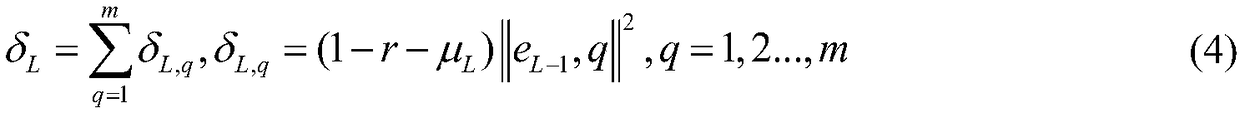Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
35 results about "Study Type" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Clinical study design is the formulation of trials and experiments, as well as observational studies in medical, clinical and other types of research (e.g., epidemiological) involving human beings.
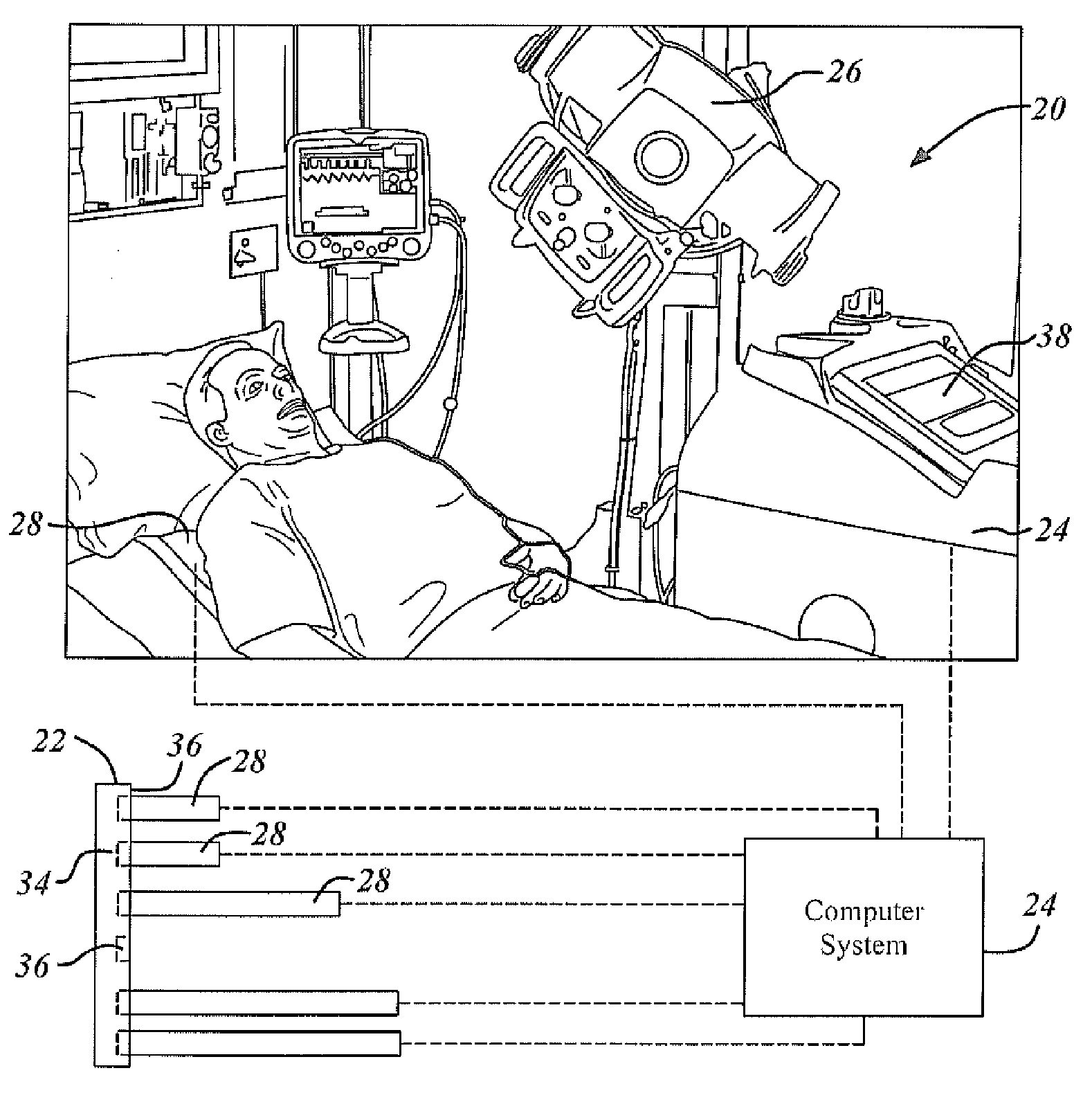
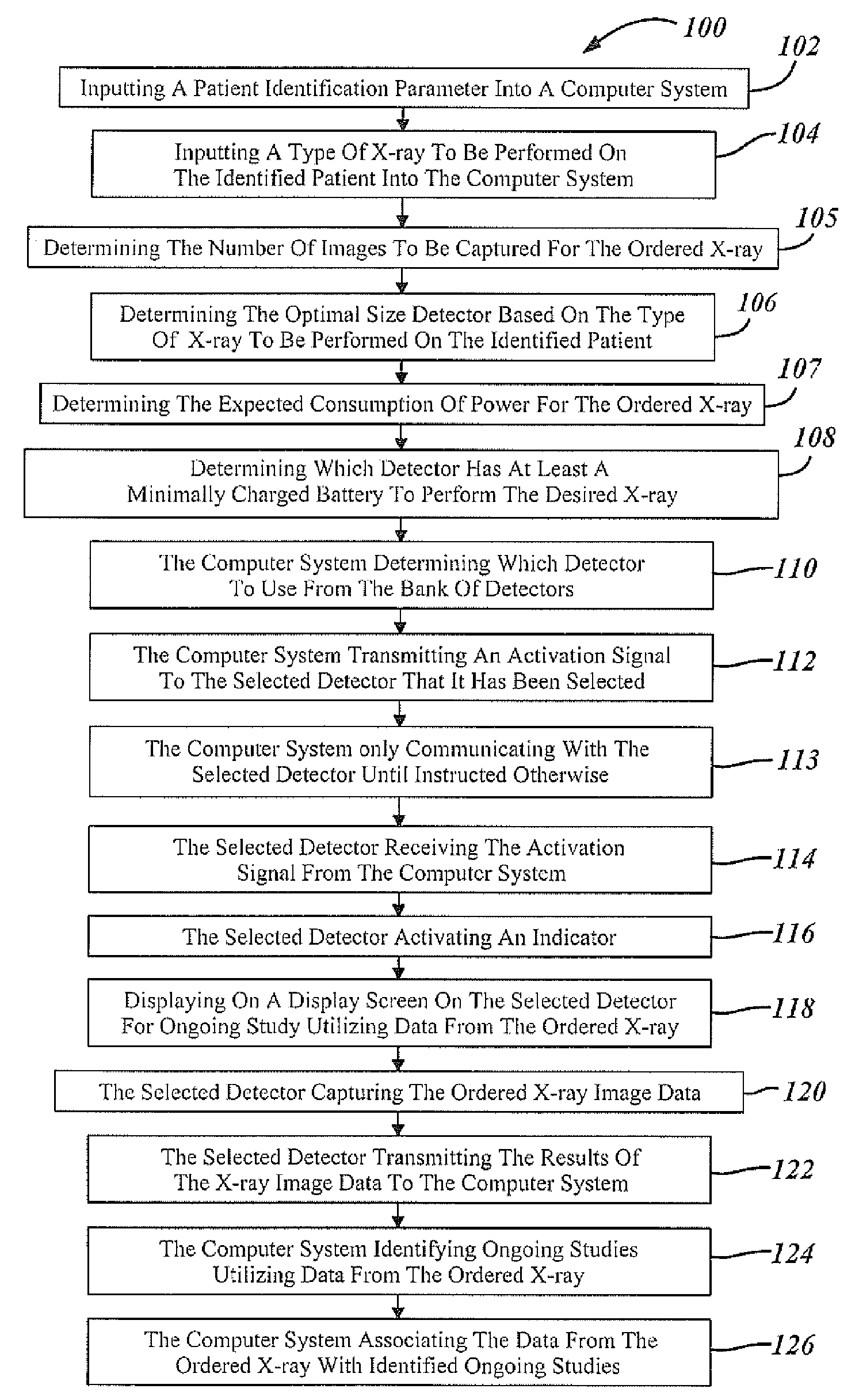
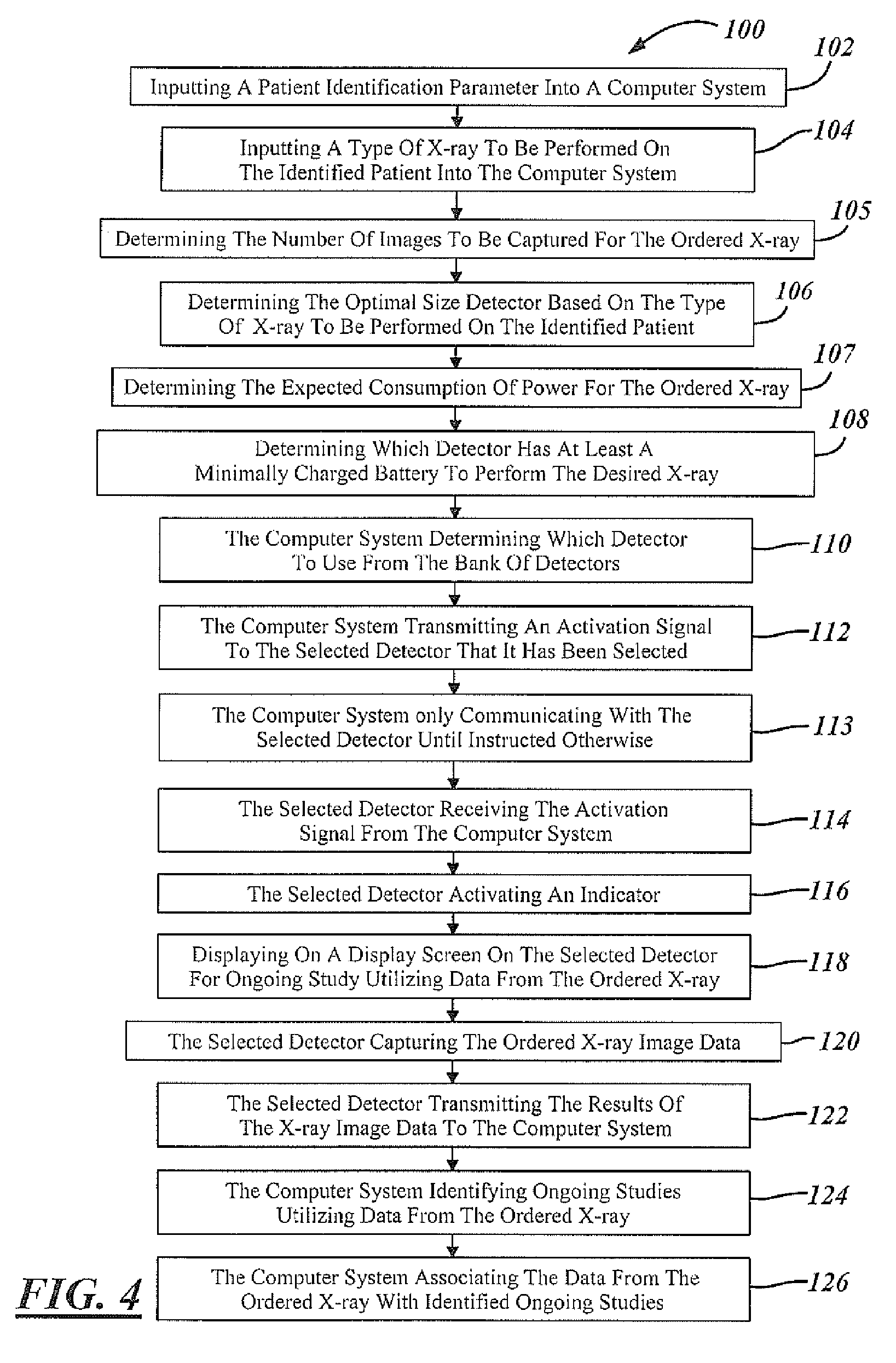
Automatic detector selection by study type
Methods of using an X-ray system having a central computer system, an X-ray source and a bank of wireless detectors. The methods include an automated X-ray system that requires the input of a type of X-ray to be performed on a patient, and the systems determine and identify which detector from the detector bank is appropriate for the ordered test.
Owner:GENERAL ELECTRIC CO
Systems, Methods and Computer Program Products for Preparing, Documenting and Reporting Chemical Process Hazard Analyses
InactiveUS20070122911A1Efficiently meetFacilitate complianceData processing applicationsBiological testingStudy TypeSolution analysis
A process hazard analysis (PHA) is performed in a data processing system. A chemical process to be evaluated is selected, after which a study type to be performed on the chemical process is also selected. The study type is conducted to evaluate the chemical process for the presence of hazard scenarios and any associated deficiencies requiring recommendations. After conducting the study, resolution plans to address the recommendations are generated. The study type used in the analysis may be customized or may be a known study type. The study type may use a risk matrix to generate a risk ranking for the hazard scenario. The risk matrix used may be created by the user, or may be a known risk matrix from another study type. After a resolution plan is generated, a resolution database may also be generated.
