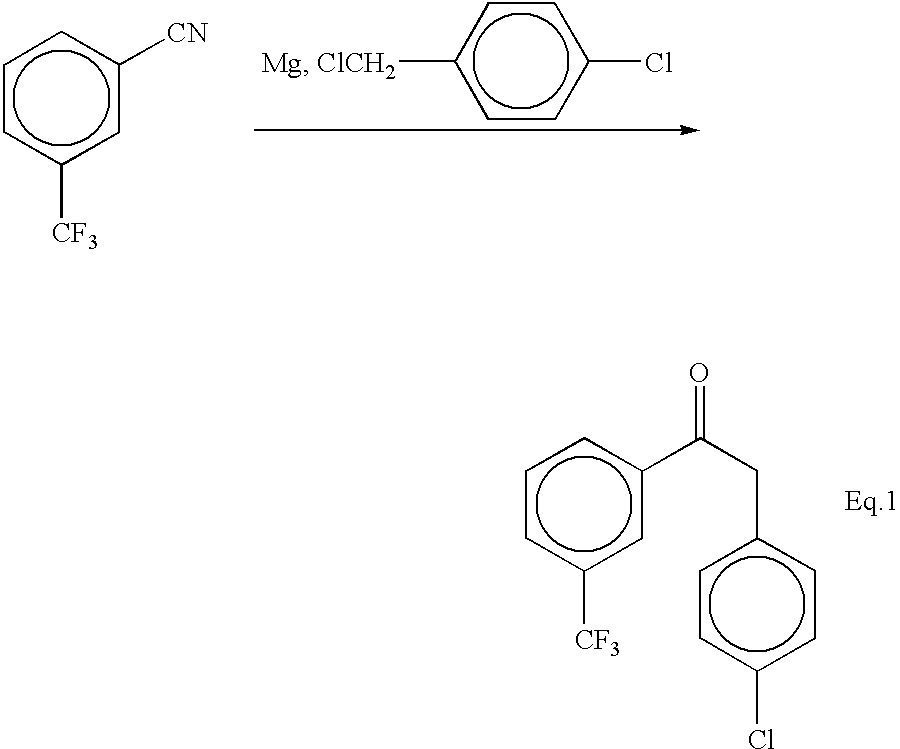
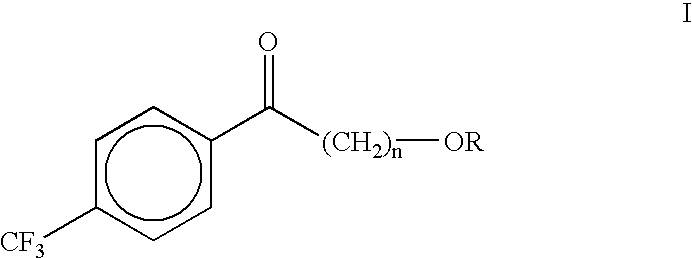
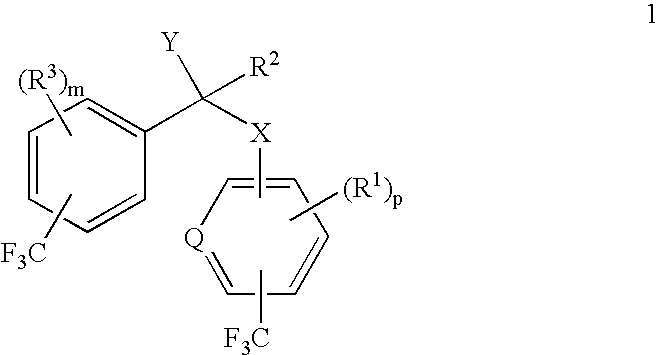
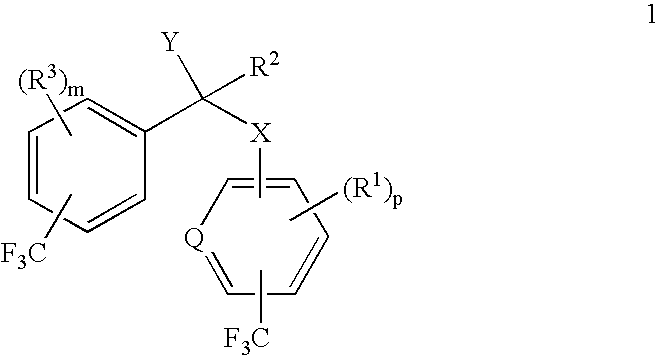
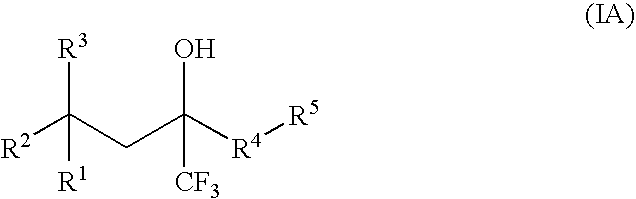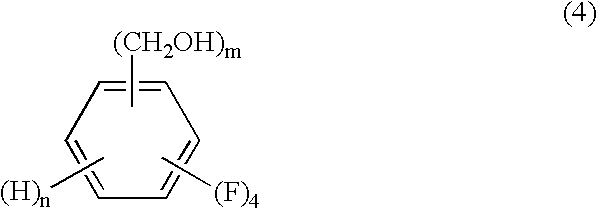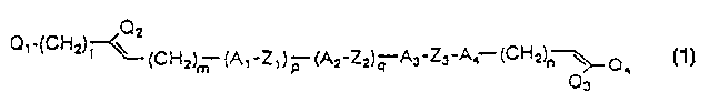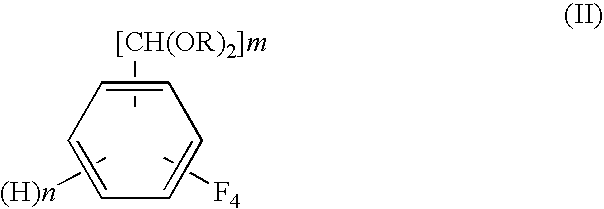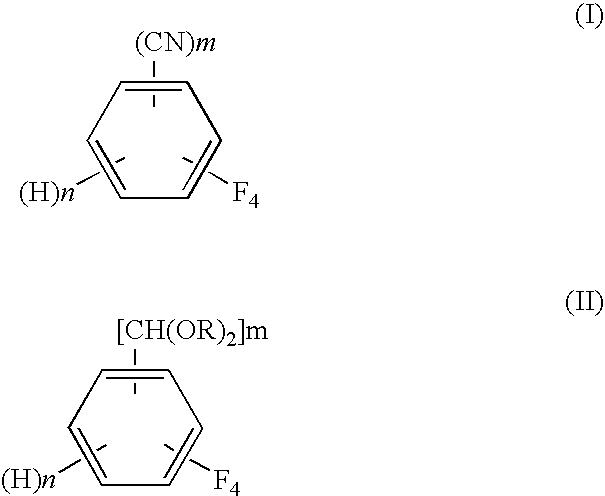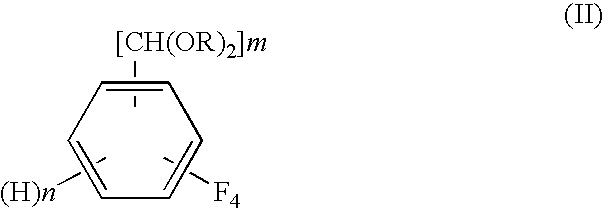Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
52results about "Preparation by nitrile reduction and hydrolysis" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
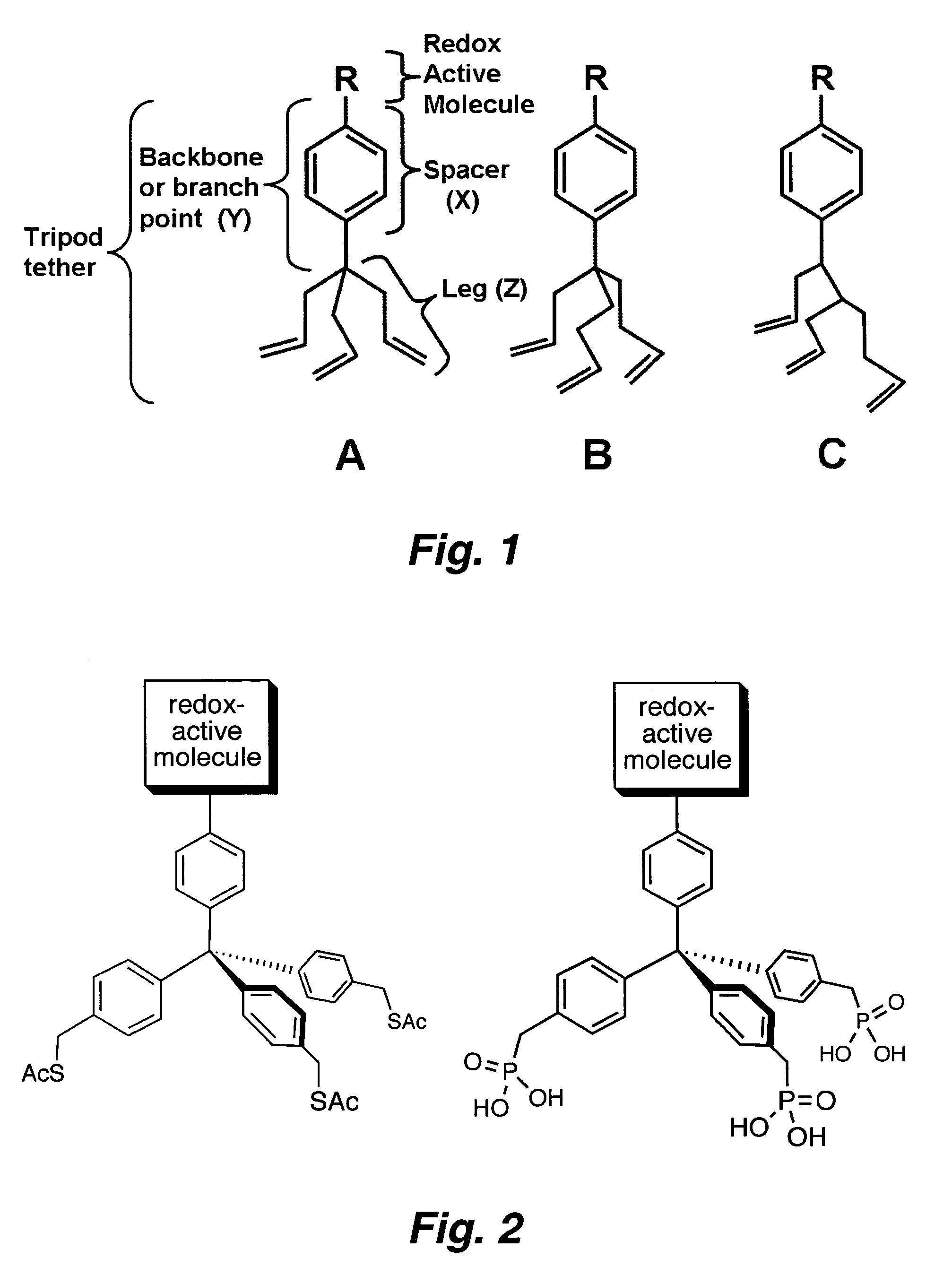
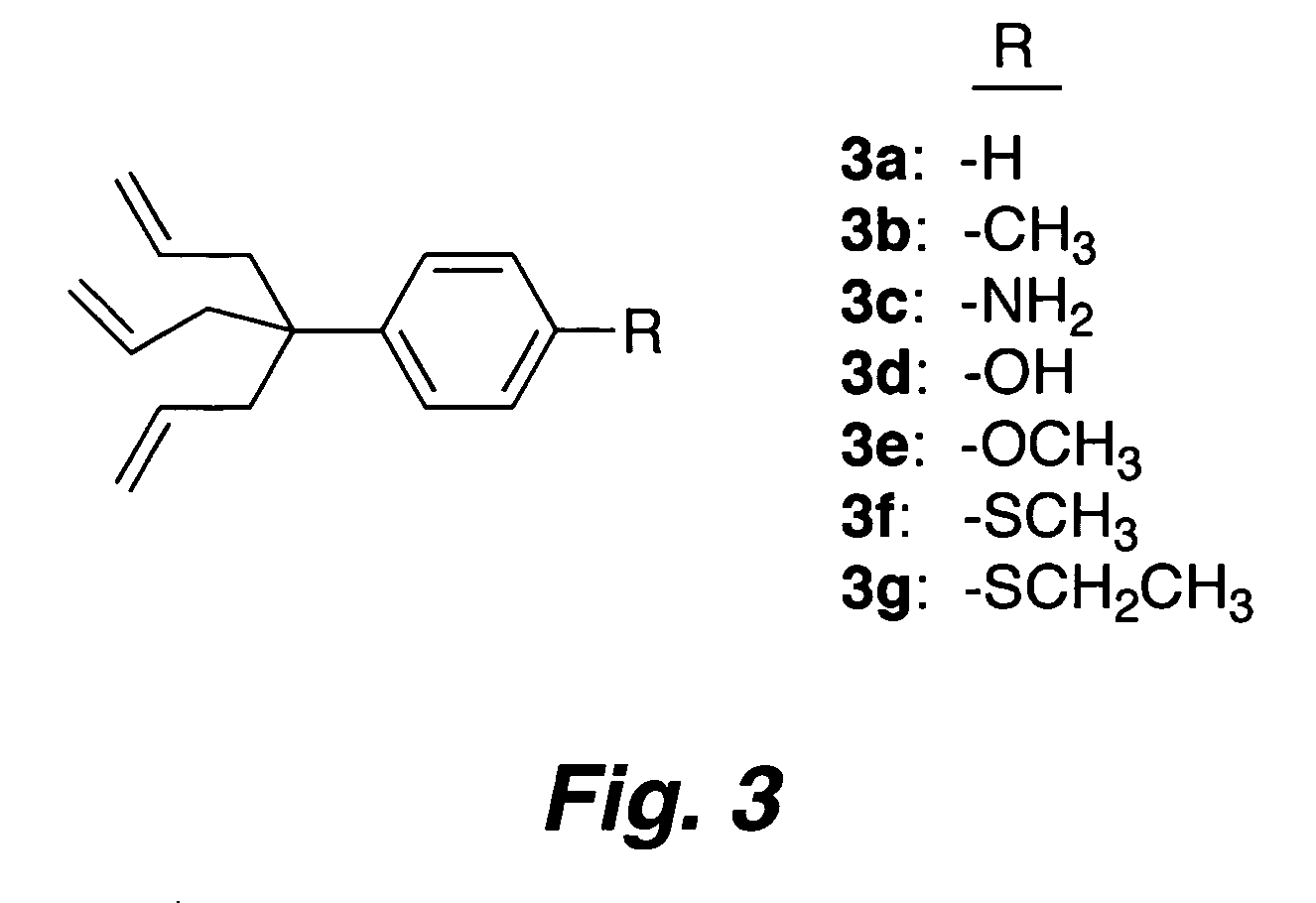
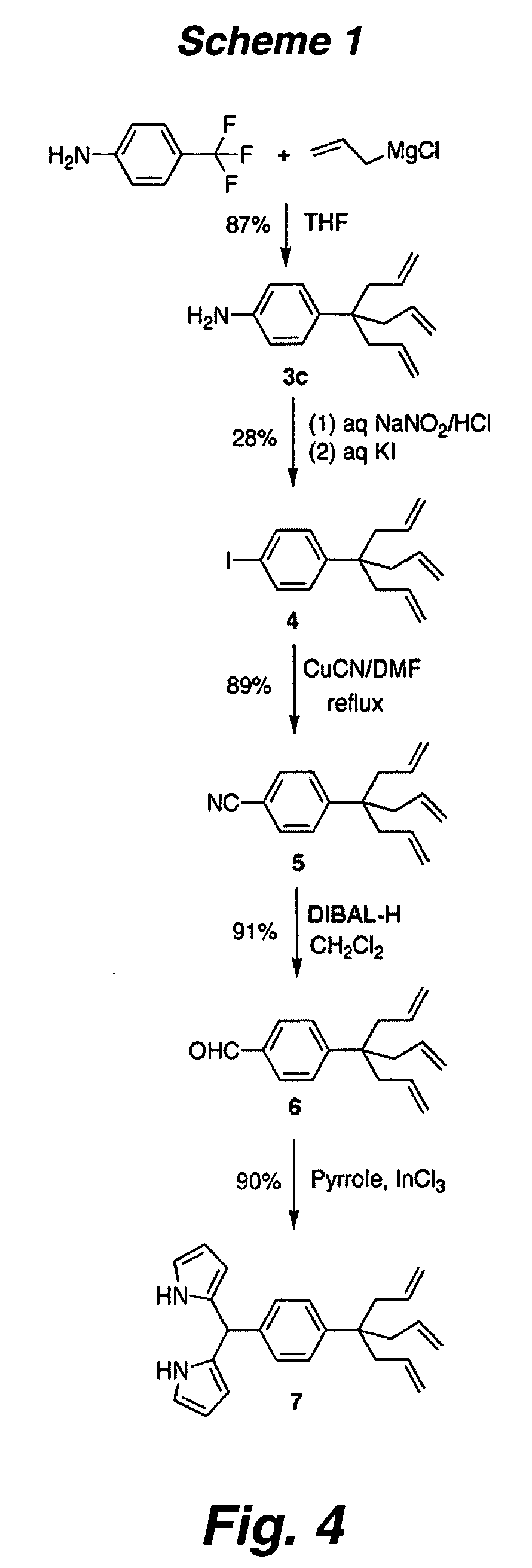
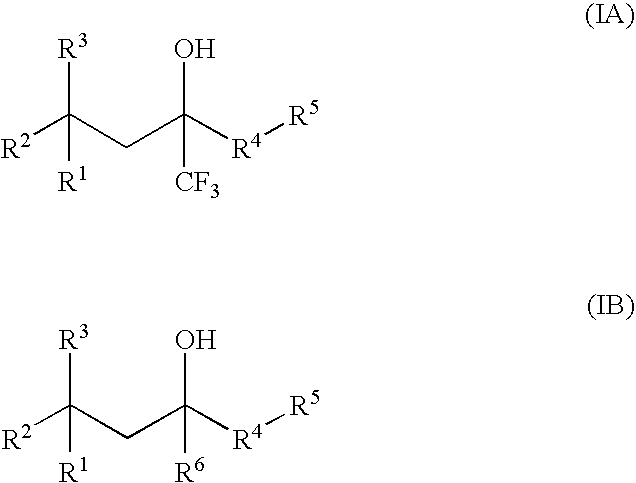
Multypodal tethers for high-density attachment of redox-active moieties to substrates
InactiveUS20070108438A1Increase charge storage densityHigh charge densityMaterial nanotechnologyNanoinformaticsHigh densityCombinatorial chemistry
This invention provides redox-active molecules attached to polypodal (e.g., bipodal, tripodal, quadrapodal, pentapodal, etc.) tethers that can be used for attachment of the redox-active molecules to a substrate (e.g., an electrode). The tethered redox-active molecules are useful for the fabrication of memory devices.
Owner:RGT UNIV OF CALIFORNIA +2
Triaryl methane compounds and analogues thereof useful for the treatment or prevention of sickle cell disease or diseases characterized by abnormal cell proliferation
InactiveUS6028103AReducing sickle erythrocyte dehydrationDelaying occurrenceHalogenated hydrocarbon active ingredientsBiocideAbnormal cellSickled erythrocytes
The present invention provides a class of chemical compounds useful as efficacious drugs in the treatment of sickle cell disease and diseases characterized by unwanted or abnormal cell proliferation. The active compounds are substituted triaryl methane compounds or analogues thereof where one or more of the aryl groups is replaced with a heteroaryl, cycloalkyl or heterocycloalkyl group and / or the tertiary carbon atom is replaced with a different atom such as Si, Ge, N or P. The compounds inhibit mammalian cell proliferation, inhibit the Gardos channel of erythrocytes, reduce sickle erythrocyte dehydration and / or delay the occurrence of erythrocyte sickling or deformation.
