Decellularized Biomaterial from Non-Mammalian Tissue
a biomaterial and non-mammalian technology, applied in the direction of drugs, peptide/protein ingredients, prosthesis, etc., can solve the problems of life-threatening, the use of conventional tissue scaffold products is not without drawbacks, and the biomechanical function of damaged tissue is restored. a true challenge, the effect of preserving structural and functional integrity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Processing Axolotl Dermis
[0098]Axolotl dermis samples can be decellularized by preparing excised samples from healthy or healing axolotl dermal tissue and then subjecting the samples to hypo / hyperosmotic soaks for cell lysis, solvent dehydration, and oven drying. Specific processing of these grafts includes storage in 15-26% NaCl, multiple hypo / hyperosmotic soaks (utilizing NaCl solutions and water), and then solvent dehydration using ethanol, and then evaporation of the solvent either with air drying or oven drying at 37° C.
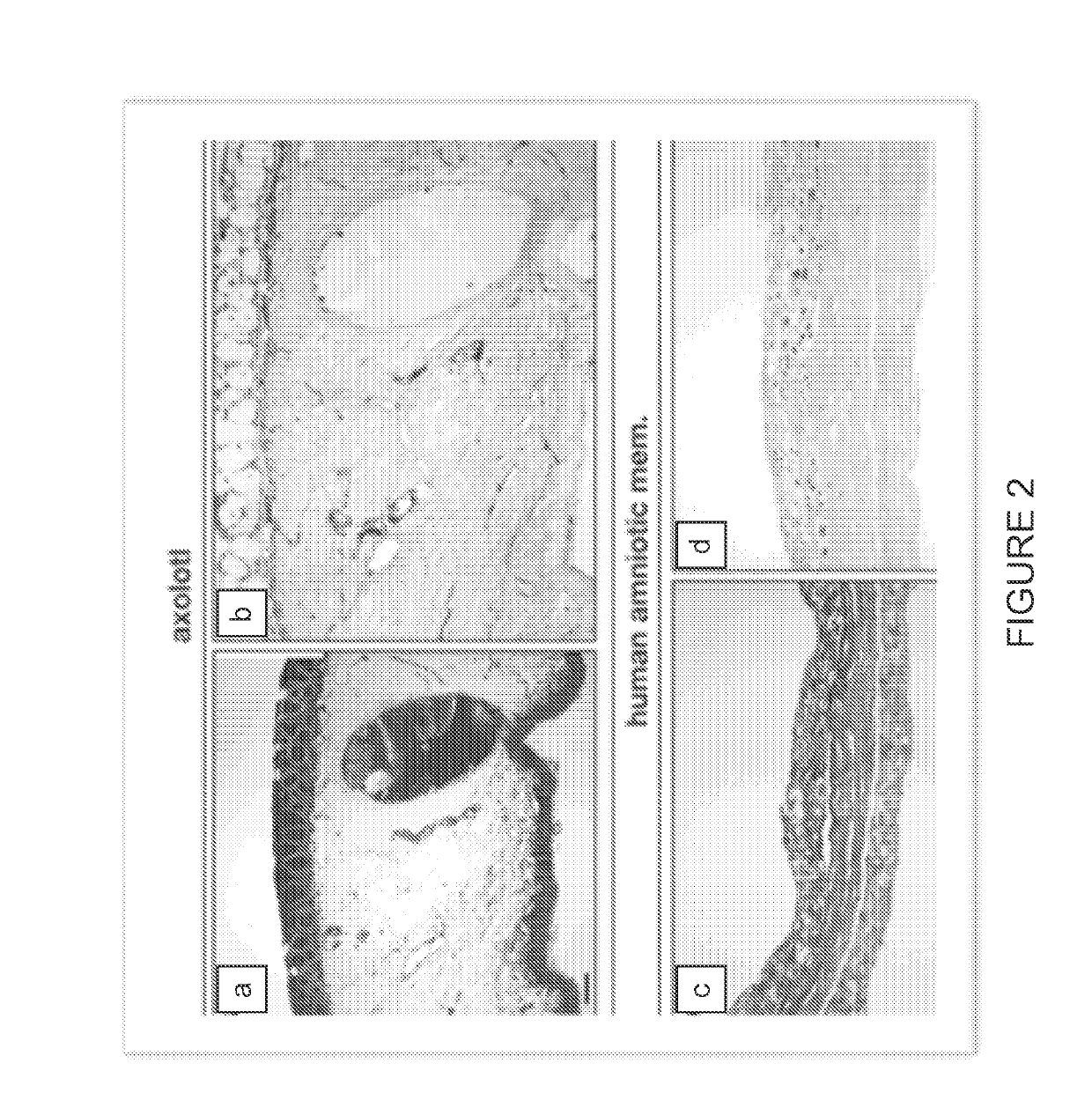
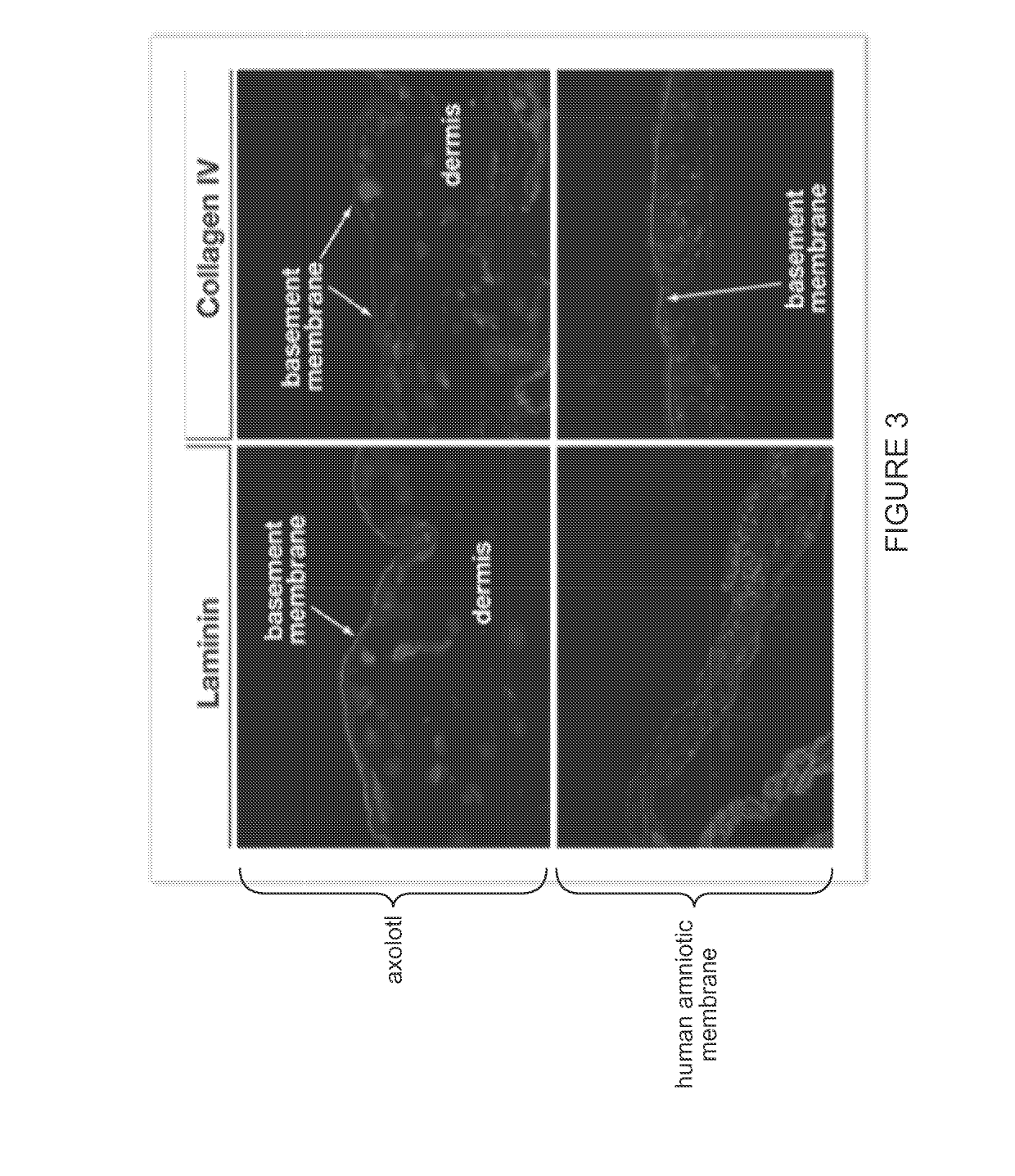
[0099]Histological examination of native axolotl dermal tissue was performed to identify the presence of the notable ECM elements, such as the basement membrane. See FIG. 1 and FIG. 4. Comparative histological and immunohistochemical analysis of native axolotl dermal tissue and human amniotic membrane was performed to compare the ECM structure and constituents, and to assess relative concentration and distribution of critical constituents. See FIG. 2, FIG. 3, an...
example 2
Splitting and Lamination of Acellular Dehydrated Axolotl Dermis
