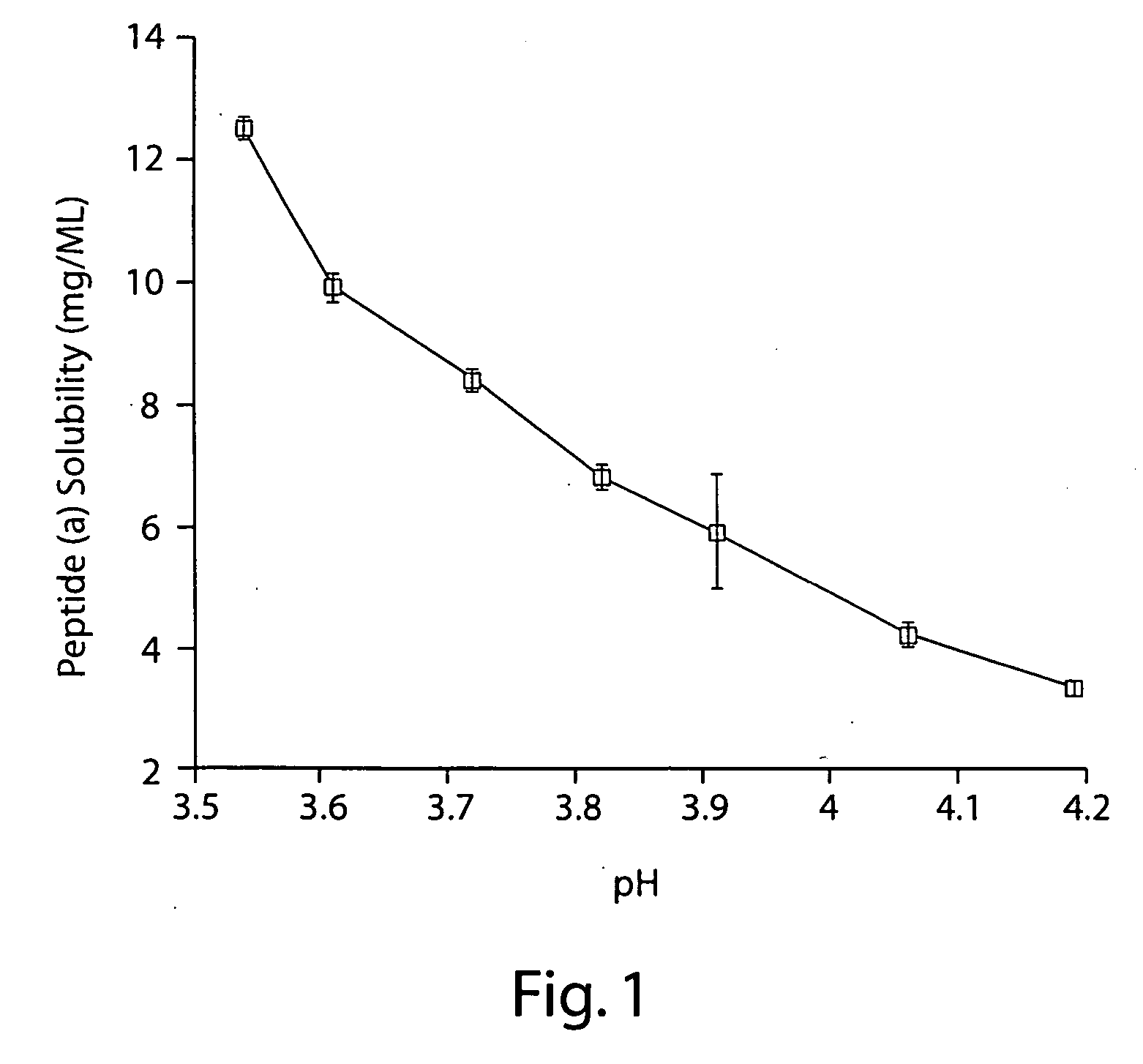
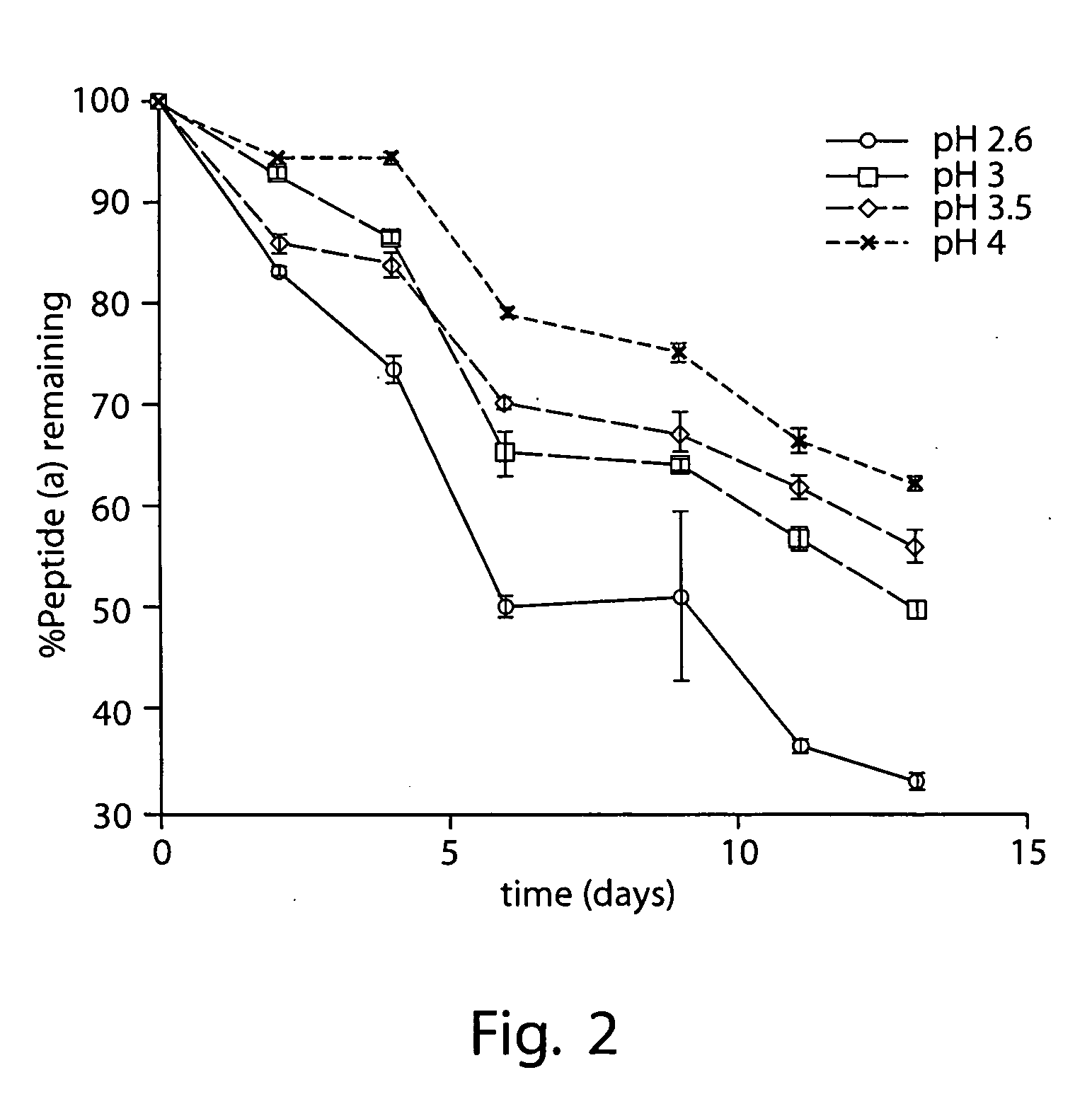
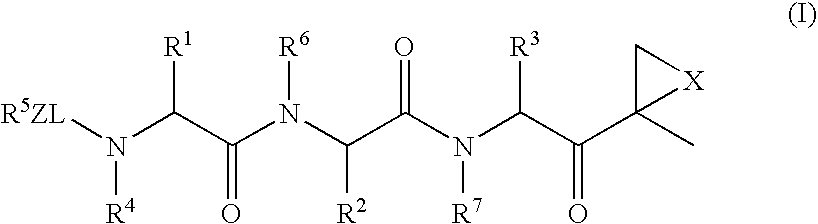
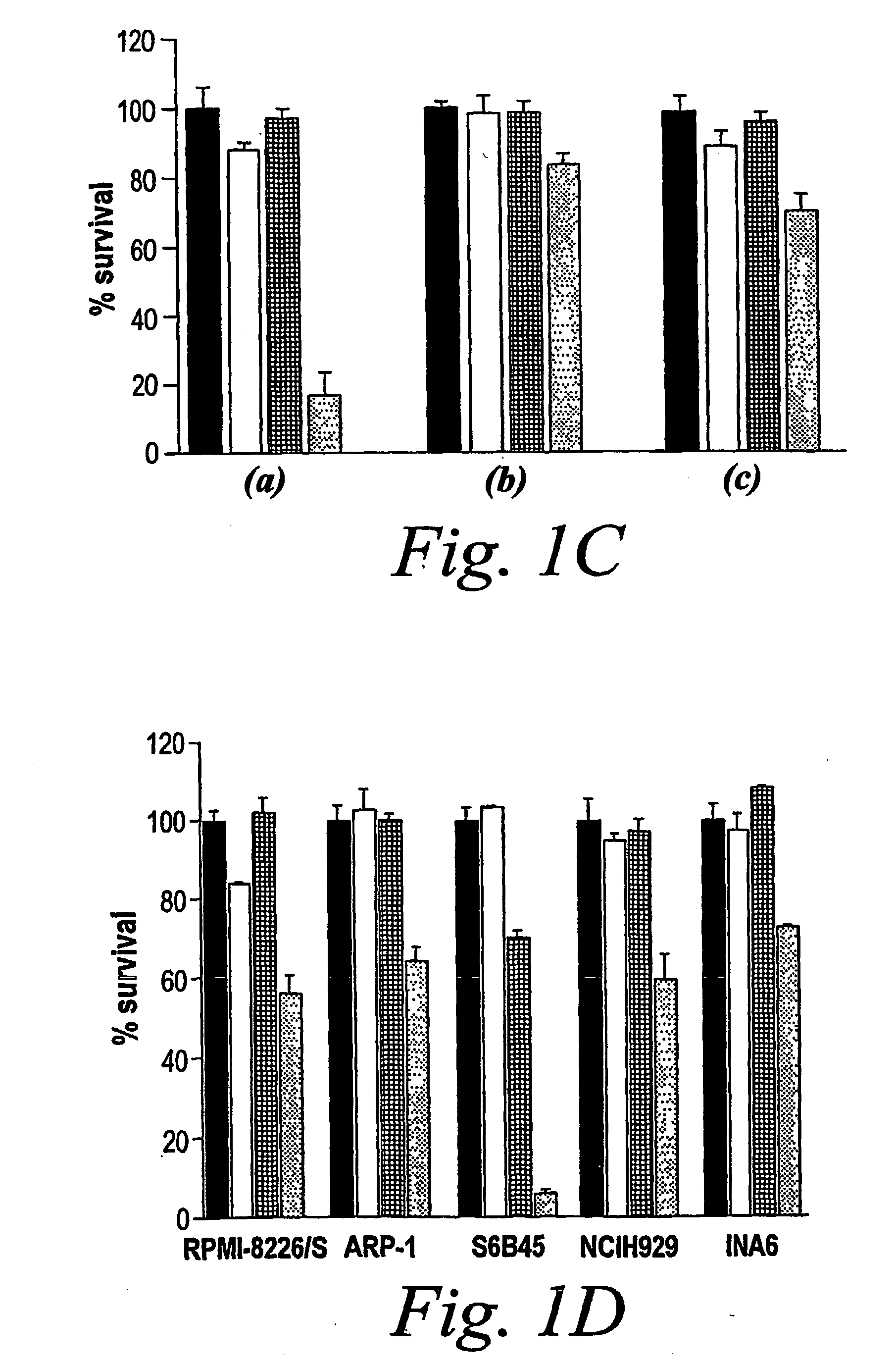
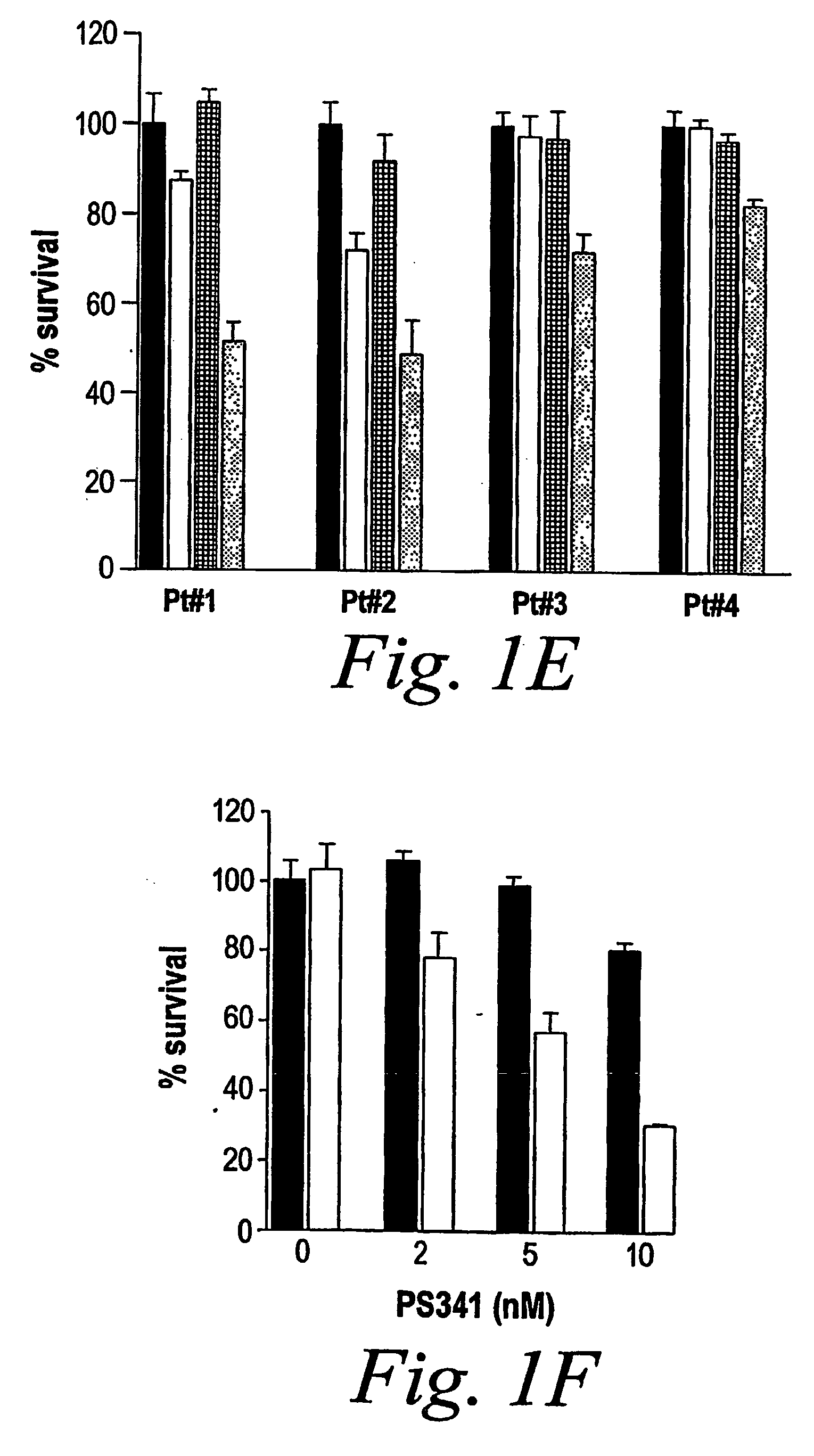
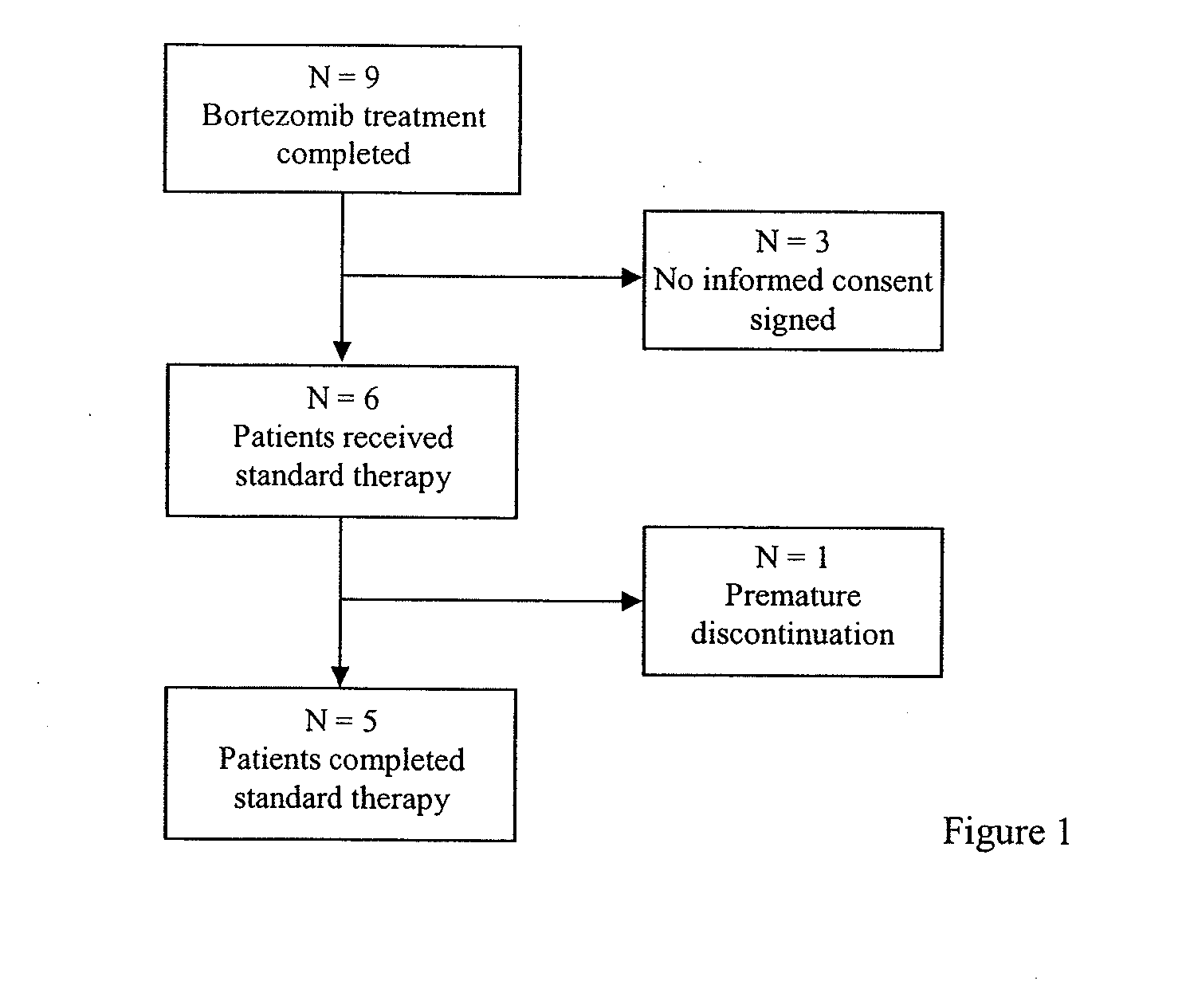
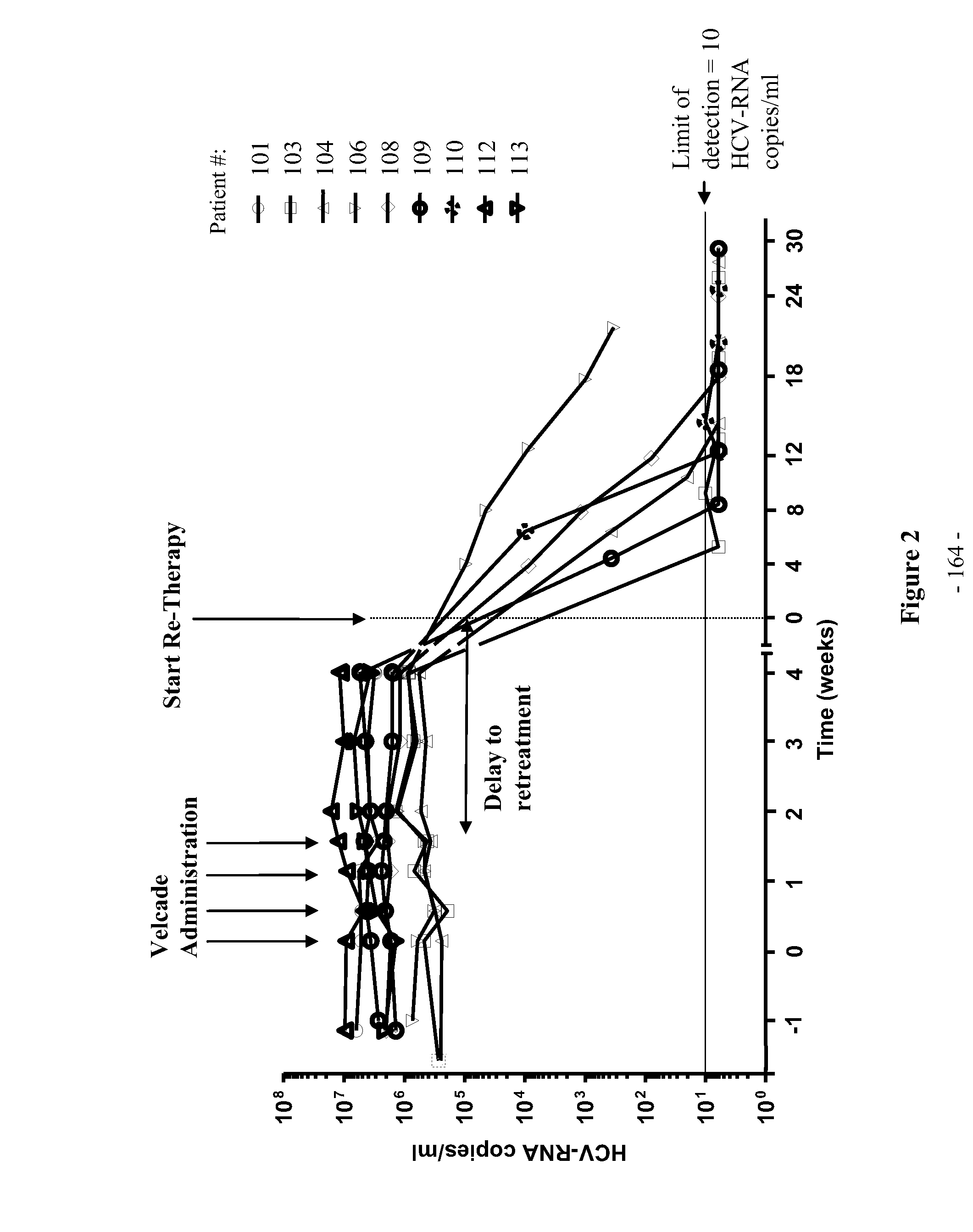
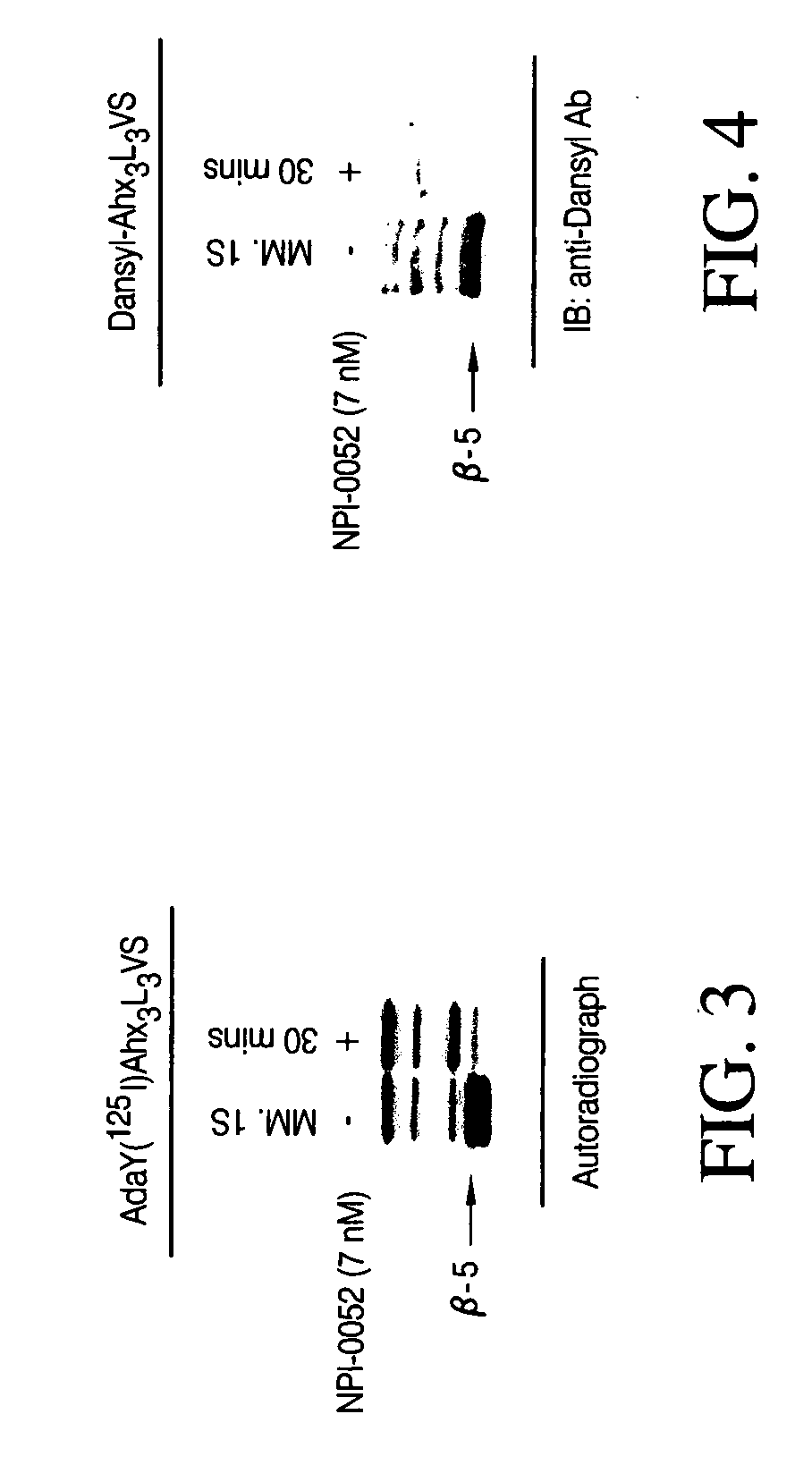
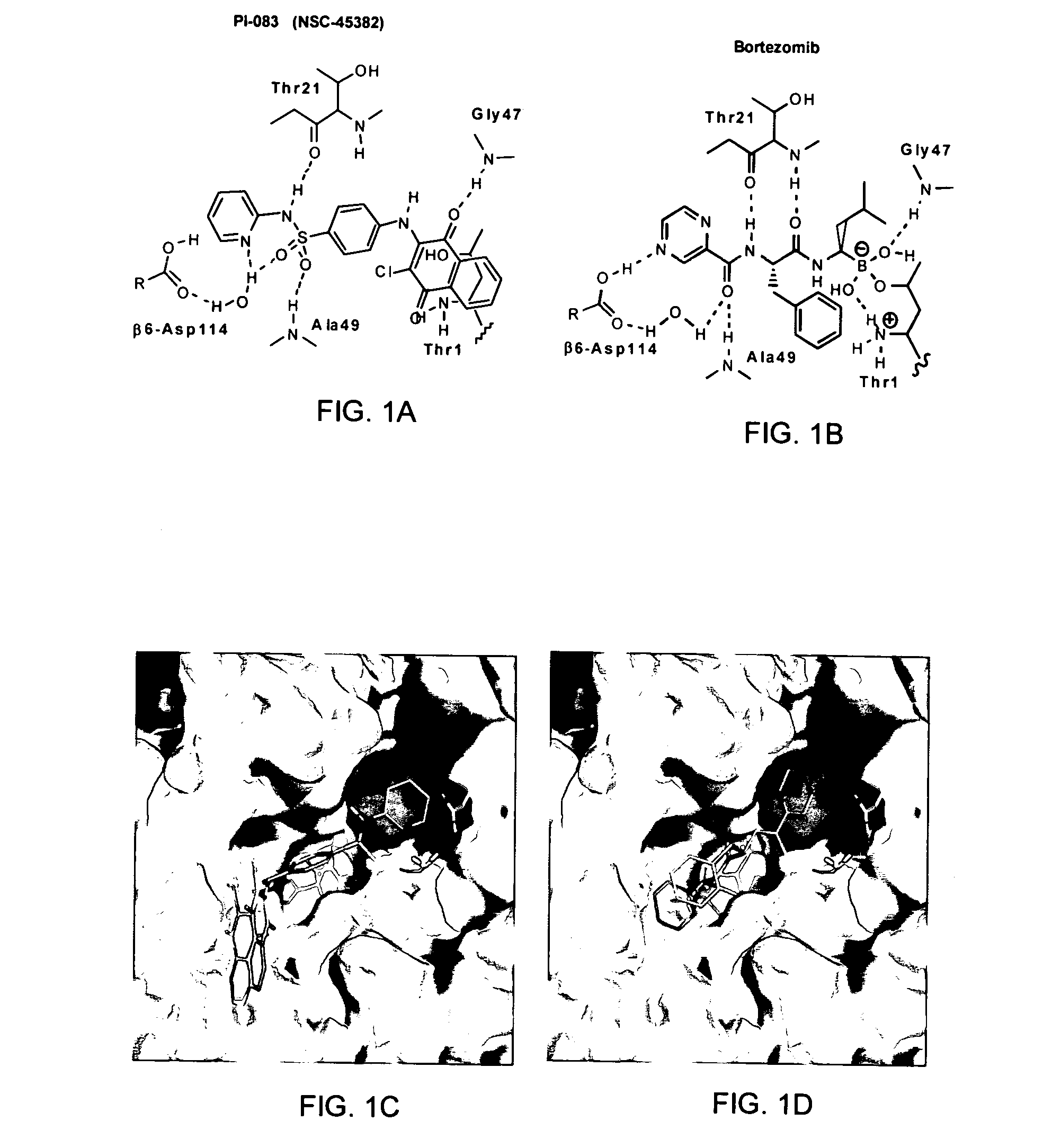
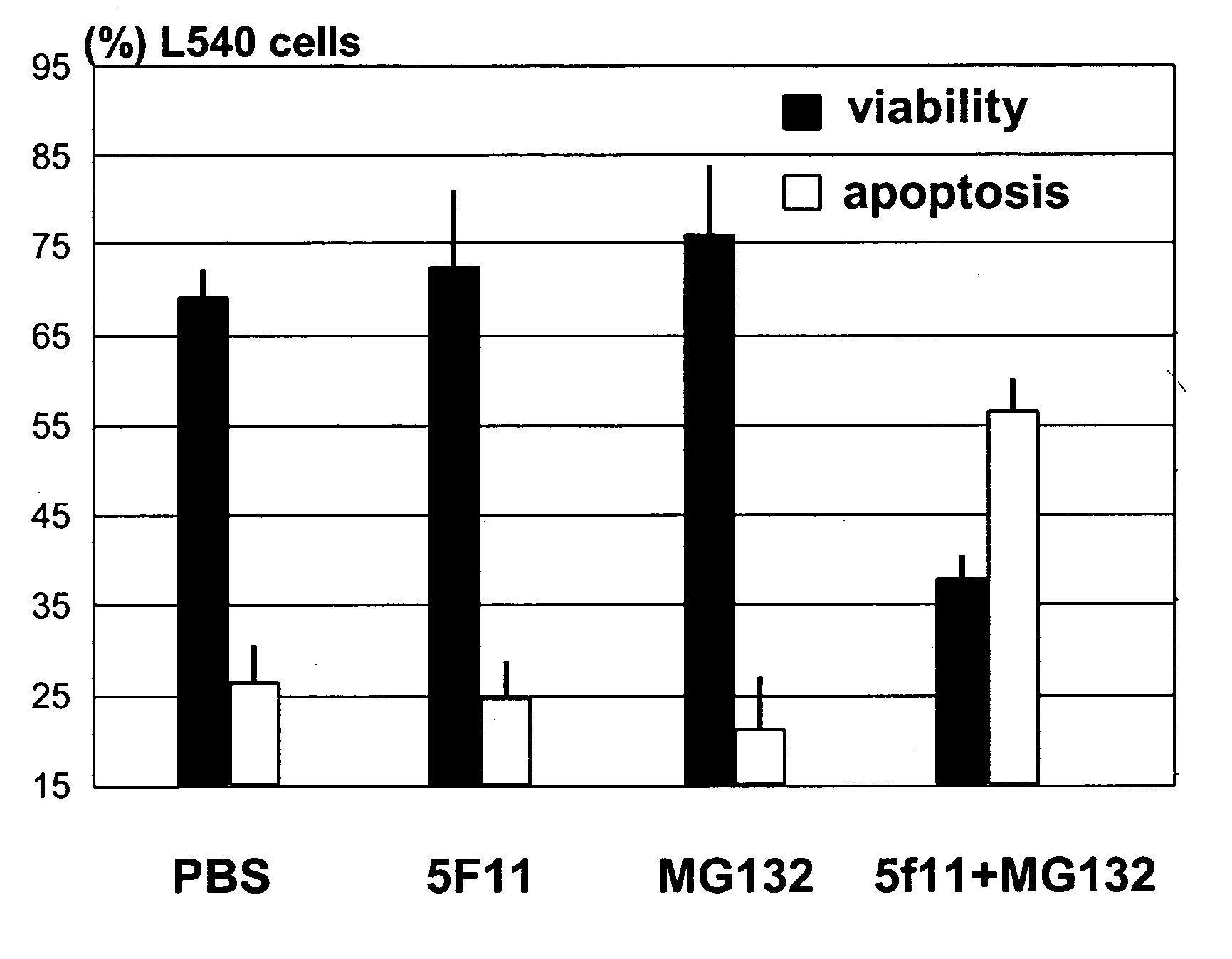
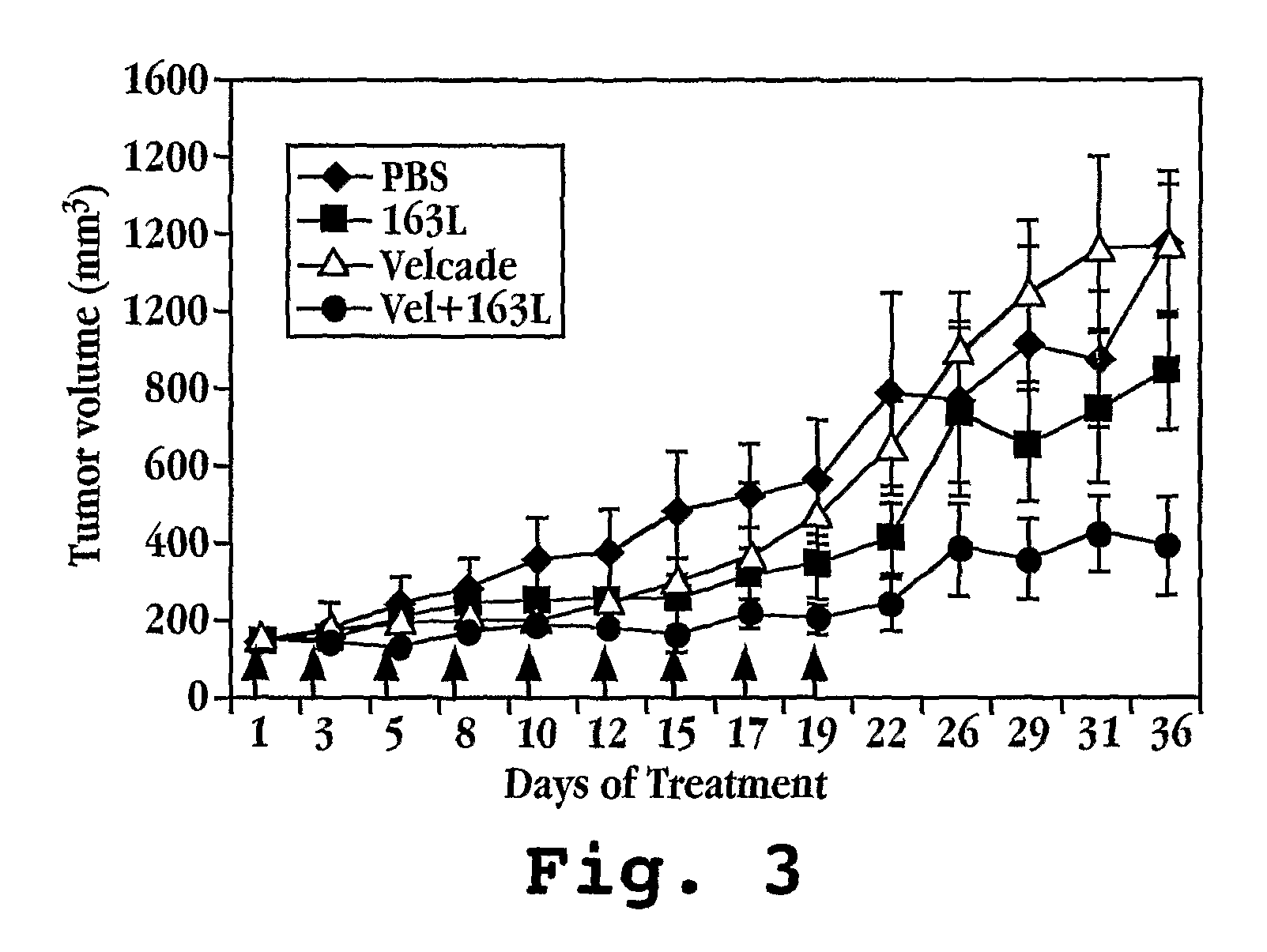
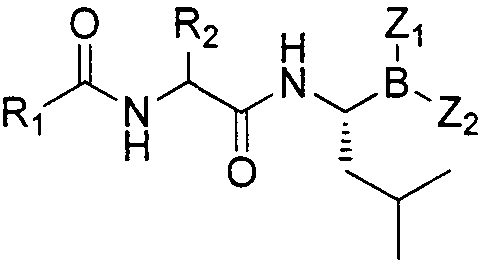
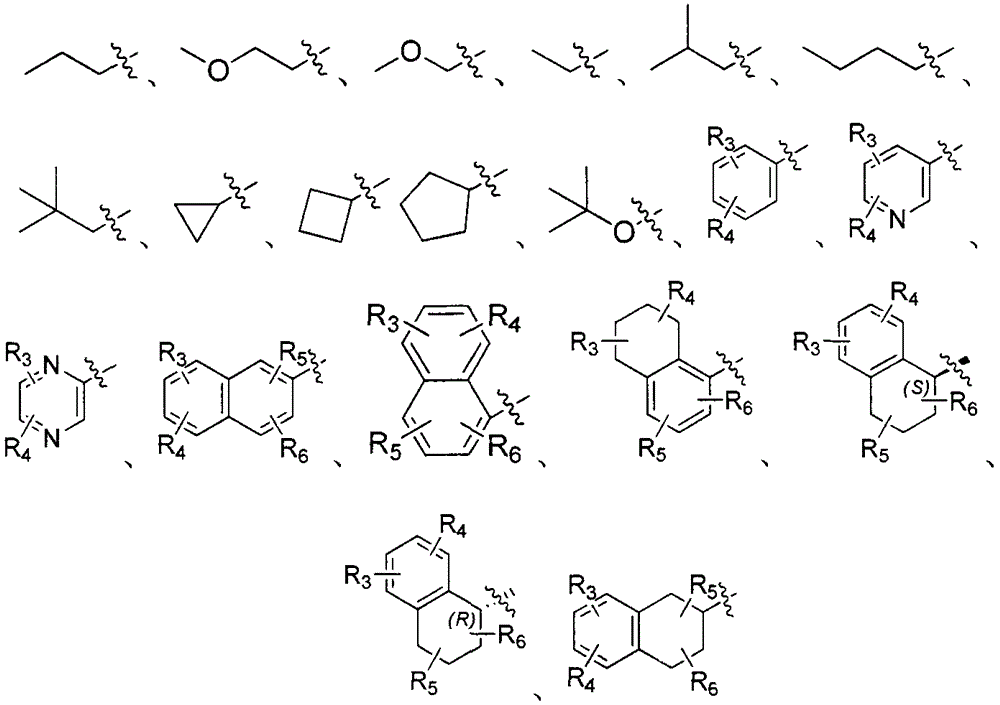
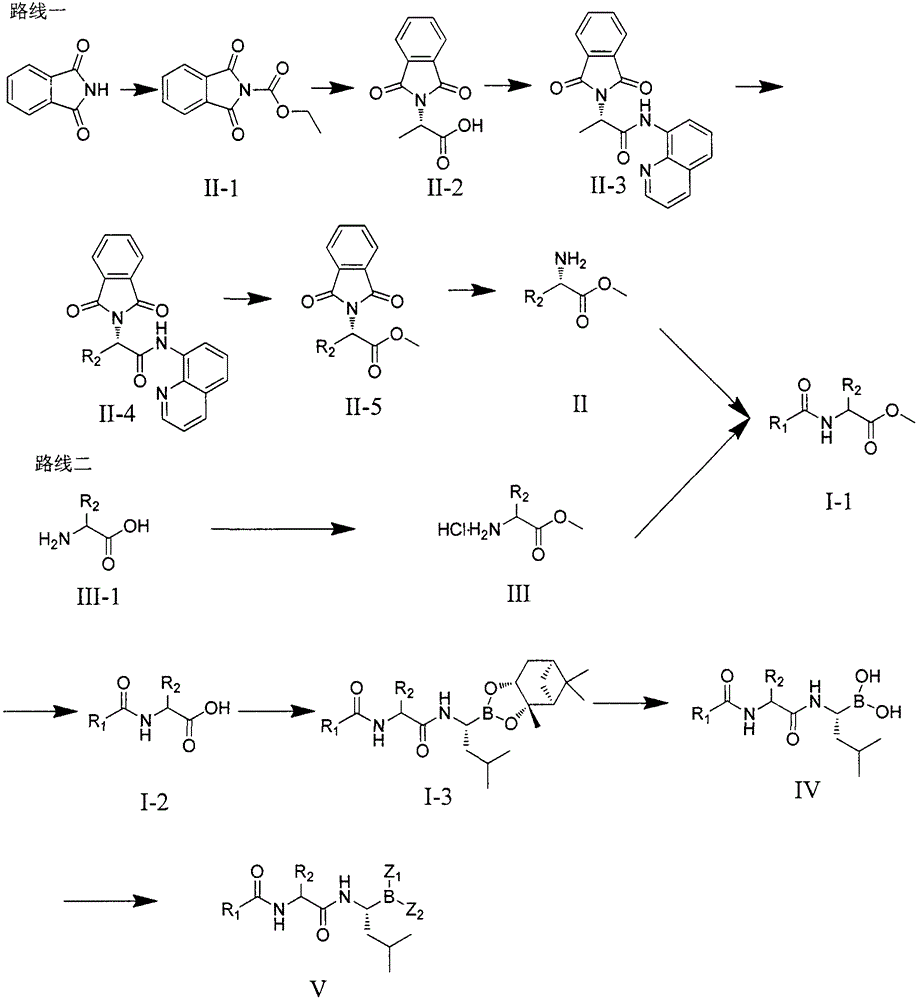
