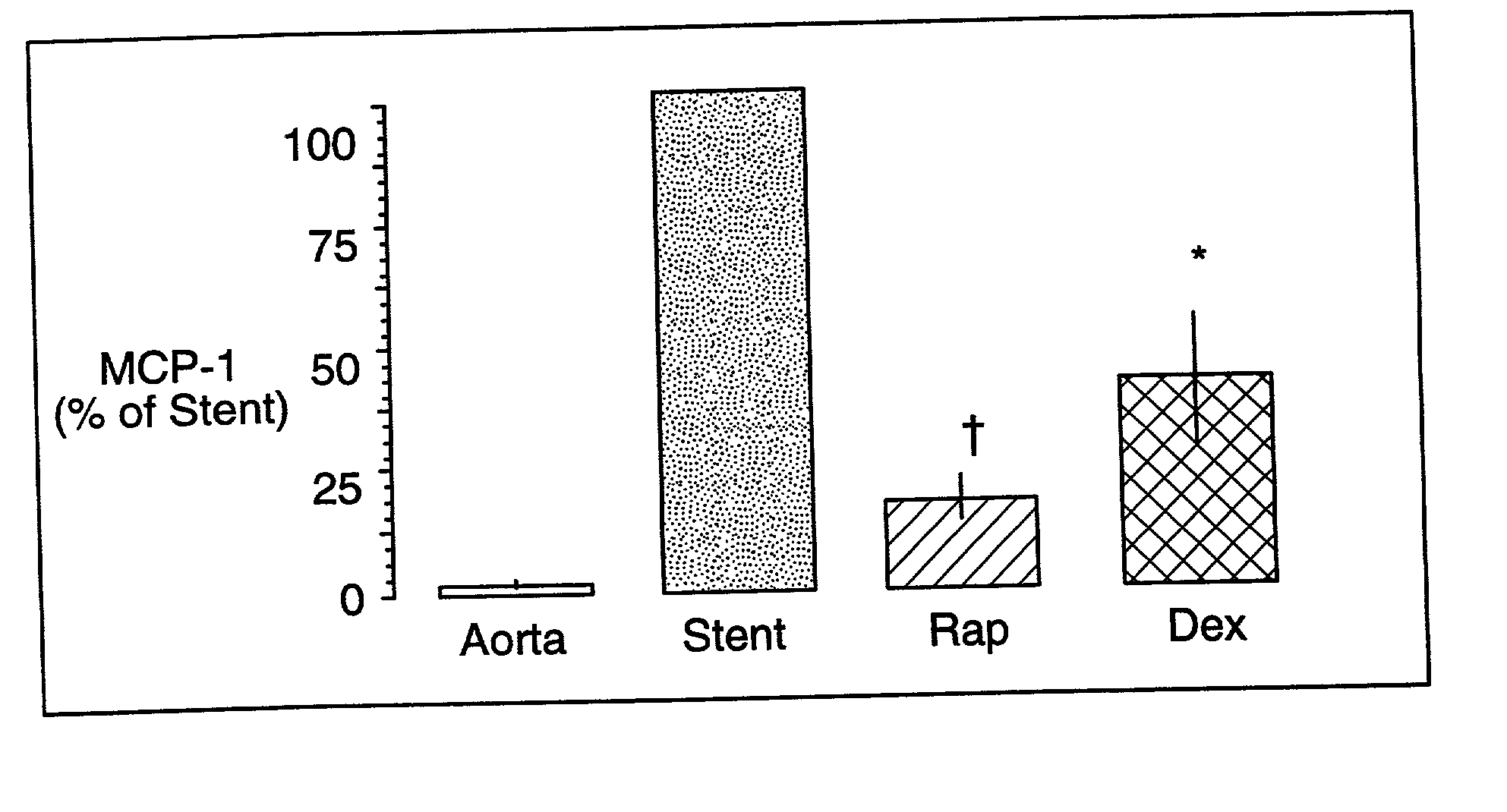
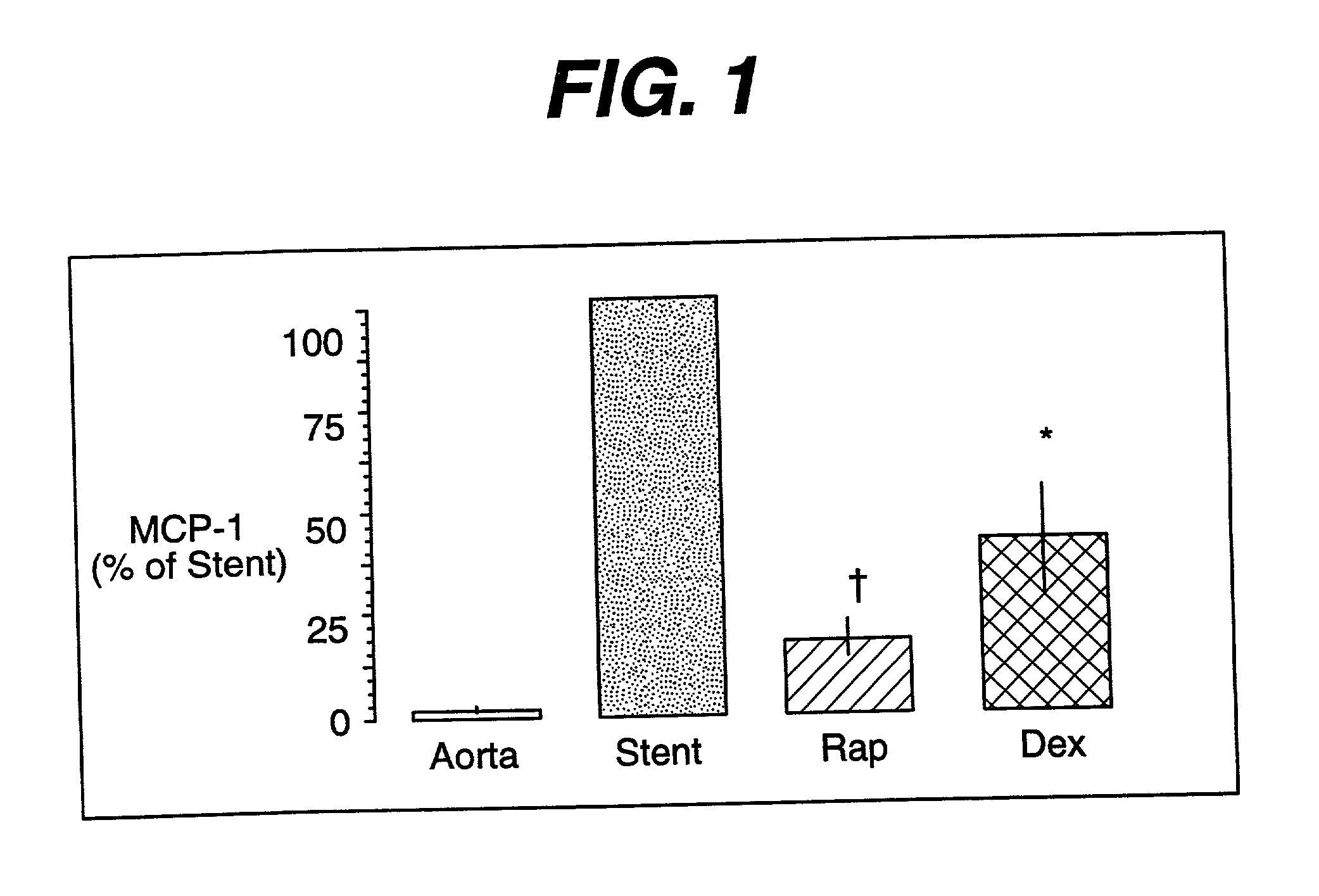
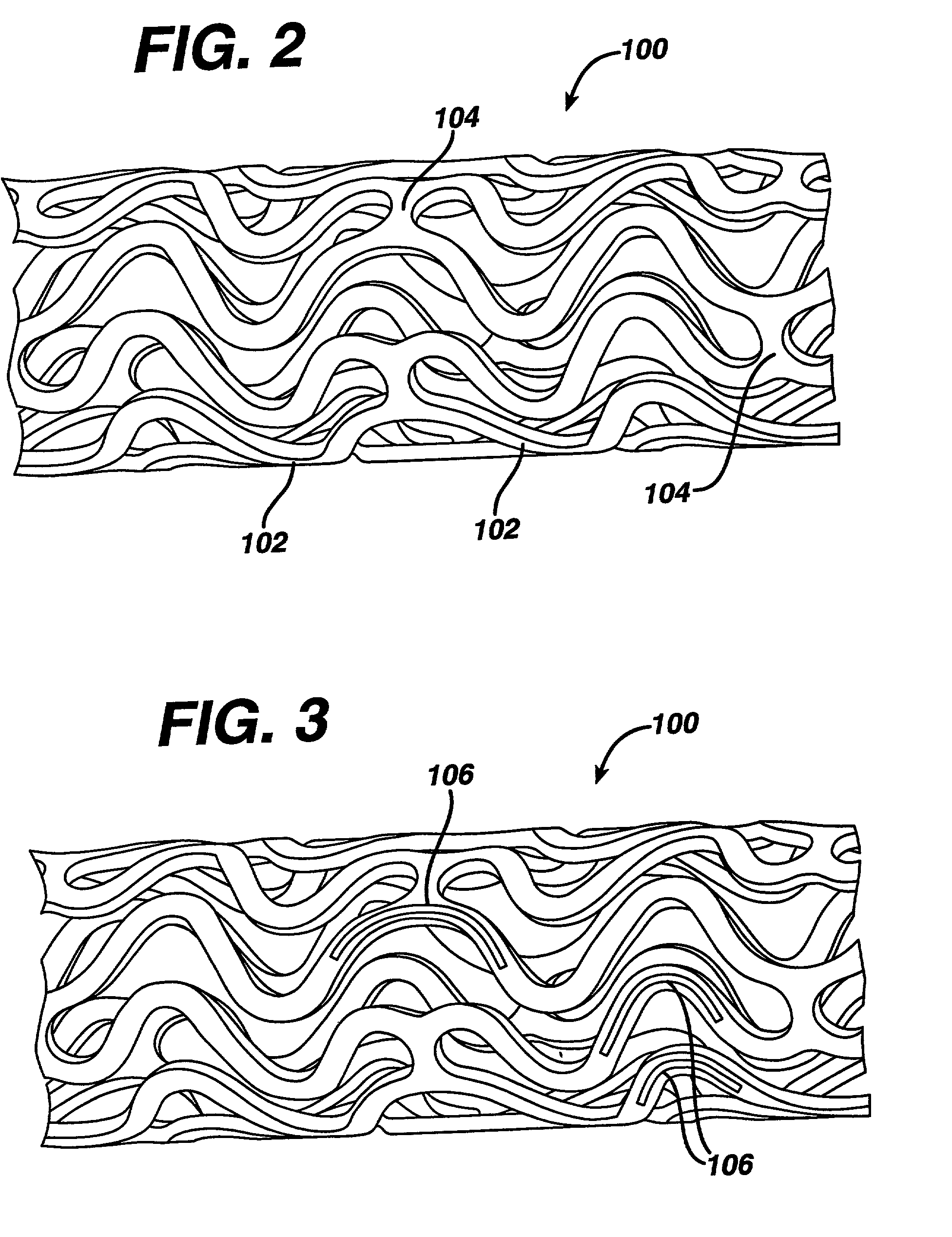
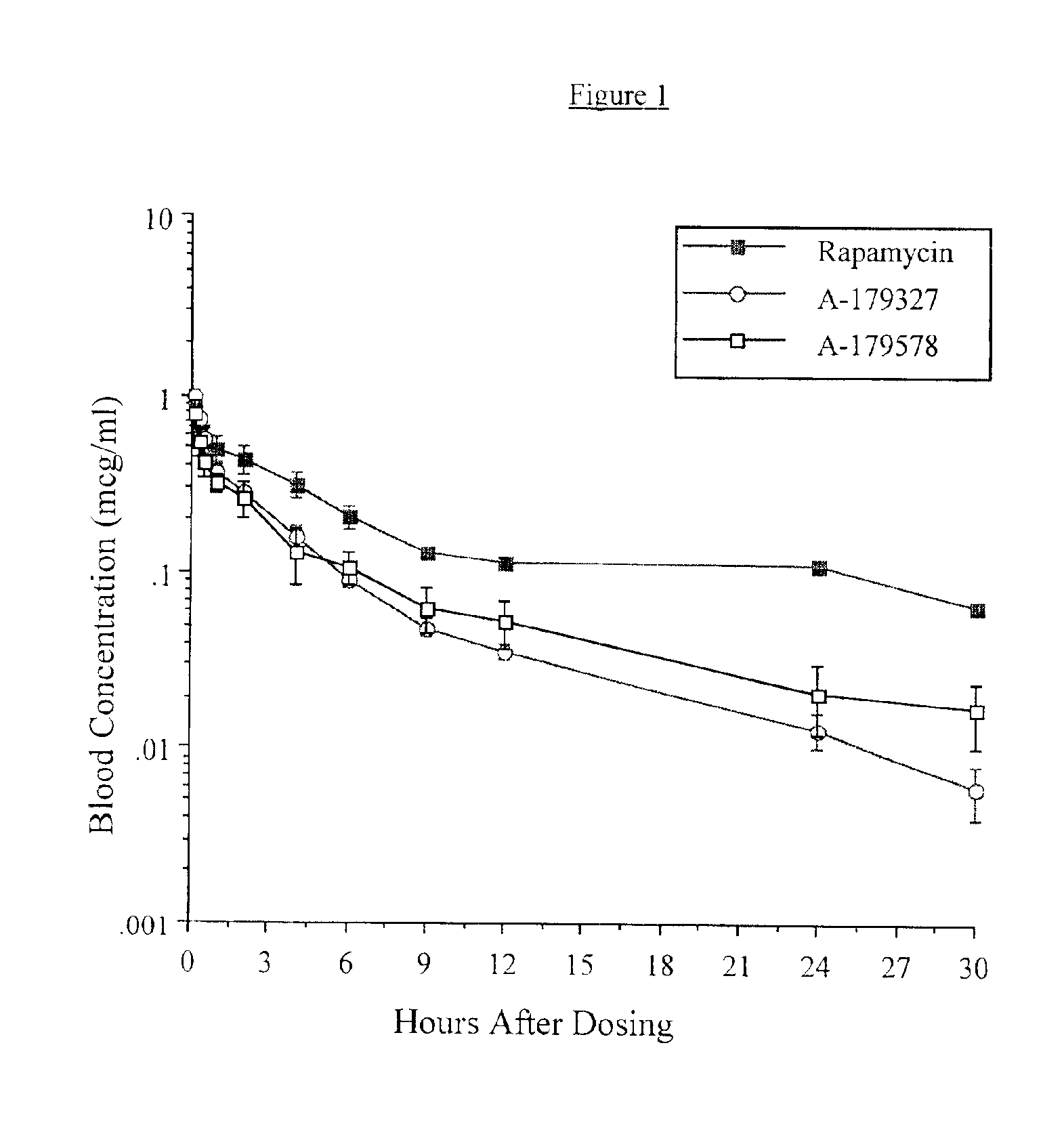
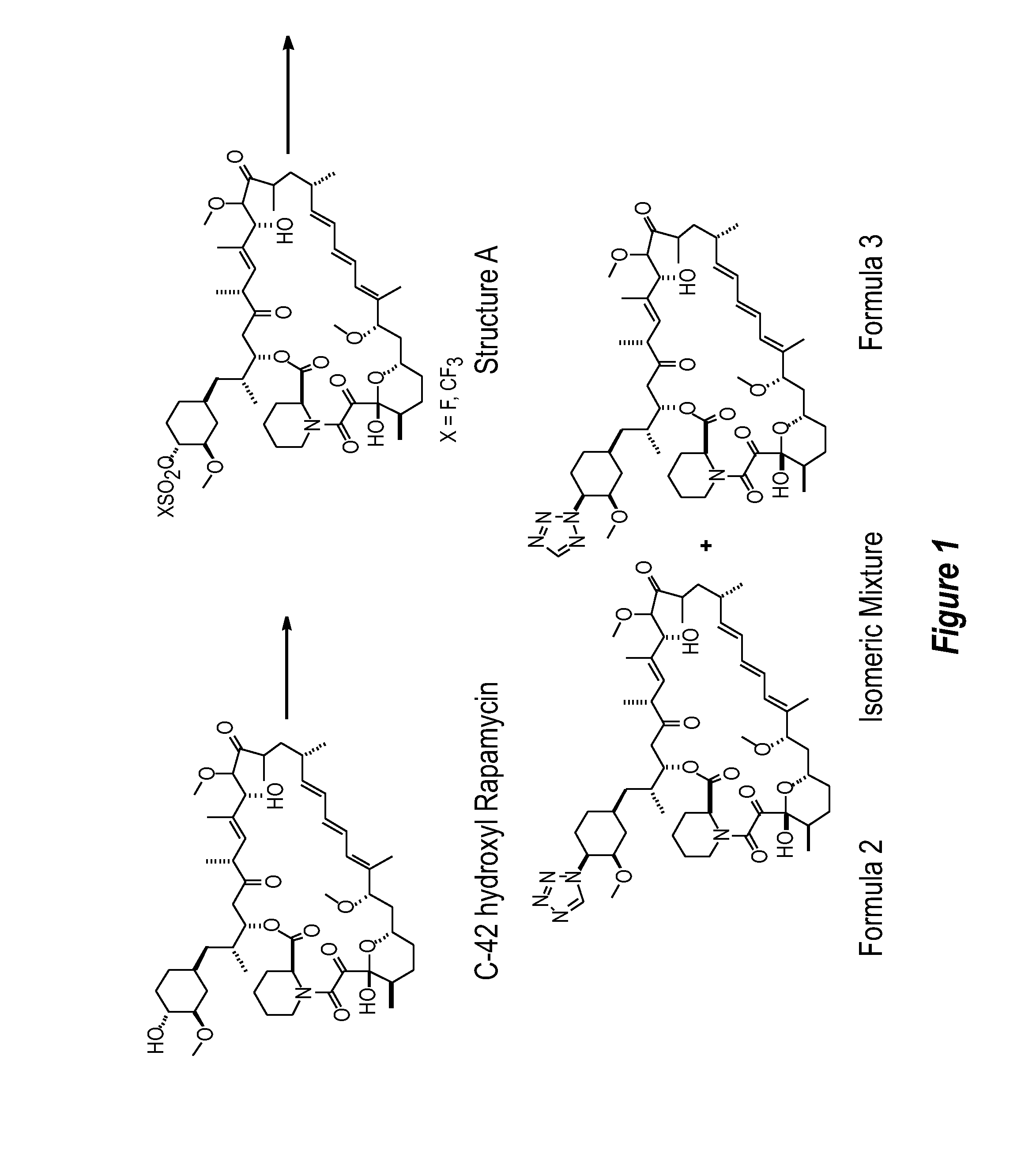
Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
538 results about "Sirolimus" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Sirolimus is used with other medications to prevent rejection of a kidney transplant.
Sutures and surgical staples for anastamoses, wound closures, and surgical closures
InactiveUS8016881B2Improve featuresControlled release rateSuture equipmentsStentsSurgical stapleMicroparticle
The invention relates to sutures and surgical staples useful in anastomoses. Various aspects of the invention include wound closure devices that use amphiphilic copolymer or parylene coatings to control the release rate of an agent, such as a drug or a biological material, polymerizing a solution containing monomers and the agent to form a coating, using multiple cycles of swelling a polymer with a solvent-agent solution to increase loading, microparticles carrying the agent, biodegradable surgical articles with amphiphilic copolymer coatings, and sutures or surgical staples the deliver a drug selected from the group consisting of triazolopyrimidine, paclitaxol, sirolimus, derivatives thereof, and analogs thereof to a wound site.
Owner:MIRUS LLC
Drug/drug delivery systems for the prevention and treatment of vascular disease
InactiveUS20020007215A1Prevent proliferationGood effectOrganic active ingredientsStentsVascular diseaseWhole body
A drug and drug delivery system may be utilized in the treatment of vascular disease. A local delivery system is coated with rapamycin or other suitable drug, agent or compound and delivered intraluminally for the treatment and prevention of neointimal hyperplasia following percutaneous transluminal coronary angiography. The local delivery of the drugs or agents provides for increased effectiveness and lower systemic toxicity.
