Method for preparing phenol compound
A technology for phenolic compounds and catalysts is applied in the field of preparing phenolic compounds, which can solve the problems of reducing the selectivity and difficulty of phenolic compounds, and achieve the effects of high efficiency, simple preparation and high reaction conversion rate.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
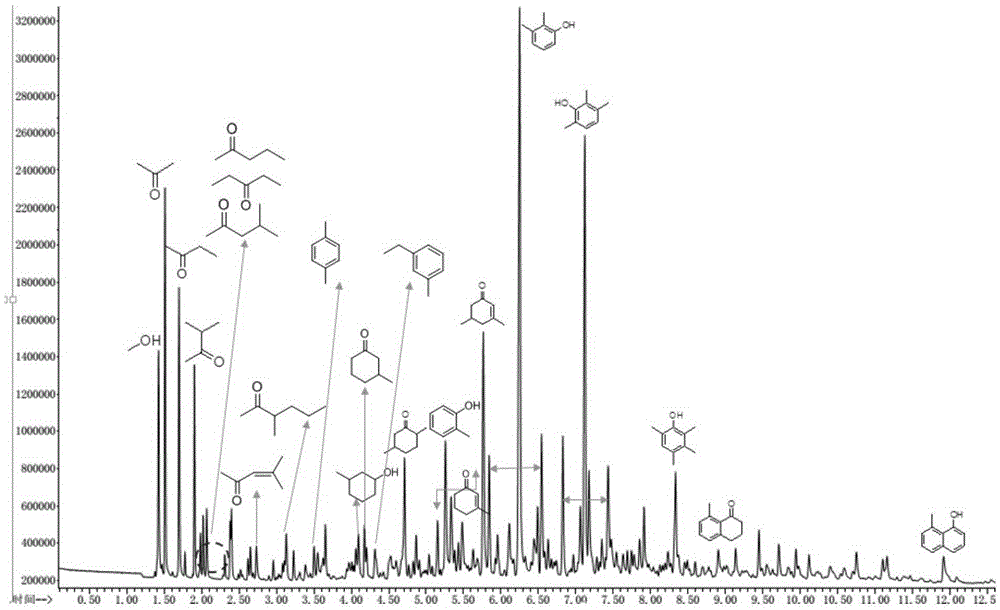
[0027] Weigh 200g of ammonium cerium nitrate and bake it at 500°C for 5h under air atmosphere to obtain cerium dioxide. Fill the 14-25 mesh catalyst with cerium oxide forming sieve into a reaction tube with an inner diameter of 7mm, fill it with a 15cm bed, and under normal pressure, prepare the reaction solution of acetic acid, formaldehyde and ethanol according to the molar ratio of 1:5:1. Raw materials, the feed rate is 0.03mL·min -1 , nitrogen as the carrier gas, the flow rate is 30mL·min -1 , reacted at 350°C for 12h, and separated the liquid after the reaction, and the oil phase and water phase obtained were analyzed by chromatography respectively. The conversion of acetic acid was 70% and the overall selectivity to phenolic compounds was 75%.
Embodiment 2
[0029] Weigh 200g of ammonium cerium nitrate and bake it at 500°C for 5h under air atmosphere to obtain cerium dioxide. Fill the 14-25 mesh catalyst of ceria forming sieve into a reaction tube with an inner diameter of 8 mm, fill it with a 25 cm bed, and under normal pressure, use the reaction solution prepared by acetic acid and formaldehyde according to the molar ratio of 1:5 as the raw material, and feed The speed is 0.03mL·min -1 , nitrogen as the carrier gas, the flow rate is 30mL·min -1 , reacted at 350°C for 12h, and separated the liquid after the reaction, and the oil phase and water phase obtained were analyzed by chromatography respectively. The conversion of acetic acid was 80%, and the overall selectivity to phenolic compounds was 73%.
Embodiment 3
[0031] Weigh 50g of cerium nitrate and dissolve it in 4L of water, use 25wt% ammonia water as a precipitating agent, adjust the pH of the system to 9, centrifuge the obtained precipitate, dry it, and roast it at 500°C for 3 hours to obtain cerium oxide. Fill the 14-25 mesh catalyst with cerium oxide forming sieve into a reaction tube with an inner diameter of 7mm, fill it with a 9cm bed, and under normal pressure, the reaction solution prepared by acetic acid, formaldehyde and methanol according to the molar ratio of 1:5:1 is Raw materials, the feed rate is 0.03mL·min -1 , nitrogen as the carrier gas, the flow rate is 30mL·min -1 , reacted at 350°C for 12h, and separated the liquid after the reaction, and the oil phase and water phase obtained were analyzed by chromatography respectively. The conversion of acetic acid was 100%, and the overall selectivity to phenolic compounds was 85%.
PUM
| Property | Measurement | Unit |
|---|---|---|
| height | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com