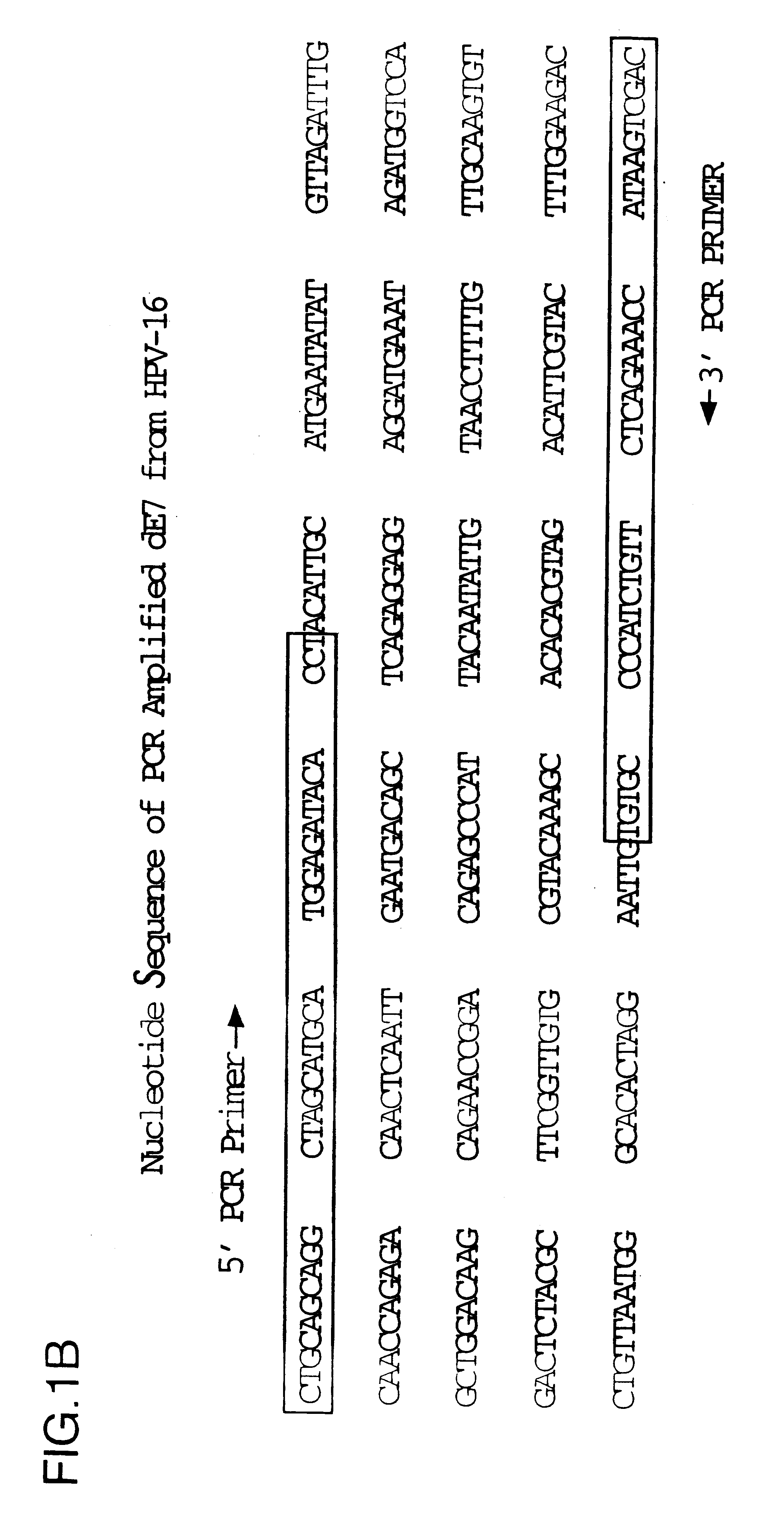
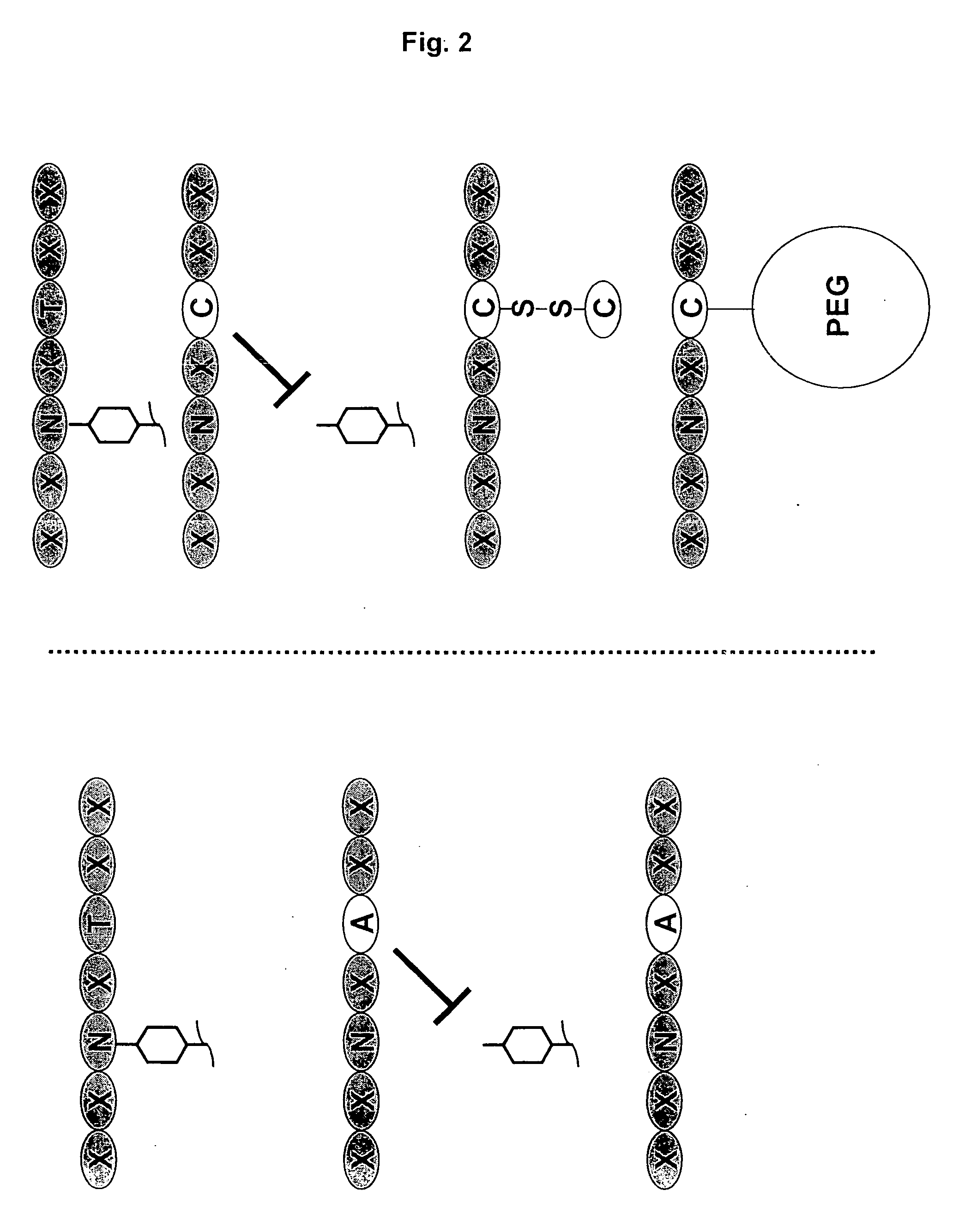Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
3051results about How to "Improving immunogenicity" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Broad-spectrum delta-endotoxins
InactiveUS6713063B1Improved insecticidal activity and broader host-range activityImproving immunogenicityBiocidePeptide/protein ingredientsAureobasidium sp.Toxin
Disclosed are novel synthetically-modified B. thuringiensis chimeric crystal proteins having improved insecticidal activity and broader insect host range against coleopteran, dipteran and lepidopteran insects. Also disclosed are the nucleic acid segments encoding these novel peptides. Methods of making and using these genes and proteins are disclosed as well as methods for the recombinant expression, and transformation of suitable host cells. Transformed host cells and transgenic plants expressing the modified endotoxin are also aspects of the invention.
