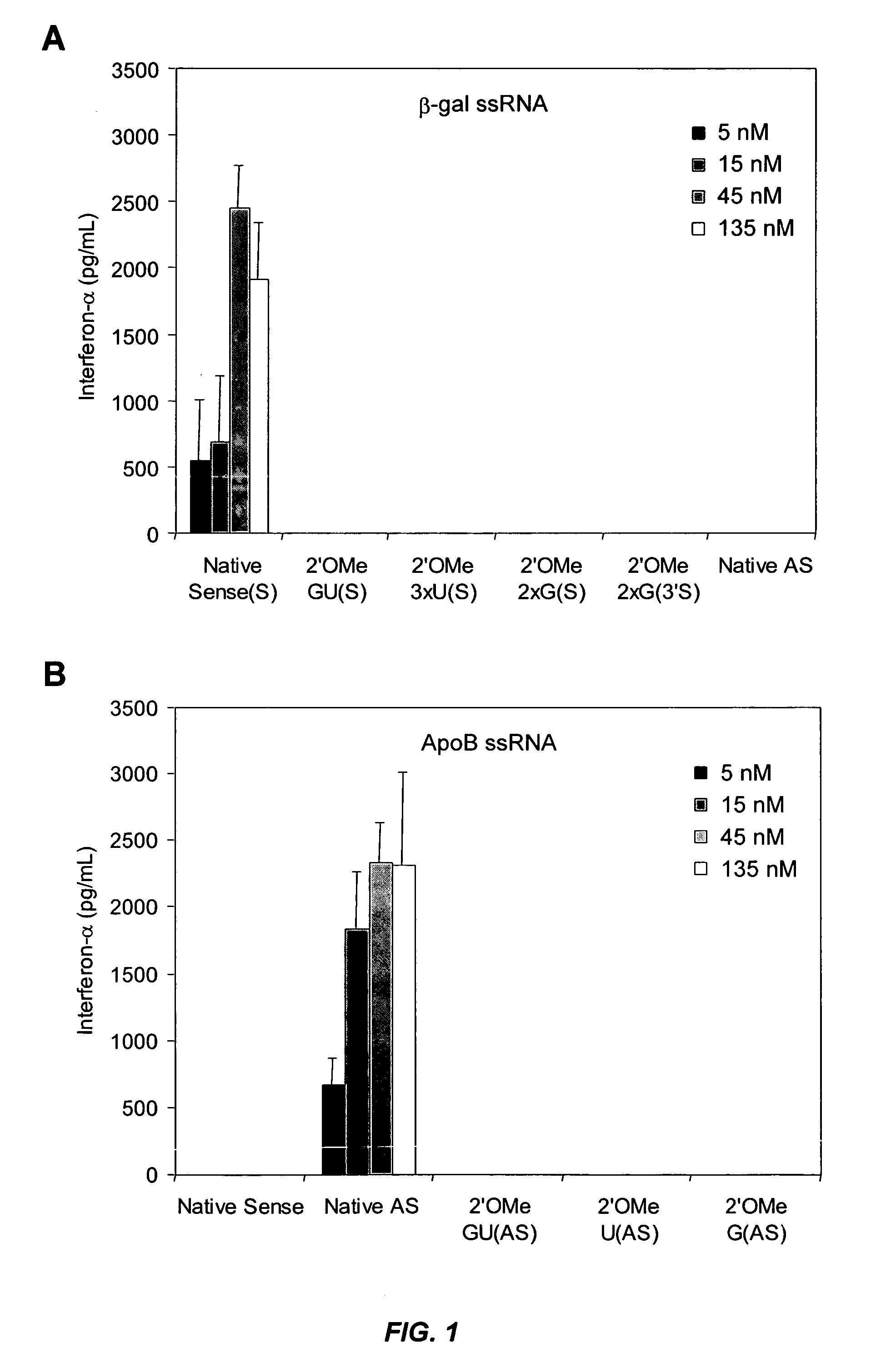
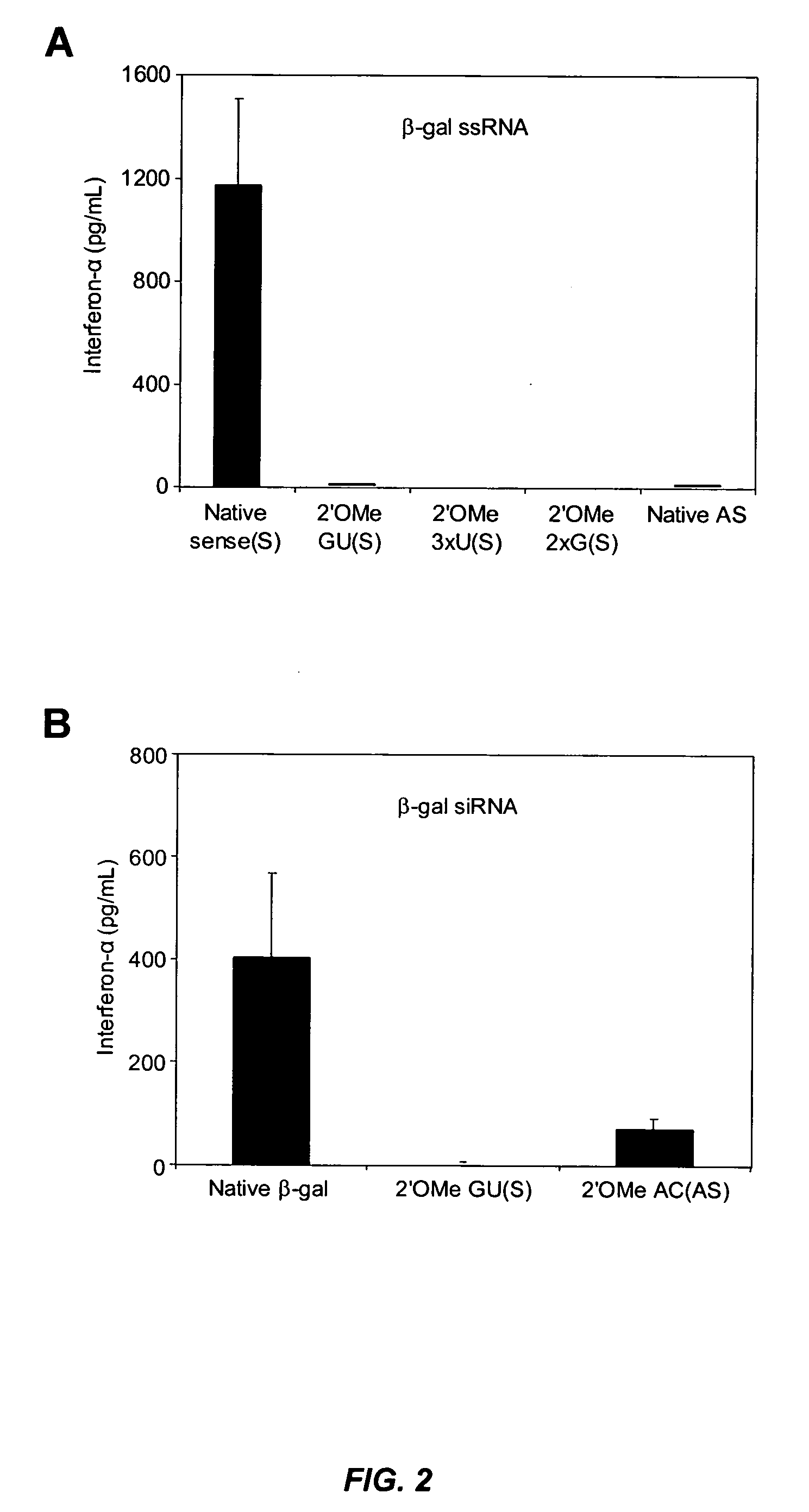
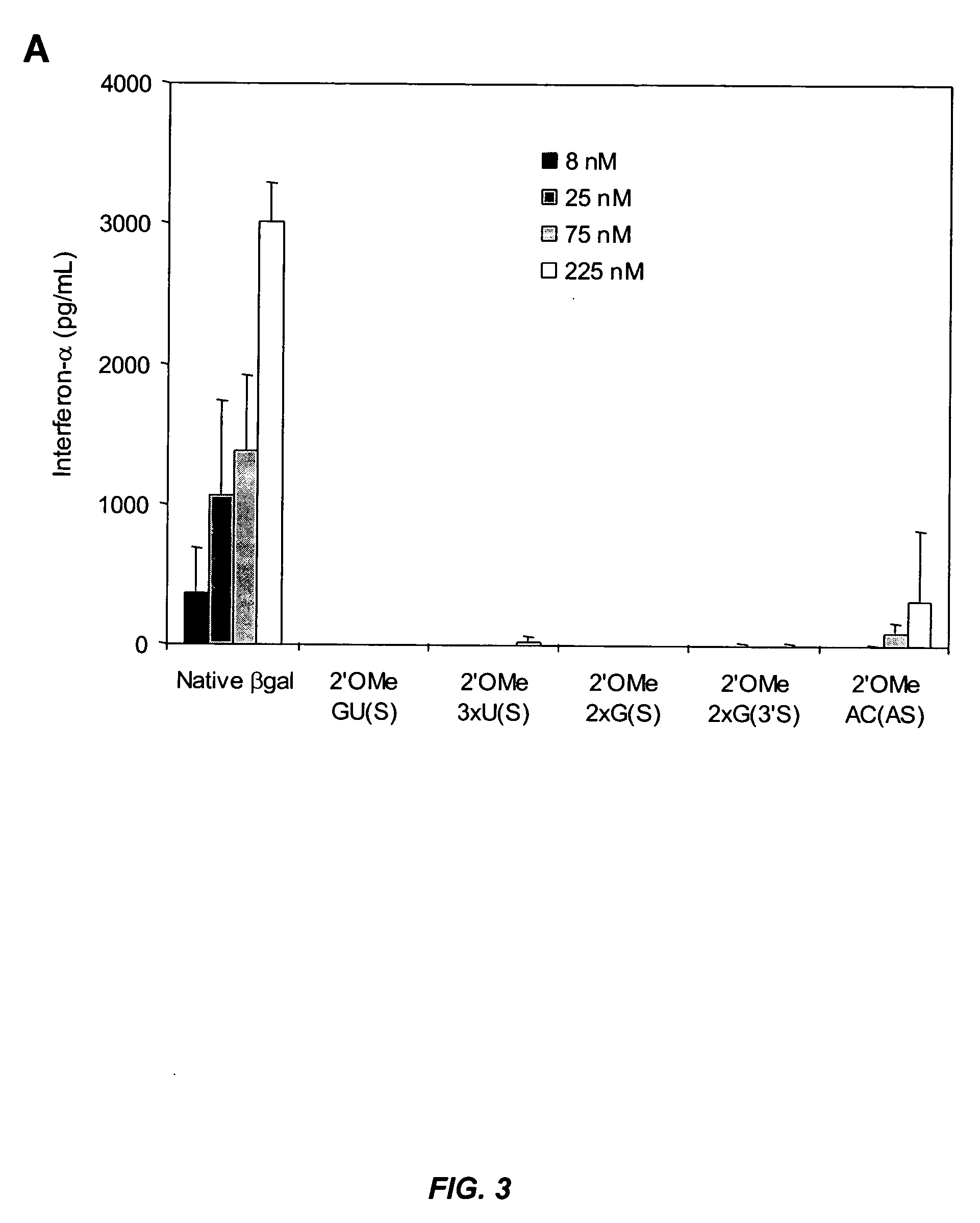
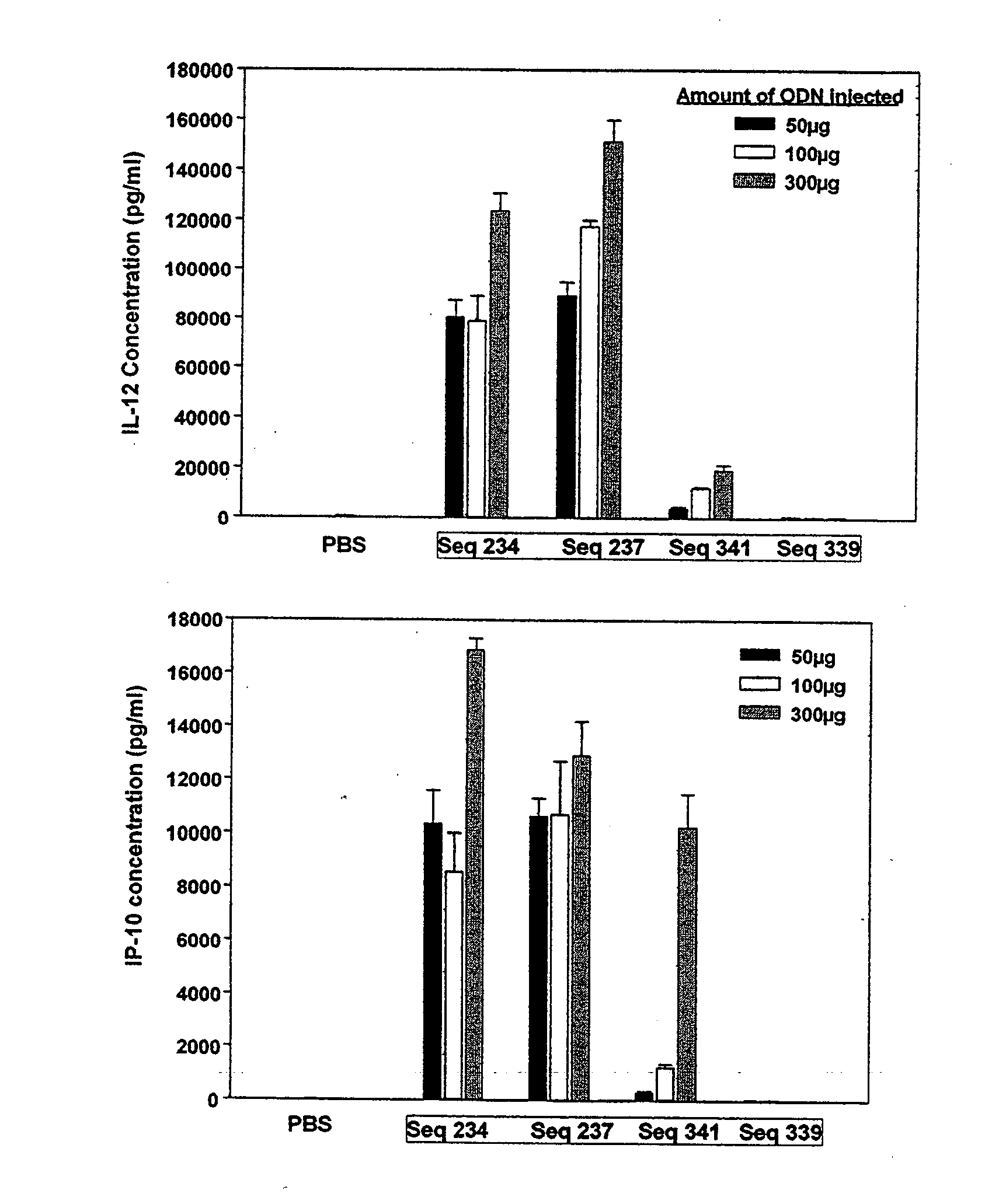
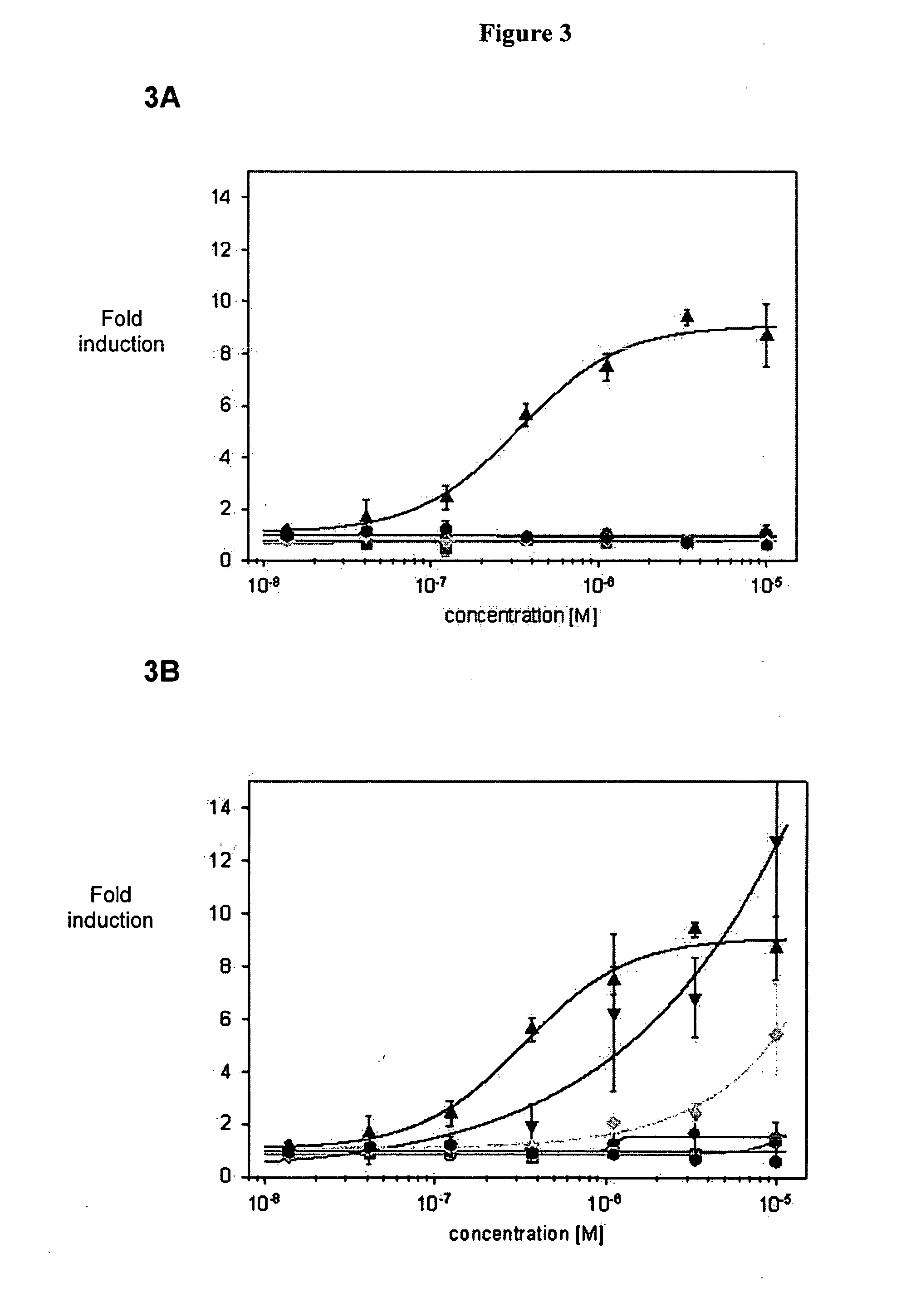
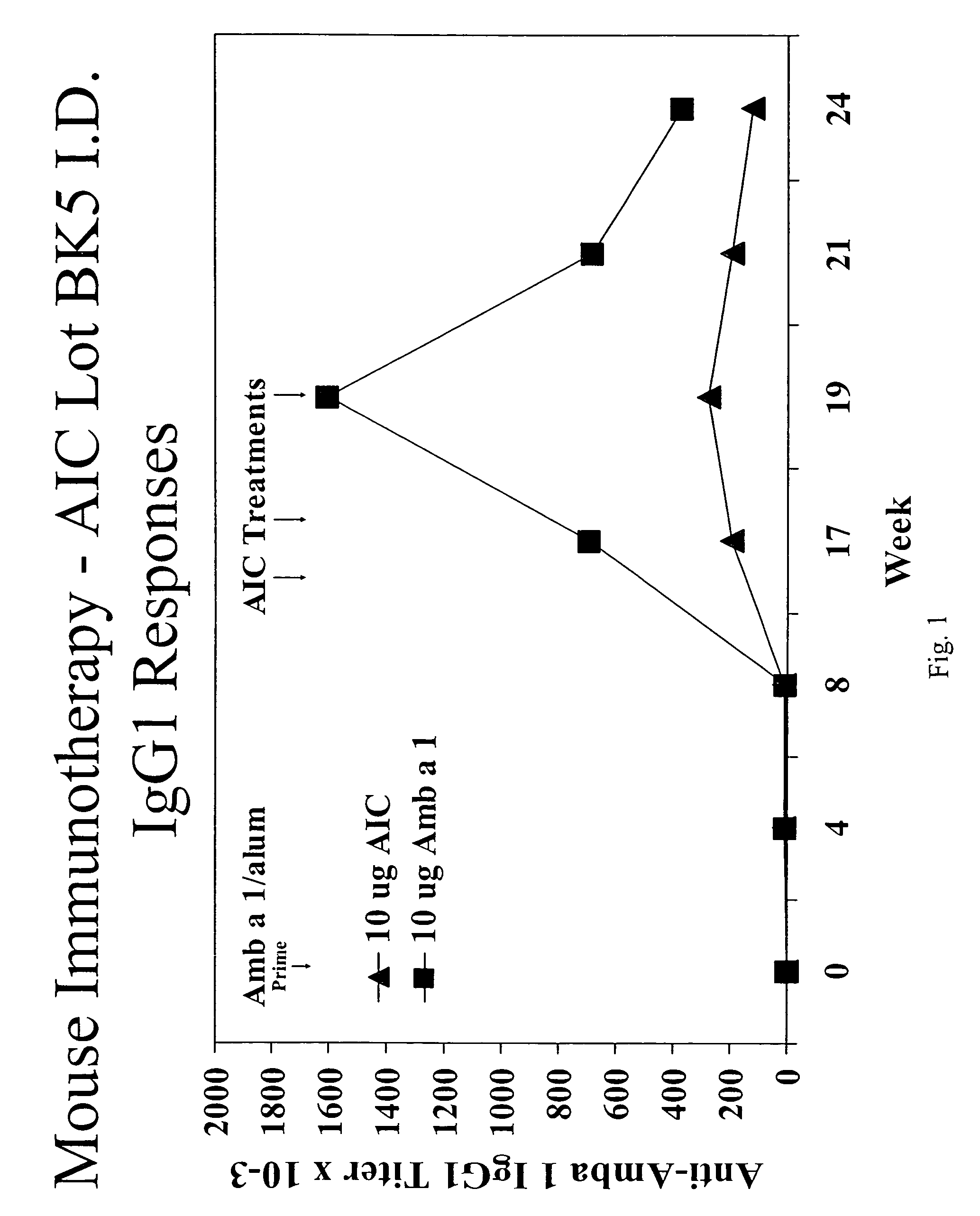
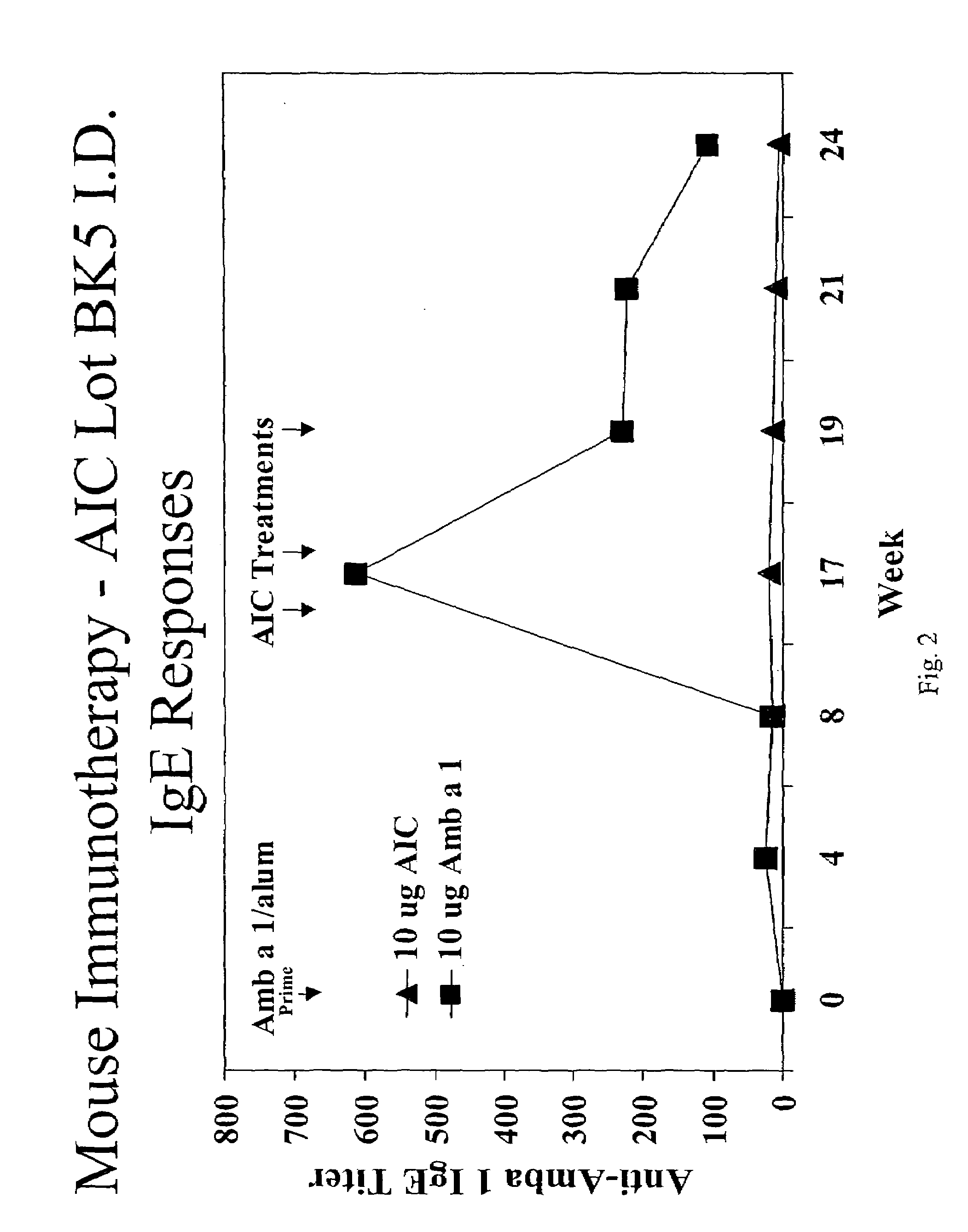
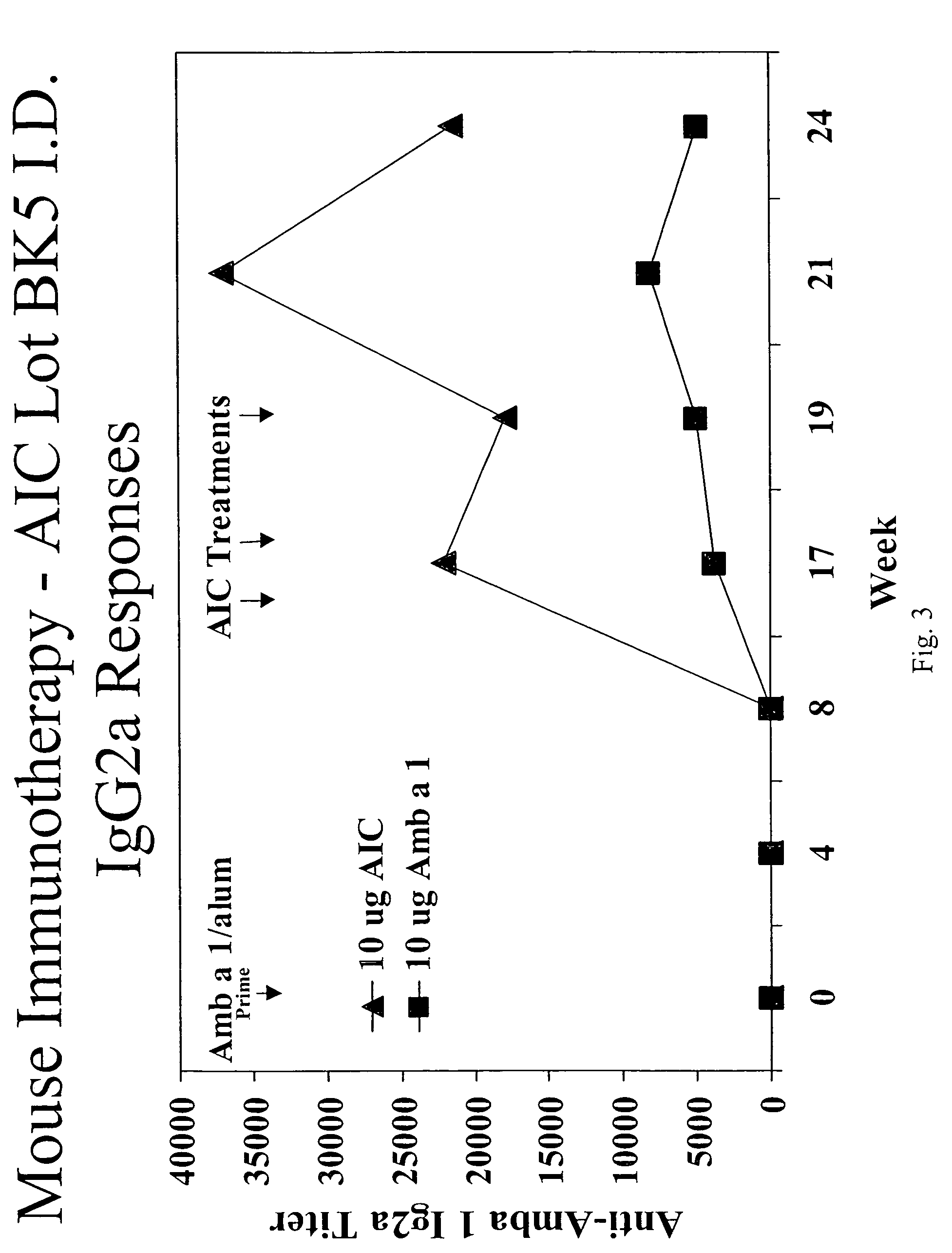
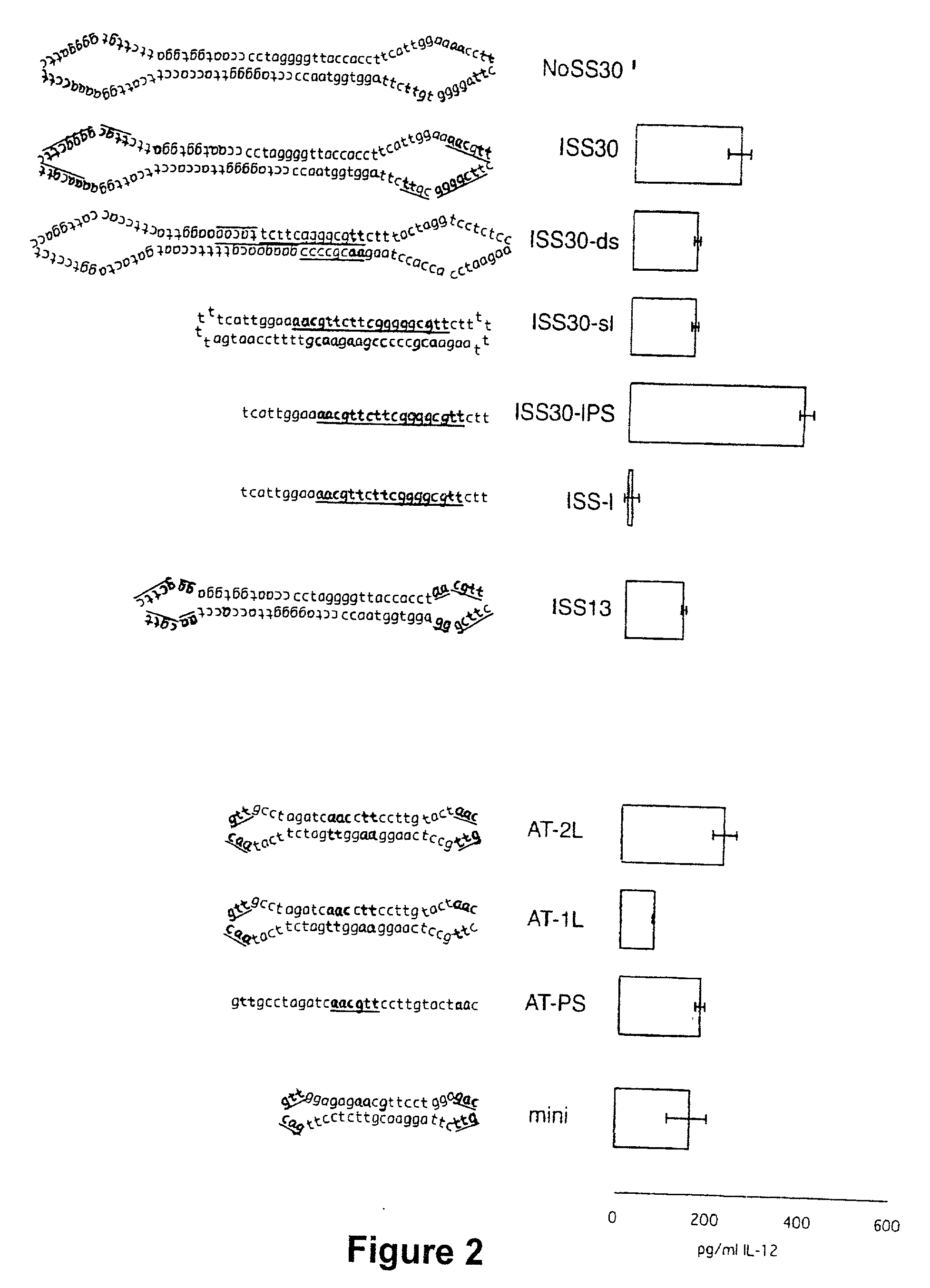
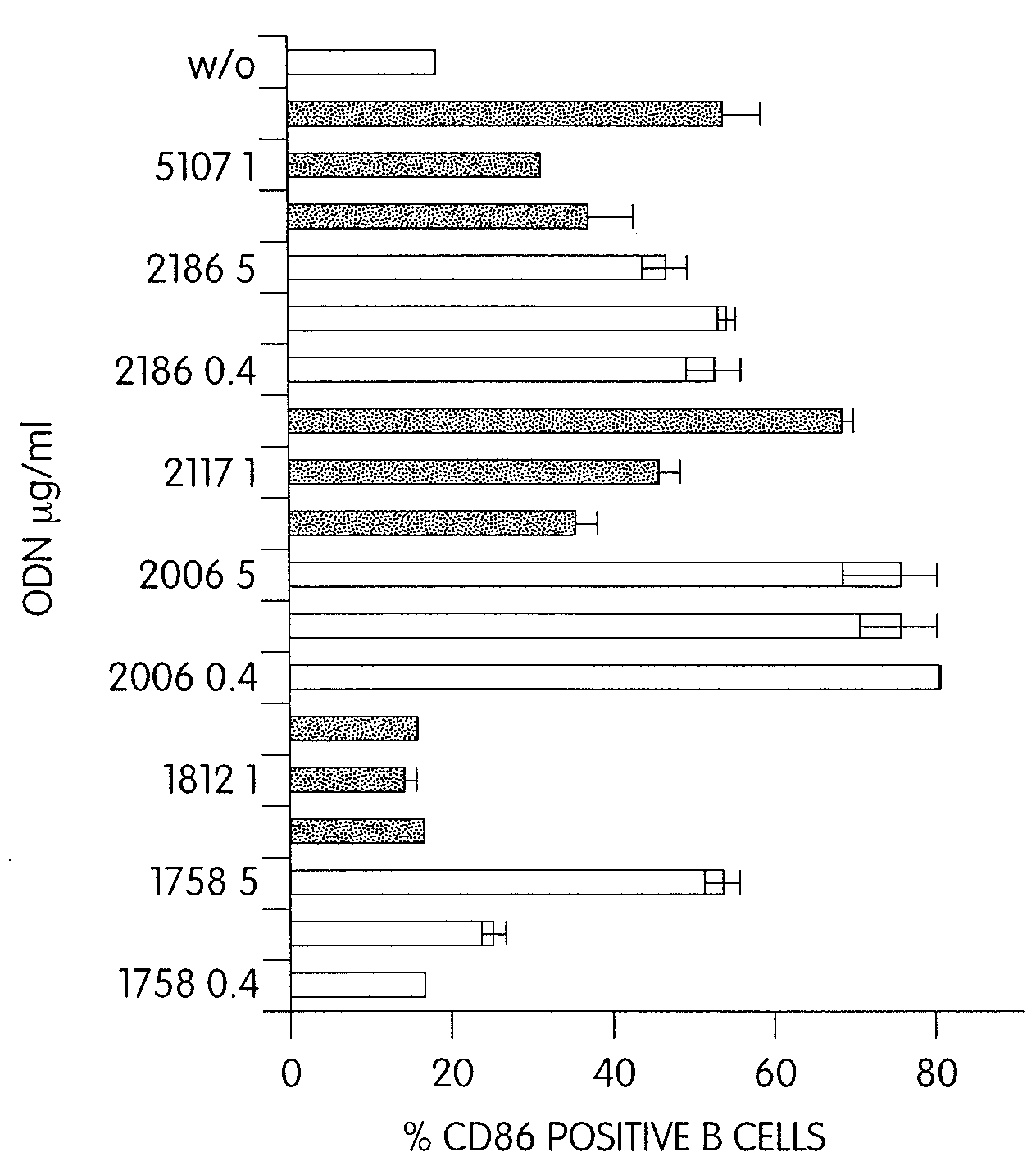
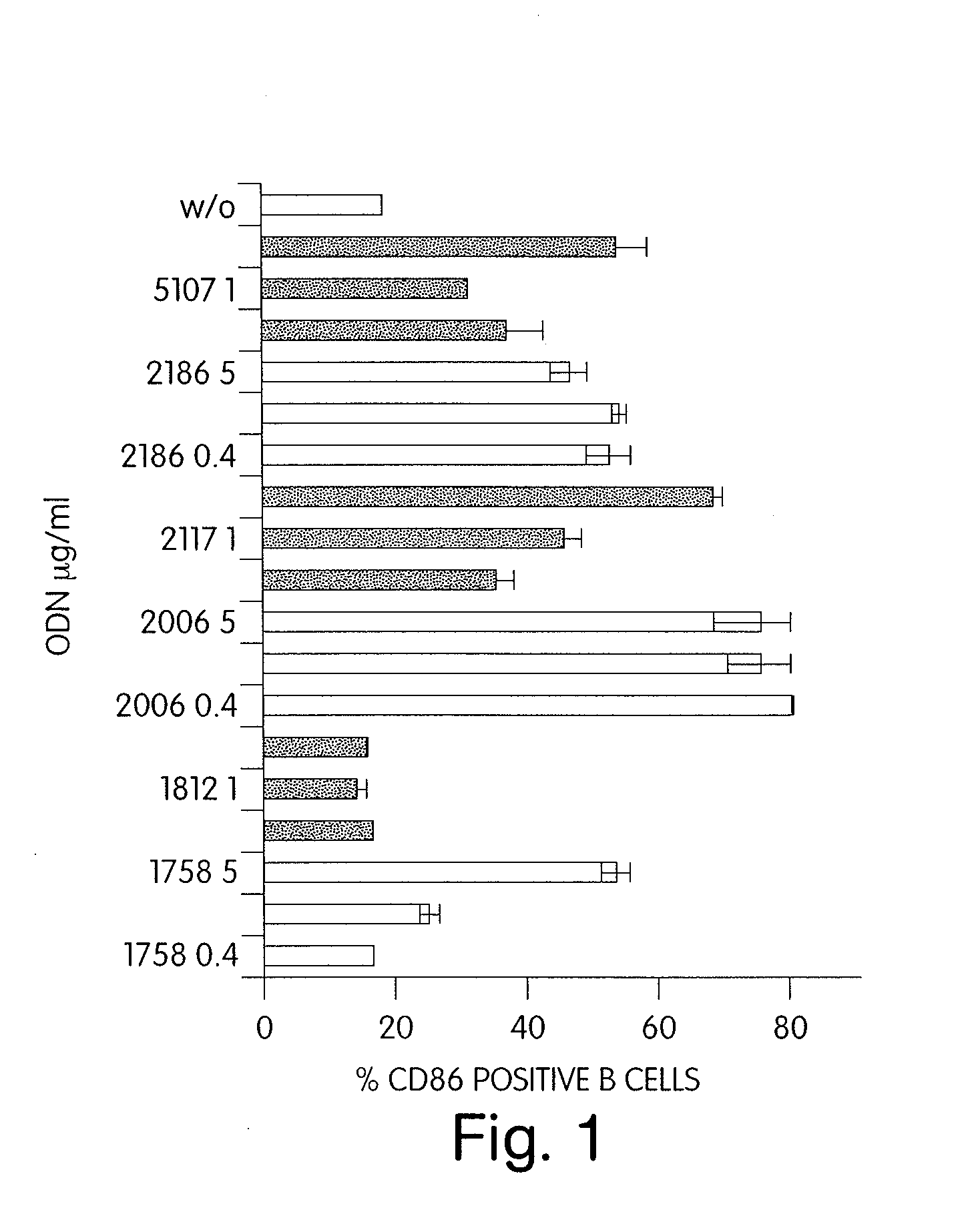
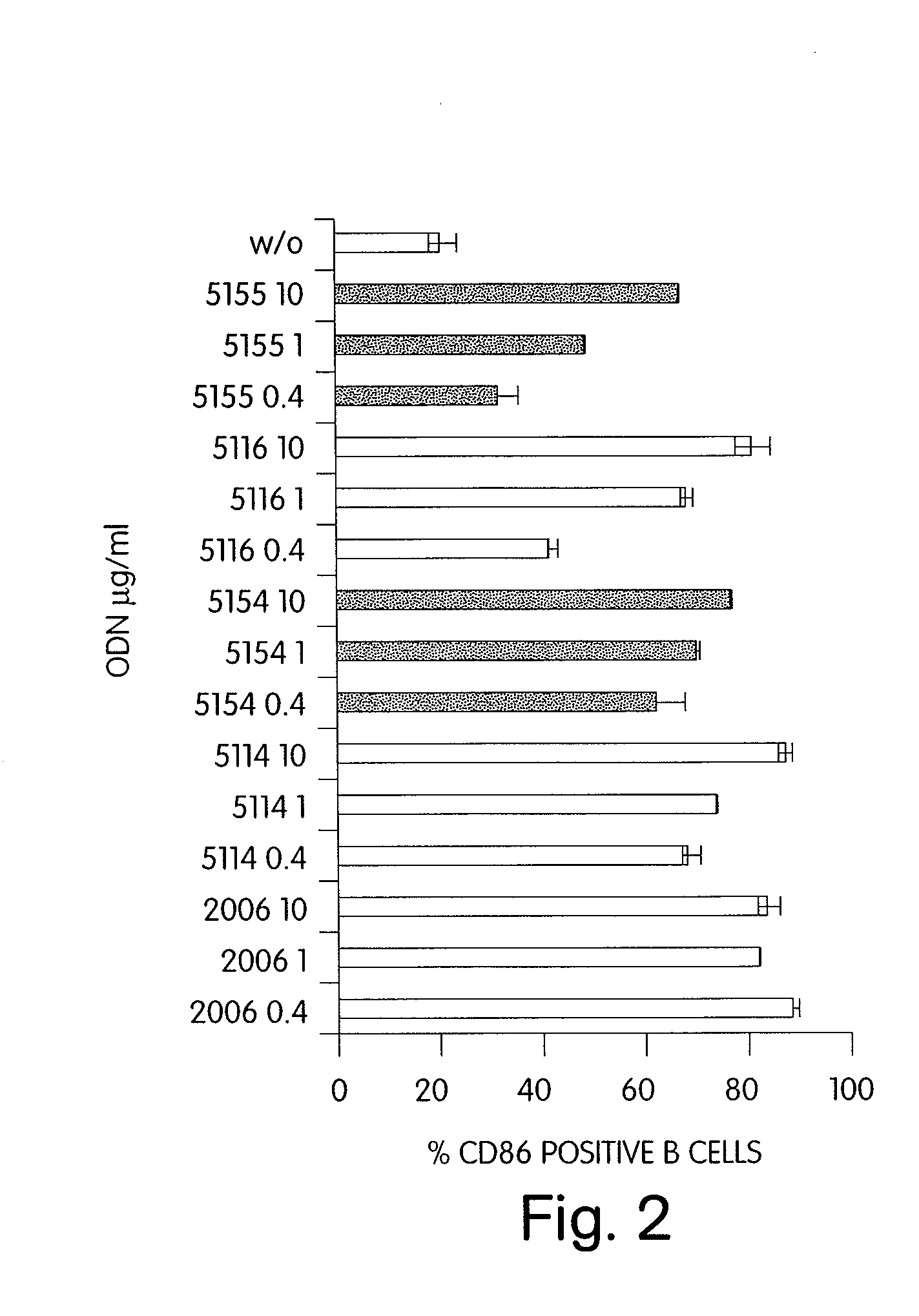
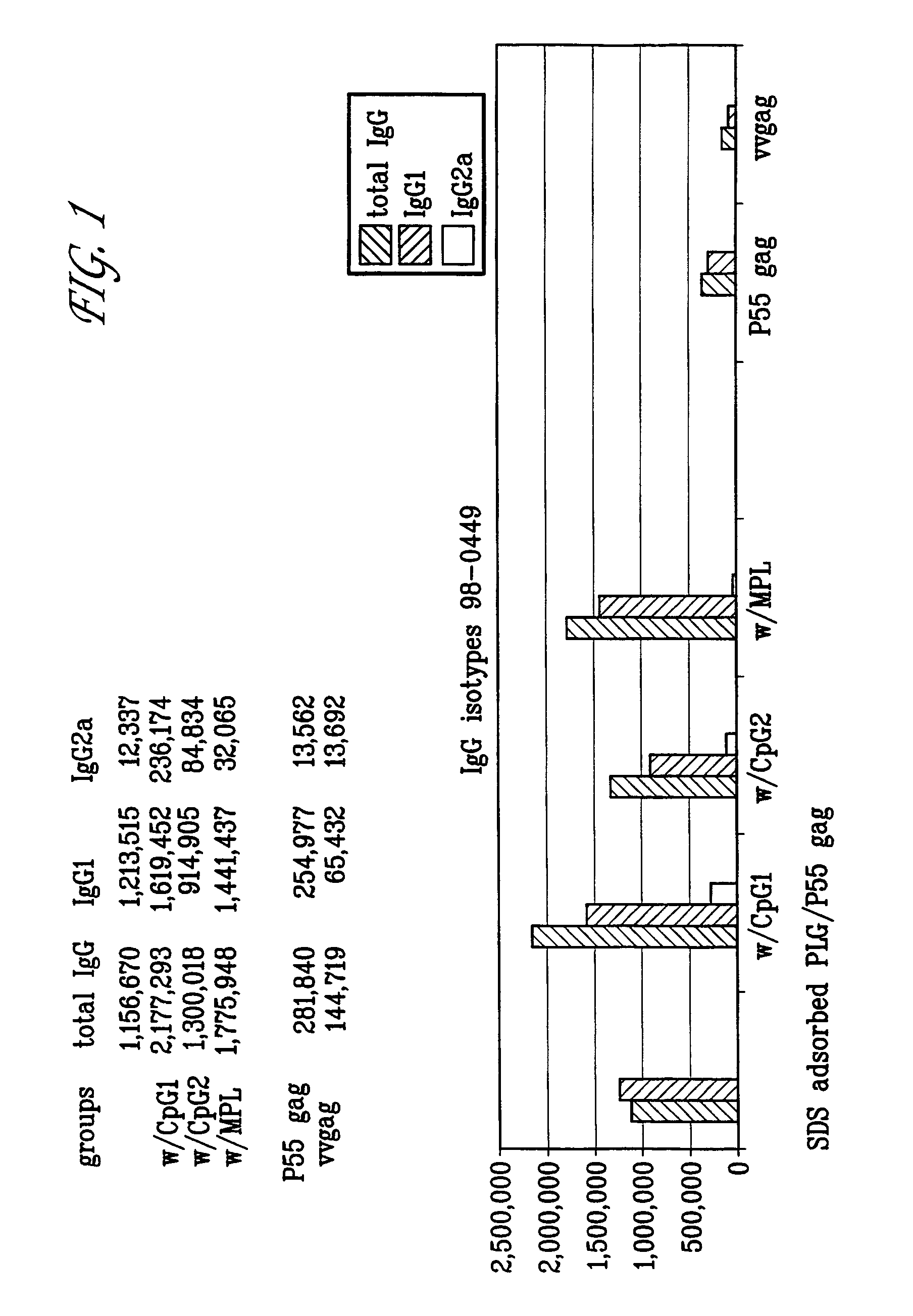
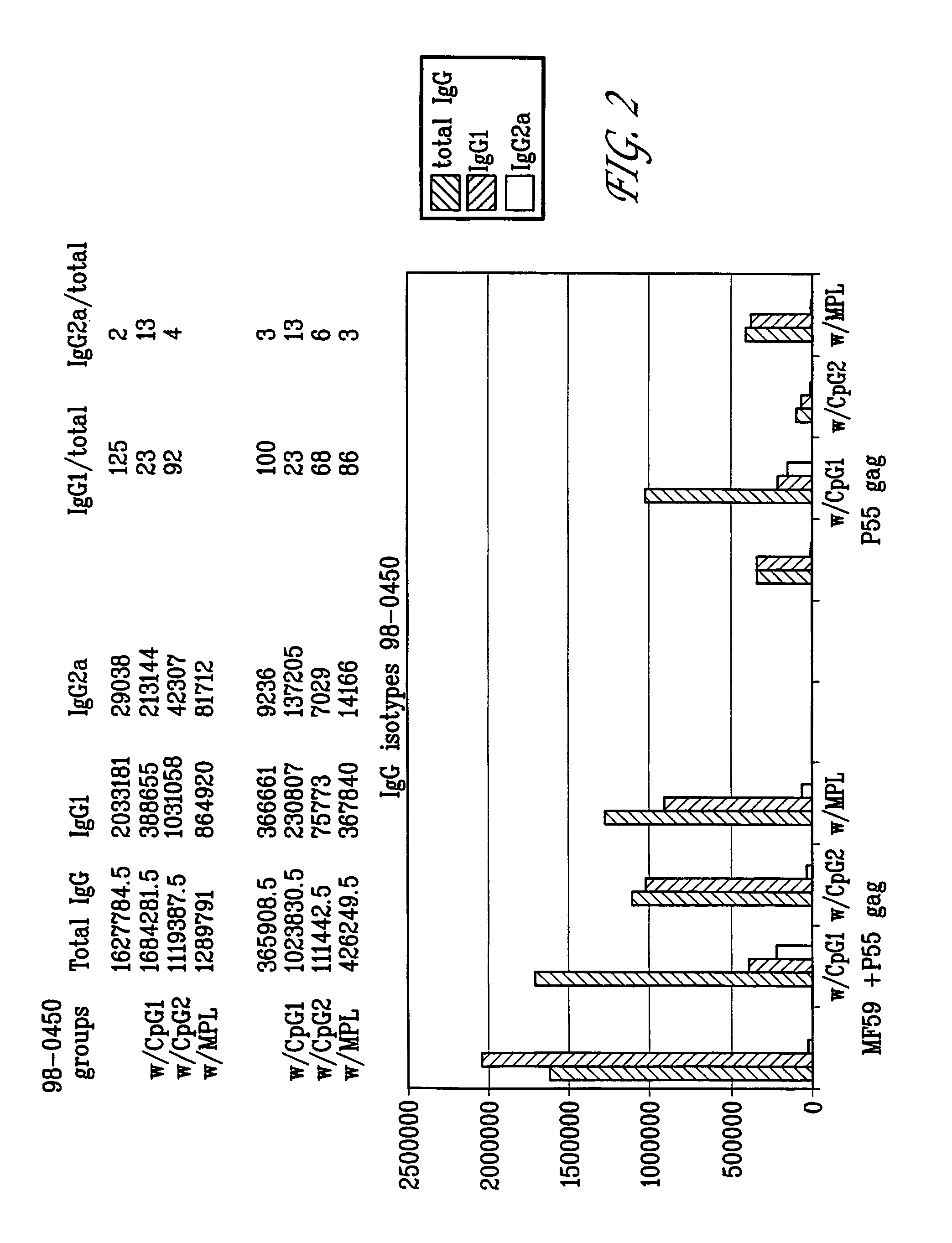
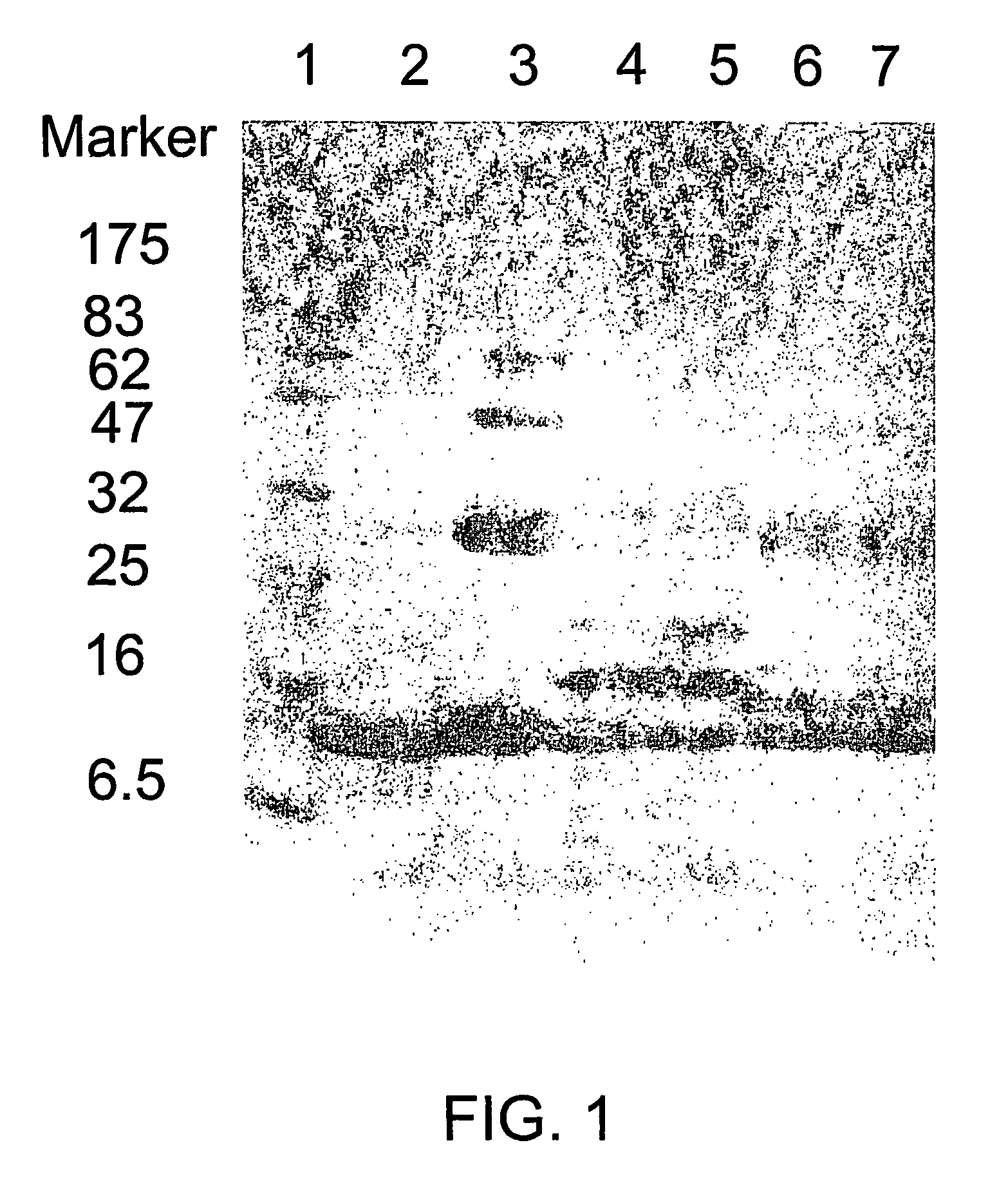
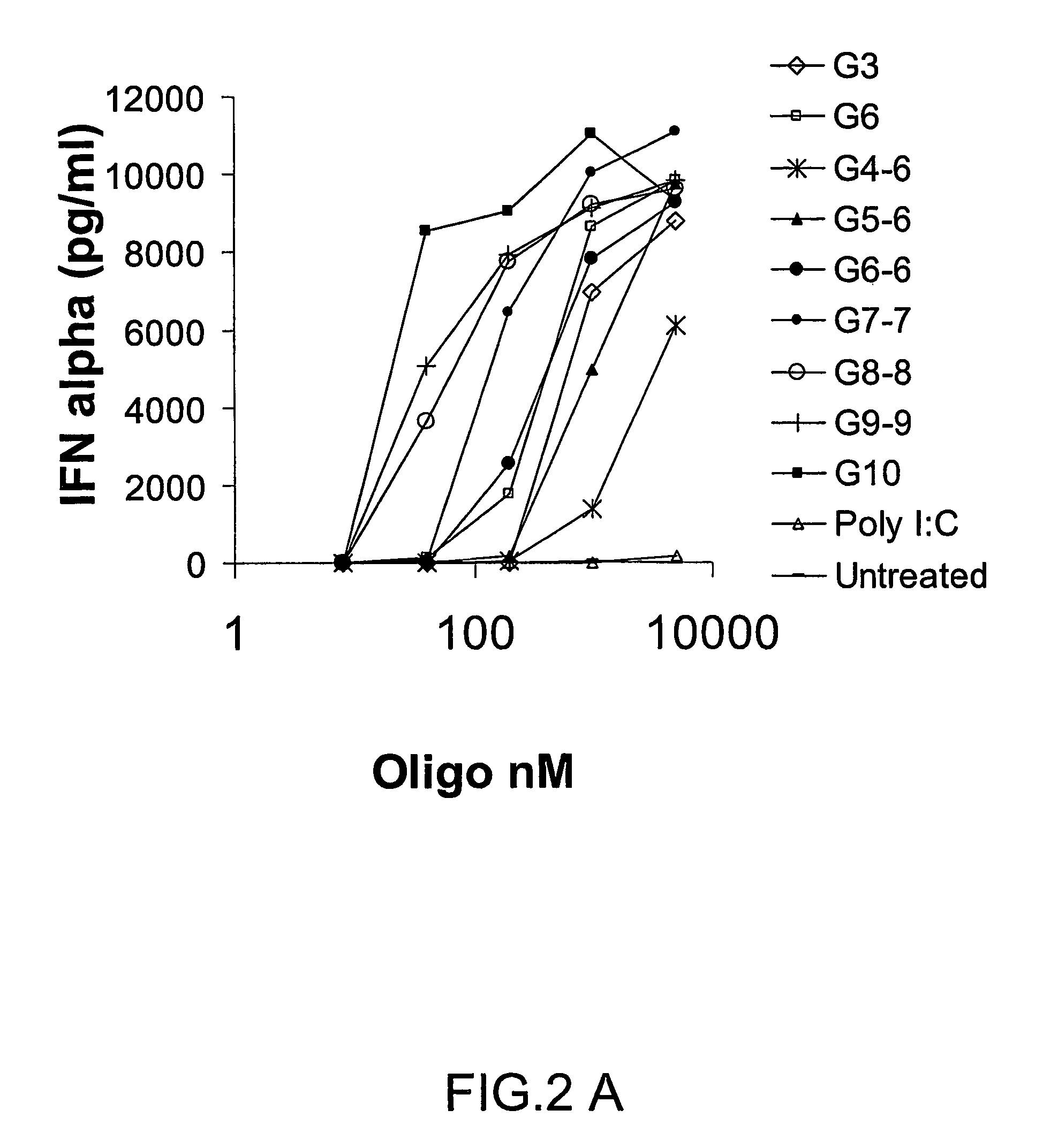
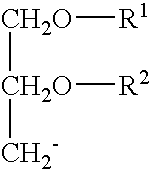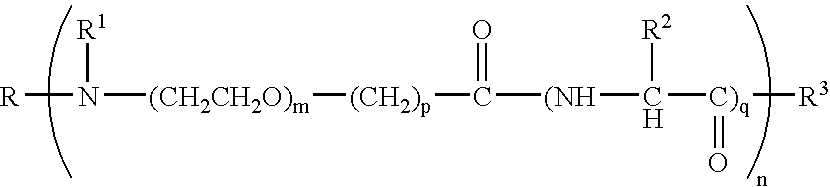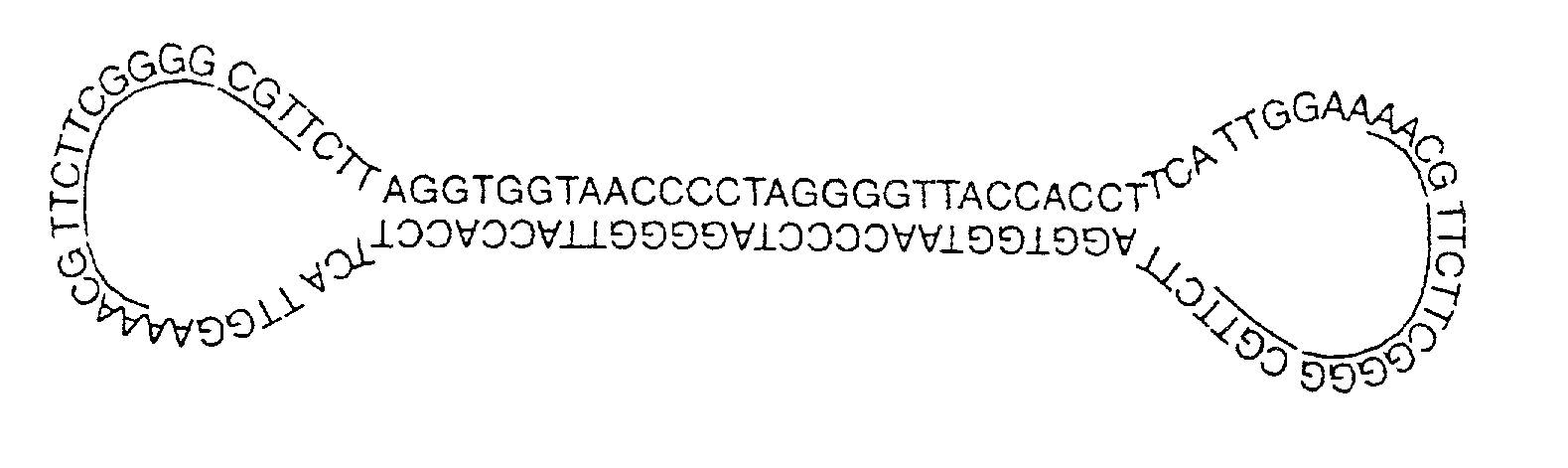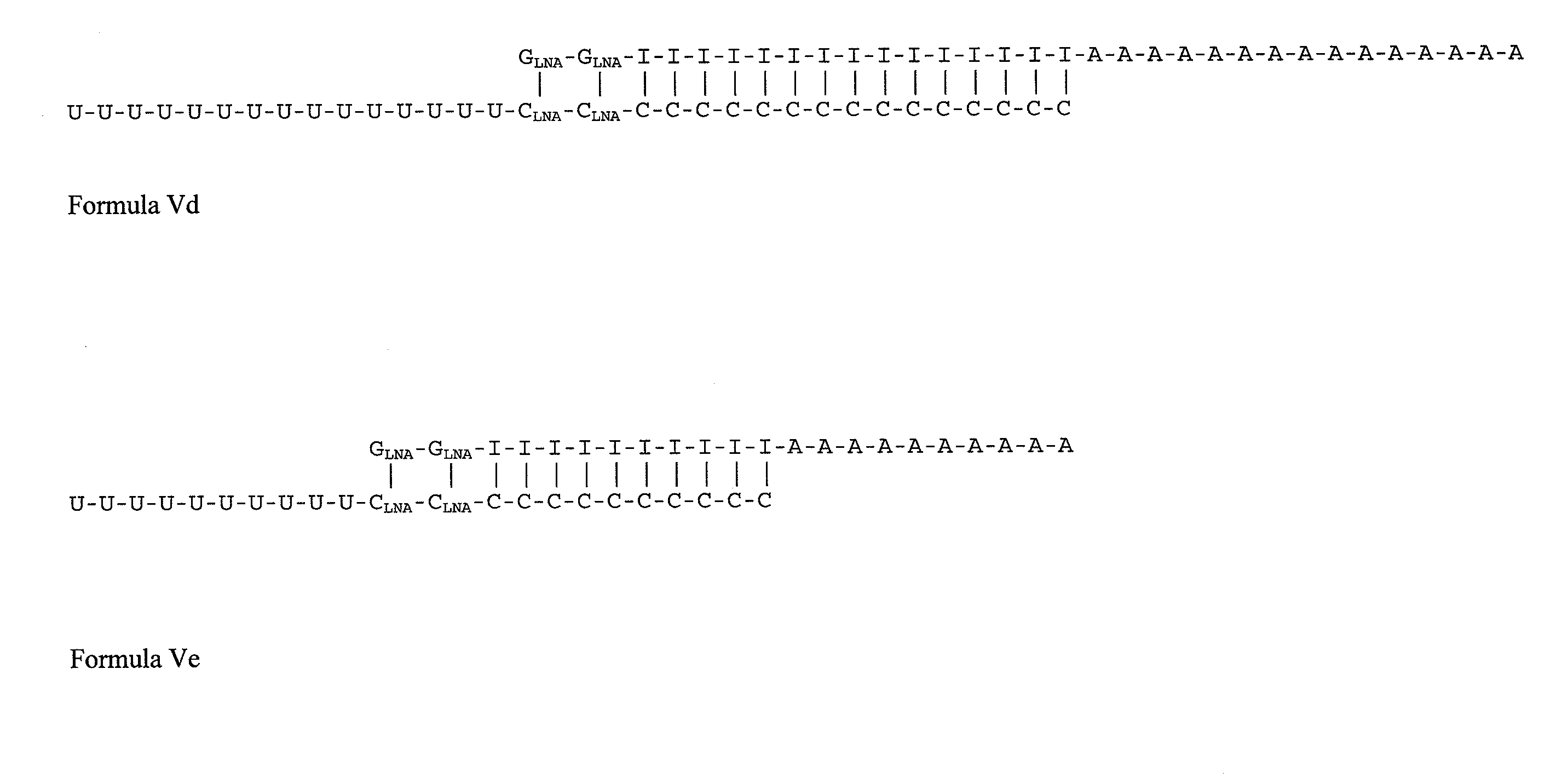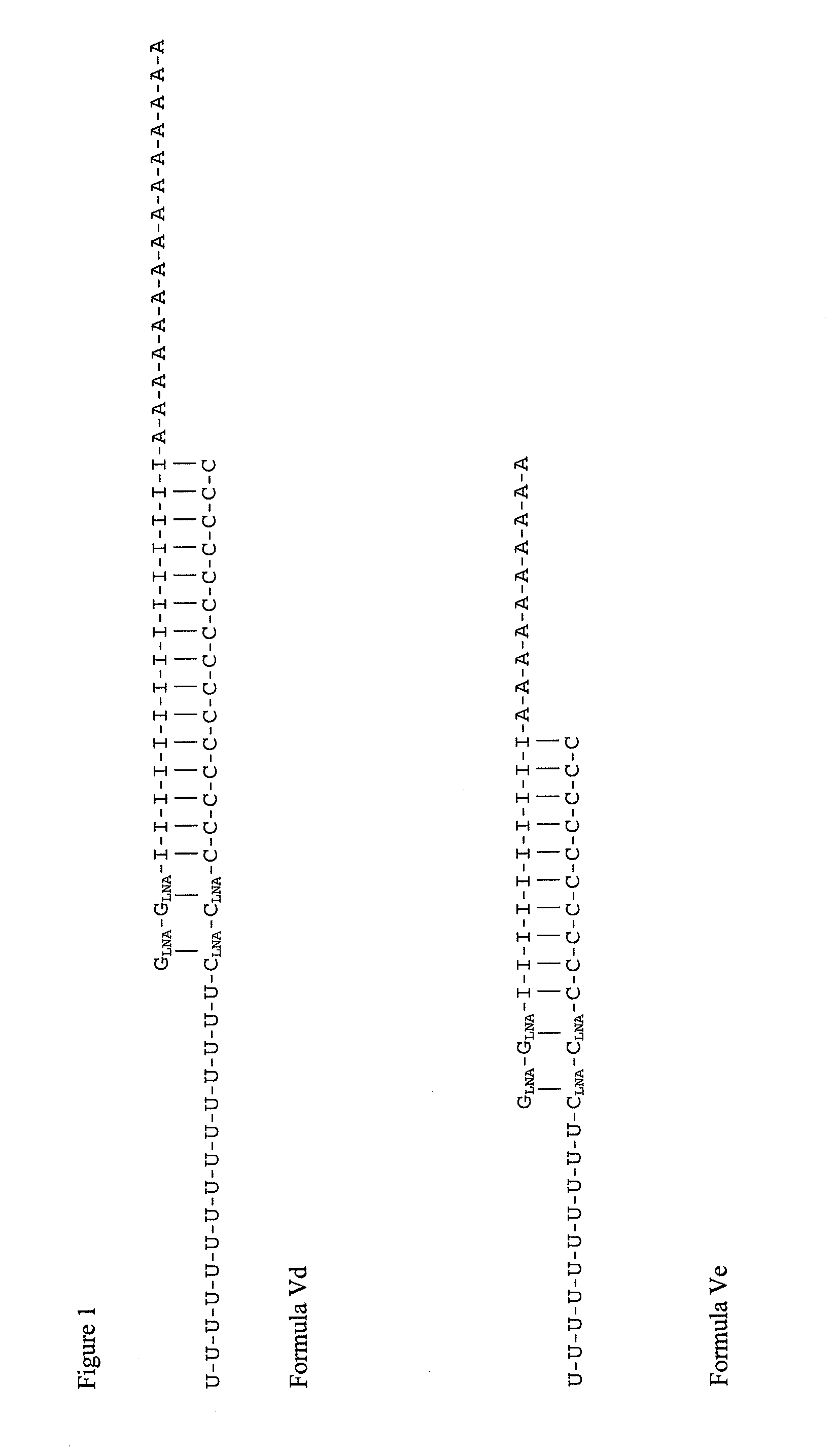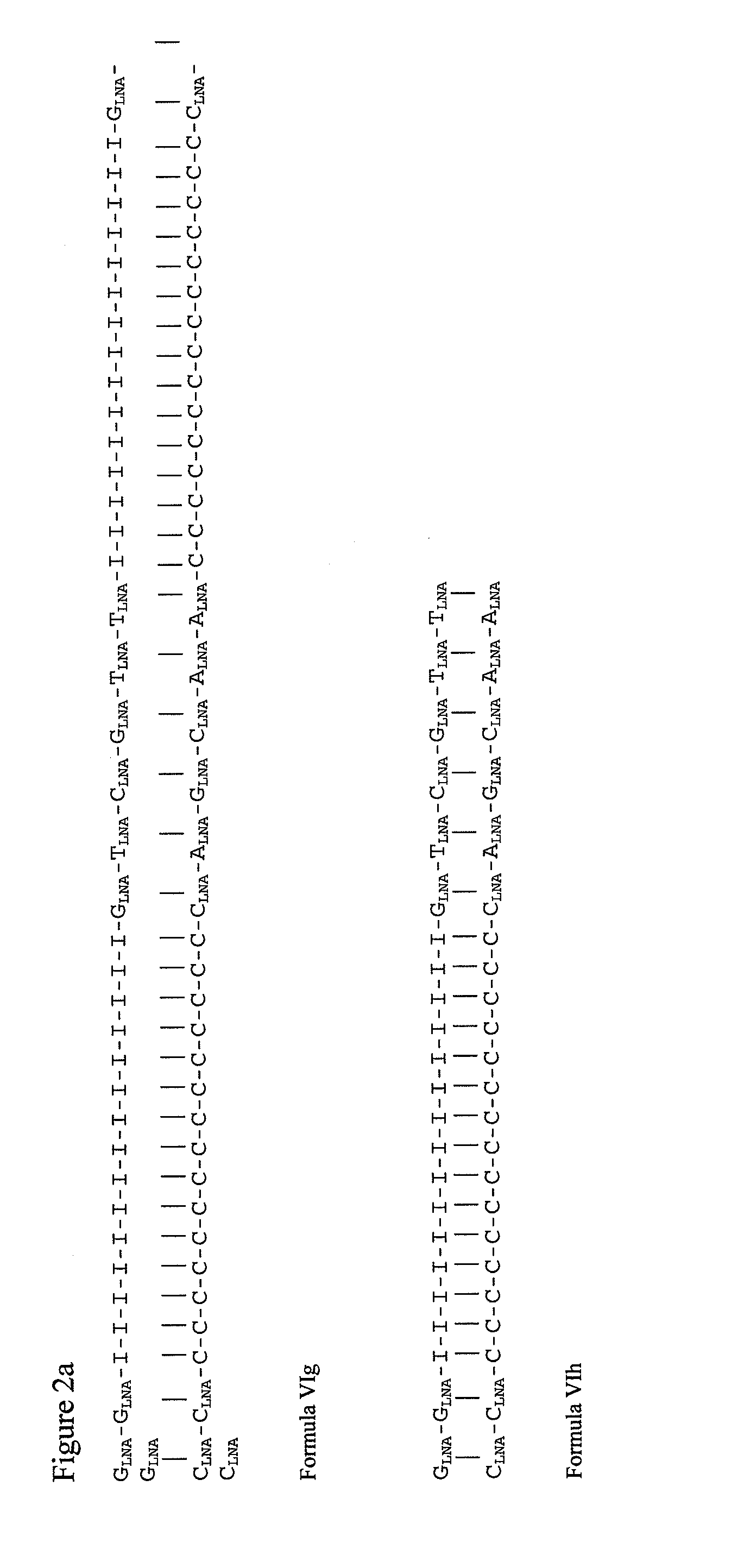Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
303 results about "Immunologic Stimulation" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
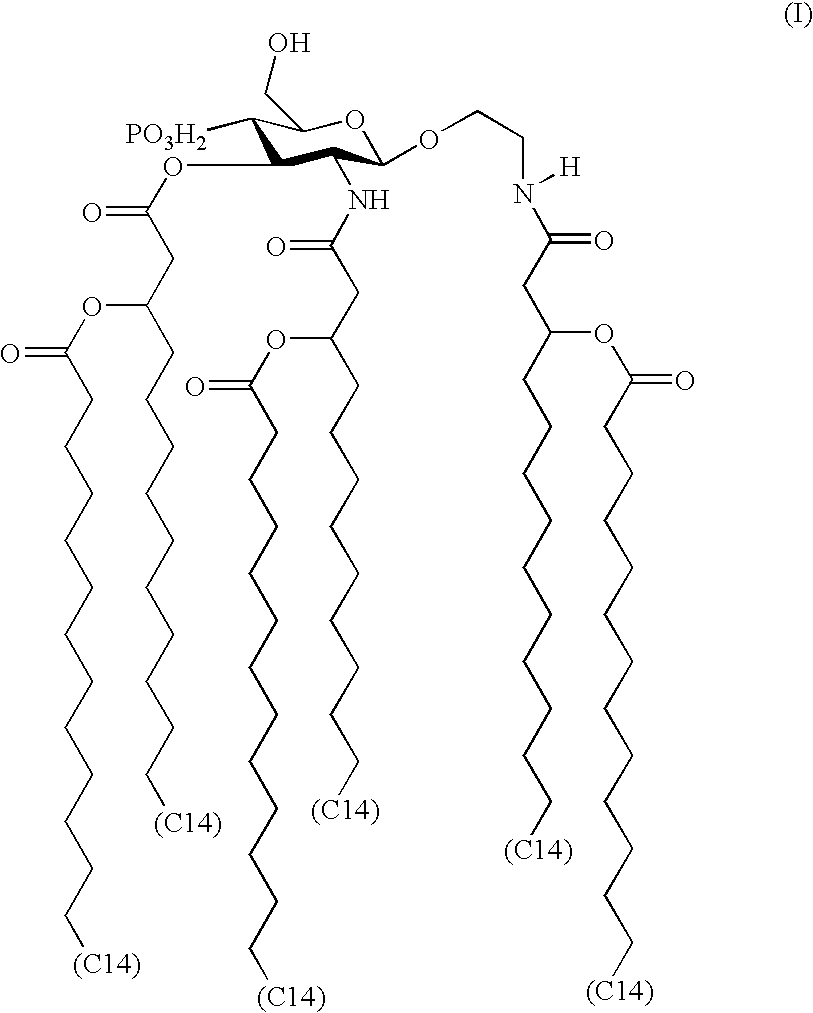
Modified siRNA molecules and uses thereof
ActiveUS20070135372A1Decrease immunostimulatory propertyImmunostimulatory activity of siRNA can be abolishedOrganic active ingredientsSugar derivativesLipid formationLipid particle
The present invention provides chemically modified siRNA molecules and methods of using such siRNA molecules to silence target gene expression. Advantageously, the modified siRNA of the present invention is less immunostimulatory than its corresponding unmodified siRNA sequence and retains RNAi activity against the target