Anthracycline bitriazole and copper fluoroborate complex with 4-methylbenzeneboronic acid catalyzing effect and preparation method of anthracycline bitriazole and copper fluoroborate complex
A technology of anthracycline bistriazole copper and copper complexes, which is applied in the field of inorganic synthesis, can solve the problems of expensive palladium catalysts, and achieve the effects of being suitable for large-scale production, low production cost, and high purity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
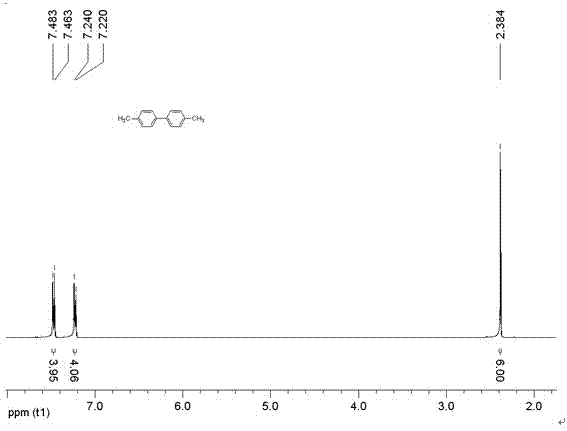
[0028] Preparation of 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole (tatrz) ligand
[0029] The preparation method of 1-[9-(1H-1,2,4-triazol-1-yl)anthracene-10-yl]-1H-1,2,4-triazole of the present invention is characterized in In the "one-pot method", the organic compound is prepared by heating 9,10-dibromoanthracene, triazole, potassium carbonate and copper oxide in a polar solvent; wherein 9,10-dibromoanthracene: three Azole: potassium carbonate: the molar ratio of copper oxide is 2:10:30:1;
[0030]
[0031] In the present invention, the molar ratio of 9,10-dibromoanthracene (I): triazole (II): potassium carbonate: copper oxide is preferably 2:10:30:1; the reaction temperature is 120° C., and the reaction time is 150 hours. The polar solvent described in the present invention is DMF.
Embodiment 2
[0033] Cu(BF 4 ) 2 and 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole (tatrz) in a molar ratio of 1: 1;
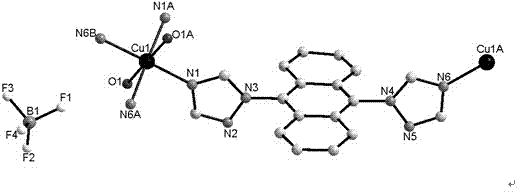
[0034] tatrz (0.0624 g, 0.2 mmol) and Cu(BF 4 ) 2 (0.0691 g, 0.2 mmol) in H 2 O (6 mL) and CH 3 CN (4 mL) was stirred in a mixed solvent at room temperature for half an hour and then filtered, and the filtrate was volatilized at room temperature to form yellow rod-shaped crystals analyzed by X-ray single crystal diffraction. Yield: 35% (calculated based on tatrz). Elemental analysis (C 36 h 28 B 2 CuF 8 N 12 o 2 ) Theoretical value (%): C, 48.16; H, 3.14; N, 18.72. Found: C, 48.15; H, 3.16; N, 18.69.
[0035] Example 2
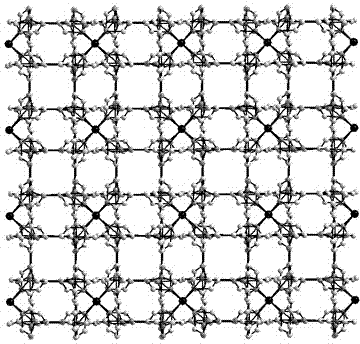
[0036] The crystal structure was determined using an APEX II CCD single crystal diffractometer, using graphite monochromatized Mokα rays (λ = 0.71073 ?) as the incident radiation, and ω -2 θ Diffraction points are collected by scanning, and the unit cell parameters are obtained by least square method correction. The ...
Embodiment 3
[0040] Cu(BF 4 ) 2 and 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole (tatrz) in a molar ratio of 1: 1;
[0041] We also tried other ratios such as Cu(BF 4 ) 2 and 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole (tatrz) in a molar ratio of 2: 1, no matter how long the hydrothermal reaction time is, no crystalline compound can be obtained. Therefore Cu(BF 4 ) 2 and 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole (tatrz) in a molar ratio of 1: 1 is the best response ratio.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com