Ionic liquid based mixtures for gas storage and delivery
a technology of ionic liquid and gas storage, which is applied in the direction of liquid degasification, separation process, and other chemical processes, can solve the problems of toxic gas storage under high pressure in metal cylinders, gas safety and environmental challenges, and inability to meet the needs of storage and delivery systems, etc., and achieve the effect of reducing the solubility of unreacted gas
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Mixture Formed from Reaction of Lewis Acidic Ionic Liquid BMIM+Al2Cl7− and PH3
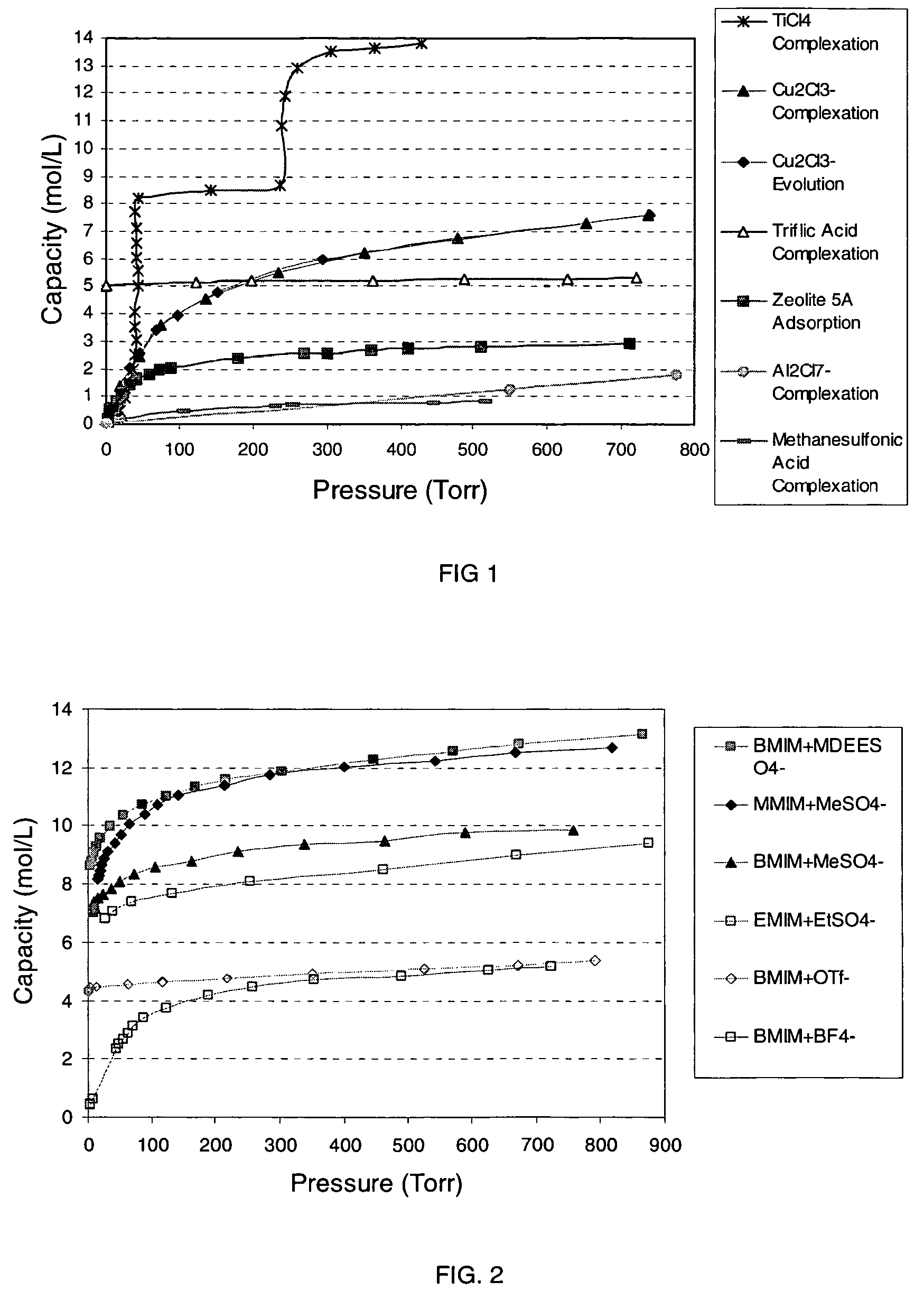
[0068]Molecular modeling was used to calculate a binding energy, ΔErxn, for this Lewis acidic ionic liquid with PH3. The ionic liquid was modeled as an ion-pair, using 1,3-dimethylimidazolium as the cation, Al2Cl7− as the anion, and it was assumed that one equivalent of PH3 reacted per equivalent of Al2Cl7− anion (concentration of Al2Cl7−=3.2 mol / L). Structures were determined based on minimum energy geometry optimization using Density Functional Theory (DFT) at the BP level with a double numerical (DN**) basis set. This Lewis acidic ionic liquid was calculated to have a ΔErxn of 0.71 kcal / mol, which suggests that the reaction is slightly unfavorable, although within the general limitations of error. To clarify the results of modeling, the following reaction was run.
[0069]In a glove box, 9.07 g of AlCl3 (2 equivalents) was slowly added to 5.94 g (1 equivalent) of 1-butyl-3-methylimidazolium chloride (BMIM...
example 2
Mixture Formed from Reaction of Lewis Acidic Ionic Liquid BMIM+CuCl2− and PH3
[0072]In a glove box, 3.10 g of CuCl was slowly added to a flask charged with 5.46 g of BMIM+Cl− (1:1 stoichiometry) (assuming the anion CuCl2− is formed from the reaction stoichiometry 1 equivalent CuCl to 1 equivalent BMIM+Cl−). The mixture was stirred overnight and stored. A glass insert was charged with 7.71 g of the ionic liquid (density=1.4 g / mL) and placed into a 50 mL reactor, and the general procedure for measuring PH3 reaction was followed. The Lewis acidic ionic liquid reacted with 7.6 mmol of PH3 at room temperature and 674 Torr, corresponding to 1.4 mol PH3 / L of ionic liquid. Equilibrium data points were not obtained and % reversibility and working capacity were not determined.
example 3
Mixture Formed from Reaction of Lewis Acidic Ionic Liquid BMIM+Cu2Cl3− and PH3
[0073]Molecular modeling was used to approximate the effectiveness of BMIM+Cu2Cl3− as a reactive liquid. The ionic liquid was modeled as an ion-pair, using 1,3-dimethylimidazolium as the cation, Cu2Cl3− as the anion, and it was assumed that one equivalent of PH3 reacted with each equivalent of copper (concentration of Cu reactive groups=9.7 mol / L). Structures were determined based on minimum energy geometry optimization using Density Functional Theory (DFT) at the BP level with a double numerical (DN**) basis set. This Lewis acidic ionic liquid was calculated to have an average ΔErxn of −5.5 kcal / mol for its reaction with PH3. The results indicate that this ionic liquid should bind PH3 more strongly than BMIM+Al2Cl7− of Example 1. Since ΔGrxn is smaller in magnitude than ΔErxn and the optimum ΔGrxn for the pressure range 20 to 760 Torr at room temperature is ca. −3 kcal / mol, the result suggests that the ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Gibb's free energy | aaaaa | aaaaa |
| decomposition temperature | aaaaa | aaaaa |
| decomposition temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com