sequence. The present invention also provides nucleic acid-lipid particles comprising a modified siRNA, a cationic lipid, and a non-cationic lipid, which can further comprise a conjugated lipid that inhibits aggregation of particles. The present invention further provides methods of silencing gene expression by administering a modified siRNA to a mammalian subject. Methods for identifying and / or modifying an siRNA having immunostimulatory properties are also provided.
Owner:ARBUTUS BIOPHARMA CORPORAT ION
Compositions and methods for oligonucleotide formulations
InactiveUS20080045473A1Improve the level ofFacilitate cross-linkingSugar derivativesGenetic material ingredientsDiseaseOligonucleotide
The present invention relates generally to immunostimulatory nucleic acids, compositions thereof and methods of using the immunostimulatory nucleic acids. In particular the invention relates to palindrome-containing immunostimulatory nucleic acids and the use of these nucleic acids in treating disease.
Owner:ADIUTIDE PHARMA
Immunostimulatory Combinations for Vaccine Adjuvants
This invention discloses immunostimulatory combinations of Tumor Necrosis Factor Receptor Superfamily (TN-FRSF) agonists, Toll-Like Receptor (TLR) agonists, “domain present in NAIP, CIITA, HET-E, TP-I (NACHT)-Leucine Rich Repeat (LRR)” or “NLR” agonists, RIG-I-Like Helicase or “RLH” agonists, purinergic receptor agonists and cytokine / chemokine receptor agonists, together with delivery methods. The combinations, when used alone at the site of pathology, provide immunostimulation that induces host humoral and cellular immunologic responses to