Owner:EASTMAN CHEM CO
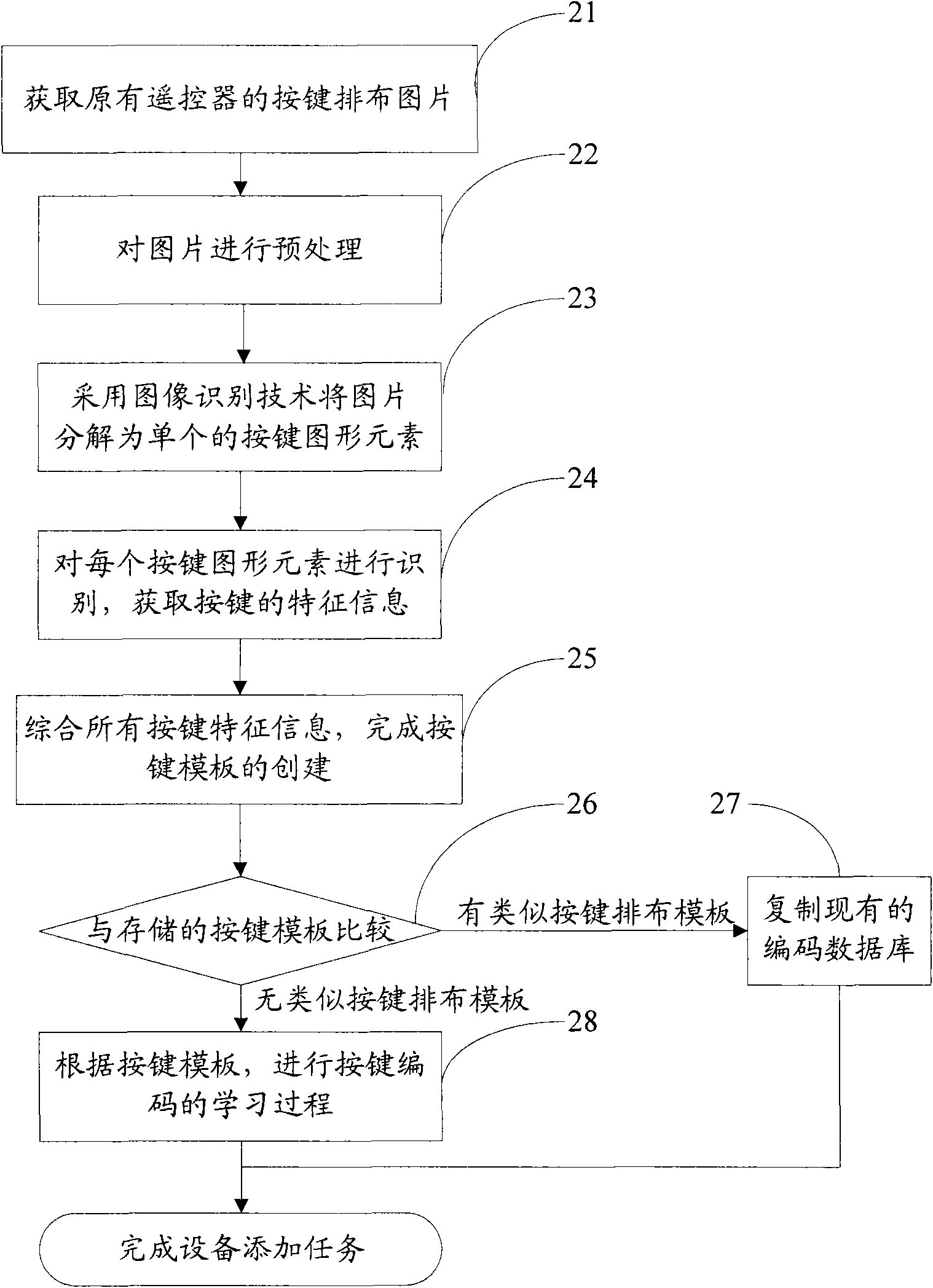
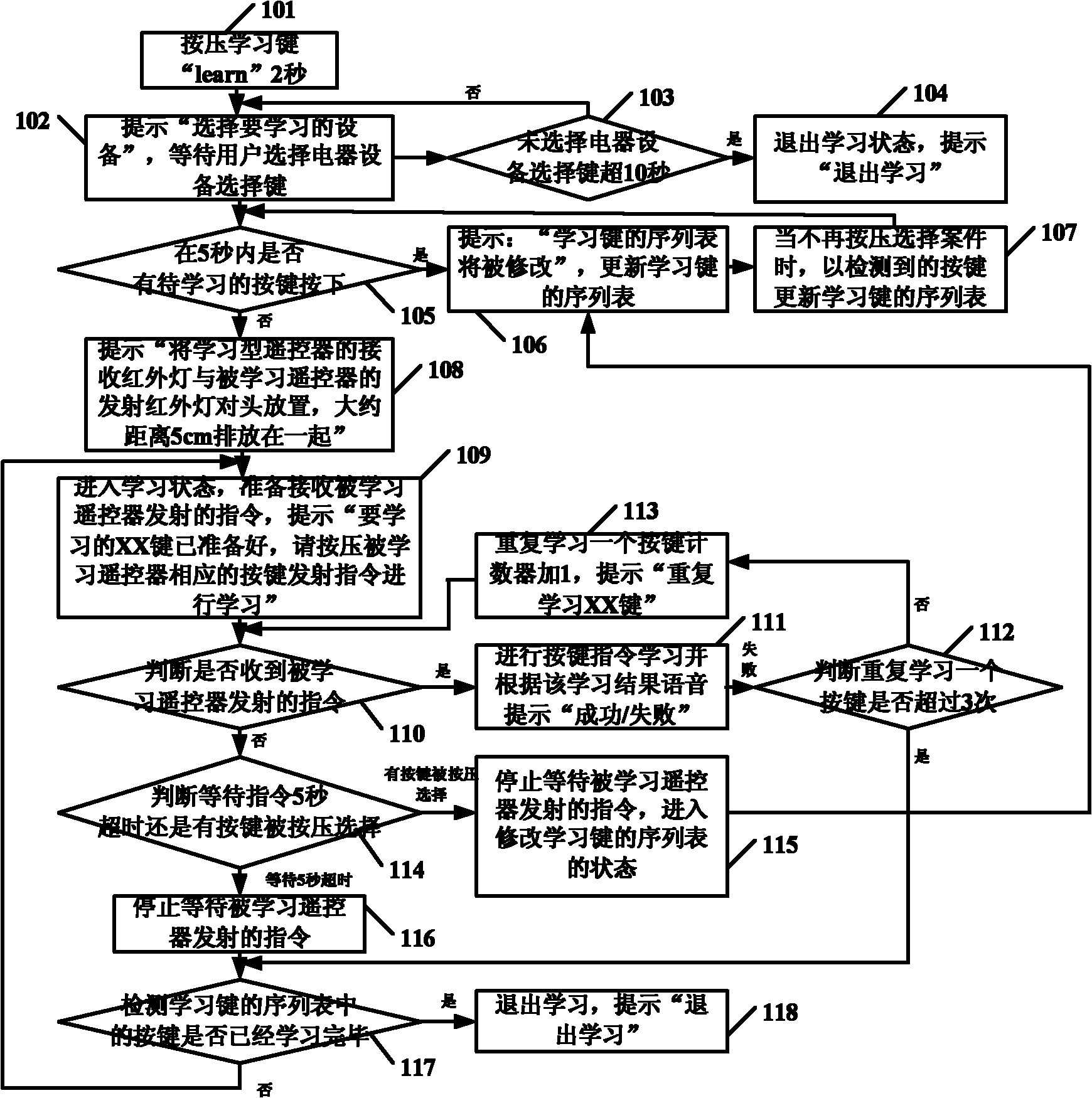
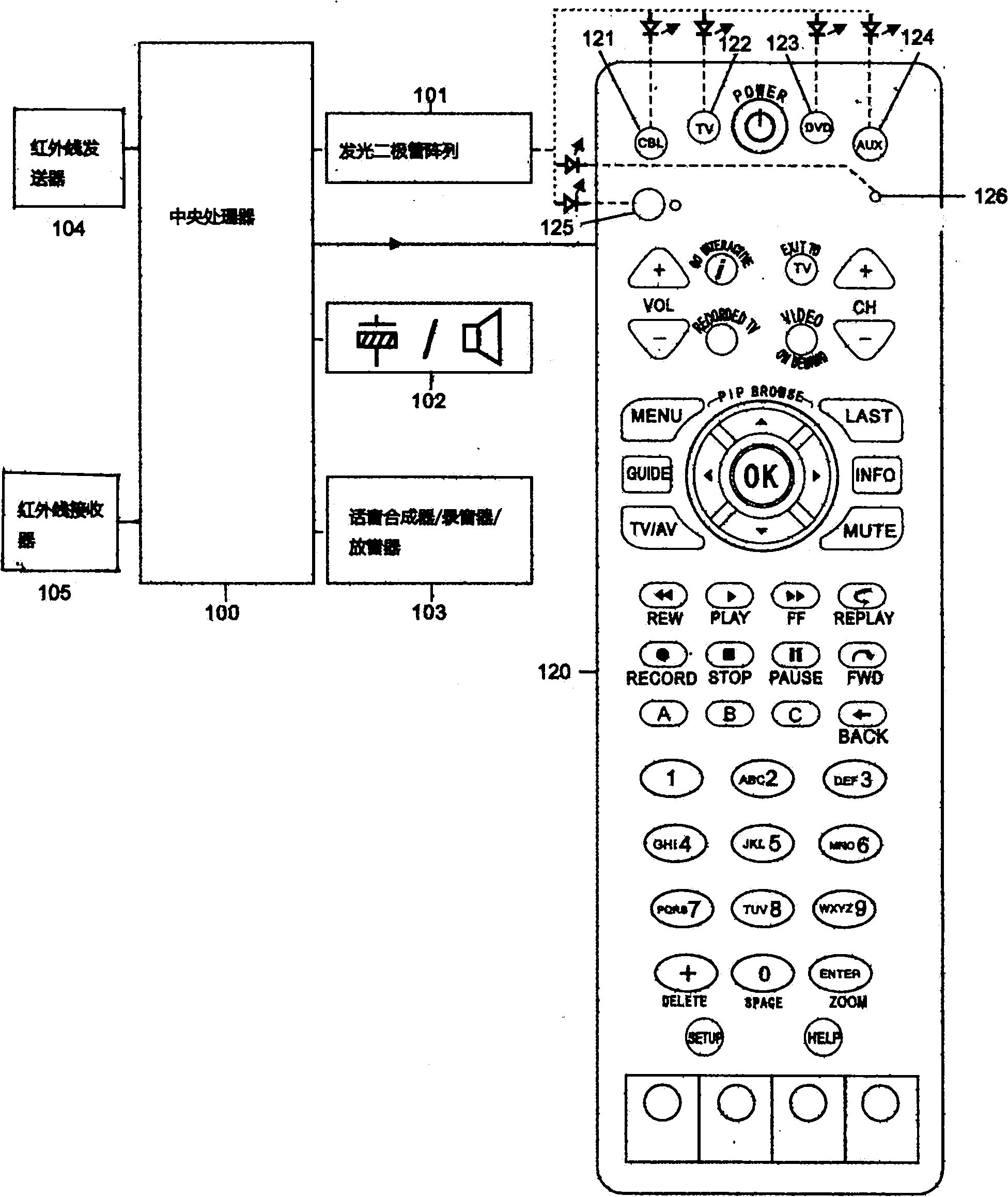
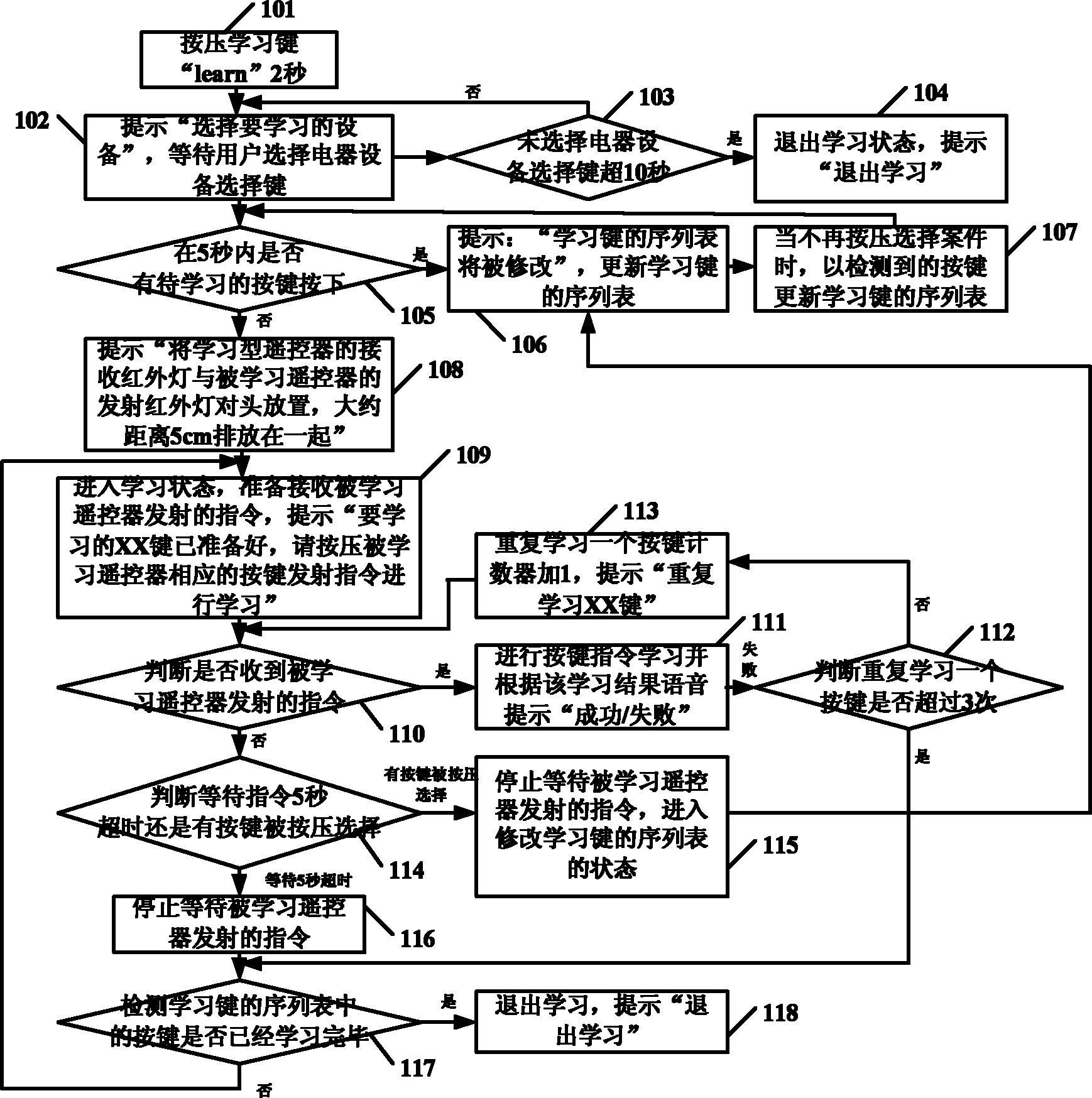
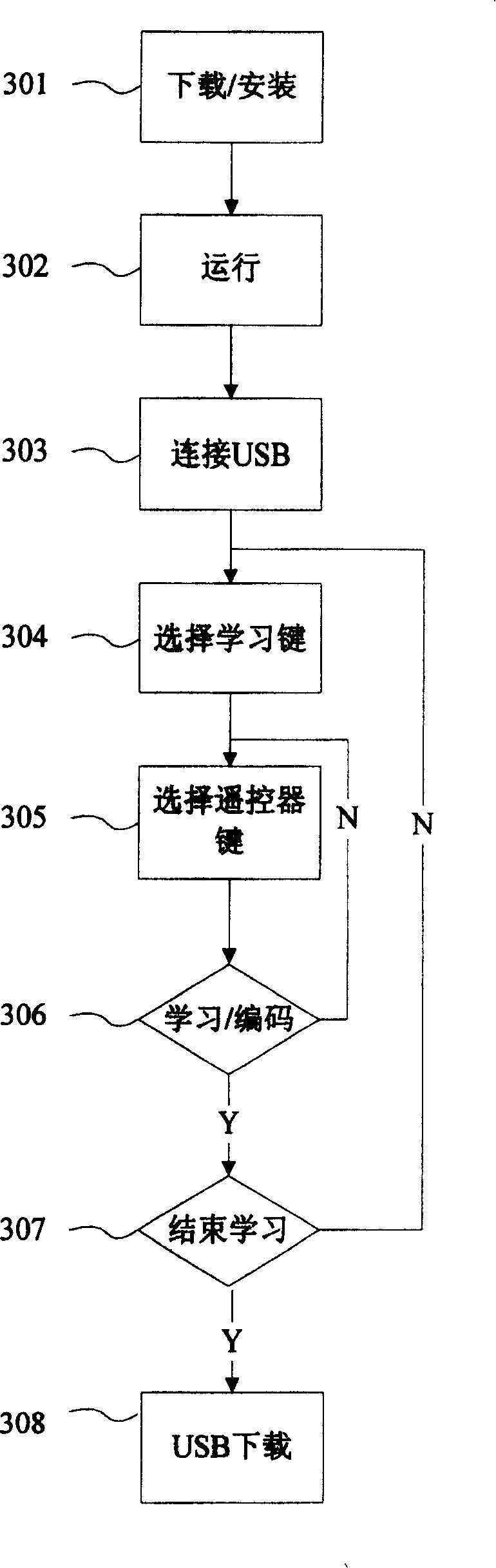
Study type remote controller and press key template establishment method thereof
InactiveCN101799975AEasy to useNon-electrical signal transmission systemsCharacter and pattern recognitionKey pressingControl signal
The invention provides a study type remote controller and a press key template establishment method thereof. The study type remote controller comprises a remote control signal sending device, a display screen, an image pick-up device and a processing unit, wherein the image pick-up device is used for obtaining press key distribution pictures of other remote controllers, and the processing unit used for analyzing and processing the press key distribution pictures and generating press key templates. The press key template establishment method of the study type remote controller comprises the following steps: step a, obtaining the press key distribution pictures of the original remote controller; and step b, analyzing and processing the press key distribution pictures of the remote controller to generate the press key templates. The study type remote controller and the press key template establishment method thereof provided by the invention can directly convert the picture information into the press key template information, so the remote control operation interface of the study type remote controller is similar to or even identical to that of the original remote controller, and the invention brings great convenience for the use by users.
Owner:TCL CORPORATION
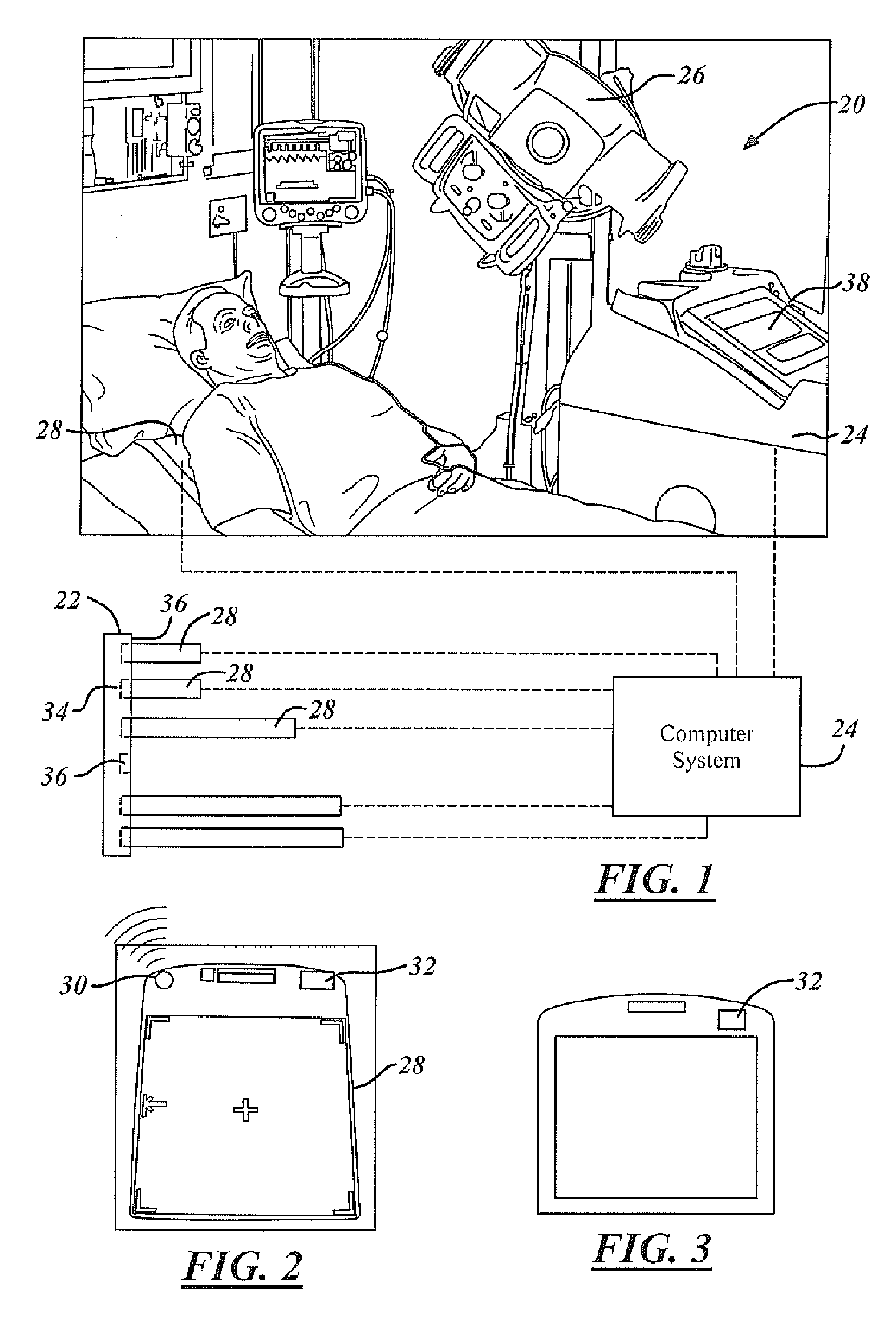
Automatic detector selection by study type
Methods of using an X-ray system having a central computer system, an X-ray source and a bank of wireless detectors. The methods include an automated X-ray system that requires the input of a type of X-ray to be performed on a patient, and the systems determine and identify which detector from the detector bank is appropriate for the ordered test.
Owner:GENERAL ELECTRIC CO
