Production of recombinant AAV using adenovirus comprising AAV rep/cap genes
a technology of aav and adenovirus, which is applied in the field of adenoviruses, can solve the problems of not being able to easily scale up for industrial production, not being able to produce high levels of raav in packaging cell lines, and not being able to meet the requirements of large-scale industrial applications, and achieves high levels of raav. high yield, high yield of raav vector
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Cell Lines and Viruses and Maintenance and Propagation Thereof
[0119] All cell lines are maintained in Dulbecco's Modified Eagle's Medium (DMEM; Gibco BRL) supplemented with 10% FBS (Hyclone) and 50 μg / ml of penicillin, 50 μg / ml of streptomycin, and 10 μg / ml of neomycin (Gibco BRL). Human embryonic kidney cell line 293 is obtained from ATCC (CRL 1573). 293-derived 84-31 cells (1) which express adenovirus E1 / E4orf6 proteins, and HeLa-derived B50 cells (7) which express AAV-2 Rep and Cap proteins from the native p5 promoter, are obtained from Dr. Guangping Gao, Institute for Human Gene Therapy, University of Pennsylvania. 293-CG3 is a 293-derived cell line carrying stably integrated copies of AAV ITRs flanking GFP as marker gene (Chen et al., unpublished data). Human adenovirus type 5 (ATCC VR-5) and derived recombinant adenoviruses are propagated on 293 cells and purified through CsCl gradient centrifugation according to the method of Jones and Shenk with modification (2).
example 2
Construction of Plasmids and Generation of Recombinant Adenoviruses
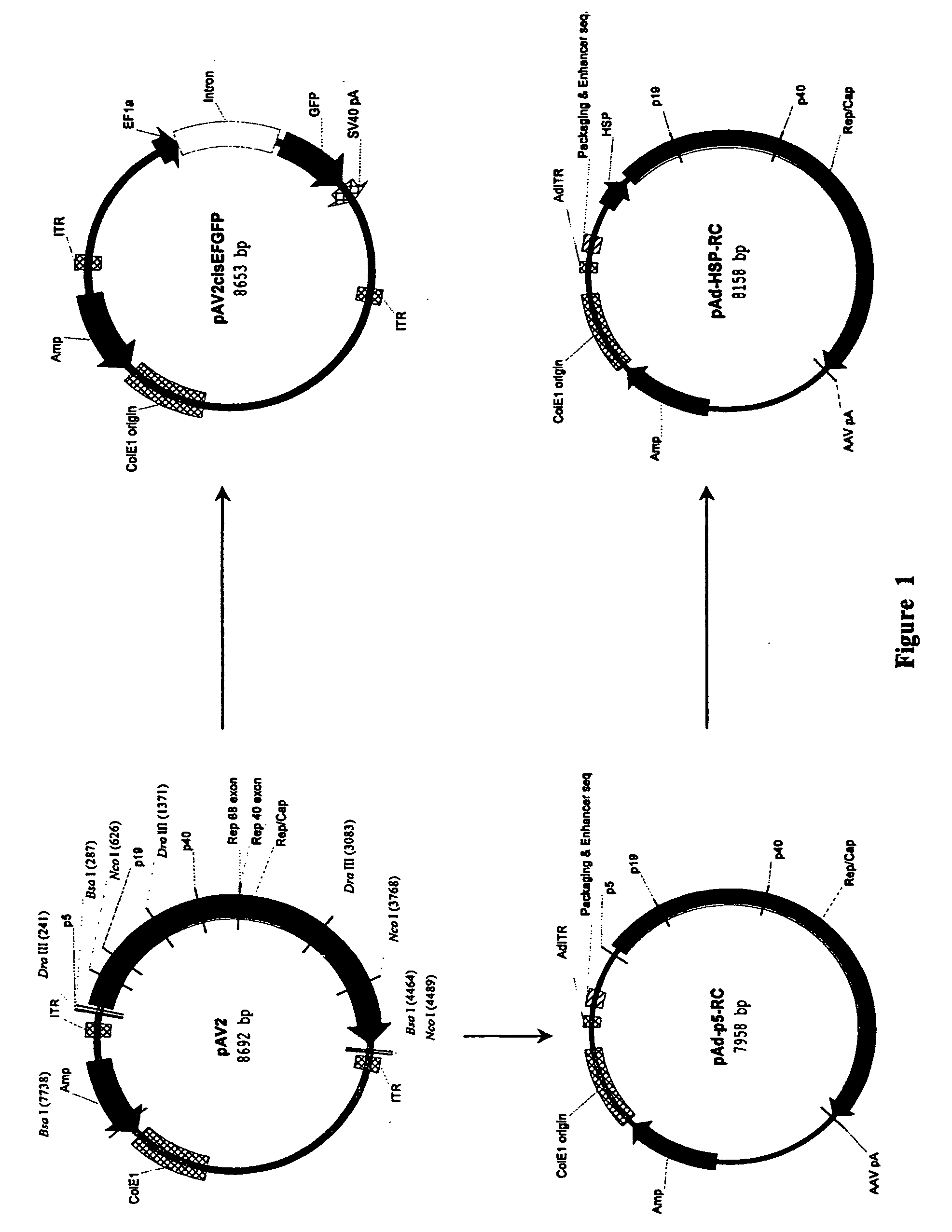
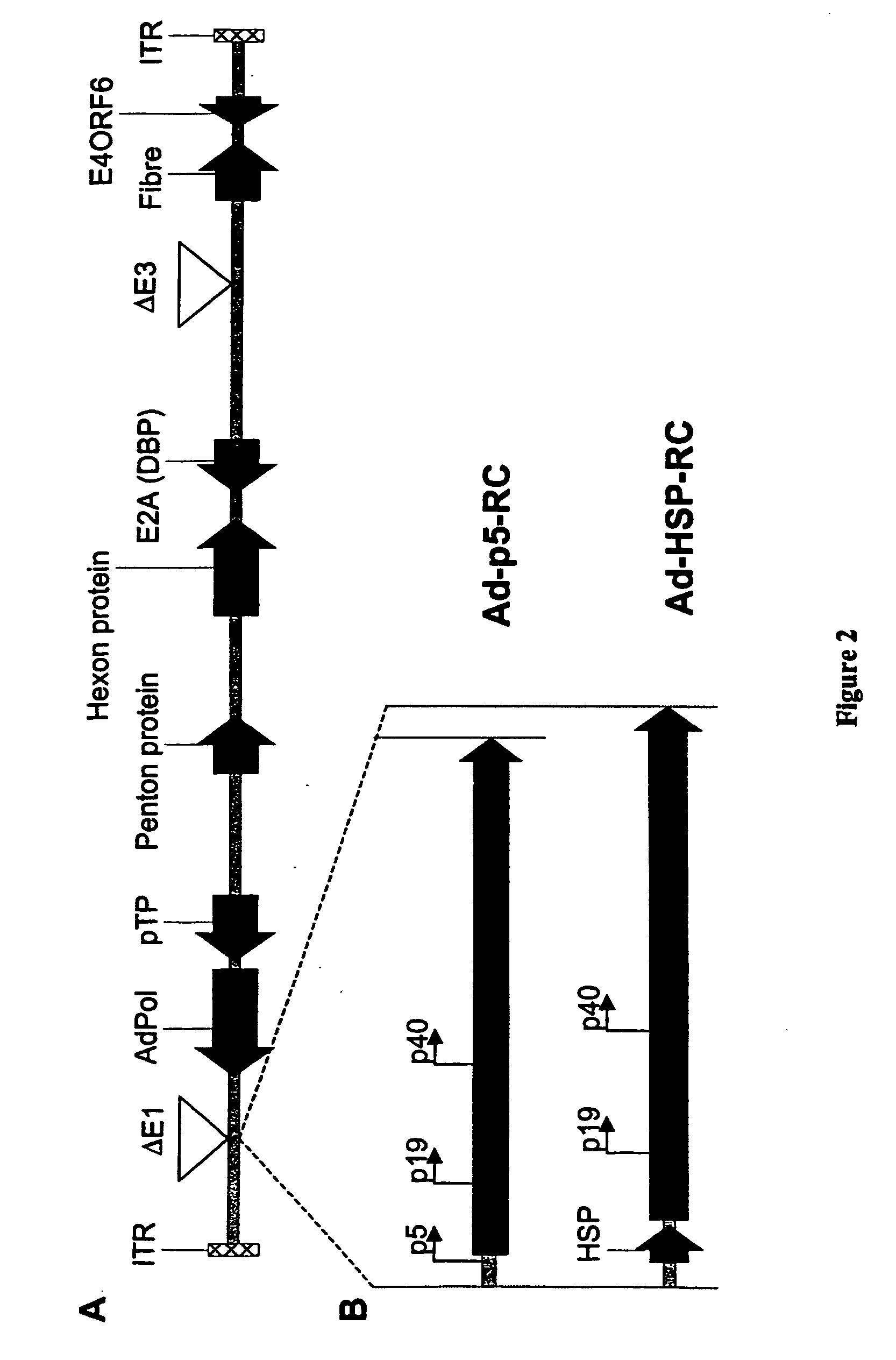
[0120] Standard recombinant DNA techniques are employed to create recombinant plasmids (3). DNA containing the rep and cap sequences of pAV2 (ATCC 37216) between DraIII site (nucleotide 241, upstream of the AAV-2 p5 promoter) and NcoI site (nucleotide 4489, downstream of the polyA signal) is removed and replaced through multiple cloning steps with a DNA cassette containing GFP under the transcriptional control of elongation factor 1 alpha (EF1α) promoter and upstream of the SV40 polyA signal to create pAV2cisEFGFP (FIG. 1). The AAV-2 rep and cap genes located between a Dra III site (nucleotide 241, upstream of the p5 promoter) and a BsaI site (nucleotide 4464, downstream of the polyA signal) are further subcloned to obtain pAd-p5-RC (FIG. 1). A small DNA fragment between nucleotides 241 and 287 of pAd-p5-RC containing the p5 promoter is removed and replaced with a Drosophila melanogaster minimal heat shock protein (...