eliminate pathogens or neoplasms. Alternatively, when the combinations are used with a defined antigens, these combinations can induce focused humoral and cellular immunologic responses useful as prophylactic and / or ameliorative therapeutic modalities for infections and the treatment of neoplastic disorders.
Owner:RGT UNIV OF CALIFORNIA
Humanized immunomodulatory monoclonal antibodies for the treatment of neoplastic disease or immunodeficiency
InactiveUS20080025980A1Induces proliferation and activationAvoids adverse immunogenic responseHybrid immunoglobulinsAntiviralsDiseaseAntiendomysial antibodies
Owner:CURETECH LTD +1
Immunostimulatory oligonucleotides
InactiveUS20080009455A9Promote digestionStimulate immune responseSugar derivativesGenetic material ingredientsNucleotideBiochemistry
Owner:COLEY PHARMA GMBH +1
Immunomodulatory compositions containing an immunostimulatory sequence linked to antigen and methods of use thereof
InactiveUS7223398B1Reduced antibody productionReduce productionGenetic material ingredientsAntiviralsNucleotideImmuno modulation
The invention provides classes of immunomodulatory compositions which comprise an average of one or more immunostimulatory sequence (ISS) containing polynucleotide conjugated, or attached, to antigen. The extent of conjugation affects immunomodulatory properties, such as extent of antigen-specific antibody formation, including Th1-associated antibody formation, and thus these various conjugate classes are useful for modulating the type and extent of immune response. The invention also includes methods of modulating an immune response using these compositions.
