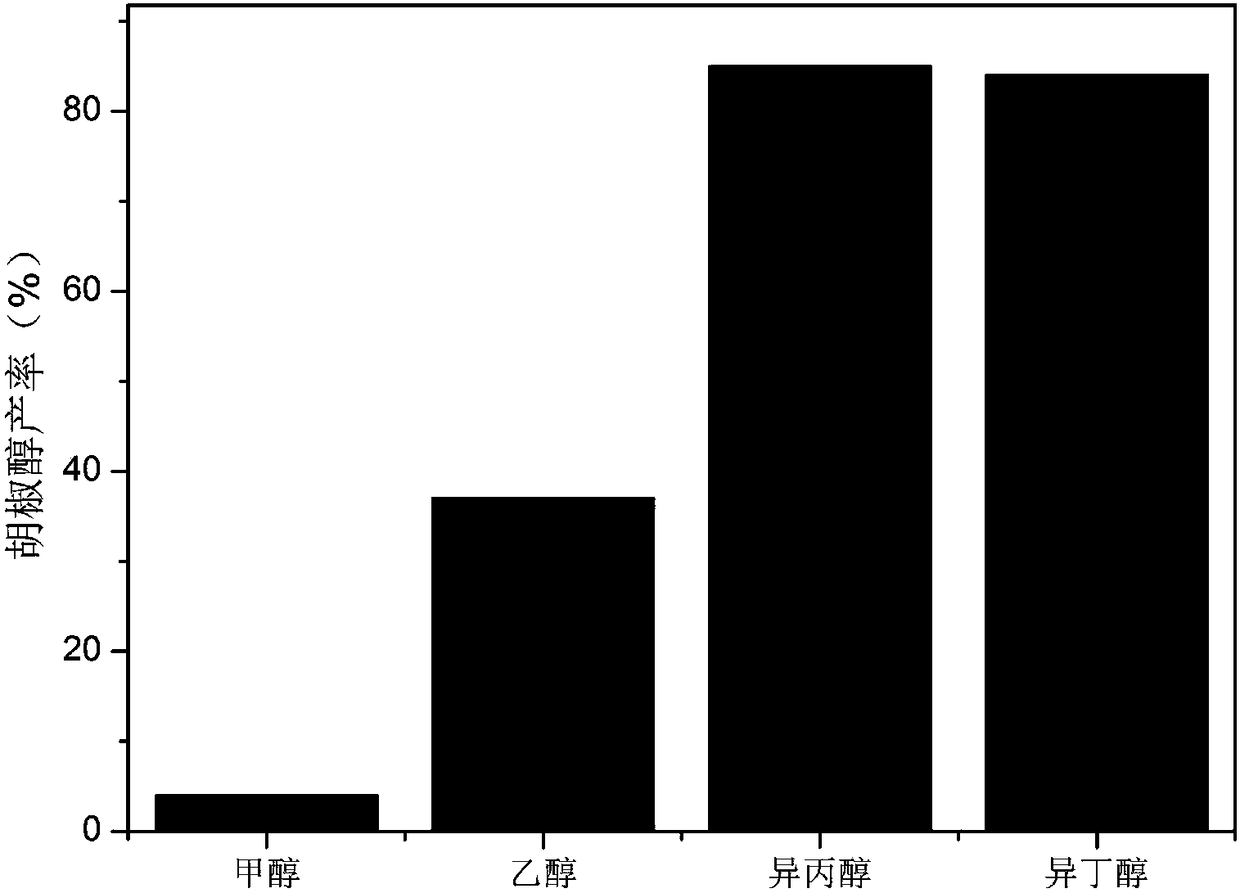
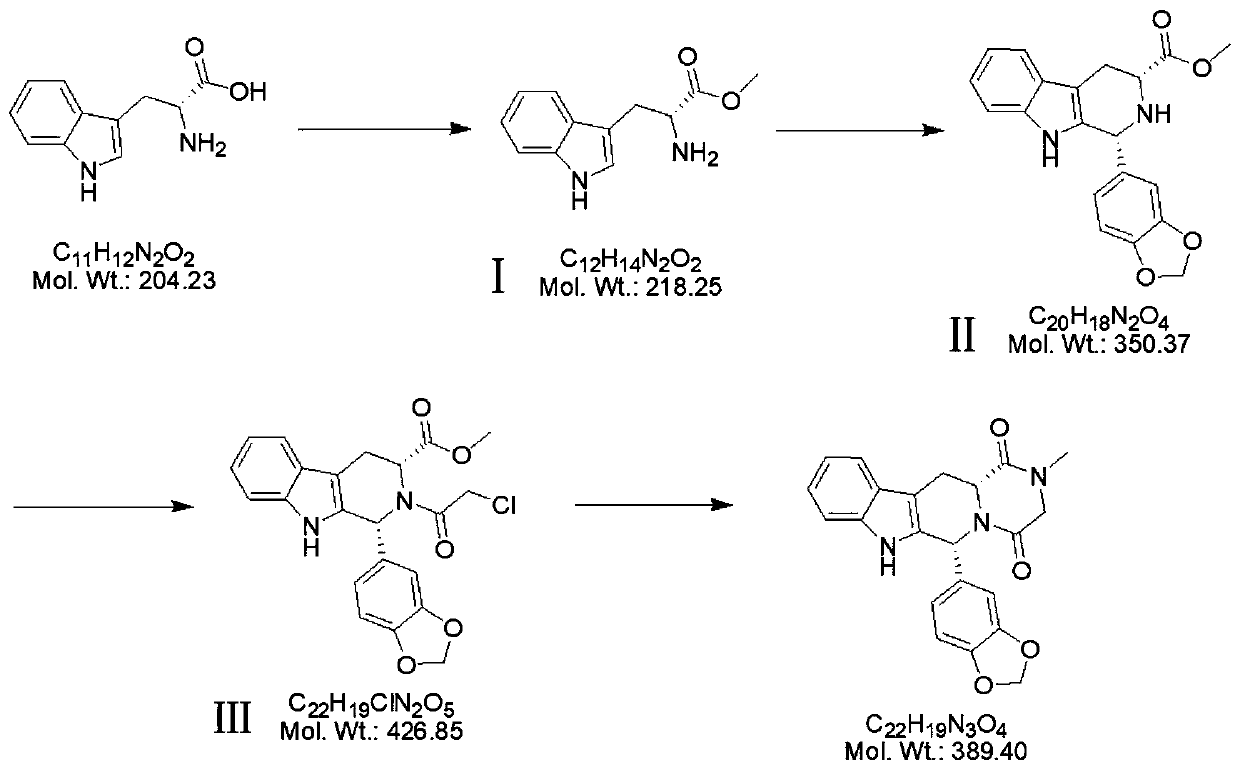
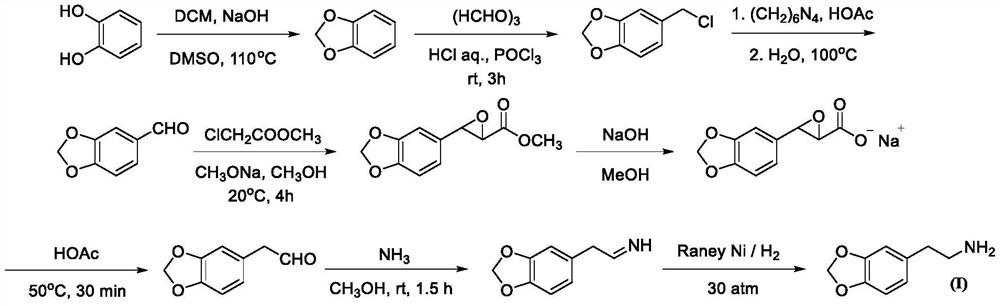
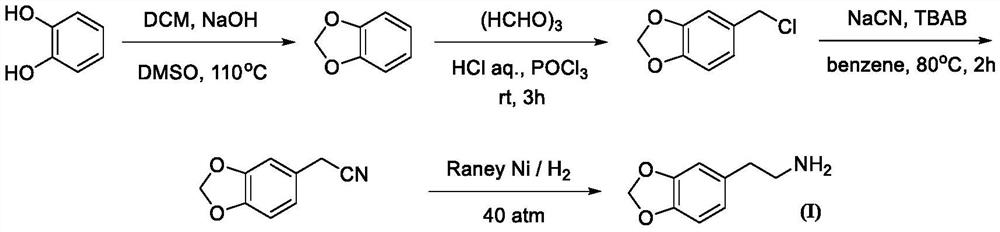
Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
35 results about "Piperonal" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
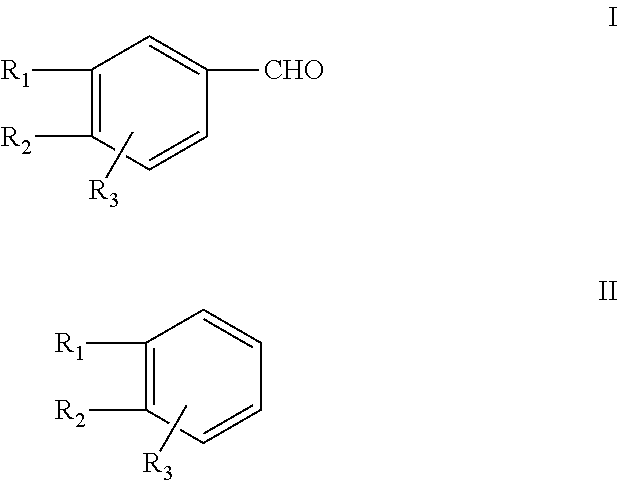
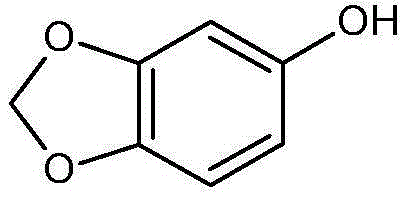
Piperonal, also known as heliotropin, is an organic compound which is commonly found in fragrances and flavors. The molecule is structurally related to other aromatic aldehydes such as benzaldehyde and vanillin.
