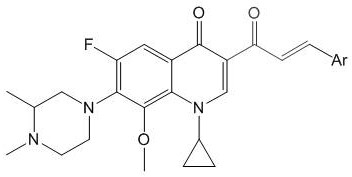
Acrylketone derivative of N-methyl gatifloxacin and preparation method and application of acrylketone derivative
A technology of gatifloxacin and acrylone, which is applied to the acrylone derivative of N-methyl gatifloxacin and its preparation field, can solve the problems such as the uncertainty of the effect of the C-3 carboxyl group of fluoroquinolone, and achieve an increase in anti-tumor effect. Activity and anti-drug resistance, the effect of reducing side effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
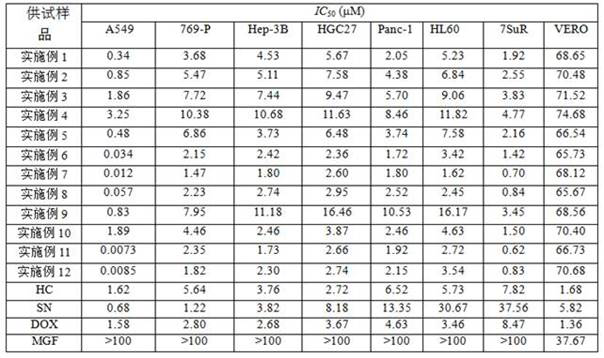
Image
Examples
Embodiment 1
[0031] 1-cyclopropyl-6-fluoro-7-(3,4-dimethylpiperazin-1-yl)-3-cinnamoyl-8-methoxy-quinoline-4(1 H )-ketone (I-1), its chemical structural formula is:
[0032]
[0033] That is, Ar in formula I is phenyl.
[0034] The preparation method of this compound is:
[0035] (1) Using N-methylgatifloxacin shown in Formula II as a raw material, react with carbonyldiimidazole (CDI) to prepare N-methylgatifloxacin imidazole amide compound shown in Formula III, and its specific preparation Methods as below:
[0036]
[0037] Take 1-cyclopropyl-6-fluoro-7-(3,4-dimethylpiperazin-1-yl)-8-methoxy-quinoline-4(1 H 20 g (51.0 mmol) of )-keto-3-carboxylic acid II was dissolved in 500 mL of anhydrous acetonitrile, 15.2 g (94.0 mmol) of carbonyldiimidazole was added, and the mixed reaction was stirred and refluxed in a water bath until the raw material II disappeared. Leave it at room temperature, collect the resulting solid by filtration, and recrystallize with acetone to obtain the light...
Embodiment 2
[0046] 1-cyclopropyl-6-fluoro-7-(3,4-dimethylpiperazin-1-yl)-3-(4-methoxycinnamoyl)-8-methoxy-quinoline-4 (1 H )-ketone (I-2), its chemical structural formula is:
[0047]
[0048] That is, Ar in formula I is p-methoxyphenyl.
[0049] The preparation method of this compound is:
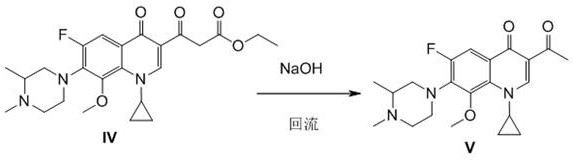
[0050] (1) 1-cyclopropyl-6-fluoro-7-(3,4-dimethylpiperazin-1-yl)-8-methoxy-quinoline-4(1 H )-ketone-3-ethanone V is prepared with reference to steps (1)-(3) of Implementation 1, the solvent in step (1) is replaced by tetrahydrofuran, the mixture of N-methylgatifloxacin and carbonyldiimidazole The molar ratio is 1:1.0;
[0051] (2) Take 1-cyclopropyl-6-fluoro-7-(3,4-dimethylpiperazin-1-yl)-8-methoxy-quinoline-4(1 H 1.2 g (3.0 mmol) of )-keto-3-ethanone V was dissolved in 20 mL of absolute ethanol, and 0.57 g (4.2 mmol) of 4-methoxybenzaldehyde and base catalyst piperidine (0.1 mL) were added. The mixed reactants were refluxed for 20 h, left at room temperature, and the resulting solid was coll...
Embodiment 3
[0053] 1-cyclopropyl-6-fluoro-7-(3,4-dimethylpiperazin-1-yl)-3-(3,4-dioxymethylenecinnamoyl)-8-methoxy- Quinoline-4(1 H )-ketone (I-3), its chemical structural formula is:
[0054]
[0055] That is, Ar in formula I is 3,4-(dioxymethylene)phenyl.
[0056] The preparation method of this compound is:
[0057] (1) 1-cyclopropyl-6-fluoro-7-(3,4-dimethylpiperazin-1-yl)-8-methoxy-quinoline-4(1 H )-keto-3-ethanone V is prepared with reference to steps (1)-(3) of Implementation 1, and the solvent in step (1) is replaced by dioxane, N-methylgatifloxacin and carbonyl The mol ratio of diimidazole is 1:2.0;
[0058] (2) Take 1-cyclopropyl-6-fluoro-7-(3,4-dimethylpiperazin-1-yl)-8-methoxy-quinoline-4(1 H )-keto-3-ethanone V1.2 g (3.0 mmol) was dissolved in 20 mL of absolute ethanol, 0.53 g (3.5 mmol) of 3,4-dioxymethylene benzaldehyde and base catalyst piperidine (0.1 mL). The mixed reactants were refluxed for 20 h, left at room temperature, and the resulting solid was collected b...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com