Method for studying remote controller and studying type remote controller
InactiveCN101986363AHigh degree of intelligenceFlexible and short operating stepsElectric signal transmission systemsNon-electrical signal transmission systemsStudy TypeStudy methods
Owner:SAISHENGYUAN ELECTRONICS SHENZHEN
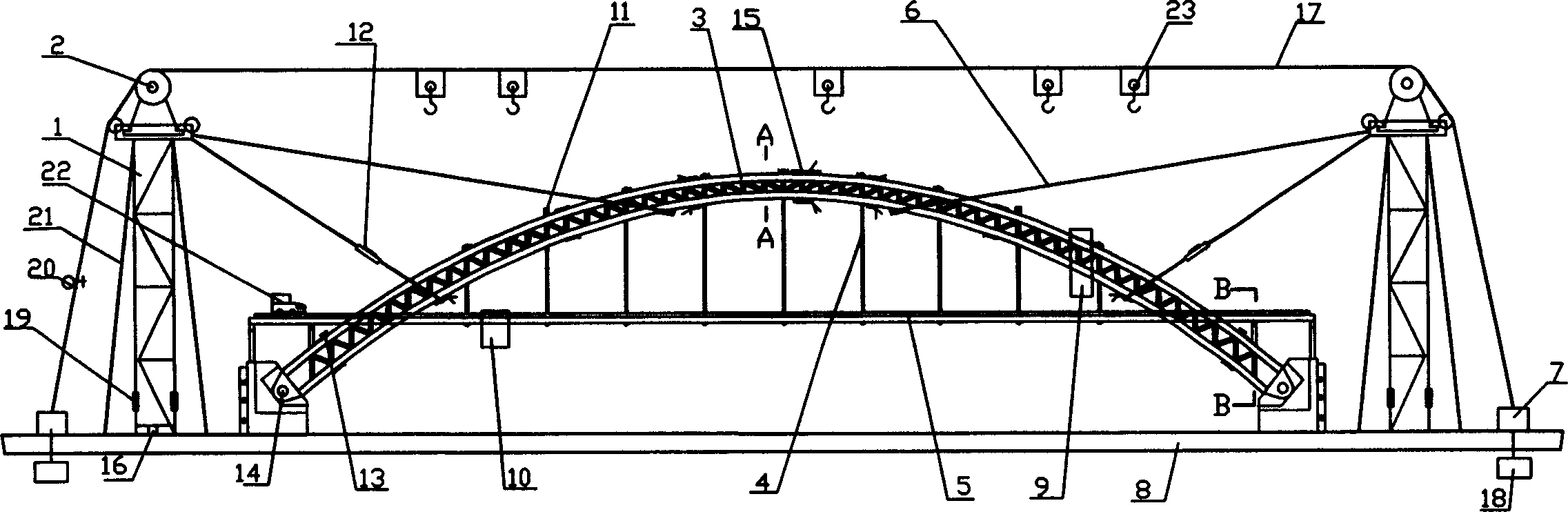
Arch bridge expiremental stage
The invention relates to arch bridge test-bed, comprises the stage board and two tower frame equipped on the stage board, each top of the tower frame has cable saddles, between which there are cords, on which suspend claw equipped; on the stage board which is between two tower frames the arch bridge model which is used to installation and experiment equipped. The invention provide a basic chance for opening comprehend, design and study types of experiments to the students, it can train proficient manager, inspector and surveillant in the numbers, let the students have chance to undertake the design and study types of experiments.
Owner:WUHAN UNIV OF TECH
Studying and up grading system and method for enhanced
ActiveCN1818988AEasy to identifyImprove conversion performanceElectric signal transmission systemsNon-electrical signal transmission systemsStudy TypeGrade system
An enhanced study type of the multi-purpose remote controller study and upgrade ability system and method, the system includes: study type of the multi-purpose remote controlling equipment, the data processing equipment and the fountain controller equipment, the data processing equipment includes a personal computer and a applied program operating on the personal computer, the data processing equipment transform the undee data of the order coding to the coding that accord with the request format of the study type of the multi-purpose remote controller inner sending code equipment, then write the coding into the study type of the multi-purpose remote controller to finish the study. The advantage of the invention is: enhancing the identified and transforming ability of the new order coding format by upgrading the applied program software continually, that becomes to possibility for all type of code for the study type of the multi-purpose remote controller.
