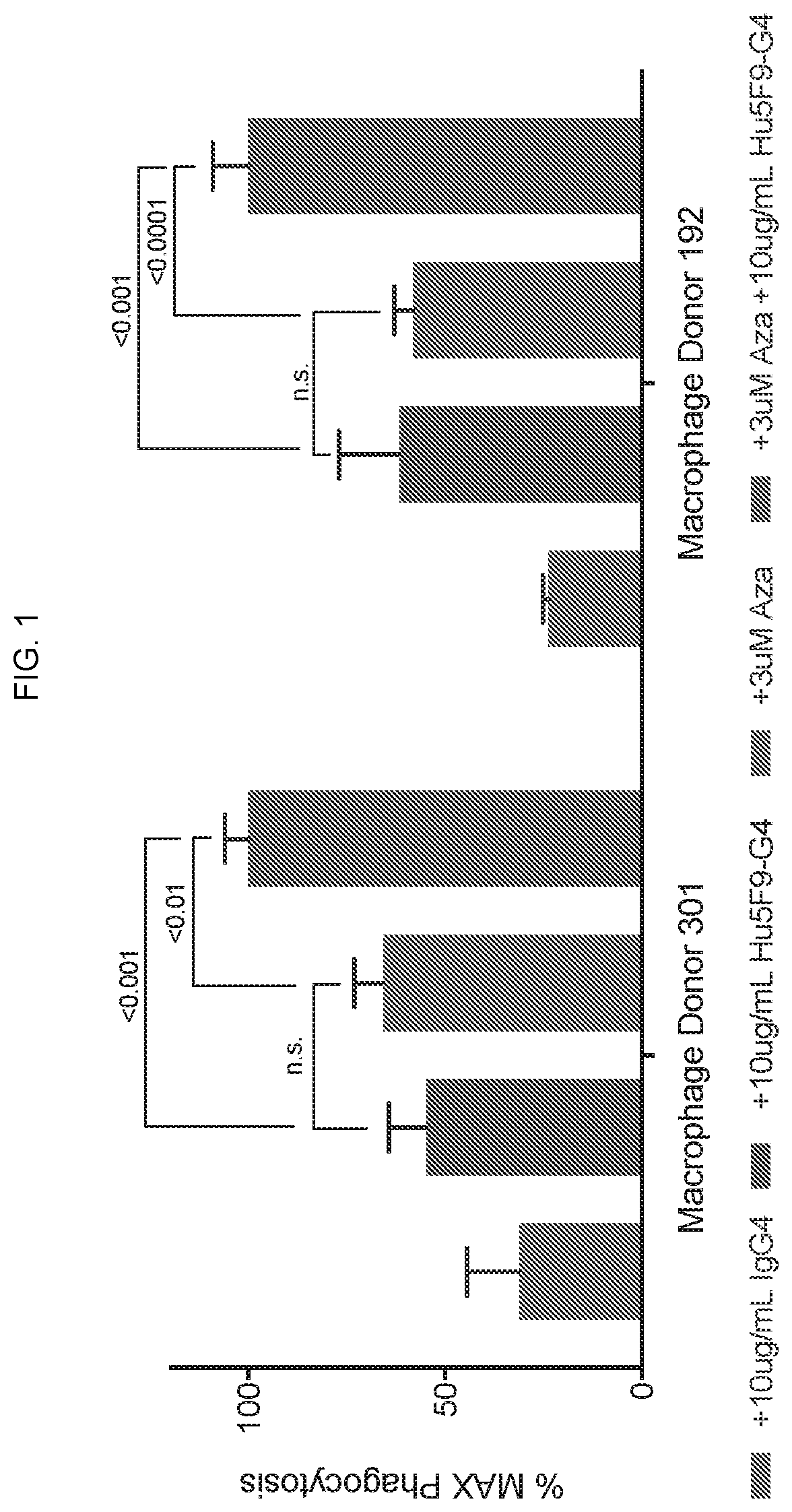
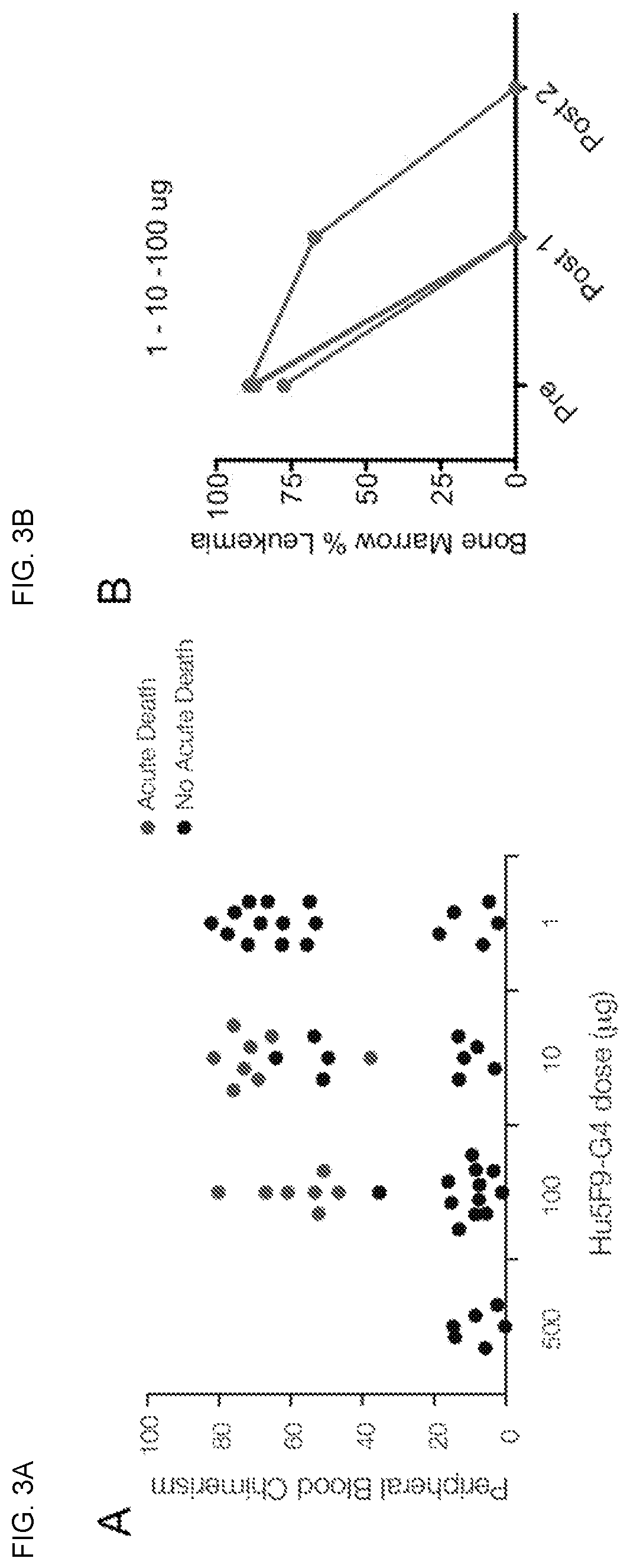
Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
138 results about "Anti-agents" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
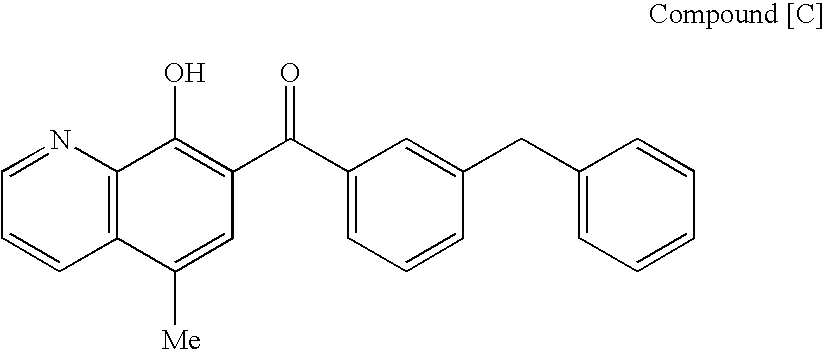
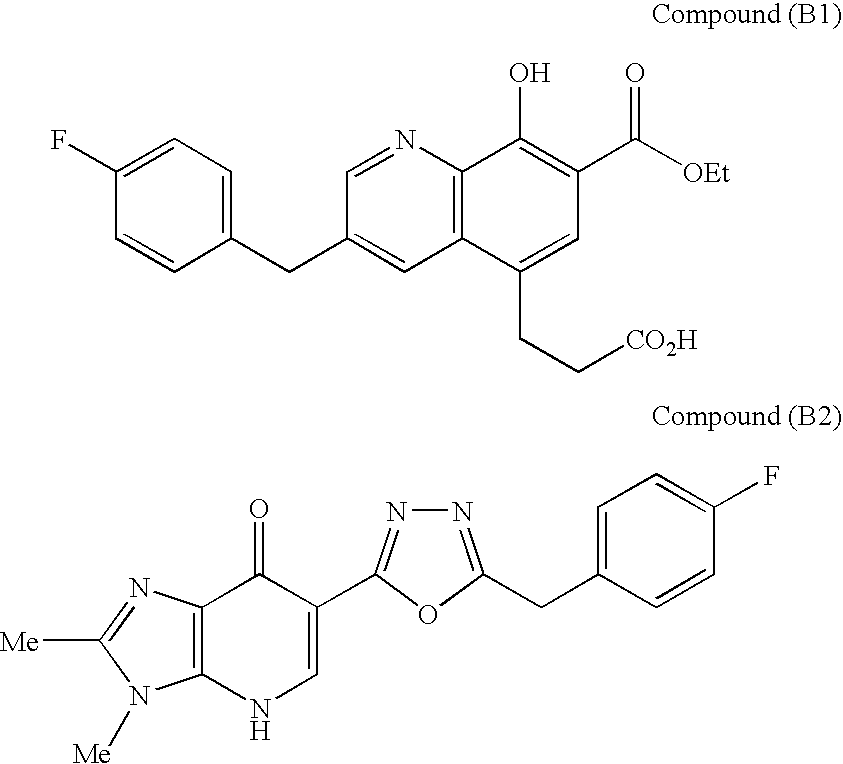
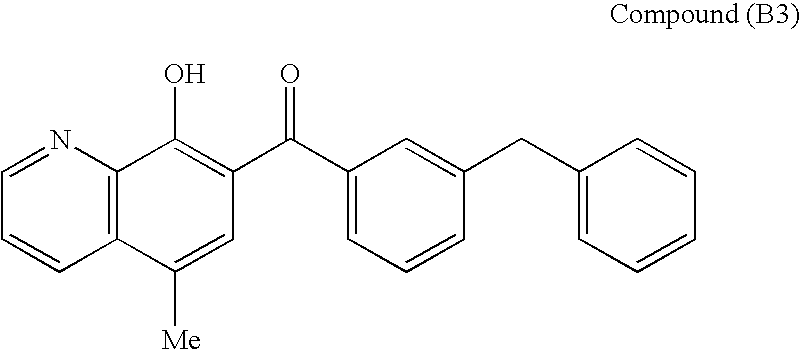
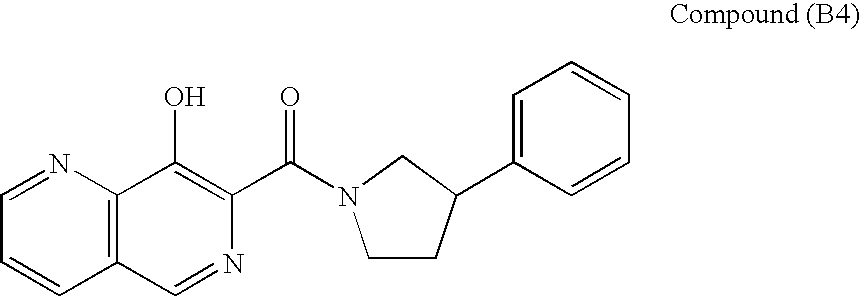
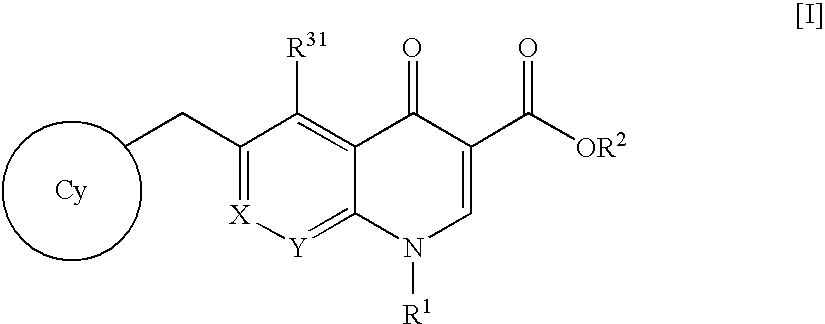
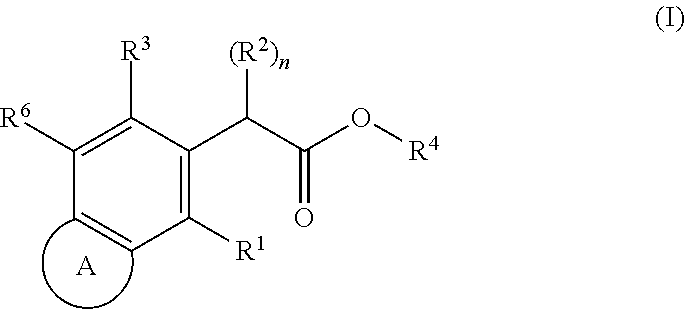
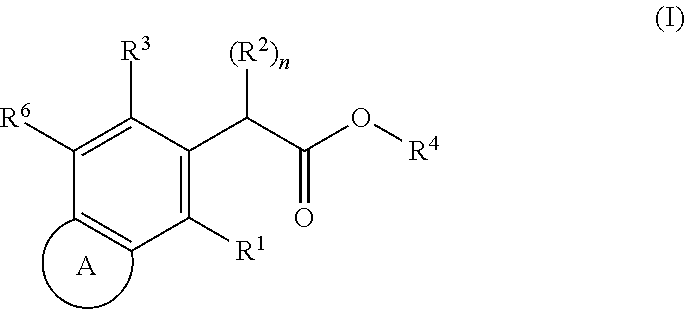
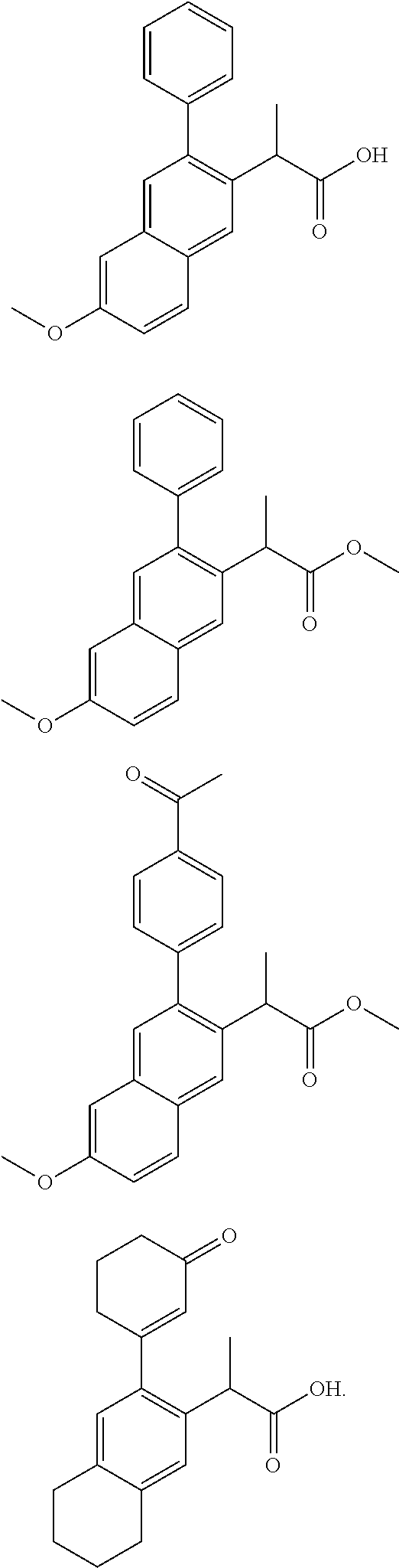
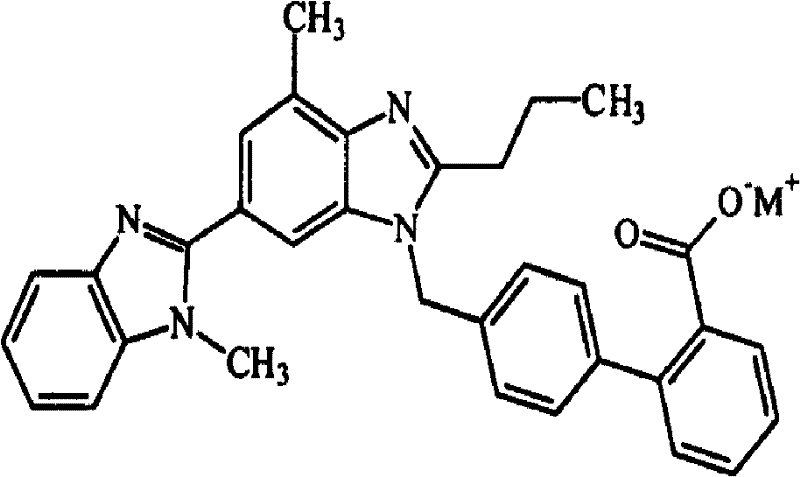
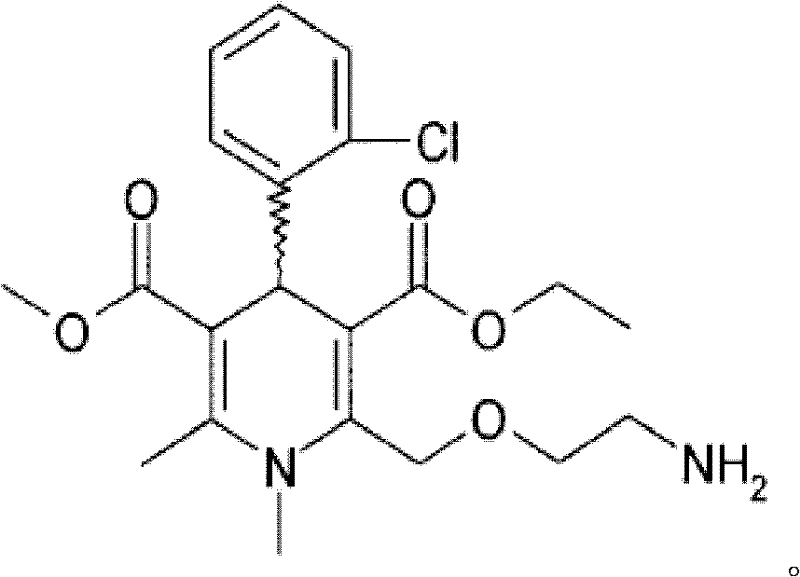
SUBSTITUTED SPIROPYRIDO[1,2-a]PYRAZINE DERIVATIVE AND PHARMACEUTICAL USE OF SAME AS HIV INTEGRASE INHIBITOR
InactiveUS20140221380A1Strong inhibitory activityLess side effectsBiocideOrganic chemistryPyrazinePharmaceutical drug
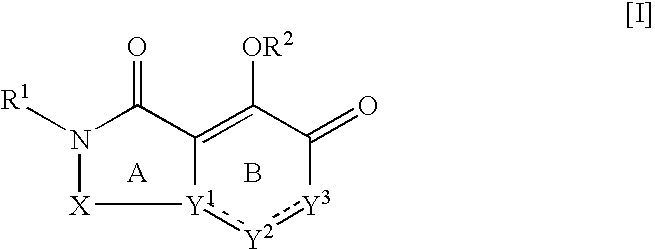
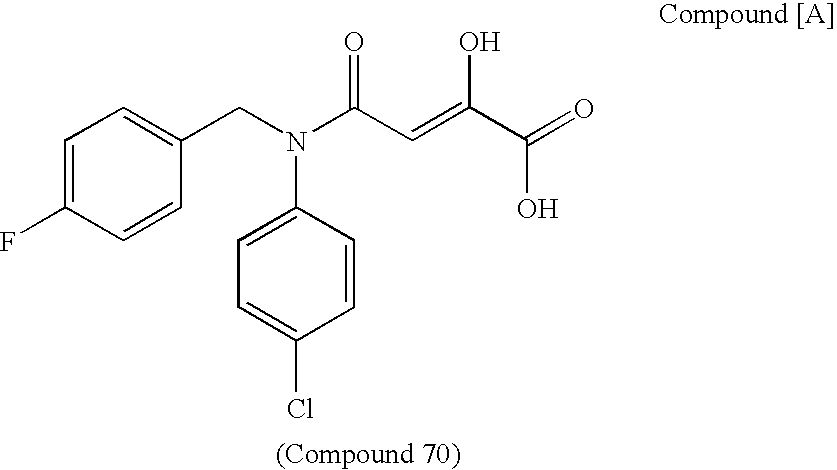
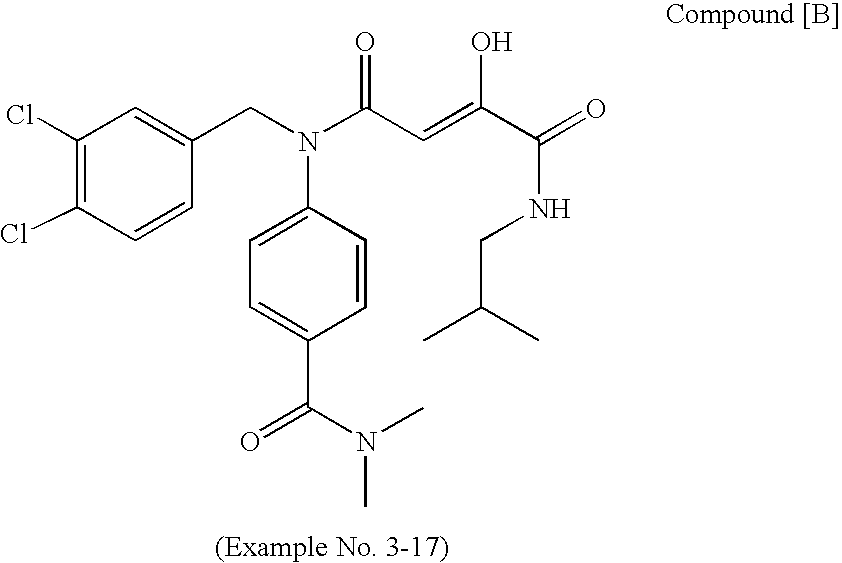
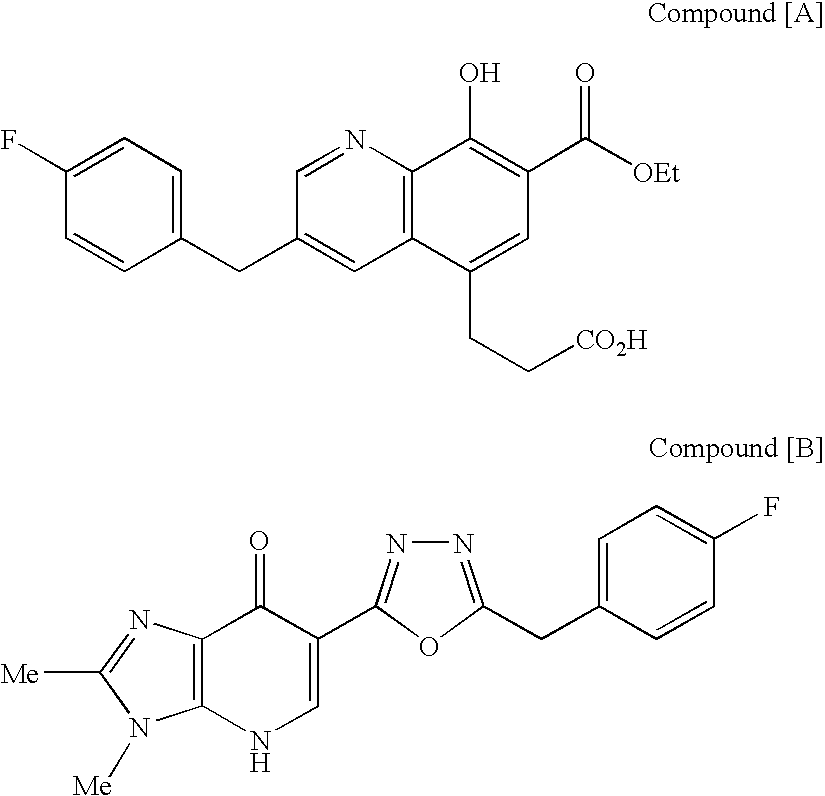
[Summary][Problem] Provided is a substituted spiropyrido[1,2-a]pyrazine derivative or a pharmaceutically acceptable salt thereof, which is useful as an anti-HIV agent.[Solving Means] The present invention relates to a compound represented by the following formula [I] or [II] or a pharmaceutically acceptable salt thereof:wherein each symbol is as defined in the specification.
