Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
119 results about "Protozoal Infection" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Protozoal infections are more common in puppies, seniors, canines with suppressed immune systems and in dogs that are in close-quarters (shelters, kennels, and pet stores). Some protozoal infections are zoonotic, which means an infected dog can transmit the infection to humans. Protozoal infections in dogs are caused by protozoans.
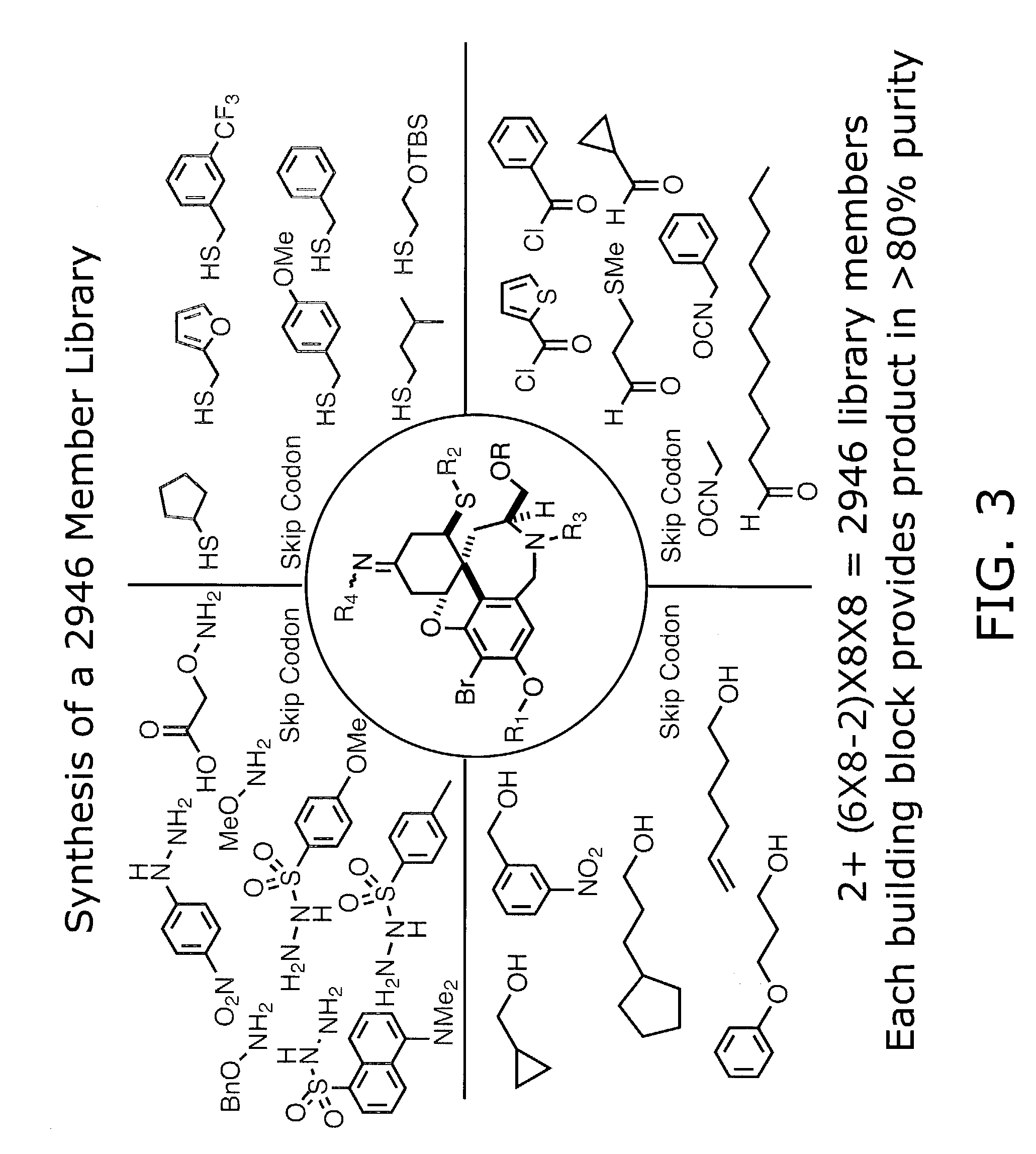
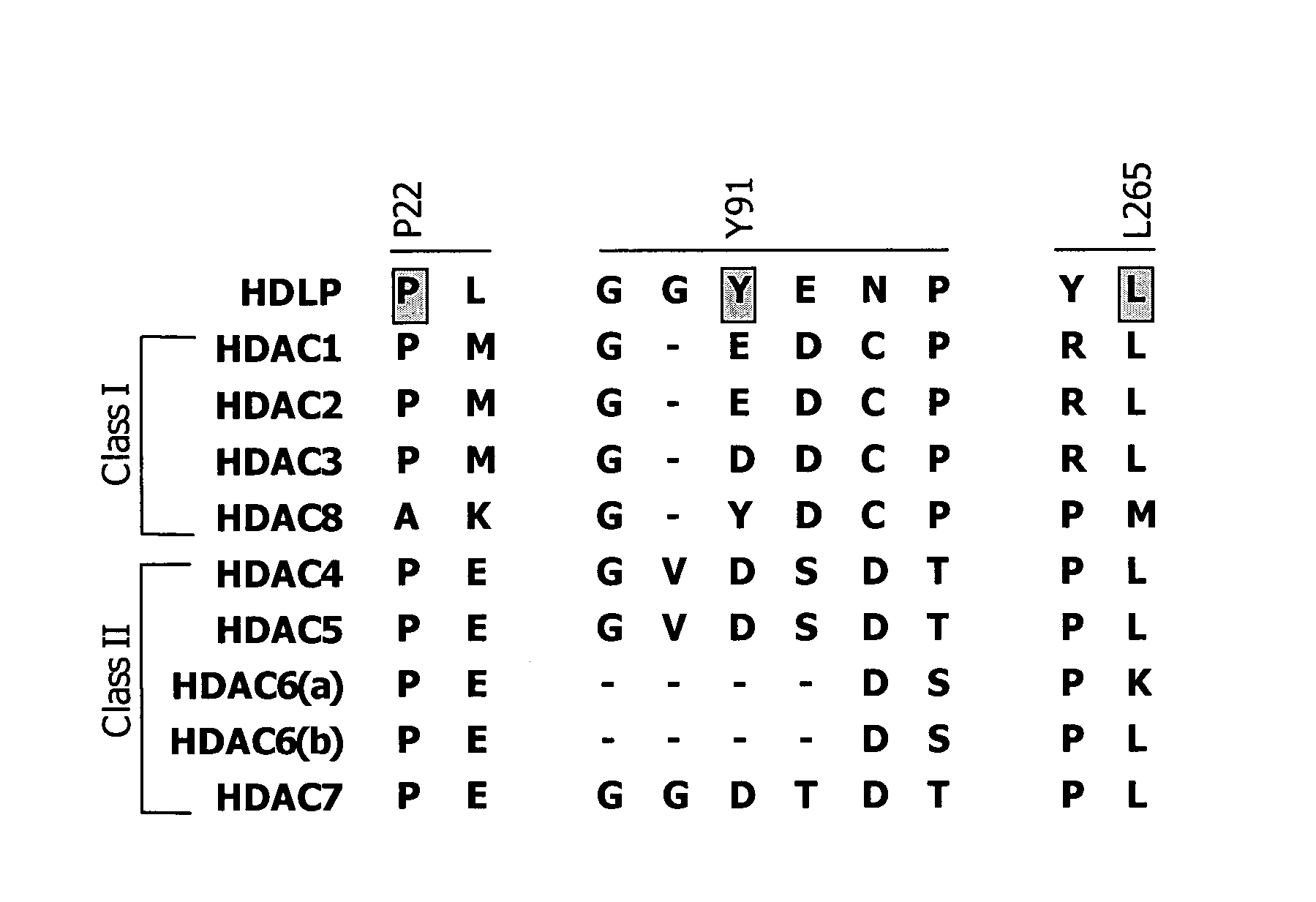
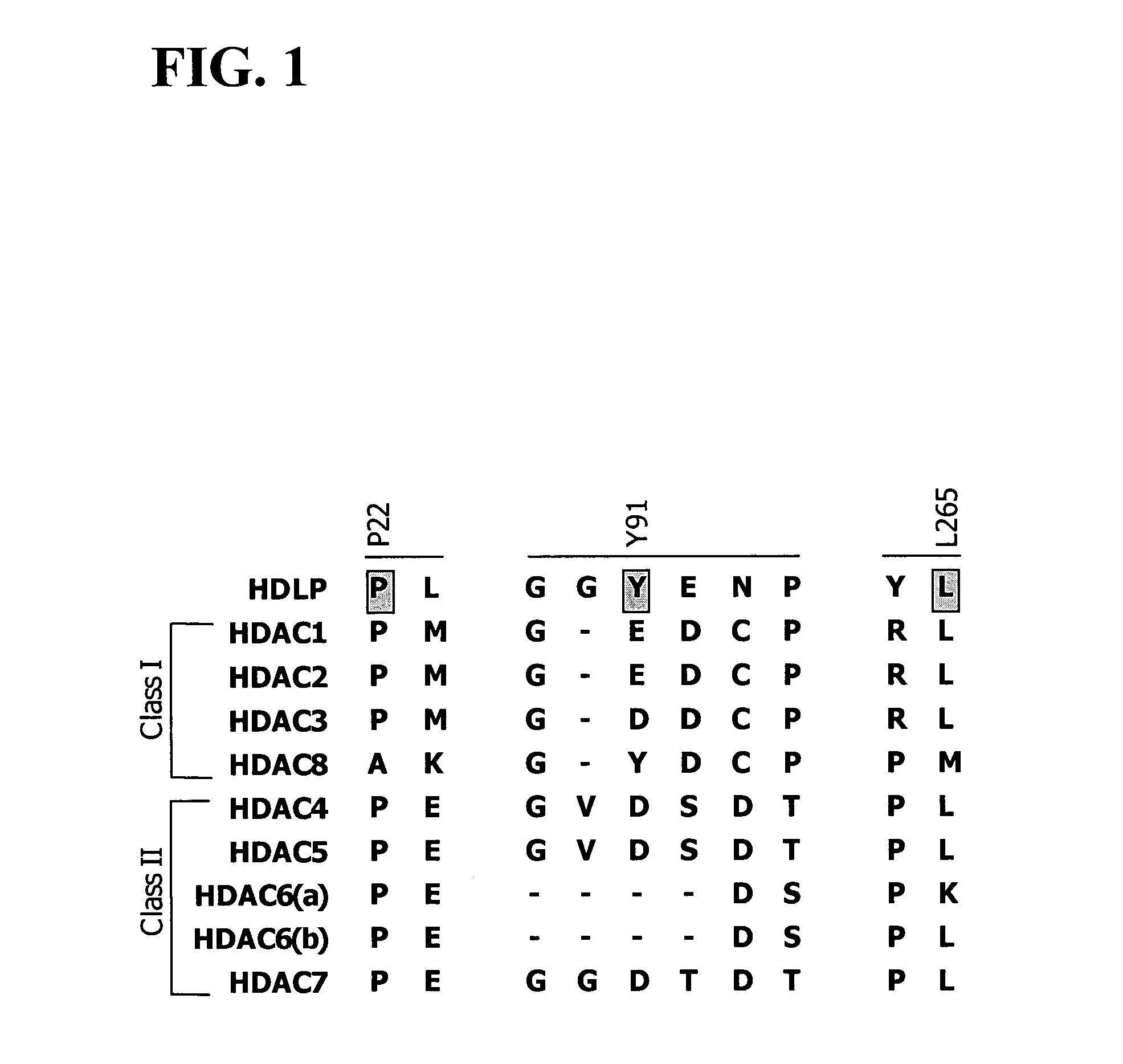
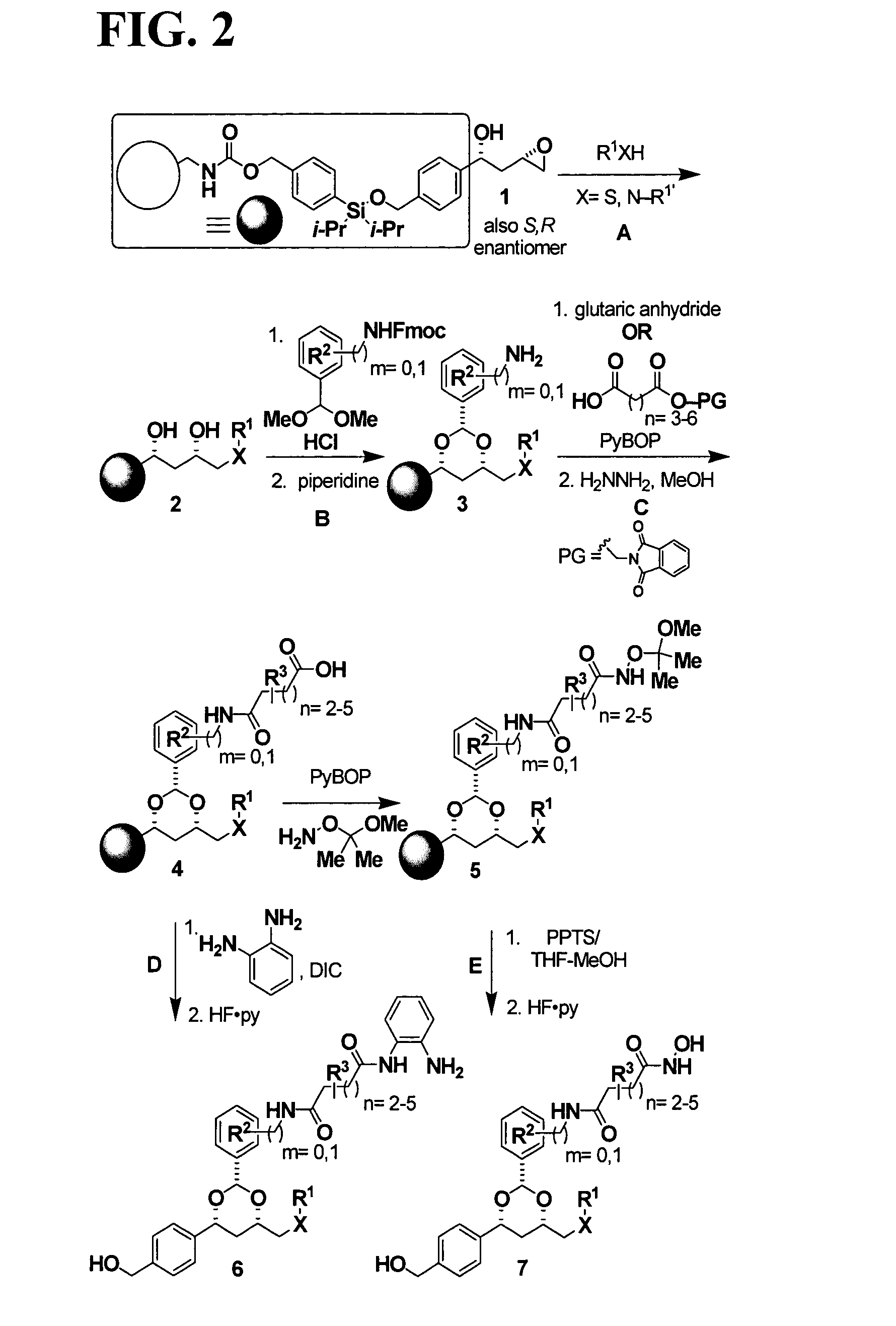
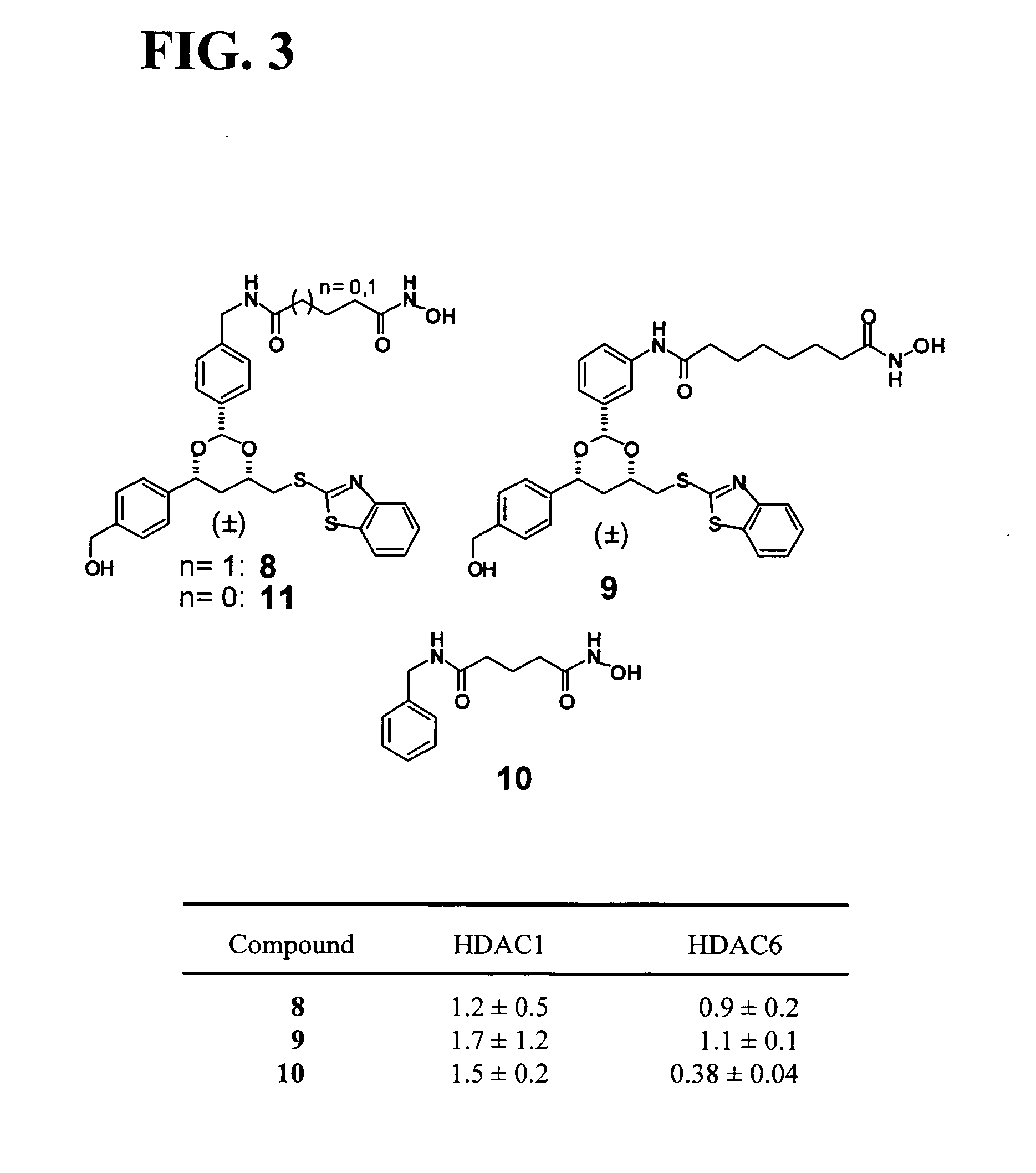
Dioxanes and uses thereof
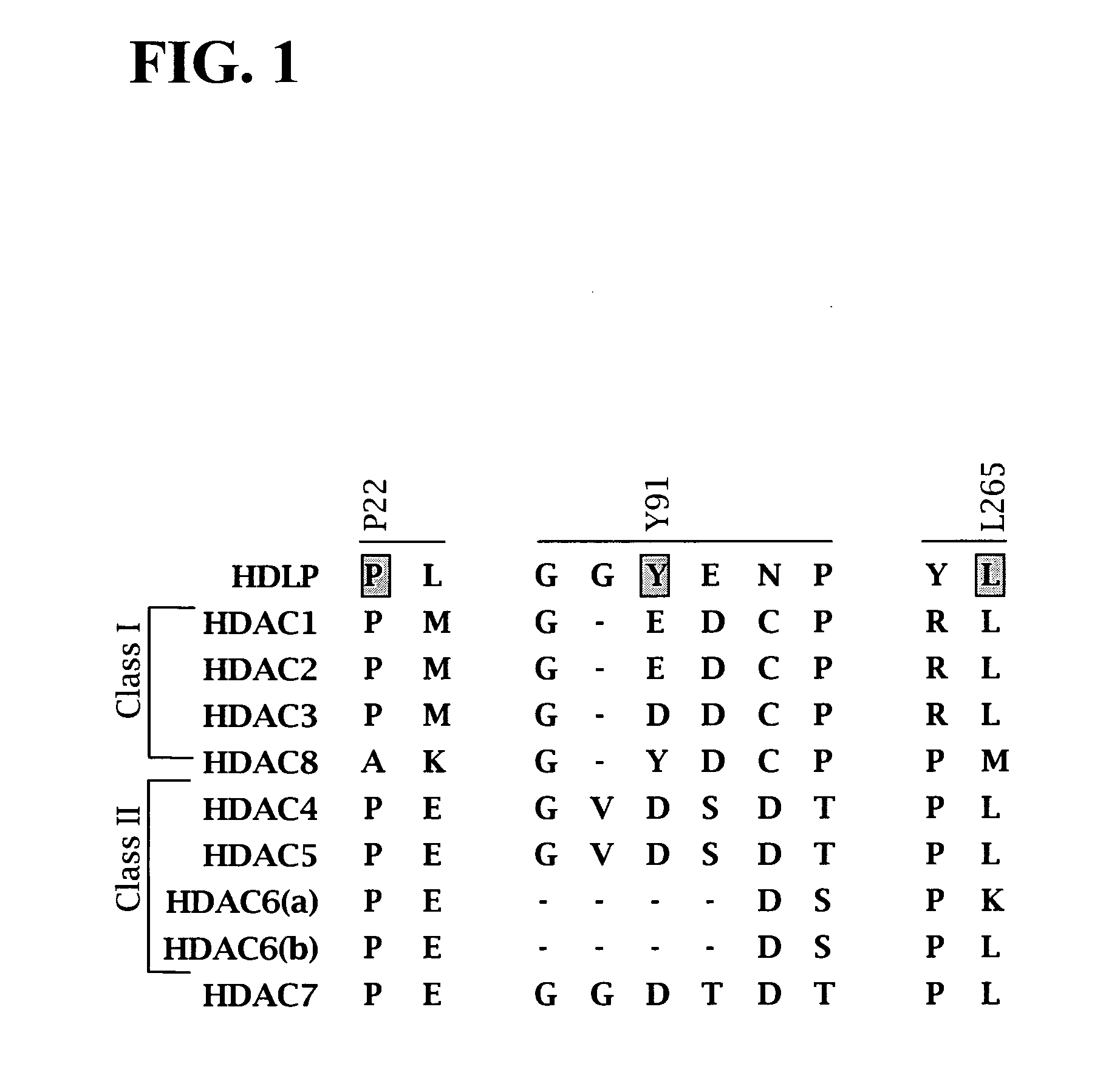
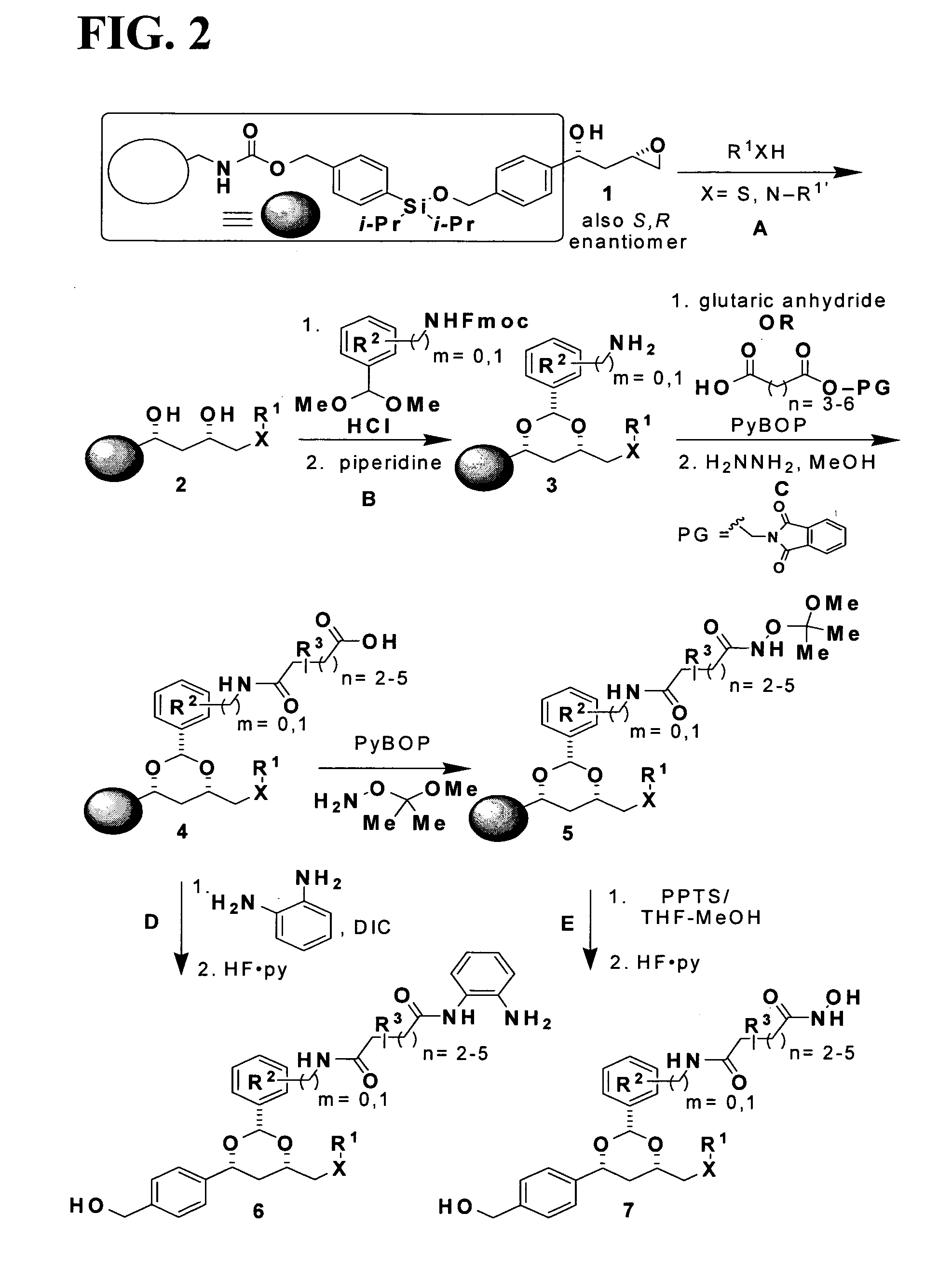
In recognition of the need to develop novel therapeutic agents and efficient methods for the synthesis thereof, the present invention provides novel compounds of general formula (I):and pharmaceutically acceptable derivatives thereof, wherein R1, R2, R3, n, X and Y are as defined herein. The present invention also provides pharmaceutical compositions comprising a compound of formula (I) and a pharmaceutically acceptable carrier. The present invention further provides