Owner:MONSANTO TECH LLC
Broad-spectrum insect resistant transgenic plants
InactiveUS6281016B1Improve insecticidal effectBroad-range specificityBiocideNanotechAureobasidium sp.Toxin
Disclosed are novel synthetically-modified B. thuringiensis chimeric crystal proteins having improved insecticidal activity against coleopteran, dipteran and lepidopteran insects. Also disclosed are the nucleic acid segments encoding these novel peptides. Methods of making and using these genes and proteins are disclosed as well as methods for the recombinant expression, and transformation of suitable host cells. Transformed host cells and tansgenic plants expressing the modified endotoxin are also aspects of the invention.
Owner:MONSANTO CO (MONSANTO CY)
Broad-spectrum delta -endotoxins
InactiveUS6110464AImprove insecticidal effectBroad-range specificityNanotechBacteriaAureobasidium sp.Toxin
Disclosed are novel synthetically-modified B. thuringiensis chimeric crystal proteins having improved insecticidal activity against coleopteran, dipteran and lepidopteran insects. Also disclosed are the nucleic acid segments encoding these novel peptides. Methods of making and using these genes and proteins are disclosed as well as methods for the recombinant expression, and transformation of suitable host cells. Transformed host cells and transgenic plants expressing the modified endotoxin are also aspects of the invention.
Owner:MONSANTO TECH LLC
Modified Antibody Constant Region
ActiveUS20100298542A1Improving immunogenicityImprove propertiesAntipyreticAnalgesicsHigh concentrationHinge region
The present inventors succeeded in improving the antibody constant region to have increased stability under acid conditions, reduced heterogeneity originated from disulfide bonds in the hinge region, reduced heterogeneity originated from the H chain C terminus, and increased stability at high concentrations as well as in discovering novel constant region sequences having reduced Fcγ receptor-binding, while minimizing the generation of novel T-cell epitope peptides. As a result, the present inventors successfully discovered antibody constant regions with improved physicochemical properties (stability and homogeneity), immunogenicity, safety, and pharmacokinetics.
Owner:CHUGAI PHARMA CO LTD
Inducing cellular immune responses to human papillomavirus using peptide and nucleic acid compositions
InactiveUS20070014810A1Reduce the possibilityImproving immunogenicitySugar derivativesViral antigen ingredientsEpitopeT cell
This invention uses our knowledge of the mechanisms by which antigen is recognized by T cells to identify and prepare human papillomavirus (HPV) epitopes, and to develop epitope-based vaccines directed towards HPV. More specifically, this application communicates our discovery of pharmaceutical compositions and methods of use in the prevention and treatment of HPV infection.
Owner:GENIMMUNE NV +1
Insect cells or fractions as adjuvant for antigens
Disclosed and claimed is an adjuvant for immunogenic, immunological, antigenic or vaccine compositions. The adjuvant is composed of insect cells or fractions thereof. Disclosed and claimed are also methods for preparing and using the adjuvant and compositions containing the adjuvant. Advantageously, a recombinant baculovirus containing DNA encoding and expressing an epitope of interest or antigen can be infected into insect cells such as insect cells derived from a Lepidopteran species such as S. frugiperda for expression, and the infected insect cells or a fraction thereof can be used with the expressed epitope of interest or antigen as an inventive antigen or in an inventive immunological, antigen or vaccine composition.
Owner:MERIAL LTD
Inducing immune responses to influenza virus using polypeptide and nucleic acid compositions
ActiveUS20080032921A1Cost reductionImproving immunogenicityVirusesPeptide/protein ingredientsNucleotideImmunology
Owner:EPIMMUNE
Engineered listeria and methods of use thereof
InactiveUS20070207171A1Enhance survivalImprove survivalBacterial antigen ingredientsCell receptors/surface-antigens/surface-determinantsNucleotideAntibiotics
The invention provides a bacterium containing a polynucleotide comprising a nucleic acid encoding a heterologous antigen, as well as fusion protein partners. Also provided are vectors for mediating site-specific recombination and vectors comprising removable antibiotic resistance genes.
Owner:ANZA THERAPEUTICS INC
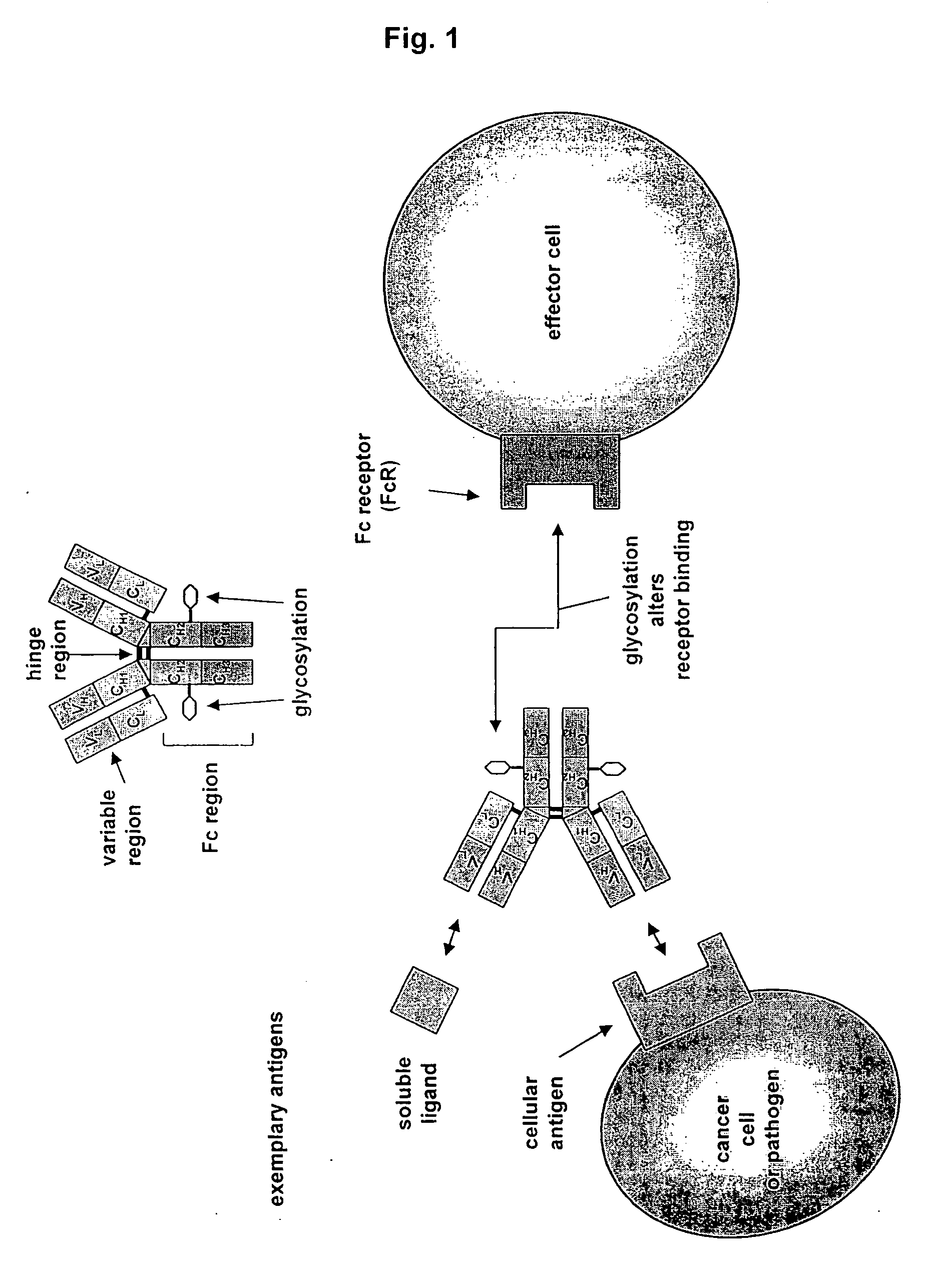
Antibodies having altered effector function and methods for making the same
ActiveUS20070048300A1Reduce bindingReduce effector functionAntipyreticAntiviralsAntibodyGlycosylation
The invention provides a method of producing aglycosylated Fc-containing polypeptides, such as antibodies, having desired effector function. The invention also provides aglycosylated antibodies produced according to the method as well as methods of using such antibodies as therapeutics.