Owner:JAPAN TOBACCO INC
Nitrogen-containing fused ring compound and use thereof as HIV integrase inhibitor
InactiveUS20050054645A1Effective anti-HIV agentStrong inhibitory activityBiocideOrganic chemistryIntegrase inhibitorSide effect
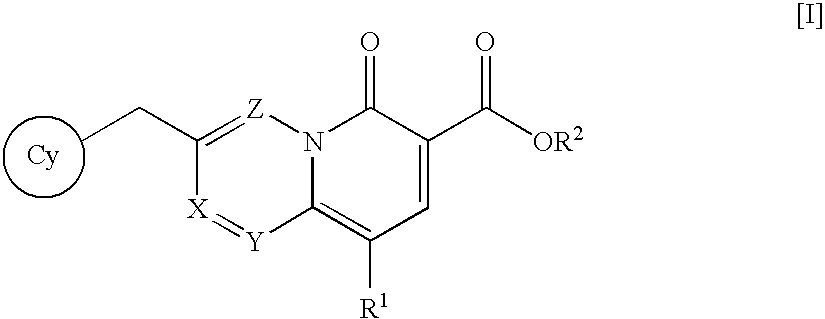
The present invention relates to a nitrogen-containing fused ring compound represented by the following formula [I]wherein each symbol is as defined in the specification, or a pharmaceutically acceptable salt thereof, and an anti-HIV agent containing such compound. The compound of the present invention has an HIV integrase inhibitory activity, and is useful as an agent for the prophylaxis or treatment of AIDS, or as an anti-HIV agent. In addition, by the combined use with other anti-HIV agents such as a protease inhibitor, a reverse transcriptase inhibitor and the like, it can be a more effective anti-HIV agent. Becuae it shows integrase-specific high inhibitory activity, the compound can be a pharmaceutical agent safe on human body, which causes only a fewer side effects.
Owner:JAPAN TOBACCO INC
4-oxoquinoline compound and use thereof as pharmaceutical agent
Owner:JAPAN TOBACCO INC
Compositions and methods for binding lysophosphatidic acid
ActiveUS20100034814A1Low effective concentrationSugar derivativesMicrobiological testing/measurementComplementarity determining regionVariable domain
Compositions and methods for making and using anti-LPA agents, for example, monoclonal antibodies, are described. Variable domain and complementarity determining region amino acid sequences of several monoclonal antibodies against LPA are disclosed, as is a consensus anti-LPA monoclonal antibody variable domain sequence.
Owner:APOLLO ENDOSURGERY INC
Combination therapy
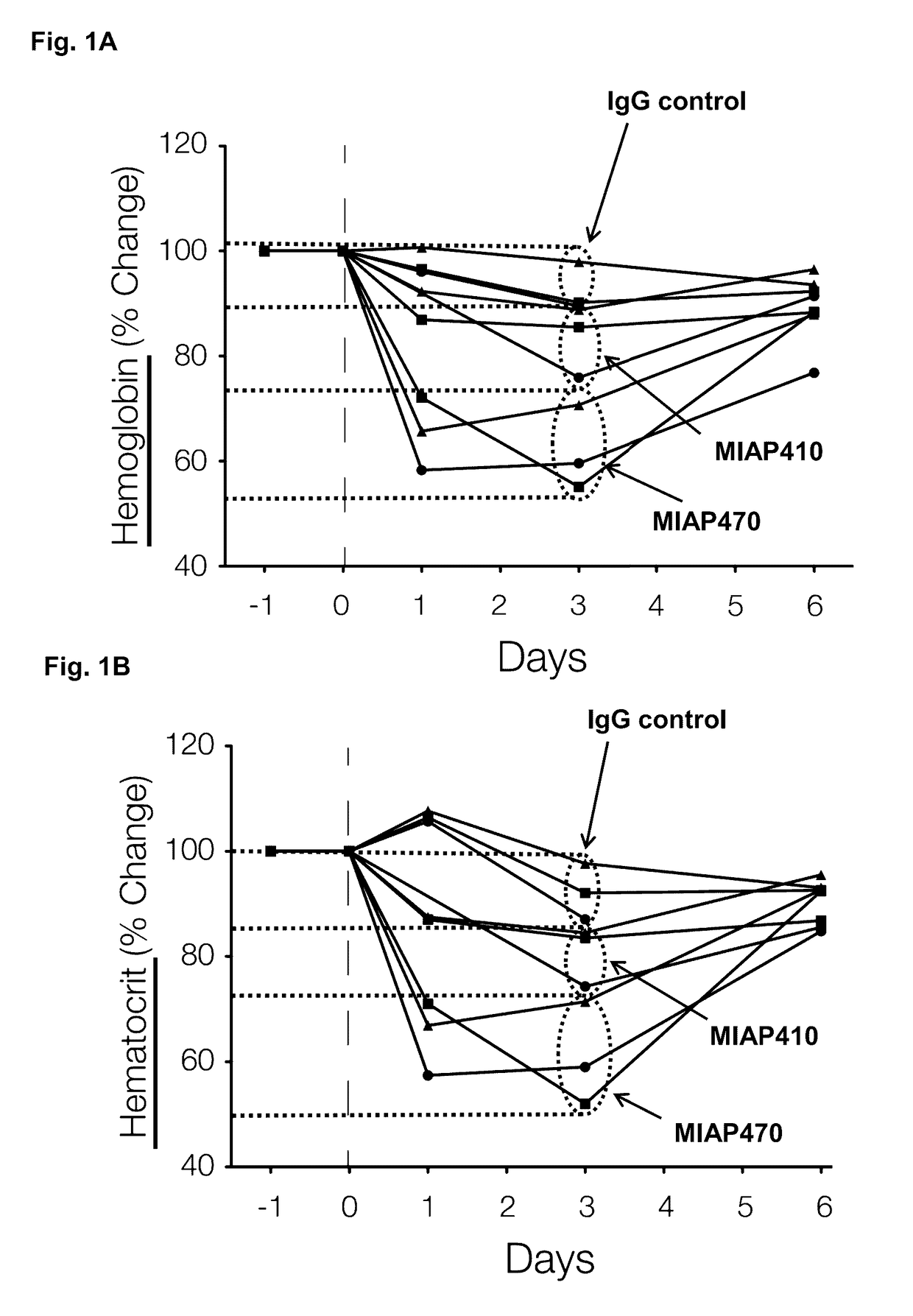
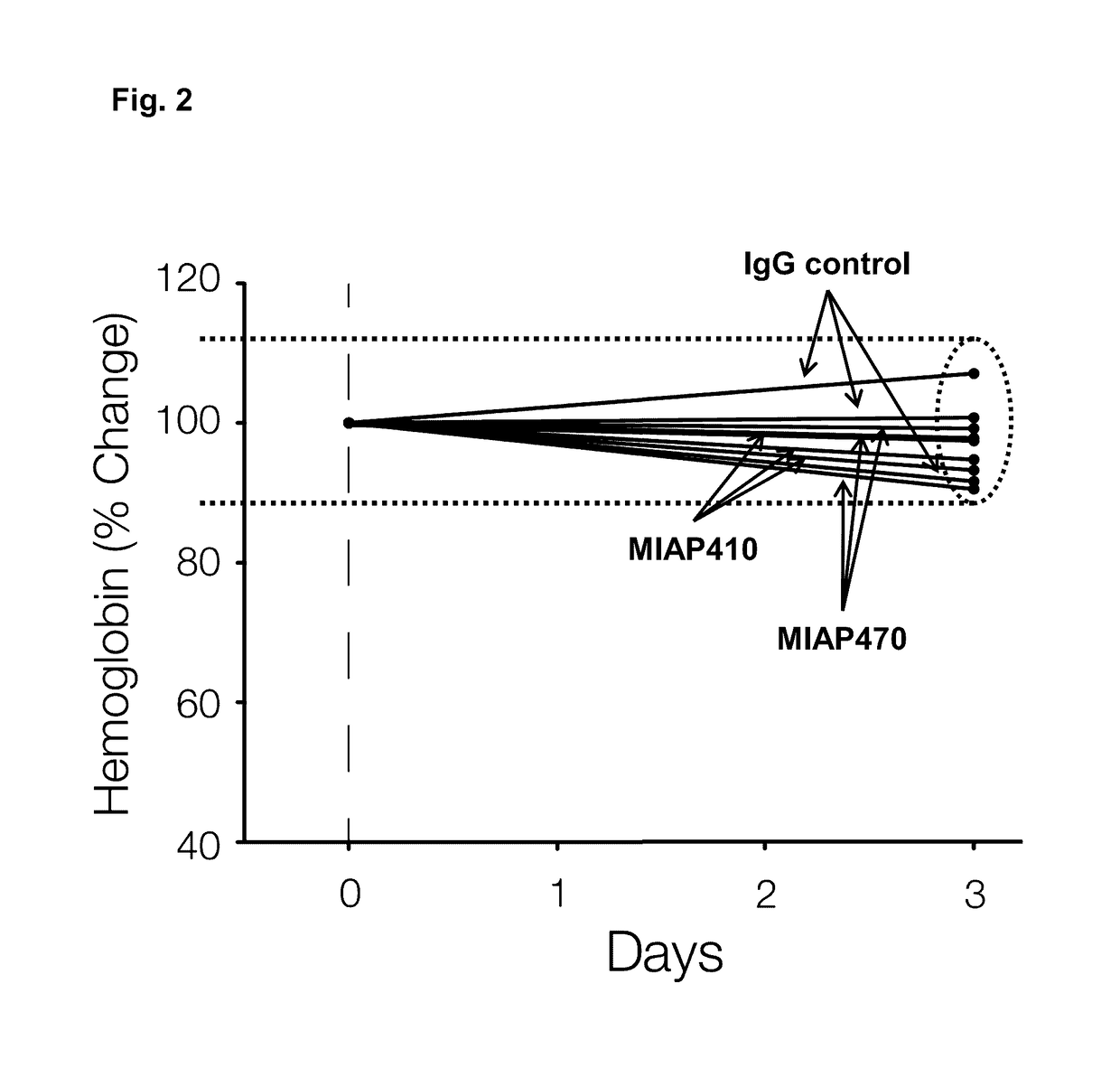
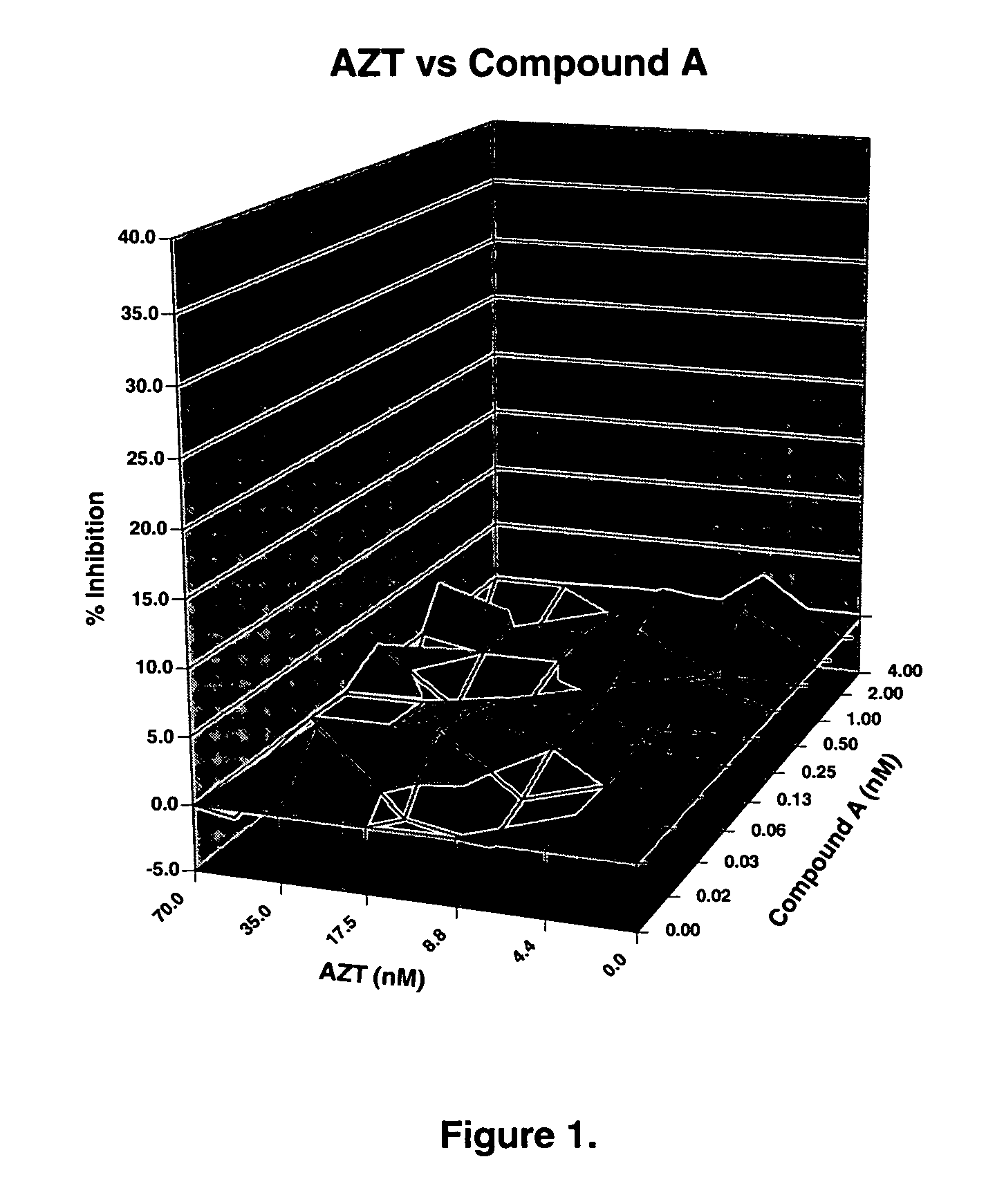
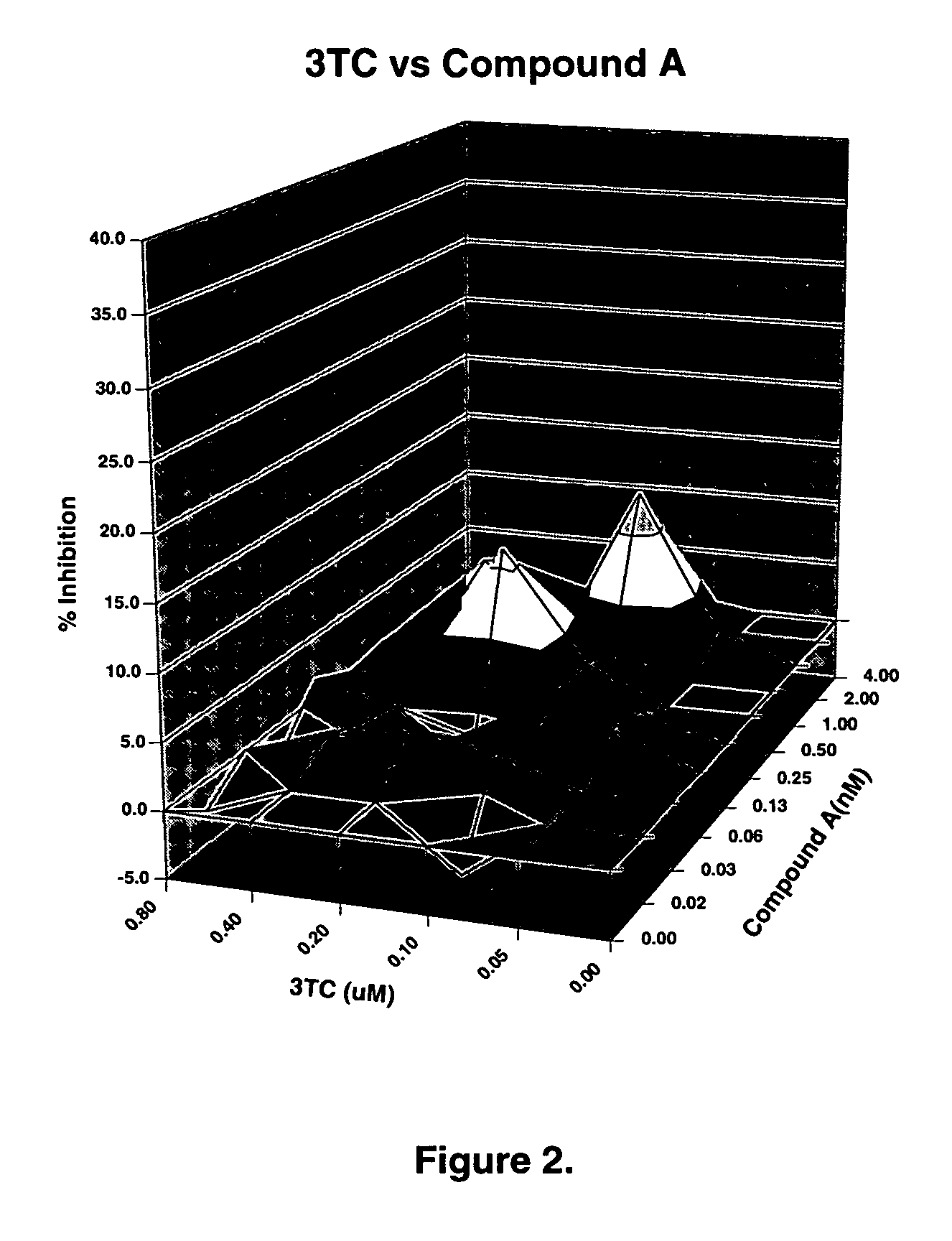
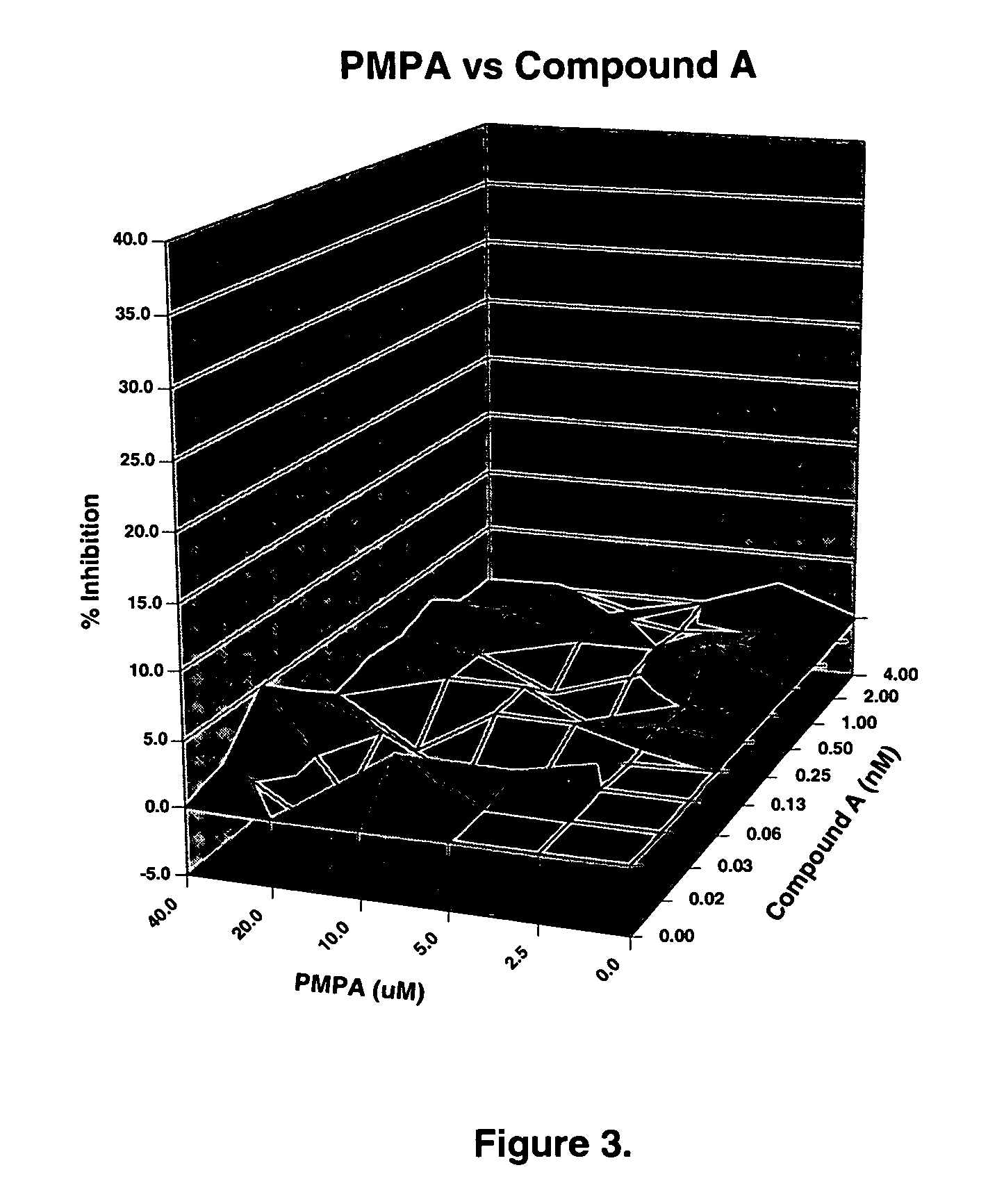
ActiveUS20050288326A1Low effective doseLow cytotoxicityBiocideAntiviralsSide effectReverse transcriptase
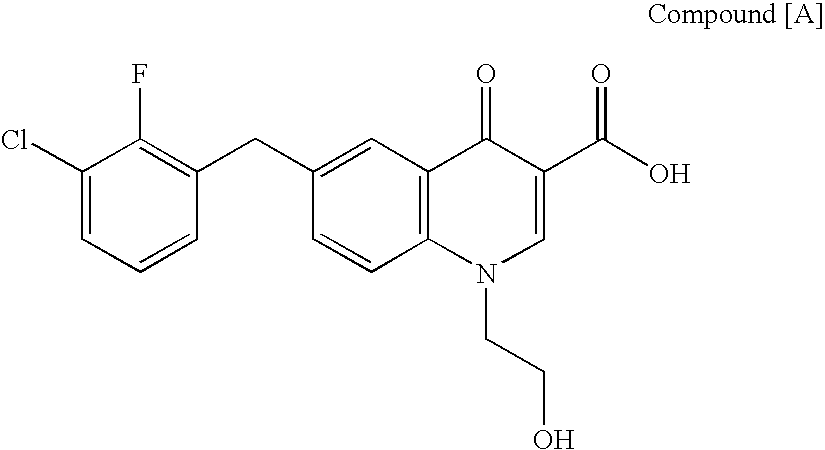
