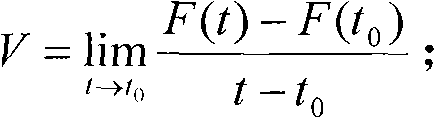Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
55results about How to "Troubleshoot configuration issues" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
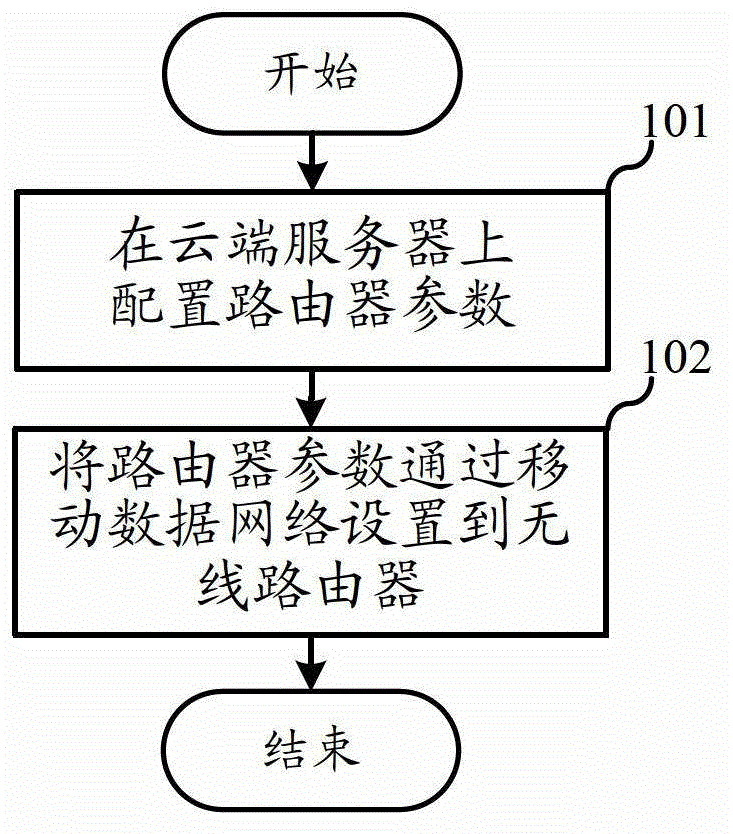
Configuration method of wireless router based on mobile data network and system thereof
InactiveCN102752389ASpeed up the setup processSave resourcesData switching switchboardsIp addressWireless router
The invention relates to the field of communication, and discloses a configuration method of a wireless router based on a mobile data network and a system thereof. The user can solve the configuration problem simply by accessing the website, and does not need to access the incomprehensible IP address; and the configured router parameters are not lost due to the router replacement, factory setting recovery and the like. According to the invention, the method comprises the following steps that: a user terminal accesses a cloud server through Internet, and configures the router parameters of the wireless router on the cloud server; and the cloud server sets the router parameters to the wireless router through the mobile data network.
Owner:林兆旭
Switch configuration recovery method in SDN (Software Defined Networking)
ActiveCN105933253ATroubleshoot configuration issuesReduce transmission delayData switching networksRecovery methodData information
The invention discloses a switch configuration recovery method in SDN. The method is realized by that in a physical topology system, each OpenFlow switch is connected with one central switch, and the central switch backs received configuration information up in a nonvolatile memory according to a standard protocol format; and when certain switch is restarted due to fault, the connected central switch is asked for data information which matches the switch, and data is recovered to a memory. Compared with the prior art, the configuration information of the switches are backed up in the near central switch, transmission time delay is reduced, the configuration information of the switches can be recovered rapidly, the problem in recovering configuration of memory information of the switches in real time in the SDN framework is solved, and the method of the invention is highly practical and easy to popularize.
Owner:SHANDONG INSPUR SCI RES INST CO LTD
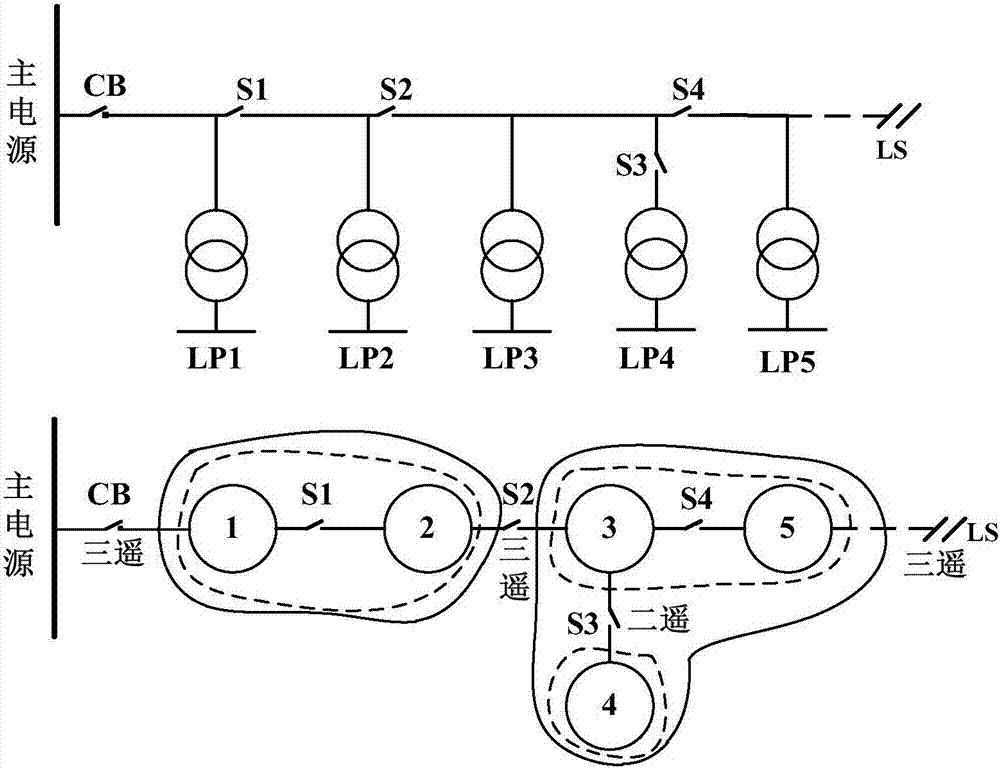
Optimal configuration method for feeder terminals based on genetic algorithm
ActiveCN107545516ANumber of solutionsTroubleshoot configuration issuesData processing applicationsEmergency protective circuit arrangementsConsequence analysisTotal investment
The invention provides an optimal configuration method for feeder terminals based on an genetic algorithm, which is characterized in that an objective function is built according to the total investment of two-remote feeder terminals and three-remote feeder terminals under constraint conditions of a power supply reliability index of a power distribution network and power failure time of a load point, reliability evaluation is performed by adopting a fault consequence analysis method, influences imposed on the reliability of a power distribution system and the power failure time of a load pointby the two-remote feeder terminals and the three-remote feeder terminals are sufficiently considered, and finally the objective function is solved in an optimized manner by adopting the genetic algorithm. The optimal configuration method can solve a configuration problem in number and position of the two-remote feeder terminals and the three-remote feeder terminals, and provides a reference for scientifically and reasonably performing power distribution automation plan design and transformation.
Owner:CHINA ELECTRIC POWER RES INST +3
Method and apparatus for multiuser QOS dispatching
InactiveCN1949748ASolve resource problemsTroubleshoot configuration issuesData switching networksDistributed computingSoftware
The invention discloses a method and device for making QOS scheduling for multiple users, belonging to communication technical field. And the invention advances a method for implementing MDRR algorithm by token bucket and finally implementing users'QOS scheduling, comprising the concrete steps of: according to the configured QOS informatiojn table, regularly refreshing token number in token bucket; finding the to-be-scheduled user number in scheduling matrix in round robin mode; according to the user number, sending out scheduling request; obtaining the previous scheduling result feedback, and executing the scheduling request. And the invention also provides a device for implementing QOS scheduling for multiple users, comprising configuration module, token bucket refreshing module, scheduling matrix updating module, round robin module, scheduling request sending module and token removing module.
