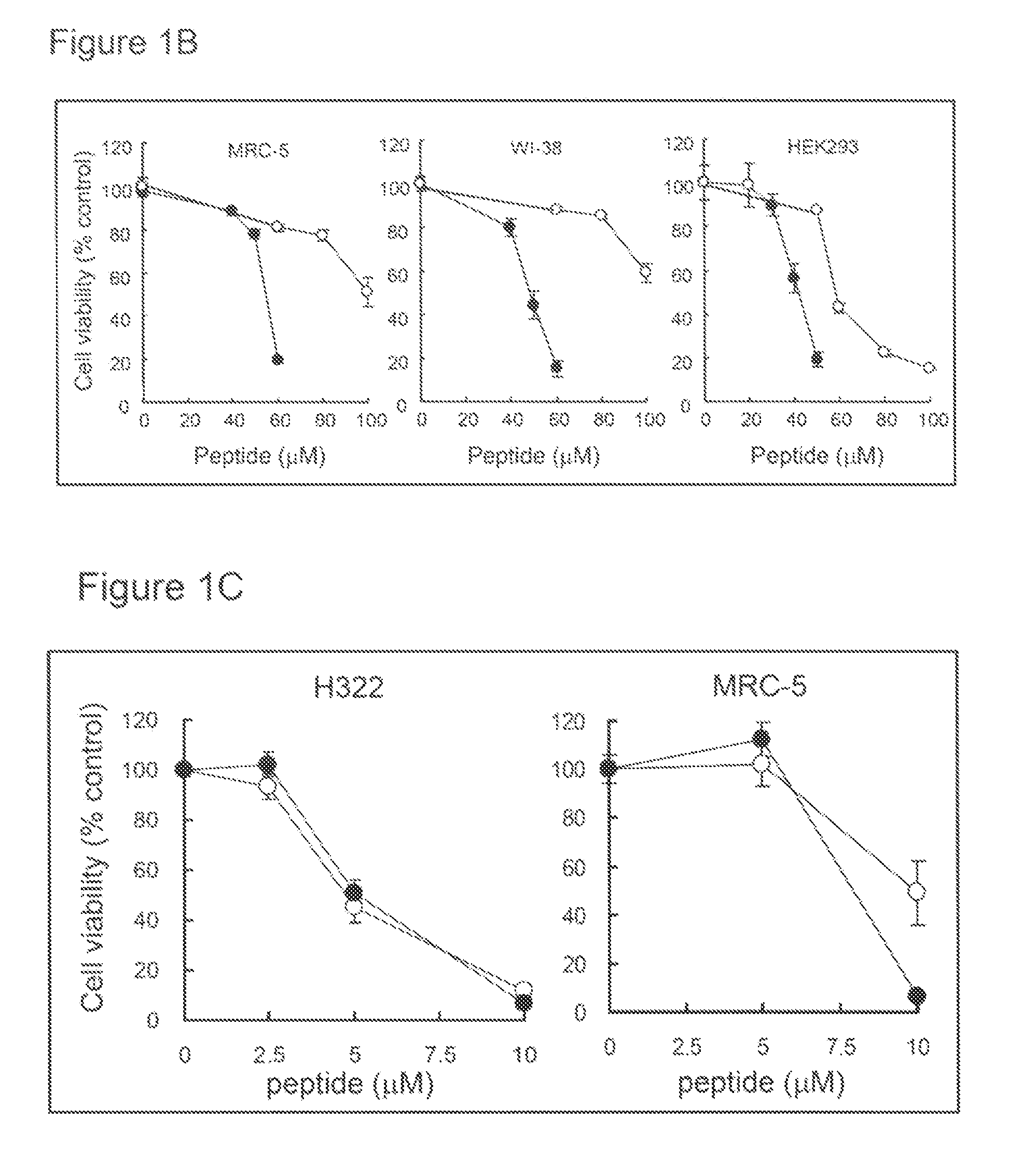
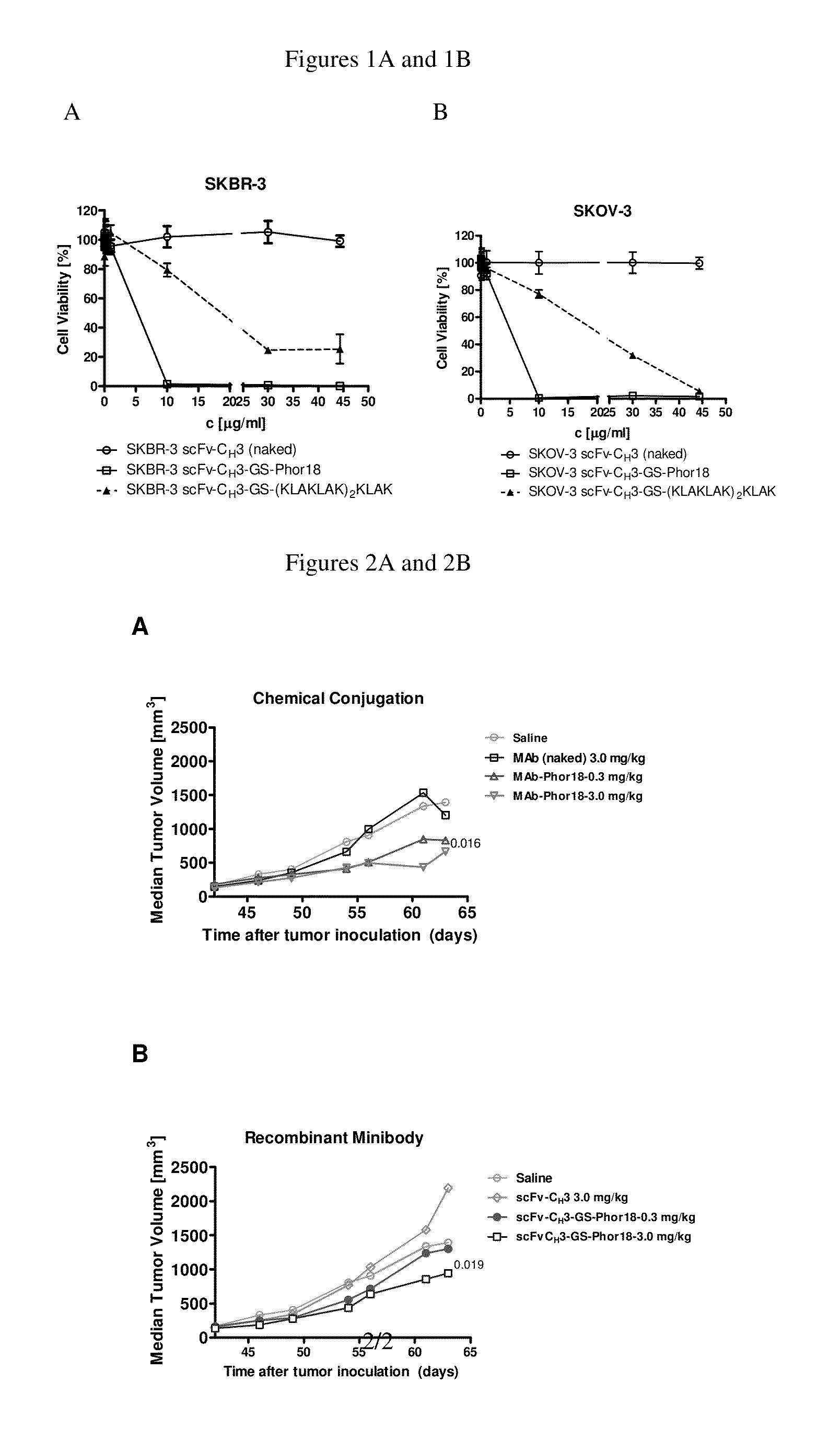
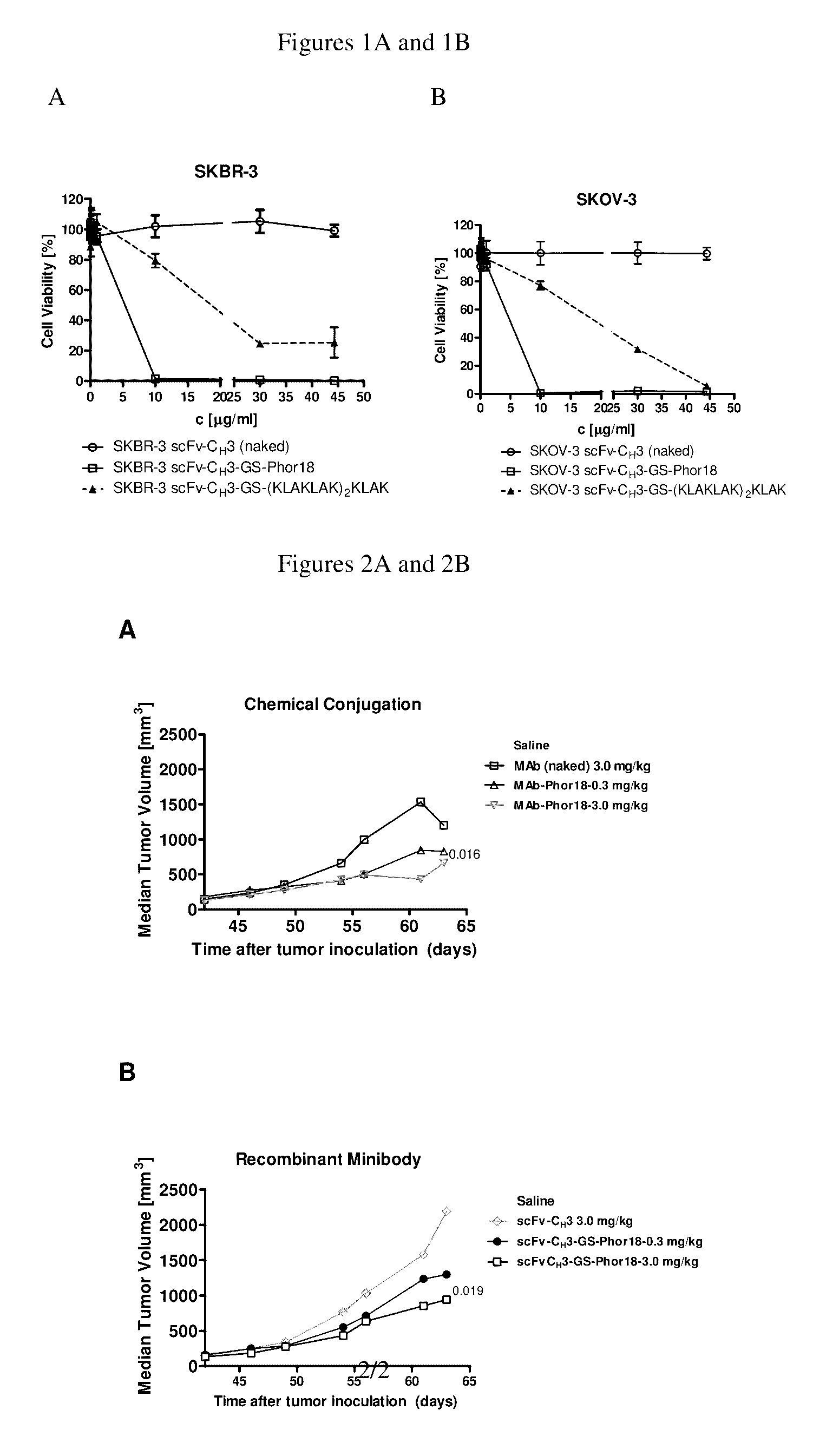
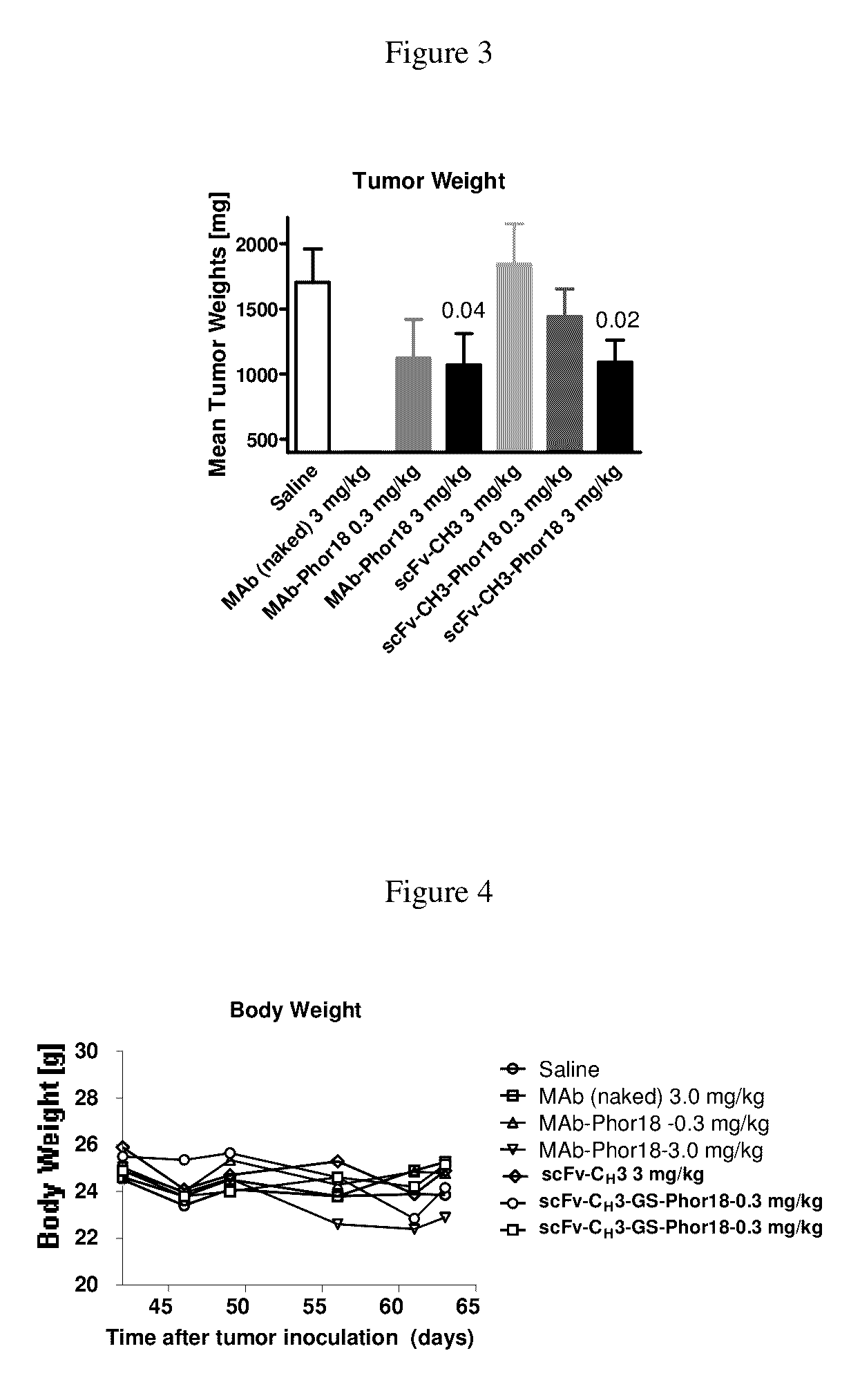
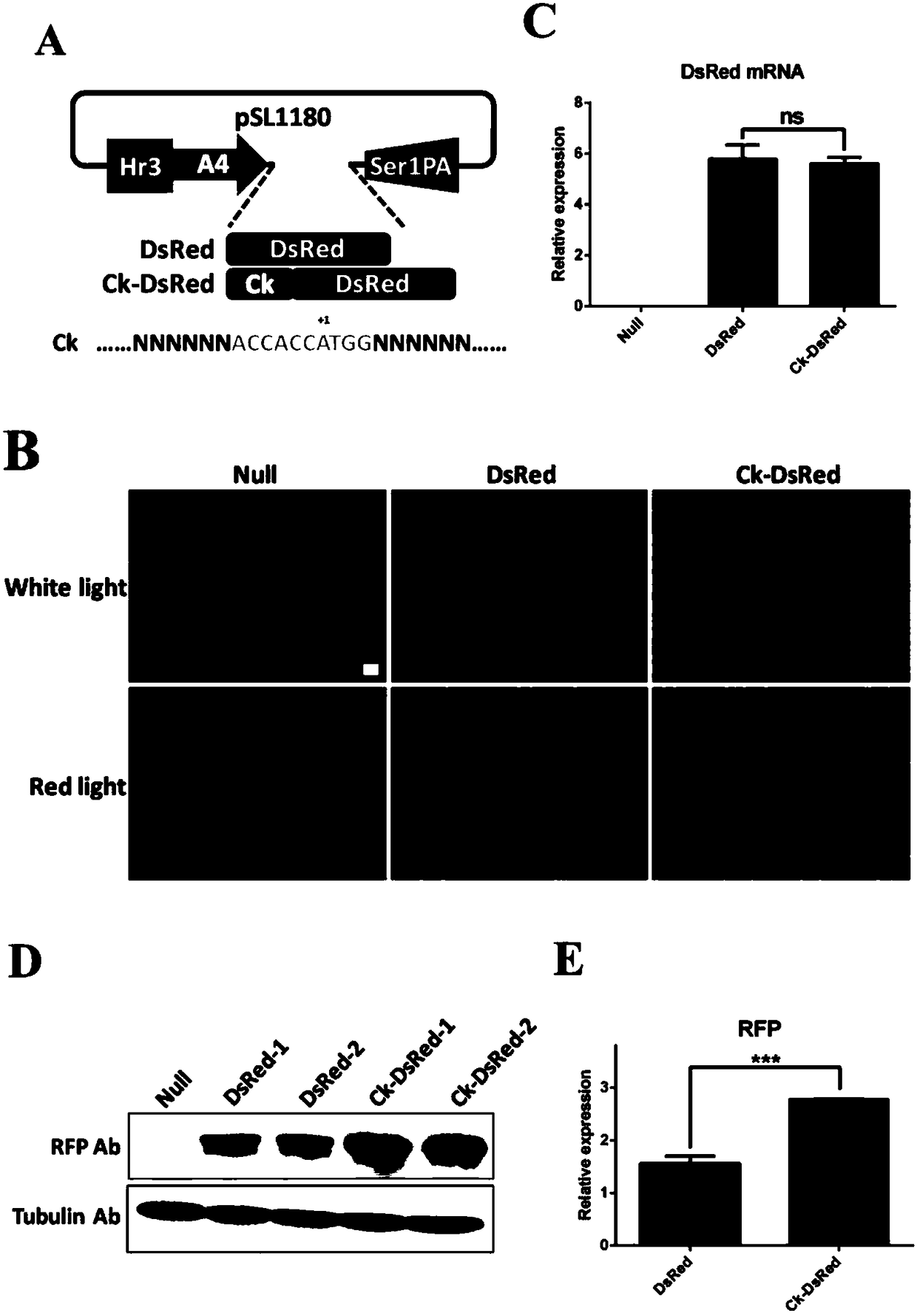
Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
54 results about "Lytic peptide" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
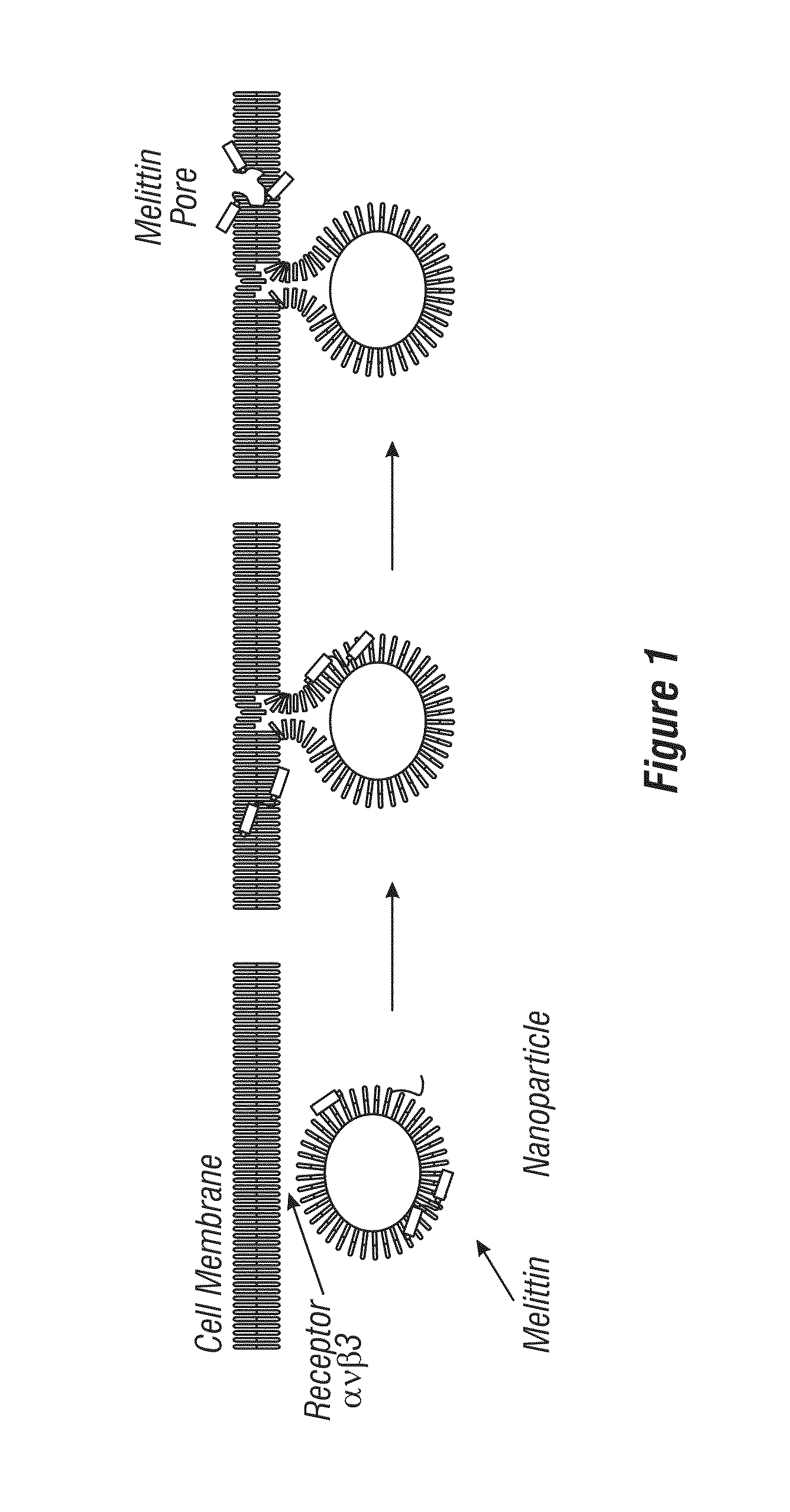
Nanoparticle delivery systems comprising a hydrophobic core and a lipid/surfactant layer comprising a membrane-lytic peptide
Owner:WASHINGTON UNIV IN SAINT LOUIS
Ligand/lytic peptide compositions and methods of use
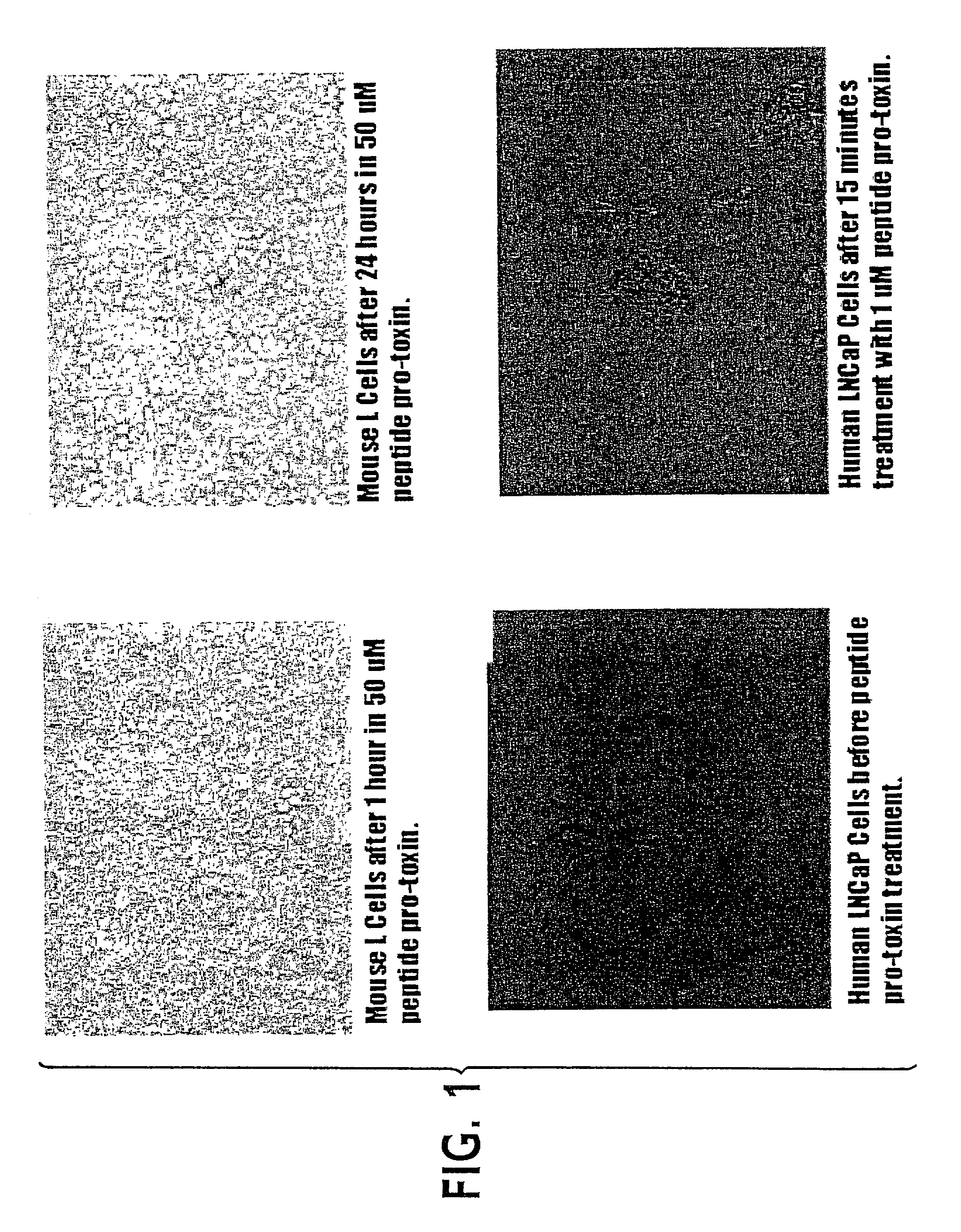
InactiveUS6635740B1Prevents sexual maturationInhibition of maturationHormone peptidesPeptide/protein ingredientsLytic peptideAbnormal tissue growth
Amphipathic lytic peptides are ideally suited to use in a ligand / cytotoxin combination to specifically inhibit cells that are driven by or are dependent upon a specific ligand interaction; for example, to induce sterility or long-term contraception, or to attack tumor cells, or to selectively lyse virally-infected cells, or to attack lymphocytes responsible for autoimmune diseases. The peptides act directly on cell membranes, and need not be internalized. Administering a combination of gonadotropin-releasing hormone (GnRH) (or a GnRH agonist) and a membrane-active lytic peptide produces long-term contraception or sterilization in animals in vivo. Administering in vivo a combination of a ligand and a membrane-active lytic peptide kills cells with a receptor for the ligand. The compounds are relatively small, and are not antigenic. Lysis of gonadotropes has been observed to be very rapid (on the order of ten minutes.) Lysis of tumor cells is rapid. The two components-the ligand and the lytic peptide-may optionally be administered as a fusion peptide, or