Owner:BIOGEN IDEC MA INC
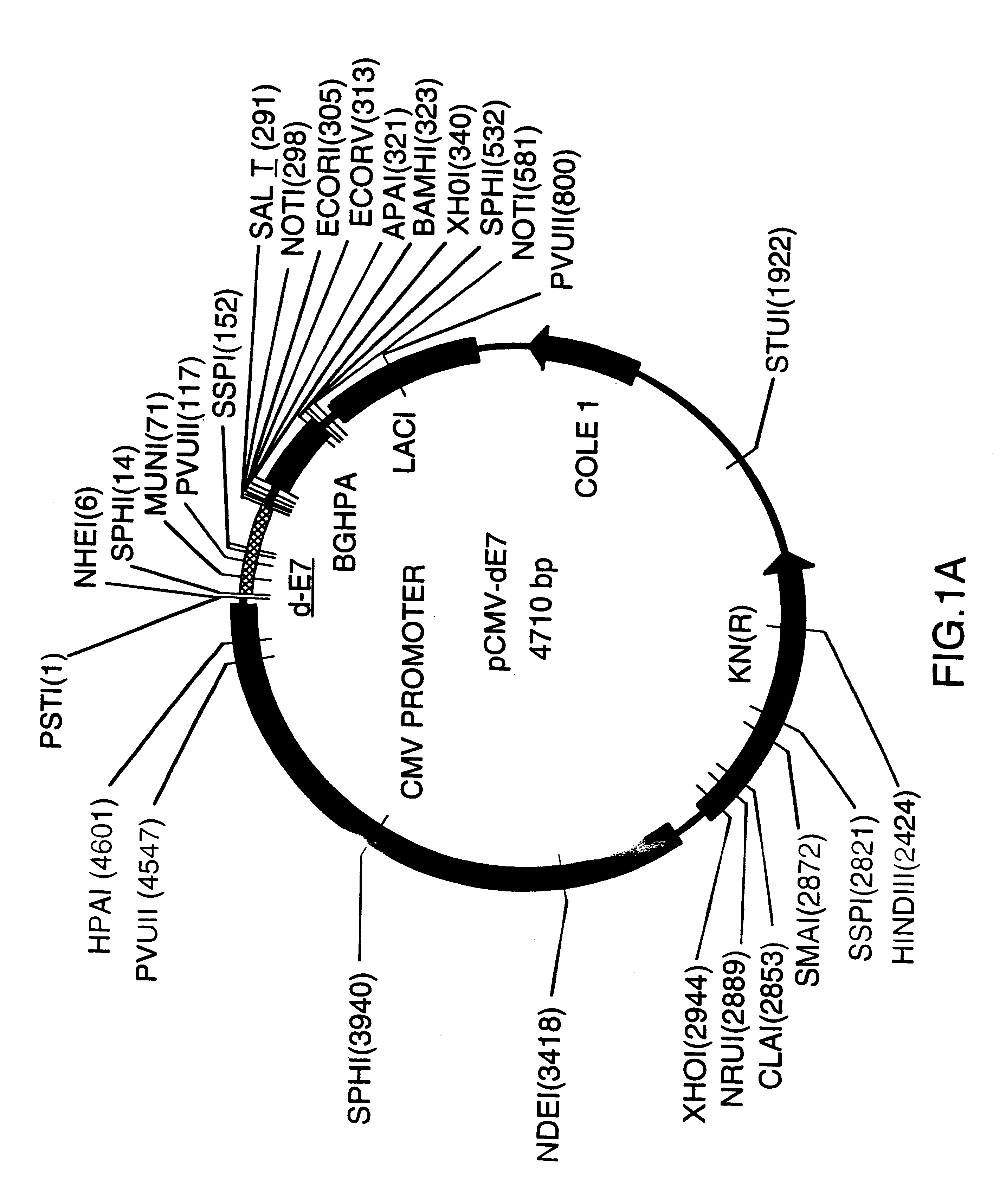
Vectors for DNA immunization against cervical cancer
InactiveUS6235523B1Improving immunogenicitySlow and sustained releaseOrganic active ingredientsVirusesAntigenDna immunization
Vectors for DNA immunization against cervical cancer comprise a nucleic acid molecule encoding at least one non-toxic T-cell epitope of the E6 and / or E7 antigens of a strain of human papilloma virus (HPV) associated with cervical cancer, such as HPV-16, and a promoter operatively coupled to the nucleic acid molecule for expression of the nucleic acid molecule in a host to which the vector is administered.
Owner:CONNAUGHT LAB
Mixture of poly-pneumococcal capsular polysaccharide-protein conjugates and preparation method of mixture
ActiveCN103495161AImproving immunogenicityIncrease productionAntibacterial agentsBacterial antigen ingredientsAdjuvantImmunogenicity
The invention discloses a mixture of poly-pneumococcal capsular polysaccharide-protein conjugates. The mixture contains 13 pneumococcal capsular polysaccharide-protein conjugates and an immunity-enhancement adjuvant, wherein each pneumococcal capsular polysaccharide-protein conjugate is formed by combining corresponding serum-type pneumococcal capsular polysaccharide with a same protein carrier through a covalent bond; the 13 pneumococcal capsular polysaccharides are obtained by purifying and extracting 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F and 23F bacteria; the protein carrier is A chain of a diphtherin mutant CRM197 obtained from expression of genetic recombinant escherichia coli. Meanwhile, the invention discloses a preparation method of the mixture. The mixture, compared to conventional bacterial fermentation total-chain diphtherin mutant CRM197, is easy to purify, high in yield and low in cost; experiments prove that the mixture disclosed by the invention is immunogenic, and is applicable to clinical inoculation.
Owner:KANVAX BIOPHARM
Recombinant newcastle disease virus RNA expression systems and vaccines
InactiveUS6451323B1Enhance immunogenicityAttenuate phenotypeSsRNA viruses negative-senseFungiViral vectorRna expression
This invention relates to genetically engineered Newcastle disease viruses and viral vectors which express heterologous genes or mutated Newcastle disease viral genes or a combination of viral genes derived from different strains of Newcastle disease virus. The invention relates to the construction and use of recombinant negative strand NDV viral RNA templates which may be used with viral RNA-directed RNA polymerase to express heterologous gene products in appropriate host cells and / or to rescue the heterologous gene in virus particles. In a specific embodiment of the invention, the heterologous gene product is a peptide or protein derived from the genome of a human immunodeficiency virus. The RNA templates of the present invention may be prepared by transcription of appropriate DNA sequences using any DNA-directed RNA polymerase such as bacteriophage T7, T3, SP6 polymerase, or eukaryotic polymerase I.
Owner:MT SINAI SCHOOL OF MEDICINE
Fusion proteins of Mycobacterium tuberculosis antigens and their uses
InactiveUS6627198B2Improving immunogenicityPeptide/protein ingredientsAntibody mimetics/scaffoldsAntigenMycobacterial antigen
The present invention relates to fusion proteins containing at least two Mycobacterium tuberculosis antigens. In particular, it relates to bi-fusion proteins which contain two individual M. tuberculosis antigens, tri-fusion proteins which contain three M. tuberculosis antigens, tetra-fusion proteins which contain four M. tuberculosis antigens, and penta-fusion proteins which contain five M. tuberculosis antigens, and methods for their use in the diagnosis, treatment and prevention of tuberculosis infection.
