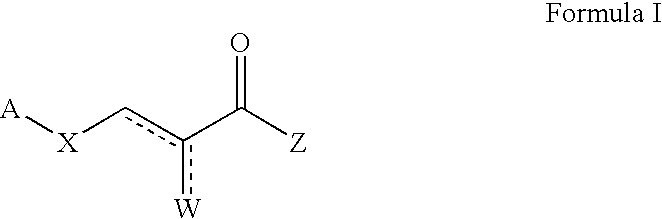
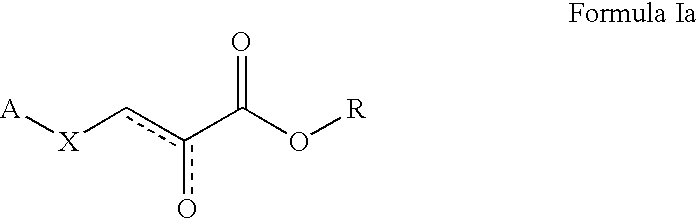
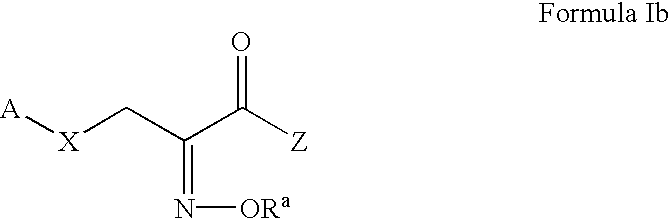
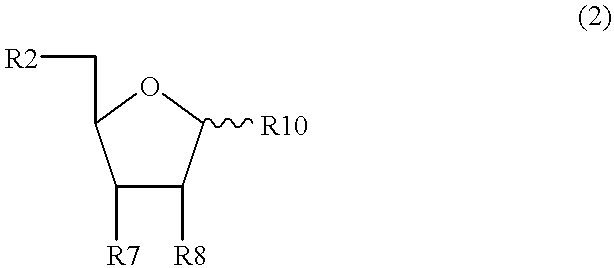
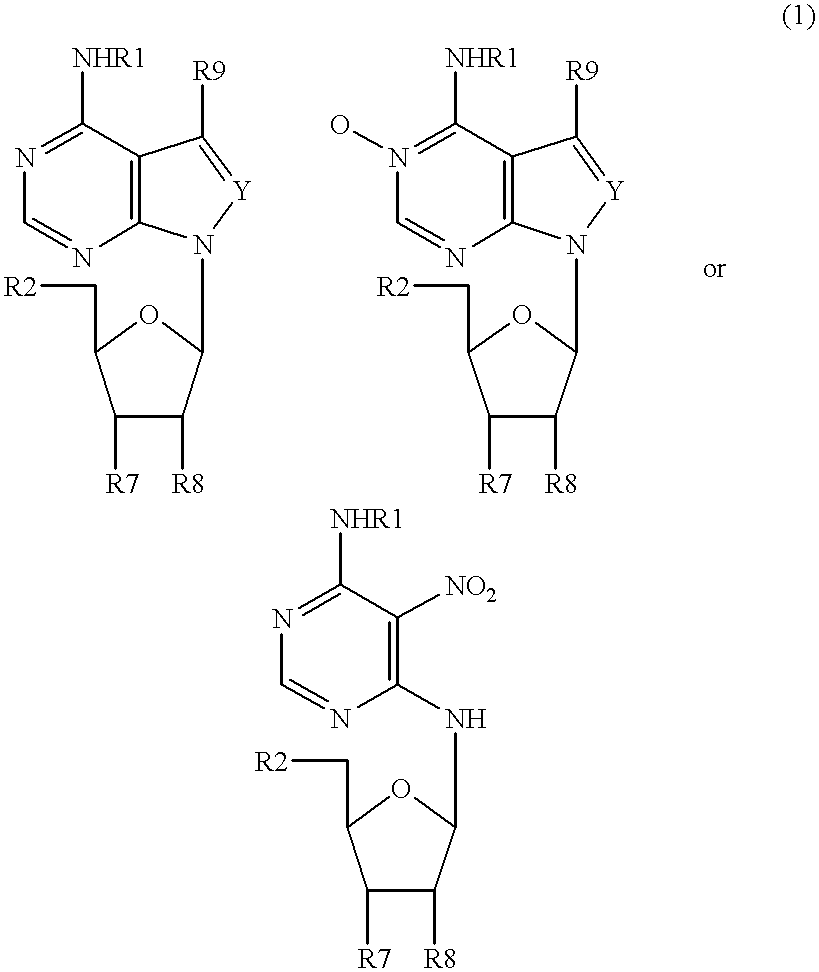
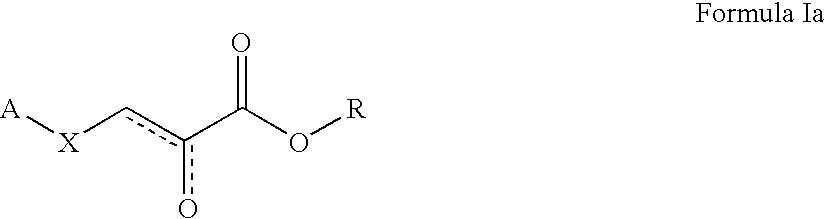
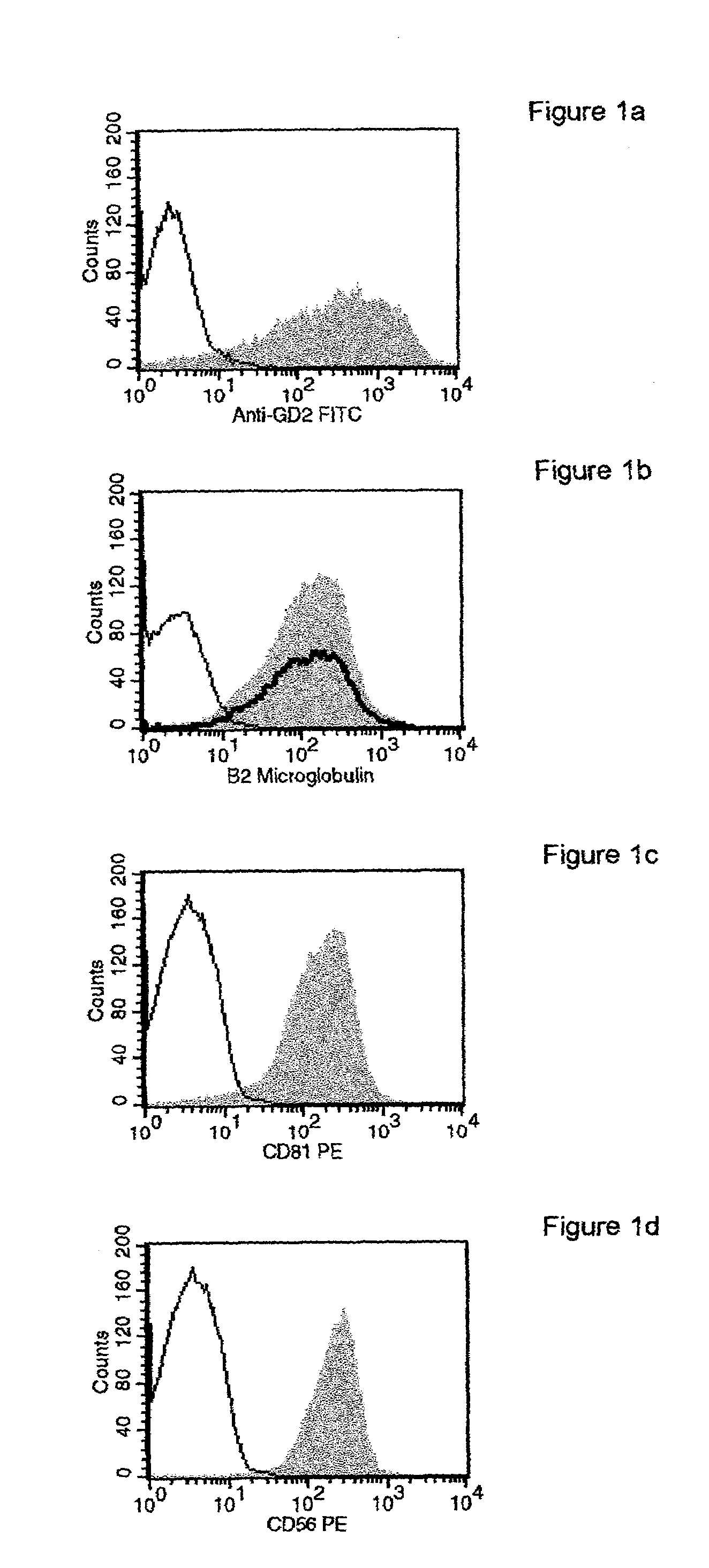
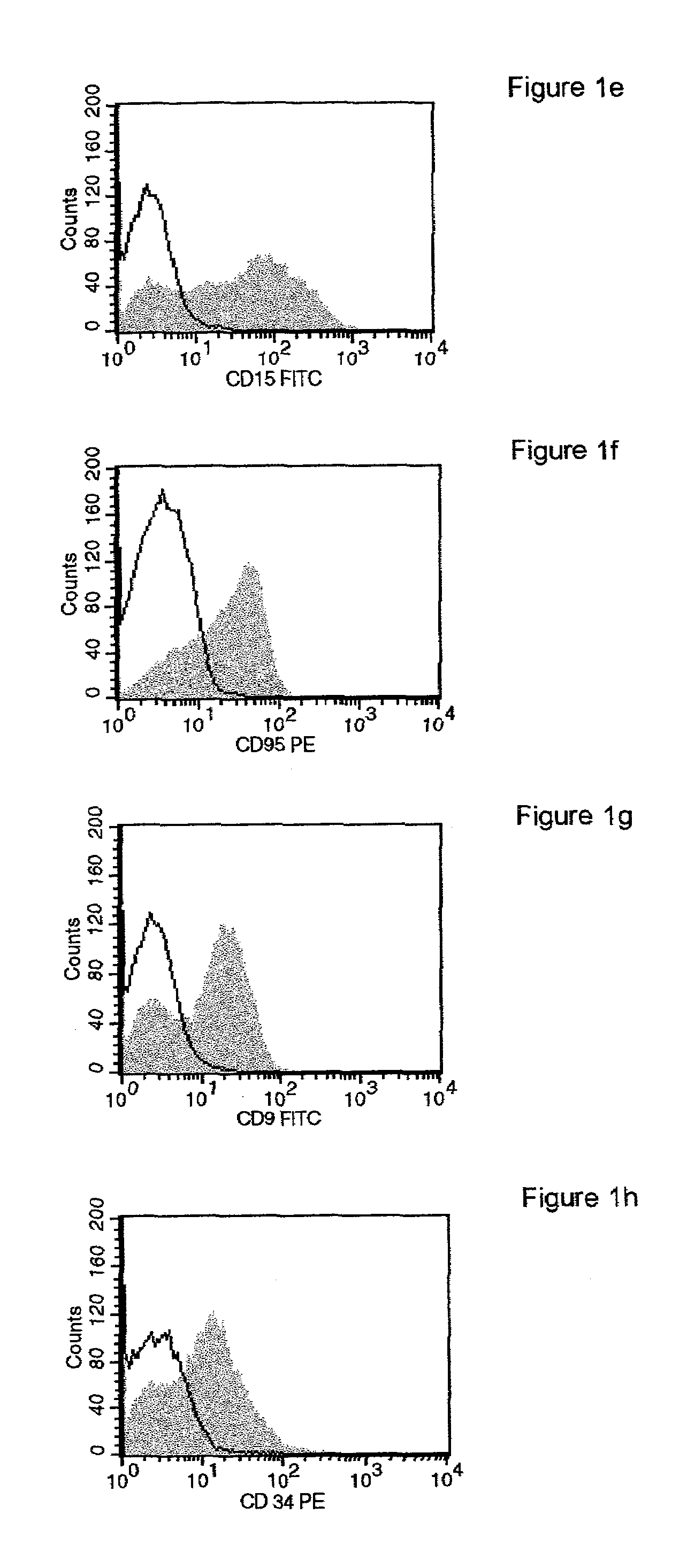
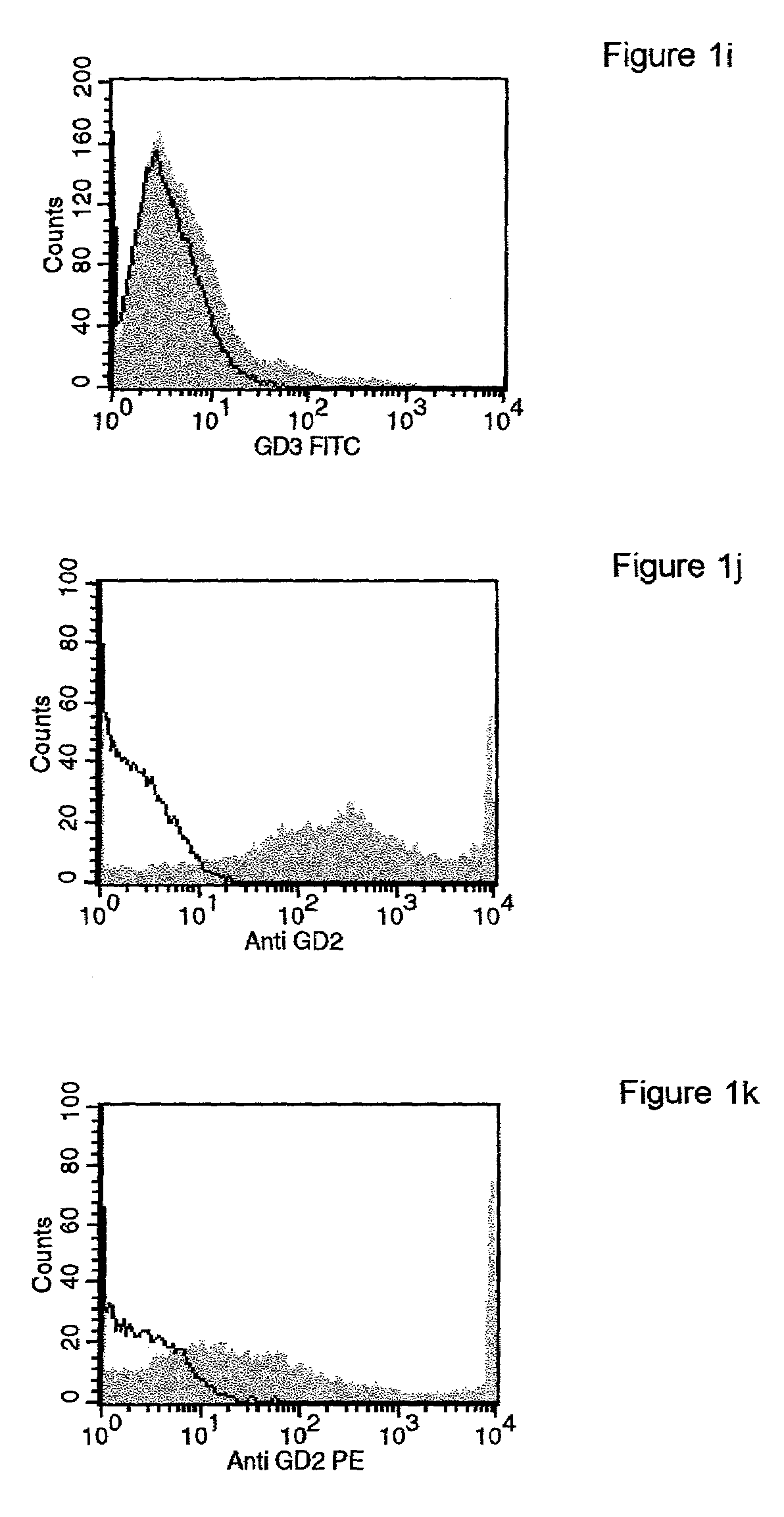
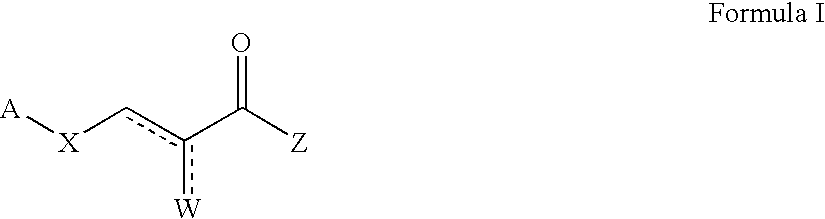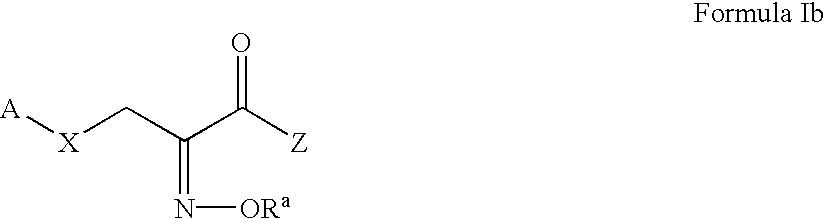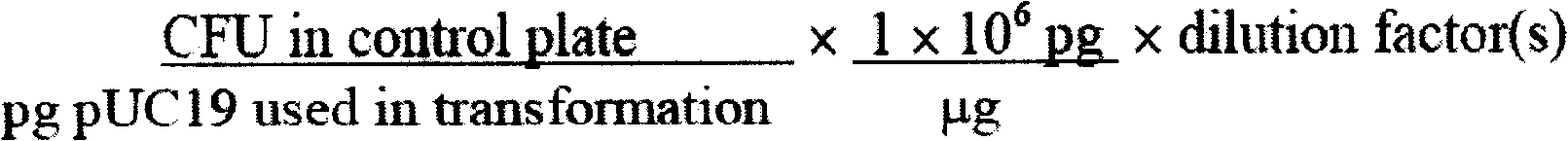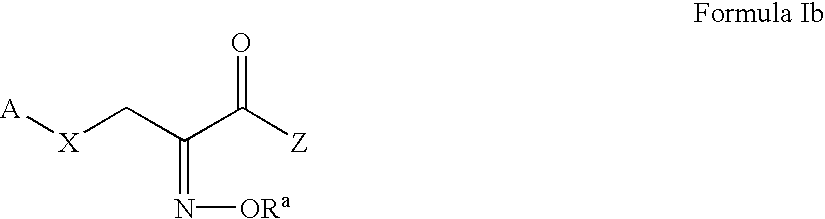Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
909 results about "Competent cell" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Competent cell. [¦käm·pəd·ənt ′sel] (cell and molecular biology) A cell that is able to incorporate exogenous deoxyribonucleic acid and undergo genetic transformation.
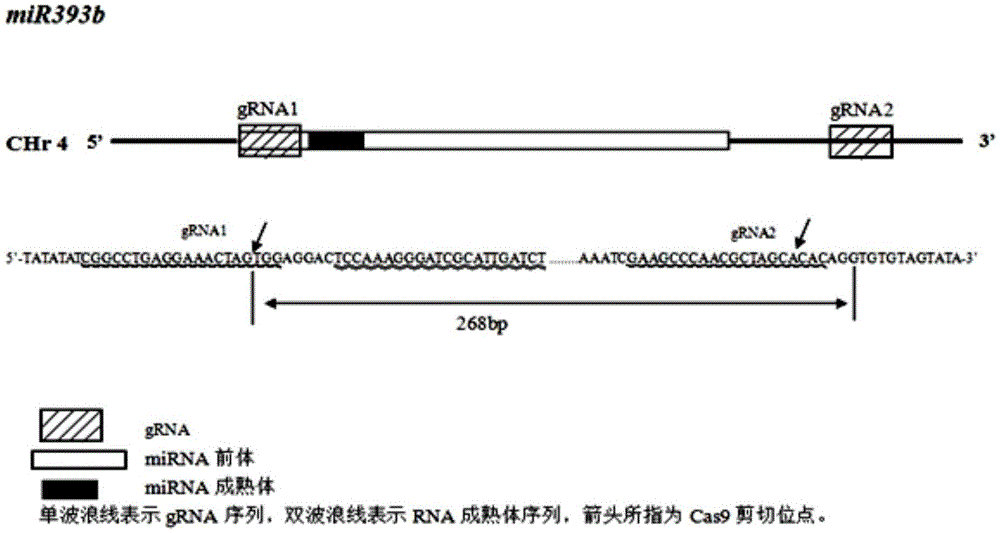
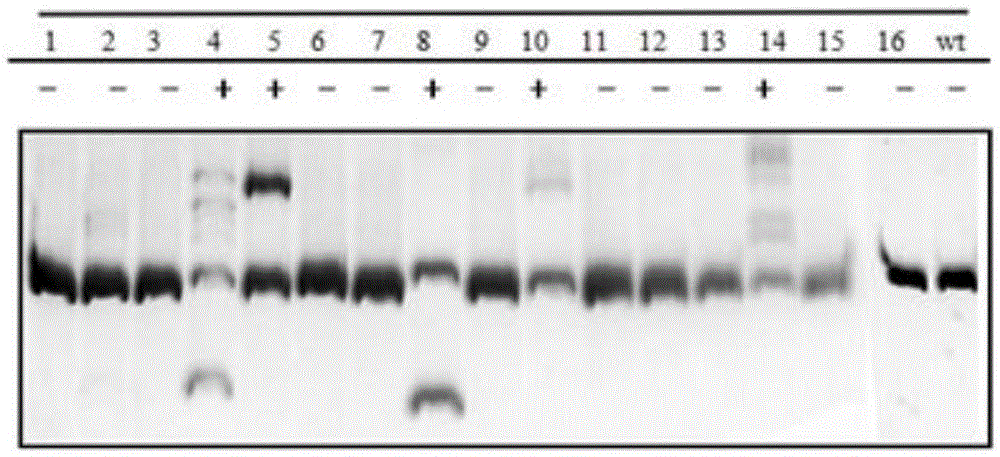
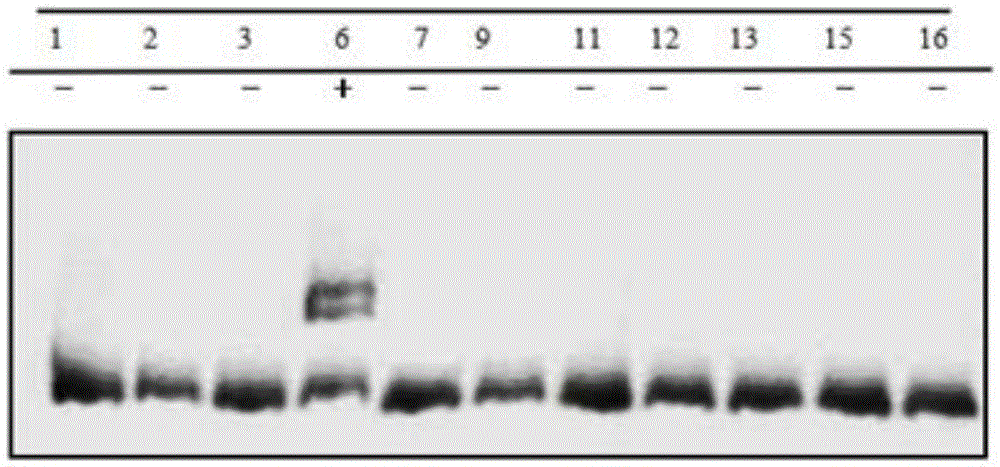
Gene editing method for knocking out rice MIRNA393b stem-loop sequences with application of CRISPR(clustered regulatory interspersed short palindromic repeat)-Cas9 system
InactiveCN105647962AKnockout worksNucleic acid vectorVector-based foreign material introductionEscherichia coliEnzyme digestion
The invention relates to construction of rice transgenic materials and aims to provide a gene editing method for knocking out rice MIRNA393b stem-loop sequences with application of a CRISPR(clustered regulatory interspersed short palindromic repeat)-Cas9 system. The gene editing method comprises steps as follows: gRNA target sites are selected for cloning and GG linking, enzyme digestion is performed after amplification, and a product is linked with a pGREB 32 vector; escherichia coli competent cells are transformed; plasmids with a correct sequencing result are used for transforming agrobacteria, transgenic plants are obtained through mediated transformation of rice calli, and transgenic positive lines are obtained; the T0-generation mutant plant seeds are collected for seeding, and the T1-generation plants are subjected to homozygote screening; homozygous lines which are discovered to be negative through MIRNA393b expression are rice mutants completely losing the MIRNA393b stem-loop sequences and MIRNA393b stem-loop sequence expression. According to the gene editing method, MIRNA stem-loop sequences can be effectively knocked out, and loss-of-function mutants of different members in the same MIRNA family can be prepared; the mutant plant propagates to obtain a large number of seeds and is an ideal material for acquiring rice MIRNA393b gene functions successfully.