Owner:WYETH LLC
Medical devices containing rapamycin analogs
InactiveUS6890546B2Reduce probabilityReduces restenosis in vasculatureBiocideAntimycoticsArteriovenous graftsMedical treatment
A medical device comprising a supporting structure having a coating on the surface thereof, the coating containing a therapeutic substance, such as, for example, a drug. Supporting structures for the medical devices that are suitable for use in this invention include, but are not limited to, coronary stents, peripheral stents, catheters, arterio-venous grafts, by-pass grafts, and drug delivery balloons used in the vasculature. Drugs that are suitable for use in this invention include, but are not limited to,
Owner:ABBOTT LAB INC
Sutures and surgical staples for anastamoses, wound closures, and surgical closures
ActiveUS20050038472A1Good storage stabilityRemove moistureSuture equipmentsPowder deliverySurgical stapleMicroparticle
The invention relates to sutures and surgical staples useful in anastomoses. Various aspects of the invention include wound closure devices that use amphiphilic copolymer or parylene coatings to control the release rate of an agent, such as a drug or a biological material, polymerizing a solution containing monomers and the agent to form a coating, using multiple cycles of swelling a polymer with a solvent-agent solution to increase loading, microparticles carrying the agent, biodegradable surgical articles with amphiphilic copolymer coatings, and sutures or surgical staples the deliver a drug selected from the group consisting of triazolopyrimidine, paclitaxol, sirolimus, derivatives thereof, and analogs thereof to a wound site.
