched concrete structure on the whole, and two ends of the arched concrete bottom plate (1) is connected with the arch supports (2) into a whole; the anchor bar piles (3) are arranged at the lower end of the arched concrete bottom plate (1) at intervals, and each anchor bar pile (3) keeps perpendicular to the lower end of the bottom plate (1); and the arch supports (2) at the two ends are positioned at the lower ends of protection slopes (9) at the two ends. The design method is completed by five steps, makes full use of the natural 'pan bottom'-shaped river beds, avoids touching geostress concentration areas at the bottom of the valley due to trapezoid excavation, and adopts the inverted arch structure to finally transfer most of load to rock mass through the arch supports instead of bearing all the load by the river beds, thus improving the stress concentration phenomenon at the bottom of the valley.
Owner:POWERCHINA XIBEI ENG
Method for preparing healthy oil capsicum
InactiveCN101243860APrevent browningPrevent oxidationEdible oils/fatsFood preparationFlavorCancer prevention
The invention relates to a preparation method of healthy oil capsicum, which overcomes the drawback in the prior art that the acrylamide with carcinogenesis possibility is produced during the high temperature baking, the roasting or the frying craft. The prepared main materials and the ingredients are blended or respectively cooked by using vacuum low temperature equipment. The preparation method of the healthy oil capsicum has the advantages that: the conditions producing excessive heat because of the high temperature frying, internal heat, carcinogenesis substance acrylamide easily because of the high temperature frying are eliminated radically and the aim of low temperature frying, cancer prevention, no internal heat is achieved; the browning and oxidation of the capsicum can be effectively avoided by the application of the vacuum low temperature equipment, the loss and the damage of the nutritional ingredient are avoided and the natural color, flavor and the frying taste of conventional oil capsicum are retained.
Owner:吴承霖
Method for preparing graphene/nickel nanocomposite material by utilizing gamma ray
ActiveCN103011151ASuitable for quantitative productionMild reaction conditionsMaterial nanotechnologyGrapheneRadiation chemistryGamma ray
The invention discloses a method for preparing a graphene / nickel nanocomposite material by utilizing a gamma ray, belongs to the field of crossing of the nanocomposite material and radiation chemistry, and in particular relates to a method for preparing the graphene / nickel nanocomposite material by utilizing the gamma ray. The problem that the graphene-nickel nanocomposite material prepared by the conventional method is high in cost and severely damages the environment is solved. The method comprises the following steps of: 1, preparing oxidized graphite; 2, preparing mixed solution; 3, reacting under the irradiation of the gamma ray to obtain a product; and 4, centrifugally separating, cleaning and drying the product to obtain the graphene / nickel nanocomposite material. The method is applied to the field of preparation of the graphene / nickel nanocomposite material.
Owner:TECHN PHYSICS INST HEILONGJIANG ACADOF SCI
Suppression method for zero-sequence voltage caused by dual inverter switching dead area
ActiveCN104506071AEnhanced inhibitory effectThe implementation method is simpleElectronic commutation motor controlAC motor controlPower inverterVoltage vector
The invention discloses a suppression method for zero-sequence voltage caused by a dual inverter switching dead area, belongs to the technical field of inverter control and aims to solve the problems that a system has zero-sequence voltage and system performances are affected due to addition of dead time when common direct-current bus dual inverter switching signals are switched. Based on SVPWM (space vector pulse width modulation) performed by a switch combination without zero-sequence voltage, the suppression method includes the steps: firstly, acquiring three-phase voltage signals after RC (resistance-capacitance) low-pass filter of dual inverters by a voltage sensor; processing the three-phase voltage signals to obtain system zero-sequence voltage; performing dead area compensation calculation according to the system zero-sequence voltage to obtain a compensation voltage vector, offsetting the system zero-sequence voltage by the zero-sequence voltage generated by the compensation voltage vector, and suppressing the system zero-sequence voltage. The suppression method is used for suppressing the zero-sequence voltage caused by the dual inverter switching dead area.