The present invention relates to a combination therapy for treating an HIV infection or inhibiting integrase comprising (S)-6-(3-Chloro-2-fluorobenzyl)-1-(1-hydroxymethyl-2-methylpropyl)-7-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (“Compound A”) or a pharmaceutically acceptable solvate or salt thereof in combination with at least one other anti-HIV agent. In some embodiments of the present invention, the other anti-HIV agents are chosen from reverse transcriptase inhibitors and protease inhibitors. In certain embodiments of the present invention, the other anti-HIV agents are chosen from AZT, 3TC, PMPA, efavirenz, indinavir, nelfinavir, a combination of AZT / 3TC, and a combination of PMPA / 3TC. Since Compound A has a high inhibitory activity specific for integrases, when used in combinations with other anti-HIV agents it can provide a combination therapy with fewer side effects for humans.
Owner:JAPAN TOBACCO INC
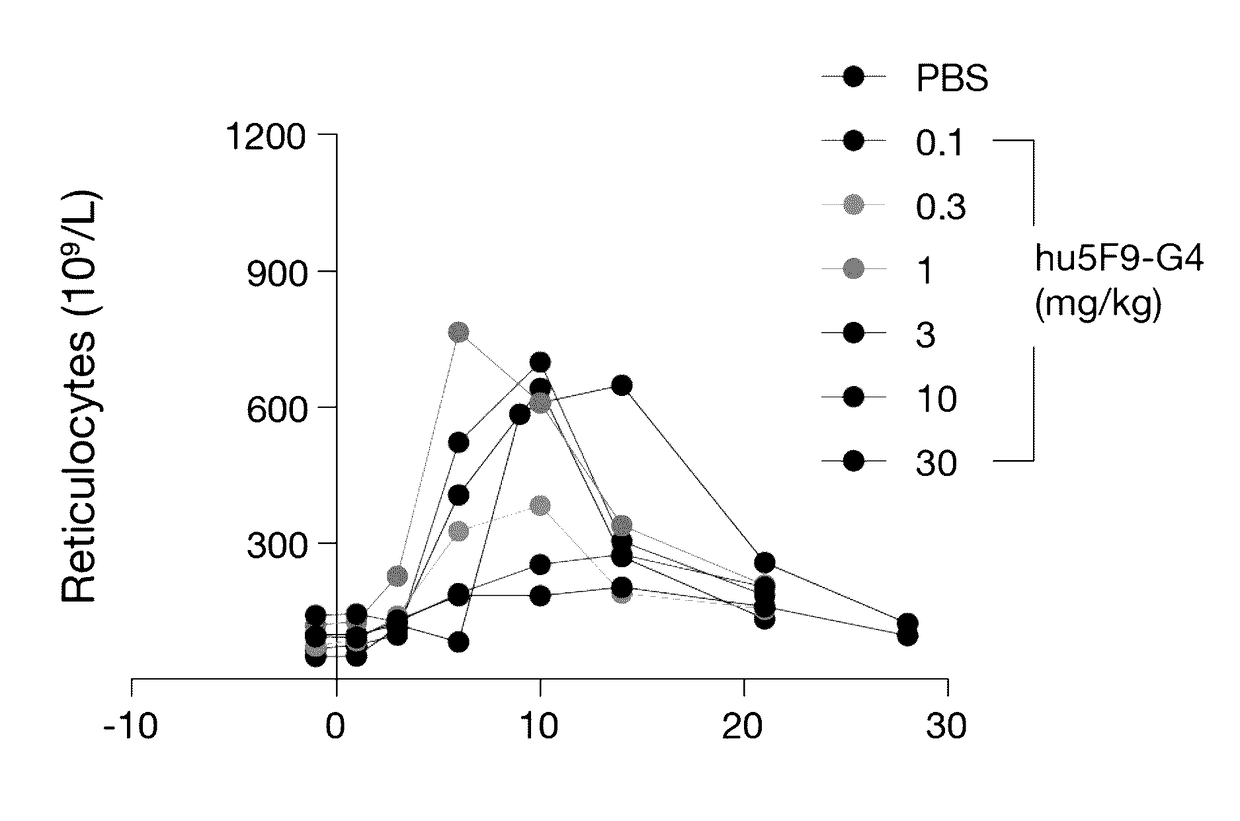
Methods for achieving therapeutically effective doses of anti-CD47 agents for treating cancer
ActiveUS9623079B2Low toxicityIncrease productionPeptide/protein ingredientsAntibody mimetics/scaffoldsPharmaceutical drugOncology
Owner:THE BOARD OF TRUSTEES OF THE LELAND STANFORD JUNIOR UNIV
Combination therapy
ActiveUS8633219B2Low effective doseEffective treatmentBiocideOrganic chemistrySide effectCombined Modality Therapy
Owner:JAPAN TOBACCO INC
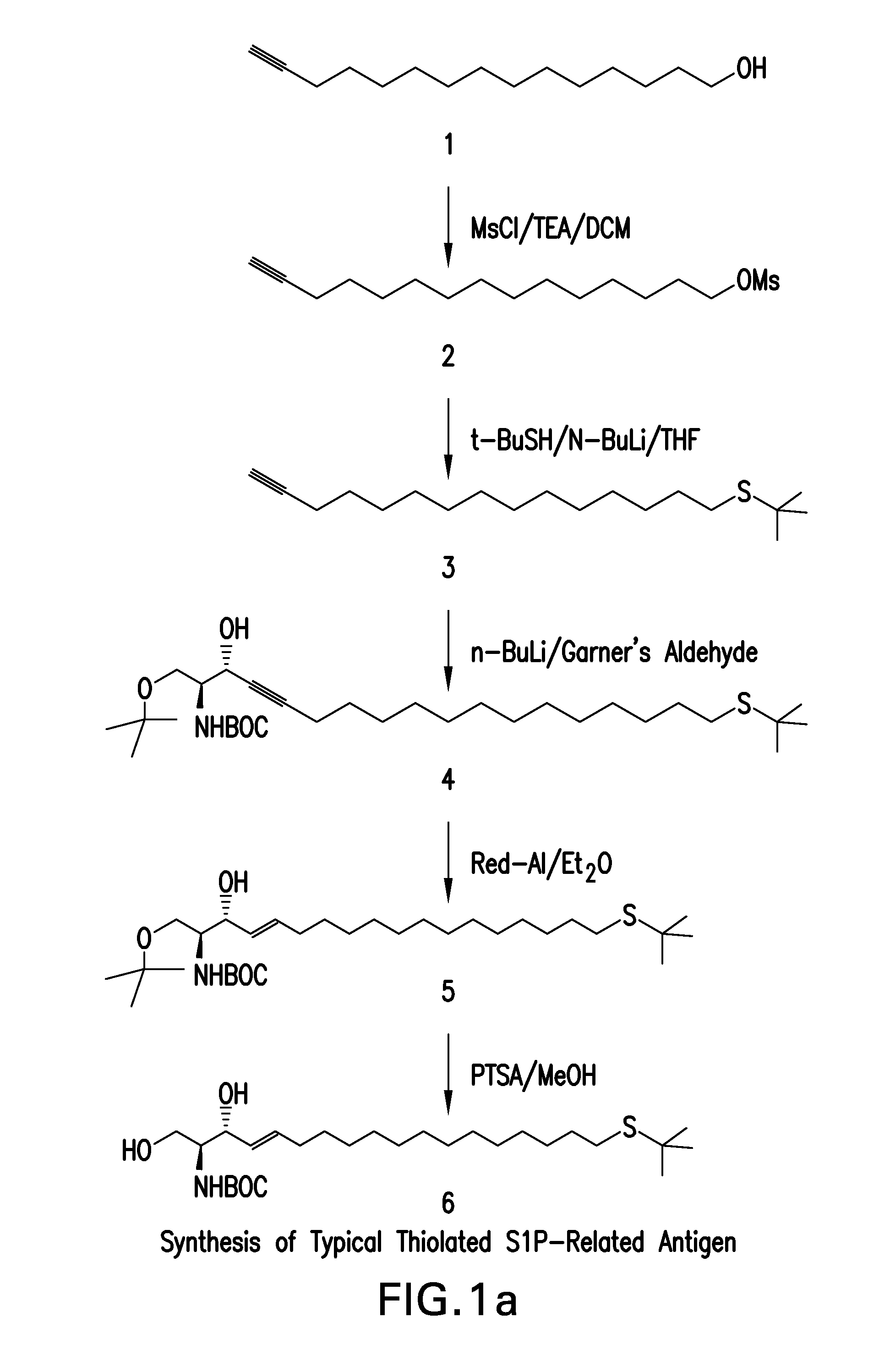
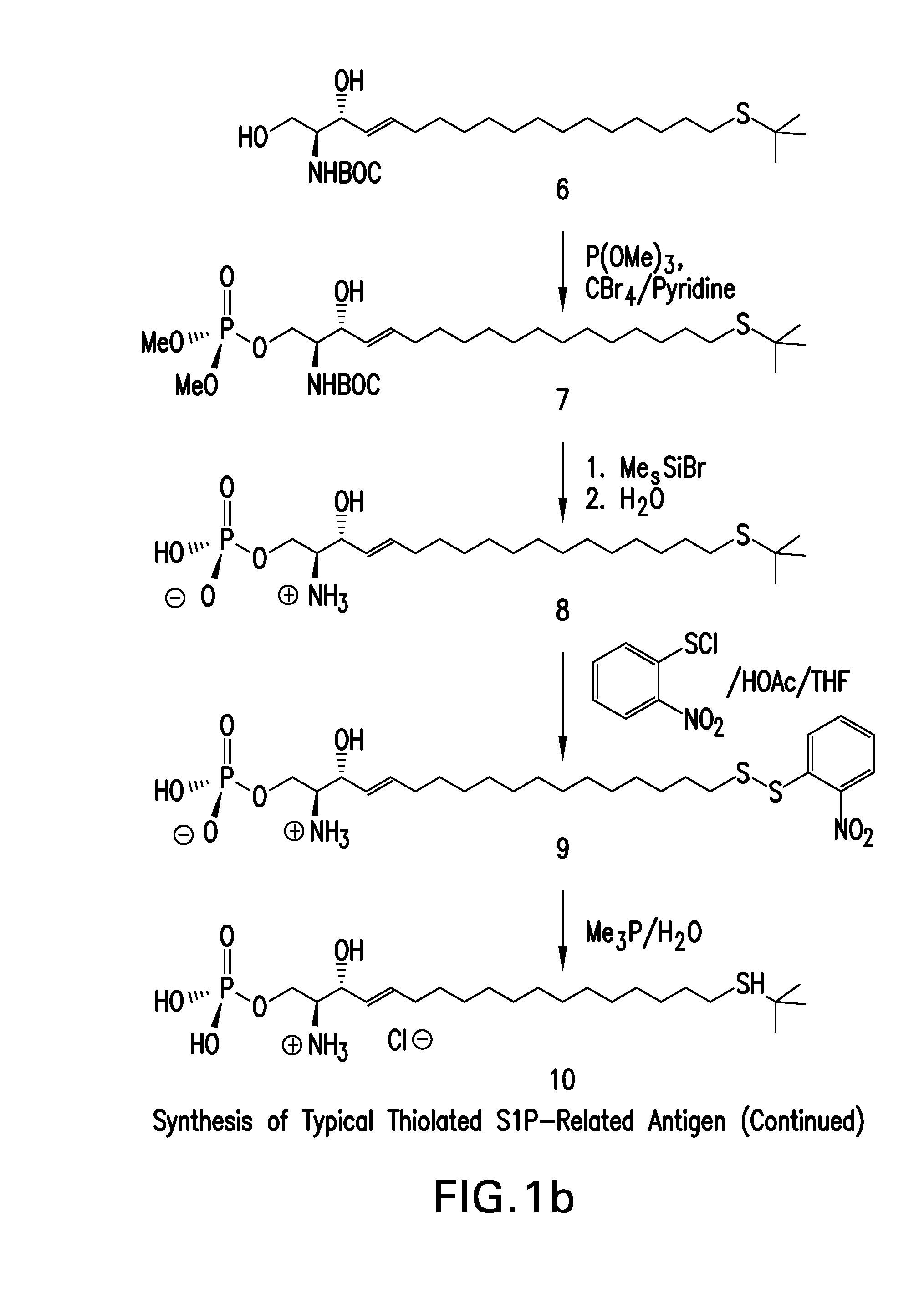
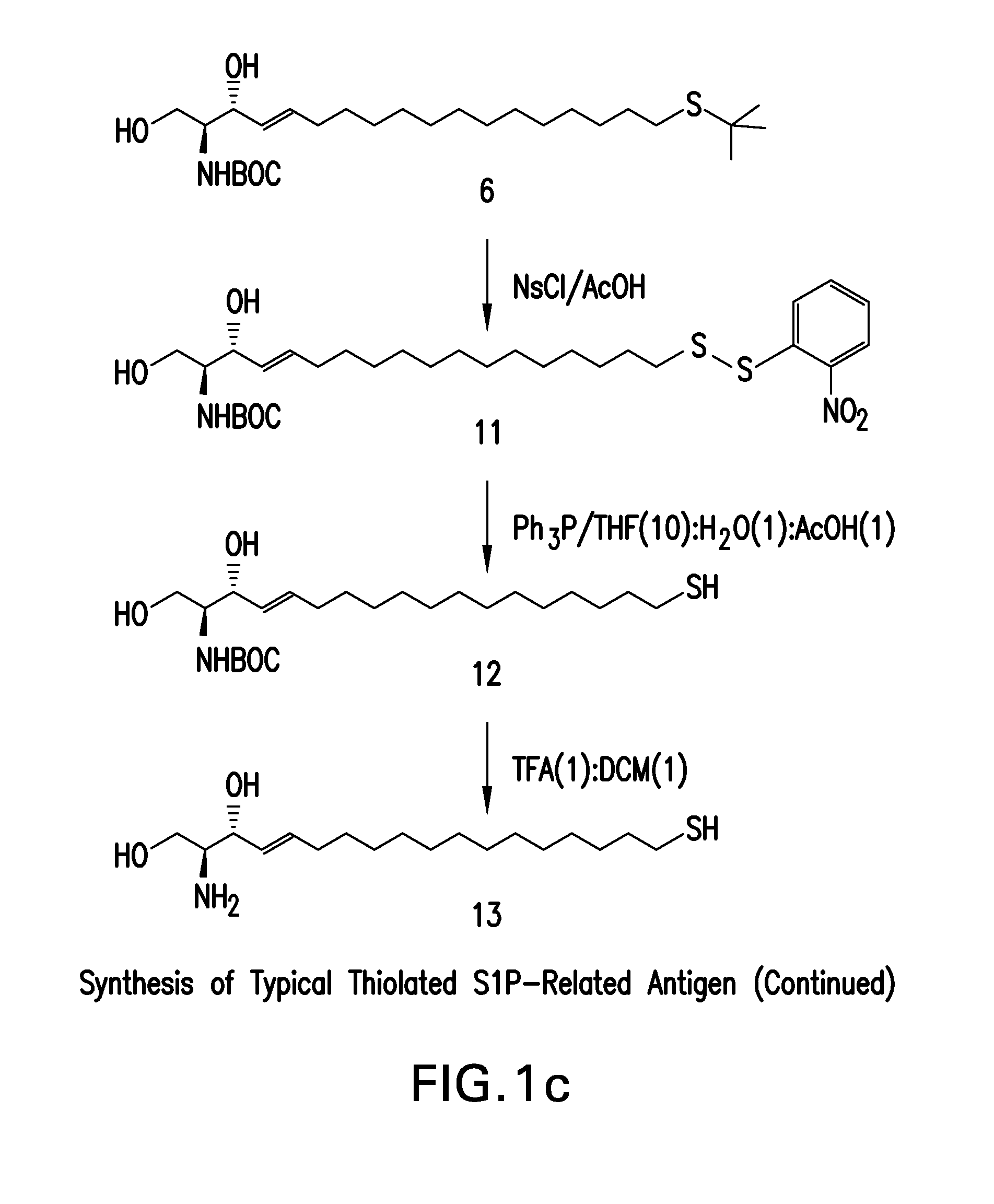
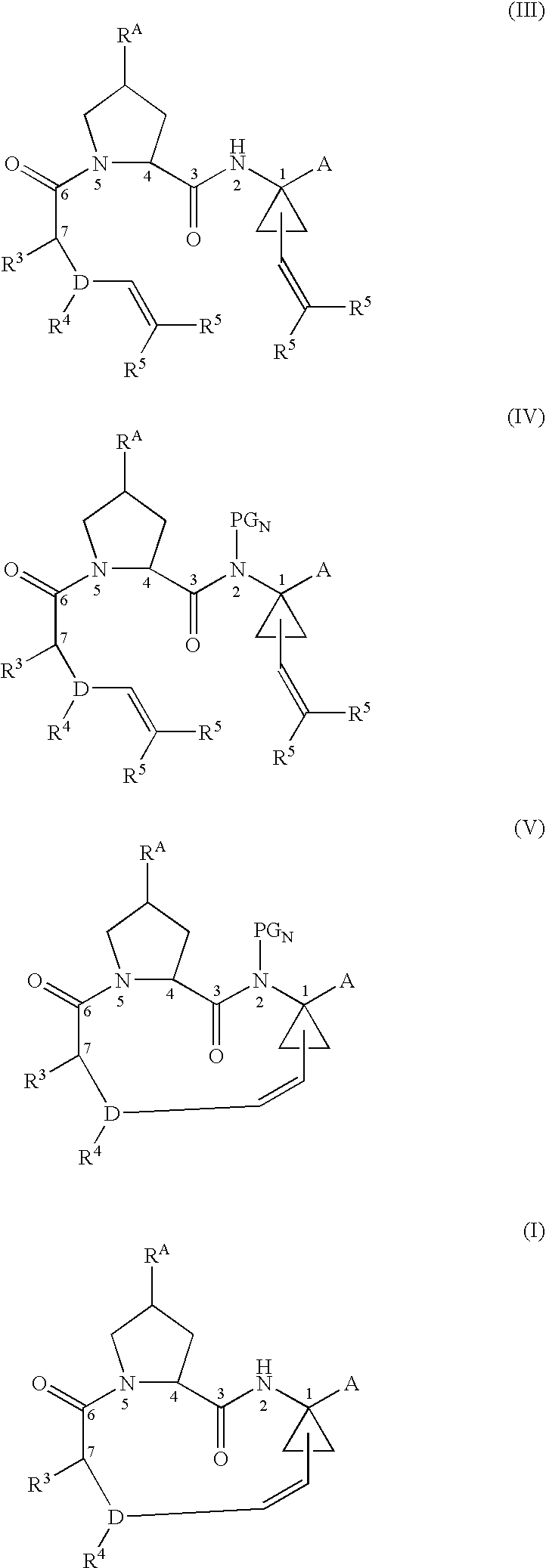
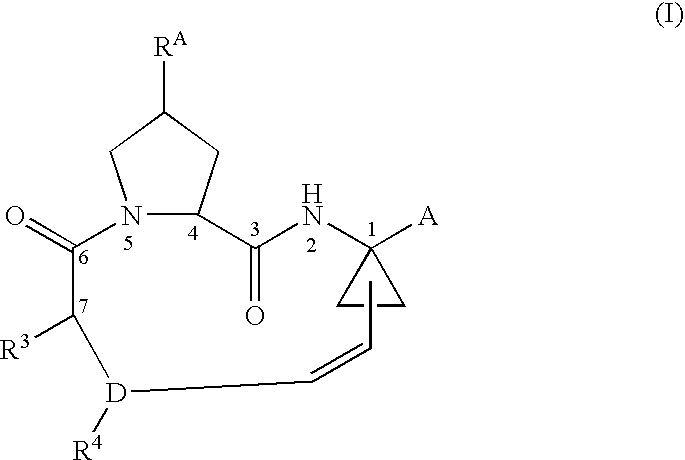
Ring-Closing Metathesis Process for the Preparation of Macrocyclic Peptides
Disclosed is a process for preparing a compound of formula (I) by protecting the secondary amide nitrogen atom in the compound of formula (III) to obtain (IV) wherein PGN is a suitable nitrogen protecting group, ring-closing the compound of formula (IV) by cyclizing it in the presence of a suitable catalyst in a suitable organic solvent to obtain (V), and then deprotecting the resulting compound of formula (V) to obtain (I), as outlined in the following scheme. The compounds of formula (I) are active agents for the treatment of hepatitis C viral (HCV) infections or are intermediates useful for the preparation of anti-HCV agents.