Owner:BEIJING HUAWEI DIGITAL TECH
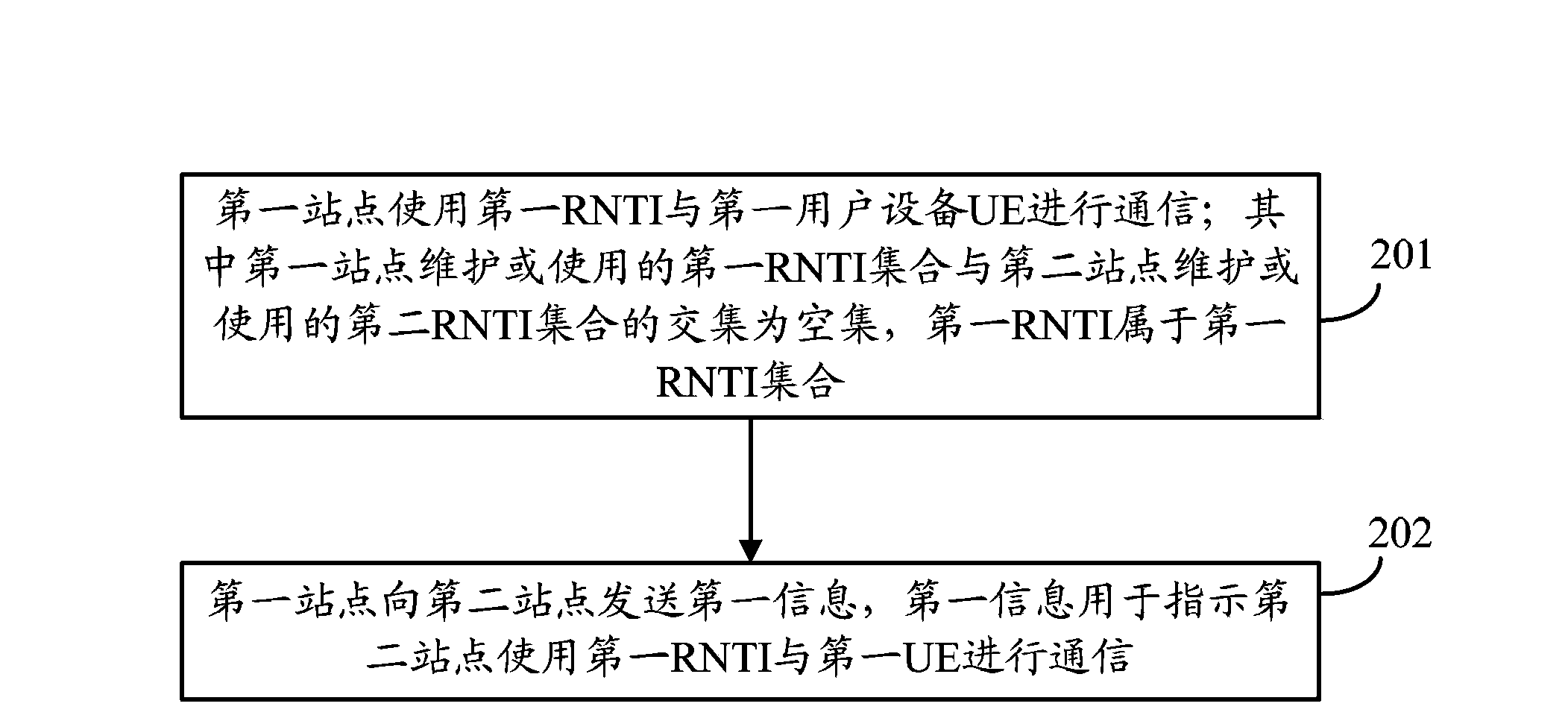
Wireless network temporary identification configuration method, station site, and UE
ActiveCN103888963ATroubleshoot configuration issuesConnection managementTelecommunicationsWireless network
The invention provides a wireless network temporary identification configuration method, a station site, and UE. The method comprises: a first station site using a first RNTI to communicate with first UE; the intersection of a first RNTI set maintained or used by the first station site and a second RNTI set maintained or used by a second station site being an empty set, and the first RNTI belongs to the first RNTI set; and the first station site sending first information to the second station site, the first information being used for indicating the second station site to use the first RNTI to communicate with the first UE. According to the embodiments of the invention, at a scene when one piece of UE communicates with multiple station sites, by configuring different RNTIs for the UE, the UE is enabled to communicate with the multiple station sites by use of the different RNTIs such that the problem of RNTI configuration under the condition that one piece of UE communicates with multiple station sites in the prior art is solved.
Owner:HUAWEI TECH CO LTD
Method, device and system for treating conflict of data dispatching time and measuring time
ActiveCN101651954AGuaranteed reliabilityIncrease speedConnection managementData transmissionControl channel
The invention discloses a method, a device and a system for treating conflict of a data dispatching time and a measuring time. The method comprises the following steps: determining an auxiliary frequency point TS0 of UE used for measuring a time under a CELL_DCH state; and controlling data sending treatment on a business channel or a control channel by the auxiliary frequency point TS0 used for measuring the time according to a preset strategy. The invention can solve the problem of the conflict of the data dispatching time and the measuring time of UE under the CELL_DCH state aiming to a conventional business, and HSDPA or HSUPA business, thereby ensuring the reliability and the measuring performance of data transmission.
Owner:北京万海云科技有限公司
Optimal configuration method and device for feeder terminal
ActiveCN108154249ANumber of solutionsTroubleshoot configuration issuesForecastingComputer terminalDependability
The invention provides an optimal configuration method and device for a feeder terminal. The method comprises the following steps that: creating a network topology structure; coding a switch installation terminal situation, randomly generating an initial population matrix, and independently setting an iteration frequency p and a population individual number k as 1; carrying out minimum two-remotearea and minimum three-remote area division on individuals in the population to obtain the adaptive value of the individual; and determining and updating a global optimal individual. By use of the technical scheme provided by the invention, the annual blackout time of a user and the power supply reliability requirement of a system are met, so that the amount and position configuration of a two-remote feeder terminal and a three-remote feeder terminal can be optimized to a maximum degree, and the optimal configuration of the feeder terminal of an overhead line can be greatly improved.