Owner:DYNAVAX TECH CORP
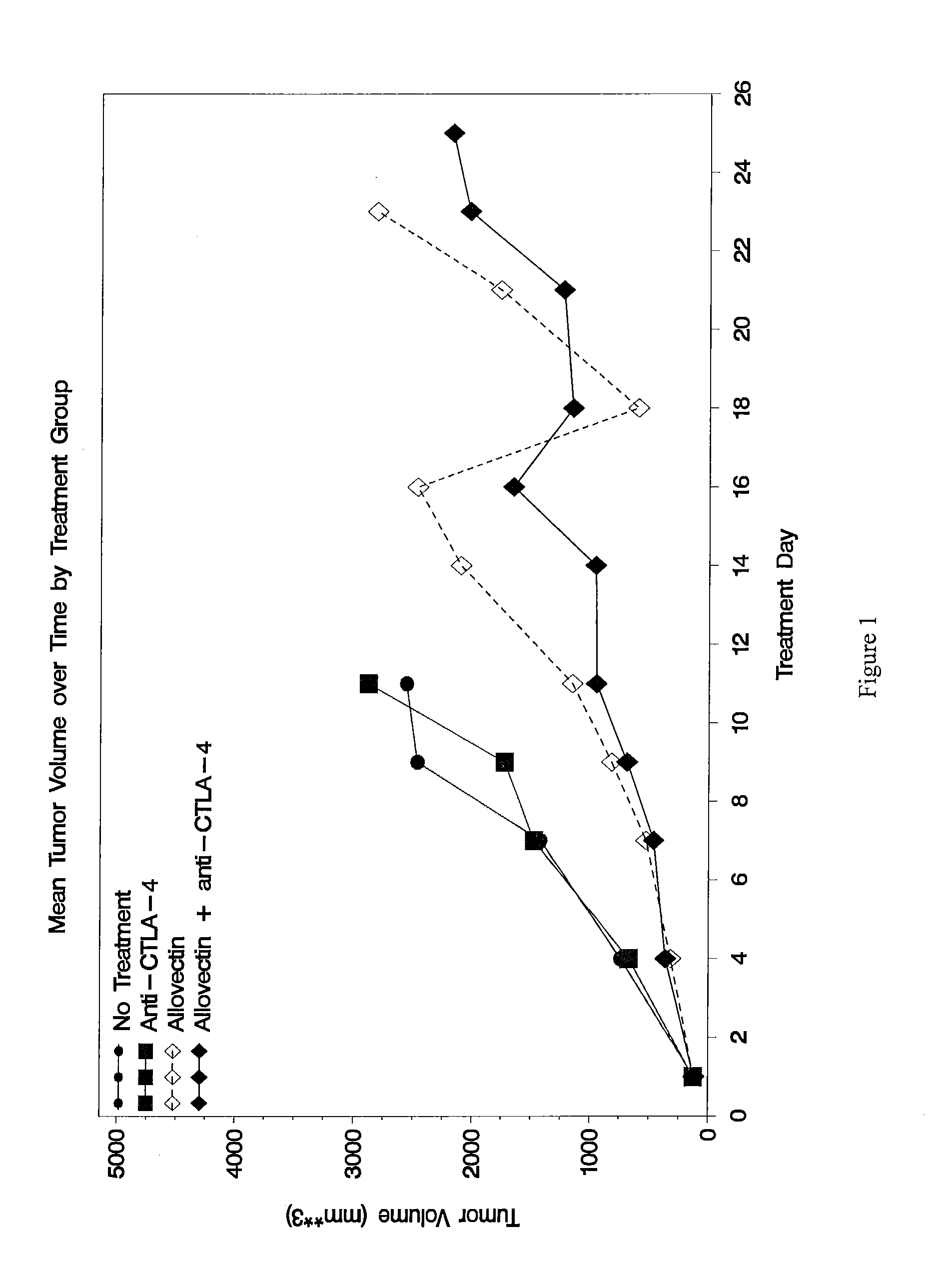
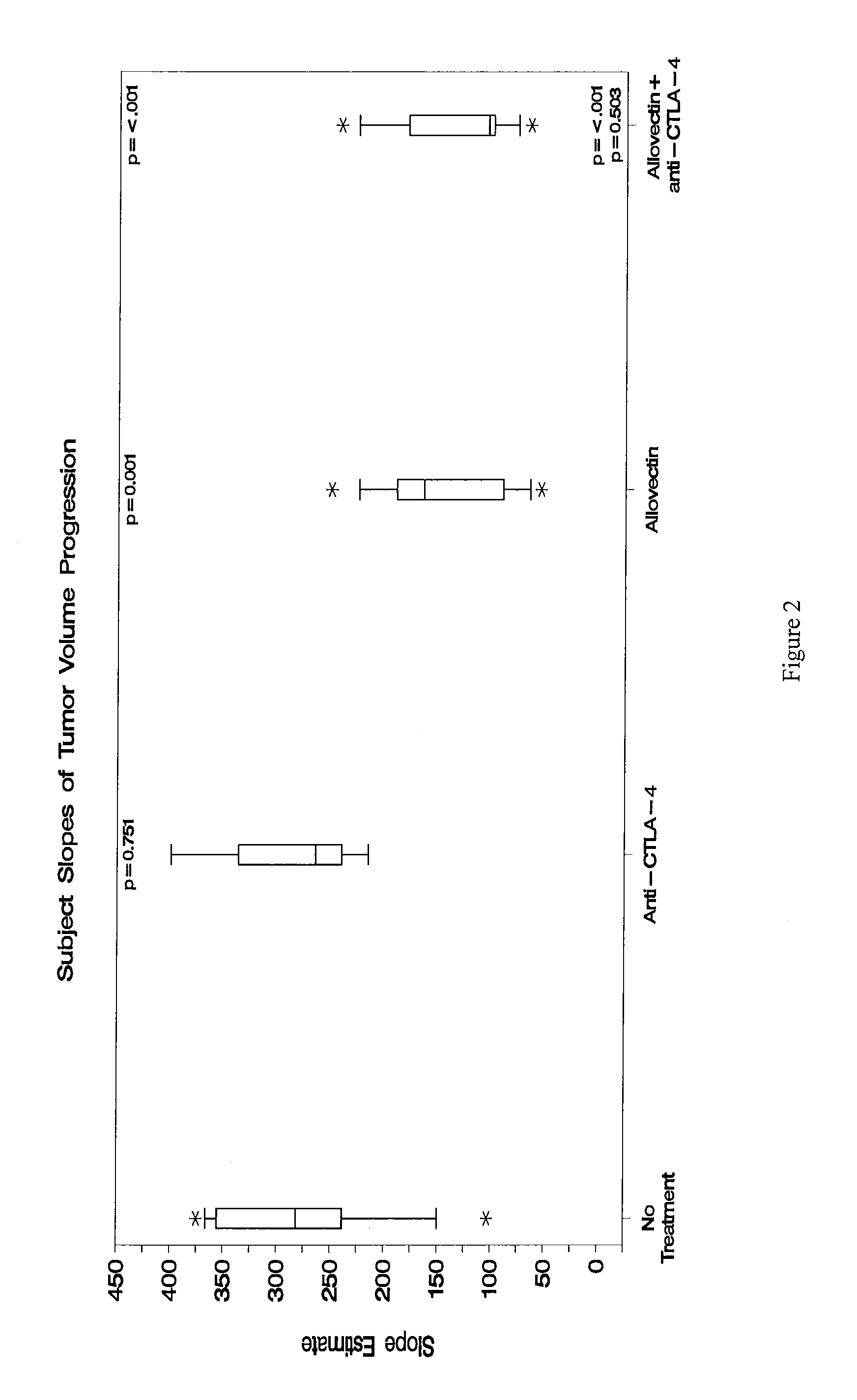
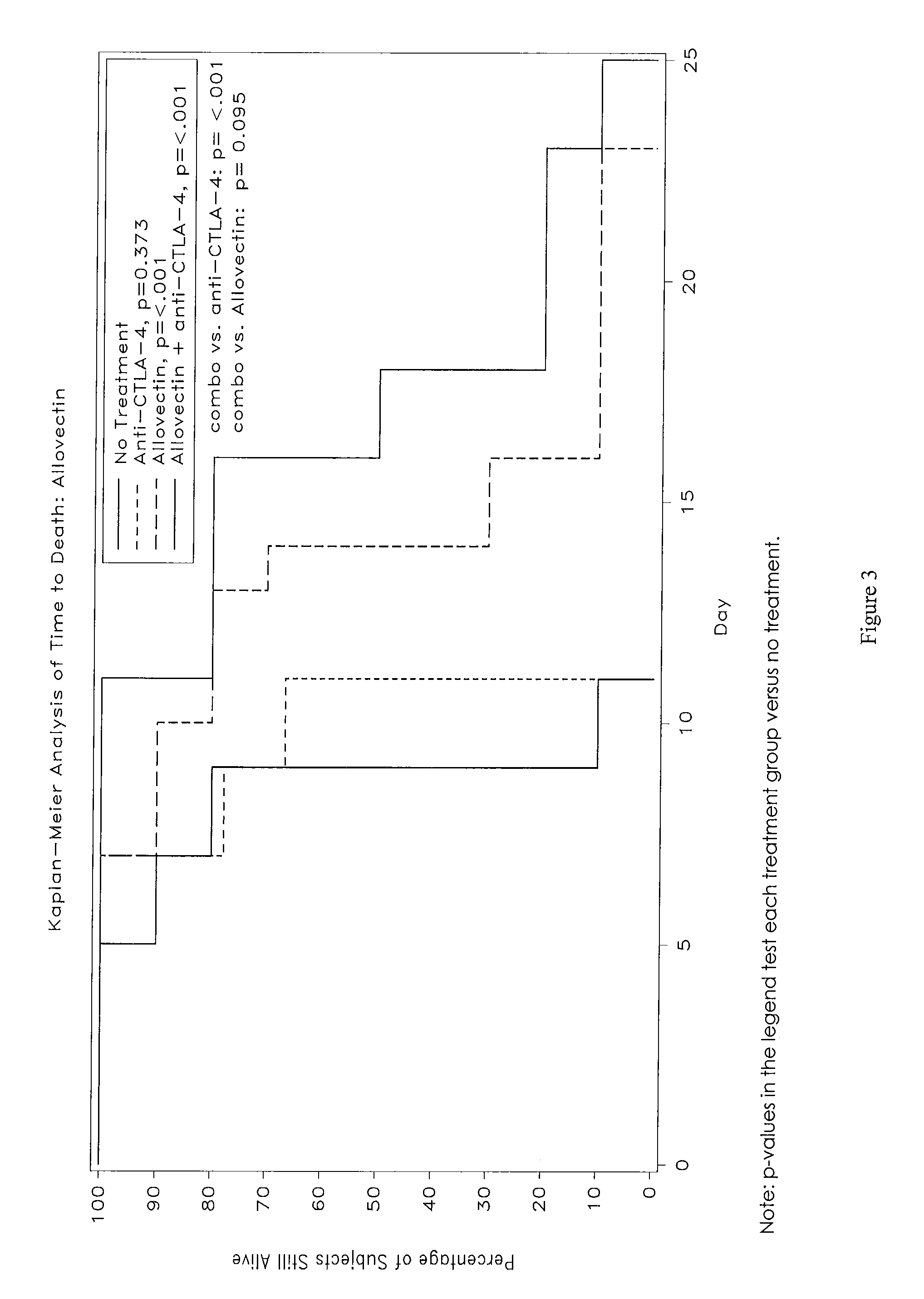
Anti-Ctla-4 Antibody and Cpg-Motif-Containing Synthetic Oligodeoxynucleotide Combination Therapy for Cancer Treatment
InactiveUS20090117132A1Improve immune activityReducing cytotoxic side effectAntibody ingredientsAntineoplastic agentsThird-line therapyPhases of clinical research
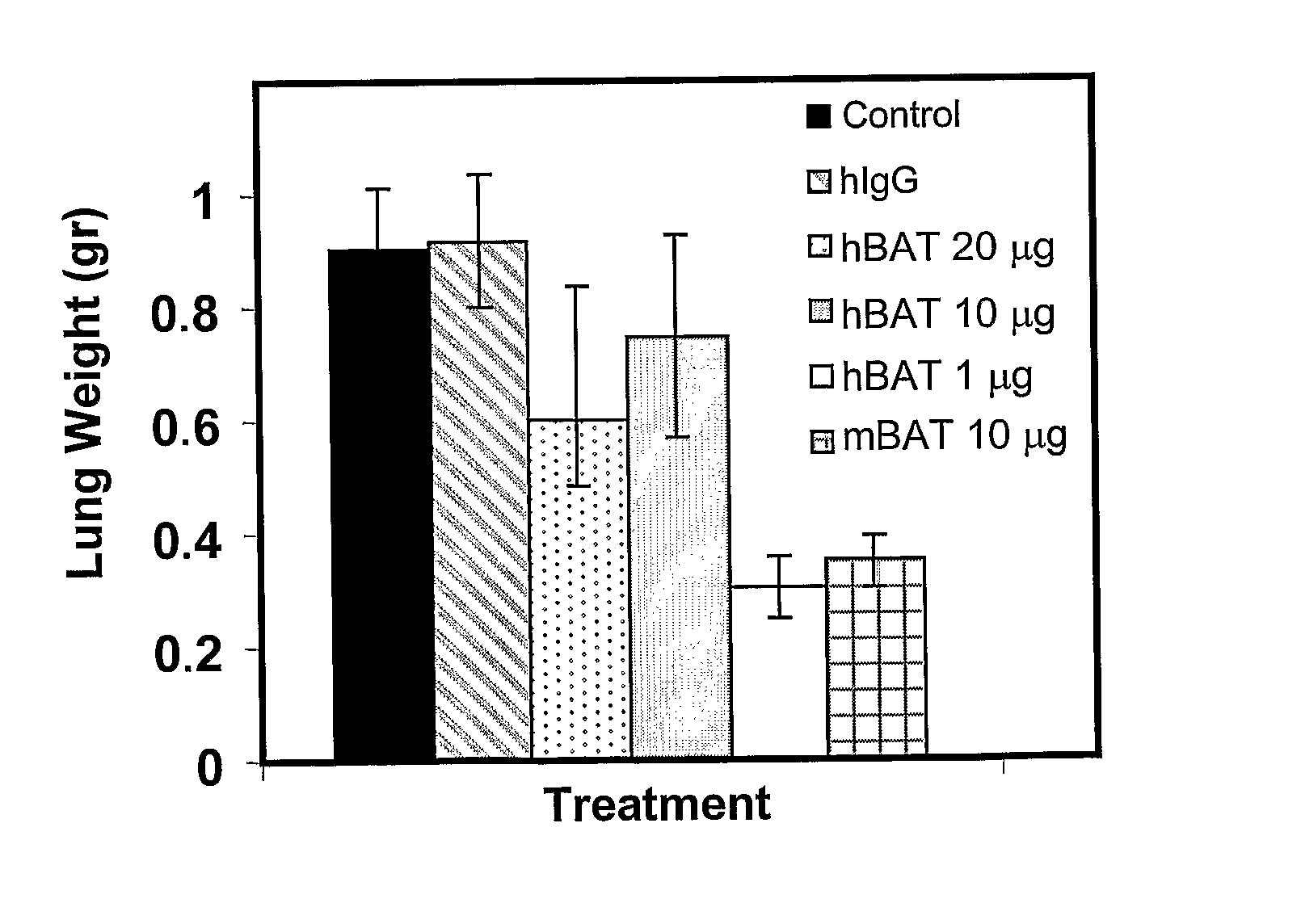
The invention relates to administration of an anti-CTLA-4 antibody, particularly human antibodies to human CTLA-4, such as those having amino acid sequences of antibodies 3.1.1, 4.1.1, 4.8.1, 4.10.2, 4.13.1, 4.14.3, 6.1.1, 11.2.1, 11.6.1, 11.7.1, 12.3.1.1, 12.9.1.1, and MDX-010, in combination with an immunostimulatory nucleotide, i.e, CpG ODN PF3512676, for treatment of cancer. The invention relates to administering a combination of an anti-CTLA-4 antibody and CpG ODN PF3512676 as neoadjuvant, adjuvant, first-line, second-line, and third-line therapy of cancer, whether localized or metastasized, and at any point(s) along the disease continuum (e.g, at any stage of the cancer).
Owner:PFIZER INC +1
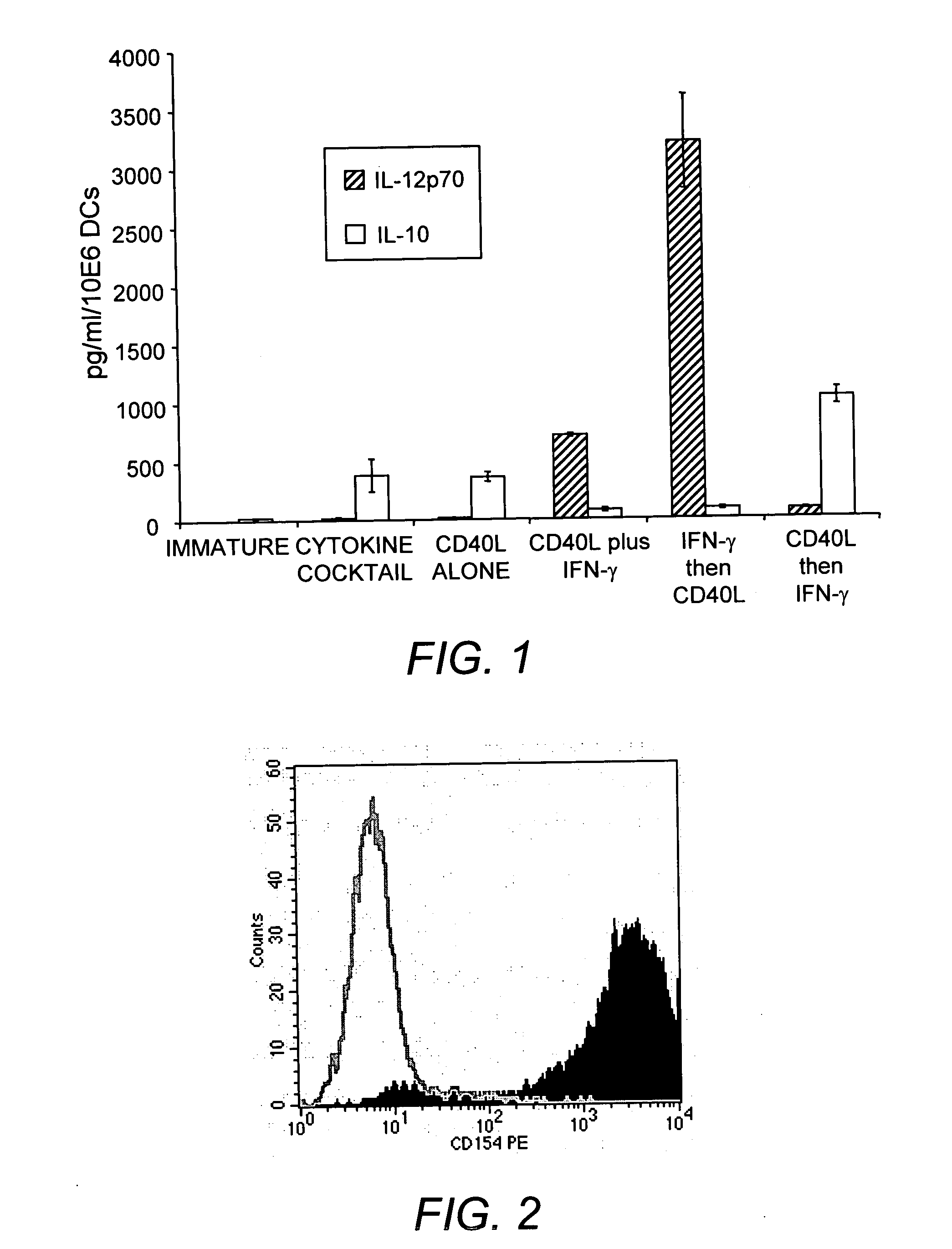
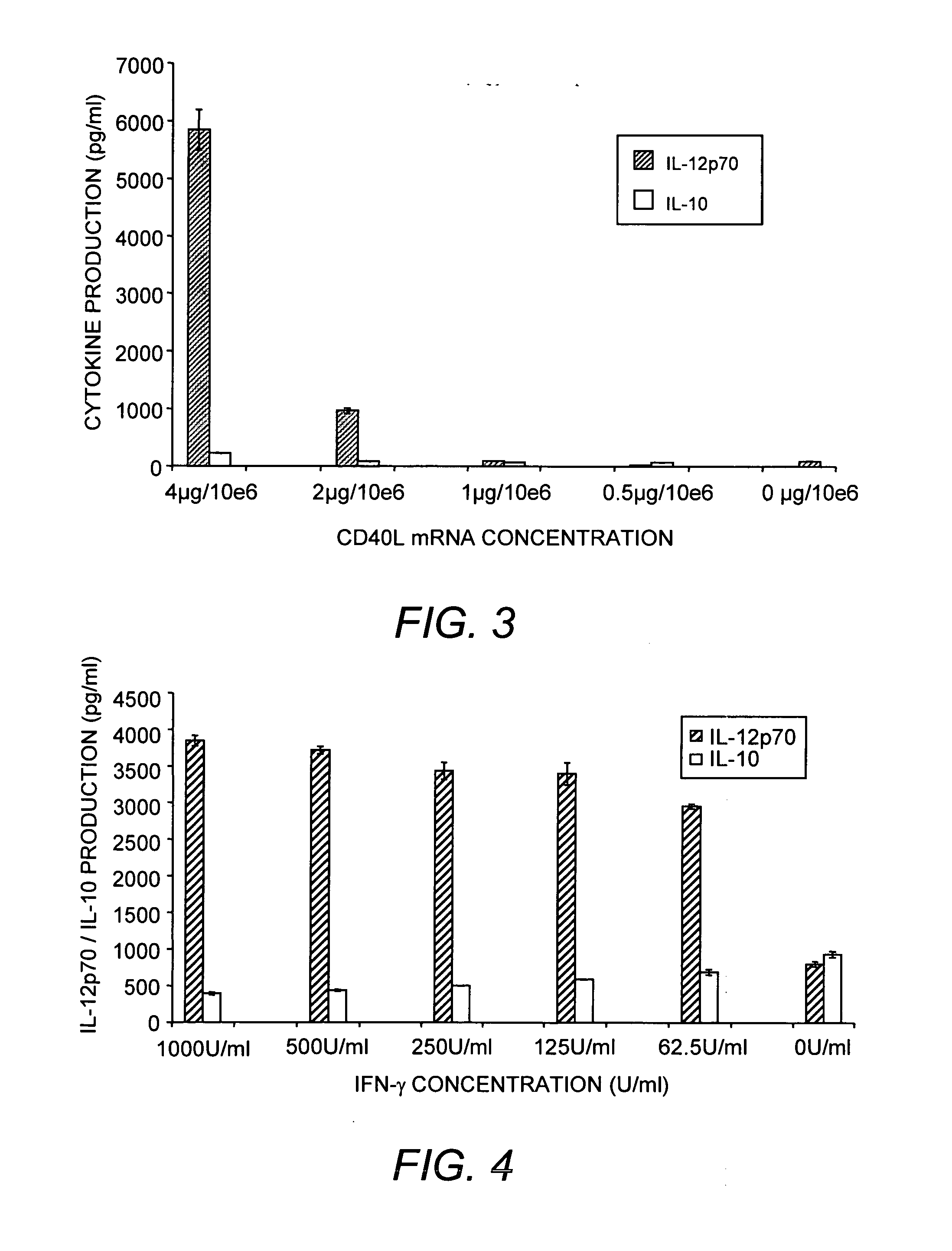
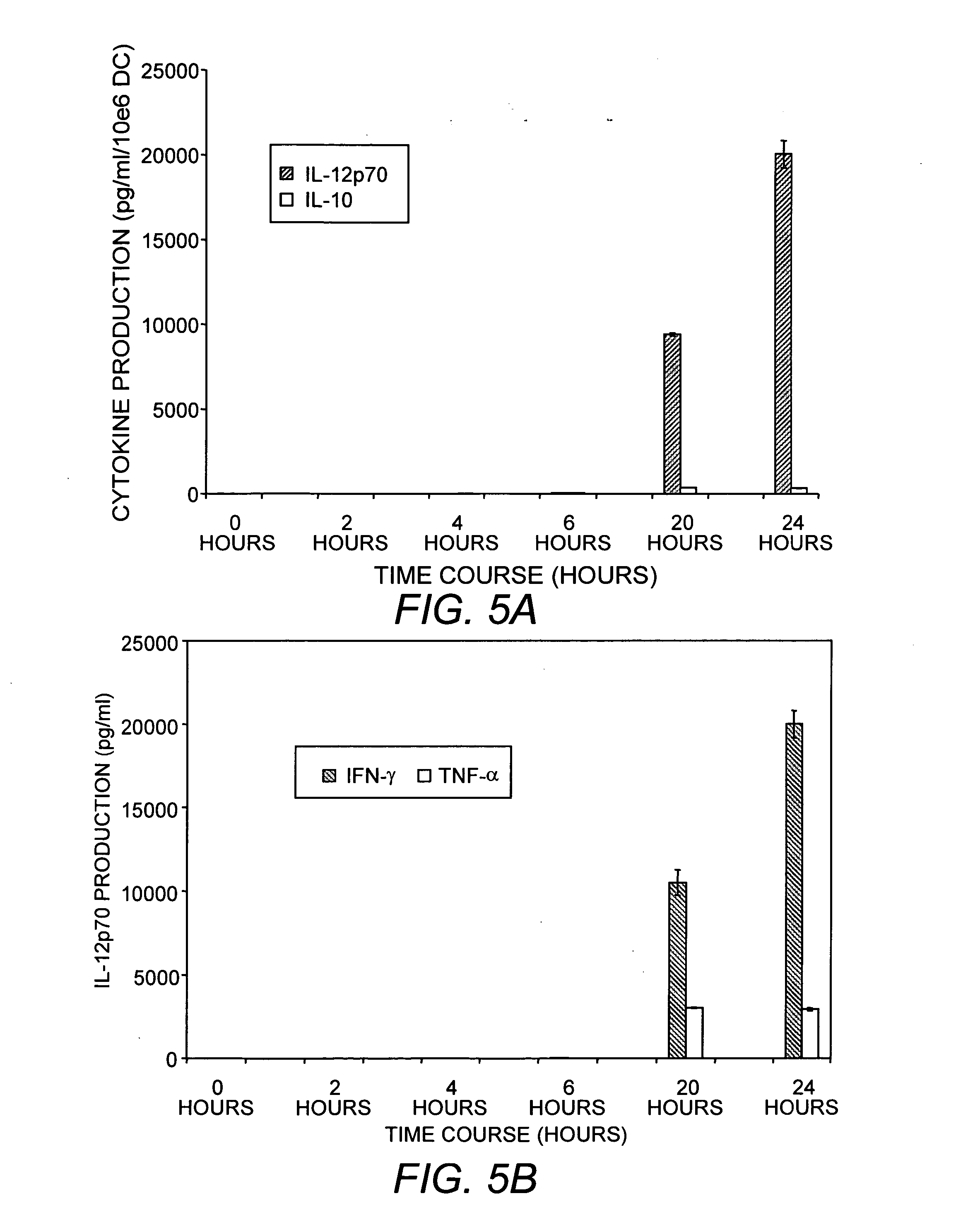
Mature dendritic cell compositions and methods for culturing same
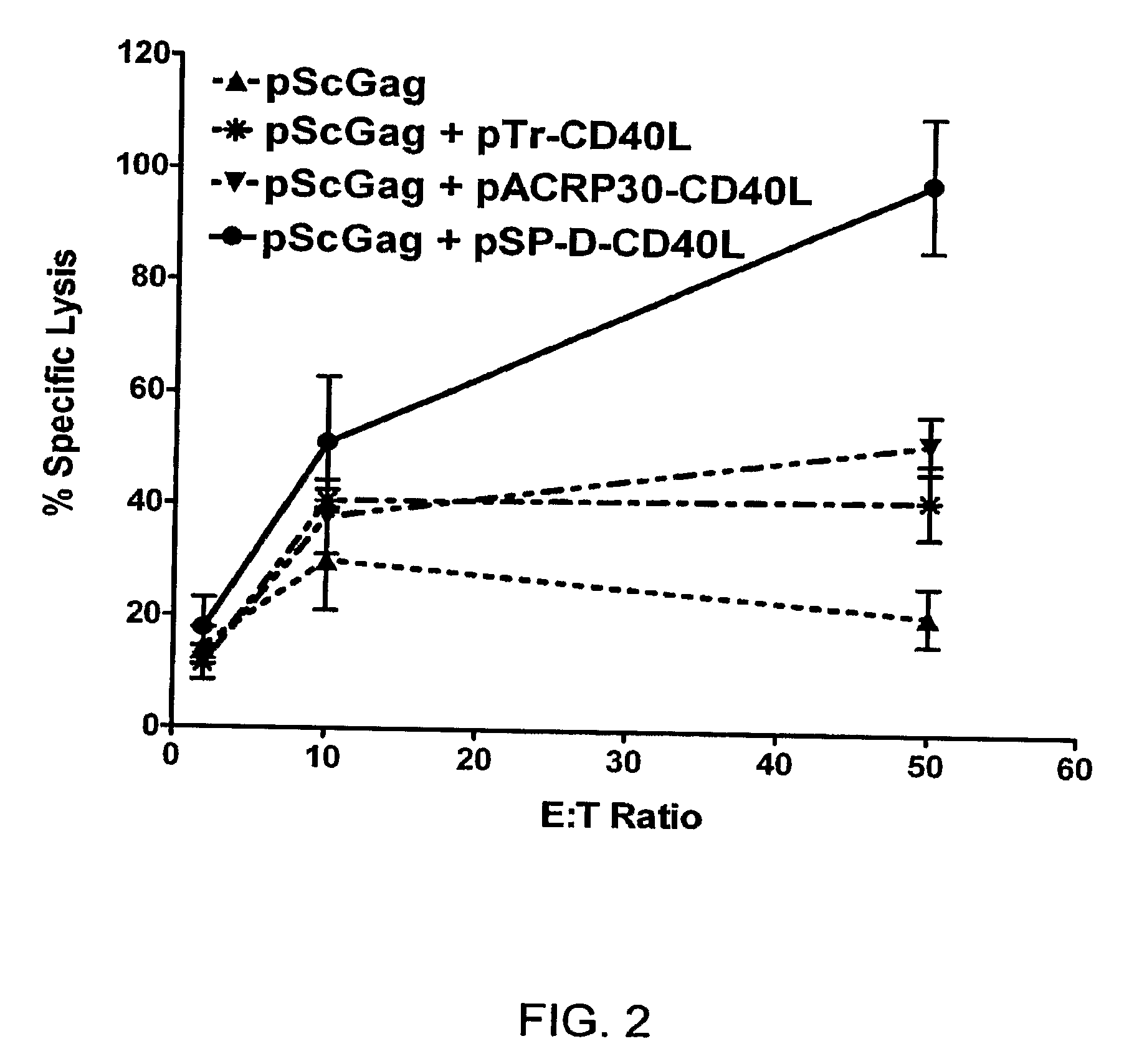
This invention provides methods to prepare and use immunostimulatory cells for enhancing an immune response. The invention provides a method for preparing mature dendritic cells (DCs), comprising the sequential steps of: (a) signaling isolated immature dendritic cells (iDCs) with a first signal comprising an interferon gamma receptor (IFN-γR) agonist and / or a tumor necrosis factor alpha receptor (TNF-αR) agonist to produce signaled dendritic cells; and (b) signaling said signaled dendritic cells with a second transient signal comprising an effective amount of a CD40 agonist to produce CCR7+ mature dendritic cells. Also provided by this invention are enriched populations of dendritic cells prepared by the methods of the invention. Such dendritic cells have enhanced immunostimulatory properties and increased IL-12 secretion and / or decreased IL-10 secretion. CD40 signaling can be initiated by one or more of polypeptide translated from an exogenous polynucleotide encoding CD40L (e.g., mRNA or DNA), an agonistic antibody to CD40 receptor or by CD40 ligand polypeptide. The enriched populations can be further modified by the administration of an immunogen to the DC. The DC will take up and process the immunogen on its cell surface.
Owner:COIMMUNE INC +1
Immunostimulatory siRNA molecules and uses therefor
InactiveUS20060025366A1Modulate immune responseNervous disorderActivity regulationGene expressionCell biology
The present invention provides siRNA molecules and methods of using such siRNA molecules to modulate an immune response and to silence expression of a target gene.
