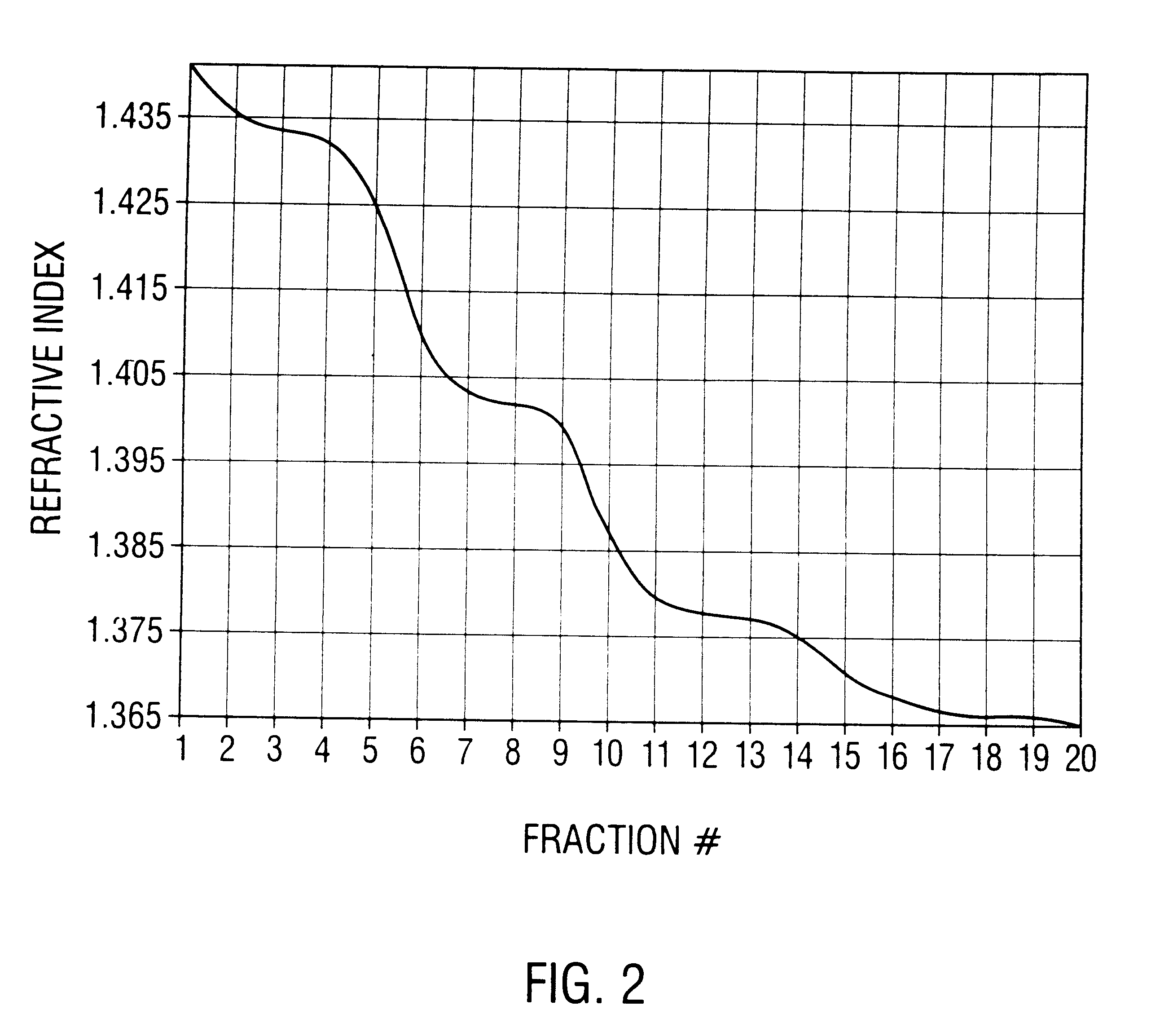
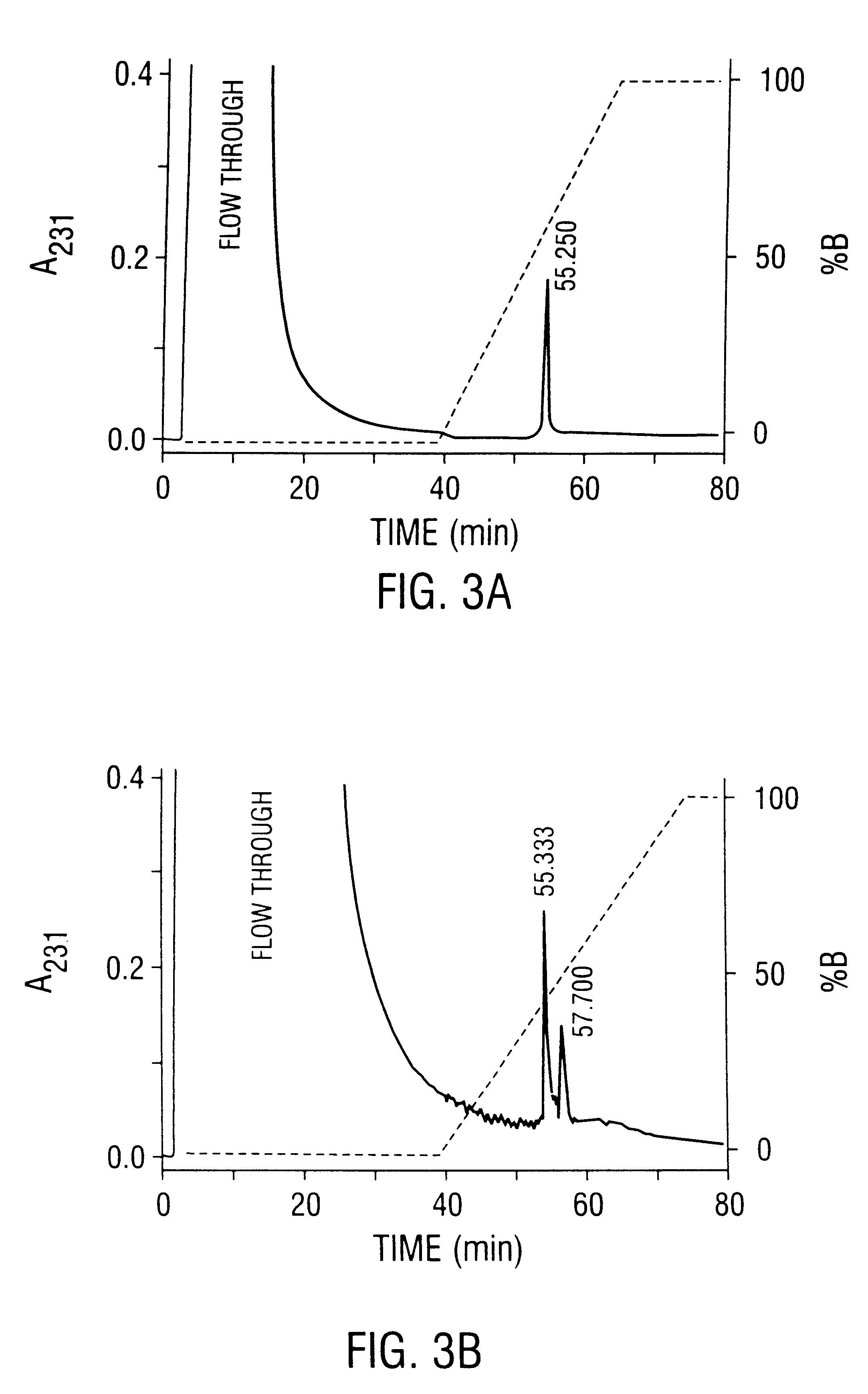
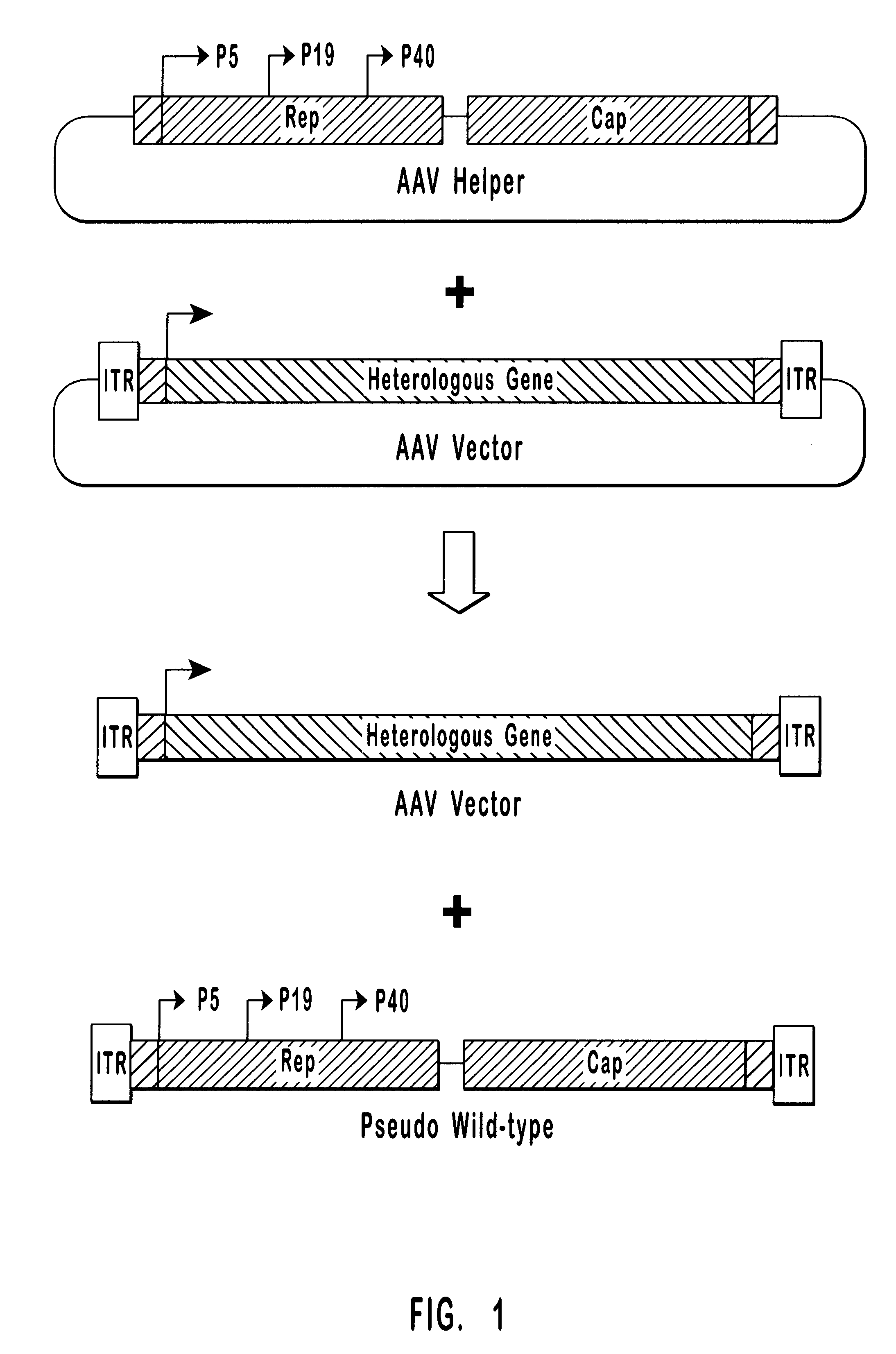
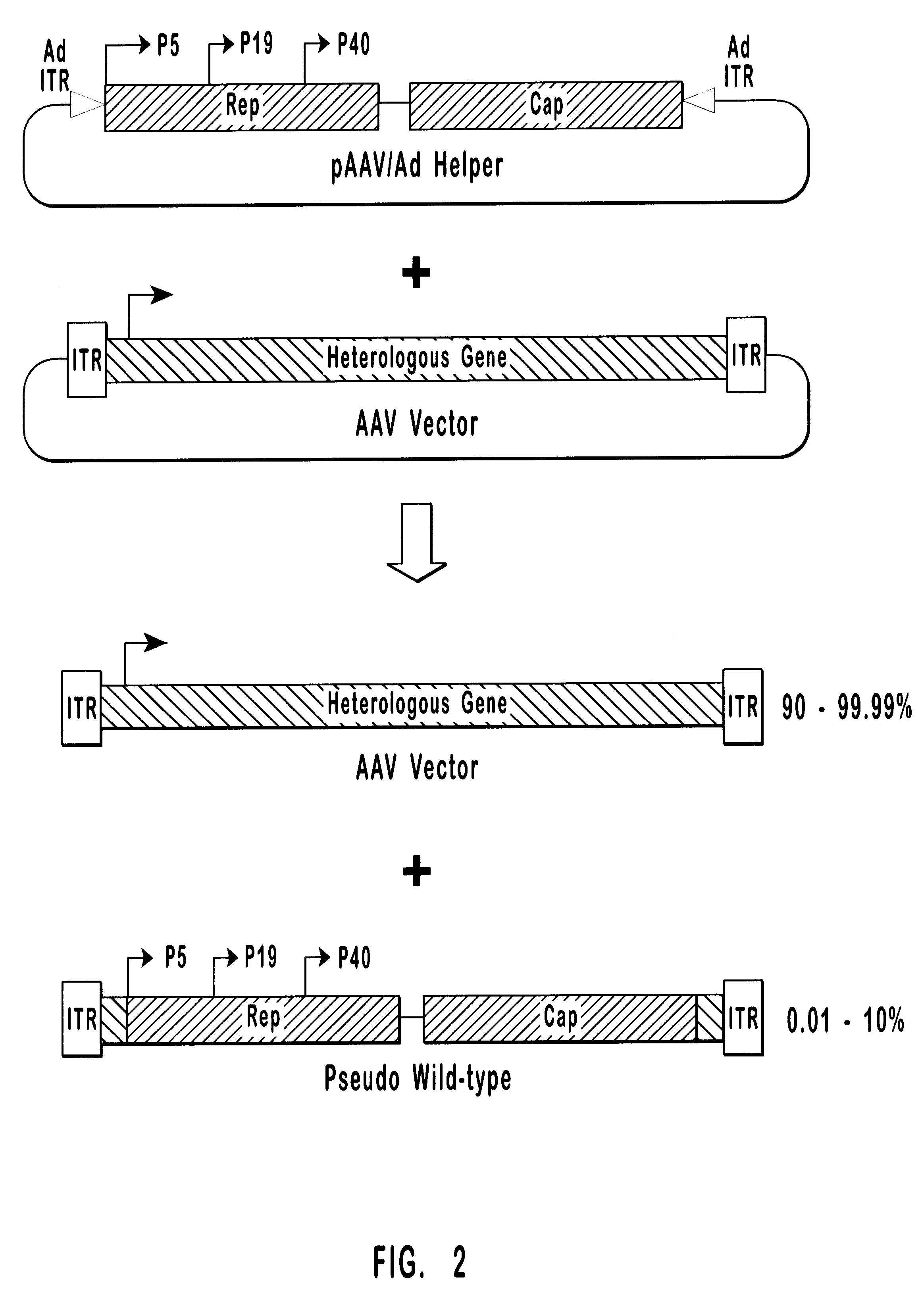
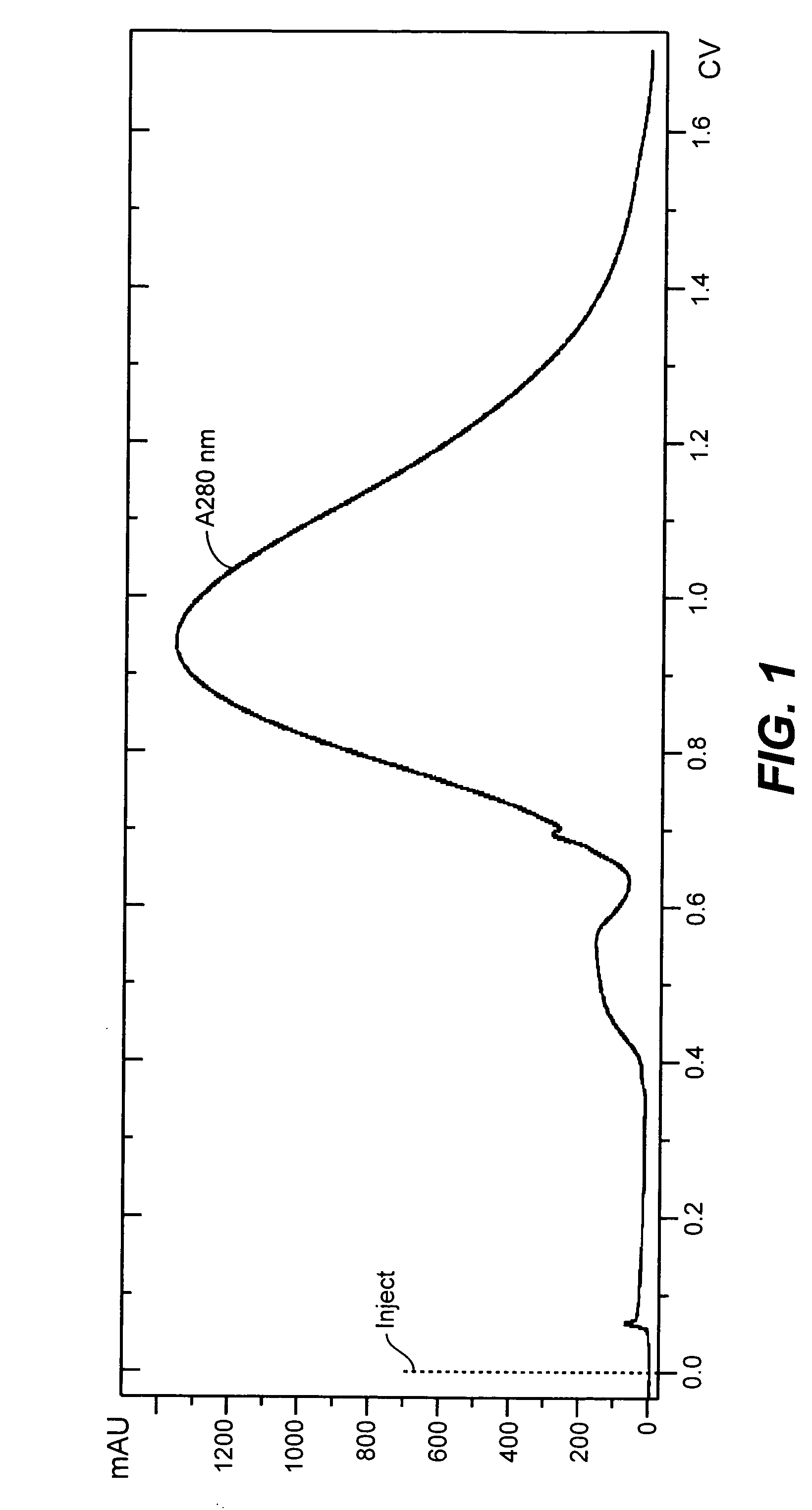
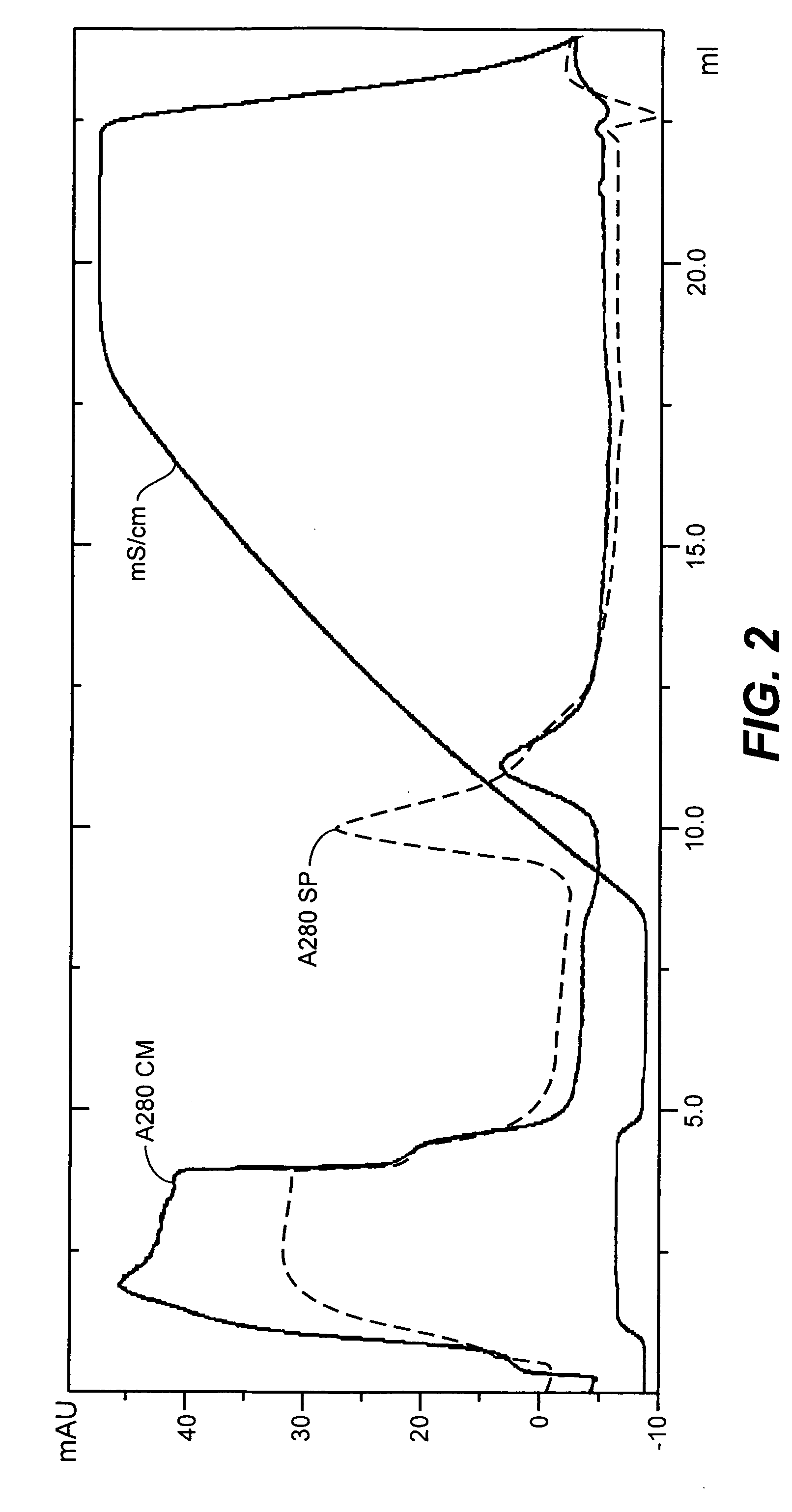
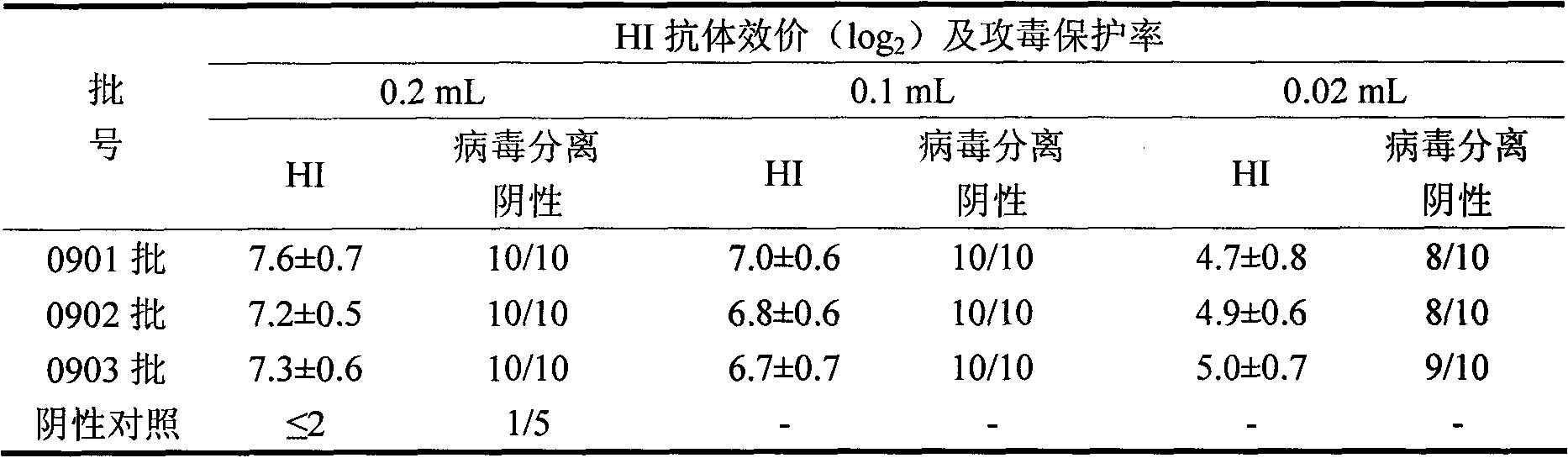
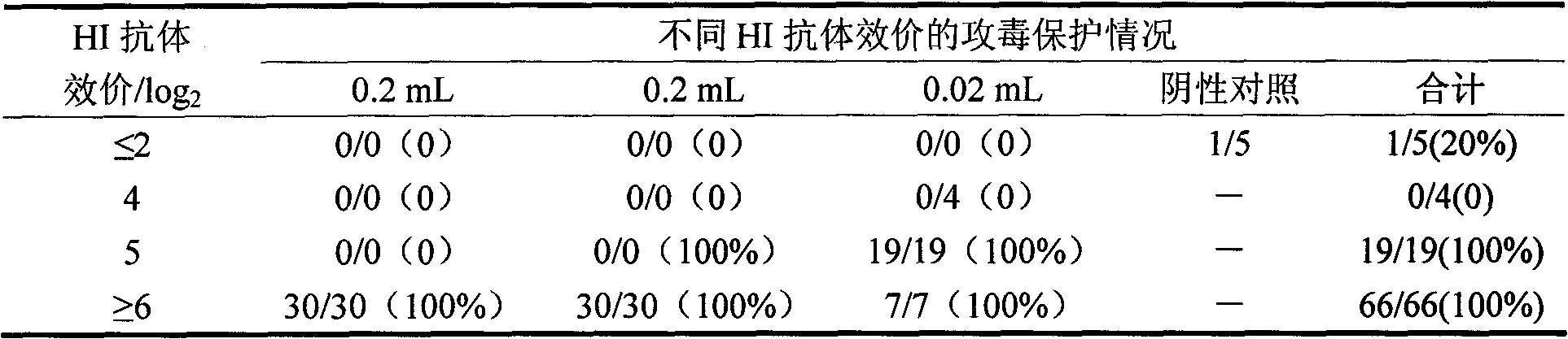
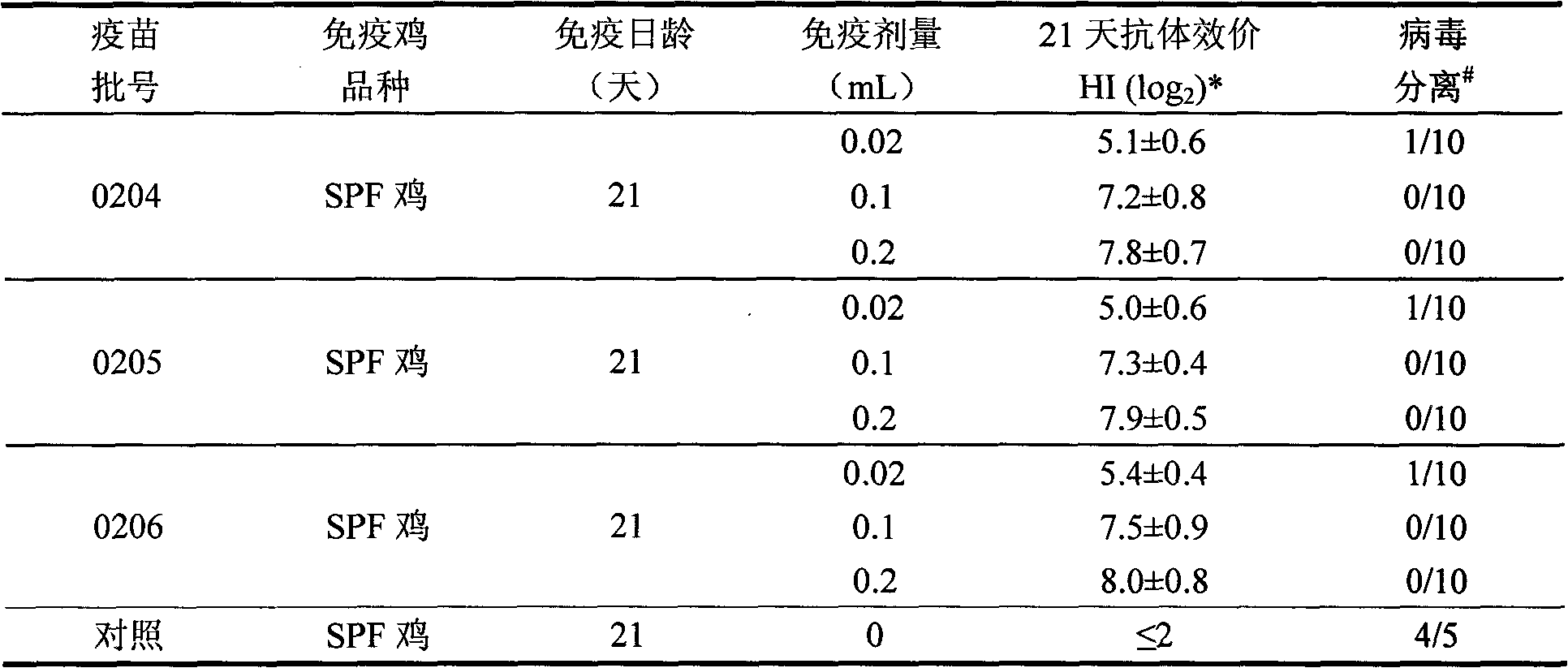
Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
839 results about "High titer" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
A high titer (high levels of ASO) is not specific for any type of poststreptococcal disease, but it does indicate if a streptococcal infection is or has been present. Serial (several given in a row) ASO testing is often performed to determine the difference between an acute or convalescent blood sample.
Methods for generating high titer helper-free preparations of released recombinant AAV vectors
InactiveUS6989264B2Genetic therapy composition manufactureGroup 5/15 element organic compoundsGene deliveryHeterologous
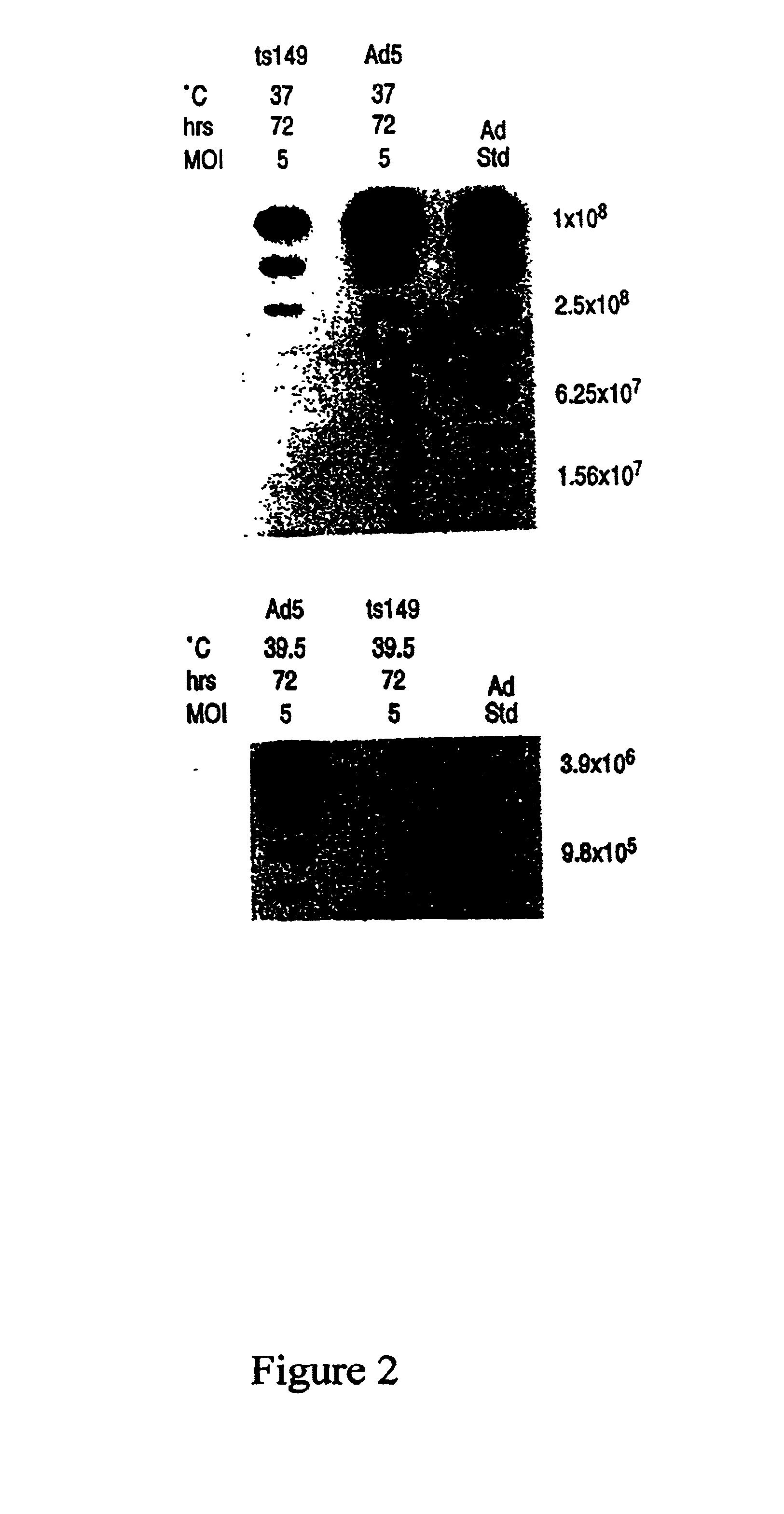
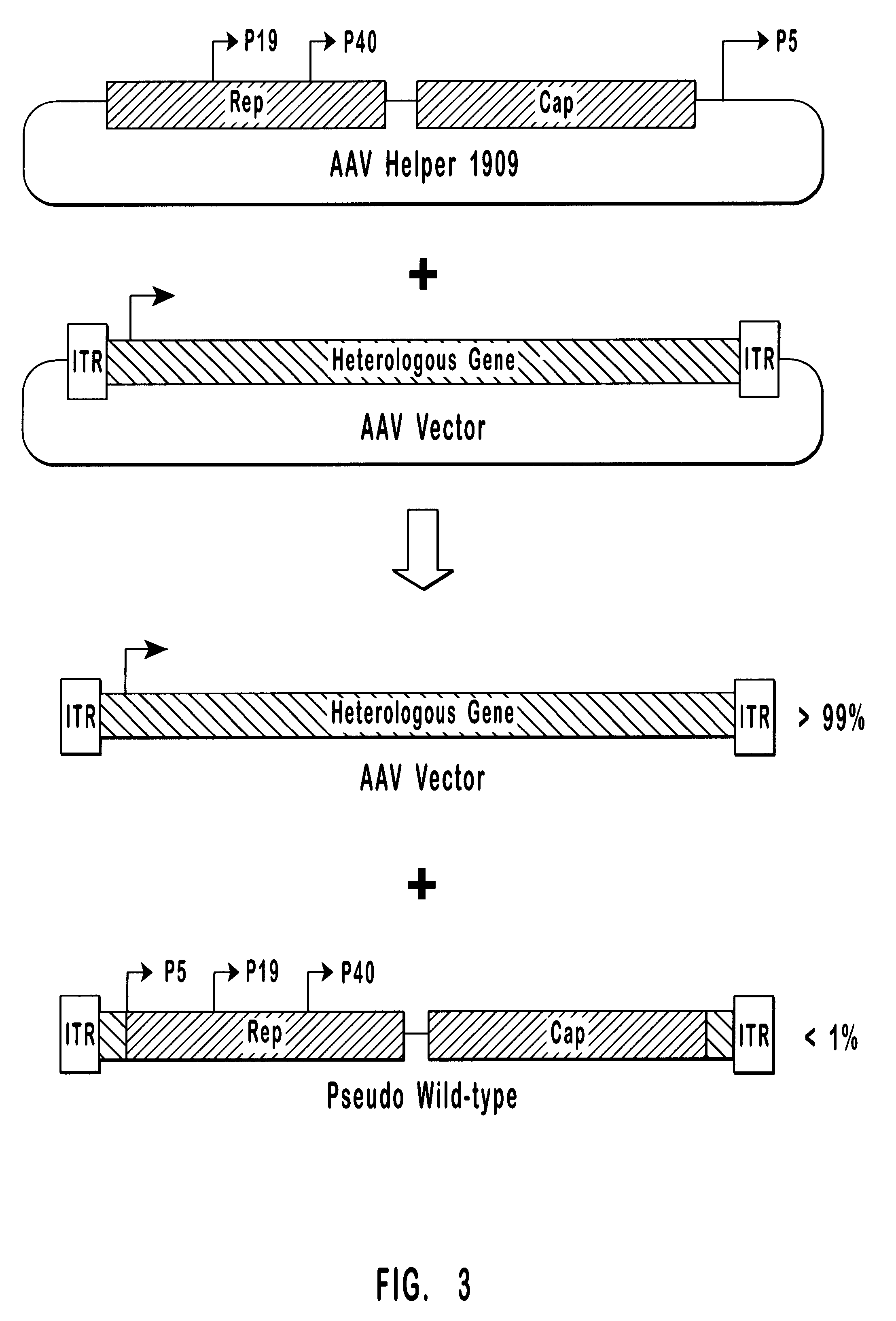
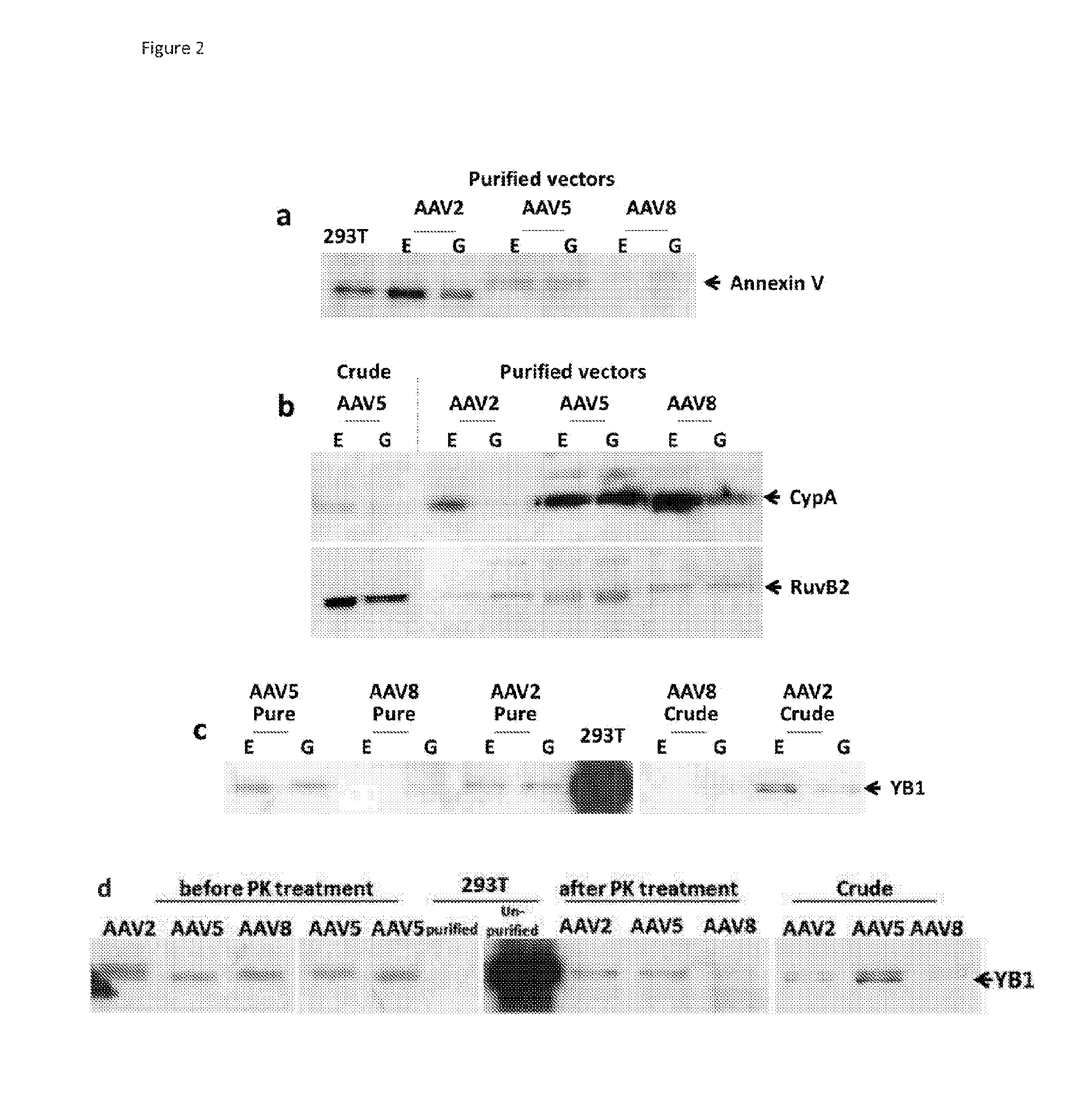
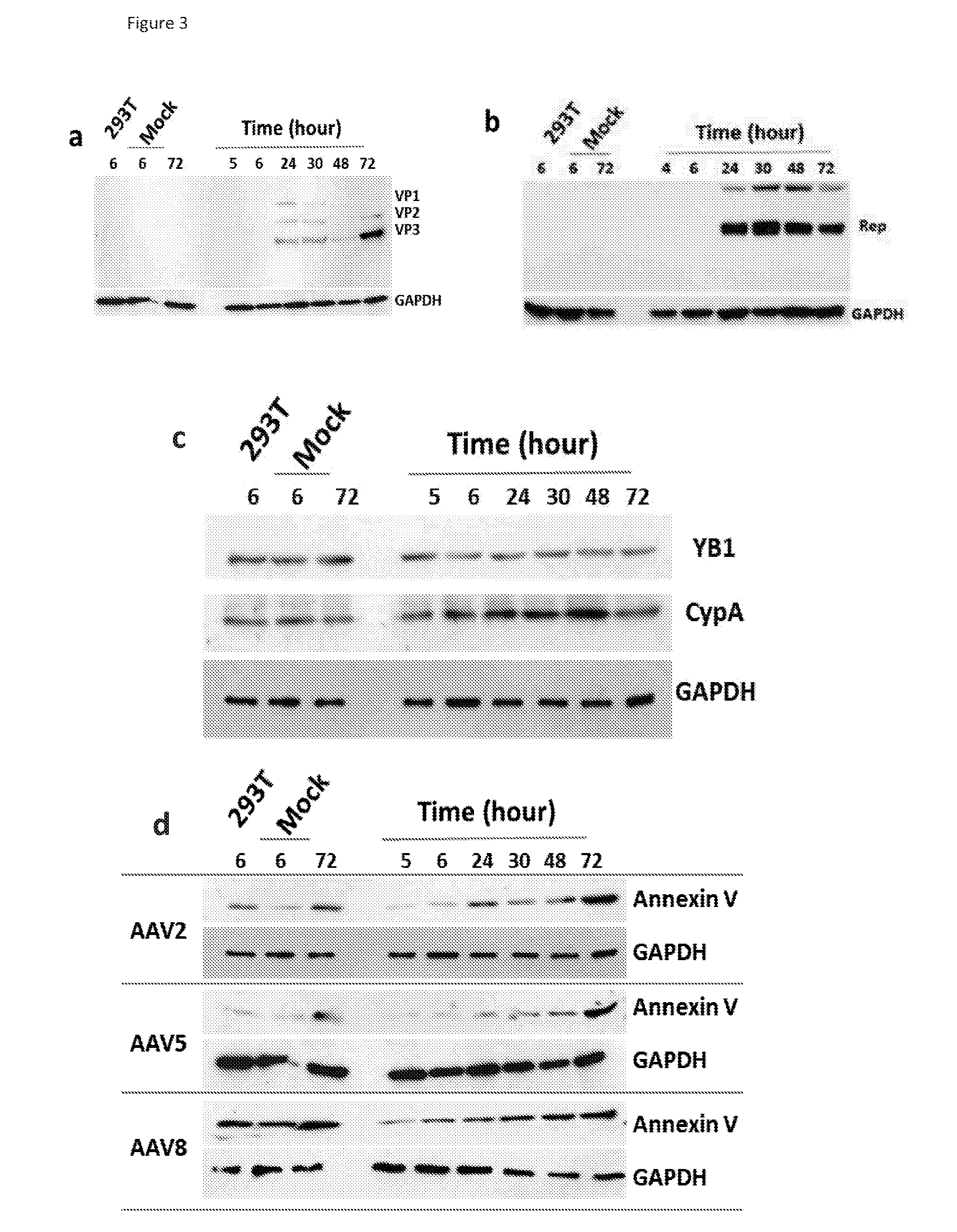
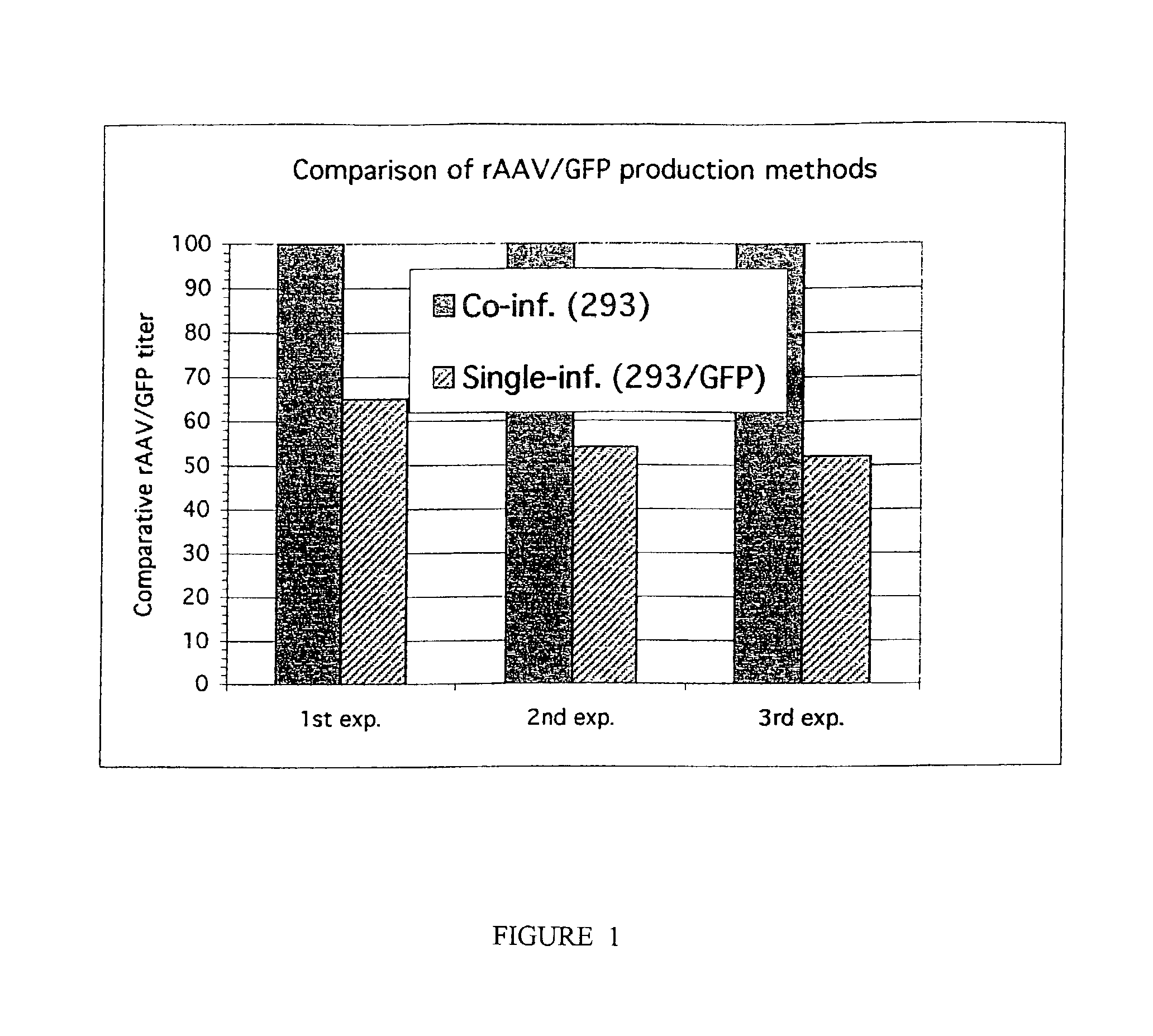