Owner:CHINA ELECTRIC POWER RES INST +3
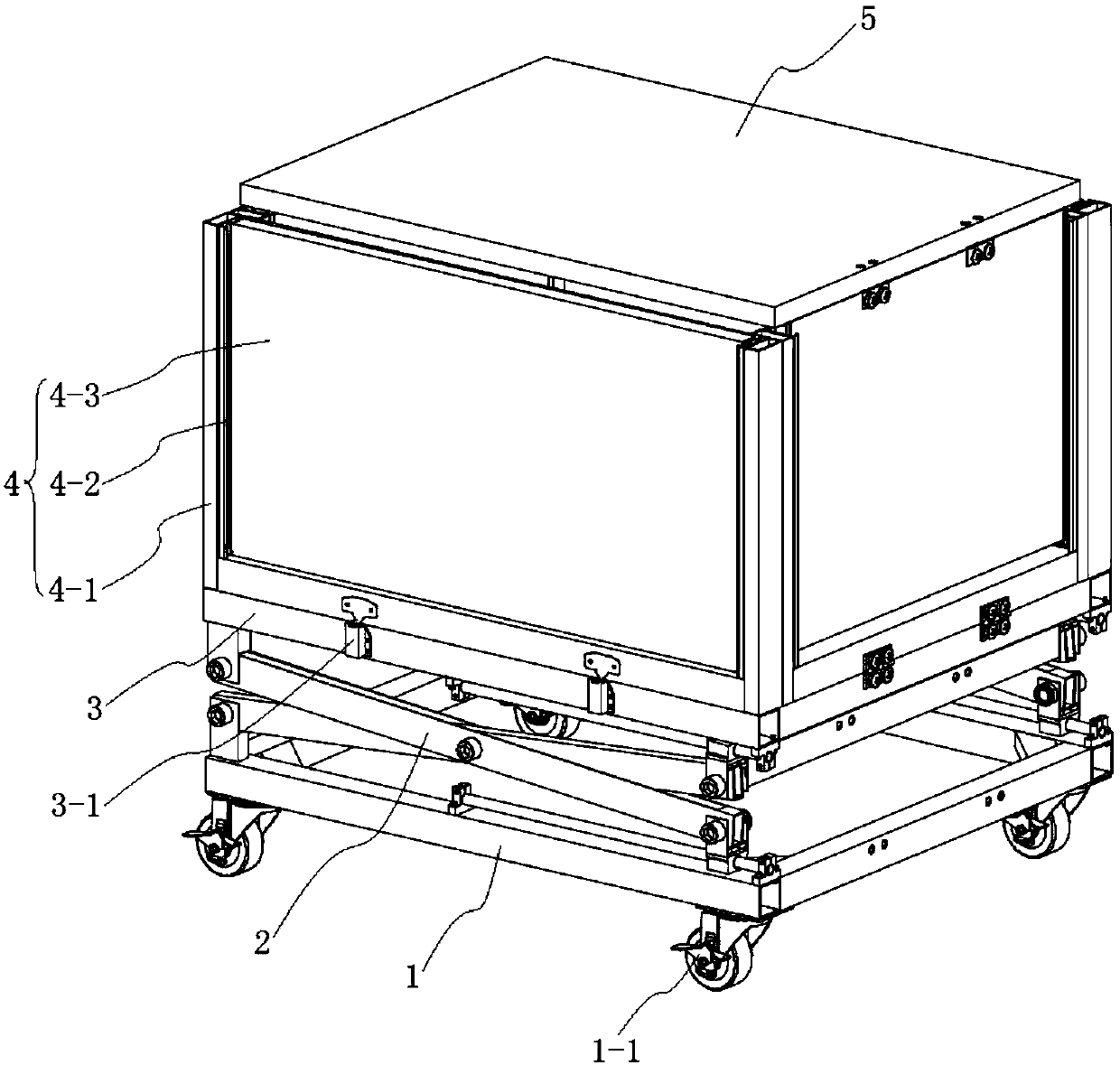
Machine packaging box that also can serve as expandable working table and using method of machine packaging box
ActiveCN107934156ALow costIncrease the areaPackaging vehiclesContainers for machinesEngineeringShear mode
The invention discloses a machine packaging box that also can serve as an expandable working table and a using method of the machine packaging box and belongs to the technical field of packaging. Themachine packaging box comprises a base, a shear mode lifting mechanism and a packaging box main body, wherein the packaging box main body is mounted on the base through the shear mode lifting mechanism and is of a cuboid structure composed of a box body bottom plate, box body side plates and a box body top plate; the four sides of the box body bottom plate are rotatably connected with four groupsof box body side plates; the rotational angle of each of two groups of opposite box body side plates is 90 degrees; the rotational angle of each of the other two groups of opposite box body side plates is 180 degrees; and the box body top plate is rotatably connected with one of the other two groups of opposite box body side plates with the rotational angle being 180 degrees. According to the machine packaging box, the original structure deforms to form the working table, so that the packaging box can package transportation products, also can be used as the working table of a machine and realizes two purposes, so that the cost of purchasing another working table is saved, resource waste caused by discard of the packaging box is avoided, and the machine packaging box has high expansion performance and practical value.
Owner:CHANGZHOU INST OF TECH
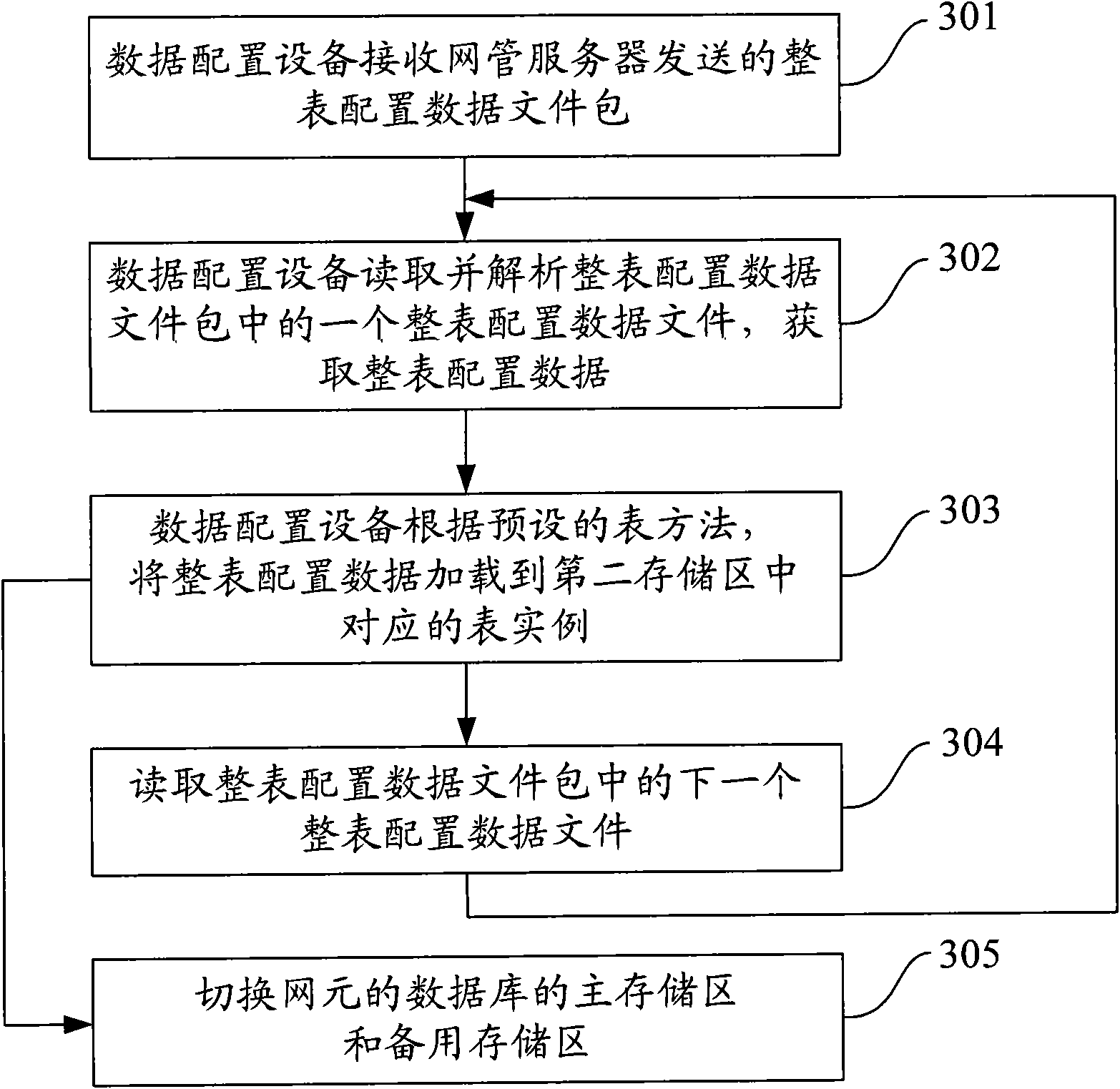
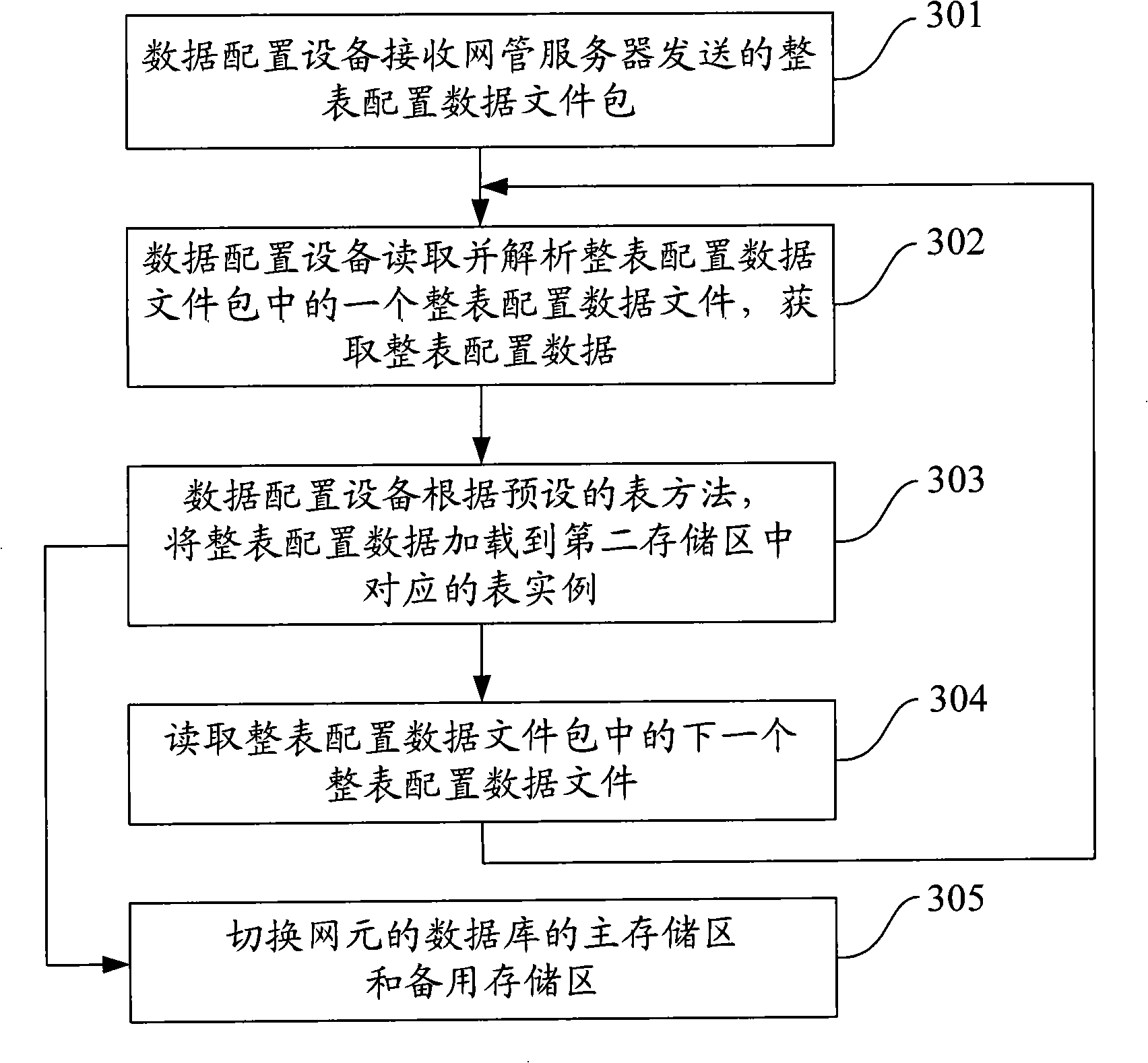
Data configuration method, equipment and system
ActiveCN101860458ATroubleshoot configuration issuesEasy to handleData switching networksNetworked systemData file
The invention discloses a data configuration method, data configuration equipment and a data configuration system. The data configuration method is applied to a network system comprising a network management server, the data configuration equipment and at least one network element, wherein the network element comprises a database. The method comprises that: the data configuration equipment receives a data file sent by the network management server, wherein the data file comprises configuration data for configuring the database of the network element; the data configuration equipment resolves the data file, and acquires the configuration data in the data file; and the data configuration equipment loads the configuration data to the database of the network element according to a preset table method. The data configuration equipment can load the configuration data to the database of the network element by resolving the data file, so the processing flow is simple, the problems of high difficulty and high cost brought by developing a universal structural language of the database are reduced, and the problem of universal data configuration can be quickly solved.
