Method of treatment of dopamine-related dysfunction
a dopamine receptor and dopamine-related technology, applied in the direction of aerosol delivery, drug composition, inorganic non-active ingredients, etc., can solve the problems of insufficient dopaminergic activity of parkinson's disease, eventual loss of efficacy, dyskinesia, etc., to prevent the induction of tolerance and high affinity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Synthesis of 8,9-dihydroxy-1,2,3,11b-tetrahydrochromeno[4,3,2-de]isoquinoline Hydrobromide (Dinoxyline)
[0057] With reference to the following described experimental procedures, melting points were determined with a Thomas-Hoover melting point apparatus and are uncorrected. 1H NMR spectra were recorded with a Varian VXR 500S (500 MHZ) NMR instrument and chemical shifts were reported in values (ppm) relative to TMS. The IR spectra were recorded as KBr pellets or as a liquid film with a Perkin Elmer 1600 series FTIR spectrometer. Chemical ionization mass spectra (CIMS) were recorded on a Finnigan 4000 quadruple mass spectrometer. High resolution CI spectra were recorded using a Kratos MS50 spectrometer. Elemental analysis data were obtained from the microanalytical laboratory of Purdue University, West Lafayette, Ind. THF was distilled from benzophenone-sodium under nitrogen immediately before use and 1,2-Dichloroethane was distilled from phosphorous pentoxide before use.
1,2-Dimethox...
example 2
Additional Variations of the Subject D1 Agonists
1. Hexahydrobenzo[α]phenanthridines
[0070] Additional variations of hexahydrobenzo[α]phenanthridines are set forth with reference to Formula II and are synthesized as described in U.S. Pat. No. 5,420,134 incorporated herein by reference.
2. Dinapsoline
[0071] Additional variations of dinapsoline are shown in Table 1 as Compounds 1-47. The compounds in Table 1 are set forth with reference to Formula III and are synthesized as described in U.S. Pat. No. 5,959,110 incorporated herein by reference.
TABLE 1Cmpd.NumberRR1R2R3R4R5X1HHCH3HHHOH2HHHCH3HHOH3HHHHCH3HOH4HHC6H5HHHOH5CH3HCH3HHHOH6C3H7HHCH3HHOH7HHC2H5HHHOH8HHHC2H5HHOH9HHHCH3CH3HBr10C3H7HCH3CH3HHOH11C2H5HHCH3CH3HBr12CH3HHHC2H5HOH13C4H9HHOHHHOH14HHCH3OHHHOH15HHHFHHOH16HHOHHHHBr17HHBrHHHOH18HCH3HBrHHOCH319HCH3HHBrHOCH320HCH3CH3BrHHOCH321CH3HFHHHOH22CH3HHFHHOH23CH3HHHFHOH24C2H5HHOHHHF25C2H5HCH3OHHHF26C2H5HCH3OHCH3HF27C3H7HHCH3OHHCl28C3H7HHCH3CH3OHCl29C3H7HC2H5OHHHOH30C3H7HHHOHHOH31C4H...