Owner:CORIXA CORP
Engineered Listeria and methods of use thereof
InactiveUS20070207170A1Improve survivalIncreasing expression and secretionBiocideBacteriaHeterologous AntigensSite-specific recombination
The invention provides a bacterium containing a polynucleotide comprising a nucleic acid encoding a heterologous antigen, as well as fusion protein partners. Also provided are vectors for mediating site-specific recombination and vectors comprising removable antibiotic resistance genes.
Owner:ANZA THERAPEUTICS INC +1
Treatment of alzheimer's disease
InactiveUS7179463B2Improve AD treatment efficacyImproving immunogenicityFungiNervous disorderDisease causeAntibody
Owner:BIOARCTIC NEUROSCI AB
Nucleic acid library design and assembly
InactiveUS20080064610A1Reduce solubilityImproving immunogenicityVector-based foreign material introductionLibrary creationSolubilityENCODE
Owner:CODON DEVICES
Fusion proteins of mycobacterium tuberculosis antigens and their uses
InactiveUS6544522B1Improving immunogenicityAntibacterial agentsPeptide/protein ingredientsAntigenMycobacterial antigen
The present invention relates to fusion proteins of Mycobacterium tuberclosis antigens. In particular, it relates to two fusion proteins, each of which contains three individual M. tuberculosis antigens, and a fusion protein of two M. tuberculosis antigens, their coding sequences, and methods for their use in the treatment and prevention of tuberculosis.
Owner:CORIXA CORP
Compositions, methods and kits for enhancing the immunogenicity of a bacterial vaccine vector
ActiveUS20060233835A1Improving immunogenicityOrganic active ingredientsBacterial antigen ingredientsImmunogenicityTGE VACCINE
The present invention comprises methods for enhancing the immunogenicity of a bacterial vaccine vector and an antigen by passaging the bacterial vaccine vector through an animal.
Owner:THE TRUSTEES OF THE UNIV OF PENNSYLVANIA
Hybrids of M. tuberculosis antigens
InactiveUS20020176867A1Improving immunogenicityEasy to transportBacteriaPeptide/protein ingredientsMycobacterial antigenMycobacterium
The present invention discloses fusion proteins of the immunodominant antigens ESAT-6 and Ag85B from Mycobacterium tuberculosis or homologues thereof, and a tuberculosis vaccine based on the fusion proteins, which vaccine induces efficient immunological memory.
Owner:STATENS SERUM INST
RNA respiratory syncytial virus vaccines
InactiveUS6060308AFaster replicationImprove efficiencySsRNA viruses negative-senseBiocideF proteinViral Vaccine
A vector comprising a first DNA sequence which is complementary to at least part of an alphavirus RNA genome and having the complement of complete alphavirus DNA genome replication regions, a second DNA sequence encoding a paramyxovirus protein, particularly a respiratory syncytial virus fusion (RSV F) protein or a RSV F protein fragment that generates antibodies that specifically react with RSV F protein, the first and second DNA sequences being under the transcriptional control of a promoter is described. Such vector may be used to produce an RNA transcript which may be used to immunize a host, including a human host, to protect the host against disease caused by paramyxovirus, particularly respiratory syncytial virus, by administration to the host.
Owner:CONNAUGHT LAB
mRNA and vaccine for coding a SARS-CoV-2 viral antigen and preparation method of vaccine
ActiveCN111218458AImproving immunogenicityRapid R&DSsRNA viruses positive-senseViral antigen ingredientsTGE VACCINECoding region
The invention provides mRNA and a vaccine for coding a SARS-CoV-2 viral antigen and a preparation method of the vaccine, and relates to the technical field of vaccines. The mRNA for coding the SARS-CoV-2 viral antigen at least contains at least one of S protein and N protein for coding SARS-CoV-2 virus and / or a coding region of a fragment of the at least one protein, and the mRNA is delivered intoa body to enable the body to generate an immune reaction.
Owner:LIVERNA THERAPEUTICS INC
Adeno-associated virus vector for boosting immunogenicity of cells
InactiveUS7001765B2Improving immunogenicityBiocideGenetic material ingredientsAdeno associate virusVaccine Immunogenicity
The present invention provides an Adeno-Associated Virus (AAV) vector having a foreign DNA coding for a protein that boosts immunogenicity of cells. The invention also provides a vaccine containing such a vector and the use of both.
Owner:MEDIGENE
Recombinant SARS-CoV-2 vaccine using human replication-defective adenovirus as vector
ActiveCN111218459AReduce loadSimple manufacturing methodSsRNA viruses positive-senseViral antigen ingredientsProtective antigenCoronavirus vaccination
The invention provides a SARS-CoV-2 vaccine using human type-5 replication-defective adenovirus as a vector. The vaccine uses E1 and E3 to be combined with replication-defective human type-5 adenovirus as the vector and HEK293 cells integrating adenovirus E1 gene as a packaging cell line, and a protective antigen gene carried is the 2019 SARS-CoV-2 S protein gene (Ad5-nCoV) which is subjected to optimization design. After the S protein gene is optimized, the expression level in transfected cells is increased significantly. The vaccine has good immunogenicity in mouse and guinea pig models, andcan induce a body to produce a strong cellular and humoral immune response in a short time. Studies on the protective effect of hACE2 transgenic mice show that after 14 days of single immunization ofAd5-nCoV, the viral load in lung tissue can be significantly reduced, and it is indicated that the vaccine has a good immunoprotective effect on the 2019 SARS-CoV-2. In addition, the vaccine is quick, simple and convenient to prepare, and can be mass-produced in a short period of time to respond to sudden outbreaks.
Owner:ACADEMY OF MILITARY MEDICAL SCI +1
Use of bioadhesives and adjuvants for the mucosal delivery of antigens
InactiveUS20050281843A1Efficient methodImproving immunogenicitySsRNA viruses negative-senseBacterial antigen ingredientsAntigenAdjuvant
Owner:NOVARTIS VACCINES & DIAGNOSTICS INC
Intradermal delivery of biological agents
InactiveUS20090012494A1Significant comprehensive benefitsMinimal expertiseBiocidePeptide/protein ingredientsActive agentWhole body
The present invention relates to methods for intradermally delivering one or more biologically active agents such as vaccines and therapeutic agents into the dermis layer of the skin of a subject to obtain systemic delivery or an immune response using a microneedle drug delivery device containing the agent to be delivered. The methods employ a microneedle device with a row of hollow microneedles. The microneedles penetrate the skin of the subject and assume an anchored state in which the microneedles are anchored in the skin and project laterally from the device. A pivotal motion is then performed with the device so that the skin in which the microneedles are engaged is lifted above the initial plane of the surface of the skin while the biologically active agent is delivered. The methods of the invention elicit increased humoral and / or cellular response as compared to conventional vaccine delivery routes, facilitating dose sparing.