Owner:DATANG MOBILE COMM EQUIP CO LTD
Method and system for configuring information delivery
InactiveCN103731437ATroubleshoot configuration issuesRealize business configurationTransmissionConfiguration itemClient-side
The application discloses a method and a system for configuring information delivery. The method comprises the steps that a server is used for searching configuring file information of a user according to identity information of the login user received by a client and reading delivery configuring permission of the user from the configuring file information; as for the user with the delivery configuring permission, configuration data corresponding to the user is acquired according to the identity information of the user, order information is acquired according to configuring rule information in the configuration data, and an operation interface of the configuration data and the order information is established and sent to the client; the server is used for receiving configuration indicating information of the user on the operation interface in the client and updating the configuration file information according to a configuration item corresponding to the configuration indicating information. With the adoption of the method and the system, information delivery is configured after a delivery service line varies and delivery group members are grouped and regulated.
Owner:阿里巴巴(成都)软件技术有限公司
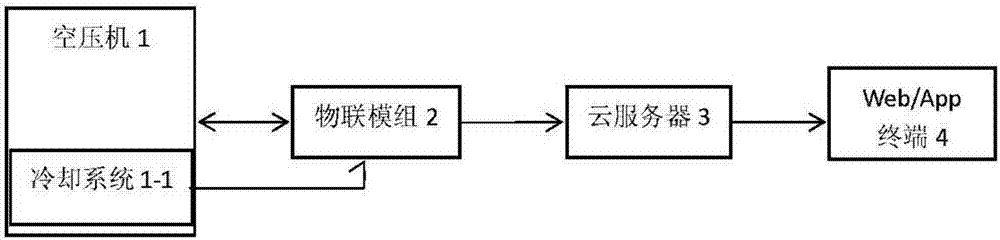
Remote and automatic screw rod-type air compressor cooling system fault judgment method
ActiveCN107247432ASolve the workloadSolve operational problemsProgramme controlComputer controlThe InternetEngineering
The invention relates to a remote and automatic screw rod-type air compressor cooling system fault judgment method. The method is characterized by comprising steps: 1) an Internet of things module is selected or set and is bound in an air compressor; 2) a two-way communication relationship between the Internet of things module and a cloud server is built, the Internet of things module is used to transmit acquired operation data of the air compressor to the cloud server, the operation data are saved in the cloud server, and the Internet of things module is used to read various versions of communication protocols for the air compressor saved in the cloud server; 3) a temperature sensing head of the Internet of things module is used to detect the temperature of a cooling system, the detected real-time temperature signals of the cooling system are transmitted to the cloud server; and 4) after the cloud server receives the temperature signals of the cooling system, the detected temperature signals are compared with a rated value according to a selected air compressor communication protocol, whether the temperature of the air compressor is over high is judged, whether fault happens to the air compressor is further judged, and thus, remote and automatic screw rod-type air compressor cooling system fault judgment is formed. The method has the advantages of small workload, easy operation, high accuracy and saved human resources.