Owner:MIRUS LLC
Injectable compositions of nanoparticulate immunosuppressive compounds
InactiveUS20060210638A1Improve complianceImprove efficacyPowder deliveryBiocideDepressantCompound (substance)
The invention is directed to an injectable nanoparticulate immunosuppressant composition for the formation of a subcutaneous or intramuscular depot. The invention is also directed to an injectable composition of nanoparticulate tacrolimus and / or sirolimus which eliminates the need to use polyoxyl 60 hydrogenated castor oil (HCO-60) and / or polysorbate 80 as a solubilizer. This invention further discloses a method of making an injectable nanoparticulate tacrolimus and / or sirolimus composition and is also directed to methods of treatment using the injectable nanoparticulate formulations comprising tacrolimus, sirolimus, or combination thereof for a subcutaneous or intramuscular depot for the prophylaxis of organ rejection and for the treatment of psoriasis or other immune diseases
Owner:ELAN PHRMA INT LTD
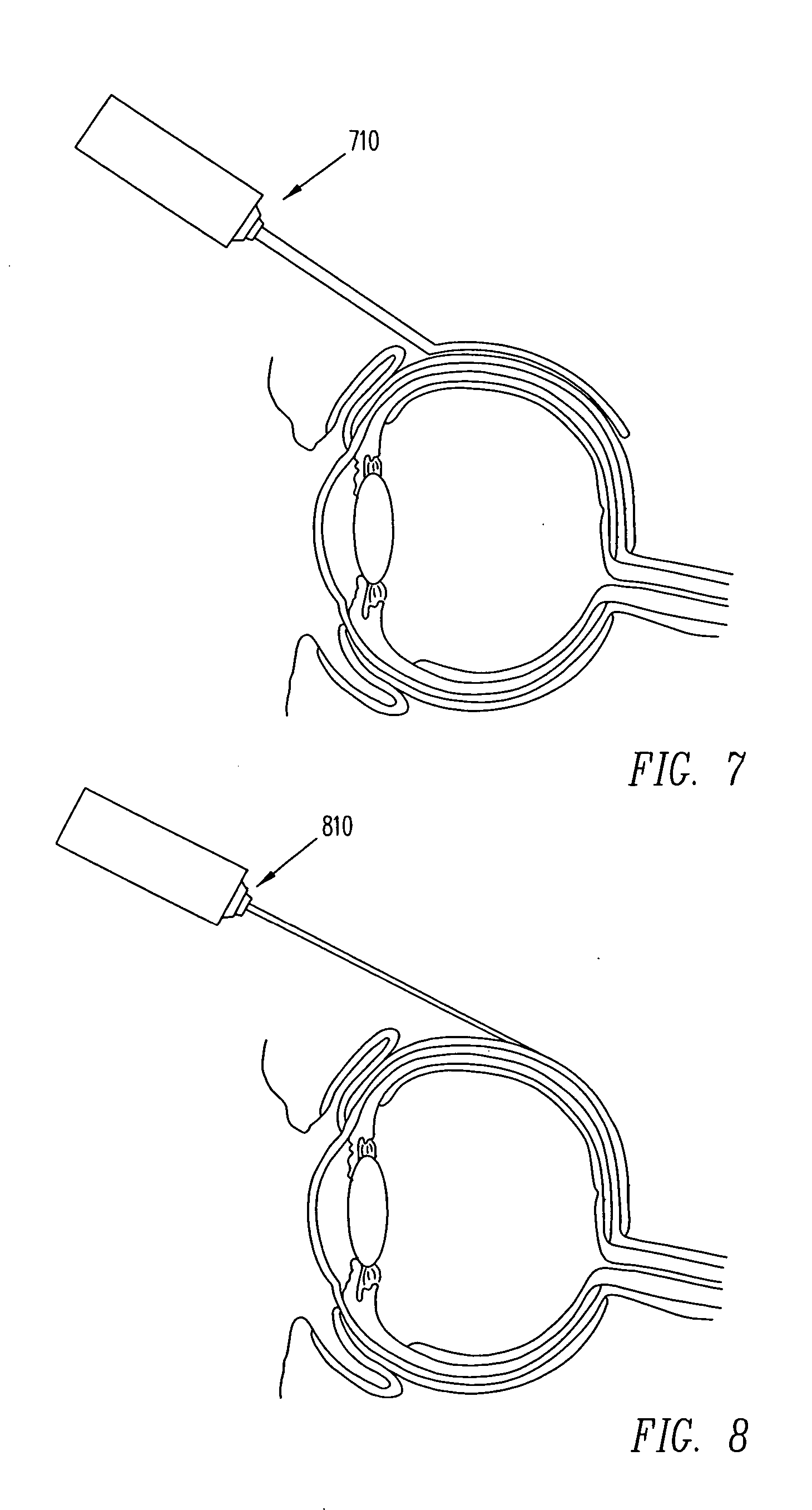
Transscleral delivery
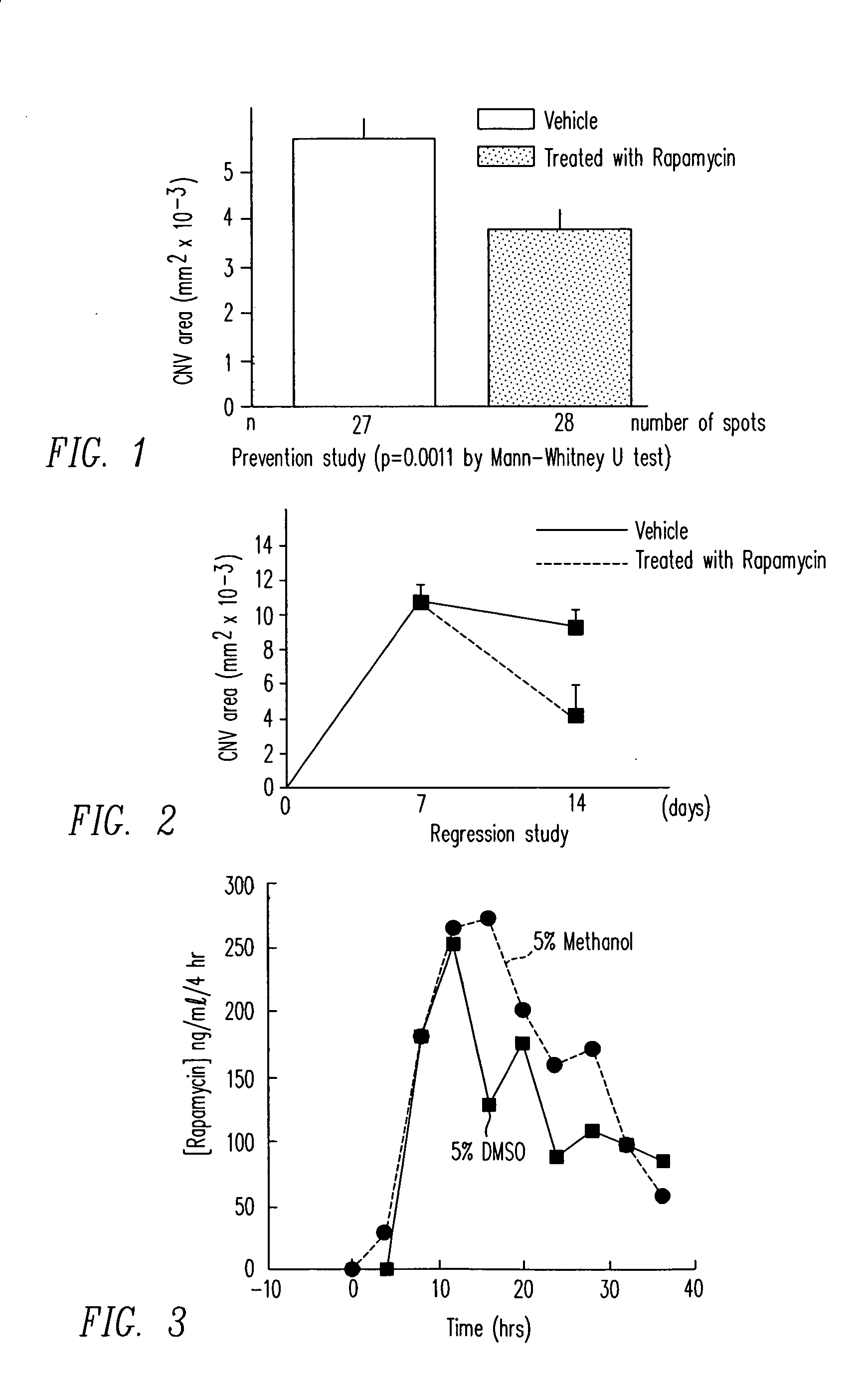
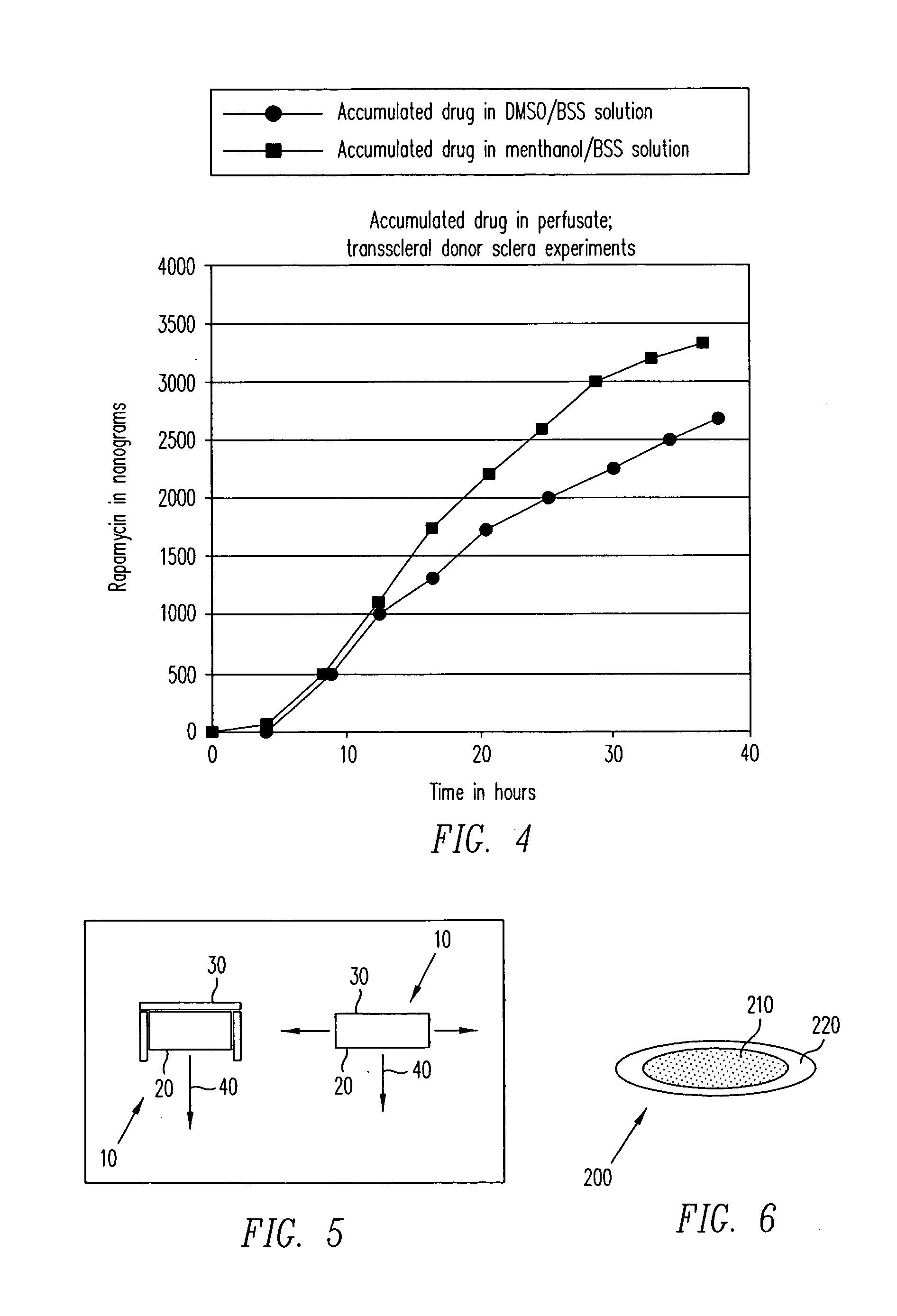
Diseases associated with the tissues in the posterior segment of the eye can be effectively treated by administering therapeutic agents transsclerally to those tissues. Compositions, devices, and methods for delivering therapeutic agents so that they cross the sclera and reach these tissues include injecting solutions or suspensions adjacent to or within the sclera and implanting solid structures containing the therapeutic agent adjacent to or within the sclera. These methods may be used for administering rapamycin or related compounds to treat choroidal neovascularization associated with age-related macular degeneration.