Compositions and methods for controlling insects
Pest control compositions, blends, and formulations are disclosed. The blends contain, in a synergistic combinations, at least two ingredients such as Lilac Flower Oil, D-Limonene, Thyme Oil, Lime Oil, Black Seed Oil, Wintergreen Oil, Linalool, Tetrahydrolinalool, Vanillin, Isopropyl myristate, Piperonal (aldehyde), Geraniol, Geraniol 60, Triethyl Citrate, and Methyl Salicylate.
Owner:TYRATECH
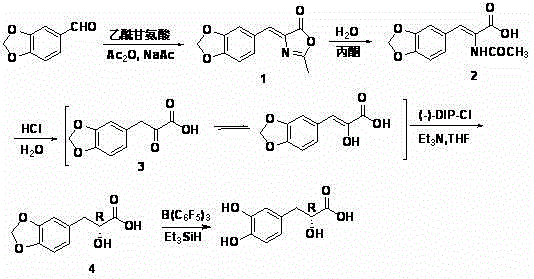
Asymmetric synthesis method of (+)-tanshinol
InactiveCN102924265ARaw materials are easy to getSimple and fast operationOrganic compound preparationCarboxylic compound preparationPropanoic acidCombinatorial chemistry
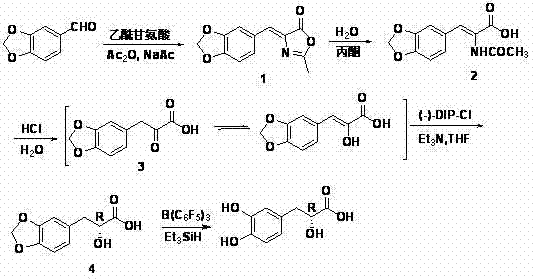
The invention relates to an asymmetric synthesis method of (+)-tanshinol, which comprises the following steps: carrying out Knoevenagel condensation on the raw material heliotropin, hydrolyzing for ring opening to obtain beta-(3,4-3,4-methylenedioxyphenyl)pyruvic acid, carrying out key asymmetric reduction reaction to obtain R-beta-(3,4-dibenzyloxyphenyl)-alpha-hydracrylic acid, and finally, deprotecting to obtain the (+)-tanshinol. The method has the advantages of accessible raw material and high optical purity of the product, is simple to operate, and can implement large-scale preparation.
Owner:FOURTH MILITARY MEDICAL UNIVERSITY
Preparing method of phosphodiesterase 5 inhibitor tadalafil
ActiveCN103980275AGood removal effectHigh chiral purityOrganic chemistryPhosphodiesterase 5 inhibitorTadalafil
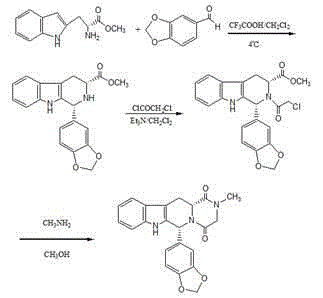
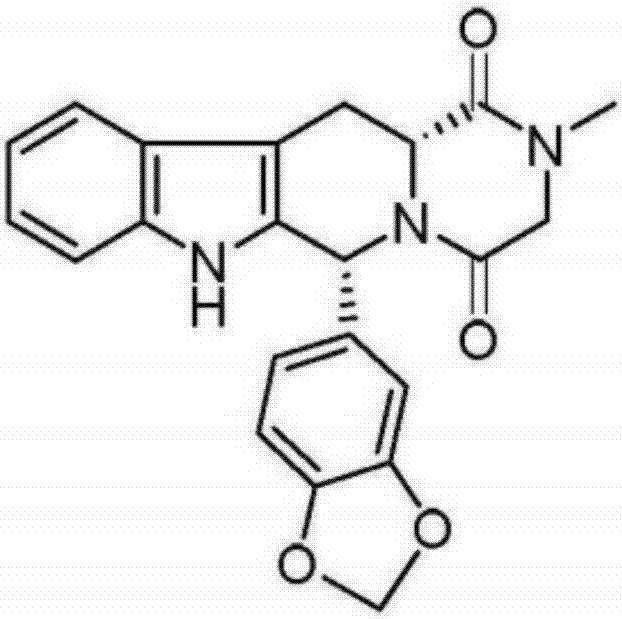
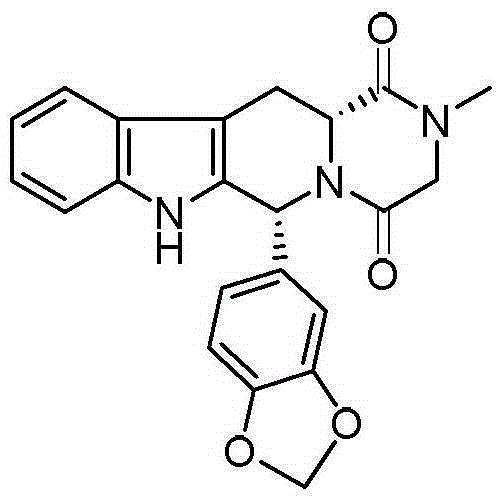
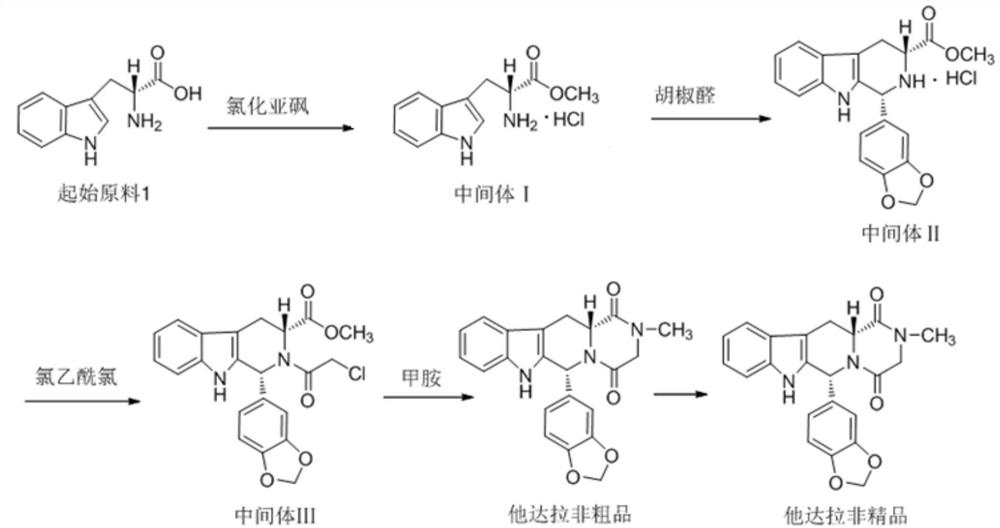
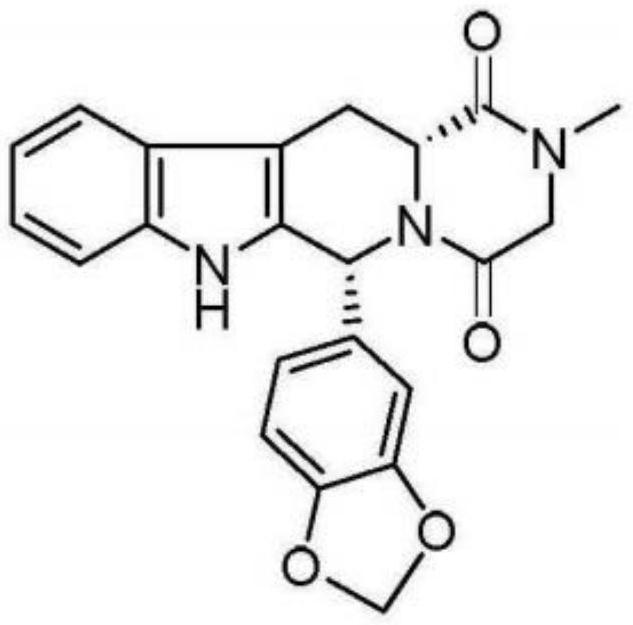
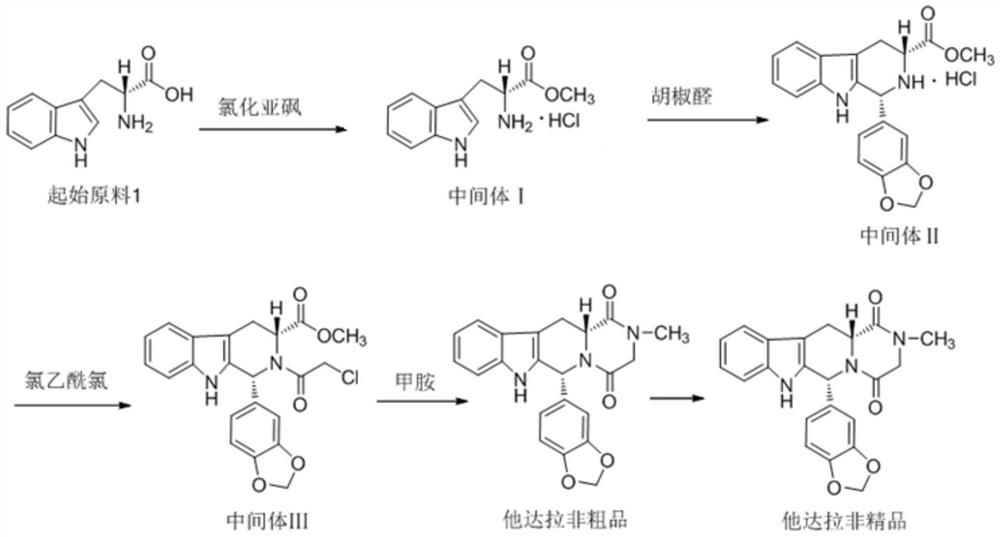
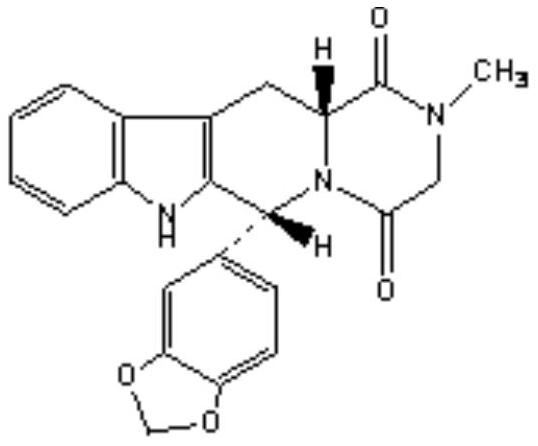
The invention relates to a preparing method of a phosphodiesterase 5 inhibitor tadalafil. D-methyl tryptophanate hydrochloride is adopted as an initial raw material, and is subjected to cyclization with heliotropin, N-acylation, aminolysis-cyclization, and other reactions to obtain a tadalafil crude product. The tadalafil crude product is recrystallized to obtain a tadalafil finished product. The method has characteristics of mild reaction conditions, short reaction time, high yield, good product stability and convenience for industrial production.