Owner:广东蘑菇物联科技有限公司
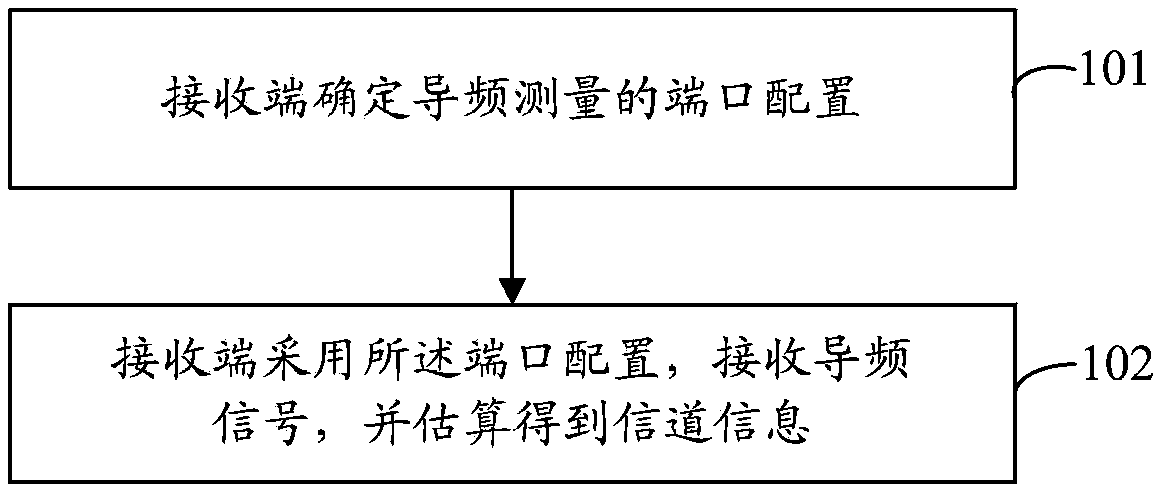
Pilot frequency measurement method and device
InactiveCN103581932ATroubleshoot configuration issuesAvoid overspendingRadio transmissionChannel estimationFrequency measurementsVIT signals
A method for measuring pilot is disclosed in the present invention. The method comprises the steps of: a receiving end determining the port configuration for measuring pilot; the receiving end receiving the pilot signal using the port configuration, and estimating the obtained channel information. Accordingly, a receiving end for measuring pilot is also disclosed in the present invention. Even if the number of antenna ports increases rapidly, a part of the ports selected therein as required can be still configured to the ports for transmitting the pilot signal. Without limiting the number of the ports for transmitting the pilot signal, the problem of the pilot configuration in the case that the number of antenna ports increases rapidly can be solved.
Owner:ZTE CORP
Table batch processing method, system and device and storable medium
PendingCN110287459ATroubleshoot configuration issuesImprove efficiencyNatural language data processingSpecial data processing applicationsBatch processingComputer science
The invention relates to the technical field of data processing, and provides a table batch processing method, system and device and a storage medium, wherein the method comprises the steps of S110, generating a business table of a corresponding format according to a preset business demand; S120, carrying out pattern parameter identification on the cells in the business table, and classifying the cells of the business table according to the identified pattern parameters, wherein the pattern parameters of the cells of all the categories are the same; S130, configuring the style of each category of cells according to a preset service demand, wherein for each category of cells, one of the plurality of cells of the type is determined as a mode cell to carry out corresponding cell style configuration, and a cell format brush is called to uniformly refresh and configure the rest cells in the type for each cell of the configured mode cell according to the style of the mode cell. By utilizing the method, the problems of low table configuration efficiency, high error rate and the like in the prior art can be solved.
Owner:PING AN TECH (SHENZHEN) CO LTD
Two-stage scheduling method for multi-period multi-product evanescent product supply chain network design
ActiveCN110852667ASolve the problem of site selectionTroubleshoot configuration issuesSustainable waste treatmentBuying/selling/leasing transactionsSite selectionIndustrial engineering
The invention relates to the technical field of evanescent supply chain networks, in particular to a two-stage scheduling method for multi-period multi-product evanescent supply chain network design.The method comprises a site selection model step, a configuration model formulating step based on the site selection model and a cyclic feedback program constructing step according to the site selection model and the configuration model. Aiming at the multi-period multi-product evanescent product supply chain network design problem, the invention provides a two-stage scheduling solving method, which comprises the following steps of: in the first stage, establishing a facility site selection model in the whole supply chain network on a strategic decision-making level; tactical configuration decision of the material configuration level is carried out in the second stage, so that a configuration model is established, and the site selection model and the configuration model are integrated intoone model for solving the site selection and configuration problems of suppliers, a mixed production inspection center, a mixed storage recovery center, a disposal and recycling center and retailers.
Owner:ZHEJIANG GONGSHANG UNIVERSITY
Method and device for configuring data source for display unit dynamically
ActiveCN101833938ATroubleshoot configuration issuesEasy flow controlSpectral/fourier analysisCathode-ray tube indicatorsTraffic capacityGraphics
The invention discloses a method and device for configuring data source for display unit dynamically. The method comprises the step 204 using data flow feedforward control, wherein data flow rate is monitored dynamically, secondly unit flow is displayed according to unit time, and unit relative data volume is calculated and displayed dynamically. The mathematic model is as follows: assuming that the function of data volume Q changed with the time t is F (t) = Q, the flow rate in the time t0 is shown below; assuming that the rate for displaying unit data volume by the screen is q and the function of data volume Qp corresponding to one display unit and time t is Qp = f(t), f(t) = dF(t) / (dt*q); and the device comprises a data flow monitoring unit, a dynamic data configuration unit and a dynamic interpolation compensation unit. The invention adopts data flow feedforward control to solve the data source problem configuration problem of the display units of the display medium and provide data source with stable and controllable flow rate and flow for the display units; and the method is particularly suitable for the waveform with high demand on real-time display or for the treatment and display of graphic data sources. The method and device for configuring data source for display unit dynamically of the invention can be widely used in the display process of wave graphics.
Owner:GUANGDONG BIOLIGHT MEDITECH CO LTD
Business monitoring method and device
ActiveCN108429632AGuaranteed stabilityTroubleshoot configuration issuesData switching networksBusiness PersonnelBusiness data
The application provides a business monitoring method and device. The business monitoring method comprises the following steps: calculating the corresponding relationship between business scenarios and business rules according to historical business data; obtaining the real business scenario and real business rule of the real-time business data aiming at the real-time business data; finding the business rule corresponding to the real business scenario in the corresponding relationship; and outputting a alert prompt if the real business rule is different from the found business rule. By means of the business monitoring method and device, the business personnel can look over the influence scope of the business rule in time and solve the configuration problem; therefore, the stability of thebusiness is ensured.
Owner:ADVANCED NEW TECH CO LTD
Maritime broadband multimedia intelligent communication system and method
PendingCN111601259AAchieve adaptiveTroubleshoot configuration issuesTelevision conference systemsParticular environment based servicesTelecommunications linkHeterogeneous network
The invention provides a maritime broadband multimedia intelligent communication system and method, which take groups as units for maritime operation, and each group is guided by one command ship witha plurality of task ships, thereby achieving the broadband multimedia networking communication between random ships and between a ship and a command center. Wide-area interconnection is achieved between the fleet and the command center and between different fleets through satellite communication, and a center network with the command ship as the core is established in the same fleet in a broadband multimedia clustering mode. When the task ships of the same fleet communicate with each other, a communication link is established through the cluster base station of the command ship of the fleet;and when the task ship communicates with a ship of another fleet, communication can be completed through a heterogeneous network formed by the cluster link and the satellite link. The central networkwith the command ship as the core solves the problem of configuration during random networking of the command ship and the task ship, and network self-adaptation of random matching of the command shipand the task ship is achieved by designing proper routes on the command ship and the task ship.
Owner:TIANJIN AEROSPACE ZHONGWEI DATA SYST TECH CO LTD
Network flow distribution management system based on Internet
PendingCN111817980ARelieve stressImprove transmission efficiencyData switching networksInternet trafficProcessing element
The invention discloses a network flow distribution management system based on the Internet. The network flow distribution management system comprises an information acquisition and processing unit and a flow distribution management unit, the information acquisition and processing unit is in interactive communication connection with a plurality of network user terminals, and the output end of theinformation acquisition and processing unit is in telecommunication connection with the input end of the flow distribution management unit. According to the invention, dual independent network flow distribution management is carried out on a plurality of user terminals and applications of the terminals; the problem of network flow distribution configuration is deeply solved; the network pressure is relieved, the network transmission efficiency is improved, the response speed of the network application is increased, meanwhile, the time period adjusting unit is arranged in the flow distributionmanagement unit, network flow distribution can be adjusted and configured for the user terminal and the application of the terminal according to time period information according to time period requirements, and the flexibility of network flow distribution management is improved.