Owner:ARBUTUS BIOPHARMA CORPORAT ION
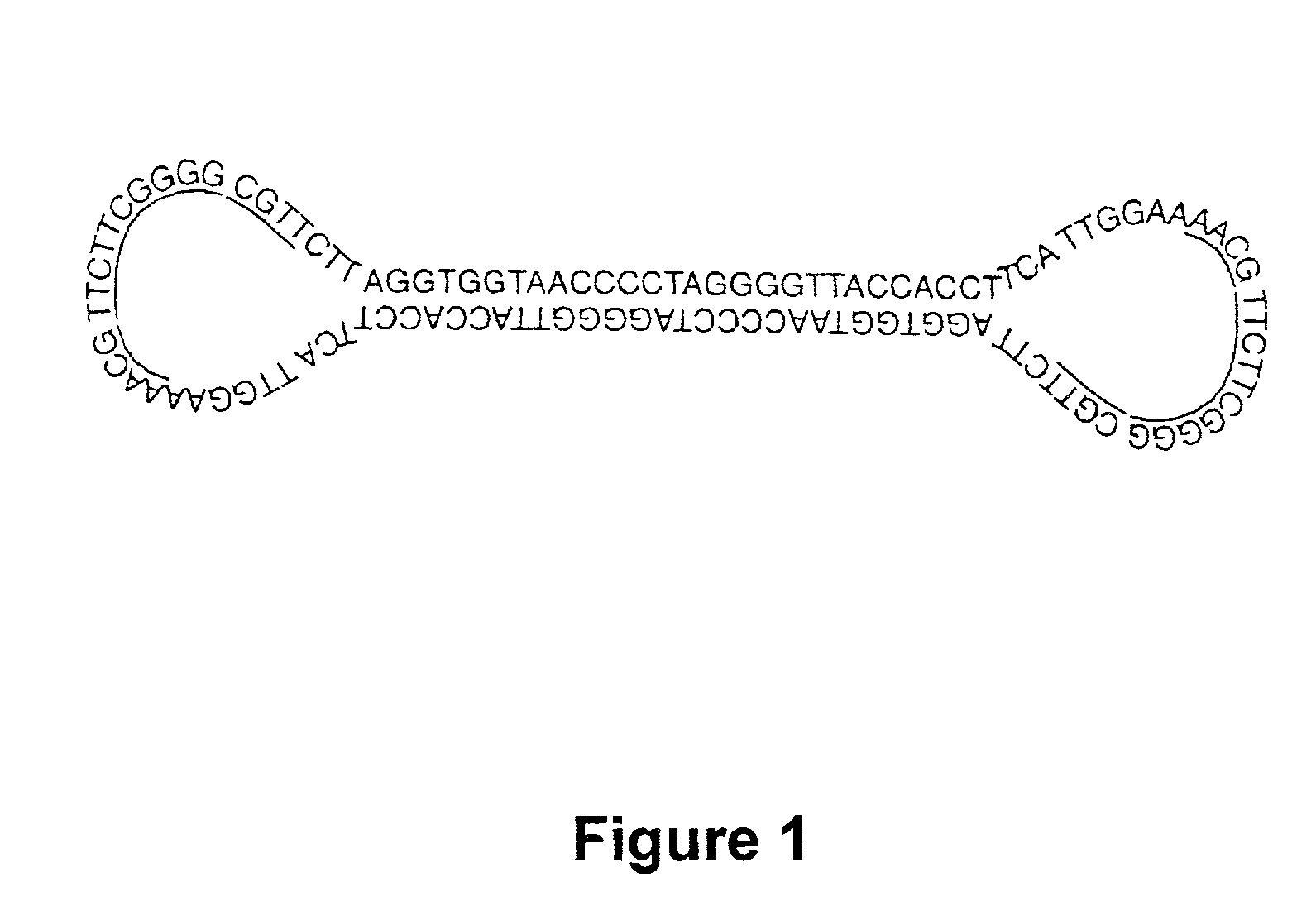
Covalently closed nucleic acid molecules for immunostimulation
InactiveUS20030125279A1Suppress stimulatory effectImprove the level ofOrganic active ingredientsSugar derivativesNucleic acid moleculeSingle strand
Short deoxyribonucleic acid molecules that are partially single-stranded, dumbbell-shaped, and covalently closed, which contain one or more unmethylated cytosine guanosine motif (CpG motif) and exhibit immunomodifying effects. Such molecules can be used for immunostimulation applications in humans or vertebrates.
Owner:GILEAD SCI INC
CPG-like nucleic acids and methods of use thereof
InactiveUS20080226649A1Enhancing neutrophil proliferationOrganic active ingredientsSugar derivativesAllergyPurine
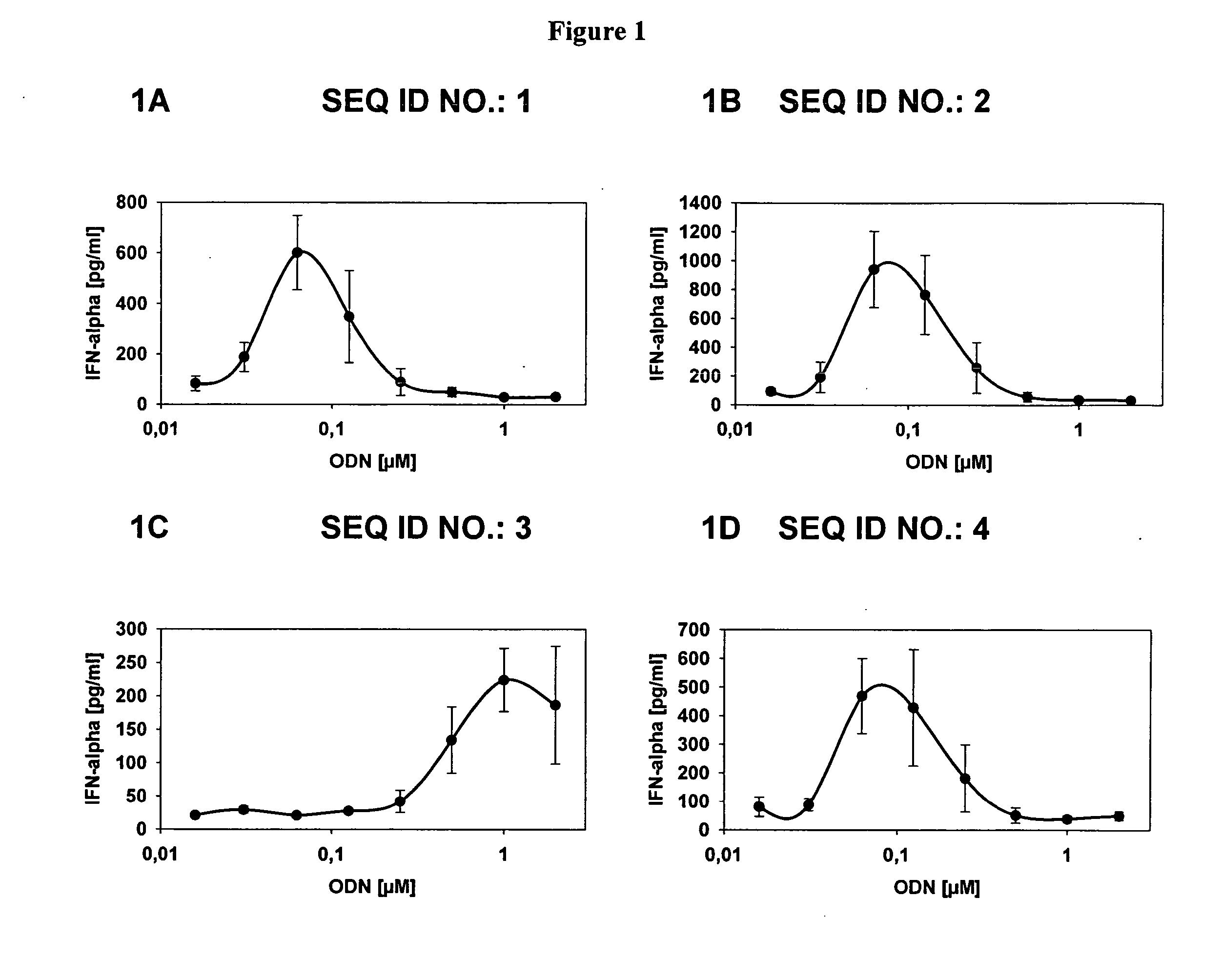
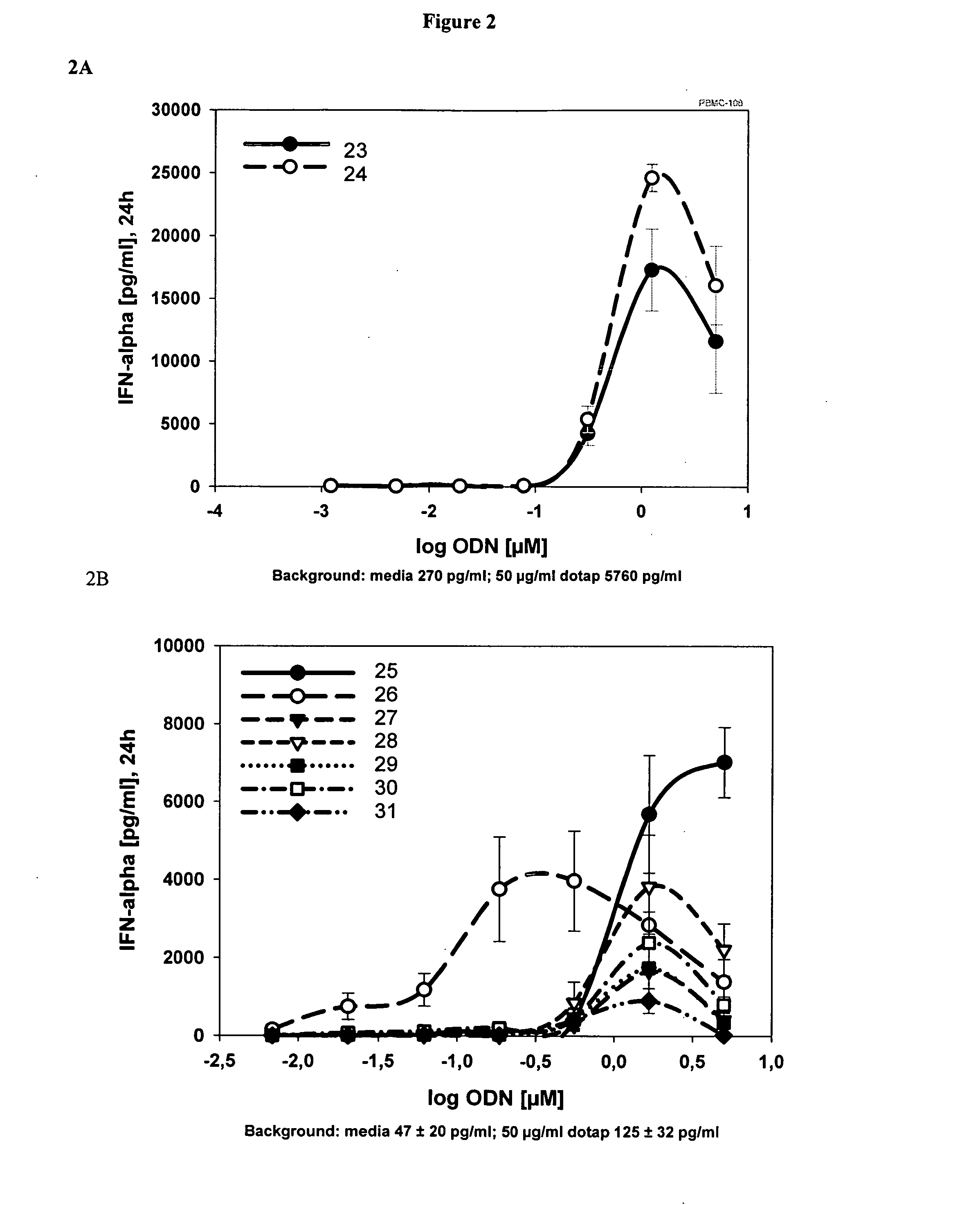
Immunostimulatory compositions described as CpG-like nucleic acids are provided, including nucleic acids having immunostimulatory characteristics of CpG nucleic acid, despite certain substitutions of C, G, or C and G of the CpG dinucleotide. The substitutions can include, among others, exchange of methylated C for C, inosine for G, and ZpY for CpG, where Z is cytosine or dSpacer and Y is inosine, 2-aminopurine, nebularine, or dSpacer. Also provided are methods for inducing an immune response in a subject using the CpG-like nucleic acids. The methods are useful in the treatment of a subject that has or is at risk of developing an infectious disease, allergy, asthma, cancer, anemia, thrombocytopenia, or neutropenia.
Owner:COLEY PHARMA GMBH
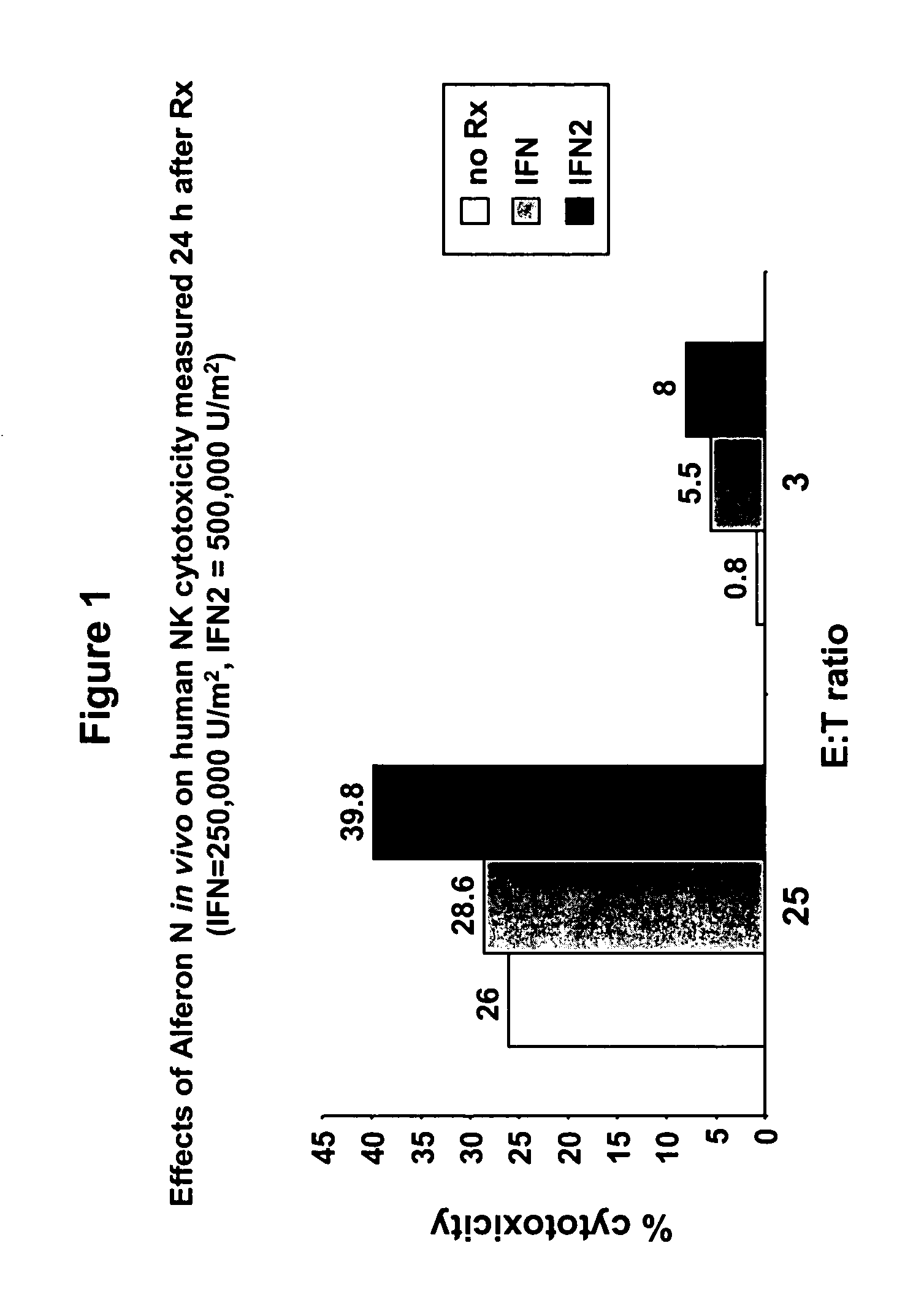
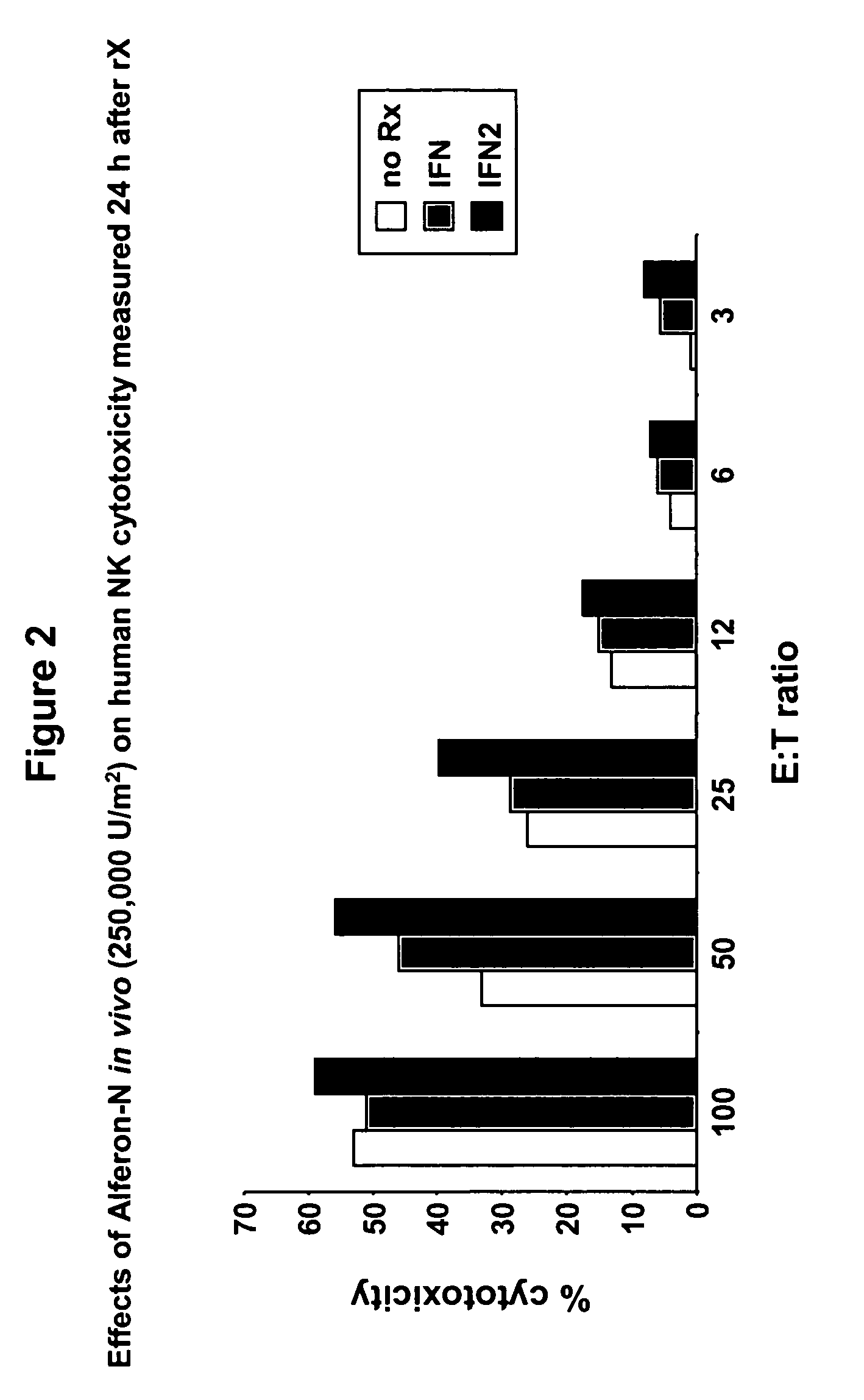
Interferon immunotherapy
InactiveUS7041301B1Inhibit progressStrong cytotoxicityPeptide/protein ingredientsPeptide preparation methodsHuman patientInterferon alpha
A method for reducing the recurrence of a resectable malignant tumor includes administering an immunostimulatory dosage of an α-interferon composition, then surgically resecting the malignant tumor. A method for treating a human patient having a non-resectable malignant tumor includes administering an immunostimulatory dosage of an α-interferon composition to the patient and treating the patient with effective non-surgical medical methodologies to diminish the tumor. An article of manufacture combines an α-interferon composition within a packaging material and a package label or insert indicating that administration of an immunostimulatory dosage of an α-interferon composition followed by surgical resection of a malignant tumor can be effective for treating a human patient having the malignant tumor.
Owner:MAYO FOUND FOR MEDICAL EDUCATION & RES
Mature dendritic cell compositions and methods for culturing same
This invention provides methods to prepare and use immunostimulatory cells for enhancing an immune response. The invention provides a method for preparing mature dendritic cells (DCs), comprising the sequential steps of: (a) signaling isolated immature dendritic cells (iDCs) with a first signal comprising an interferon gamma receptor (IFN-γR) agonist and / or a tumor necrosis factor alpha receptor (TNF-αR) agonist to produce signaled dendritic cells; and (b) signaling said signaled dendritic cells with a second transient signal comprising an effective amount of a CD40 agonist to produce CCR7+ mature dendritic cells. Also provided by this invention are enriched populations of dendritic cells prepared by the methods of the invention. Such dendritic cells have enhanced immunostimulatory properties and increased IL-12 secretion and / or decreased IL-10 secretion. CD40 signaling can be initiated by one or more of polypeptide translated from an exogenous polynucleotide encoding CD40L (e.g., mRNA or DNA), an agonistic antibody to CD40 receptor or by CD40 ligand polypeptide. The enriched populations can be further modified by the administration of an immunogen to the DC. The DC will take up and process the immunogen on its cell surface.
Owner:COIMMUNE INC +1
Double-stranded locked nucleic acid compositions
InactiveUS20090041809A1High thermostabilityIncrease stabilityAntibacterial agentsOrganic active ingredientsDouble strandNucleoside
Immunostimulatory agents, including nucleic acids having one or more than one locked nucleic acid (LNA) nucleosides are provided. The nucleic acids may further comprise CpG motifs. The nucleic acids may be double stranded, and may comprise dsRNA.
Owner:NVENTA BIOPHARMACEUTICALS CORP
Synergistic Anti-tumor efficacy using alloantigen combination immunotherapy
InactiveUS20130280265A1Increased activationOrganic active ingredientsAntibody ingredientsImmunotherapeutic agentEfficacy
The present disclosure provides combinations of immunotherapeutics and methods for treating medical conditions that are characterized by the lack of an effective immune response, for example as would result following a down-regulation of MHC class I, such as in cancer. The immunotherapeutic compositions of the invention, which can be used to treat the medical conditions, include one or more immunostimulatory antibodies or molecules having specificity for CTLA-4, PD-1, PD-L1, PD-L2, CD40, OX40, CD137, GITR, ILT2, or ILT3, or ligands for these molecules (e.g., an isolated fully-human monoclonal antibody) in association with one or more alloantigens, such as, vector(s) capable of expressing protein(s) or peptide(s) that stimulate T-cell immunity against tissues or cells, formulated in a pharmaceutically acceptable carrier. The proteins or peptides may comprise class I major histocompatibility complex (MHC) antigens, β2-microglobulins, or cytokines. The MHC antigen may be foreign to the subject. The MHC antigen may be HLA-B7.
Owner:VICAL INC
Microemulsions with adsorbed macromolecules and microparticles
InactiveUS8206749B1Powerful toolStimulate immune responseAntibacterial agentsDigestive systemHydroxybutyric acidAdjuvant
Microparticles with adsorbent surfaces, methods of making such microparticles, and uses thereof, are disclosed. The microparticles comprise a polymer, such as a poly(α-hydroxy acid), a polyhydroxy butyric acid, a polycaprolactone, a polyorthoester, a polyanhydride, and the like, and are formed using cationic, anionic, or nonionic detergents. The surface of the microparticles efficiently adsorb biologically active macromolecules, such as DNA, polypeptides, antigens, and adjuvants. Also provided are compositions of an oil droplet emulsion having a metabolizable oil and an emulsifying agent. Immunogenic compositions having an immunostimulating amount of an antigenic substance, and an immunostimulating amount of an adjuvant composition are also provided. Methods of stimulating an immune response, methods of immunizing a host animal against a viral, bacterial, or parasitic infection, and methods of increasing a Th1 immune response in a host animal by administering to the animal an immunogenic composition of the microparticles, and / or microemulsions of the invention, are also provided.
Owner:NOVARTIS VACCINES & DIAGNOSTICS INC
Melan-A- carrier conjugates
InactiveUS7537767B2Improve responseMore immunogenicSsRNA viruses negative-senseBiocideDiseaseAllergy
Owner:CYTOS BIOTECHNOLOGY AG
Synthetic immunogenic but non-deposit-forming polypeptides and peptides homologous to amyloid beta, prion protein, amylin, alpha-synuclein, or polyglutamine repeats for induction of an immune response thereto
InactiveUS7479482B2Reduce formationAvoid formingHormone peptidesNervous disorderPassive ImmunizationsAmyloid beta
The present invention relates to immunogenic but non-depositing-forming polypeptides or peptides homologous to amyloid β, prion, amylin or α-synuclein which can be used alone or conjugated to an immunostimulatory molecule in an immunizing composition for inducing an immune response to amyloid β peptides and amyloid deposits, to prion protein and prion deposits, to amylin and amylin deposits, to α-synuclein and deposits containing α-synuclein, or to polyglutamine repeats and deposits of proteins containing polyglutamine repeats. Described are also antibodies directed against such peptides, their generation, and their use in methods of passive immunization to such peptides and deposits.
Owner:NEW YORK UNIV
Soluble T cell receptor
InactiveUS20020142389A1High affinityStrong specificityBacteriaPeptide/protein ingredientsHeterologousExtracellular Structure
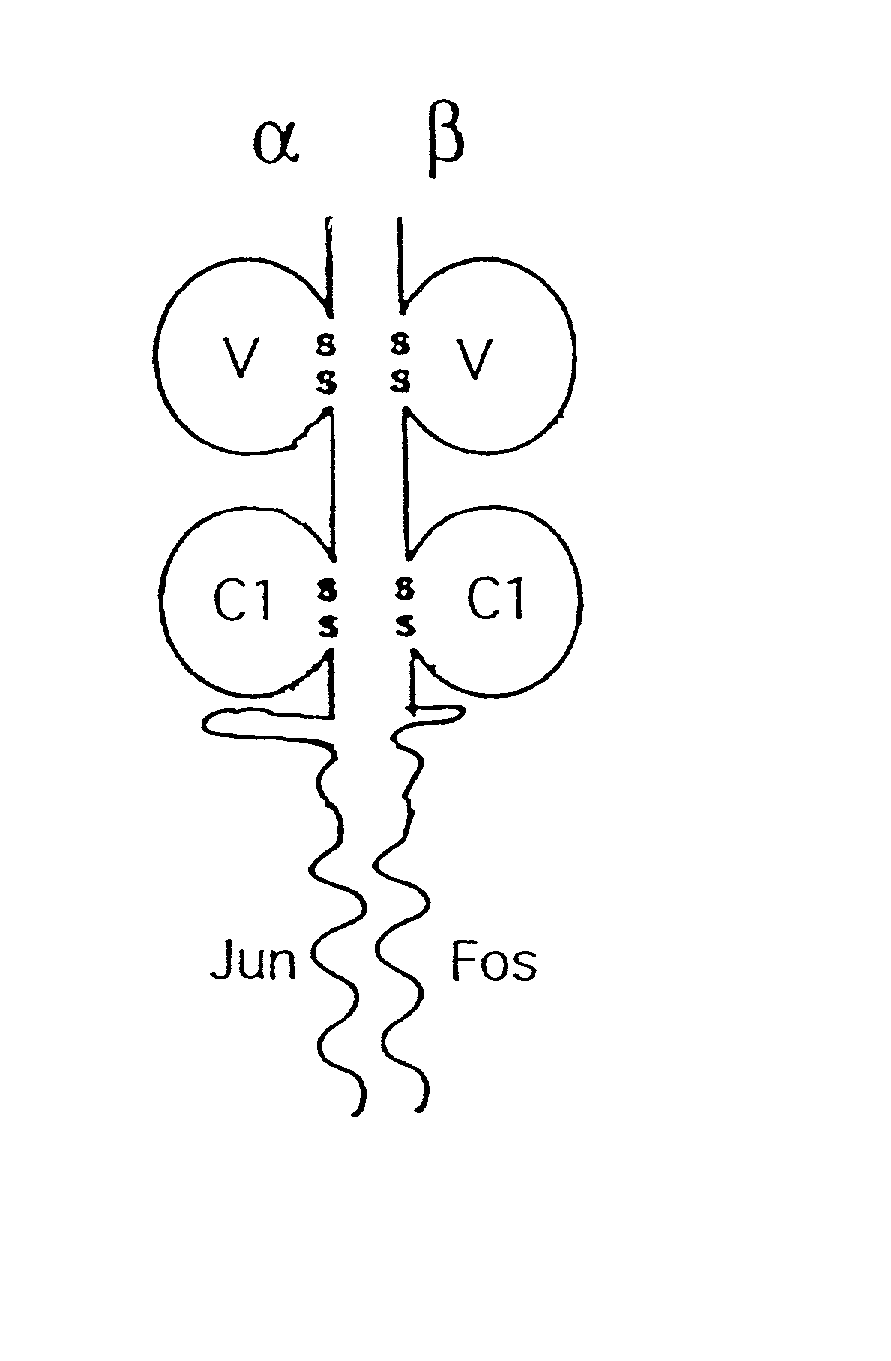
The present invention relates to a recombinant soluble T cell receptor. The T cell receptor (TCR) is refolded and comprises a recombinant TCR alpha or gamma chain extracellular domain having a first heterologous C-terminal dimerisation peptide; and a recombinant TCR beta or delta chain extracellular domain having a second C-terminal dimerisation peptide which is specifically heterodimerised with the first dimerisation peptide to form a heterodimerisation domain, which may be a coiled coil domain. The invention also provides nucleic acid sequences encoding the recombinant TCR and a method for producing the recombinant TCR. The TCR may be labelled with a detectable label so as to enable the detection of specific MHC-peptide complexes. Alternatively, it can be linked to a therapeutic agent such as a cytotoxic agent or an immunostimulating agent so as to deliver such an agent to the site of a specific MHC-peptide complex.
