Catheter injectable depot compositions and uses thereof
a technology of catheter and depot composition, which is applied in the field of depot composition, can solve the problems of reluctance of patients to accept such an implant or drug delivery system, important limitations of their use in the body of various animals, and the inability of materials to meet the demand for biodegradable implants, etc., to achieve the effect of improving shear thinning behavior, reducing injection force, and increasing composition homogeneity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0187]A gel vehicle for use in a catheter injectable depot of the composition was prepared as follows. A glass vessel was tared on a Mettler PJ3000 top loader balance. Poly(D,L-lactide-co-glycolide) (PLGA), available as 50:50 Resomer® RG502 (PLGA RG 502), was weighed into the glass vessel. The glass vessel containing PLGA was tared and the corresponding solvent was added. Amounts expressed as percentages for various polymer / solvent combinations are set forth in Table 1, below. The polymer / solvent mixture was manually stirred with a stainless steel square-tip spatula, resulting in a sticky amber paste-like substance containing white polymer particles. The vessel containing the polymer / solvent mixture was sealed and placed in a temperature controlled incubator equilibrated to 39° C. The polymer / solvent mixture was removed from the incubator when it appeared to be a clear amber homogeneous gel. Incubation time intervals ranged from 1 to 4 days, depending on solvent and polymer type and...
example 2
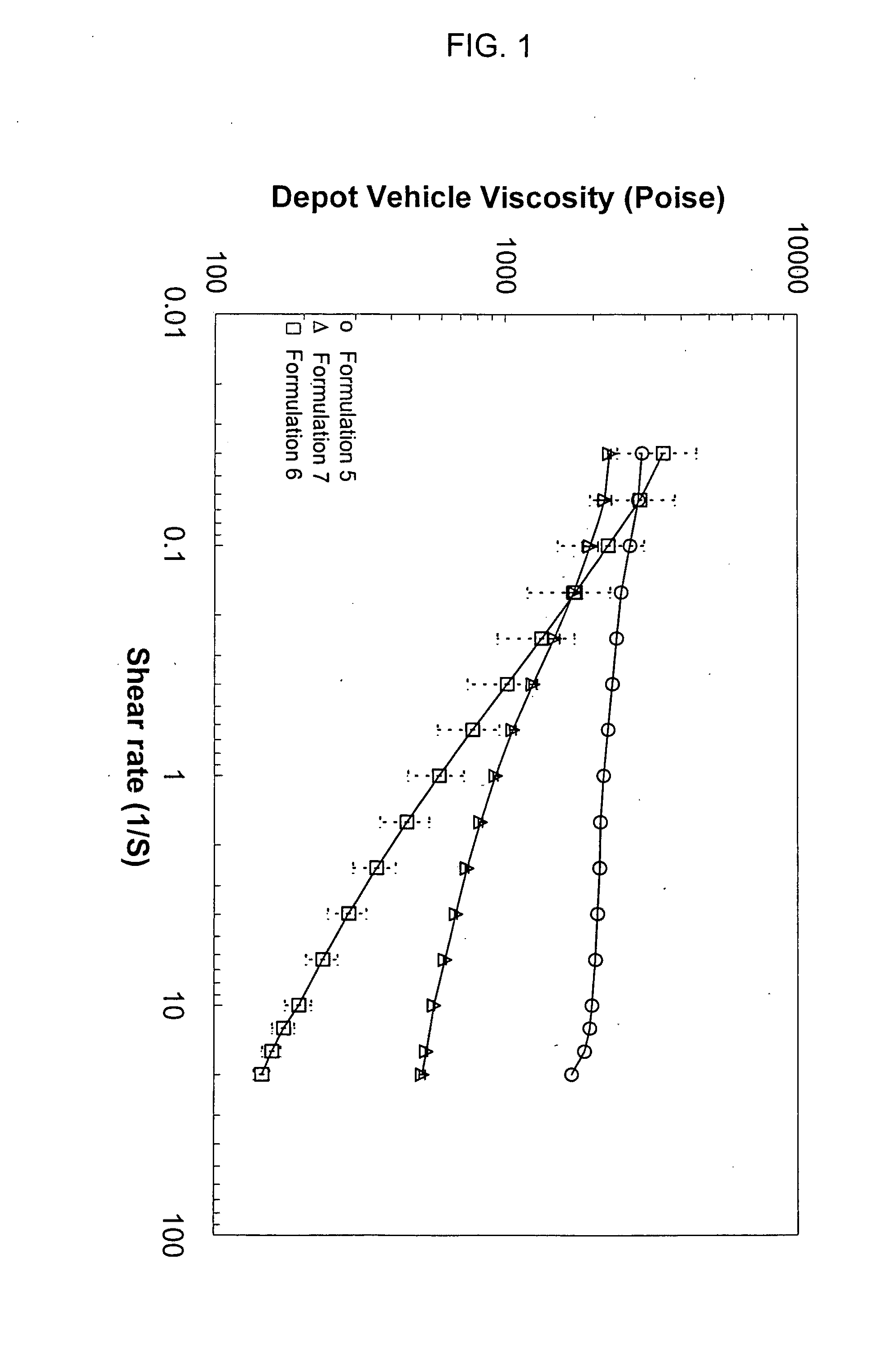
[0189]Rheological behavior was tested for depot vehicles formulated with different solvents. A vehicle comprising 50 wt. % polymer (PLGA RG502) and 50 wt. % solvent (benzyl alcohol) was prepared according to the procedures outlined in Example 1. For comparative purposes, solvent comprising benzyl benzoate (e.g., formulation 5) or benzyl benzoate combined with ethanol (e.g., formulation 7) were also prepared. Table 2 lists the formulations used in the test.
