Cloning and sequencing of pyruvate decarboxylase (PDC) genes from bacteria and uses therefor
a technology of pyruvate decarboxylase and pdc, which is applied in the field of cloning and sequencing of pdc genes from bacteria and uses therefor, can solve the problem that organisms rarely produce all of the necessarily enzymes at the most desirable levels, and achieve the effect of improving the efficiency of substrate utilization and the economics of the fermentation process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Identification and Characterization of PDC Gene from Gram-Negative Bacterium Zymobacter palmae
[0155] In this example, the identification and characterization of a PDC gene from the Gram-negative bacterium Z. palmae is described.
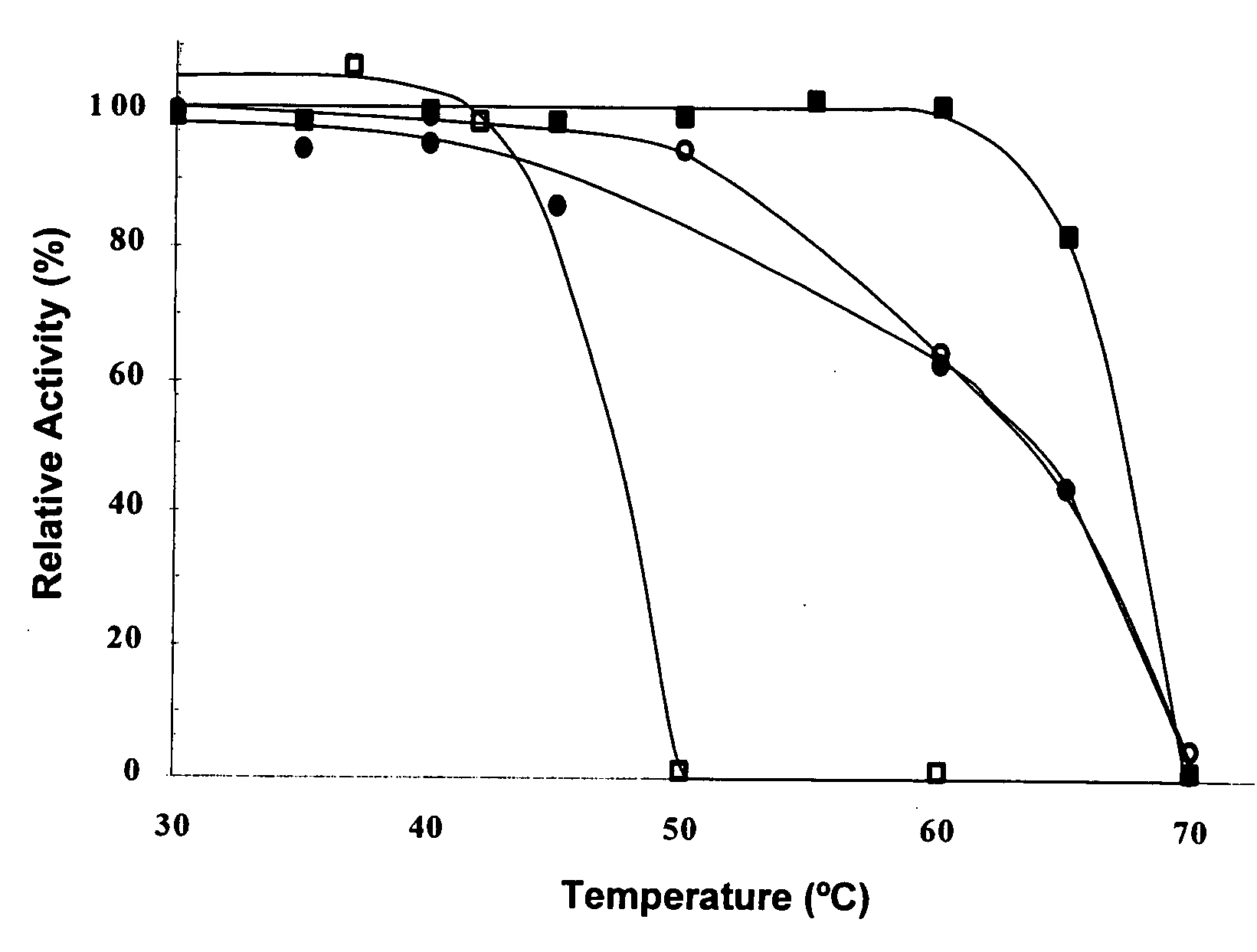
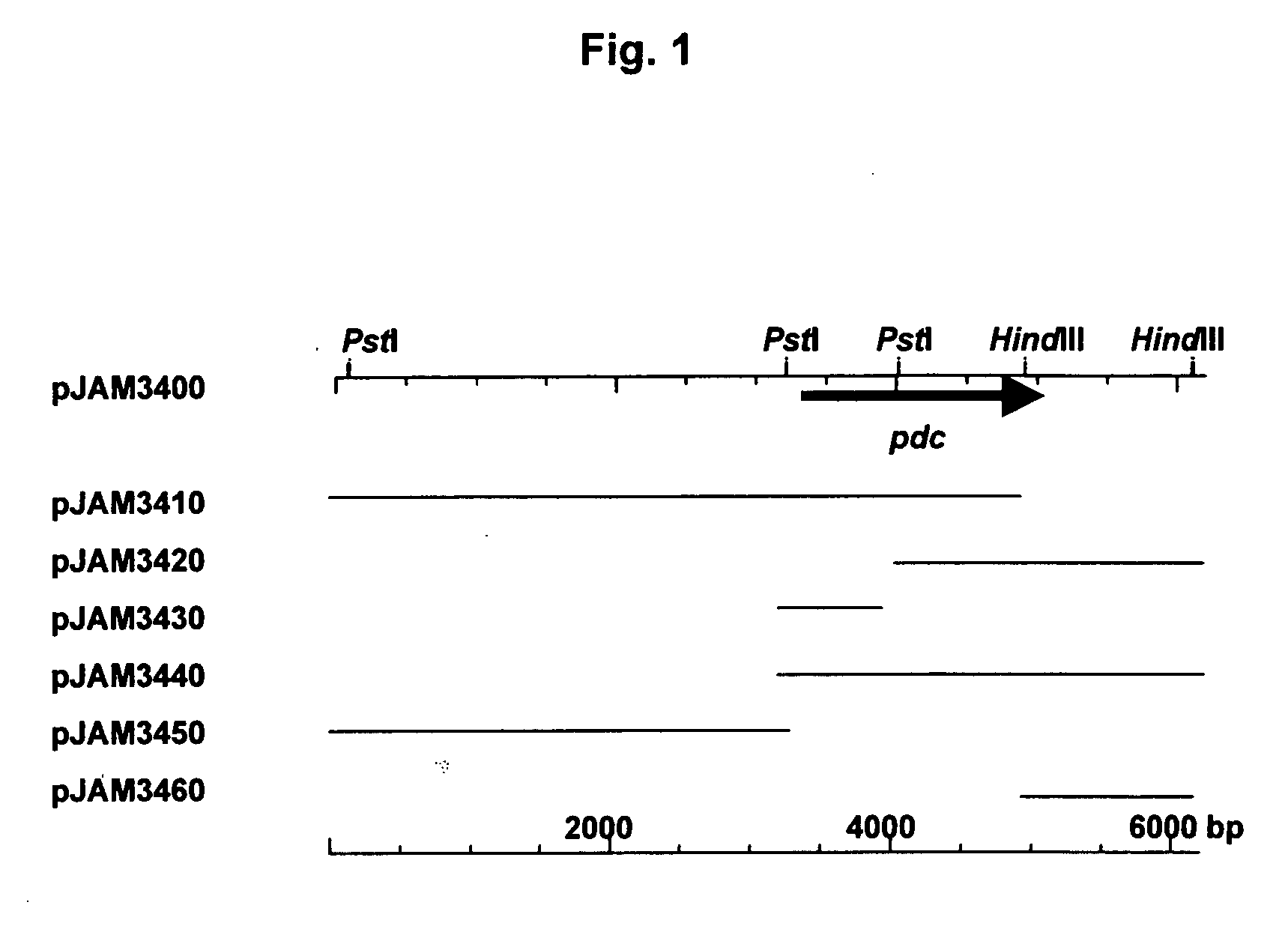
