Levo-ulifloxacin injection and preparation method thereof
A technology for injections and water for injection, which is applied in pharmaceutical formulations, medical preparations containing active ingredients, and drug delivery to achieve the effects of fast absorption, small poisoning effect, and reliable effect.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0052] The preparation of embodiment 1 (S)-(-)-ulifloxacin
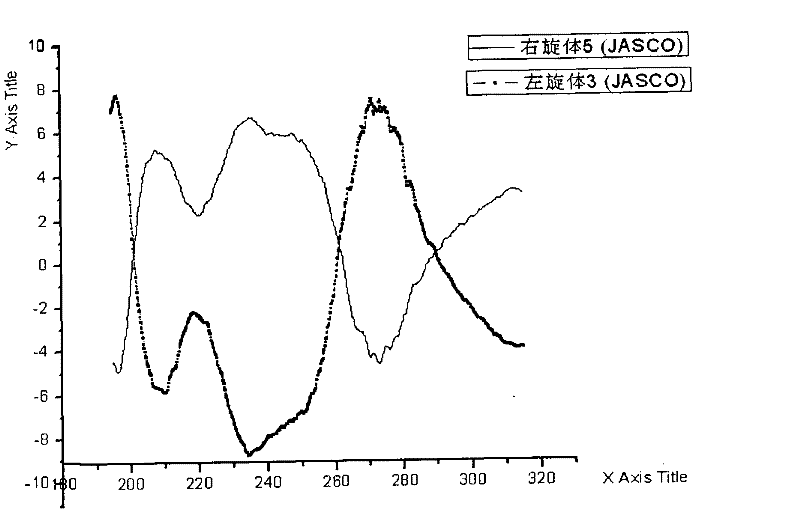
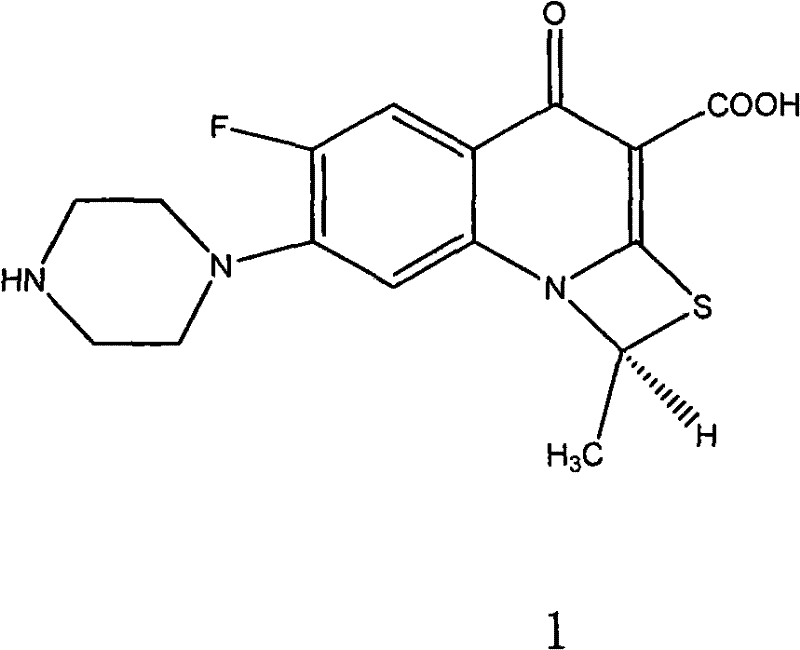
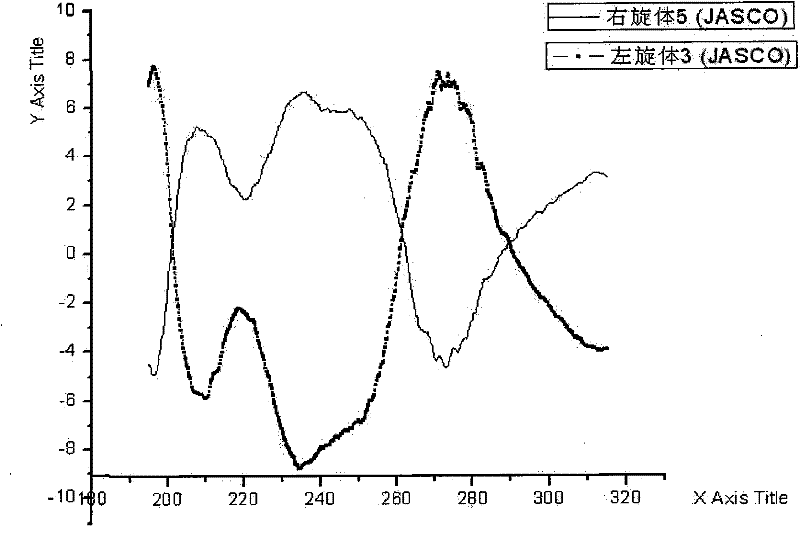
[0053] Dissolve 105 g of racemic ulifloxacin in 1500 mL of dimethyl sulfoxide, add dropwise a solution of 27 g of D-tartaric acid dissolved in 405 mL of dimethyl sulfoxide under stirring, stir at room temperature for 20 hours, precipitate and filter, and obtain The solid was dried under vacuum to obtain 86 grams, and the solid was recrystallized in dimethyl sulfoxide to obtain 37 grams of levofloxacin-D-tartrate, which was analyzed by element C49.08%, H5.06%, N9. 50%, S7.44% (molecular composition: C 16 h 16 FN 3 o 3 S 1 / 2C 4 h 6 o 6 ·H 2 O, calculated value C48.86%, H4.78, N9.50%, S7.25%); add this salt into water to form a suspension, adjust the pH value to 7-8 with 2% NaOH aqueous solution under stirring, precipitate and filter Dry to obtain (S)-(-)-ulifloxacin 24.5 grams, its chemical name: (S)-(-)-6-fluoro-1-methyl-4-oxo-7-(1-piperazine Base)-1H, 4H-[1,3]thiazetidino[3,2-a]quinoline-3-carboxylic acid
...
Embodiment 2
[0059] Example 2 Preparation of 0.1 mg / ml levofloxacin glucose infusion
[0060] Take 1 gram of levo-ulifloxacin, add it to a 20-liter batching bucket, add 7 L of water, add about 5.5 milliliters of 5% (W / W) lactic acid solution under stirring, stir until it dissolves, and measure the pH to 5.56, add 800 grams of glucose, Stir to dissolve, add activated carbon, heat up, keep warm and stir at about 50°C for 30 minutes, cool to 30°C, filter for decarburization, add water for injection to 10L, then filter through a 0.22μm filter membrane, fill and cap , sterilized at 110°C for 30 minutes.
[0061] The infusion solution prepared in this example can be filled into specifications of 25mg:250ml; 50mg:500ml.
Embodiment 3
[0062] Example 3 Preparation of 0.1mg / ml levofloxacin sodium chloride infusion
[0063] Take 1 gram of levofloxacin, add it to a 20-liter batching bucket, add 1000 ml of water, add about 5.5 ml of 5% (W / W) lactic acid solution under stirring, stir until it dissolves, add 90 grams of sodium chloride, and stir to dissolve. Add activated carbon, heat up to 50°C, heat and stir for 30 minutes, cool to 30°C, filter for decarburization, add water for injection to 10L, then filter through a 0.22μm filter membrane, fill, cap, and extinguish at 110°C Bacteria for 30 minutes.
[0064] The infusion solution prepared in this example can be filled into specifications of 25mg:250ml; 50mg:500ml.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com