Microorganisms and methods for the biosynthesis of (2-hydroxy-3methyl-4-oxobutoxy) phosphonate
a biosynthesis method and phosphonate technology, applied in the field of biosynthesis processes, can solve the problems of cost-effective methods for generating renewable pta that have not yet been developed, and the biosynthesis pathway of pta is severely limited by the properties of known enzymes
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example i
Exemplary Pathway for Producing (2-Hydroxy-3-methyl-4-oxobutoxy)phosphonate
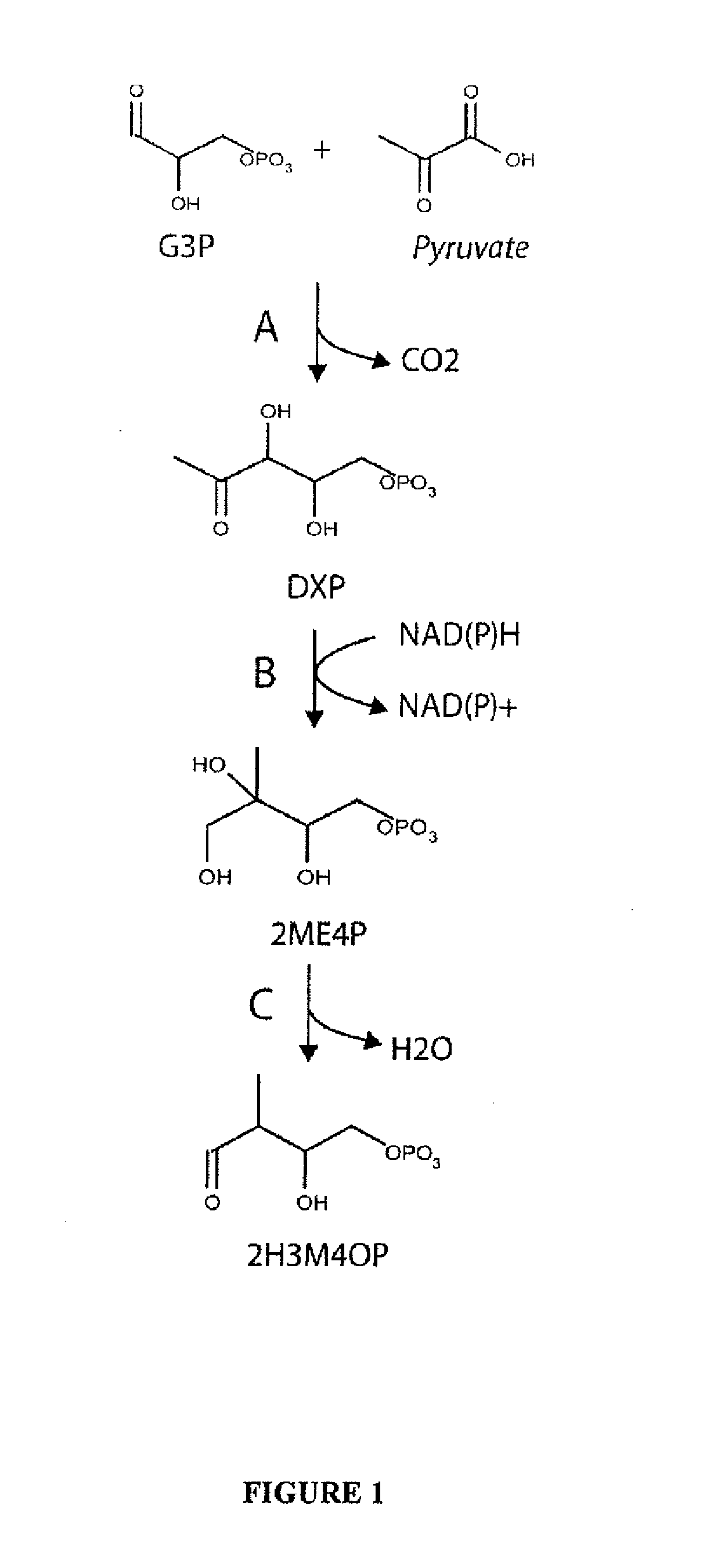
[0103]This example describes an exemplary pathway for producing the terephthalic acid (PTA) precursor (2-hydroxy-3-methyl-4-oxobutoxy)phosphonate (2H3M4OP).
[0104]The precursor to the p-toluate and PTA pathways is 2H3M4OP. This chemical can be derived from central metabolites glyceraldehyde-3-phosphate (G3P) and pyruvate in three enzymatic steps as shown in FIG. 1. The first two steps are native to E. coli and other organisms that utilize the methylerythritol phosphate (non-mevalonate) pathway for isoprenoid biosynthesis. Pyruvate and G3P are first condensed to form 1-deoxy-D-xylulose 5-phosphate (DXP) by DXP synthase. Subsequent reduction and rearrangement of the carbon backbone is catalyzed by DXP reductoisomerase. Finally, a novel diol dehydratase transforms 2-C-methyl-D-erythritol-4-phosphate to the p-toluate precursor 2H3M4OP.