[0156] Briefly, a pyruvate decarboxylase (pdc) operon was isolated from a genomic library of Zymobacter palmae using a degenerate oligonucleotide probe based on the N-terminal amino acid sequence of the purified protein (FIG. 1). A 1.7-kb PstI to BamHI subclone of this fragment (pJAM3440) was found to be required for PDC activity in recombinant E. coli. Based on DNA sequence, a 1668-bp ORF (55 mol % G+C content) was identified in this region that encoded a putative PDC polypeptide (FIG. 1). A canonical Shine-Dalgarno sequence (GGAGG) was 10 bp upstream of the translation start codon (ATG). In addition, a putative −35 and −10 promoter (TTcACt-N17-atTAAT, where N is any nucleotide and uppercase letters match the bacterial promoter consensus) was located 16 to...
example 2
Identification and Characterization of PDC Gene from Gram-Negative Aerobic Bacterium Acetobacter pasteurianus
[0158] In this example, the identification and characterization of a PDC gene from the Gram-negative bacterium Acetobacter pasteurianus is described.
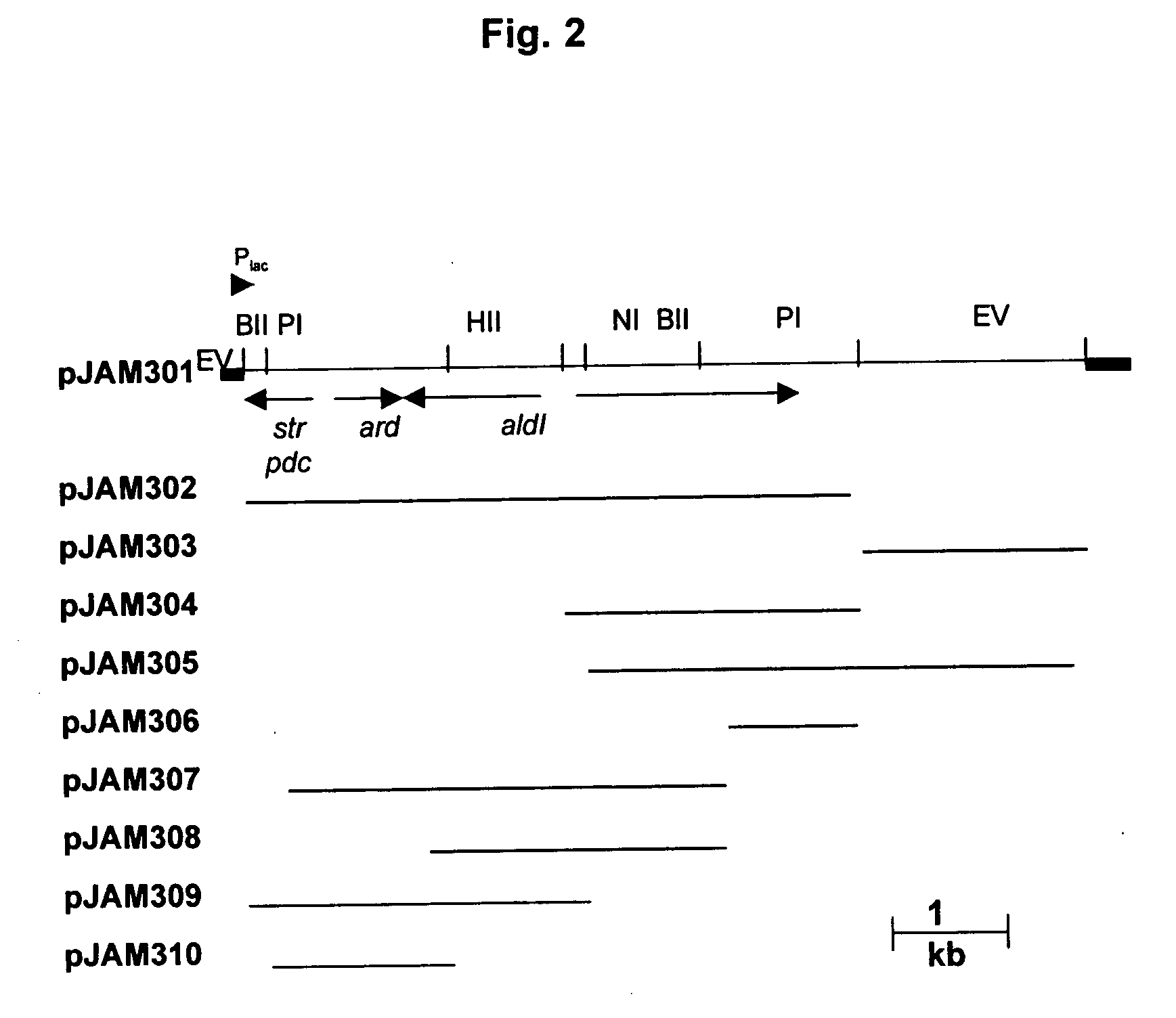