Owner:BOEHRINGER INGELHEIM INT GMBH
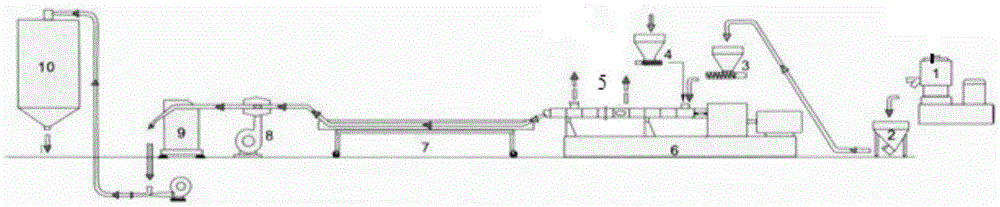
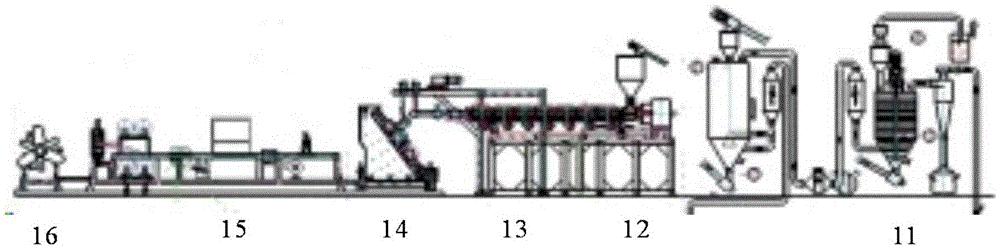
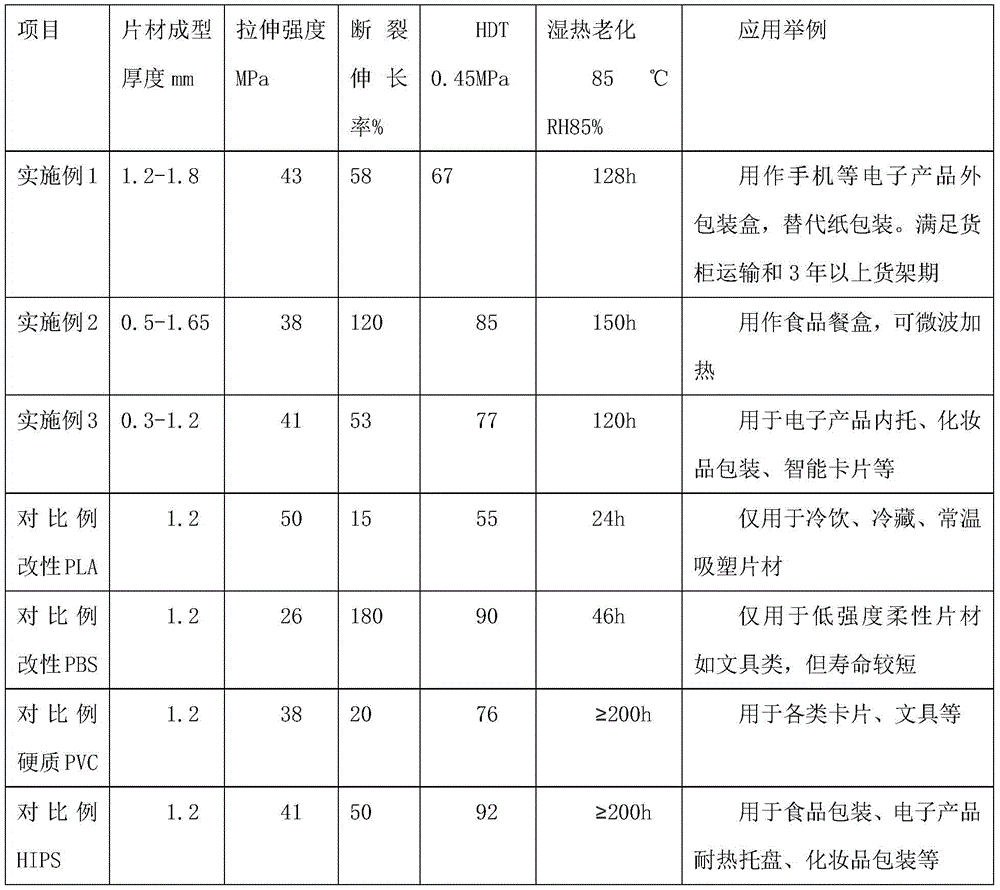
Biodegradable heat-resistant sheet and preparation method thereof
The invention provides a biodegradable heat-resistant sheet and a preparation method thereof. The biodegradable heat-resistant sheet is prepared from, by weight, 20-50% of polylactic acid, 35-55% of PBS, 0-10% of polyester, 1-25% of nucleation enhancer, 0.5-5% of an auxiliary and 0-0.5% of an anti-UV agent. The biodegradable heat-resistant sheet and the preparation method solve the problems that existing polylactic acid sheets are high in brittleness, not resistant to heat, high in sheet rolling cost, narrow in application range and the like. The biodegradable sheet is low in cost, can be efficiently produced, has good physical and mechanical properties, machinability and heat resistance, and can be widely applied to the fields such as electronic products, cosmetics and disposable heat-resistance articles. The application range of biodegradable products is widened.
Owner:SHENZHEN GREENNATURE BIODEGRADABLE TECH LTD
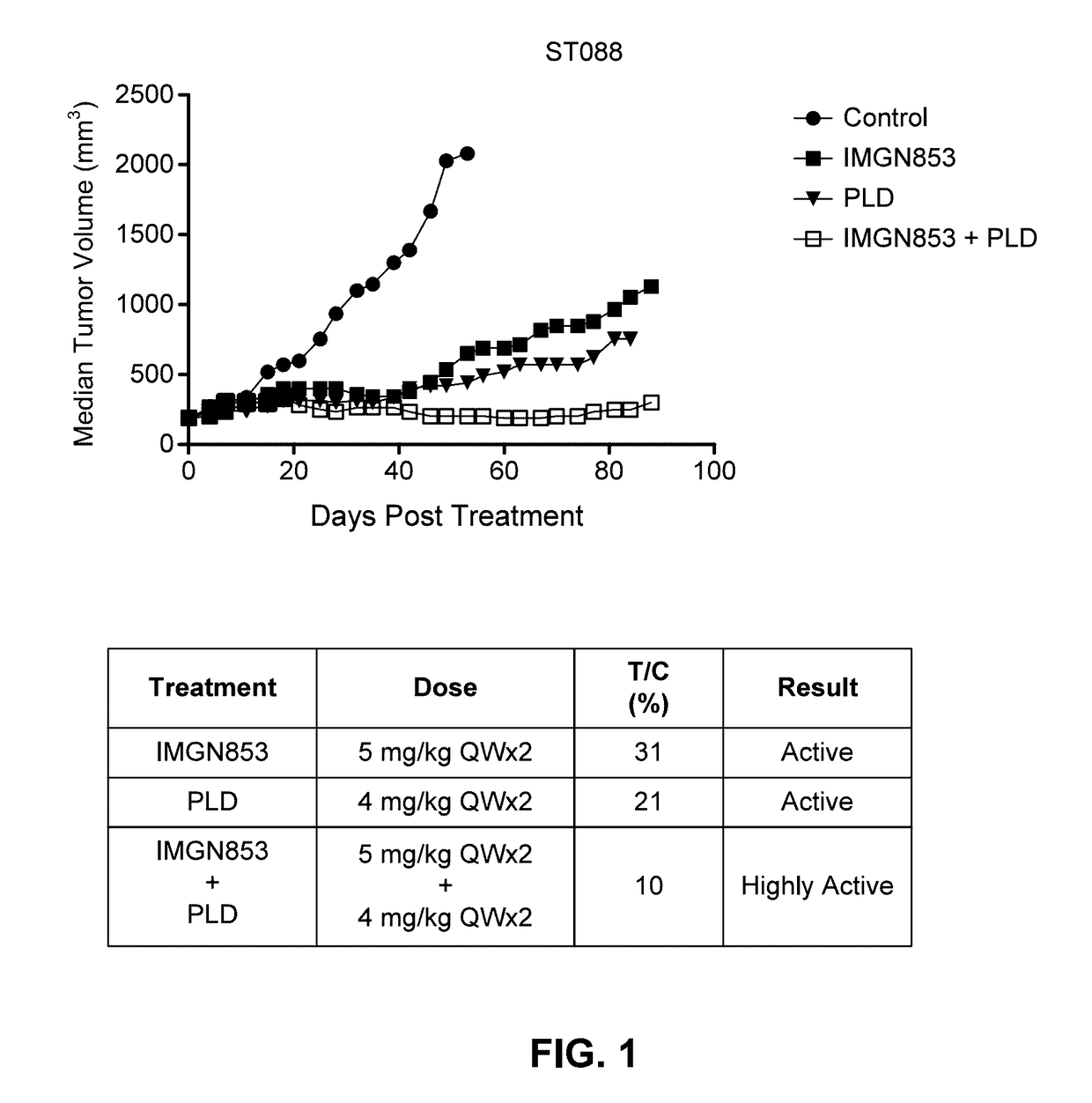
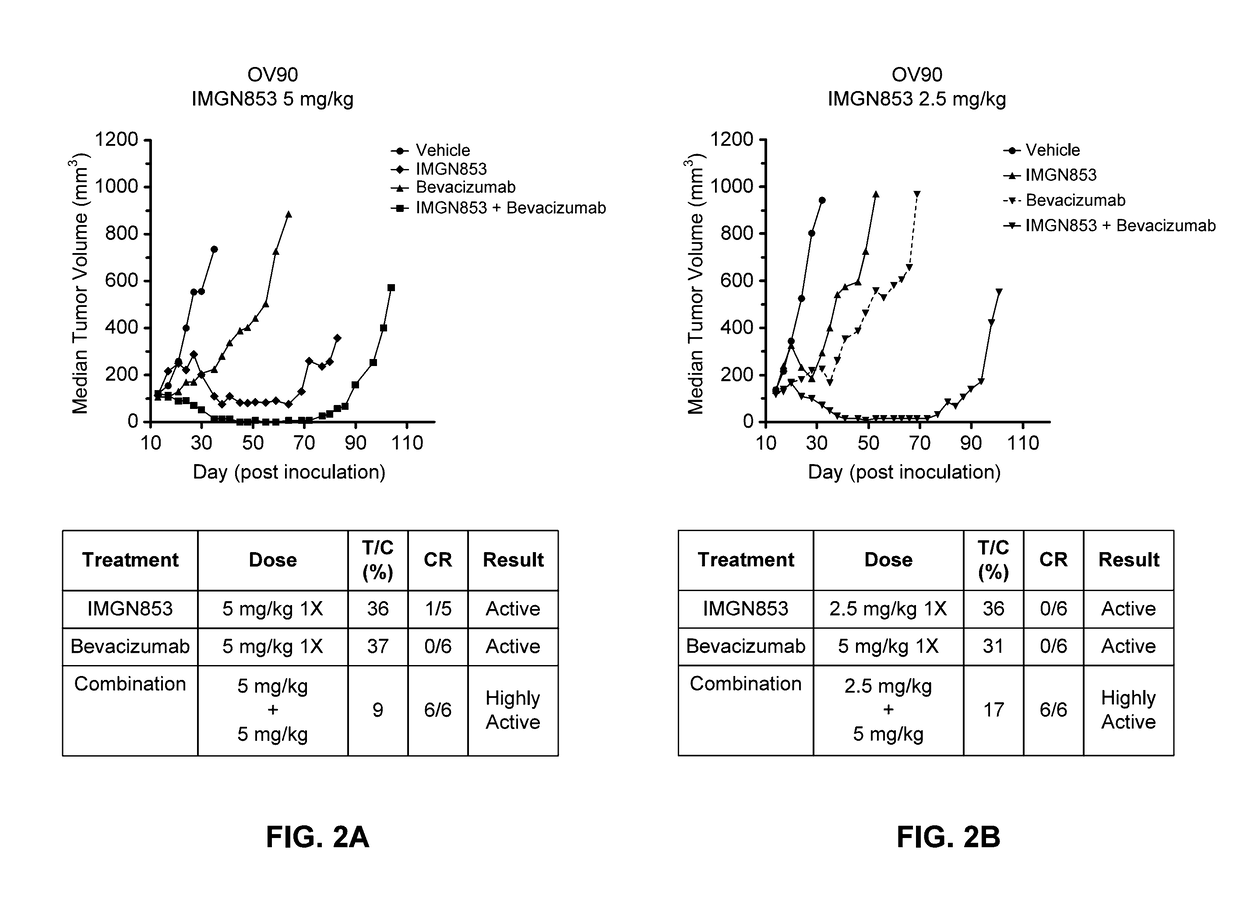
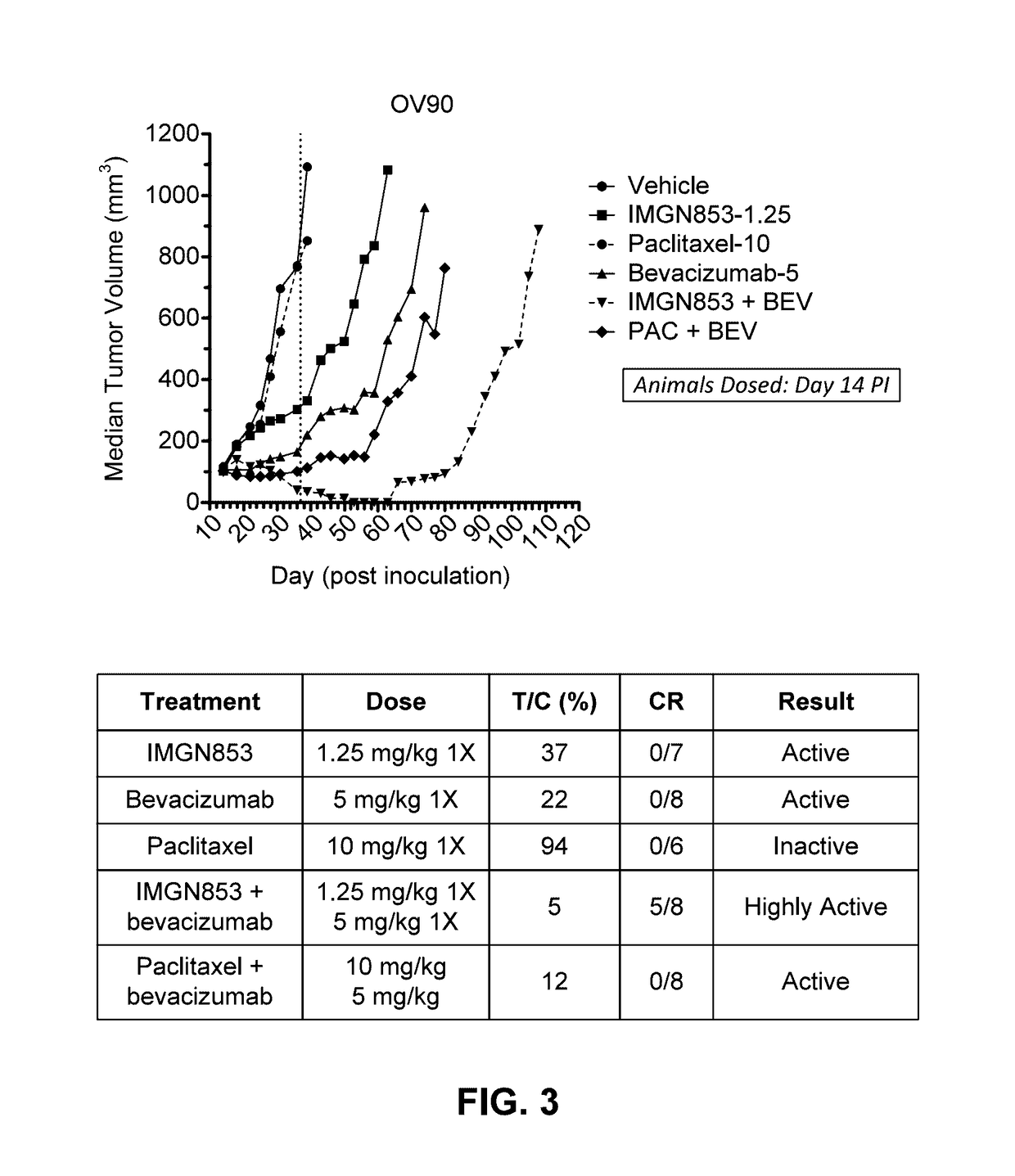
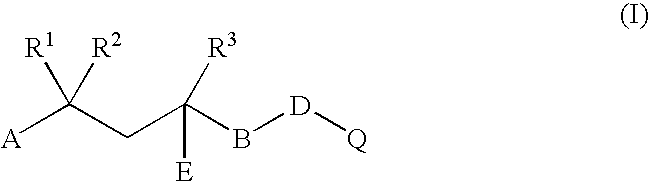
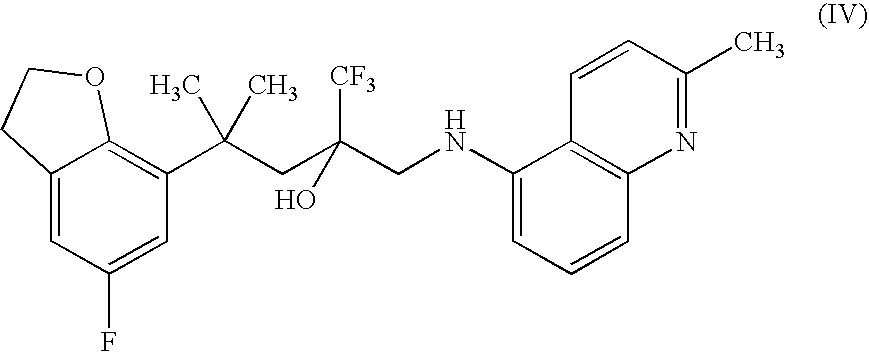