Owner:HARVARD COLLEGE PRESIDENT & FELLOWS OF +2
Glucocorticoid mimetics, methods of making them, pharmaceutical compositions, and uses thereof
Compounds of Formula (IA) and Formula (IB)wherein R1, R2, R3, R4, R5, and R6 are as defined herein for Formula (IA) or Formula (IB), or a tautomer, prodrug, solvate, or salt thereof; pharmaceutical compositions containing such compounds, and methods of modulating the glucocorticoid receptor function and methods of treating disease-states or conditions mediated by the glucocorticoid receptor function or characterized by inflammatory, allergic, or proliferative processes in a patient using these compounds.
Owner:BOEHRINGER INGELHEIM PHARMA INC
Oligo(p-phenylene vinylene) amphiphiles and methods for self-assembly
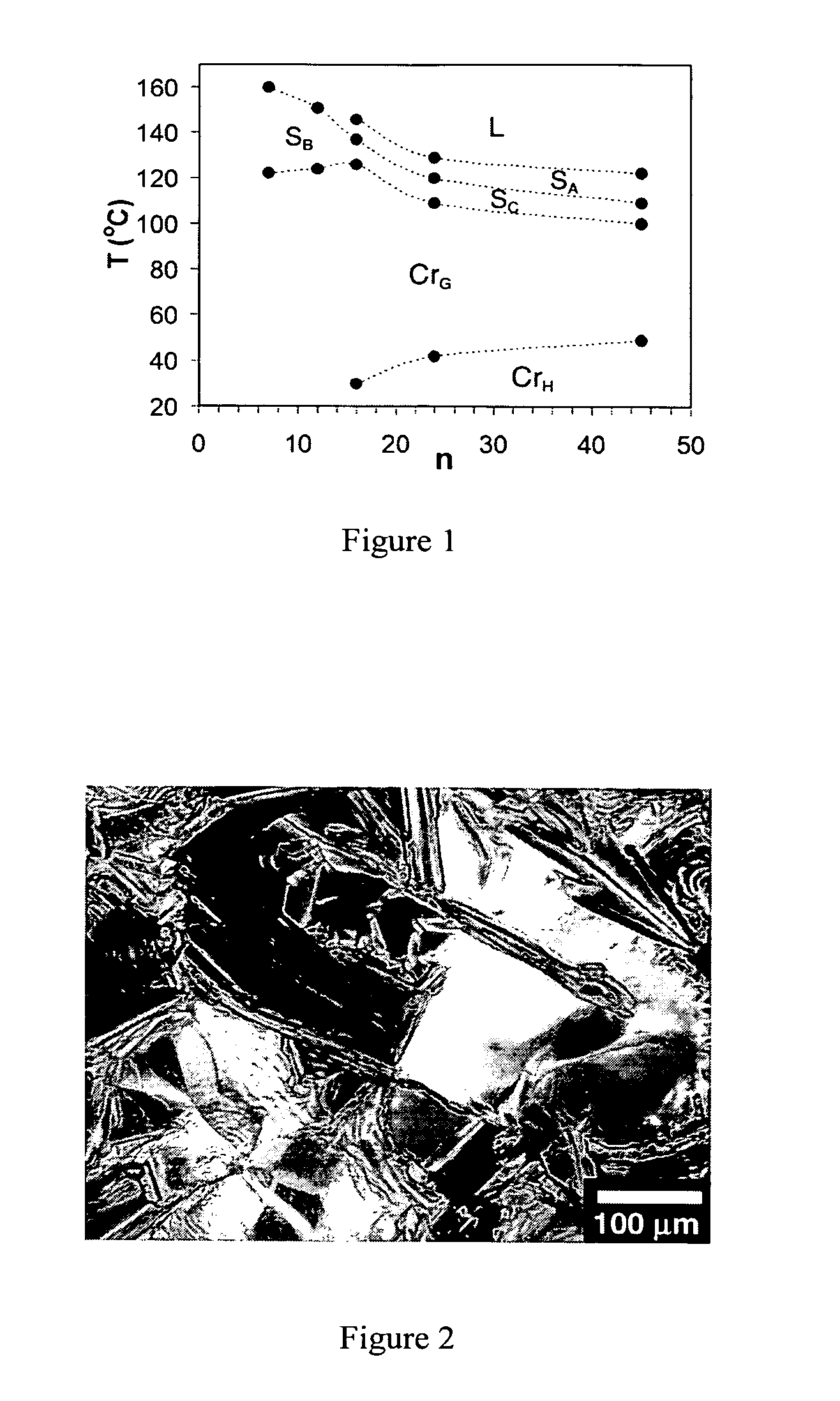
InactiveUS20050214952A1High carrier mobilityLiquid crystal compositionsOrganic compound preparationAmphiphileSelf-assembly
Owner:NORTHWESTERN UNIV
Process for producing tetrafluorobenzenemethanols
InactiveUS6624336B1High purityHigh yieldOrganic compound preparationHydroxy compound preparationOrganic chemistryCyclopropanecarboxylic acid
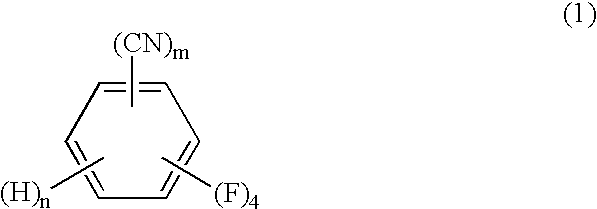
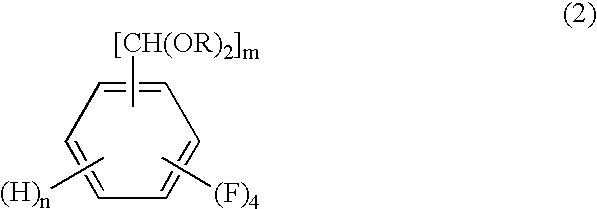
The present invention relates to a process by a series of reactions using tetrafluorocyanobenzens as material for producing tetrafluorobenzenemethanols, tetrafluorobenzenecarbaldehyde dialkylacetals and tetrafluorobenzenecarbaldehydes in a high purity and a high yield which are useful as intermediates in the production of cyclopropanecarboxylic acid esters having insecticidal action, and also relates to a novel tetrafluorobenzenecarbaldehyde dimethylacetal.
Owner:SHOWA DENKO KK
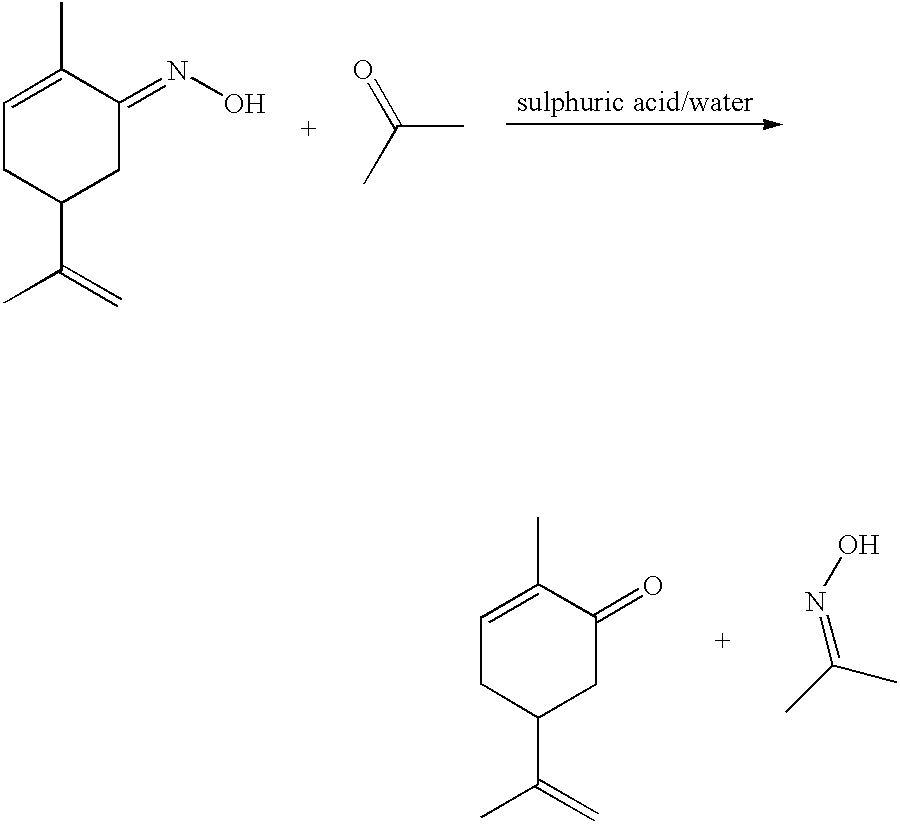
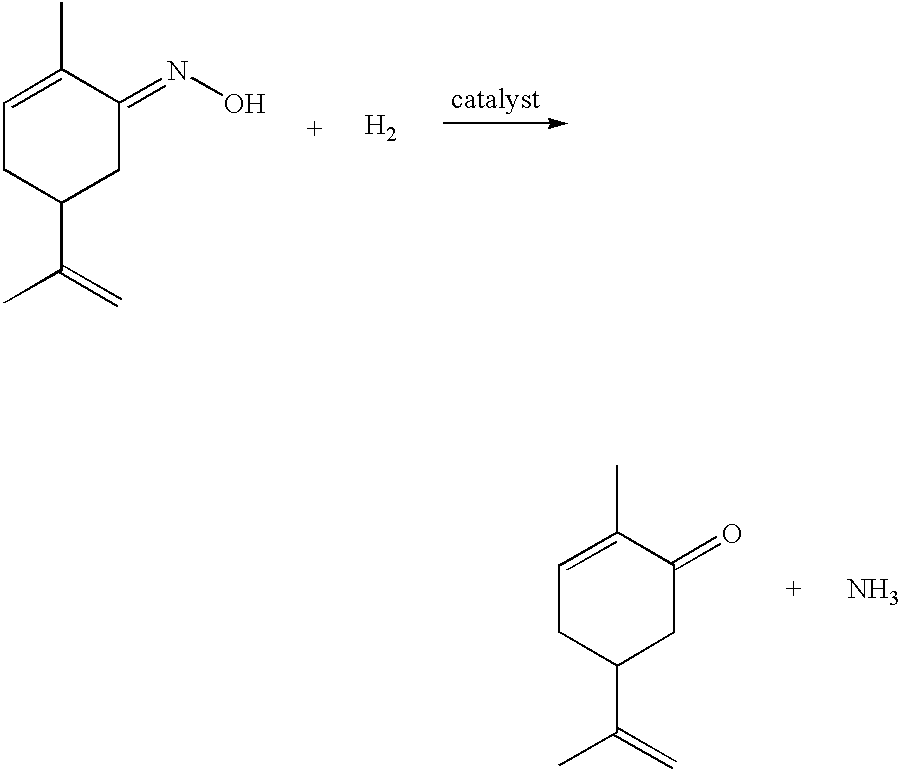
Preparation of carvone
InactiveCN1342135APhysical/chemical process catalystsOrganic compound preparationPtru catalystInorganic materials
A process for the preparation of carvone comprises hydrogenating carvoxime in the presence of a selecively poisoned catalyst. Preferred catalysts include noble metals supported on inorganic materials poisoned with a catalyst modifier. In a preferred embodiment of the process defined herein, the crude carvone reaction product produced in accordance with the process of the invention, is purified by treating the crude carvone product with an organometallic compound M(X)n wherein M is a polyvalent metal, n is the valence of M and X denotes an inorganic or organic atom or group.
