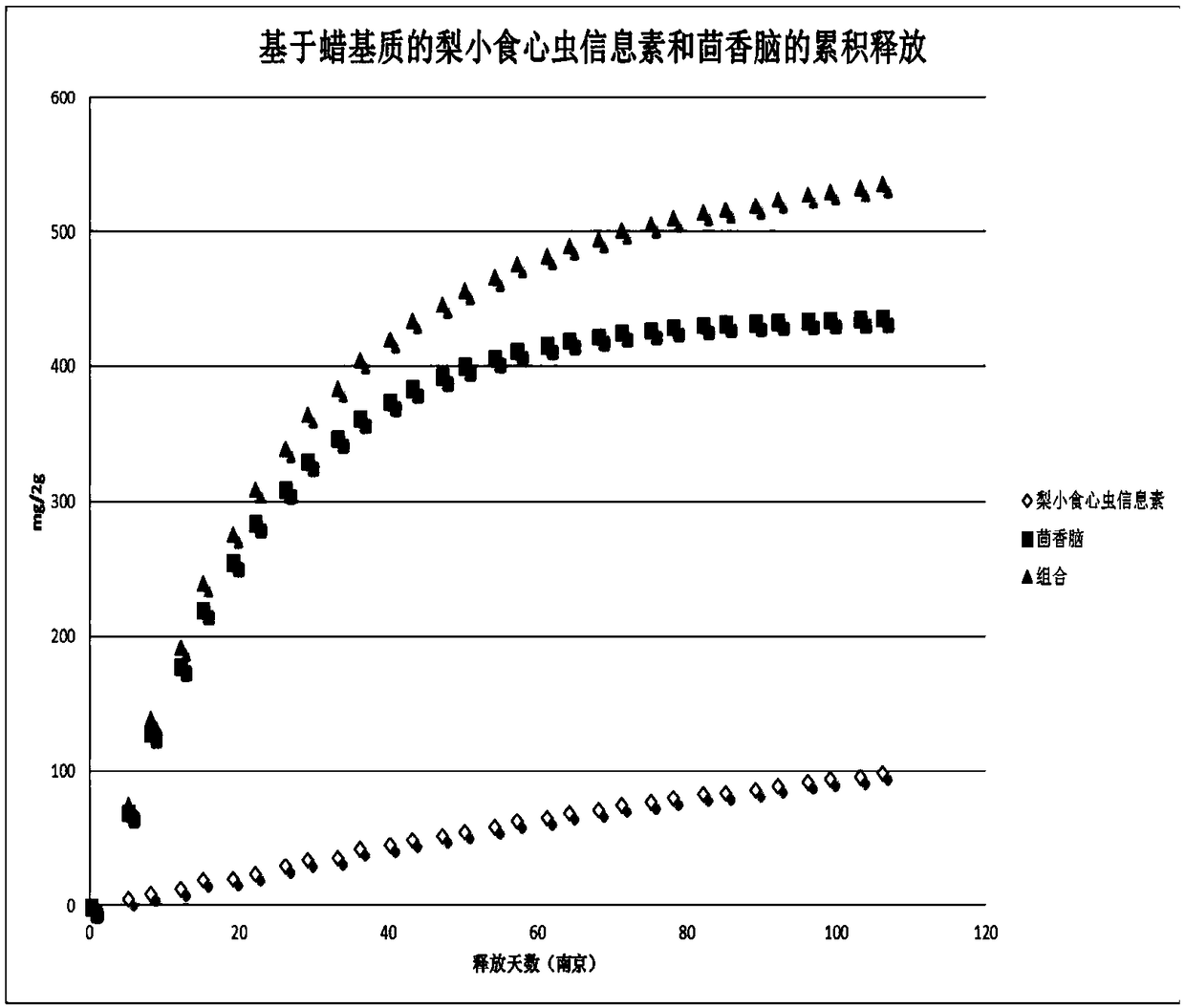
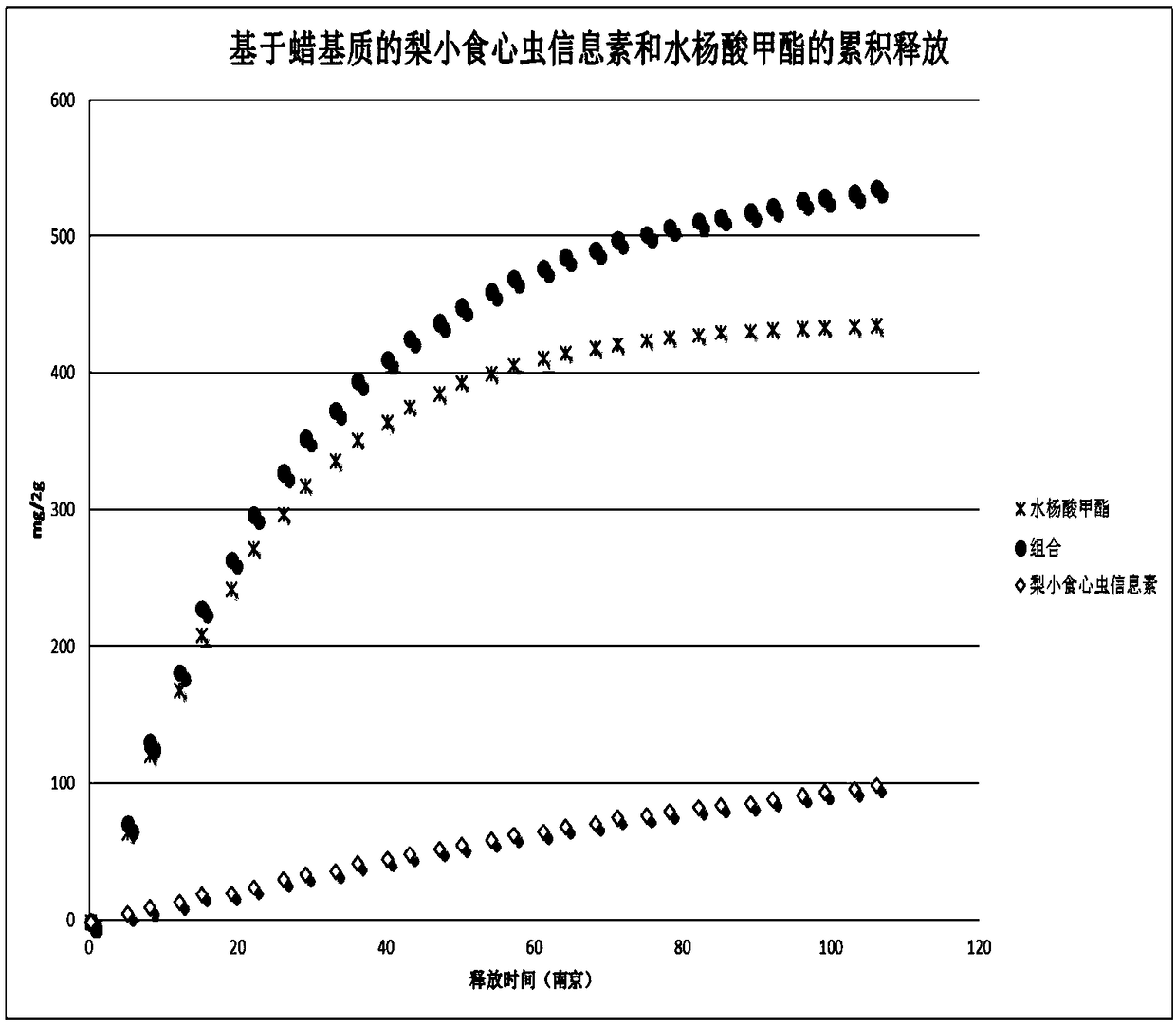
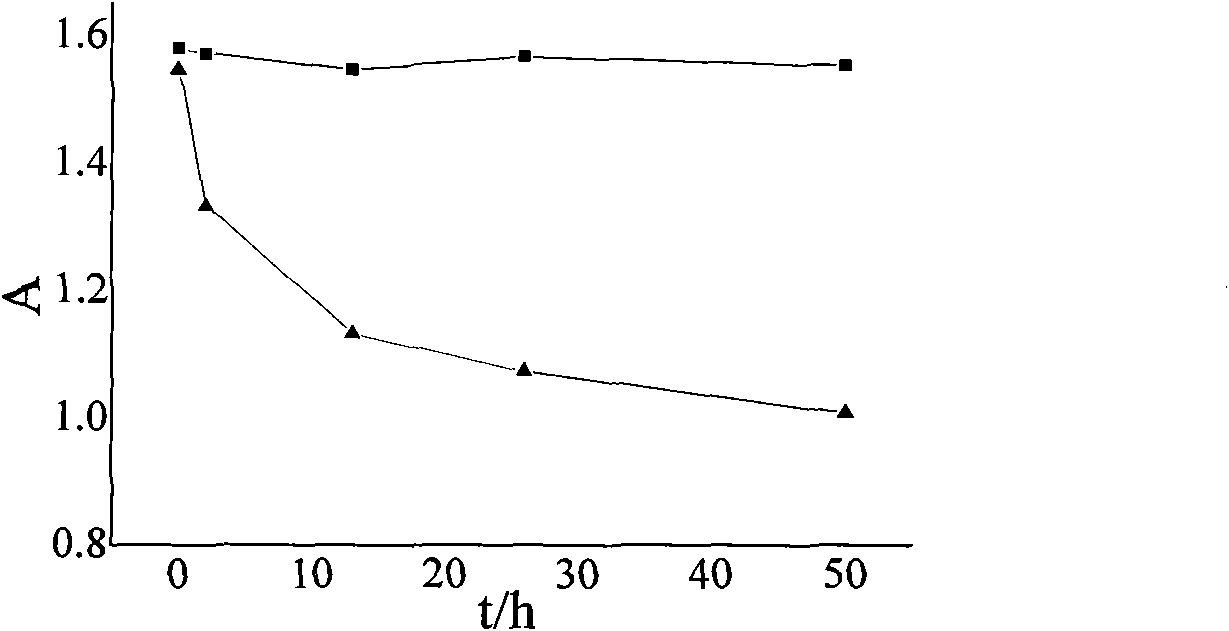
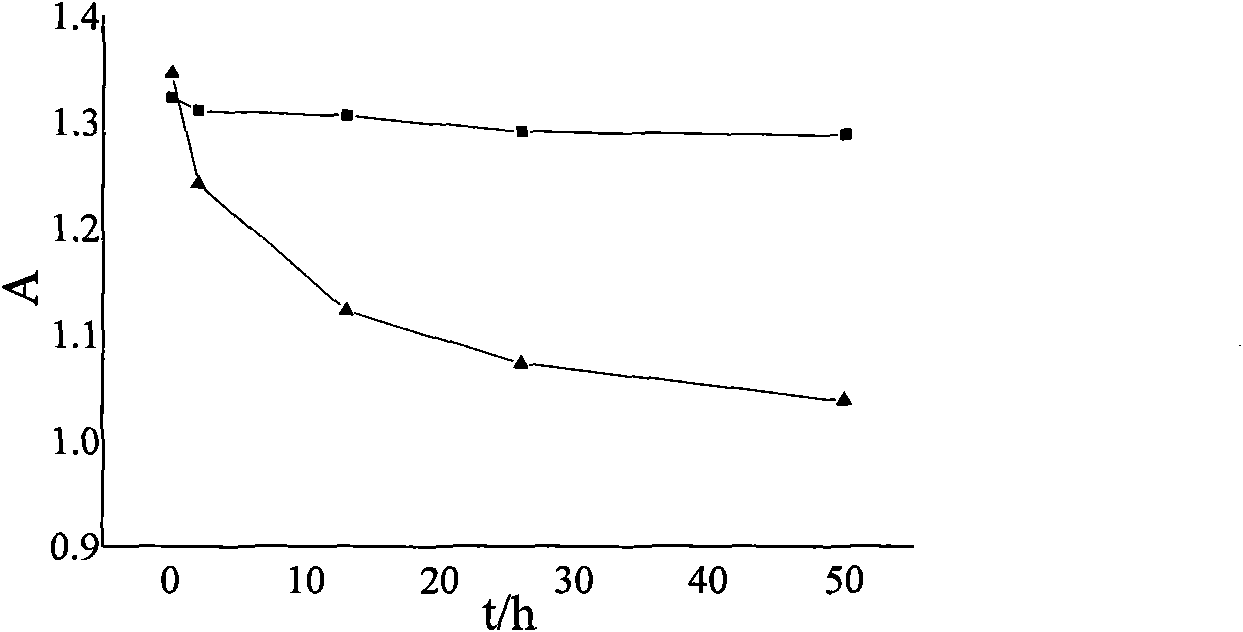
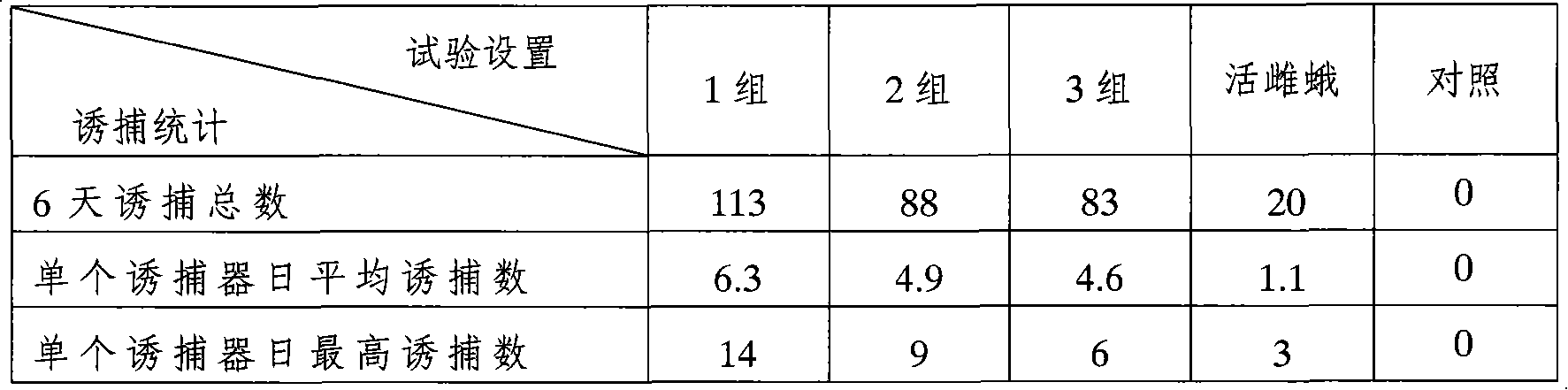
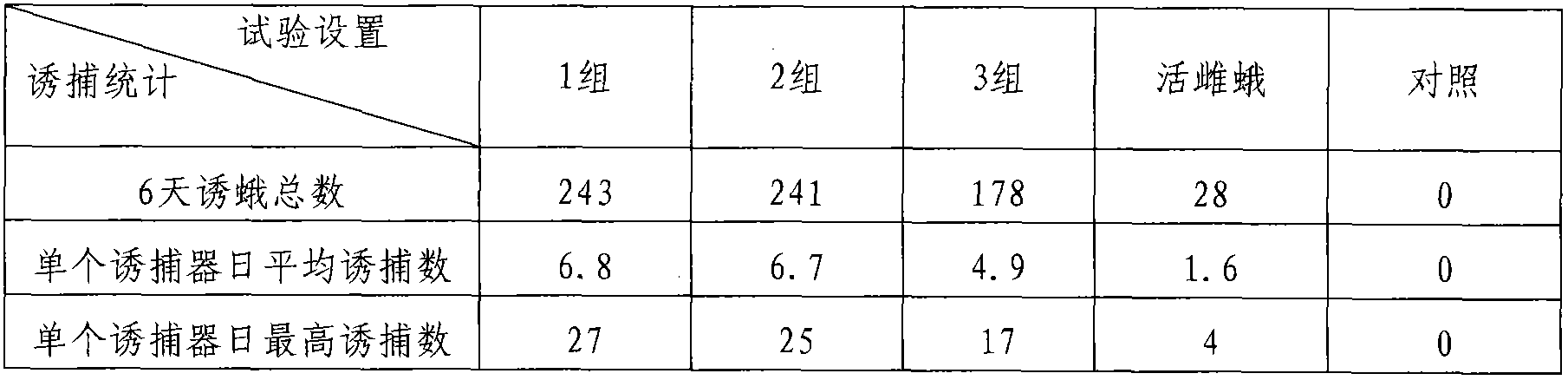
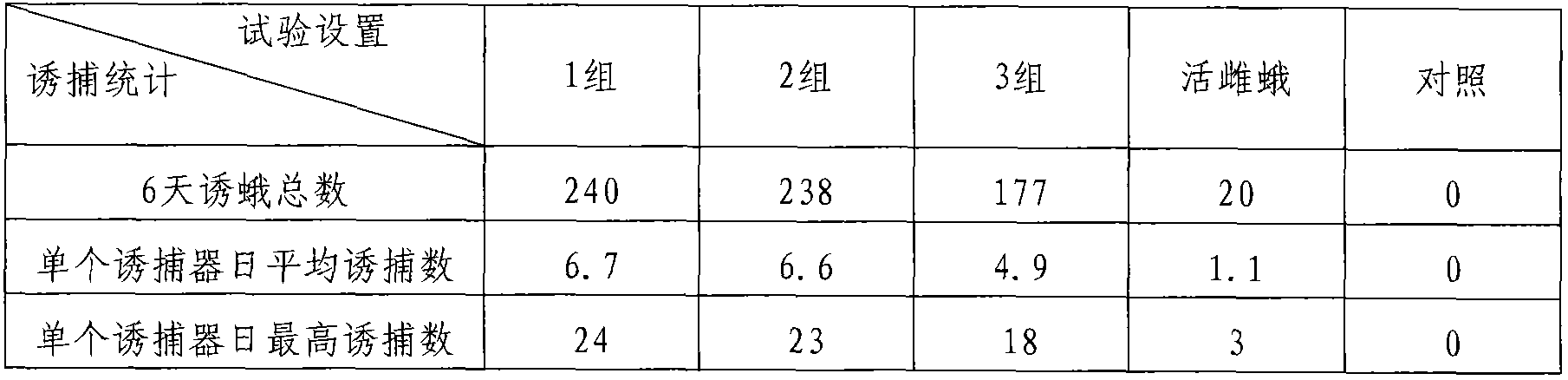
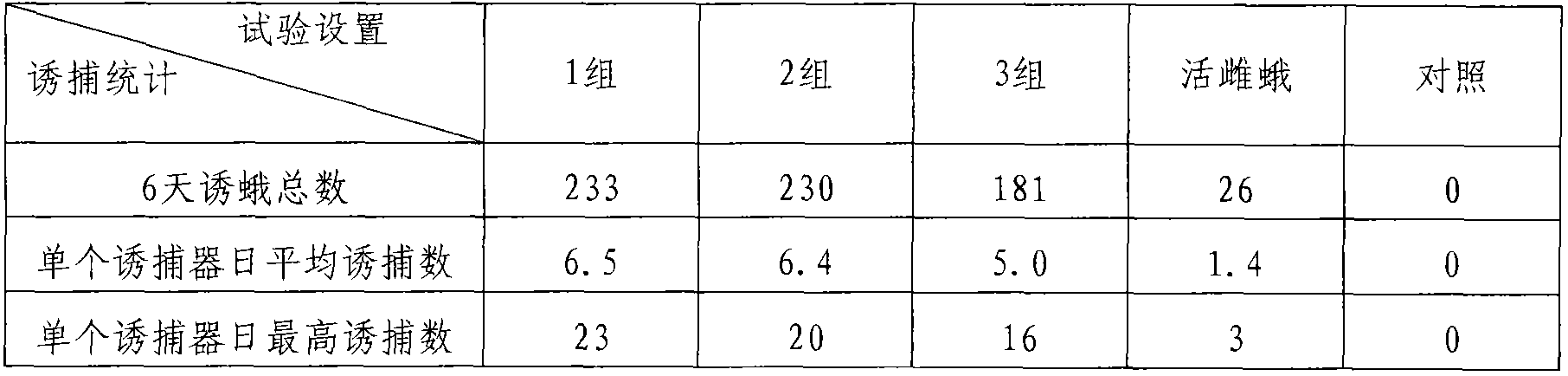
Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
87results about "Chemosterilants" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Pesticidal compositions
Owner:DOW AGROSCIENCES LLC
Pesticidal compositions
Owner:CORTEVA AGRISCIENCE LLC
Peptide analogue juvenile hormone synthesis inhibitor containing phenylpropyl alcohol-glycerol-bright tripeptide fragment
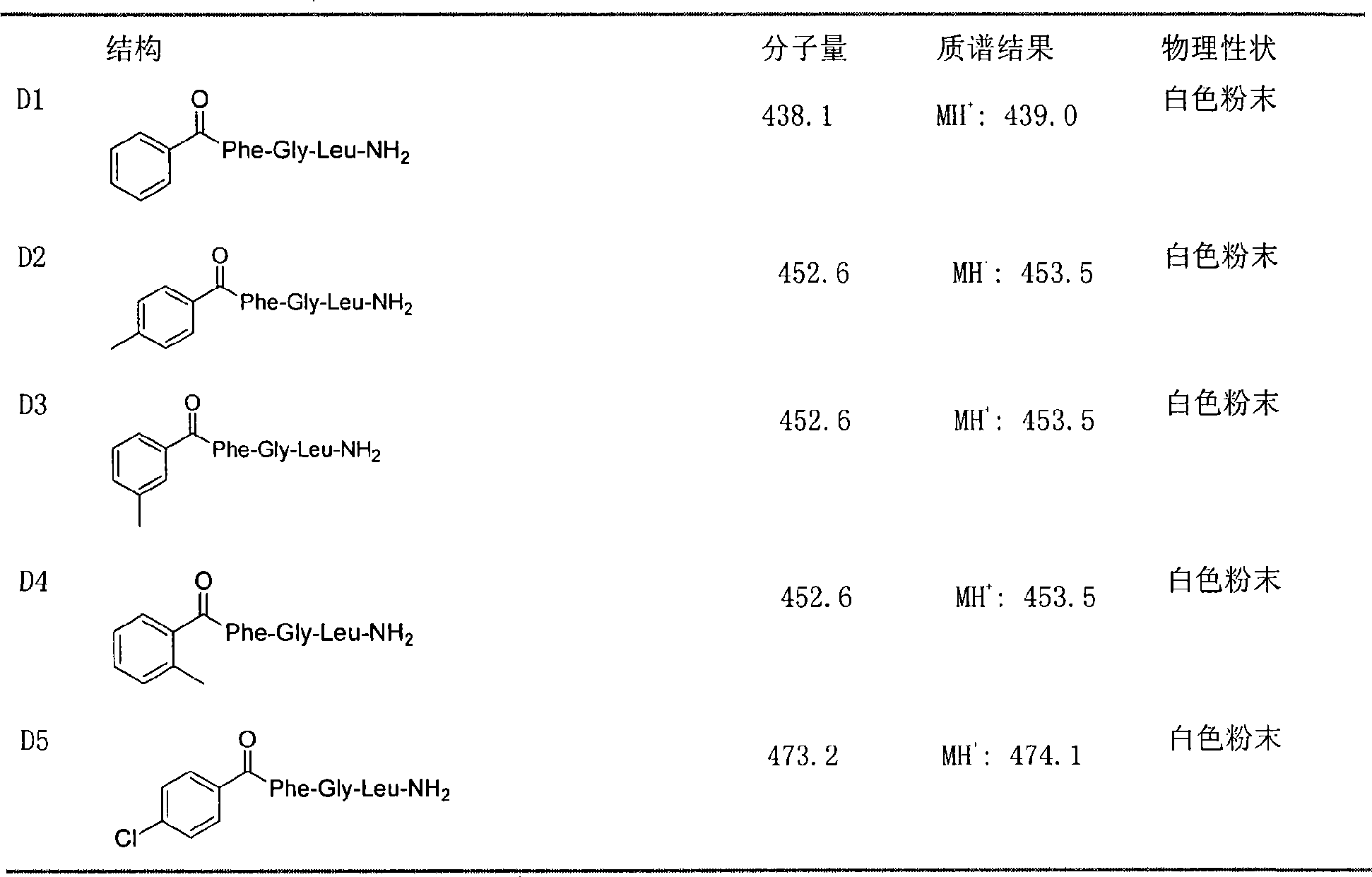
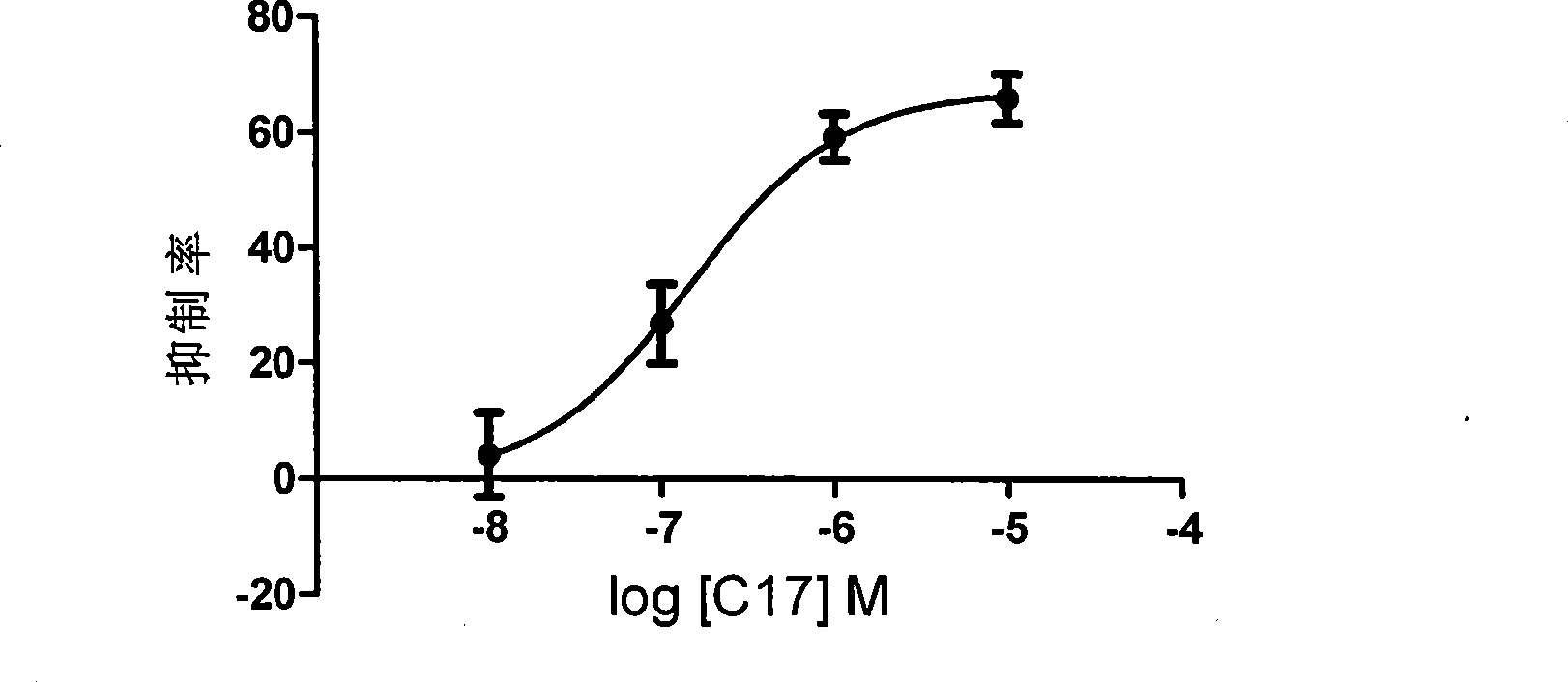
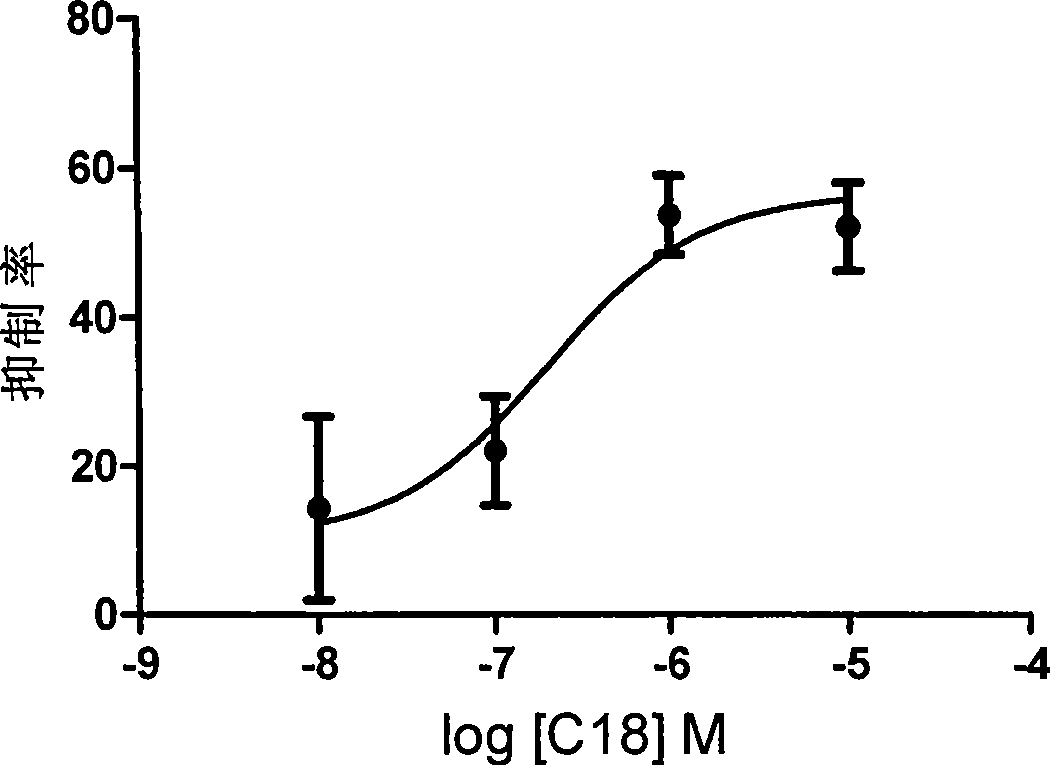
The invention discloses a novel peptide analogue juvenile hormone synthesis inhibitor which has a structure general formula D: R-Phe-Gly-Leu-NH2, wherein the R stands for (substituted) benzoyl, (substituted) benzene acetyl, (substituted) hydrocinnamoyl, (substituted) butylbenzene, (substituted) benzene valeryl, (substituted) benzene hexanoyl, (substituted) benzene acryloyl, diacid, fatty acid, hydrophobic amino acid, or the like. For the compound with the formula D, a tripeptide fragment Phe-Gly-Leu-NH2 of AST is used as a primer, and an N-end modification method is adopted to obtain a novel peptide analogue compound containing Phe-Gly-Leu-NH2 tripeptide which has simple structure. The activity of most compounds with the formula D is superior to that of the tripeptide fragment Phe-Gly-Leu-NH2, and the activity of an individual compound is equal to or superior than the activity of core pentapeptide of the natural AST. In the further application study, the partial compounds with the formula D have better in-vivo activity, thereby being the novel peptide analogue juvenile hormone synthesis inhibitor.