compounds capable of inhibiting histone deacetylatase activity and methods for treating disorders regulated by histone deacetylase activity (e.g., cancer and protozoal infections) comprising administering a therapeutically effective amount of a compound of formula (I) to a subject in need thereof. The present invention additionally provides methods for modulating the glucose-sensitive subset of genes downstream of Ure2p. The present invention also provides methods for preparing compounds of the invention.
Owner:PRESIDENT & FELLOWS OF HARVARD COLLEGE
Inhibitors of nucleoside phosphorylases and nucleosidases
The present invention relates to compounds of the general formula (I) which are inhibitors of purine nucleoside phosphorylases (PNP), purine phosphoribosyltransferases (PPRT), 5′-methylthioadenosine phosphorylases (MTAP), 5′-methylthioadenosine nucleosidases (MTAN) and / or nucleoside hydrolases (NH). The invention also relates to the use of these compounds in the treatment of diseases and infections including cancer, bacterial infections, protozoal infections, and T-cell mediated disease and to pharmaceutical compositions containing the compounds.
Owner:IND RES LTD +1
Compositions comprising trimetrexate and methods of their synthesis and use
InactiveUS6258821B1Improve stabilityEnsure long-term stabilityOrganic active ingredientsBiocideDiseaseOxygen
This invention is directed to the novel composition of matter trimetrexate ascorbate, to compositions comprising trimetrexate ascorbate, and to compositions comprising trimetrexate and ascorbic acid. These compositions are useful in the treatment of diseases in mammals such as, but not limited to, cancer, bacterial and protozoal infections, malaria, psoriasis, and rheumatoid arthritis. The invention is further related to methods of stabilizing trimetrexate to degradation caused by heat, light, oxygen, or water.
Owner:MEDIMMUNE ONCOLOGY
Inhibitors of nucleoside phosphorylases and nucleosidases
The present invention relates to compounds of the general formula (I) which are inhibitors of purine muclioside phosphorylases (PNP), purine phosphoribosyltransferases (PPRT), 5′-methylthioadenosine phosphorylases (MTAP), 5′-methylthioadenosine mucliosidases (MTAN) and / or nucleoside hydrolases (NH). The invention also relates to the use of these compounds in the treatment of diseases and infections including cancer, bacterial infections, protozoal infections, and T-cell mediated disease and to pharmaceutical compositions containing the compounds.