Owner:HARBIN INST OF TECH
Method for the extraction of an ir-image and thermal imaging camera
InactiveUS20110199489A1Disturbing noise can be avoidedDisturbing noiseTelevision system detailsCharacter and pattern recognitionLower limitData stream
A thermal imaging camera (1) which, in order to extract IR-images (2) from a stream of crude data (12) of crude IR-images (3), performs a compensation (21) of pixel measurements (6) of the crude IR-images (3) with pixel-background values (8) pixel by pixel, with a new calculation (19) of the pixel-background value (8) being performed when the variation of the pixel measurements (6) in the measurement sequence of the crude data stream (12) for the pixels exceeds a lower limit for variations.
Owner:TESTO AG
Easily-degradable alkali-free viscosity reducer for thick oil and preparation method thereof
ActiveCN102732241AEasy to degradeImprove surface activityFluid removalPipeline systemsAlkali freeReducer
The invention relates to an easily-degradable alkali-free viscosity reducer for thick oil and a preparation method thereof. The viscosity reducer consists of propylene glycol block polyether, fatty alcohol ammonium sulfate, fatty alcohol, a penetrating agent and water; and in terms of the total mass of the viscosity reducer, the sum of mass percent of propylene glycol block polyether, fatty alcohol ammonium sulfate, fatty alcohol and the penetrating agent is 45 to 60 wt%. The invention also provides the preparation method for the viscosity reducer, and the method comprises the following steps: successively adding propylene glycol block polyether, fatty alcohol ammonium sulfate, fatty alcohol and the penetrating agent into water at a temperature of 50 to 80 DEG C under normal pressure, wherein a mass ratio of the four above-mentioned raw materials to water is 15: 15: 5: 10: 55 to 25: 25: 4: 6: 40; and carrying out continuous stirring for 60 to 90 min at a rotating speed of 60 to 120 r / min while adding the four raw materials so as to obtain the easily-degradable alkali-free viscosity reducer for thick oil. Surfactant used in the viscosity reducer is nonionic and anionic surfactant, the structure of benzene rings is not included in a molecule, and therefore, the characteristics of easy degradability, no toxicity, greenness and environment friendliness are obtained.
Owner:PETROCHINA CO LTD
Preparation method of bone mesenchymal stem cell carrying NK4 gene and application thereof
InactiveCN101912618AGrowth inhibitionImprove tumor inhibition rateGenetic material ingredientsDigestive systemBALB/cGastric carcinoma
The invention discloses a preparation method of bone mesenchymal stem cells carrying NK4 genes and application thereof in medicine for treating gastric cancer. The preparation method comprises the following steps: extracting the NK4 genes from an HGF plasmid, using an in-Fusion technology to directionally clone the NK4 genes to a slow virus expression carrier plasmid, constructing an NK4-EGFP fusion gene recombinant slow virus carrier plasmid (pGC-FU-NK4) and then co-transfecting a 293T cell with the pGC-FU-NK4, a pHelper1.0 carrier and a pHelper2.0 carrier, packaging to produce NK4 overexpression slow virus particles (Lenti-NK4), using slow virus (Lenti-NK4) carrying NK4-EGFP fusion gene to transfect the bone mesenchymal stem cells to establish in vitro bone mesenchymal stem cells to stably express the NK4 genes. Balb / C nude mice animal experiments prove that the NK4 gene therapy taking BMSCs as a carrier can obviously inhibit the growth of subcutaneous transplant tumor of gastric cancer with high tumor inhibition efficiency, and the NK4 gene therapy is a good gene therapeutic method for gastric cancer.
Owner:祝荫
Jelly spinning polyethylene/epoxy resin composite fiber and its preparing method
InactiveCN1749452AHigh heat distortion temperatureImprove heat resistanceWet spinning methodsConjugated synthetic polymer artificial filamentsFiberEpoxy
The present invention discloses a kind of freezing spun composite polyethylene / epoxy resin fiber and its preparation process. The freezing spun composite polyethylene / epoxy resin fiber is prepared with powdered ultrahigh molecular weight polyethylene in average molecular weight greater than 1 MDa in 100 weight portions, epoxy resin in 1-10 weight portions, curing agent in 0.01-1 weight portions, antioxidant in 0.1-5 weight portions and antioxidant assistant in 0.01-2 weight portions. The present invention is superior in that the epoxy resin as modifier has excellent performance and few damage on ultrahigh molecular weight polyethylene and the freezing spun composite polyethylene / epoxy resin fiber may be produced in available ultrahigh molecular weight polyethylene fiber producing apparatus after slightly improvement.