Owner:CHINA AGRI UNIV
Novel pentapeptide analogue and application thereof
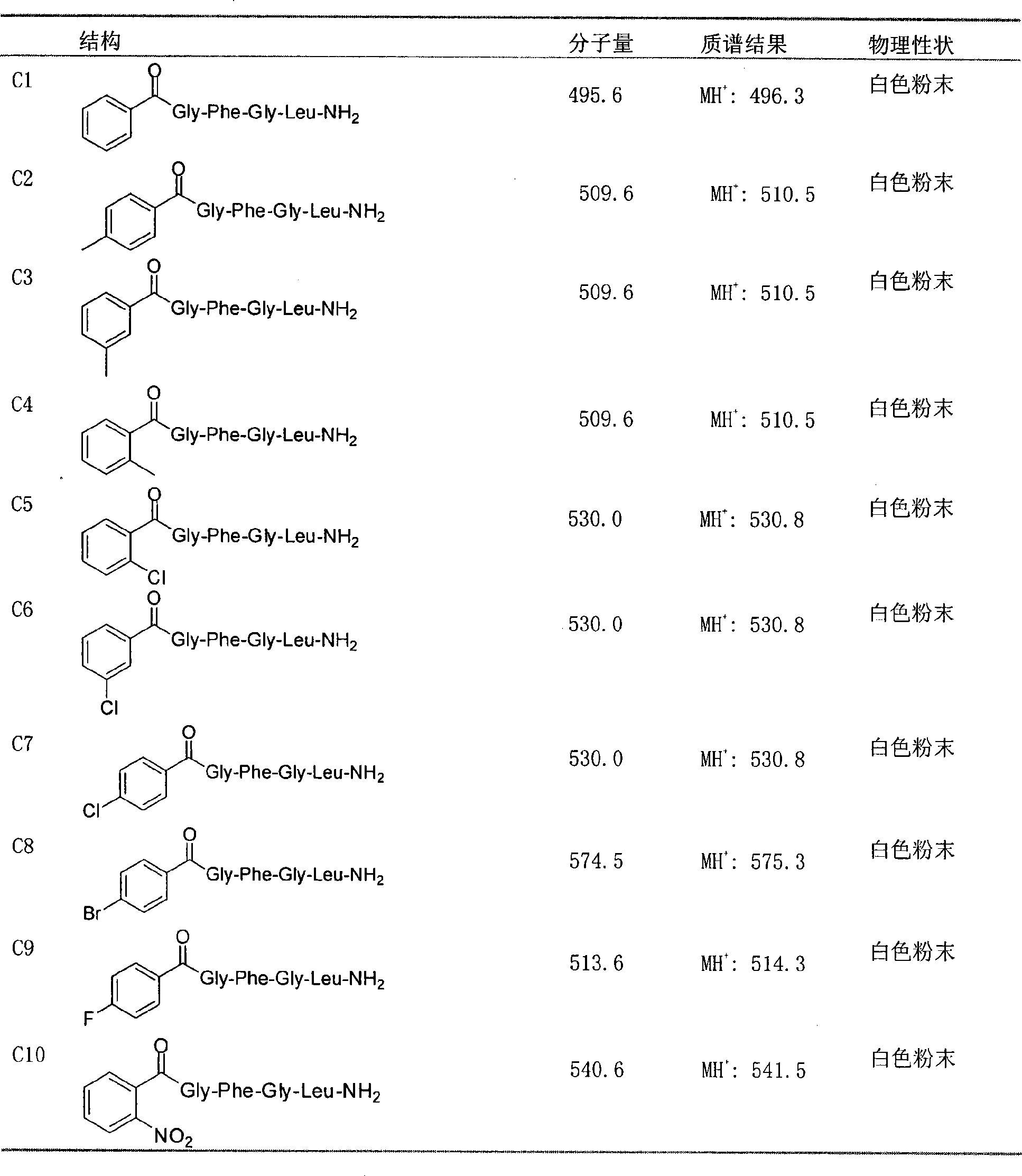
The invention discloses a novel pentapeptide analogue and application thereof. The structure general formula of the compound provided by the invention is C: CR-Xaa-Phe-Gly-Leu-NH2, wherein the Xaa stands for the Gly, Asp, Asn, Ser or Ala; and the R stands for (substituted) benzoyl, (substituted) benzene acetyl, (substituted) hydrocinnamoyl, (substituted) butylbenzene, (substituted) benzene valeryl, (substituted) benzene hexanoyl, (substituted) benzenesulfonyl, 9-dimethoxycarbonyl, or benzyloxycarbonyl. Various organic acids with aromatic rings are introduced to replace Y / F in AST core pentapeptide (Y / FXFGLa) to prepare the pentapeptide analogue with simple structure and good inhibited juvenile hormone synthesis in-vitro activity, and the activity of partial compound with the formula C is superior to that of natural core pentapeptide. The further research indicates that partial compound with the formula C has better in-vivo activity, and the compound with the formula C conforms to the characteristic of an insect growth regulator and has better development and application prospects.
Owner:CHINA AGRI UNIV
Novel juvenile hormone synthesis inhibitor-phenylpropyl alcohol-glycerol-bright tripeptide amide analogue
InactiveCN101519430ASuppress generationConsistent with the properties of growth regulatorsBiocideChemosterilantsChemical structureAlcohol
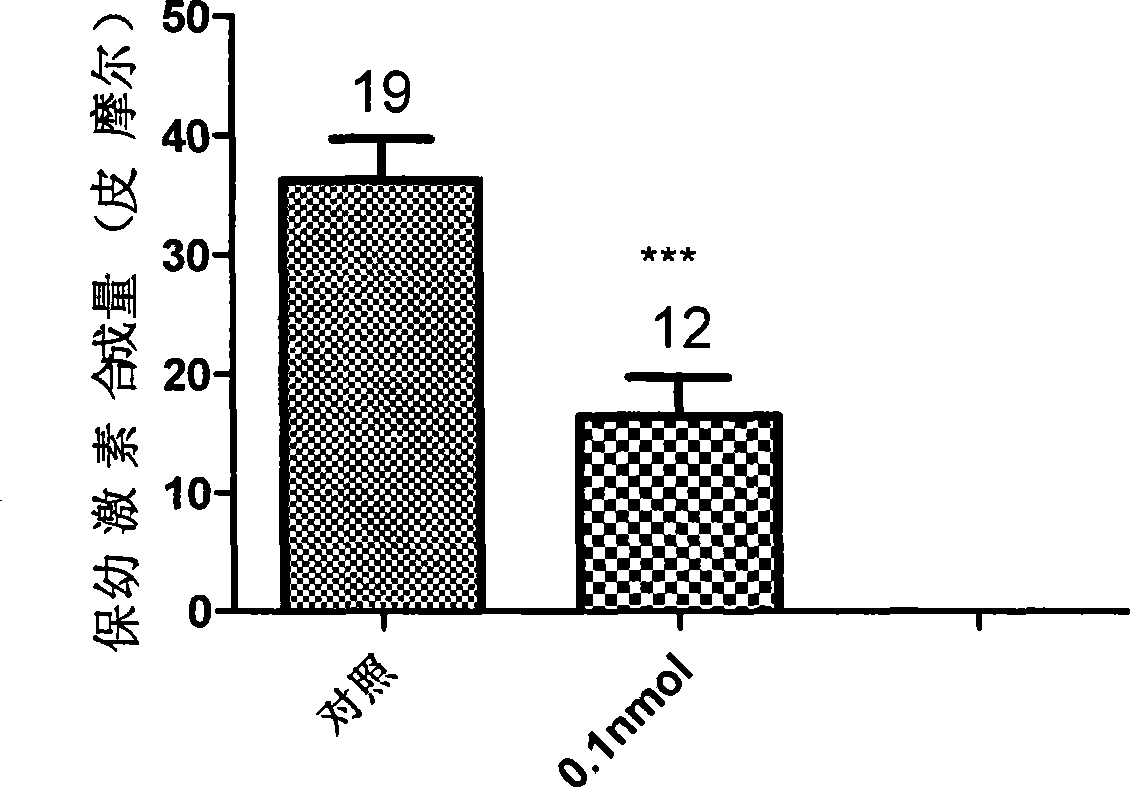
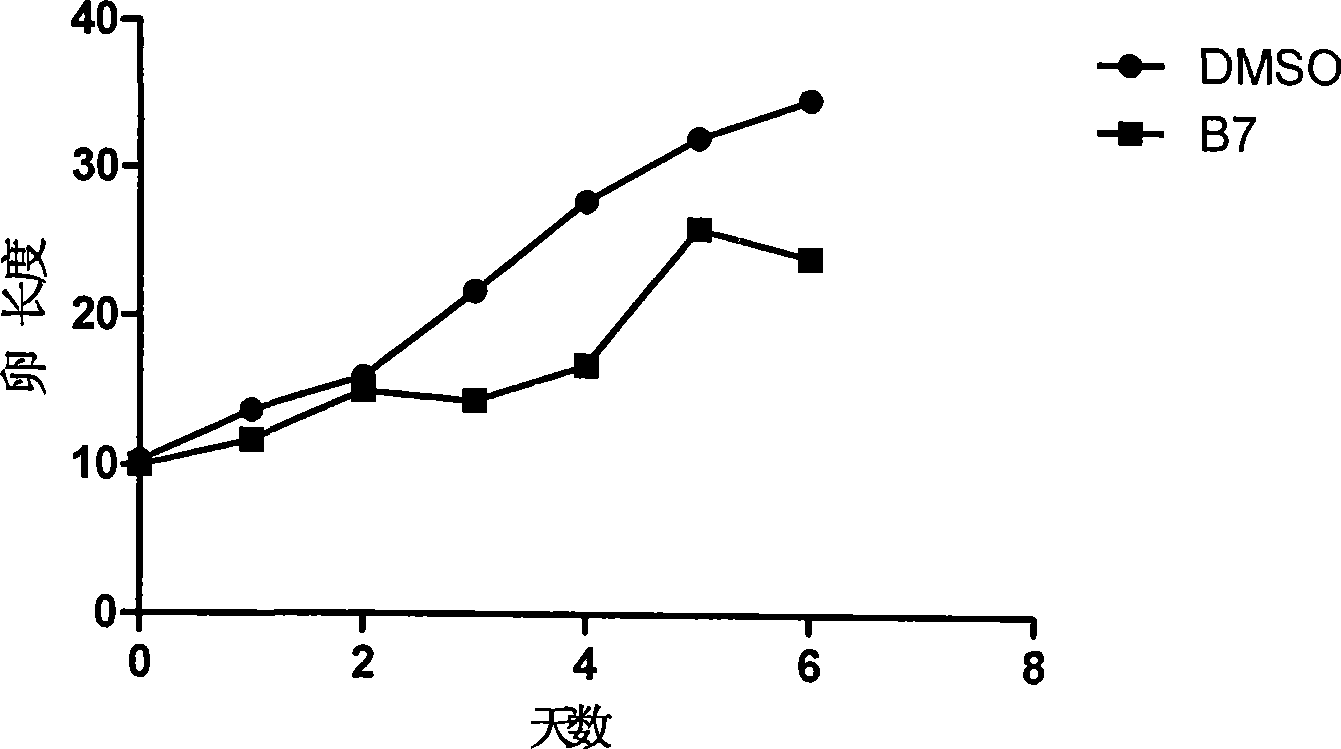
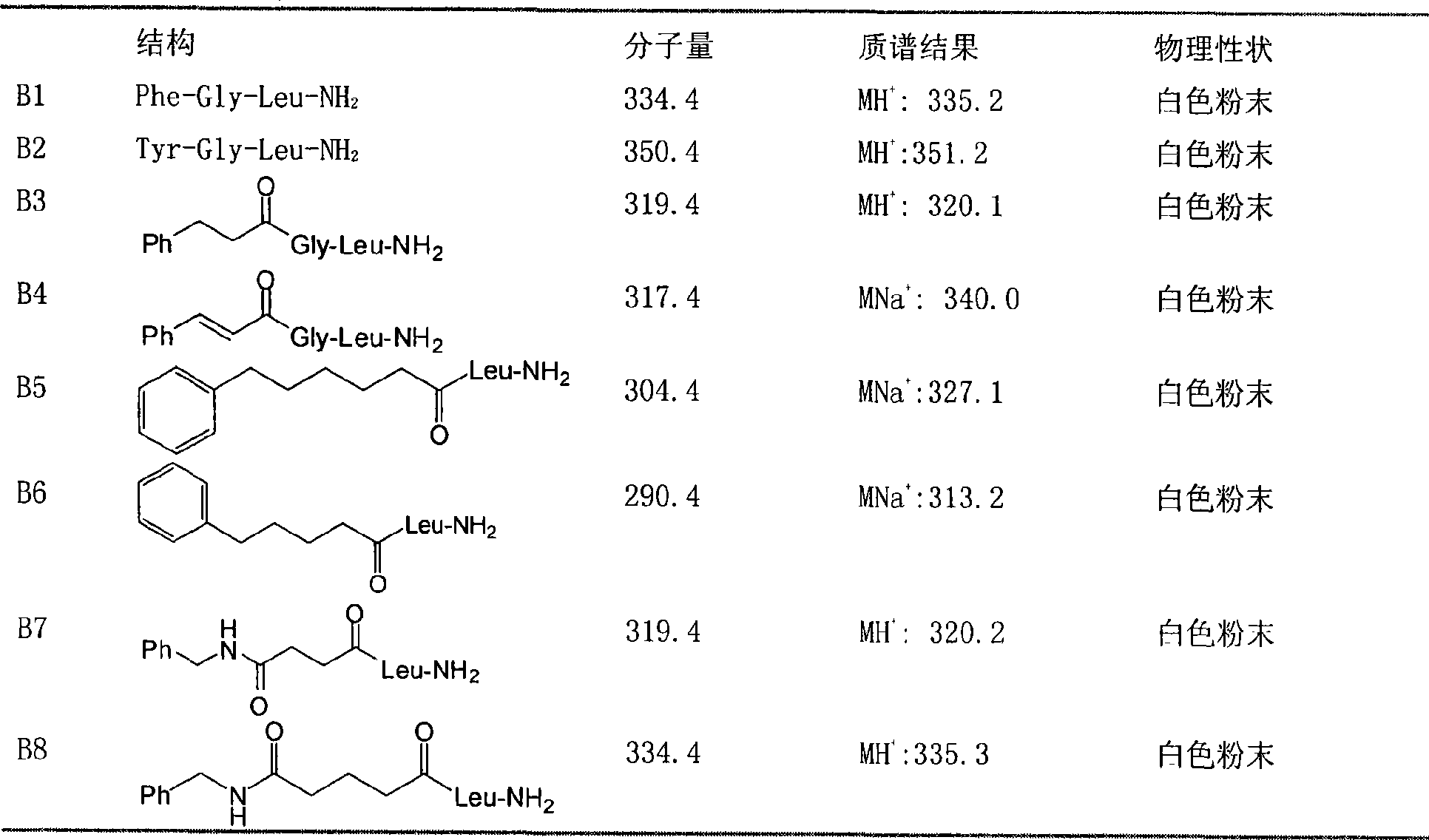
The invention discloses a juvenile hormone synthesis inhibitor with novel structure, which is a phenylpropyl alcohol-glycerol-bright tripeptide amide analogue. The structure general formula of the compound provided by the invention is B: R-Leu-NH2, wherein the R stands for Phe-Gly-, Tyr-Gly-, PhCH2CH2CO-Gly-, PhCH=CHCO-Gly-, Ph(CH2)5CO-, Ph(CH2)4CO-, PhCH2NHCO(CH2)2CO-, PhCH2NHCO(CH2)3CO- or PhCH2NHCOCH2CO-. The compound with formula B simulates the chemical structure of a PHe-Gly-Leu-NH2 fragment of an A type AST and has simple structure and low synthesis cost. The compound has better inhibition activity for the juvenile hormone synthesis of black beetles. In the further application study, the compound obviously influences reproductive systems of female black beetles, inhibits the generation of black beetle eggs, conforms to the characteristic of an insect growth regulator and has better development and application prospects.