Owner:ZHEJIANG UNIV
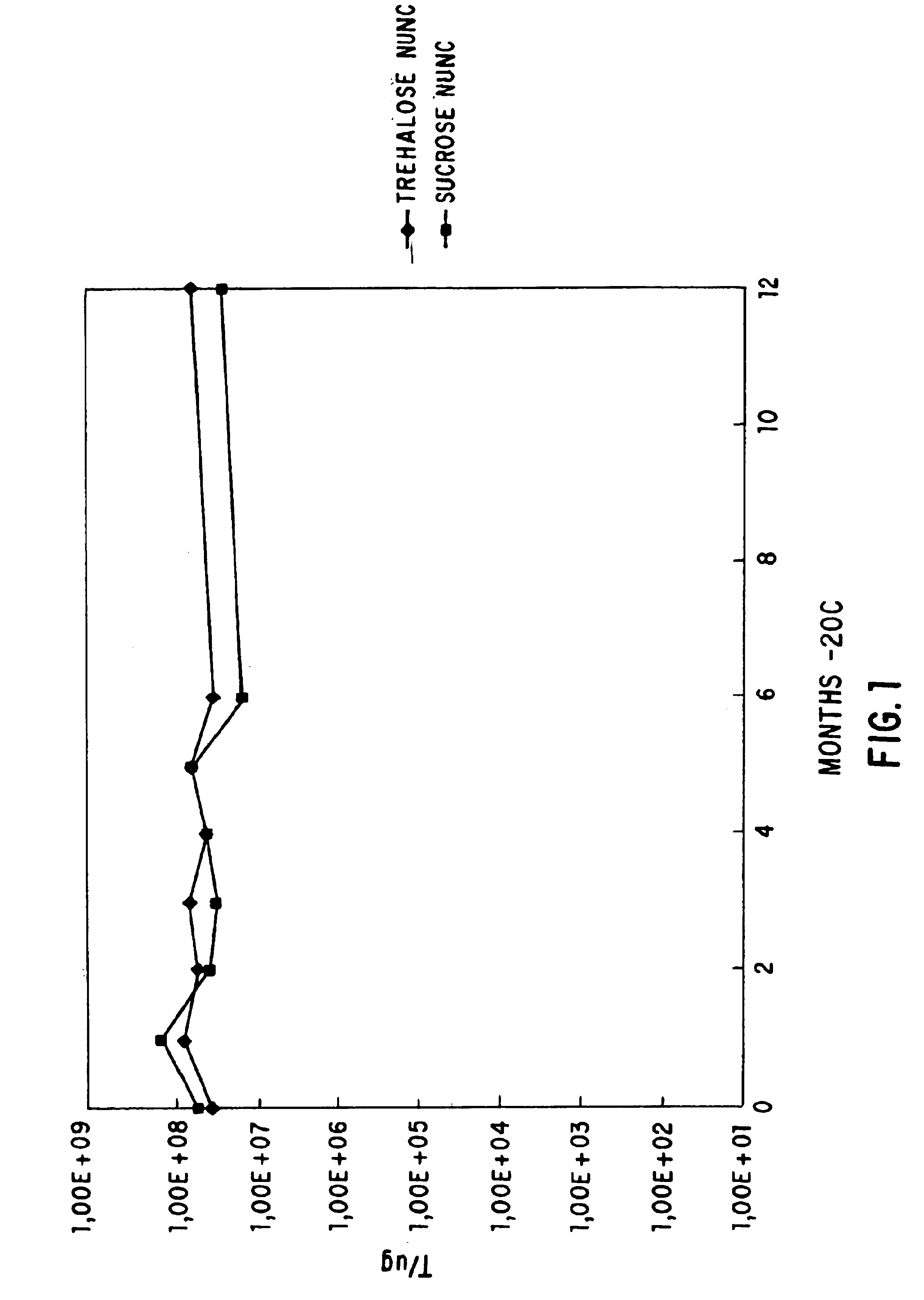
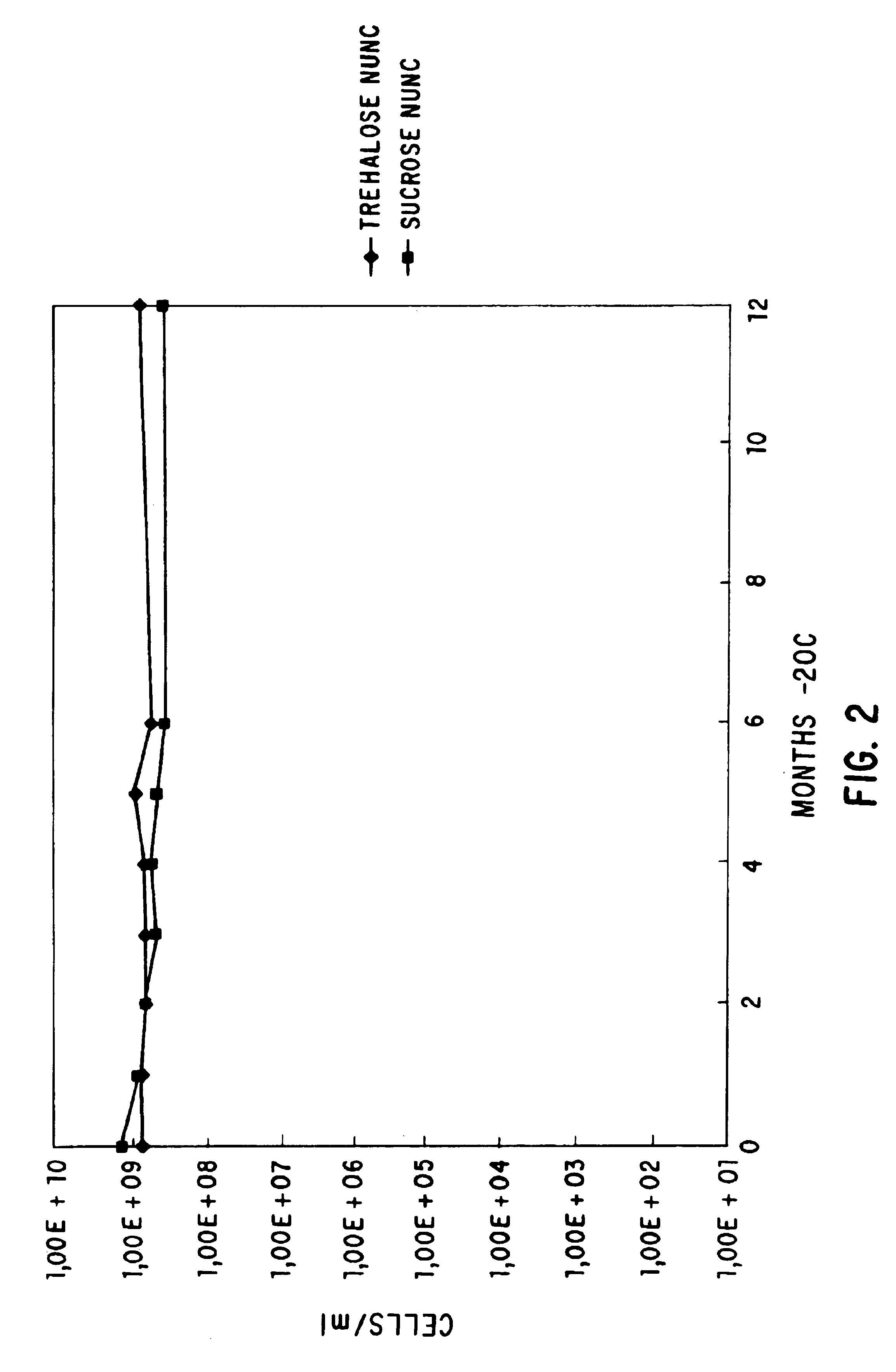
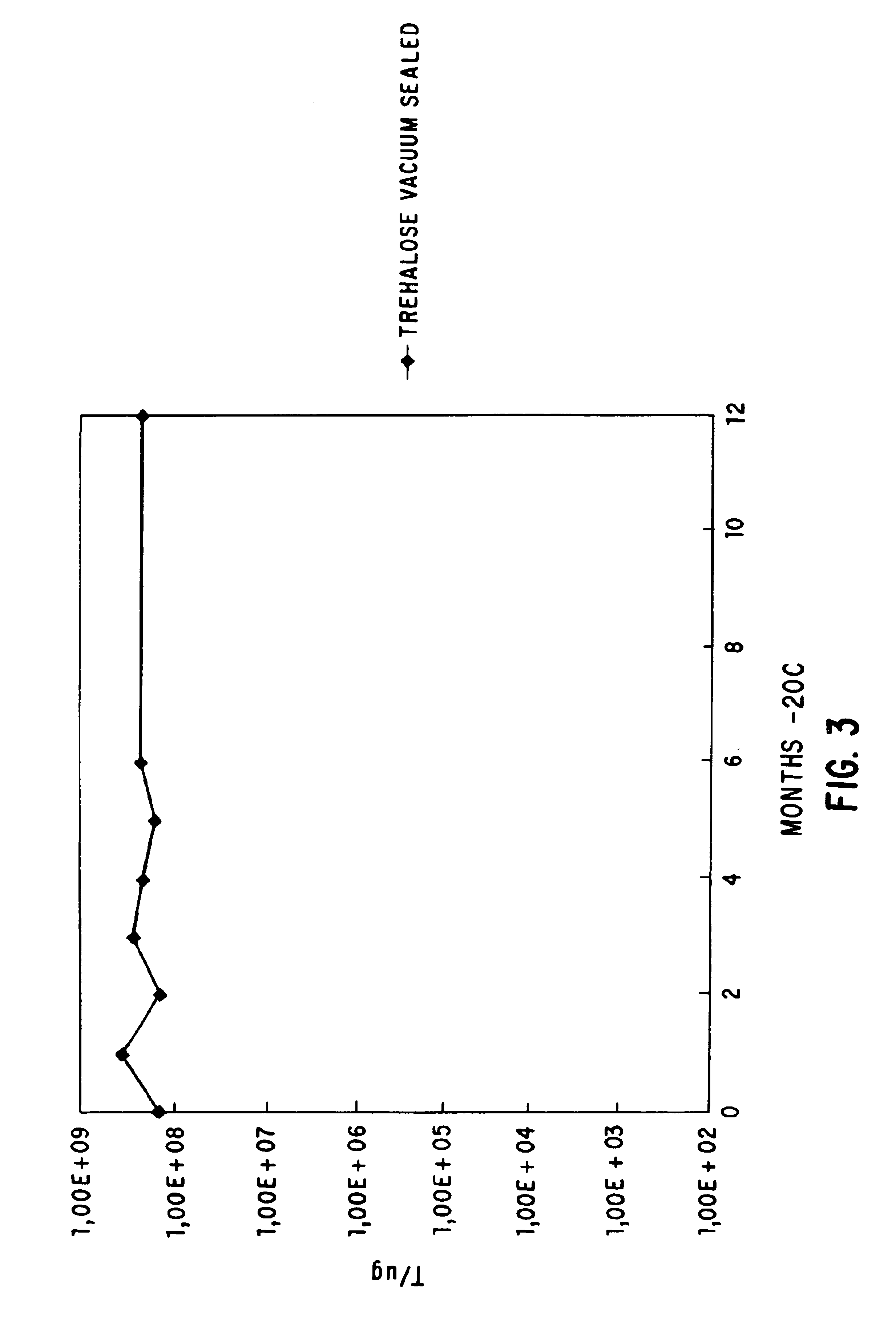
Methods for lyophilizing competent cells
This invention relates to a method for producing cells which are competent for transformation and which may be stably stored for extended periods of time at various temperatures. The method involves growing cells in a growth conducive medium, rendering said cells competent, and lyophilizing said competent cells. The invention further relates to competent cells produced by such a method, to methods of transforming said cells with a DNA molecule, and to a method of producing a desired protein or polypeptide from said transformed cells.
Owner:LIFE TECH CORP
Porcine circovirus II-type recombinant baculovirus as well as preparation method and application thereof
ActiveCN103122352AImprove expression levelHigh expressionGenetic material ingredientsAntiviralsEscherichia coliSpecific immunity
The invention discloses porcine circovirus II-type recombinant baculovirus as well as a preparation method and application thereof. ORF2 gene is artificially synthesized by referring to a PCV2b isolated strain ORF2 gene sequence; the synthesized ORF2 gene is connected to pFBDPHmHNM1P10eGFP plasmid by adopting the plasmid as a framework vector, so that a baculovirus transfer vector pFBDPHm 30RF2 is obtained. The baculovirus transfer vector pFBDPHm30RF2 is mixed with DH10Bac escherichia coli competent cells, and the positive bacterial colony is selected to obtain a recombinant rod granule rBac-PVR30RF2; the rod granule is transferred with a sf9 cell to obtain the recombinant baculovirus QP-Ac-30RF2. The recombinant baculovirus can be used for efficiently expressing the PCV20RF2 protein and forming virus-like particles. The VLP which is expressed and packaged by the recombinant baculovirus disclosed by the invention is used for preparing inactivated vaccine, and the organism is induced to generate specific immunity response after a 28-day-aged piglet is immunized, and the pig body can be completely protected from virulent attacks of the porcine circovirus.
Owner:HUAZHONG AGRI UNIV
Gene editing method using CRISPR/Cas9 system to create pink fruit tomato
InactiveCN107312795AOvercoming the problem of being unable to knock out genes in a targeted mannerGenetic stabilityHorticulture methodsPlant tissue cultureEscherichia coliCompetent cell
The invention relates to construction of a tomato transgenic material, and aims to provide a gene editing method using a CRISPR / Cas9 system to mutate a tomato SlMYB12 gene in order to transform red fruit tomato into pink fruit tomato. The method includes the following steps: designing an oligo primer corresponding to the gRNA target site of the SlMYB12 gene, and recombining and connecting an oligo dimer with a Cas9 / gRNA vector; transforming Escherichia Coli competent cells, transforming a plasmid with a correct sequencing result into Agrobacterium tumefaciens, and mediating and transforming tomato calluses to obtain a transgenic plant in order to obtain a transgenic positive strain; and carrying out gene sequencing and phenotype observation to determine a pure mutant strain which is an SlMYB12 function completely lost tomato mutant for pink fruits. The method can effectively knock out the transcriptional translation of the SlMYB12 gene, and is an ideal gene editing system for successfully transforming the tomato red fruits into the pink fruits.