Owner:VICTORIA LINK LTD
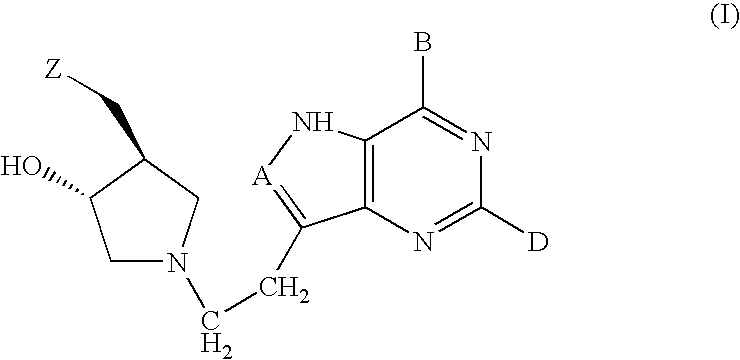
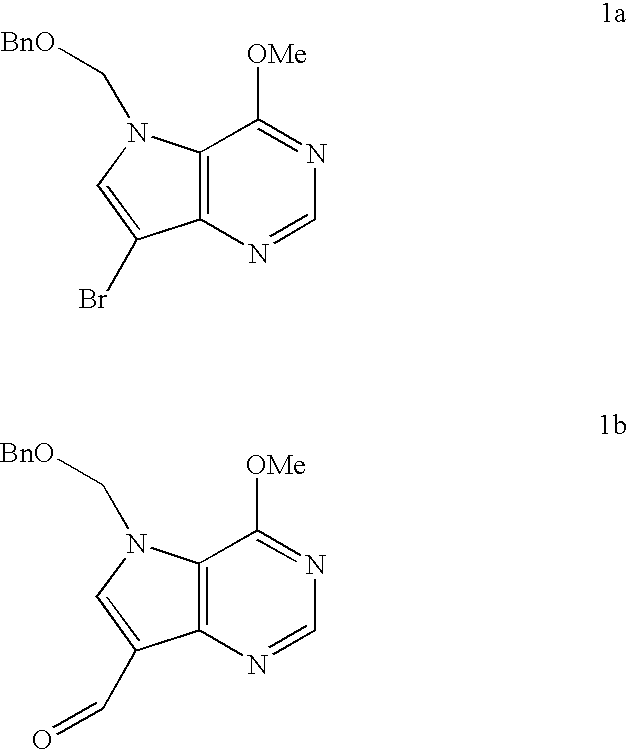
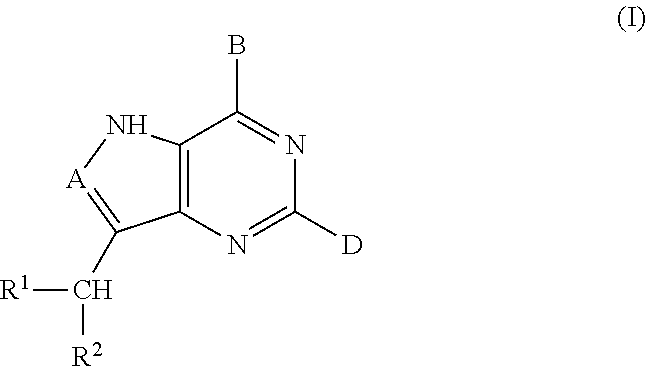
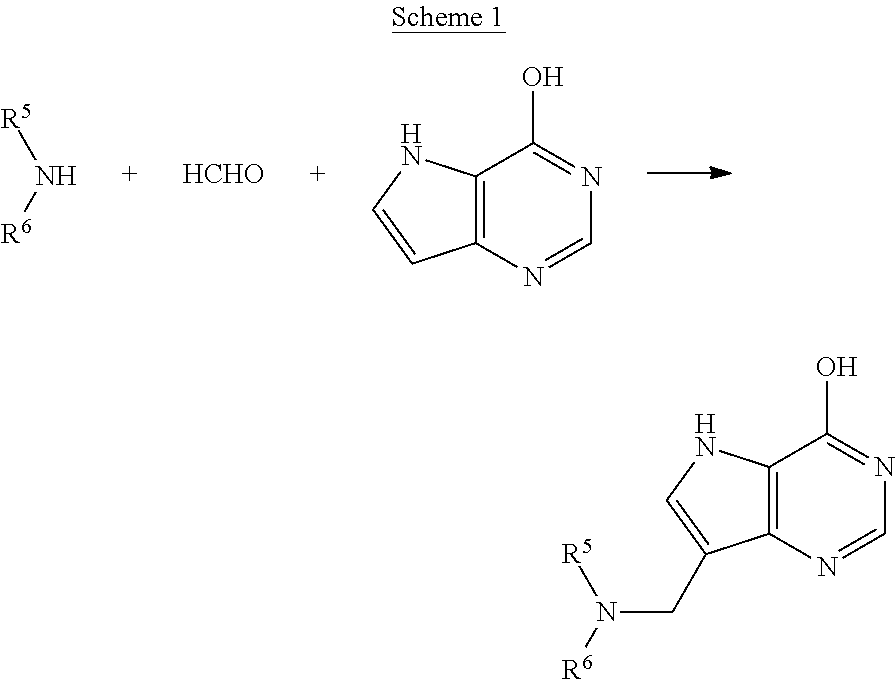
5h-pyrrolo[3,2-D] pyrimidine inhibitors of nucleoside phosphorylases and nucleosidases
The present invention relates to compounds of the general formula (I) which are inhibitors of purine muclioside phosphorylases (PNP), purine phosphoribosyltransferases (PPRT), 5′-methylthioadenosine phosphorylases (MTAP), 5′-methylthioadenosine mucliosidases (MTAN) and / or nucleoside hydrolases (NH). The invention also relates to the use of these compounds in the treatment of diseases and infections including cancer, bacterial infections, protozoal infections, and T-cell mediated disease and to pharmaceutical compositions containing the compounds.
Owner:VICTORIA LINK LTD
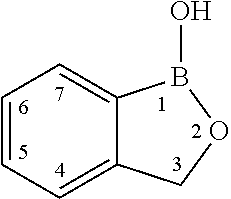
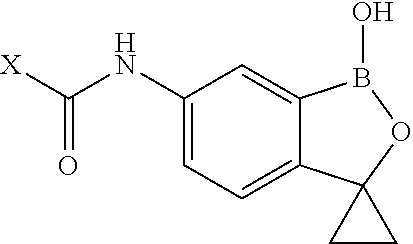
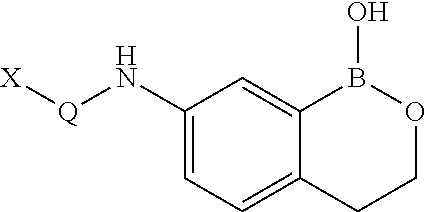
Boron-containing small molecules as Anti-protozoal agents
Owner:ANACOR PHARMA INC
Method for Treating Oncological, Virulent and Somatic Diseases, Method for Controlling Treatment Efficiency, Pharmaceutical Agents and Composition for Carrying Out Said Treatment
The invention relates to medicine and veterinary science and discloses a novel method for treating oncological, virulent and somatic diseases whose main target for therapeutic action is embodied in the form of DNA which freely circulates in blood plasma (and other liquid media) and originates from tumoral and mutant cells or cells infected by bacteria, fungi or protozoan and from different microorganisms which reside in the organism thereof. Said invention also relates to novel pharmaceutical compositions and to the use thereof for treating oncological diseases and infectious states provoked by bacteria, fungi and protozoa and non-infectious somatic diseases and states produced by accumulation of somatic mutations in cells of organism. Medicinal and immunological compositions, sorption and physico-chemical engineering and the method for using them in order to treat malignant tumors and other diseases are also disclosed.
Owner:GENKIN DMITRY DMITRIEVICH +1
Dioxanes and uses thereof
Owner:PRESIDENT & FELLOWS OF HARVARD COLLEGE
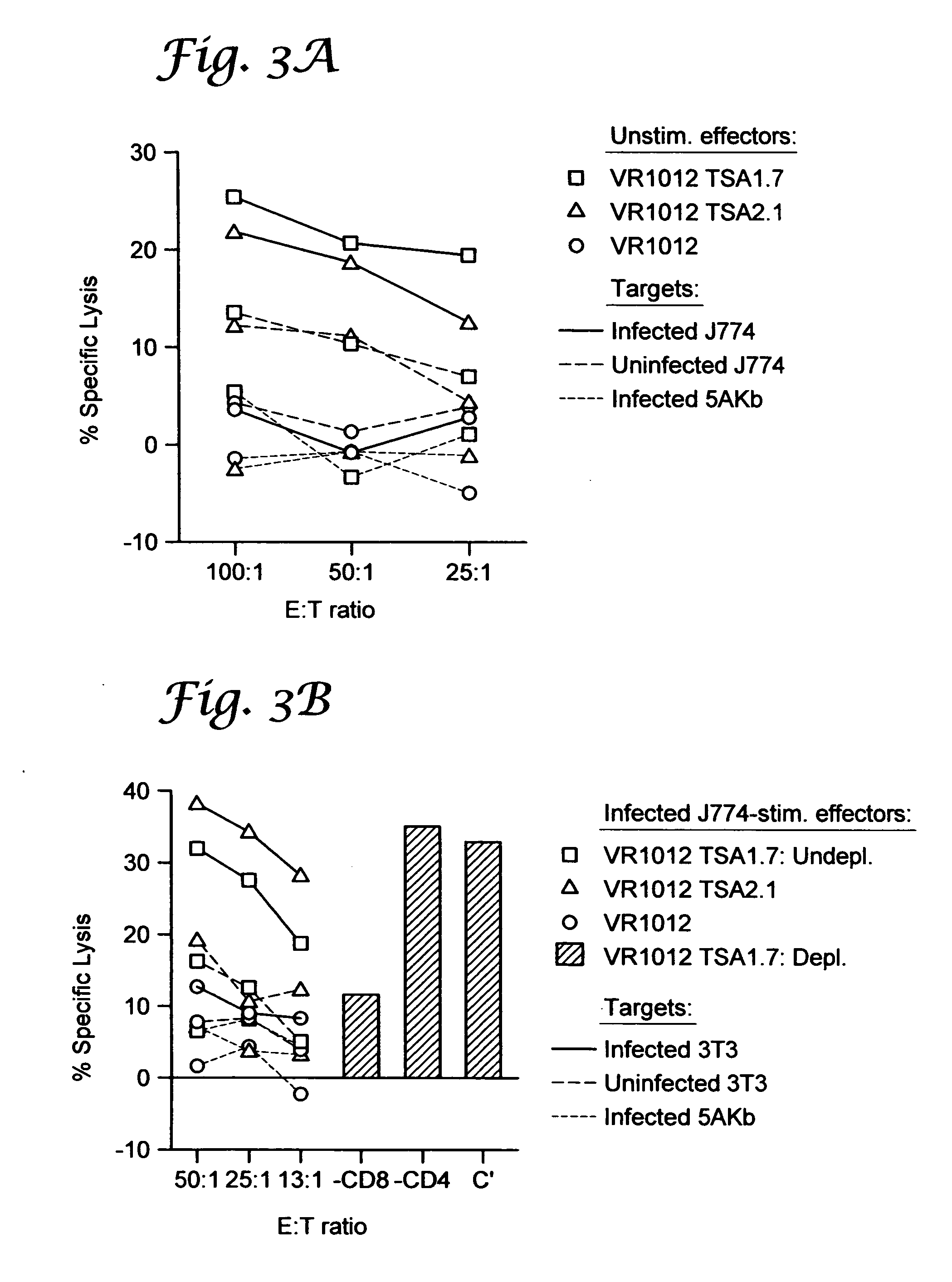
Prophylactic and therapeutic immunization against protozoan infection and disease
InactiveUS20050158347A1Convenient introductionIncrease stimulationProtozoa antigen ingredientsSugar derivativesMammalCell membrane
Polypeptide and polynucleotide vaccines effective to treat or prevent infection of a mammal, such as a dog, a cat, or a human, by a protozoan. Methods of treatment and prevention are also provided, including therapeutic administration of the vaccine to an infected mammal to prevent progression of infection to a chronic debilitating disease state. Preferred embodiments of the polynucleotide vaccine contain nucleotide coding regions that encode polypeptides that are surface-associated or secreted by T. cruzi. Optionally the efficacy of the polynucleotide vaccine is increased by inclusion of a nucleotide coding region encoding a cytokine. Preferred embodiments of the polypeptide vaccine include immunogenic peptides that contain membrane transducing sequences that allow the polypeptides to translocate across a mammalian cell membrane.