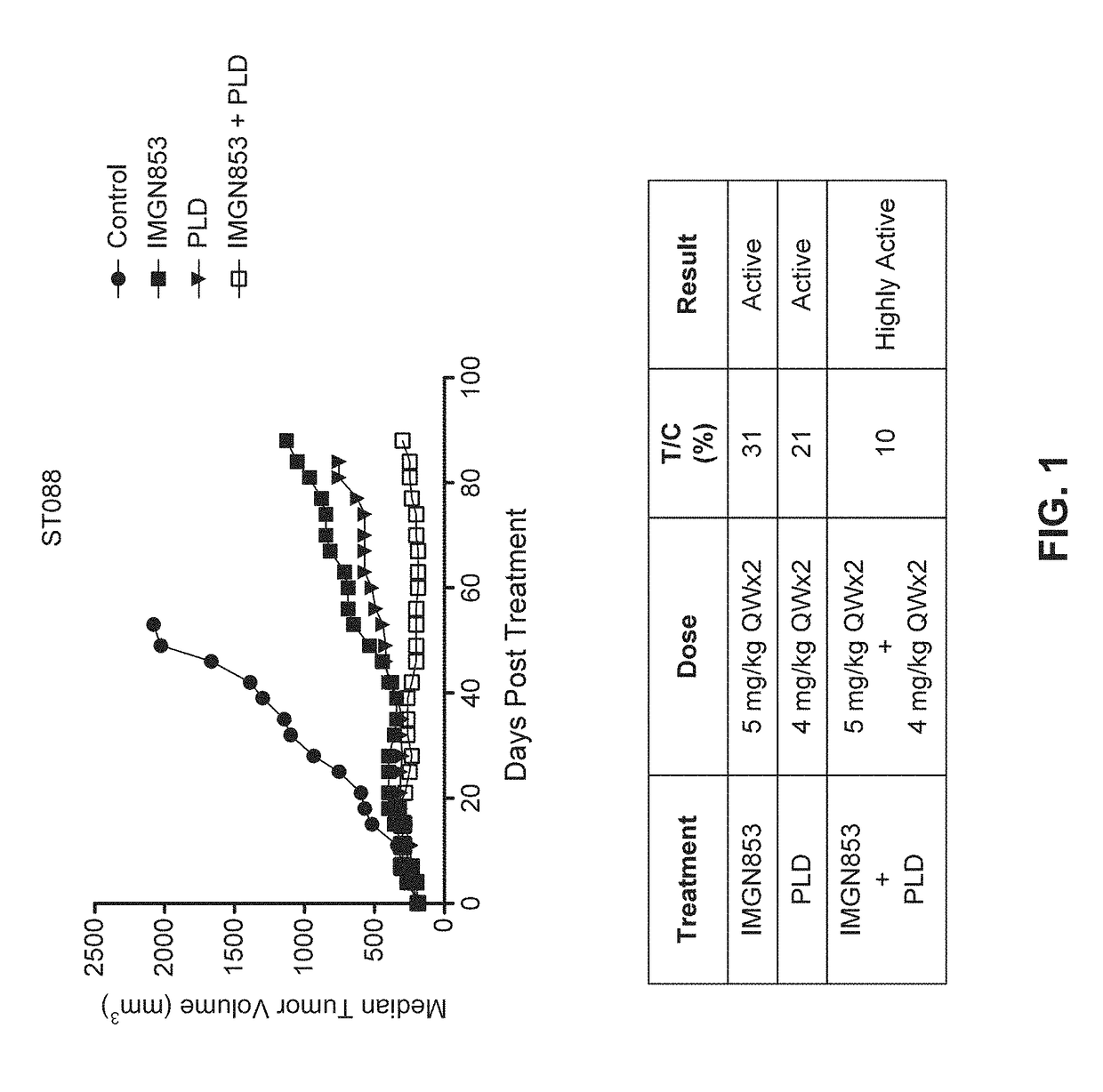
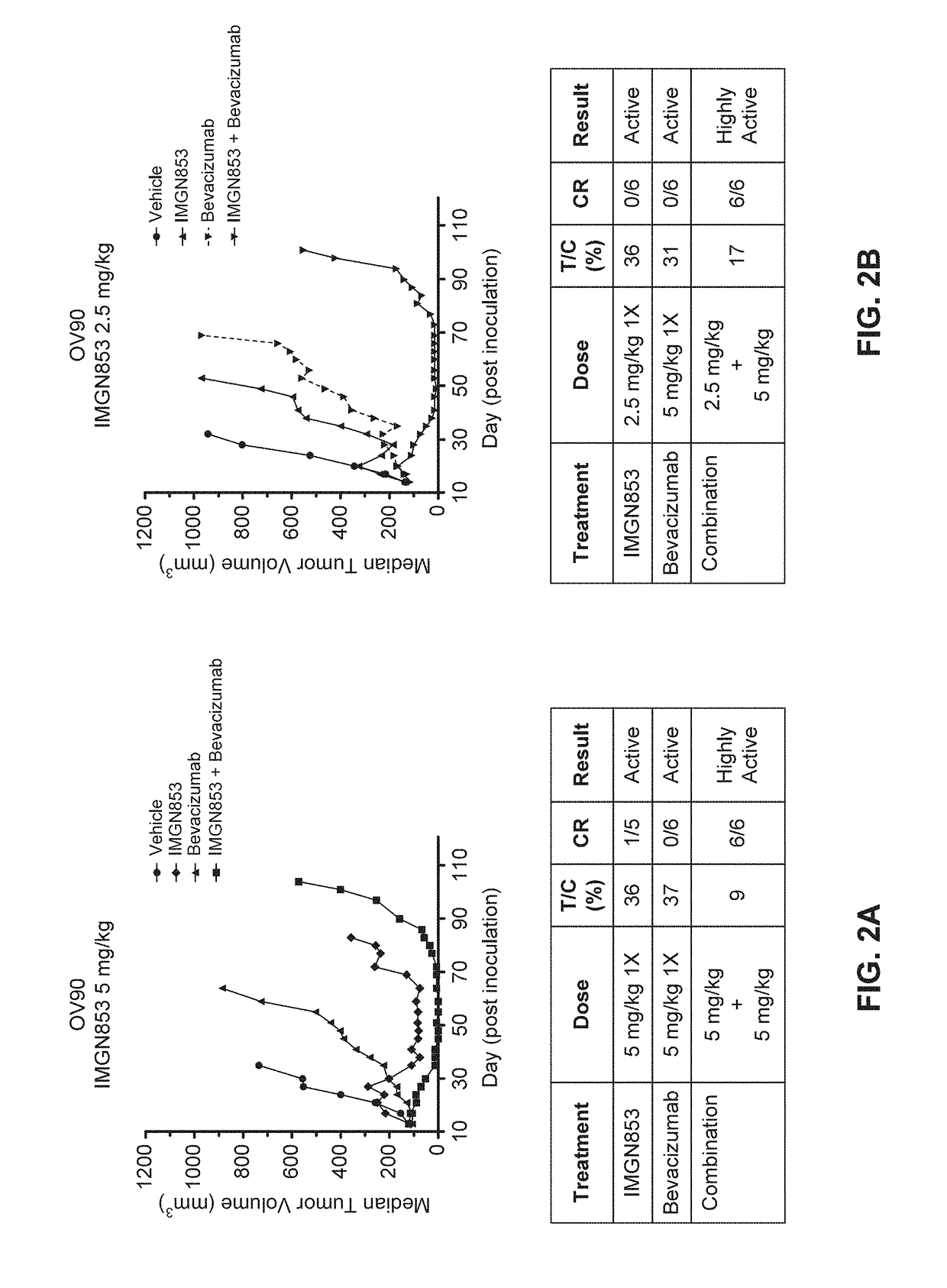
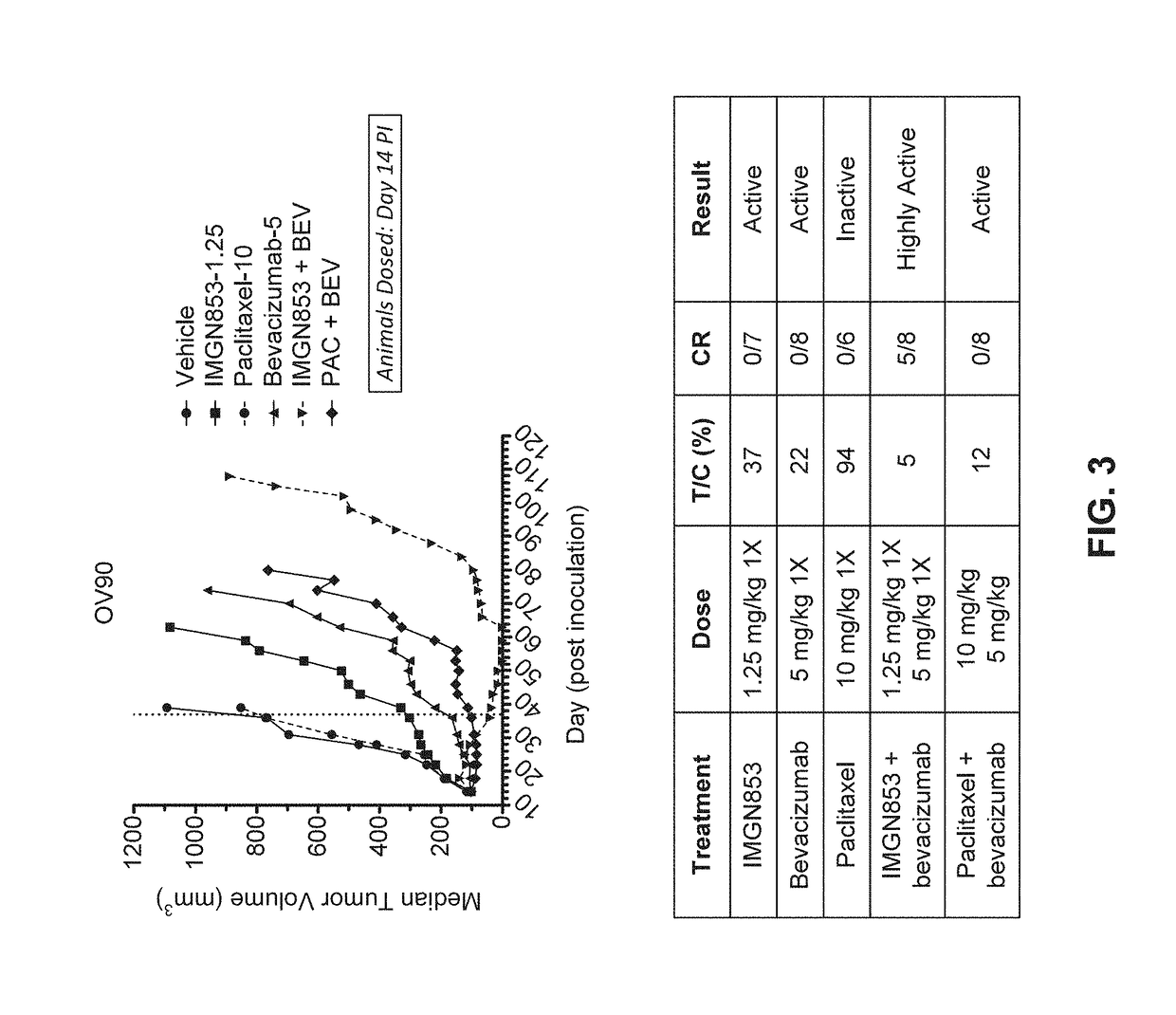
Therapeutic Combinations Comprising Anti-FOLR1 Immunoconjugates
ActiveUS20170095571A1Synergistic efficacyImprove efficacyHeavy metal active ingredientsOrganic active ingredientsClinical efficacyCurative effect
Therapeutic combinations of immunoconjugates that bind to FOLR1 (e.g., IMGN853) with anti-VEGF agents (e.g., bevacizumab), a platinum-based agent, and / or doxorubicin are provided. Methods of administering the combinations to treat cancers, e.g., ovarian cancers, with greater clinical efficacy and / or decreased toxicity are also provided.
Owner:IMMUNOGEN INC
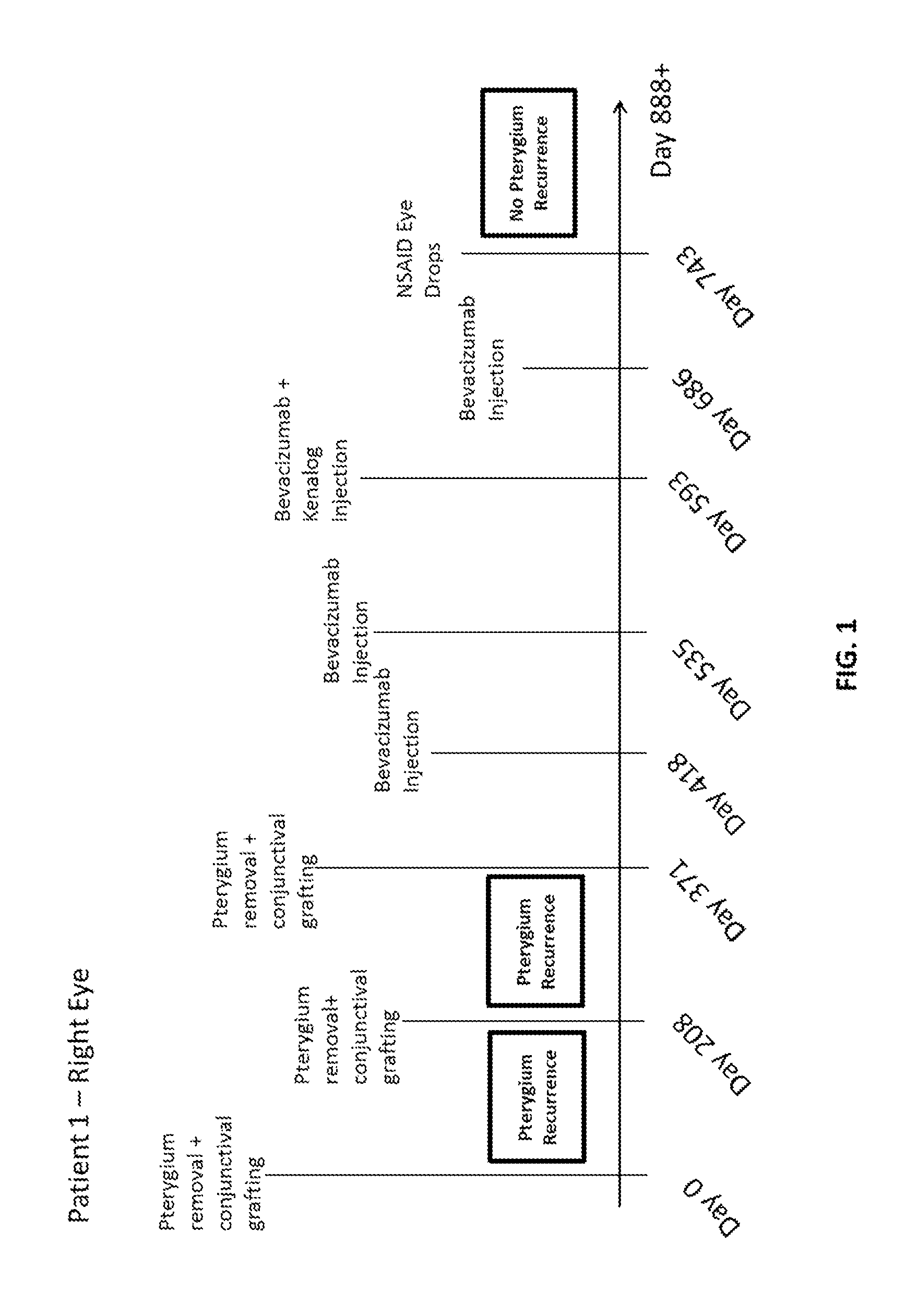
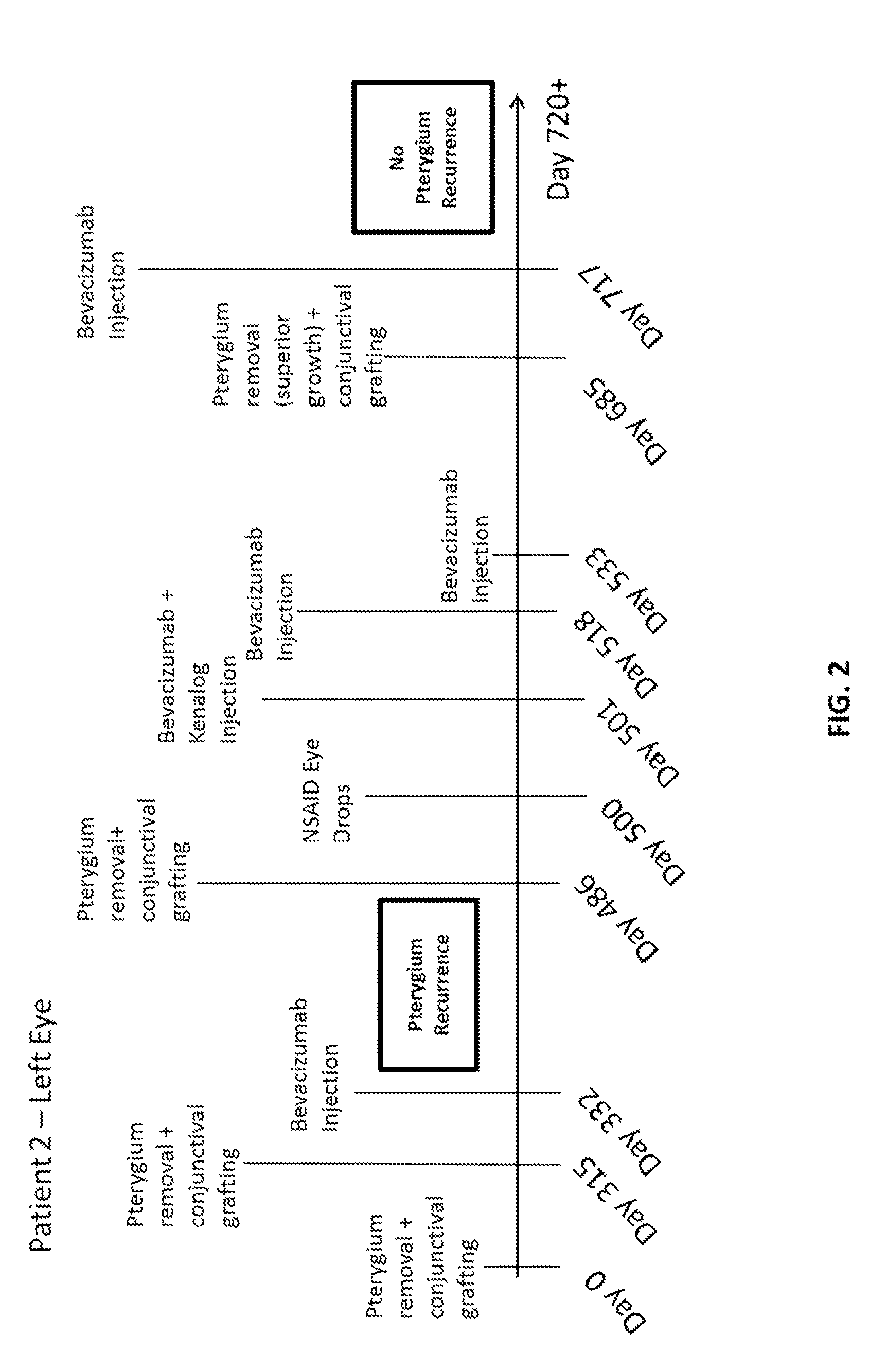
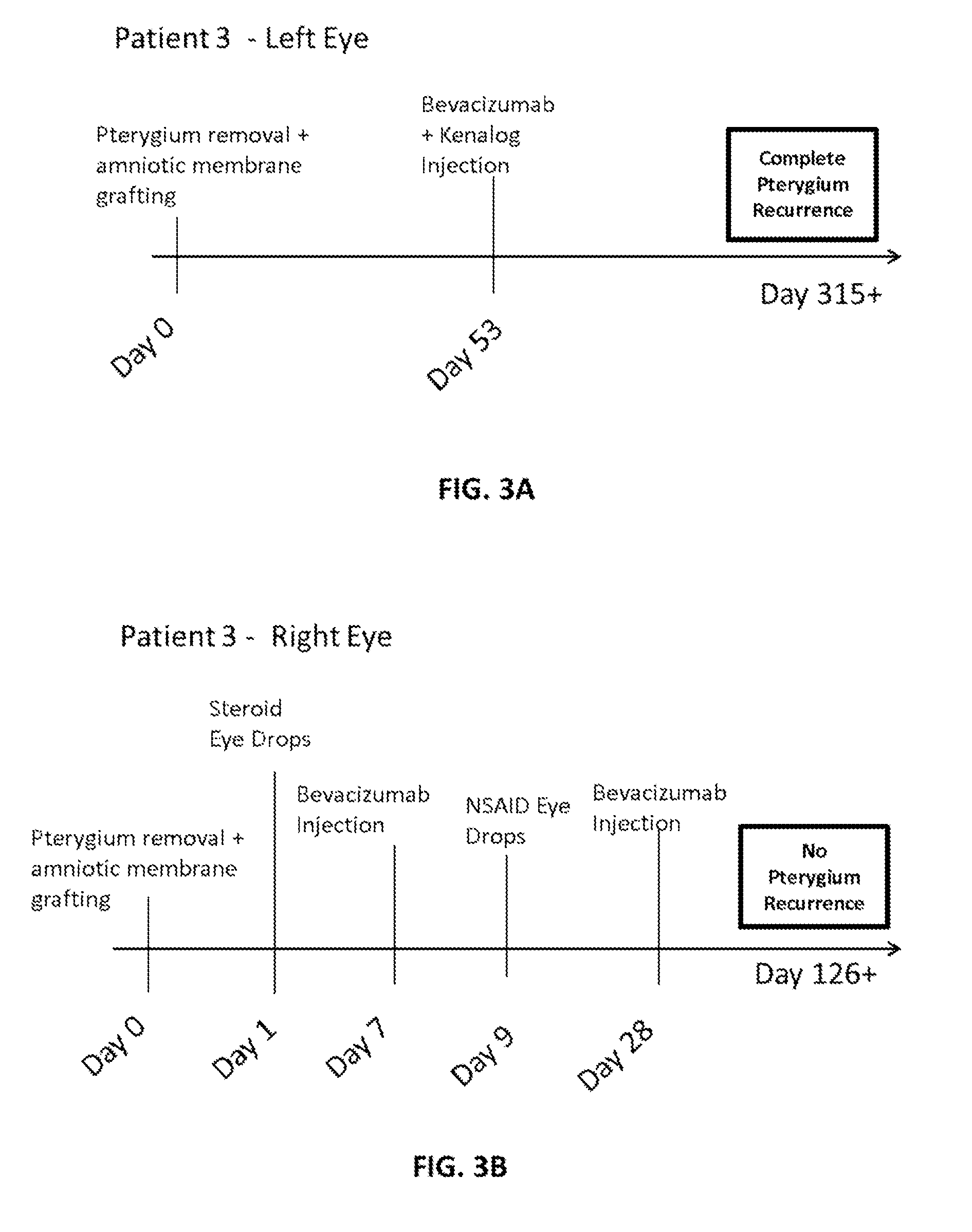
Methods of treating pterygium
Methods for treating pterygium recurrence following pterygiectomy, and for treating keloid recurrence, following surgical removal of the keloid, are disclosed. The methods include administering an anti-VEGF agent (e.g., antibody (e.g., bevacizumab) or small molecule inhibitor of VEGF signaling), or a combination therapy that includes co-administering an anti-VEGF agent, with an anti-inflammatory steroid and / or a non-steroidal anti-inflammatory drug (NSAID) to a subject.