Owner:ZHEJIANG ACADEMY OF AGRICULTURE SCIENCES
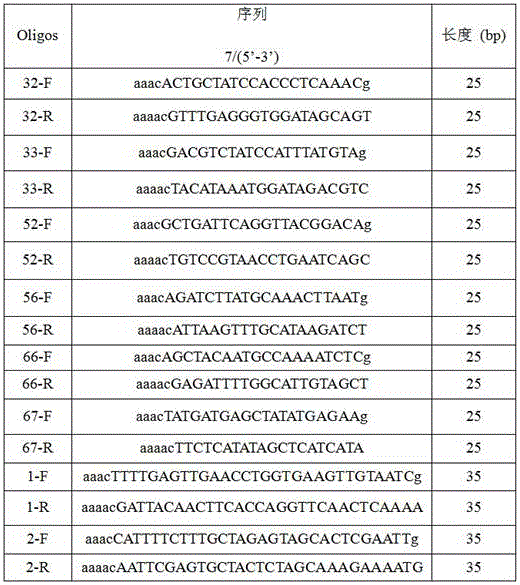
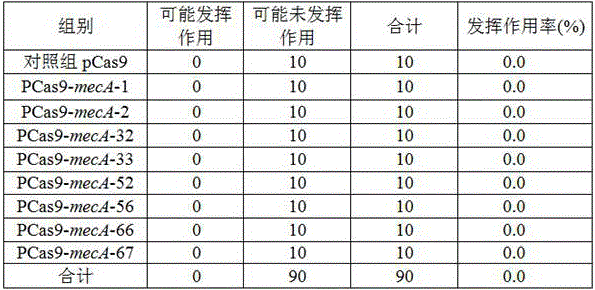
Method for eliminating mecA plasmids based on CRISPR/Cas9 technology
InactiveCN106167808AAvoid conversionEasy to operateNucleic acid vectorVector-based foreign material introductionBiotechnologyMethicillin resistance gene
The invention discloses a method for eliminating mecA plasmids based on CRISPR / Cas9 technology. The method comprises: selecting an MRSA strain to perform PCR amplification to a DNA sequence of a C terminal of mecA gene coding transpeptidase; carrying out gel extraction for mecA genes; connecting the mecA genes with a T-pMD19 (simple) carrier and preparing DH5[alpha] competent cells; electro-transforming the DH5[alpha] competent cells through the obtained T-pMD19-mecA plasmids; extracting the T-pMD19-mecA plasmids and performing sequencing and verification; performing double digestion treatment for the T-pMD19-mecA plasmids and performing gel extraction to the mecA genes; connecting pET-21a (+) plasmids with the mecA genes; designing and synthesizing oligos; constructing pCas9 :: mecA plasmids; electro-transforming the obtained pET-21a (+)-mecA plasmids into an escherichia coli expression strain BL21 (D3); and electro-transforming the pCas9 :: mecA plasmids and the pCas9 plasmids into BL21 (D3)+pET-21a(+)-mecA competent bacteria and BL21 (D3)+pET-21a(+) competent bacteria. The method is simple to operate and good in specificity, and can effectively block spread of mecA to eliminating the mecA strain.
Owner:ZHENGZHOU UNIV
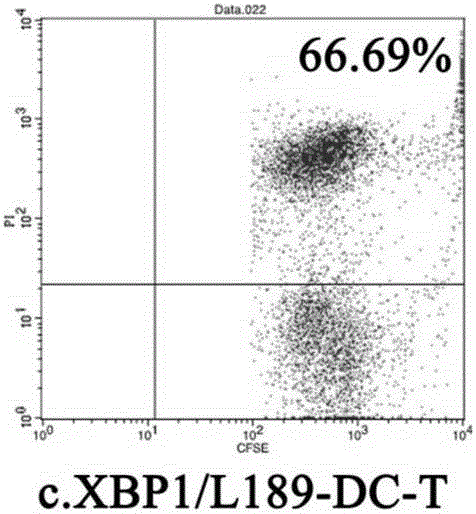
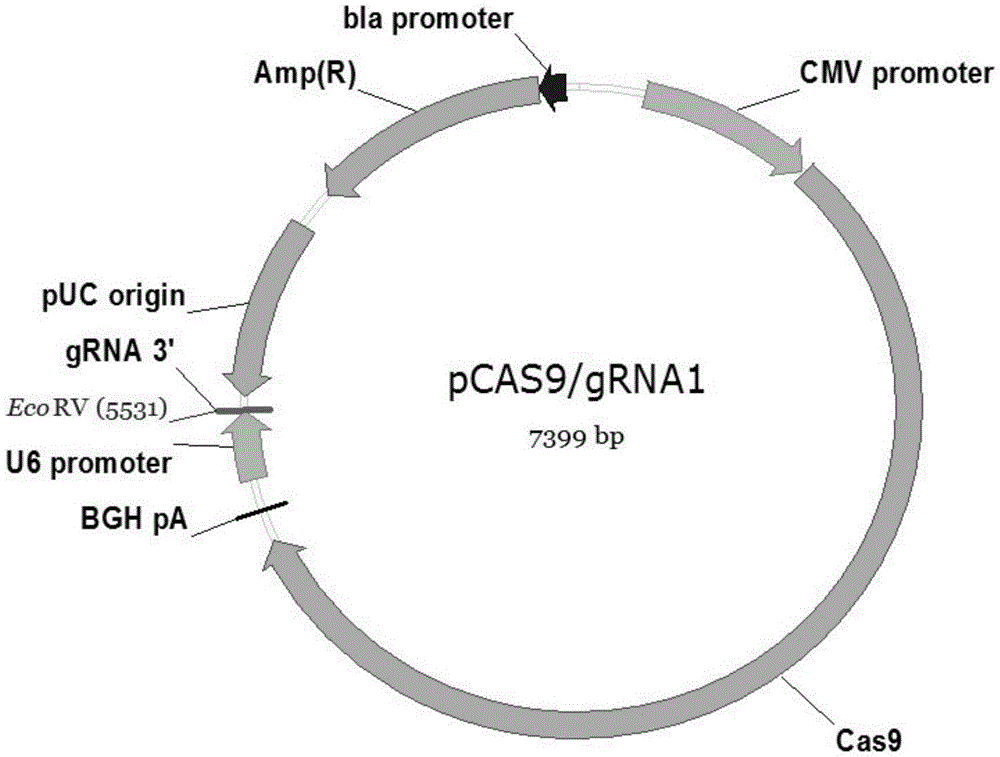
High-efficiency knockout method for XBP1 gene in DC cell
InactiveCN105602987AGuaranteed immune stimulationEffective anti-tumor immunityBlood/immune system cellsVector-based foreign material introductionCompetent cellXBP1
The invention provides a high-efficiency knockout method for the XBP1 gene in a DC cell. The high-efficiency knockout method for the XBP1 gene in the DC cell comprises the following steps: designing of a gene knockout target and oligonucleotide; annealing of oligonucleotide; enzyme digestion of a linearized vector; connection and reaction of the linearized vector with double-strand oligonucleotide; transformation of competent cells and knockout of the XBP1 gene in the DC cell; etc. According to the invention, the knockout method provided by the invention employs an improved CRISPR / pCas9 gene knockout system, uses the XBP1 as a target gene for designing of a CRISPR targeting sequence and preparation of pCas9 / gRNA1-XBP1 plasmid and further allows the plasmid to transfect the DC cell under the treatment action of an L189 drug; thus, the XBP1 gene in the DC cell can be effectively knocked out, the immunostimulation effect of the DC cells can be effectively guaranteed, and antineoplastic immunization effect of the DC cell is given to effective play.
Owner:SHENZHEN MORECELL BIOMEDICAL TECH DEV CO LTD
Process for producing freeze dried competent cells and use thereof in cloning
InactiveUS20020081565A1Improve efficiencyAvoid utilizationBacteriaUnicellular algaeCompetent cellFreeze-drying
A process for producing lyophilized competent cells wherein competent cells are that can be stored or shipped as freeze-dried cells at temperatures between 0° C. and 8° C. and remain suitable for cloning genes or DNA fragments. The process includes culturing cells, rending the cells competent, and lyophilizing the cells. Once lyophilized, the cells can be stored or shipped as freeze-dried cells. The lyophilized cells are prepared for transformation protocols by being re-hydrated in a solution of dimethyl sulfoxide. Once re-hydrated, the transformation efficiency of the competent cells is at least 5x105 transformations per microgram of DNA.
Owner:SIGMA ALDRICH CO LLC
Goat TLR4 gene knock-out vector and construction method thereof
InactiveCN106755097AThe method is simple and fastHigh knockout efficiencyNucleic acid vectorVector-based foreign material introductionEscherichia coliCompetent cell
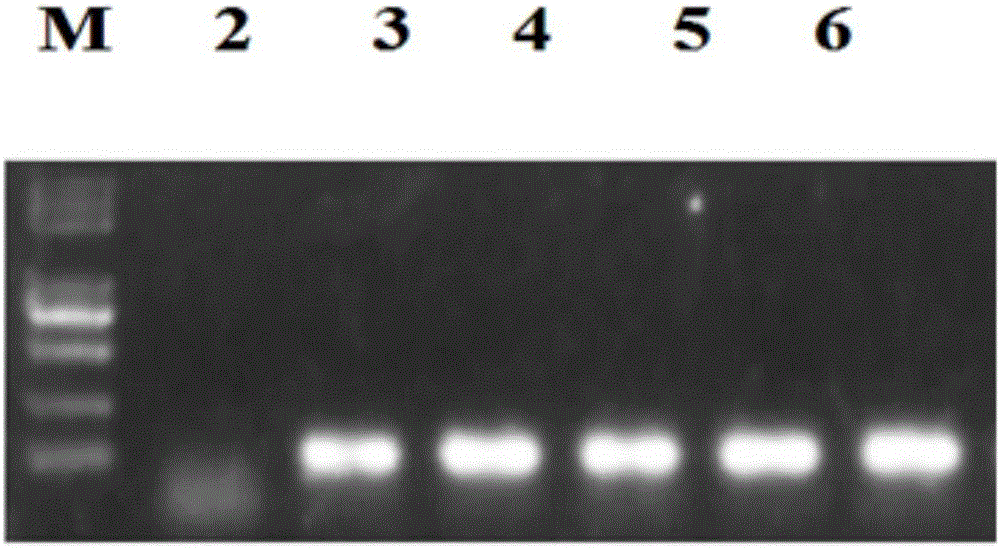
The invention discloses a goat TLR4 gene knock-out vector and a construction method thereof. The construction method comprises the following steps: firstly, designing an sgRNA fragment of the TLR4 gene by adopting a CRISPR / cas9 system, synthesizing an sgRNA nucleotide sequence, constructing and simultaneously expressing the sgRNA and plasmid PYSY-sgRNA of Cas9 D10A, connecting and transforming to an Escherichia coli DH5 alpha competent cell, and verifying the transformant; and judging and proving by enzyme digestion and sequencing that the TLR4 gene knock-out vector is constructed correctly. The invention adopts the CRISPR / cas9 for constructing the vector, and provides a theoretical basis for subsequently acquiring a goat TLR4 gene deletion type alveolar epithelial cell system, and studying the immune response molecular mechanism of mycoplasma pneumonia infection.
Owner:INST OF ANIMAL HUSBANDRY & VETERINARY MEDICINE ANHUI ACAD OF AGRI SCI
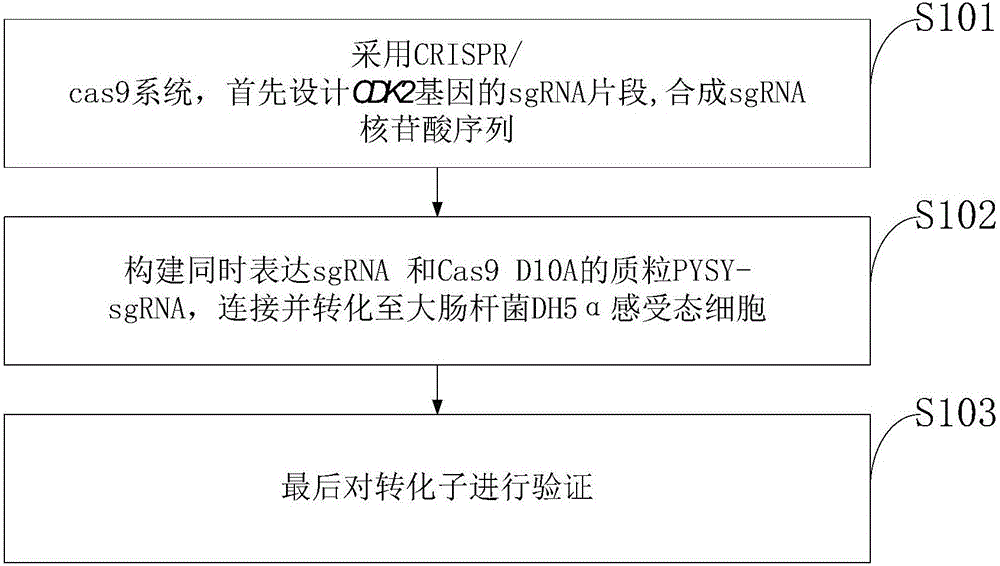
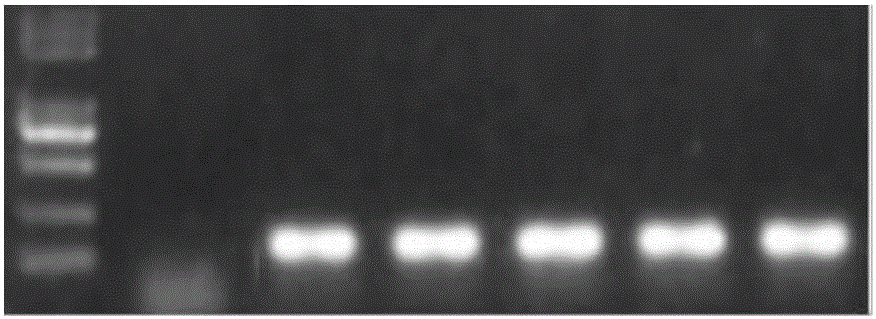
Goat CDK2 (Cyclin-dependent kinases 2) gene knockout vector and construction method thereof
InactiveCN106834347AThe method is simple and fastHigh knockout efficiencyNucleic acid vectorVector-based foreign material introductionEscherichia coliRestriction enzyme digestion
The invention discloses a goat CDK2 (Cyclin-dependent kinases 2) gene knockout vector and a construction method thereof. A CRISPR / Cas9 system is adopted; an SgRNA segment of a CDK2 gene is designed at first and an SgRNA nucleotide sequence is synthesized; a plasmid PYSY-sgRNA for expressing SgRNA and Cas9D10A at the same time is constructed; the plasmid PYSY-sgRNA is connected and transformed to an escherichia coli DH5alpha competent cell; finally, a transformant is verified; restriction enzyme digestion and sequencing identification prove that the construction of the CDK2 gene knockout vector is accurate. The vector constructed by CRISPR / Cas9 is adopted, and a theoretical basis is provided for subsequently obtaining a goat CDK2 gene deletion type cell line and researching a molecular mechanism of cell apoptosis molecules triggered by mycoplasma pneumonia infection.