Owner:SANTEN PHARMA CO LTD
Treatment of Asthma and Chronic Obstructive Pulmonary Disease With Anti-proliferate and Anti-inflammatory Drugs
InactiveUS20080175887A1Promote absorptionOrganic active ingredientsPowdered material dispensingDiseaseObstructive Pulmonary Diseases
Embodiments of the present invention provide a method for treatment of respiratory disorders such as asthma, chronic obstructive pulmonary disease, and chronic sinusitis, including cystic fibrosis, interstitial fibrosis, chronic bronchitis, emphysema, bronchopulmonary dysplasia and neoplasia. The method involves administration, preferably oral, nasal or pulmonary administration, of anti-inflammatory and anti-proliferative drugs (rapamycin or paclitaxel and their analogues).
Owner:LUTONIX INC
Local vascular delivery of trichostatin a alone or in combination with sirolimus to prevent restenosis following vascular injury
ActiveUS20050136090A1Minimize potential risk of damageReduce frictionBiocideSurgeryPercent Diameter StenosisBlood vessel
Medical devices, and in particular implantable medical devices, may be coated to minimize or substantially eliminate a biological organism's reaction to the introduction of the medical device to the organism. The medical devices may be coated with any number of biocompatible materials. Therapeutic drugs, agents or compounds may be mixed with the biocompatible materials and affixed to at least a portion of the medical device. These therapeutic drugs, agents or compounds may also further reduce a biological organism's reaction to the introduction of the medical device to the organism. In addition, these therapeutic drugs, agents and / or compounds may be utilized to promote healing, including the formation of blood clots. Also, the devices may be modified to promote endothelialization. Various materials and coating methodologies may be utilized to maintain the drugs, agents or compounds on the medical device until delivered and positioned. In addition, the devices utilized to deliver the implantable medical devices may be modified to reduce the potential for damaging the implantable medical device during deployment. Medical devices include stents, grafts, anastomotic devices, perivascular wraps, sutures and staples. In addition, various polymer combinations may be utilized to control the elution rates of the therapeutic drugs, agents and / or compounds from the implantable medical devices.
Owner:WYETH
Drug eluting coatings for medical implants
ActiveUS20040037886A1Minimizing restenosisMinimizing thrombosisSuture equipmentsBiocideEverolimusCyclosporins
Drug eluting coating compositions are composed of at least one therapeutic agent dispersed in modified, biologically active binders. The therapeutic agents included in the coating composition are paclitaxel, sirolimus, tacrolimus, everolimus, actinomycin-D, dexamethasone, mycophenolic acid, cyclosporins, estradiol, and derivatives and analogs thereof. These therapeutic agents are applied to the surface of the medical device by a modified, biologically active binders. By using these biologically active binders, the therapeutic agents can be applied to at least one surface of a medical implant without using inert polymer carriers.
Owner:BIOVENTION INC
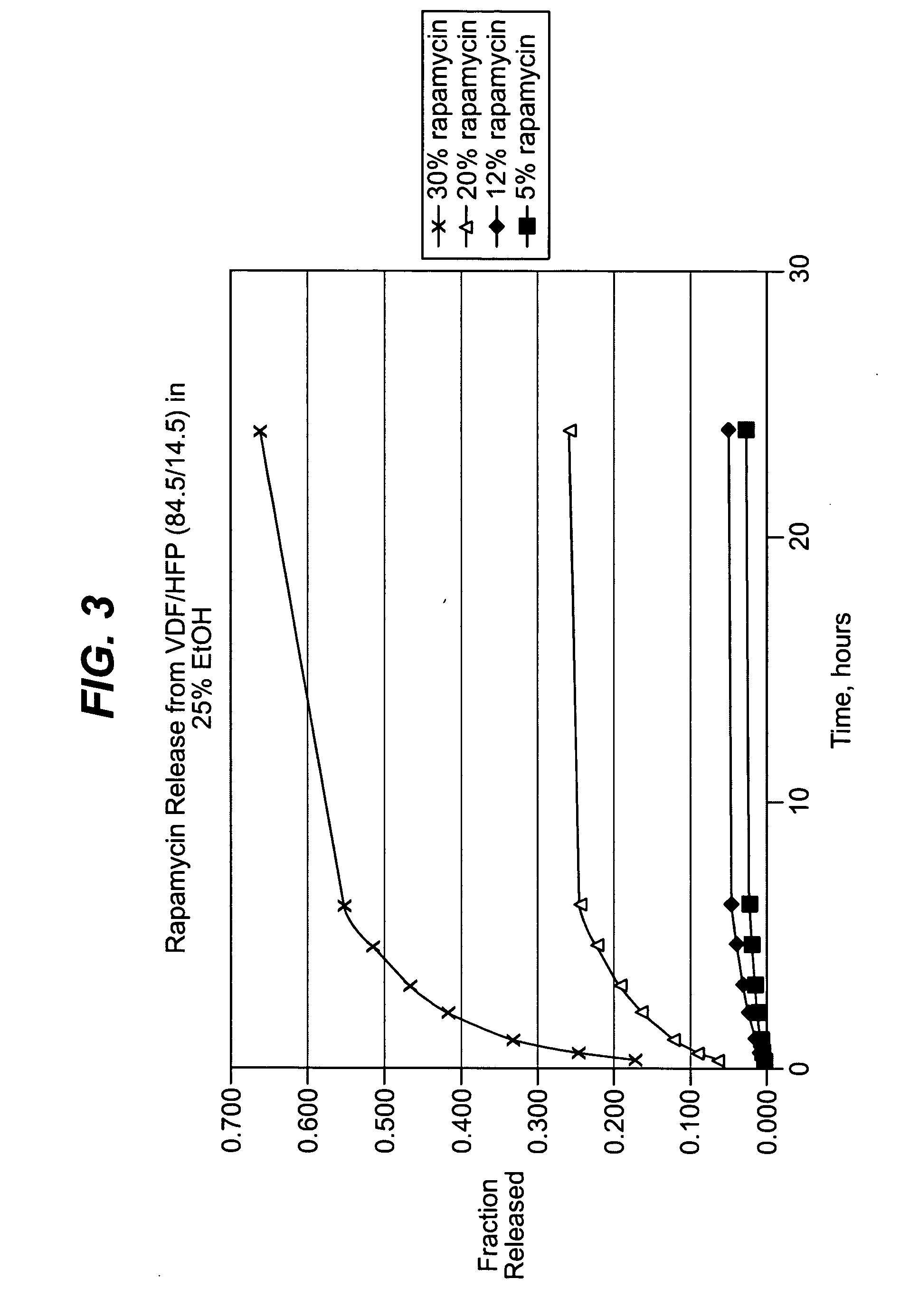
Solution formulations of sirolimus and its analogs for CAD treatment
Medical devices, and in particular implantable medical devices, may be coated to minimize or substantially eliminate a biological organism's reaction to the introduction of the medical device to the organism. The medical devices may be coated with any number of biocompatible materials. Therapeutic drugs, agents or compounds may be mixed with the biocompatible materials and affixed to at least a portion of the medical device. These therapeutic drugs, agents or compounds may also further reduce a biological organism's reaction to the introduction of the medical device to the organism. In addition, these therapeutic drugs, agents and / or compounds may be utilized to promote healing, including the formation of blood clots. Therapeutic agents may also be delivered to the region of a disease site. In regional delivery, liquid formulations may be desirable to increase the efficacy and deliverability of the particular drug. Also, the devices may be modified to promote endothelialization. Various materials and coating methodologies may be utilized to maintain the drugs, agents or compounds on the medical device until delivered and positioned. In addition, the devices utilized to deliver the implantable medical devices may be modified to reduce the potential for damaging the implantable medical device during deployment. Medical devices include stents, grafts, anastomotic devices, perivascular wraps, sutures and staples. In addition, various polymer combinations may be utilized to control the elution rates of the therapeutic drugs, agents and / or compounds from the implantable medical devices.
Owner:WYETH LLC
Method for production of antibodies to specific sites of rapamycin
InactiveUS6709873B1Immunoglobulins against fungi/algae/lichensBiological testingPolyclonal antibodiesDivinyl sulfone
Owner:ISODIAGNOSTIKA
Implantable shunt or catheter enabling gradual delivery of therapeutic agents
InactiveUS20080051691A1Reduce deliveryAvoid infectionBiocideNervous disorderTherapeutic effectFlow diverter
An implantable catheter or shunt for draining fluid from a body cavity. The catheter or shunt body has a wall structure that carries one or more therapeutic agents in a manner enabling release of the therapeutic agent from the wall structure in situ after surgical implantation of the catheter or shunt body. The therapeutic agent can be gradually released over time to prevent infection, inhibit tissue ingrowths, and / or provide some other desired medicinal purpose. As an example, the therapeutic agent can be rapamycin or an mTOR inhibitor. According to some contemplated embodiments of the present invention, the therapeutic agent carried by the catheter / shunt is rechargeable or refillable in situ so that the therapeutic agent can be gradually released from the catheter / shunt over the expected useful life of the catheter / shunt.
Owner:WYETH LLC
Composition
The invention provides microparticles comprising an immunosuppressant, such as tacrolimus, sirolimus, pimecrolimus, ciclosporin, everolimus or a derivative thereof, and optionally a pharmaceutically acceptable excipient or carrier, such as a saccharide, amino acid, a sugar alcohol or a mixture thereof, and having a median geometric diameter of less than, or equal to, about 10 μm and which have a tap density of less than or equal to about 0.3 g / cm3.