Owner:PHAM RANDAL TANH HOANG
Quinolizinone compound and use thereof as HIV integrase inhibitor
InactiveUS20060084665A1Less side effectsStrong inhibitory activityBiocideOrganic chemistrySide effectReverse transcriptase
A pharmaceutical agent having an anti-HIV action, particularly, a pharmaceutical agent having an integrase inhibitory action, is provided. The present invention relates to a quinolizinone compound represented by the following formula [I]wherein each symbol is as defined in the specification, a pharmaceutically acceptable salt thereof, and an anti-HIV agent containing same as an active ingredient. The compound of the present invention has an HIV integrase inhibitory action and is useful as an anti-HIV agent for the prophylaxis or therapy of AIDS. Moreover, by a combined use with other anti-HIV agents such as protease inhibitors, reverse transcriptase inhibitors and the like, the compounds can become a more effective anti-HIV agent. Since the compound has a high inhibitory activity specific for integrases, the compound can provide a safe pharmaceutical agent for human with a fewer side effects.
Owner:JAPAN TOBACCO INC
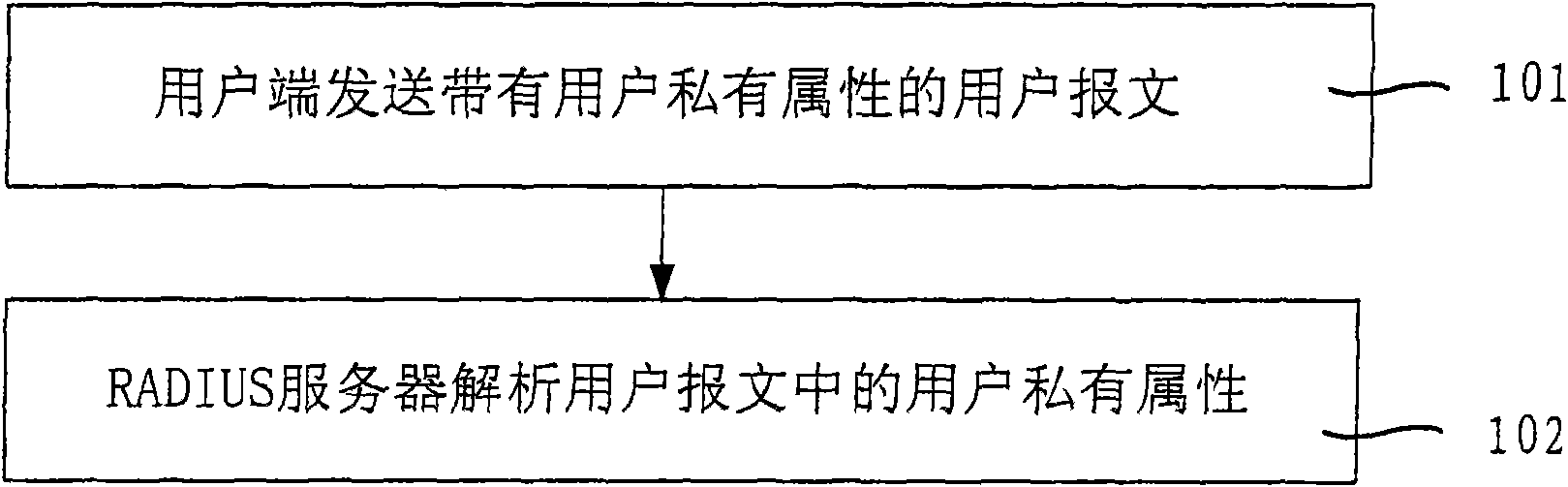
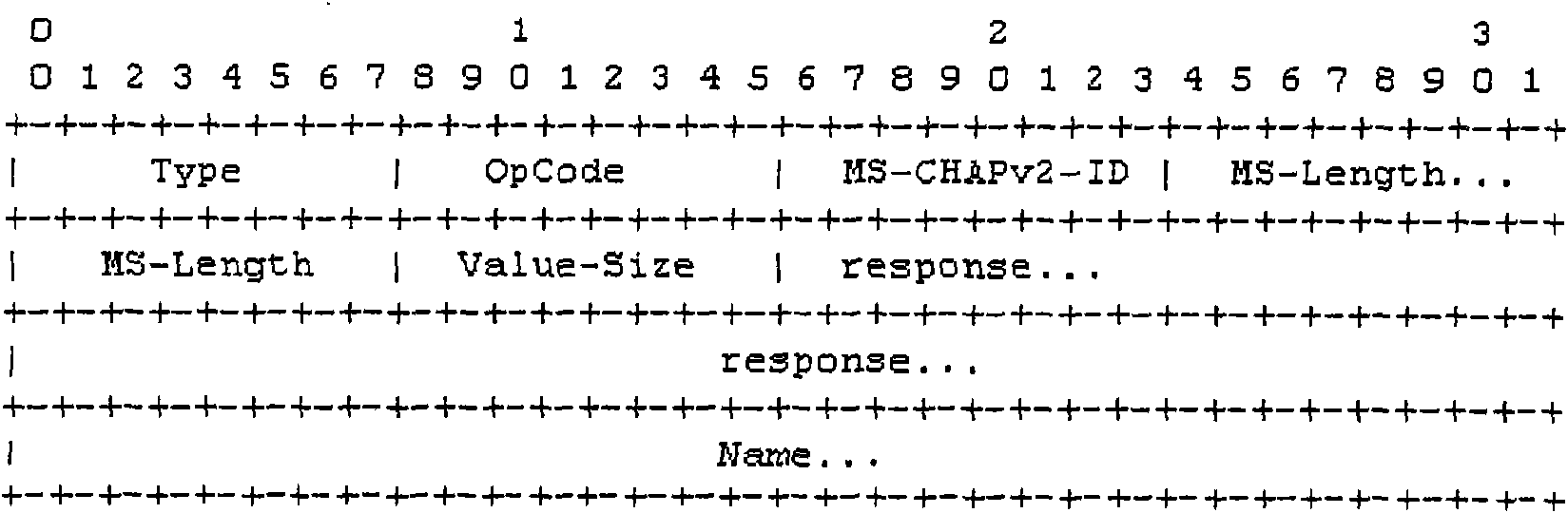
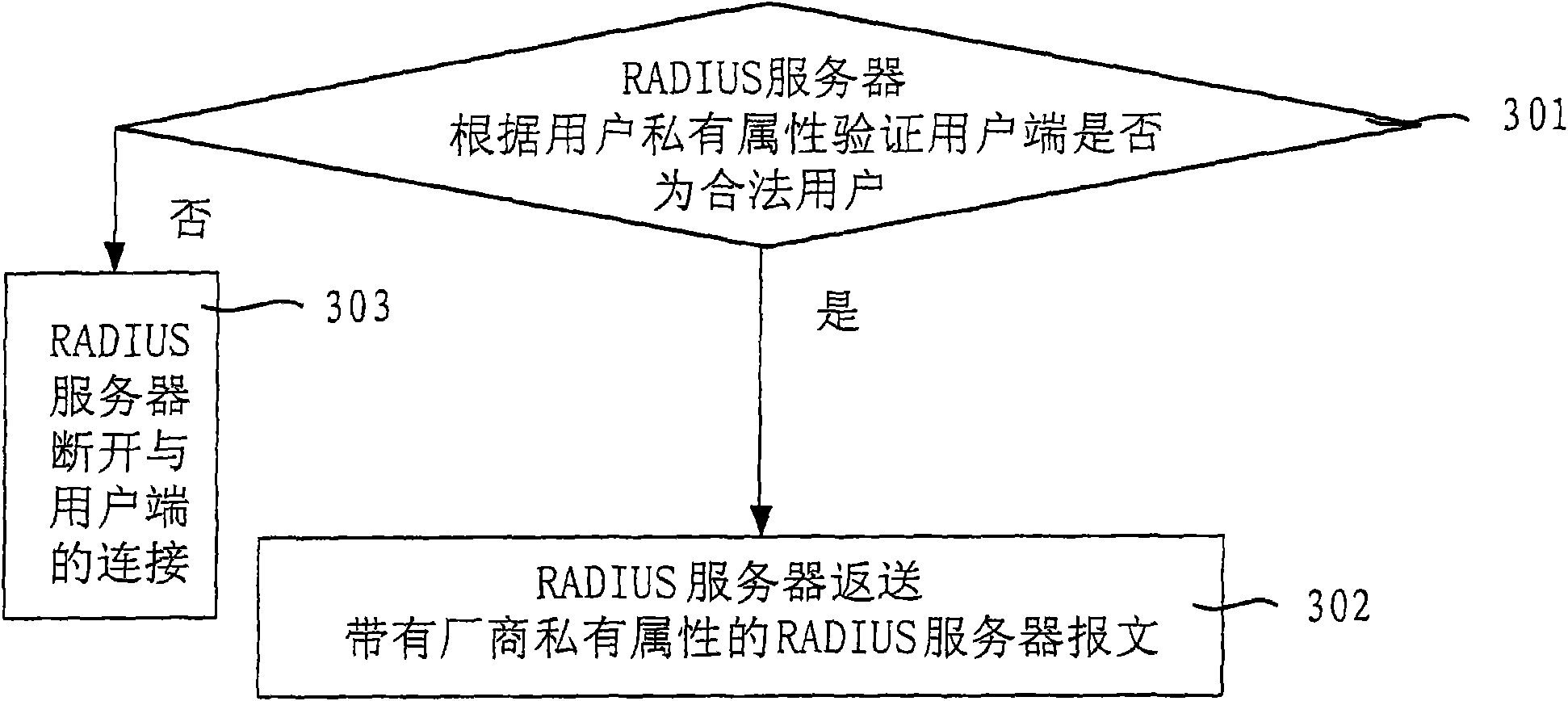
Method and system for delivering private attribute information
InactiveCN101640685AImprove securityImprove compatibilityUser identity/authority verificationOperational systemMessage delivery
The invention relates to a method and a system for delivering private attribute information, wherein the method comprises the following steps: a user terminal sends a user message with user private attribute; and a RADIUS server analyzes the user private attribute in the user message. The user private attribute is sent to the RADIUS server by the user message and the RADIUS server sends private attribute of a manufacturer to the user terminal, therefore, the embodiment of the invention enhances the safety and the compatibility of the user terminal and the RADIUS server during the delivery ofthe message comprising the private attribute and improves the capacity and the efficiency of message delivery. The user terminal obtains the address of a downloaded software, a user terminal system upgrading or anti-agent identifier and the like according to the private attribute of the manufacturer, thereby ensuring that the version of an operating system of the user terminal can be updated in time and the system is upgraded successfully, preventing the operating system of the user terminal from easily being attacked by virus, and avoiding the information of the user terminal being cracked or pirated by other persons.