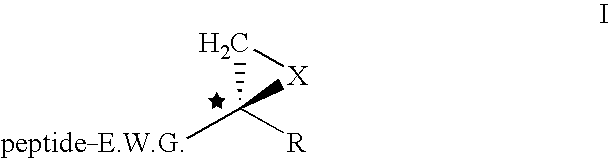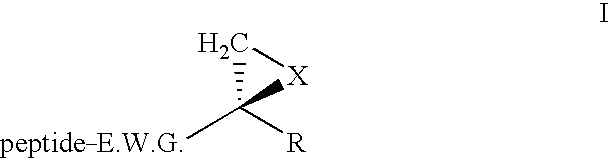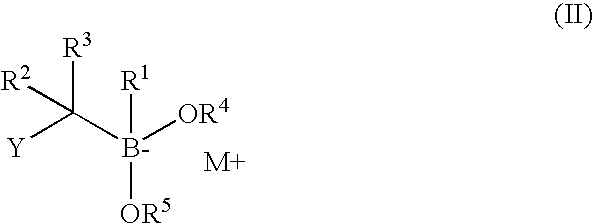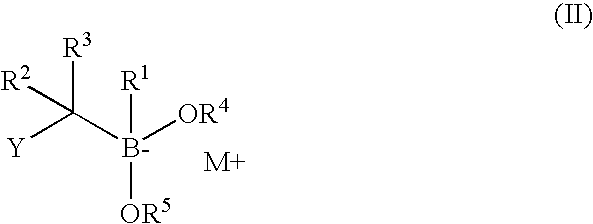Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
215 results about "Proteasome inhibitor" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Proteasome inhibitors are drugs that block the action of proteasomes, cellular complexes that break down proteins. They are being studied in the treatment of cancer; and three are approved for use in treating multiple myeloma.