Owner:CHINA AGRI UNIV
Release-controlled preparation for simultaneously interfering pest mating and attracting beneficial insects and application
ActiveCN108967423AQuick releaseReduce releaseBiocideChemosterilantsPaleontologyPhases of clinical research
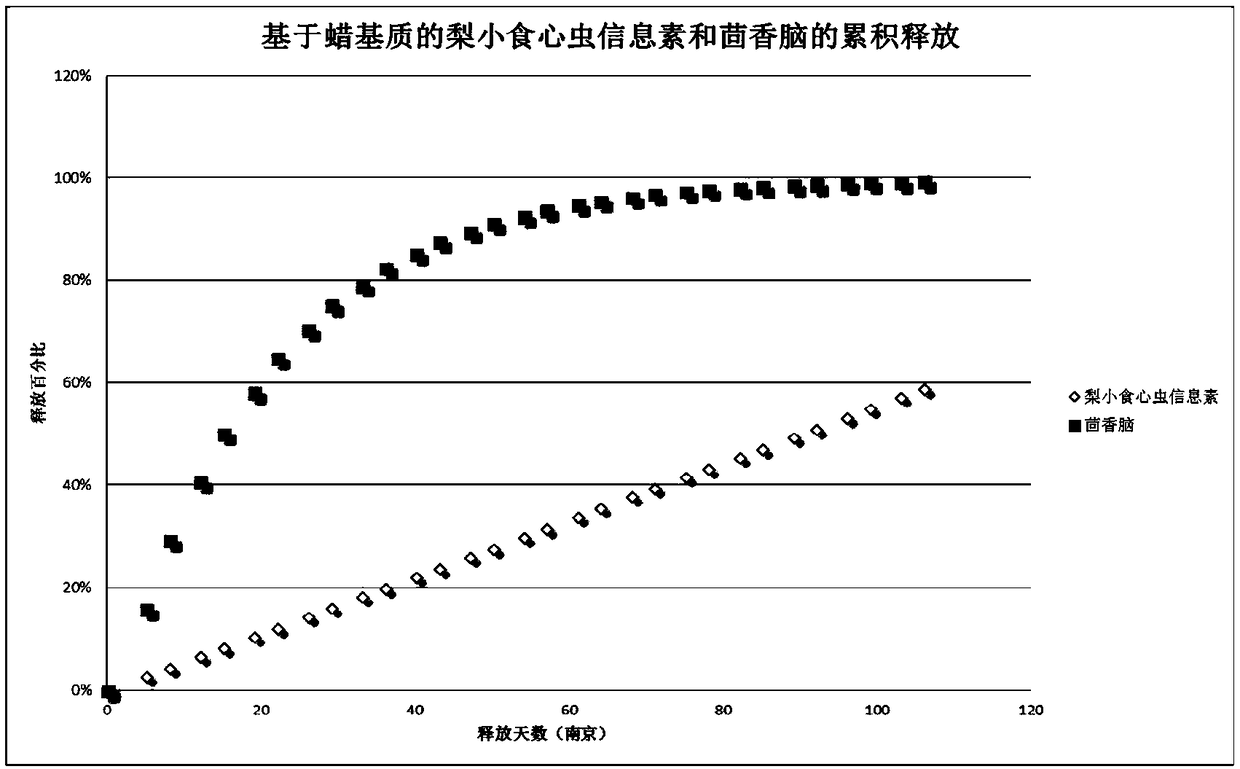
The invention provides a release-controlled preparation for simultaneously interfering pest mating and attracting beneficial insects. The release-controlled preparation comprises pheromone for interfering the pest mating and an attractant for attracting the beneficial insects. The release-controlled preparation has the advantages that the release-controlled preparation is released into the surrounding environments in a gas form, so as to interfere the mating of the target insects in the effective stage, and attract the beneficial insects, such as bees; more importantly, the release rate of thepheromone for interfering the mating and the release rate of the attractant for attracting the target insects are different, so as to ensure that the different volatiles can complete the respective functions in the same carrier at different stages.
Owner:NANJING SINO GREEN BIOTECH
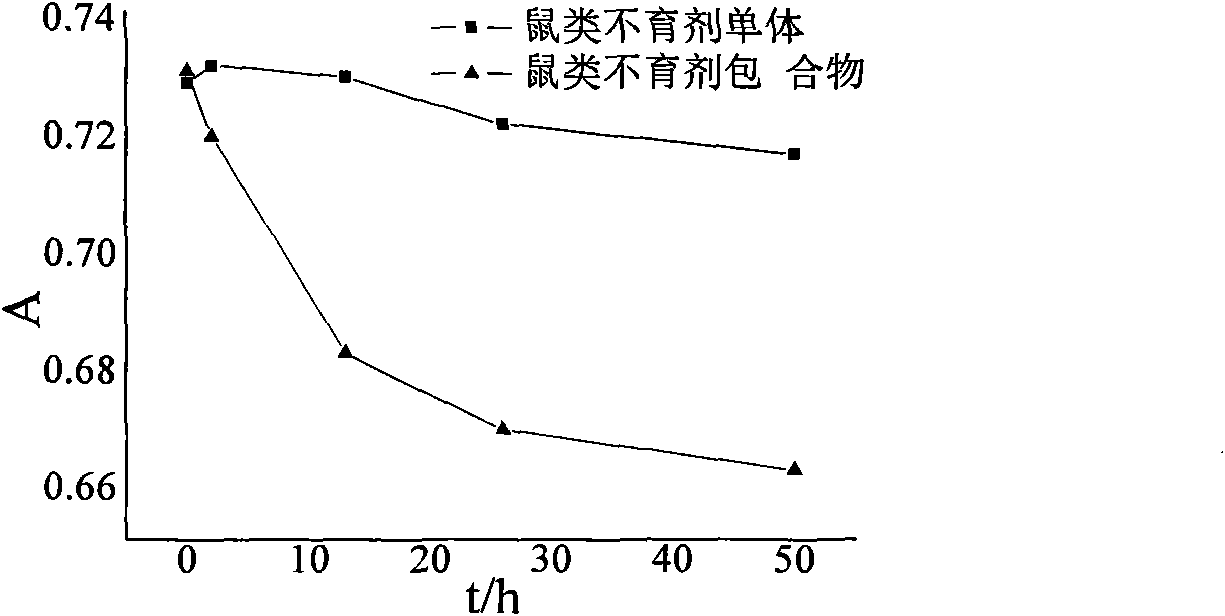
Cyclodextrin inclusion compound of rodents sterilant and preparation method thereof
InactiveCN101869113AGood water solubilityImproves UV stabilityBiocideChemosterilantsSolubilityMass ratio
The invention relates to the field of medicaments, in particular to a cyclodextrin inclusion compound of a rodents sterilant and a preparation method thereof. By the method, the rodents sterilant is mixed with cyclodextrin or a derivative of the cyclodextrin in a mass ratio of 1:1-200 to prepare the inclusion compound. The obtained inclusion compound not only has high water solubility, but also has a high ultraviolet stability. The invention initially provides the cyclodextrin inclusion compound of the rodents sterilant. The cyclodextrin inclusion compound has a tubular structure formed by glucosamine residues to form a unique conformation which is externally hydrophilic and internally hydrophobic, and can form a compound supramolecular system with an indissolvable compound to play a solubilization role and solve the problem of low water solubility of the conventional rodents sterilant; moreover, the inclusion compound has the high ultraviolet stability to meet the requirement of field administration; in addition, the cyclodextrin compound has no toxicity or low toxicity and is a good carrier for performing a medicinal effect research.
Owner:INST OF PLANT PROTECTION CHINESE ACAD OF AGRI SCI
Male sterile rodenticide and production thereof
A male sterile rodenticide is proportionally prepared from triptopolyglycoside and rat attractant prepared from bean cake powder or corn flour, bran, peanut cake powder or sesame cake powder, fish powder and oil. It has no secondary poisoning.
Owner:丁柯中
Fruit fly attractive toxicant and preparing technology
The invention relates to an insecticide product killing injurious insects, in particular to fruit fly attractive toxicant and a preparing technology thereof. The fruit fly attractive toxicant comprises fruit and vegetable food, yeast, vinegar, sweetening agents, insecticide and water or comprises a drink, vinegar, sweetening agents and insecticide. The whole attractive toxicant preparing process is nonhazardous and wholly environmentally friendly, cost is low, almost all families can prepare the attractive toxicant, and the attractive toxicant has a very good effect for trapping and killing fruit flies.
Owner:刘向阳
Sex attractant for holcocerus artemisiae
The present invention provides a sand sage cossus sex-attractant. It is characterized by that its effective component is one kind or several kinds selected from the following substances: cis-5-lanolin alcohol acetate (Z5-12:Ac), trans-3-cis-5-lanolin alcohol acetate (E3,Z5-12:Ac) and cis-5-tetradecylene alcohol acetate (Z5-14:Ac). The invented sand sage cossus sex-attractant can be used for effectively protecting natural enemy of sand sage cossus and can raise the effect for controlling sand sage cossus.
Owner:BEIJING FORESTRY UNIVERSITY +3
Female chemically sterilizing rat bait
InactiveCN101664039AImprove palatabilityImprove controlBiocideChemosterilantsEconomic thresholdEmbryo
The invention relates to a female chemically sterilizing rat bait, which is characterized by comprising 0.0002 to 0.002 percent of cabergoline, 0 to 1 percent of edible oil, 0.1 to 5 percent of ediblesugar and / or milk powder and the balance of one of unhulled rice, rice, wheat, corn, yellow bean the pea nut based on total 100 mass percent. The rat bait is high in palatability, long in control effect, safe and low in pollution, can reduce the population quantity of pest rats by over 80 percent within 3 months, ensures that most embryos are dead and that the larva survival rate is extremely lowor even zero and is simple in production process, low in cost, suitable to be promoted in large scale for controlling the population quantity of the pest rats and capable of controlling the hazard ofthe pest rats to be below an economic threshold for a long time.
Owner:GUANGDONG ENTOMOLOGICAL INST
Holcocerus vicarious walker sex attractant
InactiveCN102017945AUnderstand what happenedUnderstand situationBiocideChemosterilantsAlcoholEconomic benefits
The invention relates to a holcocerus vicarious walker sex attractant, aiming at solving the problem of prevention and control of holcocerus vicarious walker. The active ingredient of the holcocerus vicarious walker sex attractant comprises the following components: more than one of cis-7-tetradecene alcohol acetic ester, cis-3-tetradencene alcohol acetic ester, cis-3-trans-5-tetradecene alcohol acetic ester and cis-3-tetradencene alcohol in different weight portions. The sex attractant can be used for pest forcast, mating disruption or trapping massive holcocerus vicarious walker adults, chemical pesticide is not used, thus being environment-friendly and safe and being convenient to use, and meanwhile cost is low, duration is long, and the aim of effectively preventing and controlling holcocerus vicarious walker is achieved, thus having obvious biological benefit and economic benefit.