This invention provides methods and compositions for producing high titer, substantially purified preparations of recombinant adeno-associated virus (AAV) that can be used as vectors for gene delivery. At the onset of vector production, AAV producer cells of this invention typically comprise one or more AAV packaging genes, an AAV vector comprising a heterologous (i.e. non-AAV) transgene of interest, and a helper virus such as an adenovirus. The AAV vector preparations produced are generally replication incompetent but are capable of mediating delivery of a transgene of interest (such as a therapeutic gene) to any of a wide variety of tissues and cells. The AAV vector preparations produced according to this invention are also substantially free of helper virus as well as helper viral and cellular proteins and other contaminants. The invention described herein provides methods of producing rAAV particles by culturing producer cells under conditions, such as temperature and pH, that promote release of virus. Also provided is a quantitative, high-throughput assay useful in the assessment of viral infectivity and replication, as well as in the screening of agent that affect viral infectivity and / or replication.
Owner:TARGETED GENETICS CORPORTION
High titer production of highly linear poly (alpha 1,3 glucan)
ActiveUS20130244287A1Improve mechanical propertiesMore amenable to preparing fibersTransferasesFermentationSucroseSaccharophagus degradans
Owner:NUTRITION & BIOSCIENCES USA 4 INC
Method of preparing recombinant adeno-associated virus compositions
InactiveUS6660514B1Reduce concentrationImprove yield and recoveryMicrobiological testing/measurementEnzymologyBiochemistryAdeno-associated virus
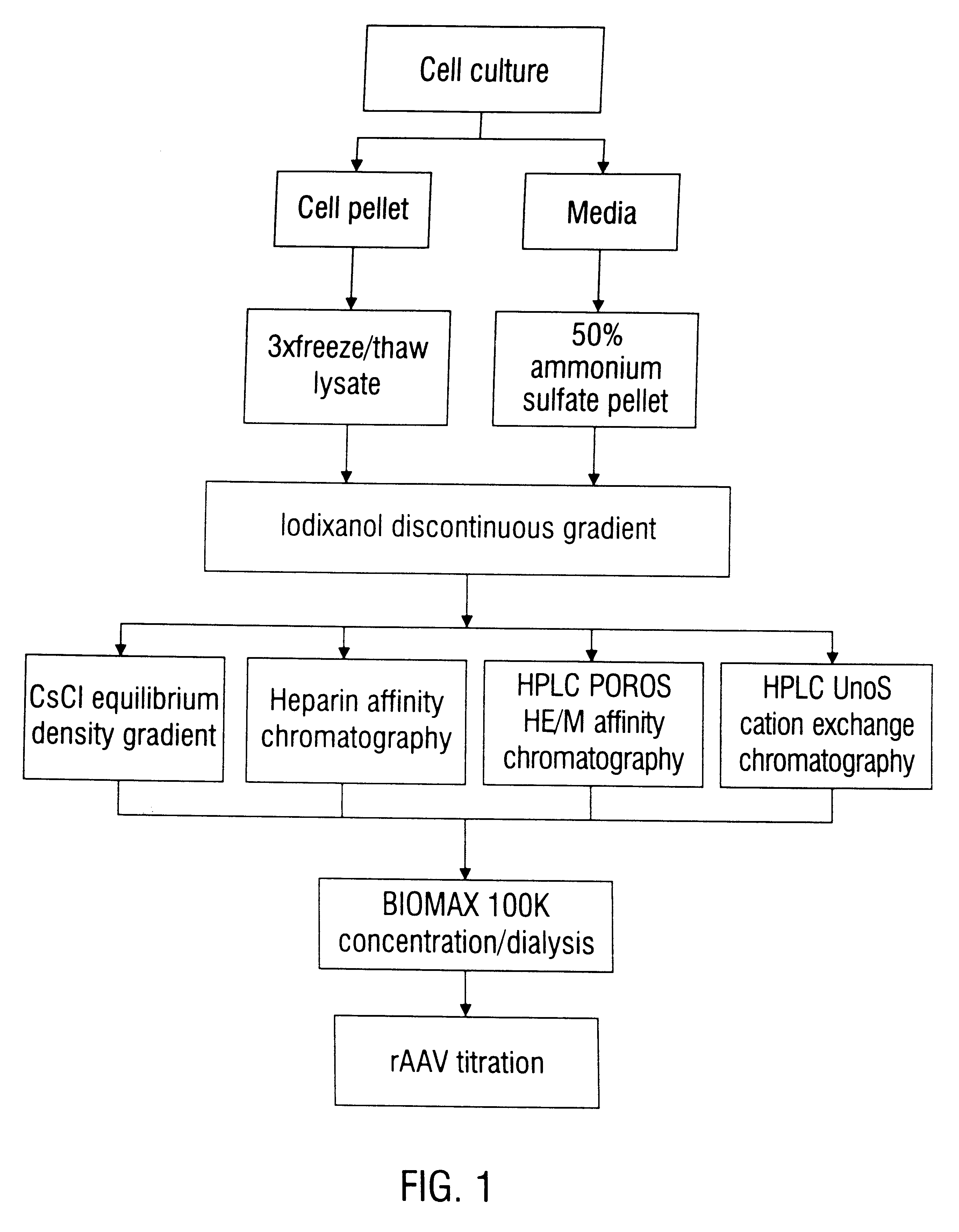
Disclosed are methods for the isolation and purification of high-titer recombinant adeno-associated virus (rAAV) compositions. Also disclosed are methods for reducing or eliminating the concentration of helper adenovirus in rAAV samples. Methods are disclosed that provide highly-purified rAAV stocks having titers up to about 10<13 >particles / ml at particle-to-infectivity ratios of less than 100 in processes that are accomplished about 24 hours or less.
Owner:UNIV OF FLORIDA RES FOUNDATION INC
High-efficiency wild-type-free AAV helper functions
InactiveUS6376237B1Efficient productionEliminate productionAntibody mimetics/scaffoldsVirus peptidesWild typeNucleic acid
The present invention provides methods and compositions for producing high titer, wild-type-free preparations of recombinant AAV ("rAAV") virions. The compositions of the present invention include novel nucleic acids encoding AAV helper functions and AAV helper function vectors. The present invention also includes host cells transfected by the claimed nucleic acids, methods of using the claimed vectors, and rAAV virions produced by such methods.