Treatment of protein degradation disorders
ActiveUS20060239909A1Augmenting protein stressReduce sensitivityCompounds screening/testingBiocideProteasome degradationCompound (substance)
The invention relates to methods of treating protein degradation disorders, such cellular proliferative disorders (e.g., cancer) and protein deposition disorders (e.g., neurodegenerative disorders). The invention provides methods and pharmaceutical compositions for treating these diseases using aggresome inhibitors or combinations of aggresome inhibitors and proteasome inhibitors. The invention further relates to methods and pharmaceutical compositions for treating multiple myeloma. New HDAC / TDAC inhibitors and aggresome inhibitors are also provided as well as synthetic methodologies for preparing these compounds.
Owner:DANA FARBER CANCER INST INC +1
Composition for enzyme inhibition
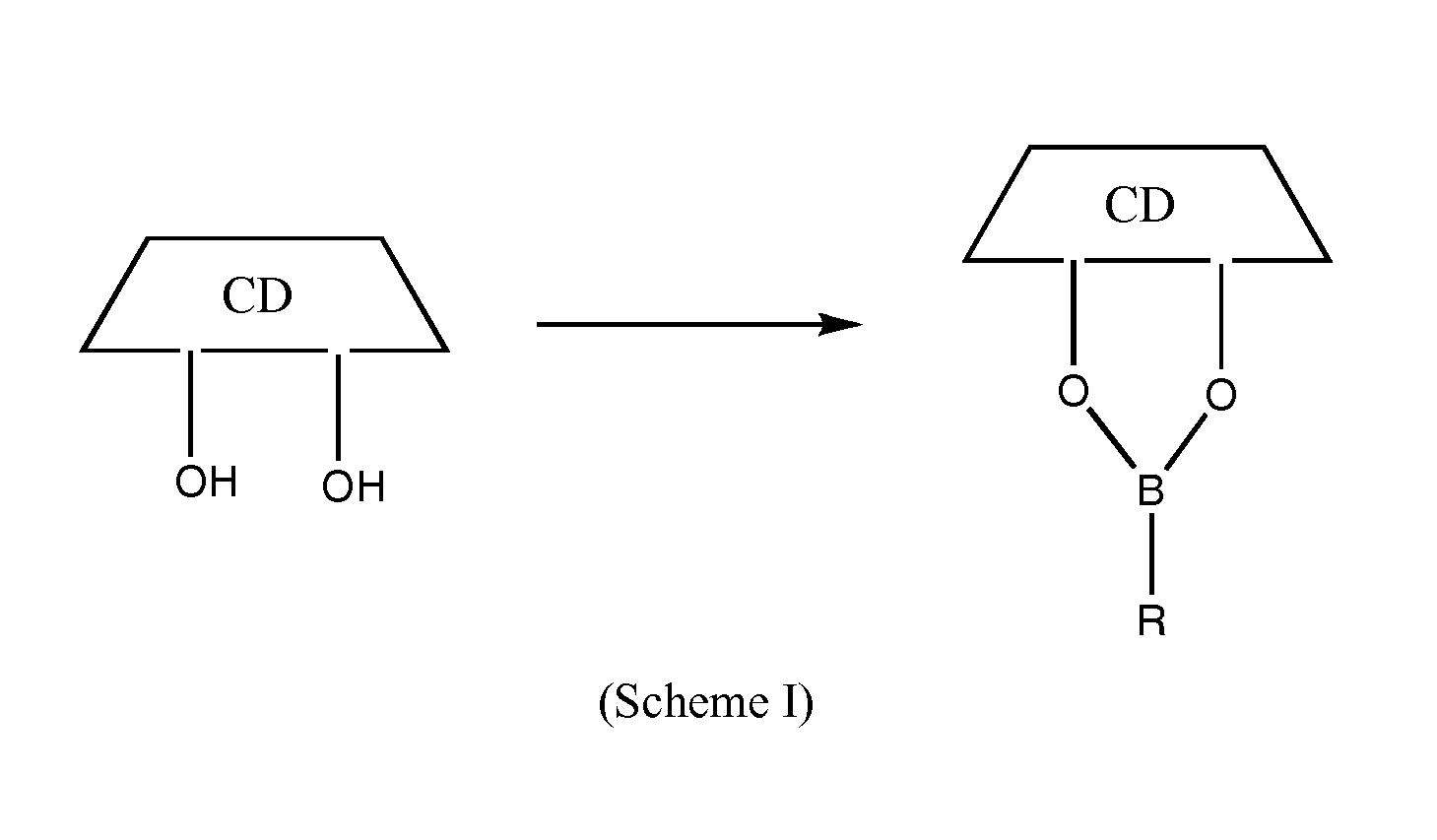
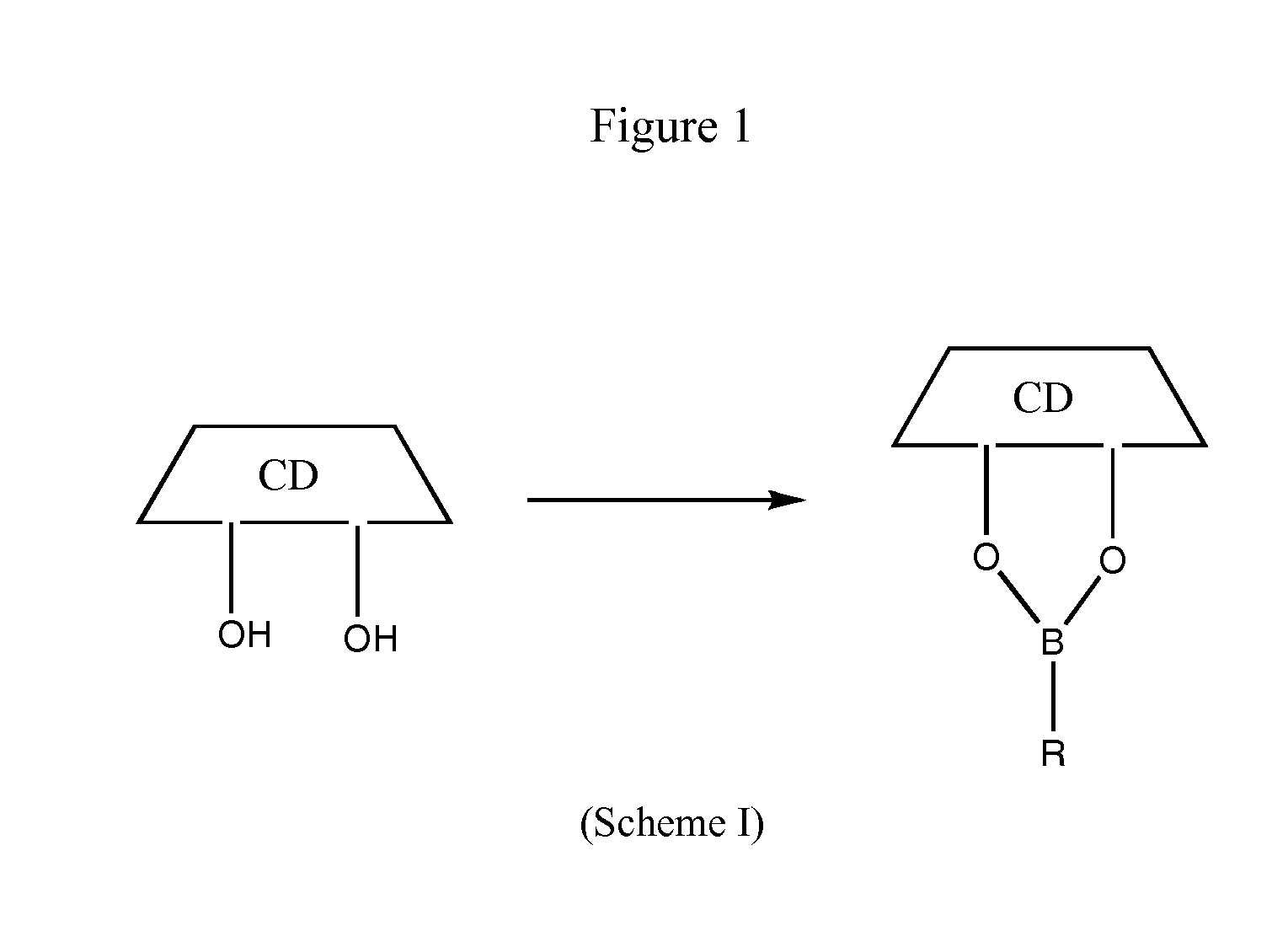
ActiveUS7737112B2Improve solubilityAntibacterial agentsOrganic active ingredientsSolubilityEnzyme inhibition
Compositions comprising one or more practically insoluble proteasome inhibitors and a cyclodextrin, particularly a substituted cyclodextrin, substantially increase the solubility of these proteasome inhibitors and facilitate their administration. Such compositions optionally comprise a buffer. Methods of treatment using such compositions are also disclosed.
Owner:ONYX THERAPEUTICS INC
Composition for enzyme inhibition
ActiveUS20060128611A1SolubilityImprove solubilityAntibacterial agentsOrganic active ingredientsSolubilityProteasome degradation
Compositions comprising one or more practically insoluble proteasome inhibitors and a cyclodextrin, particularly a substituted cyclodextrin, substantially increase the solubility of these proteasome inhibitors and facilitate their administration. Such compositions optionally comprise a buffer. Methods of treatment using such compositions are also disclosed.
Owner:ONYX THERAPEUTICS
Compounds for enzyme inhibition
InactiveUS20070105786A1Inhibiting and reducing HIV infectionAffecting levelBiocideNervous disorderEnzyme inhibitionAziridine
Peptide-based compounds including heteroatom-containing, three-membered rings efficiently and selectively inhibit specific activities of N-terminal nucleophile (Ntn) hydrolases associated with the proteasome. The peptide-based compounds include an epoxide or aziridine, and functionalization at the N-terminus. Among other therapeutic utilities, the peptide-based compounds are expected to display anti-inflammatory properties and inhibition of cell proliferation. Oral administration of these peptide-based proteasome inhibitors is possible due to their bioavailability profiles
Owner:ONYX THERAPEUTICS
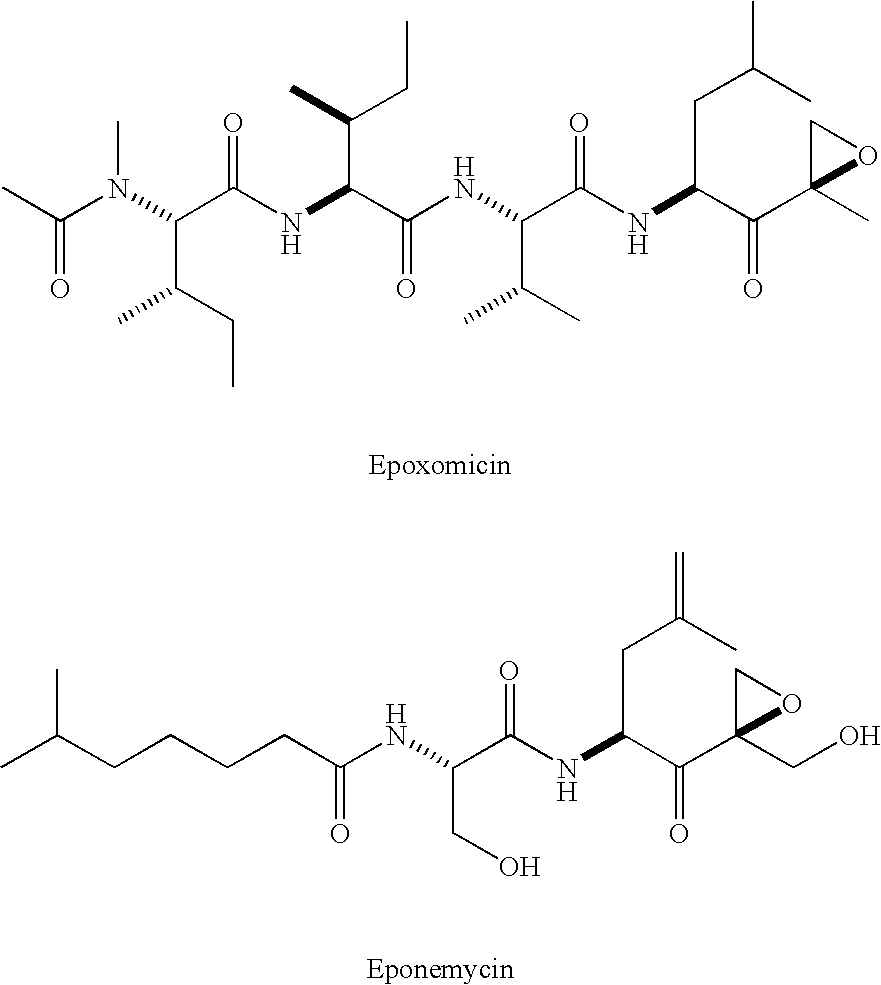
Eponemycin and epoxomicin analogs and uses thereof
InactiveUS20050101781A1Useful in treatmentBiocidePeptide/protein ingredientsPharmaceutical drugOncology
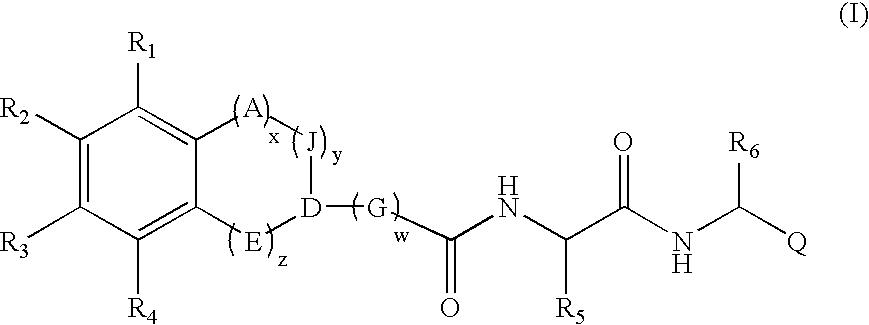
The present invention provides compounds haviang formula (1): wherein R1-R6, A, J, D, E, G, Q, w, x, y, and z are as described generally and in classes and subclasses herein, and additionally provides pharmaceutical compositions thereof, and methods for the use thereof in the treatment of cancer and / or inflammatory disorders, and more generally as proteasome inhibitors.