they may be administered separately, with the ligand administered slightly before the lytic peptide, to activate cells with receptors for the ligand, and thereby make those cells susceptible to lysis by the lytic peptide. The compounds may be used in gene therapy to treat malignant or non-malignant tumors, and other diseases caused by clones or populations of "normal" host cells bearing specific receptors (such as lymphocytes), because genes encoding a lytic peptide or encoding a lytic peptide / peptide hormone fusion may readily be inserted into hematopoietic stem cells or myeloid precursor cells.
Owner:BOARD OF SUPERVISORS OF LOUISIANA STATE UNIV & AGRI & MECHANICAL COLLEGE
Virus derived antimicrobial peptides
InactiveUS6887847B2Lowering molar concentration requiredMinimal toxicityAntibacterial agentsBiocideBacteroidesLytic peptide
The invention is directed to peptides having antimicrobial activity (antimicrobial peptides). The antimicrobial peptides of the present invention are analogs of the Lentivirus Lytic Peptide 1 (LLP1) amino acid sequence. The invention is further directed to peptides referred to as the Lytic Base Unit (LBU) peptides derived from the LLP1 analogs, also having antimicrobial activity. In addition, the present invention is also directed to methods of using the peptides in a variety of contexts, including the treatment or prevention of infectious diseases. The antimicrobial LLP1 analog peptides and the LBU peptides (collectively eLLPs) may be highly active under high salt conditions and in biologic fluids. In addition, the eLLPs are effective when presented either in soluble form, or when attached to a solid surface. Furthermore, the peptides of the present invention are selectively active against a wide variety of bacterial pathogens and exhibit minimal toxicity to eukaryotic cells in vitro and in vivo.