Owner:湖北省医药工业研究院有限公司
Synergistic pest-control compositions
Pest control compositions, blends, and formulations are disclosed. The blends contain, in a synergistic combinations, at least two ingredients such as Lilac Flower Oil, D-Limonene, Thyme Oil, Lime Oil, Black Seed Oil, Wintergreen Oil, Linalool, Tetrahydrolinalool, Vanillin, Isopropyl myristate, Piperonal (aldehyde), Geraniol, Geraniol 60, Triethyl Citrate, and Methyl Salicylate.
Owner:TYRATECH
Improved tadalafil preparation method
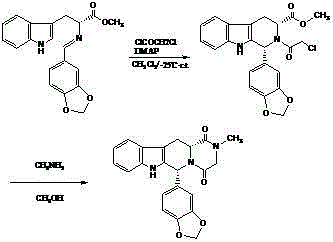
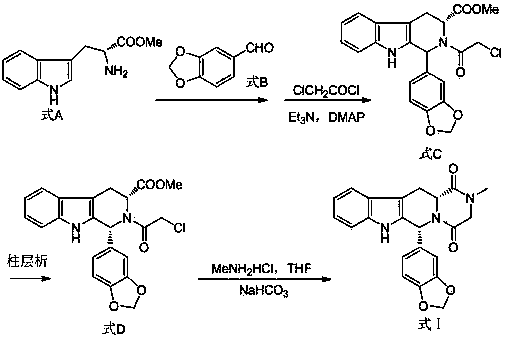
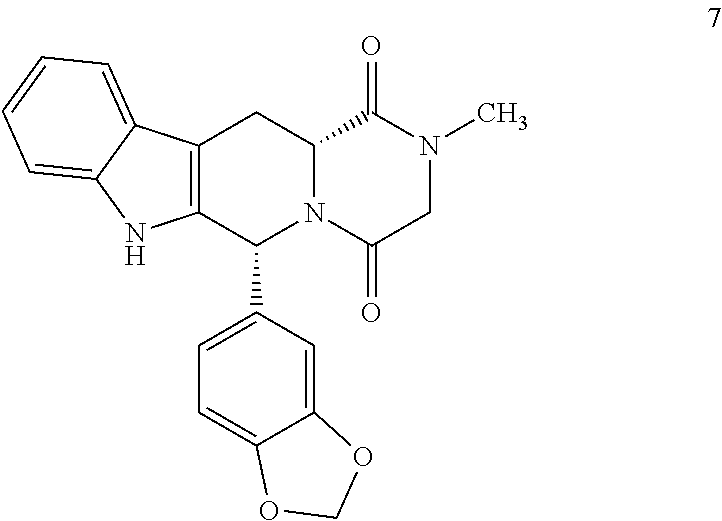
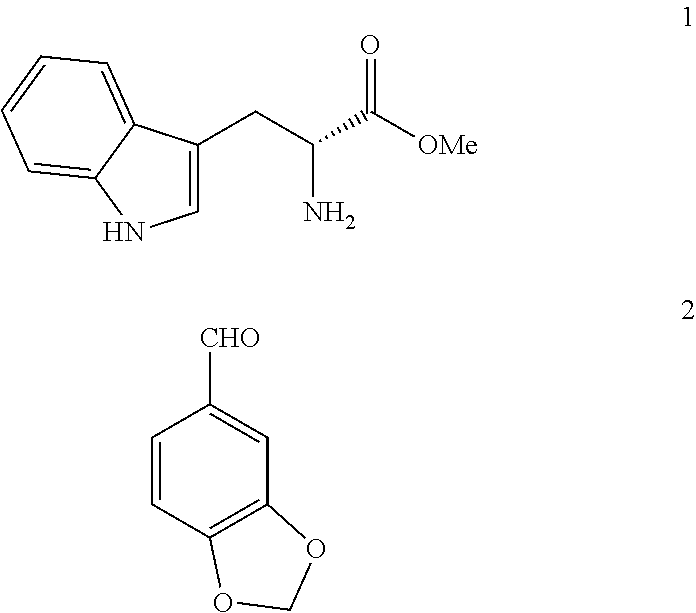
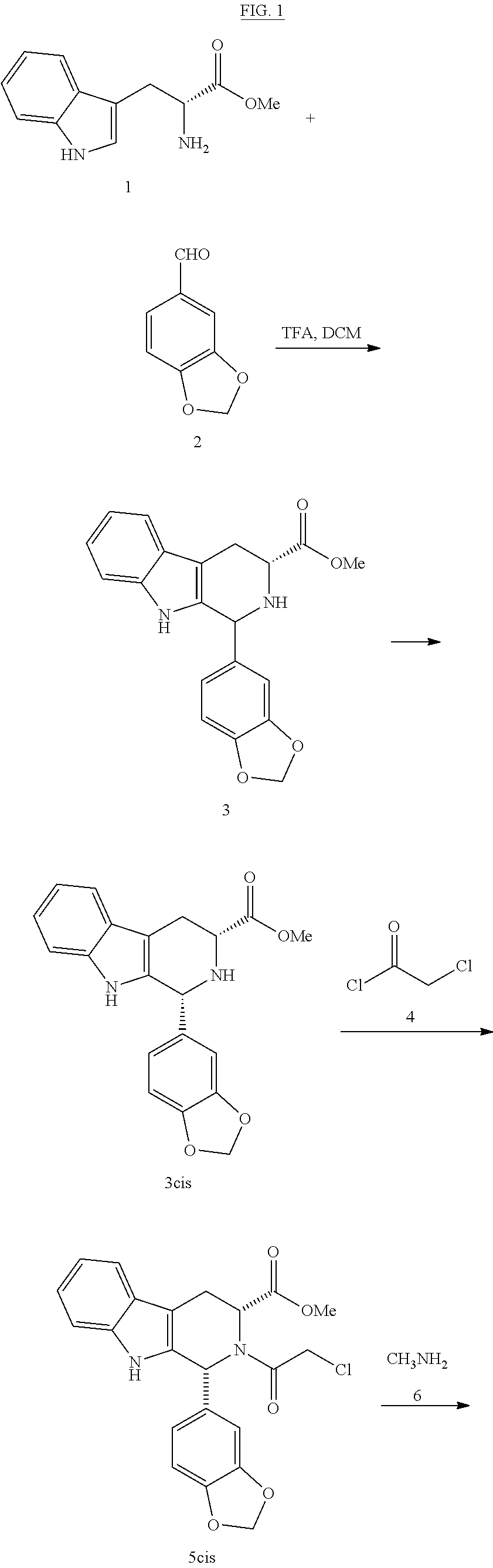
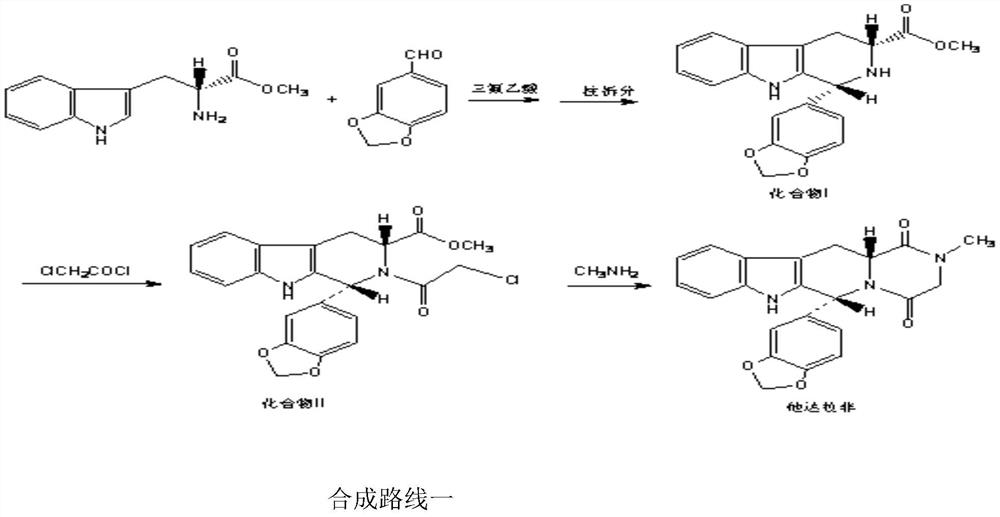
The invention belongs to the field of preparation of chemical raw medicaments, and more in particular relates to an improved preparation method for a phosphodiesterase 5 inhibitor tadalafil. A specific synthesis route is shown in the specification. The method comprises the following steps of performing Pictet-Spengler cyclization reaction and chloroacetyl chloride acylation on starting reactants (D-tryptophan methyl ester hydrochloride and piperonal) to obtain an intermediate product, directly performing subsequent reaction on the intermediate product without purification, preparing an intermediate 1-(1,3-benzodioxol-5-yl)-2-(chloracetyl)-2,3,4,9-tetrahydro-1H-pyridino-[3,4,-B]indol-3-thiophenate methyl by using a one-pot reaction method, performing column chromatography purification to obtain a single cis-isomer, and finally reacting the single cis-isomer with methylamine hydrochloride in the presence of an inorganic base to obtain the tadalafil.
Owner:ANHUI WANBANG MEDICAL TECH
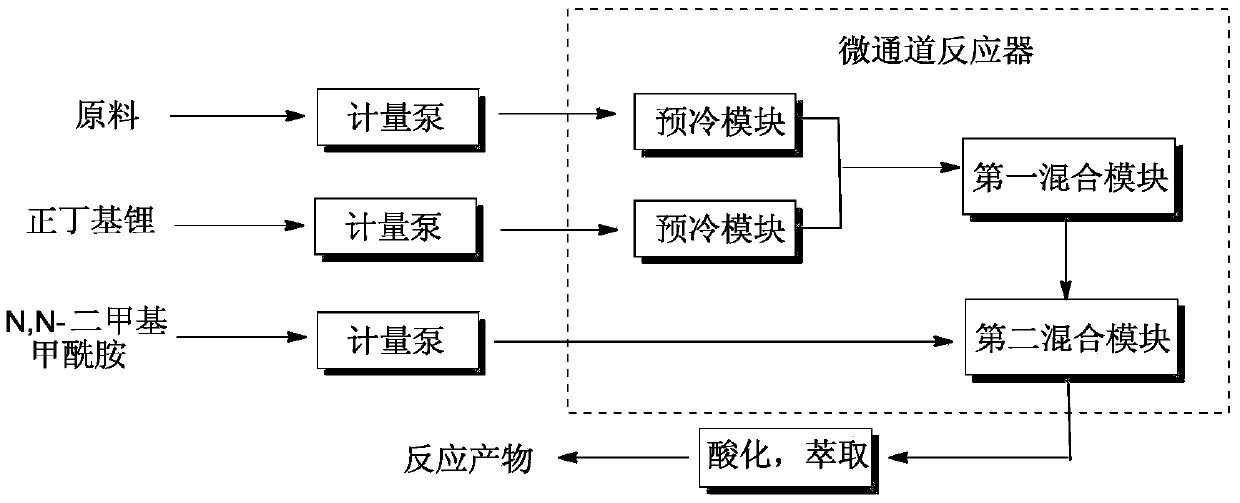
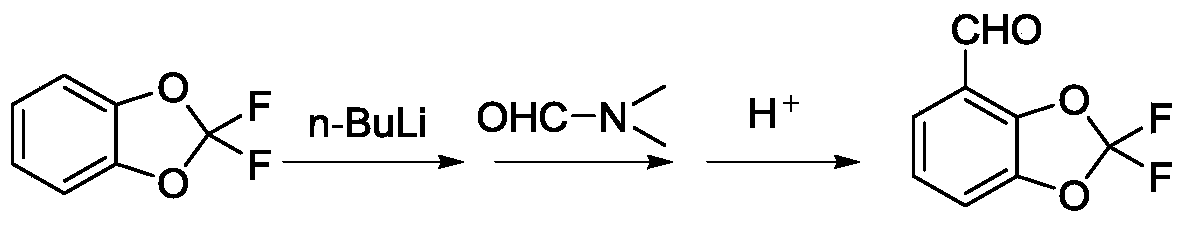
Method for preparing difluoro piperonal by utilizing continuous flow microchannel reactor