Owner:JAKOBSEN BENT KARSTEN +4
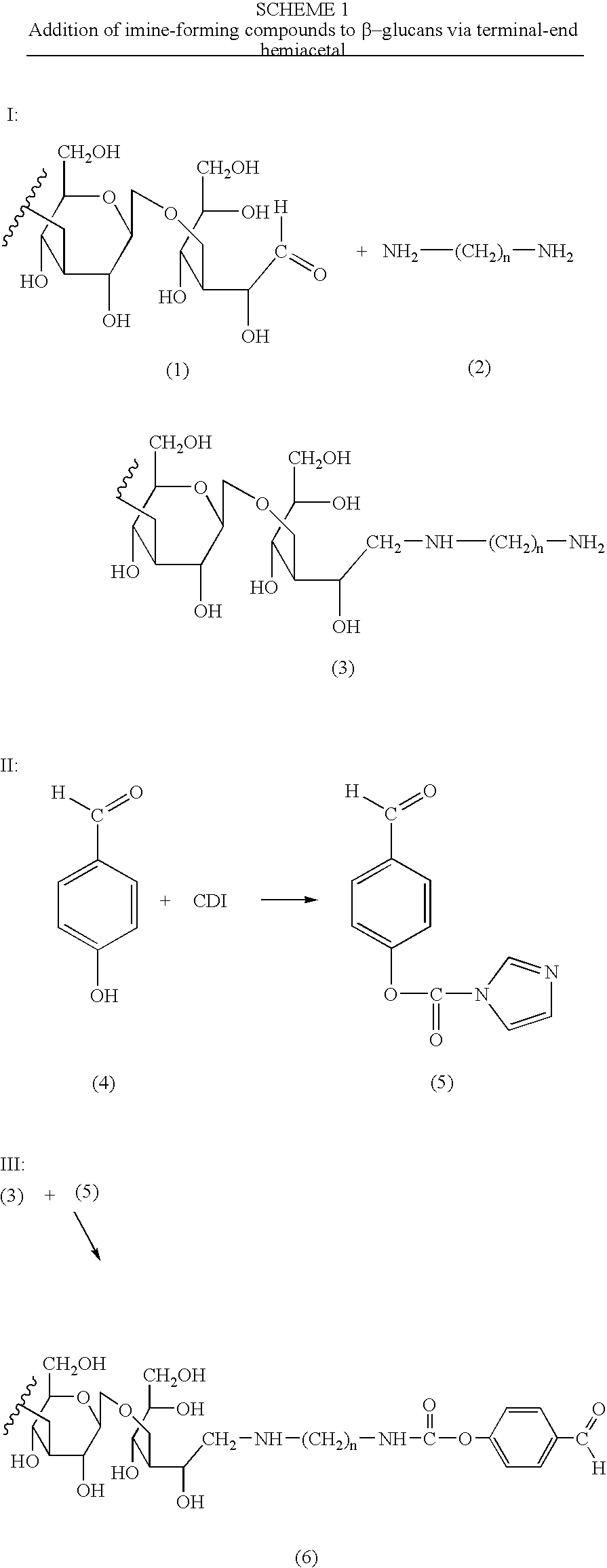
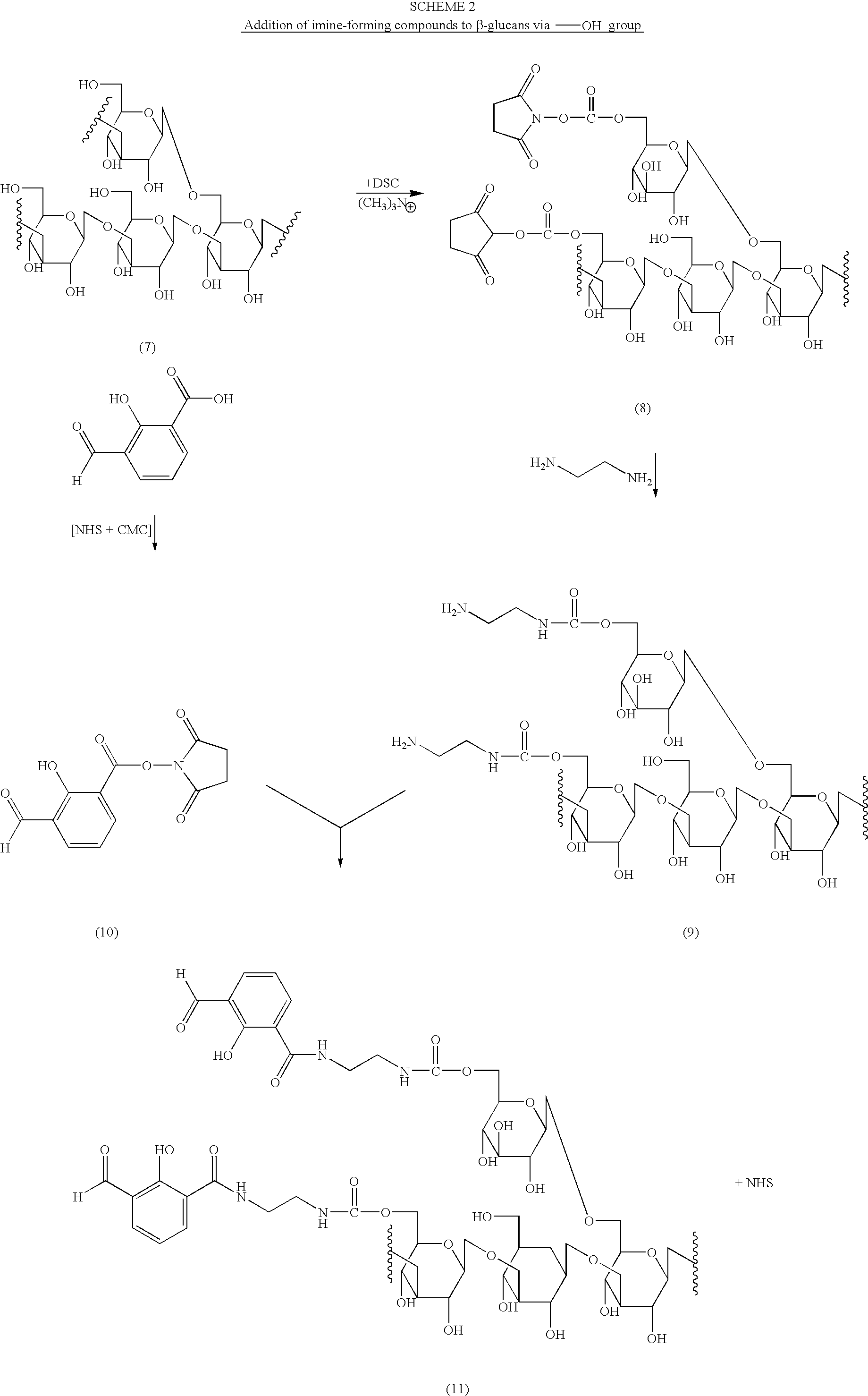
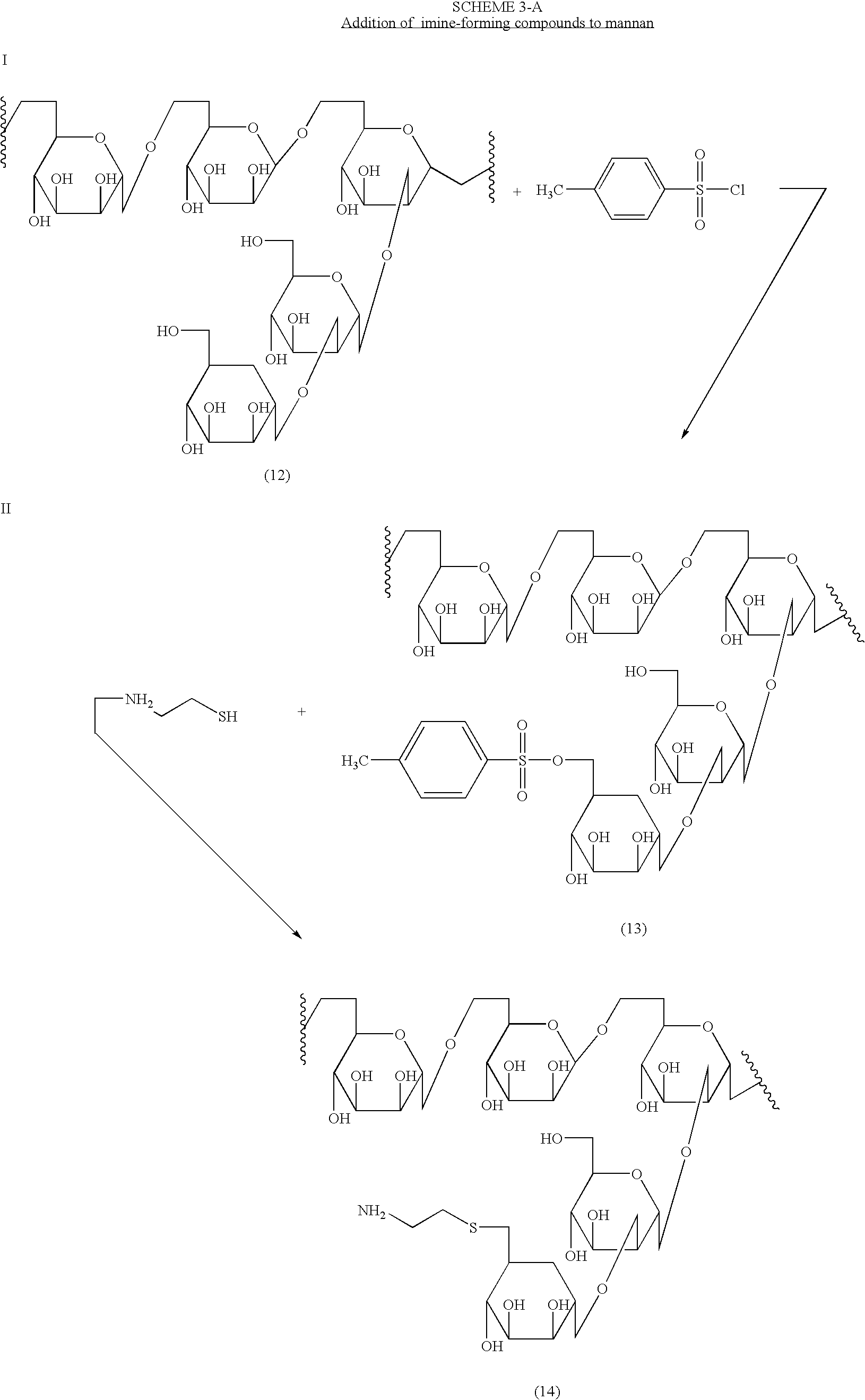
Imine-forming polysaccharide adjuvants and immunostimulants
InactiveUS7196073B2Improve acceleration performanceEnhance immune responseOrganic active ingredientsBiocideAdjuvantReceptor
The present invention relates to polysaccharide conjugates that comprise: a polysaccharide that binds to surface-receptors present on Antigen Presenting Cells, conjugated to one or more compounds having stable carbonyl groups covalently attached, either directly or via a bifunctional linker. The conjugates are useful as immuno-stimulants and adjuvants.
Owner:GALENICA PHARMA
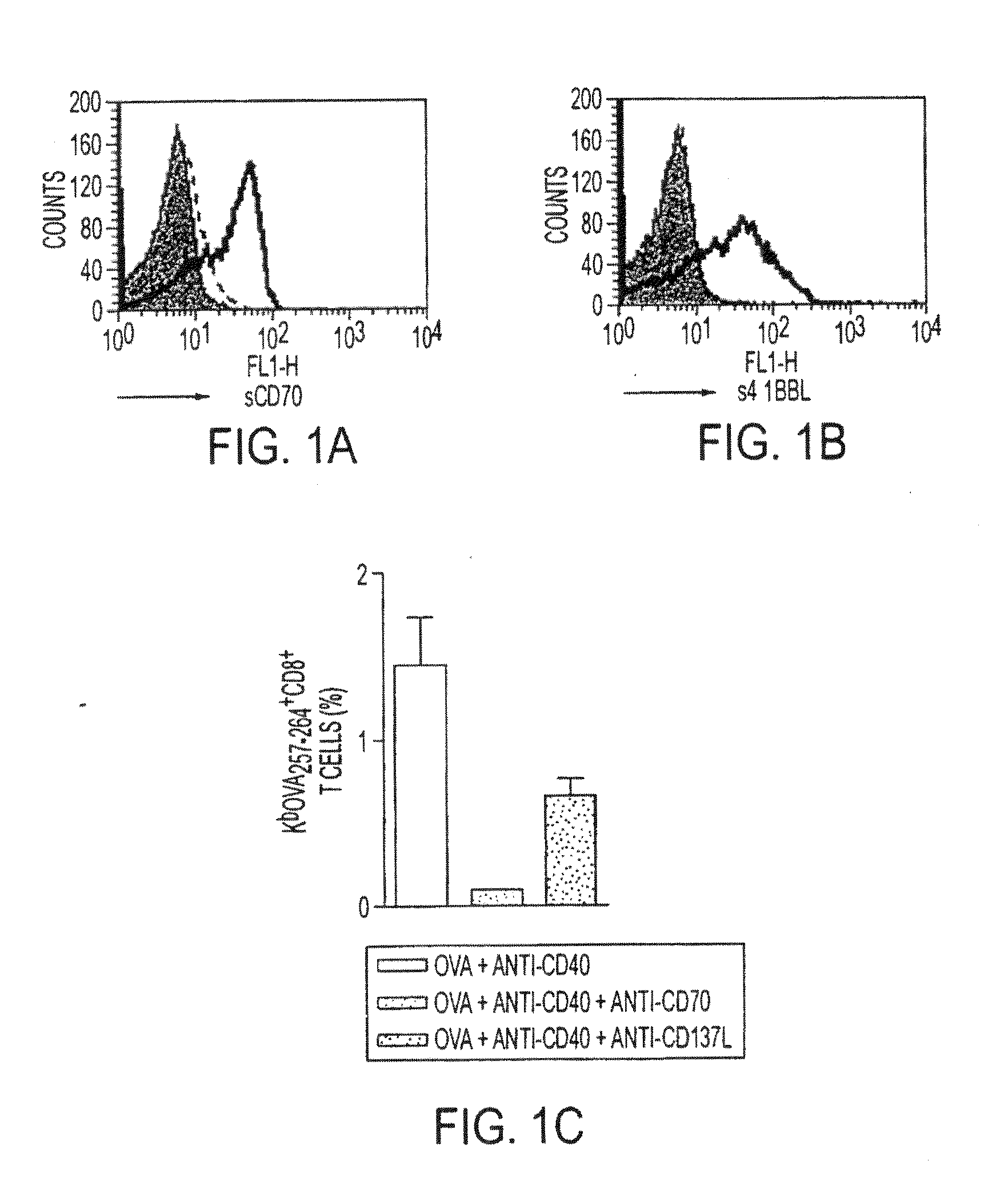
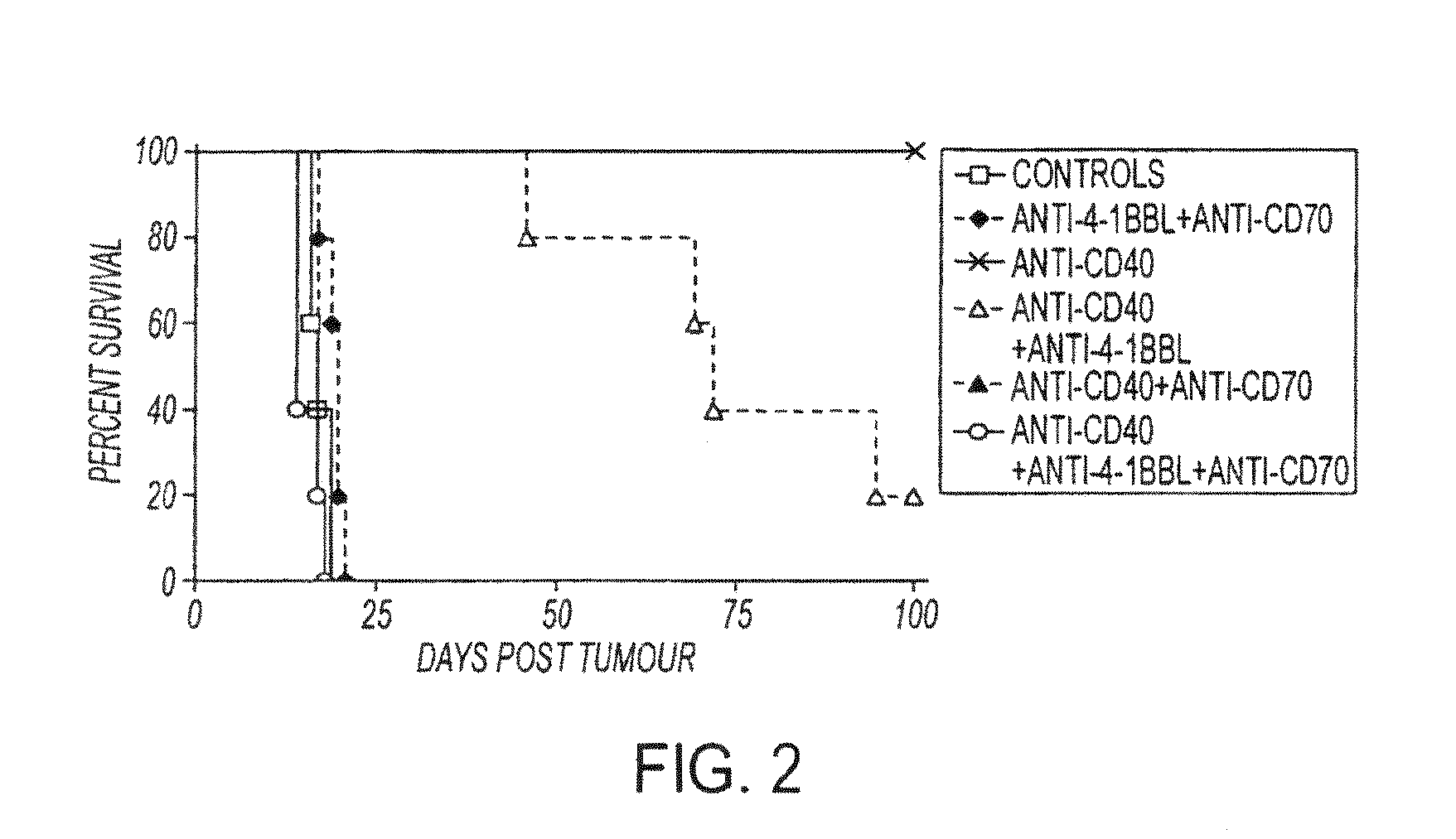
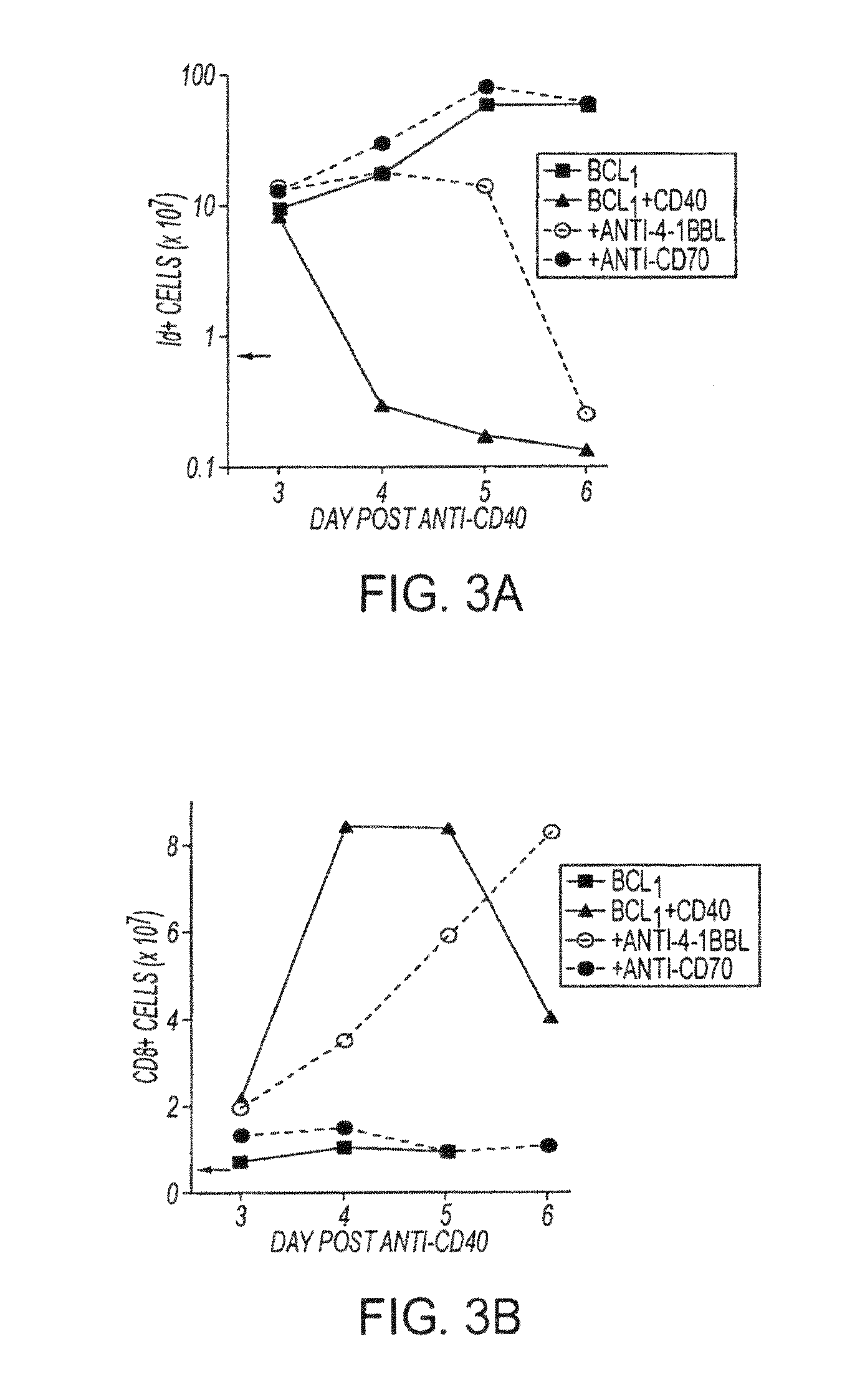
Human immune therapies using a cd27 agonist alone or in combination with other immune modulators
InactiveUS20130336976A1Promotes strong expression of 4-1BBImprove responseAntibacterial agentsAntimycoticsIMMUNE STIMULANTSCD8
Methods of inducing T cell proliferation and expansion in vivo for treating conditions wherein antigen-specific T cell immune response are therapeutically desirable such as cancer, infection, inflammation, allergy and autoimmunity and for enhancing the efficacy of vaccines are provided. These methods comprise the administration of at least one CD27 agonist, preferably an agonistic CD27 antibody, alone or in association with another moiety such as immune stimulant or immune modulator such as an anti-CD40, OX-40, 4-1BB, or CTLA-4 antibody or an agent that depletes regulatory cells, or a cytokine. These mono and combination therapies may also optionally include the administration of a desired antigen such as a tumor antigen, an allergen, an autoantigen, or an antigen specific to an infectious agent or pathogen against which a T cell response (often CD8+) is desirably elicited.
