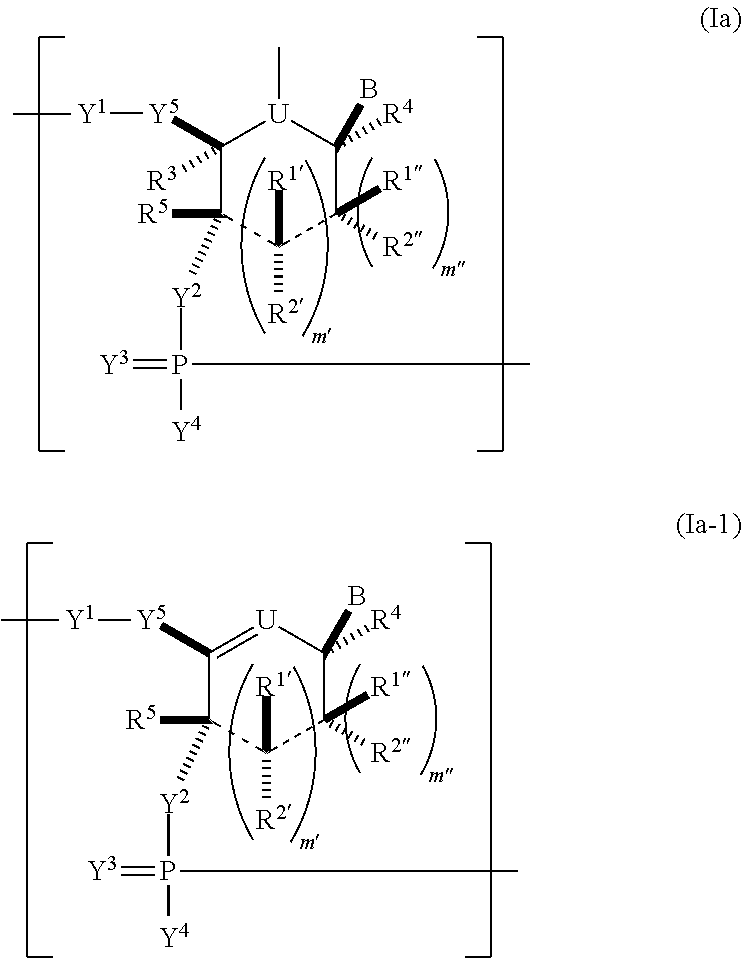
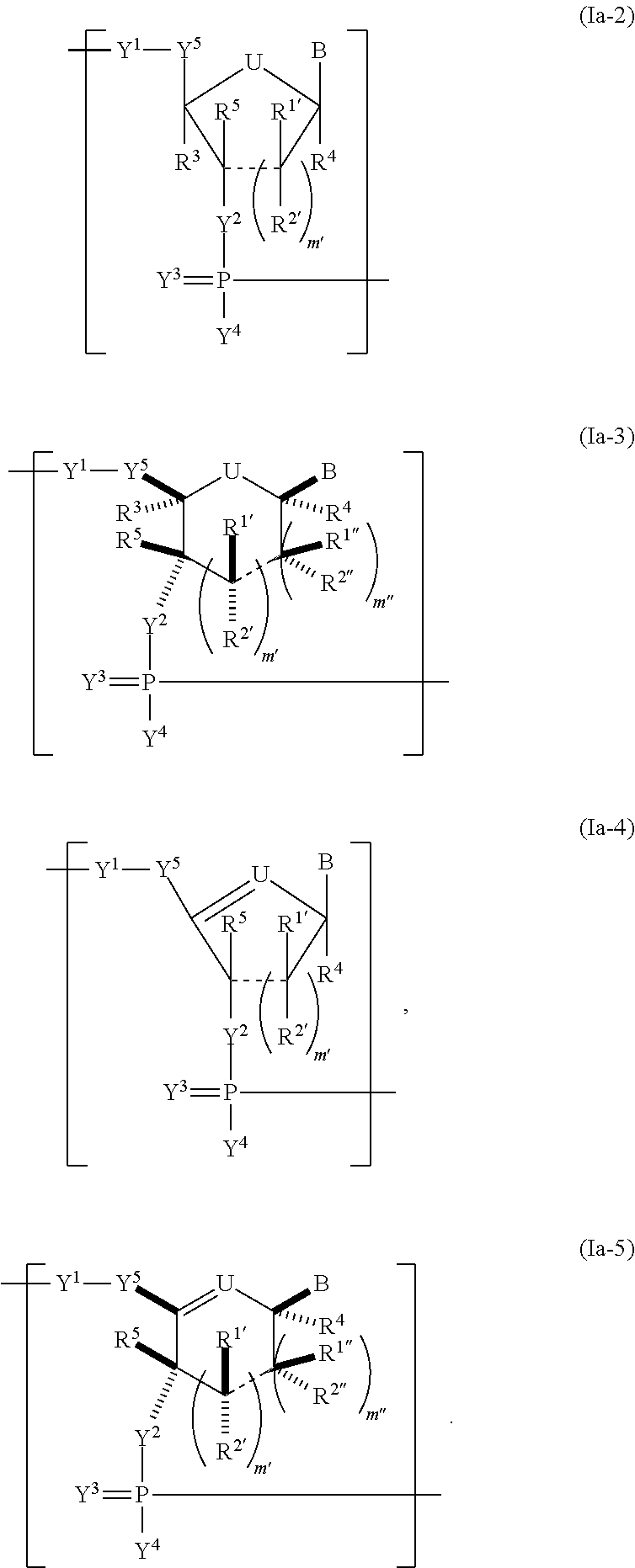
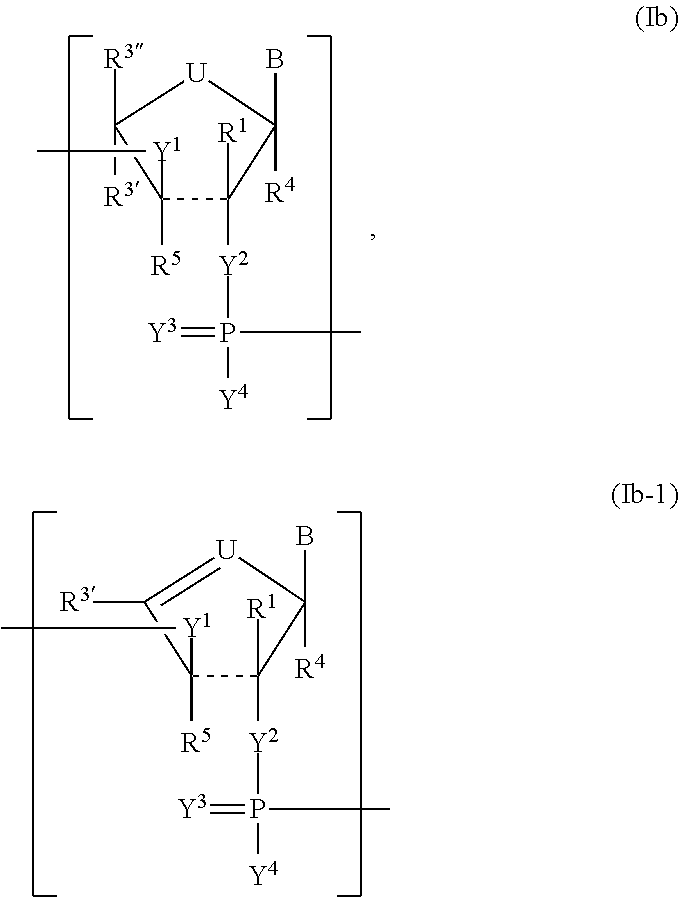
Modified polynucleotides encoding cd28 molecule
a technology of cd28 and polynucleotides, which is applied in the direction of peptide/protein ingredients, dna/rna fragmentation, peptide delivery, etc., can solve the problems of affecting the dna expression of host cells, and each step represents an opportunity for error and damage to the cell, and is known to be difficult to obtain dna expression in cells
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Modified mRNA Production
[1140]Modified mRNAs (mmRNA) according to the invention may be made using standard laboratory methods and materials. The open reading frame (ORF) of the gene of interest may be flanked by a 5′ untranslated region (UTR) which may contain a strong Kozak translational initiation signal and / or an alpha-globin 3′ UTR which may include an oligo(dT) sequence for templated addition of a poly-A tail. The modified mRNAs may be modified to reduce the cellular innate immune response. The modifications to reduce the cellular response may include pseudouridine (ψ) and 5-methyl-cytidine (5meC, 5mc or m5C). (See, Kariko K et al. Immunity 23:165-75 (2005), Kariko K et al. Mol Ther 16:1833-40 (2008), Anderson B R et al. NAR (2010); each of which are herein incorporated by reference in their entireties).
[1141]The ORF may also include various upstream or downstream additions (such as, but not limited to, β-globin, tags, etc.) may be ordered from an optimization service such as, ...
example 2
PCR for cDNA Production
[1158]PCR procedures for the preparation of cDNA are performed using 2×KAPA HIFI™ HotStart ReadyMix by Kapa Biosystems (Woburn, Mass.). This system includes 2×KAPA ReadyMix12.5 μl; Forward Primer (10 uM) 0.75 μl; Reverse Primer (10 uM) 0.75 μl; Template cDNA 100 ng; and dH20 diluted to 25.0 μl. The reaction conditions are at 95° C. for 5 min. and 25 cycles of 98° C. for 20 sec, then 58° C. for 15 sec, then 72° C. for 45 sec, then 72° C. for 5 min. then 4° C. to termination.
