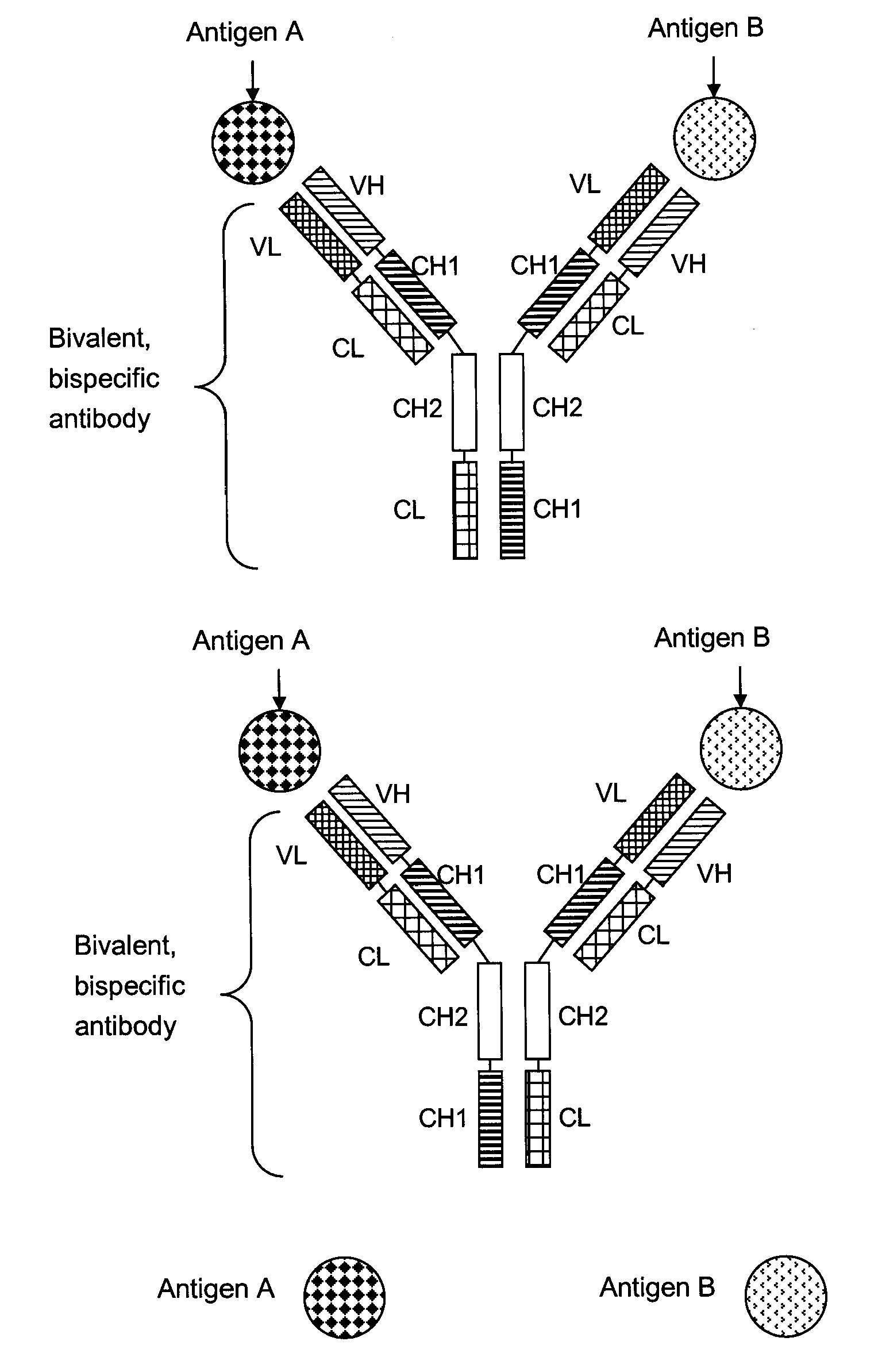
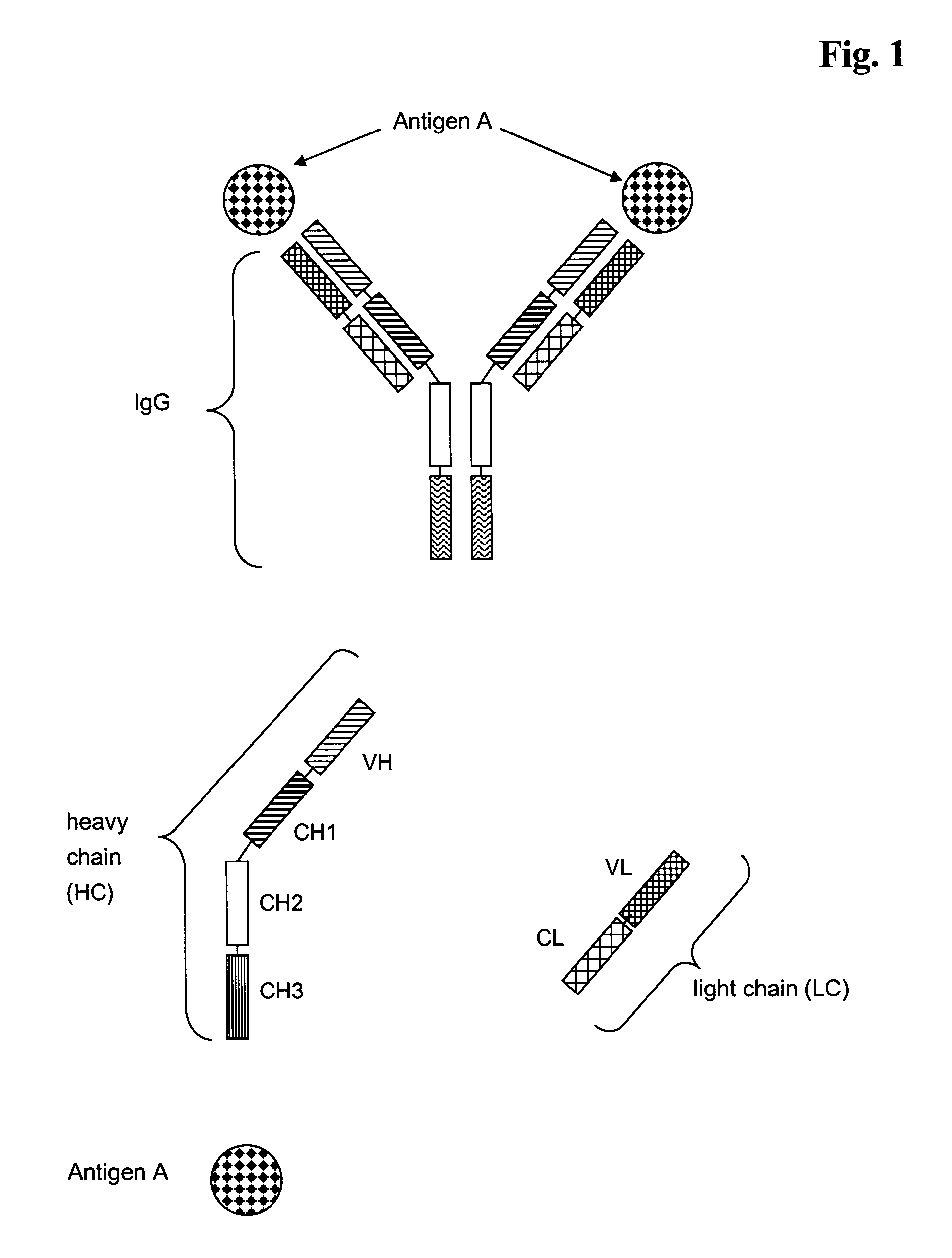
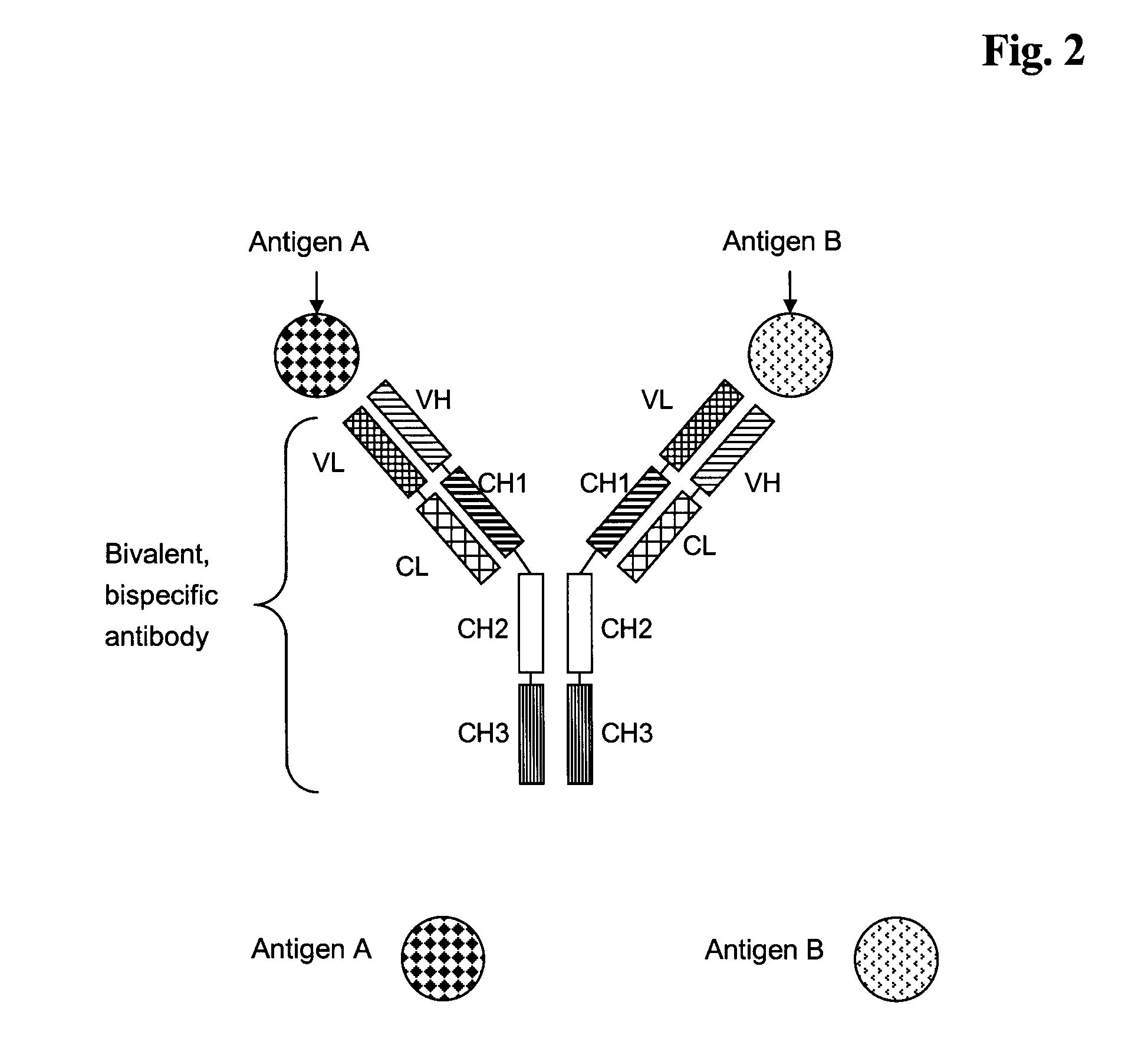
Bivalent, bispecific antibodies
a technology of bispecific antibodies and antibodies, applied in the field of bivalent, bispecific antibodies, can solve the problems of mismatched byproducts, poor antibody stability, aggregation and increased immunogenicity, and no data describing the progression towards the clinic are currently available, so as to facilitate purification and improve the yield of recombinant production
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples
Materials & General Methods
[0120]General information regarding the nucleotide sequences of human immunoglobulins light and heavy chains is given in: Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md. (1991). Amino acids of antibody chains are numbered and referred to according to EU numbering (Edelman, G. M., et al., Proc. Natl. Acad. Sci. USA 63 (1969) 78-85; Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md., (1991)).
Recombinant DNA Techniques
[0121]Standard methods were used to manipulate DNA as described in Sambrook, J. et al., Molecular cloning: A laboratory manual; Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y., 1989. The molecular biological reagents were used according to the manufacturer's instructions.
Gene Synthesis
[0122]Desired gene segments were prepared from oligonu...
examples 1
Production, Expression, Purification and Characterization of Monospecific, Bivalent Antibody, Wherein the Variable Domains VL and VH are Replaced by Each Other (Abbreviated Herein as VL-VH Exchange Antibody)
example 1a
Making of the Expression Plasmids for the Monospecific, Bivalent VL-VH Exchange Antibody
[0153]The sequences for the heavy and light chain variable domains of the monospecific, bivalent VL-VH exchange antibody including the respective leader sequences described in this example are derived from a human antibody heavy chain (SEQ ID NO: 1, plasmid 4843-pUC-HC-IGF-1R) and a light chain (SEQ ID NO: 2, plasmid 4842-pUC-LC-IGF-1R) described in WO 2005 / 005635, and the heavy and light chain constant domains are derived from a human antibody (C-kappa and IgG1).
[0154]The gene segments encoding the antibody leader sequence, light chain variable domain (VL) and the human heavy chain constant domain 1 (CH1) were joined and fused to the 5′-end of the Fc domains of the human γ1-heavy chain constant domains (Hinge-CH2-CH3). The DNA coding for the respective fusion protein resulting from the exchange of the VH domain by the VL domain (VH-VL exchange) was generated by gene synthesis and is denoted ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com