Owner:UNIVERSITY OF PITTSBURGH
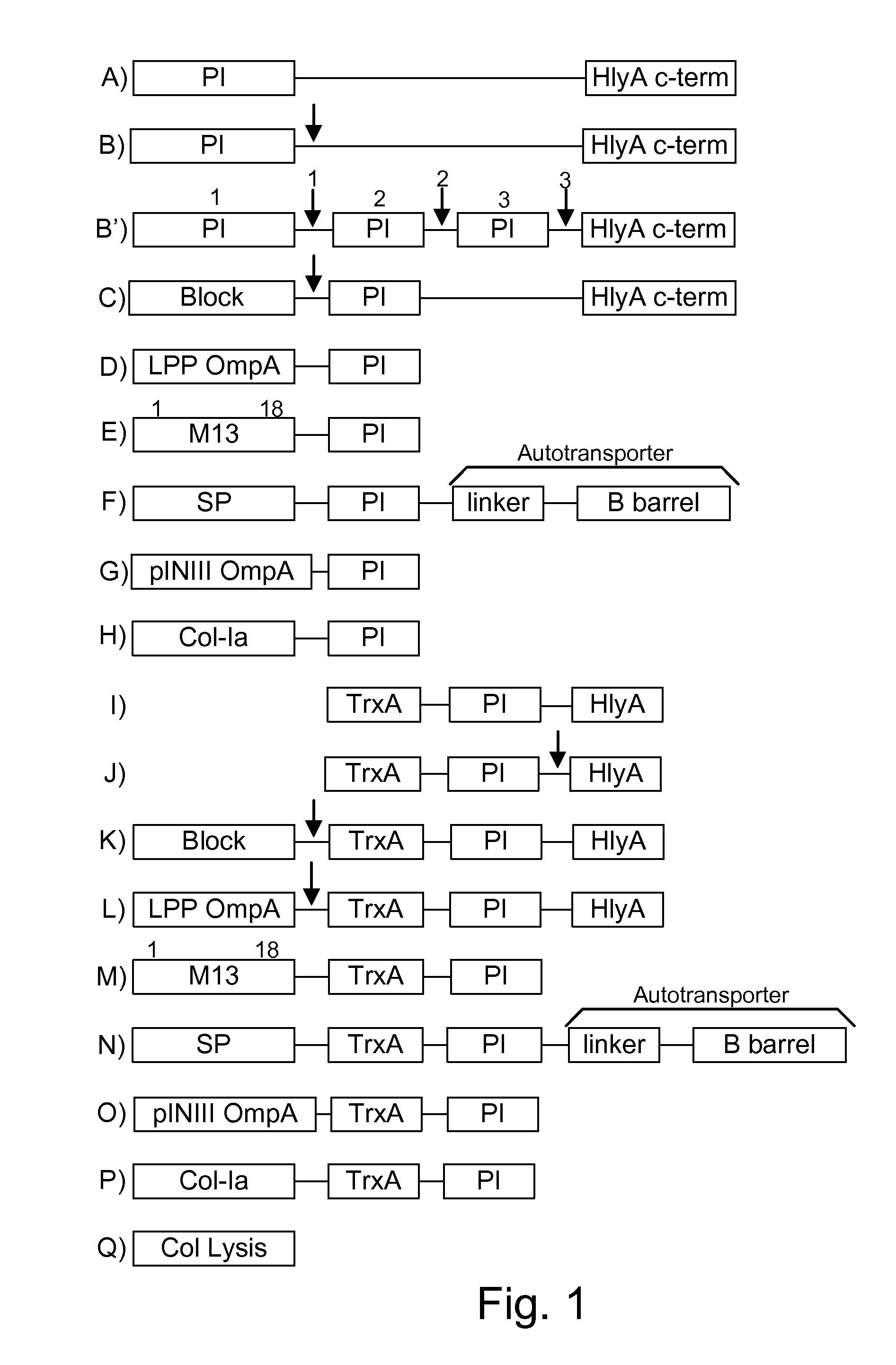
Immunization and/or treatment of parasites and infectious agents by live bacteria
ActiveUS8771669B1Reducing eliminatingReducing or eliminating the targeted parasite, infectious diseaseVirusesBacteriaLytic peptideHuntingtons chorea
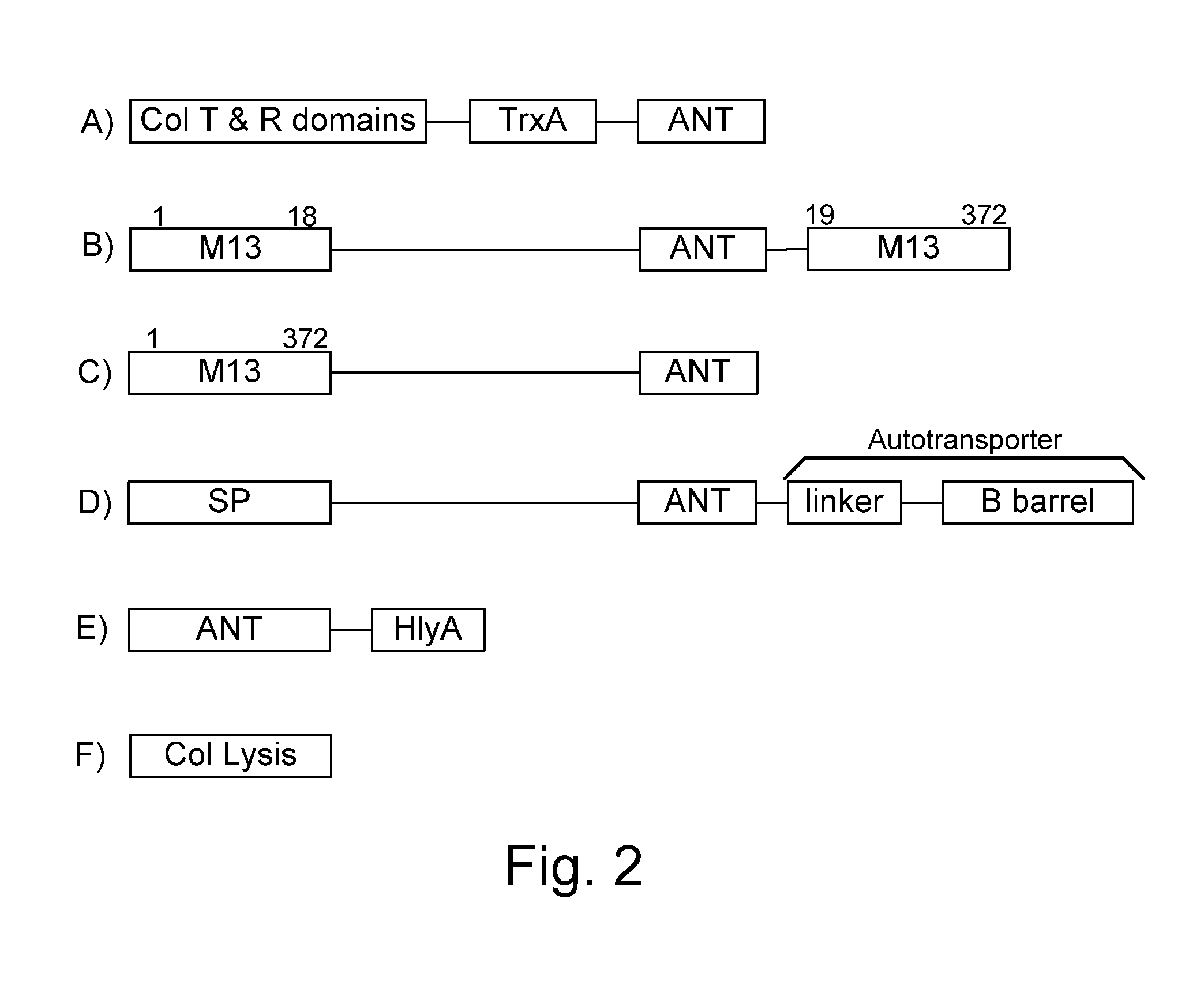
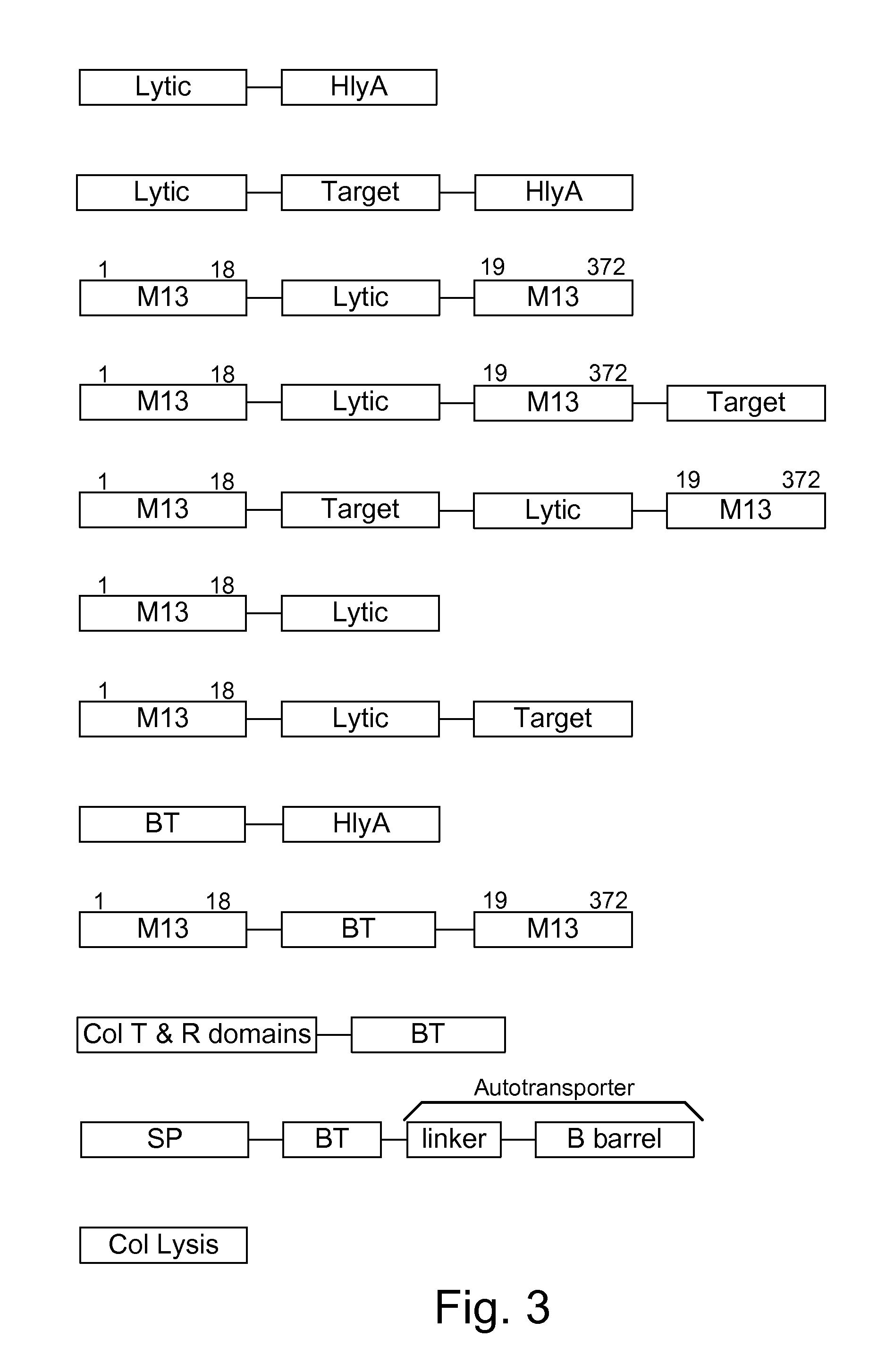
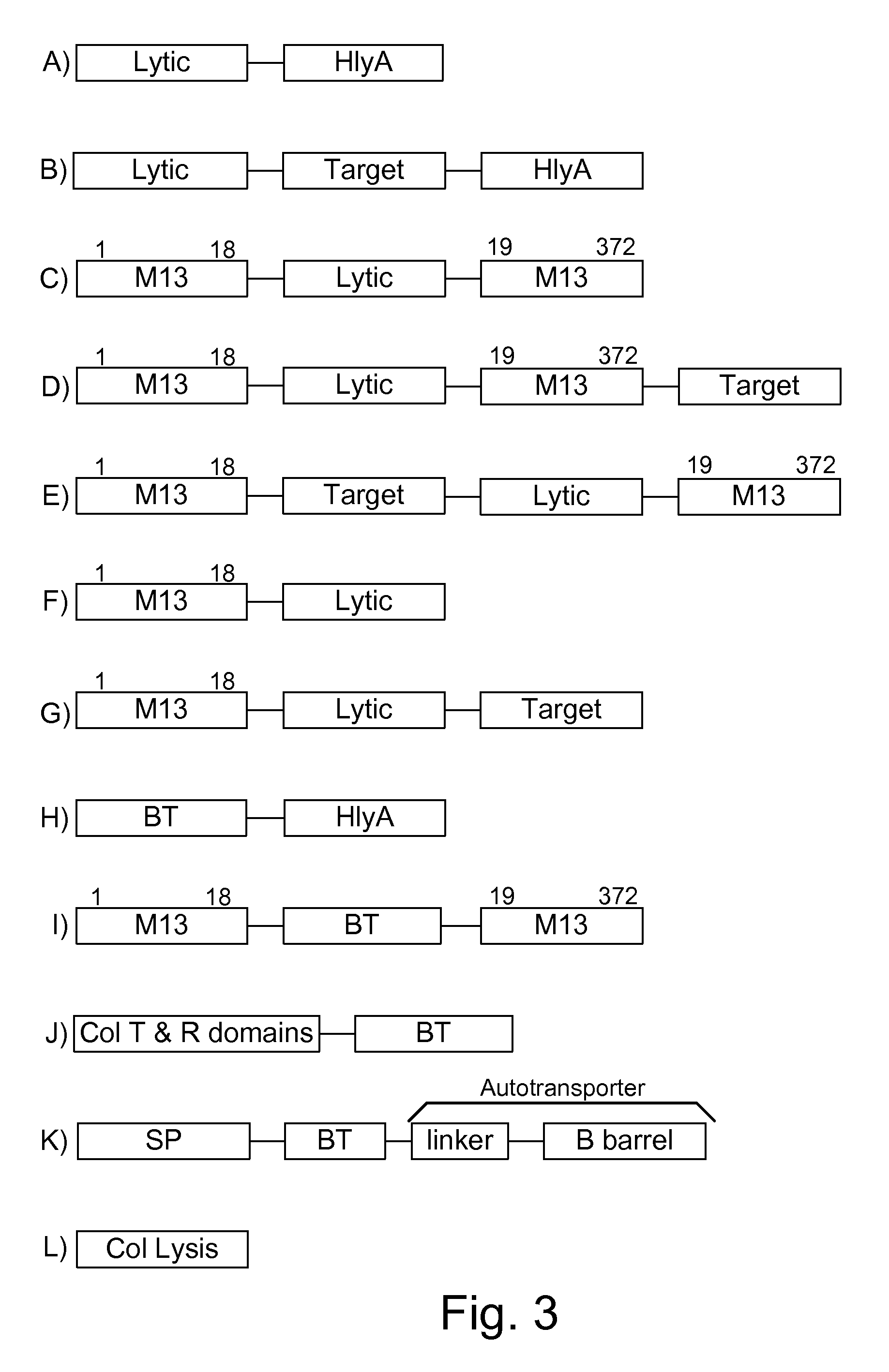
Chimeric proteins are expressed, secreted or released by a bacterium to immunize against or treat a parasite, infectious disease or malignancy. The delivery vector may also be attenuated, non-pathogenic, low pathogenic, or a probiotic bacterium. The chimeric proteins include chimeras of, e.g., phage coat and / or colicin proteins, bacterial toxins and / or enzymes, autotransporter peptides, lytic peptides, multimerization domains, and / or membrane transducing (ferry) peptides. The active portion of the immunogenic chimeric proteins can include antigens against a wide range of parasites and infectious agents, cancers, Alzheimer's and Huntington's diseases, and have enhanced activity when secreted or released by the bacteria, and / or have direct anti-parasite or infectious agent activity. The activity of the secreted proteins is further increased by co-expression of a protease inhibitor that prevents degradation of the effector peptides. Addition of an antibody binding or antibody-degrading protein further prevents the premature elimination of the vector and enhances the immune response.