Owner:EISIA R&D MANAGEMENT CO LTD
Compositions and methods useful for treating diseases
ActiveUS20120225851A1Blocking unwanted cell survival activityBiocideOrganic chemistryDiseaseAutoimmune disease
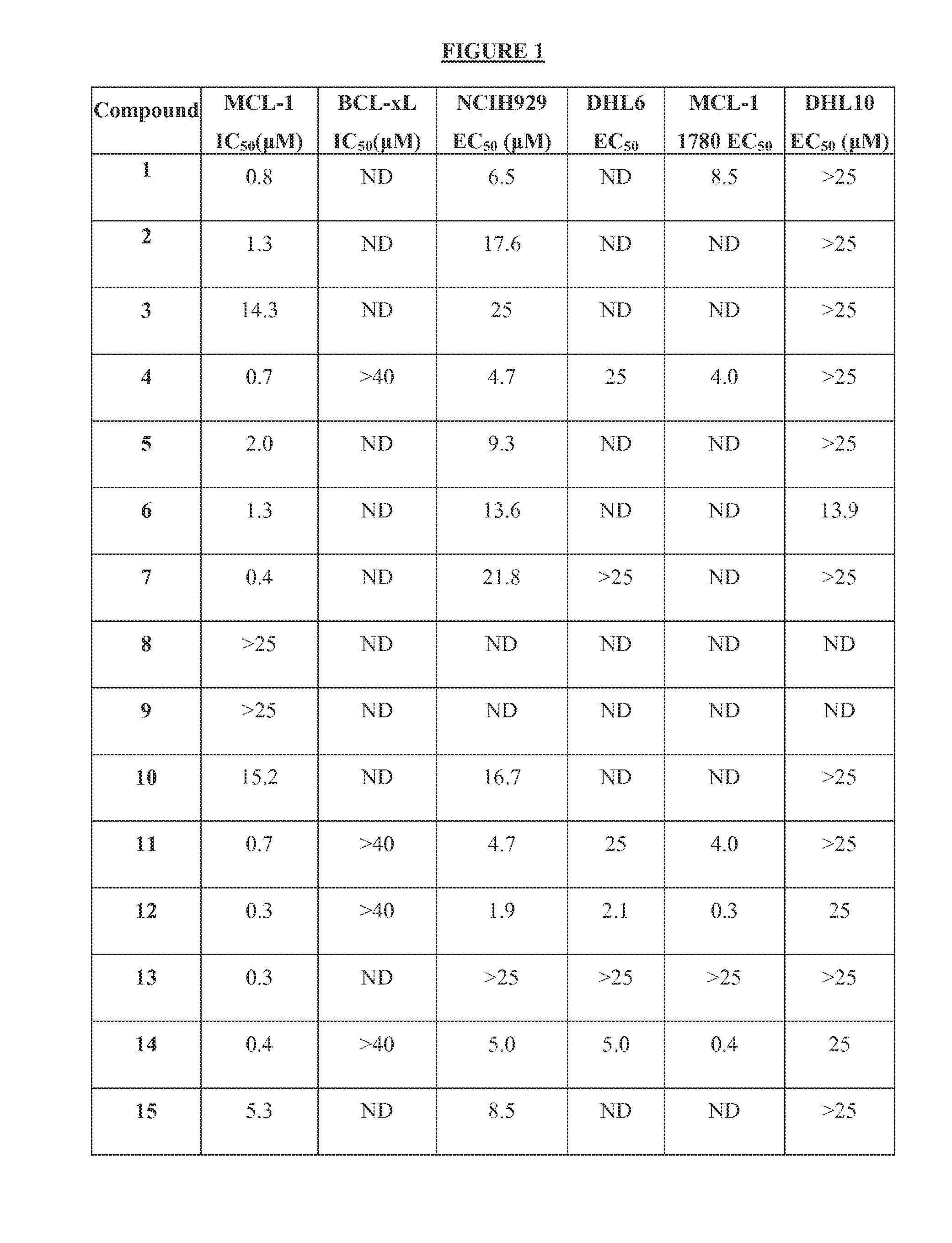
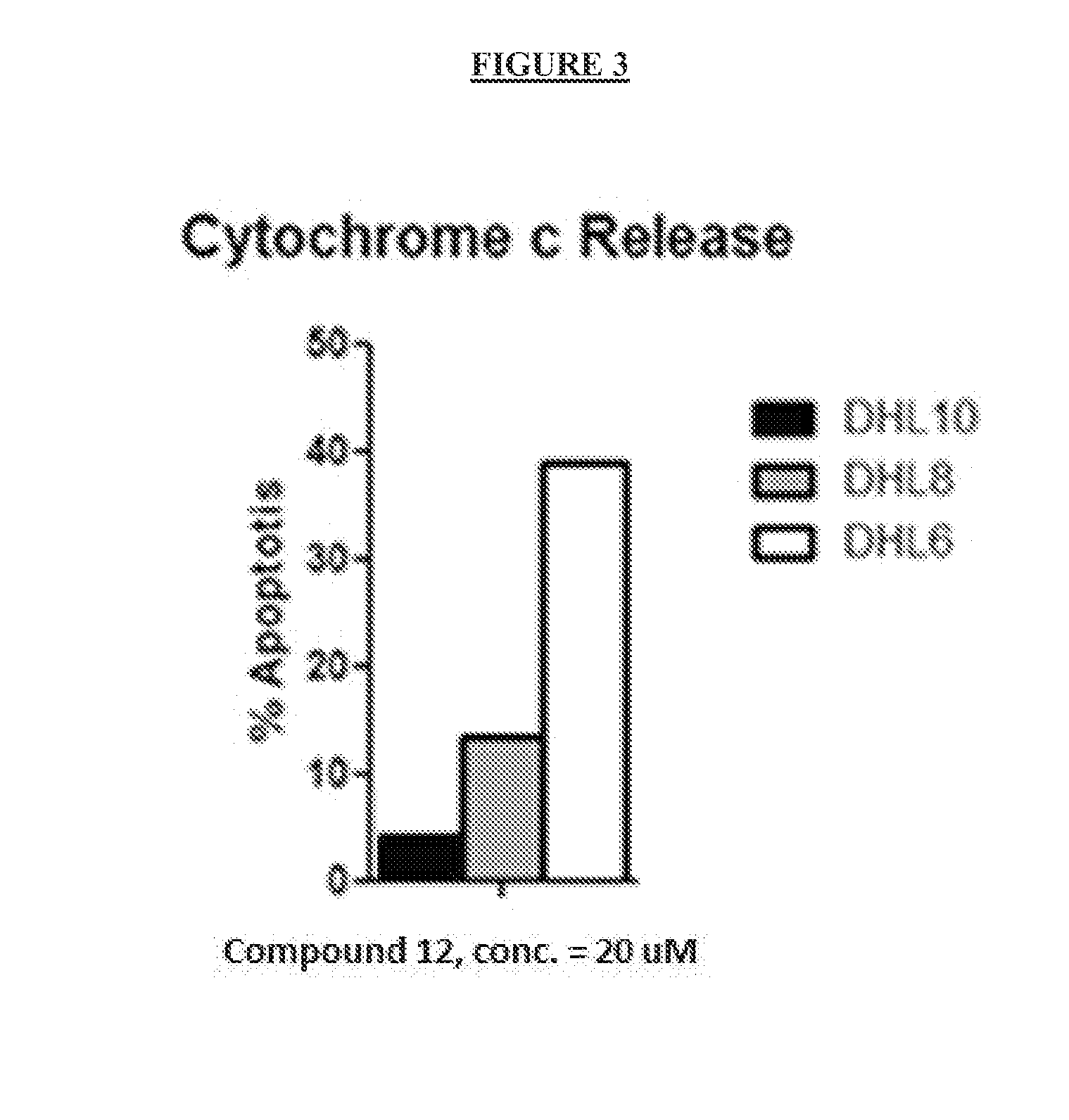
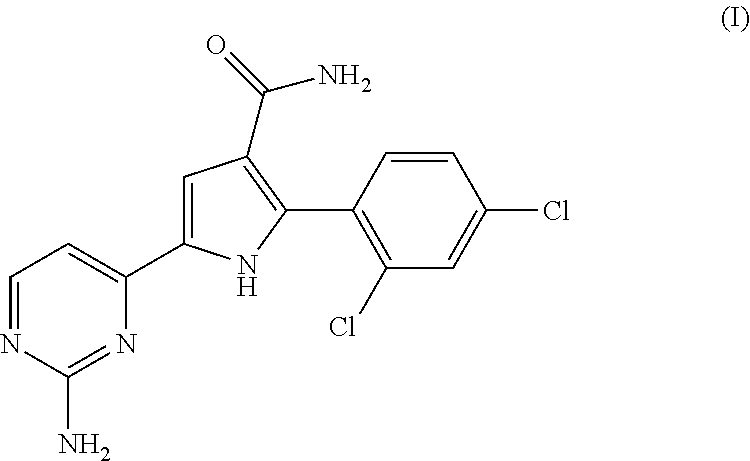
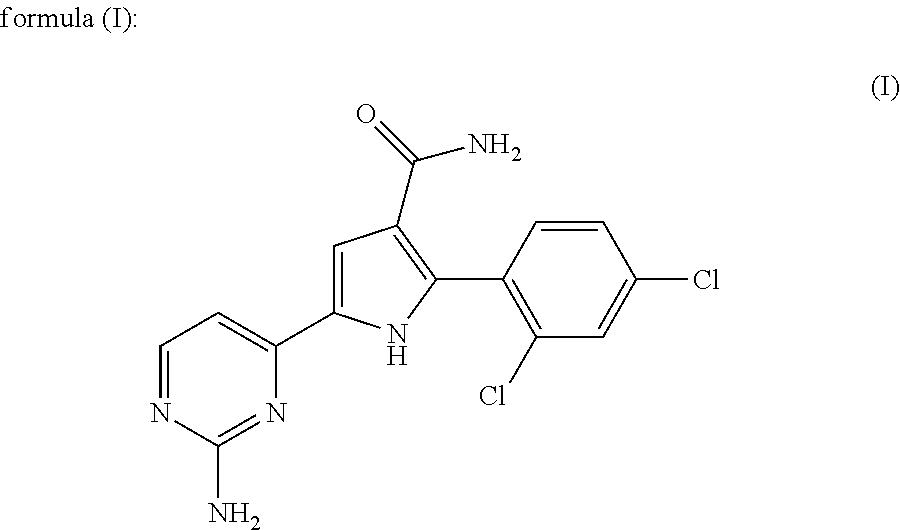
The present invention relates to a chemotherapeutic cancer treatment in which compounds of Formula Ia′, Ib′, Ic′, or II′ (referred to as a group as BH3Is) are administered to a mammal for the treatment of B-cell Lymphoma or other hematopoietic cancers, including diseases associated with MCL-1. In another aspect, the invention provides a method for treating particular types of hematopoietic cancers, such as B-cell lymphoma, using a combination of one or more compounds selected from the group consisting of compounds or Formula Ia, Ib, Ic, or II in combination with other therapies, for example, a class of therapeutics known as 26S proteosome inhibitors, such as, for example, Bortezomib. In another aspect the present invention relates to autoimmune treatment with pharmaceutical compositions comprising one or more compounds of Formula Ia′, Ib′, Ic′, or II′. In another aspect, this invention relates to methods for identifying compounds, for example, compounds of the BH3 mimic class, that have unique in vitro properties that predict in vivo efficacy against B-cell lymphoma tumors and other cancers as well as autoimmune disease.
Owner:EUTROPICS PHARMA
Compounds for Enzyme Inhibition
InactiveUS20090203698A1Inhibiting and reducing HIV infectionAffecting levelBiocideNervous disorderEnzyme inhibitionAziridine
Peptide-based compounds including heteroatom-containing, three-membered rings efficiently and selectively inhibit specific activities of N-terminal nucleophile (Ntn) hydrolases associated with the proteasome. The peptide-based compounds include an epoxide or aziridine, and functionalization at the N-terminus. Among other therapeutic utilities, the peptide-based compounds are expected to display anti-inflammatory properties and inhibition of cell proliferation. Oral administration of these peptide-based proteasome inhibitors is possible due to their bioavailability profiles.
Owner:ONYX THERAPEUTICS INC
Treatment of inflammatory and autoimmune diseases
InactiveUS20010051654A1Low rate and severityEffective treatmentBiocideCarbohydrate active ingredientsDiseaseGlucocorticoid
This invention is directed to the treatment of inflammatory and autoimmune diseases by administering proteasome inhibitors, ubiquitin pathway inhibitors, agents that interfere with the activation of NF-kappaB via the ubiquitin proteasome pathway, or mixtures thereof. The invention is further directed to the treatment of inflammatory and autoimmune diseases by administering an effective combination of a glucocorticoid and a proteasome inhibitor, ubiquitin pathway inhibitor, agent that interferes with the activation of NF-kappaB via the ubiquitin proteasome pathway, or mixture thereof. Pharmaceutical compositions comprising a combination of a glucocorticoid and a proteasome inhibitor, ubiquitin pathway inhibitor, agent that interferes with the activation of NF-kappaB via the ubiquitin proteasome pathway, or mixture thereof are also contemplated within the scope of the invention.
Owner:ELLIOTT PETER J +2
Methods and compositions for treating cancer using proteasome inhibitors
ActiveUS20050267037A1Restore sensitivityAvoid stress responseBiocideCell receptors/surface-antigens/surface-determinantsCancer cellProteasome degradation
The present invention relates to therapeutic compositions for treating cancer or preventing the growth of cancer cells, e.g., tumor growth, in a subject. The present invention also relates to methods for treating cancer, e.g., inhibiting tumor growth, in a subject who has become resistant to treatment, by administering to a subject an effective amount of a proteasome inhibitor and an effective amount of a therapeutic agent, e.g., a chemotherapeutic agent. The present invention further relates to methods for purging bone marrow, i.e., removing cancer cells from bone marrow, by exposing the bone marrow cells to a proteasome inhibitor and a therapeutic agent, e.g., a chemotherapeutic agent.
Owner:DANA FARBER CANCER INST INC
Combination therapy for treating hepatitis viral infection
Disclosed are methods of using proteasome inhibitors (PI) in combinations with other pharmaceutically active agents for treating viral hepatitis infections, for example, for treating therapy-resistant and -refractory viral hepatitis infections. Also disclosed are pharmaceutical compositions and kits of pharmaceutical compositions which can be used for treating viral hepatitis infections, for example, for treating therapy-resistant and refractory viral hepatitis infections.
Owner:SCHUBERT ULRICH
Selective proteasome inhibitors for treating diabetes
InactiveUS20100240581A1Treat and prevent diabetesChronic inflammationBiocideAntipyreticProteasome degradationProteasome inhibitor
Methods of modulating chronic low-grade inflammation are provided. More particularly, methods of treating diabetes, such as for example, type-2 diabetes mellitus, in a mammal by administering an effective amount of a selective proteasome inhibitor are provided. Also provided are unit dosage forms of such inhibitors.
Owner:THE TRUSTEES OF COLUMBIA UNIV IN THE CITY OF NEW YORK
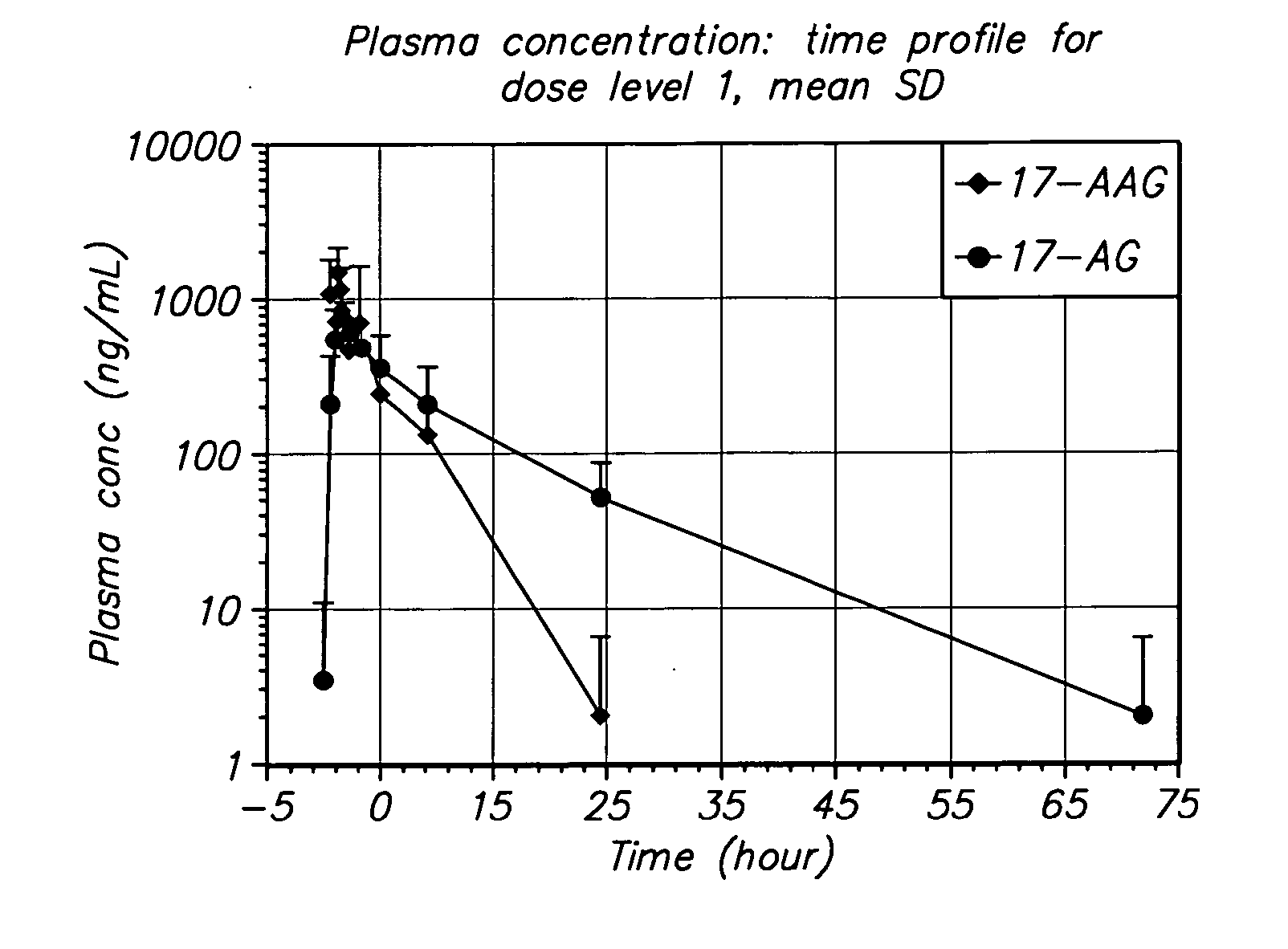
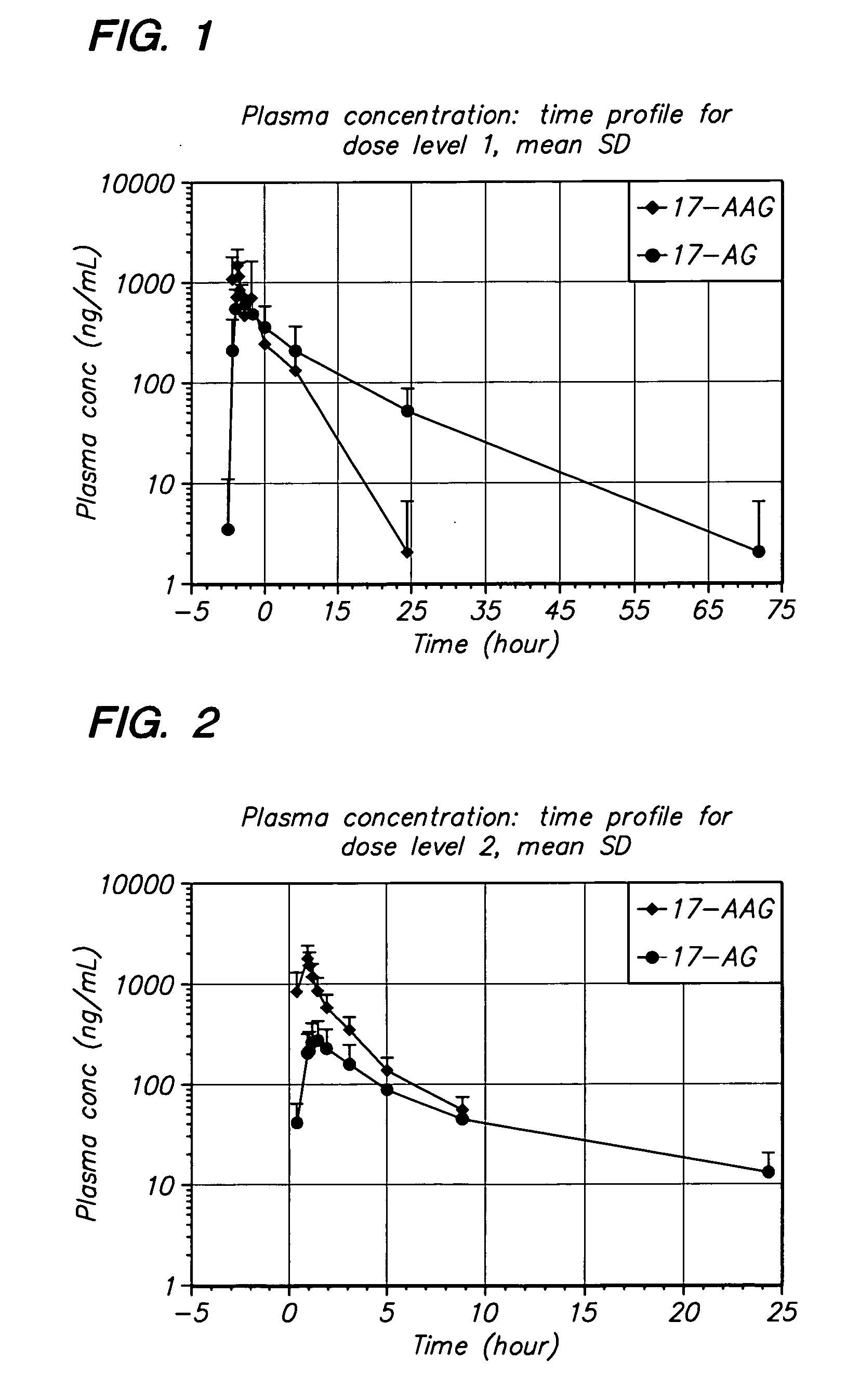
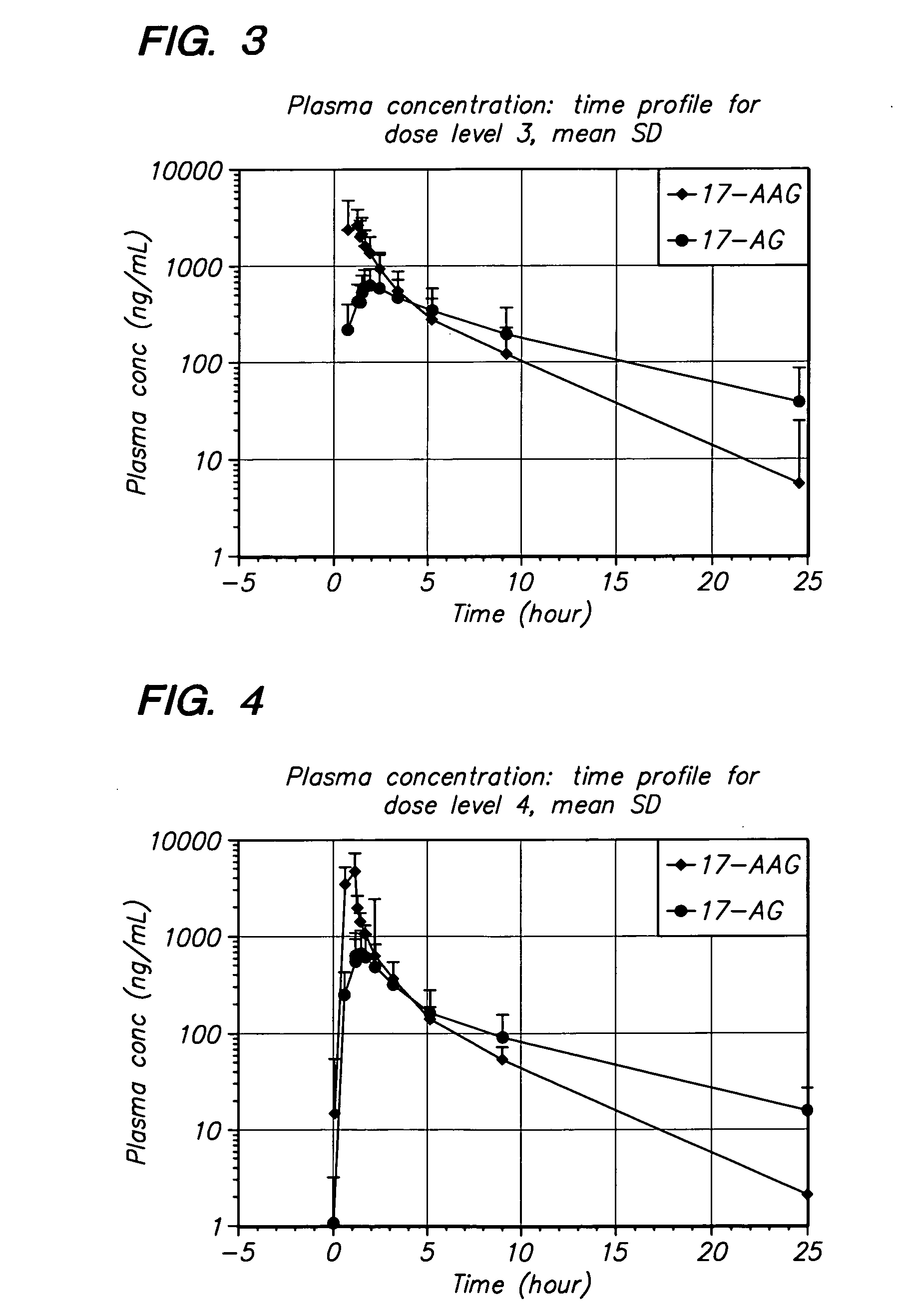
Method of treating multiple myeloma using 17-AAG or 17-AG or a prodrug of either in combination with a proteasome inhibitor
A method for treating multiple myeloma in a subject by administering to the subject 17-allylamino-17-demethoxy-geldanamycin or 17-amino geldanamycin, or a pro drug of either 17-AAG or 17-AG, in combination with a proteasome inhibitor.