Owner:INNOVATA LTD
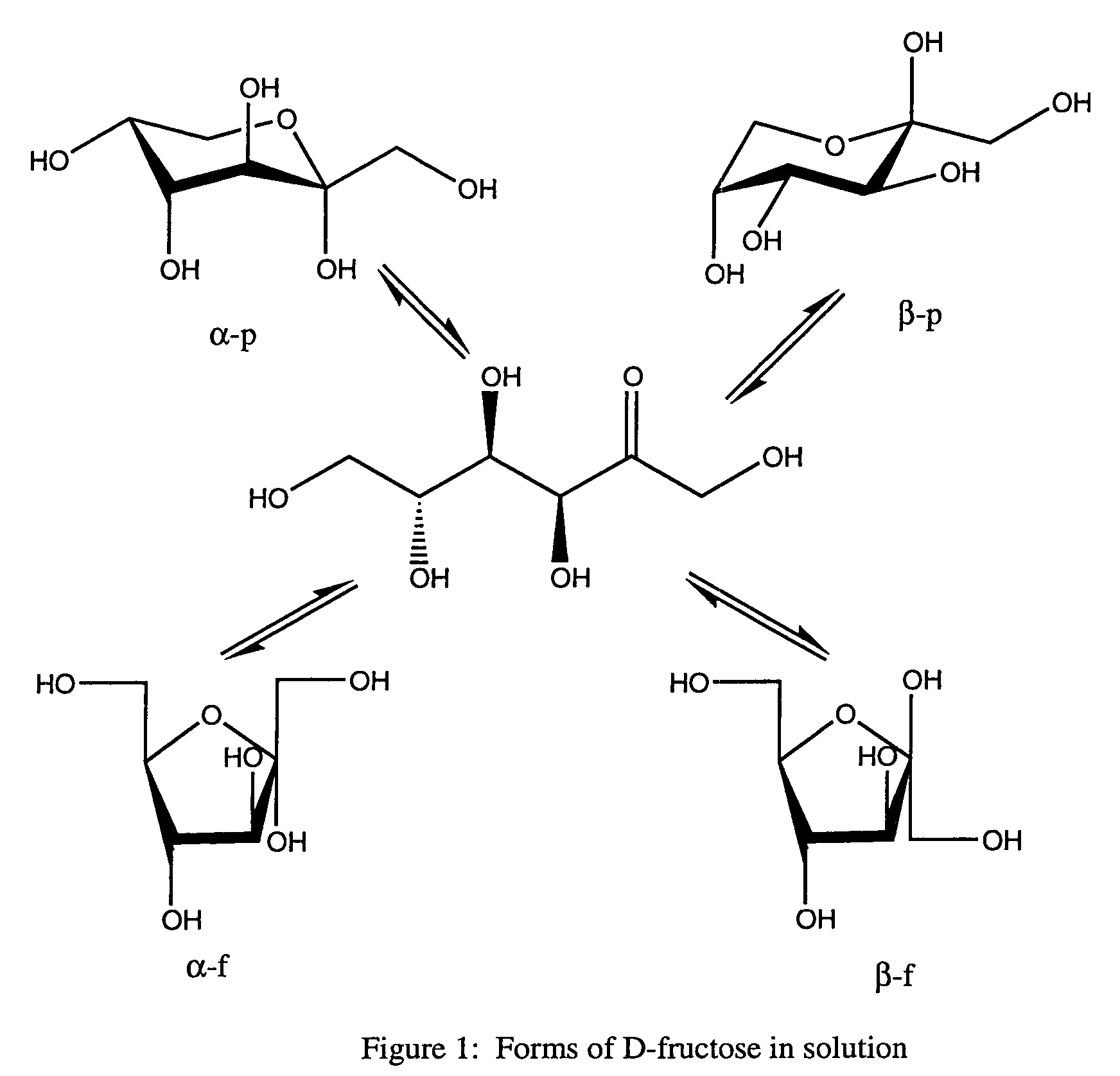
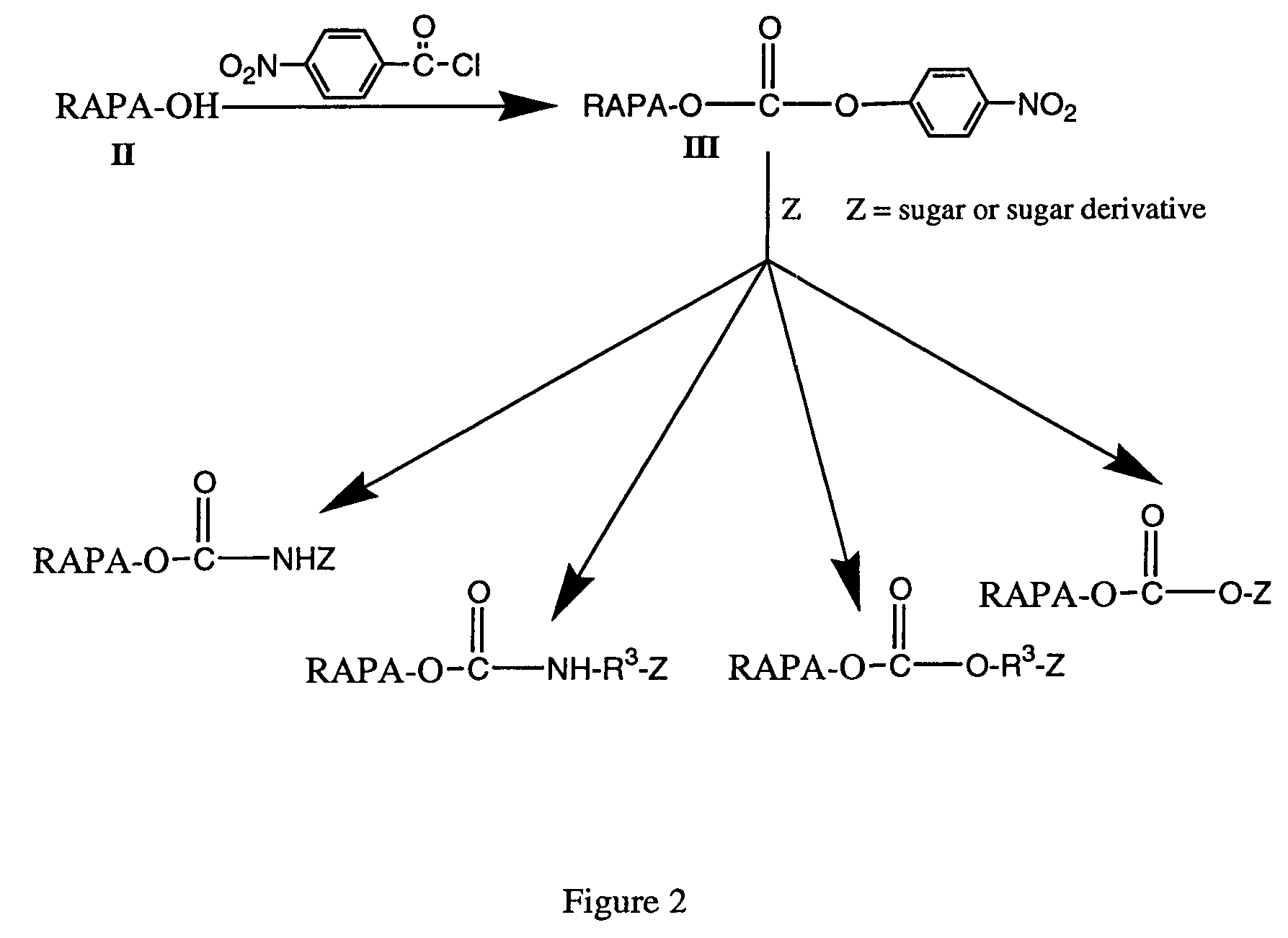
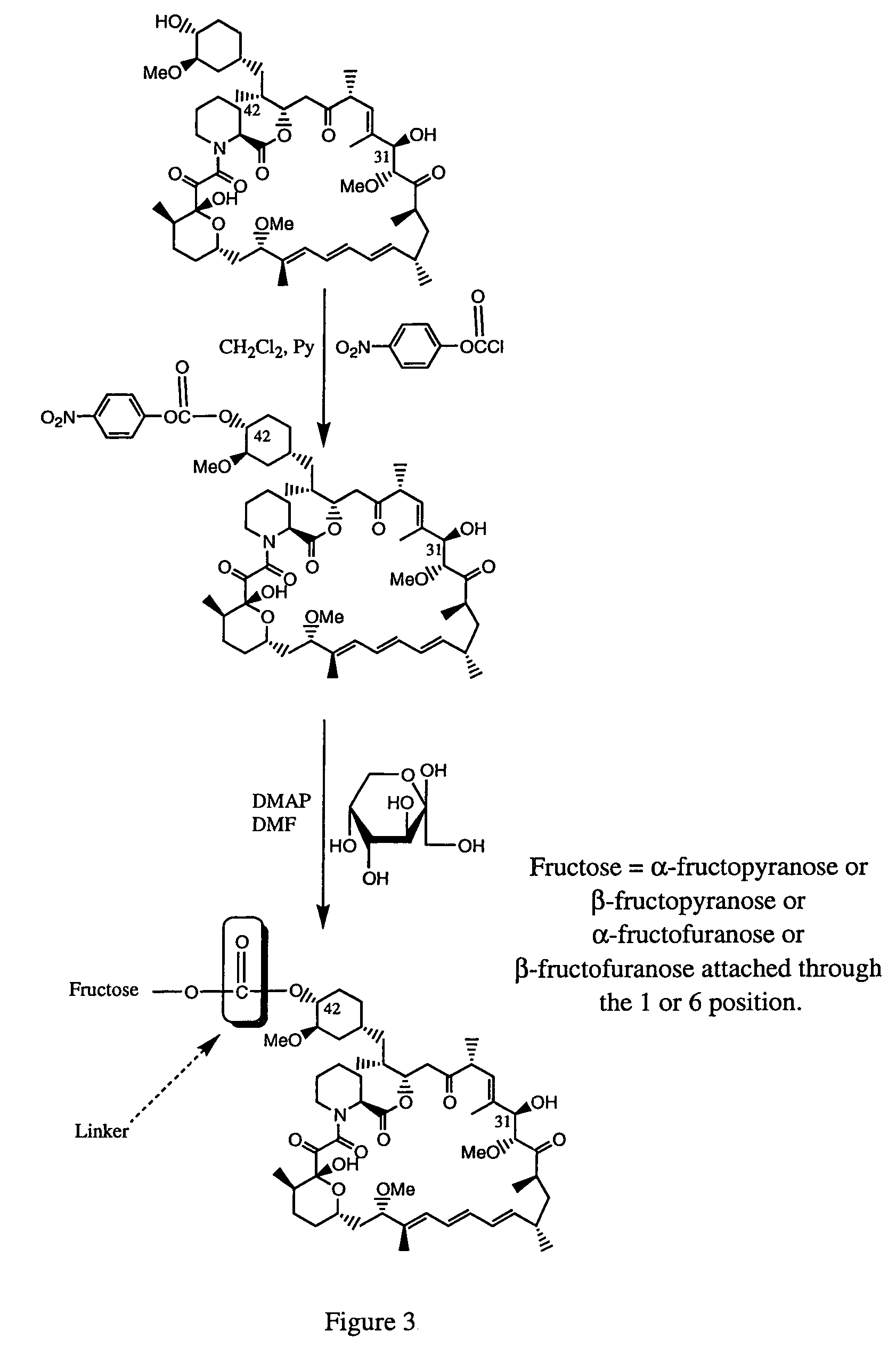
Rapamycin carbohydrate derivatives
InactiveUS7160867B2Low toxicityMore water solubleAntibacterial agentsBiocidePseudosugarsCarbohydrate derivative
This invention provides modified rapamycins that have specific monosaccharide(s), oligosaccharide(s), pseudosugar(s) or derivatives thereof attached through a linker to create rapamycin carbohydrate derivatives having enhanced pharmacokinetic and / or pharmacodynamic profiles. For example, administration of the rapamycin carbohydrate derivative results in altered pharmacokinetic profiles and reduced toxicities. Thus, the present invention provides compounds with characteristics that are distinct from other drugs in its class such as rapamycin.
Owner:ISOTECHNIKA INC
Rapamycin coated expandable devices
Medical devices may be utilized for local and regional therapeutic agent delivery. These therapeutic agents or compounds may reduce a biological organism's reaction to the introduction of the medical device to the organism. In addition, these therapeutic drugs, agents and / or compounds may be utilized to promote healing, including the prevention of thrombosis. The drugs, agents, and / or compounds may also be utilized to treat specific disorders, including restenosis, vulnerable plaque, and atherosclerosis in type 2 diabetic patients.
Owner:CARDINAL HEALTH SWITZERLAND 515 GMBH
Treatment of ocular disease
A formulation to treat ocular conditions such as dry eye disease, as well as other conditions, is disclosed. Rapamycin and / or ascomycin is administered intraocularly, such as by topical application, injection into the eye, or implantation in or on the eye. For example, a topical administration may contain between about 50 pg / ml drug to about 50 μg / ml drug in a formulation which may be applied at bedtime or throughout the day. For injection, a dose of about 50 pg / ml to about 200 μg / ml may be used. Rapamycin and / or ascomycin may also be administered in milligram quantities as a surgical implant, for example, in a diffusible walled reservoir sutured to the wall of the sclera, or may be contained within an inert carrier such as microspheres or liposomes to provide a slow-release drug delivery system.