Owner:BERMUDES DAVID G DR
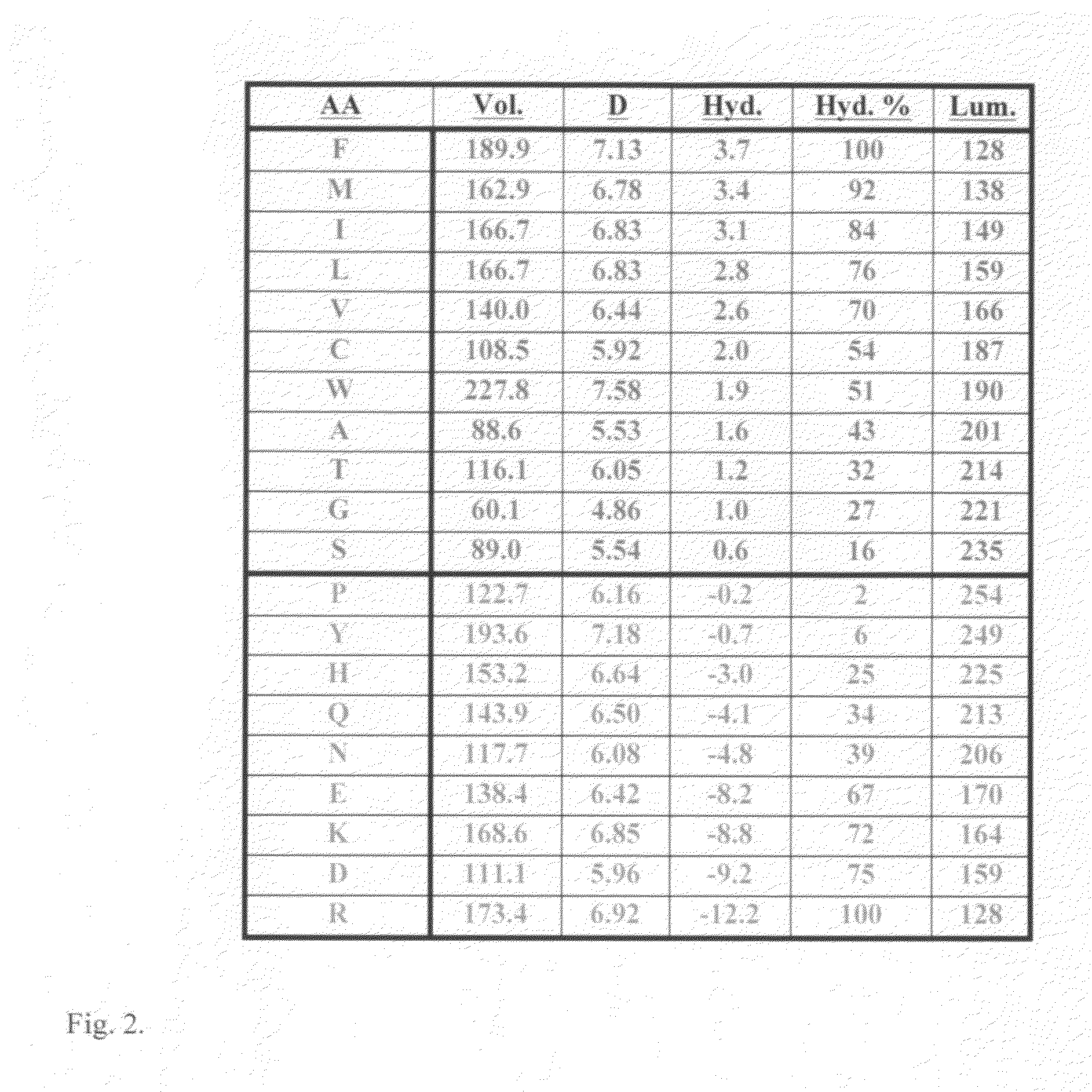
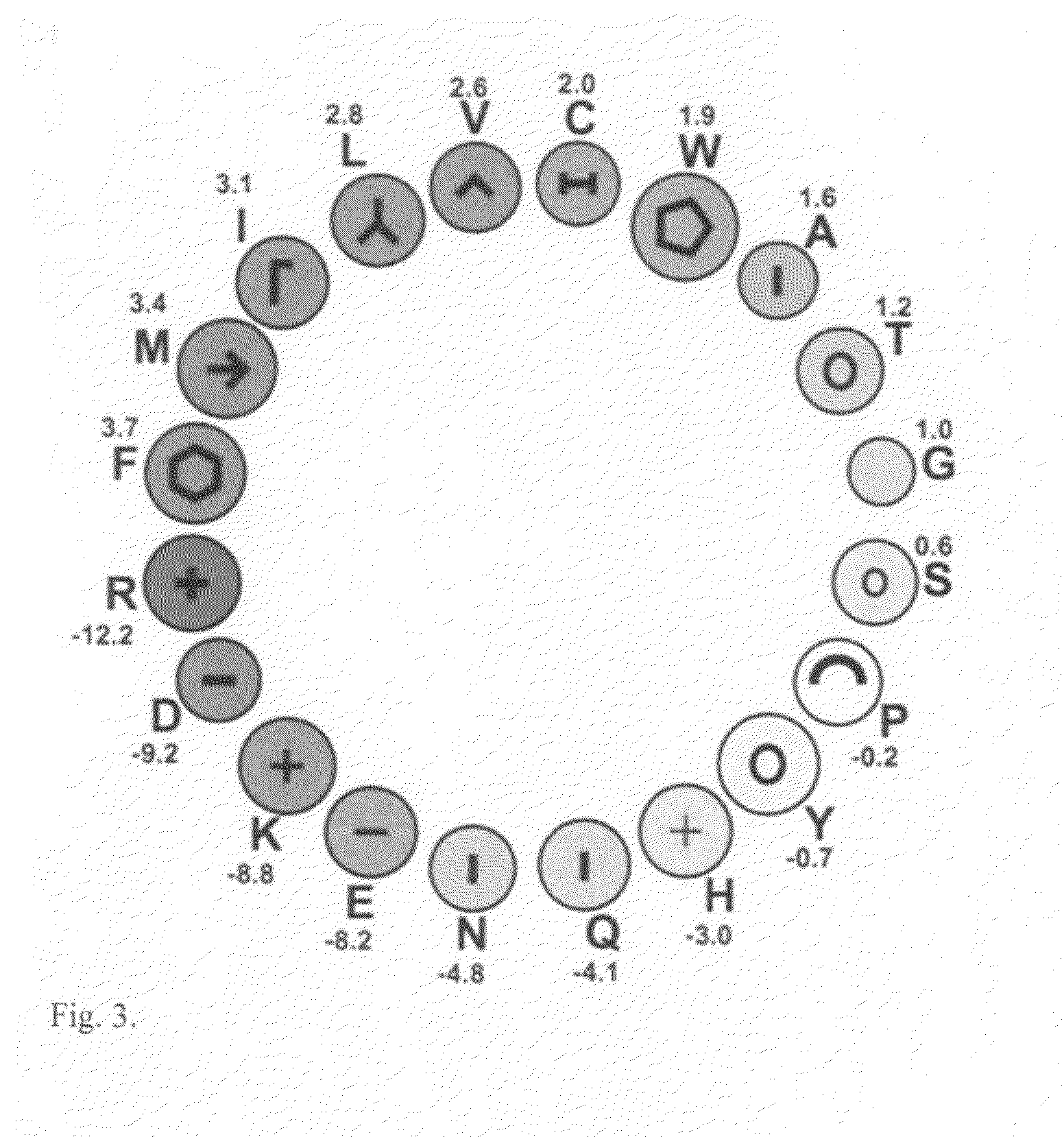
Non-naturally occurring synthetic lytic peptides
InactiveUS6559281B1Improve the immunityImprove stabilityPeptide-nucleic acidsPeptide/protein ingredientsLytic peptideCrystallography
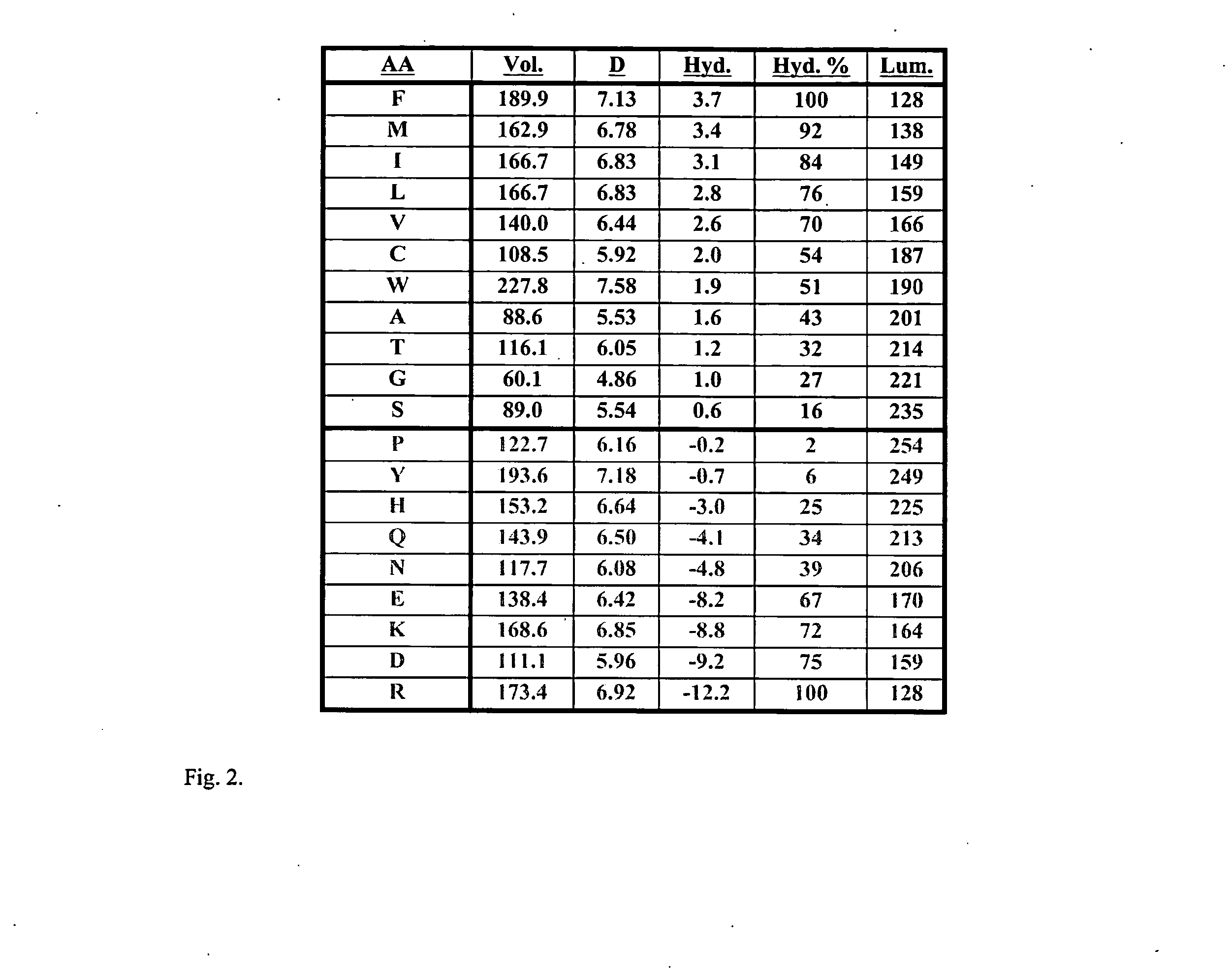
Non-naturally occurring lytic peptides which contain a phenylalanine residue and one or more alanine, valine and lysine residues, and optionally contain chemically masked cysteine or serine residues possess an amphipathic structure which allows them to promote cell lysis in certain pathologic organisms, and particularly in prokaryotes. Peptides having a beta-pleated sheet secondary structure and lacking cysteine residues form one embodiment of these lytic peptides.
Owner:DEMEGEN
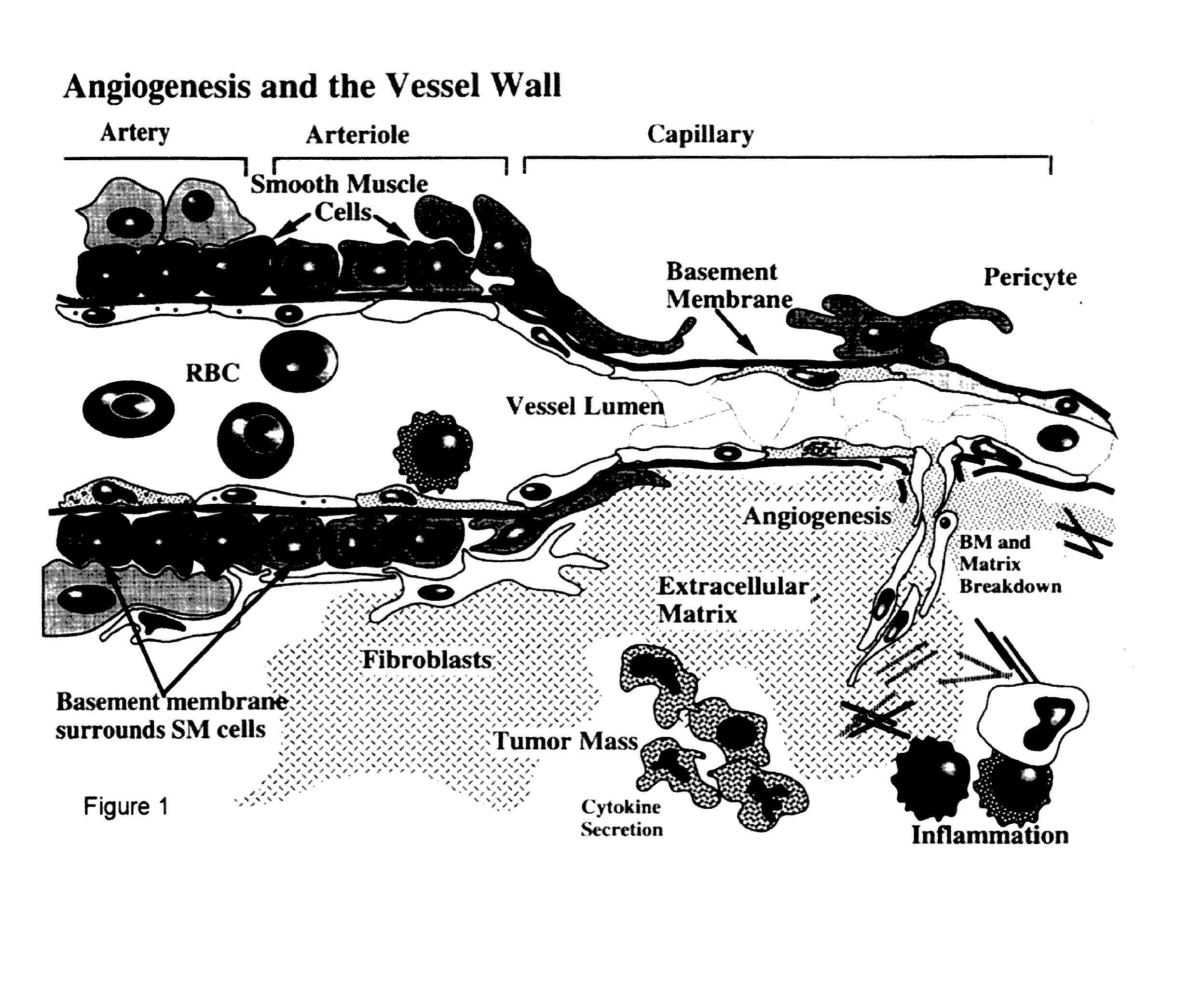
Anti-angiogenic peptides and their uses
InactiveUS20120270770A1Enhance and inhibit angiogenesisEffective therapySenses disorderNervous disorderLytic peptideDisease
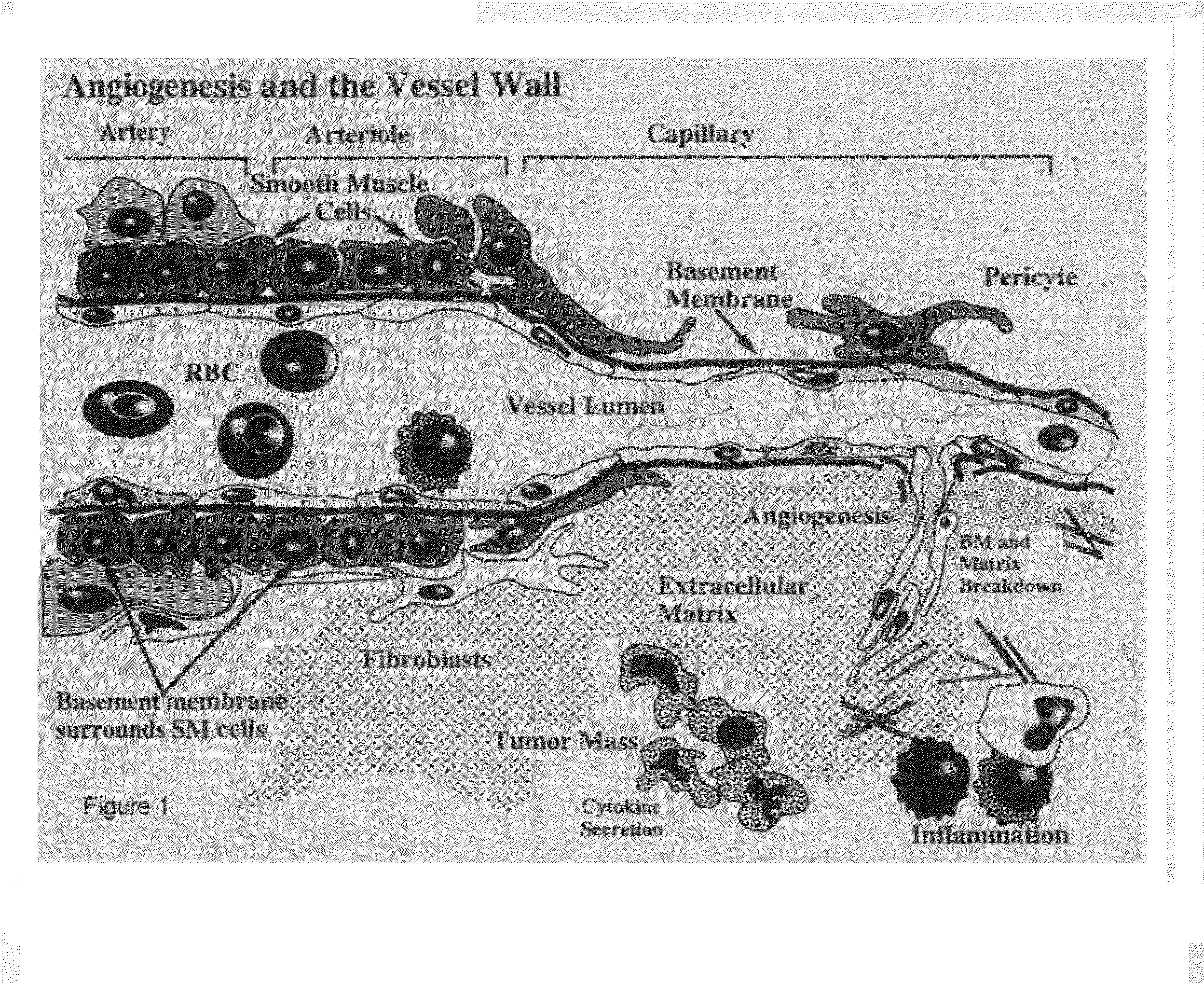
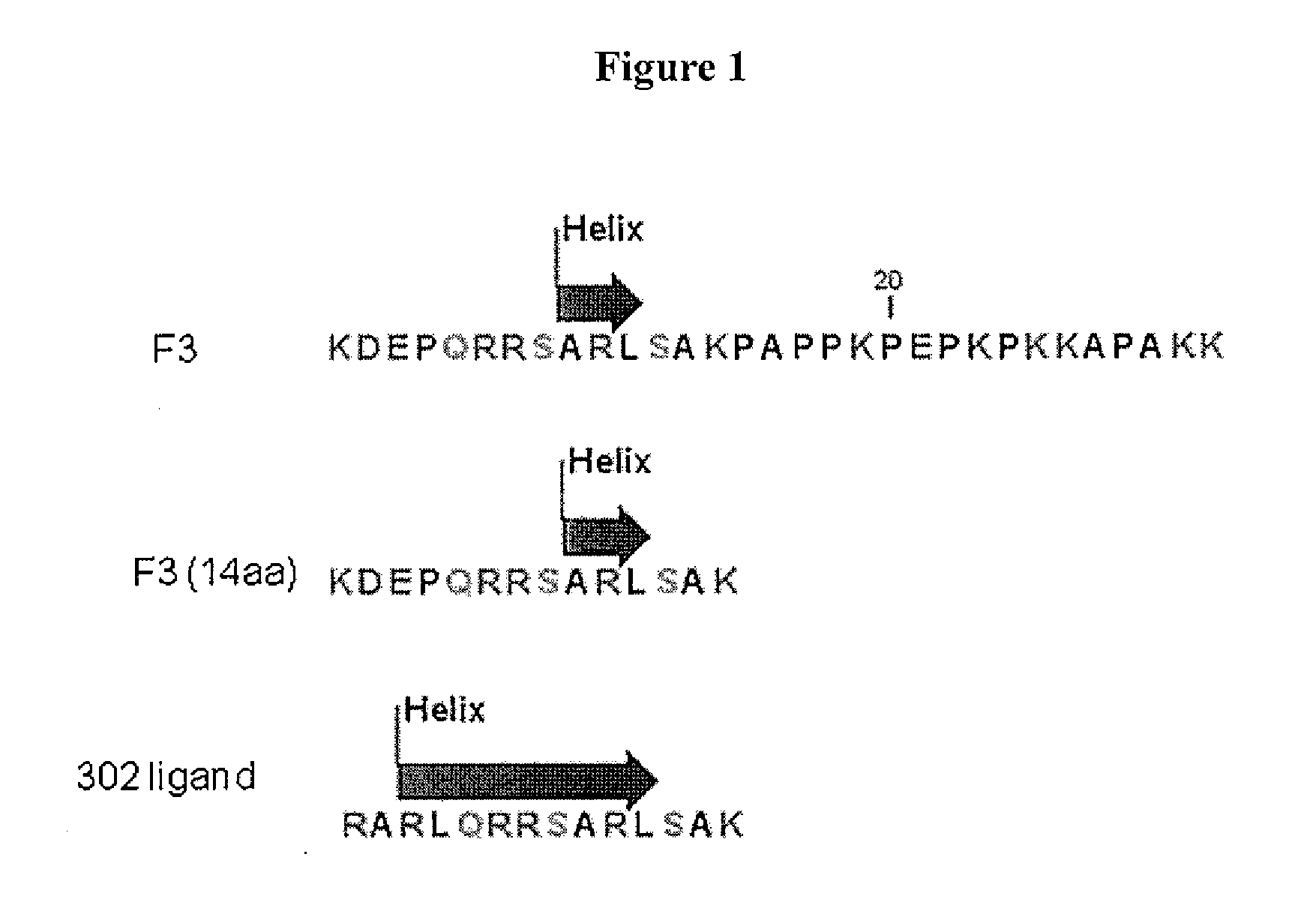
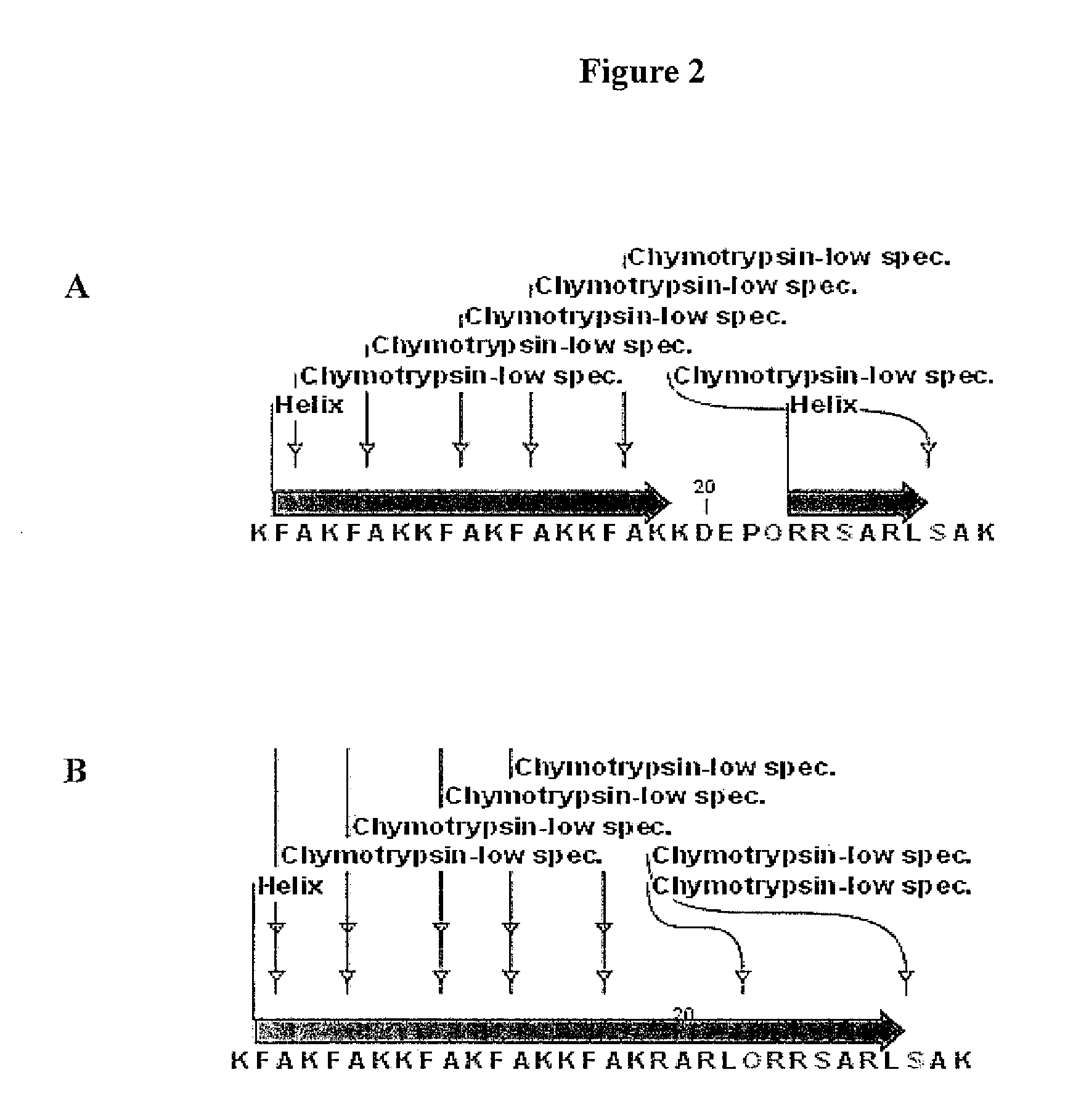
This invention relates to novel synthetic lytic peptide fragments of full-length peptides with the capacity to modulate angiogenic activity in mammals. The invention also relates to the use of such peptides in pharmaceutical compositions and in methods for treating diseases or disorders that are associated with angiogenic activity.