Owner:KOSAN BIOSCI
Compositions and methods for treating neoplastic diseases
InactiveUS20070225350A1Increased apoptosisReduce cell viabilityBiocideBoron compound active ingredientsDiseaseCancer research
Disclosed herein are compositions and methods for treating neoplastic diseases. Included are compositions and methods that are effective against multiple myeloma cells resistant to conventional and bortezomib treatment. Furthermore, combination treatment with two different proteosome inhibitors is shown to be synergistic for treating multiple myeloma.
Owner:TRIPHASE RES & DEV I +1
Proteasome inhibitors for selectively inducing apoptosis in cancer cells
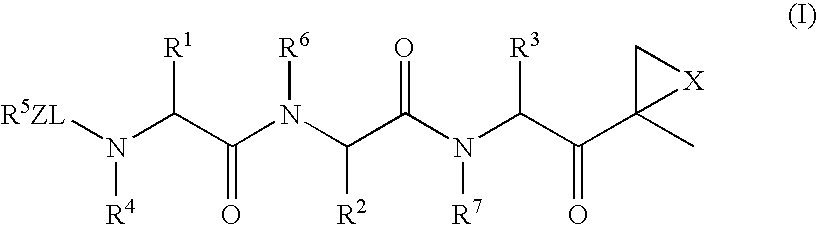
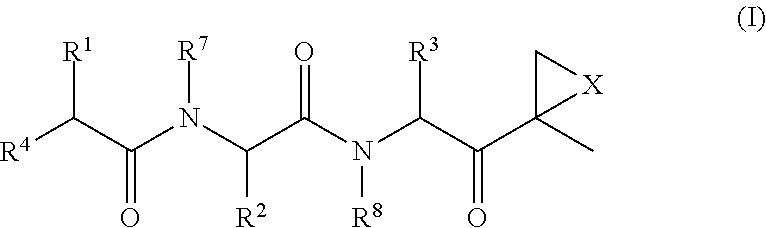
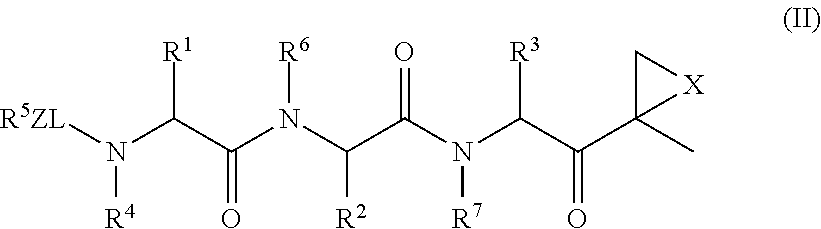
The subject invention concerns compounds having activity as inhibitors of proteasomes and methods of using the subject compounds. In one embodiment, a compound of the invention has the chemical structure shown in formula I:whereinR1 is an organic cyclic ring structure bonded to a sulfonamide structure;R2 is H, halogen, alkyl, —NR6R7, or heteroalkyl;R3 is H, halogen, —OH, —O-alkyl, alkyl, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, —NO2, —NH2 or substituted amines;R4 is H, alkyl, heteroalkyl, aryl, or heteroaryl, any of which can be optionally substituted with one or more of —NO2, alkyl, heteroalkyl, aryl, or heteroaryl, or halogen;R5 is H, —OH, halogen, alkyl, aryl, heteroaryl, cycloalkyl, heterocycloalkyl, —O-alkyl, —O-aryl, heteroalkyl, —NO2, —NH2, or substituted amine; andR6 and R7 are independently H, O, alkyl, aryl, heterocycloalkyl, or heteroaryl, or together can form a heterocycloalkyl or a heteroaryl, any of which can be optionally substituted with one or more of —NO2, alkyl, heteroalkyl, aryl, or halogen;or a pharmaceutically acceptable salt or hydrate thereof.In another embodiment, a compound of the invention has the chemical structure shown in formula II:whereinQ, W, X, Y, Z are each independently carbon, oxygen, or nitrogen;R1 is H, or X1R8;R2 is heteroalkyl, which can be optionally substituted with one or more of —OH, halogen, —C(O)OR4, alkyl, heteroalkyl, heterocycloalkyl, or heteroaryl;R3 is heterocycloalkyl, aryl, heteroaryl, any of which can be optionally substituted with one or more of a halogen or —OH; andR4 is H or alkyl;R5 is halogen, alkyl or nitro;R6 is nitro, X2R9 or a halogen;R7 is H or alkyl;R8 is H, alkyl, aryl, CH2-alkyl-aryl, -alkyl-C(O)OH, or alkyl-tetrazole (aromatic and aliphatic heterocyclic groups);R9 is H or alkyl;X1 is oxygen, nitrogen, or sulfur;X2 is oxygen, nitrogen, or sulfur;or a pharmaceutically acceptable salt or hydrate thereof.
Owner:UNIV OF SOUTH FLORIDA +1
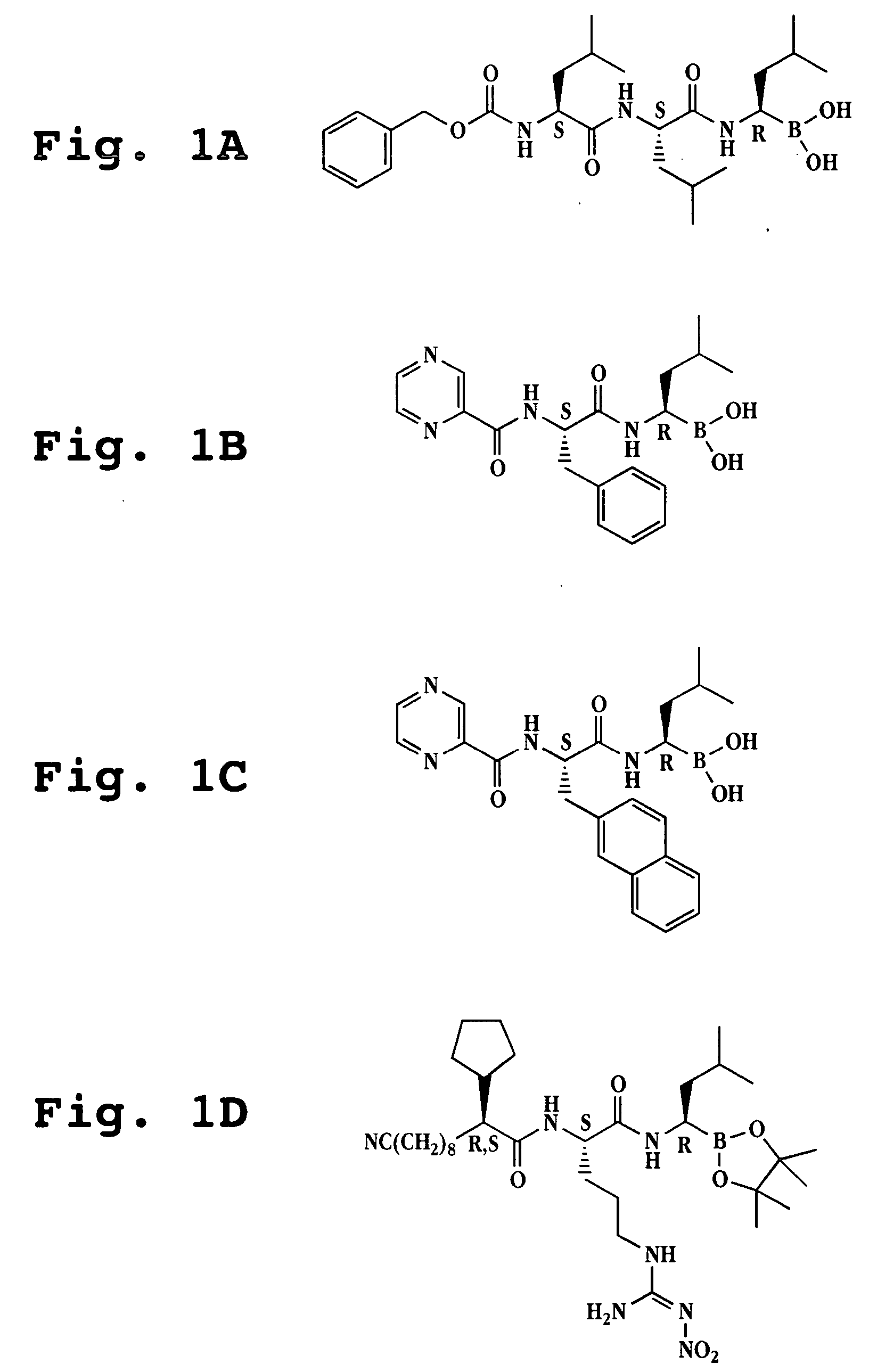
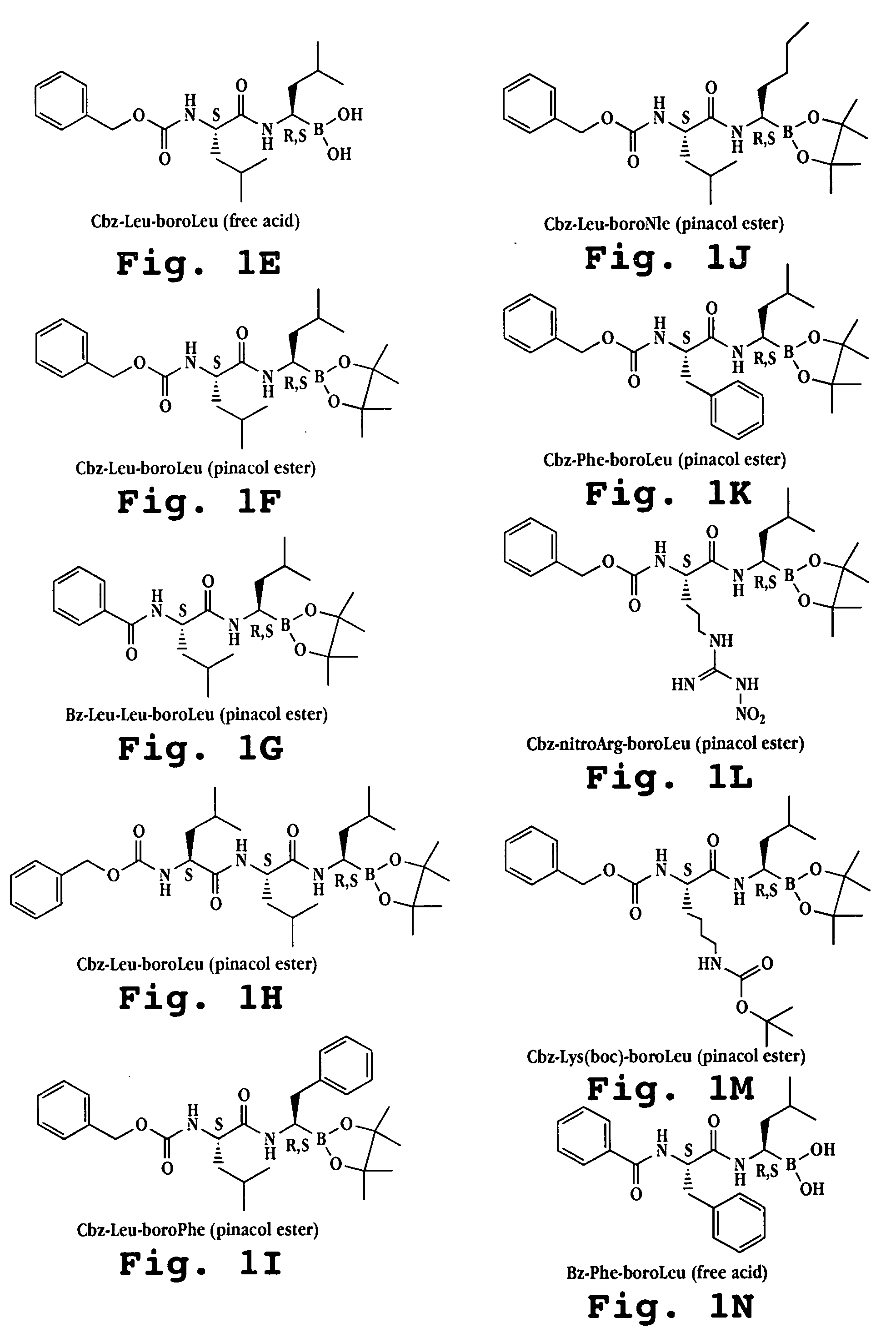
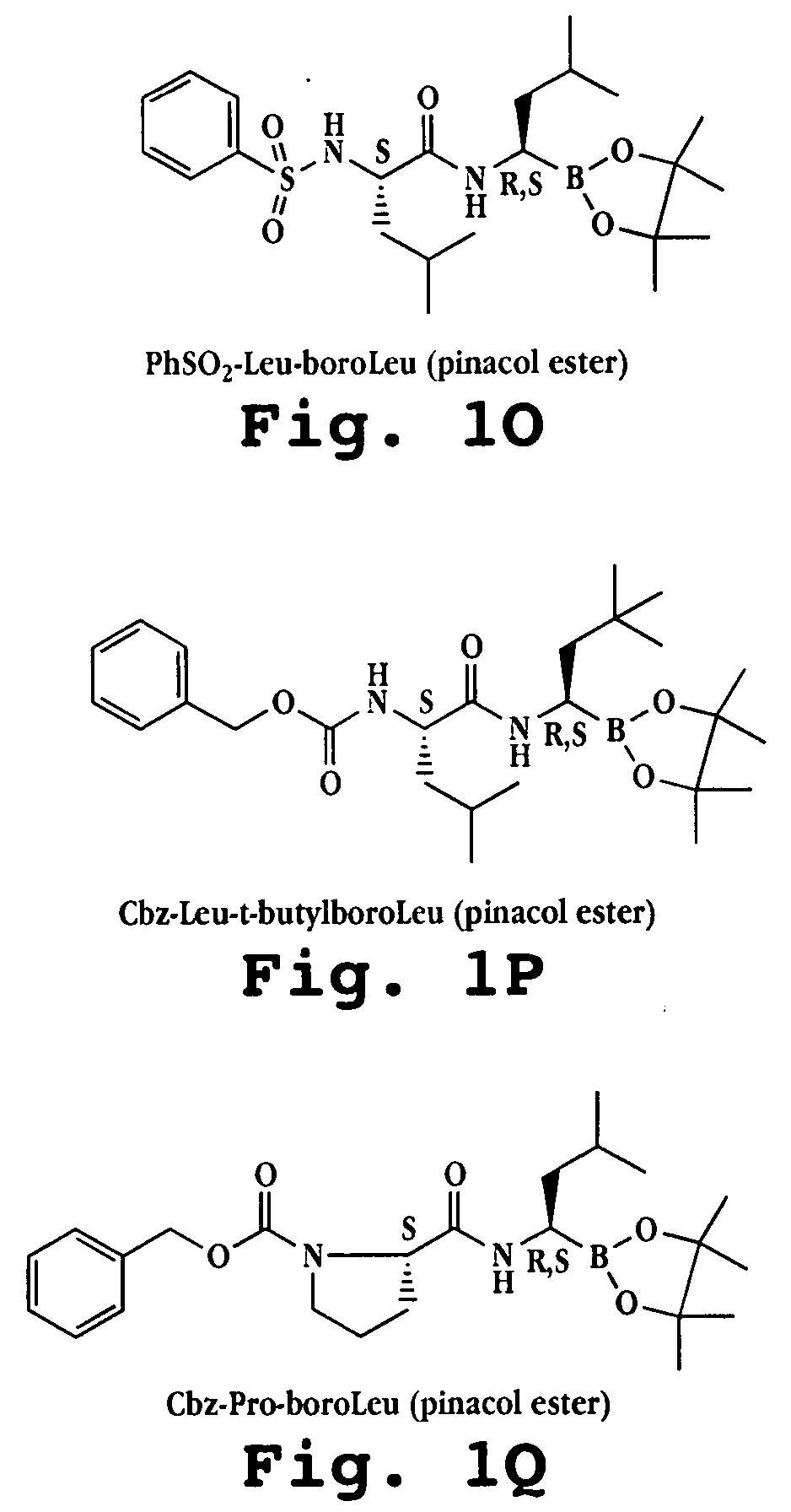
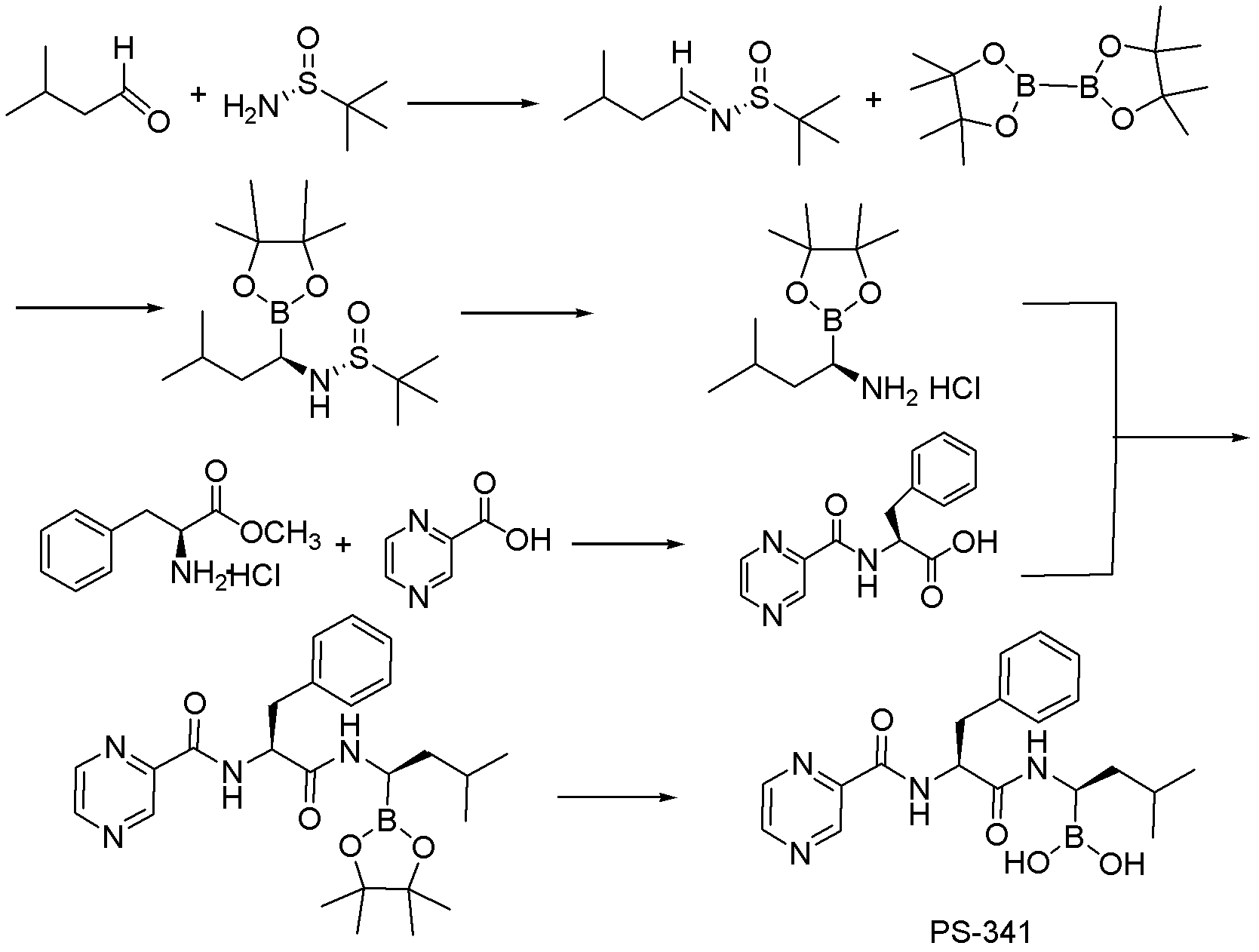
Synthesis of boronic ester and acid compounds
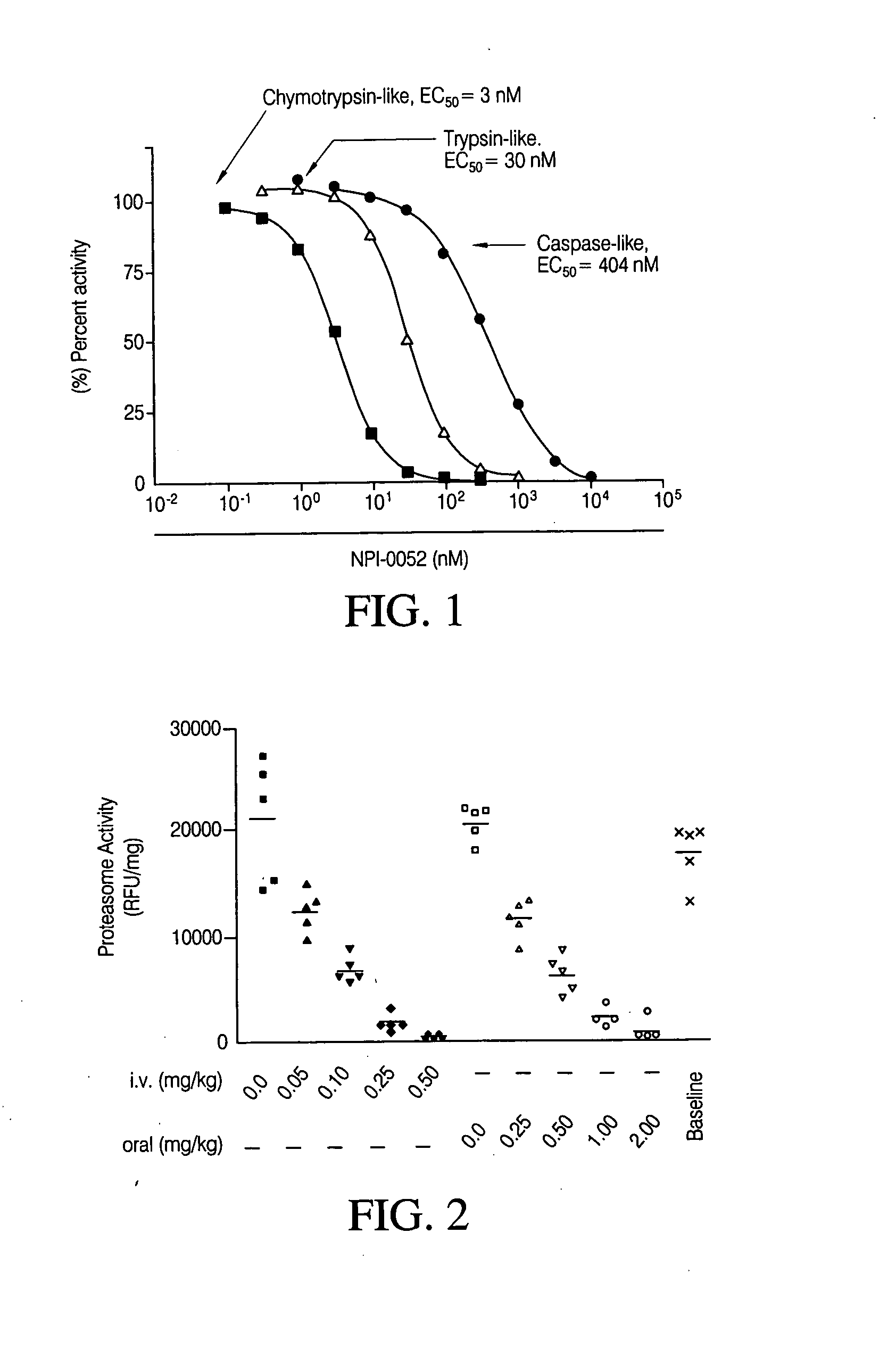
The invention relates to the synthesis of boronic ester and acid compounds. More particularly, the invention provides improved synthetic processes for the large-scale production of boronic ester and acid compounds, including the peptide boronic acid proteasome inhibitor bortezomib.
