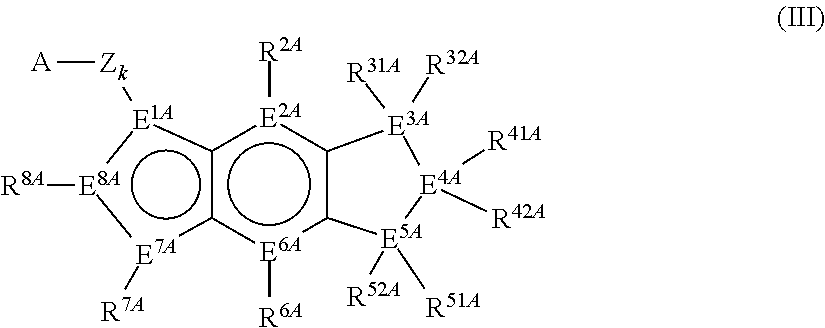
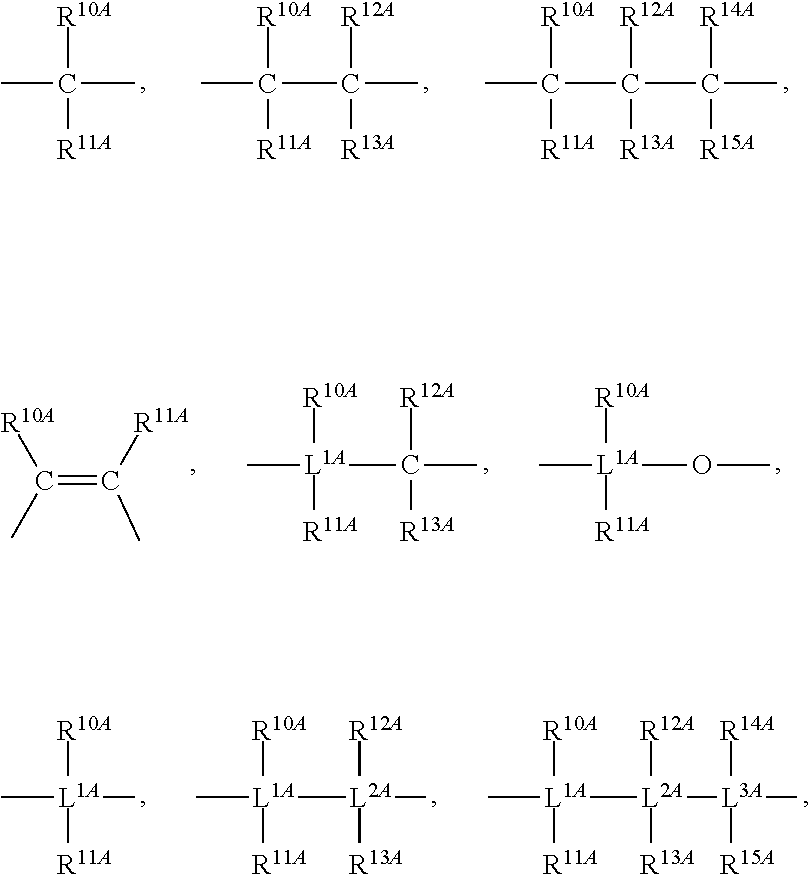Mono-hydroindacenyl complexes
a technology of monohydroindacenyl complexes and complexes, which is applied in the field of monohydroindacenyl complexes, can solve the problems of unfavorable blending, unfavorable blending, and high percentage of comonomers, and achieves the effect of high productivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Synthesis of [η5-1-(2-methylpyridine)-2-methyl-3-benzyl-1,5,6,7-tetrahydro-s-indacen-1-yl]dichlorochromium (Compound A)
[0176]
1.1 Preparation of 2-methyl-3,5,6,7-tetrahydro-s-indacen-1(2H)-one
[0177]
[0178]Methacryloyl chloride (50 ml, 0.5 mol) was added to a suspension of 133.5 g (1 mol) AlCl3 in 500 ml CH2Cl2 at −78° C. and stirred for 20 min. Then 59 g (0.5 mol) indane was added at the same temperature. The mixture was allowed to warm to room temperature and then was stirred overnight. Next day the mixture obtained was poured carefully into a mixture of ice (1000 g) and HCl (200 ml). The organic phase was separated, washed with water and 5% NaHCO3, and dried over MgSO4. Solvent was evaporated and residue was distilled in vacuum giving 77.6 g of product (83%), b.p. 118-120° C. / 0.5 torr.
[0179]NMR 1H (CDCl3): 7.59 (s, 1H); 7.28 (s, 1H); 3.34 (dd, 1H); 2.92 (m, 4H); 2.80-2.65 (group of signals, 2H); 2.13 (m, 2H); 1.42 (d, 3H).
[0180]13C 208.90, 152.82, 152.45, 143.96, 134.91, 121.85, 199...
example 2
Synthesis of [η5-1-(2-methylpyridine)-3-benzyl-1,5,6,7-tetrahydro-s-indacen-1-yl]dichlorochromium (Compound B)
2.1. Preparation of 3,5,6,7-tetrahydro-s-indacen-1(2H)-one
[0188]
[0189]The mixture of 11.8 g indane (0.1 mol) and 10 ml 3-chloropropionyl chloride (0.12 mol) was added to suspension of 16 g AlCl3 (0.55 mol) in 100 ml CH2Cl2 at 0° C. After then reaction mixture was poured into ice and HCl, the organic layer was isolated; the aqueous one was extracted with CH2Cl2 (2*50 ml). The combined organic phase was washed by NaHCO3, dried over MgSO4 and evaporated. The residue was added carefully to conc. H2SO4 (100 ml), warmed to 70° C. and stirred after then 30 min at the same temperature. The mixture was cooled and poured into ice. Precipitate was filtered, washed by water and dried, to give 12.4 g of product (72%).
2.2 Preparation of 1,2,3,5-tetrahydro-s-indacene
[0190]
[0191]The mixture of 3,5,6,7-tetrahydro-s-indacen-1(2H)-one (12.4 g, 0.072 mol), p-toluenesulfonyl hydrazide (13.4 g, 0...
example 3
3.1 Preparation of Catalytic Solution
[0193]A solution of 46.0 mg of [η5−-1-(2-methylpyridine)-2-methyl-3-benzyl-1,5,6,7-tetrahydro-s-indacen -1-yl]dichlorochromium (Compound A) in 15 ml cyclohexane and 4.0 ml PMAO (7% solution from Akzo Nobel) were mixed. The obtained suspension was stirred for 15 minutes at room temperature. The obtained suspension (30.5 ml) had red brown colour and the concentration was 3.19 μmol [η5−-1-(2-methylpyridine)-2-methyl-3-benzyl-1,5,6,7-tetrahydro-s-indacen-1-yl]dichlorochromium / ml.
3.2 Polymerisation
[0194]Under argon a 3.1 l-steelautoclave was filled with 450 ml cyclohexane at room temperature and with 1300 ml butane at 5° C. The temperature was raised up to 80° C. with a speed frequency of 350° C. / h. By addition of 7.8 l of ethylene the recruit pressure was raised to 4 bar. Then, 300 mg Triisobutylaluminium (20% TIBA in cyclohexene) were added. After 5 minutes of stirring the 61.9 μmol of catalyst solution as obtained in step 3.1 were added and the cat...
PUM
| Property | Measurement | Unit |
|---|---|---|
| glass transition temperature | aaaaa | aaaaa |
| density | aaaaa | aaaaa |
| polydispersity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com