Owner:HANGZHOU BREMAX TECH +1
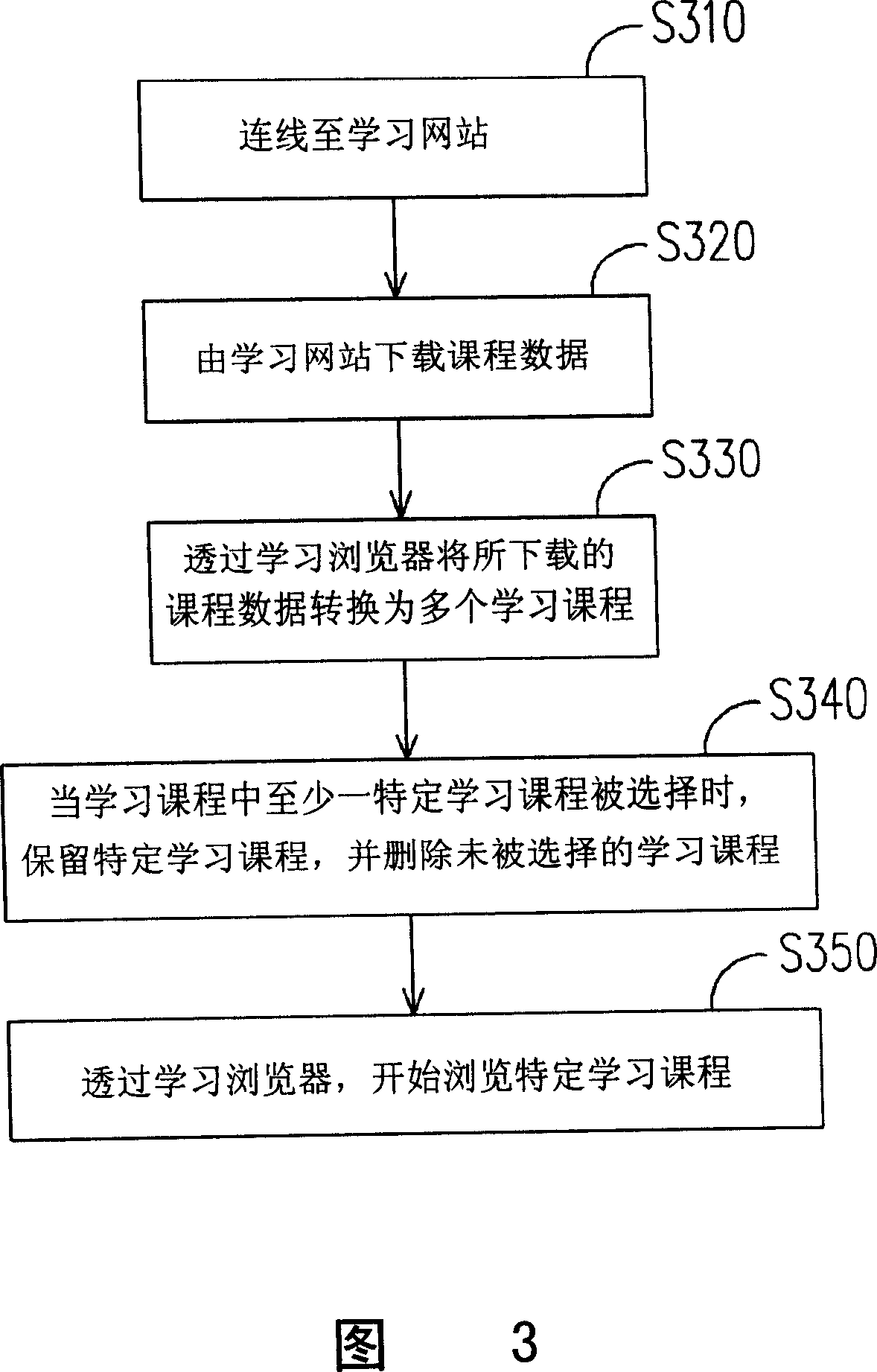
Learning method and portable learning system based on network
InactiveCN101064068AEasy updateLearning diversityElectrical appliancesSpecial data processing applicationsStudy TypeStudy methods
This invention relates to one study method and portable study system based on network, which comprises the following steps: the study browser converts the download programs into multiple study programs; providing multiple selections with multiple study types. This portable study system device can download program data through connection socket to make learner update study program.
Owner:INVENTEC(NANJING) TECH CO LTD
Decoding/coding method of infrared pulse signal
InactiveCN104217576ASave storage spaceNon-electrical signal transmission systemsStudy TypeOperating system
The invention provides a decoding / coding method of infrared pulse signal. In the method, a studying-type infrared remote-controlling device receives a remote-controlling command of buttons, wherein the remote command is sent by an infrared controller. A controlling command sent by the infrared controller is stored in a manner of a format including a code length + a controlling code + an index number + library content. If the library contents are same, a repeatedly storing operation is not carried out and only the library content which is read at the first time is stored. Same library content is enabled to be corresponding to the same index number. When the controlling command is restored, the library content, which is corresponding to the index number, is read according to the index number, so that when the remote-controlling command is stored, stored data can be compressed so that a storage space of the data can be effectively compressed.
Owner:广州市河东电子有限公司
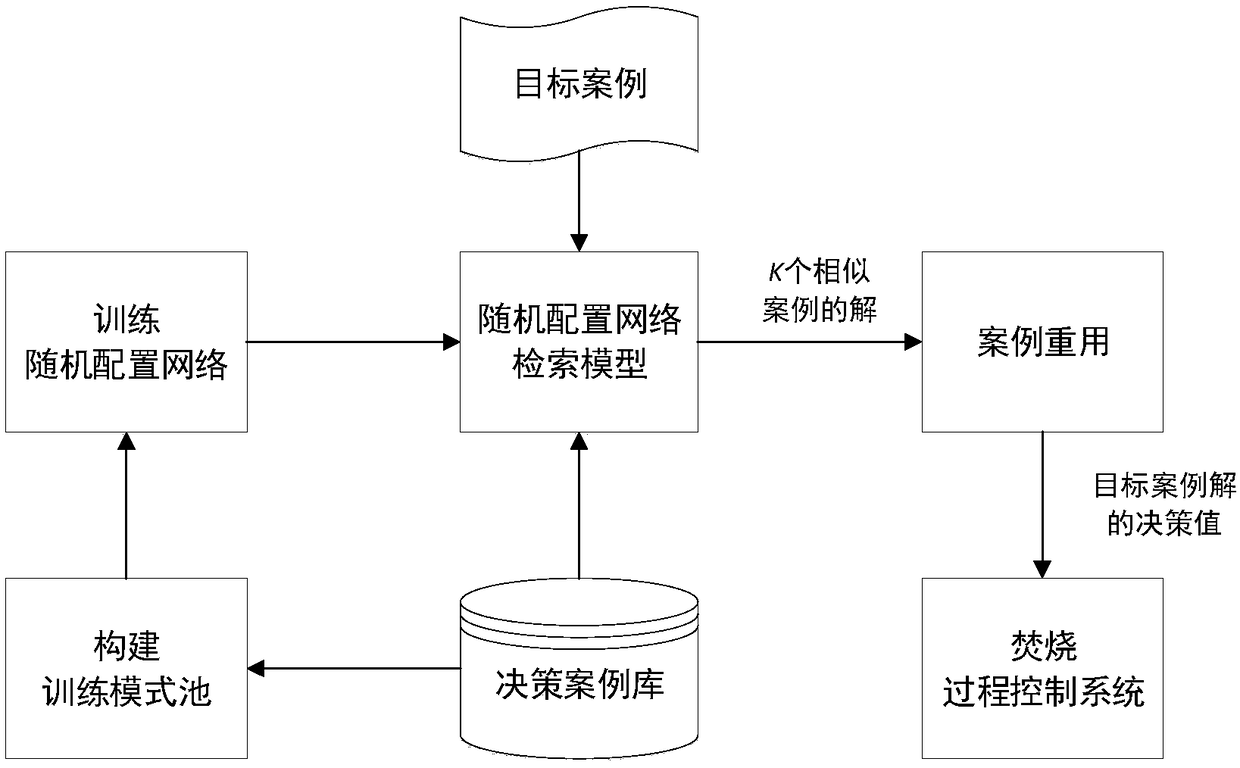
Automatic combustion optimized decision method for waste incineration process
InactiveCN108224446AAvoid subjectivityShorten the timeIncinerator apparatusCombustion systemCombustion
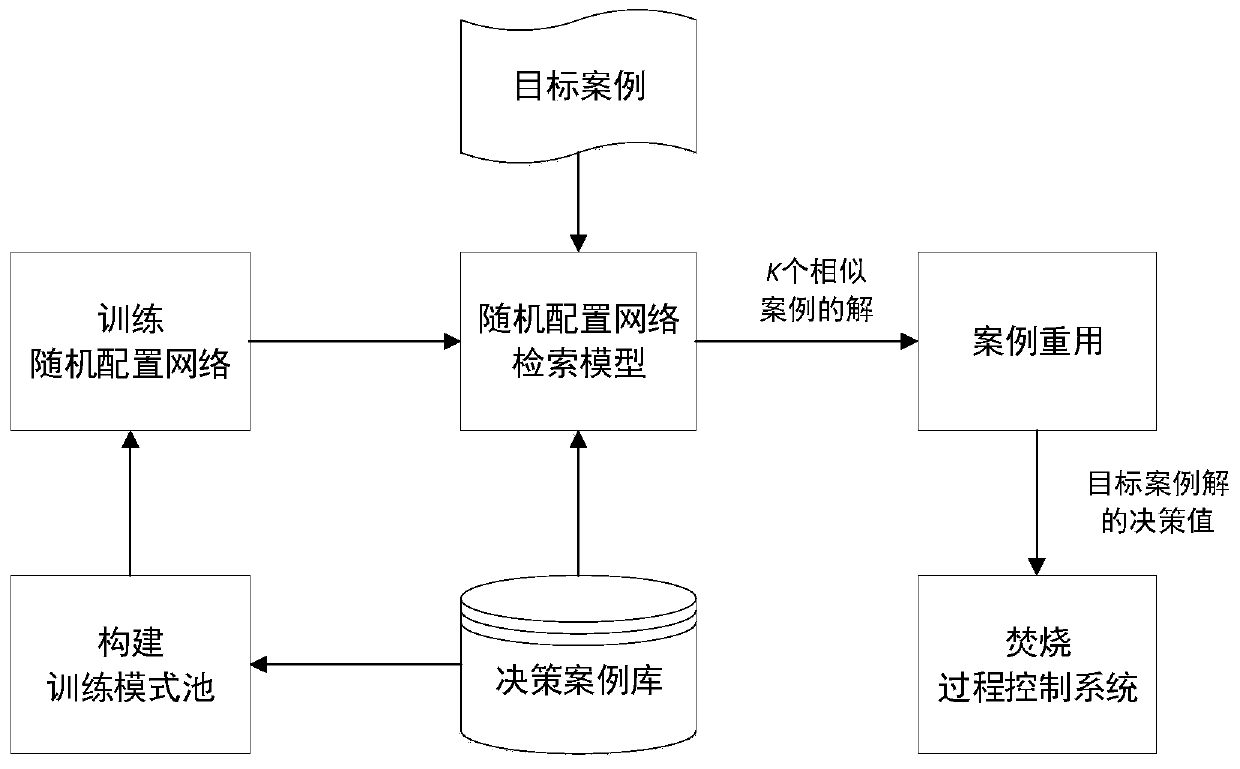
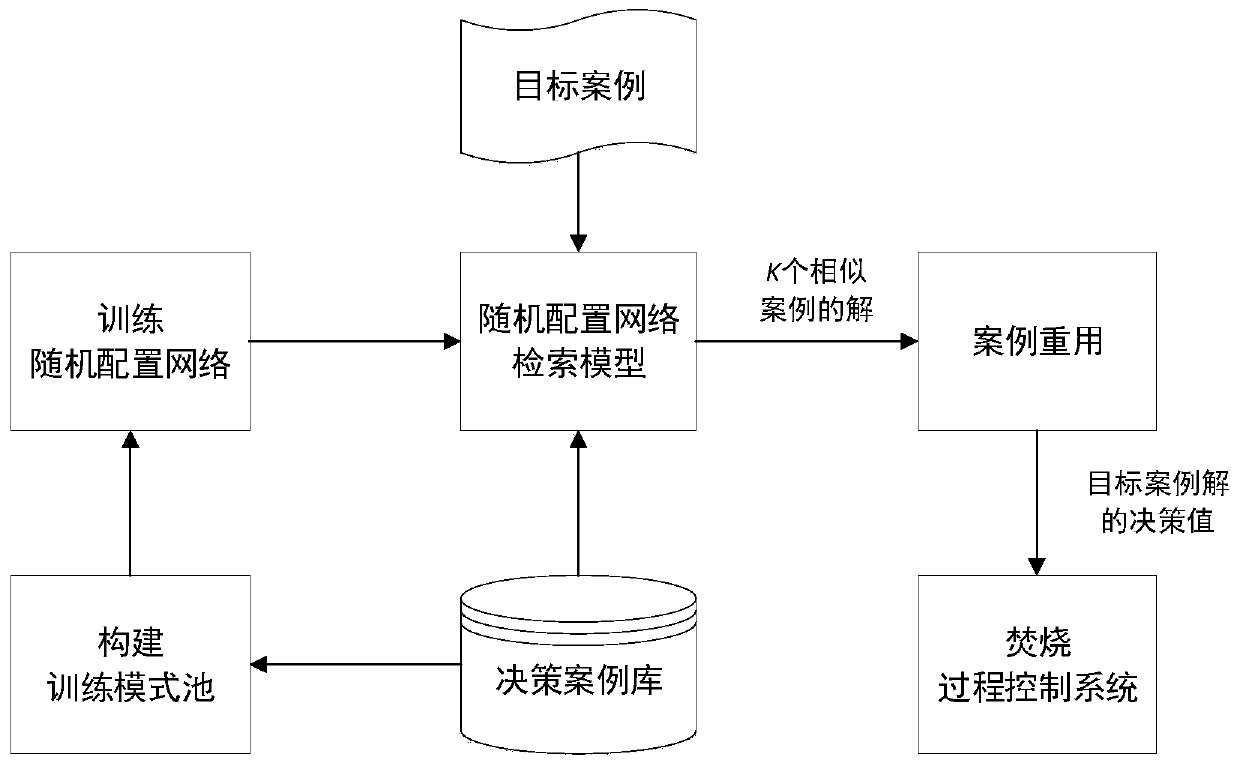
The invention relates to the technical field of urban solid waste incinerator optimal control, in particular to an automatic combustion optimized decision method for the waste incineration process. Bymeans of the optimized decision of key manipulated variables (feeder speed and fire grate speed), an automatic combustion system operates within an ideal range, and the method mainly comprises the following steps that (1) a decision case library is set up according to historical data of the incineration process; (2) a training pattern pool is constructed; (3) a stochastic allocation network is trained according to a study type pseudometric measuring criterion, and therefore a stochastic allocation network retrieval model is obtained; (4) a target case is input into the stochastic allocation network retrieval model, and K similar case solutions are obtained; (5) the average value of the K similar case solutions is obtained through case reusing, and therefore the decision value of a targetcase solution (feeder speed and fire grate speed) is obtained and output to an incineration process control system; and (6) the fourth step and the fifth step are repeated to achieve the automatic combustion real-time optimized decision process of the incineration process.