Owner:MILLENNIUM PHARMA INC
Method of treating CD30 positive lymphomas
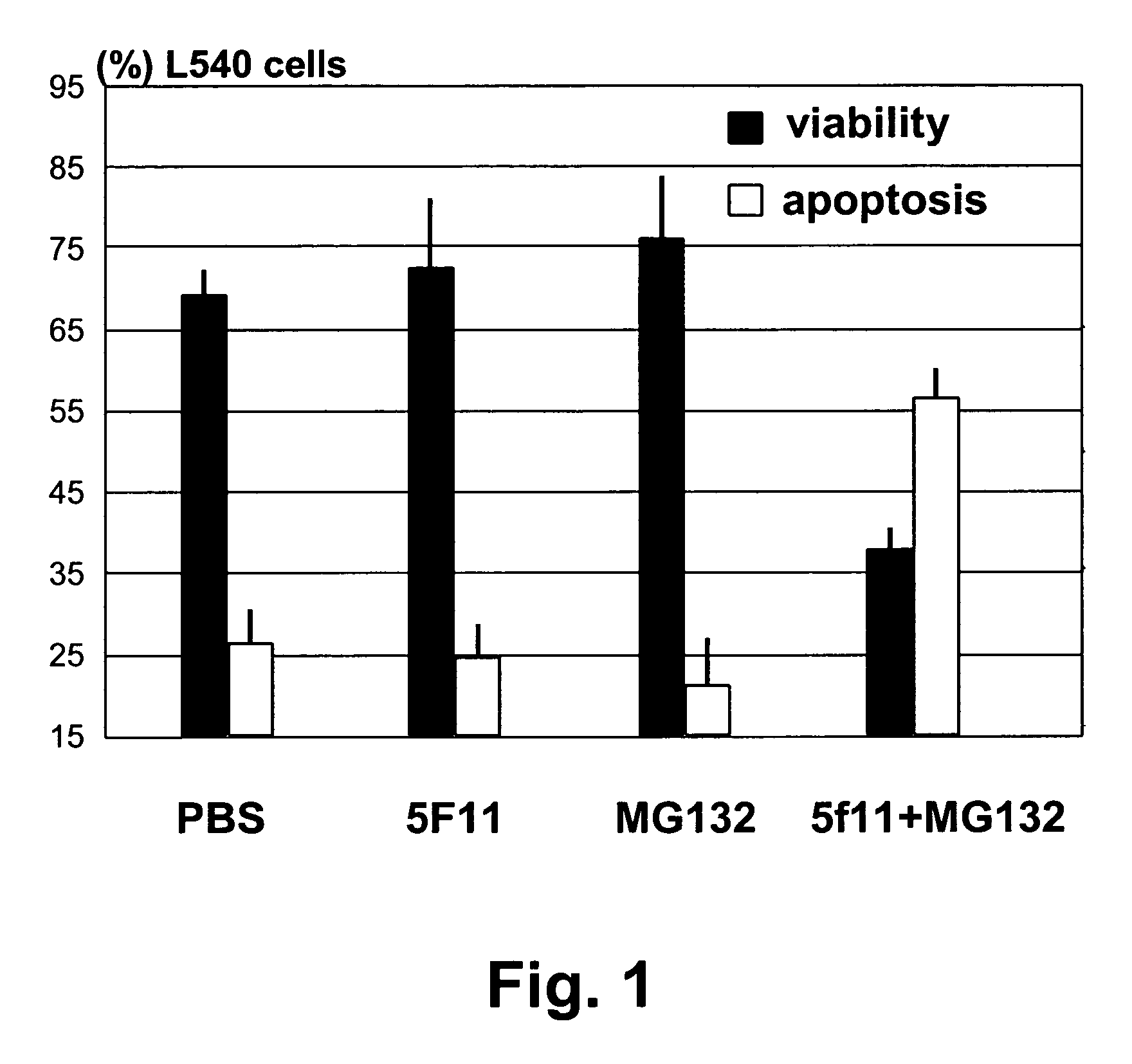
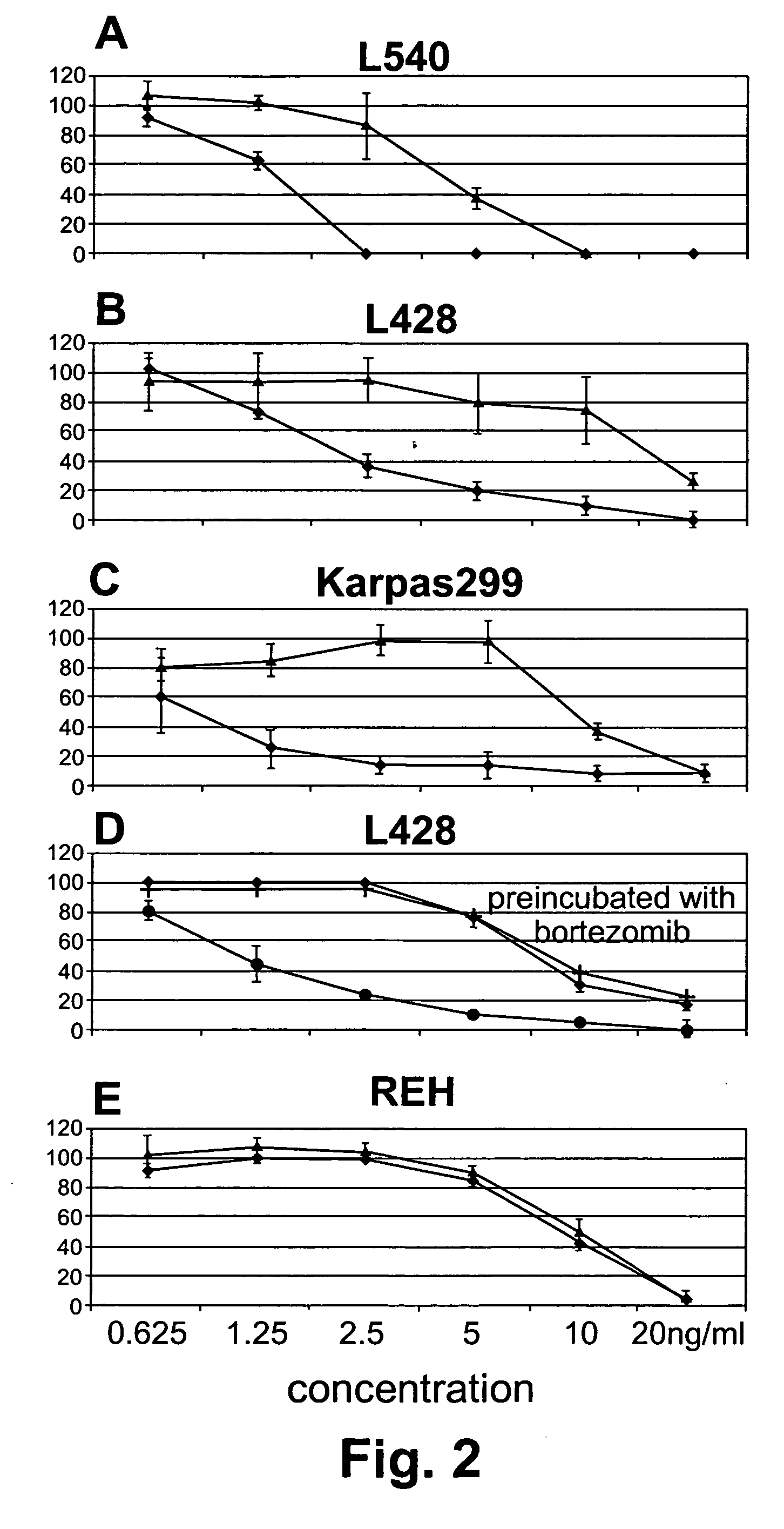
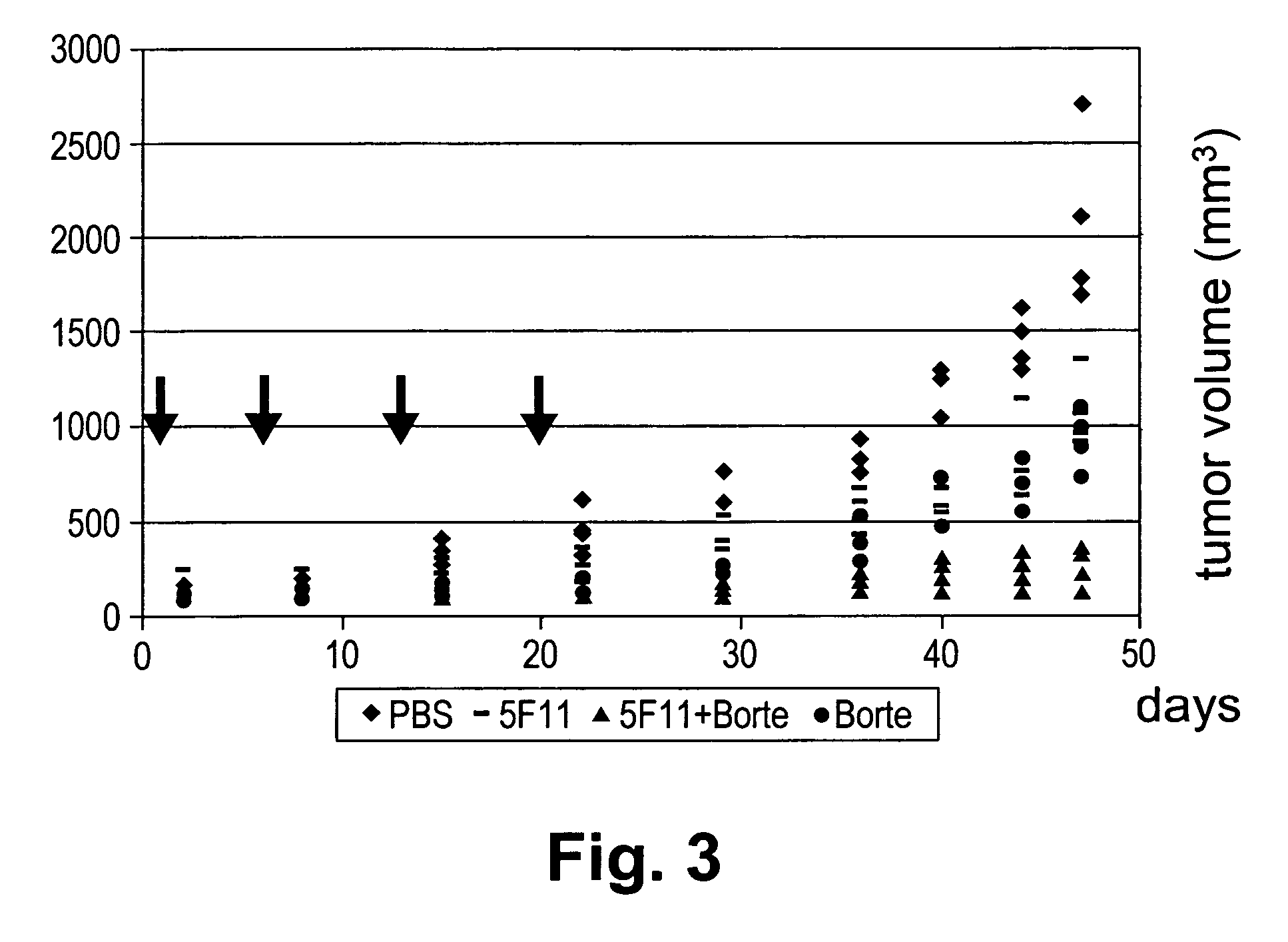
InactiveUS20060177442A1Good curative effectSuppress NF-kB activationBiocideDipeptide ingredientsProteasome degradationAntibody
Methods for treating lymphomas characterized by expression of CD30 using anti-CD30 antibodies and proteasome inhibitors in combination are disclosed.