Owner:PEYMAN GHOLAM A DR
Sirolimus having specific particle size and pharmaceutical compositions thereof
Owner:RANBAXY LAB LTD
Crystalline frap complex
InactiveUS6532437B1Reduce the numberHigh resolutionTransferasesDigital computer detailsCrystallographyTernary complex
The invention relates to the human protein FRAP, and in particular to the FKBP12-rapamycin binding domain thereof and to the ternary complex formed by the FRB domain, rapamycin and FKBP12. A new crystalline composition comprising the ternary complex, coordinates defining its three dimensional structure in atomic detail, and uses thereof are disclosed.
Owner:CORNELL RES FOUNDATION INC
Rapamycin reservoir eluting stent
ActiveUS20110009953A1Reduced elutionMaintain vessel patencyStentsSurgeryMedical deviceBiological organism
Implantable medical devices may be utilized to locally delivery one or more drugs or therapeutic agents to treat a wide variety of conditions, including the treatment of the biological organism's reaction to the introduction of the implantable medical device. These therapeutic agents may be released under controlled and directional conditions from a stent so that the one or more therapeutic agents reach the correct target area, for example, the surrounding tissue.
Owner:CARDINAL HEALTH SWITZERLAND 515 GMBH
Methods and compositions related to synthetic nanocarriers with rapamycin in a stable, super-saturated state
PendingUS20160128987A1Induce immune toleranceDurable immune toleranceBiocideGranular deliveryPolyesterNanocarriers
Disclosed are compositions and methods that provide synthetic nanocarriers that comprise hydrophobic polyester carrier material and rapamycin that is in a stable, super-saturated amount. In some embodiments, the synthetic nanocarriers are also initially sterile filterable. In other embodiments, the rapamycin is present in the synthetic nanocarrier compositions in an amount that is less than 50 weight % rapamycin / hydrophobic polyester carrier material in the composition.
Owner:SELECTA BIOSCI
Local vascular delivery of probucol alone or in combination with sirolimus to treat restenosis, vulnerable plaque, aaa and stroke
InactiveUS20080241215A1Minimize potential risk of damageReduce frictionBiocideSurgeryDiseasePercent Diameter Stenosis
Medical devices, and in particular implantable medical devices, may be coated to minimize or substantially eliminate a biological organism's reaction to the introduction of the medical device to the organism. The medical devices may be coated with any number of biocompatible materials. Therapeutic drugs, agents or compounds may be mixed with the biocompatible materials and affixed to at least a portion of the medical device. These therapeutic agents or compounds may also further reduce a biological organism's reaction to the introduction of the medical device to the organism. In addition, these therapeutic drugs, agents and / or compounds may be utilized to promote healing, including the prevention of thrombosis. The drugs, agents, and / or compounds may also be utilized to treat specific disorders, including vulnerable plaque. Therapeutic agents may also be delivered to the region of a disease site. In regional delivery, liquid formulations may be desirable to increase the efficacy and deliverability of the particular drug. Also, the devices may be modified to promote endothelialization. Various materials and coating methodologies may be utilized to maintain the agents or compounds on the medical device until delivered and positioned. In addition, the devices utilized to deliver the implantable medical devices may be modified to reduce the potential for damaging the implantable medical device during deployment. Medical devices include stents, grafts, anastomotic devices, perivascular wraps, sutures and staples. In addition, various polymer combinations may be utilized to control the elution rates of the therapeutic drugs, agents and / or compounds from the implantable medical devices.
Owner:CORDIS CORP
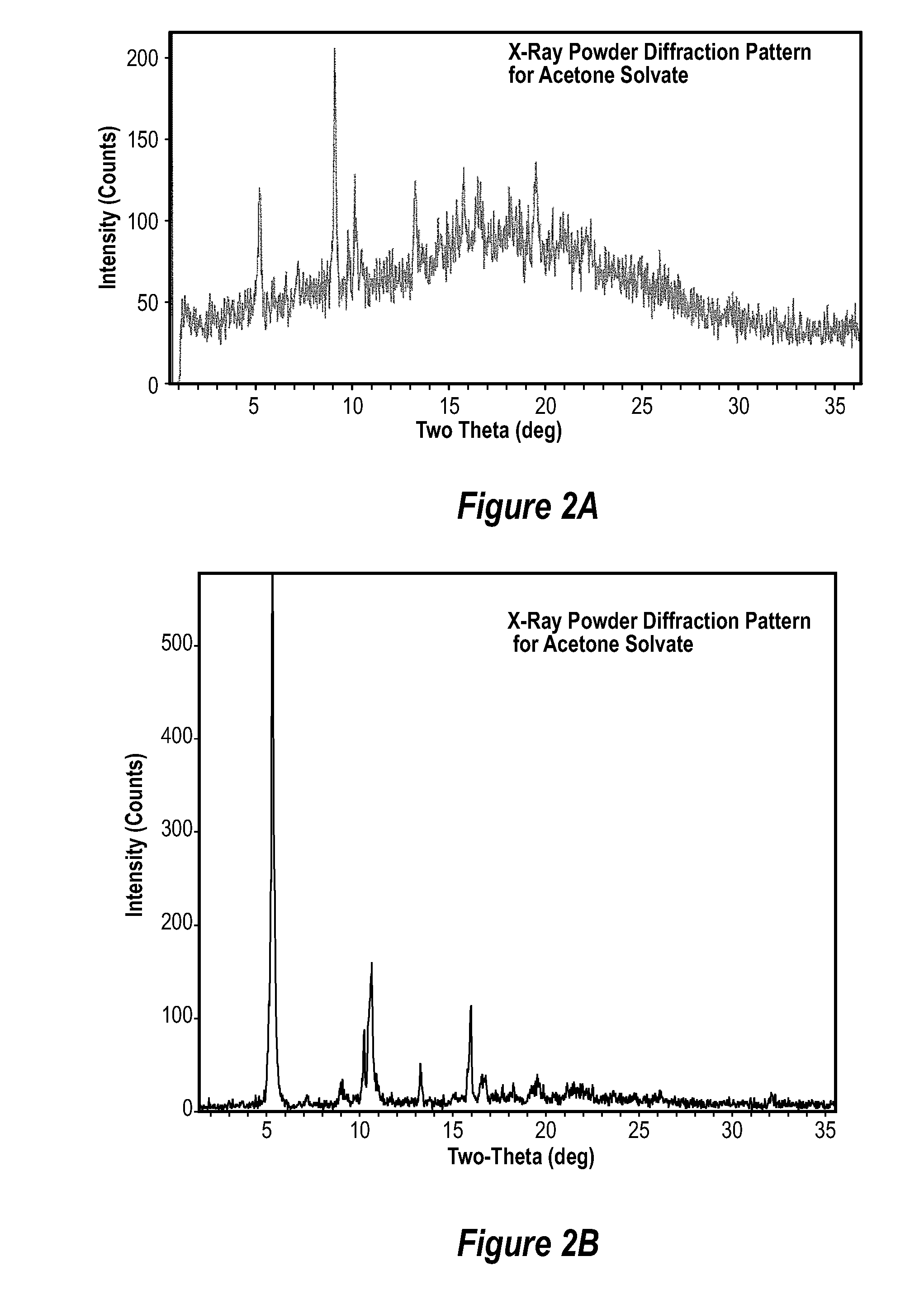
Methods of manufacturing crystalline forms of rapamycin analogs
A process for preparing a crystalline rapamycin analog includes: combining the rapamycin analog with an organic medium to form a mixture; incubating the mixture until the rapamycin analog crystallizes; and recovering the crystalline rapamycin analog. The organic medium can be a solvent, and the process can include causing the rapamycin analog to dissolve into the solvent, and incubating the solvent until the rapamycin analog crystallizes. The following can also be performed: forming a slurry of crystalline rapamycin analog; stirring the rapamycin analog mixture until the rapamycin analog crystallizes; saturating the rapamycin analog solution; forming a supersaturated rapamycin analog solution; combining an antisolvent with the rapamycin analog and the solvent to form a biphasic mixture, and incubating the biphasic mixture to cause a liquid-liquid phase split.
Owner:ABBOTT LAB INC
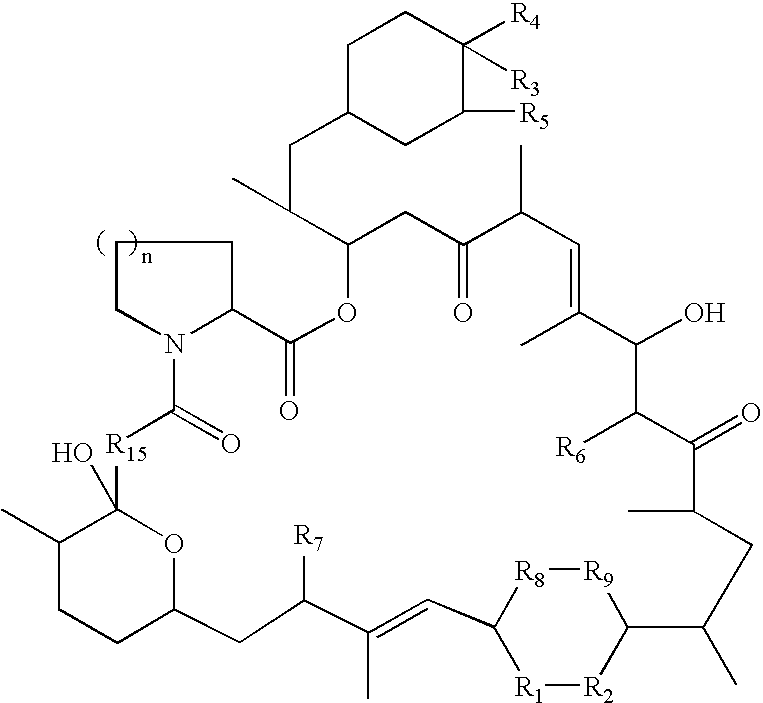
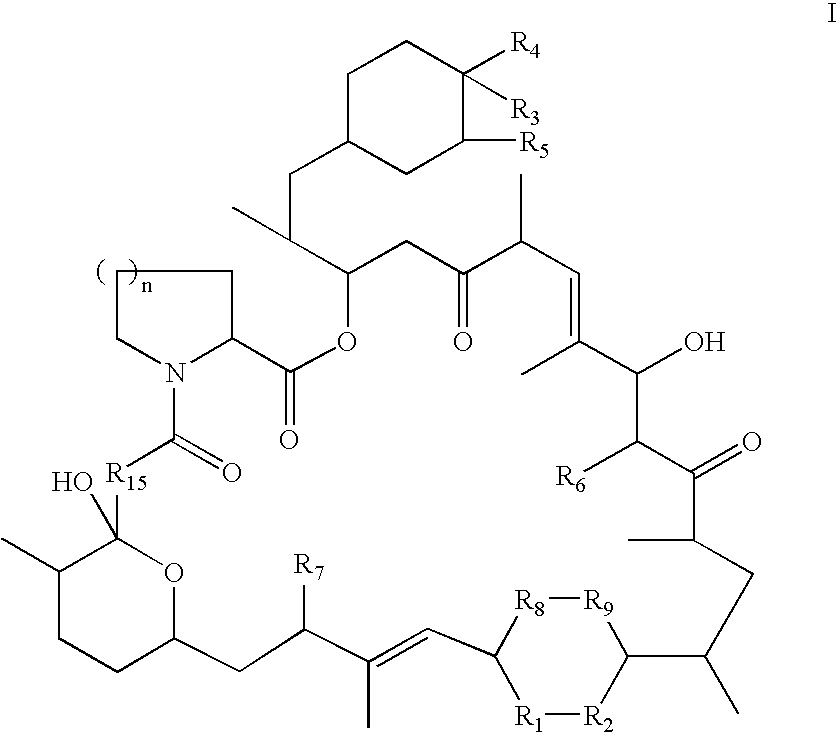
Rapamycin derivatives and the uses thereof in the treatment of neurological disorders
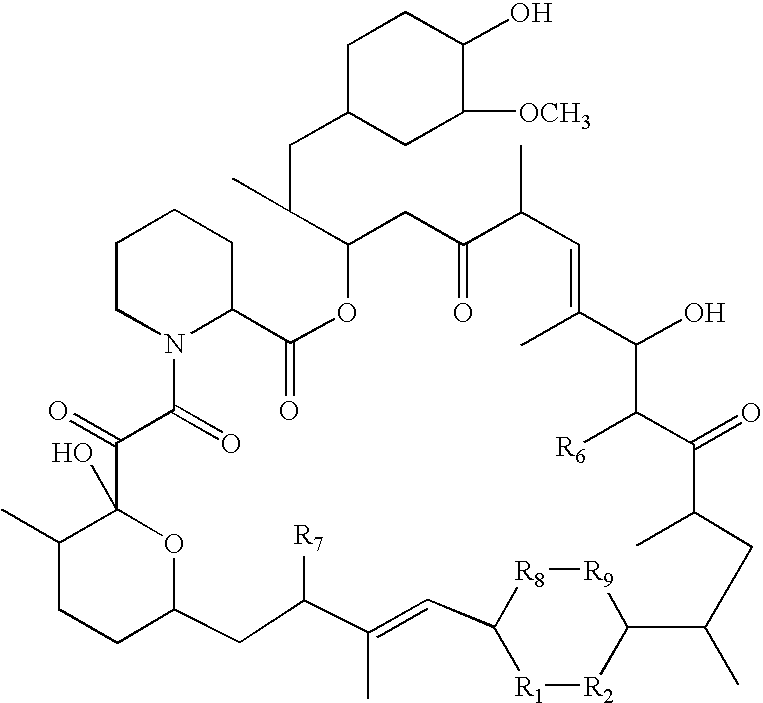
The present invention provides compounds of the following structure, wherein R1-R9, R15, and n are defined herein. These compounds are useful in treating neurological disorders or complications due to stroke or head injury, and as neuroprotective and neuroregenerative agents.
