Classical swine fever virus E2 subunit vaccine and preparation thereof
A technology for swine fever virus and swine fever vaccine, which can be applied in viral peptides, antiviral agents, botanical equipment and methods, etc., and can solve problems such as cost burden
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
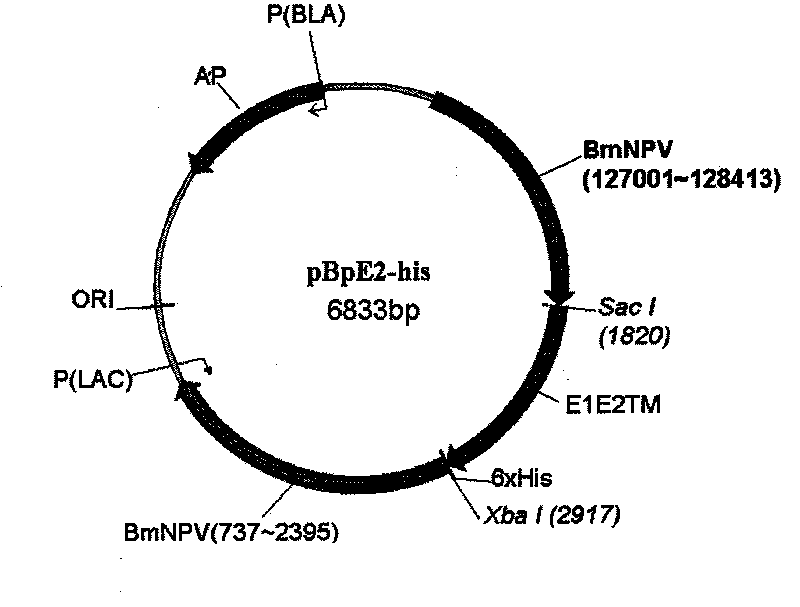
[0038] Example 1: Construction of a plasmid comprising the E2 subunit recombination gene
[0039] Amplification of E1E2TM gene of Subgroup 2.1 virus
[0040] First, use reverse transcription polymerase superscript II reverse transcriptase (Invitrogen, CA, USA) to carry out reverse transcription polymerase chain reaction, and use two E1-E2 gene-specific primers (as shown in Table 1), from TD / 96 / TWN type The swine fever virus gene body amplifies the E1-E2 gene sequence 2354-3466bp (SEQ ID NO: 2), wherein the nucleotide sequence includes a part of the N-terminal 60 nucleotide sequence of the E1 gene and the C-terminal to the TM region of the E2 gene (E1E2TM) 1026 nucleotides, a total of 1086 nucleotides. Then, the second reverse transcription polymerase chain reaction uses the first reaction product, and the reverse primer (5'-TTGAG CTCAC CATGT TGAGA GGACA GGT-3') (SEQ ID NO: 5) to reverse transcribe into cDNA, and finally The amount of product was amplified by polymerase chain...
Embodiment 2
[0045] Example 2: Homologous recombination
[0046] 5 μg of the plasmid pBpE2-his that has been colonized was mixed with 50 μg of the semi-purified gene body of baculovirus using 200 μl of commercial Liposome (Invitrogen, USA), and 1 ml of TC-100 culture medium without serum components was added, and A monolayer of silkworm cells (BmN) grown on a 30mm culture dish was added to allow co-transfection (Transfection) into the cells, and a recombination reaction naturally occurred in the cells. After about 4-7 days, anti-histidine monoclonal antibody (Invitrogen, USA) or anti-E2 monoclonal antibody (WH303, Veterinary Laboratory Agency, UK) can be used to dilute 200 times and then stained to screen out virus plaques with expressive ability. After continuous 10-fold dilution and infection of cells for three times, the virus with the highest potency was used as the seed virus strain after a large number of subclone colonization to determine the potency.
Embodiment 3
[0047] Embodiment 3: Taking silkworm as protein production platform
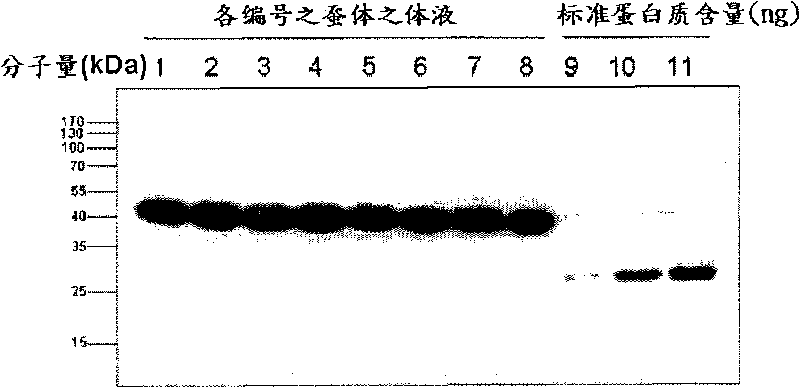
[0048] The selected recombinant baculovirus can infect the silkworm and express the E2 protein. The silkworm is a hybrid of the original species (OJ03×OJ04). The silkworm eggs of the second species are used for feeding. Generally, when the silkworm is reared to the fifth instar about the 23rd day, 10 μl of virus liquid (10 μl) can be injected at the third back hole. 7.0 TCID 50 / ml), about 3-5 days after infection, the body fluid can be collected when the silkworm shows the onset, and the concentration of E2 protein can be detected. About 0.3-0.5ml of body fluid can be extracted from each silkworm, and its body fluid is analyzed for antigen concentration by western blot method.
[0049] Take 20 μl of body fluid, use 4-1 2% SDS-PAGE gradient gel electrophoresis analysis, and then transfer protein to nitrocellulose membrane (nitrocellulose membrane), and then fill (blocking) with 5% non-fat milk powder. Th...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com