Owner:快思特国际公司
Bisalkenyl derivatives, liquid crystalline compound and liquid crystal composition
InactiveCN1110470CExtended temperature rangeLower threshold voltageLiquid crystal compositionsSilicon organic compoundsLiquid crystallineCrystallography
Liquid crystalline compounds expressed by the following formula (1), liquid crystal compositions thereof obtained by combination with specified liquid crystal compounds, and liquid crystal display devices using them,wherein, A1, A2, A3 and A4 denote each independently trans-1,4-cyclohexylene group etc.; Z1, Z2 and Z3 denote each independently -(CH2)2- etc.; Q1 and Q2 denote each independently H, F, Cl or Br; Q3 and Q4 denote each independently F, Cl or Br; 1, m and n denote each independently an integer of from 0 to 5; and p and q denote each independently an integer of 0 or 1.
Owner:CHISSO CORP
Oligo(p-phenylene vinylene) amphiphiles and methods for self-assembly
InactiveUS7371887B2StructuredHigh carrier mobilityLiquid crystal compositionsOrganic compound preparationAmphiphileSelf-assembly
Owner:NORTHWESTERN UNIV
Propellane derivates and synthesis
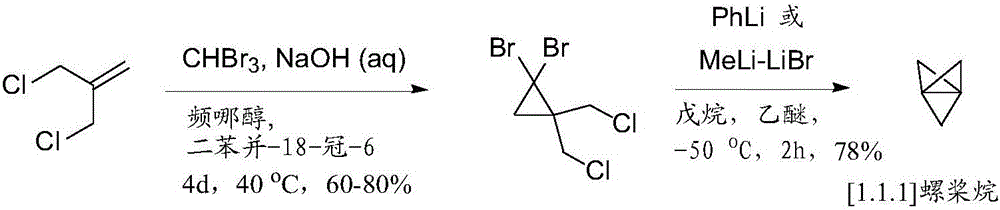
ActiveCN106232567AGroup 4/14 element organic compoundsCarbamic acid derivatives preparationPropellaneReagent
Disclosed herein are compounds of the general Formula (I), and methods of synthesizing substituted bicyclo[1.1.1 jpentanes. The synthetic methods described herein use a [1.1.1]propellane, a Group VIII transition metal compound, a hydride source and a reagent that can contribute a substituent to form a substituted bicycIo[1.1.1 ]pentane, such as a compound of the general Formula (I).
Owner:RECURIUM IP HLDG LLC
Preparation of carvone
InactiveUS6500989B1Low level of undesirable impurityPhysical/chemical process catalystsOrganic compound preparationInorganic materialsReaction product
A process for the preparation of carvone comprises hydrogenating carvoxime in the presence of a selecively poisoned catalyst. Preferred catalysts include noble metals supported on inorganic materials poisoned with a catalyst modifier. In a preferred embodiment of the process defined herein, the crude carvone reaction product produced in accordance with the process of the invention, is purified by treating the crude carvone product with an organometallic compound M(X)n wherein M is a polyvalent metal, n is the valence of M and X denotes an inorganic or organic atom or group.
Owner:QUEST INTERNATIONAL
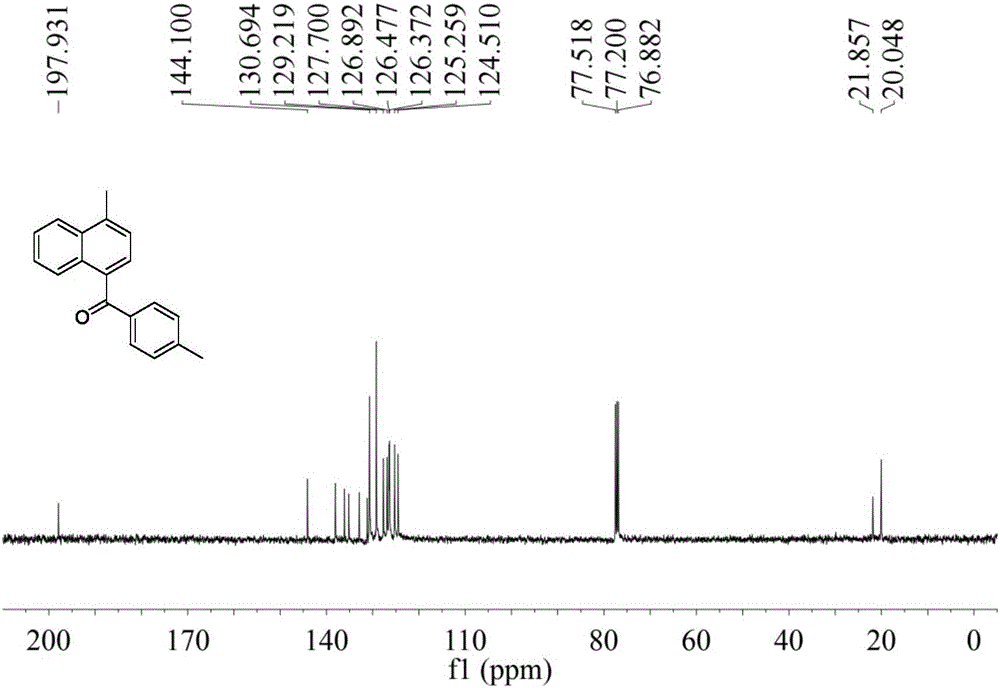
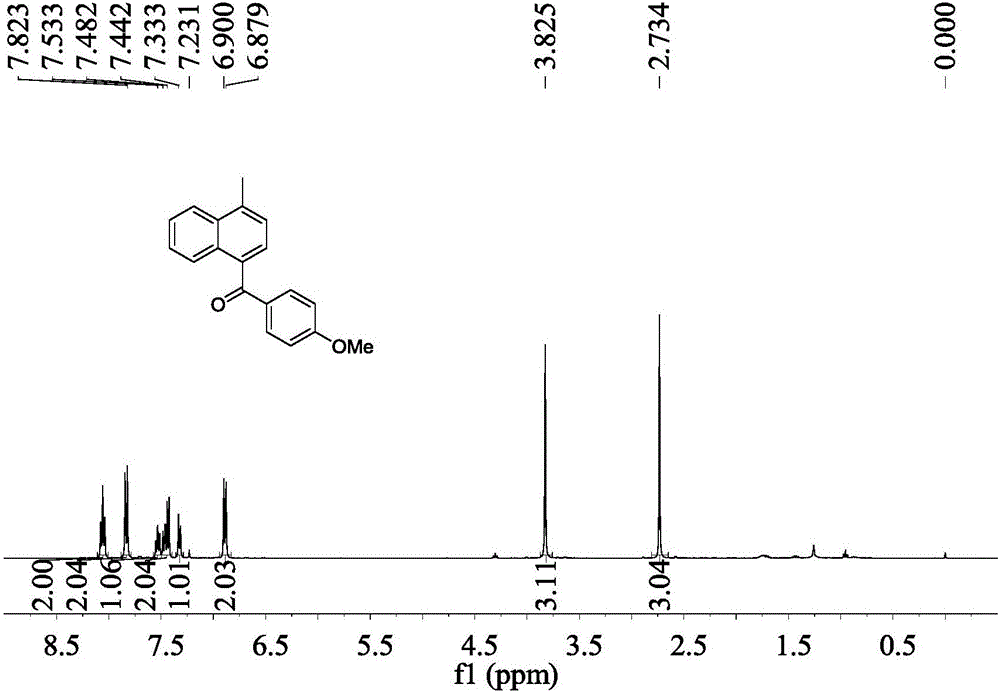
Preparation method of Alpha-naphthyl containing diarylketone compound
ActiveCN106478387AHigh yieldMild conditionsOrganic compound preparationPreparation by nitrile reduction and hydrolysisBenzeneOrganic solvent
The invention belongs to the technical field of pharmaceutical chemical intermediates and related chemistry and provides a preparation method of Alpha-naphthyl containing diarylketone compound; the preparation method comprises: reacting, under the action of a metal catalyst, a ligand and an alkali, 1-(chloromethyl)naphthalene and its derivatives as materials with phenylacetonitrile compounds in an anhydrous organic solvent, and converting, under the action of an oxidant, into the Alpha-naphthyl containing diarylketone compound. The preparation method has the advantages that the synthetic route is short, the conditions are mild, operating is simple, it is probable to industrialize the method, the yield of diarylketone products is high, it is possible, on naphthalene rings of the diarylketone compound synthesized by using the method, to further functionalize methyl so that the functional molecules, such as drug intermediates, optical materials, are acquired.