Owner:JAYNES JESSE MICHAEL
Ligand/lytic peptide compositions and methods of use
InactiveUS20040018967A1Inhibition of maturationLysis of tumor cells is rapidAntibacterial agentsOrganic active ingredientsLytic peptideAutoimmune condition
Amphipathic lytic peptides are ideally suited to use in a ligand / cytotoxin combination to specifically inhibit cells that are driven by or are dependent upon a specific ligand interaction; for example, to induce sterility or long-term contraception, or to attack tumor cells, or to selectively lyse virally-infected cells, or to attack lymphocytes responsible for autoimmune diseases. The peptides act directly on cell membranes, and need not be internalized. Administering a combination of gonadotropin-releasing hormone (GnRH) (or a GnRH agonist) and a membrane-active lytic peptide produces long-term contraception or sterilization in animals in vivo. Administering in vivo a combination of a ligand and a membrane-active lytic peptide kills cells with a receptor for the ligand. The compounds are relatively small, and are not antigenic. Lysis of gonadotropes has been observed to be very rapid (on the order of ten minutes.) Lysis of tumor cells is rapid. The two components-the ligand and the lytic peptide-may optionally be administered as a fusion peptide, or they may be administered separately, with the ligand administered slightly before the lytic peptide, to activate cells with receptors for the ligand, and thereby make those cells susceptible to lysis by the lytic peptide. The compounds may be used in gene therapy to treat malignant or non-malignant tumors, and other diseases caused by clones or populations of "normal" host cells bearing specific receptors (such as lymphocytes), because genes encoding a lytic peptide or encoding a lytic peptide / peptide hormone fusion may readily be inserted into hematopoietic stem cells or myeloid precursor cells.
Owner:BOARD OF SUPERVISORS OF LOUISIANA STATE UNIV & AGRI & MECHANICAL COLLEGE
Layered Nanoparticles for Sustained Release of Small Molecules
InactiveUS20090061006A1Strong specificityHigh selectivityPowder deliveryPeptide/protein ingredientsLytic peptideSide effect
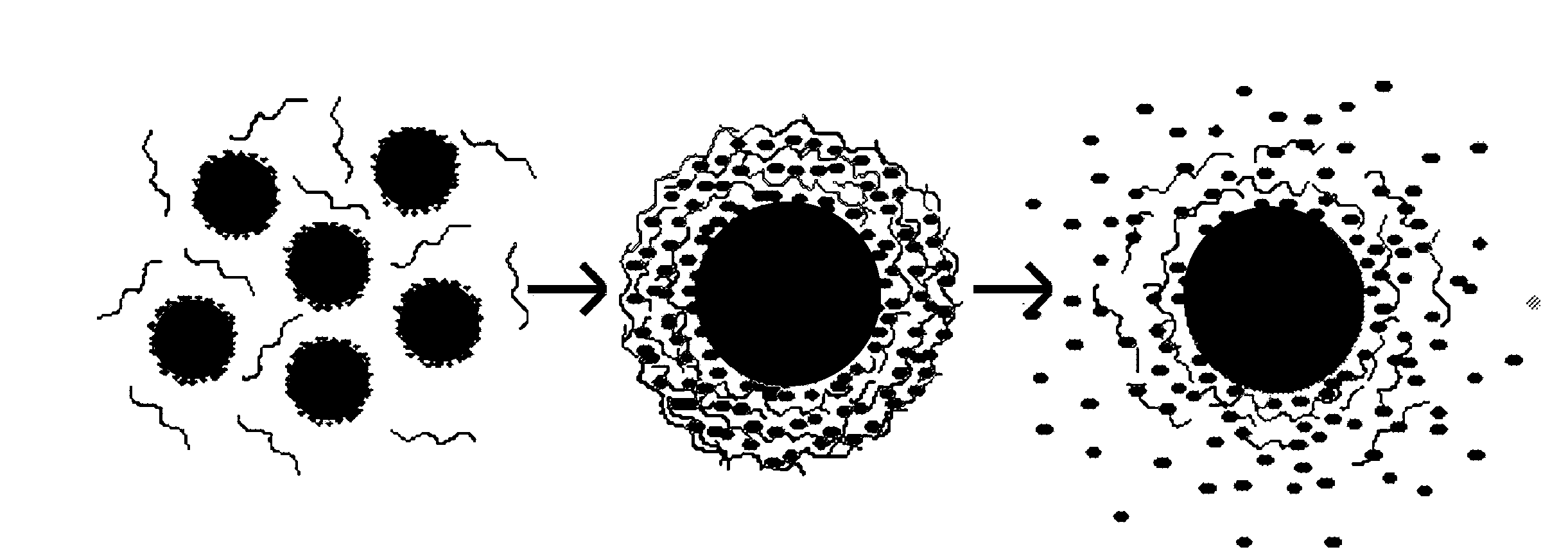
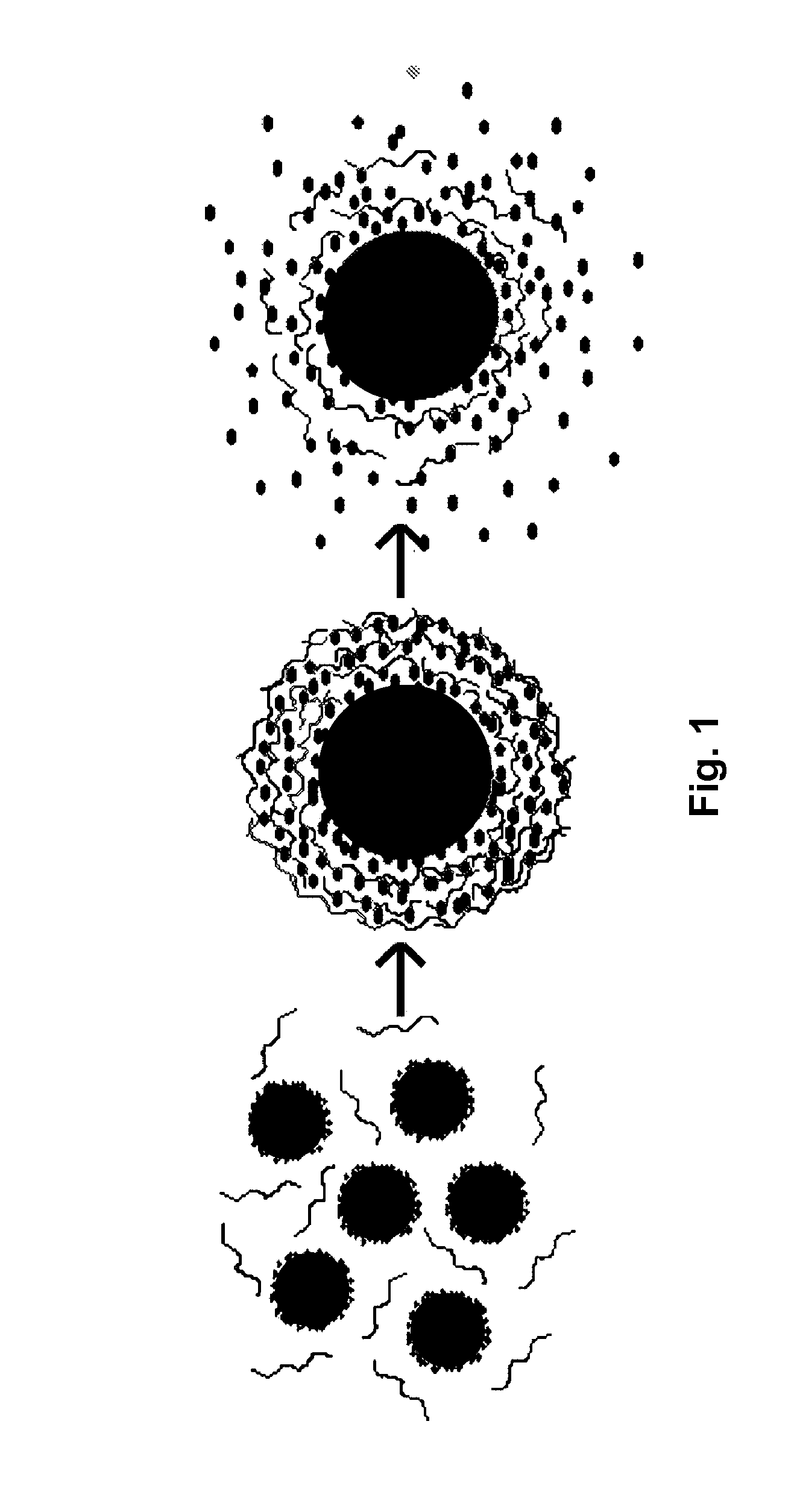
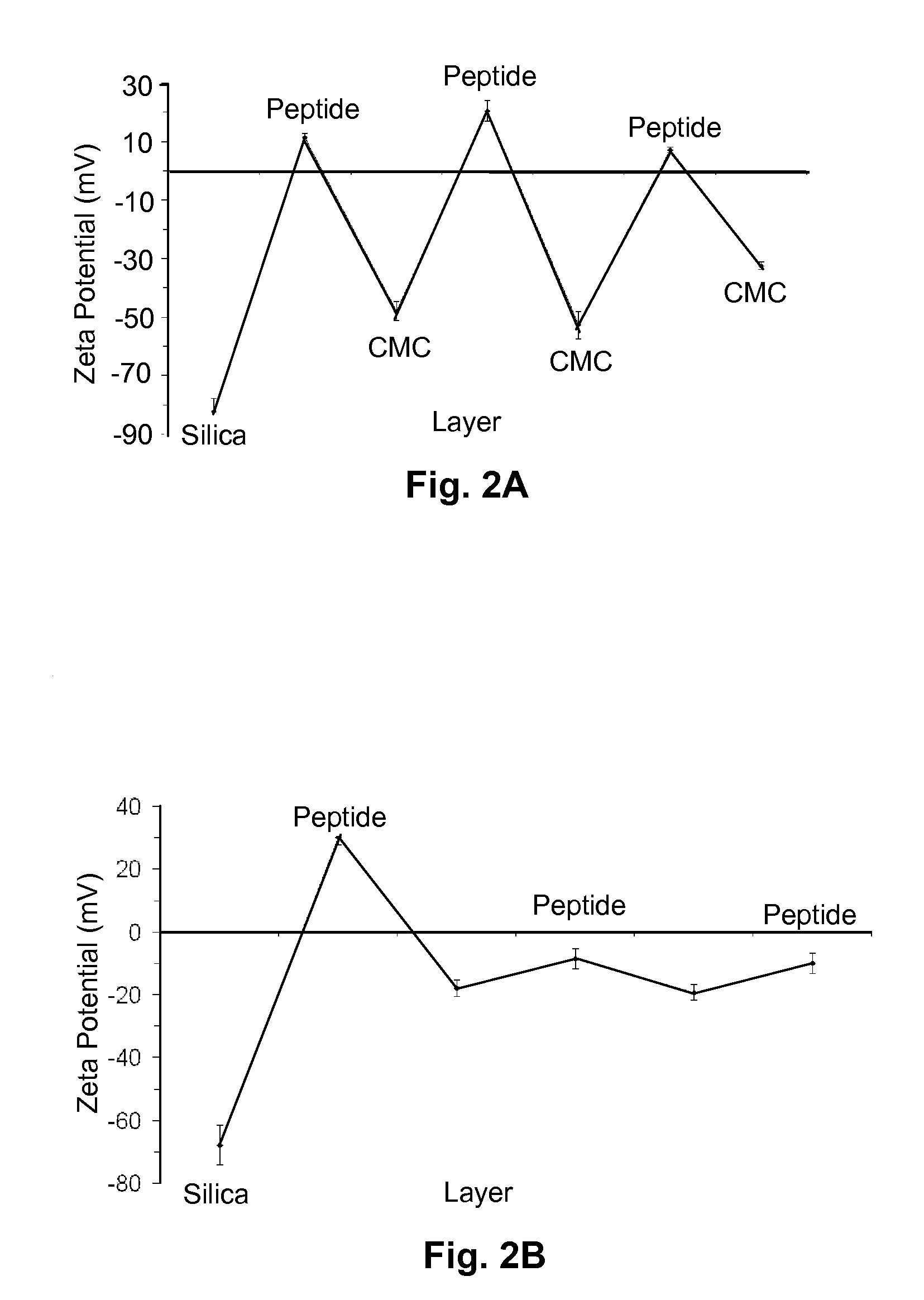
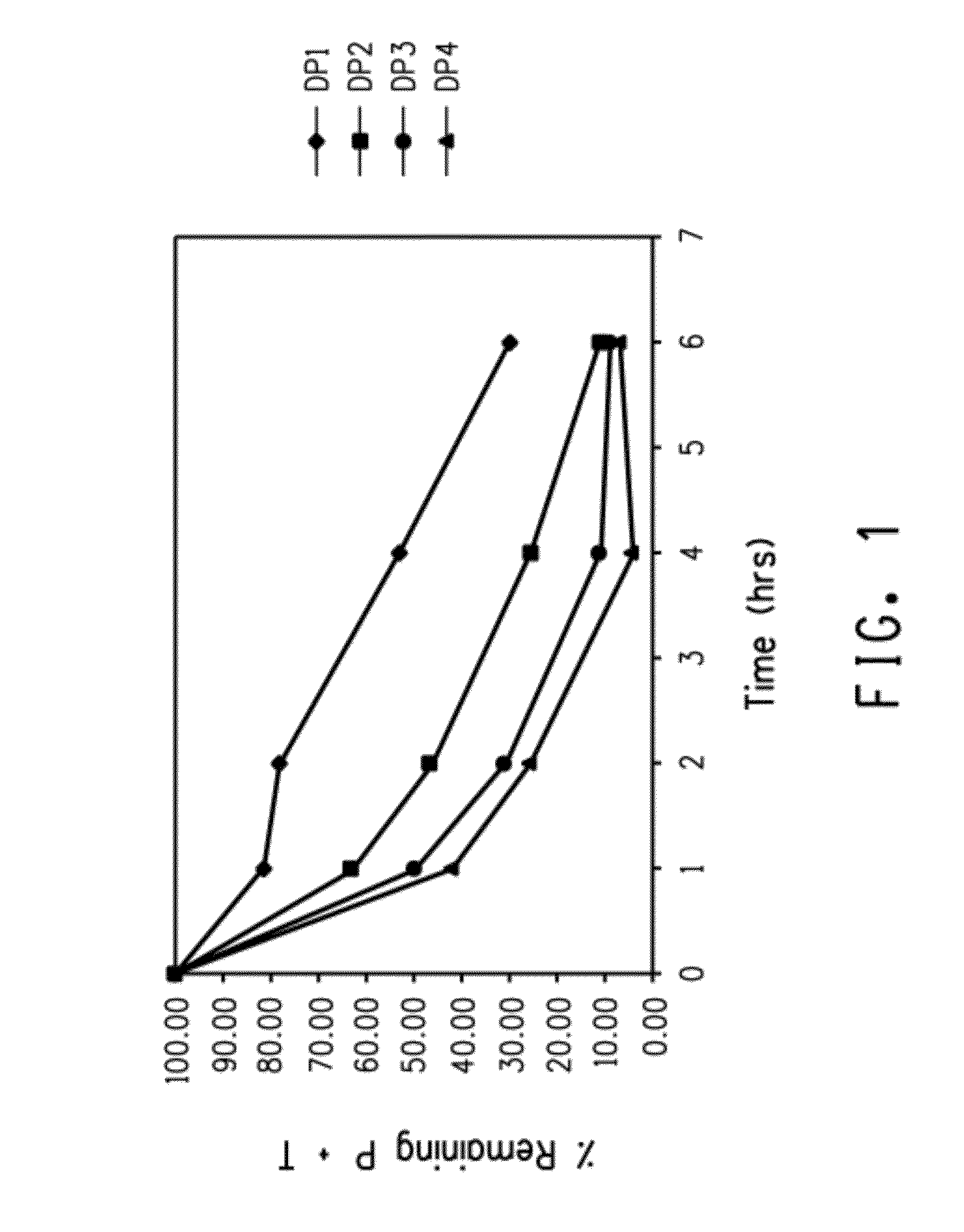
Nanoparticle compositions and methods are disclosed for the sustained release of small molecules, such as pharmaceutical compounds in vivo, for example ligand-lytic peptide conjugates. The construction of the nanoparticles helps to prevent self-aggregation of the molecules, and the consequent loss of effectiveness. The system employs layer-by-layer self-assembly of biocompatible polyelectrolyte layers, and layers of charged small molecules such as drug molecules, to form a multilayer nanoparticle in which the drug or other small molecule itself acts as one of the alternating charged layers in the multilayer assembly. The small molecules can then be released over time in a sustained manner. The LbL nano-assemblies can specifically target cancers, metastases, or other diseased tissues, while minimizing side effects.
