Plate heat exchanger and method of using
a heat exchanger and plate technology, applied in heat exchange apparatus, refining by heating/cooling, lighting and heating apparatus, etc., can solve the problems of high temperature, high temperature, and high temperature of heat exchangers, and achieve the effect of lowering the enthalpy and high enthalpy
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
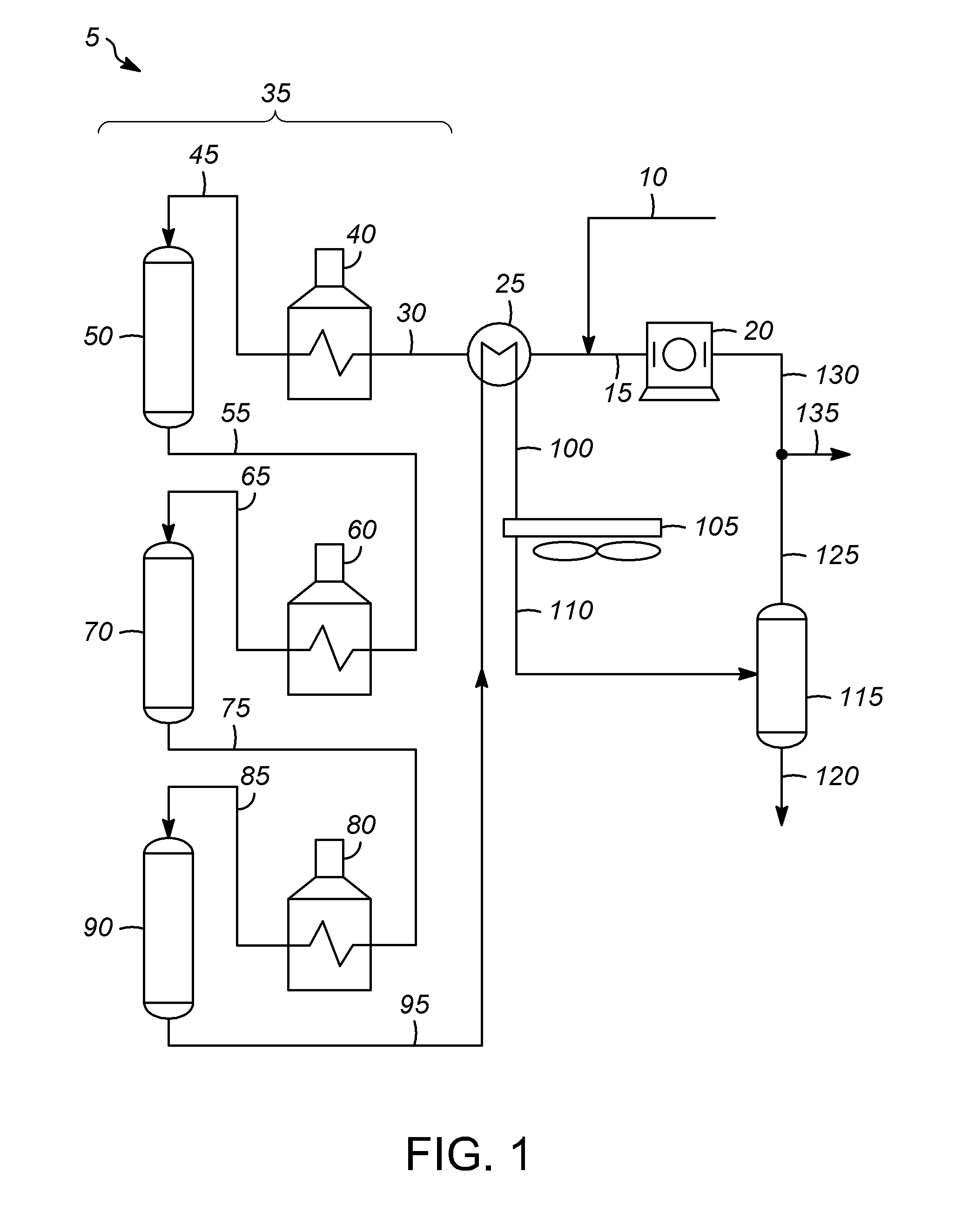
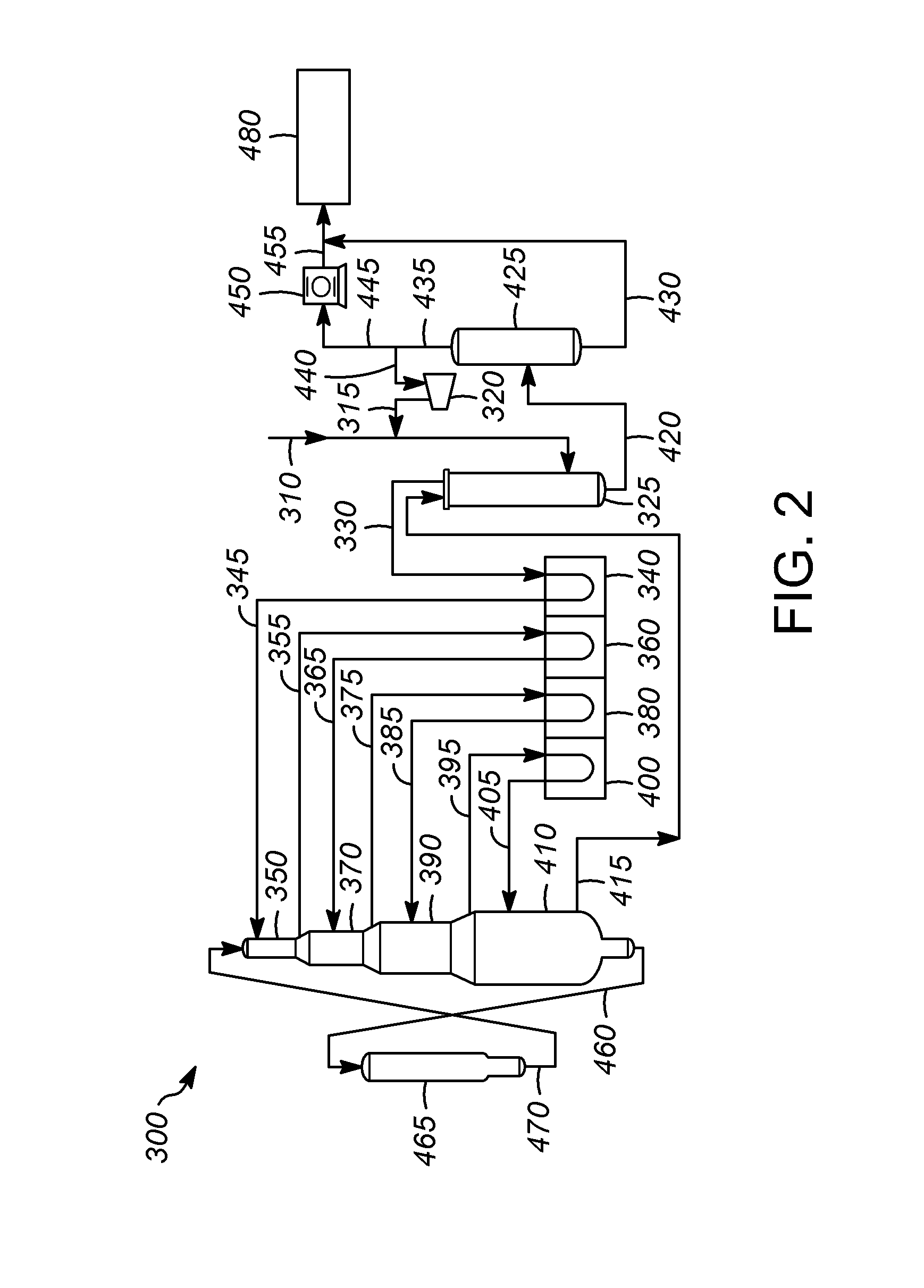
[0028]Processes for treating a hydrocarbon cold stream including one or more different hydrocarbons and which may include other components and / or impurities typically include flowing the hydrocarbon stream through various pieces of equipment. The equipment may be included as part of a larger oil refinery complex capable of performing one or more particular types of hydrocarbon conversion or treatment processes for converting or treating one or more components of the hydrocarbon cold stream to form a desired product. Heat is applied to the hydrocarbon stream and / or the equipment during operation. Heat may be applied to the hydrocarbon stream while it is within or before entering the equipment to raise the temperature thereof to a processing temperature. Particular process parameters or operating conditions, such as temperature, pressure, and space velocity, are typically process specific and are selected to promote the particular reactions or treatment steps of the particular process...
PUM
| Property | Measurement | Unit |
|---|---|---|
| temperature | aaaaa | aaaaa |
| temperatures | aaaaa | aaaaa |
| temperatures | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com