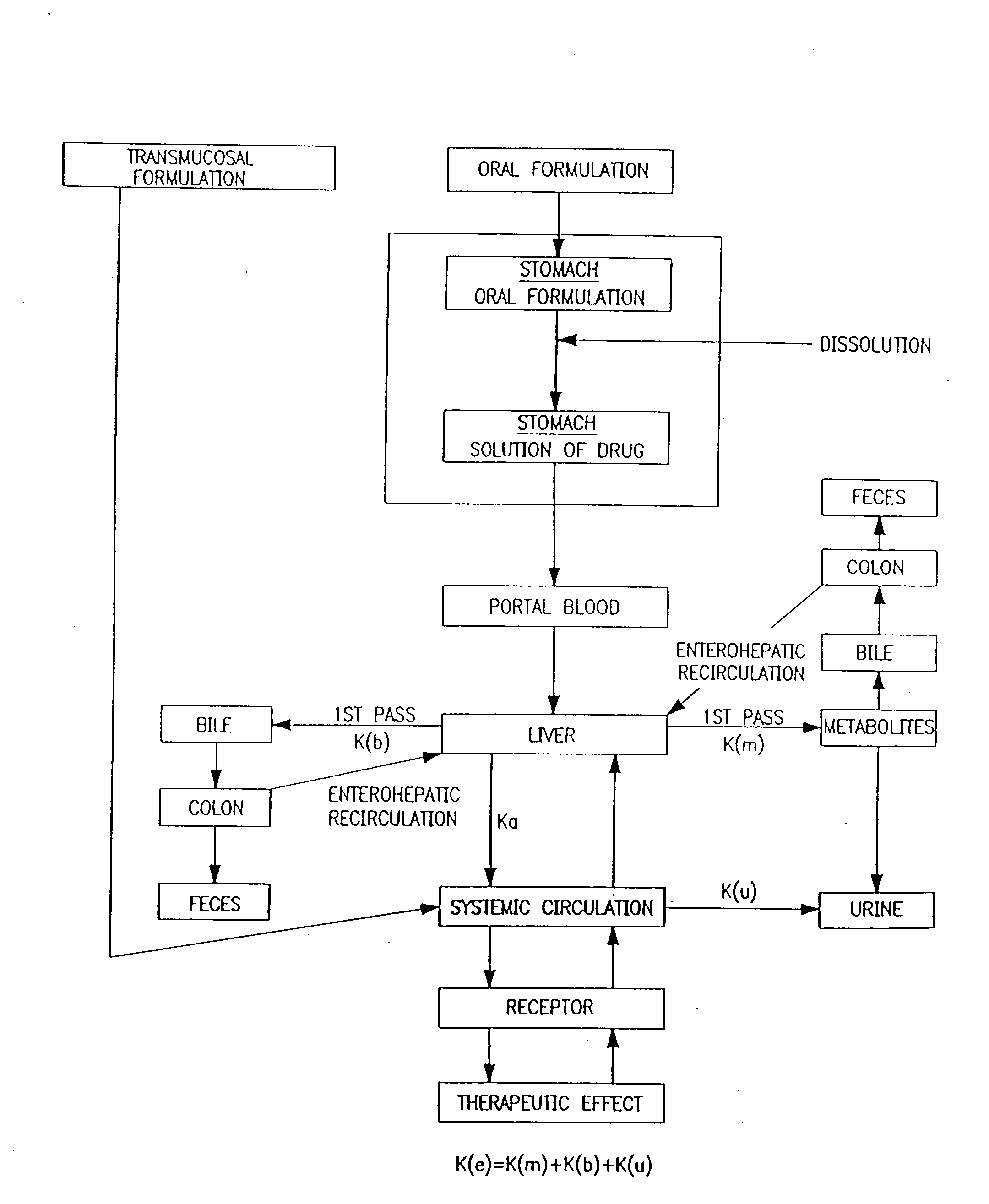
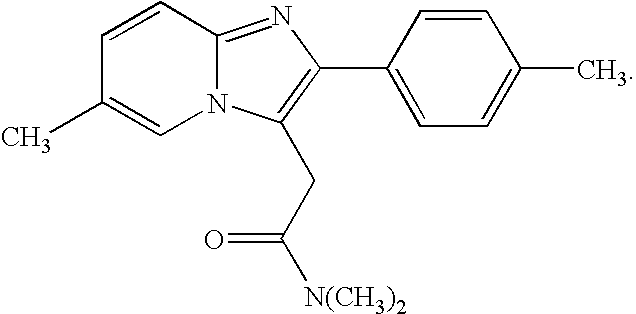
Buccal, polar and non-polar spray containing zolpidem
a technology of zolpidem and zolpidem, which is applied in the directions of drug composition, aerosol delivery, nervous disorders, etc., can solve the problems of presenting their own problems, and achieve the effect of rapid absorption through the oral mucosa and fast onset of
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Biologically Active Peptides Including Peptide Hormones
[0073]
A. Cyclosporine lingual spraymost preferredAmountspreferred amountamountcyclosporine 5-5010-3515-25water 5-207.5-50 9.5-12 ethanol 5-607.5-50 10-20polyethylene glycol20-6030-4535-40flavors0.1-5 1-42-3B. Cyclosporine Non-Polar lingual spraymost preferredAmountspreferred amountamountcyclosporine 1-50 3-40 5-30Migylol202530-40Polyoxyethylated202530-40castor oilButane25-8030-7033-50flavors0.1-5 1-42-3C. Cyclosporine non-polar bite caosulemost preferredAmountspreferred amountamountcyclosporine 1-35 5-2510-20olive oil25-6035-5530-45polyoxyethylated25-6035-5530-45oleic glyceridesflavors0.1-5 1-42-3D. Cyclosporine bite capsulemost preferredAmountspreferred amountamountcyclosporine 5-5010-3515-25polyethylene20-6030-4535-40glycolglycerin 5-307.5-25 10-20propylene glycol 5-307.5-25 10-20flavors0.1-10 1-83-6E. Sermorelin (as the acetate) lingual sprayAmountspreferred amountmost preferredsermorelin (as the.01-5 .1-3 .2-1.0acetate...
example 2