Owner:UNIV OF GEORGIA RES FOUND INC
Boron-containing small molecules as antiprotozoal agents
Owner:ANACOR PHARMA INC
Inhibitors of nucleoside phoshorylases and nucleosidases
The present invention relates to compounds of the general formula (I) which are inhibitors of purine nucleoside phosphorylases (PNP), purine phosphoribosyltransferases (PPRT), 5′-methylthioadenosine phosphorylases (MTAP), 5′-methylthioadenosine nucleosidases (MTAN) and / or nucleoside hydrolases (NH). The invention also relates to the use of these compounds in the treatment of diseases and infections including cancer, bacterial infections, protozoal infections, and T-cell mediated disease and to pharmaceutical compositions containing the compounds.
Owner:VICTORIA LINK LTD
Targeted oxidative therapeutic formulation
Pharmaceutical formulation, its method of preparation, and its use. The pharmaceutical formulation contains peroxidic species or reaction products resulting from oxidation of an alkene, such as geraniol, by an oxygen-containing oxidizing agent, such as ozone; a penetrating solvent, such as dimethyl sulfoxide; a dye containing a chelated metal, such as hematoporphyrin; and an aromatic redox compound, such as benzoquinone. The pharmaceutical formulation is used to treat horses infected with Sarcocystis protozoal infections.
Owner:TORQUIN LLC
Neuroprotective iron chelators and pharmaceutical compositions comprising them
Novel iron chelators exhibiting neuroprotective and good transport properties are useful in iron chelation therapy for treatment of a disease, disorder or condition associated with iron overload and oxidative stress, eg. a neurodegenerative or cerebrovascular disease or disorder, a neoplastic disease, hemochromatosis, thalassemia, a cardiovascular disease, diabetes, a inflammatory disorder, anthracycline cardiotoxicity, a viral infection, a protozoal infection, a yeast infection, retarding ageing, and prevention and / or treatment of skin ageing and skin protection against sunlight and / or UV light. The iron chelator function is provided by a 8-hydroxyquinoline, a hydroxypyridinone or a hydroxamate moiety, the neuroprotective function is imparted to the compound e.g. by a neuroprotective peptide, and a combined antiapoptotic and neuroprotective function by a propargyl group.
Owner:TECHNION RES & DEV FOUND LTD +1
Antipathogenic synthetic piptides and compositions comprising them
Non-hemolytic cytolytic agents selected from peptides, complexes of bundled peptides, mixtures of peptides or random peptide copolymers have a selective cytolytic activity manifested in that they have a cytolytic activity on pathogenic cells, being cells which are non-naturally occurring within the body consisting of microbial pathogenic organisms and malignant cells; and are non-hemolytic, having no cytolytic effect on red blood cells. The peptides may be cyclic derivatives of natural peptides such as pardaxin and mellitin and fragments thereof in which L-amino acid residues are replaced by corresponding D-amino acid residues, or are diastereomers of linear peptides composed of varying ratios of at least one positively charged amino acid and at least one hydrophobic amino acid, and in which at least one of the amino acid residues is a D-amino acid. Pharmaceutical compositions comprising the non-hemolytic cytolytic agents can be used for the treatment of several diseases caused by pathogens including antibacterial, fungal, viral, mycoplasma and protozoan infections and for the treatment of cancer.
Owner:YEDA RES & DEV CO LTD
Bisubstituted carbocyclic cyclophilin binding compounds and their use
InactiveUS20020127605A1Quick identificationAntibacterial agentsUrea derivatives preparationMedical disorderDisease
The present invention relates to novel, non-peptidic small organic compounds having an affinity for cyclophilin (CyP)-type immunophilin proteins. In the compounds of this invention, at least two carbo- or heterocyclic groups are attached to a central saturated, partially saturated, or aromatic 5-6 membered carbocyclic ring by a combination of straight or branched linker chains. The invention further relates to pharmaceutical compositions comprising one or more of the said compounds, and to the uses of these compounds and compositions for binding CyP-type proteins, inhibiting their peptidyl-prolyl isomerase activity, and for research, development, and therapeutic applications in a variety of medical disorders, such as neurological disorders, hair loss disorders, ischemic disorders, and disorders caused by viral or protozoan infection.
Owner:GUILFORD PHARMACEUTICALS INC
Boron-containing small molecules as Anti-protozoal agent
Owner:ANACOR PHARMA INC
Acyclic amine inhibitors of nucleoside phosphorylases and hydrolases
The invention relates to compounds of the general formula (I) which are inhibitors of purine nucleoside phosphorylases (PNPs) and / or nucleoside hydrolases (NHs). The invention also relates to the use of these compounds in the treatment of diseases and infections including cancer, bacterial infections, protozoal infections, and T-cell mediated disease and to pharmaceutical compositions containing the compounds.
Owner:VICTORIA LINK LTD
Trypanosoma Antigens, Vaccine Compositions, and Related Methods
ActiveUS20090324634A1Reduces and eliminates riskLess considerationAntibody mimetics/scaffoldsFusions for enhanced expression stability/foldingProtozoaTrypanosoma antigen
The present invention relates to the intersection of the fields of immunology and protein engineering, and particularly to antigens and vaccines useful in prevention of infection by Trypanosoma protozoa. Provided are recombinant protein antigens, compositions, and methods for the production and use of such antigens and vaccine compositions.
Owner:IBIO
Antipathogenic synthetic peptides and compositions comprising them
Non-hemolytic cytolytic agents selected from peptides, complexes of bundled peptides, mixtures of peptides or random peptide copolymers have a selected cytolytic activity manifested in that they have a cytolytic activity on pathogenic cells, being cells which are non-naturally occurring with the body consisting of microbial pathogenic organisms and malignant cells; and are non-hemolytic, having no cytolytic effect on red blood cells. The peptides may be cyclic derivatives of natural peptides such as pardaxin and mellitin and fragments thereof in which L-amino acid residues are replaced by corresponding D-amino acid residues, or are diastereomers of linear peptides composed of varying ratios of at least positively charged amino acid and at least one hydrophobic amino acid, and in which at one of the amino acid residues is a D-amino acid. Pharmaceutical compositions comprising the non-hemolytic cytolytic agents can be used for the treatment of several diseases caused by pathogens including antibacterial, fungal, viral mycoplamsa and protozoan infections and for the treatment of cancer.
Owner:YEDA RES & DEV CO LTD
Interleukin-10 peptides and antibodies thereof for inhibiting adverse effects of protozoan infection
ActiveUS20140017248A1Easy maintenanceStay highBiocideEgg immunoglobulinsInterleukin 10White blood cell
The present disclosure is directed to interleukin-10 (IL-10) peptides and isolated antibodies that specifically bind to the IL-10 peptides. The IL-10 peptides and the isolated antibodies may be administered alone or as an animal feed additive to treat gastrointestinal protozoan infection in animals.
Owner:WISCONSIN ALUMNI RES FOUND
Boron-containing small molecules as antiprotozoal agents
Owner:ANACOR PHARMA INC
Compositions having means for targeting at least one antigen to dendritic cells
A composition that can be used as a vaccine containing means for targeting at least one antigen to dendritic cells and as adjuvants a granulocyte macrophage colony stimulating factor and a CpG oligodeoxynucleotide and / or a CpG-like oligodeoxynucleotide. This composition can used to treat cancers, infectious diseases caused by bacterial, viral, fungal, parasitic or protozoan infections, allergies and / or autoimmune diseases.
Owner:INSTITUT CURIE +4
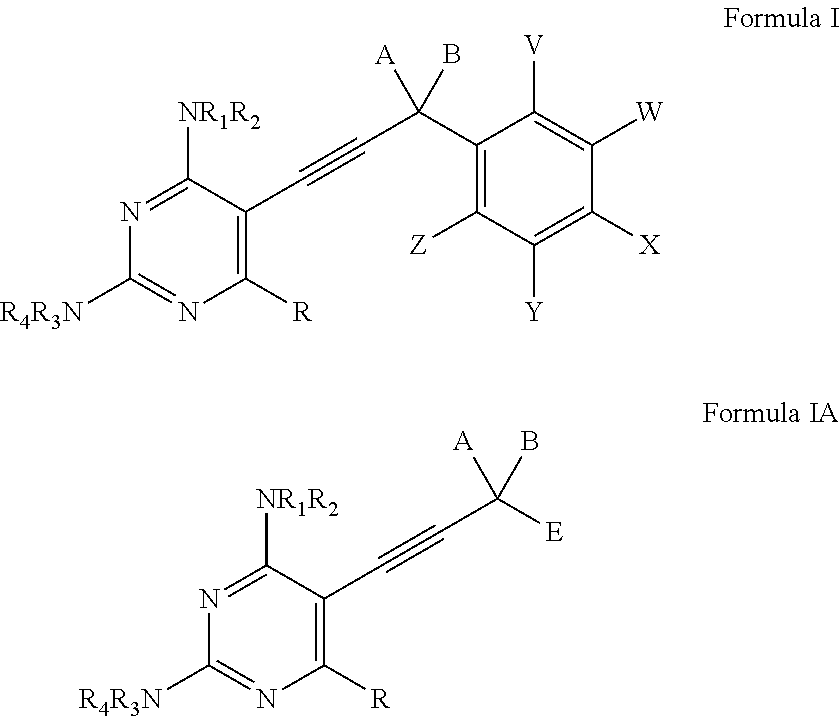
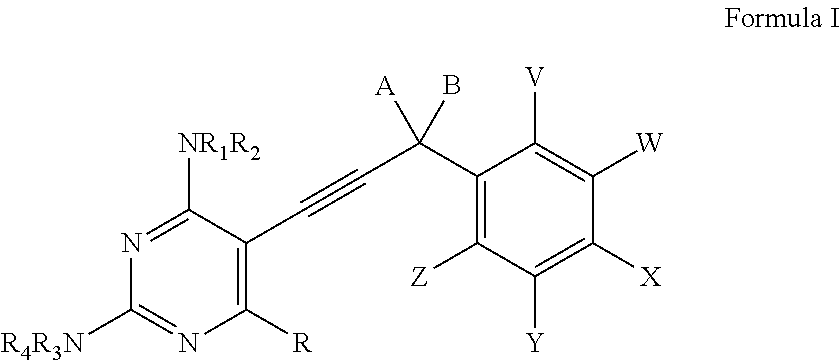
Heterocyclic and Cyclic Analogs of Propargyl-Linked Inhibitors of Dihydrofolate Reductase
ActiveUS20150225353A1BiocideOrganic active ingredientsDihydrofolate reductase inhibitorDihydrofolate reductase deficiency
Compounds of Formula I and Formula IA are inhibitors of dihydrofolate reductase and are suitable for use in compositions and methods for dihydrofolate reductase inhibition or, more specifically, treatment of a fungal infection, a bacterial infection or a protozoal infection, and, in specific embodiments, treatment of a fungal infection caused by C. albicans or C. glabrata:wherein R, R1, R2, R3, R4, A, B, E, V, W, X, Y and Z are as defined herein.