Owner:BOARD OF SUPERVISORS OF LOUISIANA STATE UNIV & AGRI & MECHANICAL COLLEGE +1
Compositions and methods for contraception in or sterilization of mammals
InactiveUS6680058B1Prevents sexual maturationInhibition of maturationHormone peptidesPeptide/protein ingredientsLytic peptideMammal
Amphipathic lytic peptides are ideally suited to use in a ligand / cytotoxin combination to induce sterility or long-term contraception in mammals. The peptides act directly on cell membranes, and need not be internalized. Administering a combination of gonadotropin-releasing hormone (GnRH) (or a GnRH agonist) and a membrane-active lytic peptide produces long-term contraception or sterilization in mammals in vivo. The compounds are relatively small, and are not antigenic. Lysis of gonadotropes has been observed to be very rapid (on the order of ten minutes.) The two components-the ligand and the lytic peptide-may optionally be administered as a fusion peptide, or they may be administered separately, with the ligand administered slightly before the lytic peptide, to activate cells with receptors for the ligand, and thereby make those cells susceptible to lysis by the lytic peptide.
Owner:BOARD OF SUPERVISORS OF LOUISIANA STATE UNIV & AGRI & MECHANICAL COLLEGE
Acid-cleavable linkers exhibiting altered rates of acid hydrolysis
InactiveUS8609621B2Improve efficiencyIncrease hydrolysis ratePolypeptide with localisation/targeting motifBacteriaCrystallographyAcid hydrolysis
Owner:DUPONT US HLDG LLC
Immunization and/or treatment of parasites and infectious agents by live bacteria
ActiveUS9486513B1Reducing or eliminating the targeted parasite, infectious diseaseSsRNA viruses negative-sensePeptide/protein ingredientsHuntingtons choreaAntiparasitic
Chimeric proteins are expressed, secreted or released by a bacterium to immunize against or treat a parasite, infectious disease or malignancy. The delivery vector may also be attenuated, non-pathogenic, low pathogenic, or a probiotic bacterium. The chimeric proteins include chimeras of, e.g., phage coat and / or colicin proteins, bacterial toxins and / or enzymes, autotransporter peptides, lytic peptides, multimerization domains, and / or membrane transducing (ferry) peptides. The active portion of the immunogenic chimeric proteins can include antigens against a wide range of parasites and infectious agents, cancers, Alzheimer's and Huntington's diseases, and have enhanced activity when secreted or released by the bacteria, and / or have direct anti-parasite or infectious agent activity. The activity of the secreted proteins is further increased by co-expression of a protease inhibitor that prevents degradation of the effector peptides. Addition of an antibody binding or antibody-degrading protein further prevents the premature elimination of the vector and enhances the immune response.
Owner:BERMUDES DAVID GORDON
Preparation method and application of functional photoacoustic probe based on assembly of gold nanoparticles
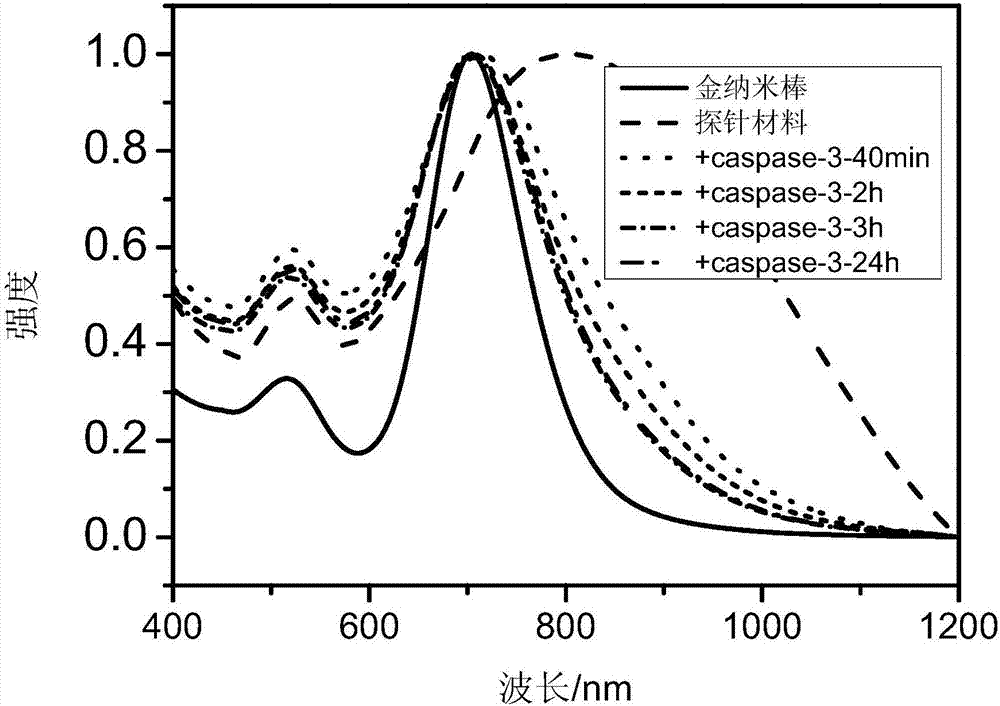
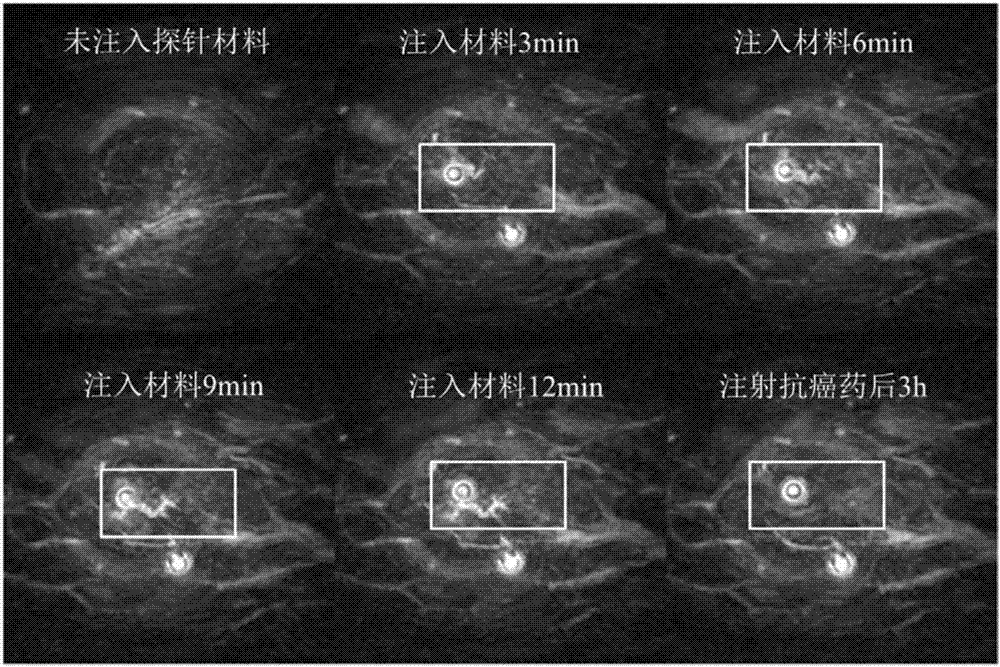
InactiveCN106924764AReach UV absorptionEchographic/ultrasound-imaging preparationsMicrocapsulesChemistryBiotin-streptavidin complex
The invention relates to the field of nano materials, in particular to a preparation method and application of a functional photoacoustic probe based on assembly of gold nanoparticles. The preparation method of the functional photoacoustic probe includes the steps that peptide chains with one ends of -SH, the other ends of biotin and the middle parts of specific sequences DEVD are used for modification of the surfaces of the gold nanoparticles, meanwhile, functional micromolecules with one ends of -SH and the other ends of streptavidin are used for modification of the surfaces of gold nanorods, and the self-assembly process of the gold nanorods and the gold nanoparticles is completed by the specific biological coupling reaction of the streptavidin and the biotin. As an apoptosis enzyme caspase-3 can recognize and lyse the specific sequences DEVD of the peptide chains, the gold nanorods are separated from the gold nanoparticles, so that ultraviolet-visible absorption spectra and photoacoustic imaging signal strength begin to change; thus production of caspase-3 in tumor cells can be monitored with the method of photoacoustic imaging so as to judge the apoptosis process of the tumor cells after treatment, and the functional photoacoustic probe is also used for evaluating the effects of radiotherapy and chemotherapy, which has great significance in clinical medicine and disease diagnosis.
Owner:NANJING UNIV OF POSTS & TELECOMM
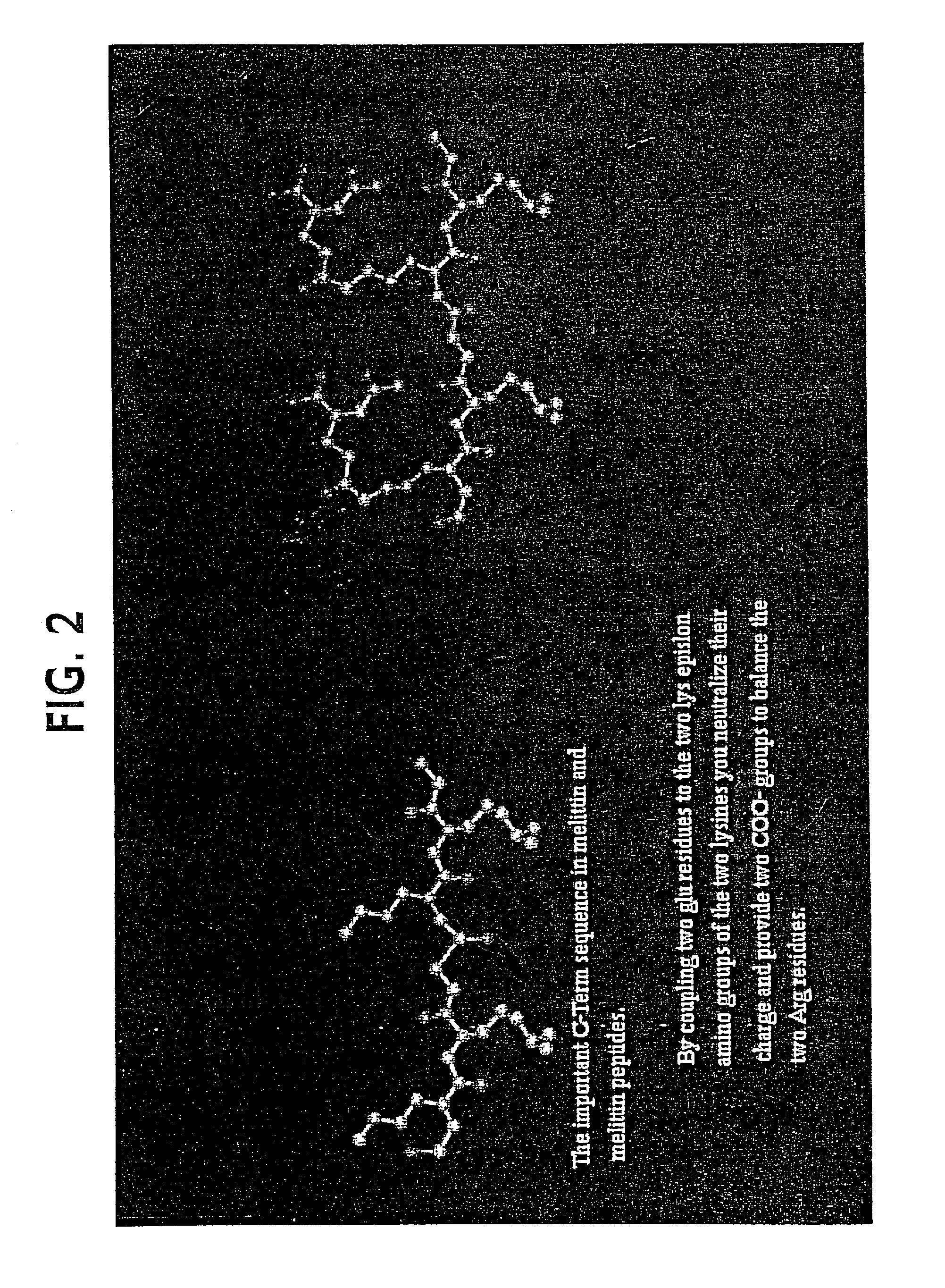
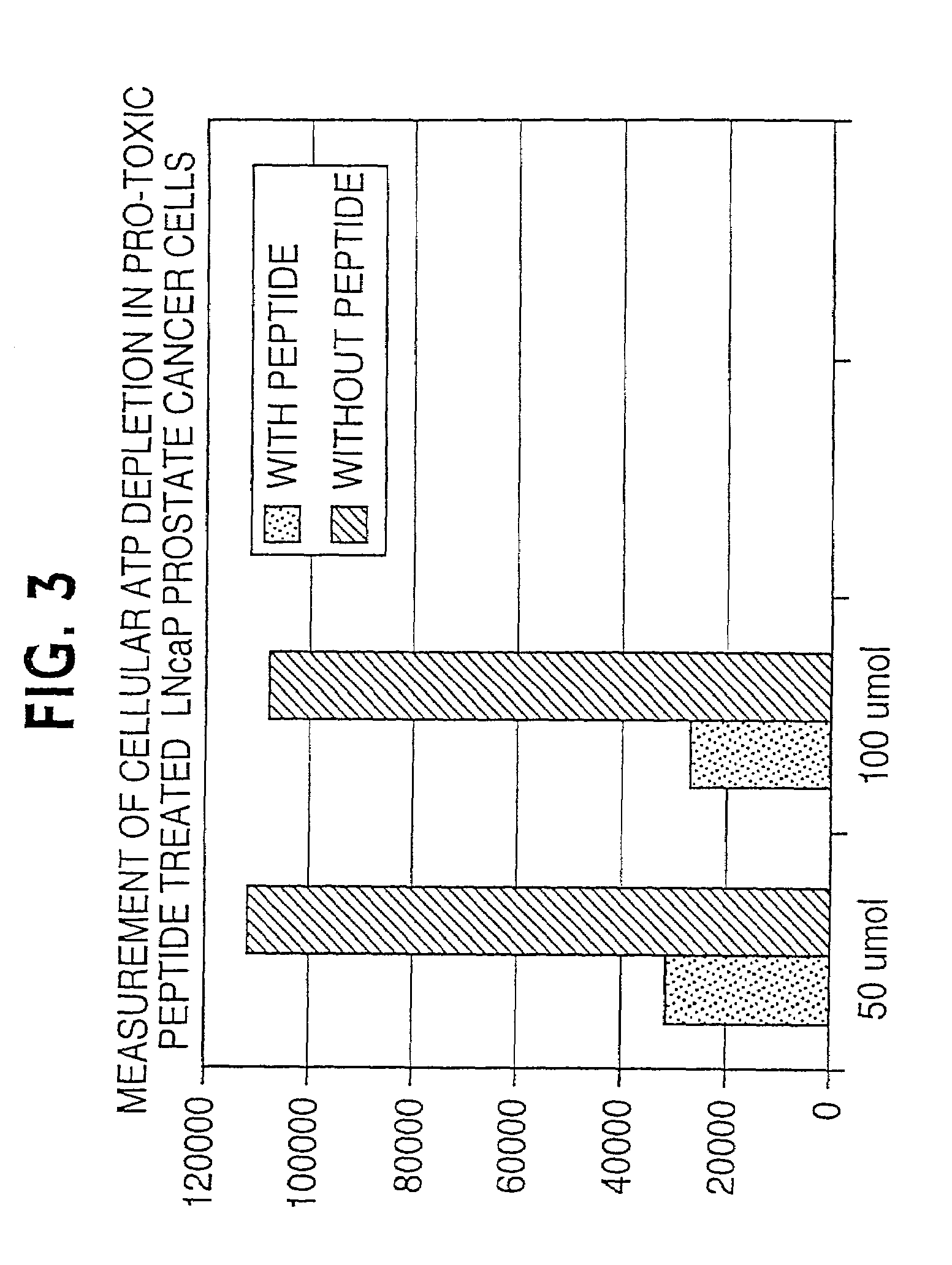
Lytic peptide prodrugs
InactiveUS7456146B2Peptide/protein ingredientsImmunoglobulins against animals/humansLytic peptideProteinase activity
A cytotoxin can be rendered non-toxic by charge neutralizing the amino acids salient to pore assembly and / or sterically inhibiting formation of the peptide's active conformation. In the presence of specific proteases, the inactive peptide or procytotoxin can be activated to assemble into its lytic conformation and selectively destroy a target cell.