Owner:INST OF ANIMAL HUSBANDRY & VETERINARY MEDICINE ANHUI ACAD OF AGRI SCI
Methods for sequential replacement of targeted region by homologous recombination
InactiveUS20110119779A1Easily employedSimple procedureOther foreign material introduction processesFermentationEscherichia coliCompetent cell
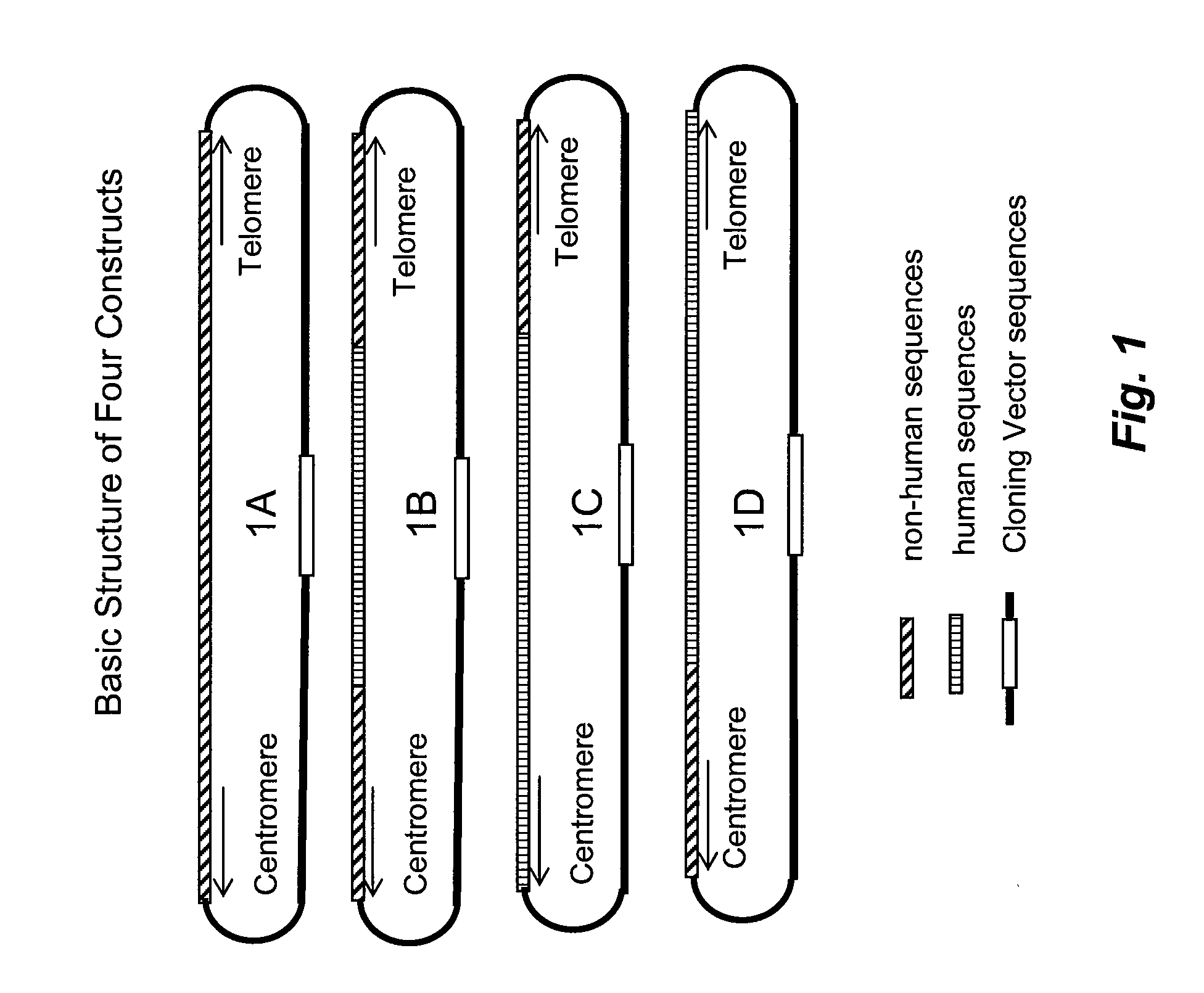
The invention provides methods and compositions for generating non-human transgenic cells and organisms that are transgenic at one or more gene sequences by separately recombining fragments of a complete gene in temporal sequence. According to the methods of the invention, a set of DNA constructs containing a non-endogenous DNA sequence flanked and / or operably linked at its ends by sequences from the non-human organism are generated by recombination in a bacterial cell, for example, in E. coli. The DNA constructs that are produced can then be introduced into a non-human homologous recombination competent cell where successive cells will contain recombined segments of a target gene, with the ultimate cell in a line containing an endogenous target gene completely replaced by genomic DNA of another species.
Owner:ABLEXIS LLC
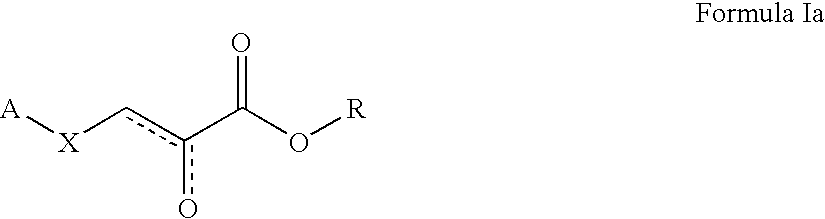
Pyruvate derivatives
Certain known and novel pyruvate derivatives are particularly active in restoring or preserving metabolic integrity in oxidatively competent cells that have been subjected to oxygen deprivation. These pyruvate-derived compounds include, but are not limited to oximes, amides, pyruvate analogues, modified pyruvate analogues, esters of pyruvate (e.g., polyol-pyruvate esters, pyruvate thioesters, glycerol-pyruvate esters and dihydroxyacetone-pyruvate esters). Such pyruvate derivatives (including single tautomers, single stereoisomers and mixtures of tautomers and / or stereoisomers, and the pharmaceutically acceptable salts thereof) are useful in the manufacture of pharmaceutical compositions for treating a number of conditions characterized by oxidative stress.
Owner:GALILEO PHARMA
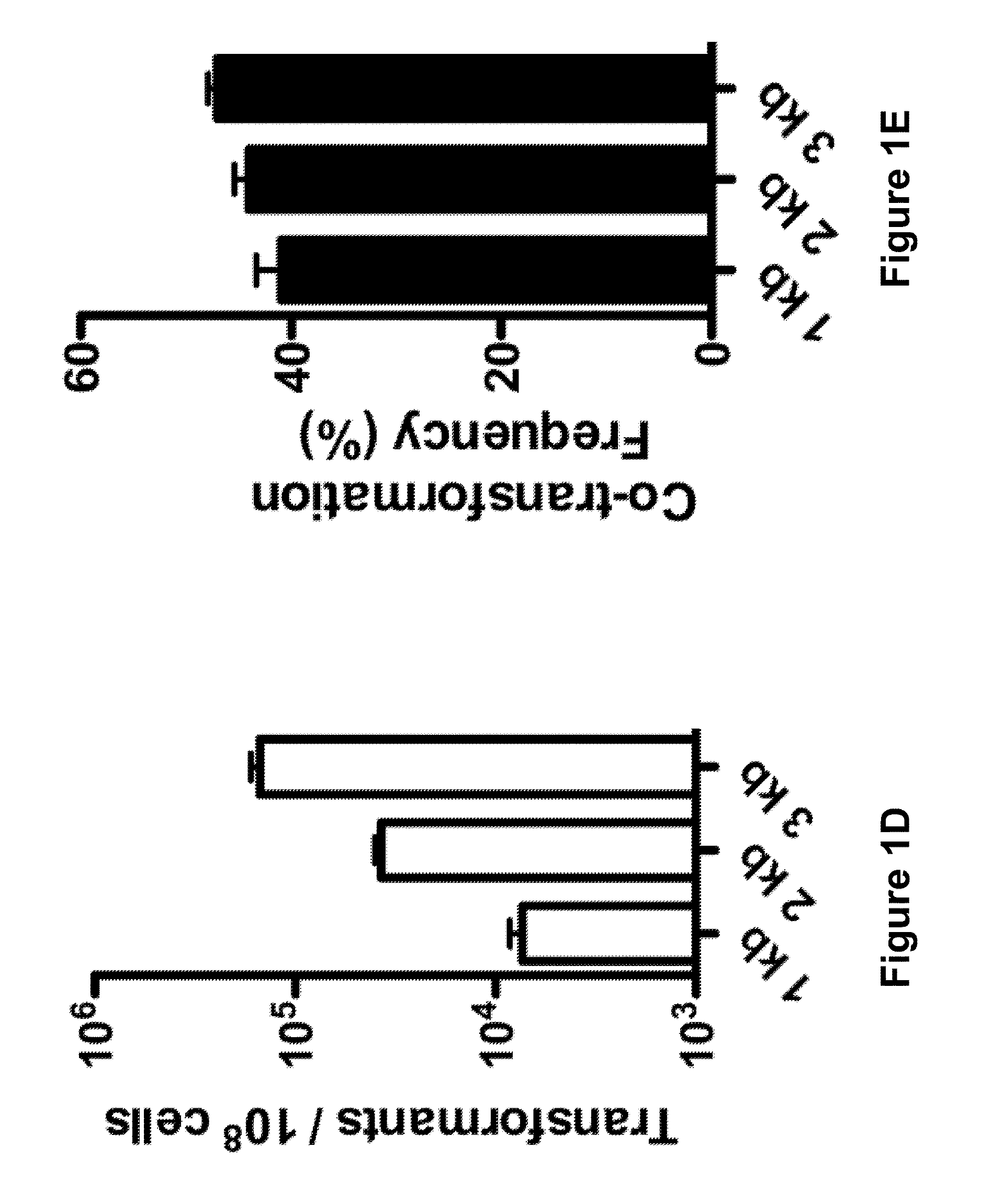
Methods and apparatus for transformation of naturally competent cells
The present invention includes compositions and methods of co-transformation of naturally competent cells. In one aspect of the invention, a method is included for introducing nucleic acid sequences into one or more naturally competent cells in parallel. In other aspects, a heterogenic pool of co-transformed naturally competent cells and an apparatus for introducing two or more populations of nucleic acid sequences into a population of naturally competent cells in parallel are also included.
Owner:TUFTS UNIV
Porcine O-type foot-and-mouth disease virus recombinant baculovirus as well as preparation method and application thereof
InactiveCN103122353AImprove expression levelHigh expressionGenetic material ingredientsAntiviralsEscherichia coliShuttle vector
The invention discloses porcine O-type foot-and-mouth disease virus recombinant baculovirus as well as a preparation method and application thereof. Sequences of VP0, VP1 and VP3 genes are artificially synthesized by referring to an FMDV (Foot And Mouth Disease Virus) O-type epidemic strain gene sequence; the VP0, VP1 and VP3 genes are connected to pFBDPHmHNM1P10eGFP plasmid by adopting the plasmid as a framework vector, so that a baculovirus transfer vector pFBDPHmVP013 is obtained. The baculovirus transfer vector pFBDPHmVP013 is mixed with DH10Bac escherichia coli competent cells, and the positive bacterial colony is selected to obtain a recombinant shuttle vector Bacmid; the shuttle vetcor Bacmid is transferred with a sf9 cell, and the recombinant baculovirus QP-Ac-FVLP is obtained by collecting the cell supernatant. The recombinant baculovirus can be used for efficiently expressing FMDVVP0, Vp1 and Vp3 proteins and forming virus-like particles. And the virus-like particles are used for preparing subunit vaccine, so that the organism is induced to generate specific immunity response after the mouse is immunized.
Owner:HUAZHONG AGRI UNIV
Method for suppressing multiple drug resistance in cancer cells
Methods for treating and preventing the onset and maintainance of multiple drug resistance (MDR) in animals undergoing chemotherapy for cancer are provided. According to the methods, target cells are depleted of adenosine 5'-monophosphate (AMP) and adenosine 5'-triphosphate (ATP) such that the cells are unable to support P-glycoprotein activity. According to one method, a population of target cells is obtained from a host and assayed for loss of methylthioadenosine phosphorylase (MTAse) activity. MTAse catabolizes methylthioadenosine to adenine for endogenous salvage incorporation into the intracellular AMP pool. MTAse deficient cells are treated with a purine synthesis inhibitor, such as L-alanosine, which starves the cells of adenine and suppresses P-glycoprotein activity. MTAse competent cells are also treated for MDR with purine synthesis inhibitors. In conjunction with treatment according to the invention, MTAse competent and deficient cells are also treated for malignancy with other anti-cancer drugs. A method for protecting non-malignant cells from adenine starvation during treatment of malignant cells according to the invention is provided.
