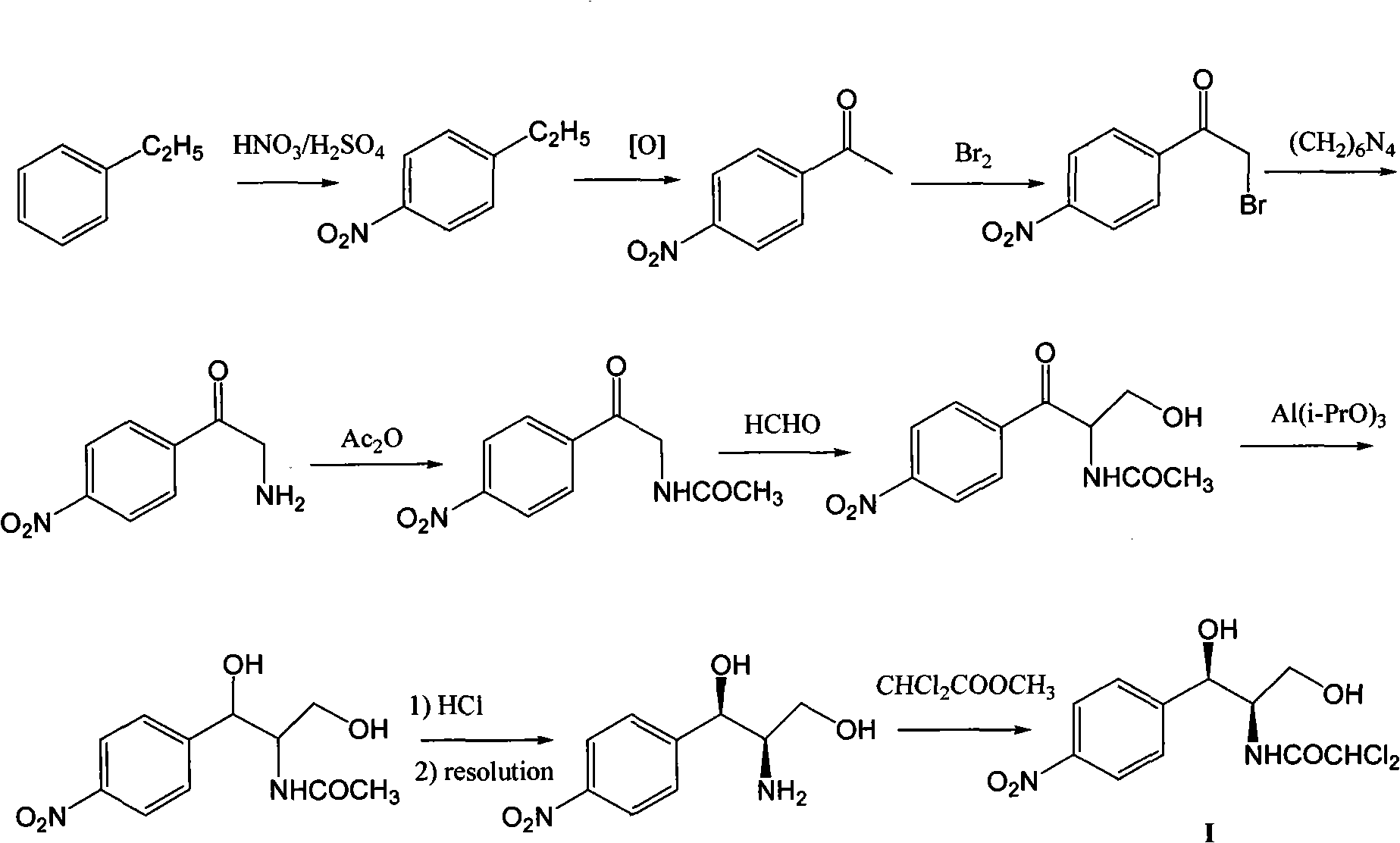
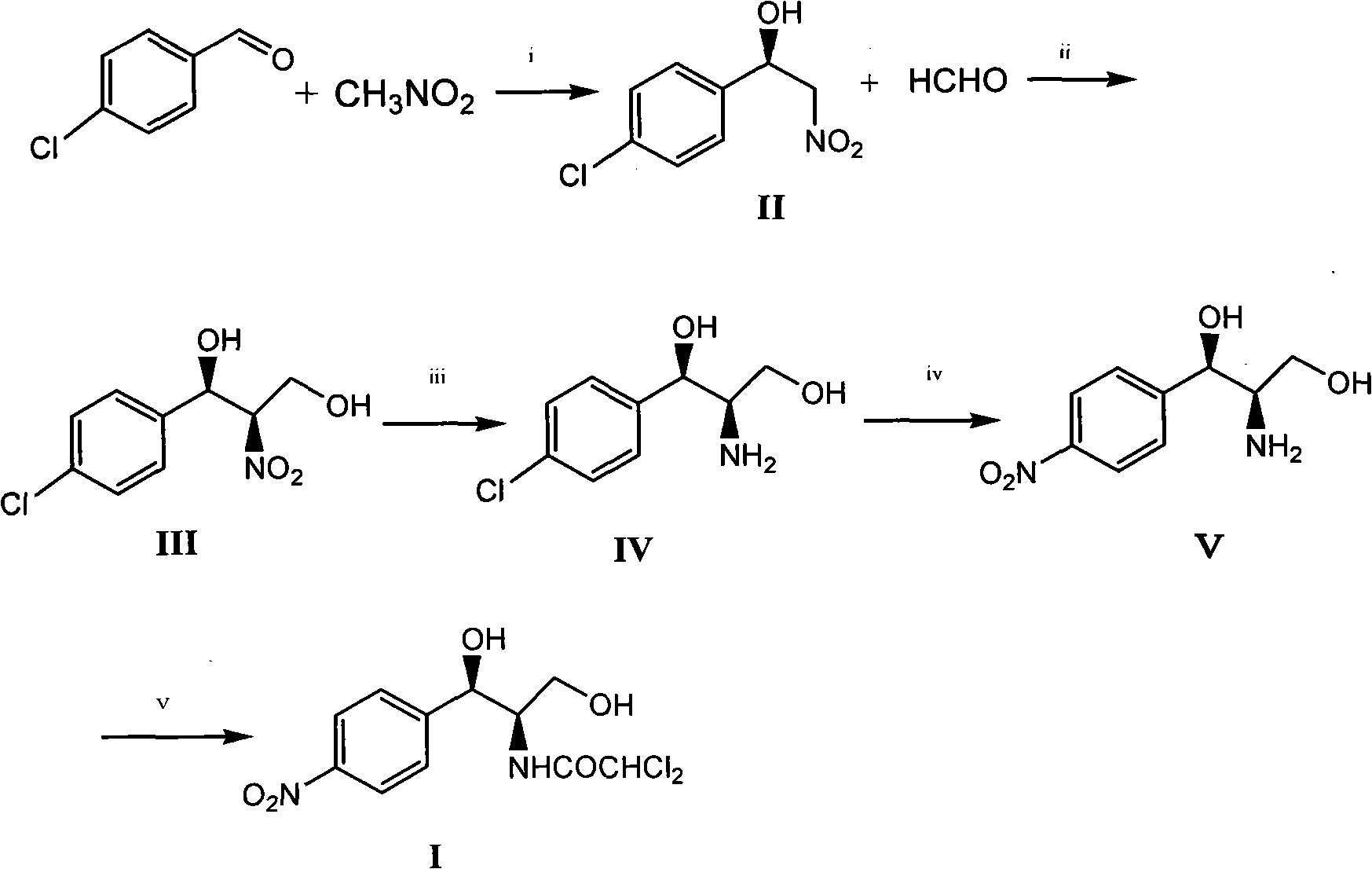
Method for synthesizing chloramphenicol from nitromethane
A technology of chloramphenicol and nitro, which is applied in the field of compound preparation, can solve the problems of production cost, increase of three wastes, and long synthesis route of chloramphenicol, and achieve the effect of cheap raw materials, easy availability of raw materials, and high total yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0035] 1 of the preparation of embodiment 1 (R)-2-nitro-1-(4-chlorophenyl) ethanol
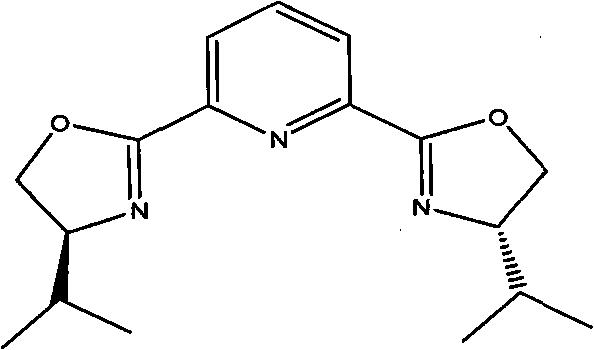
[0036] 1.8 g copper trifluoromethanesulfonate Cu(OTf) 2 (0.50mmol), 2.4 grams of ligand {2,6-bis[(S)-4-isopropyl-1-phenyl-4,5-dihydro-1H-2-imidazolyl]pyridine} (5.2mmol ) and 40 milliliters of 1,4-dioxane were added in a 250 milliliter single-necked flask, and the nitrogen flow was kept constant after replacing the air in the inside with nitrogen gas. After magnetic stirring for 2 hours, 7.0 grams of p-chlorobenzaldehyde ( 50mmol), 30.6 grams of nitromethane (500mmol) and N-methylmorpholine (0.54 milliliters, 5.0mmol), the reaction solution was stirred