A. 1-Deoxyxylulose-5-phosphate (DXP) synthase
[0105]Pyruvate and G3P are condense...
example ii
Exemplary Pathway for Synthesis of p-Toluate from (2-Hydroxy-3-methyl-4-oxobutoxy)phosphonate by Shikimate Pathway Enzymes
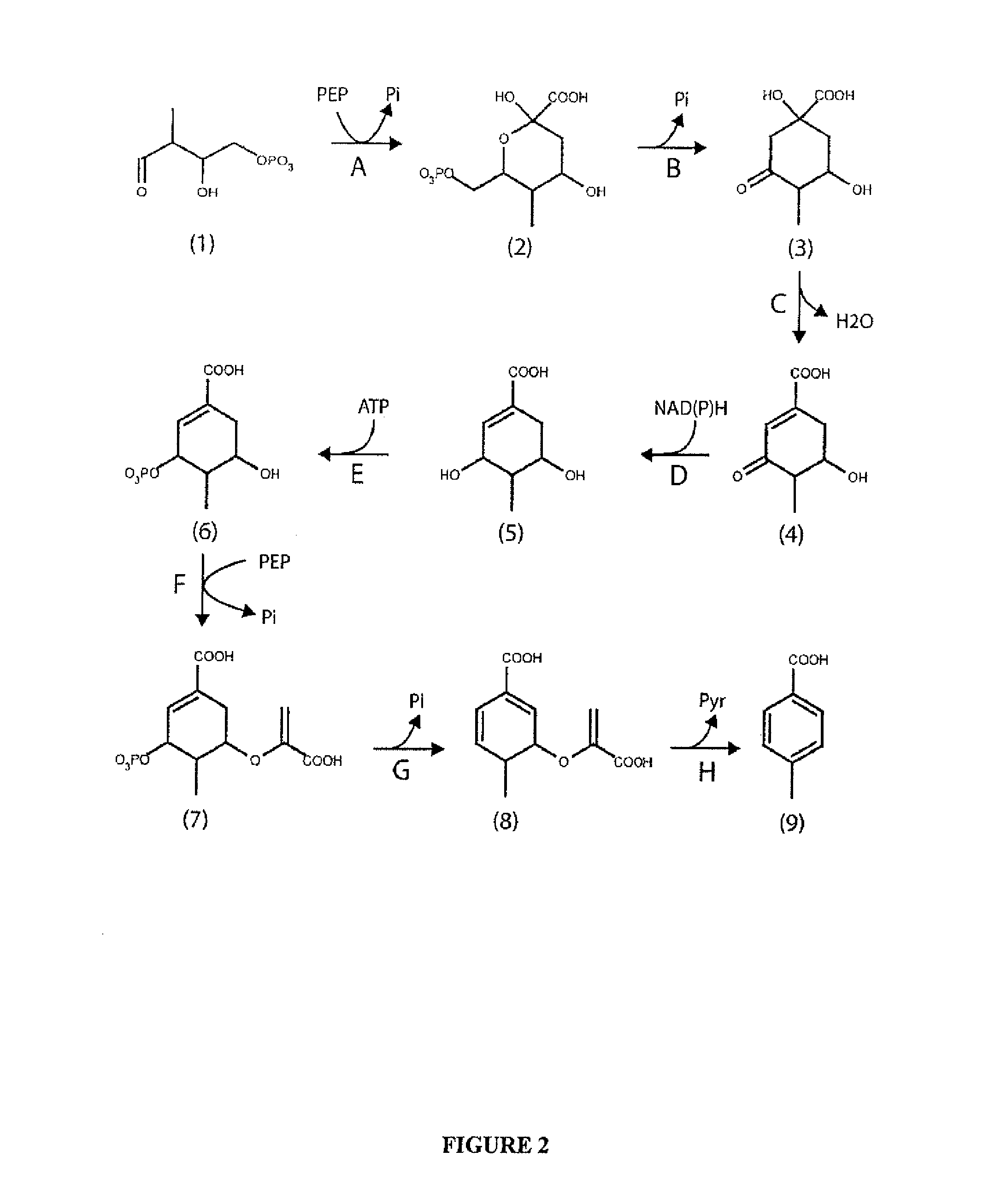
[0114]This example describes exemplary pathways for synthesis of p-toluate using shikimate pathway enzymes.
[0115]The chemical structure of p-toluate closely resembles p-hydroxybenzoate, a precursor of the electron carrier ubiquinone. 4-Hydroxybenzoate is synthesized from central metabolic precursors by enzymes in the shikimate pathway, found in bacteria, plants and fungi. The shikimate pathway is comprised of seven enzymatic steps that transform D-erythrose-4-phosphate (E4P) and phosphoenolpyruvate (PEP) to chorismate. Pathway enzymes include 2-dehydro-3-deoxyphosphoheptonate (DAHP) synthase, dehydroquinate (DHQ) synthase, DHQ dehydratase, shikimate dehydrogenase, shikimate kinase, 5-enolpyruvylshikimate-3-phosphate (EPSP) synthase and chorismate synthase. In the first step of the pathway, D-erythrose-4-phosphate and phosphoenolpyruvate are joined by DAHP synthas...
example iii
Exemplary Pathway for Enzymatic Transformation of p-Toluate to Terephthalic Acid
[0127]This example describes exemplary pathways for conversion of p-toluate to terephthalic acid (PTA).
[0128]P-toluate can be further transformed to PTA by oxidation of the methyl group to an acid in three enzymatic steps as shown in FIG. 3. The pathway is comprised of a p-toluate methyl-monooxygenase reductase, a 4-carboxybenzyl alcohol dehydrogenase and a 4-carboxybenzyl aldehyde dehydrogenase. In the first step, p-toluate methyl-monooxyngenase oxidizes p-toluate to 4-carboxybenzyl alcohol in the presence of O2. The Comamonas testosteroni enzyme (tsaBM), which also reacts with 4-toluene sulfonate as a substrate, has been purified and characterized (Locher et al., J. Bacteriol. 173:3741-3748 (1991)). 4-Carboxybenzyl alcohol is subsequently converted to an aldehyde by 4-carboxybenzyl alcohol dehydrogenase (tsaC). The aldehyde to acid transformation is catalyzed by 4-carboxybenzaldehyde dehydrogenase (tsa...
PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
| energy | aaaaa | aaaaa |
| of time | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com