Owner:WYETH LLC
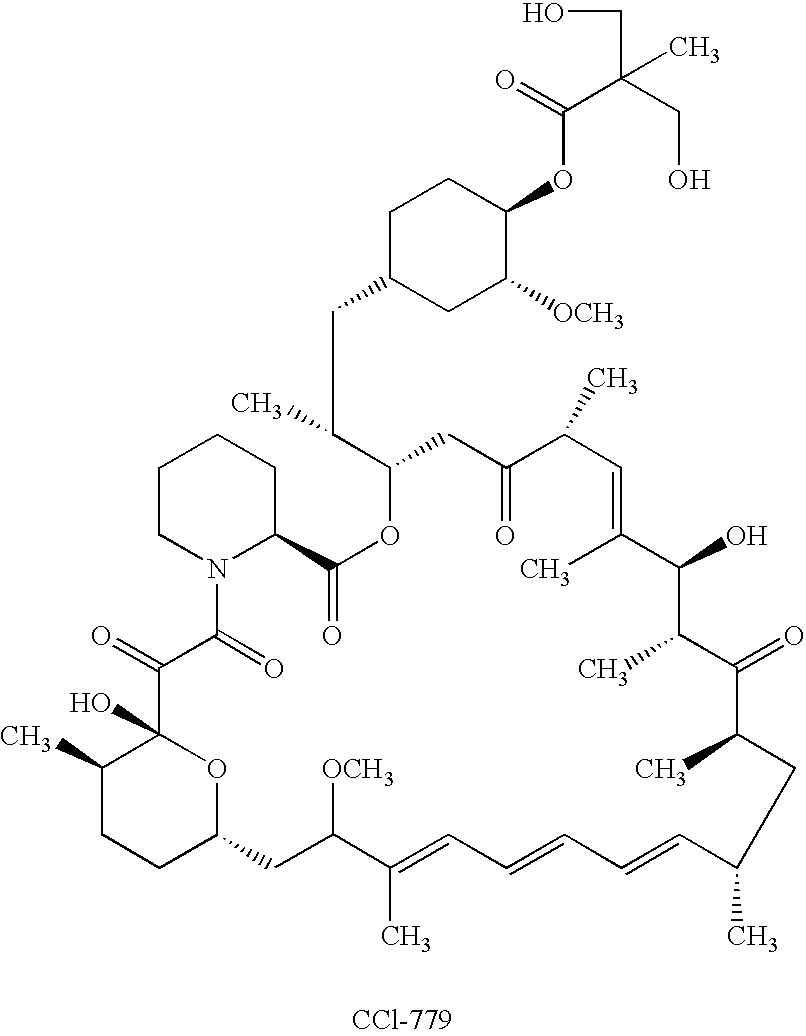
Orally bioavailable CCI-779 formulations
A CCI-779 oral dosage form is provided in which, after oral administration to a subject, the CCI-779 has a whole blood peak concentration (Cmax) of 5.4±1.8 ng / mL and an area under the curve (AUC) of about 66±about 22 ng-hr / ml and the sirolimus has a Cmax of 18.7±9.6 ng / mL and an AUC of about 600±about 228 ng-hr / ml, for a 25 mg unit dose of CCI-779. Another CCI-779 oral dosage form is provided which, after oral administration thereof to a subject, the CCI-779 has a Cmax of 5.7±1.7 ng / mL and an AUC of about 60±about 20 ng-hr / ml and the sirolimus has a Cmax of 17.1±8.1 ng / mL and an AUC of about 548±about 187 ng-hr / ml in whole blood, for a 25 mg unit dose of CCI-779. Products containing these oral dosage forms, and methods of use thereof, are also described.
Owner:WYETH LLC
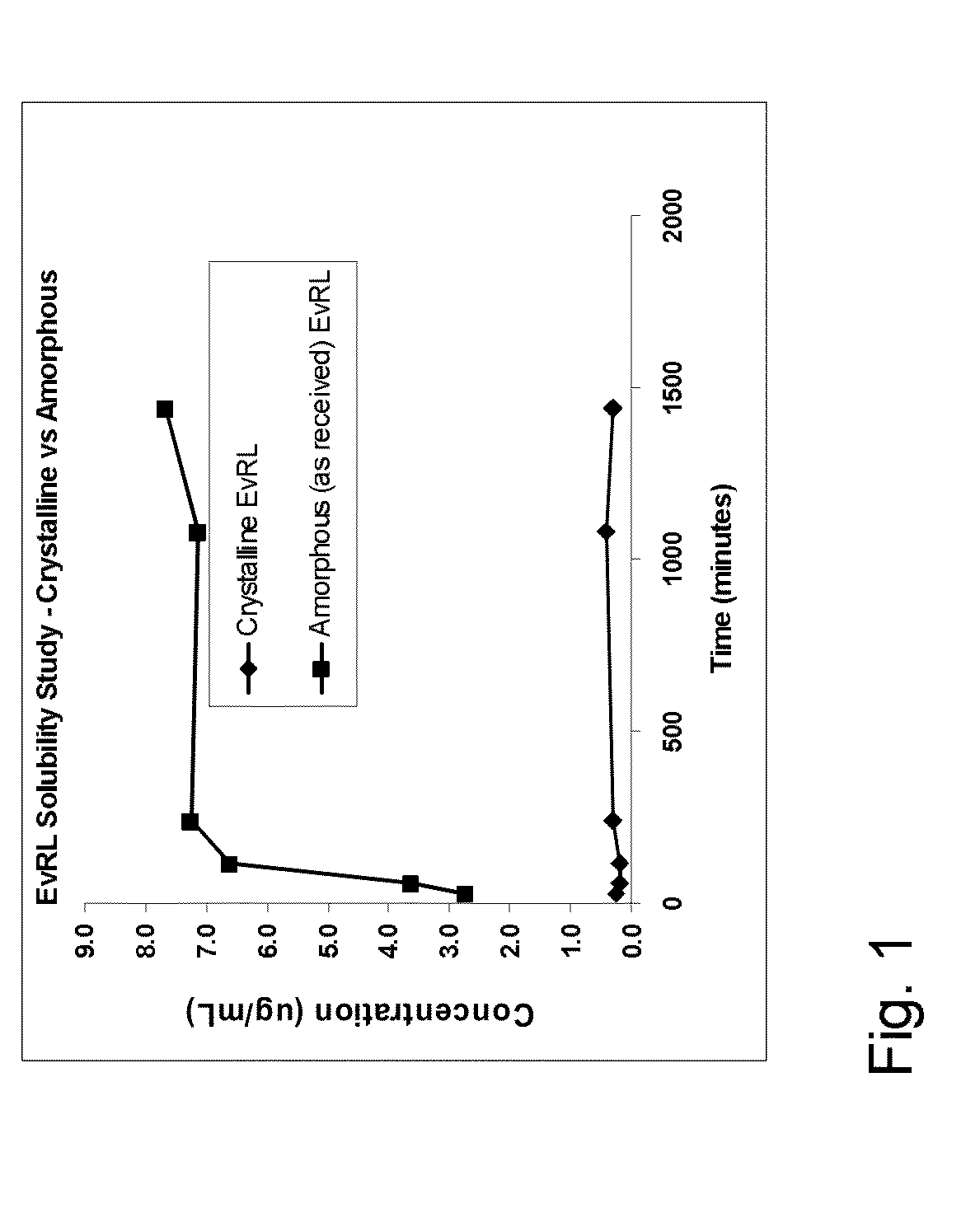
Medical Device with Crystalline Drug Coating
A medical device having a polymer-free outer surface layer comprising a crystalline drug selected from the group consisting of everolimus, tacrolimus, sirolimus, zotarolimus, biolimus, and rapamycin. The device may be produced by a method comprising the steps of providing a medical device; applying a solution of the drug to said portion of the outer surface to form a coating of amorphous drug; and vapor annealing the drug with a solvent vapor to form crystalline drug; wherein a seed layer of a crystalline form of said drug having a maximum particle size of about 10 μm or less is applied to at least said portion of the outer surface of the device before or after applying the drug solution, but before vapor annealing the amorphous coating.
Owner:BOSTON SCI SCIMED INC
Ocular solutions
InactiveUS7083803B2Reduce inflammationReduce bacterial growthBiocideSenses disorderDiseaseEverolimus
Ocular solutions containing at least one macrolide antibiotic and / or mycophenolic acid provide anti-inflammatory, anti-cell proliferation, anti-cell migration, anti-angiogenesis, antimicrobial and antifungal effects. In one embodiment, the solution is administered intraocularly after cataract surgery before insertion of a replacement intraocular lens, resulting in reduced posterior capsular opacification which may eliminate the need for a subsequent surgery. The solution may be one that is invasively administered, for example, an irrigation or volume replacement solution containing at least one macrolide antibiotic such as tacrolimus, sirolimus, everolimus, cyclosporine, and ascomycin, or mycophenolic acid. The solution may be one that is non-invasively or topically administered in the form of drops, ointments, gels, creams, etc. and may include eye lubricants and contact lens solutions. The solution may contain a supratherapeutic concentration of agent(s) so that a therapeutic concentration of a topically administered solution accumulates in a diseased ocular structure sufficient to treat the disease.