Owner:UNIV OF SOUTHAMPTON
Melanoma vaccine and methods of making and using same
Owner:WALLACK MARC K
Coordinate in vivo gene expression
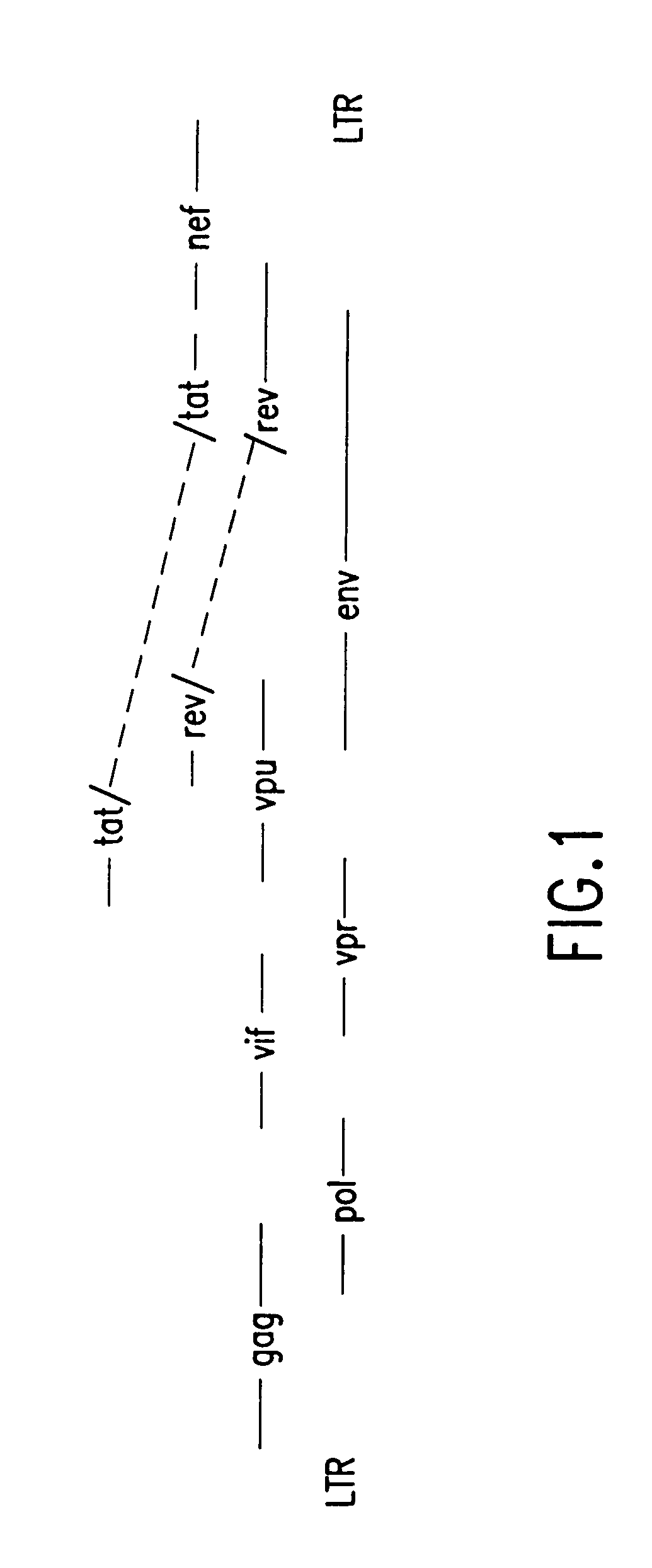
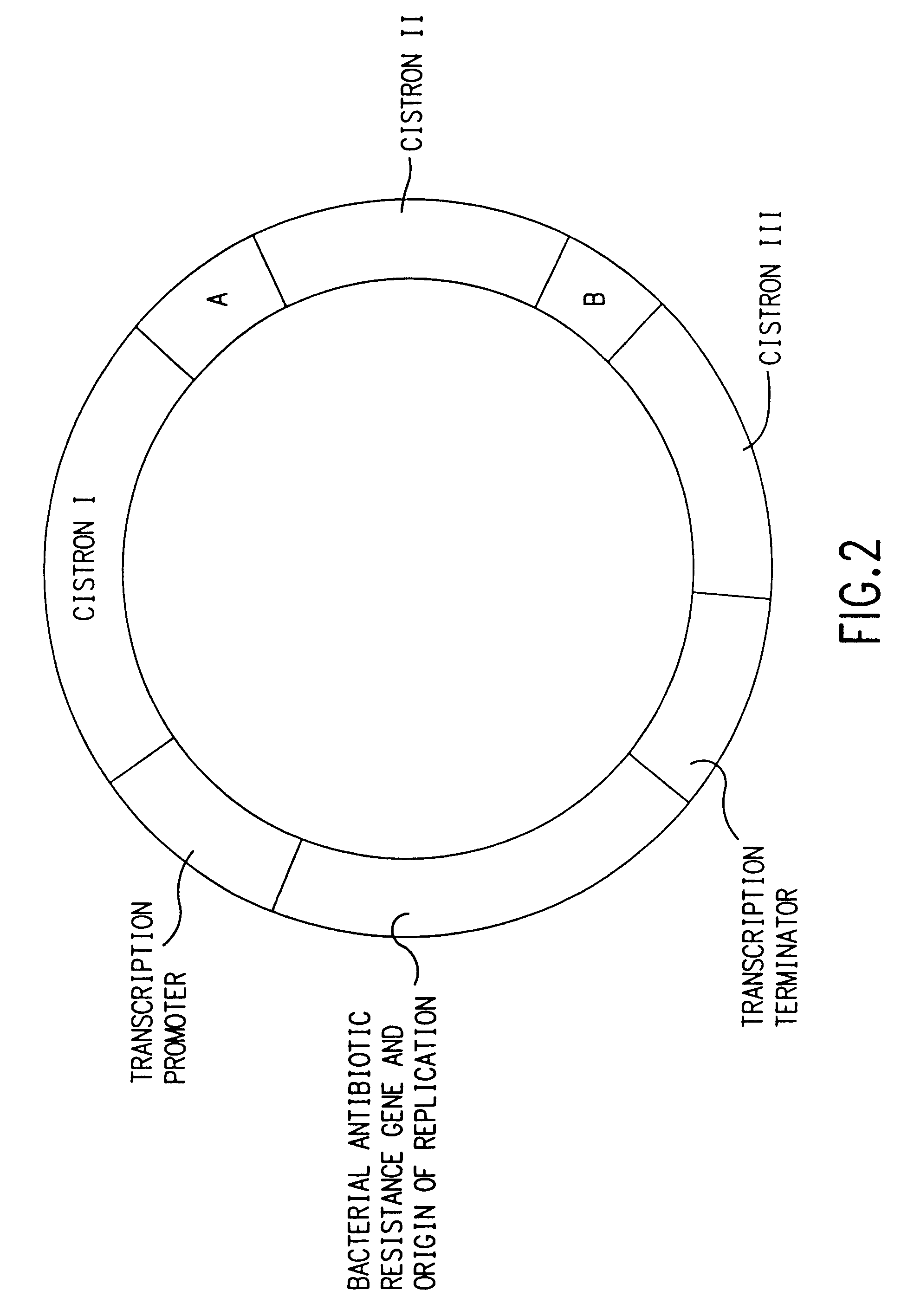
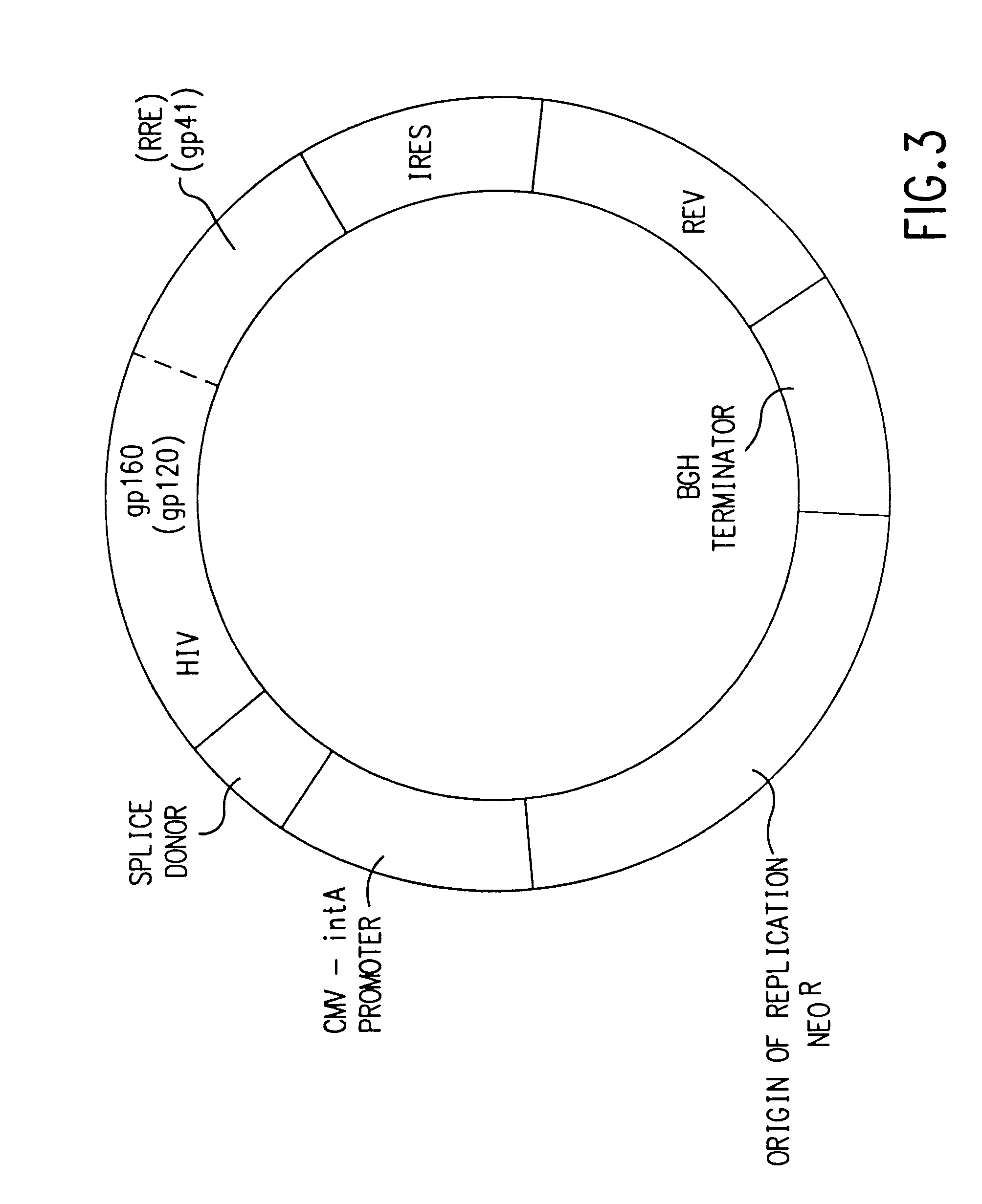
Nucleic acids, including DNA constructs and RNA transcripts, capable of inducing coordinate expression of two to three cistrons upon direct introduction into animal tissues, are bi- or tri-cistronic polynucleotides of this invention include those encoding and co-expressing HIV gene products, genes encoding antigens unrelated to HIV, and immunostimulatory gene products, including but not limited to GM-CSF, interleukins, interferon and members of the B7 family of proteins which act as T-cell costimulatory elements. The methods and polynucleotides of this invention are generally applicable to co-ordinate expression in vivo of any two or more genes in a single cell.
Owner:MERCK & CO INC
Vectors and method for genetic immunization
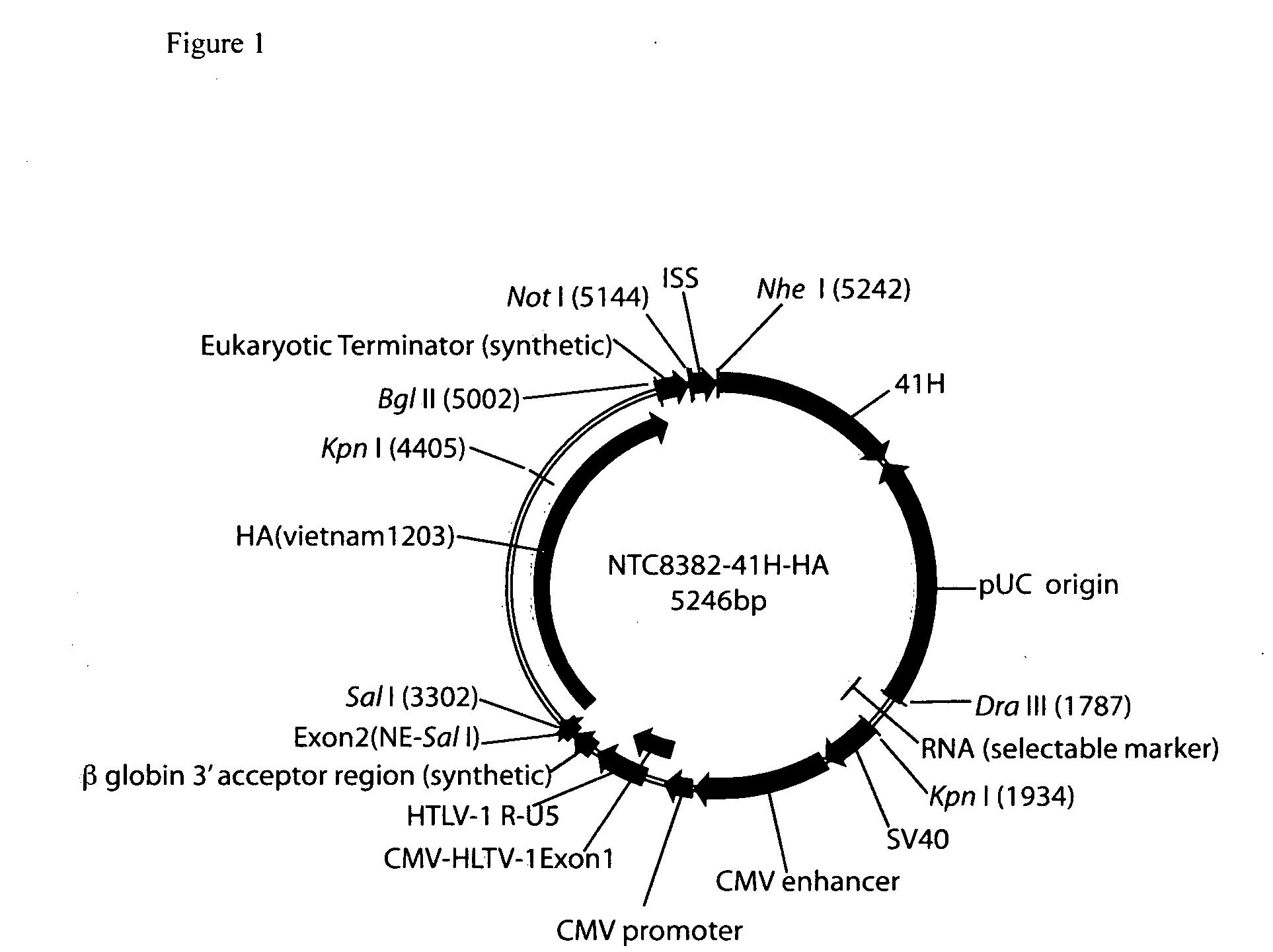
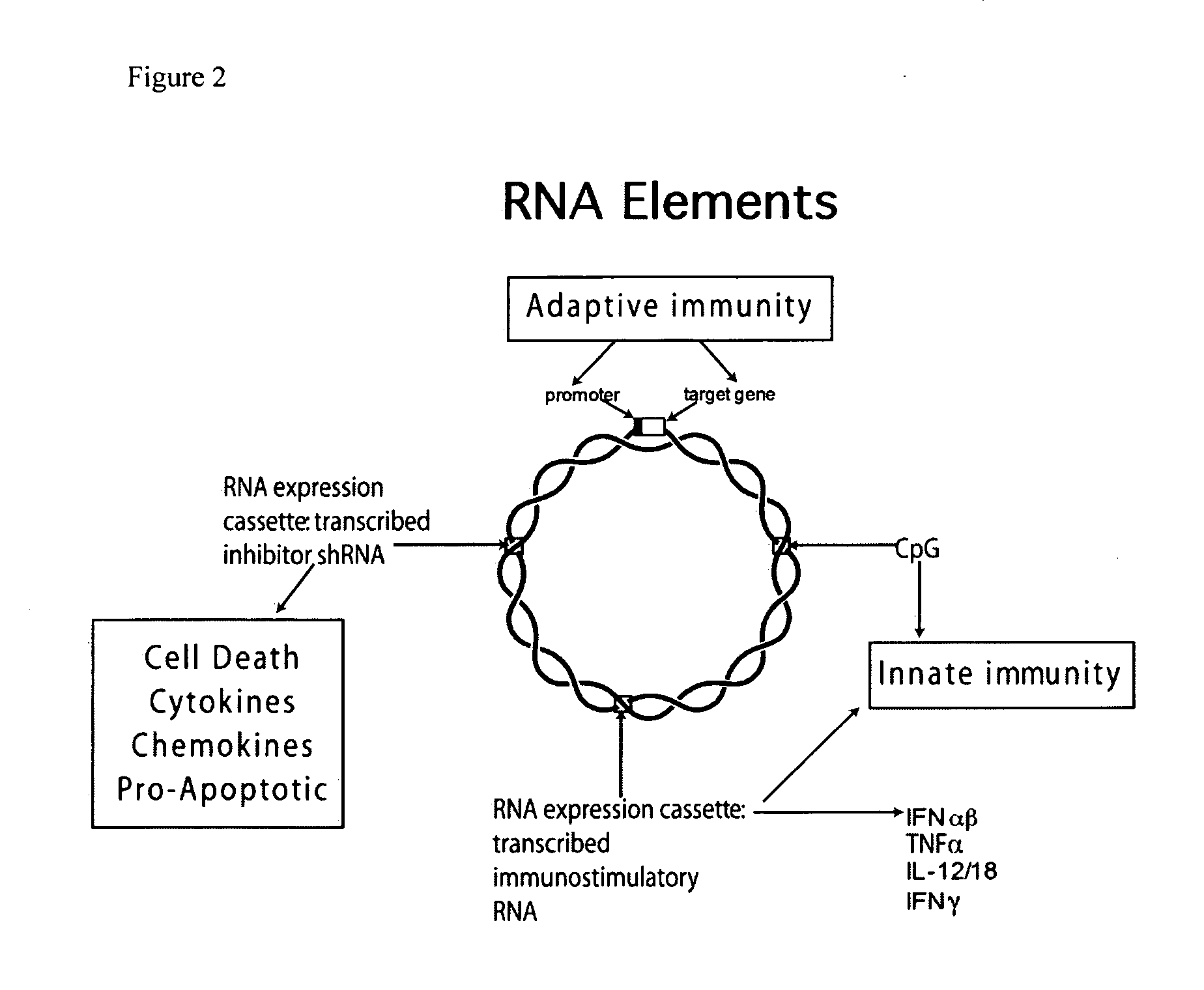
ActiveUS20100303859A1Small sizeImprove complianceSsRNA viruses negative-senseActivity regulationAntibiotic freeType 1 interferon
Improved DNA vaccine plasmids are disclosed that contain novel immunostimulatory RNA compositions. The improved plasmids eliminate all extraneous sequences, incorporate a novel antibiotic free short RNA based selectable marker, increase eukaryotic expression using a novel chimeric promoter, improve yield and stability during bacterial production, and improve immunostimulation. These vectors are utilized in immunization to elicit improved immune responses or therapy to induce type 1 interferon production.
Owner:ALDEVRON LLC
Immunostimulant compositions comprising an aminoalkyl glucosaminide phosphate and QS-21
The invention provides pharmaceutical compositions, particularly vaccine compositions, employing an adjuvant system comprising RC-529 (an aminoalkyl glucosaminide phosphate compound) and QS-21 (a saponin). Such compositions synergistically enhance the immune response in a mammal to a co-administered antigen. Also provided are methods of using the compositions in the treatment of various human diseases, including cancer, microbial infections and autoimmune disorders.
Owner:ANTIGENICS
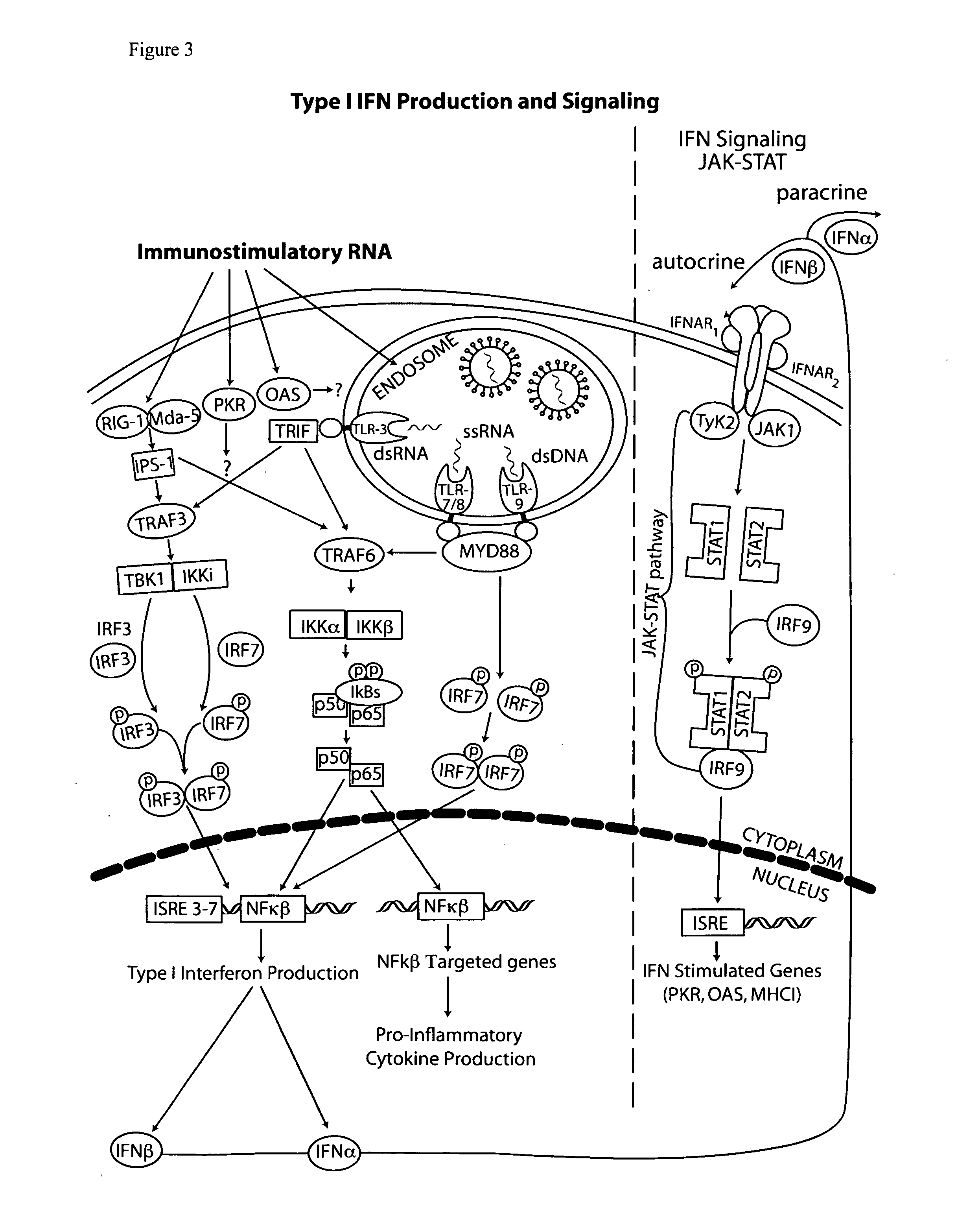
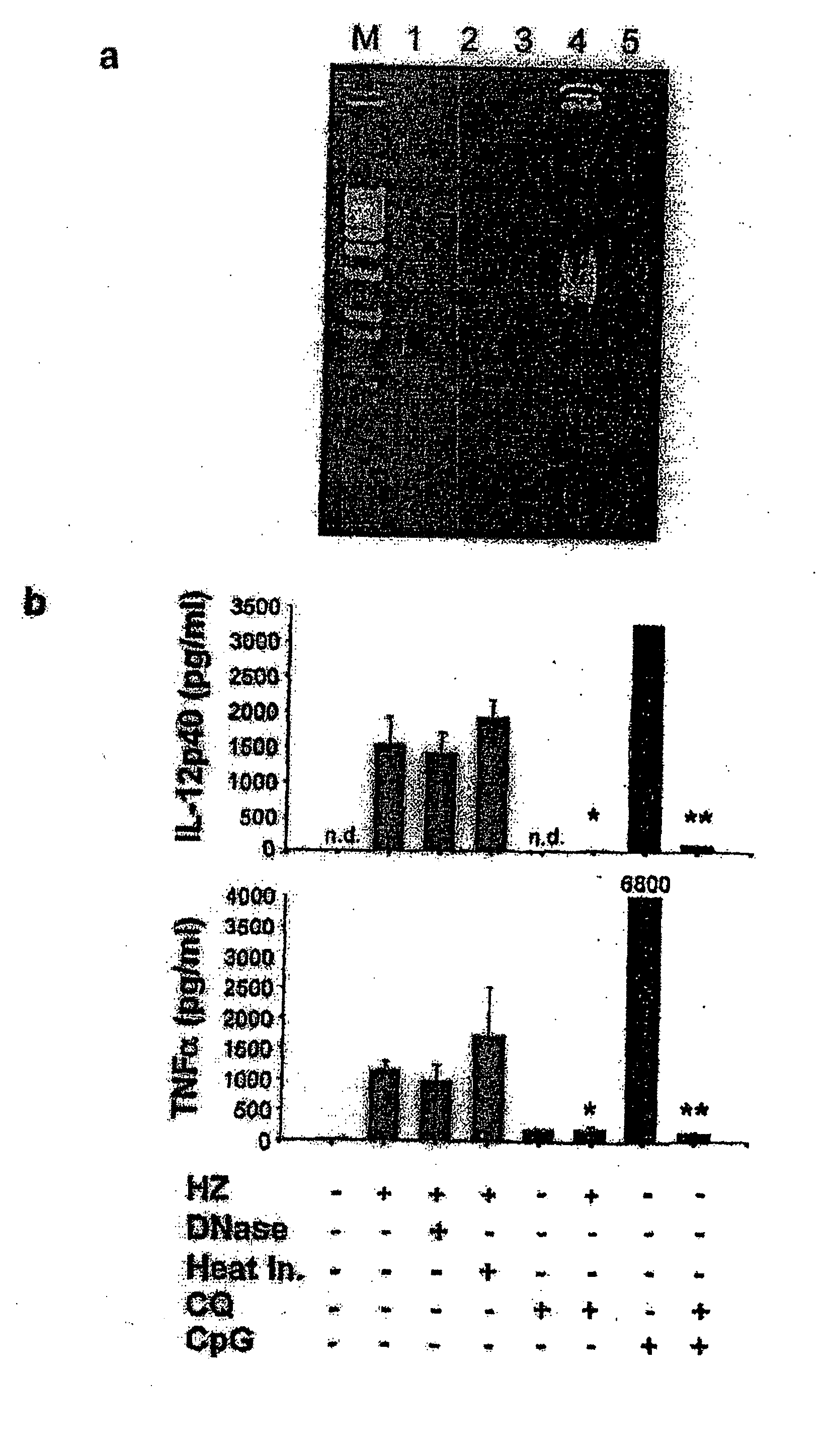
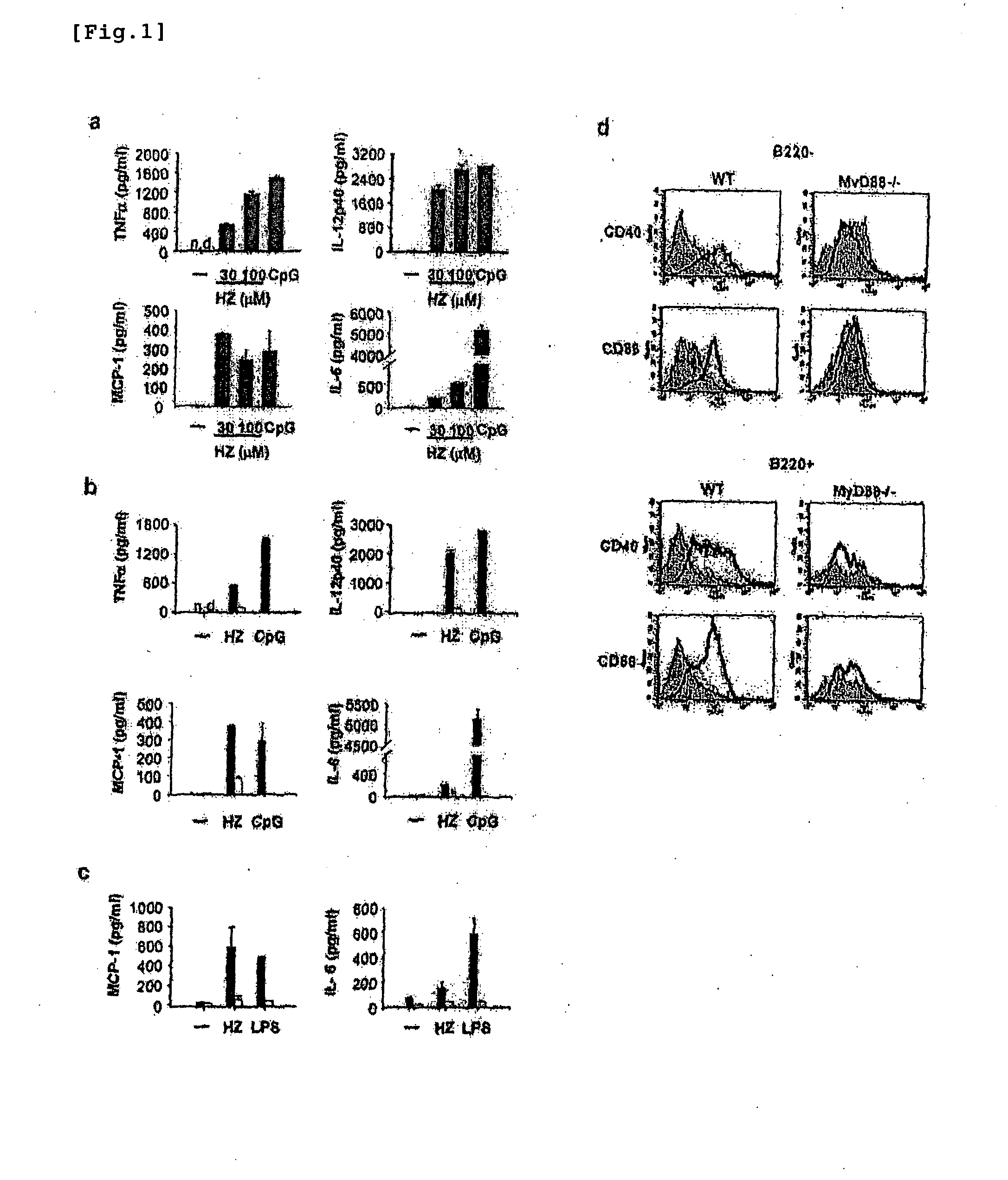
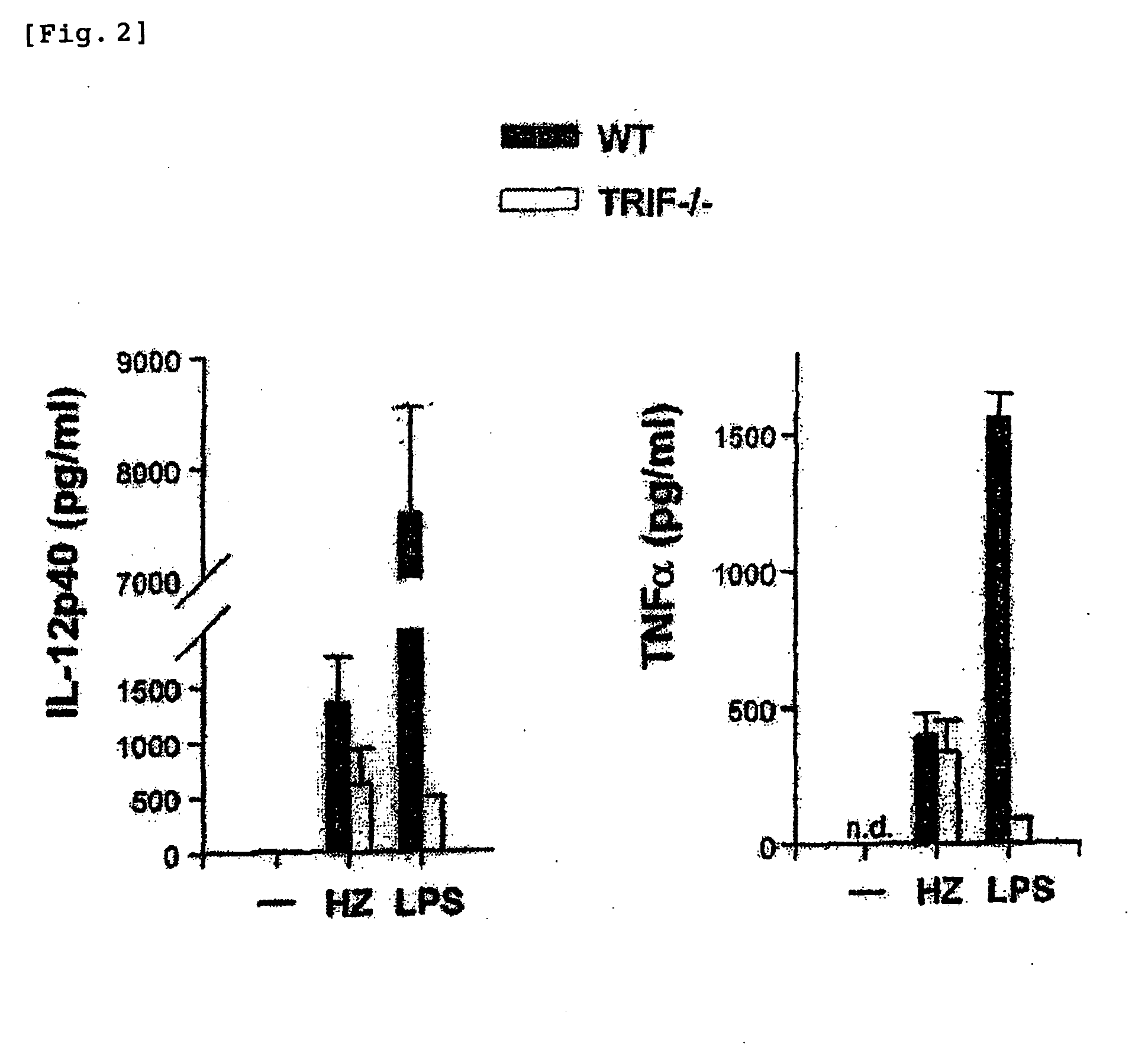
Detection/Measurement Of Malaria Infection Disease Utilizing Natural Immunity By Hemozoin Induction, Screening Of Preventative Or Therapeutic Medicine For Malaria Infection Disease, And Regulation Of Natural Immunity Induction
The instant invention is to provide a method for detecting and measuring malaria infection utilizing the induction by hemozoin (HZ); a method for screening a vaccine for malaria infection and a preventative or therapeutic agent for malaria infection using the method for detecting and measuring; and a means for regulating the induction of innate immunity using the HZ, synthetic HZ, or derivatives thereof as an adjuvant or immunostimulant. Malaria infection is detected and measured of by detecting and measuring HZ-induced, TLR9-mediated, and MyD88-dependent innate immune activity. The detection and measurement of malaria infection can be used to diagnose malaria infection. The method for detecting and measuring is also used for screening a vaccine for malaria infection and a preventative or therapeutic agent for malaria infection. Further, HZ, synthetic HZ, or derivatives thereof are used as an adjuvant or immunostimulant to regulate HZ-induced innate immune induction.
