Treating benign prostate hyperplasia with SARMS
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Interaction Between Compound VI and Human 5α-Reductase
[0271]
[0272]Testosterone can be reduced by the enzyme 5α-reductase to dihydrotestosterone (DHT). DHT binds with five-fold greater affinity to the human androgen receptor and is thought to be the mediator of androgen effects in many tissues. Since Compound VI mimics the effects of testosterone in many in vitro and in vivo systems, Compound VI was tested to determine whether it interacts with 5α-reductase. This study (1) determined if Compound VI is a substrate for 5α-reductase, and (2) determined if Compound VI has any effects on the conversion of testosterone to DHT via 5α-reductase.
[0273]Methods: COS1 (American Type Culture Collection, Manassas, Va.) cells were plated in twelve-well plates at a density of 60,000 cells / well and transiently transfected with expression vectors for human 5α-reductase (obtained from Dr. David W. Russell, Southwestern Medical Center, Dallas, Tex.). LipofectAMINE PLUSTM Reagent (Invitrogen, Carlsbad, C...
example 2
Pharmacologic Activity and Tissue Selectivity of Compound VI in Rats of Varying Hormonal Status
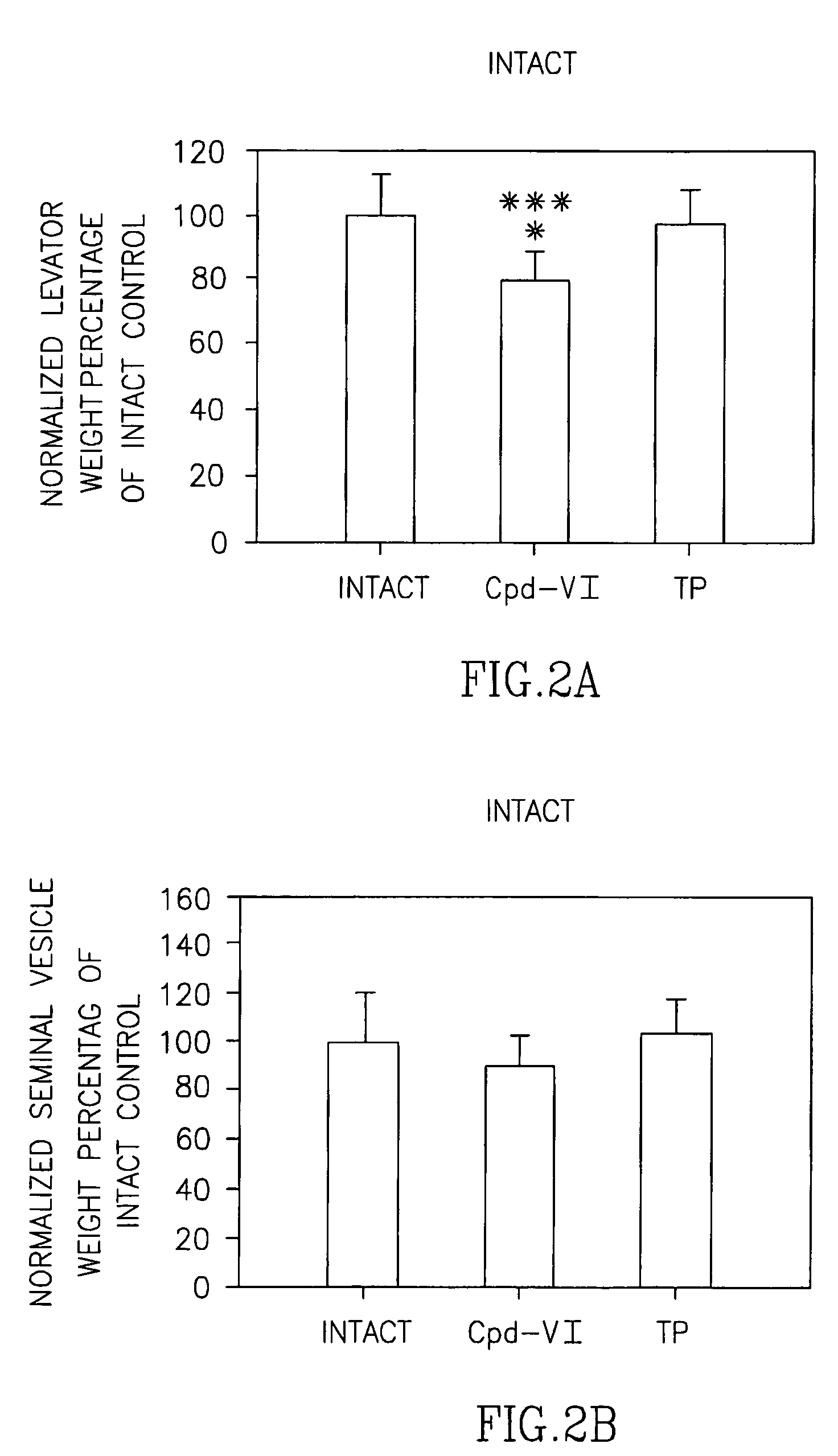
[0276]Selective androgen receptor modulators (SARMs) have a wide variety of potential therapeutic applications, including male hypogonadism, osteoporosis, muscle-wasting diseases, sexual libido and contraception. Previous studies by Applicants demonstrated that Compound VI is a potent and efficacious selective androgen receptor modulator (SARM) in castrated male rats. Applicants completed a preclinical study to compare the pharmacologic effects and tissue-selectivity of Compound VI and testosterone propionate (TP) in male rats of varying hormonal status. Male rats with normal testicular function (i.e., intact with no surgical manipulation) were included to examine the effects of Compound VI on animals with normal blood levels of testosterone. Male rats that received unilateral orchidectomy (i.e., surgical removal of one testis) were included to examine the effects of Compound VI on animals...
example 3
Pharmacologic Activity and Tissue-Selectivity of Compound II, Hydroxy-flutamide and Finasteride in Intact Male Rats
[0288]Compound II is a selective androgen receptor modulator (SARM) in castrated male rats. It behaved as an agonist in anabolic tissue while a partial agonist in androgenic tissue. When it's administered to intact male rats at the dose rate of 0.5 mg / day, Compound II significantly decreased the prostate weight to 63% of that observed in vehicle-treated intact animals without affecting the levator ani muscle weight. The tissue selectivity Compound II demonstrated in intact male rats could be explained by two possible mechanisms; 1) in the presence of endogenous testosterone, Compound II simply behaved as a partial agonist in DHT-dependent androgenic tissue; 2) Compound II is also a 5α-reductase inhibitor besides its partial agonist activity in androgenic tissues.
[0289]Methods: Male Sprague-Dawley rats were purchased from Harlan Biosciences (Indianapolis, Ind.). The anim...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com