Owner:NANOPASS TECH LTD
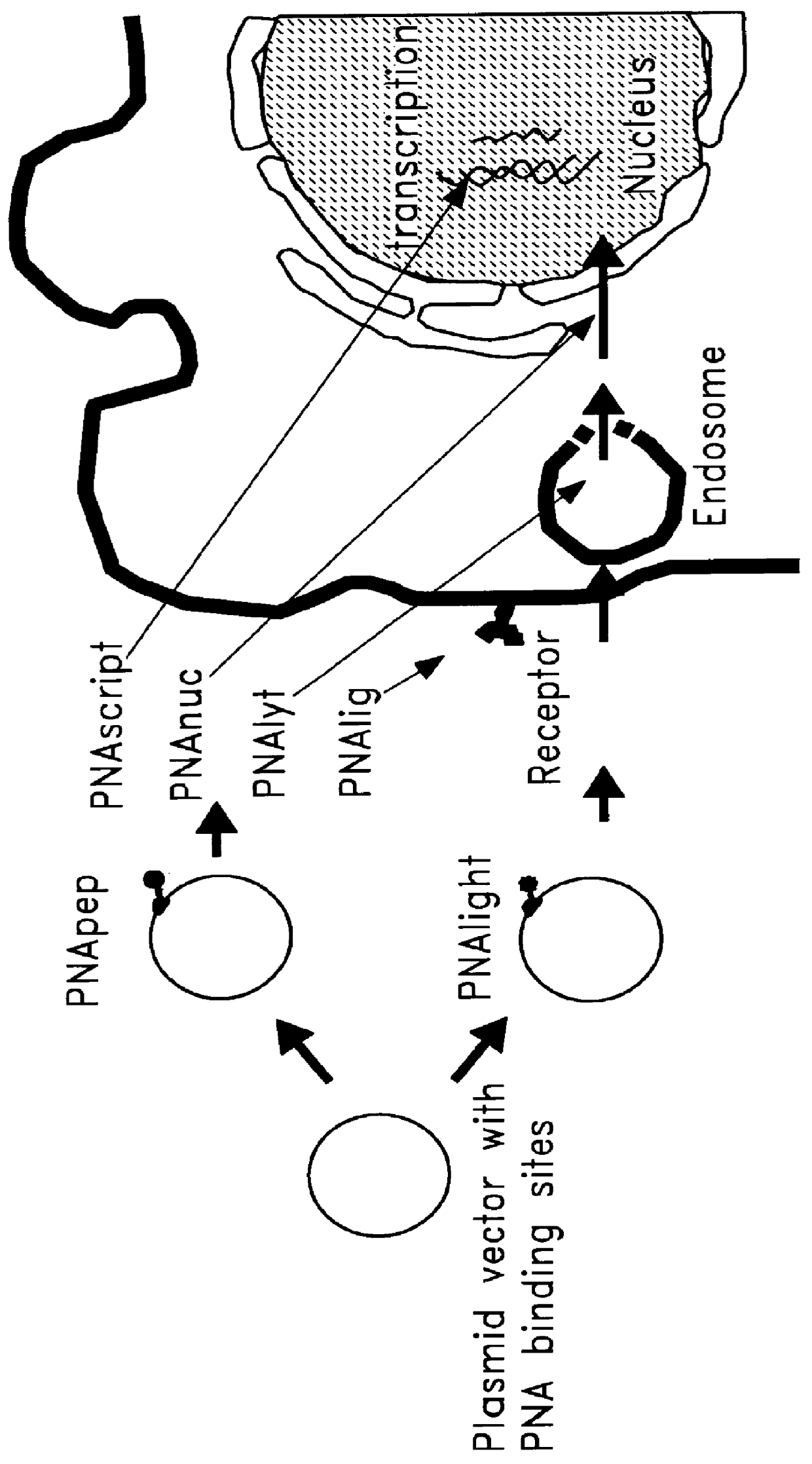
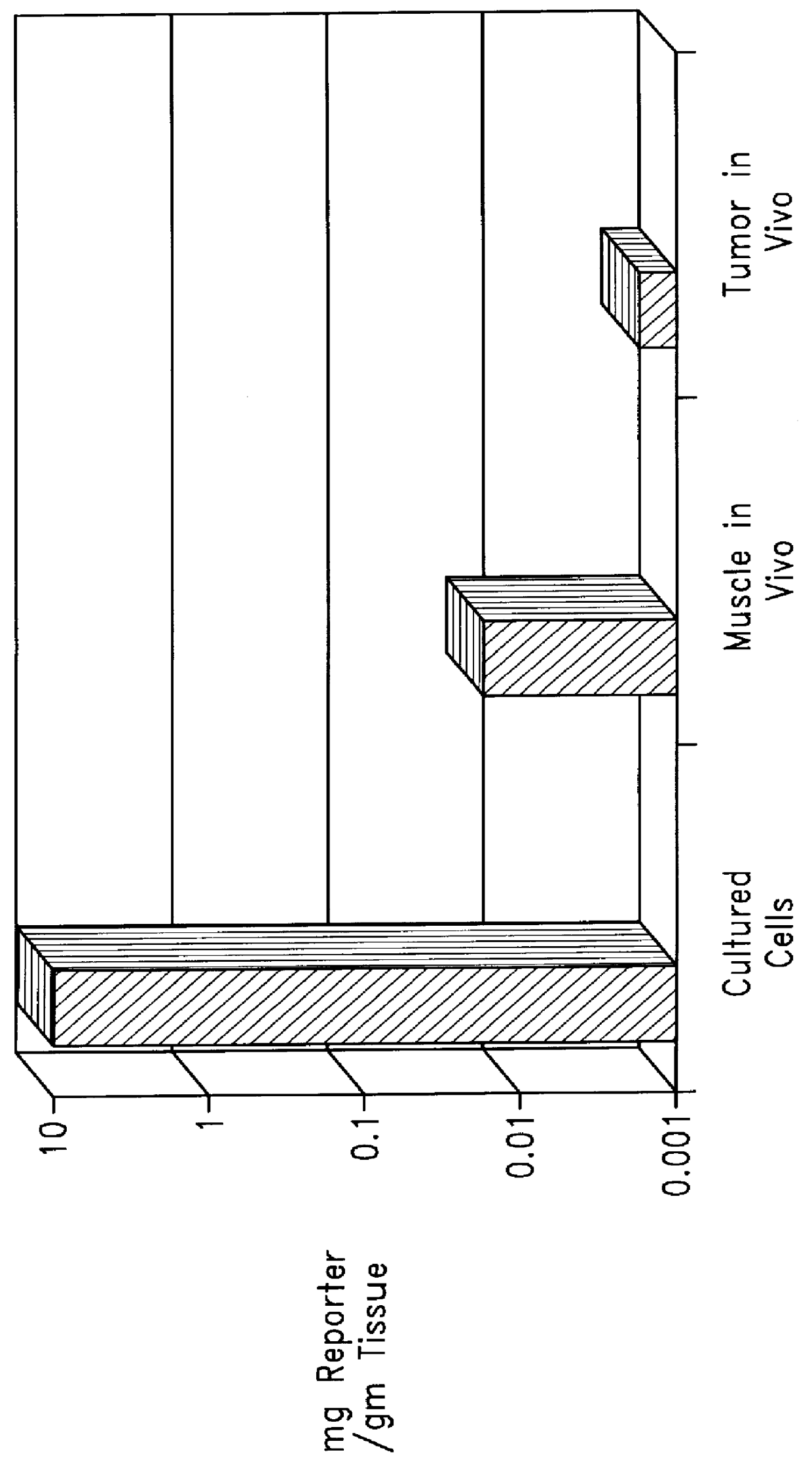
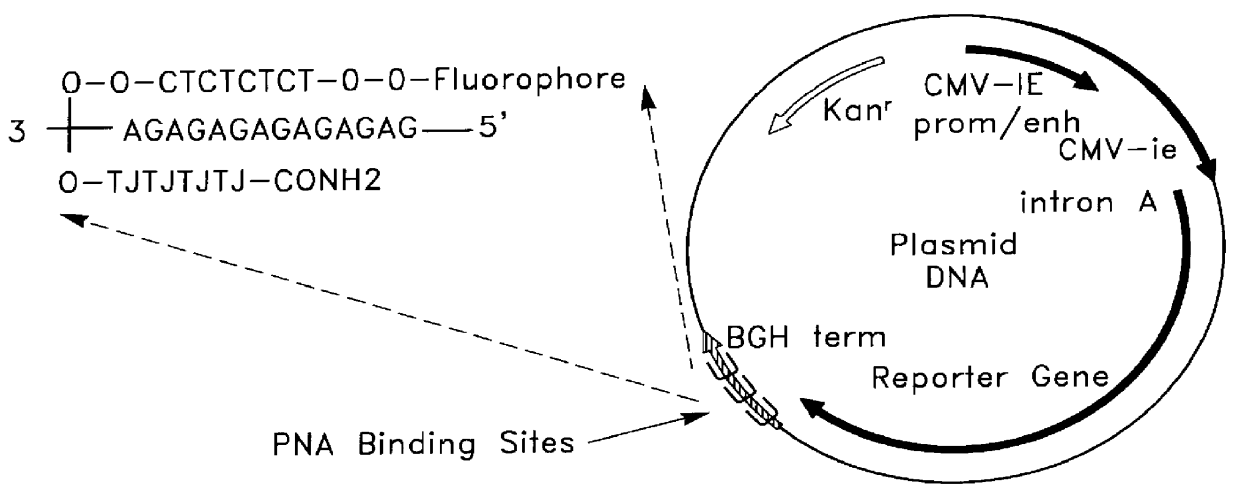
Chemical modification of DNA using peptide nucleic acid conjugates
InactiveUS6165720AReduce deliveryHigh transfection efficiencyFungiBacteriaEukaryotic plasmidsBiological activation
Complexes comprising a nucleic acid molecule and a conjugated peptide nucleic acid (PNA). The PNA may be labeled or conjugated to a protein, peptide, carbohydrate moiety or receptor ligand. These complexes are used to transfect cells to monitoring plasmid biodistribution, promote nuclear localization, induce transcriptional activation, lyse the endosomal compartment and facilitate transfection. These complexes increase the efficiency of expression of a particular gene.