Owner:GENZYME CORP
Cocal vesiculovirus envelope pseudotyped retroviral vectors
InactiveUS20120164118A1Increase serum stabilitySsRNA viruses negative-senseBiocideCocal vesiculovirusVirus-Retrovirus
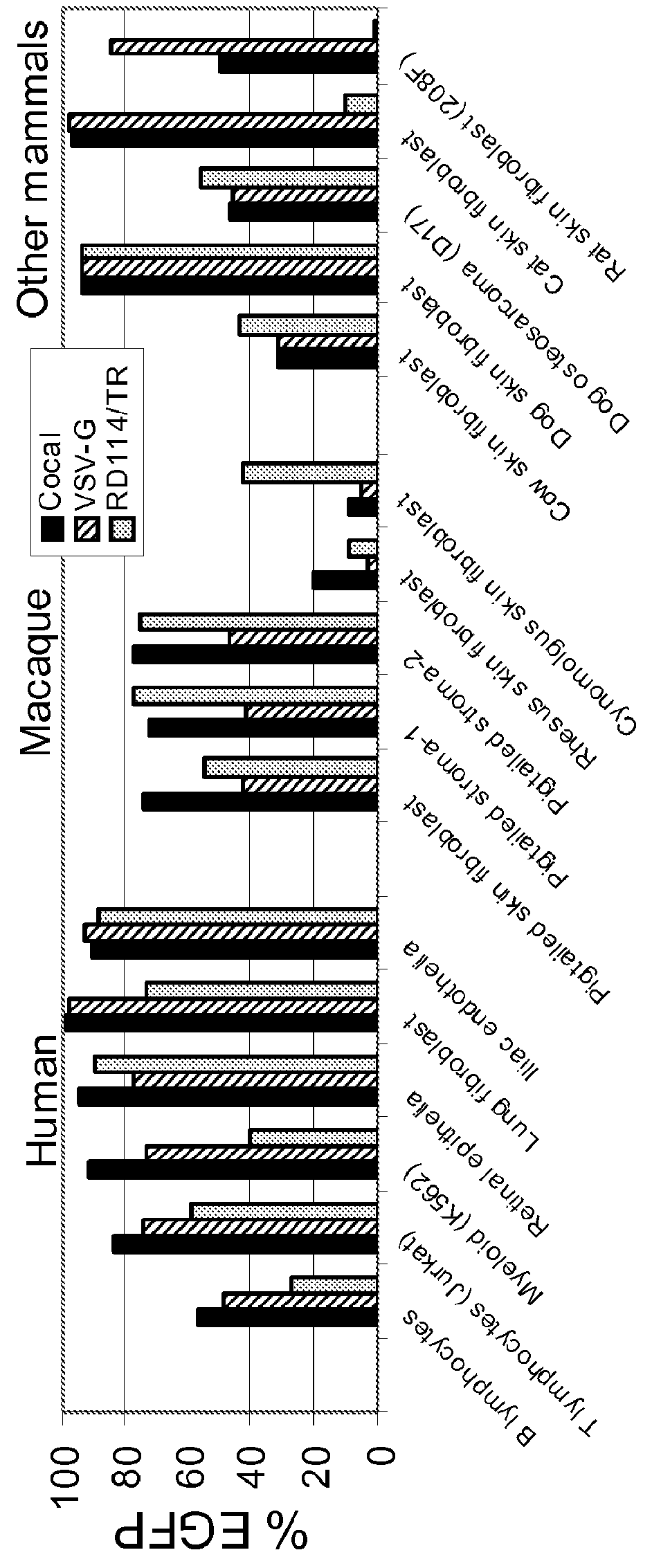
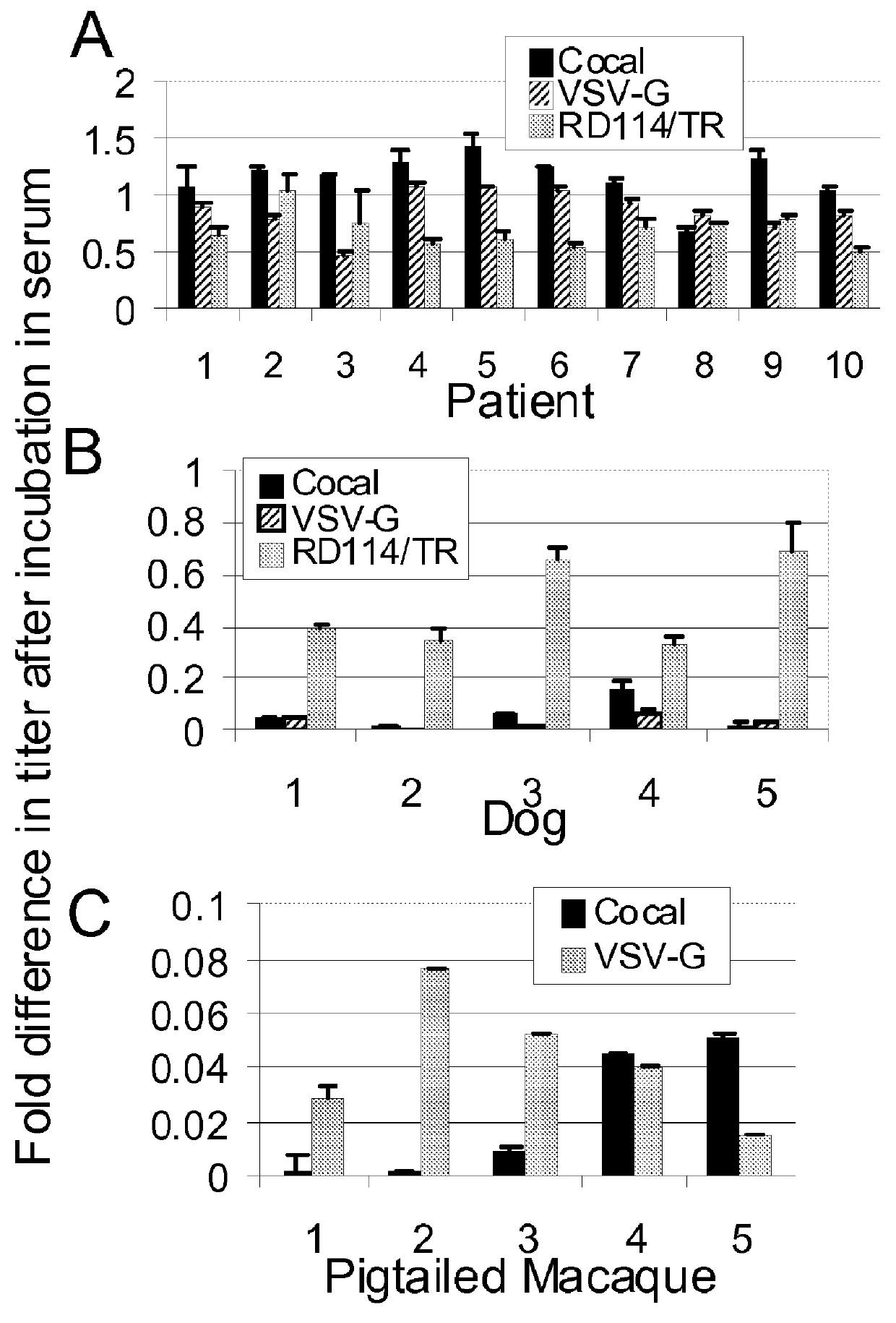
Provided herein are Cocal vesiculovirus envelope pseudotyped retroviral vectors that exhibit high titers, broad species and cell-type tropism, and improved serum stability. Disclosed Cocal vesiculovirus envelope pseudotyped retroviral vectors may be suitably employed for gene therapy applications and, in particular, for the ex vivo and in vivo delivery of a gene of interest to a wide variety of target cells.
Owner:FRED HUTCHINSON CANCER RES CENT
Recombinant vesiculoviruses and their uses
The invention provides recombinant replicable vesiculoviruses. The invention provides a method which, for the first time, successfully allows the production and recovery of replicable vesiculoviruses, as well as recombinant replicable vesiculoviruses, from cloned DNA, by a method comprising expression of the full-length positive-strand vesiculovirus antigenomic RNA in host cells. The recombinant vesiculoviruses do not cause serious pathology in humans, can be obtained in high titers, and have use as vaccines. The recombinant vesiculoviruses can also be inactivated for use as killed vaccines.
Owner:YALE UNIV
Viral purification methods
The present invention is directed to an improved method of purifying virus, particularly reovirus. Infectious virus can be extracted from a cell culture with a detergent to produce high titers of virus, and the virus can then be purified by simple steps such as filtration and column chromatography. Viruses and compositions comprising the viruses prepared according to the present invention are also provided.
Owner:ONCOLYTICS BIOTECH
Viral purification methods
ActiveUS7223585B2Simple methodViral antigen ingredientsGenetic material ingredientsPurification methodsFiltration
The present invention is directed to an improved method of purifying virus, particularly reovirus. Infectious virus can be extracted from a cell culture with a detergent to produce high titers of virus, and the virus can then be purified by simple steps such as filtration and column chromatography. Viruses and compositions comprising the viruses prepared according to the present invention are also provided.
Owner:ONCOLYTICS BIOTECH
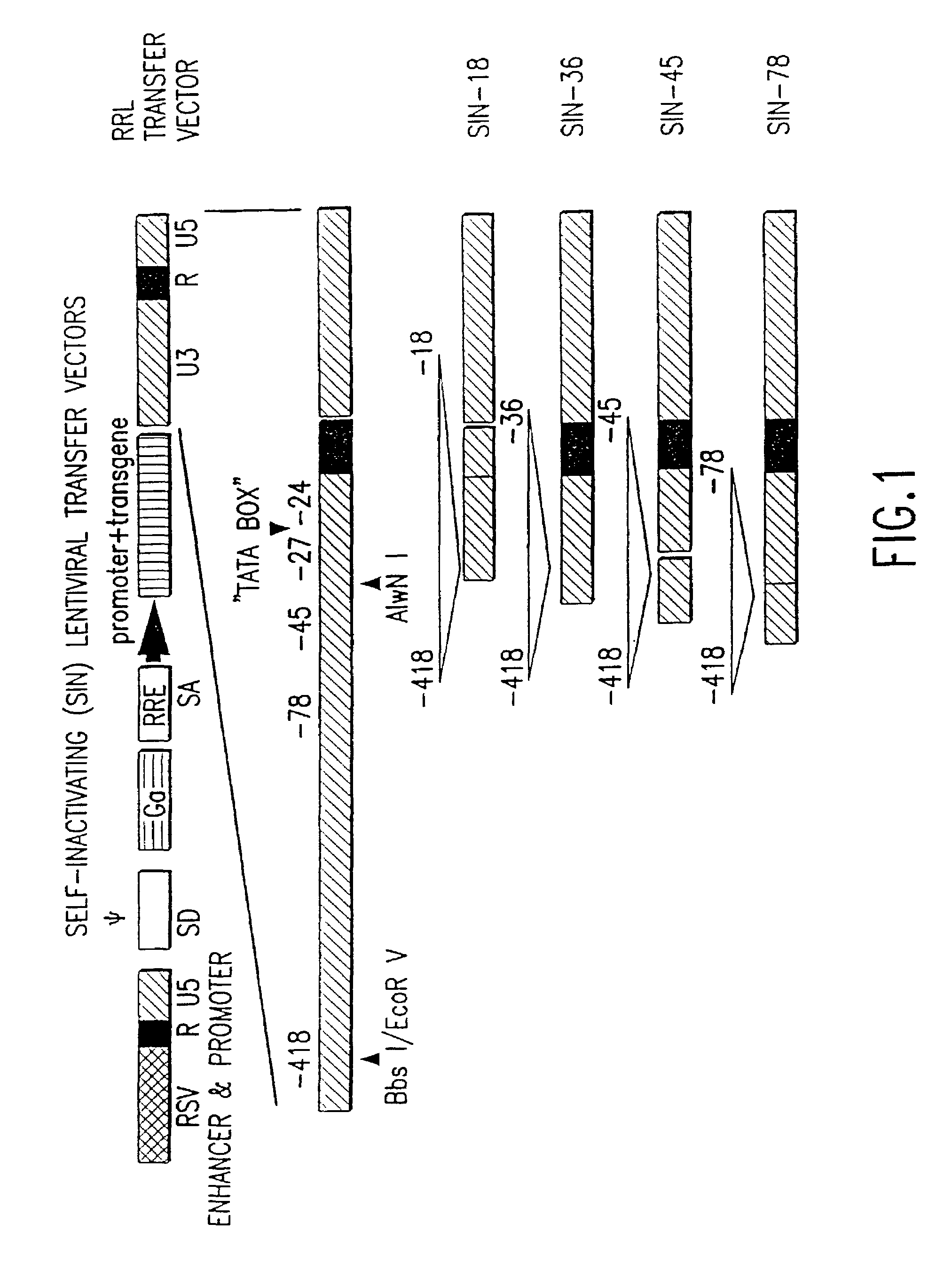
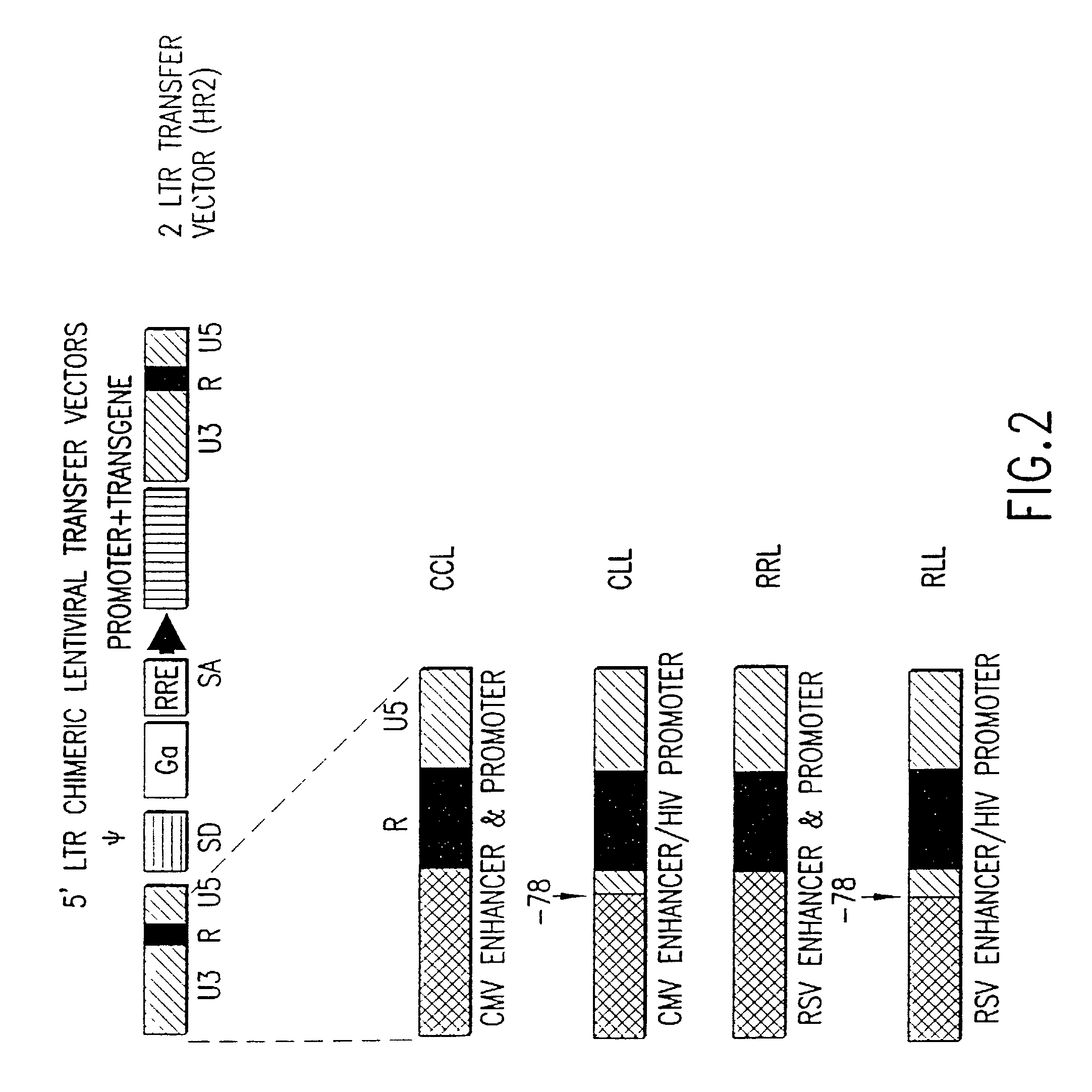
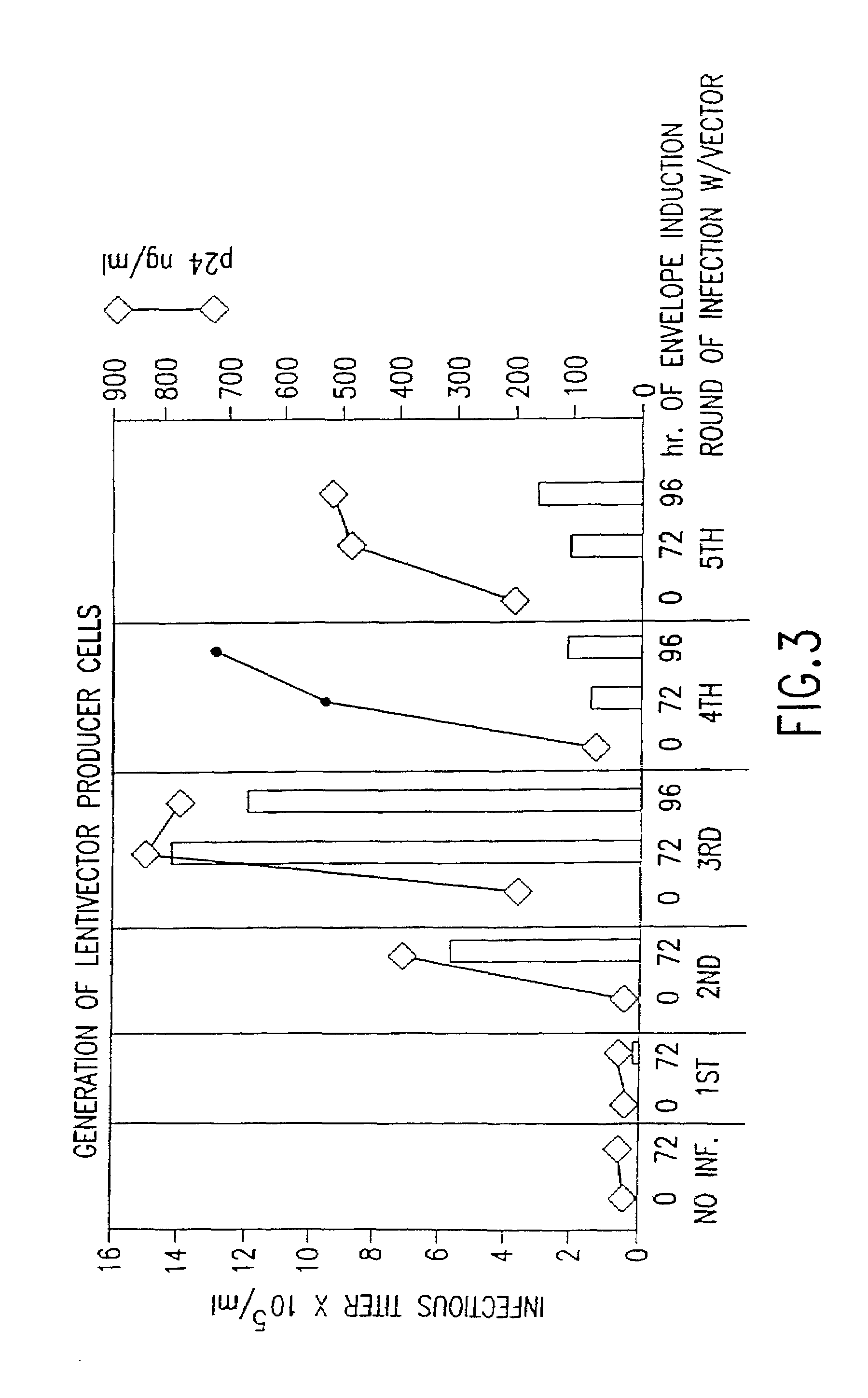
Method and means for producing high titer, safe, recombinant lentivirus vectors
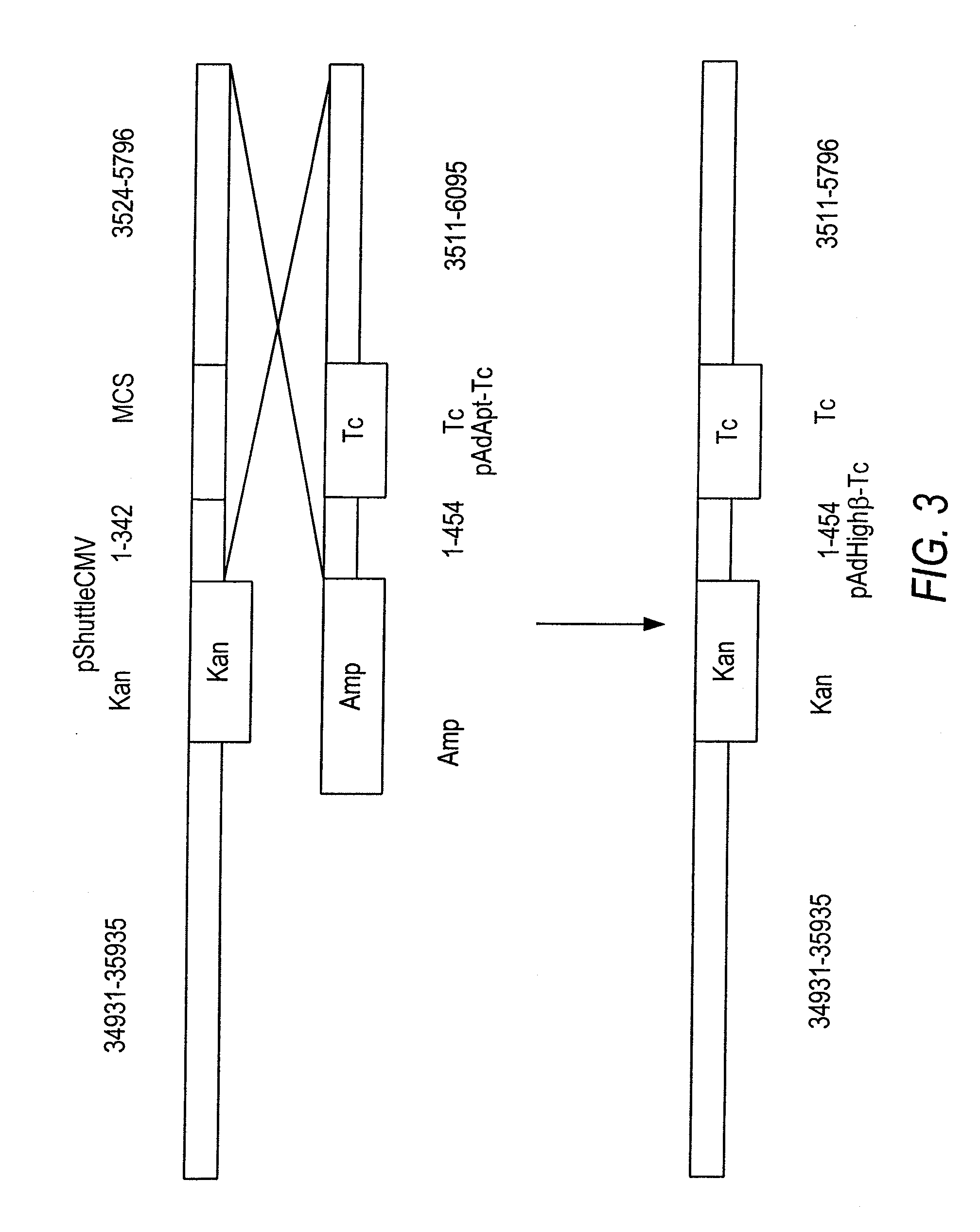
Lentiviral vectors modified at the 5′ LTR or both the 5′ and 3′ LTR are useful in the production of recombinant lentivirus vectors (See the Figure). Such vectors can be produced in the absence of a functional tat gene. Multiple transformation of the host cell with the vector carrying the transgene enhances virus production. The vectors can contain inducible or conditional promoters.