Owner:DALIAN UNIV OF TECH
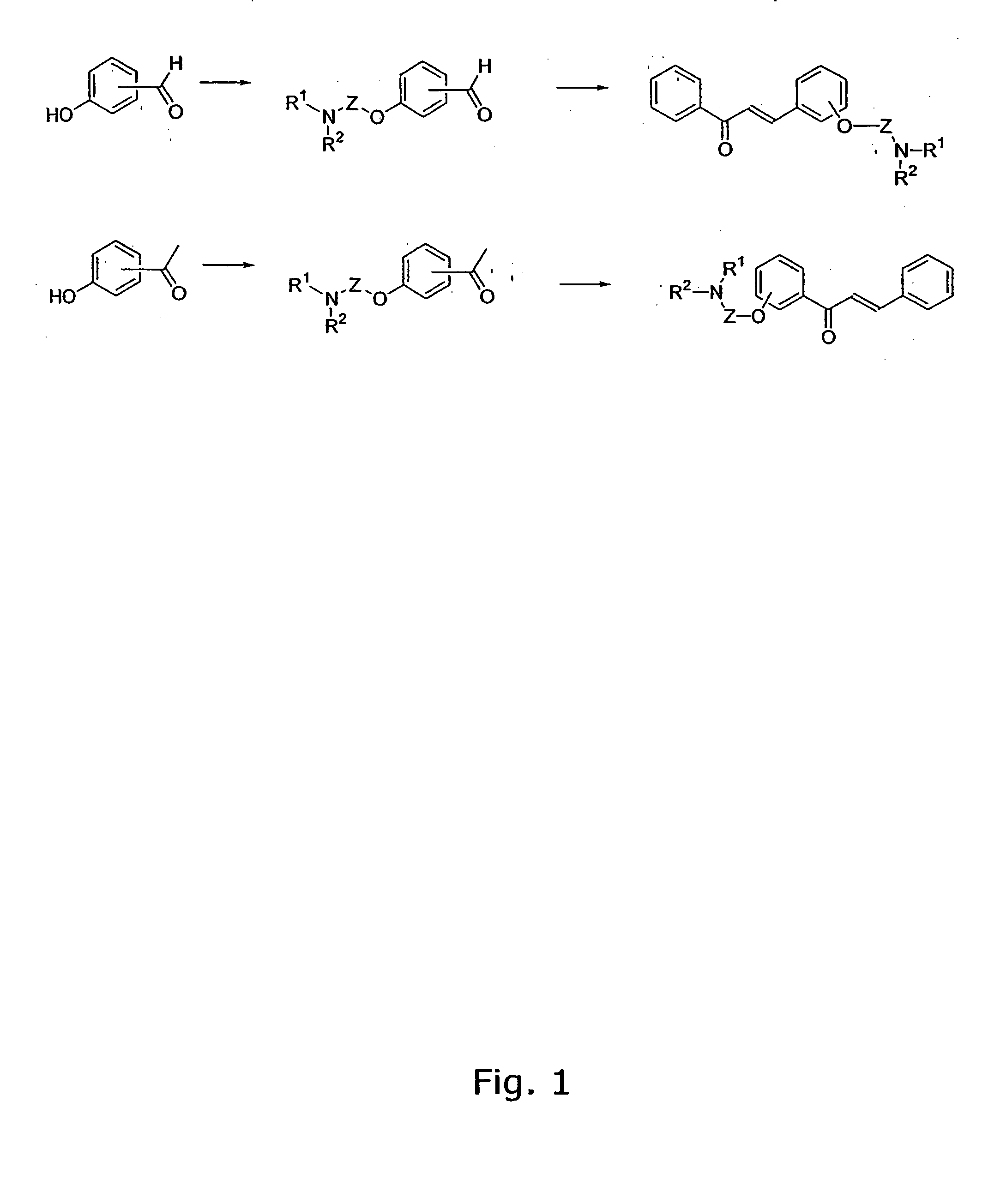
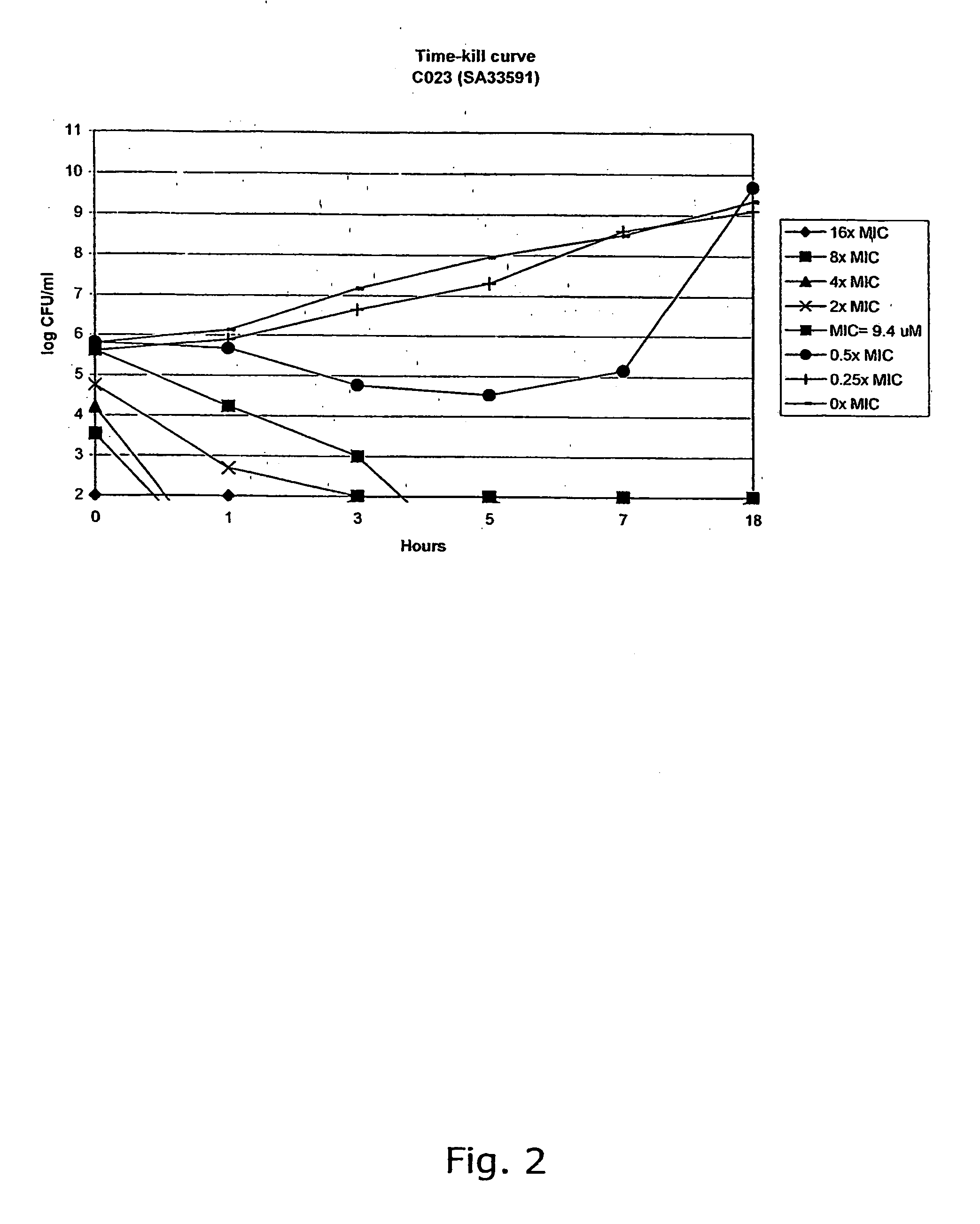
Aminoalkoxy-functional chalcones
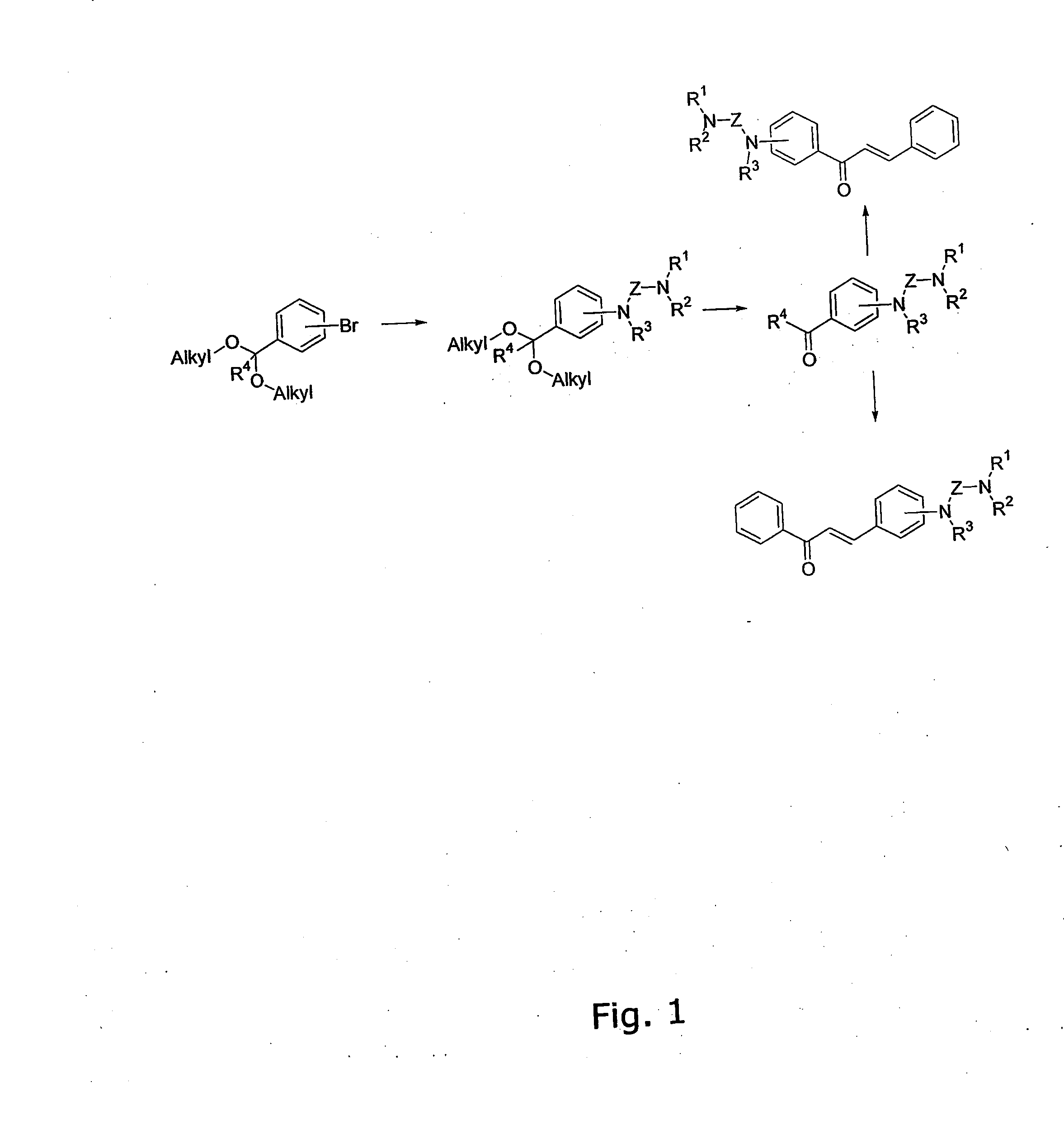
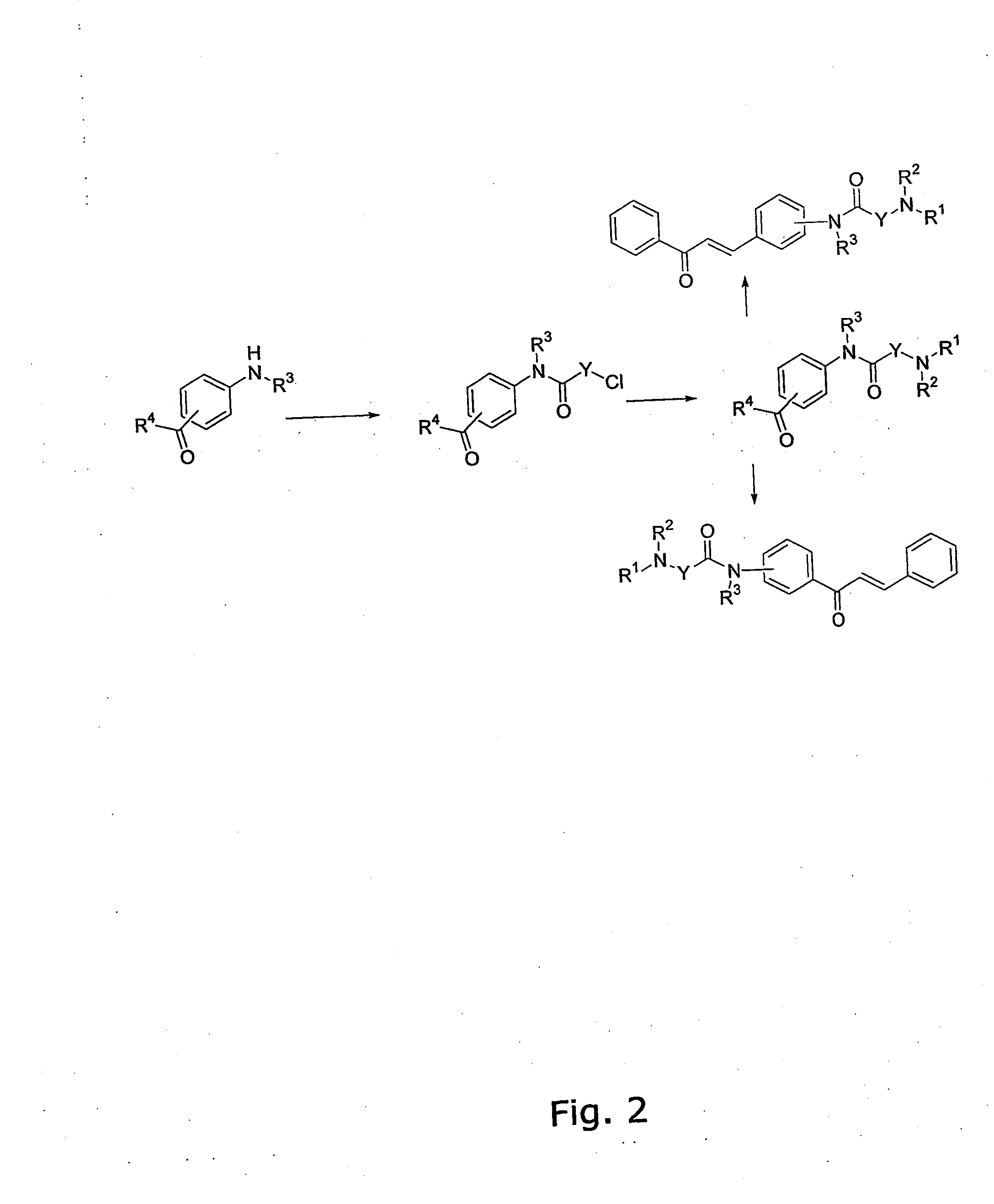
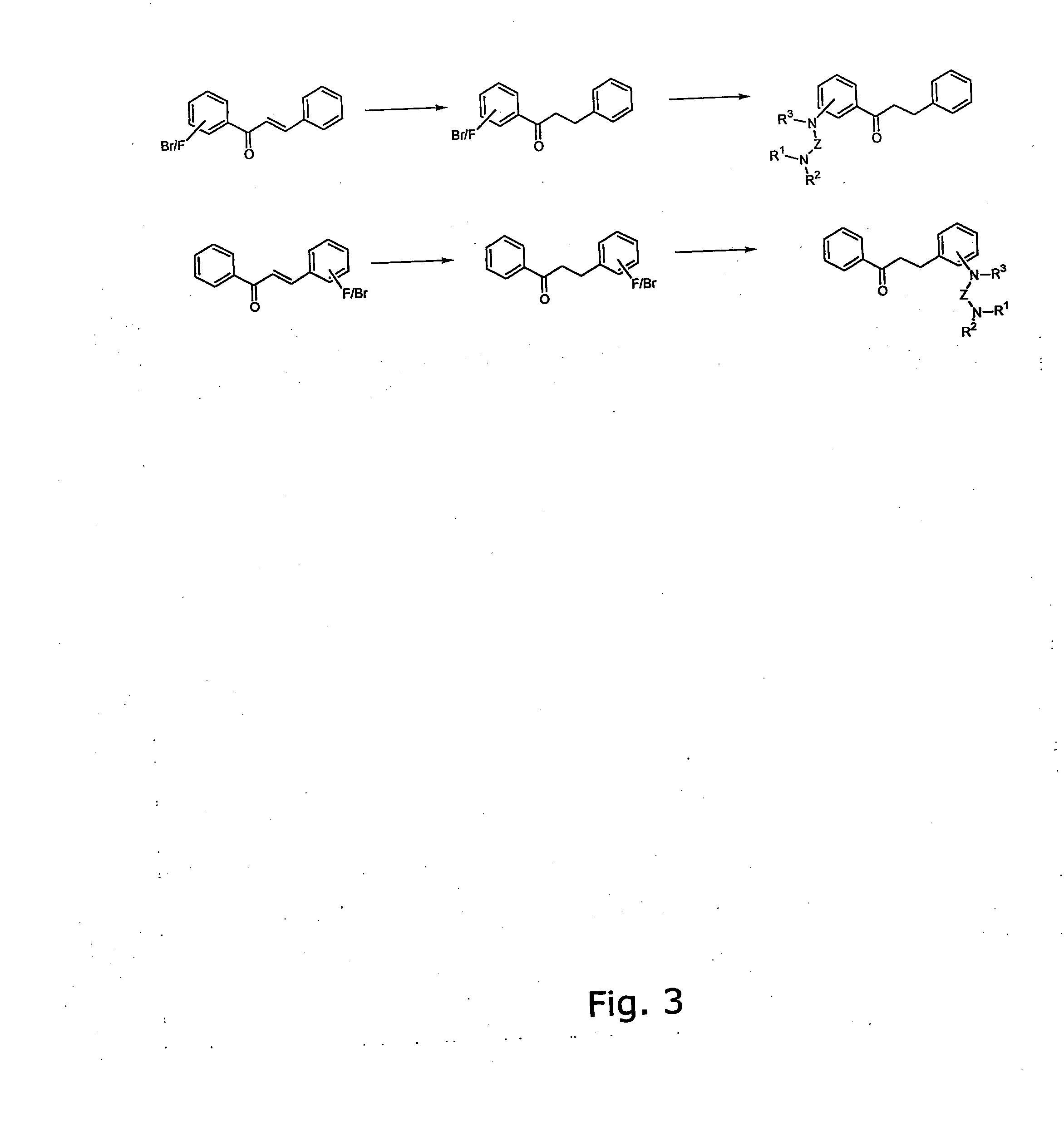
The invention provides novel alkoxyaminochalcone derivatives and analogues thereof. Use of the compounds, or compositions comprising them, as pharmaceutically active agents, in particular against bacterial and parasitic infections, is also disclosed. The invention further relates to a method for detecting inhibitory effects against e.g., bacteria, parasites, fungi, and helminths. The chalcones of the invention carry amino substituents and exhibit enhanced biological effects combined with improved metabolic and physicochemical properties, making the compounds useful as drug substances, in particular as antiparasitic, bacteriostatic, and bacteriocidal agents.