Owner:GENE THERAPY SYST +1
Adjuvants Of Immune Response
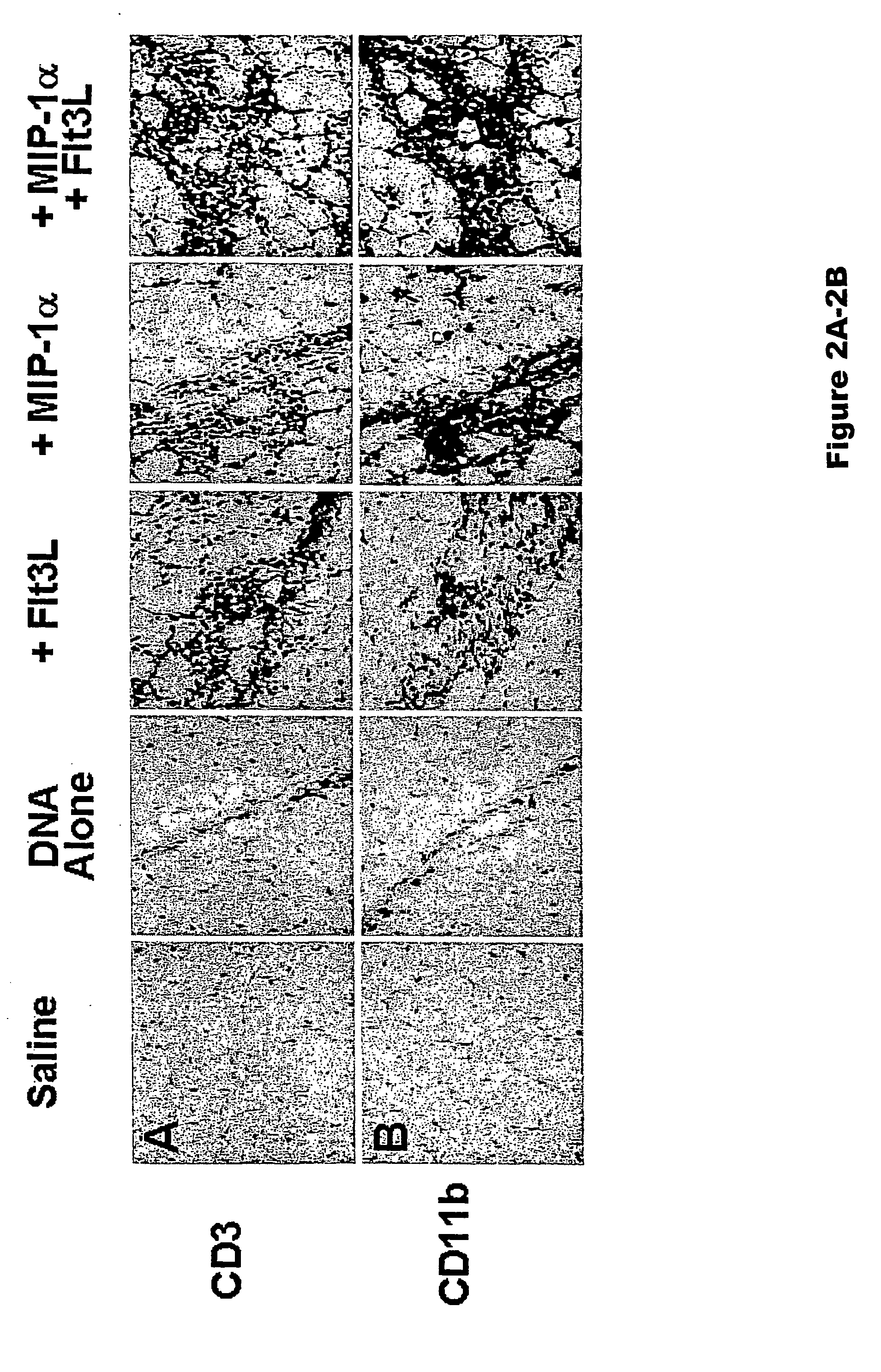
InactiveUS20070298051A1Improving immunogenicityImprove efficacyBiocideOrganic active ingredientsAdjuvantImmunogen
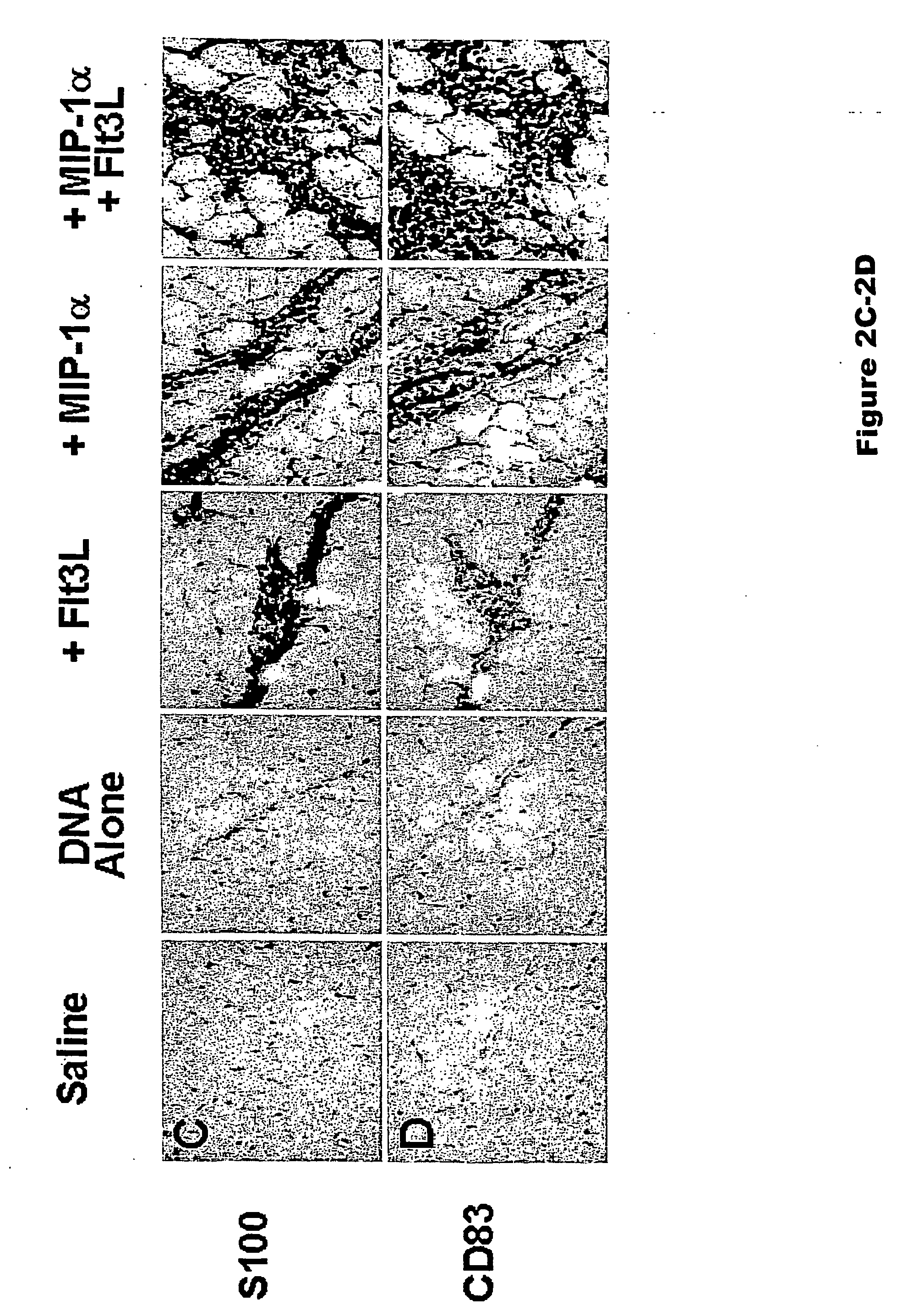
The present invention features methods to substantially increase the immunogenicity of a vaccine, preferably a DNA vaccine, and involves providing a mammal with a vaccine regimen, which includes an immunogen and Flt3L in combination with MIP-1α or MIP-3α. The methods of the present invention can be used for the prevention and treatment of various pathological states, including for example, cancer, microbial infections, autoimmune diseases, tissue rejection, and allergic reactions.