Owner:DONGHUA UNIV
Electrically-driven hedge clipping device and method
ActiveCN108522058AFlexible movementFlexible adjustmentHedge trimming apparatusCuttersElectricityControl system
The invention discloses an electrically-driven hedge clipping device, and belongs to the technical field of hedge clipping. The electrically-driven hedge clipping device comprises a lifting system, atranslation system, an overall rotating system, a tool rotating system, a clipping system and a control system; the overall rotating system comprises a rotating base, supporting beams, a telescopic cross beam, motors and speed reducers, the supporting beams are installed on the rotating base, a cross beam sliding cover is hinged to the upper ends of the supporting beams through hinging shafts, themotors and the speed reducers are installed at the outer sides of the supporting beams, the motors are connected with the speed reducers, and the speed reducers are connected with the hinging shafts;the tool rotating system comprises a lifting rod, and the lifting rod is rotatably installed at the tail end of the telescopic cross beam; the lifting system is installed on the side wall of the lifting rod, the translation system and the lifting system are connected through a sliding ring connection part, and the clipping system is installed on the lifting system. The electrically-driven hedge clipping device is adopted for an electrically-driven hedge clipping method. The electrically-driven hedge clipping device has the advantages of being simple in structure, capable of clipping a hedge from multiple angles, flexible in clipping and the like.
Owner:GUANGZHOU COLLEGE OF SOUTH CHINA UNIV OF TECH
Transient zero sequence current-based single phase earth fault discrimination method
InactiveCN107589347AAccurate analysis of flow directionThe method is simple and easy to implementFault locationInformation technology support systemTransient stateElectrical polarity
The invention discloses a transient zero sequence current-based single phase earth fault discrimination method comprising the following steps: in step A, power distribution network zero sequence voltage magnitude is detected via all power distribution terminals, whether a single phase earth fault occurs is determined, step B is executed if the fault occurs, otherwise step A is executed in a circulating manner; in step B, when the earth fault occurs, zero sequence voltage is subjected to derivation operation via the power distribution terminals, polarities of a transient zero sequence current and a zero sequence voltage derivative are compared, and a line having the single phase earth fault is determined; in step C, a line with opposite polarities is chosen as the line having the fault, andthe power distribution terminals send earth fault blocking signals carrying grade information to power distribution terminals for a line which is one level higher; in step D, according to variation of transient zero sequence current amplitudes of all detection points on the line chosen in step C, a small current earth fault is positioned.
Owner:CHENGDU HANDU TECH
Shaft furnace with central air distribution device and method for controlling air distribution capacity
InactiveCN102417945AThe implementation method is simpleEasy to retrofit on siteShaft furnaceEngineeringShaft furnace
The invention provides a shaft furnace with a central air distribution device, which is characterized in that: a guide cone in the existing shaft furnace is heightened, hollowed-out and drilled, and a pipeline and an adjustable valve for conveying reducing coal gas into the interior of the furnace are additionally arranged outside the furnace, so that the implement method is simple, and the field renovation is easy. The air distribution device has the characteristics of small size, smoothness in blanking, uniformity in distribution of central coal gas and long service life. According to the invention, the coal gas amount of the center of the shaft furnace can be adjusted, and the metallization rate of the materials on the central part of the shaft furnace is ensured.
Owner:张昭贵
Method for improving waxberry fruit soluble solid content
ActiveCN103621339AHigh soluble solids contentNo side effectsCultivating equipmentsHorticulture methodsSodium HydroseleniteEconomic benefits
The invention discloses a method for improving the waxberry fruit soluble solid content. In the waxberry fruit development process, a sodium selenite or boric acid agent is sprayed to a waxberry tree. Sodium selenite is prepared into an aqueous solution with the 20-200mg.L-1 mass concentration in use, and boric acid is prepared into an aqueous solution with the 0-2g.L-1 mass concentration in use; the using mode is that in the period from 20 days after waxberry flowers wither to 30 days before waxberry fruits mature and are picked up, the concentration of the agent is adjusted, spraying processing is carried out on the waxberry tree 2-4 times with a sprayer, spraying is carried out every 7-21 days, and spraying is carried out until the leave faces of the waxberry tree is wet each time; production management is carried out according to daily management of a normal waxberry garden. The method is easy to implement and low in cost, the effect of improving the waxberry fruit soluble solid content is obvious, and the method has the obvious economic benefits and is suitable for being popularized and applied.