Owner:LICA PHARMA
Process for the synthesis of intermediates useful for the synthesis of tubulin inhibitors
InactiveUS6951955B2Carbamic acid derivatives preparationPeptide/protein ingredientsCombinatorial chemistryTubulin Inhibitors
Owner:WYETH HOLDINGS CORP
Diamino-functional chalcones
The invention provides novel diamino-functionalised chalcone derivatives and analogues thereof. Use of the compounds, or compositions comprising them, as pharmaceutically active agents, in particular against bacterial and parasitic infections, is also disclosed. The invention further relates to a method for detecting inhibitory effects against e.g., bacteria, parasites, fungi, and helminths. The chalcones of the invention carry amino substituents and exhibit enhanced biological effects combined with improved metabolic and physicochemical properties, making the compounds useful as drug substances, in particular as antiparasitic and bacteriostatic agents.
Owner:LICA PHARMA
Process for the synthesis of alkoxyalkyl (trifluormethylphenyl) methanones
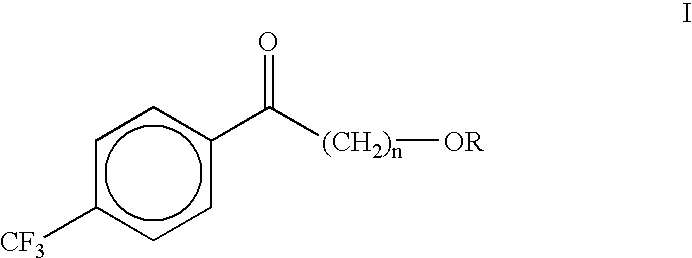
InactiveUS6380436B1High yieldRapid and cost-effective processPreparation by organometalhalide reactionOrganic compound preparationSolventAntidepressants drugs
A new process is described for the preparation of (alkoxyalkyl)(4-trifluoromethylphenyl)methanones. The process comprises reacting a 4-trifluoromethylbenzonitrile with an alkoxyalkyl Grignard in the presence of a suitable polar aprotic solvent. The compound (4-methoxybutyl)(4-trifluoromethylphenyl)methanone is useful as an intermediate in the preparation of the antidepressant drug fluvoxamine.
Owner:SUN PHARMA INDS
alpha-(Trifluoromethyl-substituted aryloxy, arylamino, arylthio or arylmethyl)-trifluoromethyl-substituted phenylacetic acids and derivatives as antidiabetic agents
InactiveUS7371888B2Halogenated hydrocarbon active ingredientsUrea derivatives preparationPhenylacetic acidMetabolic disorder
Compounds having a formula:or a pharmaceutically acceptable salt or prodrug thereof, are provided, and are useful for the treatment of metabolic disorders.
Owner:CYMABAY THERAPEUTICS
Catalytic reduction method of benzonitrile to benzoaldehyde
InactiveCN1483712AEasy to operateSave chemical process stepsPreparation by nitrile reduction and hydrolysisPtru catalystBenzaldehyde
The invention relates to a process for catalytically reducing substituted benzonitriles to substituted benzaldehydes in the presence of aqueous formic acid, of a nickel- and aluminium-containing catalyst and hydrogen.
Owner:BAYER AG
Synthesis method of 5-Methoxy-1-[4-(trifluoromethyl) phenyl]-1-pentanone
ActiveCN101602658AReduce consumptionWide choicePreparation by nitrile reduction and hydrolysisSynthesis methodsChloride
The invention discloses a synthesis method of 5-Methoxy-1-[4-(trifluoromethyl) phenyl]-1-pentanone. Firstly, parts of solvent are steamed out after magnesium chips and solvent are put in according to proportioning; then, 1-chloro-4-methoxy butane is added to react for synthesizing 4-methoxyl butyl magnesium chloride; 4-trifluoromethyl benzonitrile according to proportioning is added into the 4-methoxyl butyl magnesium chloride solution obtained in the last step to react; after reaction ends, solid products are obtained after filtering, pH value adjustment, organic phase washing by water, and solvent steaming out; then the obtained solid products after solvent is steamed out are decompressed and rectified to obtain the finished product of 5-Methoxy-1-[4-(trifluoromethyl) phenyl]-1-pentanone; the raw materials are as follows according to proportioning: 1.2-1.3 of magnesium chip, 1.1-1.2 of 1-chloro-4-methoxy butane and 1 of 4-trifluoromethyl benzonitrile. The method can obviously lower the consumption amount of the raw material, and the yield can be improved to above 80%.
Owner:青岛和兴精细化学有限公司
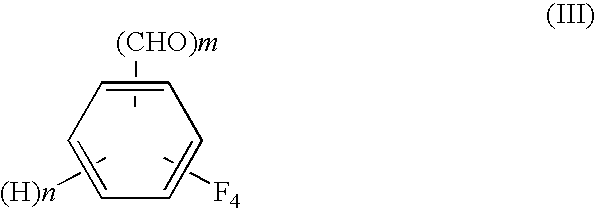
Process for preparing tetrafluorobenzene carbaldehyde alkyl acetal
InactiveUS7790931B2Improve efficiencyReduced isolationOrganic compound preparationCarbonyl compound preparation by hydrolysisAlcoholMetal catalyst
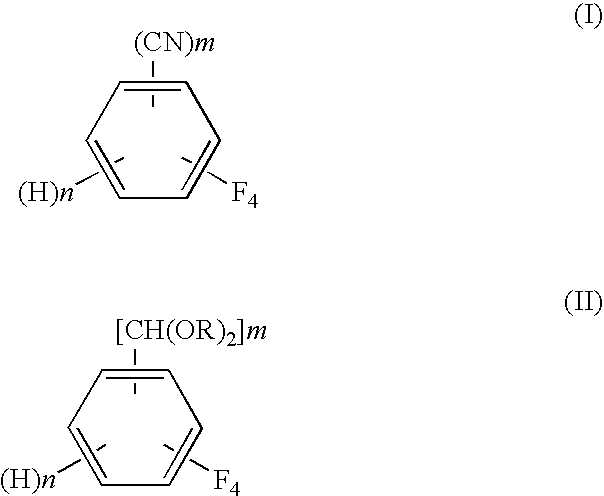
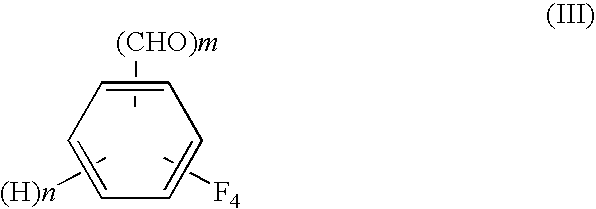
A process for preparing tetrafluorobenzene carbaldehyde alkyl acetal represented by the following formula (II), comprising reducing tetrafluorocyanobenzene represented by the following formula (I) with a metal catalyst containing a platinum group metal in the presence of an alkyl alcohol represented by R—OH (R is an alkyl group of 1 to 4 carbon atoms) and an acid; (I) wherein m is 1 or 2, n is 0 or 1, and m+n is 2, (II) wherein m and n are the same as those in the formula (I), and R is an alkyl group of 1 to 4 carbon atoms.
Owner:SHOWA DENKO KK
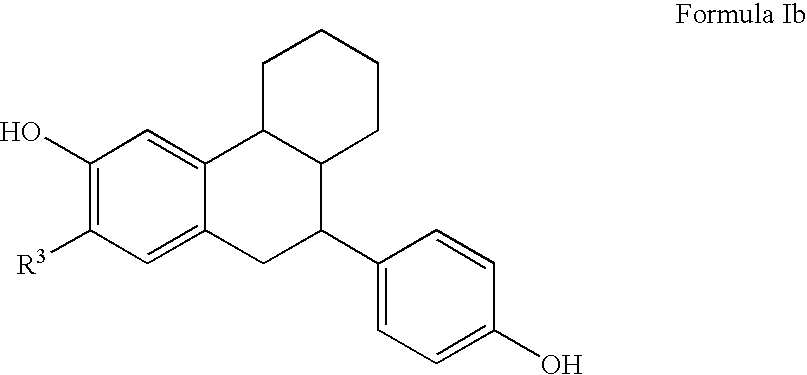
Substituted tetralins as selective estrogen receptor-β agonists
The present invention relates to novel tetralin ER-β agonist compounds, pharmaceutical compositions thereof, and use of these compounds to treat a ER-β mediated disease such as nocturia, obstructive uropathy, benign prostatic hypertrophy, obesity, dementia, hypertension, incontinence, colon cancer, prostate cancer, infertility, depression, leukemia, inflammatory bowel disease, and arthritis.
