Decitabine freeze-dried powder injection
A technology of freeze-dried powder injection and decitabine, which is applied in the field of decitabine freeze-dried powder injection, can solve the problems of poor stability of decitabine, decreased drug efficacy, and no improvement of defects, etc., to achieve thorough water drying, Avoid the reduction of curative effect and the effect of uniform and stable quality
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
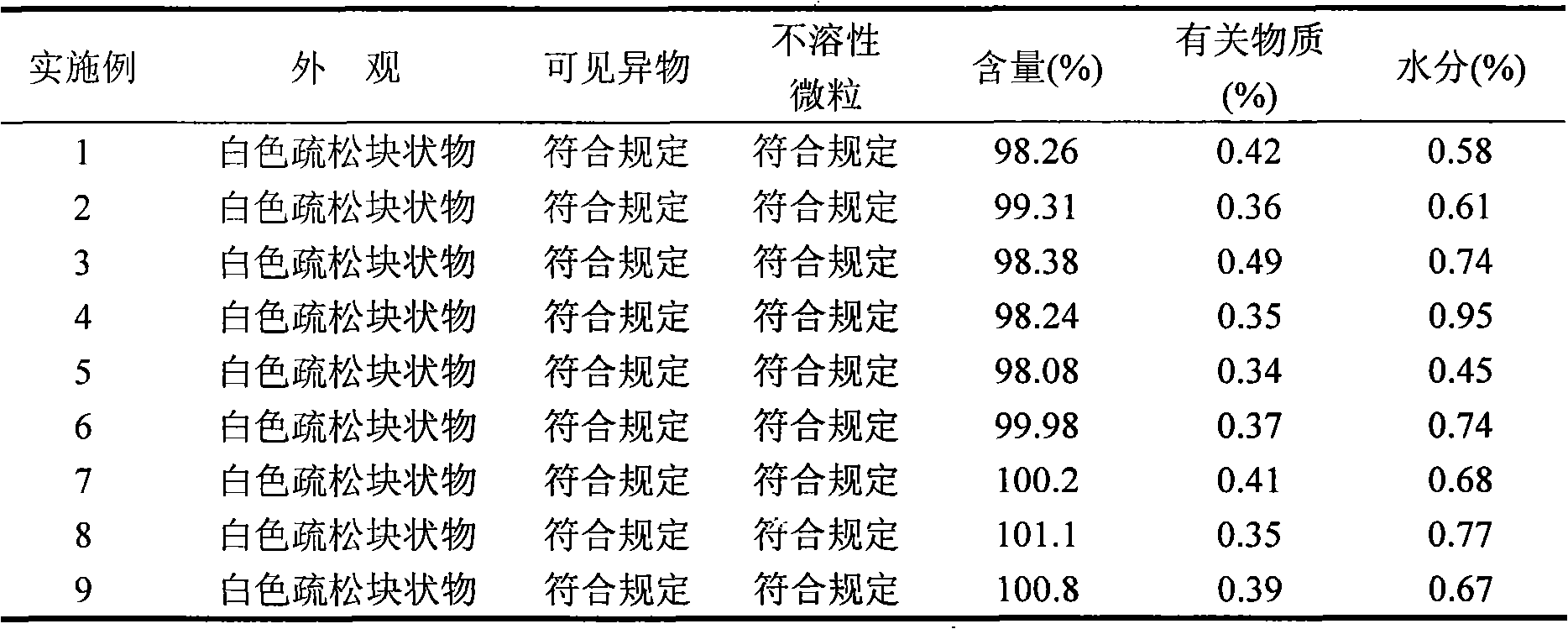
Examples
Embodiment 1
[0027] prescription
[0028] Decitabine 50g
[0029] Mannitol 200g
[0030] Potassium dihydrogen phosphate 68g
[0032] tert-butanol 3000ml
[0033] Add water for injection to 10000ml
[0034]
[0035] A total of 1000 bottles were made
[0036] Measure tert-butanol into a container, add water for injection, potassium dihydrogen phosphate, sodium hydroxide and mannitol, stir and mix evenly, cool the mixed solvent to 6°C and keep it warm, then add decitabine, stir to make it Dissolve and mix evenly. After passing the intermediate inspection, send it to a sterile room with a peristaltic pump and filter it through a 0.22 μm microporous membrane until it becomes clear. Fill it into a vial, partially plug it with a butyl rubber stopper, and put it on a plate; Put the sample to be freeze-dried in the freeze-drying box, close the door, turn on the machine, open the circulating pump, compressor and plate cooling valve, u...
Embodiment 2
[0038] prescription
[0039] Decitabine 50g
[0040] Mannitol 100g
[0041] Potassium dihydrogen phosphate 68g
[0042] Sodium hydroxide 11.6g
[0043] tert-butanol 4000ml
[0044] Add water for injection to 20000ml
[0045]
[0046] A total of 1000 bottles were made
[0047] Measure tert-butanol into a container, add water for injection, potassium dihydrogen phosphate, sodium hydroxide and mannitol, stir and mix evenly, cool the mixed solvent to 8°C and keep it warm, then add decitabine, stir to make it Dissolve and mix evenly. After passing the intermediate inspection, send it to a sterile room with a peristaltic pump and filter it through a 0.22 μm microporous membrane until it becomes clear. Fill it into a vial, partially plug it with a butyl rubber stopper, and put it on a plate; Put the sample to be freeze-dried in the freeze-drying box, close the door, turn on the machine, open the circulating pump, compressor and plate cooling valve, u...
Embodiment 3
[0049] prescription
[0050] Decitabine 50g
[0051] Potassium dihydrogen phosphate 68g
[0052] Sodium hydroxide 11.6g
[0053] tert-butanol 5000ml
[0054] Add water for injection to 10000ml
[0055]
[0056] A total of 1000 bottles were made
[0057]Measure tert-butanol into a container, add water for injection, potassium dihydrogen phosphate and sodium hydroxide, stir and mix evenly, cool the mixed solvent to 6°C and keep it warm, then add decitabine, stir to dissolve and mix Uniformity, after passing the intermediate inspection, send it to the sterile room with a peristaltic pump and filter it through a 0.22 μm microporous membrane until it becomes clear, fill it in a vial, partially plug it with a butyl rubber stopper, and put it into a plate; put it into a plate Put the sample to be freeze-dried in the freeze-drying box, close the box door, turn on the machine, turn on the circulation pump, compressor and plate cooling valve, use heat tr...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com