Owner:POMOLOGY RES INST FUJIAN ACAD OF AGRI SCI
TBM bad geological identification method based on intelligent driving model
InactiveCN110020694AThe implementation method is simpleQuick responseCharacter and pattern recognitionMachine learningAlgorithmIntelligent driver model
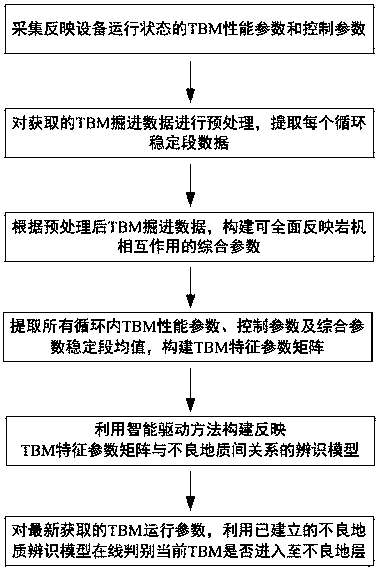
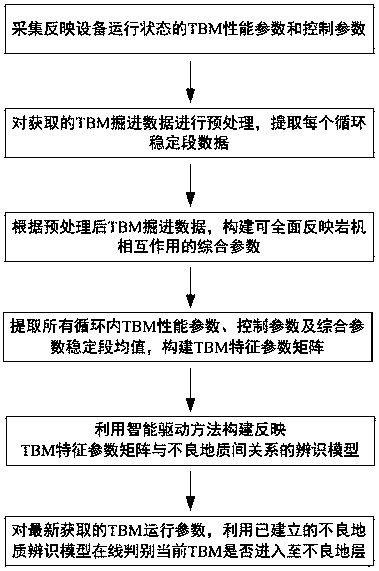
The invention provides a TBM unfavorable geology identification method based on an intelligent driving model to solve the problems of low identification degree and the like caused by the fact that a TBM driver judges geology according to experience of the TBM driver, and the method comprises the following steps: collecting TBM operation parameters reflecting an equipment operation state; preprocessing the obtained TBM operation parameters; according to the preprocessed TBM operation parameters, constructing TBM comprehensive parameters capable of comprehensively reflecting the interaction of the rock machine; extracting stable section mean value characteristics from the obtained TBM operation parameters and TBM comprehensive parameters, and a constructing TBM characteristic parameter matrix; according to the identified TBM normal stratum and the unfavorable geological section in the tunneling process, using an intelligent driving model to construct an unfavorable geological identification model between the TBM characteristic parameter matrix under the corresponding scalar section and whether there is unfavorable geology or not; and obtaining a new TBM characteristic parameter matrix for the newly obtained TBM operation parameters according to the method, and judging whether the current TBM enters bad geology or not on line through the established unfavorable geology identification model. The implementation method disclosed by the invention is simple and high in distinguishing degree.
Owner:CHINA RAILWAY ENGINEERING EQUIPMENT GROUP CO LTD
Method for performing catalytic stereoselective separation on 2,3-diphenylpropionic acid enantiomer by adopting bio-enzyme
The invention discloses a new chiral separation method of a 2,3-diphenylpropionic acid single enantiomer, i.e., a method for synthesizing the 2,3-diphenylpropionic acid single enantiomer by performing enzyme-catalyzed enantioselective hydrolysis on a 2,3-diphenylpropionic acid enantiomer. 2,3-diphenylpropionate enantiomer is hydrolyzed by utilizing high selectivity and high catalysis efficiency of Candida antarctica lipase A and a solubilization effect of cyclodextrin on a 2,3-diphenylpropionate enantiomer is utilized, so that hydrolysis reaction of the 2,3-diphenylpropionate enantiomer in a phosphate buffering solution is enhanced; the conversion rate of a substrate and the optical purity of a product are respectively up to 44.79 percent and 98.24 percent, and the stereoselectivity E is greater than 276. According to the method disclosed by the invention, the problems of low optical purity, low yield, environment pollution and the like in a general separation technique are overcome; by adopting the method, the high conversion rate and the high selectivity required by hydrolysis of the 2,3-diphenylpropionate enantiomer can be realized, so that the aims of no-toxicity and no-harmlessness of a separation chiral compound, mild reaction conditions, simpleness of equipment, convenience in operation, low cost and the like are fulfilled.
