Compositions comprising streptococcus pneumoniae polysaccharide-protein conjugates and methods of use thereof
a technology of streptococcus pneumoniae and conjugates, which is applied in the field of multivalent immunogenic compositions, can solve the problems of limited serotype coverage of prevnar® in certain regions of the world, infants and young children's poor response to unconjugated pneumococcal polysaccharides, etc., and achieve enhanced immunogenicity and superior stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Preparation of S. Pneumoniae Capsular Polysaccharides
[0484]Methods of culturing pneumococci are well known in the art. See, e.g., Chase, 1967, Methods of Immunology and Immunochemistry 1:52. Methods of preparing pneumococcal capsular polysaccharides are also well known in the art. See, e.g., European Patent No. EP 0 497 524 B 1. The process described below generally follows the method described in European Patent No. EP 0 497 524 B1 and is generally applicable to all pneumococcal serotypes.
[0485]Isolates of pneumococcal strains for serotypes 6C, 23B, and 31 were obtained from Centers for Disease Control and Prevention (Atlanta, Ga.). Strains for serotypes 3, 8, 10A, 11A, 12F, 15B, 22F, and 33F were obtained from the University of Pennsylvania (Dr. Robert Austrian). Strains for serotypes 17F and 19A were obtained from the FDA Office of Biologics (Dr. John Robbins). Serotype 7F was obtained from the State University of New York, Downstate Medical Center (Dr. Gerald Schiffman). Isolate...
example 2
Purification of Pneumococcal Polysaccharides
[0488]The purification process for the pneumococcal polysaccharides consisted of several centrifugation, depth filtration, concentration / diafiltration operations, and precipitation steps. All procedures were performed at room temperature unless otherwise specified.