Owner:深圳市高德信通信股份有限公司
A switch configuration recovery method under SDN network
ActiveCN105933253BTroubleshoot configuration issuesReduce transmission delayData switching networksRecovery methodTransmission time delay
The invention discloses a switch configuration recovery method under an SDN network. The implementation process is as follows: in a physical topology system, each OpenFlow switch is connected to a central switch, and the central switch backs up received configuration information in a standard protocol format. In the non-volatile memory; when the switch restarts due to a fault, it only needs to request the matching data information from the connected central switch and restore the data to the memory. Compared with the prior art, the switch configuration recovery method under the SDN network backs up the configuration information of the switch in the adjacent central switch, thereby reducing the transmission delay and achieving the purpose of quickly restoring the switch configuration information. Real-time recovery of configuration problems of switch memory information, strong practicability, and easy promotion.
Owner:SHANDONG INSPUR SCI RES INST CO LTD
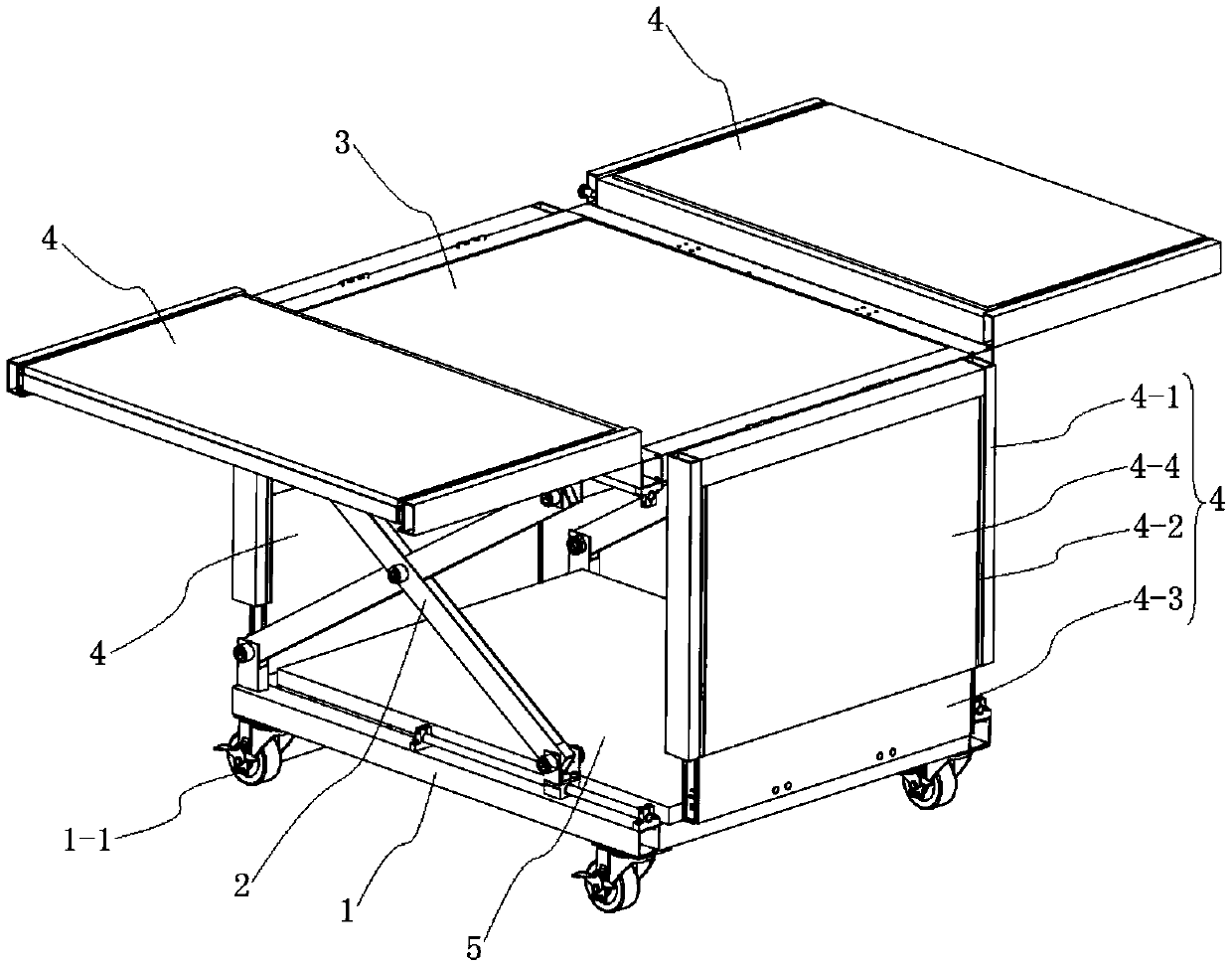
Machine packing case double as workbench and method of use thereof
The invention discloses a machine packaging box doubling as a workbench and a use method of the machine packaging box and belongs to the technical field of packaging. The machine packaging box doubling as the workbench comprises a base, a shear type lifting mechanism and a packaging box main body; the packaging box main body is installed on the base through the shear type lifting mechanism and isof a cuboid structure formed by a box body bottom plate, box body side plates and a box body top plate; the four side edges of the box body bottom plate are rotationally connected with four groups ofthe box body side plates respectively; and the box body top plate is rotationally connected with the side edge of one group of the box body side plate. As for the machine packaging box provided by theinvention, by arranging the lifting mechanism below the packaging box main body and utilizing original structure deformation of the packaging box main body, the machine packaging box has two purposesas being capable of packaging a transported product and being used by doubling as the workbench of a machine, lowers the cost of additionally purchasing the workbench, avoids resource waste caused bypackaging box abandoning, has very good practical value and is worthy of being widely popularized and used.