Owner:HUNAN INSTITUTE OF SCIENCE AND TECHNOLOGY
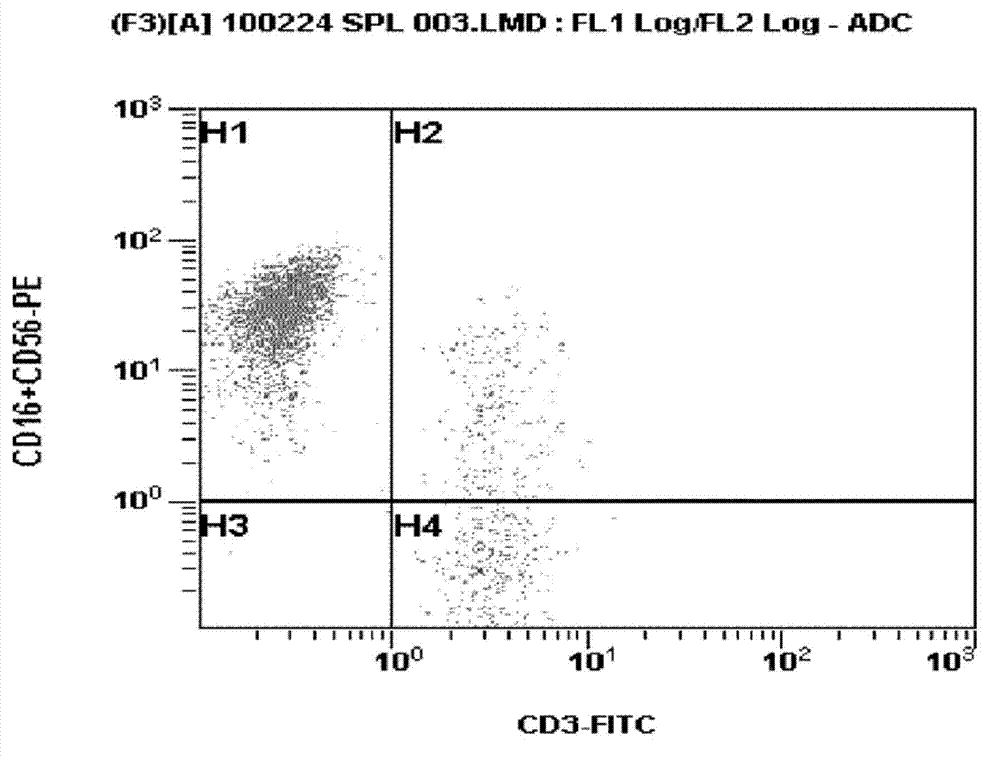
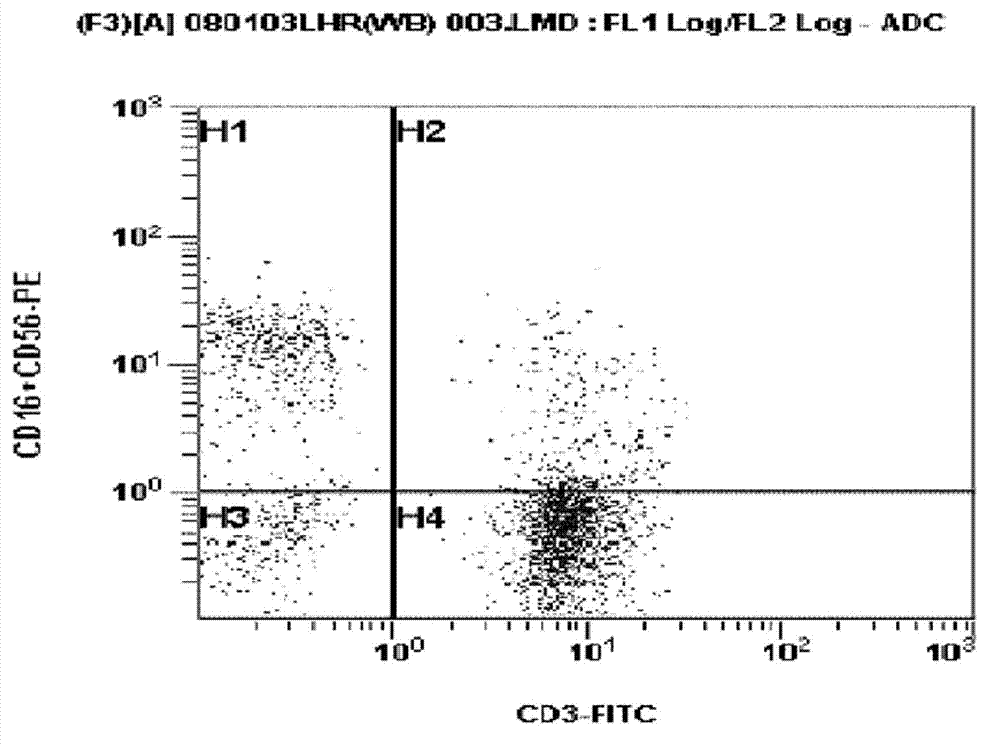
Medium composition for culturing self-activated lymphocytes and method for culturing self-activated lymphocytes using same
InactiveCN103080302ARaise the ratioLittle side effectsImmunoglobulins against cell receptors/antigens/surface-determinantsCancer antigen ingredientsCD16Interleukin II
Disclosed is a medium composition for culturing self-activated lymphocytes, which contains anti-CD3 antibody and anti-CD16 antibody in addition to interleukin 2 (IL-2), interleukin 12 (IL-12) and interleukin 18 (IL-18) in a medium, and thus can efficiently proliferate and activate NK cells, T cells and NKT cells and, at the same time, can significantly increase the ratio of NK cells in lymphocytes so as to provide immunocytes having excellent effects on the treatment of various kinds of malignant tumors, and a method for culturing self-activated lymphocytes using the medium composition.
Owner:CELLS SCI CORP
Preparation method for anhydride cured epoxy resin/palygorskite nanocomposite material
The invention relates to a preparation method for an anhydride cured epoxy resin / palygorskite nanocomposite material. The preparation method comprises the following steps of: grinding palygorskite into mineral powder; performing calcining activation treatment on the palygorskite mineral powder; heating and liquefying epoxy resin; adding the palygorskite in an amount which is 1 to 8 percent of themass of the epoxy resin, mixing and stirring; adding an anhydride curing agent in an amount which is 20 to 40 percent of the mass of the epoxy resin; stirring the obtained mixture; and vacuumizing, degassing and the like to obtain the composite material. The product is generally used for casting or encapsulating electronic and electrical products, and heating and curing. The preparation method has the advantages of obviously improving the toughness of an epoxy resin anhydride cured material and obviously improving mechanical properties and heat stability, and is easy to implement and low in cost.