ActiveCN105254610AShort reaction timeImprove reaction efficiencyOrganic chemistryOrganic solventN-Butyllithium
The invention discloses a method for preparing difluoro piperonal by utilizing a continuous flow microchannel reactor. The method for preparing the difluoro piperonal by utilizing the continuous flow microchannel reactor comprises the following steps: respectively introducing difluoro piperidine organic solvent solution and n-hexane solution of n-butyllithium into a precooling module of a microchannel reactor by virtue of a metering pump to be precooled, then enabling the difluoro piperidine organic solvent solution and n-hexane solution of n-butyllithium to enter a first mixing module for reacting, after reaction is finished, enabling reaction products to enter a second mixing module for reacting with N,N-dimethyl formamide, acidifying, extracting, and distilling, so that the difluoro piperonal is obtained. The method for preparing the difluoro piperonal by utilizing the continuous flow microchannel reactor has the advantages of short reaction time, easiness and safety in operation and low production energy consumption.
Owner:XIAN MODERN CHEM RES INST
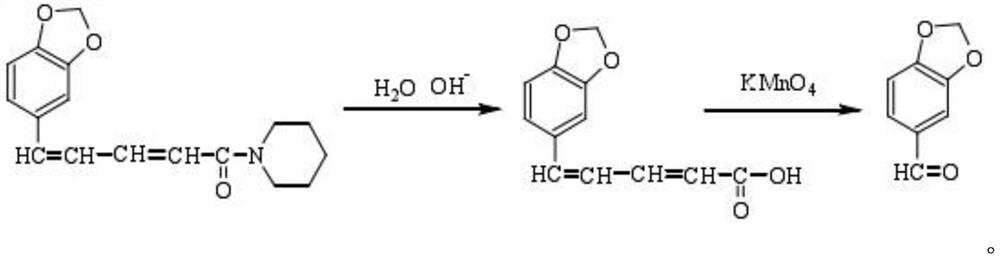
Method for synthesizing piperonal
The invention relates to a method for synthesizing piperonal, which comprises the following steps of: adding 3,4-methylenedioxy benzene acid, nitric acid, sulfate, sodium nitrite and water into a reaction kettle by one step; after introducing oxygen gas, sealing the reaction kettle and obtaining acid filtrate, a piperonal product and recovered gas comprising nitrogen oxide and the oxygen gas after reacting; introducing the oxygen gas and the obtained recovered gas comprising the nitrogen oxide and the oxygen gas into the reaction kettle filled with the 3,4-methylenedioxy benzene acid and the acid filtrate, sealing the reaction kettle and obtaining the acid filtrate, the piperonal product and the recovered gas comprising the nitrogen oxide and the oxygen gas after reacting; and carrying out the operation repeatedly. The invention has the advantages of low cost, no pollution and simple operation.