Owner:CHANGZHOU INST OF TECH
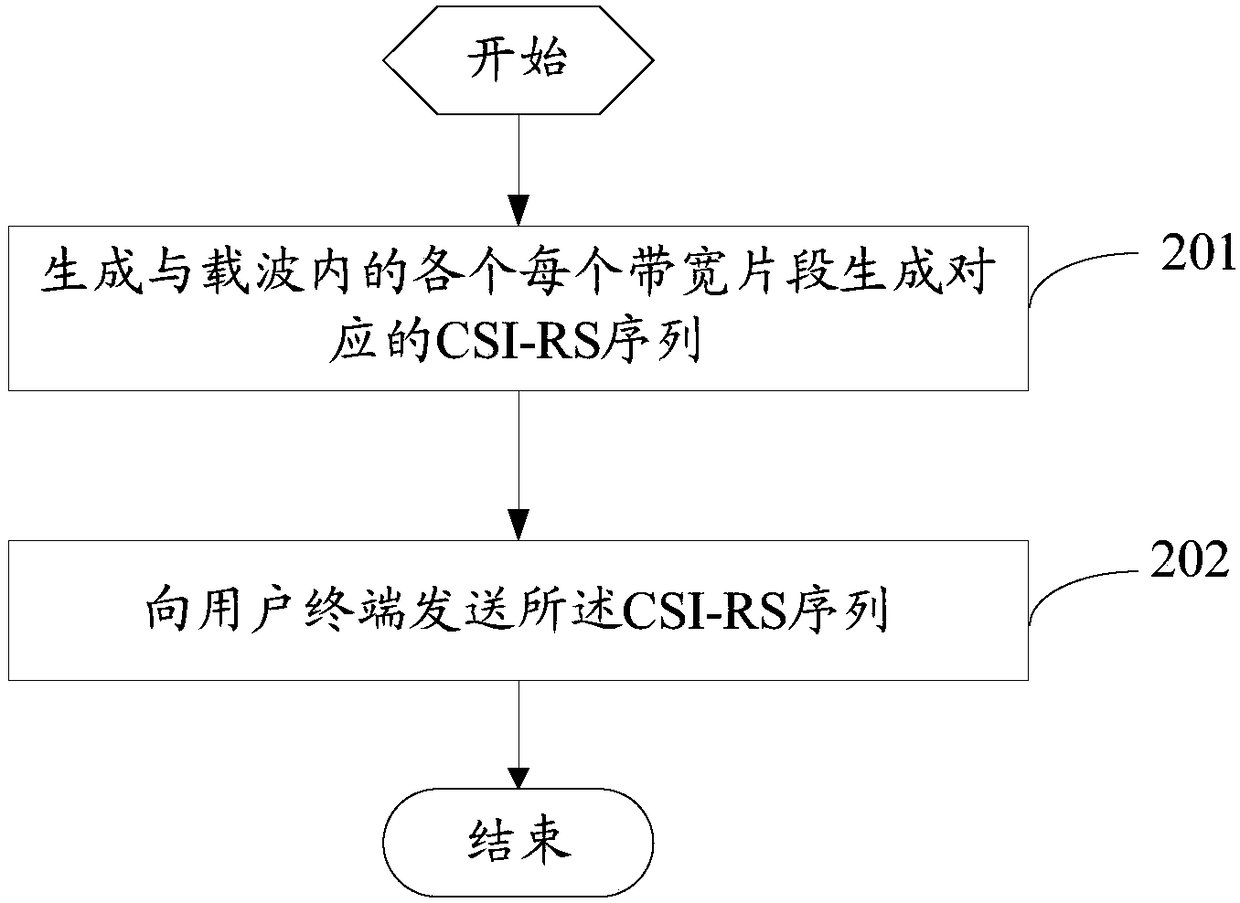
Sending method and receiving method of CSI-RS sequence, related device and system
ActiveCN108809494AImprove performanceTroubleshoot configuration issuesBaseband system detailsSignalling characterisationCarrier signalReal-time computing
The invention provides a sending method and a receiving method of a CSI-RS sequence, a related device and a system. The sending method comprises the following steps: generating the CSI-RS sequence corresponding to each bandwidth segment in a carrier; and sending the CSI-RS sequence to a user terminal. According to the sending method provided by the invention, the corresponding CSI-RS sequence is configured to each bandwidth segment, thereby solving the CSI-RS configuration problem when different carrier intervals exist in the carrier, and improving the performance of the CSI-RS sequence and the flexibility of system transmission.
Owner:VIVO MOBILE COMM CO LTD
Folding and unfolding method of large-area and small-length-width ratio satellite borne planar radar antenna
InactiveCN106785307ASolve the problem of small aspect ratio and large area planar radar antenna configuration layoutTroubleshoot configuration issuesCollapsable antennas meansAntenna arraysWidth ratioSatellite
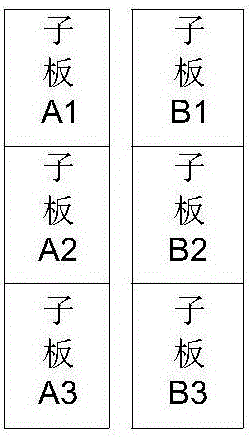
The invention relates to a folding and unfolding method of a large-area and small-length-width ratio satellite borne planar radar antenna. The unfolding process of the folding and unfolding method includes a blocking stage and an unfolding stage. The blocking stage includes the following steps that: a plane phased array antenna is divided into an A sub-array and a B sub-array in a width direction, wherein the two sub-arrays are respectively installed on two side surfaces of the body of a satellite; and the A sub-array is divided into two or three sub-plates in a length direction which are denote as A1, A2 and A3 respectively, and the B sub-array is divided into two or three sub-plates in a length direction which are denote as B1, B2 and B3 respectively. The unfolding stage includes following steps that: the sub-plates A1, A2 and A3 are unfolded through an unfolding mechanism so as to form the complete A sub-array, and the sub-plates B1, B2 and B3 are unfolded through an unfolding mechanism so as to form the complete B sub-array; and the B sub-array is rotated, so that the B sub-array and the A sub-array can be spliced together to form the complete plane phased array antenna. With the folding and unfolding method of the invention adopted, the problem that the large-area and small-length-width ratio satellite borne planar radar antenna cannot be mounted on the satellite can be solved.
Owner:SHANGHAI SATELLITE ENG INST
Preparation method of trans-N-Boc-3-amino-4-hydroxypiperidine
InactiveCN103664742AHigh ee valueResponse and application convenienceOrganic chemistryKetoneCarbonyl group
The invention discloses a preparation method of trans-N-Boc-3-amino-4-hydroxypiperidine, mainly aiming to solve the problem that the compound is needed to be chirally resolved because of having stereoisomerisms. The preparation method comprises the following steps: firstly, with a compound N-Boc-4-piperidone as a starting raw material, enabling N-Boc-4-piperidone to react with dibenzyl diazene-1, 2-dicarboxylate in the presence of a catalyst amino acid to obtain (R)-3-[N, N'-bis(carbobenzoxy)hydrazine]-N-Boc piperidine-1-ketone; secondly, enabling (R)-3-[N, N'-bis(carbobenzoxy)hydrazine]-N-Boc piperidine-1-ketone to react with a reducing reagent at the temperature of 0 DEG C to obtain (R)-3-[N, N'-bis(carbobenzoxy)hydrazine]-N-Boc piperidine-1-aocohol; thirdly, enabling (R)-3-[N, N'-bis(carbobenzoxy)hydrazine]-N-Boc piperidine-1-aocohol to react in the presence of a metal hydrogenation catalyst or samarium diiodide to obtain (R)-3-[N-(carbobenzoxy)amino]-N-Boc piperidine-1-aocohol; fourthly, reducing (R)-3-[N-(carbobenzoxy)amino]-N-Boc piperidine-1-aocohol in the presence of the metal hydrogenation catalyst or hydrogen gas to obtain the trans-N-Boc-3-amino-4-hydroxypiperidine. The total yield of the trans-N-Boc-3-amino-4-hydroxypiperidine is 33%.
Owner:上海药明康德新药开发有限公司 +3
Parameter configuration method and device of electric bus
ActiveCN106828140ATroubleshoot configuration issuesOperating modesPropulsion by batteries/cellsPower batteryElectrical battery
The invention discloses a parameter configuration method and device of an electric bus. The method comprises the following steps that a plurality of parameters of the electric bus are obtained; a battery residual quantity matrix is built according to the battery residual quantity of the electric bus at every dispatch running time, and a battery charger charging state matrix is built according to the charging states of a battery charger at every dispatch running time; order of the battery residual quantity matrix is calculated, the minimum configuration quantity of the bus is obtained, order of the battery charger charging state matrix is calculated, and the minimum configuration quantity of the battery charger is obtained; and according to a pre-build consumption model, the minimum configuration quantity of the bus and the minimum configuration quantity of the battery charger are input into the consumption model, and a parameter, corresponding to the minimum output from the consumption model, of the electric bus serves as an optimal parameter of the electric bus. According to the parameter configuration method and device of the electric bus, the technical problem that in the prior art, optimal quantity configuration of a power battery and the battery charger cannot be obtained due to the fact that the battery capacity of the electric bus and the quantity of the battery charger are not considered by electric bus operation is solved.