CNS Active Amines and Their Salts: Including but not Limited to Tricyclic Amines, GABA Analogues, Thiazides, Phenothiazine Derivatives, Serotonin Antagonists and Serotonin Reuptake Inhibitors
[0074]
most preferredAmountspreferred amountamountA. Sumatriptan succinate lingual spraysumatriptan succinate0.5-30 1-2010-15ethanol5-607.5-50 10-20propylene glycol5-307.5-20 10-15polyethylene glycol0-6030-4535-40water5-307.5-20 10-15flavors0.1-5 1-42-3B. Sumatriptan succinate bite capsulesumatriptan succinate0.01-5 0.05-3.5 0.075-1.75 polyethylene glycol25-70 30-6035-50glycerin25-70 30-6035-50flavors0.1-10 1-83-6C. Clozepine lingual sprayclozepine0.5-30 1-2010-15ethanol5-607.5-50 10-20propylene glycol5-307.5-20 10-15polyethylene glycol0-6030-4535-40water5-307.5-20 10-15flavors0.1-5 1-42-3D. Clozepine non-polar lingual spray with propellantclozepine0.5-30 1-2010-15Migylol20-85 25-7030-40Butanol5-8030-7560-70flavors0.1-5 1-42-3E. Clozepine non-polar lingual spray without propellantclo...
example 3
Sulfonylureas
[0075]
mostAmountspreferred amountpreferred amountA. Glyburide lingual sprayglyburide0.25-25 0.5-20 0.75-15 ethanol 5-60−7.5-50 10-20propylene glycol 5-307.5-20 10-15polyethylene glycol 0-6030-4535-40water2.5-30 5-20 6-15flavors0.1-5 1-42-3B. Glyburide non-polar bite capsuleglyburide0.01-10 0.025-7.5 0.1-4 olive oil30-6035-5530-50polyoxyethylated oleic30-6035-5530-50glyceridesflavors0.1-5 1-42-3
PUM
| Property | Measurement | Unit |
|---|---|---|
| weight % | aaaaa | aaaaa |
| pressure | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com