Owner:SHANXI INST OF COAL CHEM CHINESE ACAD OF SCI
Compositions and methods for controlling insects
Pest control compositions, blends, and formulations are disclosed. The blends contain, in a synergistic combinations, at least two ingredients such as Lilac Flower Oil, D-Limonene, Thyme Oil, Lime Oil, Black Seed Oil, Wintergreen Oil, Linalool, Tetrahydrolinalool, Vanillin, Isopropyl myristate, Piperonal (aldehyde), Geraniol, Geraniol 60, Triethyl Citrate, and Methyl Salicylate.
Owner:TYRATECH
Preparation method of tadalafil
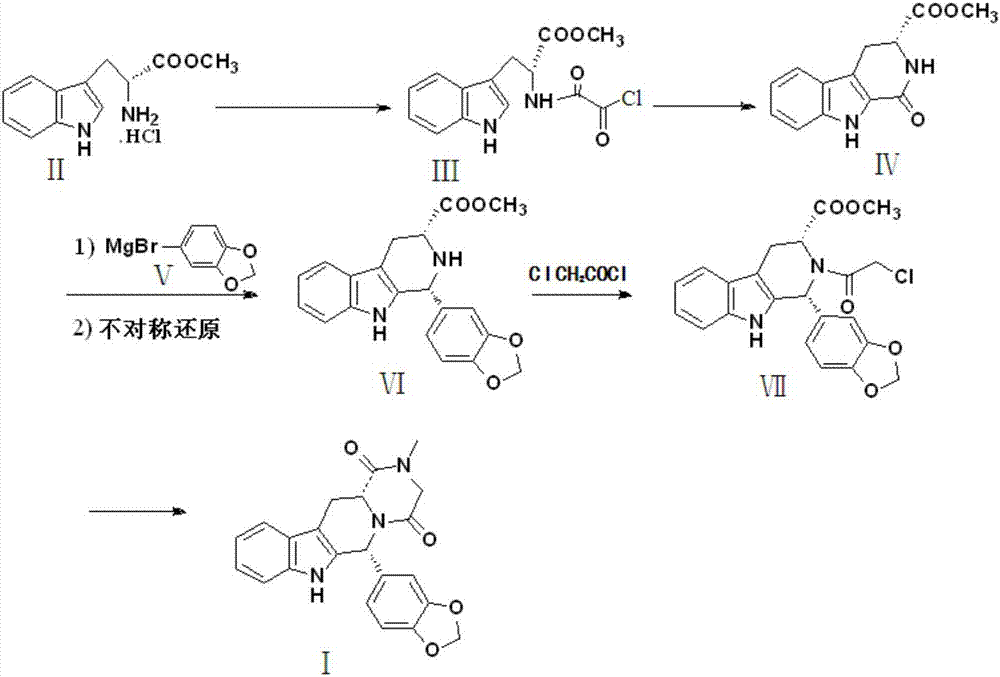
The invention discloses a preparation method of tadalafil, starting material D-Tryptophan methyl ester hydrochloride is reacted with oxalyl chloride to obtain an intermediate III, and the final product tadalafil (I) is obtained through cyclization, Grignard reaction, asymmetric reduction, substitution and condensation reaction. The use of national control chemical piperonal is avoided, an intermediate VI can be highly-selectively obtained by the asymmetric reduction, and the method has the advantages of simple postprocessing, short synthesis steps and high product total yield, and is suitable for industrialized production.
Owner:SHANDONG YUXIN PHARMA CO LTD
Method of preparing piperonal in one kettle way
The invention relates to a method of preparing piperonal in a one kettle way, which is a technical improvement of using 1,2-(methylenedioxy) benzene as a raw material to synthesize the piperonal. By the measures of selecting a proper solvent, adding an additive, diluting a reaction solution, and the like, the intermediate 3,4-(methylenedioxy) mandelic acid can be directly oxidized without being separated and purified to synthesize the piperonal in the 'one kettle' way. The invention has the characteristics of simple operation, high yield and the like, and both the purity and the quality of theproduct meet the requirements.
Owner:上海力智生化科技有限公司
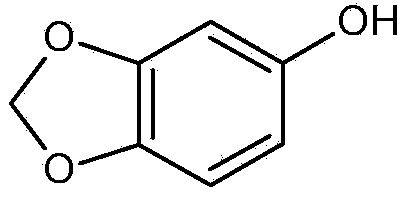
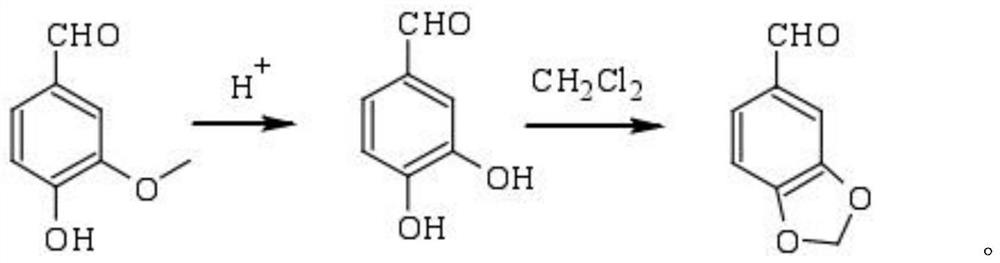
Preparation method and application of sesamol intermediate heliotropin
The invention belongs to the field of food additives and provides a preparation method of a sesamol intermediate heliotropin. The preparation method is convenient to use by taking nitric acid as a decarboxylating agent; the product, namely the sesamol intermediate heliotropin is good in purity; especially the decarboxylation efficiency is high, so that the decarboxylation reaction can be efficiently carried out to directly influence the synthesis yield of the overall preparation route; compared with other synthesis routes, the heliotropin synthesis route disclosed by the invention has considerable advantage on the cost, and the product competitiveness is relatively strong. The synthesis method disclosed by the invention has the advantages of cheap raw materials, simple reaction process and high yield and can be applied to large-scale synthesis of sesamol.
Owner:JINAN UNIVERSITY
Novel synthetic method for tadalafil
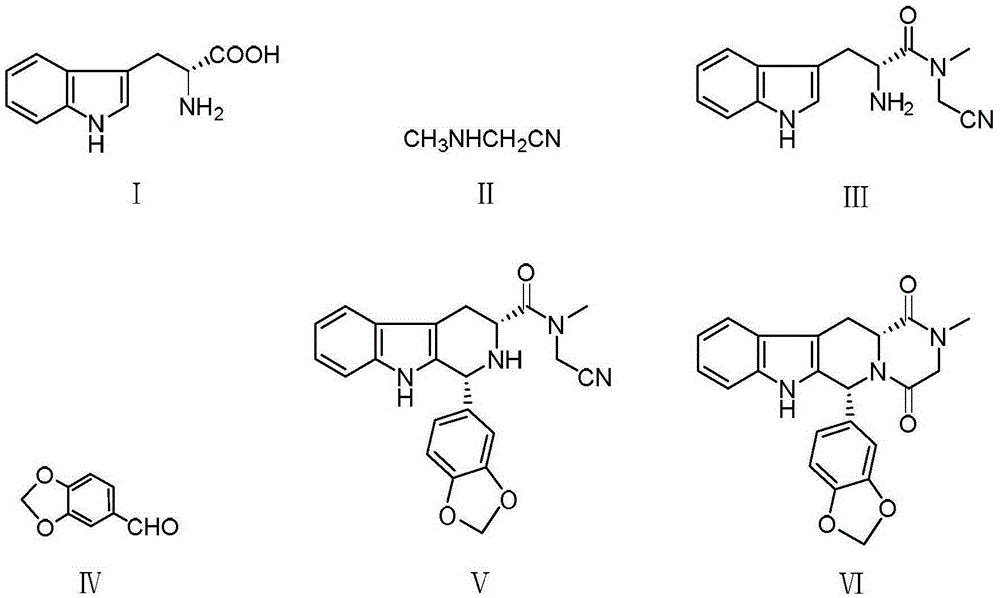
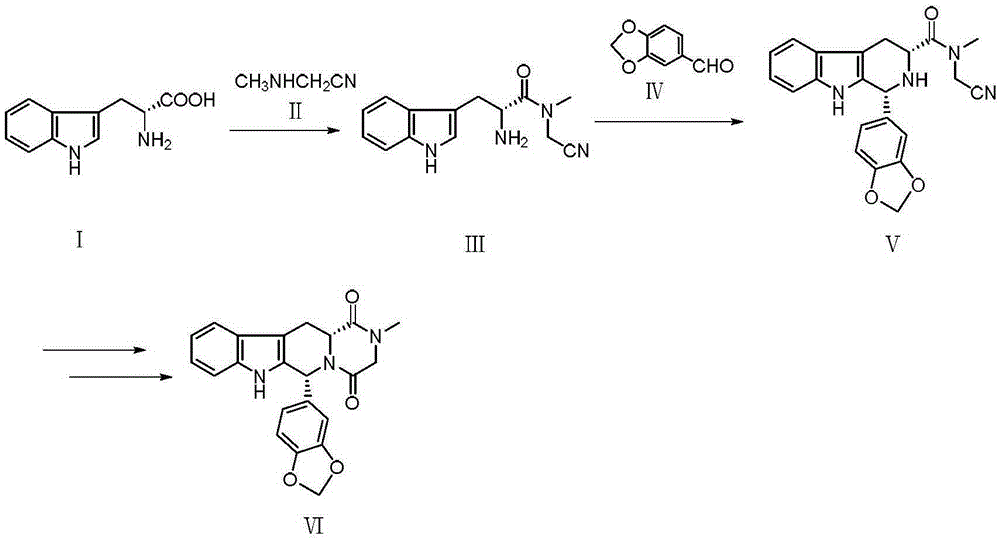
The invention relates to a synthetic method for a compound in the technical field of medicine and chemical engineering, and particularly relates to a novel synthetic method for tadalafil. The synthetic method comprises the steps of: 1) condensing an initial raw material which is D-tryptophan (I) and methylamino acetonitrile (II) or salt thereof to obtain a compound as shown in a formula III; 2) carrying out Pictet-Spengler cyclization reaction on the compound as shown in the formula III and heliotropin and carrying out crystallization to obtain a compound as shown in a formula V; and 3) hydrolyzing the compound V under an acidic or alkaline condition by cyano and then condensing the compound to obtain the target product tadalafil (VI). The formula is shown in the description. In the synthetic method provided by the invention, chemical raw materials which are great in toxicity, severe in environmental pollution and flammable and combustible such as thionyl chloride, chloroacetyl chloride, methylamine and the like. The novel synthetic method for tadalafil provided by the invention is simple in reaction method, convenient to operate and high in yield.