[0489]Inactivated broth from the fermentor cultures of S. pneumoniae were flocculated with a cationic polymer (such as BPA-1000, TRETOLITE® (Baker Hughes Inc., Houston, Tex.), Spectrum 8160, poly(ethyleneimine), and Millipore pDADMAC). The cationic polymers binded to the impurity proteins, nucleic acids and cell debris. Following the flocculation step and an aging period, flocculated solids were removed via centrifugation and multiple depth filtration steps. Clarified broth was concentrated and diafiltered using a 100 kDa to 500 kDa MWCO (molecular weight cutoff) filter. Diafiltration was accomplished using Tris, MgCl2 buffer and sodium phosphate buffer. Diafiltration removed res...
example 3
Structure Identity Analysis of Certain Pneumococcal Serotypes by NMR Testing
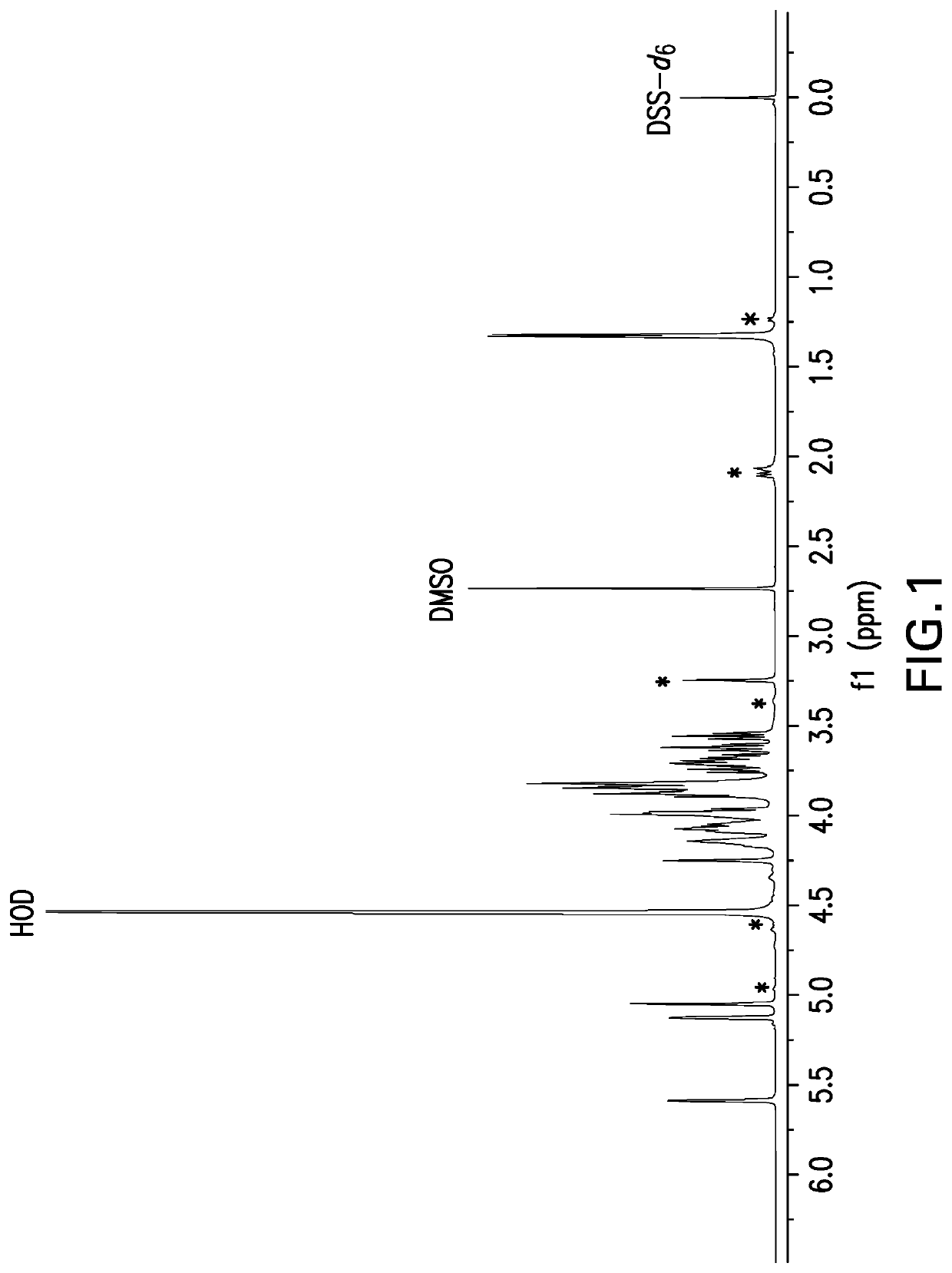
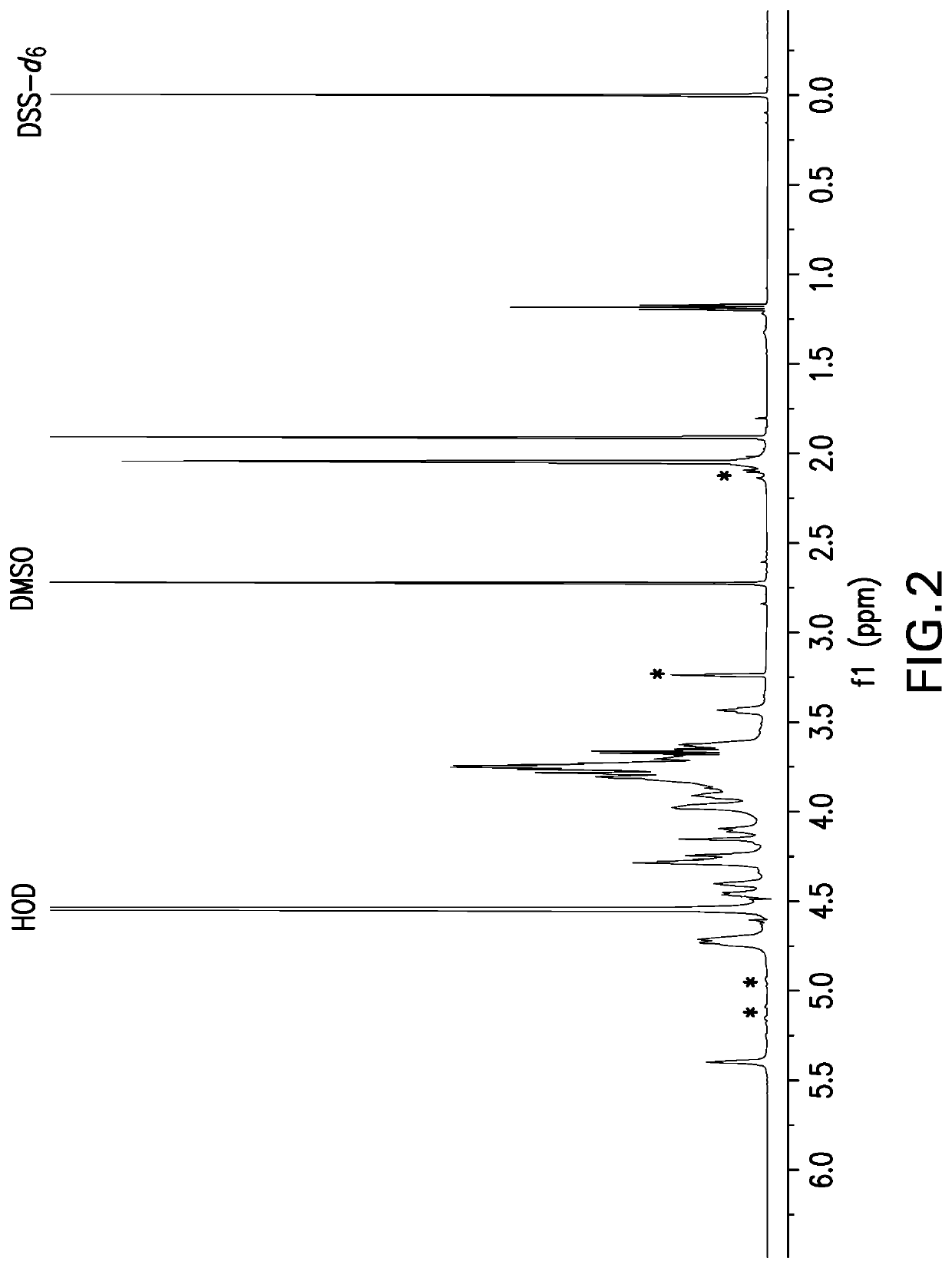
[0491]Samples for NMR analysis were prepared by dissolving polysaccharide powder at 5 mg powder / mL solution in deuterium oxide (D2O) containing 0.01% dimethyl sulfoxide (DMSO) and 0.01% 2,2-Dimethyl-2-silapentane-5-sulfonate-d6 sodium salt (DSS-d6). DMSO is an internal standard that was used for quantitative analysis and DSS-d6 was used to set the chemical shift scale to 0 ppm. A one-dimensional proton NMR data set was acquired at 50° C. and a portion of spectrum containing the anomeric resonances as then selectively written as x, y coordinates to an ASCII file for analysis using a Microsoft Excel workbook. The Y coordinates (i.e., spectral profile) were then compared to spectral profiles of capsular bacterial polysaccharides in a reference database. The reference profiles were generated in a similar manner on selected preparations of each serotype thereafter designated as the reference lot. A pairwise compa...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| Mw | aaaaa | aaaaa |
| Mn | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com