Owner:RUIJIE NETWORKS CO LTD
4-Oxoquinoline compound and use thereof as HIV integrase inhibitor
ActiveUS20060217413A1Promote absorptionIncreased riskBiocideOrganic chemistrySide effectReverse transcriptase
Owner:JAPAN TOBACCO INC
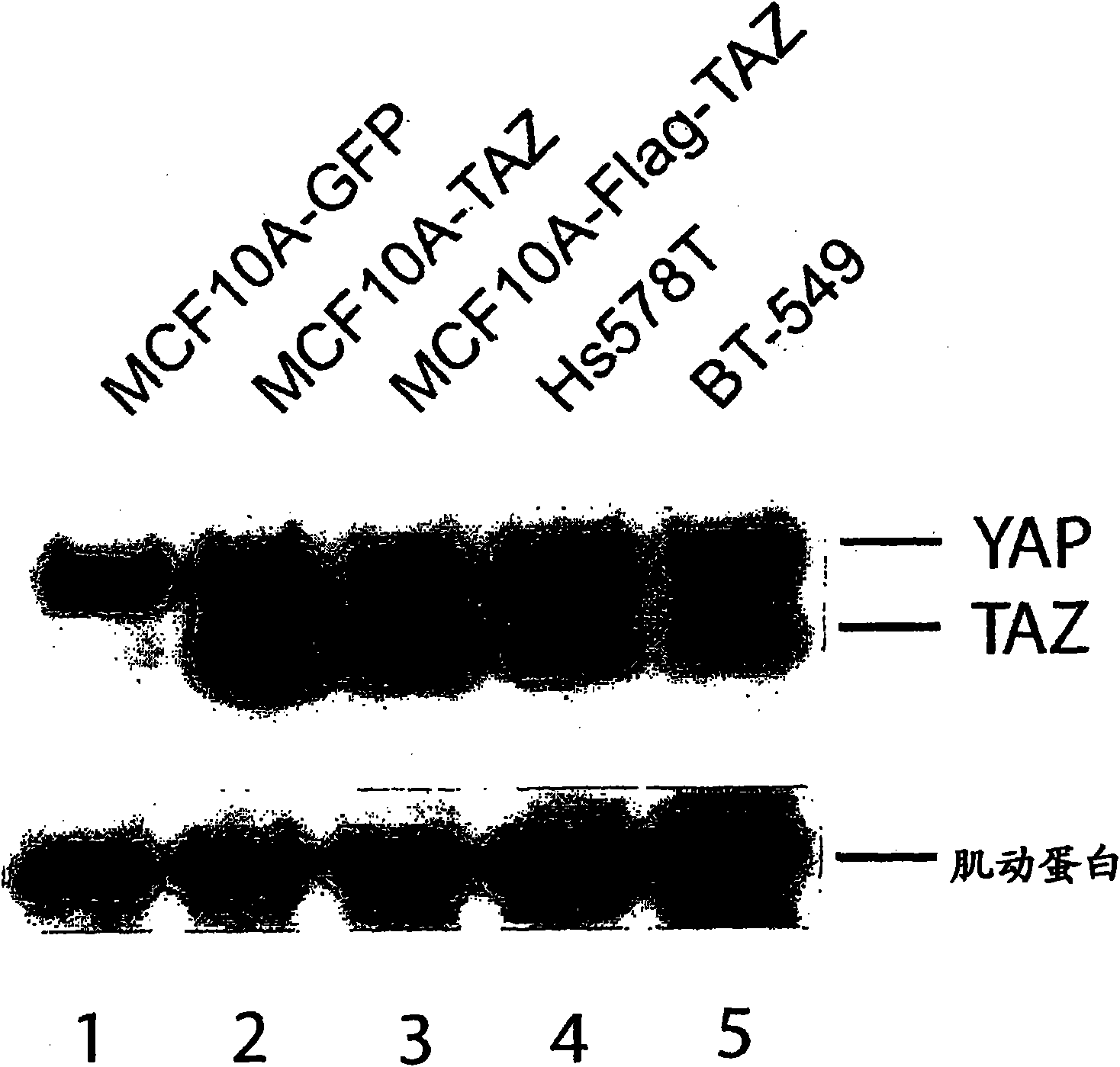
TAZ/WWTR1 for diagnosis and treatment of cancer
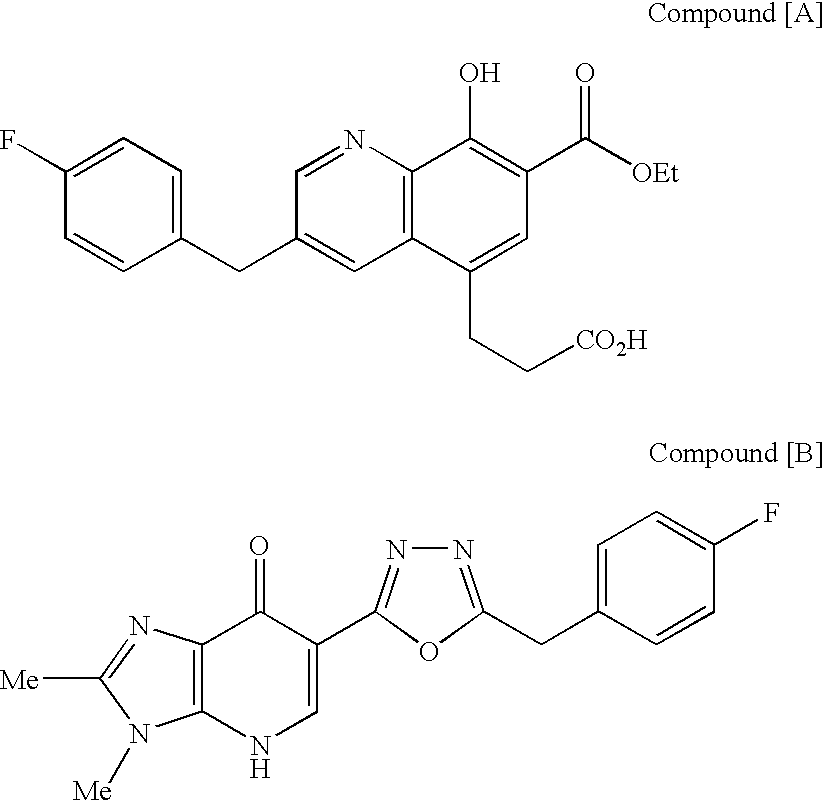
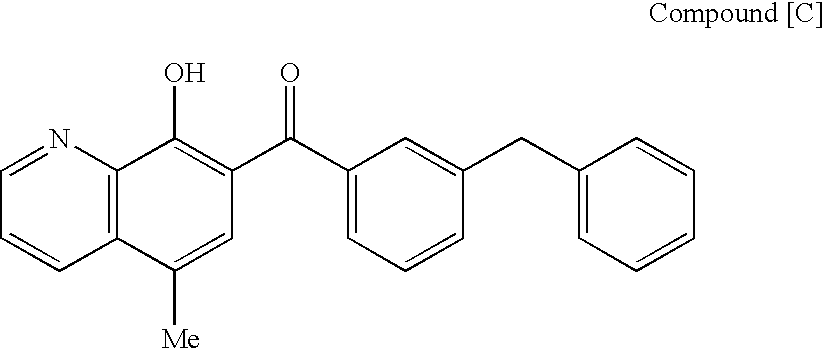
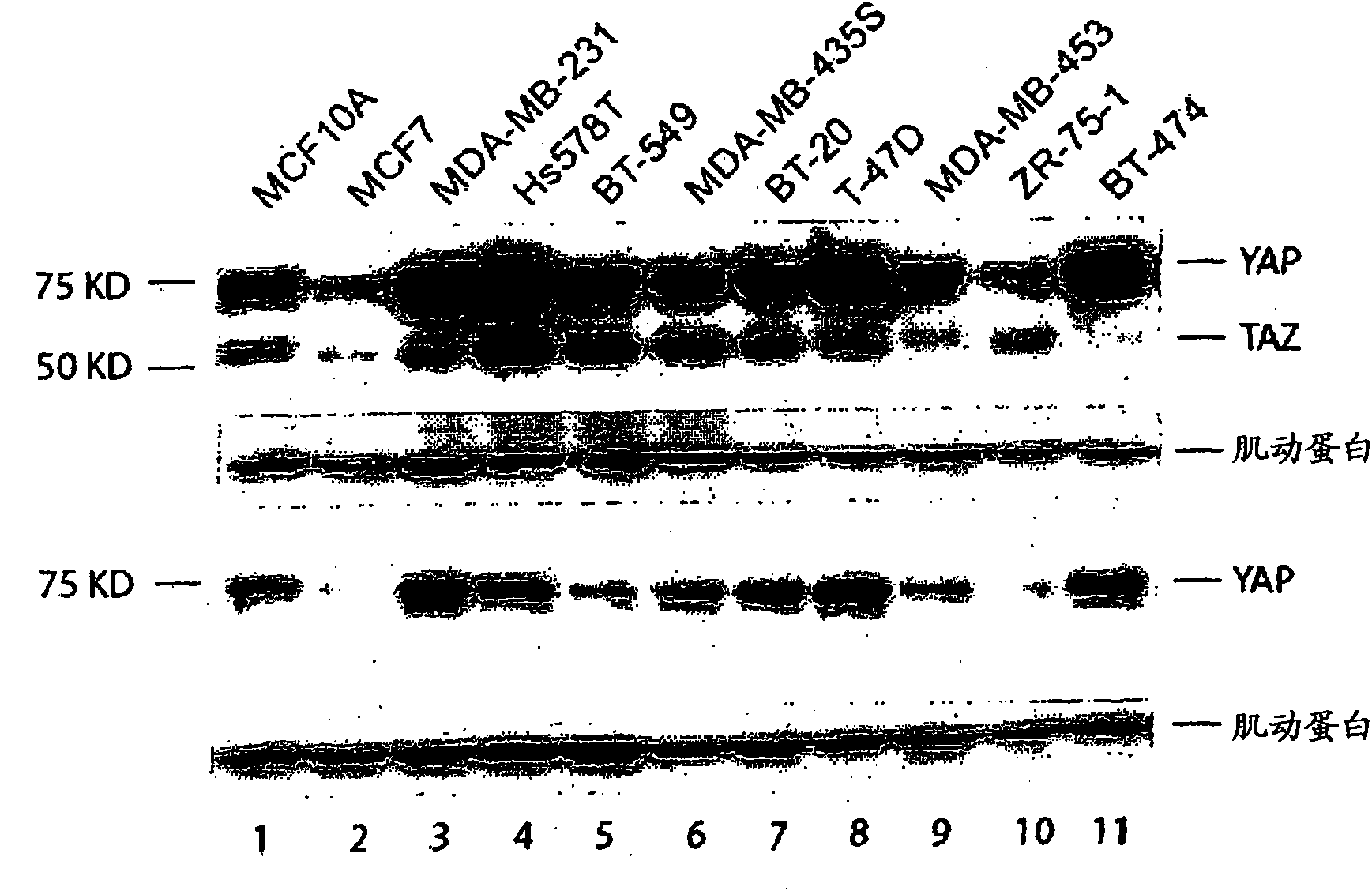
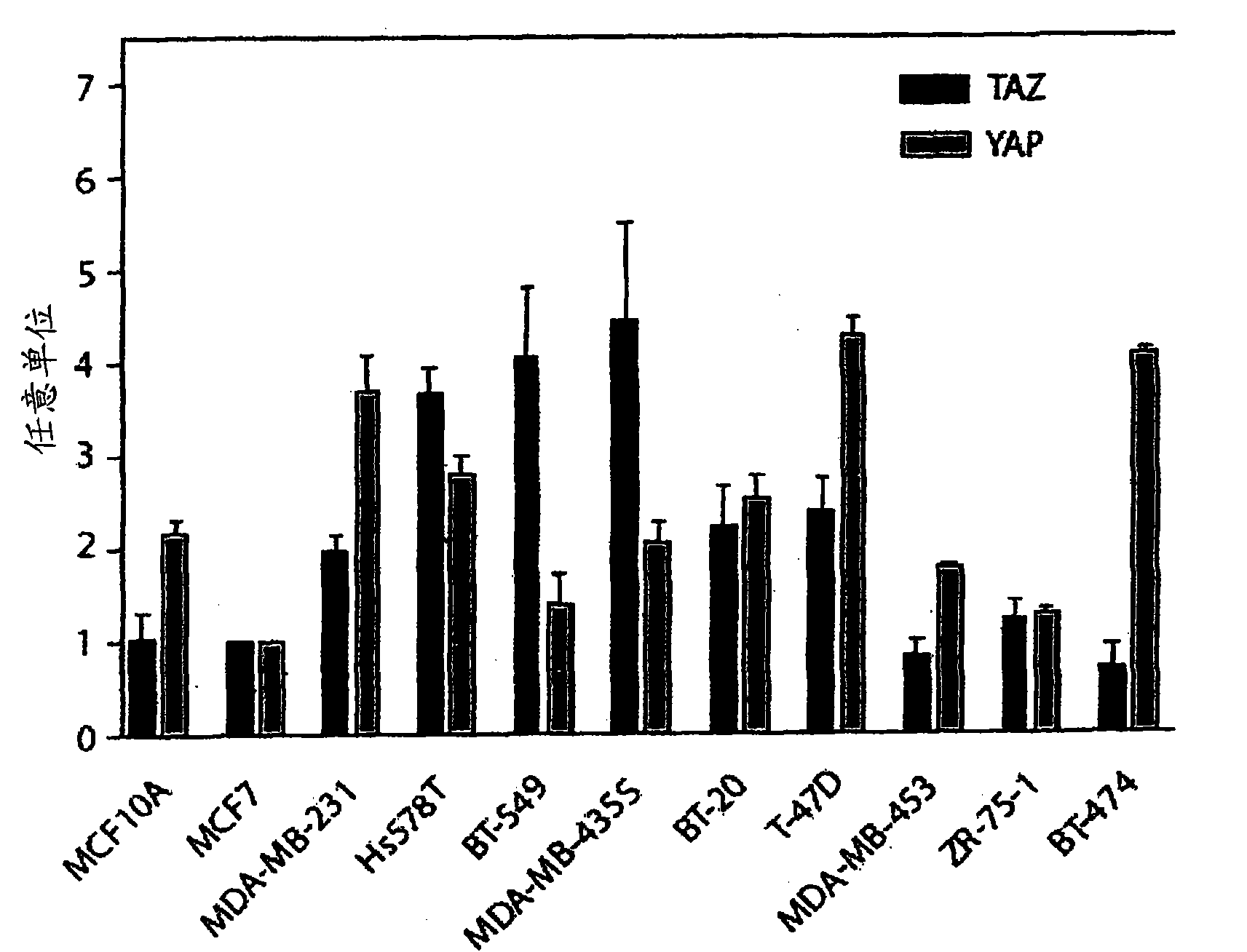
The invention provides an anti-TAZ agent for the treatment, prophylaxis or alleviation of cancer. We further provide a kit for detecting breast cancer in an individual or susceptibility of the individual to breast cancer comprising means for detection of TAZ expression in the individual or a sample taken from him or her as well as a method of detecting a cancer cell, the method comprising detecting modulation of expression, amount or activity of TAZ in the cell.
Owner:AGENCY FOR SCI TECH & RES
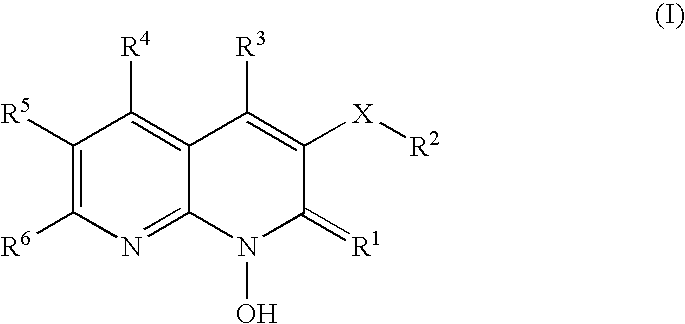
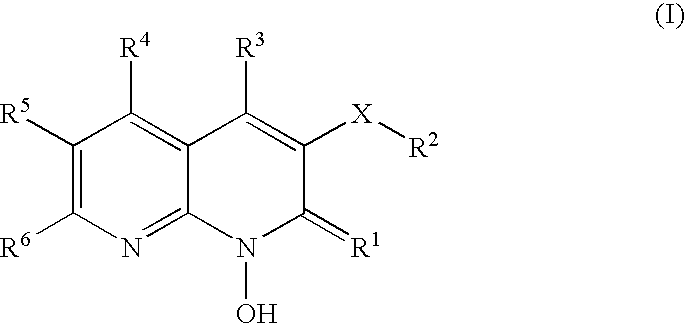
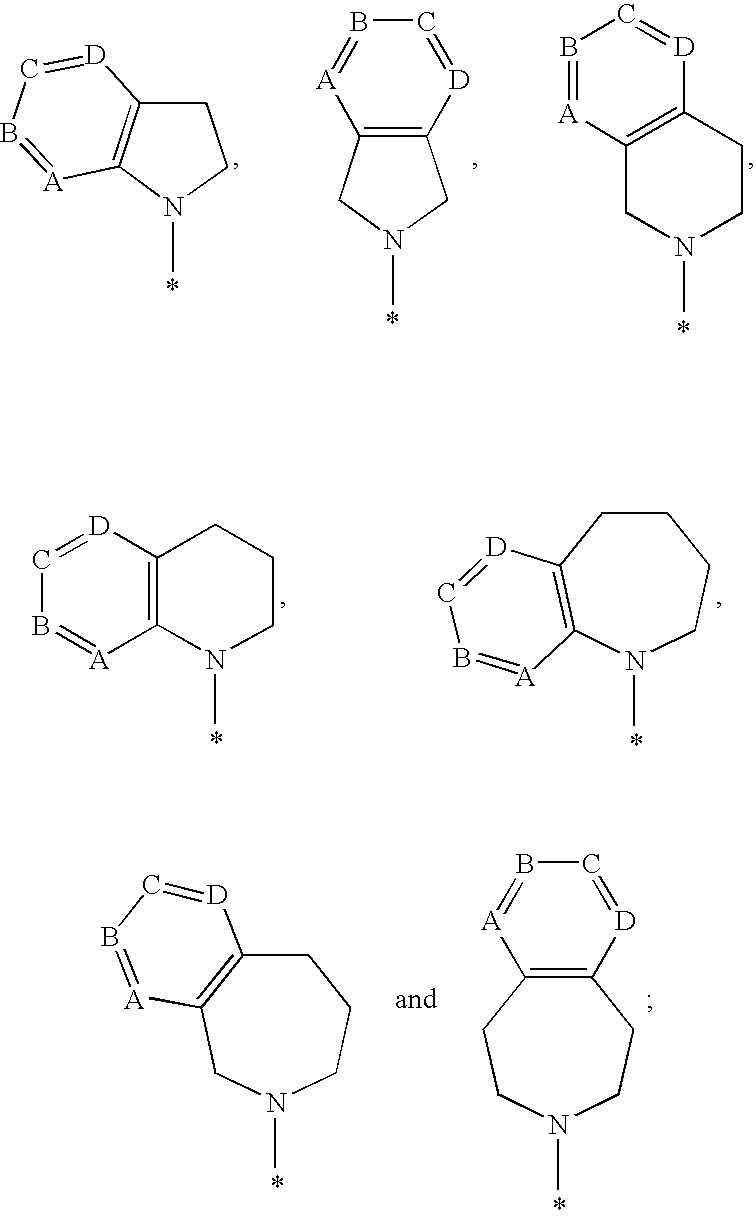
1-hydroxy naphthyridine compounds as Anti-hiv agents
1-Hydroxy naphthyridine compounds (e.g., 1-hydroxy naphthyridin-2(1H)-one compounds of Formula I are inhibitors of HIV integrase and / or HIV RNase H and inhibitors of HIV replication: (I) wherein X and R1-R6 are as defined herein. The compounds are useful in the prophylaxis and treatment of infection by HIV and in the prophylaxis, delay in the onset, and treatment of AIDS. The compounds are employed against HIV infection and AIDS as compounds per se or in the form of pharmaceutically acceptable salts. The compounds and their salts can be employed as ingredients in pharmaceutical compositions, optionally in combination with other anti-HIV agents such as HIV antivirals, immunomodulators, antibiotics and vaccines.
Owner:MERCK SHARP & DOHME CORP
Compositions and Methods for Treating, Controlling, Reducing, Ameliorating, or Preventing Allergy
InactiveUS20080064721A1Treating and controlling and reducing and ameliorating and preventing allergyBiocideSenses disorderAllergyPharmaceutical Substances
A composition for treating, controlling, reducing, ameliorating, or preventing allergy comprises a dissociated glucocorticoid receptor agonist (“DIGRA”), a prodrug thereof, a pharmaceutically acceptable salt thereof, or a pharmaceutically acceptable ester thereof. The composition can comprise an anti-allergic medicament and / or an additional anti-inflammatory agent and can be formulated for topical application, injection, or implantation. The anti-allergic medicament can comprise an antihistamine, a mast-cell stabilizer, a leukotriene inhibitor, an immunomodulator, an anti-IgE agent, or a combination thereof.
Owner:BAUSCH & LOMB INC
Methods for preventing toxic drug-drug interactions in combination therapies comprising Anti-erbb3 agents
InactiveUS20140234317A1Prevent and reduce toxicityProvide benefitsBiocideOrganic active ingredientsErlotinibDrug interaction
Methods are disclosed for preventing toxic drug-drug interactions during combination cancer therapy with a drug that is an anti-ErbB3 agent, such as an anti-ErbB3 antibody, together with a drug that is a tyrosine kinase inhibitor and / or a drug that binds to alpha-1 acid glycoprotein (e.g., erlotinib). Health care practitioners obtaining any one of the drugs are warned that when co-administering the drug that is an anti-ErbB3 agent with either or both of a drug that is a tyrosine kinase inhibitor and a drug that binds to alpha-1 acid glycoprotein, at least one of the co-administered drugs should be administered using a reduced dosage to prevent toxicity. In a reduced dosage, the amount of drug administered per unit time is reduced as compared to a dose that would be administered if the drug was administered as monotherapy. The reduced dosage can be, for example, a reduced drug dose or a reduced drug dosing frequency, or both. Compositions useful in practicing the disclosed methods are also provided.
Owner:MERRIMACK PHARMACEUTICALS INC
VHZ for diagnosis and treatment of cancers
We provide VHZ for use in a method of treatment, prophylaxis or alleviation of a cancer in an individual selected from the group consisting of: colon cancer, lung cancer, squamous cell carcinoma including lip, larynx, vulva, cervix and penis cancer, pancreatic cancer, brain cancer, oesophageal cancer, stomach cancer, bladder cancer, kidney cancer, skin cancer, ovary cancer, prostate cancer and testicular cancer. We provide an anti-VHZ agent for the treatment, prophylaxis or alleviation of such a cancer. The anti-VHZ agent may comprise SEQ ID NO:4 or SEQ ID NO: 5, or both.
Owner:AGENCY FOR SCI TECH & RES
Antistatic decontaminating car wax and preparation method of antistatic decontaminating car wax
ActiveCN104231942AExcellent protective polishing functionEasy to useOther chemical processesPolishing compositionsParaffin waxPolyethylene oxide
The invention discloses antistatic decontaminating car wax and a preparation method of the antistatic decontaminating car wax. The antistatic decontaminating car wax comprises the following raw materials in parts by weight: 15-20 parts of carnauba wax, 6-8 parts of polyethylene wax, 1-3 parts of polypropylene wax, 0.4-0.8 part of essence, 0.3-0.4 part of polyethylene oxide, 0.3-0.5 part of conductive mica powder, 0.5-1 part of anti-UV agent, 4-8 parts of marseille soap, 0.3-0.5 part of oleic acid, 6-8 parts of dimethyl silicone oil emulsifier, 25-30 parts of solvent oil, 1-2 parts of paraffin and 28-30 parts of water. Under common action of the components, the antistatic decontaminating car wax has strong dirt-removing power, antistatic function and anti-ultraviolet function, is simple in application method, and integrates a plurality of functions of decontaminating and waxing, so that time and labor are saved, the work efficiency is improved, and the labor intensity of a user is effectively reduced.
Owner:常熟紫金知识产权服务有限公司
Epichlorohydrin rubber composition
The invention relates to an epichlorohydrin rubber composition, which comprises homopolymerized epichlorohydrin rubber, copolymerized epichlorohydrin rubber, diethyl(o-)phthalate (DEP), fast extruding furnace black N550, silicon dioxide, stearamide, polyethylene wax, anti-agent agent MB, anti-aging agent RD, metal magnesium oxide, 1,2-hexylenethiourea (NA-22) and dibenzothiazole disulfide. Besides high performance in heat-resistance and oil-resistance aspects, the rubber has further improved metal corrosion resistance. The rubber is not corroded when kept at 130 DEG C for 720 hours, so the corrosion resistance of the rubber is improved greatly.
Owner:TIANJIN PENGYI GRP CO LTD
Therapeutic combinations comprising anti-FOLR1 immunoconjugates
ActiveUS10172875B2Improve efficacyOrganic active ingredientsHeavy metal active ingredientsClinical efficacyEfficacy
Owner:IMMUNOGEN INC
Stable anti-IgE humanized single anti-agent
ActiveCN101199845AEasy to usePowder deliveryAntibody ingredientsMedicineMonoclonal antibody preparation
The invention discloses a stable anti-Ige humanized monoclonal-antibody preparation, which is prepared by anti-Ige humanized monoclonal-antibody protective-agent, buffer, surfactant and isoosmotic adjustment. The preparation disclosed by the invention has the advantages of stability and convenient usage.
Owner:TAIZHOU MABTECH PHARM CO LTD
HIV replication inhibitor
InactiveUS20150361093A1Reduce usageUseful in treatmentOrganic chemistryAntiviralsPharmaceutical drugMedicinal chemistry
The present invention provides a novel compound having an antiviral activity, in particular, an HIV replication inhibiting activity, as well as a pharmaceutical composition, in particular, an anti-HIV agent.wherein ring A is substituted or unsubstituted carbocycle or substituted or unsubstituted heterocycle; R1 is substituted or unsubstituted alkyl etc.; R2 is substituted or unsubstituted alkyloxy etc.; n is 1 or 2; R3 is substituted or unsubstituted carbocyclyl or substituted or unsubstituted heterocyclyl; R4 is a hydrogen atom etc.; R6 is substituted or unsubstituted alkyl etc.