Owner:RGT UNIV OF CALIFORNIA
Natural nanometer antibody library for Bactrian camel phage display as well as construction method and usage thereof
InactiveCN104404630ASolve problems that take a long timeSolve the inability to construct the corresponding phage display libraryPeptide librariesImmunoglobulins against hormonesAntigenCompetent cell
The invention discloses a natural nanometer antibody library for Bactrian camel phage display. The natural nanometer antibody library is prepared from the following steps: sampling Bactrian camel blood and spleen which are not immunized with any antigen, extracting total mRNA, reversely transcribing into cDNA, and amplifying VHH by nested PCR (Polymerase Chain Reaction); carrying out enzyme digestion on a pMECS phage display vector and the VHH by restrictive incision enzymes Pst I and Not I and connecting two fragments; electrically transforming a connected product into a competent cell TG1. The invention further discloses a construction method and application thereof in the sieving of PCT and NAGL nanometer antibodies. The constructed natural phage display library can obtain the nanometer antibodies with specificity and a detection function through sieving, and can solve the problem that as a camel cannot be immunized due to antigen factors, the corresponding phage display library cannot be constructed, and accordingly, the nanometer antibodies cannot be obtained. Meanwhile, the library can also solve the problem that the immunization of the camel spends long time.
Owner:SOUTHEAST UNIV
A kind of beverage with anti-oxidation and immunity enhancement and preparation method thereof
The invention relates to a beverage with functions of antioxidation and immunity enhancing, and a preparation method thereof. The beverage is characterized in that: the beverage is prepared from the following raw materials: 1-3 kg of Chinese caterpillar fungus; 3-8 kg of medlar; 3-8 kg of mongolian milkvetch root; 2-6 kg of siberian solomonseal rhizome; 2-6 kg of mythic fungus; 1.5-3.5 kg of panas quinquefolium; 100-150 mg of a grape seed extract and 45-70 g of stevioside; purified water is added to the raw materials to prepare 650 L of the beverage. According to the present invention, the combination of the raw materials used in the beverage has good synergistic reaction; the raw materials collectively provide the effects of vital energy benefiting and blood nourishing, yang reinforcingand body fluid regeneration; the beverage can act on a plurality of immune competent cells, and has functions of antioxidation and immunity enhancing; the beverage can replace drugs so as to prevent disease, such that the bitterless beverage capable of prevention of the disease is provided; the beverage is easy to be drunken, and easy to be accepted by the people.
Owner:北京卓创首机通讯科技有限公司
Pyruvate derivatives
Certain known and novel pyruvate derivatives are particularly active in restoring or preserving metabolic integrity in oxidatively competent cells that have been subjected to oxygen deprivation. These pyruvate-derived compounds include, but are not limited to oximes, amides, pyruvate analogues, modified pyruvate analogues, esters of pyruvate (e.g., polyol-pyruvate esters, pyruvate thioesters, glycerol-pyruvate esters and dihydroxyacetone-pyruvate esters). Such pyruvate derivatives (including single tautomers, single stereoisomers and mixtures of tautomers and / or stereoisomers, and the pharmaceutically acceptable salts thereof) are useful in the manufacture of pharmaceutical compositions for treating a number of conditions characterized by oxidative stress.
Owner:MONSANTO TECH LLC
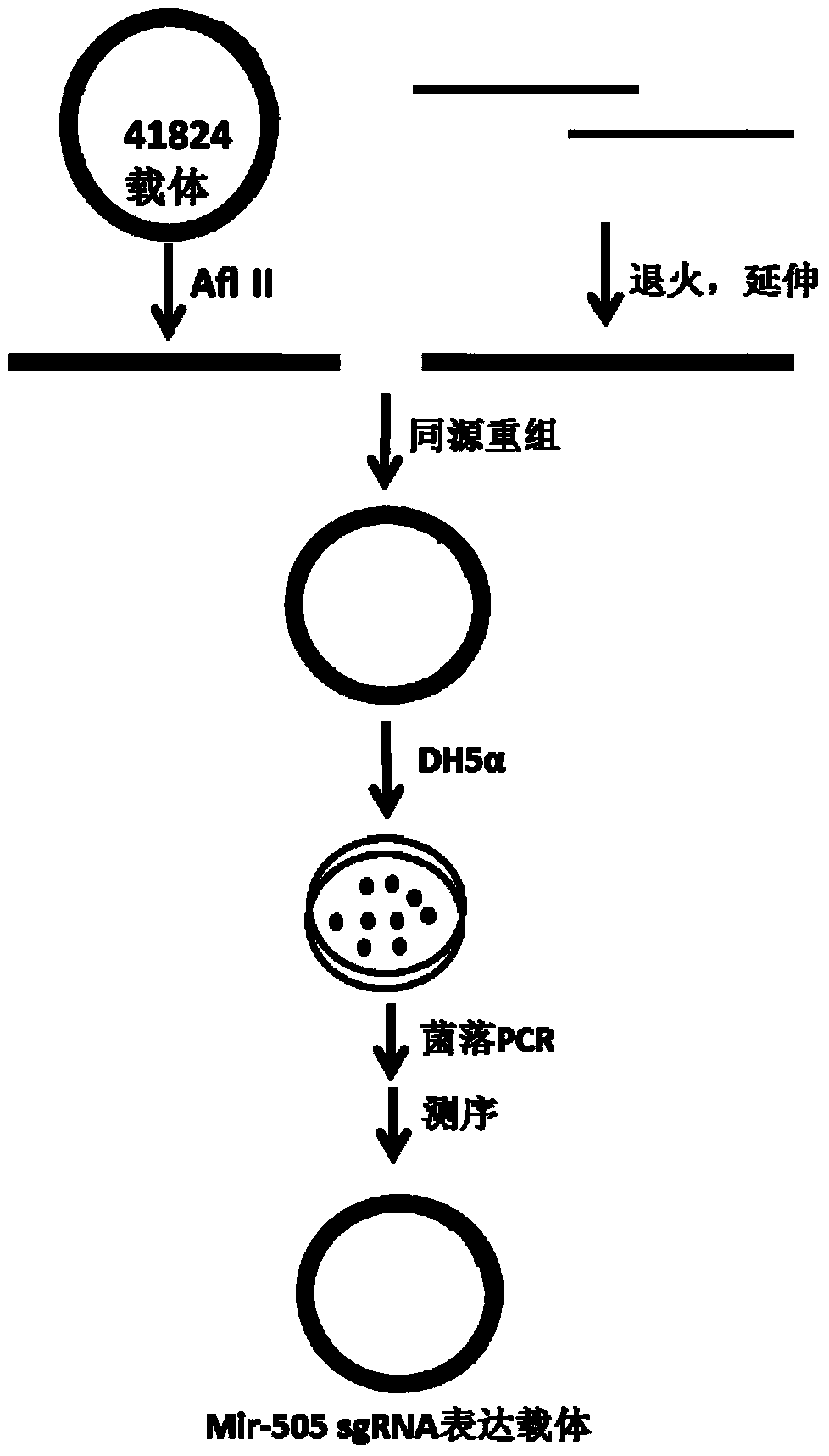
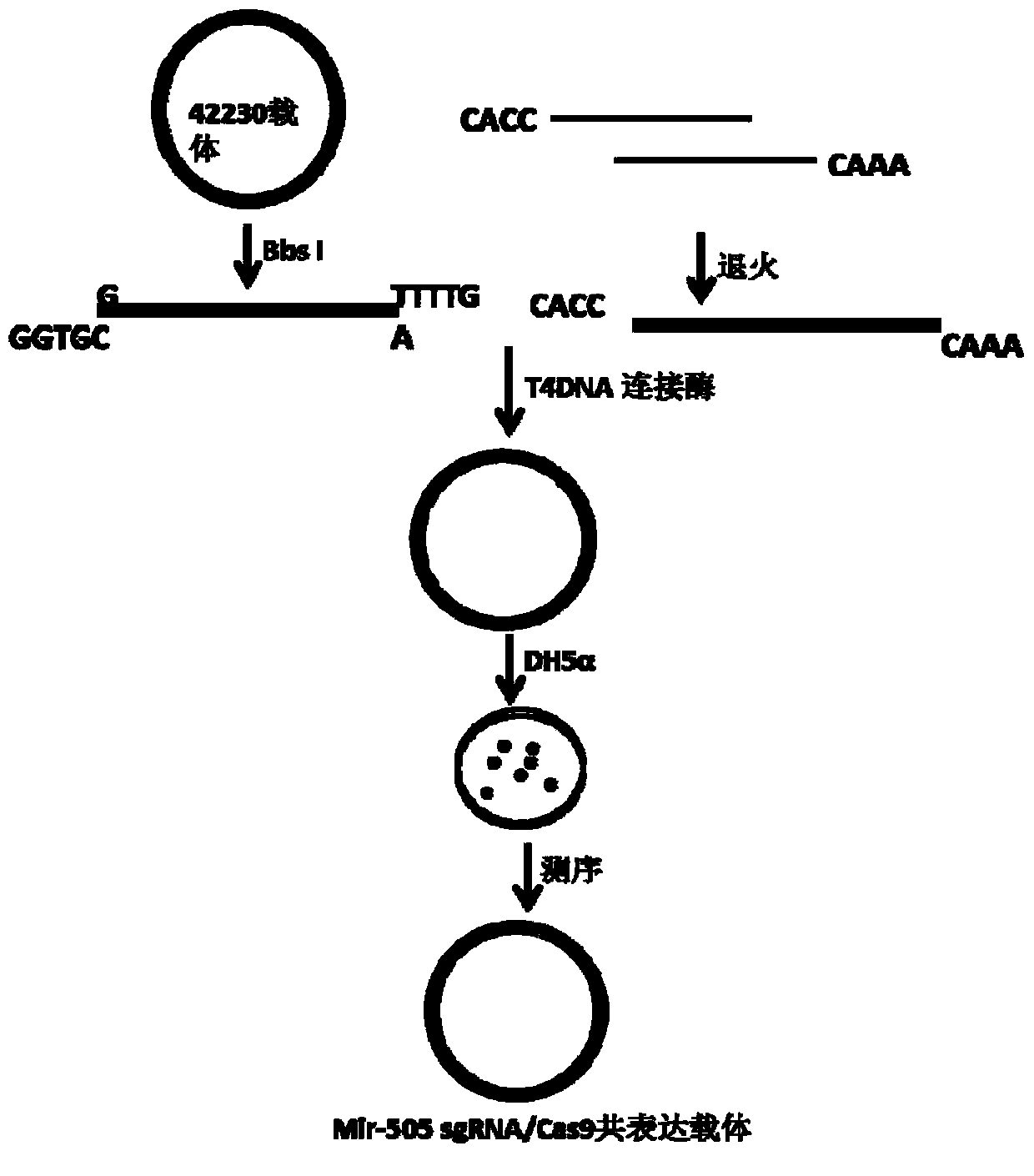
Method for knocking out mir-505 from mammal cell line
InactiveCN104212836AReduced shear efficiencyImprove shear efficiencyVector-based foreign material introductionEscherichia coliEnzyme digestion
The invention relates to a method for knocking out mir-505 from a mammal cell line, which comprises the following steps: synthesizing an sgRNA nucleotide single chain in vitro, treating to obtain an insertion element, inserting the sgRNA into a 41824 vector or 42230 vector by homologous recombination or T4 connection, transforming into competent cells of Escherichia coli, carrying out bacterial colony PCR (polymerase chain reaction) detection and sequencing confirmation on the vector to obtain an expression vector, denaturing the PCR product, annealing to form a heterologous hybrid double chain, and determining the shear efficiency of the CRISPR-Cas9 system on the mir-505 gene by using a T7E1 enzyme digestion test. The method provides references for selecting the sgRNA expression vector.
Owner:DONGHUA UNIV
Production technology for preparing freeze-drying genetic engineering bacterium competence cell and protective agent formula
InactiveCN101264062AReduce moisture contentNot prone to oxidationPowder deliveryGenetic material ingredientsBiotechnology researchFreeze-drying
The invention discloses a manufacturing technique for preparing frozen-out gene engineering bacteria competent cells. The invention also relates to a prescription of protecting agent, which is wide in application by taking as the host bacteria for transferring outer DNA in researching, developing, producing and checking of biotechnology. The protecting agent is one of the most fundamental matching consumption preparations. The invention utilizes vacuum freeze drying technology to deal with the poor gene engineering bacteria competent cells, and can save the cells steadily at a wide temperature range of 20 DEG C below zero to 4 DEG C for long time while keeping high transformation efficiency. The invention enables the cells to store and long-distance transport conveniently. The invention relates to a manufacturing technique for preparing frozen-out competent cells, quality inspection regulation and the frozen-out protecting agent, comprising culture conditions of gene engineering bacteria, technological processes of freezing and drying and the composition and matching of the protecting agent. The protecting agent is formed by water and one or arbitrary combination of the following materials: gelatin, degreasing milk, dextran, trehalose, sucrose, sorbitol or mannitol.