Owner:PROMILIAD BIOPHARMA INC +1
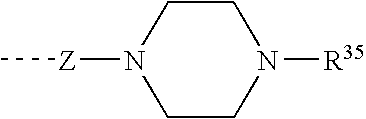
Piperazines as antimalarial agents
Owner:IDORSIA PHARM LTD
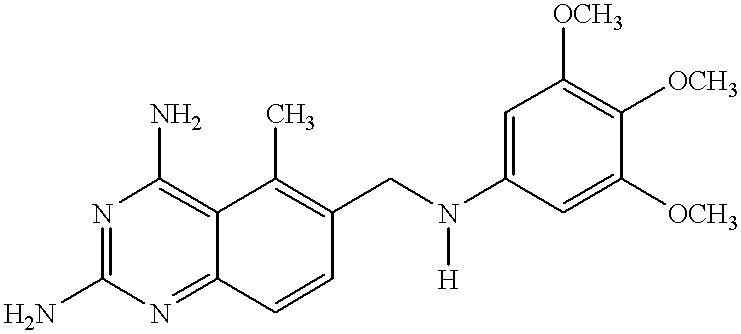
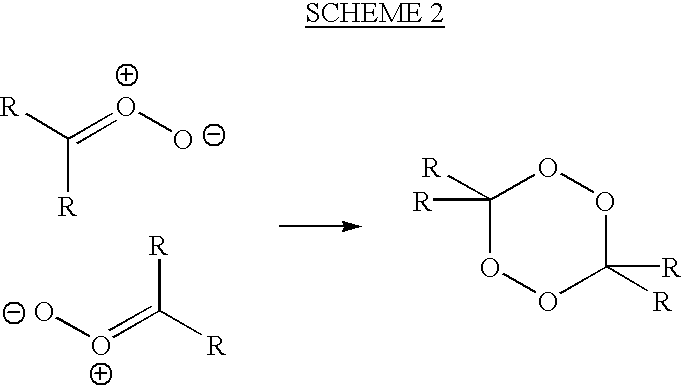
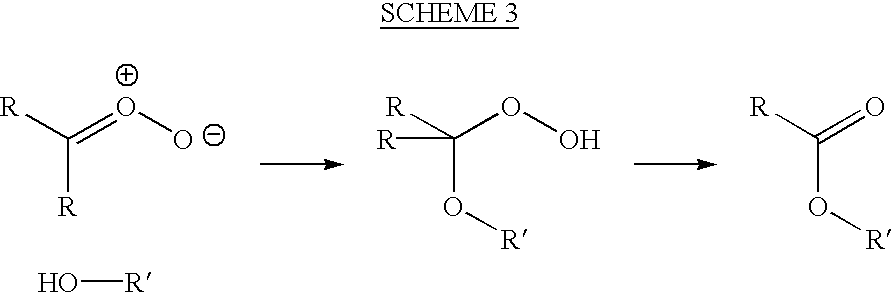
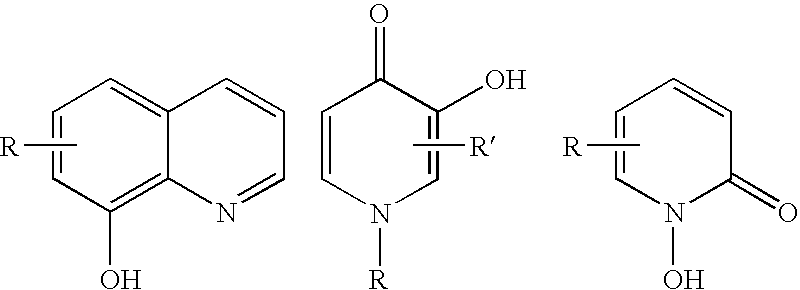
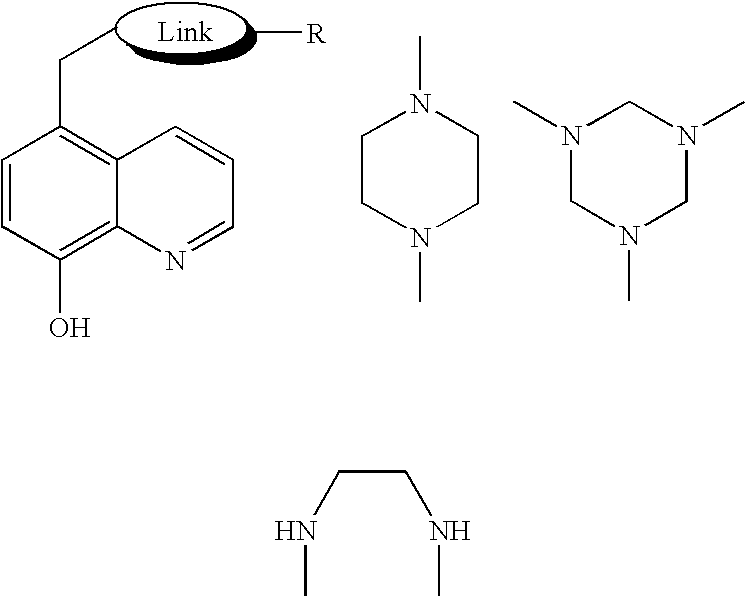
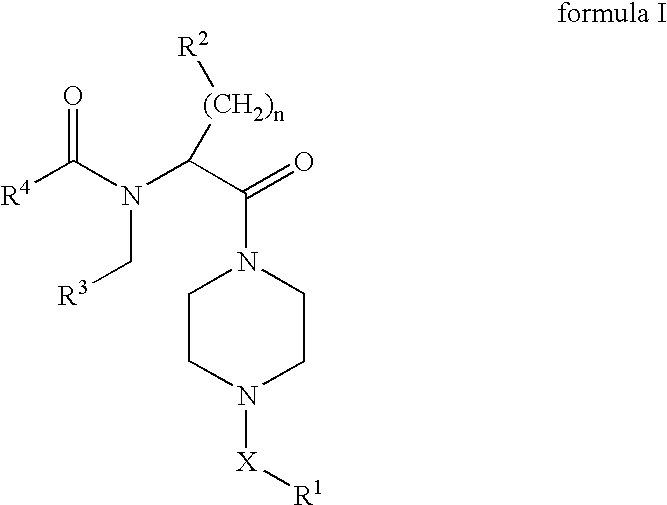
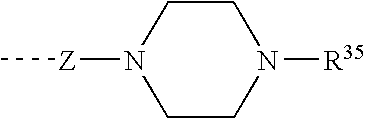
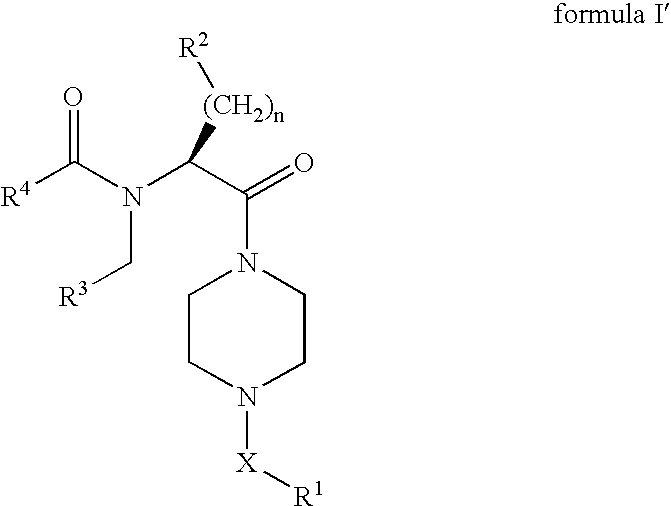
8-N-substituted-2H-isothiazolo[5,4-b]quinolizine-3,4-diones and related compounds as antiinfective agents
Owner:ACHILLION PHARMA INC
Azithromycin dosage forms with reduced side effects
ActiveUS20050123627A1Reduce gastrointestinal side effectsAntibacterial agentsBiocideOral suspensionsGLYCERYL MONOBEHENATE
An oral dosage form comprising azithromycin and an effective amount of an alkalizing agent. Preferably, said oral dosage form comprises an effective amount of an alkalizing agent and an azithromycin multiparticulate wherein said multiparticulate comprises azithromycin, a mixture of glyceryl monobehenate, glyceryl dibehenate and glyceryl tribehenate, and a poloxamer. Typically, the oral dosage form includes any suitable oral dosing means such as a powder for oral suspension, a unit dose packet or sachet, a tablet or a capsule. Additionally disclosed is an oral suspension comprising azithromycin, an effective amount of an alkalizing agent and a vehicle. Preferably, the azithromycin is in multiparticulate form wherein said multiparticulate comprises azithromycin, a mixture of glyceryl monobehenate, glyceryl dibehenate and glyceryl tribehenate, and a poloxamer. Also disclosed is a method for reducing gastrointestinal side effects, associated with administering azithromycin to a mammal, comprising contiguously administering azithromycin and an effective amount of alkalizing agent to said mammal wherein the frequency of gastrointestinal side effects is lower than that experienced by administering an equal dose of azithromycin without said alkalizing agent. Further disclosed is a method of treating a bacterial or protozoal infection in a mammal in need thereof comprising contiguously administering to said mammal a single dose of an oral dosage form wherein said oral dosage form comprises azithromycin and an effective amount of an alkalizing agent. Additionally disclosed are azithromycin multiparticulates comprising azithromycin, a surfactant; and a pharmaceutically acceptable carrier.
