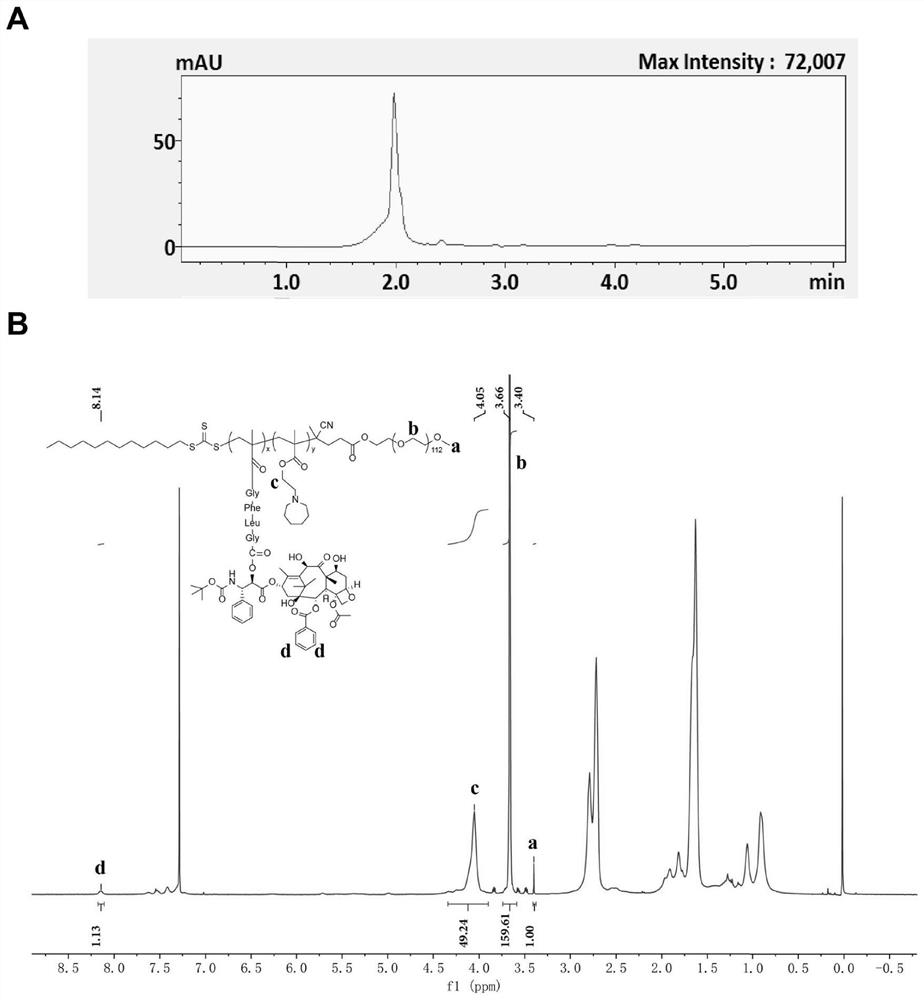
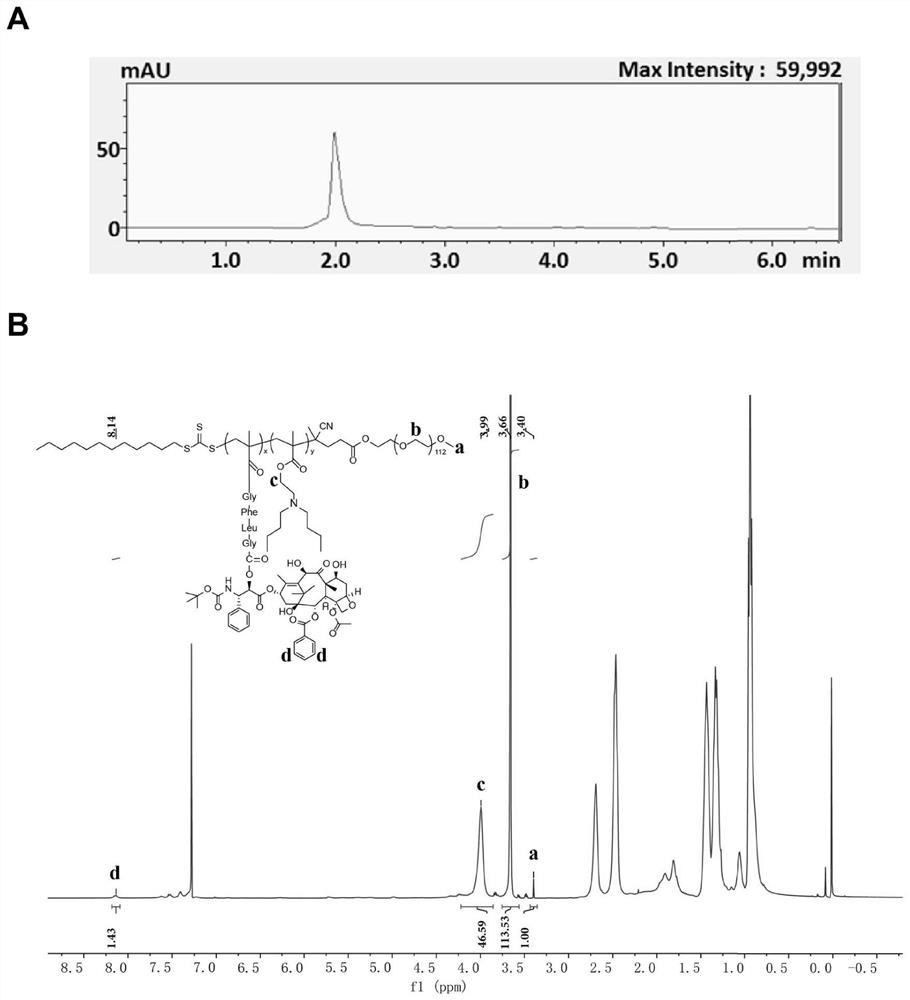
PH/cathepsin B step-by-step response polymer-drug conjugate and micelle, as well as preparation method and application of pH/cathepsin B step-by-step response polymer-drug conjugate and micelle
A technology of cathepsin and polymer, which is applied in the field of biomedicine, can solve the problems of poor response to disease signals and weak penetration promotion ability, and achieve the effects of avoiding drug release, reducing toxic and side effects, and rapid response
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0116] The preparation method of the polymer-drug conjugate micelles of the present invention can adopt solvent removal method, thin film ultrasonic method, ethanol injection method, etc., preferably solvent removal method.
[0117] For example, the polymer-drug conjugate micelles of the present invention can be prepared by the following steps:
[0118] Step 1: Weigh one or more polymer-drug conjugates according to a certain proportion, add an appropriate amount of organic solvent to ultrasonically dissolve, and set aside;
[0119] Step 2: Take ultrapure water with a volume ratio of 10-100, and quickly add the solution obtained in "Step 1" into the ultrapure water under the ultrasonic condition of the probe, and continue ultrasonication for 1-3 minutes;
[0120] Step 3: Transfer the micellar solution containing the organic solvent after the ultrasonication to an ultrafiltration tube, and perform ultrafiltration with ultrapure water for 3 to 5 times to remove the organic solven...
Embodiment 1
[0143] Embodiment 1: the synthesis of macromolecular chain transfer agent PEG-CTA