Owner:BETH ISRAEL DEACONESS MEDICAL CENT INC
THERAPEUTIC ANTIGEN-BINDING MOLECULE WITH A FcRn-BINDING DOMAIN THAT PROMOTES ANTIGEN CLEARANCE
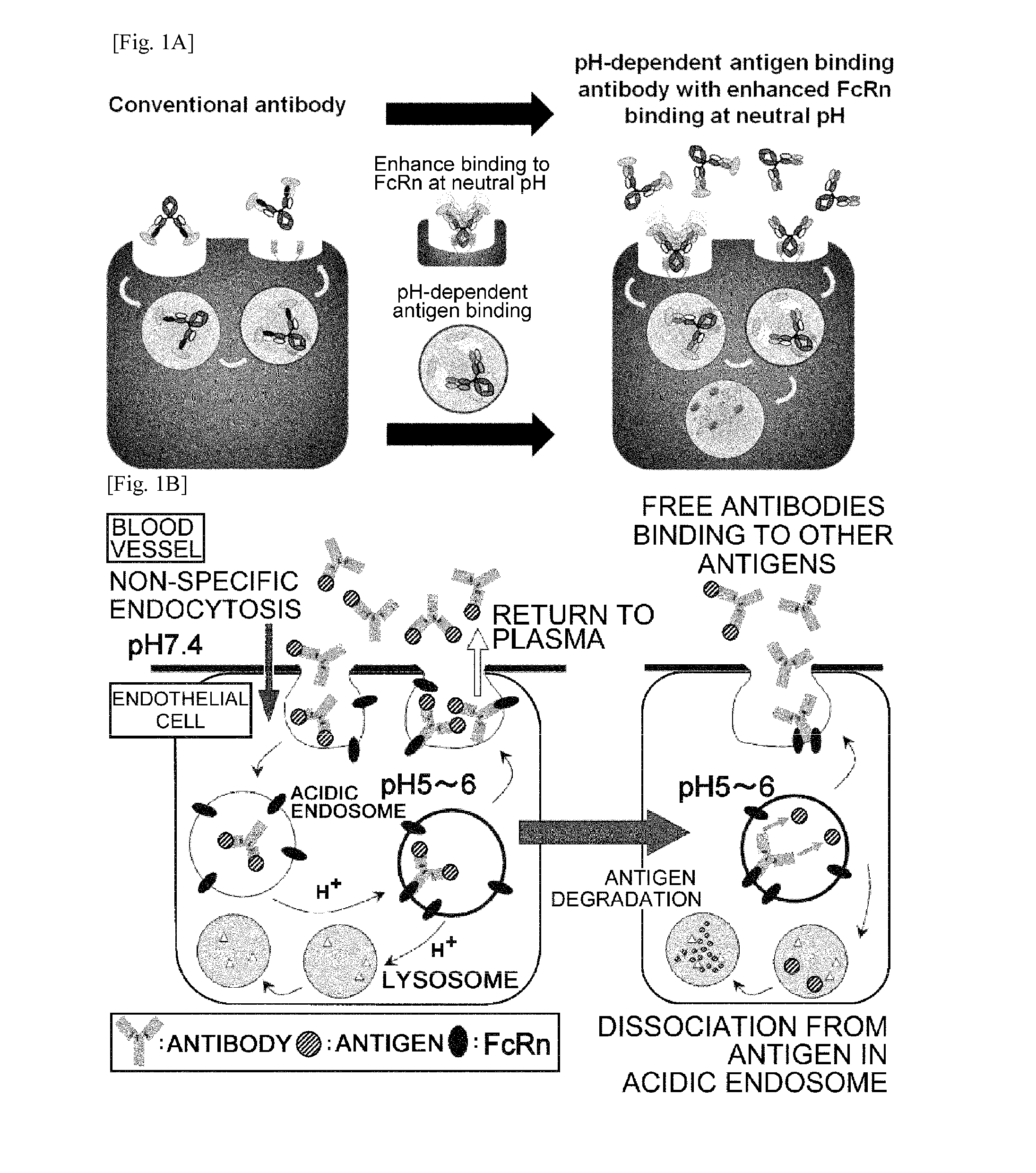
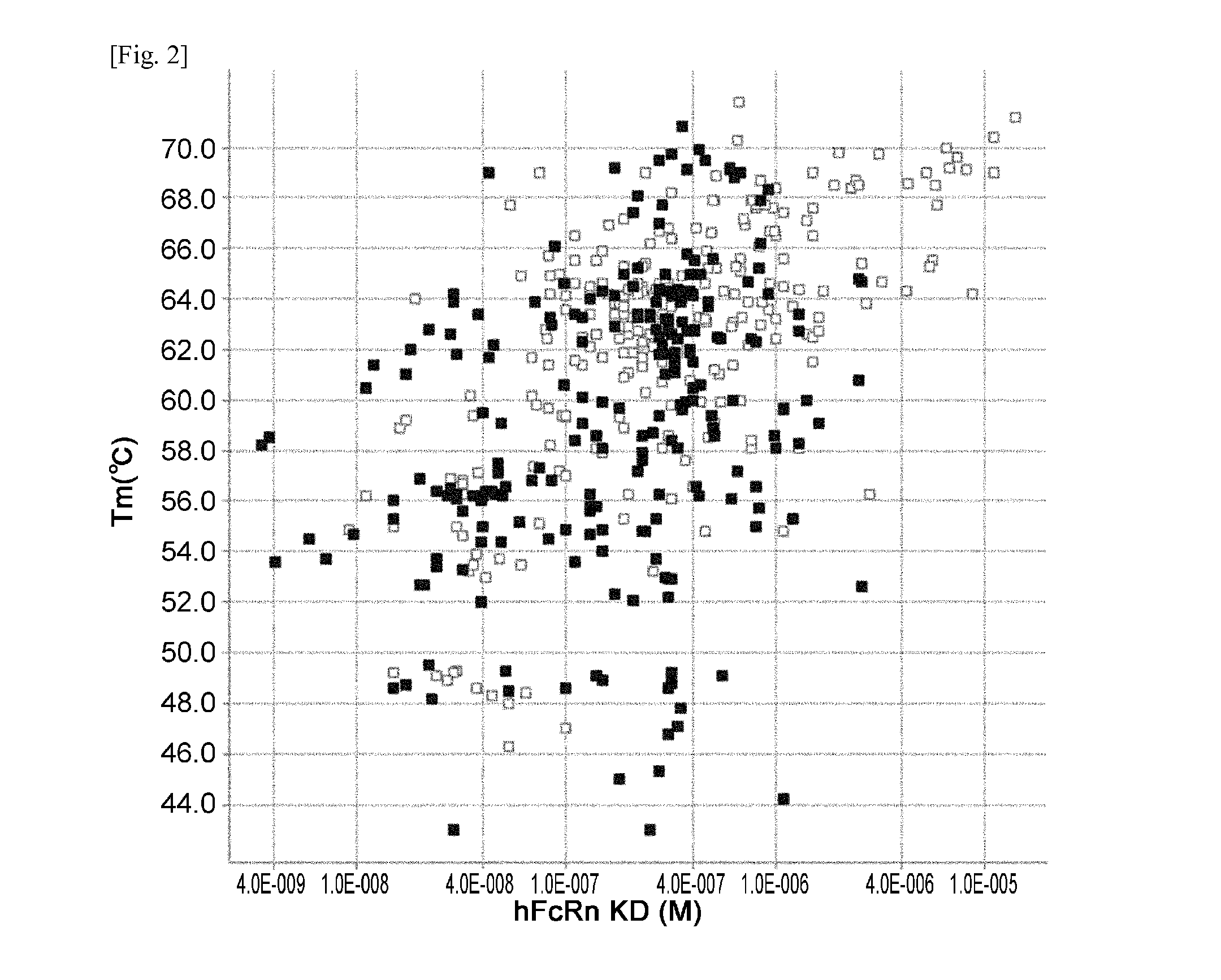
ActiveUS20140363428A1Low immunogenicityImprove stabilityAntipyreticAnalgesicsFc(alpha) receptorNeutral ph
The present invention provides: a modified FcRn-binding domain having an enhanced affinity for the Fc Receptor neonatal (FcRn) at neutral pH; an antigen-binding molecule comprising said FcRn-binding domain, which has low immunogenicity, high stability and form only a few aggregates; a modified antigen-binding molecule having an increased FcRn-binding activity at neutral or acidic pH without an increased binding activity at neutral pH for a pre-existing anti-drug antibody; use of the antigen-binding molecules for improving antigen-binding molecule-mediated antigen uptake into cells; use of the antigen-binding molecules for reducing the plasma concentration of a specific antigen; use of the modified FcRn-binding domain for increasing the total number of antigens to which a single antigen-binding molecule can bind before its degradation; use of the modified FcRn-binding domain for improving pharmacokinetics of an antigen-binding molecule; methods for decreasing the binding activity for a pre-existing anti-drug antibody; and methods for producing said antigen-binding molecules.