Owner:MILTENYI BIOTEC B V & CO KG
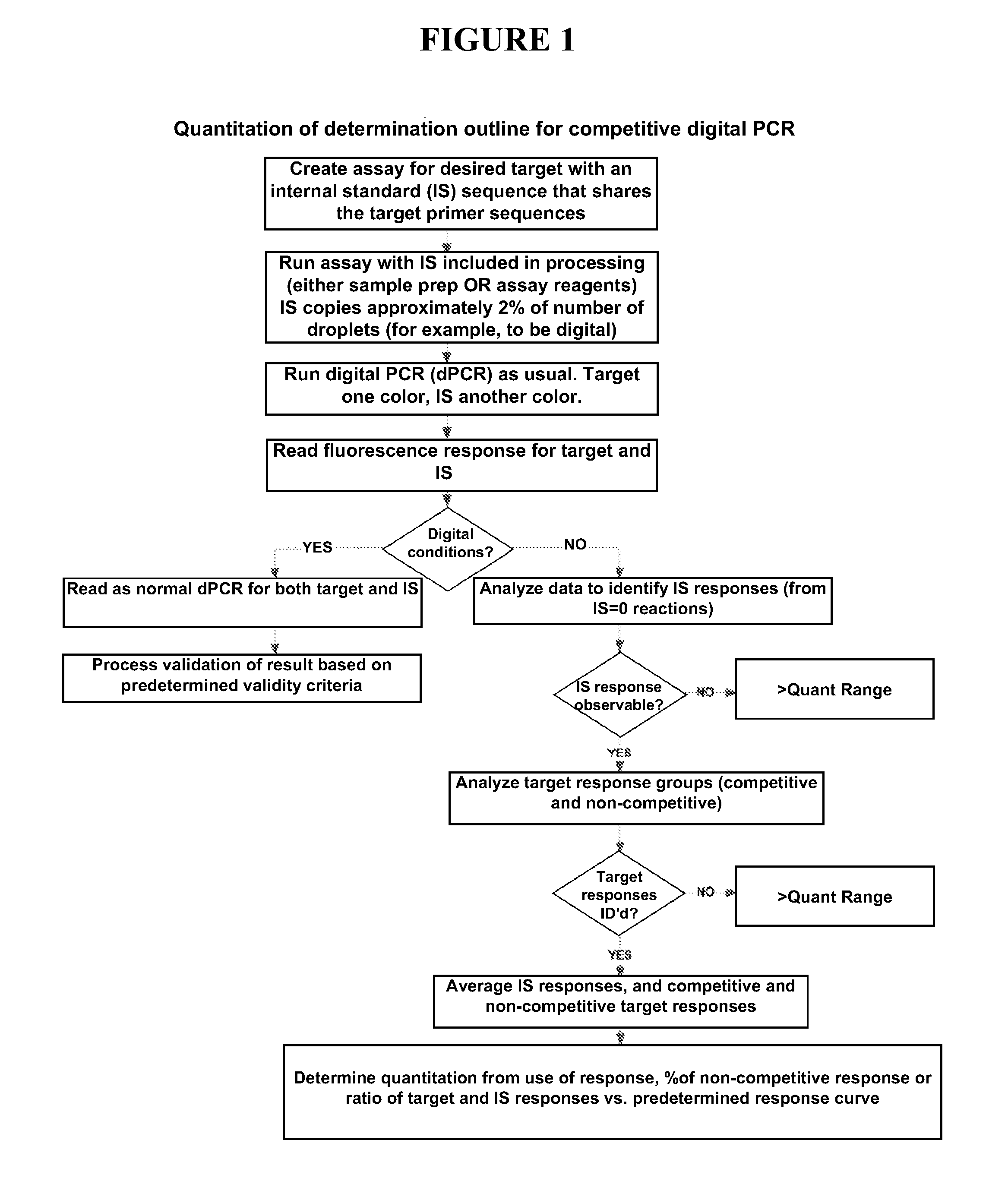
Quantitating high titer samples by digital PCR
ActiveUS20120164652A1Microbiological testing/measurementFluorescence/phosphorescenceBiologyHigh titer
The present invention provides systems, devices, methods, kits, and compositions for nucleic acid analysis using digital PCR. In particular, methods are provided to analyze high titer samples that cannot be divided into a sufficient number of partitions containing zero nucleic acid molecules per partition to allow for Poisson analysis (digital PCR analysis).
Owner:ABBOTT MOLECULAR INC
High-efficiency AA V helper functions
The present invention provides methods and compositions for producing high titer preparations of recombinant AAV ("rAAV") virions. The compositions of the present invention include AAV helper function systems and host cells. The present invention also includes methods of using AAV helper function vectors that effect the production of only small amounts of the long forms of Rep protein, and rAAV virions produced by such methods.
Owner:GENZYME CORP
Hybridoma cell line 1C11 and anti-aflatoxin general monoclonal antibody generated by same as well as applications thereof
ActiveCN101993855AHigh sensitivityHigh practical application valueMicroorganism based processesTissue cultureCell strainHybridoma cell
The invention provides a hybridoma cell line 1C11 and an anti-aflatoxin general monoclonal antibody secreted by the same as well as the applications thereof. The hybridoma cell line 1C11 can be used for preparing a high-titer aflatoxin antibody, and a mouse hydroperitoneum antibody is measured to reach 5.12*106 by using an ELISA (Enzyme-Linked Immunosorbent Assay). The anti-aflatoxin general monoclonal antibody has high sensitivity, respectively reaches the IC50 (50% inhibiting concentration) of aflatoxin B1, B2, G1 and G2 to be 1.2, 1.3, 2.2 and 18.0 pg / mL, is the antibody with highest sensitivity among currently reported four aflatoxin antibodies, is used for measuring the total aflatoxin amounts, i.e. the total amounts of the aflatoxin B1, B2, G1 and G2 and has great practical application values.
Owner:INST OF OIL CROPS RES CHINESE ACAD OF AGRI SCI
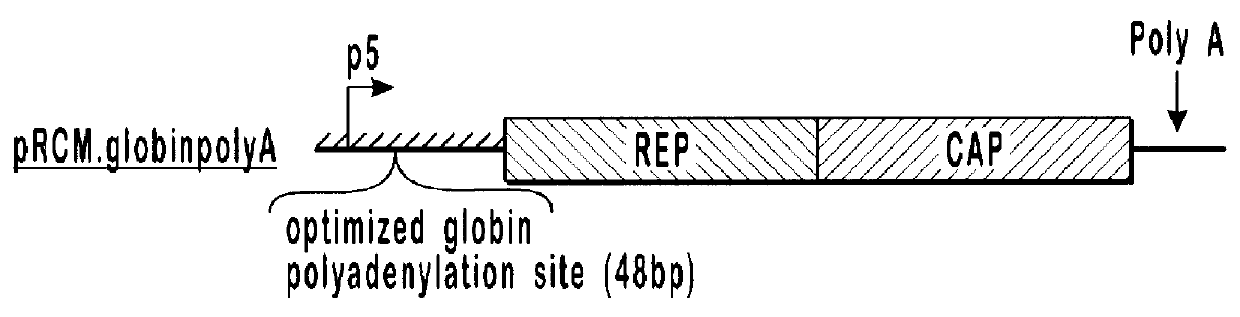
Production of recombinant AAV using adenovirus comprising AAV rep/cap genes
InactiveUS7115391B1High yieldAmenable to large-scale industrial applicationBiocideVectorsMinimal promoterDNA Intercalation
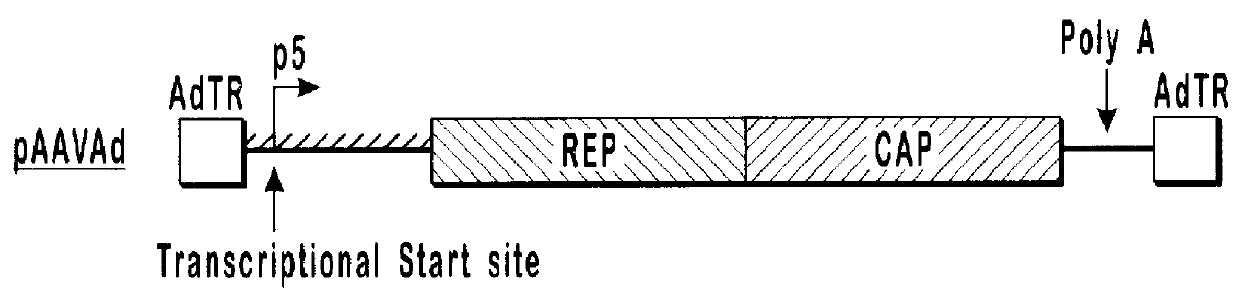
This invention relates to novel adenoviruses useful in the production of high titers of recombinant adeno-associated virus (rAAV) comprising a foreign DNA insert and methods of making these adenoviruses. The adenovirus comprises the AAV rep gene in which the p5 promoter is replaced by a minimal promoter or by no promoter. The invention also provides methods of producing high levels of rAAV as a substantially homogenous preparation and composition of rAAV.
Owner:GENOVO