[0100]Decellularized dehydrated axolotl dermis can be split, via a mechanical splitter, to isolate heterogeneous matrix into homogenous sections. Isolated sections of desired thickness then can be rehydrated and lyophilized to obtain multilayered laminate structures of desired orientation with facial surface features. More specifically, dual-sided basement membrane structure, with interior open porous matrix obtained from the reticular dermis region of the dermal matrix, can be constructed to obtain desired facial surface properties. Alternatively, isolated native section can be used in native form for desired clinical outcome. For example, open porous homogenous matrix of the reticular dermis can be used to obtain augmentation of soft tissue structures.
[0101]A laminated custom construct with sulfated gags on both facial surface and collagen IV and laminin could be obtained for desirable dual-surface, anti-adhesion and a...
example 3
Preparation of Solubilized Acellular Dehydrated Axolotl Dermis, Pericardium, Fascia Lata, Periosteum, Peritoneum, or Dura Mater
[0102]Decellularized dehydrated axolotl native or isolated section of acellular urodele connectivue tissye matrix can be prepared by sectioning decellularized soft tissue structures into 1 cm2 sections and homogenizing the sections in a Warring blender (˜100 grams of tissue) in aqueous 1M glacier acetic acid for 30-60 seconds. Preparation of sponge can be obtained by the addition of varying volumes of water followed then neutralization and lypoholization of the slurry in a mold of desired geometric shape. The resultant porosity will correlate to the volume of water added to the matrix. Additionally, a selected range of bioactive extracts can be added to the slurry prior to neutralization, including particulated or small protein constituents extracted from digested human or urodele mineralized and nonmineralized connective tissues, such as demineralized bone ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| dry weight | aaaaa | aaaaa |
| size | aaaaa | aaaaa |
| particle size distribution | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com