Owner:袁红杰 +2
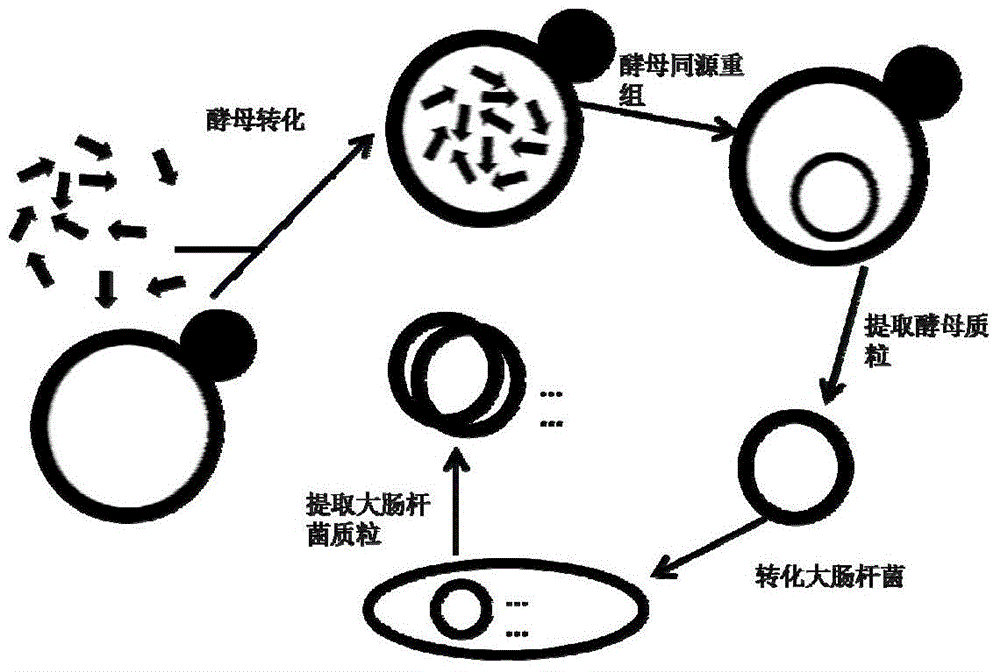
Rapid assembling method of multi-fragment DNA yeast
ActiveCN104419701AHigh speedIncrease success rateFermentationVector-based foreign material introductionEscherichia coliBiotechnology
The invention discloses a rapid assembling method of multi-fragment DNA yeast. The rapid assembling method comprises the following steps: (1) performing co-transformation on a plurality of DNA molecules with homologous arms, and linearized yeast shuttle vectors, so as to obtain yeast; (2) eluting all the transformed yeast colonies on a whole screening culture plate, centrifuging, and discarding the supernate to obtain transformed yeast cells; (3) extracting plasmid DNA of the yeast cells obtained in step (2), transforming the competent cells of colon bacillus; and (4) screening colon bacillus, and cloning to obtain large-fragment DNA. The method for assembling large-fragment DNA molecules is fast in speed, simple, convenient and feasible, high in success rate, low in cost, high in efficiency, easy to operate, beneficial in enlarging the industrialization scale and wide in application, and a plurality of small-fragment DNA molecules can be assembled into one large-fragment DNA molecule.
Owner:TIANJIN UNIV
Pyruvate derivatives
Certain known and novel pyruvate derivatives are particularly active in restoring or preserving metabolic integrity in oxidatively competent cells that have been subjected to oxygen deprivation. These pyruvate-derived compounds include, but are not limited to oximes, amides, pyruvate analogues, modified pyruvate analogues, esters of pyruvate (e.g., polyol-pyruvate esters, pyruvate thioesters, glycerol-pyruvate esters and dihydroxyacetone-pyruvate esters). Such pyruvate derivatives (including single tautomers, single stereoisomers and mixtures of tautomers and / or stereoisomers, and the pharmaceutically acceptable salts thereof) are useful in the manufacture of pharmaceutical compositions for treating a number of conditions characterized by oxidative stress.
Owner:GALILEO PHARMA
SARS vaccine and its preparation method
InactiveCN101007168AImprove securityTo achieve the purpose of surface displayAntiviralsRespiratory disorderSurface displayCompetent cell
The invention discloses a SARS vaccine and preparing method, which comprises the following steps: 1) constructing external baculoviral surface display carrier of S protein of SARS coronary virus; 2) transmitting the carrier into susceptive cell with baculoviral genome plasmid Bacmid; obtaining recombinant baculoviral genome plasmid; 3) using the plasmid to infect insect cell; purifying the recombinant plasmid; obtaining the product.
Owner:PEKING UNIV
Fluorescence quantitative PCR (Polymerase Chain Reaction) detection method for porcine transmissible gastroenteritis virus gene S and primer thereof
InactiveCN102154516ALow costReduce false positivesMicrobiological testing/measurementMicroorganism based processesCompetent cellFluorescence
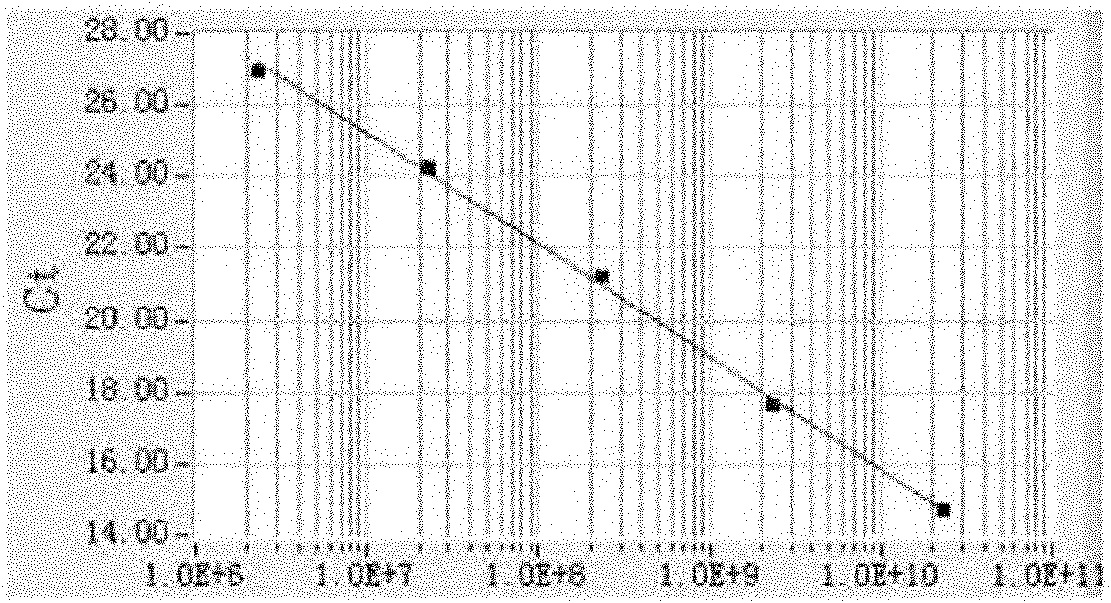
The invention discloses a fluorescence quantitative PCR (Polymerase Chain Reaction) detection method for a porcine transmissible gastroenteritis virus gene S and a primer thereof in the technical field of biotechnology. The method comprises the following steps of: cloning a PCR amplification target segment identified as a positive PCR product to a vector pMD18-T, transforming to a competent cell DH5alpha, selecting positive clone by screening blue and white spots and identifying sequencing; extracting a positive recombinant plasmid, quantifying by using an ultraviolet spectrophotometer, diluting a standard product series by 10 times of gradient until the final concentration is 1.0*10<3>-1.0*10<11> copies / mL, undergoing a fluorescence quantitative PCR by taking the standard product series as a template, and establishing a fluorescence quantitative PCR standard curve; and extracting virus RNA (Ribonucleic Acid) of a clinical excrement sample, undergoing a fluorescence quantitative PCR, and calculating the content of viruses in the sample according to a result and the standard curve, wherein the sequences of the primer are sequence 1 and sequence 2. The method and the primer have theadvantages that: a fluorescent probe does not need to be designed additionally, the cost is lowered, operation is easy and convenient, and detection can be completed within 2 hours. The detection method and the primer are suitable for any fluorescence quantitative PCR instrument, and can be applied to the detection of large-scale and high-flux samples.
Owner:SHANGHAI JIAO TONG UNIV
Isolation of neural stem cells using gangliosides and other surface markers
During the growth and study of NSCs, a range of molecules present on the surface of multipotent neural stem and progenitor cells (NSCs) were identified. These markers were identified using a number of human and murine neural stem cell lines, including retinal stem cells (RSCs). The NSC-specific markers identified included gene products as well as non-protein molecules and sugar epitopes not directly coded in the genome. Together with surface markers which were determined to be absent from the surface of hNSCs, the molecules described herein provide a means to enrich for neural stem cells, or neural progenitor subpopulations, particularly using combinatorial cell sorting strategies. These same molecules also represent targets for pharmacological manipulation of NSC populations and subpopulations, both in vivo and ex vivo. Furthermore, these molecules provide potential targets for therapeutic manipulation of other neural precursor-related cell types including malignant cell types as well as diseases originating from, or preferentially affecting, various uncommitted or replication-competent cell types.
Owner:CHILDRENS HOSPITAL OF ORANGE COUNTY +1
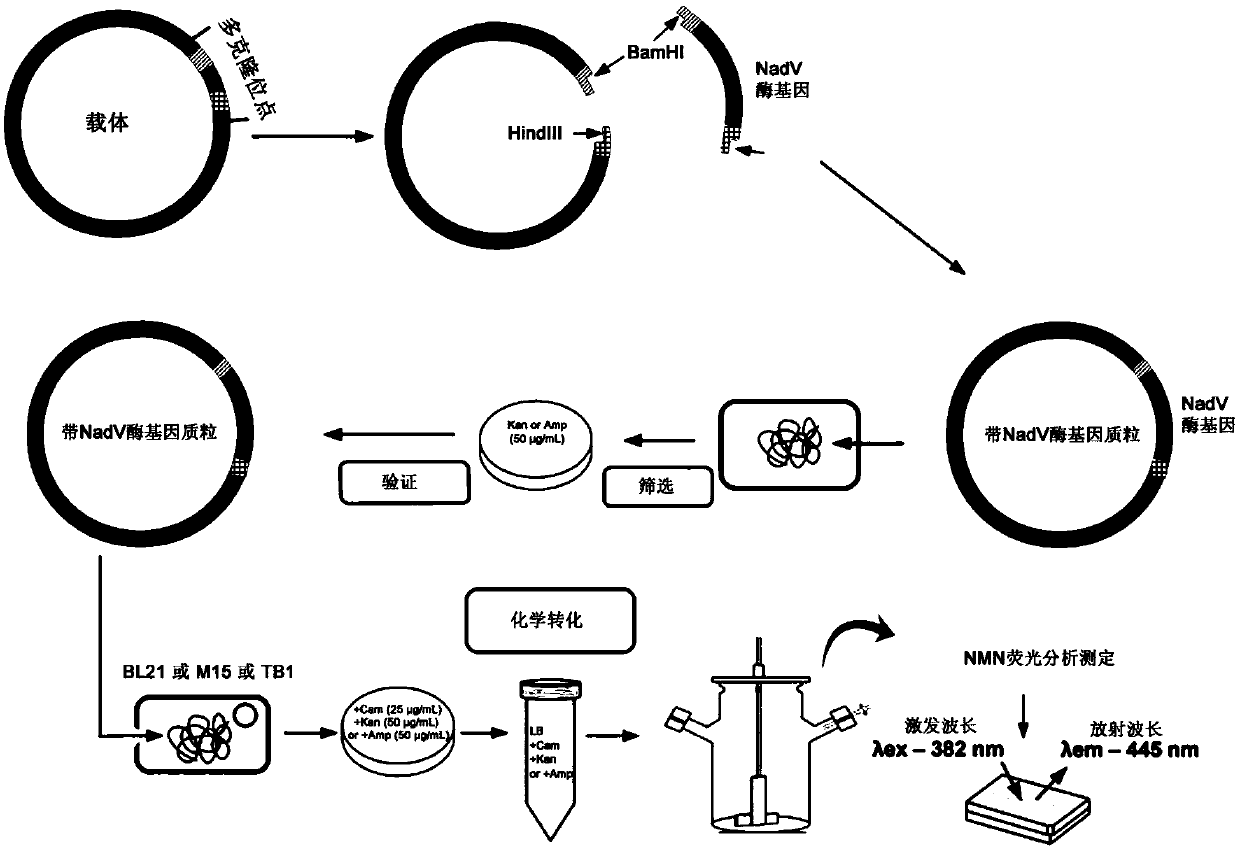
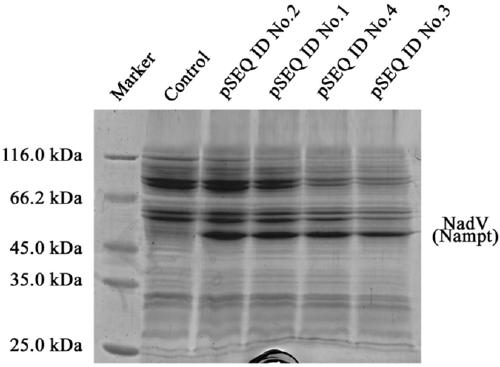
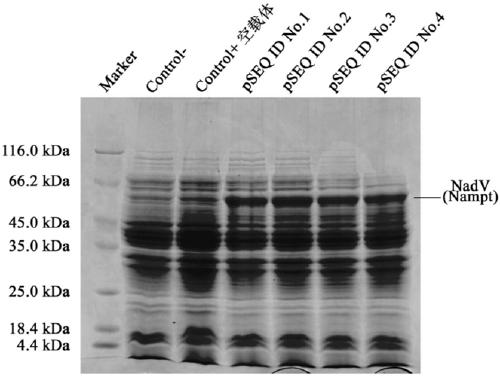
Niacinamide ribose phosphate transferase for preparing NMN, coding gene, recombinant vector and application
ActiveCN109666658ASimple processReduce investmentBacteriaMicroorganism based processesEscherichia coliNiacinamide
Owner:成都及禾生物科技有限公司
Pyruvate derivatives
Certain known and novel pyruvate derivatives are particularly active in restoring or preserving metabolic integrity in oxidatively competent cells that have been subjected to oxygen deprivation. These pyruvate-derived compounds include, but are not limited to oximes, amides, pyruvate analogues, modified pyruvate analogues, esters of pyruvate (e.g., polyol-pyruvate esters, pyruvate thioesters, glycerol-pyruvate esters and dihydroxyacetone-pyruvate esters). Such pyruvate derivatives (including single tautomers, single stereoisomers and mixtures of tautomers and / or stereoisomers, and the pharmaceutically acceptable salts thereof) are useful in the manufacture of pharmaceutical compositions for treating a number of conditions characterized by oxidative stress.