Owner:STATE GRID BEIJING ELECTRIC POWER +1
Data acquisition and transmission system and method for locally upgrading and deploying parameters
PendingCN112650517AGuaranteed accuracyAchieve upgradeVersion controlTransmissionData acquisitionLogic module
The invention discloses a data acquisition and transmission system and a method for locally upgrading and deploying parameters, and mainly relates to the field of software upgrading of embedded internet-of-things equipment. The system comprises a local upgrading subsystem and a parameter deploying subsystem, wherein the local upgrading subsystem is used for local upgrading of a data acquisition and transmission system, the local upgrading subsystem comprises an APP logic module and a BOOT guiding module, the parameter deploying subsystem is used for parameter deploying of the data acquisition and transmission system, and the parameter deploying subsystem comprises a function module. The function module comprises a parameter adjustment mode identification sub-module and a parameter adjustment instruction execution sub-module. The beneficial effects of the invention are that the method achieves the local online upgrading and parameter deployment of the IOT equipment, greatly reduces the time cost and manpower cost, solves a problem of timeliness caused by the time and region difference, and enables the equipment operation and maintenance and on-site debugging to be simpler and more convenient.
Owner:山东向明数智物联科技有限公司
Configuration method of cell code source in time-division synchronous CDMA system group network
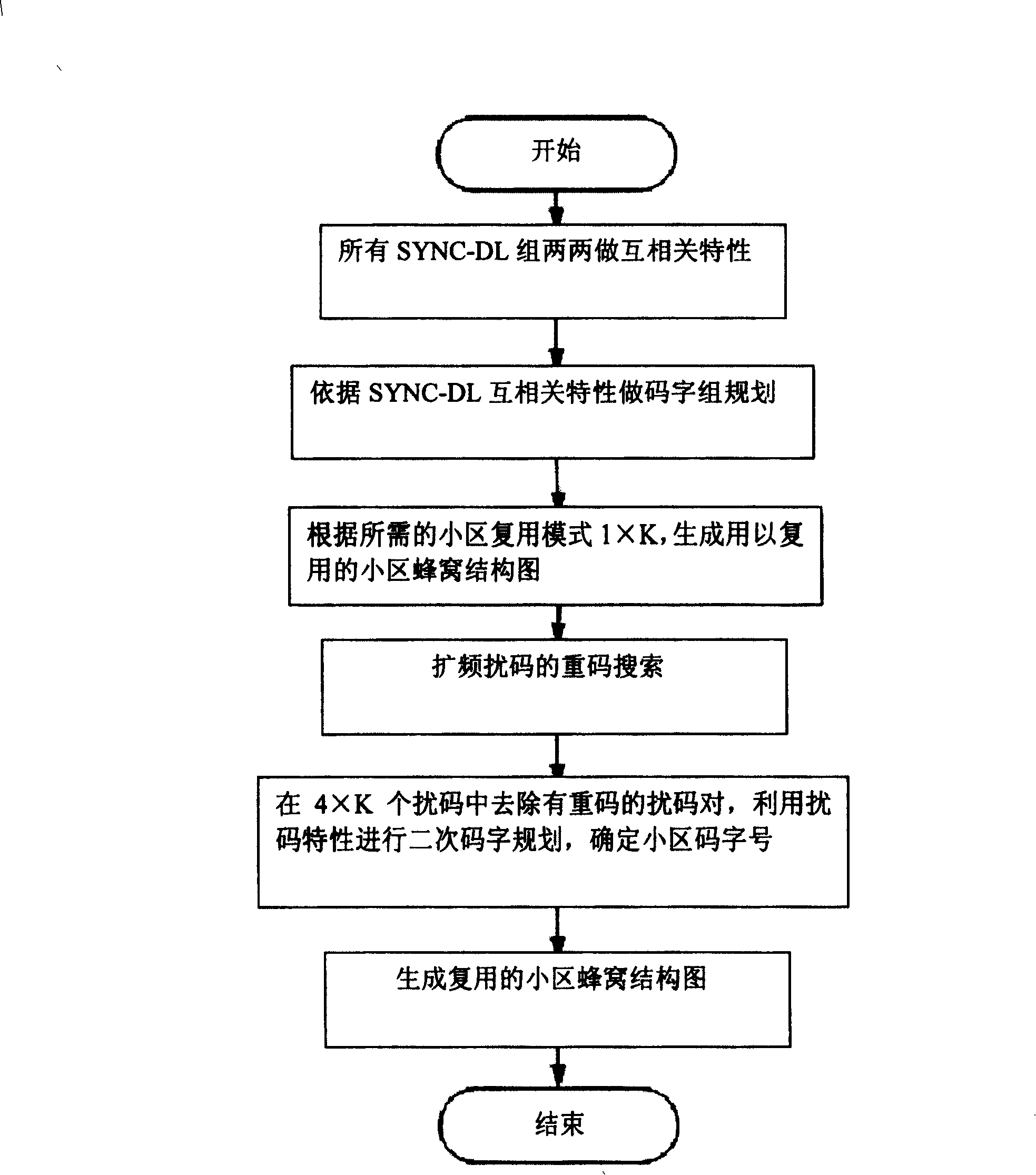
InactiveCN100407600CCorrect accessTroubleshoot configuration issuesCode division multiplexRadio transmission for post communicationComputer hardwareTD-SCDMA
This invention relates to a configuration method for local area code work resource in TD-SCDMA system network, which utilizes the SYNC-DL code property to carry out first code word design on the basis of analyzing co-relation property of code words in TD-SCDMA system code set to eliminate the coincide codes of spread spectrum interfere codes, then utilizes the property of the perturbation codes to carry out a second time code word design to get a code word configuration pattern of a honey comb after multiplexing local area code words.
Owner:JIANGSU HONGCHENG FORGING MACHINE TOOL
Information configuration method, base station and user device
InactiveCN107453885ATroubleshoot configuration issuesImplement configurationData switching networksWireless communicationRadio Link ControlUser equipment
The embodiment of the invention discloses an information configuration method, comprising the steps that a base station sends first configuration information to a centralized radio link control (C-RLC) layer established by the base station, thereby configuring the C-RLC layer, wherein the first configuration information at least comprises C-RLC layer configuration related information and control related information for the C-RLC layer to control at least one distributed radio link control (D-RLC) layer; and the base station sends second configuration information to the at least one D-RLC layer established by the base station, thereby configuring the at least one D-RLC layer, wherein the second configuration information at least comprises D-RLC layer configuration related information. The embodiment of the invention also discloses the base station and a user device.
Owner:CHINA MOBILE COMM LTD RES INST +1
Method and system for configuring information delivery
InactiveCN103731437BTroubleshoot configuration issuesRealize business configurationTransmissionConfiguration itemClient-side
Owner:阿里巴巴(成都)软件技术有限公司
Data configuration method, equipment and system
ActiveCN101860458BTroubleshoot configuration issuesEasy to handleData switching networksData fileStructure of Management Information
The invention discloses a data configuration method, data configuration equipment and a data configuration system. The data configuration method is applied to a network system comprising a network management server, the data configuration equipment and at least one network element, wherein the network element comprises a database. The method comprises that: the data configuration equipment receives a data file sent by the network management server, wherein the data file comprises configuration data for configuring the database of the network element; the data configuration equipment resolves the data file, and acquires the configuration data in the data file; and the data configuration equipment loads the configuration data to the database of the network element according to a preset table method. The data configuration equipment can load the configuration data to the database of the network element by resolving the data file, so the processing flow is simple, the problems of high difficulty and high cost brought by developing a universal structural language of the database are reduced, and the problem of universal data configuration can be quickly solved.
Owner:DATANG MOBILE COMM EQUIP CO LTD
Device and method for testing basic parameters of coal seam gas and predicting gas accumulation zone
ActiveCN110307035BTroubleshoot configuration issuesReduce adverse effectsMining devicesThermodynamicsProcess engineering
The present invention provides a device and method for testing the basic parameters of coal seam gas and predicting the gas accumulation zone, so as to overcome the defects of complex methods for testing the basic parameters of coal seam gas and few methods for predicting the gas accumulation zone on site, and provide a method for testing the basic parameters of coal seam gas The wireless device and method for parameterizing and predicting the gas accumulation zone make its data transmission stable and reliable, so it is more suitable for practical use and has industrial utilization value. It belongs to the field of mine gas control and monitoring.
Owner:GUIZHOU UNIV
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com