Owner:PEYMAN GHOLAM A DR
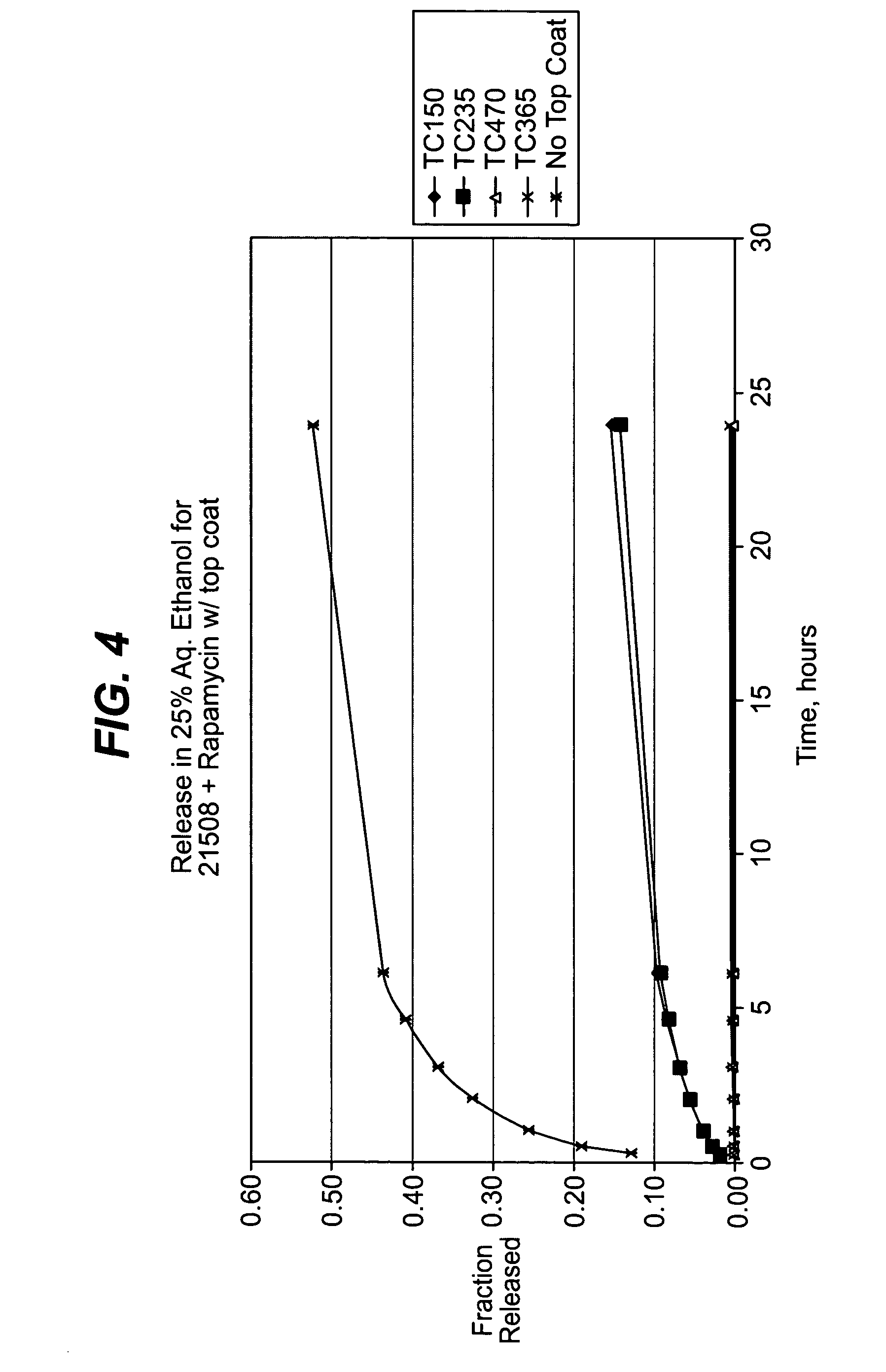
Stent with polymer coating containing amorphous rapamycin
InactiveUS20090062909A1Extensive adhesionBulk propertiesStentsSurgeryPolymer coatingsPolybutyl methacrylate
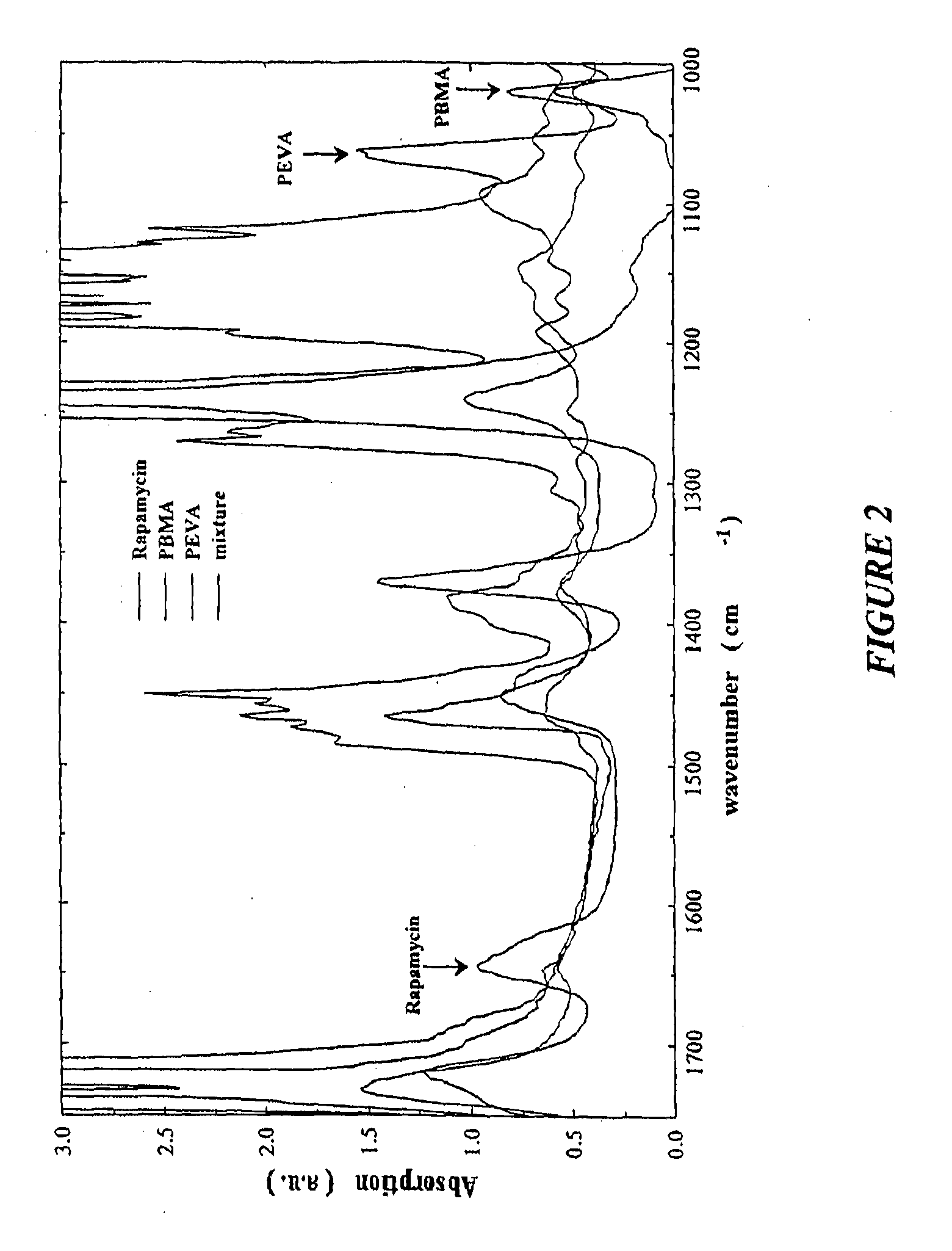
A coated coronary stent, comprising: a stainless steel sent framework coated with a primer layer of Parylene C; and a rapamycin-polymer coating having substantially uniform thickness disposed on the stent framework, wherein the rapamycin-polymer coating comprises polybutyl methacrylate (PBMA), polyethylene-co-vinyl acetate (PEVA) and rapamycin, wherein substantially all of the rapamycin in the coating is in amorphous form and substantially uniformly dispersed within the rapamycin-polymer coating.
Owner:MICELL TECH INC
Ocular solutions
InactiveUS20050063996A1Reduce turbidityReduce inflammationAntibacterial agentsBiocideEverolimusCell migration
Ocular solutions containing at least one macrolide antibiotic and / or mycophenolic acid provide anti-inflammatory, anti-cell proliferation, anti-cell migration, anti-angiogenesis, antimicrobial, and antifungal effects. In one embodiment, the solution is administered intraocularly after cataract surgery before insertion of a replacement intraocular lens, resulting in reduced posterior capsular opacification which may eliminate the need for a subsequent surgery. The solution may be one that is invasively administered, for example, an irrigation or volume replacement solution containing at least one macrolide antibiotic such as tacrolimus, sirolimus, everolimus, cyclosporine, and ascomycin, or mycophenolic acid. The solution may be one that is non-invasively or topically administered in the form of drops, ointments, gels, creams, etc. and may include eye lubricants and contact lens solutions.
Owner:PEYMAN GHOLAM A DR
Medical implants and methods for regulating the tissue response to vascular closure devices
InactiveUS20050004158A1High incidenceReducing host responseBiocideSurgeryVascular closure deviceFibrosis
Devices and methods for reducing, eliminating, preventing, suppressing, or treating tissue responses to hemostatic devices e.g., biological sealants or vascular procedures are disclosed. The invention employs a combination of resorbable, biocompatible matrix materials and a variety of therapeutic agents, such as antiproliferatives or antibiotics, applied to a vascular puncture or incision to achieve hemostasis following diagnostic or interventional vascular catheterizations and to treat neointimal hyperplasia and stenosis. A matrix of a material such as collagen provides a reservoir of a therapeutic agent such as rapamycin (sirolimus) and its derivatives and analogs for delivery at a tissue site at risk for vasculoproliferation, infection, inflammation, fibrosis or other tissue responses.
Owner:VASCULAR THERAPIES LLC (US)
Novel anti aging agents and methods to identify them
ActiveUS20100260733A1Stimulate mitochondrial functionPrevent and treat deteriorationOrganic active ingredientsSenses disorderAge related diseaseHigh-Throughput Screening Methods
The present invention discloses novel mechanisms in the aging process and describes novel methods for high-throughput screening to identify, detect, and purify agents to be used for improving mitochondrial function, maintaining the cell cycle-arrested state in senescent and post mitotic cells, and thus preventing or treating age-related diseases or disorders associated with accelerated mitochondrial function loss, telomere dysfunction, and / or deterioration of the growth-arrested state. The present invention also discloses a number of compounds or compositions identified from this method. The present invention further provides the use of low doses of rapamycin or its analogs as a mimic of caloric restriction in preventing age-related diseases or disorders.
Owner:QI HAIYAN
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com