Owner:MONSANTO TECH LLC
Recombinant humanized III-type collagen and prokaryotic expression method thereof
ActiveCN111087463APromote migrationImprove adhesionConnective tissue peptidesBacteriaEscherichia coliNucleotide
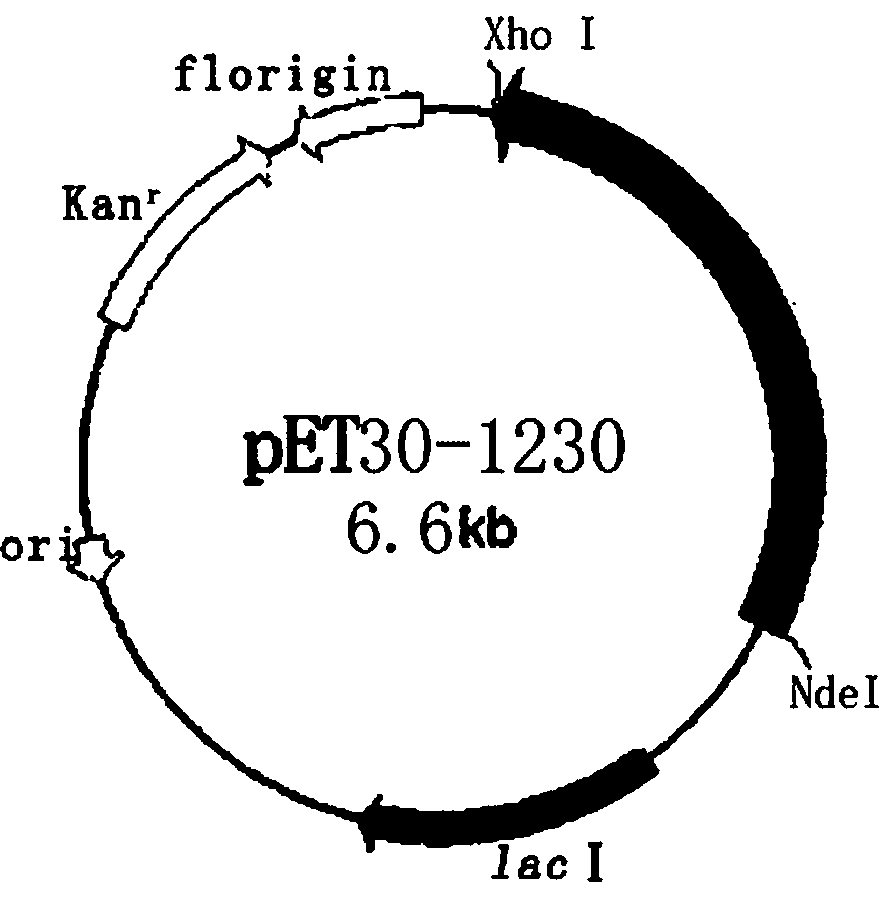
The invention relates to a recombinant human III-type collagen and a prokaryotic expression method thereof. The amino acid sequence of the humanized III-type collagen is shown as SEQ ID NO.1, and thenucleotide sequence of an encoding gene is shown as SEQ ID NO.2. The method comprises the following steps: carrying out codon optimization and design to obtain the gene sequences; inserting the gene sequences into a position between enzyme cutting sites NdeI and XhoI of an expression vector PET30a(+) to construct a recombinant expression vector pET30-1230; transforming Escherichia coli BL211(DE3)competent cells; selecting positive clones; and performing culture induction for efficient expression.
Owner:河北纳科生物科技有限公司 +1
Method for secretory expression of super-folded green fluorescent protein mediated heterologous protein in escherichia coli
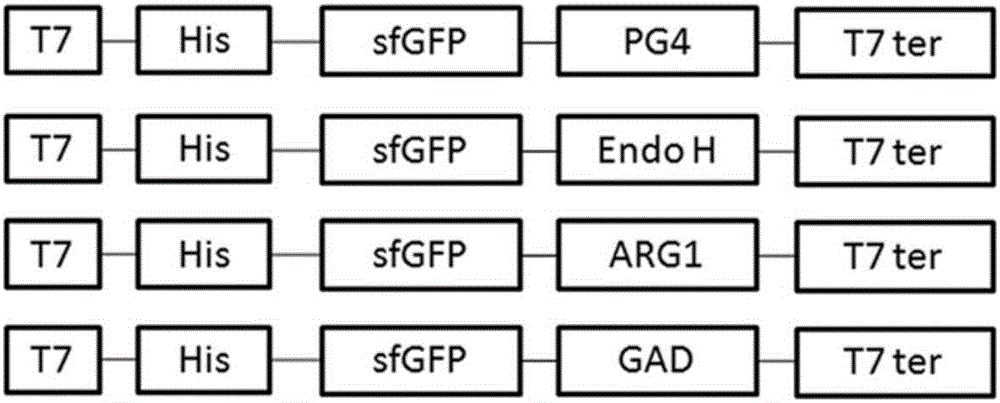
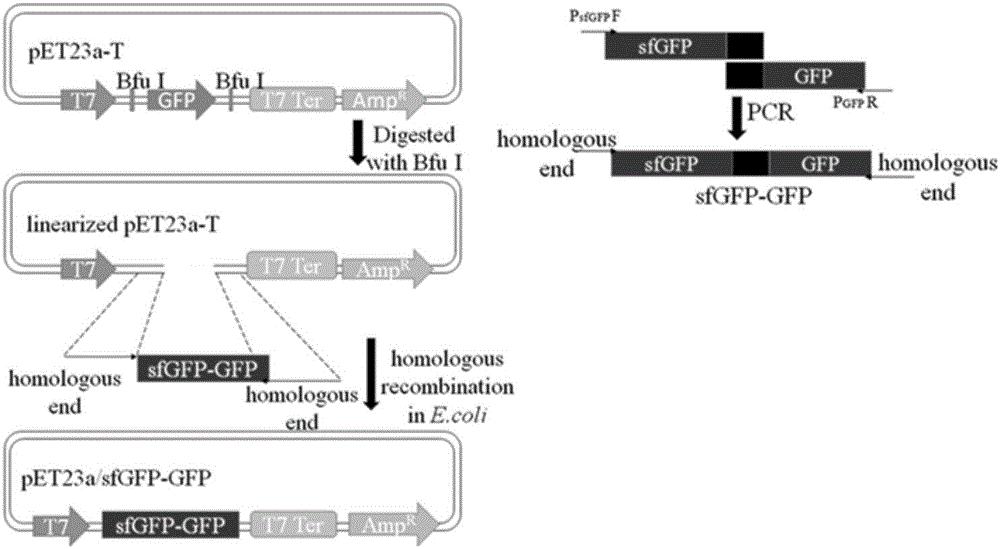
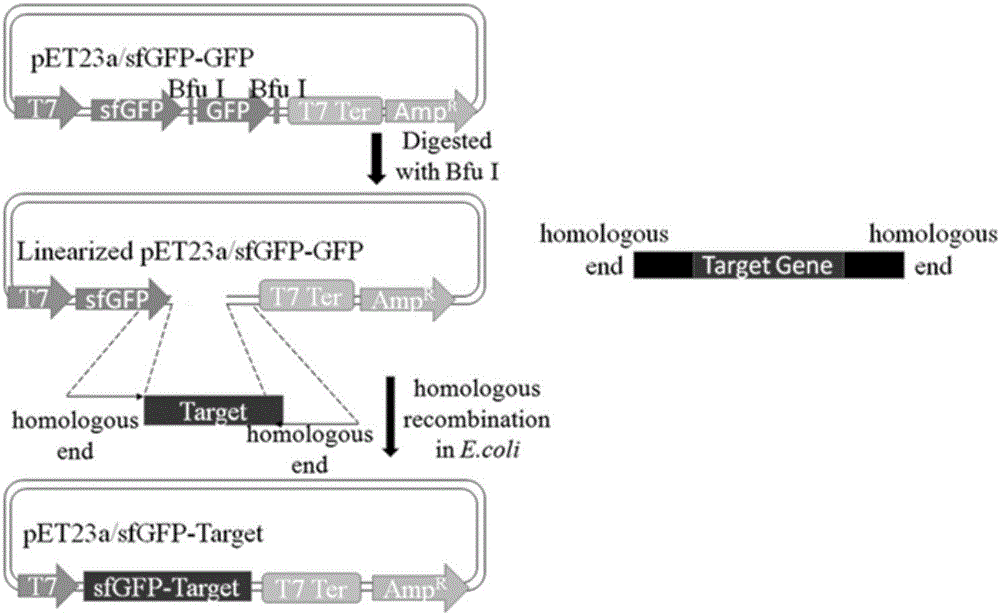
ActiveCN106591343AStrong autocrine abilityNo effect on functionMicroorganism based processesNucleic acid vectorEscherichia coliHeterologous
The invention provides a method for secretory expression of super-folded green fluorescent protein mediated heterologous protein in escherichia coli. The method includes steps: 1) constructing a secretory expression carrier pET23a / sfGFP-GFP taking sfGFP (super-folded green fluorescent protein) as a secretory tag; 2) constructing a heterologous protein gene recombinant expression carrier; 3) respectively converting and fusing the expression carriers to escherichia coli competent cell Rosetta Blue to obtain a recombinant strain; 4) expressing, culturing and performing functional verification; 5) performing high-density fermentation of the recombinant strain. According to secretion characteristics of the super-folded green fluorescent protein, extracellular secretory expression of the heterologous protein in escherichia coli in a fusion protein form is realized without mediating through signal peptides, an operation process is simplified, high autocrine performance is realized, functional influences of tag protein on target protein are avoided, expression conditions can be quickly optimized, target protein yield is increased, and the method is suitable for large-scale production. In addition, an application field of sfGFP is expanded, and a novel method is provided for extracellular secretory expression of the heterologous protein in escherichia coli.
Owner:HUBEI UNIV
Method for controlling micrometre zinc sulphide morphology by Escherichia coli biological template
InactiveCN101372357AImprove conversion rateImprove solubilityZinc sulfidesEscherichia coliCompetent cell
The invention relates to a method for controlling micron zinc sulfide shape by an escherichia coli biological template. A micron zinc sulfide material with excellent shape and a short rod-shaped structure is obtained by utilizing the space confinement effect of the biological template, taking the escherichia coli as the biological template, preparing competent cells which incubate with the zinc sulfide system and combine with thermal shock and calcination treatment. The shape control of the zinc sulfide material is carried out by the biological confinement effect existing in nature to obtain the zinc sulfide material with uniform shape and same size, which proves that the biological template can accurately regulate and control the synthesis of the material. The method has the advantages of simple process, mild conditions, available materials, low cost and that the obtained material has excellent shape characteristics. In addition, as the biological template has the characteristics of self-breeding and high shape repeatability, and the like, the method can easily realize large-scale production.
Owner:YANSHAN UNIV
Method for efficiently preparing (S)-styrene glycol from carbonyl reductase recombinant bacterium
The invention discloses a method for efficiently preparing (S)-styrene glycol from a carbonyl reductase recombinant bacterium, belonging to the technical field of biocatalysis asymmetric conversion. The invention provides a novel Candidaparapsiospis carbonyl group reductase gene scr II with Genbank sequence number of GQ411433; the scr II is inserted into a vector pET28a to construct a recombinant plasmid pETSCR II which is converted into E.coli BL21(DE3) competent cell; and a recombinant bacterium E.coli BL21 / pETSCR II with the preservation number of CCTCC NO:M209290 can be obtained by the screening of an LB flat plate containing 100 micrograms / ml kanamycin. The (S)-styrene glycol can be prepared by catalyzing 5g / L 2-carbonyl acetophenone obtained by asymmetrically reducing the recombinant bacterium; and the optical purity of products is 100 percent and the yield reaches 98.1 percent. The invention provides a novel functional gene and an effective path for efficiently preparing the (S)-styrene glycol.
Owner:JIANGNAN UNIV
Popular searches
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com