Owner:ORBIS HEALTH SOLUTIONS
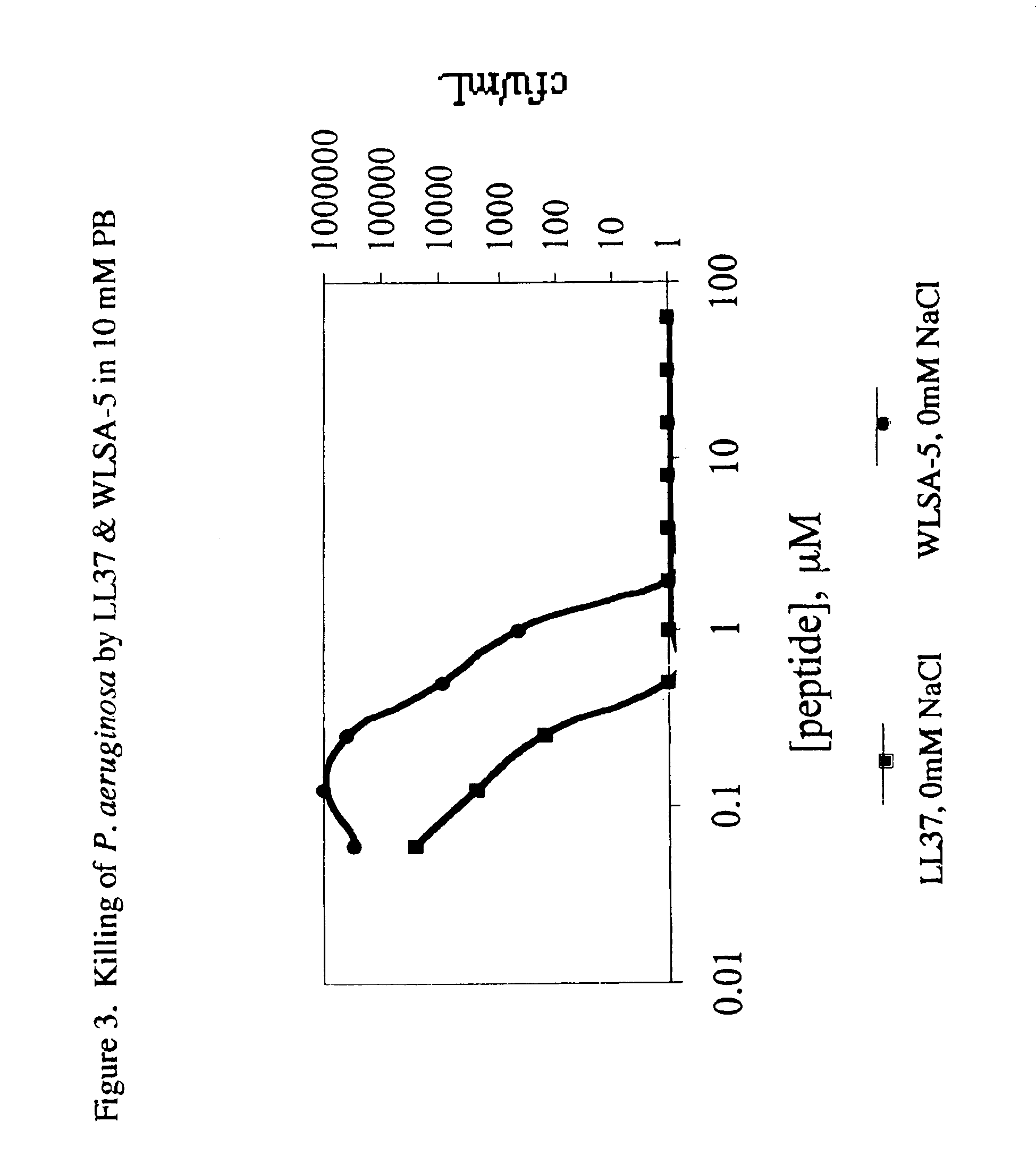
Antimicrobial lytic peptides
This invention relates to novel lytic peptides which are effective in treating citrus plant diseases including canker and greening. The invention also relates to antimicrobial compositions comprising lytic peptides and the use of such compositions in methods for treating diseases of citrus plants.
Owner:GENVOR INC
Acid-resistant solubility tag for the expression and purification of bioactive peptides
InactiveUS20090029420A1Improve acid resistanceHigh expressionBacteriaPeptide/protein ingredientsSolubilityInclusion bodies
An acid-resistant peptide solubility tag (an “inclusion body tag”) is provided that is effective in producing peptides of interest in an insoluble form. Fusion peptide constructs comprising the inclusion body tag fused to a peptide of interest are provided. An acid cleavable peptide moiety separates the inclusion body tag from the peptide of interest so that acid hydrolysis can be used during subsequent processing steps to separate the tag from the desired peptide of interest. The present inclusion body tag's resistance to acid hydrolysis facilitates easier and cleaner separation of the peptide of interest after acid hydrolysis. Specifically, a ketosteroid isomerase-derived inclusion body tag is provided that has been engineered to be more resistant to acid hydrolysis.
Owner:EI DU PONT DE NEMOURS & CO
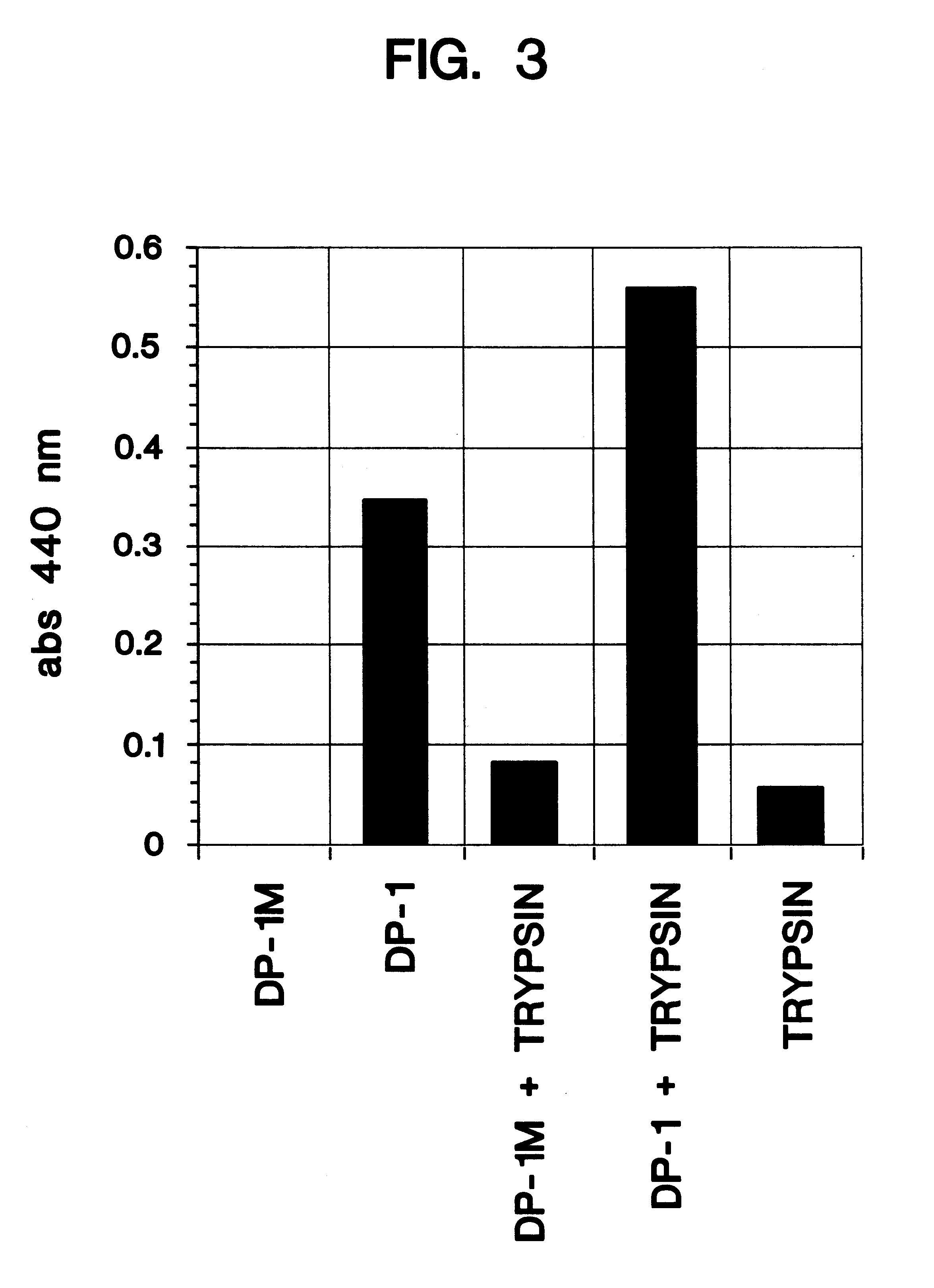
Angiogenic active lytic peptides
ActiveUS20140329753A1Enhance and inhibit angiogenesisEffective therapyPeptide/protein ingredientsDigestive systemLytic peptideDisease
This invention relates to novel synthetic lytic peptide fragments of full-length peptides with the capacity to modulate angiogenic activity in mammals. The invention also relates to the use of such peptides in pharmaceutical compositions and in methods for treating diseases or disorders that are associated with angiogenic activity.
Owner:ISSAR PHARMA LTD
Nucleolin-binding peptides, nucleolin- binding lytic peptides, fusion constructs and methods of making and using same
InactiveUS20110124564A1Reducing and decreasing and durationReducing and decreasing and and frequencyPeptide/protein ingredientsAntibody mimetics/scaffoldsAntibody conjugateBinding peptide
The invention relates to nucleolin binding peptides, nucleolin binding peptides and anti-nucleolin antibody conjugates with cytotoxic activity, fusion constructs, methods of using nucleolin binding peptides and antibodies and fusion constructs thereof, and methods of treating various disorders, undesirable conditions and diseases treatable with nucleolin binding peptides and fusion constructs, such as undesirable or aberrant cell proliferation (hyperproliferation) or hyperproliferative disorders, including tumors, cancers, neoplasia and malignancies, angiogenesis related or dependent diseases, and inflammatory diseases and inflammation.
Owner:A28 THERAPEUTICS INC
Immunization and/or treatment of parasites and infectious agents by live bacteria
ActiveUS10364435B1Reducing or eliminating the targeted parasite, infectious diseaseSsRNA viruses negative-sensePeptide/protein ingredientsBacteroidesLytic peptide
Chimeric proteins are expressed, secreted or released by a bacterium to immunize against or treat a parasite, infectious disease or malignancy. The delivery vector may also be attenuated, non-pathogenic, low pathogenic, or a probiotic bacterium. The chimeric proteins include chimeras of, e.g., phage coat and / or colicin proteins, bacterial toxins and / or enzymes, autotransporter peptides, lytic peptides, multimerization domains, and / or membrane transducing (ferry) peptides. The active portion of the immunogenic chimeric proteins can include antigens against a wide range of parasites and infectious agents, cancers, Alzheimer's and Huntington's diseases, and have enhanced activity when secreted or released by the bacteria, and / or have direct anti-parasite or infectious agent activity. The activity of the secreted proteins is further increased by co-expression of a protease inhibitor that prevents degradation of the effector peptides. Addition of an antibody binding or antibody-degrading protein further prevents the premature elimination of the vector and enhances the immune response.
Owner:BERMUDES DAVID GORDON
Selective anticancer chimeric peptide comprising an EGF receptor-binding peptide and a cytotoxic peptide
It is an object of the present invention to provide a substance usable as an anticancer agent or DDS, which has intracellular stability, which is capable of evading side effects from functional disorder with respect to normal cells, or which has instantaneous effect. The inventors developed a novel chimeric peptide targeting cancer cells which overexpress EGFR or the like using a binding peptide such as a peptide sequence binding to EGFR, and a lytic peptide sequence, thereby solving such an object. Particularly, by using a chimeric peptide including an EGF receptor-binding peptide or the like and a cytotoxic peptide, this object was solved.
Owner:KAWAKAMI KOJI
Acid-clevable linkers exhibiting altered rates of acid hydrolysis
InactiveUS20120122153A1Improve efficiencyIncrease hydrolysis rateFungiBacteriaLytic peptideAcid hydrolysis
An acid-cleavable peptide linker comprising aspartic acid and proline residues is disclosed. The acid-cleavable peptide linker provides an altered sensitivity to acid-hydrolytic release of peptides of interest from fusion peptides of the formula PEP1-L-PEP2. The inventive linker, L, is described in various embodiments, each of which provides substantially more rapid acid-release of peptides of interest than does a single aspartic acid-proline pair. In an additional aspect, a method of increasing the stability of an acid cleavable linkage to acid hydrolysis is also provided.
Owner:DUPONT US HLDG LLC
Protease-activated receptor (par) modulator assays
There is provided a method of identifying a PAR cleaving molecule comprising the steps of: (a) providing an immobilized PAR cleavage peptide linked to a reporter molecule; (b) contacting the immobilized PAR cleavage peptide with a PAR cleaving molecule; and (c) detecting release of the reporter molecule.
Owner:VENTURE LENDING & LEASING IV
Selective anticancer chimeric peptide
It is an object of the present invention to provide a substance usable as an anticancer agent or DDS, which has intracellular stability, which is capable of evading side effects from functional disorder with respect to normal cells, or which has instantaneous effect. The inventors developed a novel chimeric peptide targeting cancer cells which overexpress EGFR or the like using a binding peptide such as a peptide sequence binding to EGFR, and a lytic peptide sequence, thereby solving such an object. Particularly, by using a chimeric peptide including an EGF receptor-binding peptide or the like and a cytotoxic peptide, this object was solved.