[0159] Briefly, a chromosomal DNA was isolated from cells of A. pasteurianus using the method described by Harwood et al. (17). For Southern analysis, the genomic DNA (1 to 2 μg per lane) was cleaved with restriction enzymes (AatII, BamHI, ClaI, EcoRI, and HincII), separated by gel electrophoresis (0.8% agarose), and transferred by downward capillary action to positively charged nylon membranes. Membranes were equilibrated at 60° C. for 2 h in 5×SSC (1×SSC is 0.15 M NaCl plus 0.015 M sodium citrate) containing 1% blocking reagent, 0.1% N-lauroylsarcosine, and 0.02% sodium dodecyl sulfate (SDS). Random primers were used to label a probe with dixoigenin-11-dUTP using a 0.7-kb KpnI fragment of the Z. mobilis pdc gene as the templa...
example 3
Identification and Characterization of PDC Gene from Gram-Positive Bacterium Sarcina ventriculi
[0167] In this example, the identification and characterization of a PDC gene from the Gram-positive bacterium Sarcina ventriculi is described.
[0168] Briefly, a degenerate oligonucleotide 5′-AARGARGTNAAYGTNGARCAYATGTTYGGNGT-3′ (SEQ ID NO: 11) was synthesized based on the N-terminal amino acid sequence of PDC purified from S. ventriculi (Lowe and Zeikus, 1992) (where, R is A or G; N is A, C, G, or T; Y is C or T). This oligonucleotide was labeled at the 3′-end using terminal transferase with digoxigenin-11-dUTP and dATP as recommended by the supplier (Roche Molecular Biochemicals) and was used to screen genomic DNA from S. ventriculi.
[0169] For Southern analysis, genomic DNA was digested with BglI, EcoRI, or HincII, separated by 0.8% agarose electrophoresis, and transferred to positively charged nylon membranes (Southern, 1975). Membranes were equilibrated at 58° C. for 2 h in 5×SSC (1×S...
PUM
| Property | Measurement | Unit |
|---|---|---|
| cell cycle time | aaaaa | aaaaa |
| temperatures | aaaaa | aaaaa |
| temperatures | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com