TABLE 2BenzylBenzylPolymerBenzoateAlcoholEthanolFormulation(%)(%)(%)(%)550.050.00.00.0650.00.050.00.0745.052.80.02.2
[0190]Formulations 5, 6 and 7 were tested for viscosity under various shear rates. As indicated in FIG. 1, significant shear thinning behavior was observed when benzyl alcohol was used as the solvent (e.g., formulation 6), in contrast to formulations using benzyl benzoate (e.g., formulation 5) and benzyl benzoate with ethanol as a thixotropic agent (e.g., formulation 7), respectively.
example 3
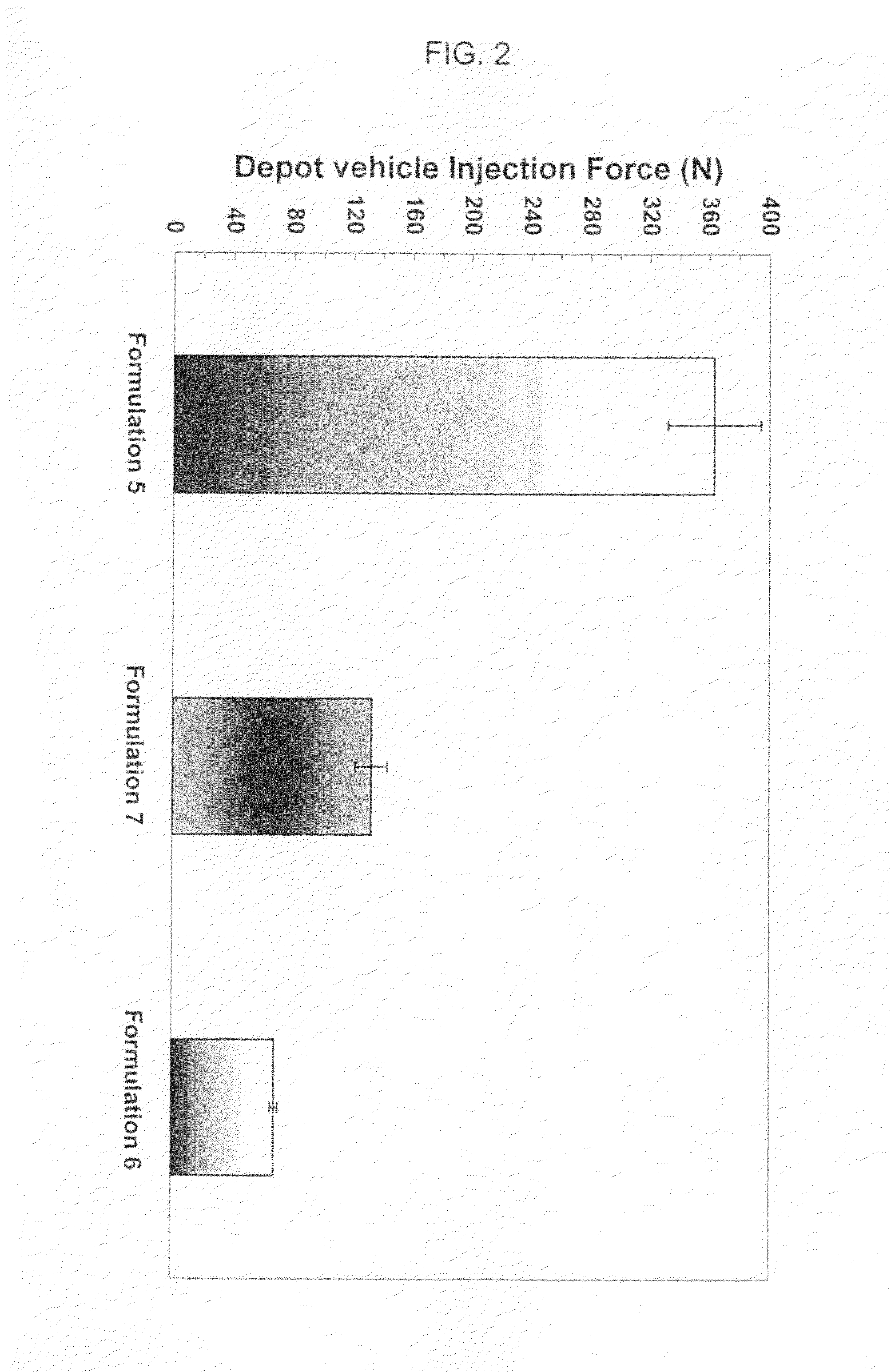
[0191]The injection force required to dispense depot vehicles was evaluated for the three formulations identified in Example 2. The formulations were injected through a 24-gauge needle at 1 ml / minute, at room temperature. As indicated in FIG. 2, significantly reduced injection force was observed when benzyl alcohol is used as the solvent (e.g., formulation 6), in contrast to formulations using benzyl benzoate (e.g., formulation 5) and benzyl benzoate with ethanol as a thixotropic agent (e.g., formulation 7), respectively. Notably, due to the shear thing behavior, the formulations using benzyl alcohol as the solvent (e.g., formulation 6), and benzyl benzoate with ethanol as a thixotropic agent (e.g., formulation 7) showed significantly reduced injection force while maintaining viscosities equal to or greater than the formulations using benzyl benzoate (e.g., formulation 5), at lower shear rate; thus maintaining the intactness of the depot after injection into the animals.
PUM
| Property | Measurement | Unit |
|---|---|---|
| miscibility | aaaaa | aaaaa |
| particle size | aaaaa | aaaaa |
| wt. % | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com