Method for generating influenza viruses and vaccines
InactiveUS20050054846A1Grows more efficientlyEfficient growth processSsRNA viruses negative-senseBiocideVirusMammalian cell
The present invention is based on the discovery that a high titer reassortant influenza virus is produced in mammalian cell culture by replacing the NS gene of the A / PuertoRico / 3 / 24 master strain with the NS gene of the A / England / 1 / 53 strain. The invention provides influenza viruses and vaccines generated in mammalian cells as well as methods for producing such. The invention further provides an influenza virus master strain and kits for generating reassortant influenza viruses in mammalian cell culture and methods of making and using the master strain.
Owner:ST JUDE CHILDRENS RES HOSPITAL INC
Staphylococcal immunotherapeutics via donor selection and donor stimulation
InactiveUS20060222651A1Immunoglobulins against bacteriaPharmaceutical delivery mechanismPassive ImmunizationsCell-Extracellular Matrix
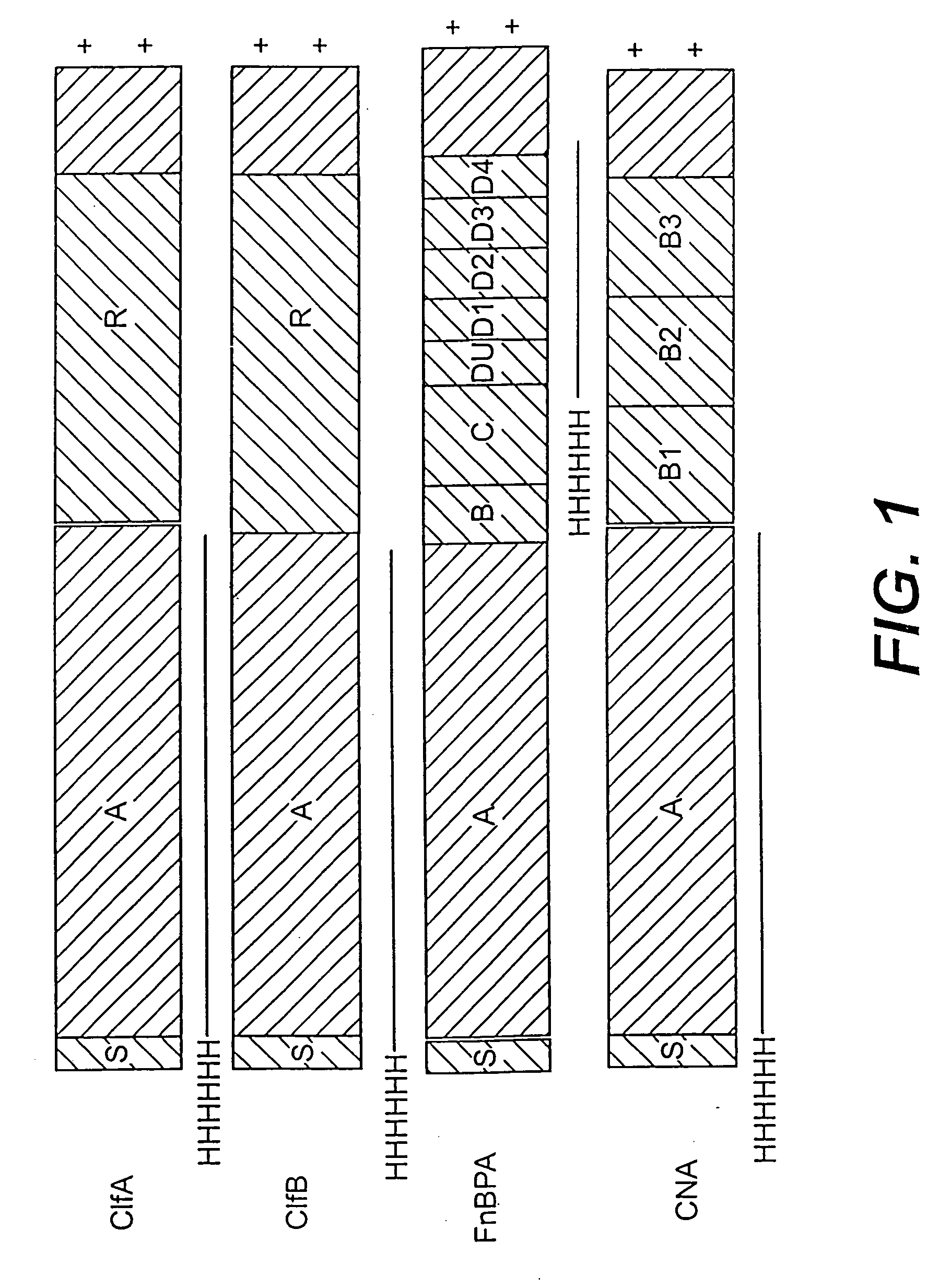
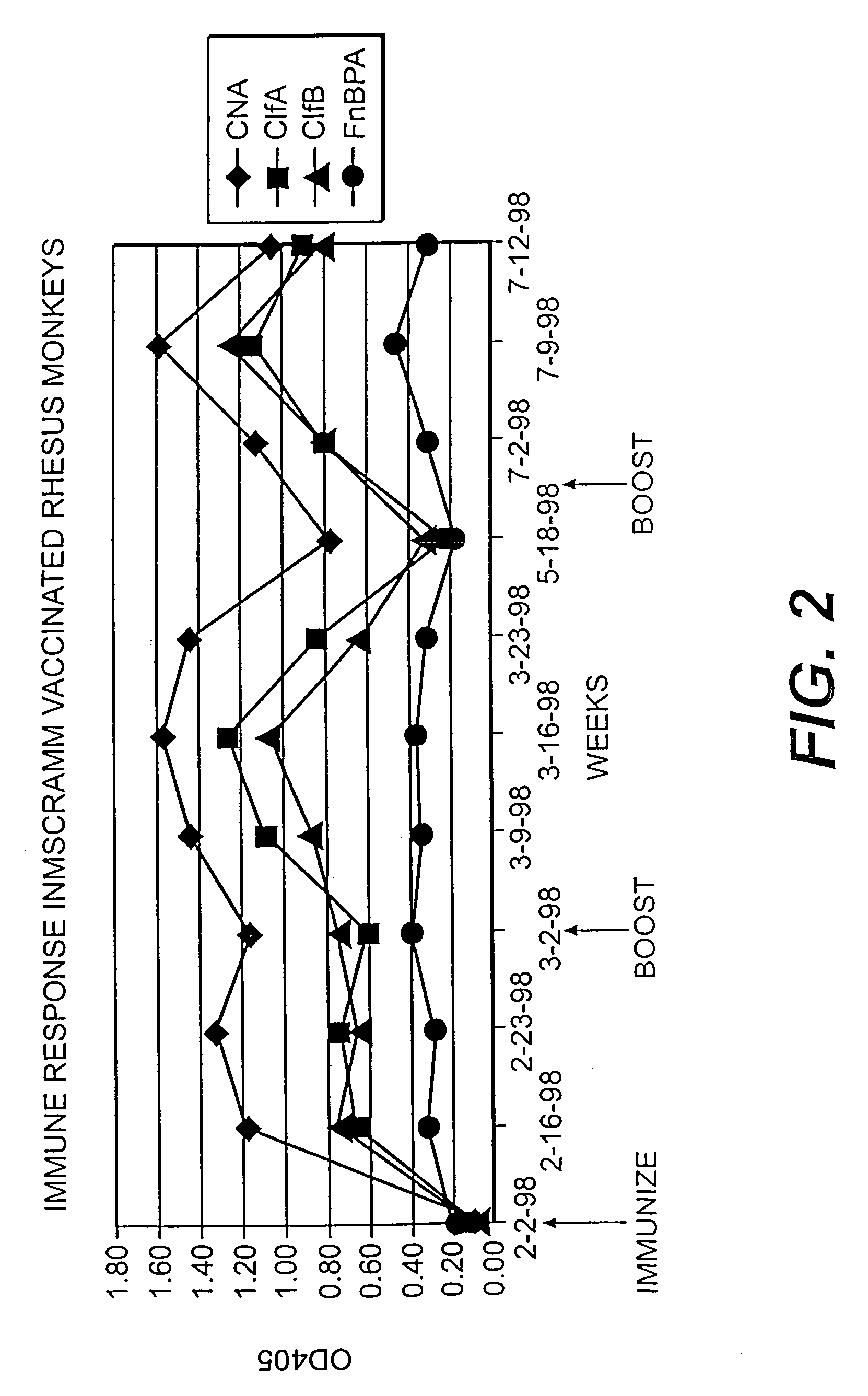
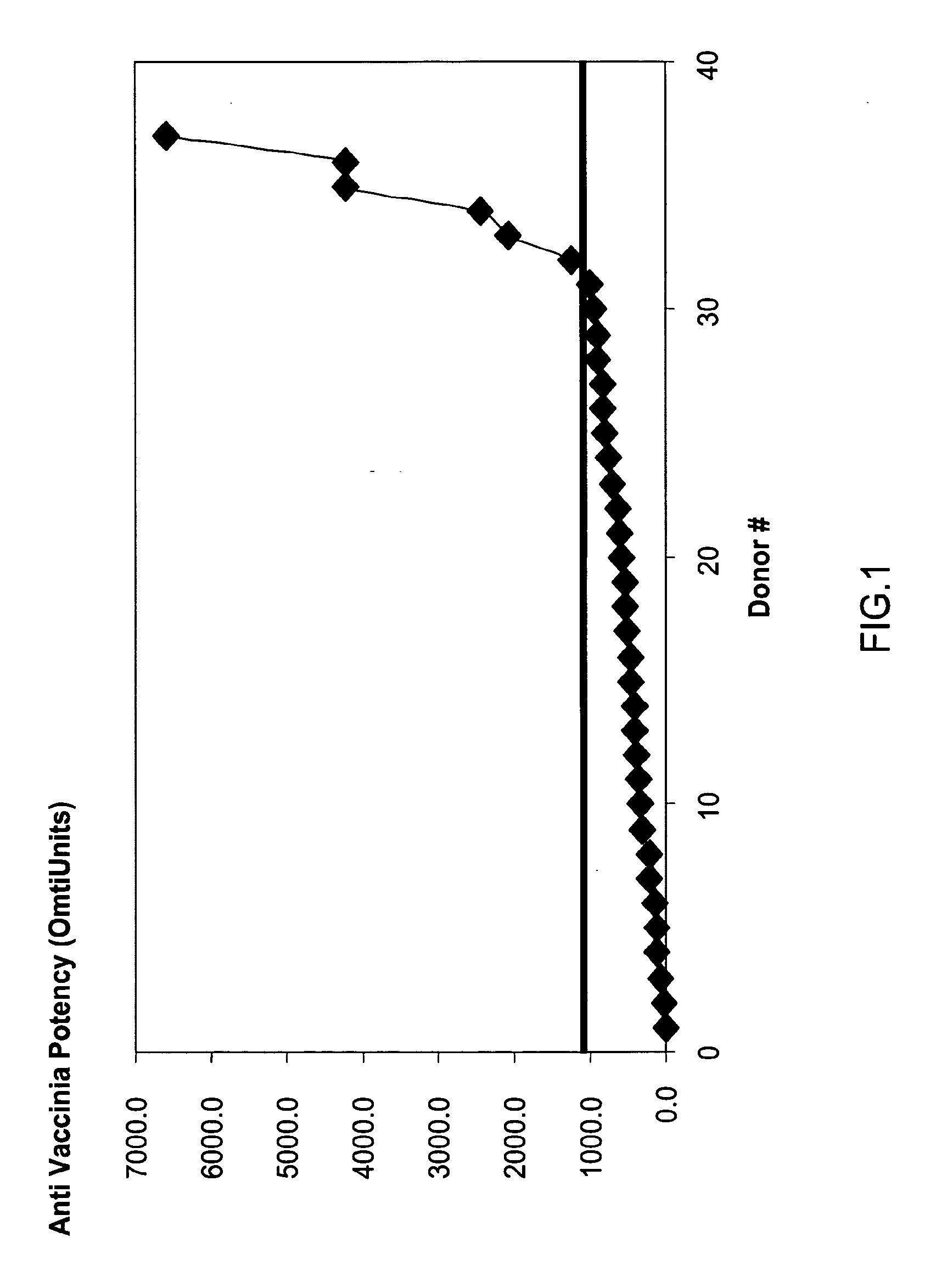
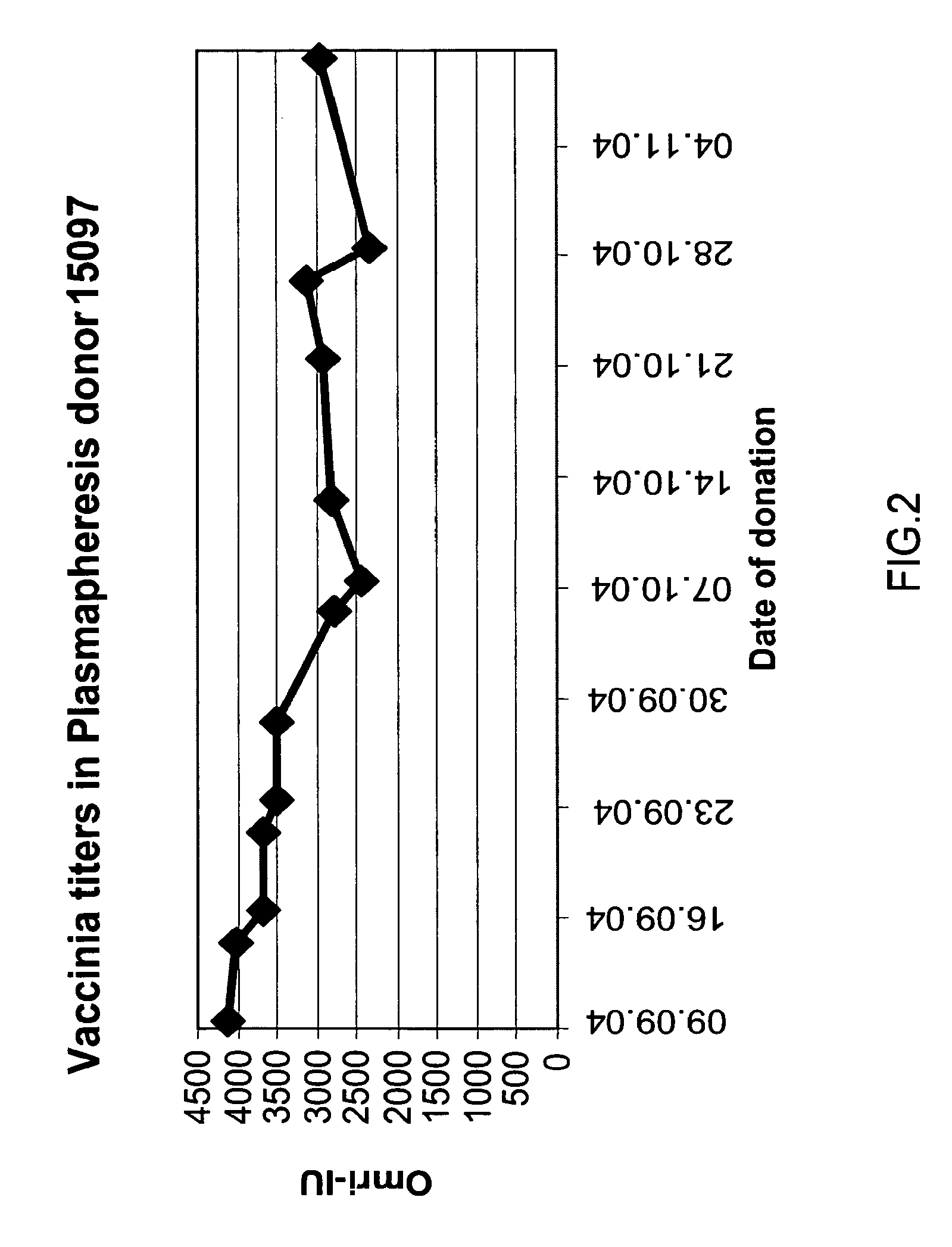
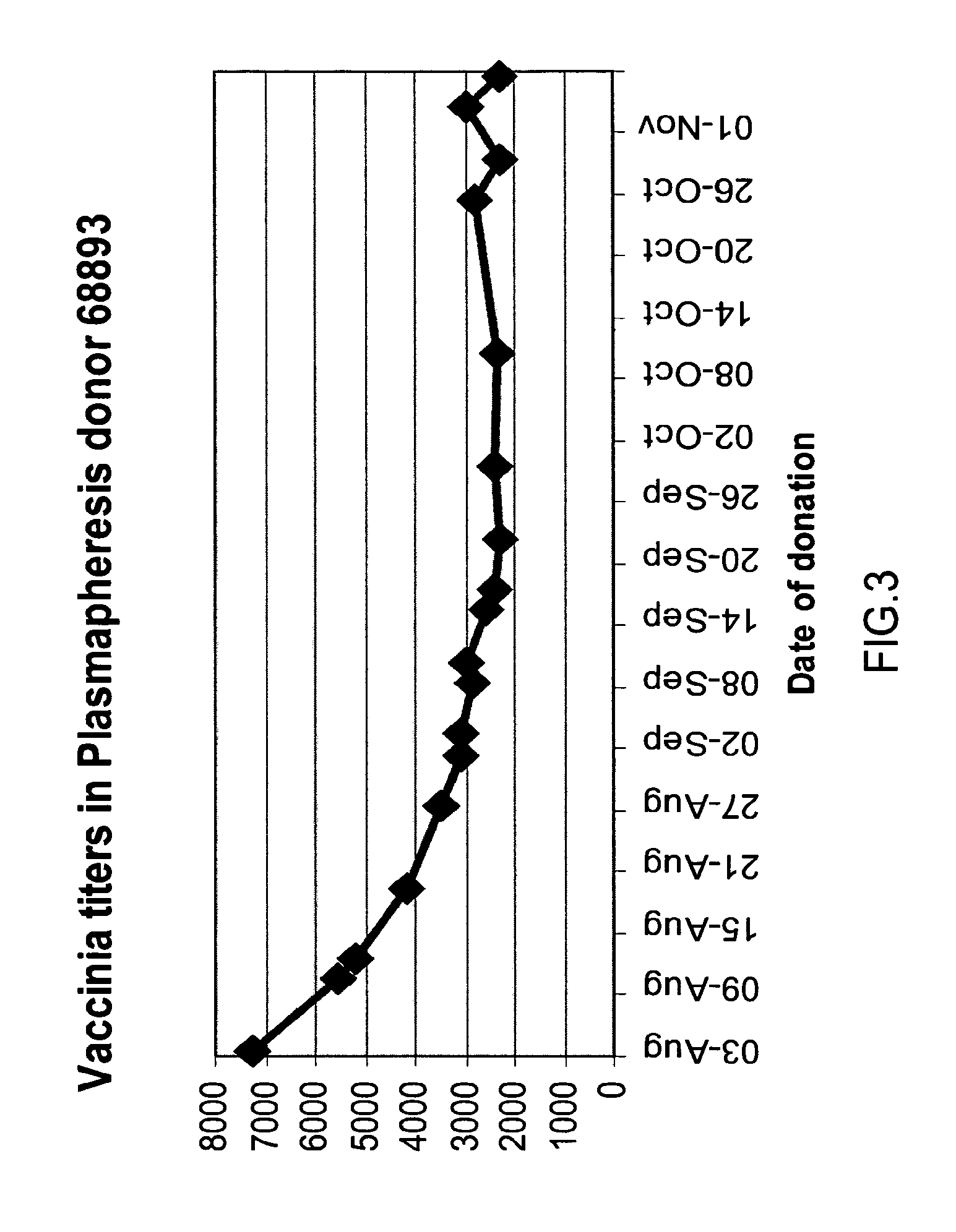
A method and composition for the passive immunization of patients infected with or susceptible to infection from Staphylococcus bacteria such as S. aureus and S. epidermidis infection is provided that includes the selection or preparation of a donor plasma pool with high antibody titers to carefully selected Staphylococcus adhesins or MSCRAMMs, or fragments or components thereof, or sequences with substantial homology thereto. The donor plasma pool can be prepared by combining individual blood or blood component samples which have higher than normal titers of antibodies to one or more of the selected adhesins or other proteins that bind to extracellular matrix proteins, or by administering carefully selected proteins or peptides to a host to induce the expression of desired antibodies, and subsequently recovering the enhanced high titer serum or plasma pool from the treated host.
Owner:PATTI JOSEPH +2
System for rapid production of high-titer and replication-competent adenovirus-free recombinant adenovirus vectors
InactiveUS20090175897A1Shorten production timeLow costSsRNA viruses negative-senseBiocideHeterologousImmunologic function
The present invention relates generally to the fields of gene therapy, immunology, and vaccine technology. More specifically, the invention relates to a novel system that can rapidly generate high titers of adenovirus vectors that are free of replication-competent adenovirus (RCA). Also provided are methods of generating these RCA-free adenoviral vectors, immunogenic or vaccine compositions comprising these RCA-free adenovirus vectors, methods of expressing a heterologous nucleic acid of interest in these adenovirus vectors and methods of eliciting immunogenic responses using these adenovirus vectors.