[0144] Accurately weigh PEG-OH (5g, 1mmol) and equimolar equivalent of CTA in a round bottom flask, add 50mL of dichloromethane, ultrasonically dissolve, then add 0.1 times the molar equivalent of 4-dimethylaminopyridine and 1.5 times the molar equivalent of dicyclohexylcarbodiimide. After reacting at room temperature in the dark for 36 hours, the precipitate was removed by ultrafiltration, and the organic filtrate was concentrated to 10 mL with a rotary evaporator. The product was purified three times in glacial ether to obtain 4.2 g of macromolecular chain transfer agent PEG-CTA with a yield of 84%.
Embodiment 2
[0145] Example 2: RAFT Polymerization and Structural Characterization of Polymer-Drug Conjugates
[0146] 1. Synthesis of polymer-drug conjugate PEG-PC7A-DTX
[0147] Weigh the PEG-CTA (50mg, 0.01mmol) in "Example 1" in the reaction bottle, add 100 times molar equivalent of C7A monomer and 5 times molar equivalent of MA-GFLG-DTX prodrug monomer, and use 1mL N,N-dimethylformamide was dissolved, and then 0.333 times the molar equivalent of initiator azobisisobutyronitrile was added, and it was fully dissolved and mixed evenly by ultrasonication. Freezing-nitrogen-filling-thawing cycles were performed 3 times to remove oxygen, and the reaction was placed at 60°C for 36 hours. After the reaction is terminated, transfer the reaction solution to a 10kD dialysis bag and dialyze with N,N-dimethylformamide for 48 hours to remove unreacted monomers and other small molecules, then dialyze against distilled water for 12 hours to remove organic solvents, and dialyze the product The polym...
PUM
| Property | Measurement | Unit |
|---|---|---|
| particle diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com