Owner:ZHEJIANG YONGNING PHARMA
Porous zirconium pyrophosphate catalyst and application thereof to piperaldehyde hydrogenation reaction
ActiveCN108314671AHigh selectivityReduce manufacturing costOrganic chemistryPhysical/chemical process catalystsChemistryPyrophosphate
The invention discloses a porous zirconium pyrophosphate catalyst and application thereof to piperaldehyde hydrogenation reaction and belongs to the technical field of solid acid catalysis. The porouszirconium pyrophosphate catalyst and the application thereof disclosed by the invention have the beneficial effects that the prepared catalyst is simple in synthetic step, raw materials used are cheap and easy to obtain, and the obtained catalyst self carries a large number of acidic sites and alkaline sites and can efficiently catalyze and reduce piperitol obtained from piperaldehyde; the catalyst is easy to separate after reaction, can be recycled for many times and meets the requirements of green and sustainable development.
Owner:JIANGNAN UNIV
Method of preparing piperonal in one kettle way
The invention relates to a method of preparing piperonal in a one kettle way, which is a technical improvement of using 1,2-(methylenedioxy) benzene as a raw material to synthesize the piperonal. By the measures of selecting a proper solvent, adding an additive, diluting a reaction solution, and the like, the intermediate 3,4-(methylenedioxy) mandelic acid can be directly oxidized without being separated and purified to synthesize the piperonal in the 'one kettle' way. The invention has the characteristics of simple operation, high yield and the like, and both the purity and the quality of the product meet the requirements.
Owner:上海力智生化科技有限公司
Piperonal preparation method
The invention relates to a preparation method of piperonal. The specific steps are: mix water, glyoxylic acid and sulfuric acid in proportion, add piperonyl cyclocyclone dropwise at a temperature of 0-5°C, stir at a high speed for 5-8 hours, add After diluting with 80-100ml of water, filter with suction to obtain solid 3,4-dimethoxymandelic acid, and the filtrate is the mother liquor containing dilute sulfuric acid; the mass ratio of water, glyoxylic acid, and sulfuric acid is 1.5-1.7:10- 11:10‑11; take 30%‑70% dilute sulfuric acid mother liquor, add solid 3,4‑dimethoxymandelic acid to it, then add 80 mL of dichloromethane, add 3% dilute nitric acid 18‑1 dropwise at 20°C 20mL, heated, and reacted for 1-3 hours until the reaction was complete; layers were separated, washed, concentrated and distilled under reduced pressure to obtain the product piperonal.
Owner:江西科美香料有限公司
Production process of tadalafil bulk drug
The invention belongs to the technical field of medicines, and particularly relates to a production process of a tadalafil bulk drug. The production process of the tadalafil bulk drug comprises the following steps: A1, carrying out an esterification reaction on methanol and D-tryptophan to obtain an intermediate I; A2, performing a condensation reaction on the intermediate I and heliotropin to obtain an intermediate II; A3, performing an acylation reaction on the intermediate II and chloroacetyl chloride to obtain an intermediate III; and A4, carrying out a cyclization reaction on the intermediate III and monomethylamine to obtain the tadalafil bulk drug. According to the method, a reaction path is reasonably selected, and meanwhile, the process details of each reaction step are deeply optimized, so high purity and yield of a product of each step of reaction can be obtained, the prepared tadalafil bulk drug is low in cost and good in stability, the economical efficiency of the whole reaction path is improved, and production cost is reduced.
Owner:SUZHOU HOMESUN PHARMA
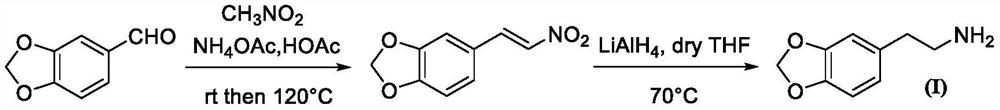
Synthesis method of homopiperony lamine
The invention belongs to the field of organic chemical synthesis, and particularly relates to a synthesis method of homopiperony lamine. The synthesis method comprises the following steps: preparing heliotropin by using catechol as a raw material; preparing beta-nitro-3, 4-dioxomethine styrene from heliotropin; and preparing the homopiperony lamine by using the beta-nitro-3, 4-dioxomethylenestyrene. The method for preparing heliotropin by using catechol as the raw material comprises the following two ways: (1) catechol -> 3,4-dihydroxy mandelic acid -> 3, 4-dihydroxy benzaldehyde -> heliotropin; and (2) catechol -> benzodioxole -> heliotropin. The used raw materials are safe, easy to obtain and low in cost; reaction conditions are mild, operation is simple and convenient, chemical yield is high, and intermediate reagents are easy to recover; and the method is suitable for industrial production.
Owner:SICHUAN UNIV +1
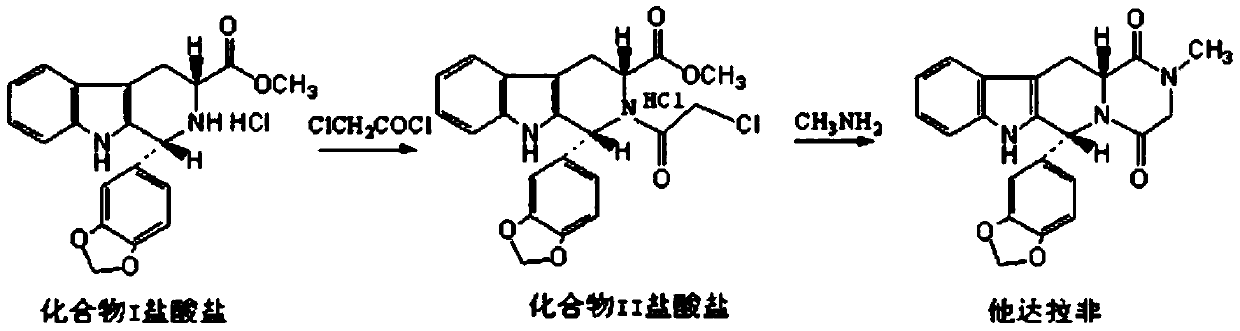
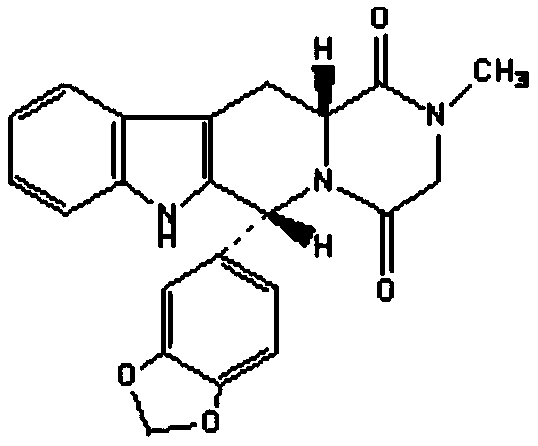
Method for preparing Tadalafil by one-pot method
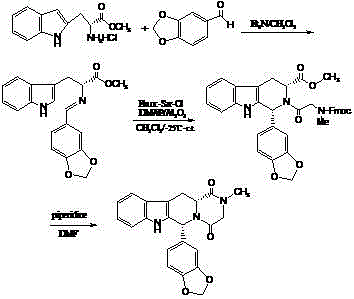
The invention discloses a method for preparing Tadalafil by a one-pot method. The method for preparing the Tadalafil by the one-pot method comprises the steps that D-Tryptophan methyl ester hydrochloride and piperonal are used as starting materials, and a single-configuration high-purity compound I hydrochloride is obtained through a Pictet-Spengler reaction; the compound I hydrochloride is subjected to an acylation reaction with chloroacetyl chloride in an organic base system of an aprotic solvent to obtain a mixed reaction solution; and the mixed reaction solution is directly added to a methylamine solution to undergo an aminolysis cyclization reaction without treatment, and the mixture is subjected to cooling crystallization to obtain the Tadalafil after the reaction. According to the method for preparing the Tadalafil by the one-pot method, the usage amount of the solvent is reduced, the amount of waste liquid discharge is effectively controlled, and environmental protection is facilitated; the production cycle is shortened, and the process efficiency is improved; and the yield is improved, and the cost is reduced. Compared with an existing process route, the method for preparing the Tadalafil by the one-pot method is more environmentally friendly, more efficient and lower in cost, and is more suitable for industrial production.