Owner:TANG DE CHU C +2
Intravenous immunoglobulin composition
ActiveUS20070037170A1High potencyLow volumePeptide/protein ingredientsAntibody mimetics/scaffoldsPopulationSmallpox virus
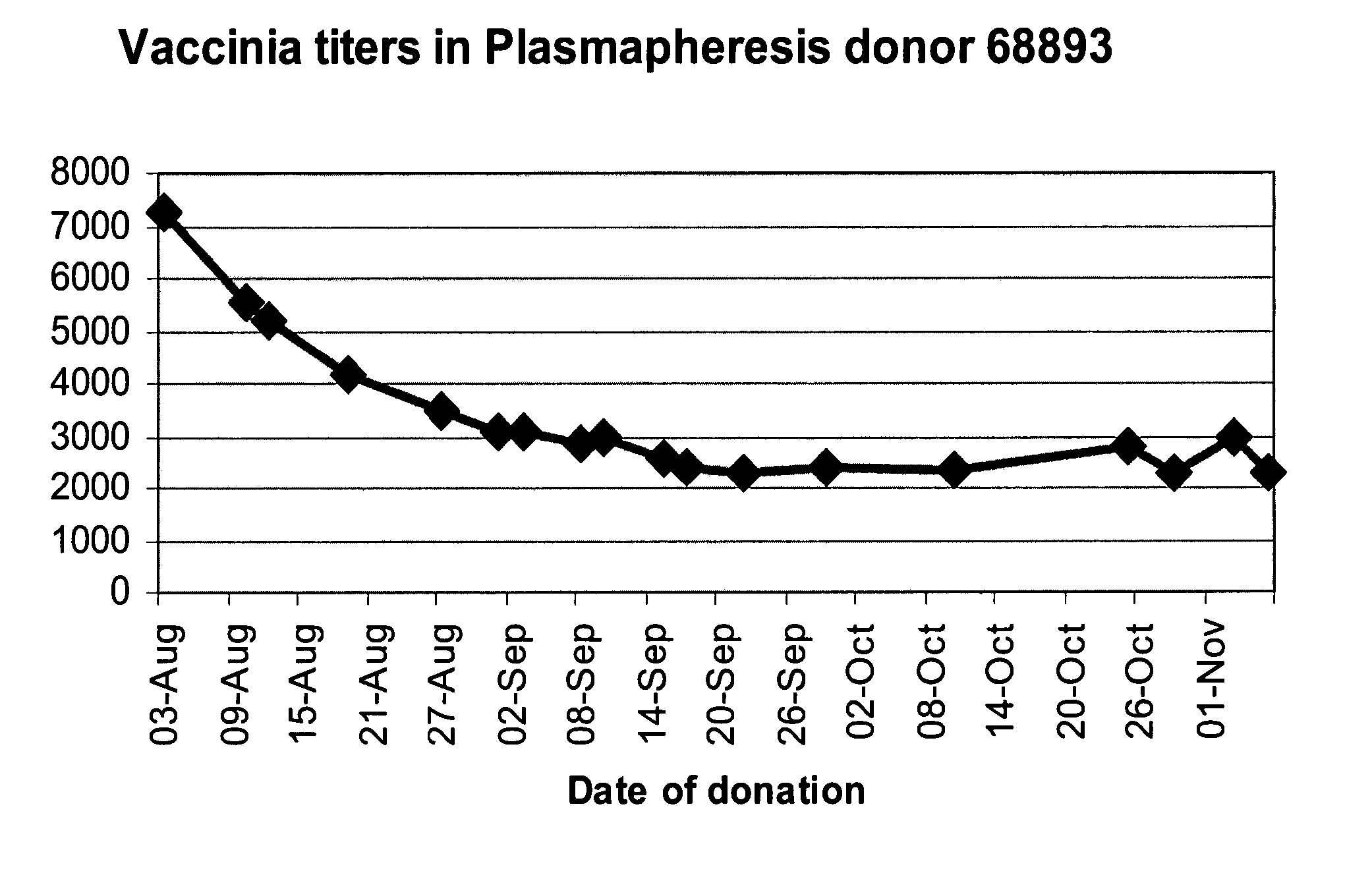
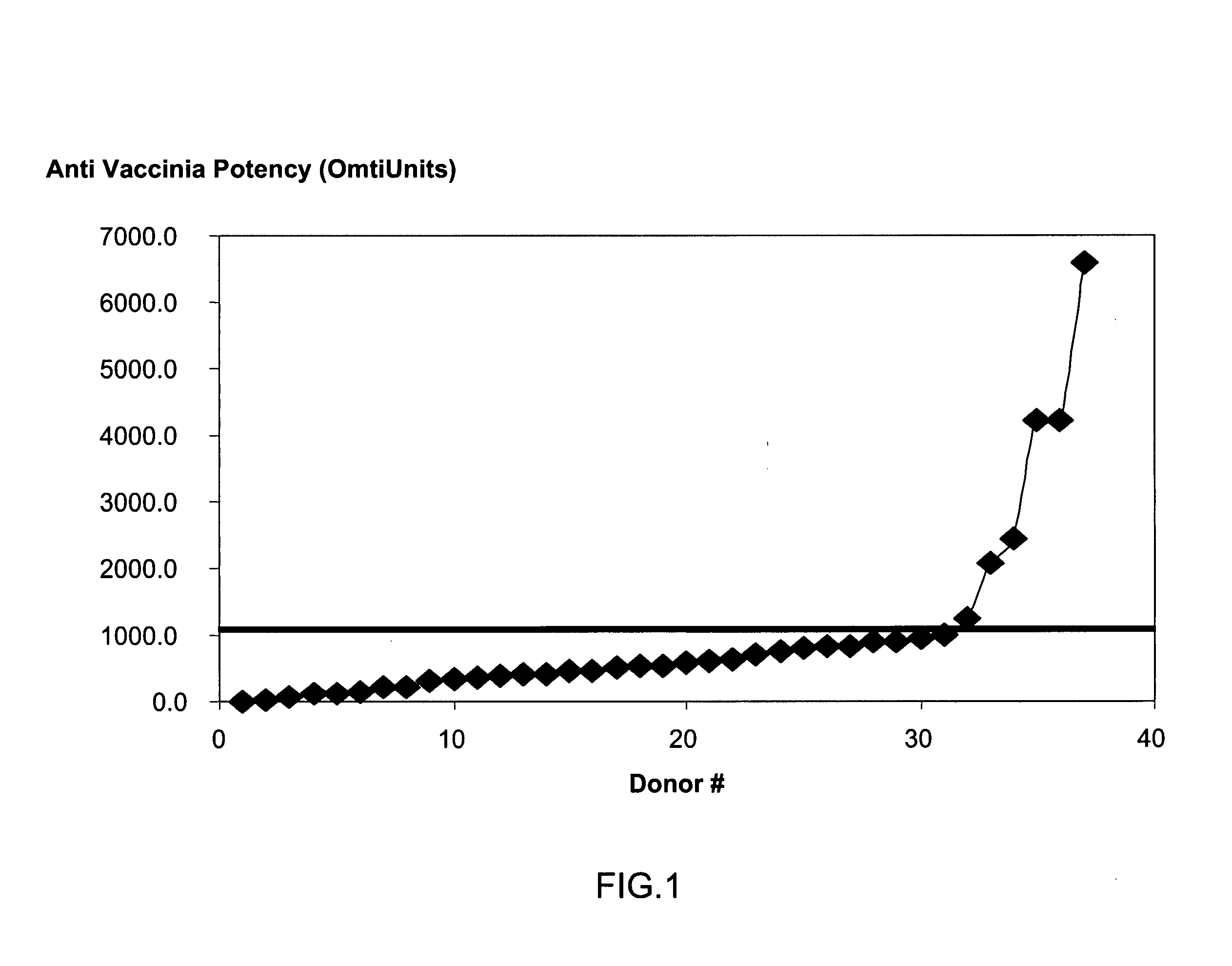
A method for preparing a concentrated, immunoglobulin composition for treating subjects vaccinated against or infected with a pathogenic microorganism, comprising: (a) selecting a population of individuals previously vaccinated against one or more antigens associated with the pathogenic microorganism; (b) determining the level of specific antibodies immunoreactive with the pathogenic microorganism in a blood or blood component of the individuals to identify very high titre individuals having a very high titre of the specific antibodies; (c) combining blood or blood components comprising immunoglobulins from the very high titre individuals; and (d) purifying and / or concentrating the product of step (c), thereby obtaining a concentrated immunoglobulin composition. Also disclosed is a concentrated immunoglobulin composition comprising specific antibodies immunoreactive with a pathogenic microorganism, characterized in that the titre of specific antibodies of the composition is at least 5 times higher than the average titre of specific antibodies of a population of individuals previously vaccinated against one or more antigens associated with the pathogenic microorganism. The composition has a relatively high protein concentration and a low percentage of protein aggregates, and is therefore suitable for both iv and im administration. In a preferred embodiment, the pathogenic microorganism is smallpox virus or vaccinia virus.
Owner:OMRIX BIOPHARM
Preparation method and product of H9N2 subtype avian influenza inactivated vaccine
ActiveCN101816785AHigh titerSimple production methodAntiviralsAntibody medical ingredientsVirus multiplicationVaccine Production
The invention relates to a preparation method and a product of an H9N2 subtype avian influenza inactivated vaccine. The technical points of the invention mainly relate to the screening, the determination and the domestication of a virus-adapted cell line, the primary amplification cultivation and the continuous cultivation of a virus-adapted cell, the preparation of virus fluid by virus-inoculated culture and the preparation of final inactivated vaccine products. Firstly, the invention avoids the virus propagating method using a large amount of chick embryos in the avian influenza production at present, thereby avoiding the problem of biological potential safety hazards, and overcoming the problem that the mass production of vaccines is enslaved to the supply of the chick embryos; secondly, the invention provides a safe, continuous and closed cell culture virus production method, is used for the preparation of the H9N2 subtype avian influenza inactivated vaccine, enables the use of the cell culture method, and can simultaneously produce high-titer viruses to meet the requirements for the immunological production; and finally, the vaccine production method of the invention is simple and fast, thereby realizing the fast vaccine supply at the epidemic situation.
Owner:扬州优邦生物药品有限公司
High titer production of adeno-associated viral vectors
InactiveUS20170029785A1Increase in AAV2 vector titresInhibiting transcription and expressionVector-based foreign material introductionSsDNA virusesGeneticsViral vector
This invention relates to adeno-associated viral (AVV) vectors, to producer cell lines for the production of AAV vectors and to methods of producing such vectors. More specifically, the invention relates to producer cell lines adapted to increase the titre of said vectors and methods of producing AAV vectors using said producer cell lines.
Owner:THE UK SEC FOR HEALTH & HER BRITANNIC MAJESTYS GOVERNMENT OF THE UK OF GREAT BRITAIN & NORTHERN IRELAND
High titer recombinant AAV production
InactiveUS7091029B2Increase final yieldEliminate stepsBiocideGenetic material ingredientsHerpes simplex virus DNAViral vector
The invention includes methods and compositions for the production of high titer recombinant adeno-associated virus (rAAV). The disclosed rAAV are useful in gene therapy applications. Methods are based on the use of recombinant herpes virus vectors and result in highly efficient production of rAAV.
Owner:APPL GENETIC TECH CORP +2
Rhodococcus ruber and application of same as immunologic adjuvant in preparing vaccine
ActiveCN109576180AWill not cause accidental infectionReduce pollutionBacteriaMicroorganism based processesSide effectShort terms
The invention discloses rhodococcus ruber and application of same as an immunologic adjuvant in preparing vaccine. The rhodococcus ruber is also called rhodococcus ruber RDC-01, and the preservation number is CGMCC (China General Microbiological Culture Collection Center) NO. 16640. The rhodococcus ruber disclosed by the invention has the function of increasing and regulating the body immunity andis capable of nonspecifically enhancing the activity of TB (Tuberculosis) lymphocyte, macrophagocyte and NK cells and inducing multiple cell factors such as interferon, and the rhodococcus ruber canbe used as the immunologic adjuvant after being inactivated so as to be added in an oil-adjuvant inactive vaccine, so that generation of an animal antibody induced by the vaccine can be obviously promoted; compared with single use of the oil-adjuvant inactive vaccine, a high-titre antibody can be generated, the use is safe, long-term and short-term toxic and side effects are not generated, and anapplication prospect in the field of preparation of vaccines for animals is good.
Owner:北京利昂盛生物技术有限公司
Production of vaccines
InactiveUS7192759B1Improve purification effectHigh yieldSsRNA viruses negative-sensePeptide/protein ingredientsSerum freeHuman cell
Novel means and methods are provided for the production of mammalian viruses comprising, infecting a culture of immortalized human cells with the virus, incubating the culture infected with virus to propagate the virus under conditions that permit growth of the virus, and to form a virus-containing medium, and removing the virus-containing medium. The viruses can be harvested and be used for the production of vaccines. Advantages are that human cells of the present invention can be cultured under defined serum free conditions, and the cells show improved capability for propagating virus. In particular, methods are provided for producing, in cultured human cells, influenza virus and vaccines derived thereof. This method eliminates the necessity to use whole chicken embryos for the production of influenza vaccines. The method provides also for the continuous or batchwise removal of culture media. As such, the present invention allows the large-scale, continuous production of viruses to a high titer.
Owner:JANSSEN VACCINES & PREVENTION BV
A type foot-and-mouth disease recombinant vaccine strains and preparation method and application thereof
ActiveCN103266091AMultiple phenotype improvements and enhancedPhenotype Improvement and EnhancementMicroorganism based processesAntiviralsAntigenDisease
The invention relates to A type foot-and-mouth disease recombinant vaccine strains prepared by using a reverse genetic manipulation technology and a preparation method and application of the strains. One strain is an A type foot-and-mouth disease recombinant vaccine strain with high titer, antigen matching property and immune protection rate, and the other strain is an A type foot-and-mouth disease recombinant non-pathogenic vaccine strain with high titer, antigen matching property and immune protection rate and without pathogenicity for a host; an antigen nucleotide sequence of each of the vaccine strains is shown as SEQ ID NO: 1; eukaryotic plasmids of viruses can be saved by using a reverse genetic manipulation system; after pigs and cattle are immunized by using the inactivated vaccines prepared from the prepared recombinant vaccine strains, the bodies can be effectively stimulated to produce immune response, and an immune protection effect is provided for the bodies of the pigs and the cattle; through a 10,000-times cattle median infectious dose (BID50) challenge assay of A type AISA topological strains, the immune protection rate reaches 100 percent, and the median protective dose (PD50) is 10.81 to 13.59; and the recombinant vaccine strains can be applied to prevention and control of A type foot-and-mouth disease viruses of China and neighboring countries.
Owner:LANZHOU INST OF VETERINARY SCI CHINESE ACAD OF AGRI SCI
Intravenous immunoglobulin composition
ActiveUS20130011388A1Reduce riskEasy to managePeptide/protein ingredientsAntibody mimetics/scaffoldsSpecific antibodyHigh titre
A concentrated, immunoglobulin composition for treating subjects vaccinated against or infected with a pathogenic microorganism, is made by (a) selecting a population of individuals previously vaccinated against antigens associated with the pathogenic microorganism; (b) identifying very high titre individuals by determining the level of specific antibodies immunoreactive with the pathogenic microorganism in the blood of the individuals; (c) combining blood from the very high titre individuals; and (d) purifying and / or concentrating the product of step (c). A concentrated immunoglobulin composition can include specific antibodies immunoreactive with a pathogenic microorganism, wherein the titre of specific antibodies is at least 5 times higher than the average titre of specific antibodies of a population of individuals previously vaccinated against antigens associated with the pathogenic microorganism. The composition has a relatively high protein concentration and a low percentage of protein aggregates. The pathogenic microorganism is preferably smallpox virus or vaccinia virus.
Owner:OMRIX BIOPHARM
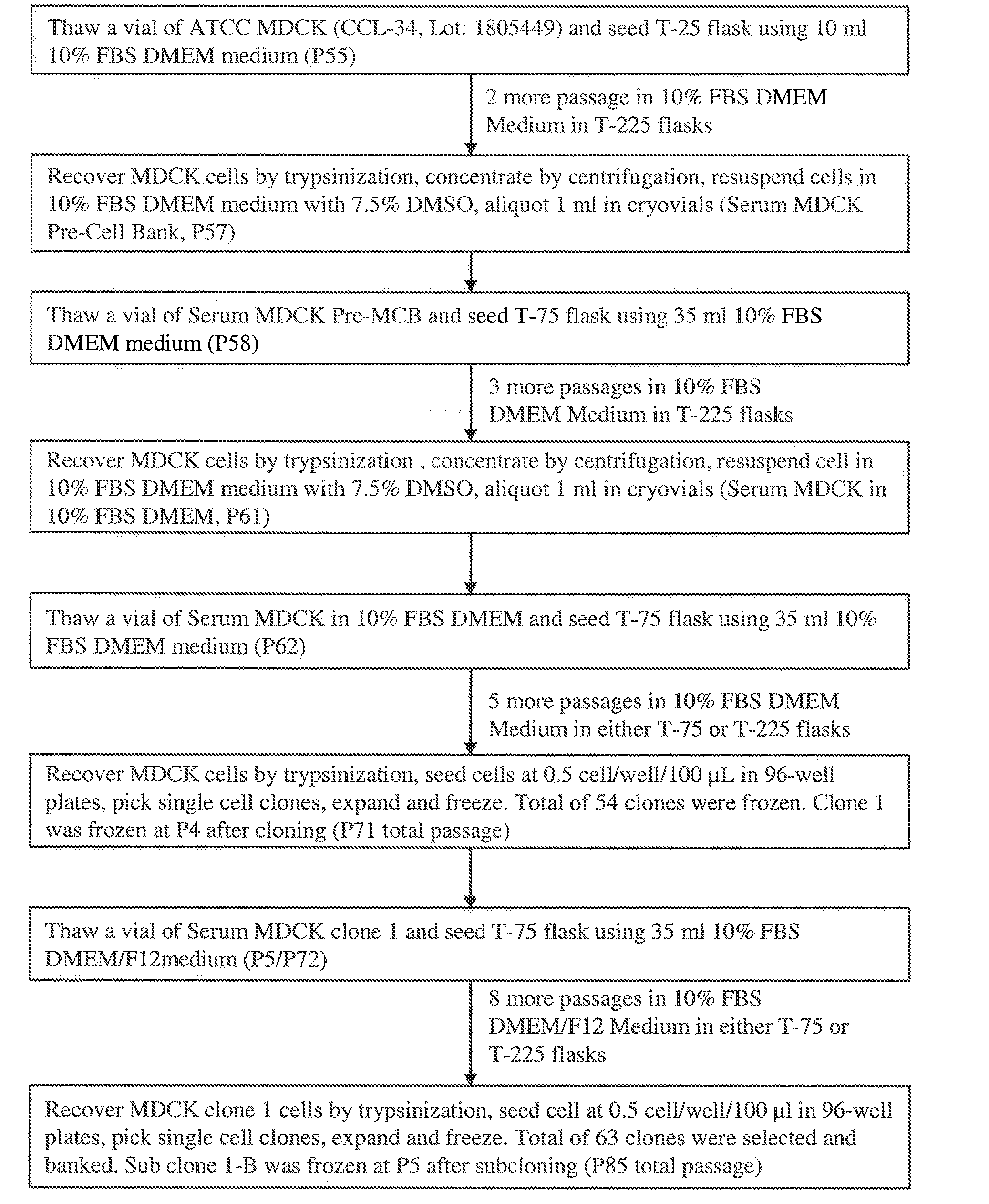
Mdck cell lines supporting viral growth to high titers and bioreactor process using the same
The present invention relates to novel MDCK cells which can be to grow viruses, e.g., influenza viruses, in cell culture to higher titer than previously possible. The MDCK cells can be adapted to serum-free culture medium. The present invention further relates to cell culture compositions comprising the MDCK cells and cultivation methods for growing the MDCK cells. The present invention further relates to methods for producing influenza viruses in cell culture using the MDCK cells of the invention.
