Therapies for acute renal failure
a renal failure and therapy technology, applied in the field of renal disease treatment methods, can solve the problems of acute renal failure, failure of other major organ systems and death, significant permanent and/or progressive damage to renal tissues, etc., to reduce the rate and/or degree of morbidity, prevent, inhibit, delay or alleviate the permanent or progressive loss of renal function, and improve survival rate
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0061]To examine the ability of OP-1 to beneficially affect the course of renal function after an acute ischemic insult to the kidney, the effect of OP-1 on creatinine levels was examined in rats in which ischemic renal damage was induced by temporarily preventing renal blood flow. In this model system, both renal arteries are clamped for 60 minutes. OP-1 is administered into the tail vein of each rat at 0.25 μg / kg at the following four times: 10 minutes before artery clamping, and then again at each of 24, 48, and 72 hours after the ischemic injury. As shown in FIG. 1, the creatinine level of placebo-treated rats rises rapidly after injury, peaks at the 24 hour measurement, and is back to normal by the 18th day post-injury. In contrast, OP-1 treatment is associated with a creatinine level which peaks at the 24 hour measurement to a level only half that of the placebo group, after which it begins to decrease. For each of the four post-injury days on which measurements were made, the...
example 2
[0062]To study the effect of post-injury OP-1 treatment on renal function in another animal model of renal failure, rats with a single kidney were treated with OP-1 2 days after norepinephrine-induced renal damage. In the model system (further described in Conger et al., Kidney Intl. 40:21-28, 1991), each rat has a kidney removed 7 days prior to renal injury, which is induced by intra-arterial infusion of norepinephrine for 90 minutes. Two days after norepinephrine administration, OP-1 is administered intravenously at 0.25 mg / kg.
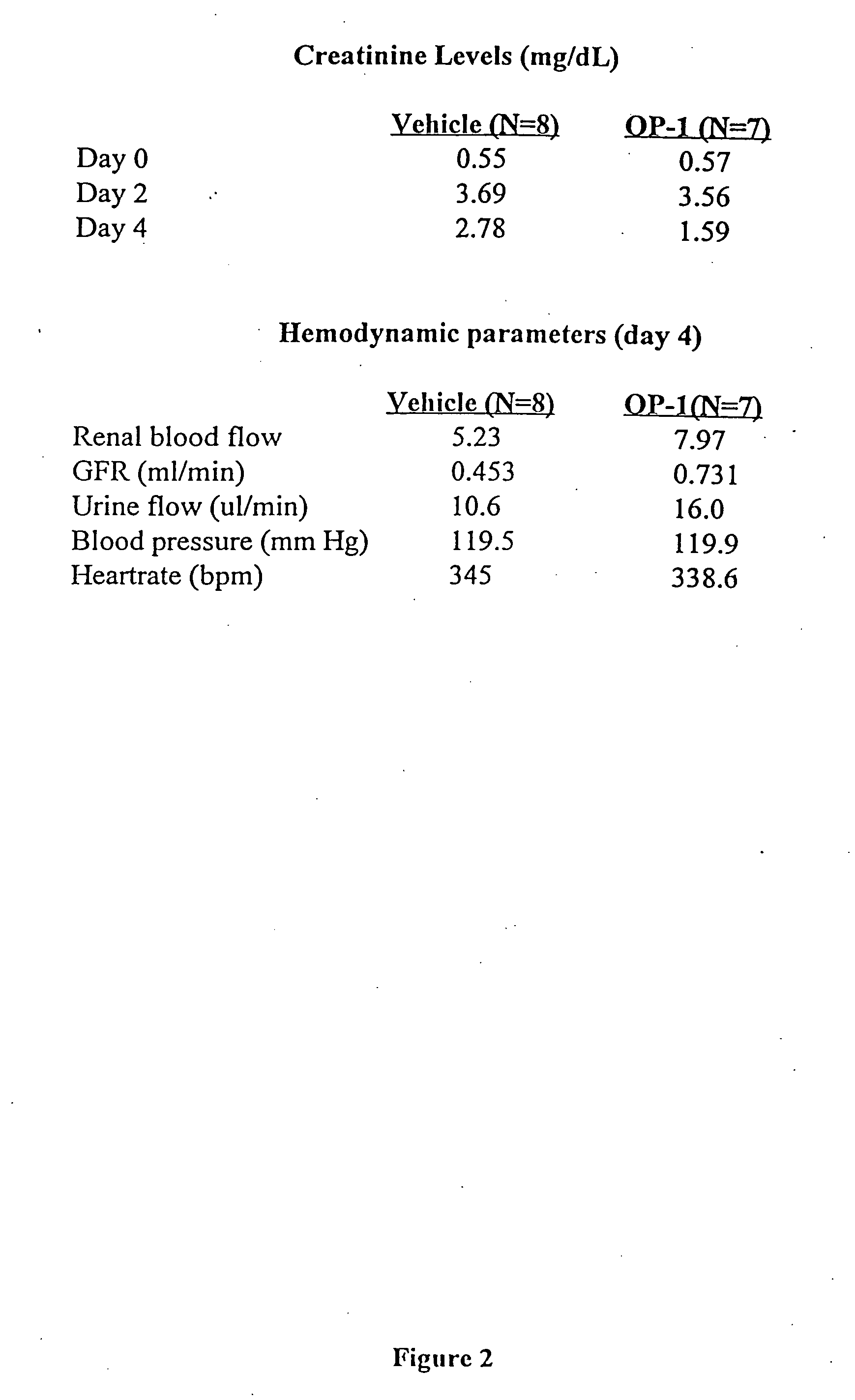
[0063]The upper table of FIG. 2 presents creatinine levels of vehicle- and OP-1 treated rats. On the fourth day after injury, the animals which had been treated with OP-1 on the second day after injury had creatinine levels which are significantly lower than the levels of the vehicle-treated controls. The data presented in the lower table of FIG. 2 shows that OP-1 treatment was also associated with improvements in renal blood flow, GFR, and urine flow as com...
example 3
[0064]Further experiments, using ischemia-reperfusion injury as a model of acute renal failure in rats, were conducted as follows:
[0065]Animal Surgical Procedures and Experimental Protocols.
[0066]Wistar male rats of 200-250 g (Pliva Breeding Laboratory, Zagreb, Croatia) were fasted for 12 h prior to surgery. After intraperitoneal administration of ketamin (20 mg / kg) anesthetic, both renal arteries were dorsally occluded for 60 minutes with microaneurysm clamps (Roboz). Vehicle buffer or OP-1 containing 20 mM sodium acetate buffer (500 μl) were administered via the tail vein. All animals were subjected to intraperitoneal administration of 1-3 ml of pre-warmed (37° C.) saline (0.9% NaCl) to compensate for any fluid loss during the surgery. Experiments were blind, and rats were terminated at different time intervals ranging from 30 min to 18 days. Blood samples (0.5 ml) were obtained from the orbital plexus at 0 h, 24 h, 48 h, and 72 h and, in some cases, at 30 min, 2 h, 8 h, and 96 h,...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weights | aaaaa | aaaaa |
| molecular weights | aaaaa | aaaaa |
| molecular weights | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com