in ice-bath cooling for 24 hours, and after thin plate chromatography detected no raw material p-chlorobenzaldehyde spots, Then the volatile solvent is removed by distillation under reduced pressure, the catalyst is removed by silica gel filtration, and the filtrate is concentrated to obtain 9.0 grams of product, the yield is 90%, and the HPLC ...
Embodiment 2
[0037] The 2 of the preparation of embodiment 2 (R)-2-nitro-1-(4-chlorophenyl)ethanol
[0038] In a 100 ml single-necked flask, add 0.5 g of 1-[2-(4S)-4-R-4,5-dihydro-2-oxazoline-ethyl]piperidine, 0.09 g of trifluoromethanesulfonate Copper (CuOTf) 2, 40 milliliters of anhydrous dimethyl sulfoxide, keep the nitrogen flow constant after replacing the air inside with nitrogen, cool with ice bath after magnetic stirring for 3 hours, add 7.0 grams of p-chlorobenzaldehyde (50mmol) successively , 30.6 grams of nitromethane (500mmol) and N-methylmorpholine (0.54 milliliters, 5.0mmol), the reaction solution was stirred in ice-bath cooling for 24 hours, after thin-plate chromatography detected no raw material p-chlorobenzaldehyde spots, then subtracted The volatile solvent was removed by distillation under pressure, the catalyst was removed by silica gel filtration, and the filtrate was concentrated to obtain 9.1 grams of product, with a yield of 91%, and the HPLC measurement e.e value ...
Embodiment 3
[0039] Example 3 Preparation of (1R, 2R)-2-nitro-1-(4-chlorophenyl)-1,3-propanediol 1
[0040] Add 40 ml of 1,4-dioxane into a 250 ml single-necked flask, replace the air inside with nitrogen to keep the nitrogen flow constant, stir magnetically and add 1.2 g of paraformaldehyde (40 mmol), 8.0 g of (R)-2 -Nitro-1-(4-chlorophenyl)ethanol (40mmol) and N-methylmorpholine (0.44 milliliters, 4mmol), the reaction solution was stirred in ice-bath cooling for 24 hours, and no raw material (R )-2-nitro-1-(4-chlorophenyl)ethanol spots, the volatile solvent was distilled off under reduced pressure, purified by silica gel filtration, and the filtrate was concentrated to obtain 4.0 g of the product, with a yield of 86%. 1 H NMR (acetone-d 6 )δ: 3.71(s, 1H), 3.85(m, 1H), 4.12(m, 1H), 4.52(m, 1H), 4.79(m, 1H), 5.01(d, J=8.7Hz, 1H), 7.15-7.26 (m, 4H).
PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
| specific rotation | aaaaa | aaaaa |
| purity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com