Owner:ELI LILLY & CO
Methods
InactiveUS20100130749A1Simple manufacturing methodHigh puritySilicon organic compoundsOrganic compound preparationMedicinal chemistryRenin inhibitor
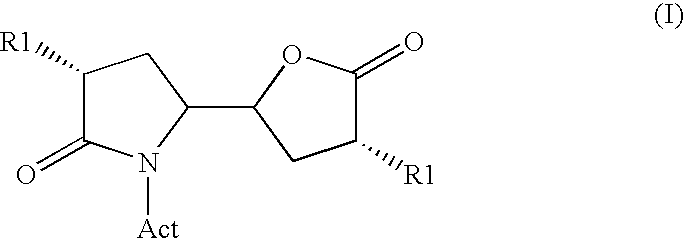
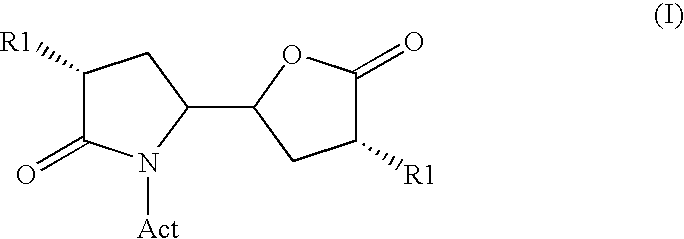
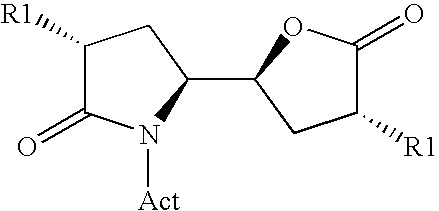
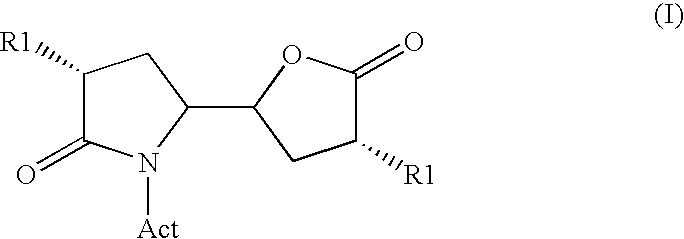
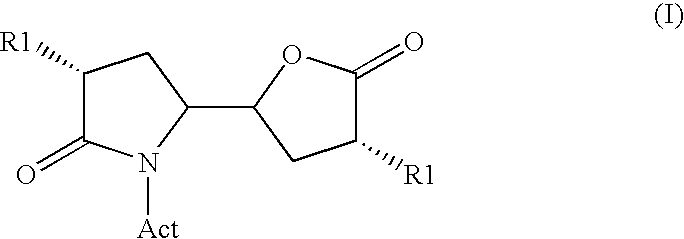
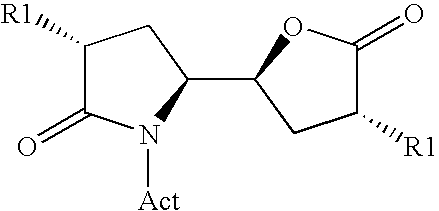
The invention related to a novel process, novel process steps and novel intermediates useful in the synthesis of pharmaceutically active compounds, especially renin inhibitors, such as Aliskiren Inter alia the invention relates to a process for the manufacture of a compound of the formula I.or a salt thereof, wherein R1 as well as Act are as defined in the specification, and processes of manufacturing this compound as well as intermediates in this process.
Owner:NODEN PHARMA DAC
Synthesis method of high-purity o-bromoacetophnones
InactiveCN105523911AImprove the production operating environmentEasy to operate and controlPreparation by nitrile reduction and hydrolysis2-bromobenzonitrileLiquid product
The present invention discloses a synthesis method of a high-purity o-bromoacetophnones. The preparation method comprises the following steps: a methylmagnesium bromide Grignard reagent is added into a container, tetrahydrofuran and 2-Bromobenzonitrile are dropwise added into the container at room temperature, and then a lot of white solids appear; a reaction is stopped until no raw material is detected by HPLC spectrum; reaction solution is dropwise added into hydrochloric acid solution, and the hydrochloric acid solution treated reaction solution is subjected to skimming, spin drying and reduced pressure distillation processes, thereby obtaining colorless liquid products--high-purity o-bromoacetophnones. The preparation method improves the production and operation environment, can synthesize the high-purity o-bromoacetophnones under the condition of normal temperature, is easy to operate and control, shortens reaction time, reduces costs, and is beneficial to industrial production.
Owner:SHANGHAI HUAXIAN NEW MATERIAL TECH CO LTD
Methods for the production of C-8 lactam lactone compounds
InactiveUS8338620B2Simple manufacturing methodInexpensive mannerGroup 4/14 element organic compoundsOrganic compound preparationMedicinal chemistryLactone formation
Owner:NODEN PHARMA DAC
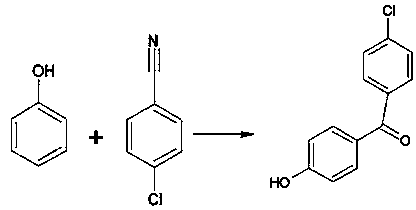
Preparation method of 4-chloro-4'-hydroxybenzophenone
InactiveCN107778153ALow costImprove responsePreparation by nitrile reduction and hydrolysisOrganic layerStrong acids
The invention relates to a preparation method of 4-chloro-4'-hydroxybenzophenone. The preparation method comprises the following steps: adding a proper amount of dichloroethane, phenol, 4-chlorobenzonitrile, anhydrous zinc chloride and gel type strong-acid ion exchange resin into a reaction kettle and stirring; controlling temperature to 50 DEG C to 120 DEG C and continuously introducing dry hydrogen chloride gas until the 4-chlorobenzonitrile reacts completely; cooling to room temperature and adding a proper amount of a hydrochloric acid solution; raising the temperature, reflowing and reacting for 5h to 7h; then cooling to room temperature and layering; recycling the dichloroethane into an organic layer and adding alkali into residues and dissolving; de-coloring by utilizing active carbon, carrying out acid precipitation, extracting and filtering and drying to obtain the 4-chloro-4'-hydroxybenzophenone. The preparation method of the 4-chloro-4'-hydroxybenzophenone, provided by the invention, is simple in route and high in yield.
Owner:江苏万隆科技有限公司
Preparation method of pentafluorobenzaldehyde
ActiveCN106397149AReduce generationReduce pollutionPreparation by nitrile reduction and hydrolysisReaction stepFormic acid
The invention relates to a preparation method of pentafluorobenzaldehyde, and belongs to the field of fine chemical product preparation. According to the preparation method, catalyst Ni-Al alloy, pentafluorobenzonitrile, and formic acid are subjected to reaction in water to prepare pentafluorobenzaldehyde. Ni-Al alloy is adopted, formic acid and water are taken as reducing agents, and pentafluorobenzaldehyde is prepared via reduction of pentafluorobenzonitrile. The preparation method is capable of realizing direction reduction of pentafluorobenzonitrile into pentafluorobenzaldehyde, so that reaction steps are reduced, generation of waste is reduced, production cost is reduced, and environmental pollution is reduced.
Owner:DALIAN QIKAI MEDICAL TECH
Process for preparing tetrafluorobenzene carbaldehyde alkyl acetal
InactiveUS20100105954A1Improve efficiencyReduce the burden onOrganic compound preparationPreparation by nitrile reduction and hydrolysisAlcoholMetal catalyst
A process for preparing tetrafluorobenzene carbaldehyde alkyl acetal represented by the following formula (II), comprising reducing tetrafluorocyanobenzene represented by the following formula (I) with a metal catalyst containing a platinum group metal in the presence of an alkyl alcohol represented by R—OH (R is an alkyl group of 1 to 4 carbon atoms) and an acid; (I) wherein m is 1 or 2, n is 0 or 1, and m+n is 2, (II) wherein m and n are the same as those in the formula (I), and R is an alkyl group of 1 to 4 carbon atoms.
Owner:SHOWA DENKO KK
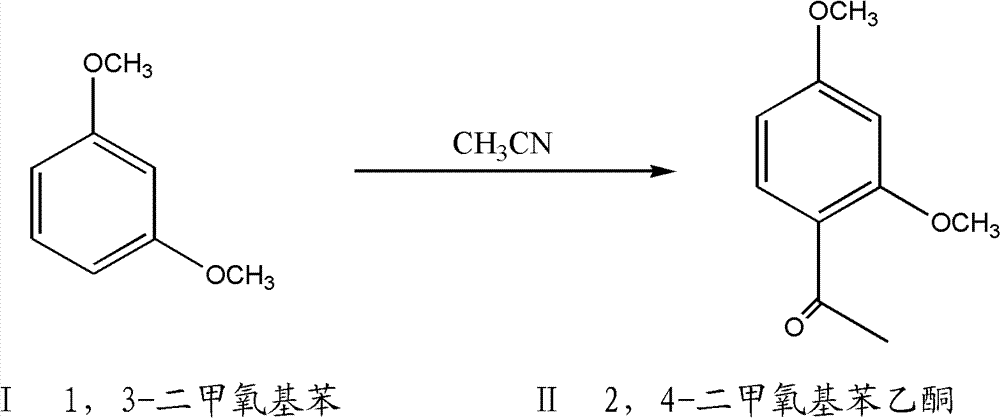
A method for preparing 2,4-dimethoxyacetophenone
ActiveCN103570518BEasy to recycleImprove securityCarbonyl compound preparation by hydrolysisPreparation by nitrile reduction and hydrolysisFiltrationEther
Owner:INSIGHT HIGH TECH JIANGSU CO LTD
Propellane derivates and synthesis
PendingCN113336653AGroup 4/14 element organic compoundsCarbamic acid derivatives preparationPropellaneCombinatorial chemistry
Disclosed herein are compounds of the general Formula (I), and methods of synthesizing substituted bicyclo[1.1.1]pentanes. The synthetic methods described herein use a [1.1.1]propellane, a Group VIII transition metal compound, a hydride source and a reagent that can contribute a substituent to form a substituted bicycIo[1.1.1]pentane, such as a compound of the general Formula (I).
Owner:RECURIUM IP HLDG LLC
Process for preparing tetrafluorobenzene carbaldehyde alkyl acetal
A process for preparing tetrafluorobenzene carbaldehyde alkyl acetal represented by the following formula (II), comprising reducing tetrafluorocyanobenzene represented by the following formula (I) with a metal catalyst containing a platinum group metal in the presence of an alkyl alcohol represented by R-OH (R is an alkyl group of 1 to 4 carbon atoms) and an acid; (I) wherein m is 1 or 2, n is 0 or 1, and m+n is 2, (II) wherein m and n are the same as those in the formula (I), and R is an alkyl group of 1 to 4 carbon atoms.
Owner:SHOWA DENKO KK
Popular searches
Solid-state devices Semiconductor/solid-state device manufacturing Functional group formation/introduction Halogenated hydrocarbon preparation Thermoelectric device junction materials Preparation from heterocyclic compounds Semiconductor devices Group 5/15 element organic compounds Aldehyde active ingredients Animal repellants
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com