Owner:四川省通园制药集团有限公司
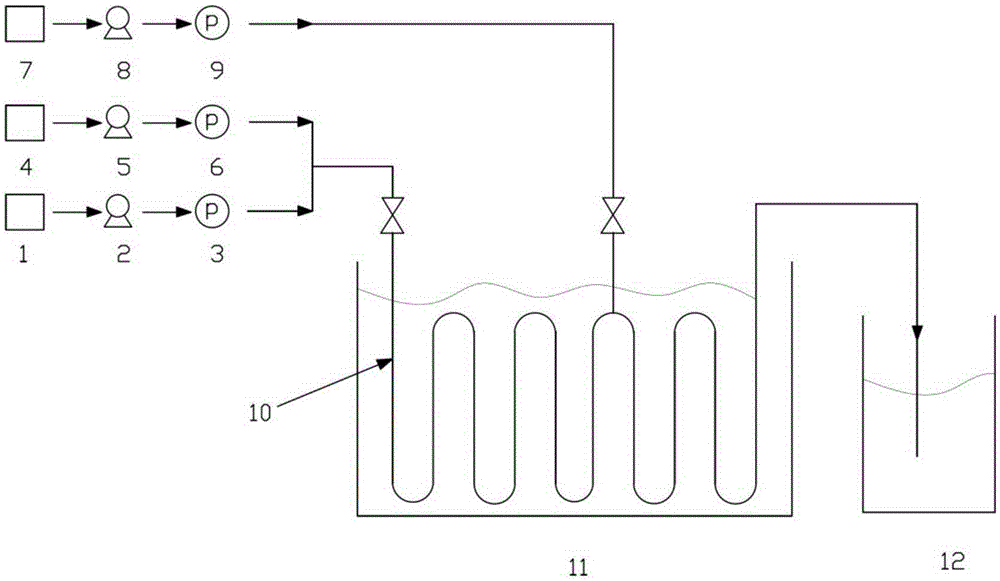
A kind of treatment method of piperonal production wastewater
ActiveCN114477666BIncrease B/C valueGood removal effectWater contaminantsTreatment involving filtrationChemical synthesisIndustrial waste water
The invention relates to the technical field of industrial wastewater treatment, and specifically discloses a treatment method for piperonal production wastewater. The method for treating the piperonal production wastewater includes: adding 1,2-methylenedioxybenzene to the piperonal production wastewater, mixing uniformly, performing distillation, collecting fractions, and separating liquids to obtain recovered materials; cooling the remaining mother liquor from the distillation , After adjusting the pH to weak acidity, adding transition metal oxides, performing high-pressure pulse treatment, filtering, and treating the treated water through the sewage treatment system and discharging. The method for treating wastewater from piperonal production provided by the invention has the advantages of simple process operation and low energy consumption, and solves the problem that the reaction raw materials and reaction products in the wastewater cannot be recovered by the traditional treatment process, and the treated wastewater can reach the level of "Chemical Synthesis Pharmaceutical Industry" "Water Pollutant Discharge Standard" discharge standard, will not cause secondary pollution, realize the comprehensive treatment and resource utilization of wastewater, has high economic and environmental benefits, and has high promotion value.
Owner:HEBEI HAILI FRAGRANCES CO LTD
A kind of preparation method of piperonal
Owner:SUZHOU HOMESUN PHARMA
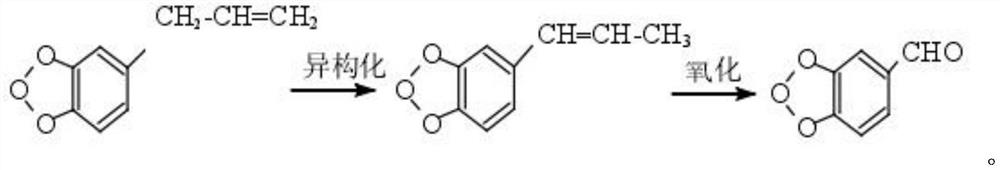
A method for biosynthesizing piperonal with recombinant escherichia coli
ActiveCN107699587BShort fermentation cycleEasy to operateBacteriaMicroorganism based processesEscherichia coliEnzyme Gene
The invention relates to a heliotropin biosynthesis method utilizing recombinant escherichia coli, and belongs to the technical filed of biological engineering. The method is characterized in that anoxidase gene derived from pseudomonas is introduced into escherichia coli. The recombinant bacterium can catalyze heliotropin synthesis from propenylbenzene, and the concentrated of the produced heliotropin is 9.15 g / L. The method in which the heliotropin is synthesized by utilizing recombinant oxidase has characteristics of a short production period, and simple extraction, and has a good industrial production prospect.
Owner:JIANGNAN UNIV +1
Efficient process for the synthesis of alkoxy substituted benzaldehydes
ActiveUS10633360B2Reduce in quantityMinimize handlingOrganic compound preparationPreparation from heterocyclic compoundsEthylenedioxyBenzaldehyde
The present invention relates to the synthesis of alkoxy substituted benzaldehydes obtained from the corresponding alkoxy substituted benzenes. Alkoxy substituted benzaldehydes are products of broad commercial interest and are used as end products and intermediates in flavor and fragrance applications and pharmaceutical ingredients. For example, 3,4-methylendioxy-benzaldehyde (also known as heliotropin or piperonal) is used widely both as a end product and intermediate for the above mentioned applications. Other examples include 3,4-dimethoxybenzaldehyde, 3,4,5-trimethoxybenzaldehyde and 3,4-ethylenedioxybenzene which are intermediates in the synthesis of active pharmaceutical intermediates.
Owner:ANTHEA AROMATICS
Preparation method of phosphodiesterase inhibitor
The invention discloses a preparation method of a phosphodiesterase inhibitor, belonging to the field of medicinal chemistry. The preparation method comprises the following steps: carrying out an esterification reaction on an initial raw material 1 and methanol to prepare an intermediate I, directly carrying out condensation cyclization on the intermediate I and heliotropin to prepare an intermediate II, and carrying out chloracetylation, aminolysis cyclization and refining to prepare a final product, namely a refined tadalafil product. The method improves product yield and quality, greatly shortens a reaction period in a preparation process, reduces operation steps and production cost, and is suitable for industrial mass production.
Owner:SHANDONG LUOXIN PARMACEUTICAL GROUP STOCK CO LTD +2
Cyanogen-free silver-plating brightener and method for preparing the same
The preparation method for cyan-free silver plating lustre-coating agent comprises: adding 0.1-1mol / L heliotropin into saturated 0.1-2mol / L NaHSO3 solution for ultrasonic oscillating; dissolving 0.1-2mol / L triethanolamine and 0.1-1mol / L butynediol into the solution, adding solvent to dilute to 1L. This invention overcomes the defects in prior art withnot sulfur-containing compound, and has well dispersion.
Owner:HARBIN INST OF TECH
Asymmetric Synthesis of (+)-Danshensu
InactiveCN102924265BRaw materials are easy to getSimple and fast operationOrganic compound preparationCarboxylic compound preparationPropanoic acidCombinatorial chemistry
The invention relates to an asymmetric synthesis method of (+)-tanshinol, which comprises the following steps: carrying out Knoevenagel condensation on the raw material heliotropin, hydrolyzing for ring opening to obtain beta-(3,4-3,4-methylenedioxyphenyl)pyruvic acid, carrying out key asymmetric reduction reaction to obtain R-beta-(3,4-dibenzyloxyphenyl)-alpha-hydracrylic acid, and finally, deprotecting to obtain the (+)-tanshinol. The method has the advantages of accessible raw material and high optical purity of the product, is simple to operate, and can implement large-scale preparation.