Owner:MEDAREX LLC
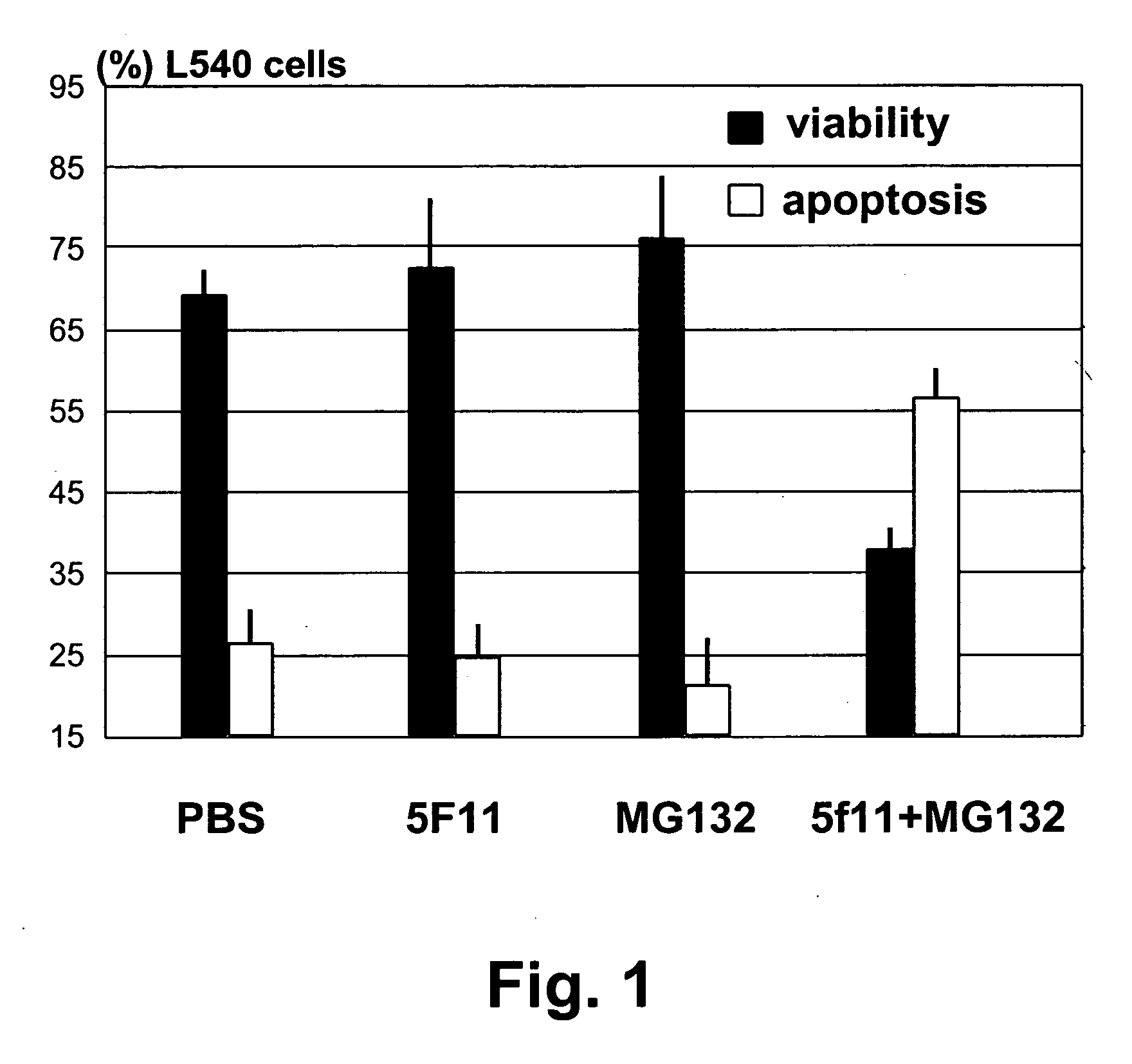
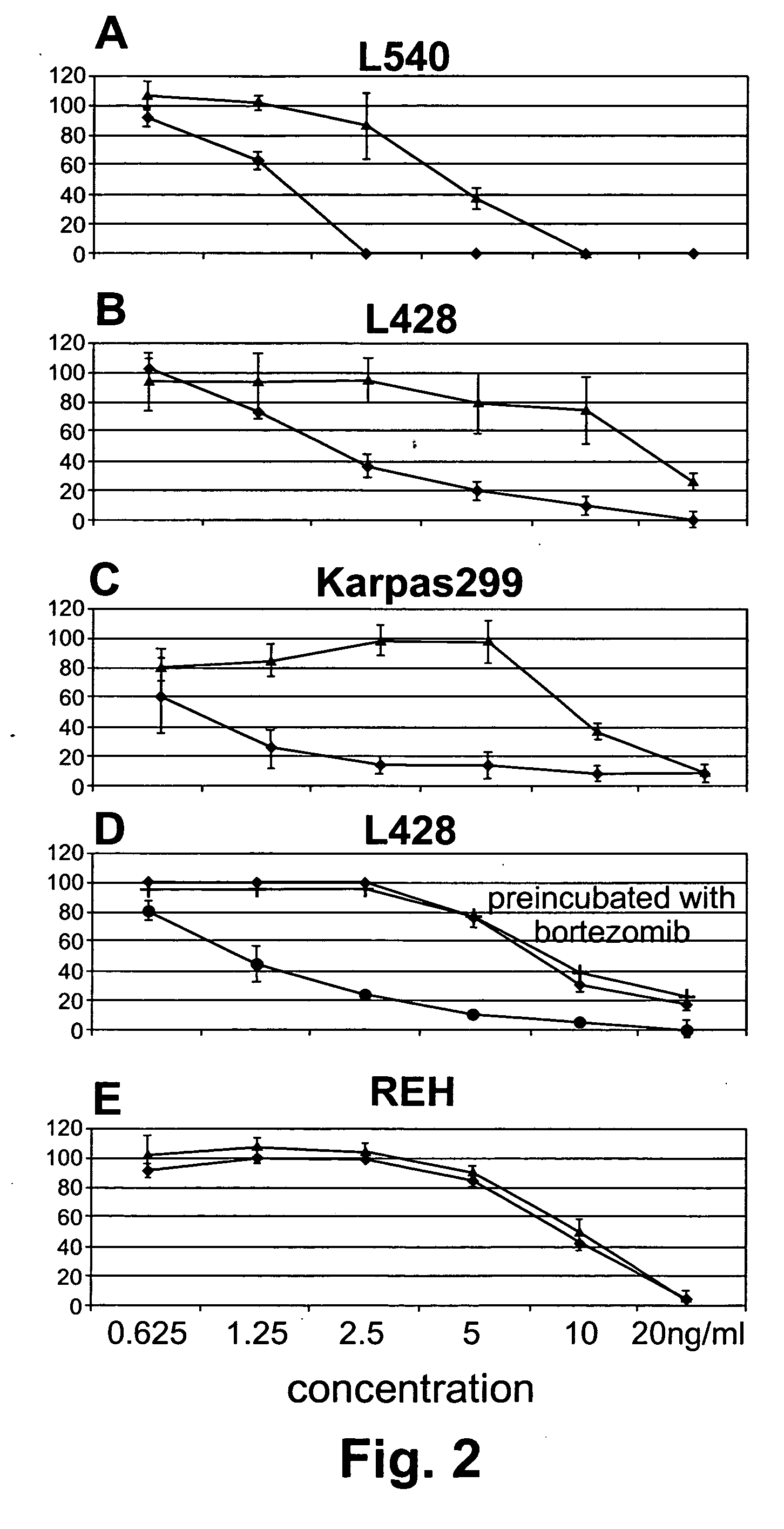
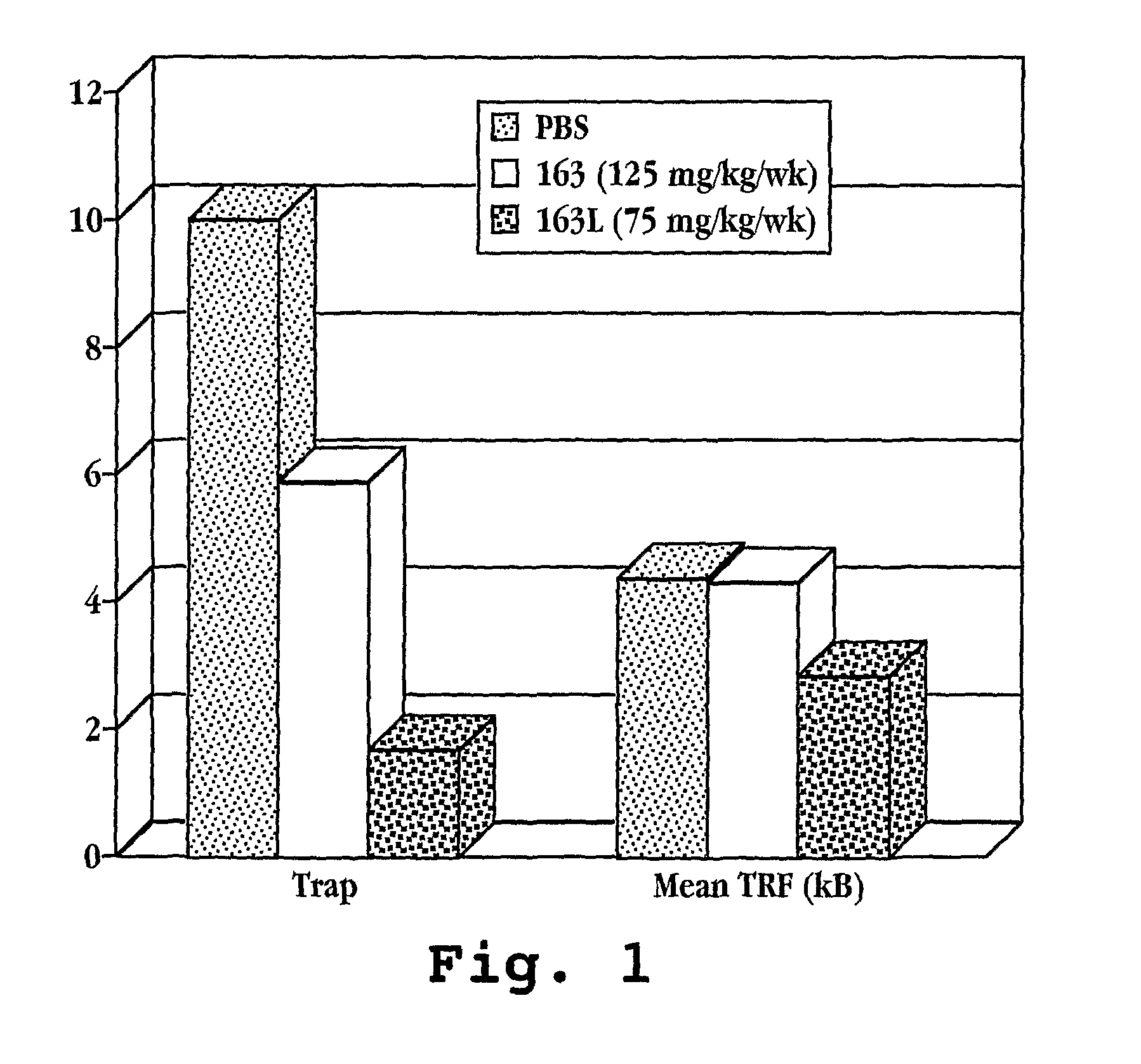
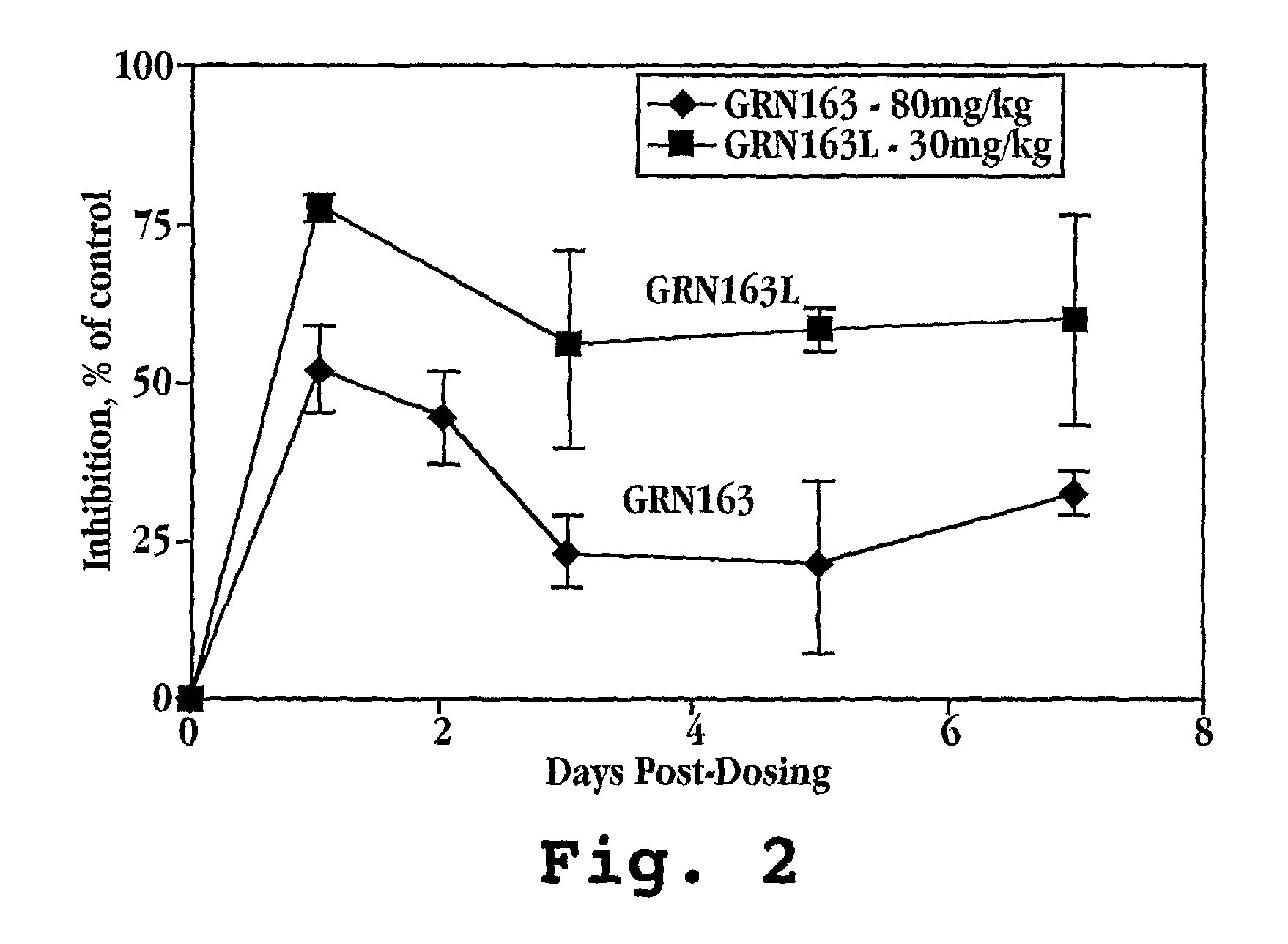
Cancer treatment by combined inhibition of proteasome and telomerase activities
A method and kit for inhibiting the proliferation of cancer cells are disclosed, based on a combination of a proteasome inhibitor and a telomerase inhibitor. When used in cancer therapy, the two compounds in combination enhance the anti-cancer treatment efficacy obtained with the proteasome inhibitor alone or the telomerase inhibitor alone. Preferably, efficacy is supraadditive or synergistic in nature relative to the combined effects of the individual agents, with minimal exacerbation of side effects.
Owner:GERON CORPORATION +1
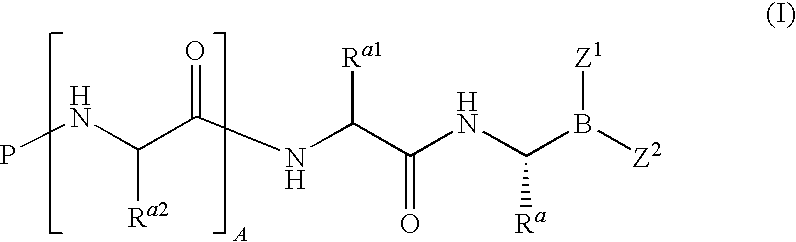
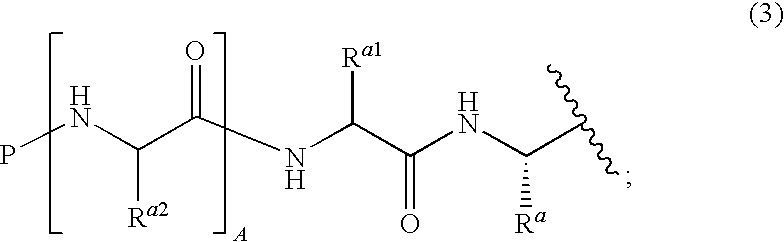
Dipeptide boric acid composed of carboxylic acid and alpha-amino acid as well as ester compound thereof, and preparation method and application of dipeptide boric acid and ester compound thereof
ActiveCN105732683AHigh yieldHigh activityBoron compound active ingredientsGroup 3/13 element organic compoundsProstate cancerProteasome inhibitor
The invention belongs to the field of drug synthesis and in particular relates to a series of novel peptide boric acids as well as an ester compound or pharmaceutical salt thereof, and a preparation method and application of the peptide boric acids as well as the ester compound or pharmaceutical salt thereof in pharmacodynamics. A structure of the peptide boric acid and the ester compound or pharmaceutical salt thereof is shown in a formula I (described in the specification). The compound provided by the invention can be used for preparing a proteasome inhibitor and can further be used for treating solid tumours and blood tumours, wherein the solid tumours are selected from non-small cell lung cancer, small cell lung cancer, lung adenocarcinoma, lung squamous carcinoma, pancreatic cancer, breast cancer, prostate cancer, liver cancer, skin cancer, epithelial cell cancer, gastrointestinal stromal tumor, nasopharynx cancer and leukemia; and the blood tumours are selected from multiple myeloma, mantle cell lymphoma and histiocytic lymphoma.
Owner:JIANGSU CHIA TAI FENGHAI PHARMA
Liposome formulations of boronic acid compounds
A liposome composition comprised of liposomes having the peptide boronic acid proteasome inhibitor compound bortezomib entrapped in the liposomes is described. The boronic acid compound is entrapped in the liposomes in the form of a boronate ester, subsequent to interaction with a liposome-entrapped polyol. In one embodiment, the liposomes have an outer coating of hydrophilic polymer chains and are used to treat a solid tumor in a subject.
Owner:ALZA CORP
Boronate ester compounds and pharmaceutical compositions thereof
The present invention provides novel compounds useful as proteasome inhibitors. The invention also provides pharmaceutical compositions comprising the compounds of the invention and methods of using the compositions in the treatment of various diseases.
Owner:TAKEDA PHARMA CO LTD
Use of Peptide Epoxyketones for Metastasis Suppression
InactiveUS20130035295A1Organic active ingredientsDipeptide ingredientsCancer researchMetastasis Suppression
The invention provides a method of repressing metastasis of a cancer compromising the administration of a peptide epoxyketone proteasome inhibitor. Furthermore, the method can be performed in combination with the administration of one or more additional therapeutics
Owner:ONYX THERAPEUTICS INC
Method of treating CD30 positive lymphomas
InactiveUS7790160B2Good curative effectSuppress NF-kB activationBiocideAntiviralsAntiendomysial antibodiesAnti antibody
Methods for treating lymphomas characterized by expression of CD30 using anti-CD30 antibodies and proteasome inhibitors in combination are disclosed.
Owner:ER SQUIBB & SONS INC
Cyclodextrin-based polymers for therapeutic delivery
InactiveUS20110178287A1Effective treatmentNanomedicinePharmaceutical non-active ingredientsCyclodextrinPolymer
Owner:CERULEAN PHARMA
Synthetic method of proteasome inhibitor bortezomib and analogs
ActiveCN103030656ALower synthesis costEasy to manufactureGroup 3/13 element organic compoundsProteasome inhibitorChemistry
Owner:PEKING UNIV
Synthesis of boronic ester and acid compounds
The invention relates to the synthesis of boronic ester and acid compounds. More particularly, the invention provides improved synthetic processes for the large-scale production of boronic ester and acid compounds, including the peptide boronic acid proteasome inhibitor bortezomib.
Owner:MILLENNIUM PHARMA INC
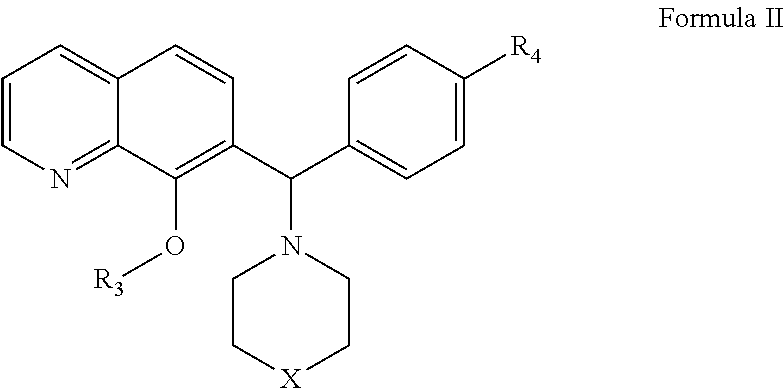
Methods and compositions useful for treating diseases involving bcl-2 family proteins with quinoline derivatives
The present invention relates to compositions and methods for cancer treatment comprising compounds of Formulae I, II, and III. In some aspects, the invention relates to the treatment of B-cell Lymphoma or other hematopoietic cancers. In other aspects, the invention provides methods for treating particular types of hematopoietic cancers, such as, for example, B-cell lymphoma, using a combination of one or more compounds of Formulae I, II, and III. Combination therapy with, for example, 26S proteasome inhibitors, such as, for example, Bortezomib, are also included. In another aspect the present invention relates to autoimmune treatment with compounds of Formulae I, II, and III. In another aspect, this invention relates to methods for identifying compounds, for example, compounds of the BH3 mimic class, that have in vitro properties that predict in vivo efficacy against B-cell lymphoma tumors and other cancers as well as autoimmune disease.
Owner:EUTROPICS PHARMA
Therapeutic combination comprising a cdc7 inhibitor and an Anti-neoplastic agent
ActiveUS20120276093A1Less side effectsStrong synergyHeavy metal active ingredientsBiocideCell Surface ProteinsAntimitotic Agent
The present invention provides a therapeutic combination comprising (a) a compound of formula (I) as set forth in the specification and (b) one or more antineoplastic agents selected from the group consisting of an alkylating or alkylating-like agent, an antimetabolite agent, a topoisomerase I inhibitor, a topoisomerase 11 inhibitor, an inhibitor of a growth factor or of a growth factor receptor, an antimitotic agent, a proteasome inhibitor, an inhibitor of an anti-apoptotic protein and an antibody directed against a cell surface protein, wherein the active ingredients are present in each case in lice form or in the form of a pharmaceutically acceptable salt or any hydrate thereof.