Owner:BEIJING UNIV OF TECH
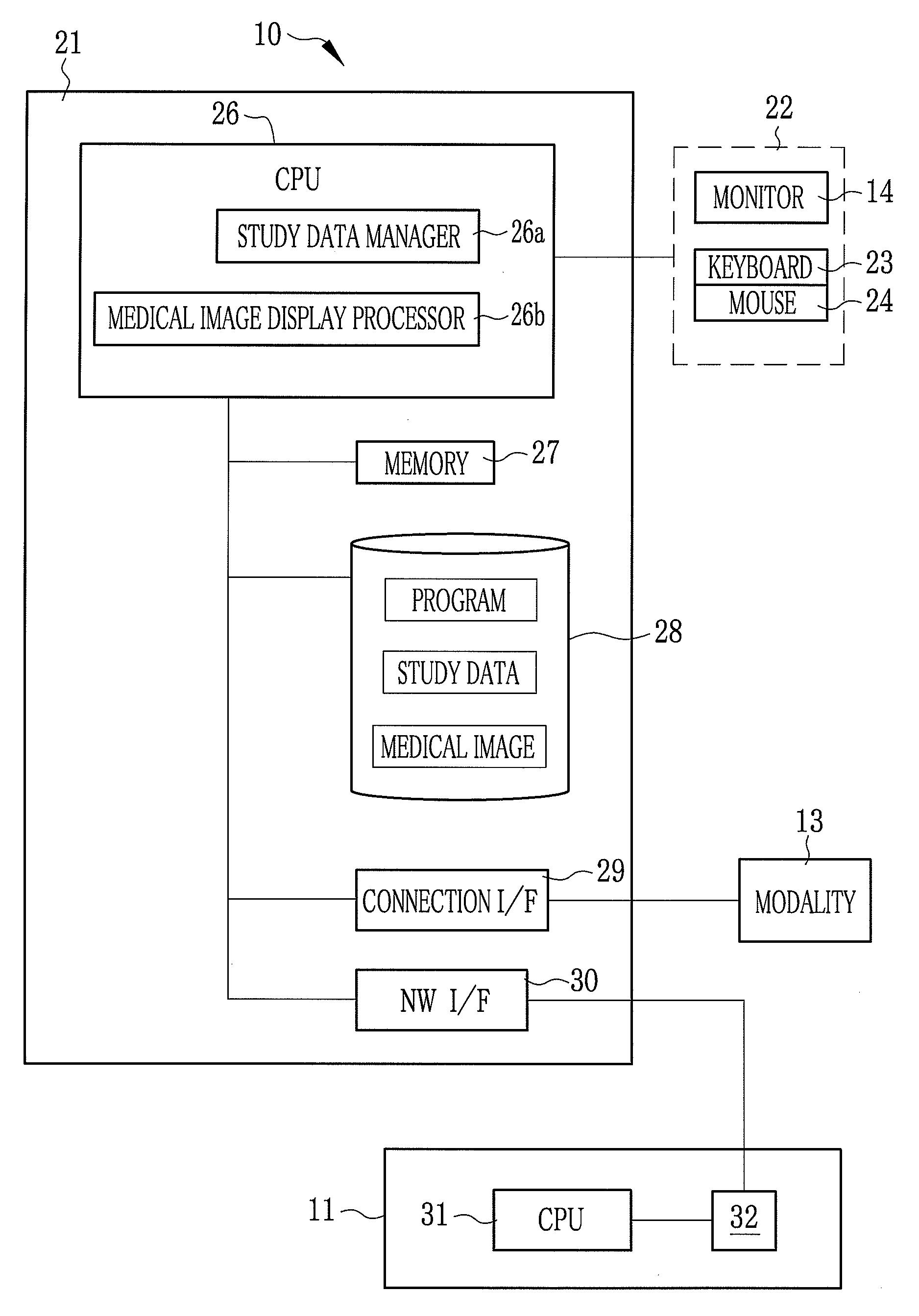
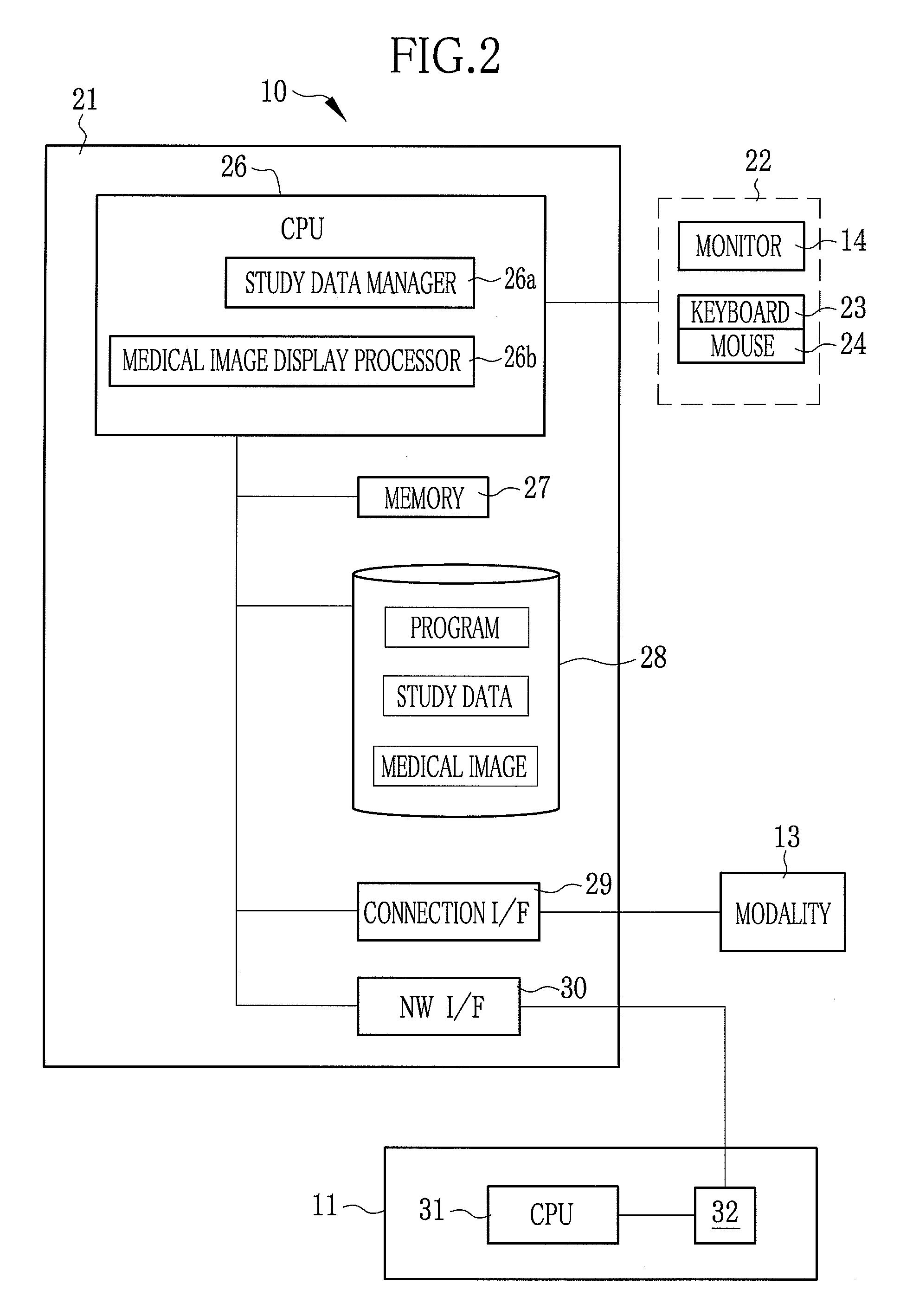
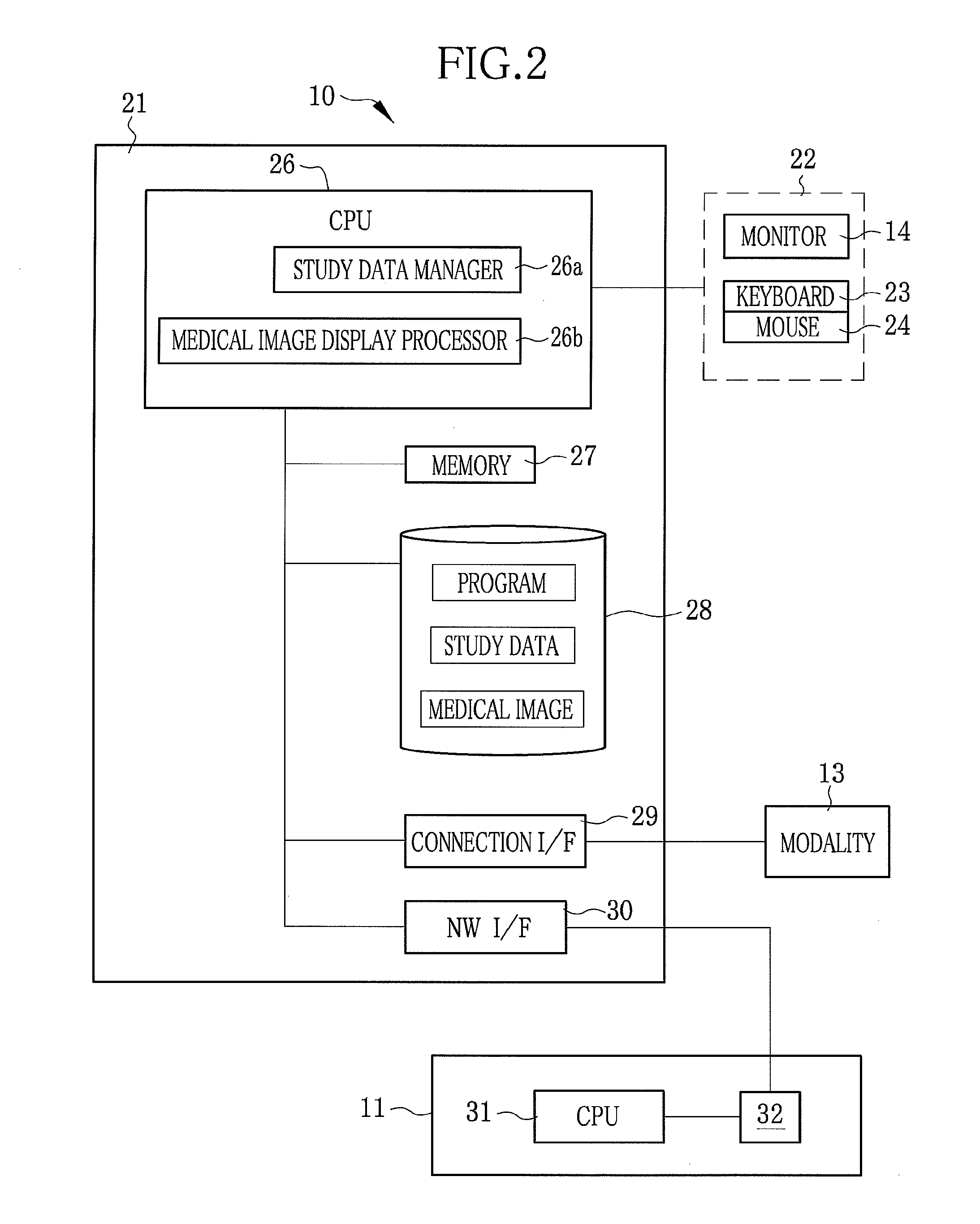
Medical study support apparatus and study list display method
InactiveUS20080241806A1Decrease productivityPrivacy protectionData processing applicationsDiagnostic recording/measuringData displayStudy Type
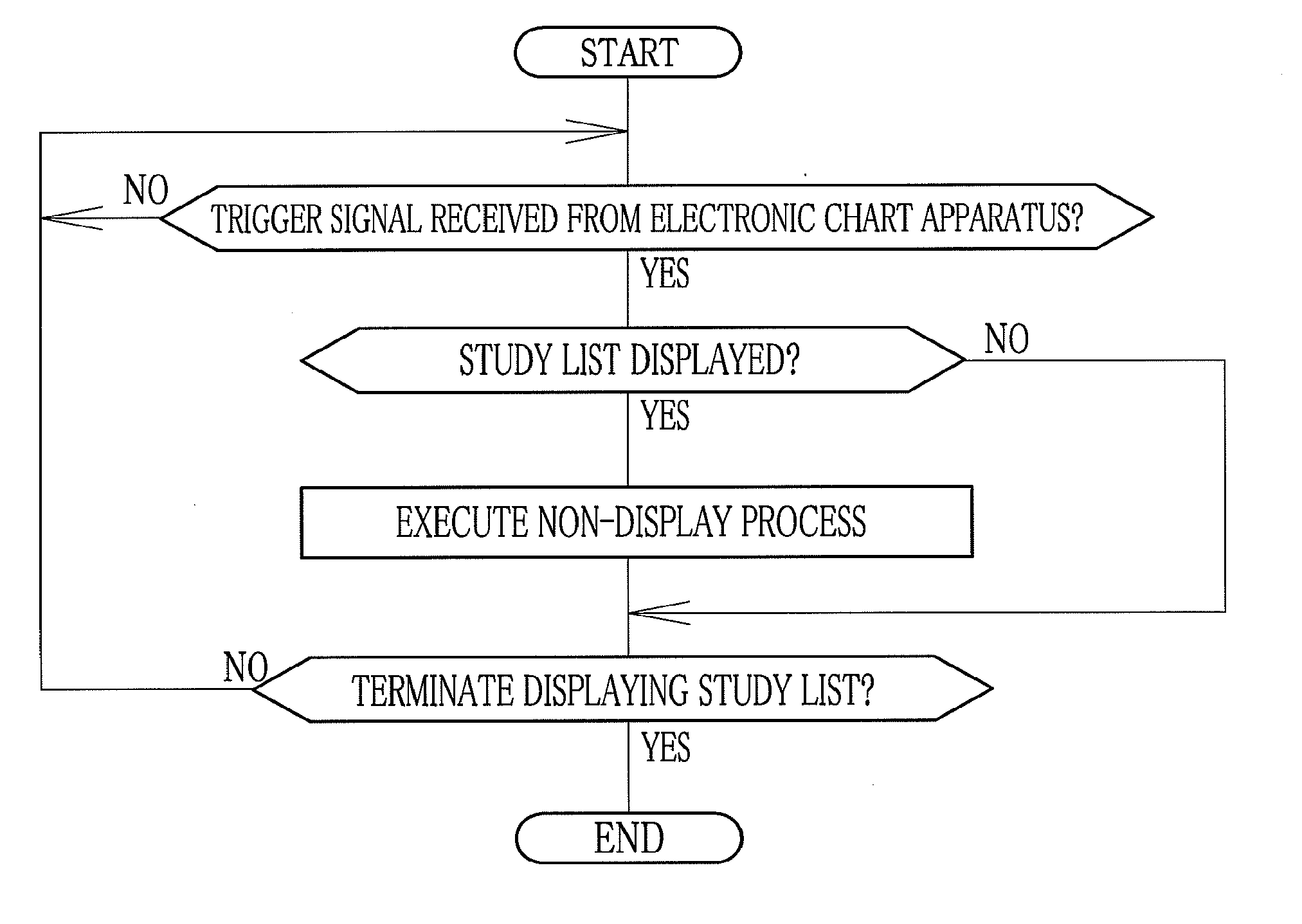
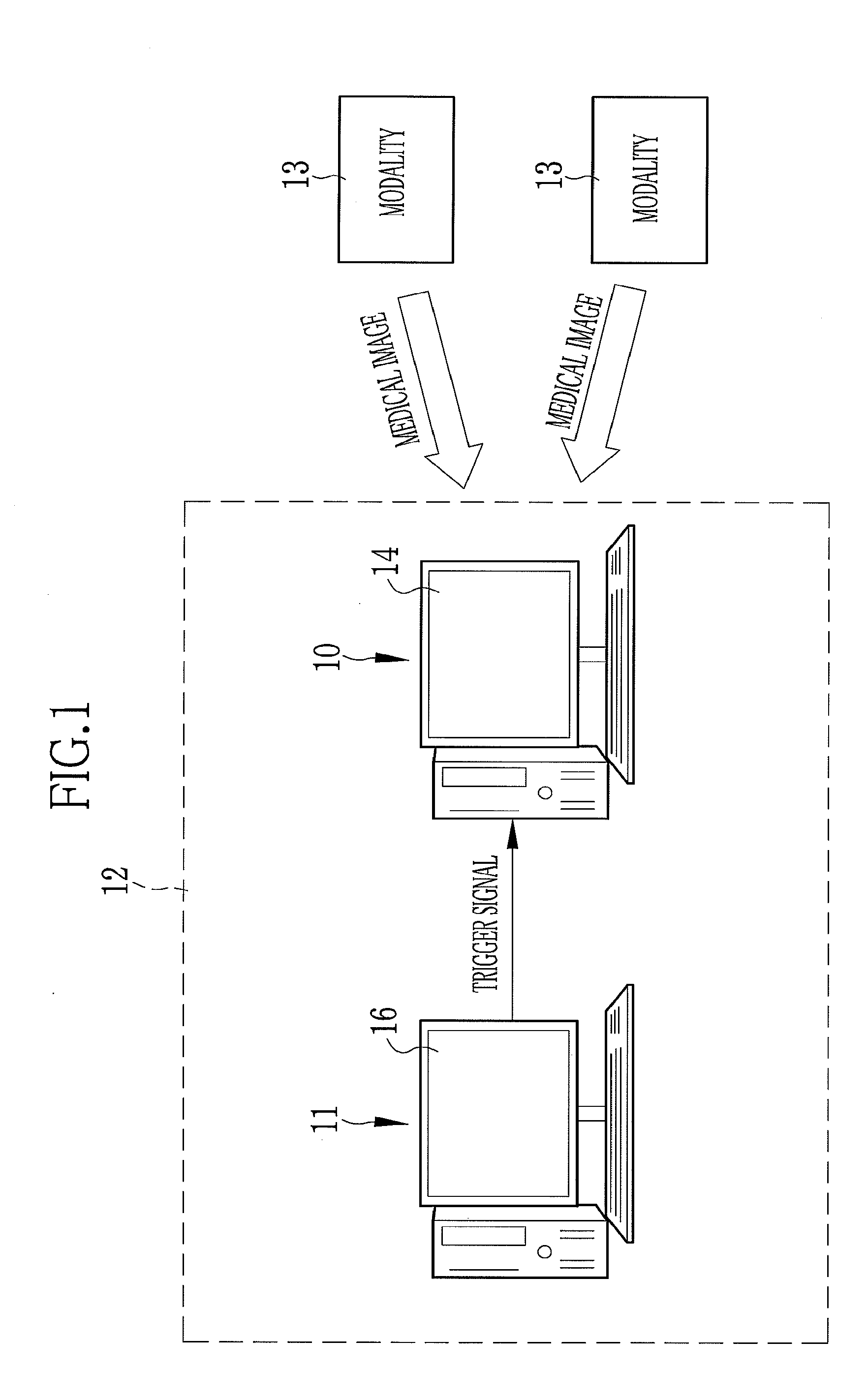
A medical study support apparatus manages study data, and displays a study list on a monitor screen. In the study list, data items on each study, including patient's personal information such as its name, birth date, sex and address as well as study items such as study ID, study date, study type and progress of study, are displayed in a lined for one case after another. The medical study support apparatus cooperates with an electronic chart apparatus. As chart data of a particular patient is displayed on its monitor, the electronic chart apparatus sends a trigger signal to the medical study support apparatus along with ID data of the particular patient. Upon receipt of the trigger signal and the ID data, the medical study support apparatus conceals the personal information on other patients than the particular patient in the study list.
Owner:FUJIFILM CORP
Medical information analysis systems and methods
InactiveUS20150324550A1Computer-assisted medical data acquisitionDrug referencesRegulatory authorityGood laboratory practice