Owner:CHUGAI PHARMA CO LTD
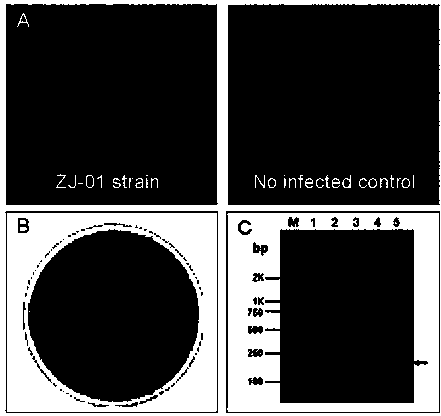
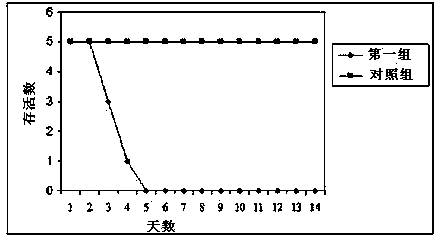
Porcine pseudorabies virus (PRV) variant PRV-ZJ01 and application thereof
ActiveCN103627678AImprove securityImproving immunogenicityMicroorganism based processesAntiviralsRabiesEngineering
The invention relates to the technical field of porcine pseudorabies viruses (PRVs) and in particular relates to a porcine PRV variant PRV-ZJ01 with collection number of CGMCCNo.8170 and an application of the porcine PRV variant PRV-ZJ01 in preparation of vaccines. The porcine PRV variant PRV-ZJ01 has the beneficial effects that a water-soluble inactivated vaccine is prepared by adopting a PRV-ZJ01 variant virus solution and is subjected to a swine immune protection test with live vaccines of Bartha-K61, Bucharest and HB-98 strains and the results show that the inactivated vaccine of the ZJ01 strain has relatively high safety and has the immune protection efficiency obviously higher than that of immunity groups of the live vaccines of the Bartha-K61, Bucharest and HB-98 strains, and the live vaccines of the Bartha-K61, Bucharest and HB-98 strains can not provide full protection for the ZJ01 very virulent strain; the inactivated vaccine of ZJ01 has relatively good immune protection effects on the PRV variant and the traditional strains; infected with 10<6.0>TCID50 (Tissue culture infectious dose 50) / ml nasal drops of the PRV-ZJ01 variant, all the 85-day-old non-immune swine can become ill and die; results prove that the virulence of the virus strain is obviously enhanced, the antigenicity is varied and the virus strain has relatively good immunogenicity after being inactivated and can be used for research and development of the vaccine of the virus strain and the diagnostic methods.
Owner:NANJING AGRICULTURAL UNIVERSITY
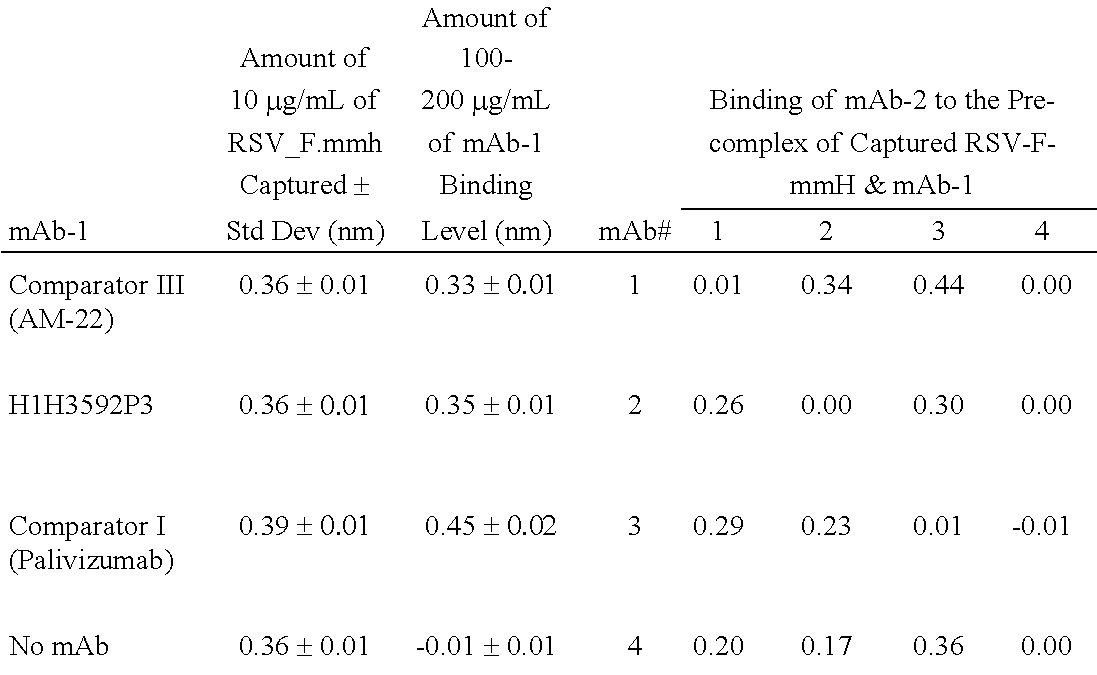
Human antibodies to respiratory syncytial virus f protein and methods of use thereof
ActiveUS20140271653A1Efficiently neutralizedGood protection levelVirusesAntipyreticF proteinCell membrane
The present invention provides fully human antibodies that bind to respiratory syncytial virus F protein, compositions comprising the antibodies and methods of use. The antibodies of the invention are useful for preventing fusion of the virus with the cell membrane and preventing cell to cell spread of the virus, thereby providing a means of preventing the infection, or treating a patient suffering from the infection and ameliorating one or more symptoms or complications associated with the viral infection. The antibodies may also be useful for diagnosis of an infection by RSV.
Owner:REGENERON PHARM INC
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com