Owner:KAWAKAMI KOJI
Composition and method for the release of protected peptidesfrom a resin
InactiveCN101437838AReduce processing timeLess amount of reagentPeptide preparation methodsOrganic solventEvaporation
The present invention provides a composition and a method for cleaving a peptide from a solid support resin. Hydrochloric acid in an organic water miscible solvent is used to cleave the peptide-resin attachment. Optionally, trifluoroethanol or hexafluoroisopropanol may be added to the cleavage composition to improve results. When using the present cleavage composition, an evaporation or other step to remove carboxylic byproducts is not necessary following the cleavage reaction. After the resin is filtered out of the cleavage mixture, the peptide may be immediately precipitated with water.
Owner:MALLINCKRODT INC
Lytic-peptide-her2/neu (human epidermal growth factor receptor 2) ligand conjugates and methods of use
ActiveUS20130157961A1Reduce and inhibit spreadReduce and inhibit and disseminationSugar derivativesSaccharide peptide ingredientsLytic peptideHuman epidermal growth factor receptor
The invention relates to conjugates that bind to Her2 / neu, methods of using conjugates that bind to Her2 / neu and methods of treating undesirable or aberrant cell proliferation or hyperproliferative disorders, such as tumors, cancers, neoplasia and malignancies that express Her2 / neu.
Owner:A28 THERAPEUTICS INC
Lytic-peptide-Her2/neu (human epidermal growth factor receptor 2) ligand conjugates and methods of use
ActiveUS9586996B2Reducing and inhibiting and growthReducing and inhibiting proliferationSugar derivativesImmunoglobulins against cell receptors/antigens/surface-determinantsLytic peptideDisease
The invention relates to conjugates that bind to Her2 / neu, methods of using conjugates that bind to Her2 / neu and methods of treating undesirable or aberrant cell proliferation or hyperproliferative disorders, such as tumors, cancers, neoplasia and malignancies that express Her2 / neu.
Owner:A28 THERAPEUTICS INC
Preparation method of polypeptide
ActiveCN108341883APrevent oxidationHigh product purityAntibody mimetics/scaffoldsIntegrin superfamilyPeptideSolid phases
The invention relates to a method for preparing polypeptide or pharmaceutically acceptable salts. The method comprises the following steps of 1, synthesizing a full-protection peptide sequence on a solid phase carrier, and obtaining peptide resin; 2, adopting a lysate for pyrolyzing the peptide resin, and obtaining crude peptide; 3, adopting an iodine oxidation method for directly performing a disulfide bond cyclization reaction on the crude peptide. The preparation method is applicable to preparing the polypeptide in large scale, and the preparation scale can reach the hectogram scale to thekilogram scale.
Owner:北京爱泰浦生物医药科技有限责任公司
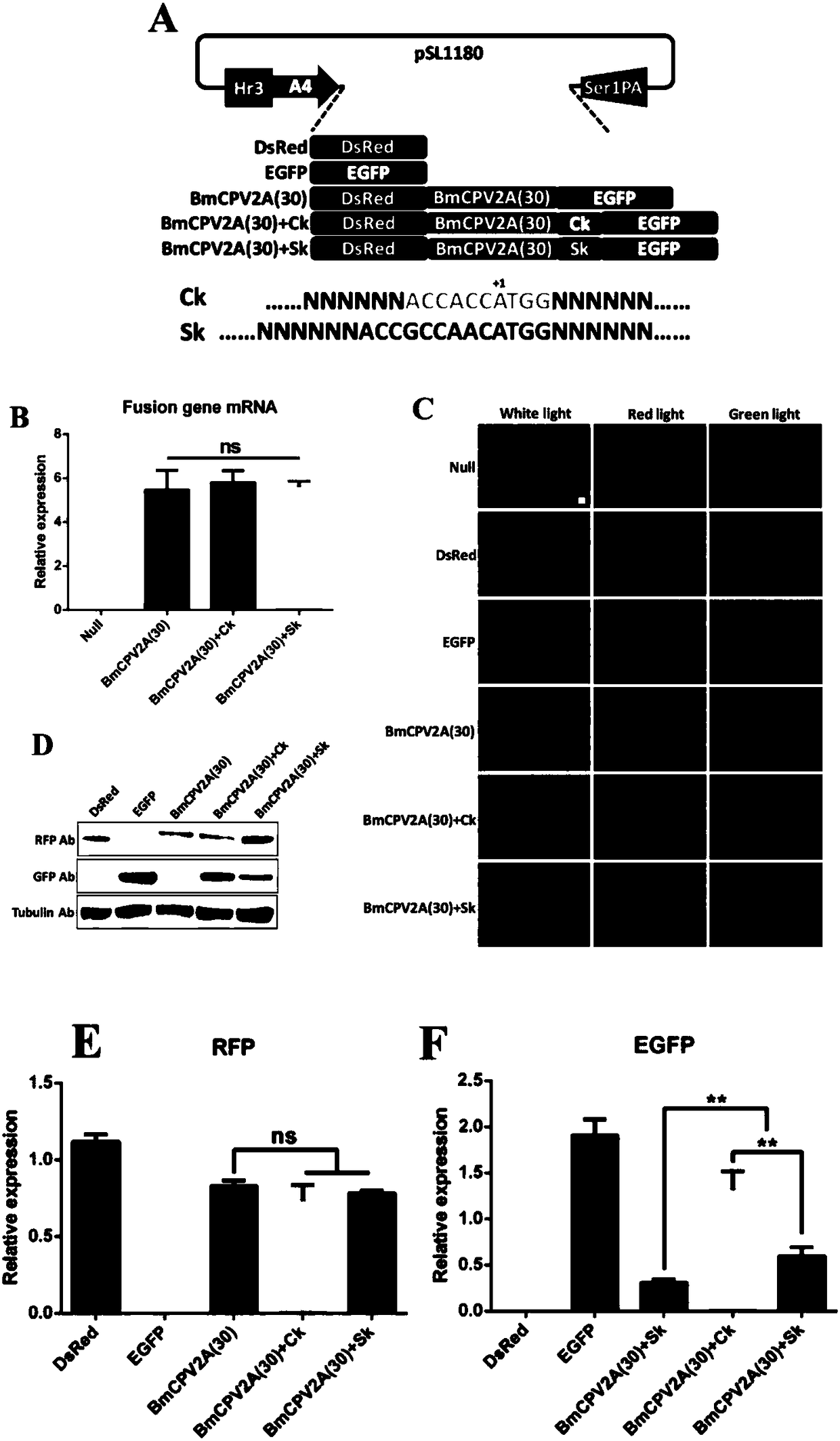
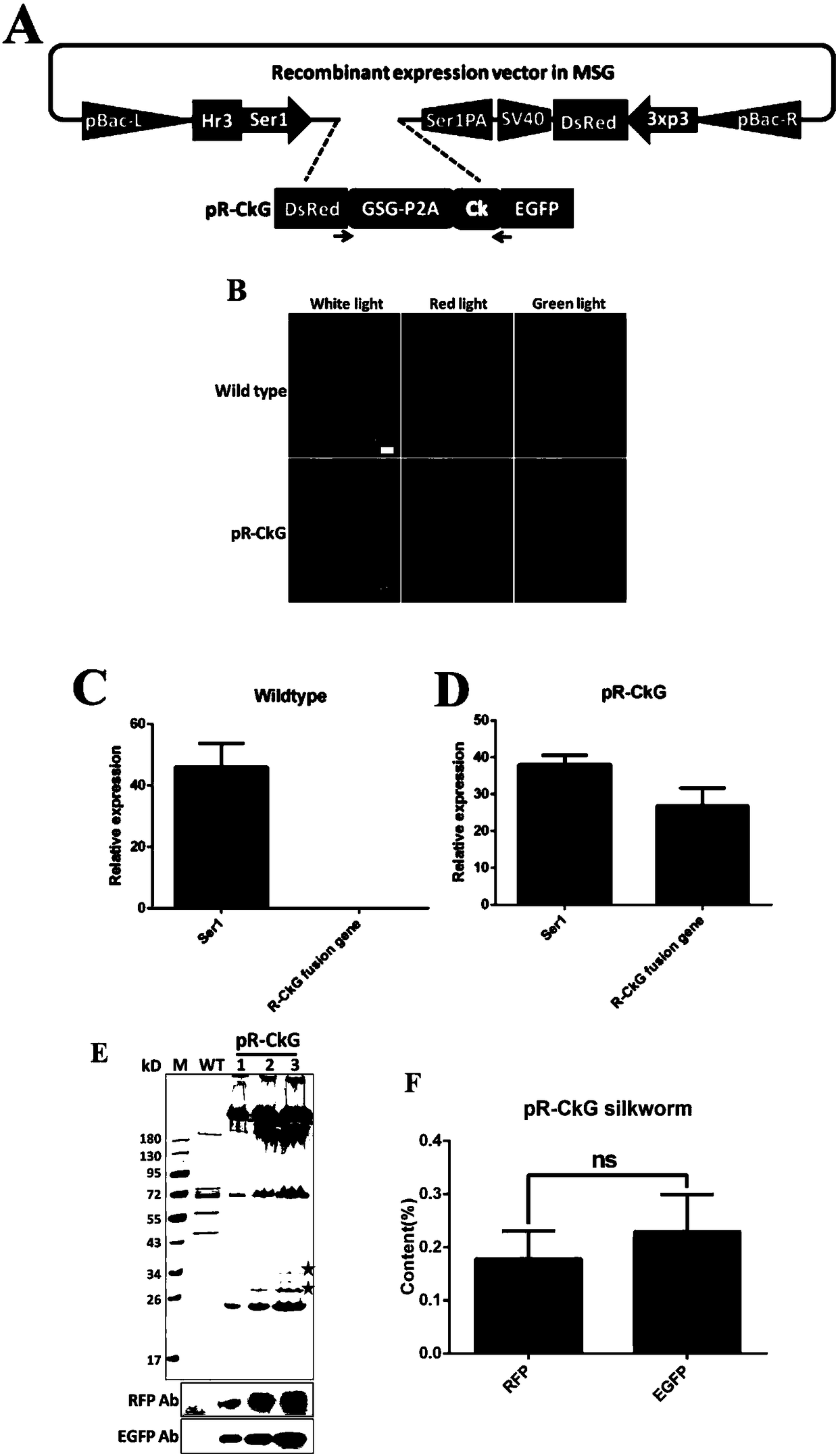
Polygene element realizing equivalent expression of two genes based on 2A lytic peptide, expression vector and application
ActiveCN108486113ANucleic acid vectorVector-based foreign material introductionLytic peptideNucleotide
The invention relates to a polygene element realizing equivalent expression of two genes based on 2A lytic peptide, an expression vector and application. The gene element is formed by Ck and the 2A lytic peptide, wherein a nucleotide sequence of the 2A lytic peptide is as shown in SEQ ID NO.38, and a nucleotide sequence of the Ck is as shown in SEQ ID NO.39. The vector containing the element can be used for equivalently performing recombination expression on the genes located on two ends of 2A so as to have significance on realizing silkworm poly-gene expression.
Owner:SOUTHWEST UNIVERSITY
Angiogenic active lytic peptides
This invention relates to novel synthetic lytic peptide fragments of full-length peptides with the capacity to modulate angiogenic activity in mammals. The invention also relates to the use of such peptides in pharmaceutical compositions and in methods for treating diseases or disorders that are associated with angiogenic activity.
Owner:ISSAR PHARMA LTD
Selective anticancer chimeric peptide
ActiveUS20130274201A1Evading side effectBacteriaPeptide/protein ingredientsFunctional disturbanceLytic peptide
It is an object of the present invention to provide a substance usable as an anticancer agent or DDS, which has intracellular stability, which is capable of evading side effects from functional disorder with respect to normal cells, or which has instantaneous effect. The inventors developed a novel chimeric peptide targeting cancer cells which overexpress EGFR or the like using a binding peptide such as a peptide sequence binding to EGFR, and a lytic peptide sequence, thereby solving such an object. Particularly, by using a chimeric peptide including an EGF receptor-binding peptide or the like and a cytotoxic peptide, this object was solved.
Owner:STELLA PHARMA CORP +1
Compound polypeptide molecule for targeted killing of cancer cells and preparation method thereof
PendingCN112724258ANeutralize electropositivityNeutralizing activityAntibody mimetics/scaffoldsPeptide preparation methodsLytic peptideGallic acid ester
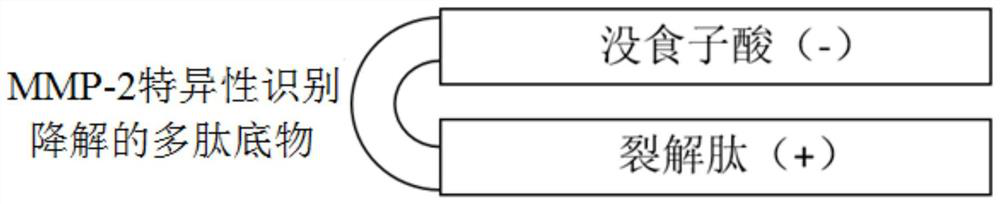
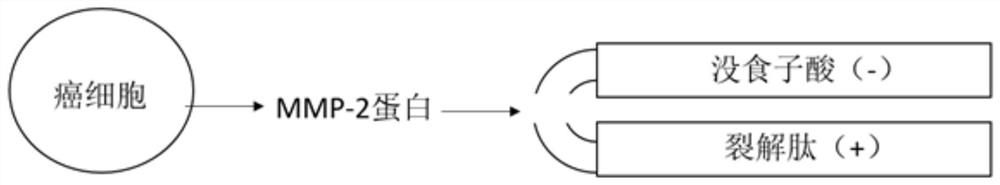
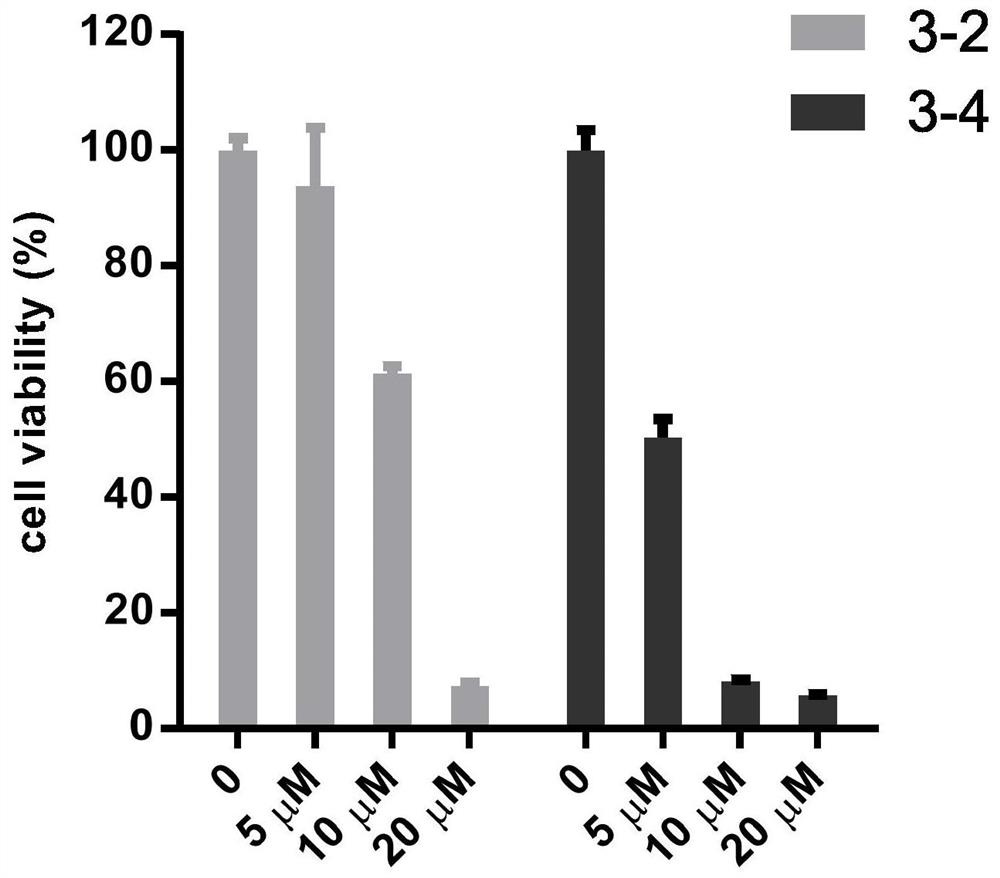
The invention belongs to the technical field of biological medicines, and particularly relates to a compound polypeptide molecule for targeted killing of cancer cells and a preparation method thereof. The compound polypeptide molecule comprises gallic acid, a cracking peptide and a polypeptide substrate which is connected between the gallic acid and the cracking peptide and is used for MMP-2 specific recognition degradation. In the composite polypeptide molecule, the electronegative gallic acid small molecule can effectively neutralize the electropositivity and the split membrane activity of the split peptide, so that the damage of the split peptide to normal cells in the in-vivo circulation process can be effectively prevented, and after the composite polypeptide molecule enters cancer tissues, the cancer cells secrete a large amount of MMP-2 protein, the MMP-2 protein can recognize and degrade a polypeptide substrate connected between gallic acid and lytic peptide, so that free lytic peptide is released, and the effect of killing cancer cells in a targeted manner can be achieved.
Owner:THE SECOND PEOPLES HOSPITAL OF SHENZHEN
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com