Owner:SHANXI AGRI UNIV +2
Synergistic method of insect growth regulator and nucleopolyhedron virus
InactiveCN1887093AImprove damage control effectImprove the level of biological controlBiocideChemosterilantsNuclear Polyhedrosis VirusNeurotoxicity
The present invention discloses the synergistic method for nucleopolyhedron virus. For sake of overcoming the demerits of single insect virus, such as slow pesticidal speed, low toxicity, etc, insect growth regulator chlorfluazuron is made to cooperate with insect virus so as to raise the pest controlling effect obviously. Compared with the traditional method of raising the pest controlling effect of virus with neurotoxic pesticide, the present invention has the advantages of high synergistic effect, high safety and raised biological control level of pests.
Owner:JIANGSU ACAD OF AGRI SCI
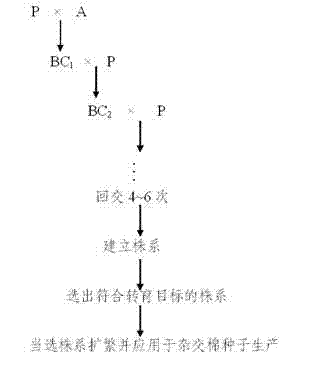
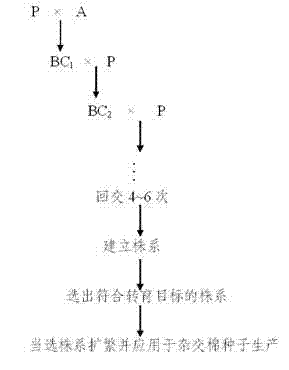
Method for producing hybrid cotton seeds by inducing male sterility of cotton with chemical male gametocide
The invention relates to a method for producing hybrid cotton seeds by inducing the male sterility of cotton with a chemical male gametocide, comprising the following steps of: (1) screening the chemical male gametocide, which has a thorough castration performance and a stable effect, and cotton varieties or strains which correspond to the chemical male gametocide; (2) backcrossing for 4-6 generations by using varieties with genes to be transformed and the screened cotton varieties, spraying each backcrossed generation by using the screened chemical male gametocide, and selecting plants, which are subjected to thorough castration and free from influences harmful to the growth and the development of cotton plants, from a colony to establish a plant line till the plant line in accordance with a transformed object is selected, wherein the selected plant line is used for producing the hybrid cotton seeds after being propagated. The method provided by the invention has a good castration effect and is beneficial to cost and labor saving; the chemical male gametocide has thorough and stable effect; and the production cost of the hybrid cotton seeds is reduced by about 30 percent compared with that of artificial castration and pollination.
Owner:JIANGSU KETENG COTTON
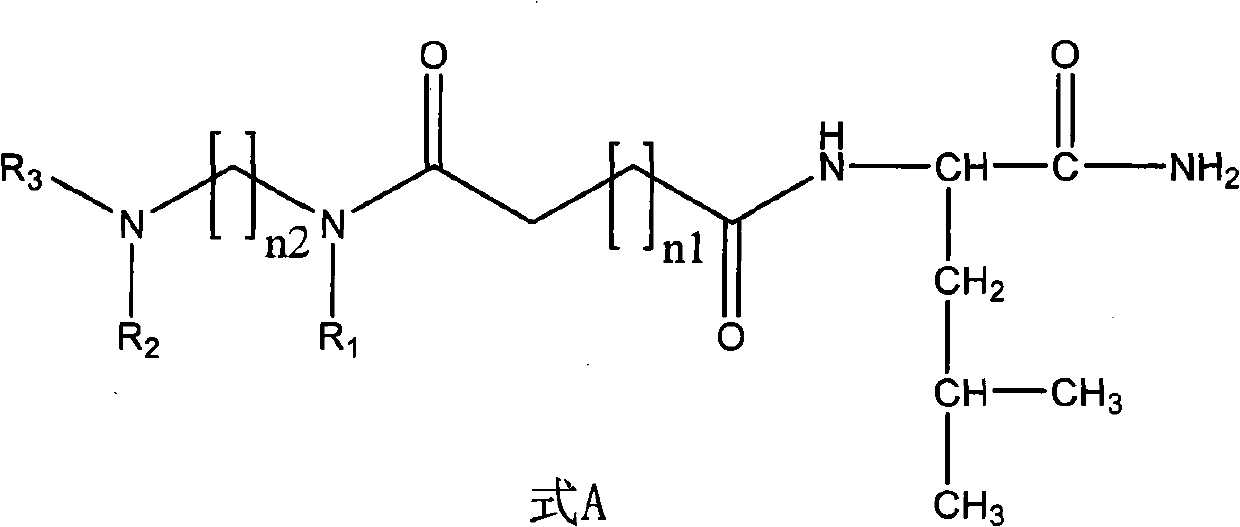
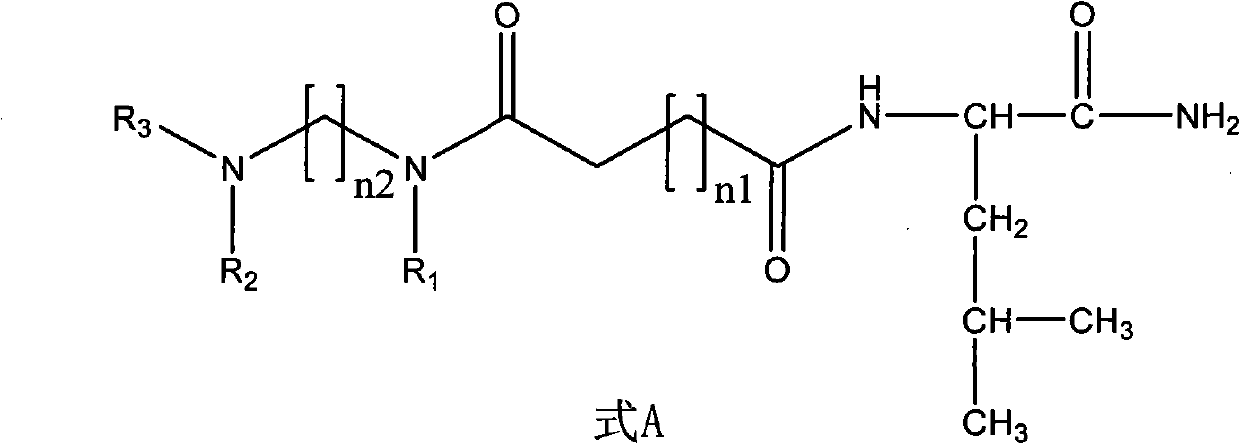
Non-peptide juvenile hormone synthetic inhibitor
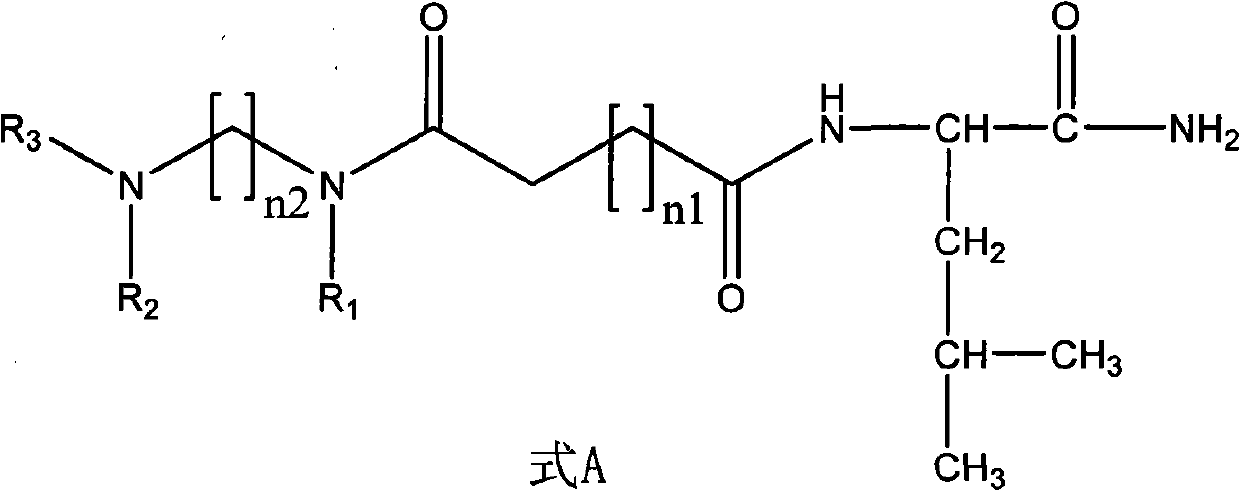
InactiveCN102093251AGood synthetic in vitro activityNovel structureBiocideChemosterilantsStructural formulaHigh activity
The invention discloses a non-peptide juvenile hormone synthetic inhibitor and belongs to the technical field of juvenile hormone synthetic inhibitors. A structural formula of an analogue is shown as a formula A. The non-peptide juvenile hormone synthetic inhibitor with a novel structure, simpleness, low cost and high activity of inhibiting juvenile hormone synthetic separation is obtained by substituting non-peptide micromolecule segments for amino acids in a core pentapeptide except for leucine. The compound of the formula A accords with the characteristics of an insect growth regulator andhas a good development application prospect.
Owner:CHINA AGRI UNIV
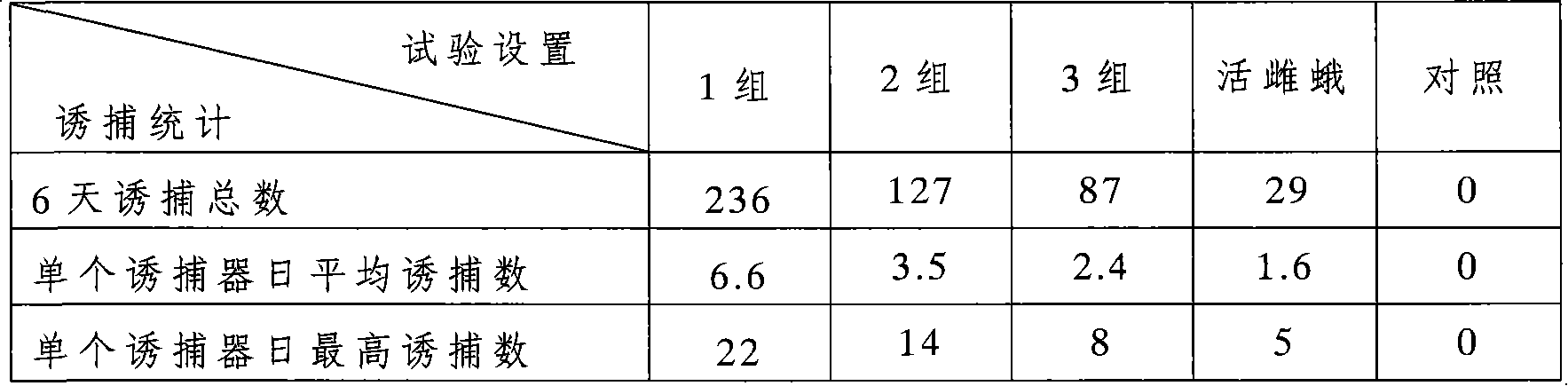
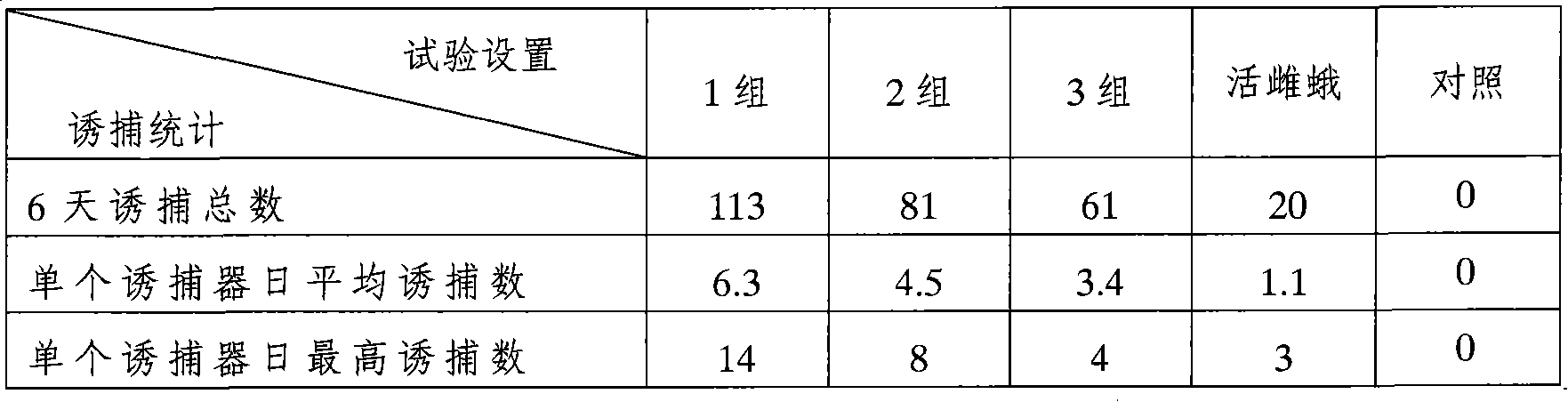
Compositions and methods for attracting noctuid moths
The invention provides compositions for attracting noctuid moths, which include at least one pheromone and at least one synergist. The synergist is a naturally-occurring phytochemical compound. The synergist is preferably β-caryophyllene, iso-caryophyllene, α-humulene, or combinations thereof. The invention also provides traps containing the composition for attracting noctuid moths, and methods for attracting, capturing, killing or sterilizing noctuid moths using the composition.