A method for evaluating drug study information to determine suitability of the drug study information for acceptance by a regulatory authority. The method includes receiving a drug study identification and study type, indicating whether the drug study is either (i) a nonclinical study that is intended to be a Good Laboratory Practice study, or (ii) a clinical study that is intended to support the efficacy indication of a drug that is the subject of the drug study information, and associating one or more checklists with the drug study. Each checklist has elements identifying one or more features to be identified in the drug study information, and a query to select a deficiency type from a predetermined list of deficiency types. The method receives the deficiency types and automatically assigns a criticality value to each based on the nature of the type of drug study.
Owner:DRUG DEV ANALYTICS
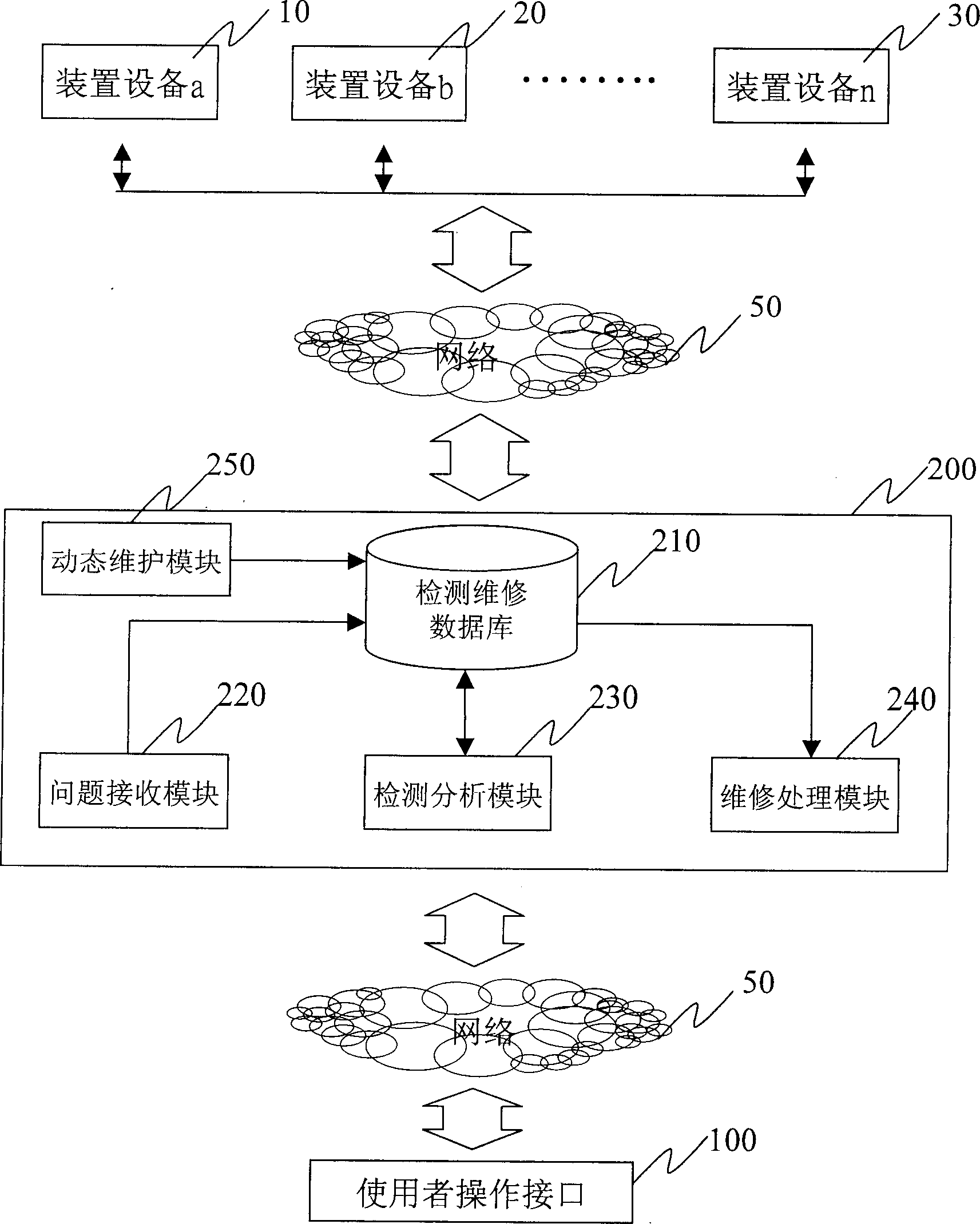
Studying-type detecting and fixing system and method
InactiveCN1510573ASimplified inspection and maintenance workError detection/correctionStudy TypeSystem study
In the present invention, maintenance and detection information of various device and equipment is received through network having been connected with, the device and equipment connected with the system can be detected and maintained on real time and at the same time, dynamic maintenance on line can be carried on in the cource to make system study continuously and keep on refreshing internal data of databank so that analysis information content in high reference value can be worked out to let detection and maintenance work optimize continuously.