Owner:PFIZER INC
Flavonoid dimers and their use
This invention relates to bis-flavonoid compounds, their synthesis and use for inhibiting multidrug resistance in chemotherapy and protozoan infection, wherein the bis-flavonoids compounds have the formula:
Owner:THE HONG KONG POLYTECHNIC UNIV
Novel Piperazines as Antimalarial Agents
The invention relates to novel piperazine derivatives and their use as active ingredients in the preparation of pharmaceutical compositions. The invention also concerns related aspects including pharmaceutical compositions containing one or more of those compounds and their use as medicaments for the treatment or prevention of protozoal infections, especially malaria.
Owner:IDORSIA PHARM LTD
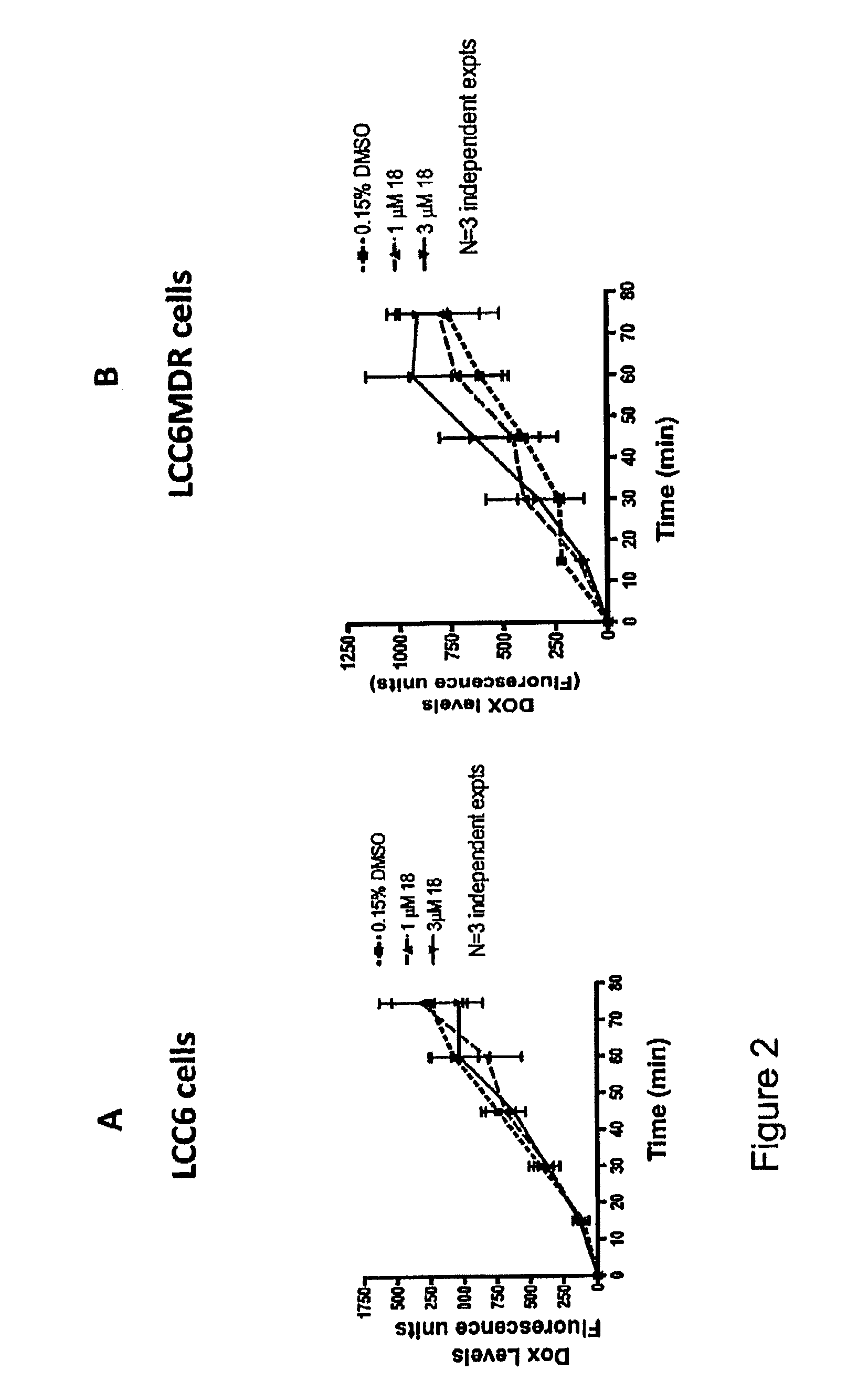
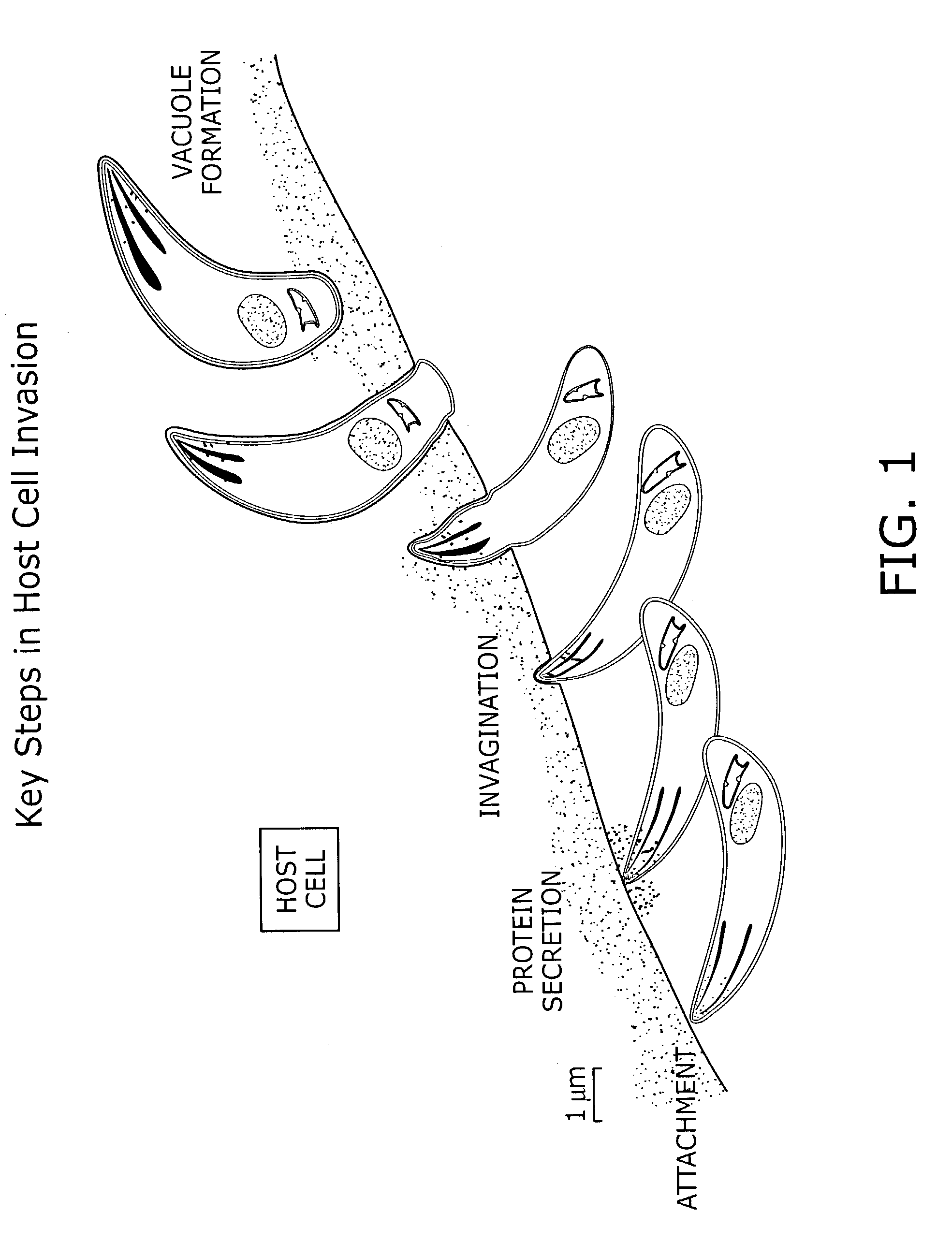
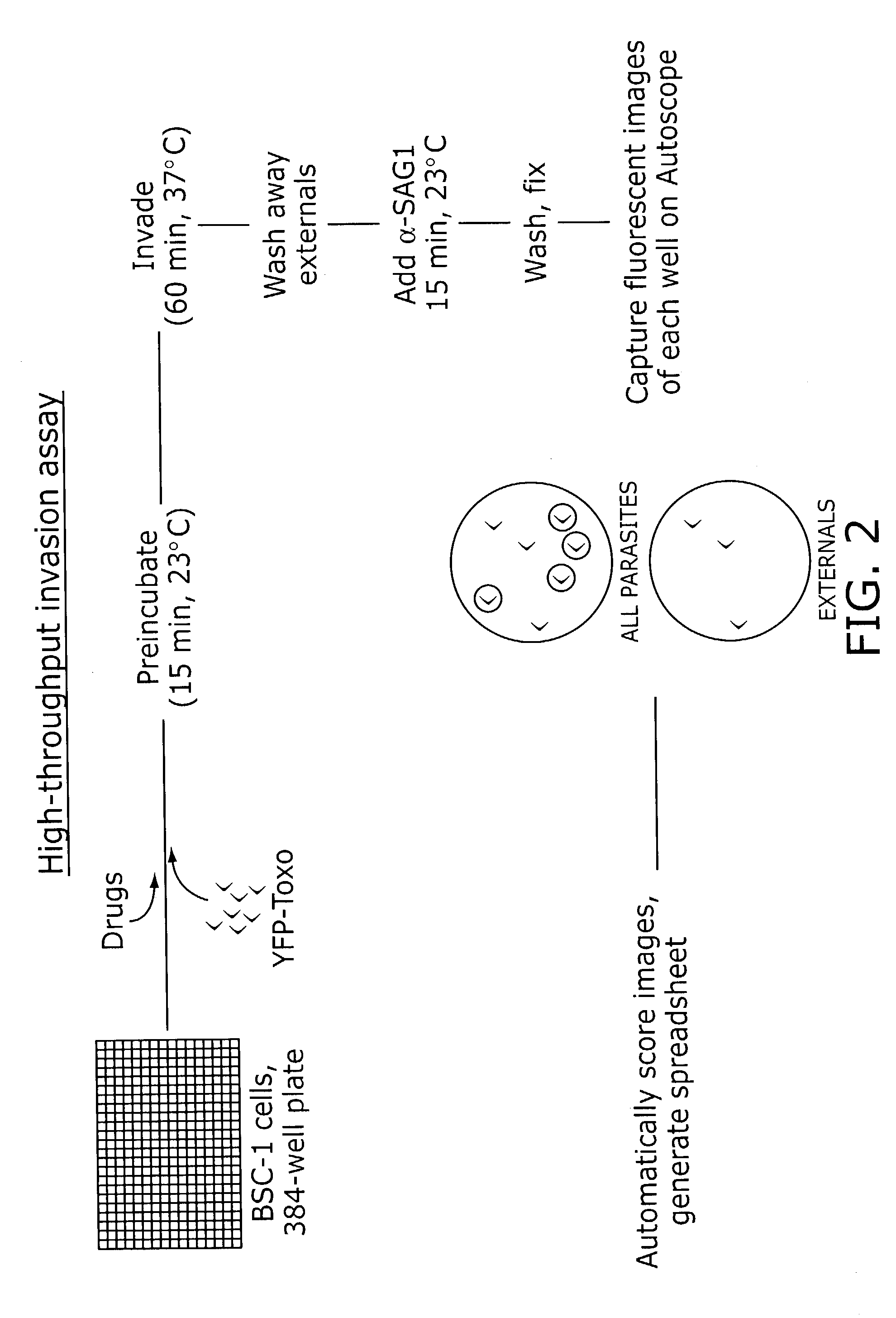
Identification of anti-protozoal agents
InactiveUS7067315B2Lower Level RequirementsImprove bioavailabilityBiocideAnimal repellantsCell invasionAnti protozoal
The present invention provides novel assay systems and methods for monitoring cell invasion by protozoal parasites. The present invention further provides methods of using these assays systems to identify compounds that treat or prevent protozoal infection. The present invention further provides pharmaceutical compositions that have anit-protozoal activity and methods of treating infections.