Owner:OSAKA UNIV
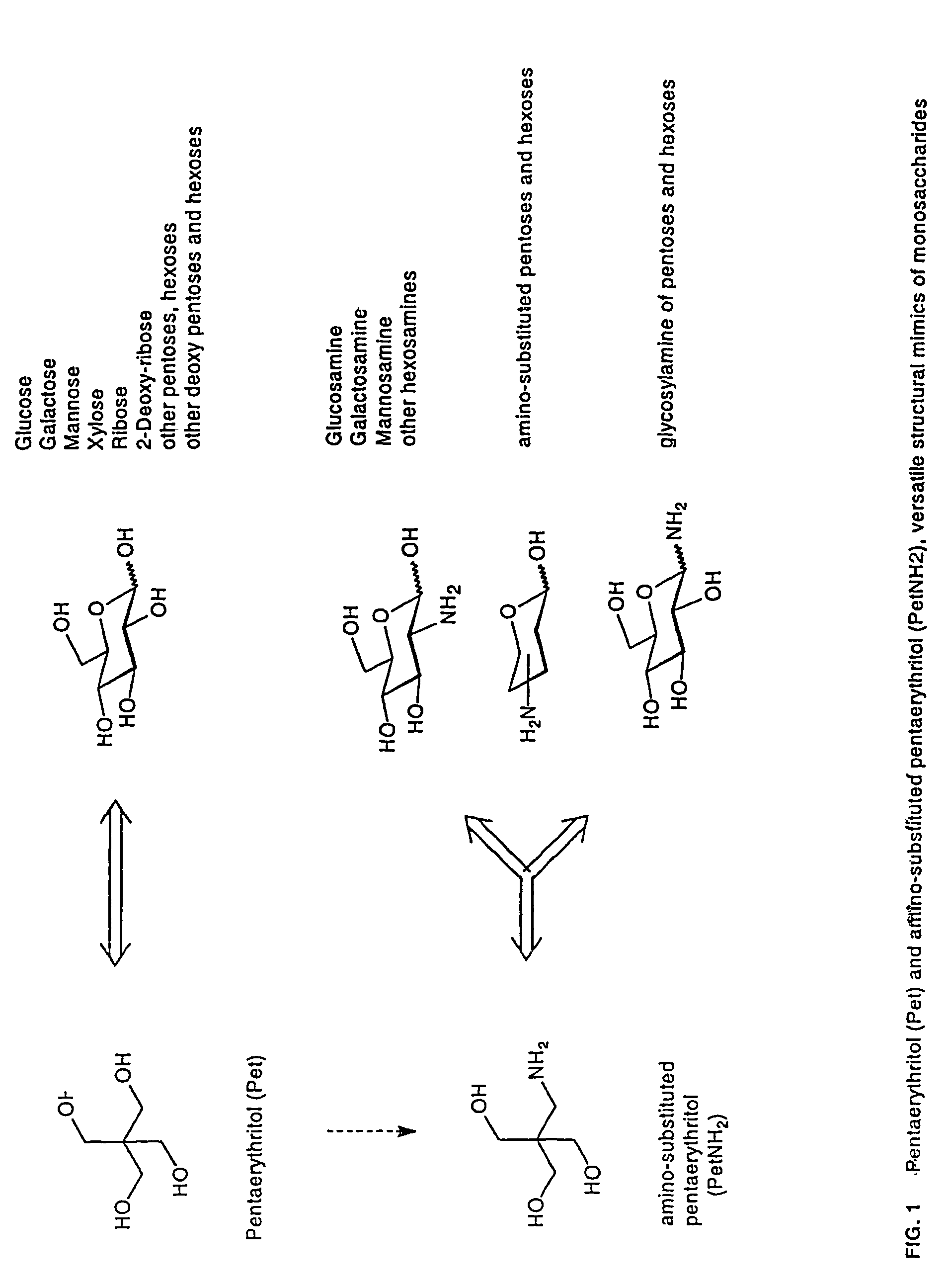
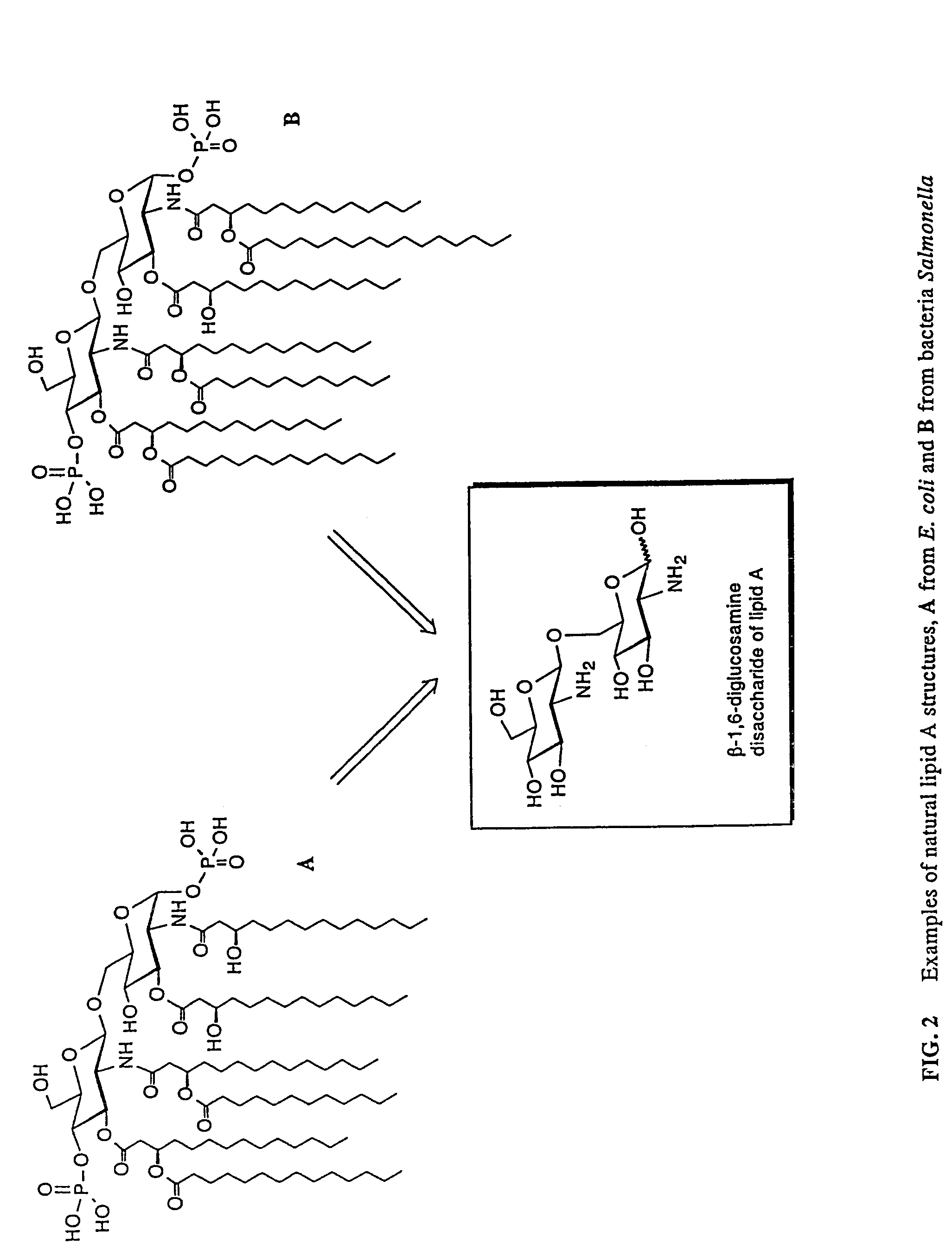
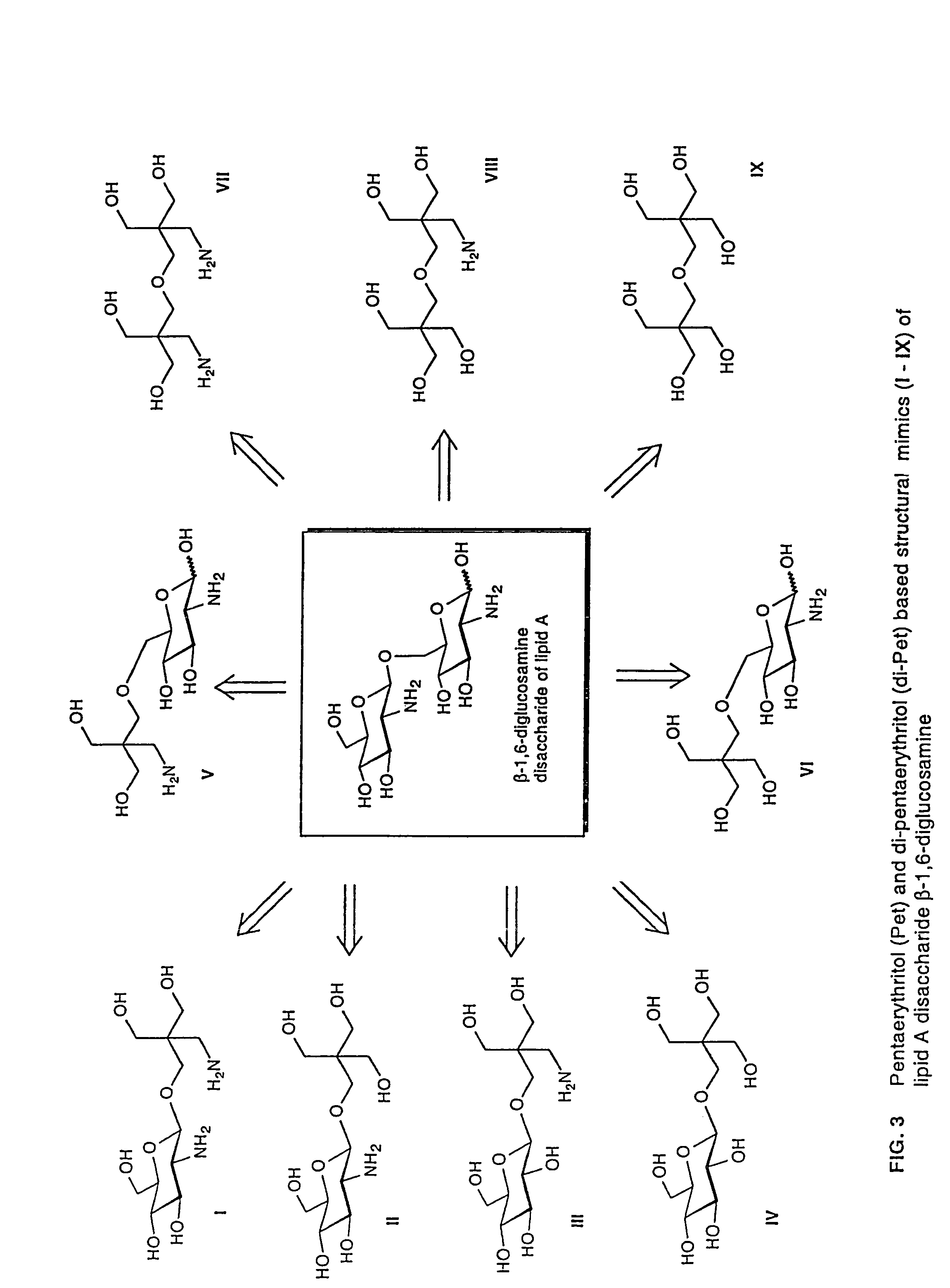
Lipid A and other carbohydrate ligand analogs
Owner:ONCOTHYREON
Immunostimulatory compositions and methods
The invention provides conjugates comprising an immune co-stimulatory polypeptide and an antigen or infectious agent. The conjugates are useful for generating or enhancing an immune response against the antigen or infectious agent. The invention also provides immune cells modified with a conjuagte that are useful for generating or enhancing an immune response to an antigen or infectious agent. The invention also provides immunostimulatory moieties comprising an immune co-stimulatory polypeptide that are useful for stimulating an immune response. The invention also provides immunotherapy methods and methods of treating or preventing infections.
Owner:UNIV OF LOUISVILLE RES FOUND INC +1
Humanized immunomodulatory monoclonal antibodies for the treatment of neoplastic disease or immunodeficiency
InactiveUS20050180969A1Induces proliferation and activationAvoids adverse immunogenic responseAnimal cellsHybrid immunoglobulinsDiseaseAntiendomysial antibodies
The present invention provides to a humanized monoclonal antibody having immunostimulatory effects. This antibody binds specifically to B lymphoblastoid cells, induces proliferation and activation of peripheral blood lymphocytes, and is capable of eliciting an anti-tumor effect upon administration to subjects suffering from cancer.
Owner:CURETECH LTD +1
Potent immunostimulatory extracts from microalgae
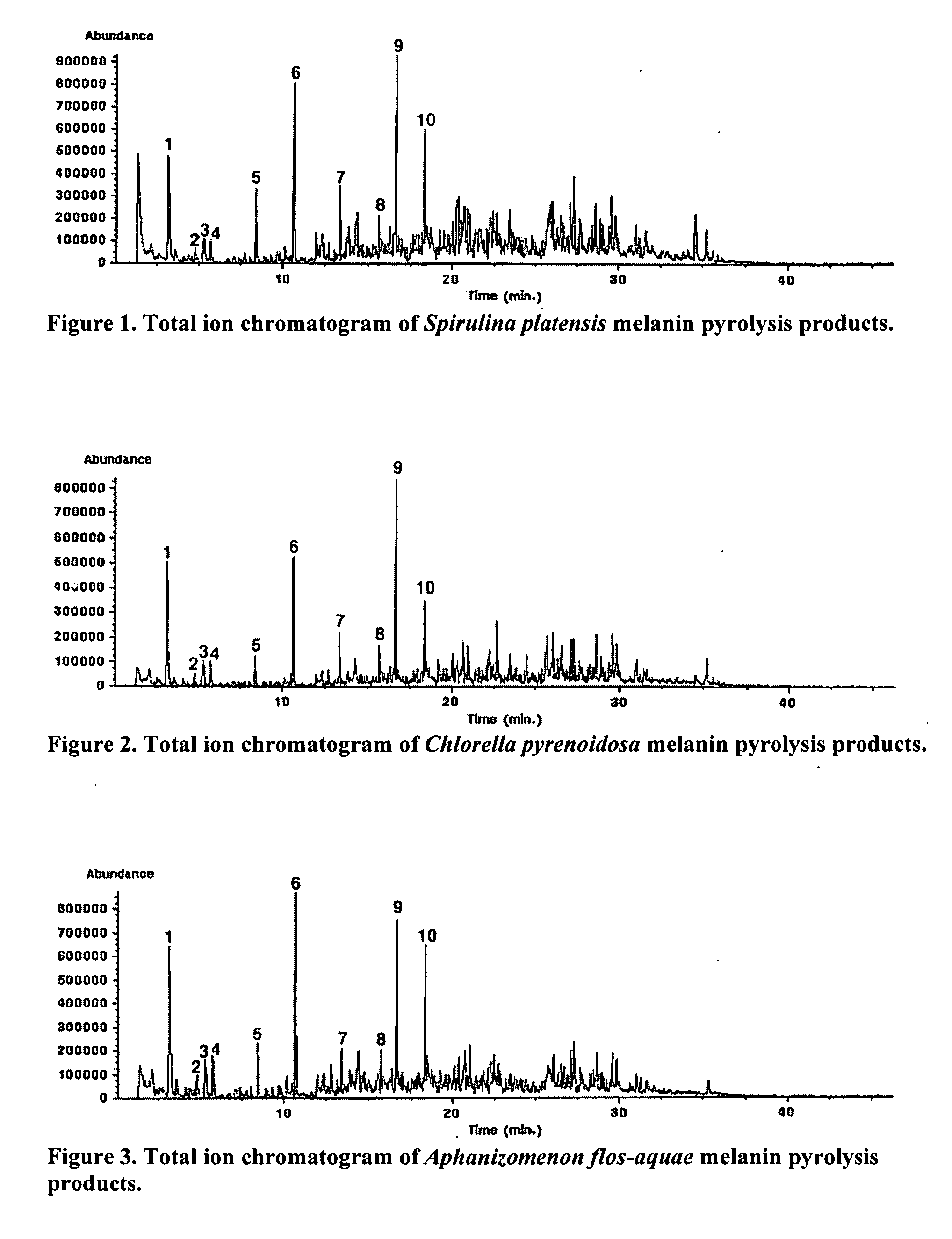
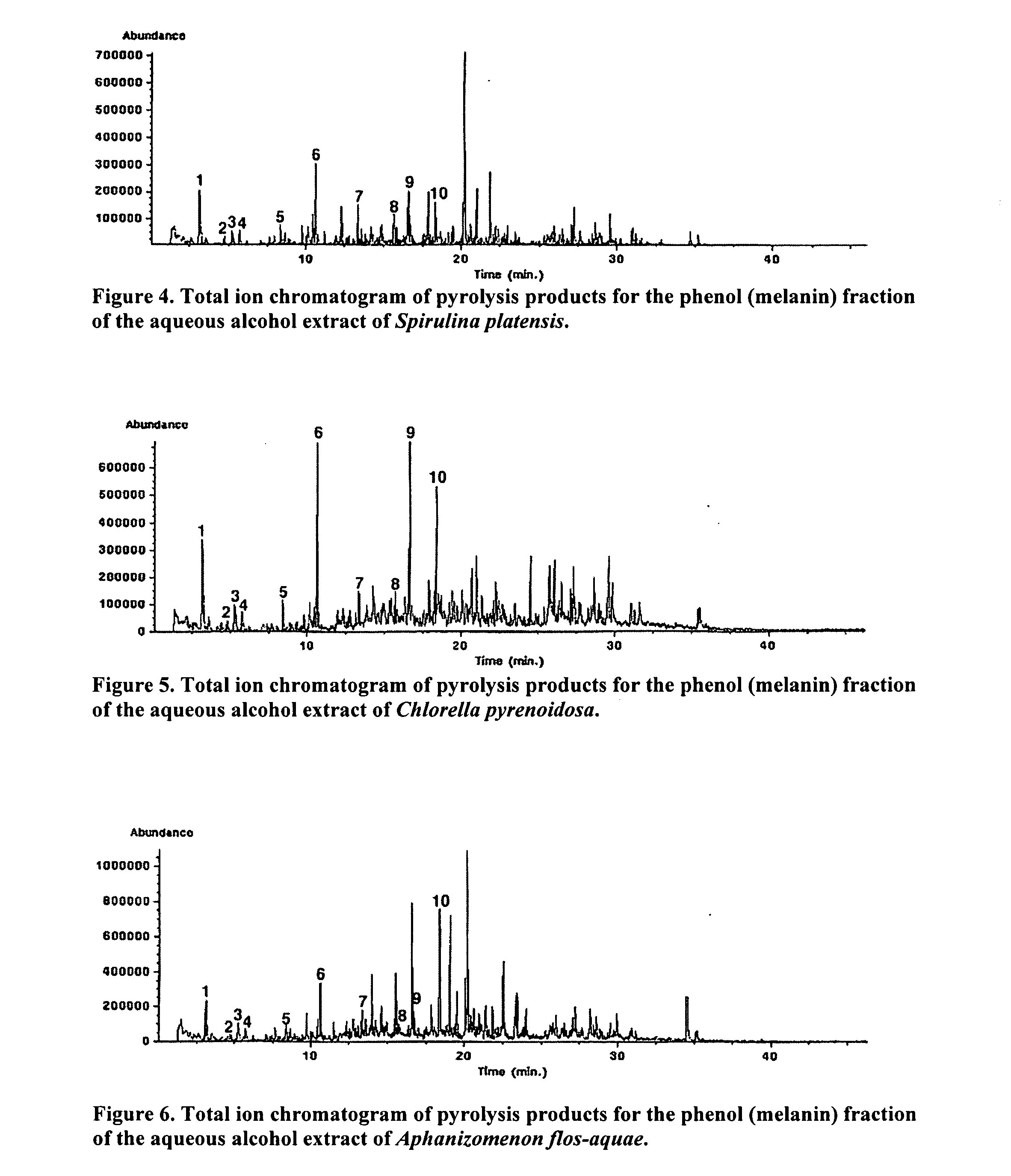
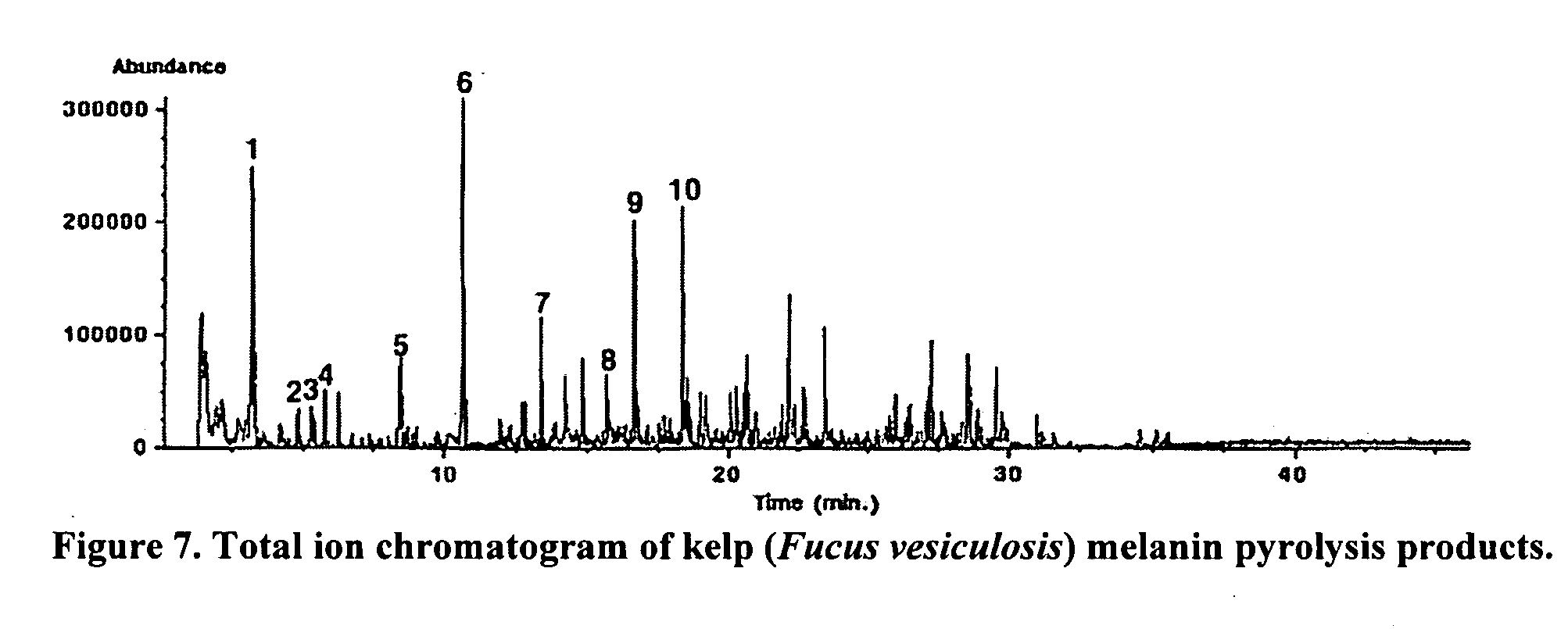
ActiveUS20060024328A1Enhance immune functionPromote wound healingBiocideAlgae medical ingredientsSolventAlgae
Immunostimulatory compounds can be extracted from microalgae or algae using various procedures. The resulting preparations exhibit extremely potent immunostimulatory activity. The extraction of these immunostimulatory agents is dependent on the solvent used and the extraction temperature. These preparations are potentially useful as a botanical or pharmaceutical preparation to improve immune function.
Owner:UNIVERSITY OF MISSISSIPPI
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com