Owner:INVENTEC CORP
Tire state monitoring method based on study-type multiple perception and device
ActiveCN107571690AReal-time monitoringDynamic MonitoringTyre measurementsSignalling/lighting devicesLearning basedStudy Type
The invention discloses a tire state monitoring method based on study-type multiple perception and a device. The tire state monitoring method comprises the steps that the minimum mileage of one or a plurality of automobiles serves as an analysis unit, data are collected from an automobile bus, ABS pulse data of coaxial tires are modified and accumulated correspondingly according to correction value obtained under a study mode; if a sum of the ABS pulse data of diagonal tires is abnormal in the process, completeness of the monitoring cycle is ensured by replacing or filling; the ABS pulse independent accumulation data of the four tires obtained in the study mode after the correction values are corrected serve as a comparing object, and the real-time tire pressure values of the four tires serve as standard, and the accurate tire pressure values of the tires of the next cycle is calculated by looking-up a table; and pre-warning and alarming quantitative processing of the tire health stateare conducted, and data outputting, state displaying and sound-light alarming processing are conducted. The invention further provides a tire monitoring device. The tire state monitoring method can monitor multi-information of the tires, and a user can obtain comprehensive tire state information.
Owner:中睿宏智汽车技术(深圳)有限公司
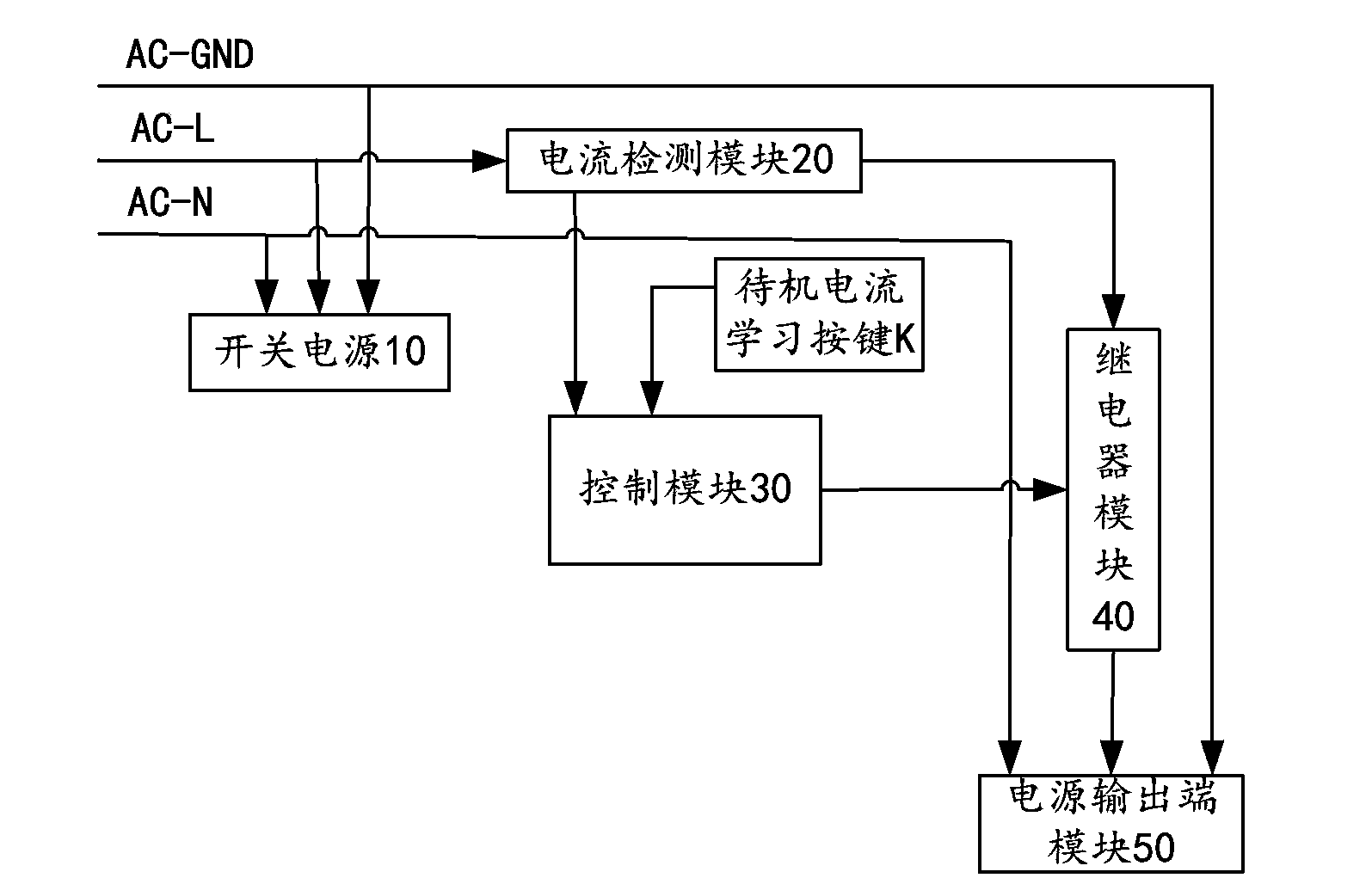
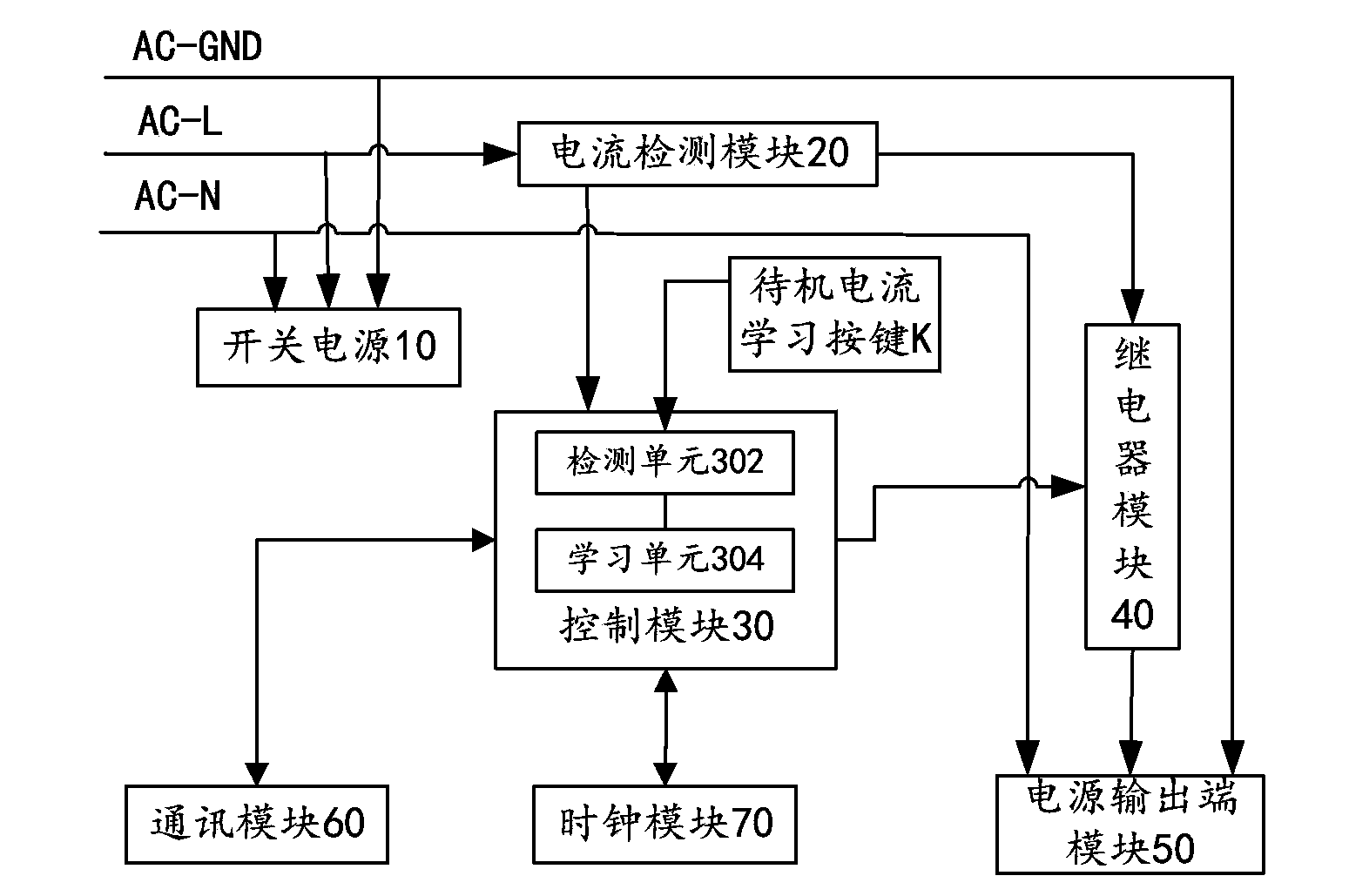
Study-type intelligent power supply management system and management method thereof
ActiveCN103699019ASolve the disadvantage that standby control can only target the same deviceEasy to controlProgramme control in sequence/logic controllersSupply managementStudy Type
The invention provides a study-type intelligent power supply management system and a management method thereof. The study-type intelligent power supply management system comprises a switching power supply, a current detection module, a control module, a relay module, a power supply output module and a standby current study button, wherein the switching power supply is used for providing voltage and current to the study-type intelligent power supply management system; the current detection module is used for detecting the magnitude of current passing through a conducting wire when electric equipment is in a standby state; the control module is respectively connected with the current detection module, the relay module and the standby current study button, and is used for reading the current data of the current detection module and controlling the on-off of the relay module according to the state of the electric equipment; the relay module is used for controlling the on-off of the power supply output module. The invention further discloses the management method of the study-type intelligent power supply management system. When the electric equipment is in the standby state for a long time or in a nonworking time, a power supply of the electric equipment is switched off, and the electric energy is saved to the greatest degree.
Owner:BEIJING ITSYNC TECH
Multifunctional desk with desktop heightened guard bar structure for reading
The invention discloses a multifunctional table equipped with a reading device by raising the table top with safety guards structure and various moderating assembled layout which can adjust and move the top movable table surface in longitudinal direction; the table can provides reading function on the book height, book apparent distance and angle corresponding with the ergonomics principle and scientific eye using state, and can solve the operating paradox and space paradox caused by several studying types proceeded at the same time, and also can induce and promote the user keep the optimum sitting posture of raising head and stiffing chest, and is convenient for users to change and moderate benefit setting posture, which can solve the shortcomings of the current table which force users incline their body and damage the eyesight and results in the stooping figure of hump back and fatigue, backache, and rachiopathy, and other pains and disease thoroughly and is favorable to increase the interest and effectiveness of the user and protect health.
Owner:洪佳恒
Studying and up grading system and method for enhanced
ActiveCN100389441CEasy to identifyImprove conversion performanceElectric signal transmission systemsNon-electrical signal transmission systemsStudy TypeGrade system
Owner:HANGZHOU BREMAX TECH +1
Multifunctional experiment table for chemistry teaching
The invention relates to a multifunctional experiment table for chemistry teaching, which comprises a table top and supporting legs, wherein a cylindrical experiment functional island is penetrated and fixed in the table top, a water tank is arranged on the table top at one side of the cylindrical experiment functional island, and the cylindrical experiment functional island is equipped with special experimental equipment satisfying the requirements for water, electricity, wind, gas, light and the like used for physical, chemical and biological experiments in middle schools. The invention overcomes the defect that an ordinary experiment table for teaching in the prior art only can satisfy the requirement of experimental operation in a single field and can not satisfy the requirement of discussing and studying type digital teaching; through the design, a computer can be placed on the table top, the special experimental equipment for physics, chemistry and biology can be intensively arranged in the cylindrical experiment functional island, the multifunctional experiment table has the advantages of simple structure and convenient assembly, the utilization ratio of a laboratory is also greatly improved, the investment of education cost is greatly lowered, and not only the experimental requirement of digital teaching is satisfied, but also a promoting action is performed for the universality of laboratory functions in the middle schools and the upgrading of laboratory functions in the future.
Owner:陈书
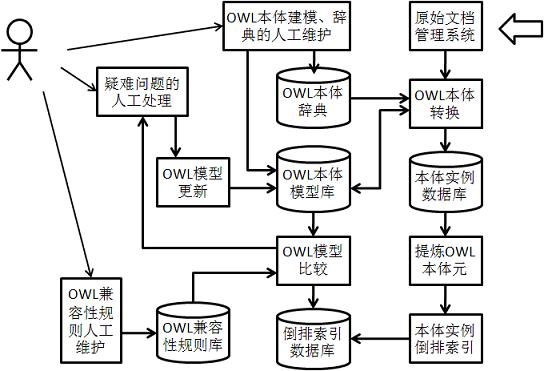
Mainstream media report hot-spot analyzing system based on studying type web ontology language (OWL) modeling
ActiveCN102436497AMake up for deficienciesSpecial data processing applicationsThe InternetStudy Type
The invention discloses a mainstream media report hot-spot analyzing system based on studying type web ontology language (OWL) modeling. The system is characterized by comprising the following functional modules: 1), an OWL ontology instance transforming module; 2), an invert index module; 3), an ontology element frequency counting module, which is used for performing the ontology element frequency counting on the OWL ontology invert index table and sequencing according to the size of the frequency; 4), an OWL seed model generating module; 5), a knowledge ontology model polymerizing module; 6), an OWL hot model counting module; 7), a news hot frequency counting module. For utilizing the method of the OWL ontology model analysis, the invention promotes the internet news hot-spot analysis to the concept hierarchy; some related hot-spot concepts are polymerized by using the relationship between the concepts to form the news hot, thereby overcoming the shortcoming due to partial hot-spot analyzing in traditional method.