Owner:COTTON INC
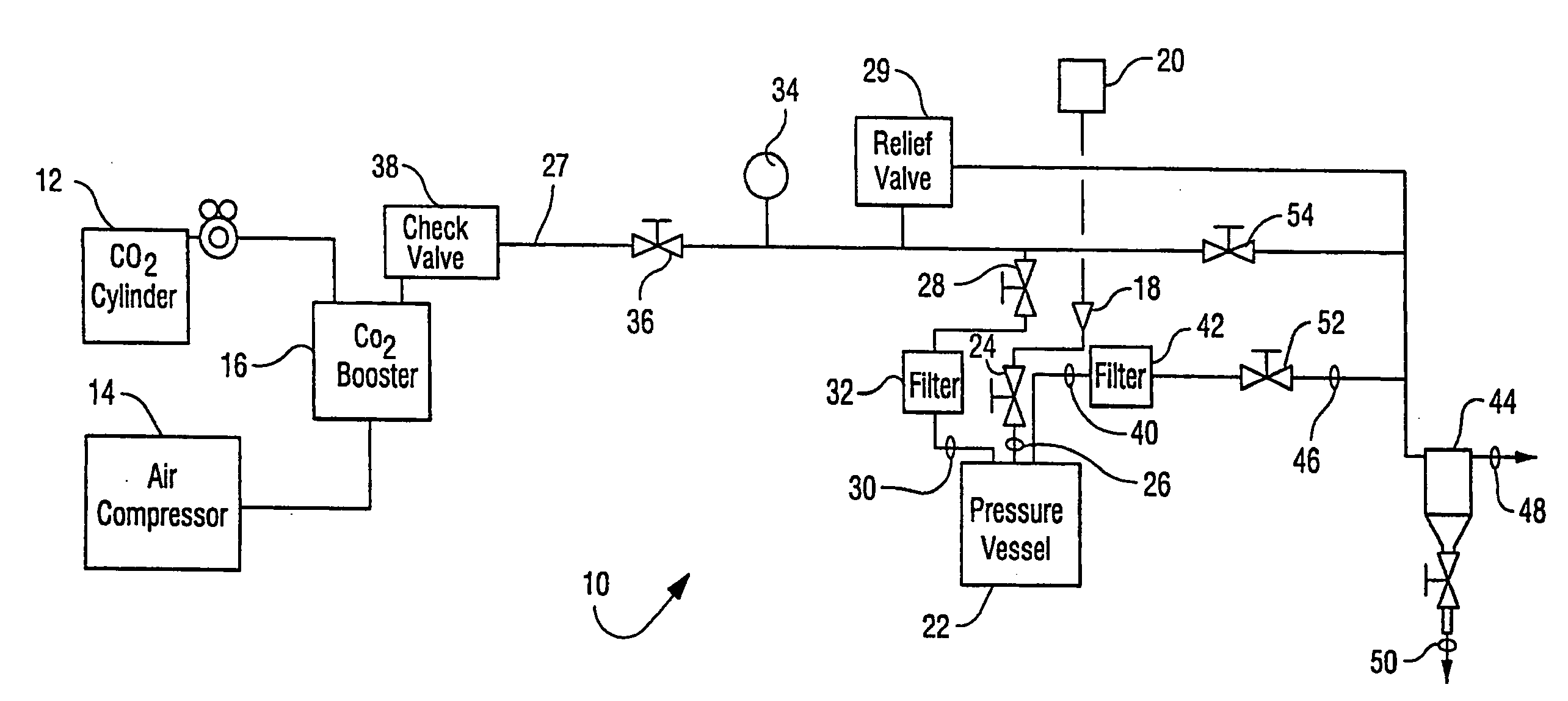
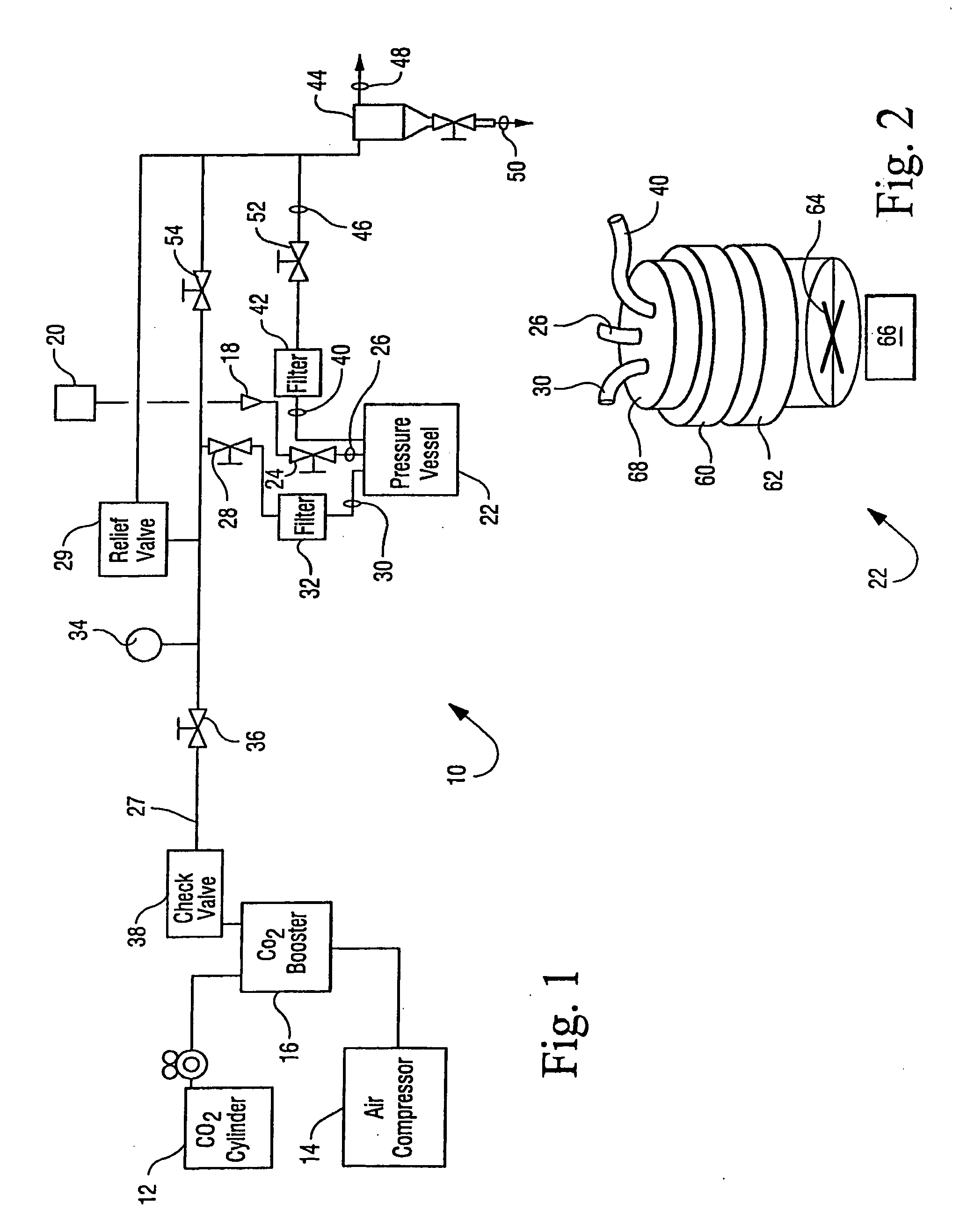
Sterilization methods and apparatus which employ additive-containing supercritical carbon dioxide sterilant
InactiveUS20090110596A1Enhances mass transfer and sterilizationImprove sterilizationBiocideChemosterilantsSporePressure cycling
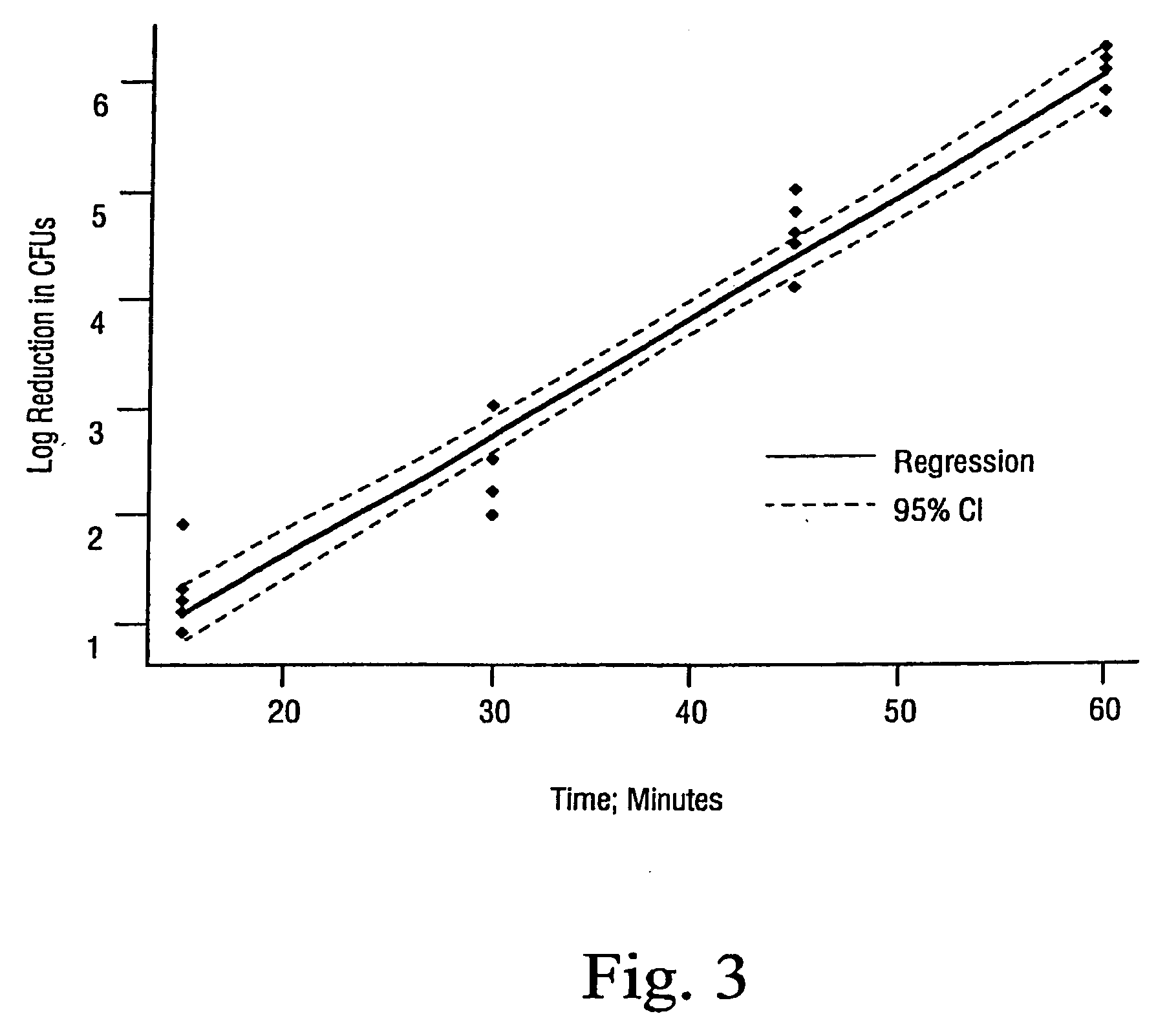
Sterilization methods and apparatus are effective to achieve a 6-log reduction in CFUs of industry standard bacteria and bacterial spores, i.e., B. stearothermophilus and B. subtilis spores, by subjecting sterilizable materials to a chemical additive-containing carbon dioxide sterilant fluid at or near its supercritical pressure and temperature conditions. Most preferably, the chemical additive-containing supercritical carbon dioxide sterilant fluid is agitated during sterilization, e.g., via mechanical agitation or via pressure cycling.
Owner:CHRISTENSEN TIMOTHY WAYNE +4
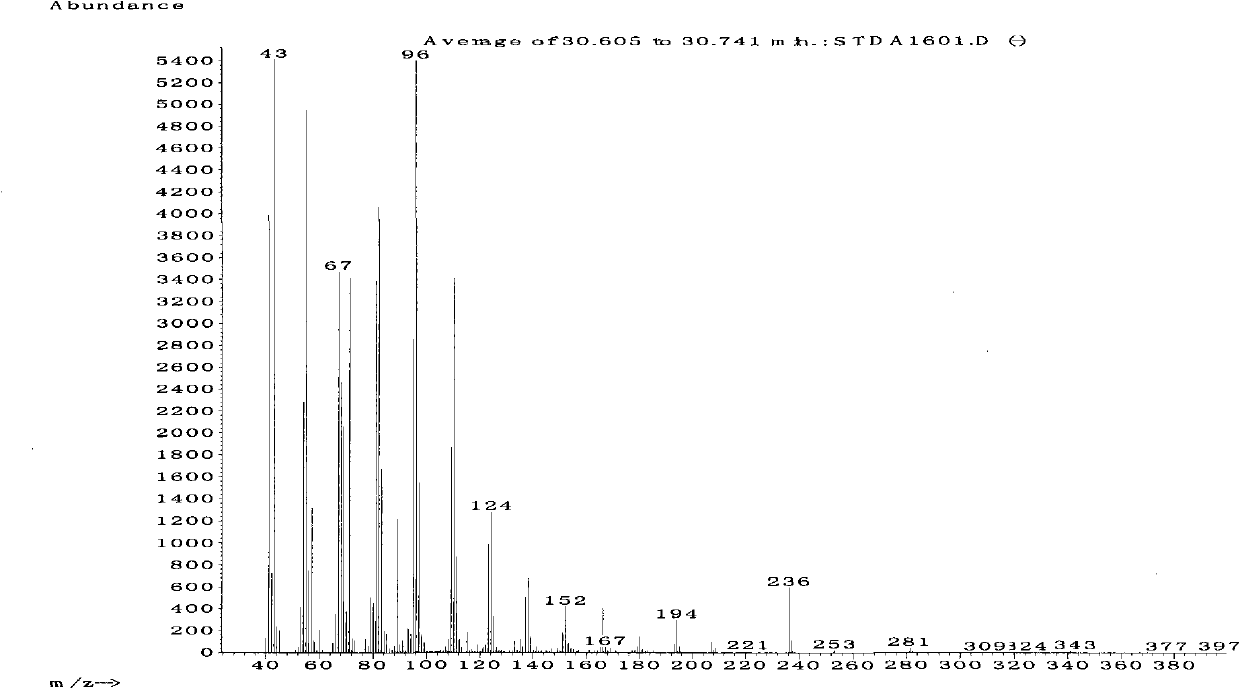
Application of (2S,8Z)-2-butyryl acyloxy-8-heptadecene
The invention discloses the application of a compound (2S,8Z)-2-butyryl acyloxy-8-heptadecene with the structural formula shown as the formula (I). The compound is used for preparing a chrysanthemum gall midge sex attractant. Shown by the experiment for the trapping effect for the chrysanthemum gall midge, the compound (2S,8Z)-2-butyryl acyloxy-8-heptadecene has strong attraction effect to the chrysanthemum gall midge. The product has proper price, favorable social and ecological benefits and suitable generalization and application.