Owner:PRESIDENT & FELLOWS OF HARVARD COLLEGE +1
Interleukin-10 peptides and antibodies thereof for inhibiting adverse effects of protozoan infection
The present disclosure is directed to interleukin-10 (IL-10) peptides and isolated antibodies that specifically bind to the IL-10 peptides. The IL-10 peptides and the isolated antibodies may be administered alone or as an animal feed additive to treat gastrointestinal protozoan infection in animals.
Owner:WISCONSIN ALUMNI RES FOUND
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com


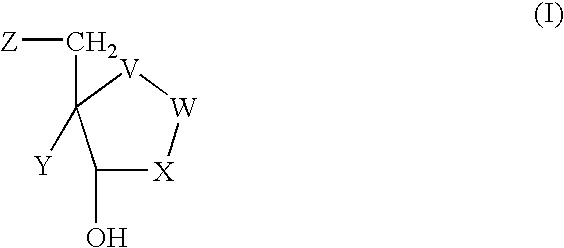
![5h-pyrrolo[3,2-D] pyrimidine inhibitors of nucleoside phosphorylases and nucleosidases 5h-pyrrolo[3,2-D] pyrimidine inhibitors of nucleoside phosphorylases and nucleosidases](https://images-eureka-patsnap-com.libproxy1.nus.edu.sg/patent_img/382d2696-421c-41ef-b7d6-97472bac8513/US07553839-20090630-D00001.png)
![5h-pyrrolo[3,2-D] pyrimidine inhibitors of nucleoside phosphorylases and nucleosidases 5h-pyrrolo[3,2-D] pyrimidine inhibitors of nucleoside phosphorylases and nucleosidases](https://images-eureka-patsnap-com.libproxy1.nus.edu.sg/patent_img/382d2696-421c-41ef-b7d6-97472bac8513/US07553839-20090630-D00002.png)
![5h-pyrrolo[3,2-D] pyrimidine inhibitors of nucleoside phosphorylases and nucleosidases 5h-pyrrolo[3,2-D] pyrimidine inhibitors of nucleoside phosphorylases and nucleosidases](https://images-eureka-patsnap-com.libproxy1.nus.edu.sg/patent_img/382d2696-421c-41ef-b7d6-97472bac8513/US07553839-20090630-C00001.png)








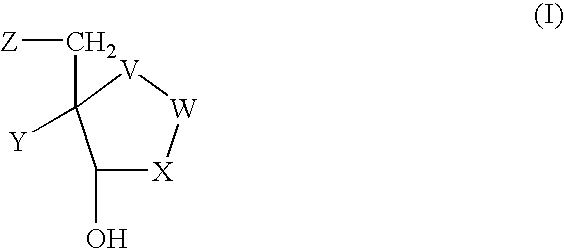
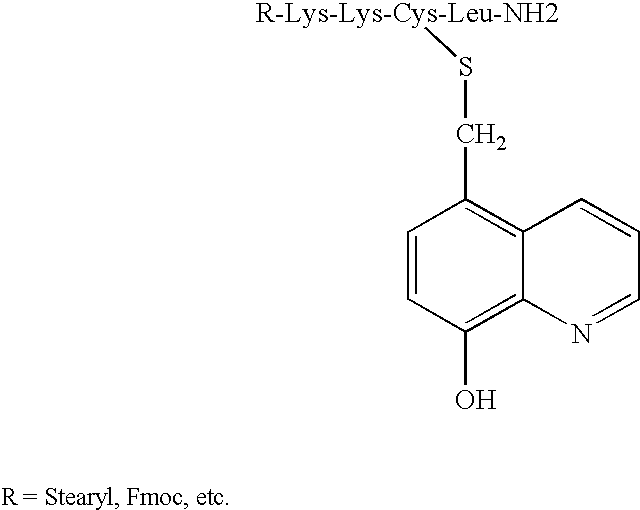

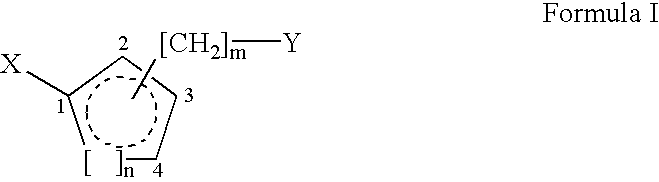
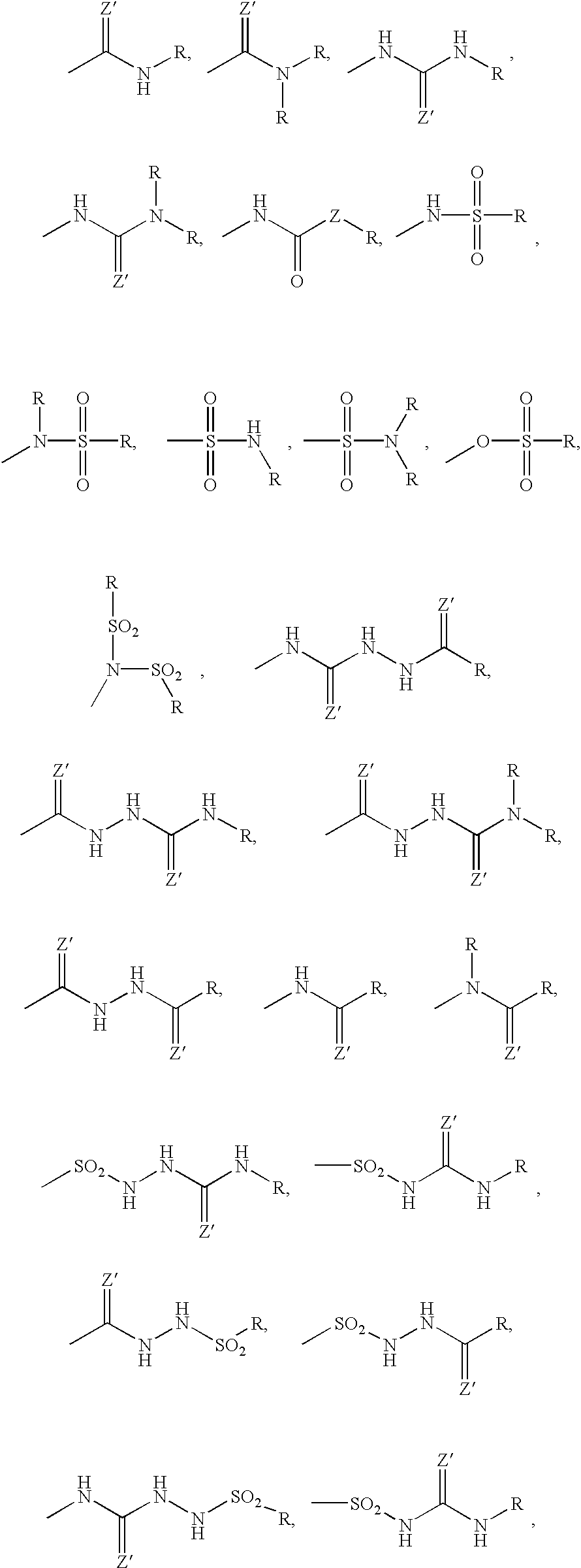













![8-N-substituted-2H-isothiazolo[5,4-b]quinolizine-3,4-diones and related compounds as antiinfective agents 8-N-substituted-2H-isothiazolo[5,4-b]quinolizine-3,4-diones and related compounds as antiinfective agents](https://images-eureka-patsnap-com.libproxy1.nus.edu.sg/patent_img/5a568bcc-1f27-474b-9176-2d1419f17c5e/US07199128-20070403-C00001.png)
![8-N-substituted-2H-isothiazolo[5,4-b]quinolizine-3,4-diones and related compounds as antiinfective agents 8-N-substituted-2H-isothiazolo[5,4-b]quinolizine-3,4-diones and related compounds as antiinfective agents](https://images-eureka-patsnap-com.libproxy1.nus.edu.sg/patent_img/5a568bcc-1f27-474b-9176-2d1419f17c5e/US07199128-20070403-C00002.png)
![8-N-substituted-2H-isothiazolo[5,4-b]quinolizine-3,4-diones and related compounds as antiinfective agents 8-N-substituted-2H-isothiazolo[5,4-b]quinolizine-3,4-diones and related compounds as antiinfective agents](https://images-eureka-patsnap-com.libproxy1.nus.edu.sg/patent_img/5a568bcc-1f27-474b-9176-2d1419f17c5e/US07199128-20070403-C00003.png)