Owner:SHIONOGI & CO LTD
Dosing parameters for CD47 targeting therapies to hematologic malignancies
ActiveUS11141480B2Immunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsDosing regimenRegimen
Owner:THE BOARD OF TRUSTEES OF THE LELAND STANFORD JUNIOR UNIV +1
Pharmaceutical composition comprising telmisartan salt and calcium ion antagonist
InactiveCN102266559AImprove solubilityHigh dissolution rateOrganic active ingredientsSenses disorderMagnesium saltDiltiazem
The present invention relates to a kind of pharmaceutical composition, it comprises telmisartan salt and calcium ion antagonist or its pharmaceutically acceptable salt and pharmaceutically acceptable carrier; Described telmisartan salt is selected from telmisartan Sodium salt, potassium salt, calcium salt, magnesium salt or amine salt of sartan, described calcium ion antagonist is selected from amlodipine, lacidipine, cilnidipine, lercanidipine, nisoldipine, nica Dipine, azedipine, barnidipine, manidipine, benidipine, verapamil, diltiazem, or a pharmaceutically acceptable salt thereof. The composition is used for preventing, delaying progress or treating patients with hypertension, angina pectoris, atherosclerosis, stroke, cardiac insufficiency, dyslipidemia, diabetes, renal function damage or hypertension accompanied by Alzheimer's disease, reducing Reduce the morbidity and / or mortality of cardiovascular and cerebrovascular diseases, reduce adverse drug reactions, and improve patients' compliance with medication.
Owner:王丽燕
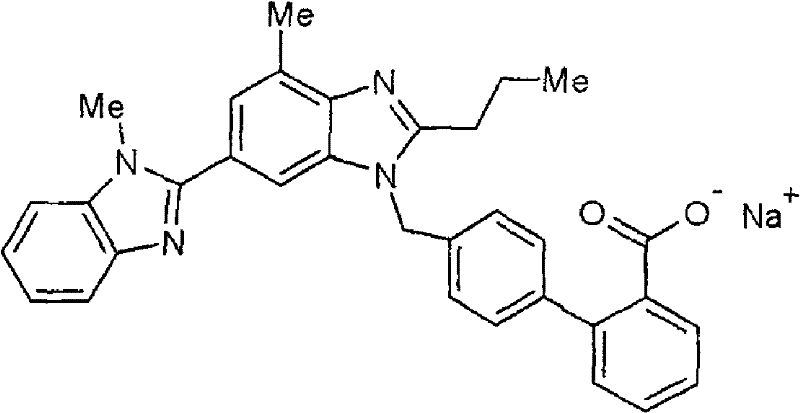
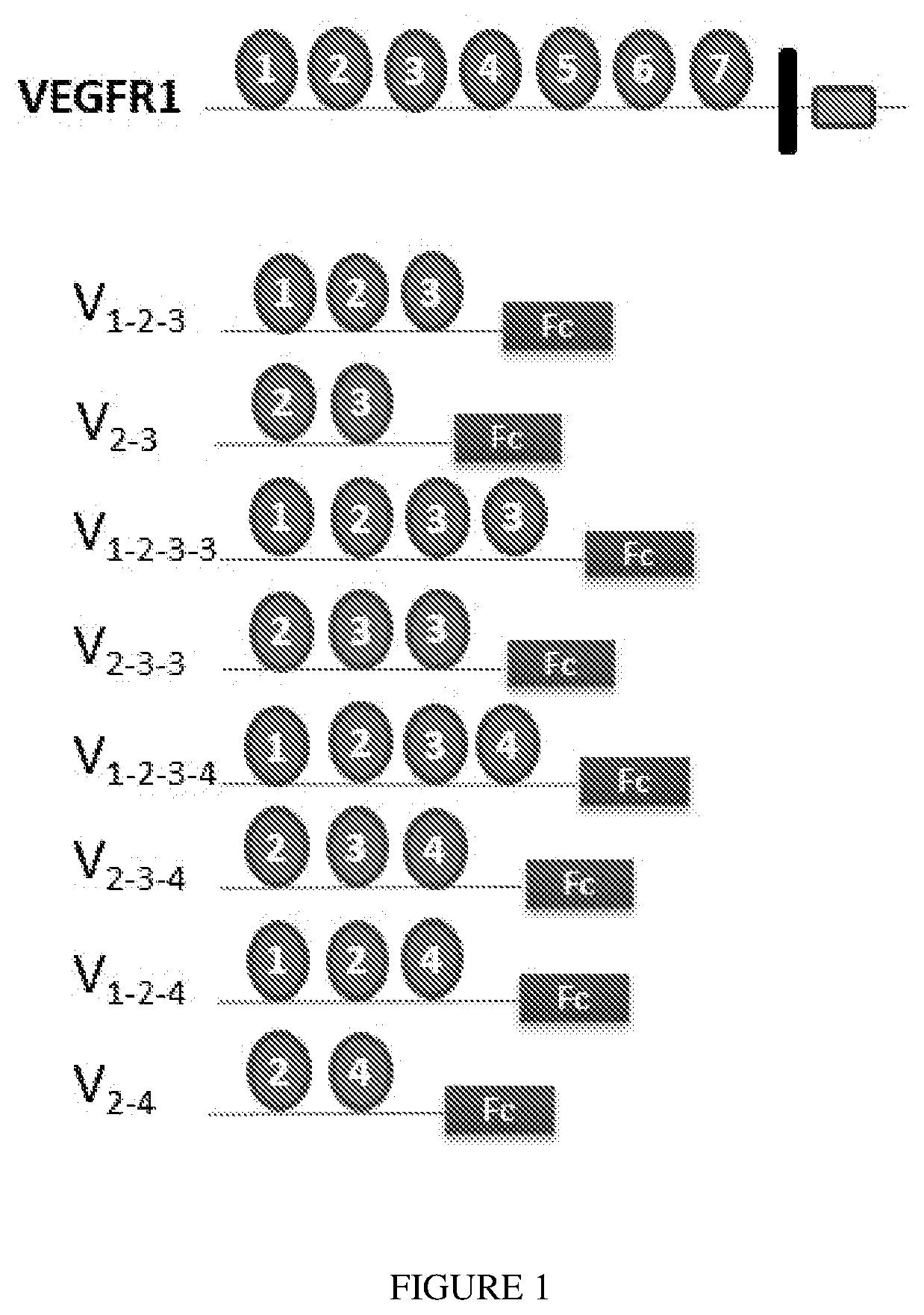
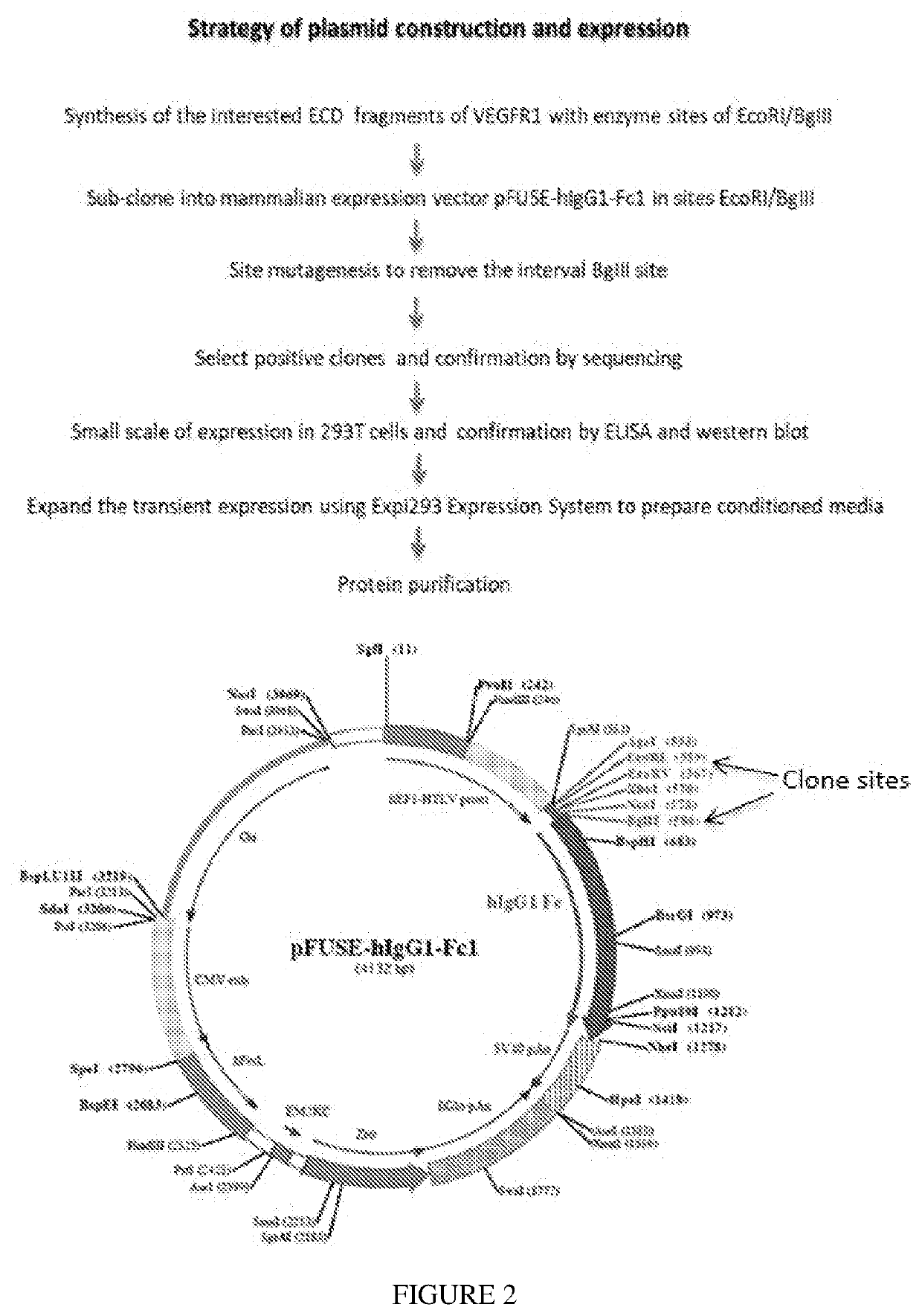
Methods and Compositions for Treatment of Angiogenic Disorders Using Anti-VEGF Agents
ActiveUS20200353041A1Inhibitory activityLong half-lifeSenses disorderPeptide/protein ingredientsDiseaseBinding domain
Provided are methods and compositions for treatment of angiogenic disorders using anti-VEGF agents. The anti-VEGF agents comprise VEGF binding domains and have the ability to bind vitreous. Provided are exemplary embodiments of Fc-IgG fusion proteins with VEGF binding domains with strong heparin-binding characteristics, strong inhibition of VEGF mitogenic activity, and improved pharmacokinetics, namely longer half-lives of the anti-VEGF agents and consequently less frequent dosing.
Owner:RGT UNIV OF CALIFORNIA
Formula of anti-wear PC material
The invention is a formula of wear-resistant PC material. The raw materials in the formula are composed by weight percentage: polycarbonate 5-15%, phenoxy tetrabromobisphenol A carbonate oligomer 1-5%, anti- Oxygen agent 1-10%, heat stabilizer 5-15%, anti-scratch agent 3-9%, lubricant 2-3%, toughening agent 1-5%, cycloalkyl polycarbonate 11-19%, Petroleum sulfonate 5‑7%, alkyl phenate 3‑5%, alkyl calcium salicylate 3‑5%, thiophosphate 1‑5%, kaolin 5‑10%, traditional Chinese medicine powder 1‑1.5% , titanium dioxide 4-5%, and zinc stearate 2-4%. At this time, the wear-resistant PC material has a good effect of modifying the face shape, sun protection and skin care.
Owner:解伟伟
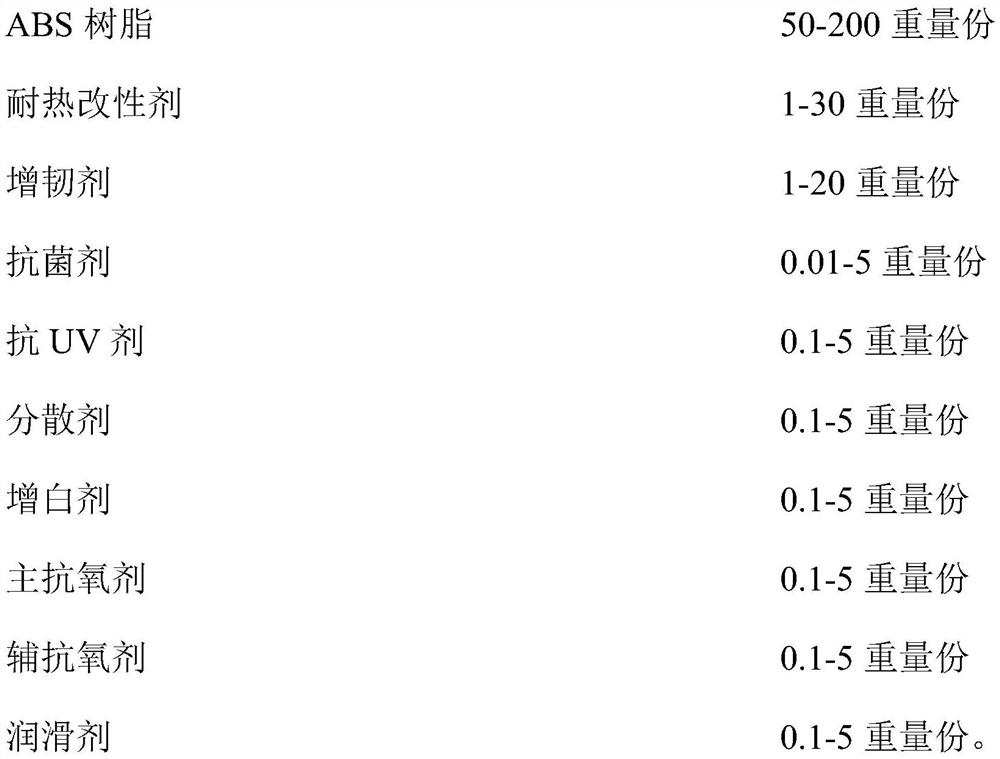
ABS material and preparation method and application thereof
The invention relates to an ABS material and a preparation method and application thereof. The ABS material comprises the following components in parts by weight: 50-200 parts of ABS resin, 1-30 partsof a heat-resistant modifier, 0.01-5 parts of an antibacterial agent, 0.1-5 parts of an anti-UV agent, 0.1-5 parts of a brightener and 1.4-40 parts of an assistant. The ABS material with excellent antibacterial property, heat resistance, UV resistance, mechanical properties and glossiness can be obtained by adopting an ABS resin, heat-resistant modifier, antibacterial agent, anti-UV agent and brightener blending system and combining the ABS resin, the heat-resistant modifier, the antibacterial agent, the anti-UV agent and the brightener in a specific ratio, and can be used in high-temperatureand high-humidity environments and occasions with antibacterial and anti-UV requirements.
Owner:GUANGZHOU SHIYUAN ELECTRONICS CO LTD
Method for detecting primary amine in anti-aging agents TMQ
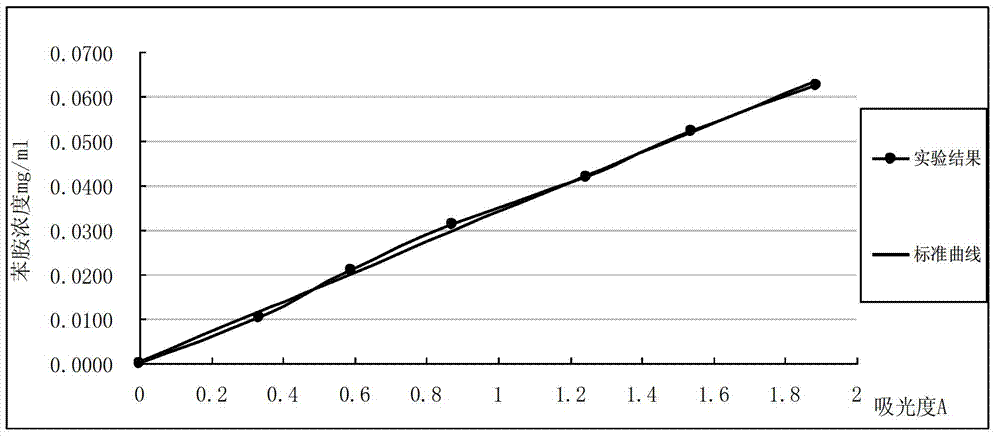
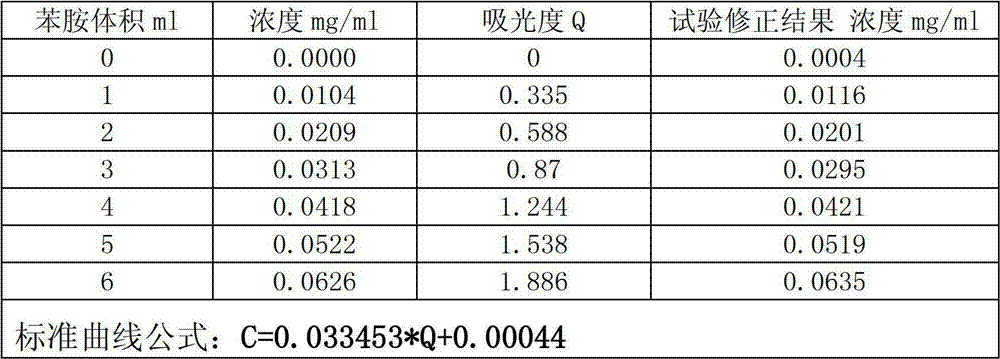
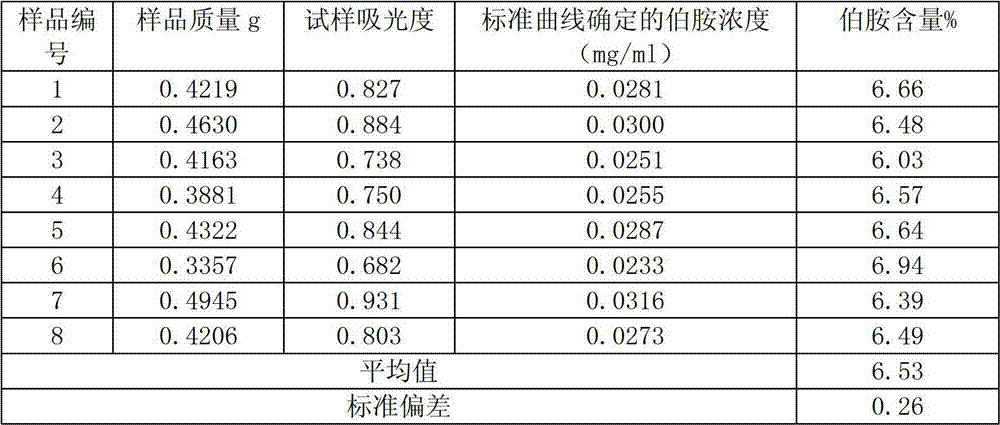
InactiveCN102879344AThe instrument is simpleSimple and fast operationColor/spectral properties measurementsCuvetteAniline
The invention relates to a method for detecting primary amine in anti-aging agents TMQ. The method comprises the following steps of: preparing aniline standard solutions with different concentrations and a standard reference solution, respectively measuring the absorbance of the different aniline standard solutions under the condition of the wavelength of 437 nanometers by using an ultraviolet and visible spectrophotometer and the 1cm optical path of a cuvette, and drawing a standard curve of the aniline standard solutions by using the standard reference solution as a comparison solution; measuring the absorbance of a sample solution by the same method by using a sample reference solution as a comparison solution; performing quantitative analysis by the aniline standard curve to obtain the content of the primary amine in the anti-agents TMQ in a sample. By the method, the content of the primary amine in the anti-aging agents TMQ can be accurately detected, instruments are simple, and the method is easy and convenient to operate and high in accuracy.
Owner:KEMAI CHEM
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com
![SUBSTITUTED SPIROPYRIDO[1,2-a]PYRAZINE DERIVATIVE AND PHARMACEUTICAL USE OF SAME AS HIV INTEGRASE INHIBITOR SUBSTITUTED SPIROPYRIDO[1,2-a]PYRAZINE DERIVATIVE AND PHARMACEUTICAL USE OF SAME AS HIV INTEGRASE INHIBITOR](https://images-eureka-patsnap-com.libproxy1.nus.edu.sg/patent_img/eaca0958-e088-450b-a622-0be890c729fb/US20140221380A1-20140807-C00001.png)
![SUBSTITUTED SPIROPYRIDO[1,2-a]PYRAZINE DERIVATIVE AND PHARMACEUTICAL USE OF SAME AS HIV INTEGRASE INHIBITOR SUBSTITUTED SPIROPYRIDO[1,2-a]PYRAZINE DERIVATIVE AND PHARMACEUTICAL USE OF SAME AS HIV INTEGRASE INHIBITOR](https://images-eureka-patsnap-com.libproxy1.nus.edu.sg/patent_img/eaca0958-e088-450b-a622-0be890c729fb/US20140221380A1-20140807-C00002.png)
![SUBSTITUTED SPIROPYRIDO[1,2-a]PYRAZINE DERIVATIVE AND PHARMACEUTICAL USE OF SAME AS HIV INTEGRASE INHIBITOR SUBSTITUTED SPIROPYRIDO[1,2-a]PYRAZINE DERIVATIVE AND PHARMACEUTICAL USE OF SAME AS HIV INTEGRASE INHIBITOR](https://images-eureka-patsnap-com.libproxy1.nus.edu.sg/patent_img/eaca0958-e088-450b-a622-0be890c729fb/US20140221380A1-20140807-C00003.png)