example 3
Transfection of 293 Cells and Selection of the 293-CG3 Stable Cell Line
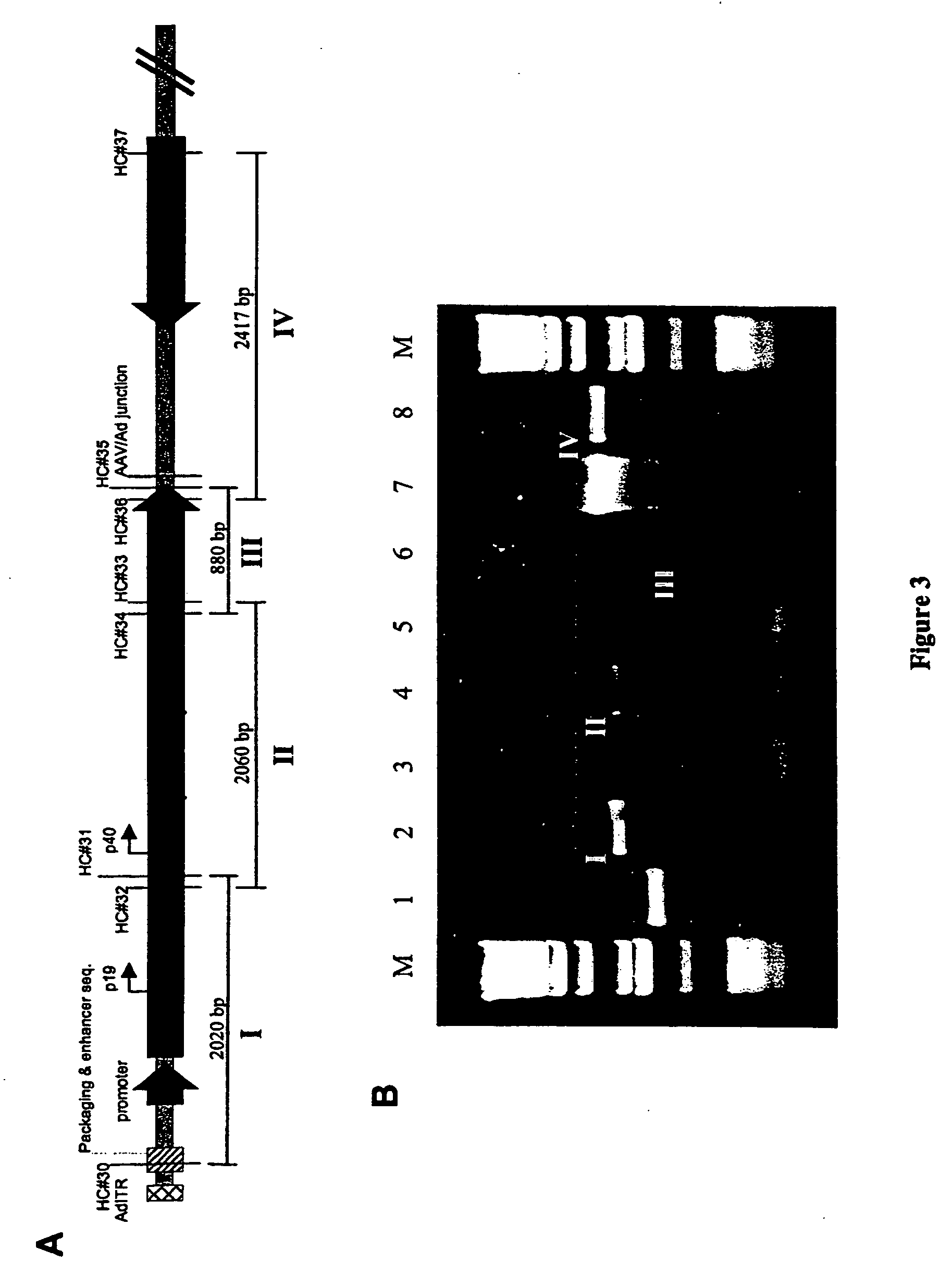
[0121] 293 cells are grown to ˜70% confluency in 6-cm tissue culture dishes and co-transfected overnight with 1 μg pIRES1neo and 10 μg pAV2cisEFGFP by the calcium phosphate transfection method. The monolayer is replenished with fresh medium containing 10% FBS and cultured for 24 hours. Following trypsinization, cells are seeded at a 1:20 dilution in fresh medium containing 10% FBS. After incubation for another 24 hours, fresh medium containing 1,250 μg / ml of G418 (Gibco BRL) is added to the cell monolayer for genetic selection of G418-resistant cells. The medium containing G418 is replaced every 3-4 days to allow formation of G418-resistant cell colonies. A total of fifty colonies are picked, six of which demonstrate constitutive GFP expression. These six clones are expanded and tested for their ability to rescue functional rAAV by transfection with pBV-EiOV-RC, a plasmid that carries adenovirus E2A, E4ORF6, and...
PUM
| Property | Measurement | Unit |
|---|---|---|
| diameter | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com