Owner:MEDIMMUNE LLC
Chimeric viral vectors for gene therapy
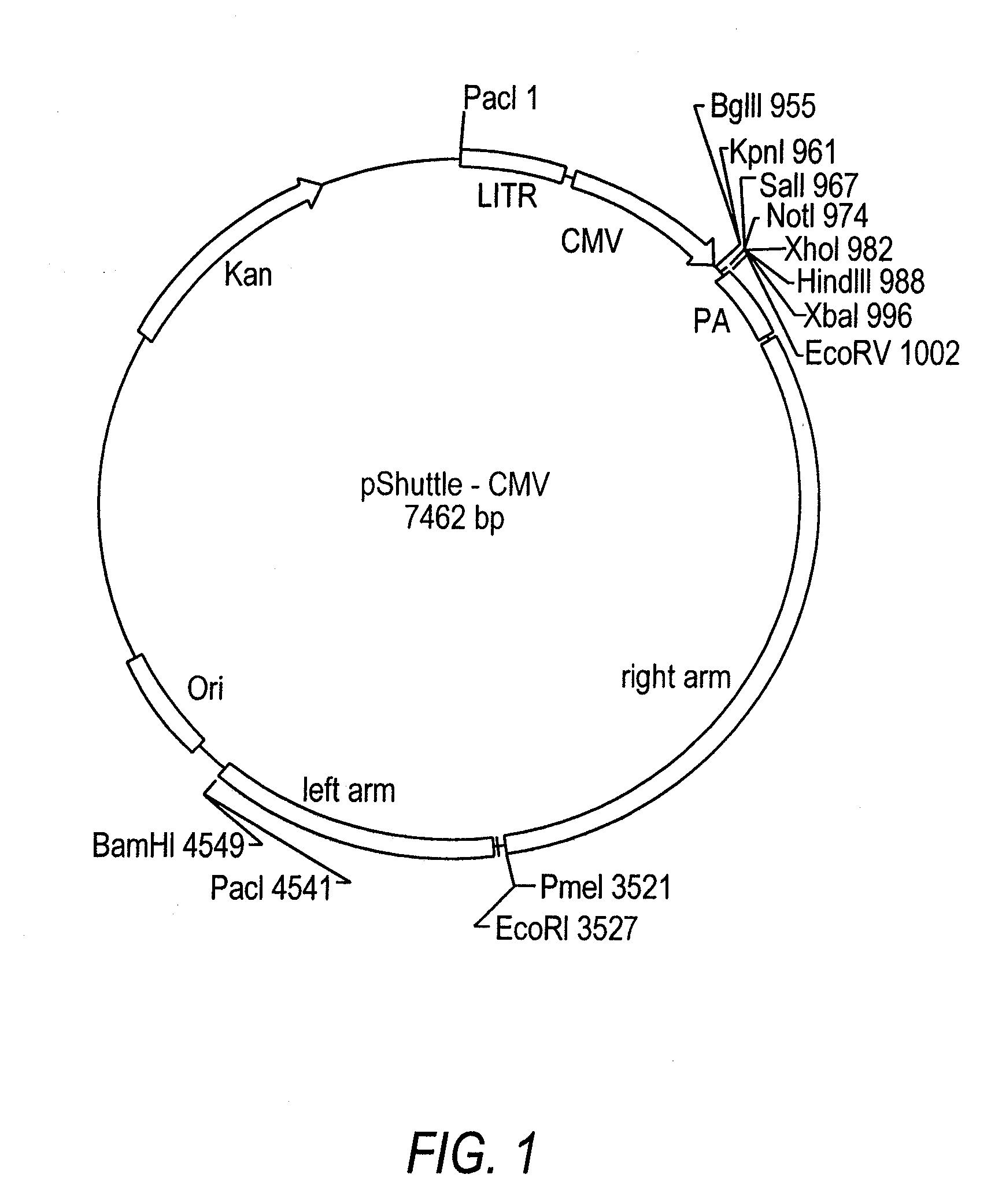
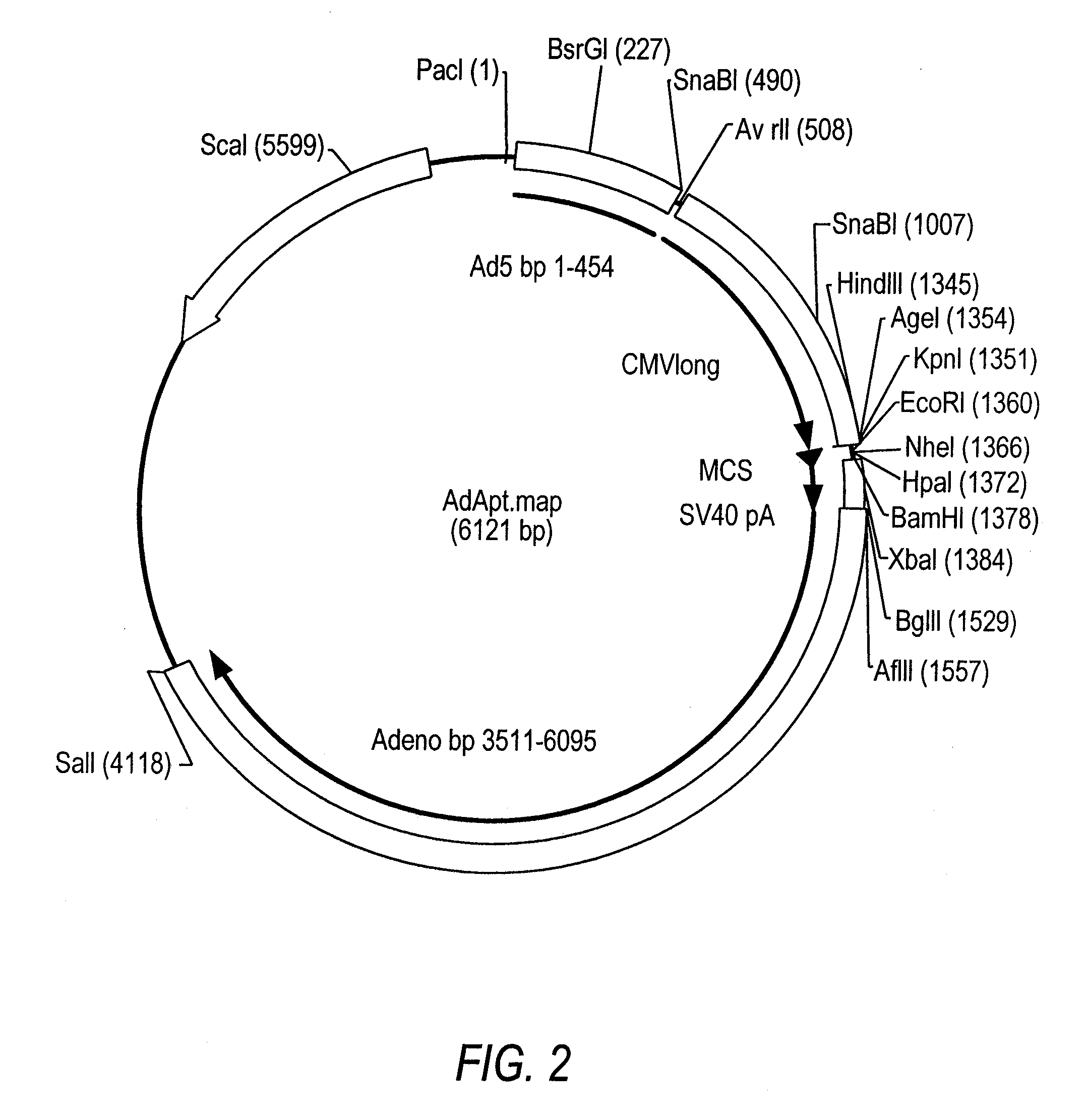
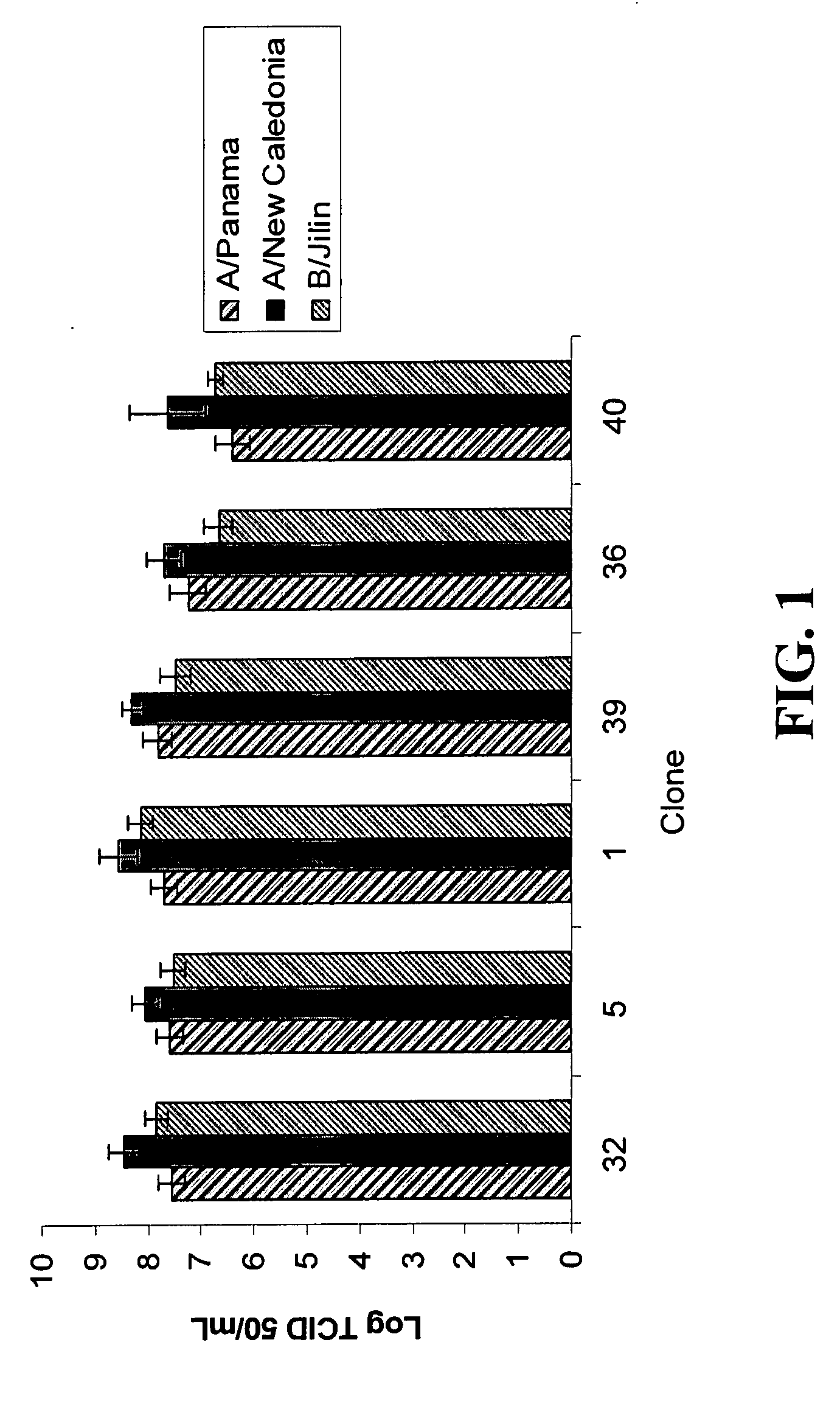
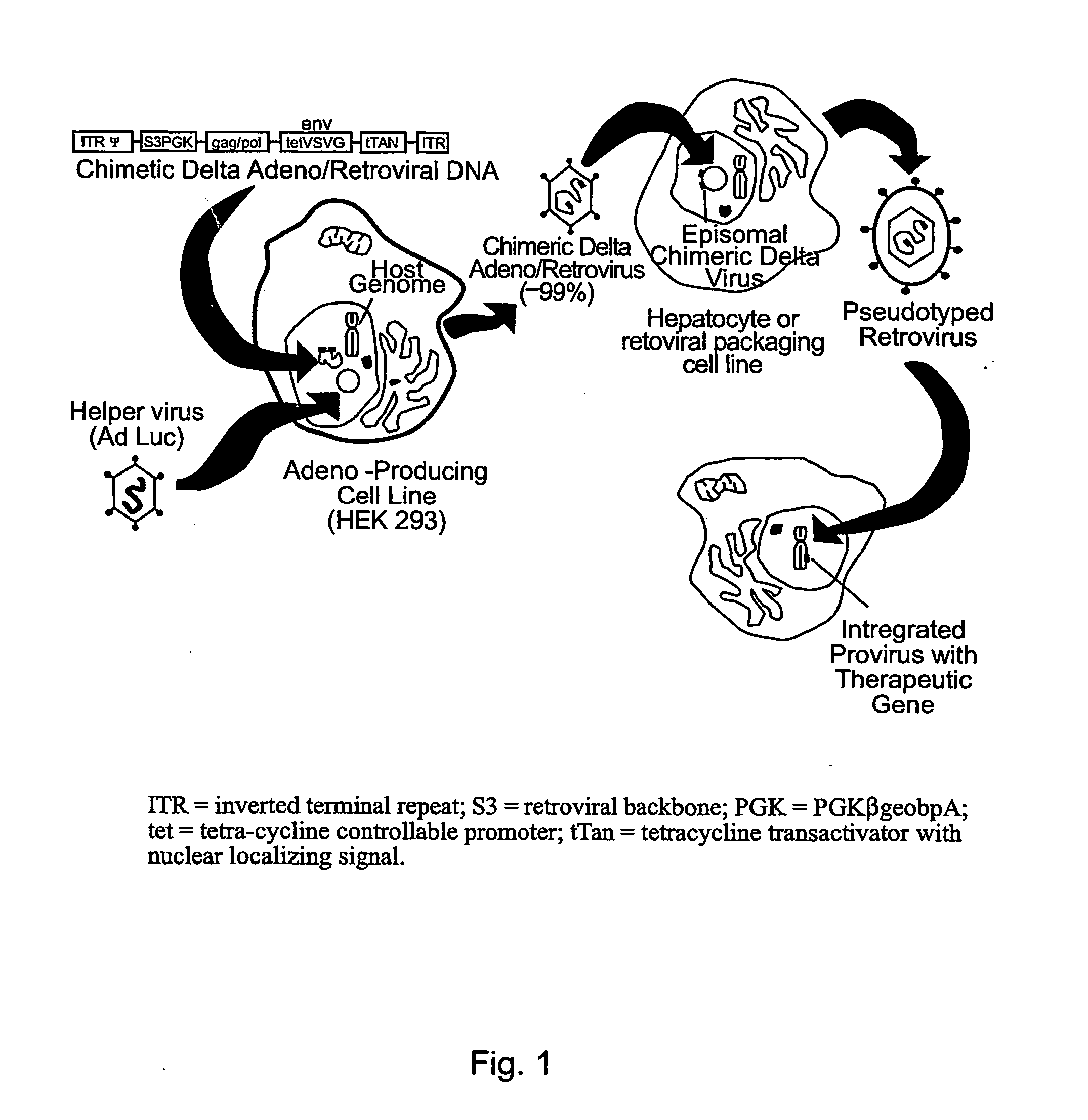
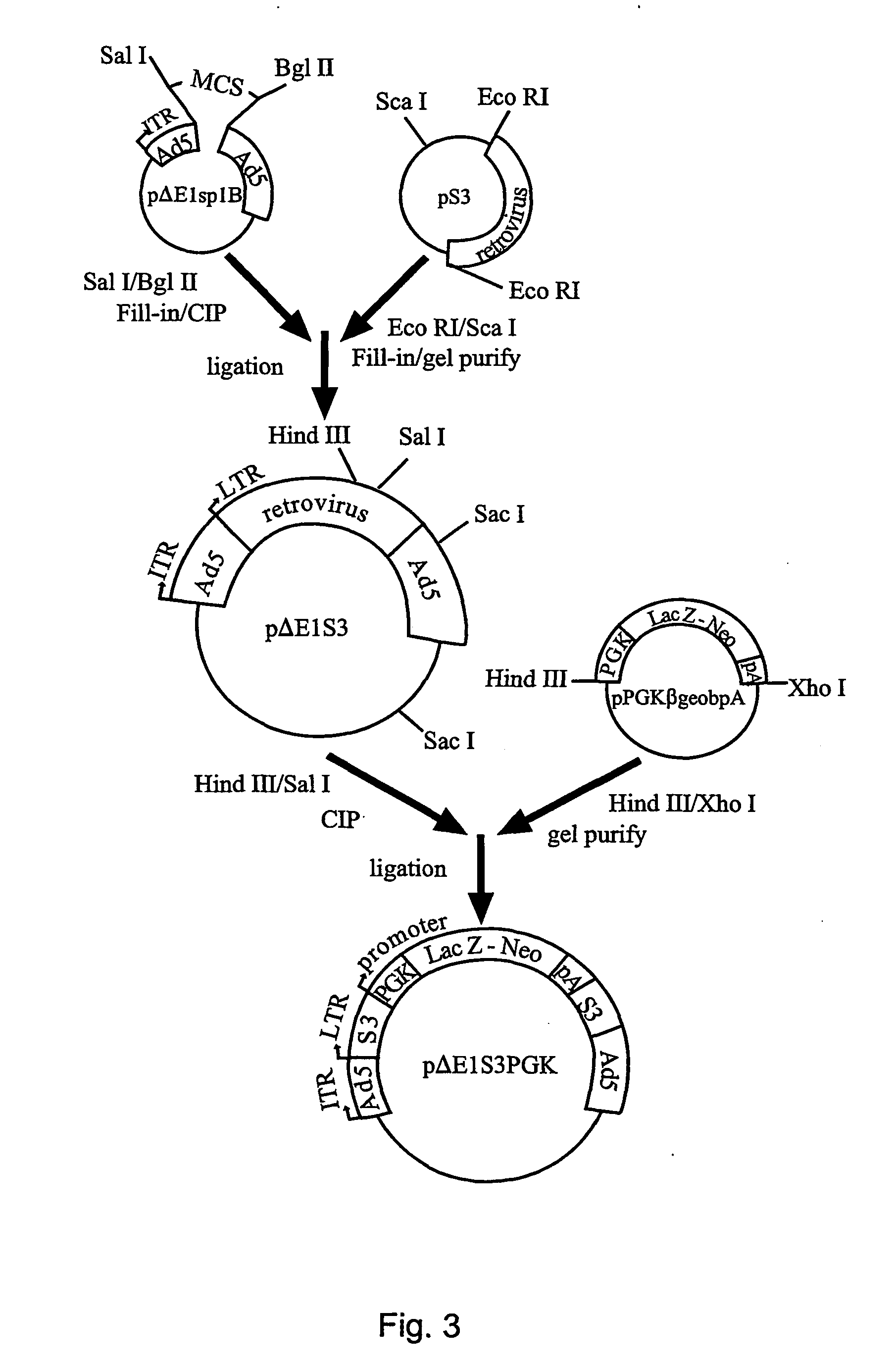
The invention relates to a single nucleic acid vector comprising both adenoviral and retroviral sequences for gene therapy. The vectors described herein (See FIG. 2) are capable of transducing all cis and trans components of a retroviral vector for the generation of high titer recombinant retroviral vectors. The chimeric vectors are used for the delivery and stable integration of therapeutic constructs and eliminate limitations currently encountered with in vivo gene transfer application.
Owner:AGUILAR CORDOVA ESTUARDO
Low serum efficiency mycoplasma gallisepticam attenuated strain culture medium and preparation method thereof
ActiveCN103074246AIncrease the titer of live bacteriaReduce allergic reactionsBacteriaMicroorganism based processesAntigenCulture mediums
The present invention relates to a low serum efficiency mycoplasma gallisepticam attenuated strain culture medium and a preparation method thereof, and belongs to the technical field of veterinary biology. The culture medium comprises: (1) a base culture medium; and (2) an auxiliary culture medium, wherein the auxiliary culture medium mainly comprises MEM, yeast extract powder, tryptone, glucose, an inorganic salt, and the like, and growth, high titer and stability of the semi-finished product can be ensured with the auxiliary culture medium. According to the present invention, a titer of the semi-finished product bacterial liquid prepared by using the preparation method is up to 10<11> CCU / ml; and the culture medium adopts reduced pig serum to culture mycoplasma gallisepticam so as to reduce allergic stress reactions on chicken by heterologous pig serum, consider animal biosafety, improve antigen titer, and reduce production cost.
Owner:兆丰华生物科技(南京)有限公司
High titer antibody production
InactiveUS20110229933A1Bioreactor/fermenter combinationsBiological substance pretreatmentsCell culture mediaAntibody production
Owner:MERCK SHARP & DOHME CORP
Hybridoma cell line 3G1 and anti-alfatoxin B1 monoclonal antibody produced by the same
ActiveCN102747043AHigh sensitivityStrong specificityBiological material analysisTissue cultureAflatoxin BELISA unit
The present invention relates to a hybridoma cell line 3G1 and an anti-alfatoxin B1 monoclonal antibody produced by the hybridoma cell line 3G1. The hybridoma cell line 3G1 (CCTCCNO.C201014) can be used for preparation of a high titer anti-aflatoxin B1 monoclonal antibody, wherein an enzyme-linked immunosorbent assay (ELISA) method is adopted to determine a titer, and the titer is 6.40*10<6>. The anti-aflatoxin B1 monoclonal antibody of the present invention has characteristics of high sensitivity and good specificity, wherein 50% inhibiting concentration on aflatoxin B1 by the monoclonal antibody is 1.6 ng / mL, cross reaction rate with aflatoxin B2 is 6.4%, and cross reaction rates with aflatoxin G1 and G2 are less than 1%. In addition, the anti-aflatoxin B1 monoclonal antibody of the present invention can be used for determination of aflatoxin B1.
Owner:INST OF OIL CROPS RES CHINESE ACAD OF AGRI SCI
Production of poliovirus at high titers for vaccine production
ActiveUS8546123B2High potencyShort processNervous disorderSsRNA viruses positive-senseSerum freeVaccine Production
Provided is a process for the production of poliovirus, comprising the steps of:a) providing a serum-free suspension culture of cells, which are primary human retina (HER) cells that have been immortalized by expression of adenovirus E1 sequences,b) infecting the cells with poliovirus, at a cell density of between 2×106 cells / ml and 150×106 cells / ml, and c) harvesting poliovirus at a time of between 12 and 48 hours after infection.
Owner:JANSSEN VACCINES & PREVENTION BV
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com