Owner:CHINA AGRI UNIV
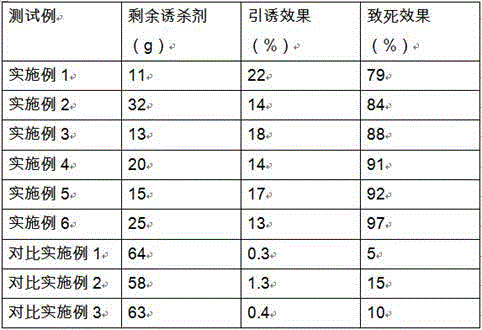
Poison bait capable of inhibiting rat reproduction and preparation method thereof
ActiveCN105994315AEffectively interfere with normal physiological processesInterfere with normal physiological processesBiocideChemosterilantsBiodegradable coatingZoology
The invention provides a poison bait capable of inhibiting rat reproduction. The poison bait comprises a component particle A and a component particle B. The component particle A is a compound bait adopting a multilayer structure, the compound bait adopting the multilayer structure comprises a degradable porous carrier which is loaded with a chemosterilant and peanut flour, a biodegradable coating layer which can internally coat the degradable porous carrier and the chemosterilant and the peanut flour loaded on the degradable porous carrier, and a food layer which comprises foods and coats the biodegradable coating layer internally in sequence from inside to outside, wherein the chemosterilant is desogestrel and ethinyloestradiol. The component particle B is a food particle. The poison bait disclosed by the invention is conducive to long-term and all-weather application, especially the sublimation, degradation or loss of effective components are avoided when the poison bait is applied and released in the field or in the wild, so that secondary pollution to the environment is not caused, and the palatability of the poison bait is good.
Owner:SHAANXI INST OF ZOOLOGY NORTHWEST INSTOF ENDANGERED ZOOLOGICAL SPECIES
Bait for controlling sterilization of female pest rats
The invention relates to a bait for controlling the sterilization of female pest rats, which is characterized by comprising the following components on the basis of total 100 mass percent: 0.005 to 0.05 percent of mifeprex, 0.1 to 1 percent of edible oil, 0.1 to 5 percent of edible sugar and / or milk powder and the balance of one of unhulled rice, rice, wheat, corn, yellow bean the pea nut. The bait for controlling sterilization of the female pest rats has the advantages of long-lasting control effect, no medicament resistance, high palatability, no environmental pollution, no safety hazard andthe like, is low in cost, simple in process, suitable to be promoted to control pest rat disaster in a large range and capable of reducing the population quantity of pest rats by over 80 percent within 6 months, reduces the pregnancy rate of female rates and even completely eradicates pregnant females.
Owner:GUANGDONG ENTOMOLOGICAL INST
Biological preventing and controlling agent for pests of storage grain
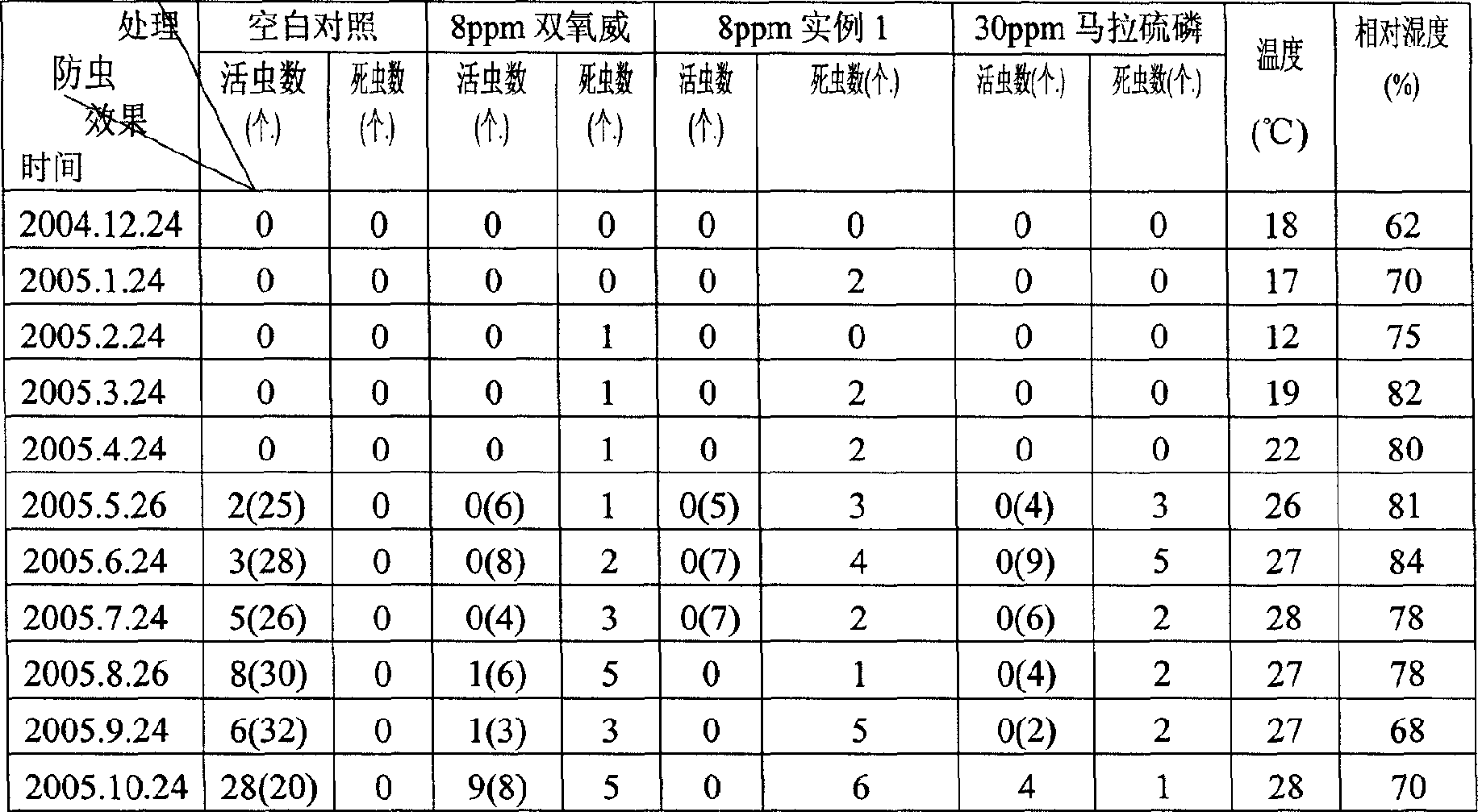
The present invention relates to a preparation process of pesticide composition for stored grain, and features that the pesticide composition consists of diflubenzuron 2-10%, fenoxycarb 10-35%, solvent 50-72%, co-solvent 8-15% and emulsifier 5-18%. It is used for stored grain and has low toxicity and broad pesticidal spectrum. The use amount of 8 ppm can result in effective period of 1 year.
Owner:GUANGDONG GRAIN SCI INST
Anti-breeding rodenticide
InactiveCN103125530AReduce in quantityImprove palatabilityBiocideChemosterilantsInterstitial cellBiology
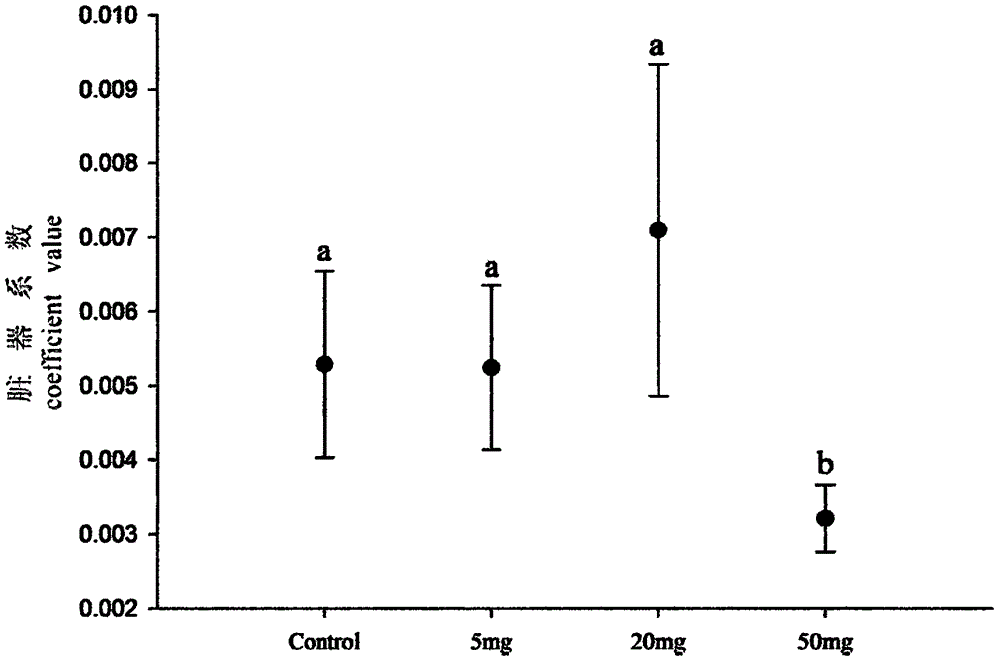
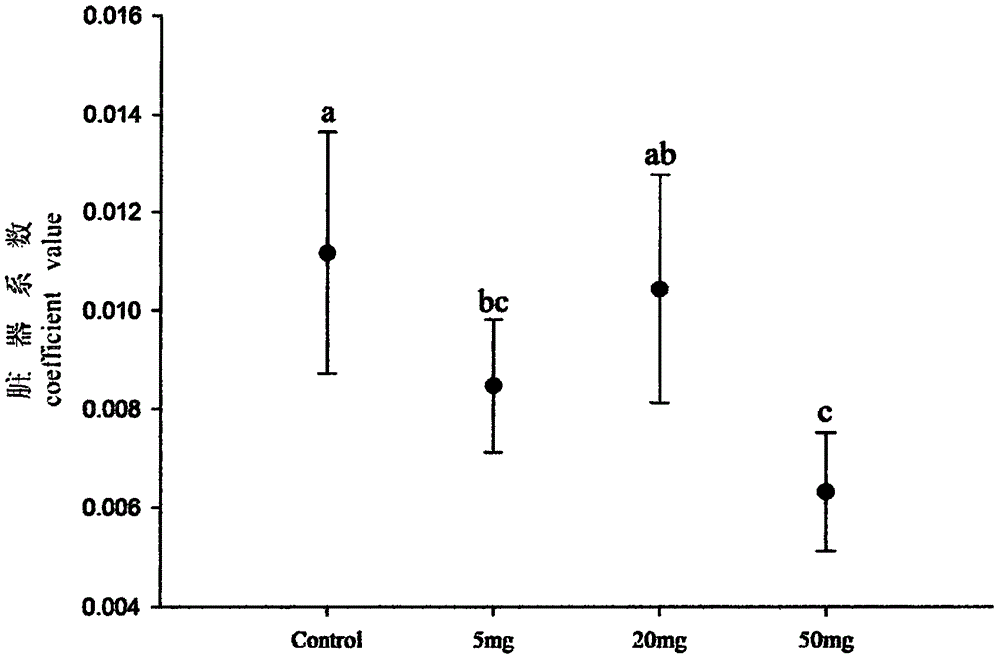
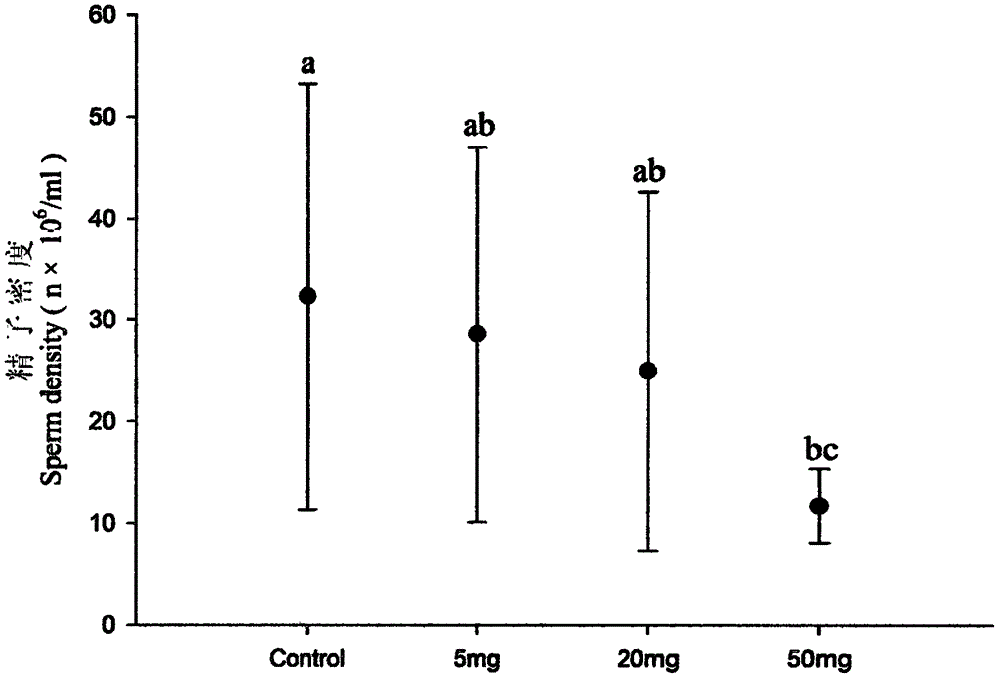
The invention discloses an anti-breeding rodenticide. Melia azedarach oil is served as an anti-breeding agent, and the weight percent of the melia azedarach oil in the anti-breeding rodenticide accouts for 0.08%-0.1%. Through researching the intrinsic rules and hormone regulation mechanism of mouse breeding as well as studying the toxicological and function mechanism of a vegetable anti-breeding agent, ideal anti-breeding agent-melia azedarach oil is screened. The anti-breeding rodenticide is low in effective dosage, makes mice infertile, and has the advantages of low toxicity index, high safety, no harm to human being and animals and no secondary poisoning. After the anti-breeding rodenticide is eaten 10 days later, main reproductive organs-testicles of male mice generate the following variation: the organ coefficient gets small, the sperm amount is reduced, the seminiferous tubule diameter is thinned, lumens are reduced, germinal epithelium thickness is thinned, and interstitial cells are decreased; and ovarial organ coefficient of female mice is obviously reduced, and the corpus luteum in an ovarial structure is contracted. Dead fetus are increased after male and female mice coition, and the breeding number is greatly decreased.
Owner:XIAN UNVERSITY OF ARTS & SCI
Biological control of insects
Provided herein are modified insects having decreased expression of a testis-specific coding region compared to a control insect. In one embodiment, the modified insect includes the characteristic of reduced fertility, reduced fecundity, or a combination thereof, when compared to the control insect. Optionally and preferably, the competiveness of the modified insect is not significantly reduced compared to the control insect. Also included herein are methods for making a modified insect, and methods for using the modified insects, including making populations of modified insects and use of the modified insects in sterile insect technique for biological control.