example 3
Unilateral 6-OHDA Lesion Model for Parkinson's Disease
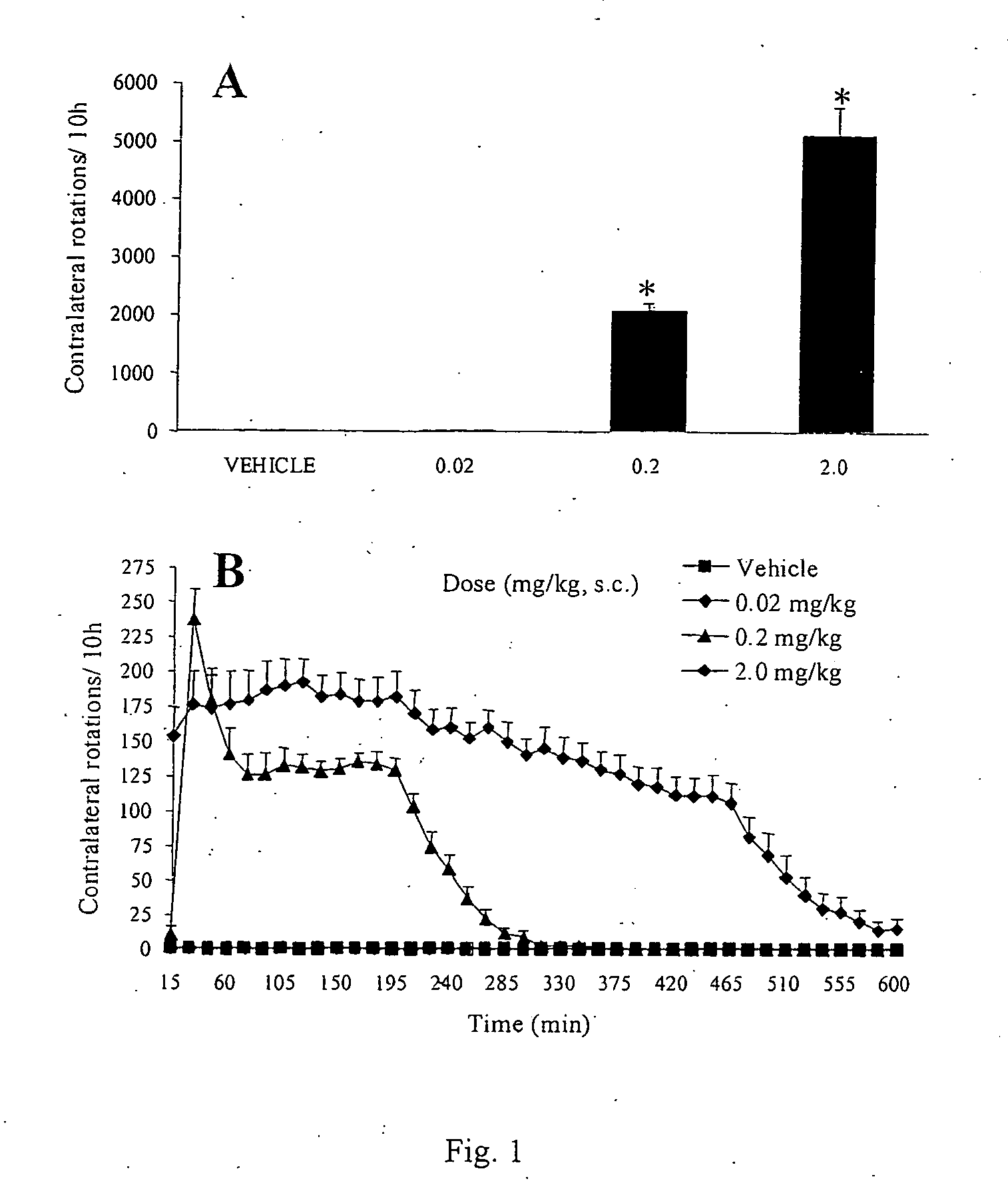
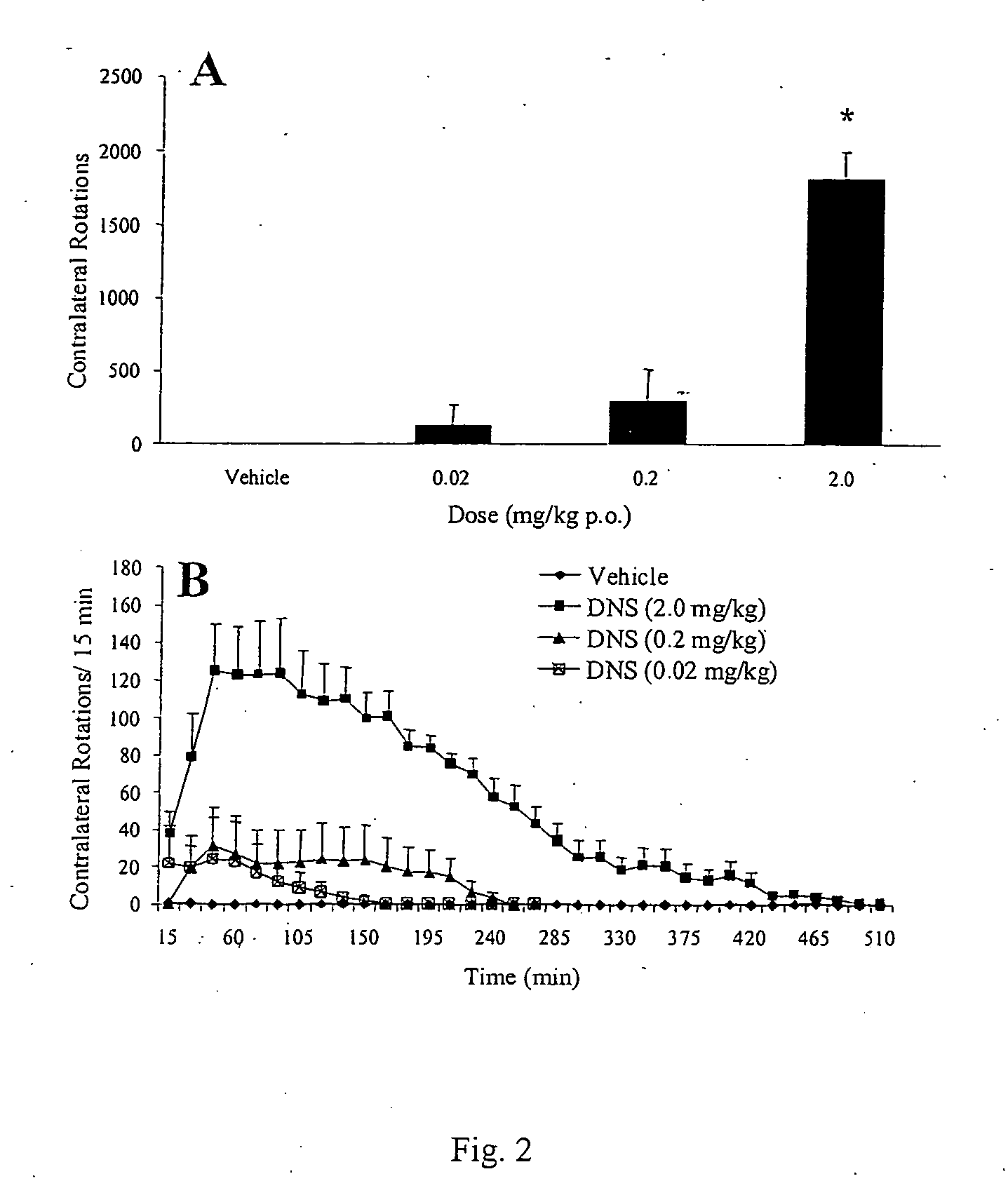
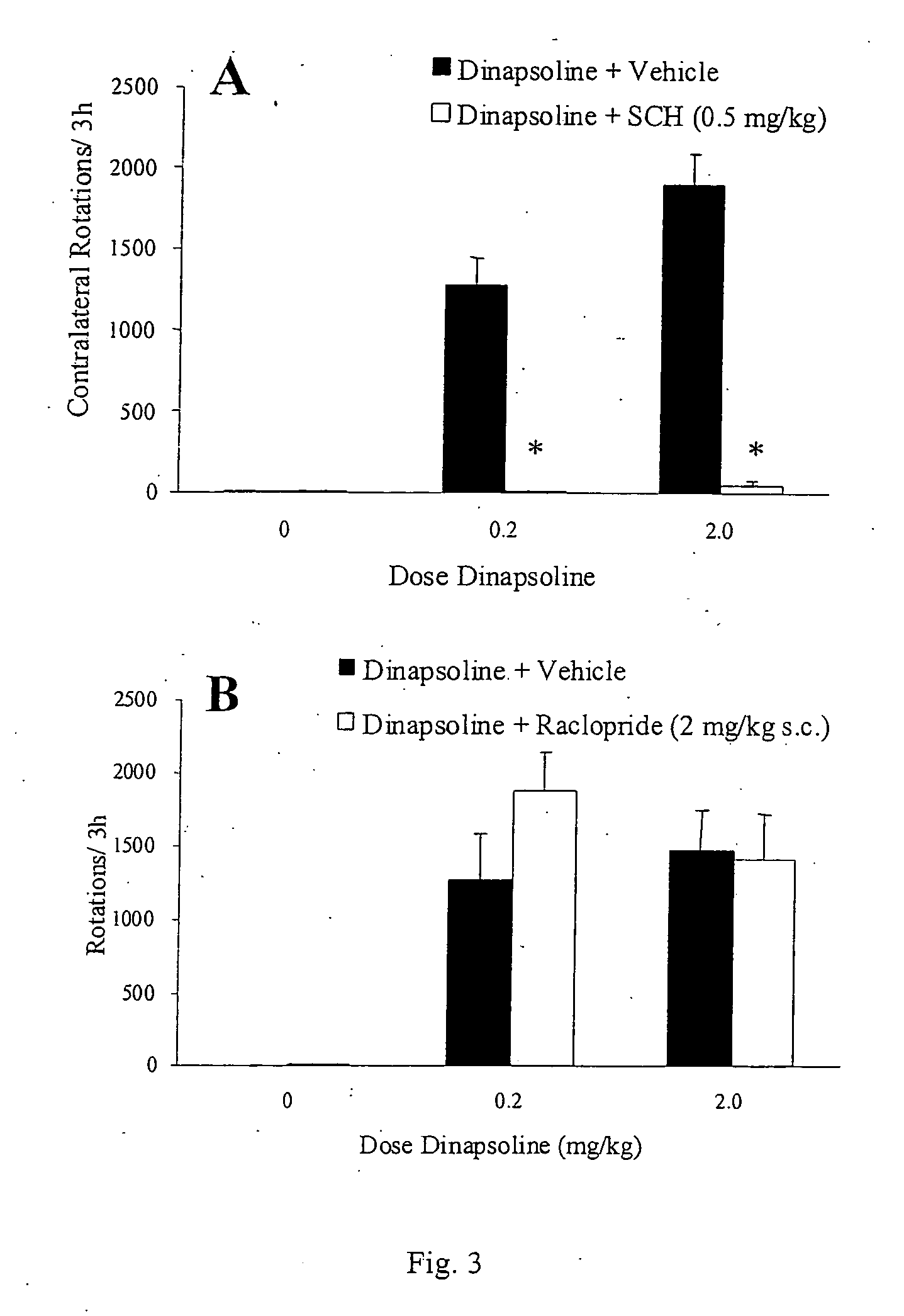
[0074] Summary. In the rat unilateral 6-hydroxydopamine (6-OHDA) rotation model of Parkinson's disease, 6-OHDA is infused unilaterally into the medial forebrain bundle, the substantia nigra, or the striatum. This treatment results in the destruction of dopamine terminals and neurons and a loss of striatal dopamine, and a profound functional dopaminergic supersensitivity develops on the lesioned side. When challenged with direct-acting dopamine receptor agonists, unilateral 6-OHDA rats turn contralaterally (away from the side of the lesion) because of the increased sensitivity of the postsynaptic dopamine receptors on the lesioned side. The experiments described below examine tolerance induced by the fall D1 agonist, dinapsoline, using the 6-OHDA model.
[0075] Subjects. Adult male Sprague-Dawley Rats (Hilltop Laboratories, Chatsworth, Calif.), weighing between 280 and 320 grams, were used as subjects. Animals were housed individu...
PUM
| Property | Measurement | Unit |
|---|---|---|
| time | aaaaa | aaaaa |
| time | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com