Curcumin Derivatives for Amyloid-Beta Plaque Imaging
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Methods and Materials
[0096]Reagents used for synthesis were purchased from Aldrich without further purification. Column chromatography was performed on silica gel (SiliCycle Inc., 60 Å, 40-63 mm) slurry packed into glass columns. Thioflavin S was purchased from Aldrich. Synthetic amyloid-b protein (1-40) was purchased from rPeptide (Bogart, Ga., 30622) and aggregates for in vitro studies were generated followed the reported procedure. BSA was purchased from Sigma, and human serum was obtained from Invitrogen (type AB). 1H and 13C NMR spectra were recorded at 400 MHz and 100 MHz respectively, and are reported in ppm downfield from tetramethylsilane. Fluorescence studies were carried out with F-4500 Fluorescense Spectrophotometer. Fluorescence quantum yields were determined using aqueous solution of Cy5.5-maleimide as a standard (y=0.23). High resolution mass spectra were obtained at the Harvard University, Department of Chemistry Instrumentation Facility. Transgenic Tg2576 mice {45} ...
example 2
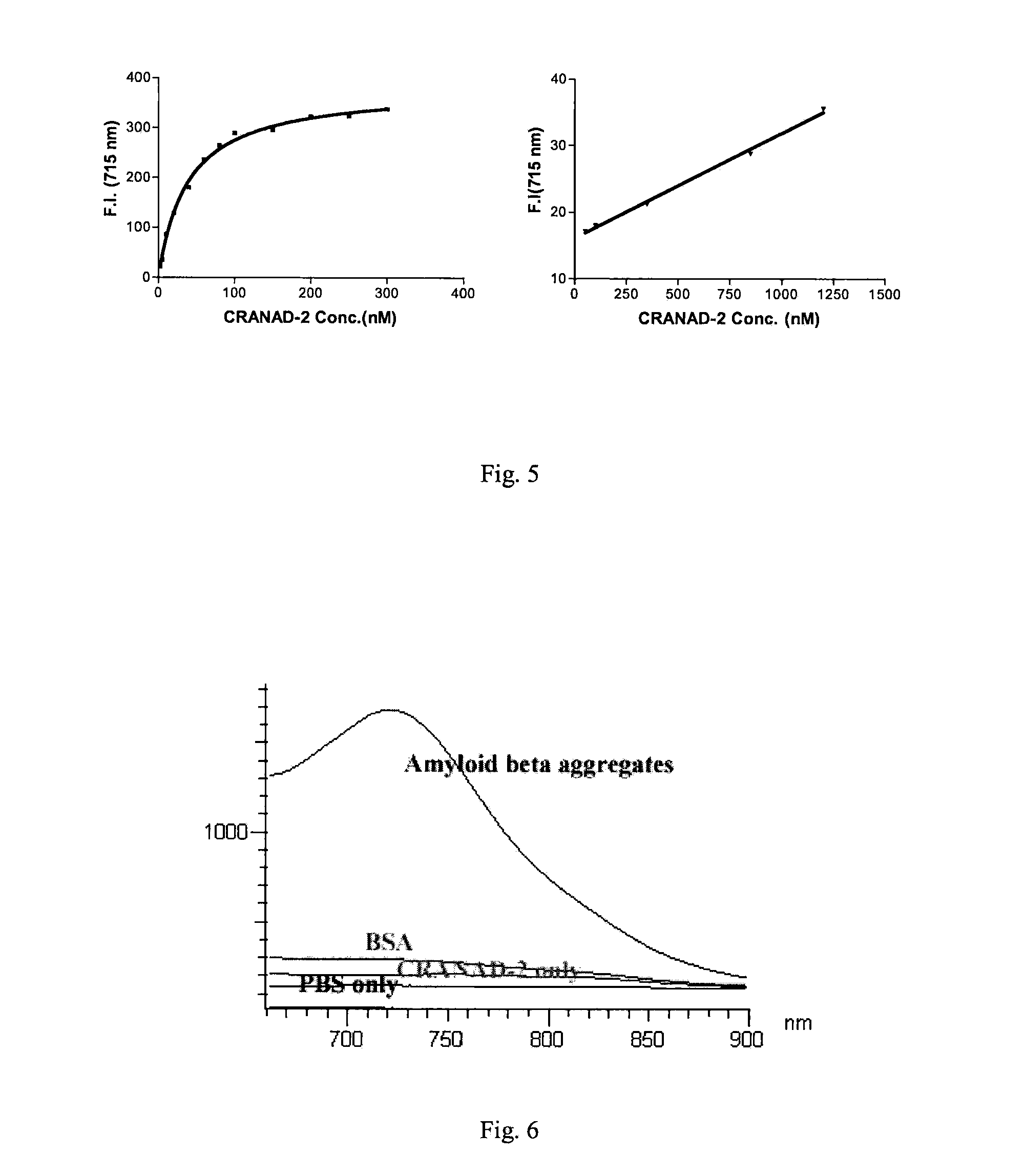
[0136]The inventors conducted tissue staining with 25 micron brain sections from 12-month old APP-PS1 mice by all the compounds listed in FIG. 12. As reported, curcumin, PiB, thioflavin T and CRANAD2 stained the plaques brightly; while as expected, CRANAD-2, CRANAD-3 and CRANAD-5 yielded good staining as well (FIG. 12B). These data indicate that these compounds are specific to Aβ plaques.
Preparation of CRANAD-3 and CRANAD-5 Compounds
[0137]Boric oxide (700.0 mg, 10.0 mmol) was dissolved in DMF (10.0 mL) at 120° C. Ensuring most of boric oxide dissolved was very crucial for high yield. To this solution, acetylacetone (1.1 mL, 10.0 mmol) was added, followed by tributyl borate (5.4 mL, 20.0 mmol) at 110° C. and stirred for 5 min. To the above borate complex, 4-N,N′-dimethylamino-benzaldehyde (3.1 g, 20.0 mmol) was added and stirred for 5 min. A mixture of 1,2,3,4-tetrahydroquinoline (0.2 mL) and acetic acid (0.4 mL) in DMF (4.0 mL) was added to the reaction mixture and...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Electric charge | aaaaa | aaaaa |
| Capacitance | aaaaa | aaaaa |
| Capacitance | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com