Owner:UNIVERSITY OF MANITOBA
Botanical mice composite sterilant preparation method
The invention discloses a preparation method of a botanical mice composite sterilant Nongda-1, which belongs to the technical field of medicine compound. By researching reasonable ratio and synergism of the medicinal materials, the medicines prepared by the pharmaceutical composition of the invention have good effect for controlling mice fertility, alkannin extracted from roots of Macrotomia euchroma is combined with quinsetrol, an unique technology is employed, so that the botanical mice composite sterilant can be prepared, and the botanical mice composite sterilant has good effect for controlling mice fertility.
Owner:INNER MONGOLIA AGRICULTURAL UNIVERSITY
Prepn process of theasaponin sterilant for preventing and control rat plague
The present invention is preparation process of theasaponis sterilant for preventing and controlling rat plague. The theasaponis sterilant is prepared with tea seed cake as main material, and through extracting with water solution of hydrophilic solvent at 50-80 deg.c for 1-3 times, merging the extracted liquid and filtering; membrane separating the filtrate to eliminate impurity and concentrating to 10-15 Baume degree (60 deg.c); adding 4 wt% concentration water solution of sodium alginate in the same weight as the concentrate; dropping the mixture into 4 wt% concentration water solution of calcium chloride in the same weight; stilling at 4-10 deg.c for 8-12 hr to cure and shape; sieving grains of diameter 4-6 mm and drying to obtain the theasaponis sterilant. The preparation process is novel, simple, efficient and low in production cost, and the theasaponis sterilant has high efficiency in preventing and controlling rat plague, low toxicity and other advantages.
Owner:FUZHOU UNIV
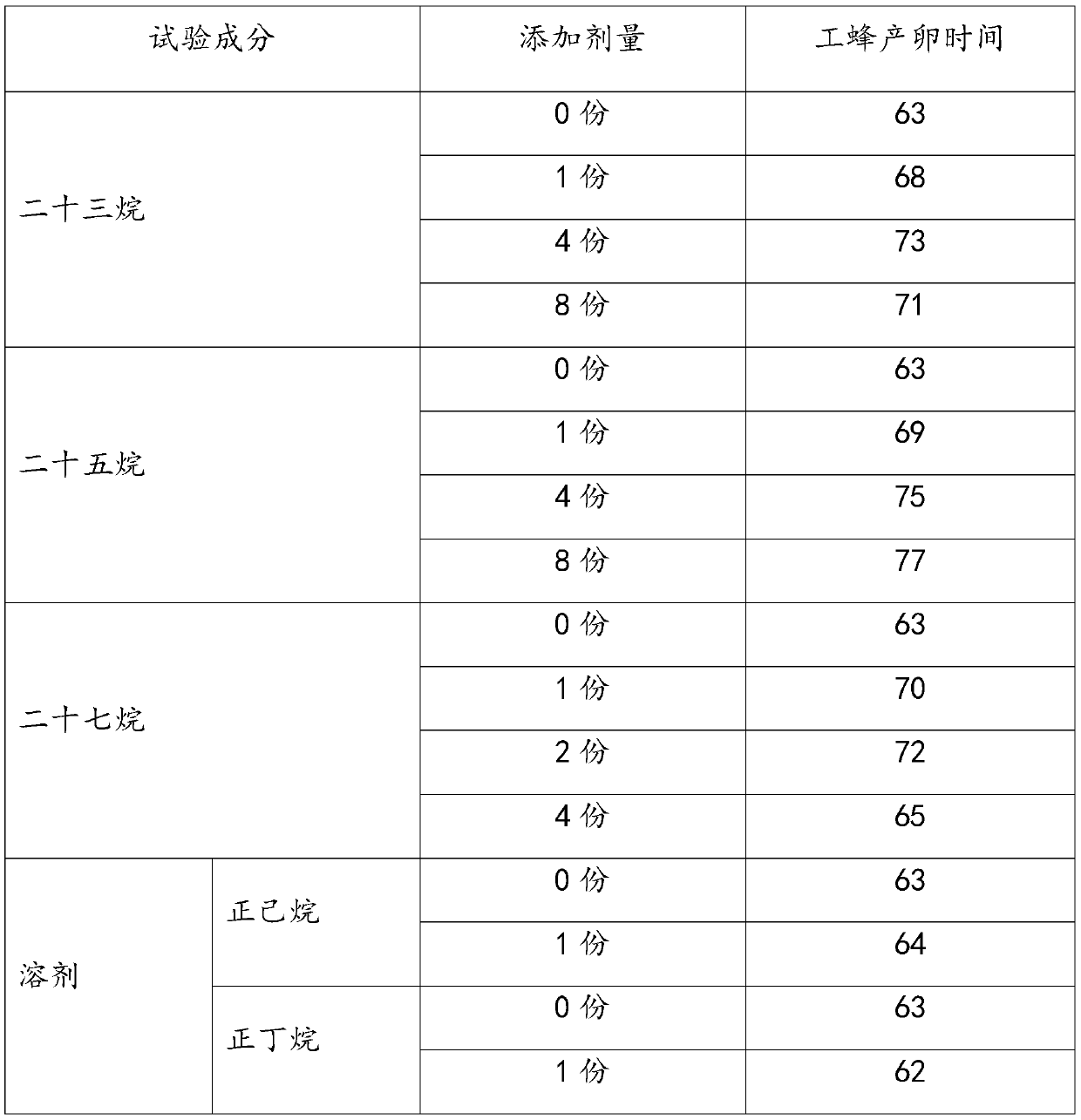
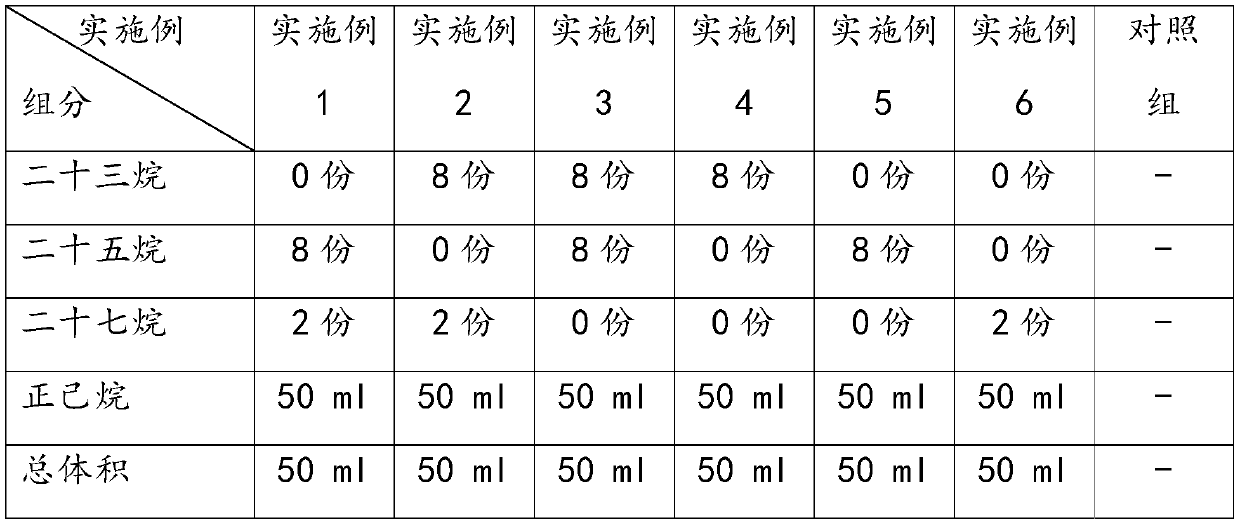
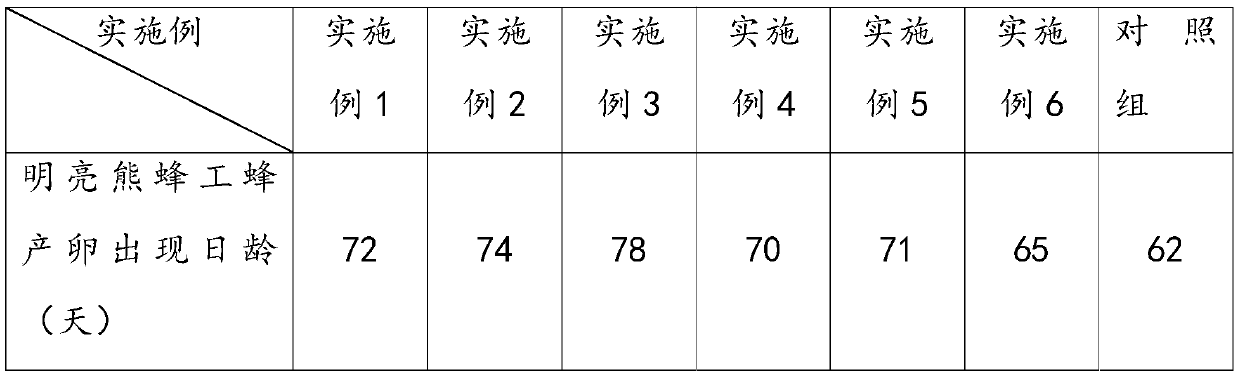
Inhibitor for inhibiting worker bees in bumblebee colony from spawning as well as preparation method and application of inhibitor
The invention provides an inhibitor for inhibiting worker bees in a bumblebee colony from spawning, and a preparation method and application thereof. The inhibitor comprises at least one of tricosane,pentacosane and heptacosane. Components of the inhibitor belong to main components of bumblebee queen pheromones, the inhibitor can effectively inhibit worker bee spawning behaviors in bumblebee colonies, has remarkable effects on prolonging the bumblebee breeding period and improving the bumblebee colony utilization rate, and is low in cost, high in cost performance and easy for practical application in bumblebee industrialized breeding.
Owner:吉林省养蜂科学研究所
Plant source antifertility agent for preventing and controlling grassland rats as well as preparation method and application thereof
InactiveCN103238635AHarm reductionIncrease productionBiocideChemosterilantsAdditive ingredientPlant Sources
The invention discloses a plant source antifertility agent for preventing and controlling grassland rats as well as a preparation method and an application thereof. The plant source antifertility agent consists of the following ingredients in parts by weight: 1-4 parts of cortex albiziae total saponins, 93-97 parts of grass meal particles and 1-3 parts of auxiliary materials. The preparation method is simple and reliable; the prepared granules have good palatability, are easy in quality standard setting, have an obvious antifertility function on male and female rats and are a novel plant source bisexual antifertility agent for preventing and controlling grassland rats.
Owner:SICHUAN AGRI UNIV
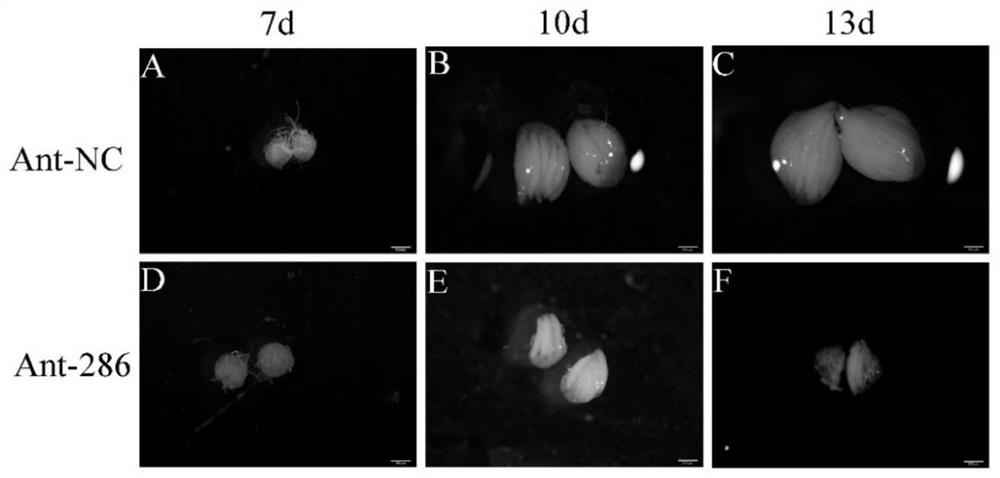
miR-286 inhibitor related to reproductive development of bactrocera dorsalis and application of miR-286 inhibitor
InactiveCN111705059AConducive to green prevention and controlDecreases egg production per femaleBiocideChemosterilantsPhysiologyBactrocera dorsalis
The invention discloses a miR-286 inhibitor related to reproductive development of bactrocera dorsalis and application of the miR-286 inhibitor. miR-286 is found to be related to development of ovaries and egg yield of female bactrocera dorsalis for the first time, the expression of miR-286 can be reduced by using the specific miR-286 inhibitor, compared with a control group, the ovary form of female adults of the bactrocera dorsalis with the miR-286 inhibitor shows obvious development delay, the ovary diameter is also inhibited, and the single female egg yield and the total egg yield are significantly reduced. Therefore, the expression of the miR-286 in the bactrocera dorsalis can be inhibited to affect normal reproductive development of the bactrocera dorsalis and reduce fertility, thereby facilitating green prevention and control of the bactrocera dorsalis.
Owner:HUAZHONG AGRI UNIV
Deratting bait for influencing sterility through male endocrine
ActiveCN103314981AImprove palatabilityImprove controlBiocideChemosterilantsEconomic thresholdActive component
The invention discloses a deratting bait for influencing sterility through the male endocrine. The deratting bait comprises 0.0001-0.01mass% of cabergoline as a single active component and acceptable auxiliary materials. A proper amount of cabergoline is mixed in an appropriate bait, and the obtained deratting bait has the advantages of good palatability to rats, lasting control effect, safety, low pollution, simple production technology, and low cost, and is suitable for the large scale implementation for controlling the number of rat populations to put the harms of the rats under an economic threshold for a long term.
Owner:INST OF ZOOLOGY GUANGDONG ACAD OF SCI
Murine sterilant bait for controlling rodent damage in farmland and grassland
The invention discloses a murine sterilant bait and a preparation method thereof. The murine sterilant bait comprises a basic bait, a warning color and the original drug of a sterilant, wherein, the basic bait comprises 10 to 30% of corn grit, 10 to 30% of naked oat powder, 60% of dry grass meal and 0.5 to 0.7% of peanut oil. According to the invention, dry grass meal and naked oat flour are targetedly added into the murine sterilant bait; a formula for the murine sterilant bait is reasonable, materials to prepare the murine sterilant bait are easily available, and it is convenient to prepare and utilize the murine sterilant bait; an fertility control effect on bandicoots in farmland and grassland is improved; palatability of the bait is good, the intake rate of bandicoots is increased, while intake of the bait by other animals is effectively avoided, and therefore, the bait has the advantage of environmental security.
Owner:CHINA AGRI UNIV
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com