[1159]The reverse primer of the instant invention incorporates a poly-T120 for a poly-A120 in the mRNA. Other reverse primers with longer or shorter poly(T) tracts can be used to adjust the length of the poly(A) tail in the mRNA.
[1160]The reaction is cleaned up using Invitrogen's PURELINK™ PCR Micro Kit (Carlsbad, Calif.) per manufacturer's instructions (up to 5 μg). Larger reactions will require a cleanup using a product with a larger capacity. Following the cleanup, the cDNA is quantified ...
example 3
In Vitro Transcription (IVT)
[1161]The in vitro transcription reaction generates mRNA containing modified nucleotides or modified RNA. The input nucleotide triphosphate (NTP) mix is made in-house using natural and un-natural NTPs.
[1162]A typical in vitro transcription reaction includes the following:
1Template cDNA1.0μg210x transcription buffer (400 mM Tris-2.0μlHCl pH 8.0, 190 mM MgCl2, 50 mM DTT,10 mM Spermidine)3Custom NTPs (25 mM each)7.2μl4RNase Inhibitor20U5T7 RNA polymerase3000U6dH20Up to 20.0 μl. and7Incubation at 37° C. for 3 hr-5 hrs.
[1163]The crude IVT mix may be stored at 4° C. overnight for cleanup the next day. 1 U of RNase-free DNase is then used to digest the original template. After 15 minutes of incubation at 37° C., the mRNA is purified using Ambion's MEGACLEAR™ Kit (Austin, Tex.) following the manufacturer's instructions. This kit can purify up to 500 μg of RNA. Following the cleanup, the RNA is quantified using the NanoDrop and analyzed by agarose gel electrophore...
PUM
| Property | Measurement | Unit |
|---|---|---|
| compositions | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com