Owner:NERVIANO MEDICAL SERVICES SRL
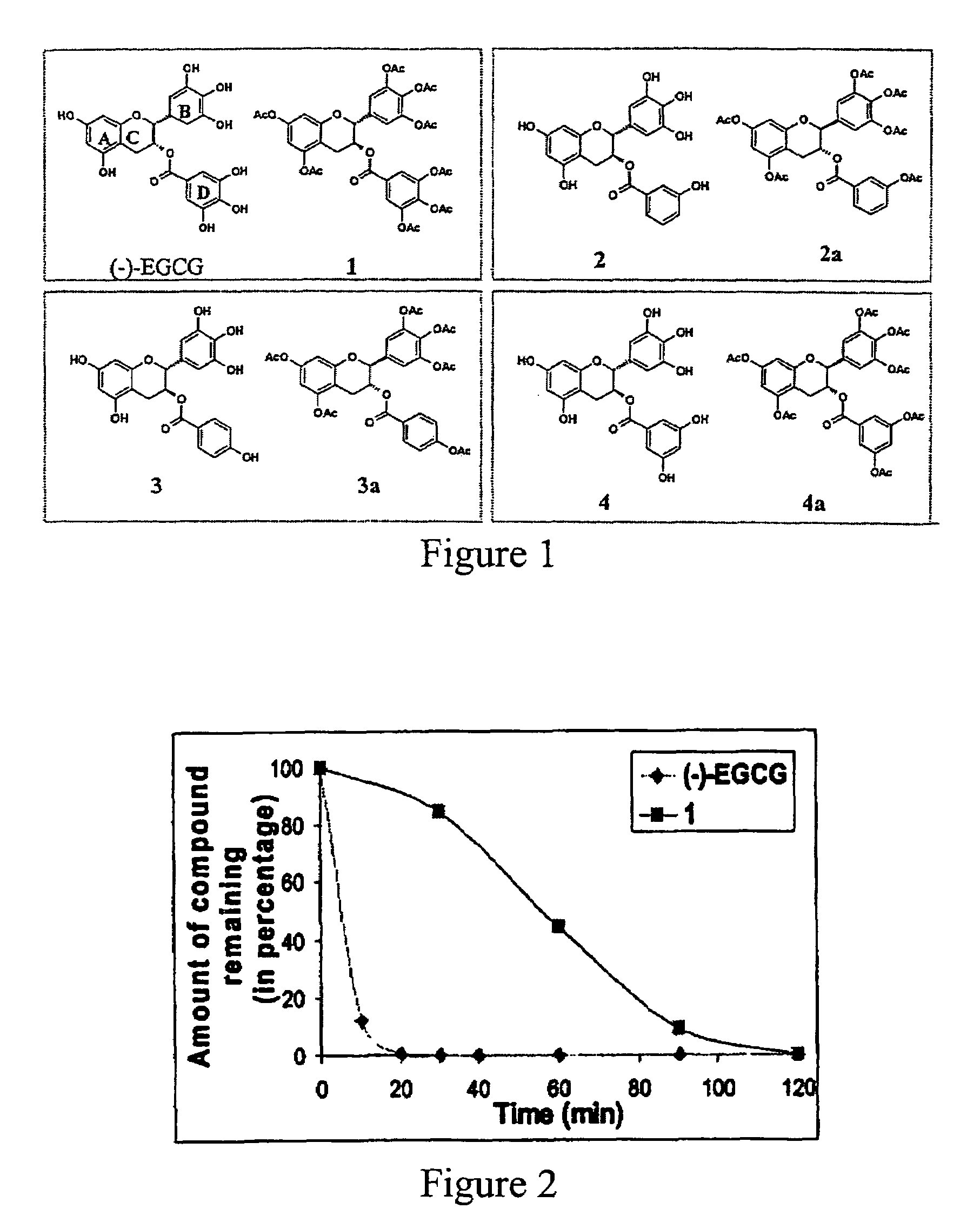
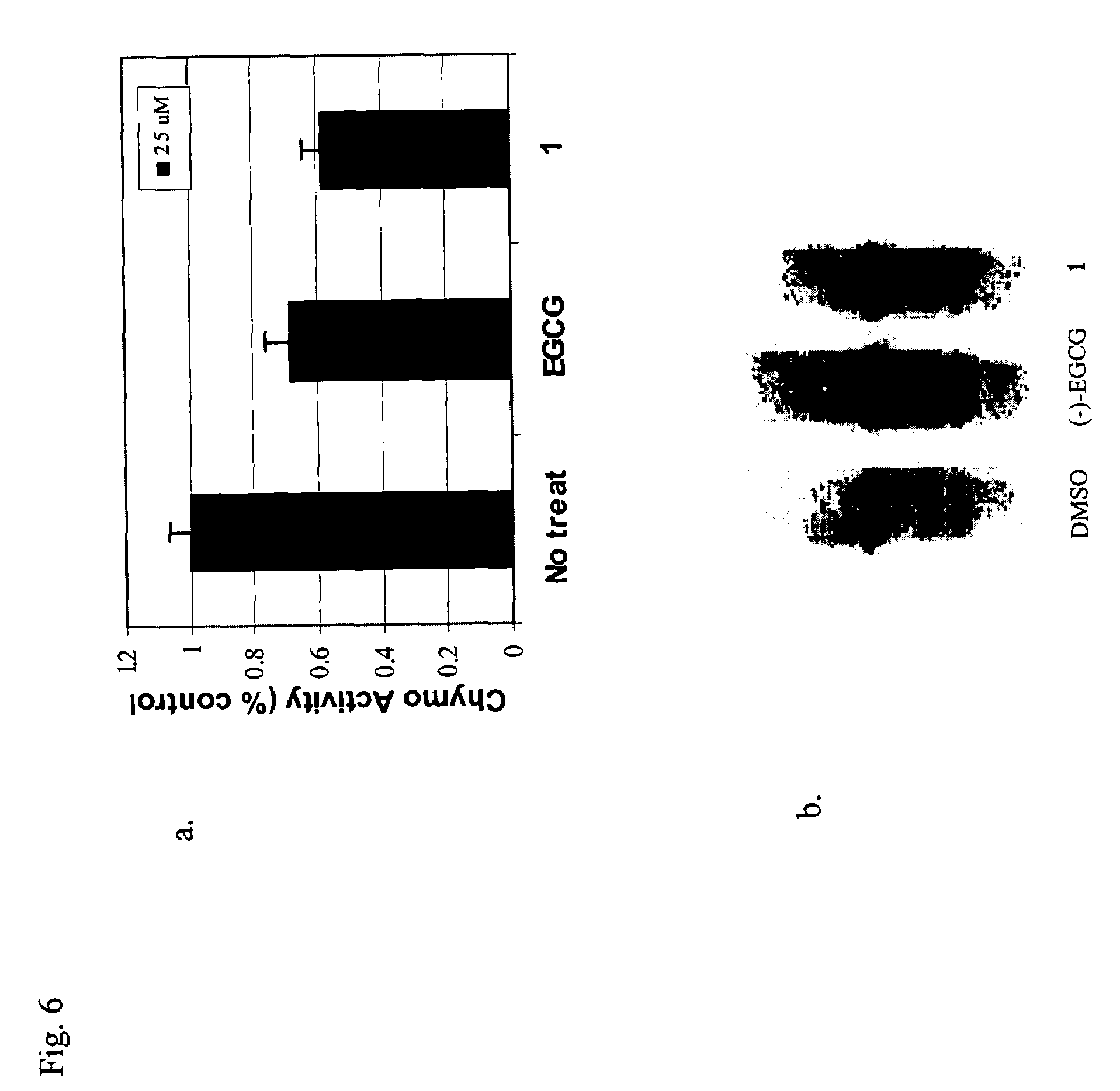
(-)-Epigallocatechin gallate derivatives for inhibiting proteasome
Owner:WAYNE STATE UNIV +3
Novel bis-Benzylidine Piperidone Proteasome Inhibitor with Anticancer Activity
ActiveUS20160106725A1Reduced potencyReducing potencyBiocideOrganic chemistryHuman papillomavirus19S regulatory particle
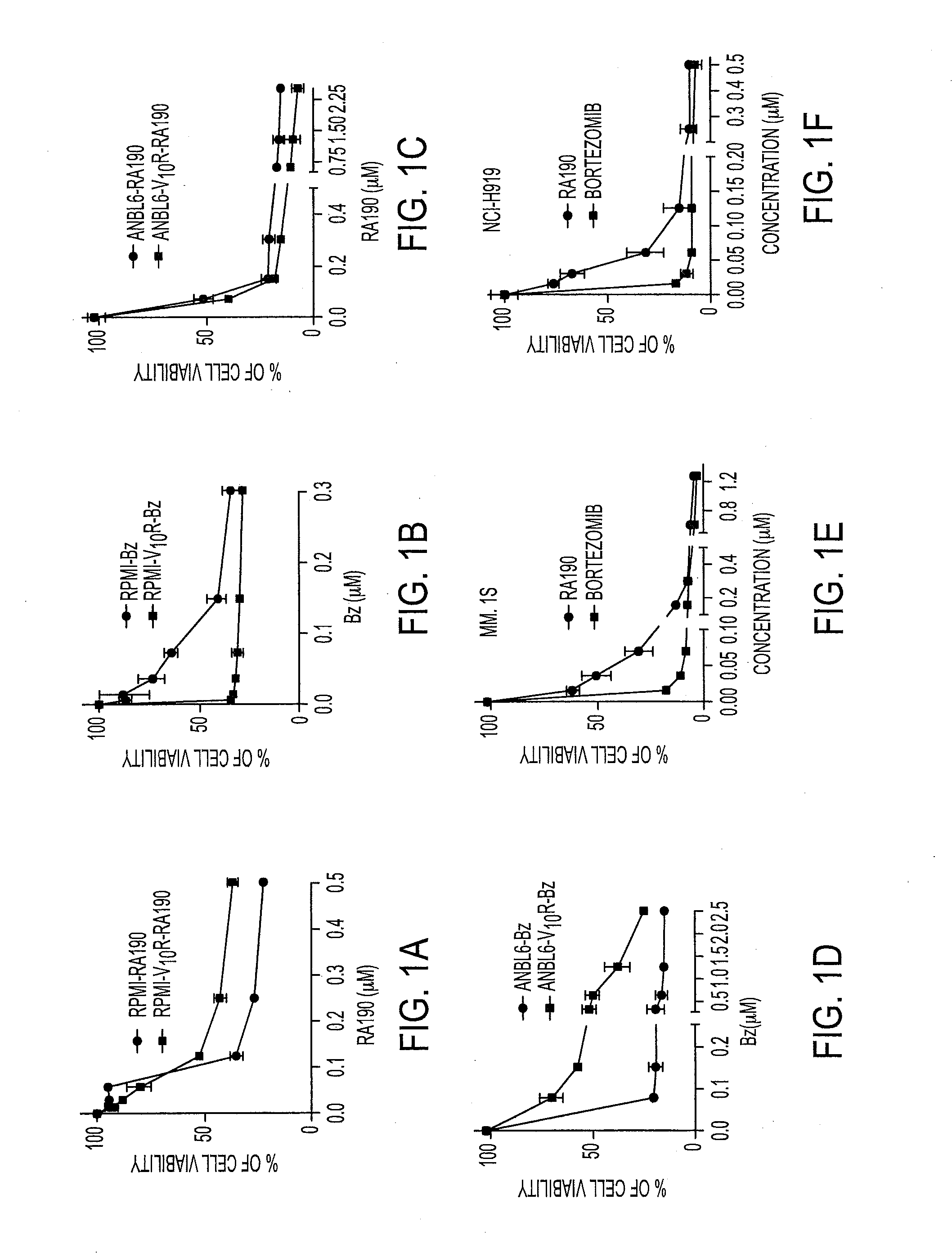
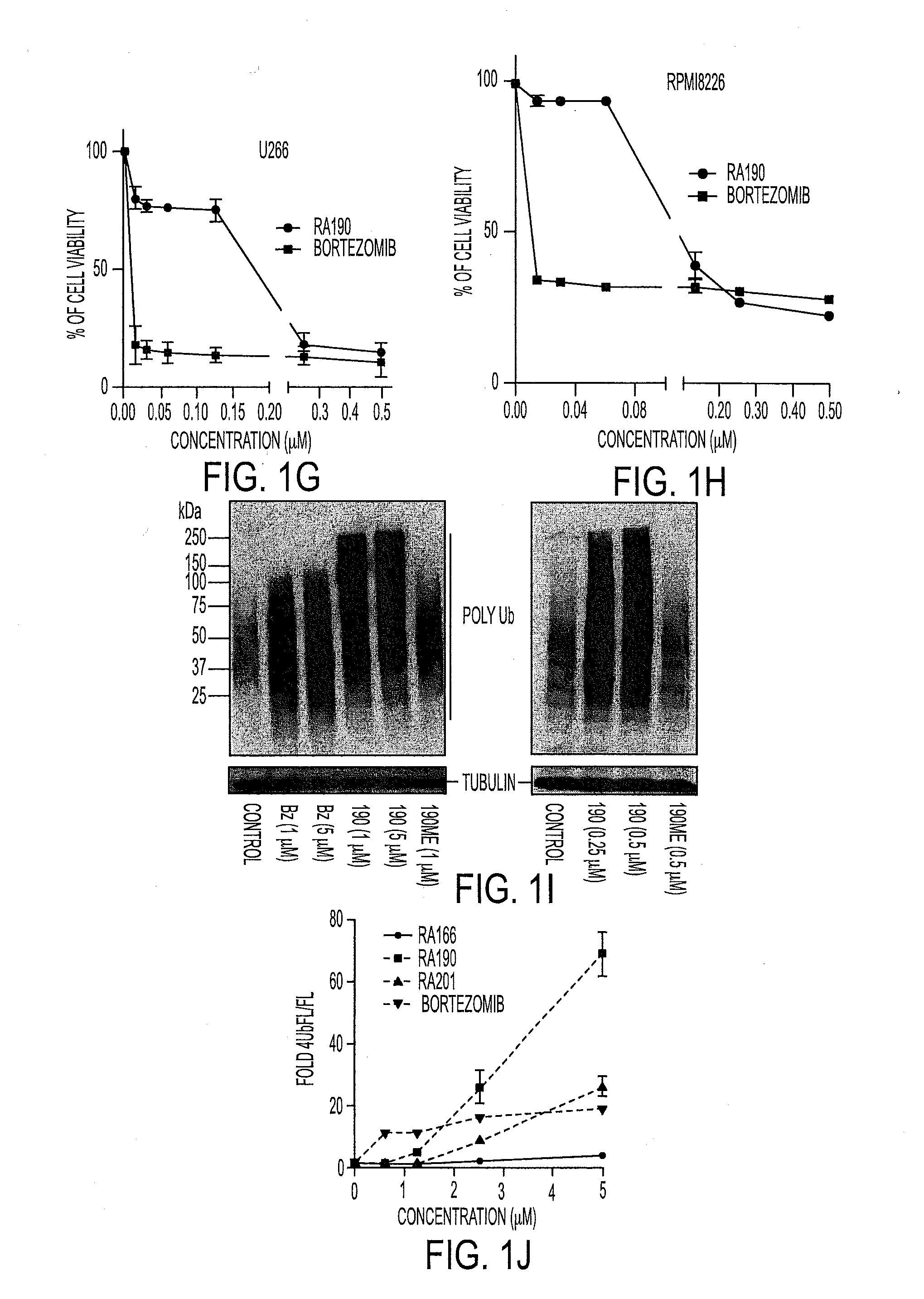
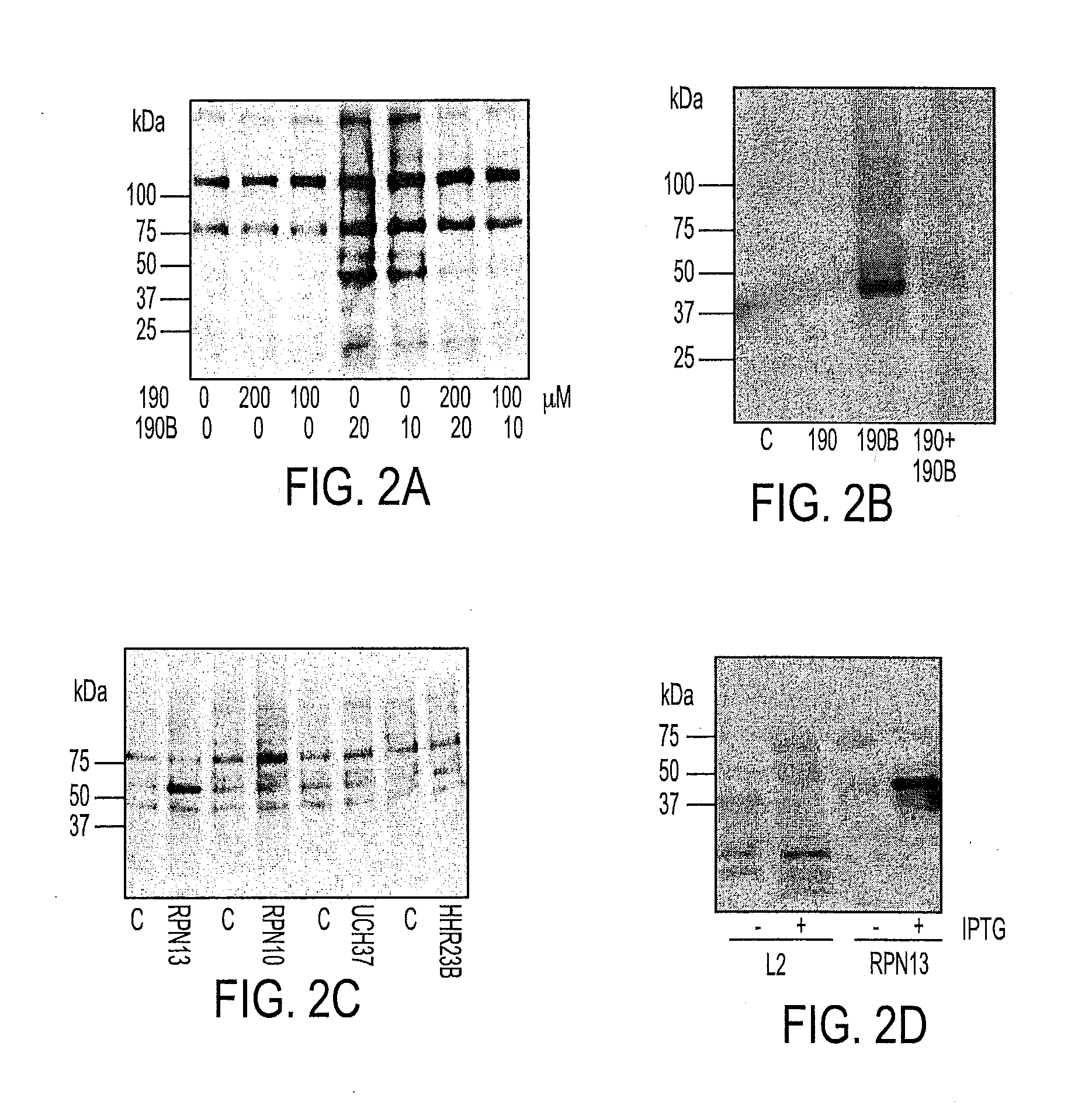
We describe a bis-benzylidine piperidone, RA190, which covalently binds to the ubiquitin receptor RPN13 (ADRM1) in the 19S regulatory particle and inhibits proteasome function, triggering rapid accumulation of polyubiquitinated proteins. Multiple myeloma lines, even those resistant to bortezomib, were sensitive to RA190 via ER stress-related apoptosis. RA190 stabilized targets of human papillomavirus (HPV) E6 oncoprotein, and preferentially killed HPV-transformed cells. After p.o. or i.p. dosing of mice, RA190 distributed to plasma and major organs excepting brain, and potently inhibited proteasome function in skin and muscle. RA190 administration i.p. profoundly reduced growth of multiple myeloma and ovarian cancer xenografts, and oral RA190 treatment retarded HPV+ syngeneic mouse tumor growth, without impacting spontaneous HPV-specific CD8+ T cell responses, suggesting its therapeutic potential. The bis-benzylidine piperidone RA190 is a new orally-available proteasome inhibitor. Multiple myeloma, cervical and ovarian cancers are particularly sensitive to RA190.
Owner:THE JOHN HOPKINS UNIV SCHOOL OF MEDICINE
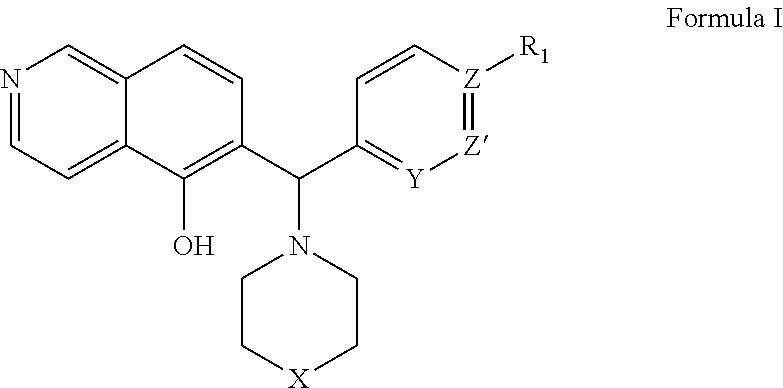
Methods and compositions useful for treating diseases involving bcl-2 family proteins with isoquinoline and quinoline derivatives
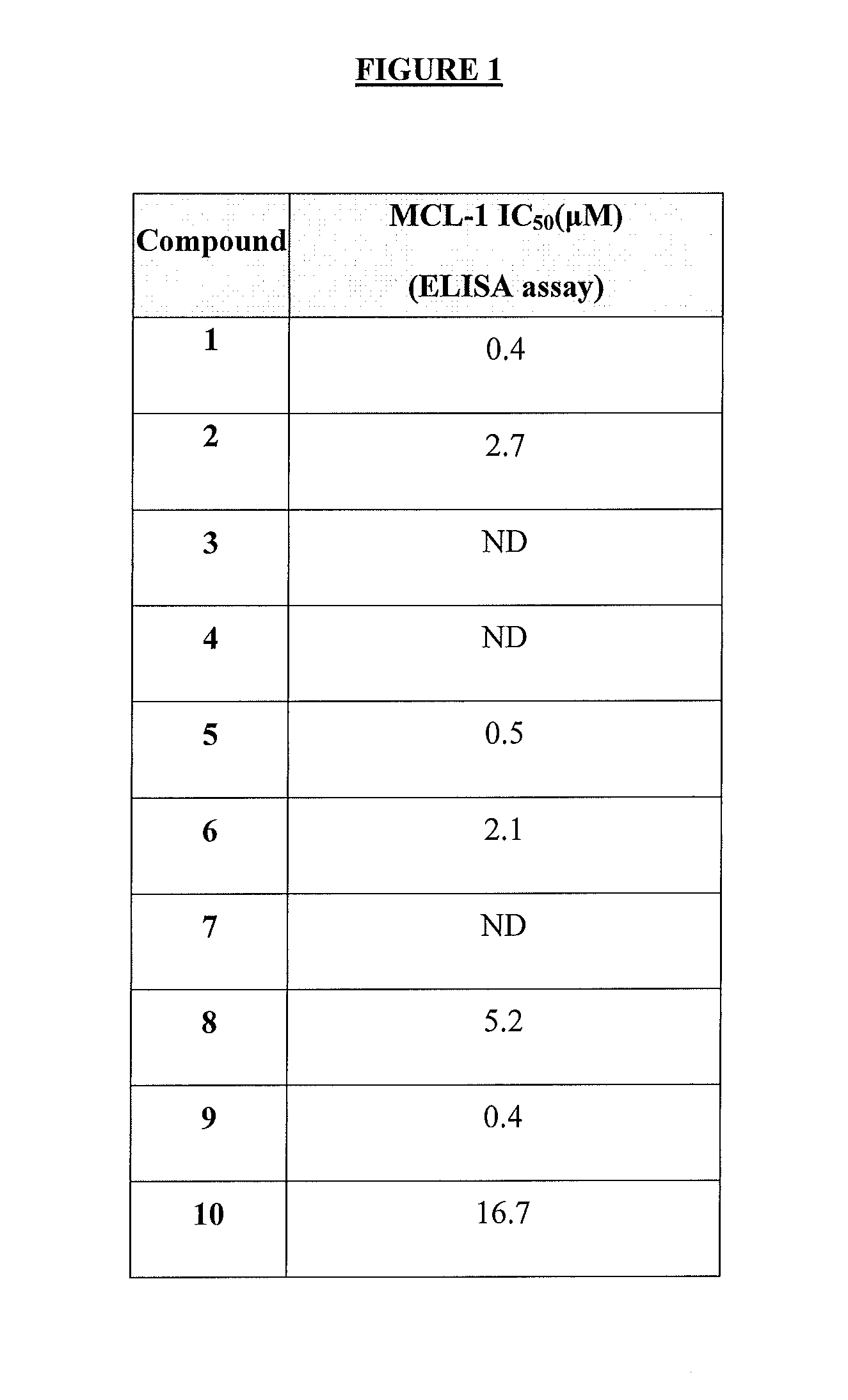
InactiveUS20160038503A1BiocideBoron compound active ingredientsAbnormal tissue growthAutoimmune condition
The present invention relates to a compositions for and methods for cancer treatment, for example, hematopoietic cancers (e.g. B-cell Lymphoma). In other aspects, the invention provides methods for treating particular types of hematopoietic cancers, such as B-cell lymphoma, using a combination of one or more of the disclosed compounds and, for example, 26S proteasome inhibitors, such as, for example, Bortezomib. In another aspect the present invention relates to autoimmune treatment with the disclosed compounds. In another aspect, this invention relates to methods for identifying compounds, for example, compounds of the BH3 mimic class, that have unique in vitro properties that predict in vivo efficacy against B-cell lymphoma tumors and other cancers as well as autoimmune disease.
Owner:EUTROPICS PHARMA
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com