Owner:GUANGZHOU INST OF GEOCHEMISTRY - CHINESE ACAD OF SCI
A stable power generation method for converting multiple kinds of ocean energy into usable electric energy
InactiveCN105804915AImprove utilization efficiencyThe implementation method is simplePV power plantsEnergy industryCouplingFuel tank
The invention relates to a stable power generation method for converting multiple kinds of ocean energy into usable electric energy. The devices employed in the method include an auxiliary oil tank, a wind wheel, a coupling, a constant delivery pump, a check valve, an energy stabilizing device, a flow valve, a hydraulic constant displacement motor, a common alternating current generator, an electromagnetic overflow valve A and an electromagnetic overflow valve B. The constant delivery pump is connected to the auxiliary oil tank and the coupling is connected to the constant delivery pump; the coupling is connected with the wind wheel; a pipeline on the other end of the constant delivery pump is connected with the check valve; the energy stabilizing device is connected to a pipeline on the other end of the check valve; the energy stabilizing device is connected with the electromagnetic overflow valve A and the electromagnetic overflow valve B; the flow valve is connected to a pipeline of the other end of the energy stabilizing device; the flow valve is connected with the hydraulic constant displacement motor; the hydraulic constant displacement motor is connected with the common alternating current generator. The method can convert unstable ocean energy into usable electric energy, is simple and feasible, reduces the production and power generation costs, increases the ocean energy utilization rate and is wide in application range.
Owner:CHINA UNIV OF PETROLEUM (EAST CHINA)
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com