Owner:FOURTH MILITARY MEDICAL UNIVERSITY
Method for preparing difluoropiperonal in continuous flow microchannel reactor
ActiveCN105254610BShort reaction timeImprove reaction efficiencyOrganic chemistryOrganic solventN-Butyllithium
The invention discloses a method for preparing difluoro piperonal by utilizing a continuous flow microchannel reactor. The method for preparing the difluoro piperonal by utilizing the continuous flow microchannel reactor comprises the following steps: respectively introducing difluoro piperidine organic solvent solution and n-hexane solution of n-butyllithium into a precooling module of a microchannel reactor by virtue of a metering pump to be precooled, then enabling the difluoro piperidine organic solvent solution and n-hexane solution of n-butyllithium to enter a first mixing module for reacting, after reaction is finished, enabling reaction products to enter a second mixing module for reacting with N,N-dimethyl formamide, acidifying, extracting, and distilling, so that the difluoro piperonal is obtained. The method for preparing the difluoro piperonal by utilizing the continuous flow microchannel reactor has the advantages of short reaction time, easiness and safety in operation and low production energy consumption.
Owner:XIAN MODERN CHEM RES INST
Process for obtaining compounds derived from tetrahydro-.beta.-carboline
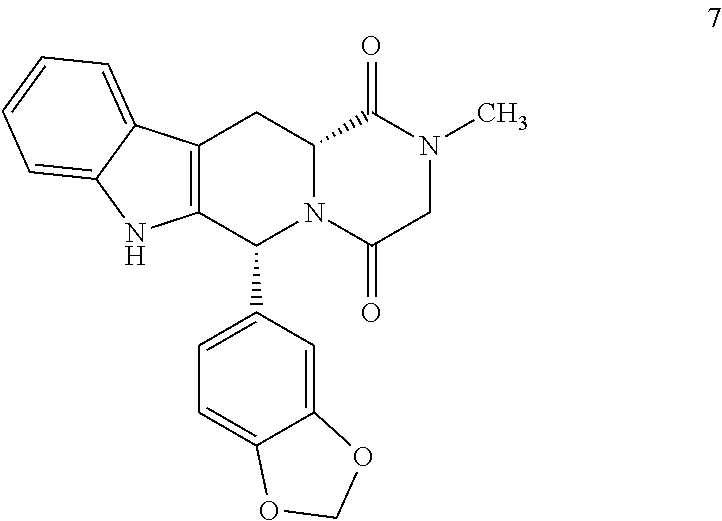
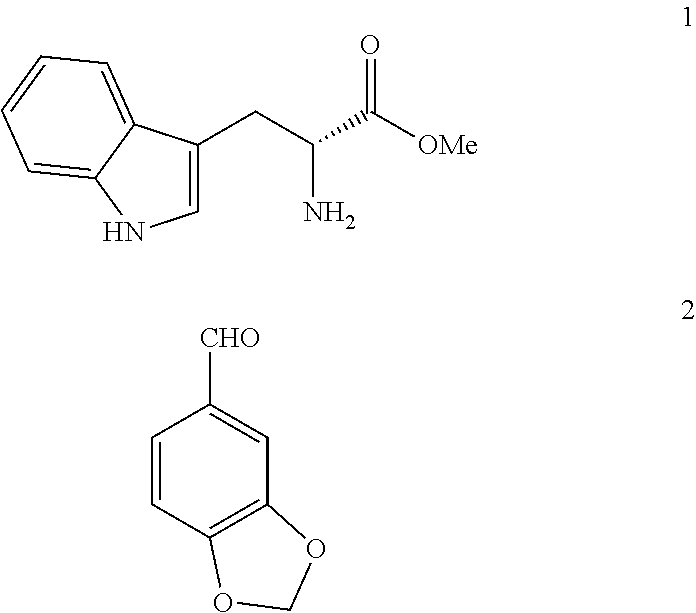
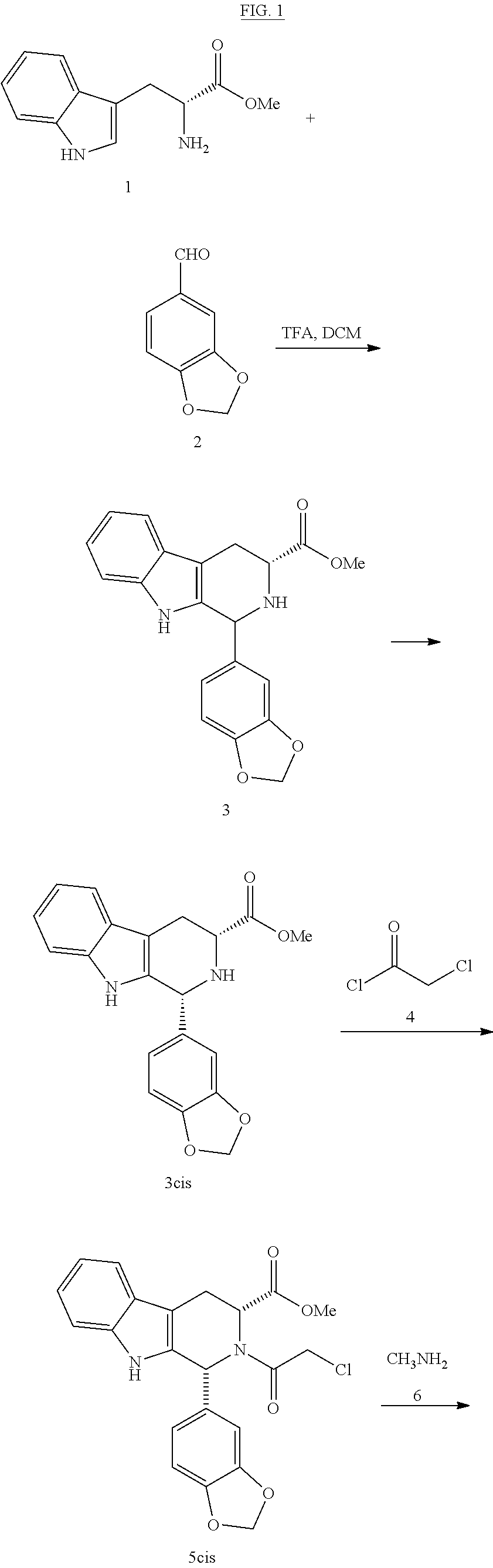
The invention relates to a process for obtaining compounds derived from tetrahydro-β-carboline, specifically tadalafil and intermediate products from the synthesis, comprising the reaction between piperonal and an alkyl ester of D-tryptophan as a salt, and in the absence of any other component, followed by haloacetylation and a final cyclization with methylamine.
Owner:INTERQUIM SA
Preparation method and application of sesamol intermediate piperonal
The invention belongs to the field of food additives and provides a preparation method of a sesamol intermediate heliotropin. The preparation method is convenient to use by taking nitric acid as a decarboxylating agent; the product, namely the sesamol intermediate heliotropin is good in purity; especially the decarboxylation efficiency is high, so that the decarboxylation reaction can be efficiently carried out to directly influence the synthesis yield of the overall preparation route; compared with other synthesis routes, the heliotropin synthesis route disclosed by the invention has considerable advantage on the cost, and the product competitiveness is relatively strong. The synthesis method disclosed by the invention has the advantages of cheap raw materials, simple reaction process and high yield and can be applied to large-scale synthesis of sesamol.
Owner:JINAN UNIVERSITY
PROCESS FOR OBTAINING COMPOUNDS DERIVED FROM TETRAHYDRO-beta-CARBOLINE
ActiveUS20130324730A1Improve liquidityHigh yieldOrganic chemistrySexual disorderD tryptophanTetrahydro-beta-carboline
The invention relates to a process for obtaining compounds derived from tetrahydro-β-carboline, specifically tadalafil and intermediate products from the synthesis, comprising the reaction between piperonal and an alkyl ester of D-tryptophan as a salt, and in the absence of any other component, followed by haloacetylation and a final cyclization with methylamine.
Owner:INTERQUIM SA
A kind of one pot method prepares the method for tadalafil
The invention discloses a method for preparing tadalafil by a one-pot method. Using D-tryptophan methyl ester hydrochloride and piperonal as starting materials, a high-purity compound I with a single configuration is obtained through Pictet-Spengler reaction Hydrochloride, and then acylated with chloroacetyl chloride in an organic base system of an aprotic solvent to obtain a mixed reaction solution. The mixed reaction solution is not treated and directly added to methylamine solution to undergo an aminolysis and cyclization reaction. After the reaction, cooling crystallization Later got tadalafil. The method of the invention reduces the amount of solvent used, thereby effectively controlling the discharge of waste liquid, which is beneficial to environmental protection; the production cycle is shortened, the process efficiency is improved; the yield is increased and the cost is reduced. Compared with the existing process route, the present invention is more environmentally friendly, has higher efficiency and lower cost, and is more suitable for industrial production.
Owner:四川省通园制药集团有限公司
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com