Owner:江苏联著实业股份有限公司
Product service model generating system based on study type web ontology language (OWL) modeling
The invention discloses a product service model generating system based on study type web ontology language (OWL) modeling and is characterized by comprising a manual processing module, an inverted index library building module and an OWL model comparison module. The manual processing module builds an OWL knowledge module of product service information materials to serve as a seed module in a manual operation mode. The inverted index library building module collects product service information from the Internet and coverts the product service information into an OWL body example, and products serve as a body element of the OWL body example to build an inverted index data base of the products. The OWL module comparison module compares the seed module with the products in the inverted index data base, and supplements properties of the same product service materials to the seed module to improve the seed module. The product service model generating system combines manual intervention and automatic study so that the OWL modeling of the product service information materials is achieved, relevant information searched from the Internet is added automatically, and a solution idea with operability is provided for building a wide and complete product service material computer model.
Owner:江苏联著实业股份有限公司
Online interaction platform
InactiveCN108053355AInteractive needs resolutionMeet interaction needsBuying/selling/leasing transactionsOnline helpStudy Type
The invention relates to the technical field of interaction platforms, and discloses an online interaction platform. Through the online interaction platform provided by the invention, interaction of online help seeking and online aid can be carried out to realize the aim of doing good, interaction demand solution (comprising study type and non-study type interaction demand solution) and doing goodin life of people can be combined through a forum mode, and love values accumulated by doing good can be used for seeking interaction demand solution, so that people can obtain return while helping others (not doing good for nothin0, benefit is brought to call on people to do good, the aim of win-win in pecuniary benefit and charity of people is realized, and then the construction of harmonious society is furthermore promoted.
Owner:程鹏
A method and system for evaluating the reliability of toxicological data
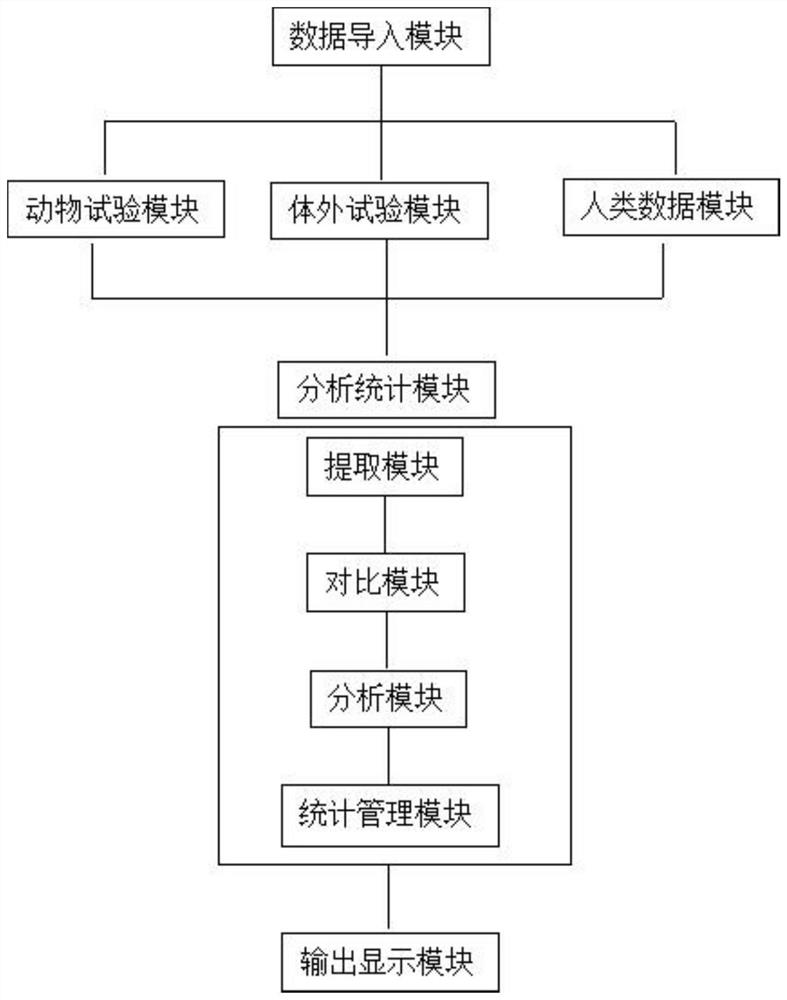
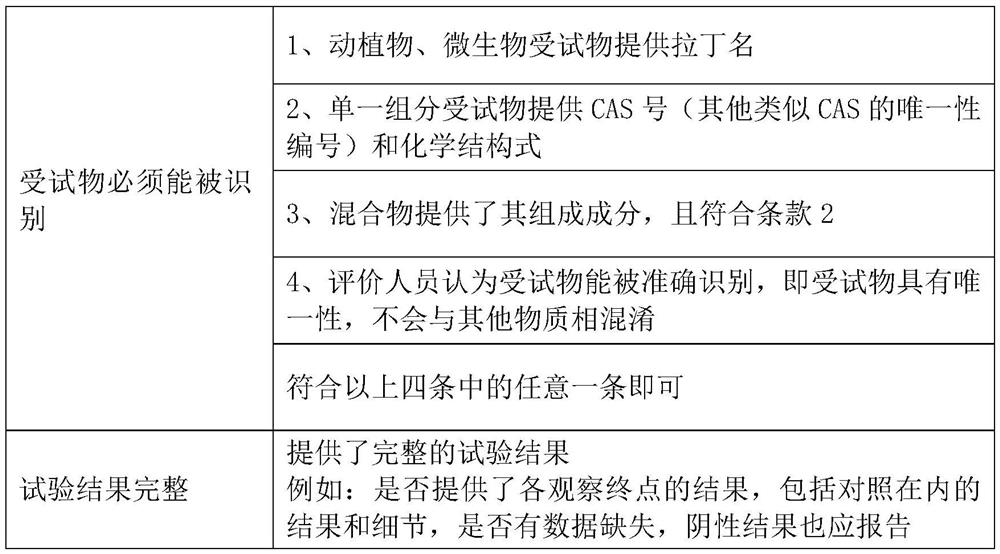
The invention discloses a method and system for evaluating the reliability of toxicological data. For the animal test data, the reliability evaluation is carried out from five parts: the test substance, experimental animals, test design, test operation and method, test results and conclusions. For in vitro test data, reliability evaluation is carried out from five parts: test substance, test system, test design, test operation and method, test result and conclusion. For human data, the reliability evaluation is carried out from five parts: research type and research object, measurement, quality control, bias control, results and conclusion. Aiming at the gap in the prior art, the invention designs a set of detailed and quantitative evaluation method and system for reliability evaluation of toxicological data.
Owner:JIANGSU PROVINCIAL CENT FOR DISEASE PREVENTION & CONTROL +1
Study type COD online analyzer
PendingCN107941732AImprove applicabilityImprove measurement accuracyChemoinformaticsTesting waterMicrocontrollerMicrocomputer system
The invention discloses a study type COD online analyzer which comprises an embedded microcomputer system, wherein the embedded microcomputer system is connected to a singlechip microcomputer system,the singlechip microcomputer system is successively connected to a spectrum scanning stepping motor, a measuring valve and a cleaning valve, the measuring valve and the cleaning valve communicate to acirculating measuring tank, the circulating measuring tank is connected to an automatic cleaning device, the cleaning valve communicates to a clean water inlet through a pipeline, the measuring valvecommunicates to a tested water sample inlet through a pipeline, and the singlechip microcomputer system is connected to a photovoltaic conversion circuit. The study type COD online analyzer which ishigh in precision, high in measuring speed and free of pollution adopts a theory of ultraviolet spectrophotometry, and meanwhile, adopts a potassium dichromate oxidization method to measure the COD content in a water body, so that the problem that a lot of chemical reagents are used in the measuring process can be solved, the deteriorating consequence on the environment caused by secondary pollution is alleviated, and the pollution discharge monitoring effectivenss and safety are improved.
Owner:WUXI DIANCHUANG TECH
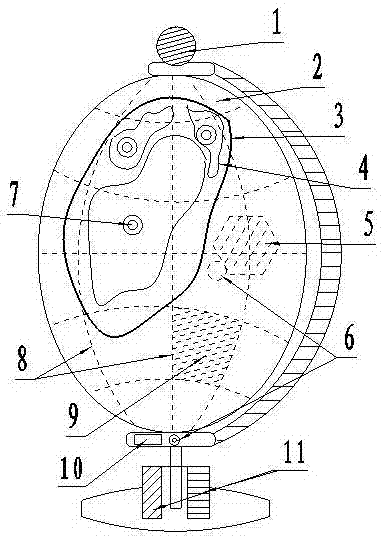
Self-electricity-generating study-type tellurion device
The invention relates to a self-electricity-generating study-type tellurion device. For the self-electricity-generating study-type tellurion device, a zinc-copper reaction electricity generating device is mounted on a base of a tellurion main body, and is connected with white-light LED lamps, a voice-control module single chip microcomputer provided with a storage battery, a red-light LED lamp, a yellow-light LED lamp and a trigger switch, wherein a microphone is mounted at the upper end of the tellurion main body, voice recording the names of the countries and the corresponding continents on the tellurion is stored in the voice-control module single chip microcomputer provided with the storage battery, if the trigger switch is touched, the red-light LED lamp and the yellow-light LED lamp simultaneously emit light, then the microphone can broadcast the name of the country of the corresponding continent in the area touched by a user, and therefore, the sound and light integrated fun study purpose is realized.
Owner:张晓光
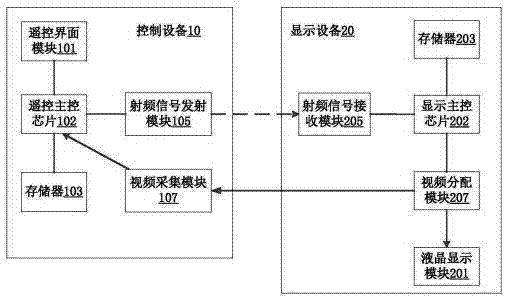
System for testing study-type remote control equipment
InactiveCN107481506ALow costEasy to produceNon-electrical signal transmission systemsSelective content distributionLiquid-crystal displayRemote control
A system for testing and learning remote control equipment, including a control device and a display device; the control device includes a radio frequency signal transmitting module, a remote control main control chip, a memory A, a remote control interface module, and a video acquisition module; the remote control main control chip is connected to the remote control interface Module, radio frequency signal transmitting module, memory A and video acquisition module; display device includes memory B, radio frequency signal receiving module, display main control chip, video distribution module, liquid crystal display module; display main control chip connects memory B, radio frequency signal receiving module and a video distribution module; the video distribution module is connected to the video acquisition module and the liquid crystal display module. The invention solves the problem of test automation of the test learning type remote control system, can realize the full braking test remote control system through programming control, and effectively saves manpower.
Owner:南京荣膺软件科技有限公司
Mainstream media report hot-spot analyzing system based on studying type web ontology language (OWL) modeling
ActiveCN102436497BMake up for deficienciesSpecial data processing applicationsLearning basedStudy Type
The invention discloses a mainstream media report hot-spot analyzing system based on studying type web ontology language (OWL) modeling. The system is characterized by comprising the following functional modules: 1), an OWL ontology instance transforming module; 2), an invert index module; 3), an ontology element frequency counting module, which is used for performing the ontology element frequency counting on the OWL ontology invert index table and sequencing according to the size of the frequency; 4), an OWL seed model generating module; 5), a knowledge ontology model polymerizing module; 6), an OWL hot model counting module; 7), a news hot frequency counting module. For utilizing the method of the OWL ontology model analysis, the invention promotes the internet news hot-spot analysis to the concept hierarchy; some related hot-spot concepts are polymerized by using the relationship between the concepts to form the news hot, thereby overcoming the shortcoming due to partial hot-spot analyzing in traditional method.
Owner:江苏联著实业股份有限公司
A real-time optimization decision-making method for automatic combustion in waste incineration process
InactiveCN108224446BAvoid subjectivityShorten the timeIncinerator apparatusCombustion systemCombustion
Owner:BEIJING UNIV OF TECH
Method for rapidly enhancing personal capability
InactiveCN1971612AClear goal orientationStrong targetingData processing applicationsStudy TypeEngineering
Owner:黄智
Medical study support apparatus and study list display method
InactiveUS20110141147A1Decrease productivityPrivacy protectionData processing applicationsGeometric image transformationData displayStudy Type
A medical study support apparatus manages study data, and displays a study list on a monitor screen. In the study list, data items on each study, including patient's personal information such as its name, birth date, sex and address as well as study items such as study ID, study date, study type and progress of study, are displayed in a lined for one case after another. The medical study support apparatus cooperates with an electronic chart apparatus. As chart data of a particular patient is displayed on its monitor, the electronic chart apparatus sends a trigger signal to the medical study support apparatus along with ID data of the particular patient. Upon receipt of the trigger signal and the ID data, the medical study support apparatus conceals the personal information on other patients than the particular patient in the study list.
Owner:FUJIFILM CORP
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com