Identification of channelrhodopsin-2 (CHR2) mutations and methods of use
a technology of channelrhodopsin and chr2 is applied in the field of molecular biology, which can solve the problems of reduced visual acuity, reduced photoreceptor loss or degeneration, and reduced photoreceptor function, so as to improve photocurrent, reduce threshold light intensity, and high temporal resolution
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
n of Labeled Mutant Chop2 Constructs
[0113]Mutations were made on a codon optimized Chop2-GFP fusion protein to create single and double mutations at the L132 (Leucine 132) and T159 (Threonine 159) sites. Several mutants were generated, for example, single mutants such as L132A, L132C, T159A, T159C, and T 159S, and double mutants such as L132C / T159C, L132C / T159S, L132A / T159C, and L132C / T159A. Chop2-GFP transgenes were cloned into a rAAV vector under the control of a CAG promoter using methods known in the art.
example 2
Analysis of Mutant Chop2 Constructs
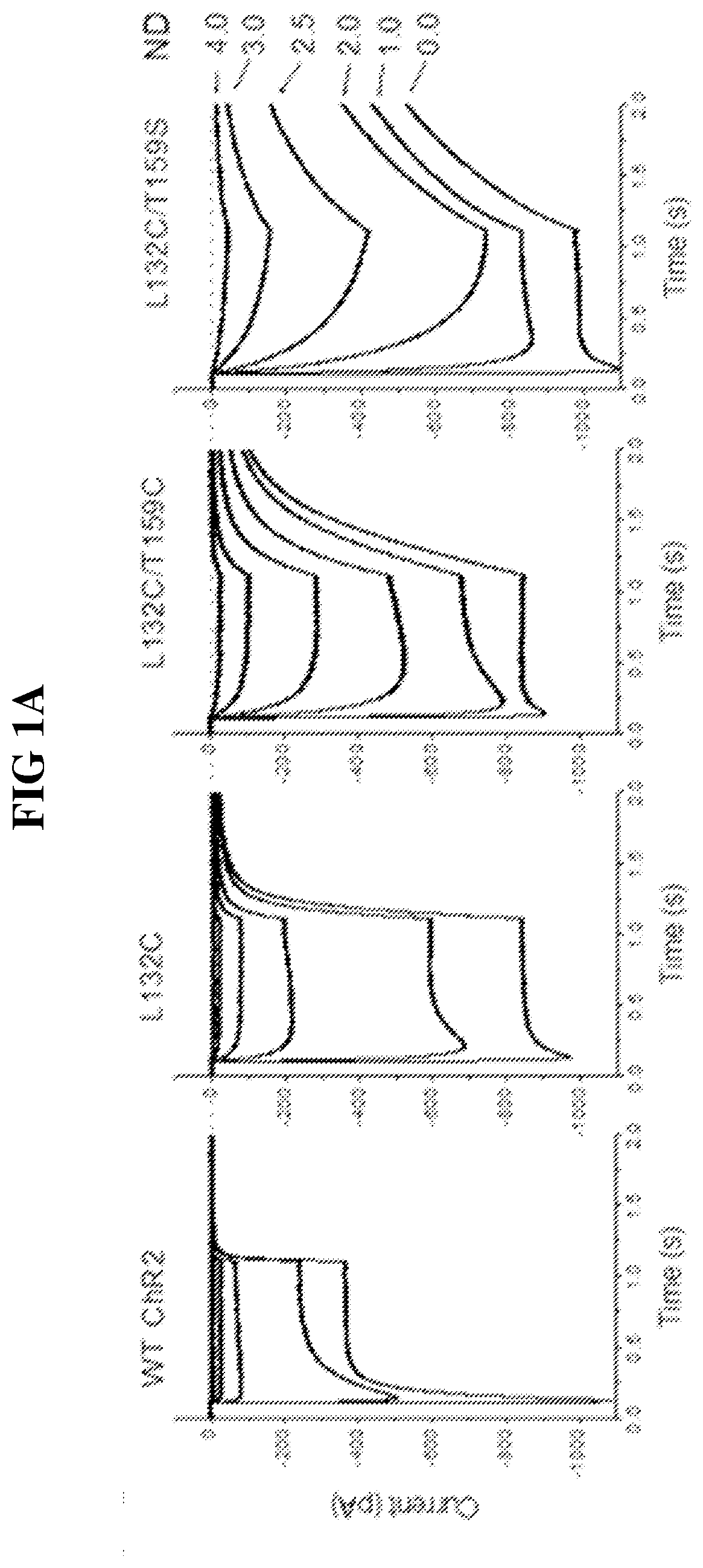
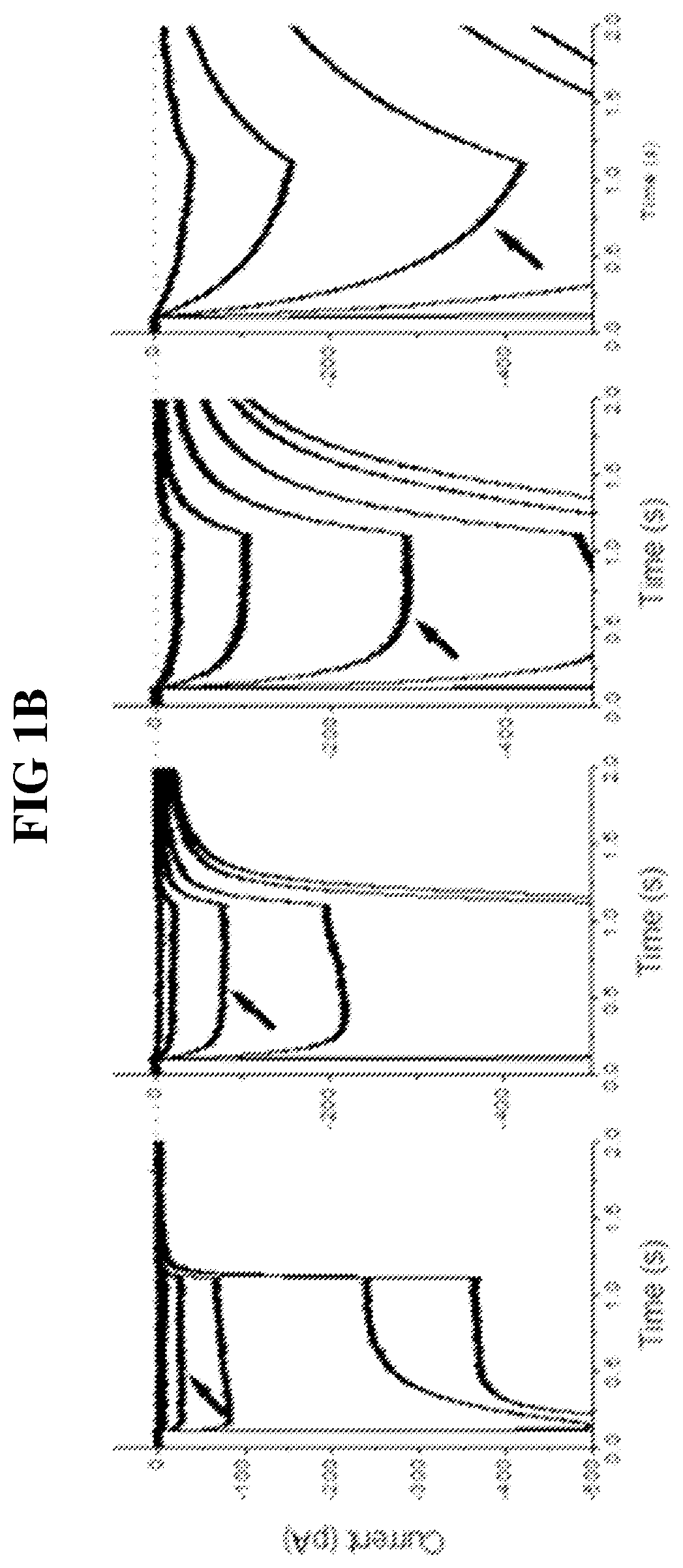
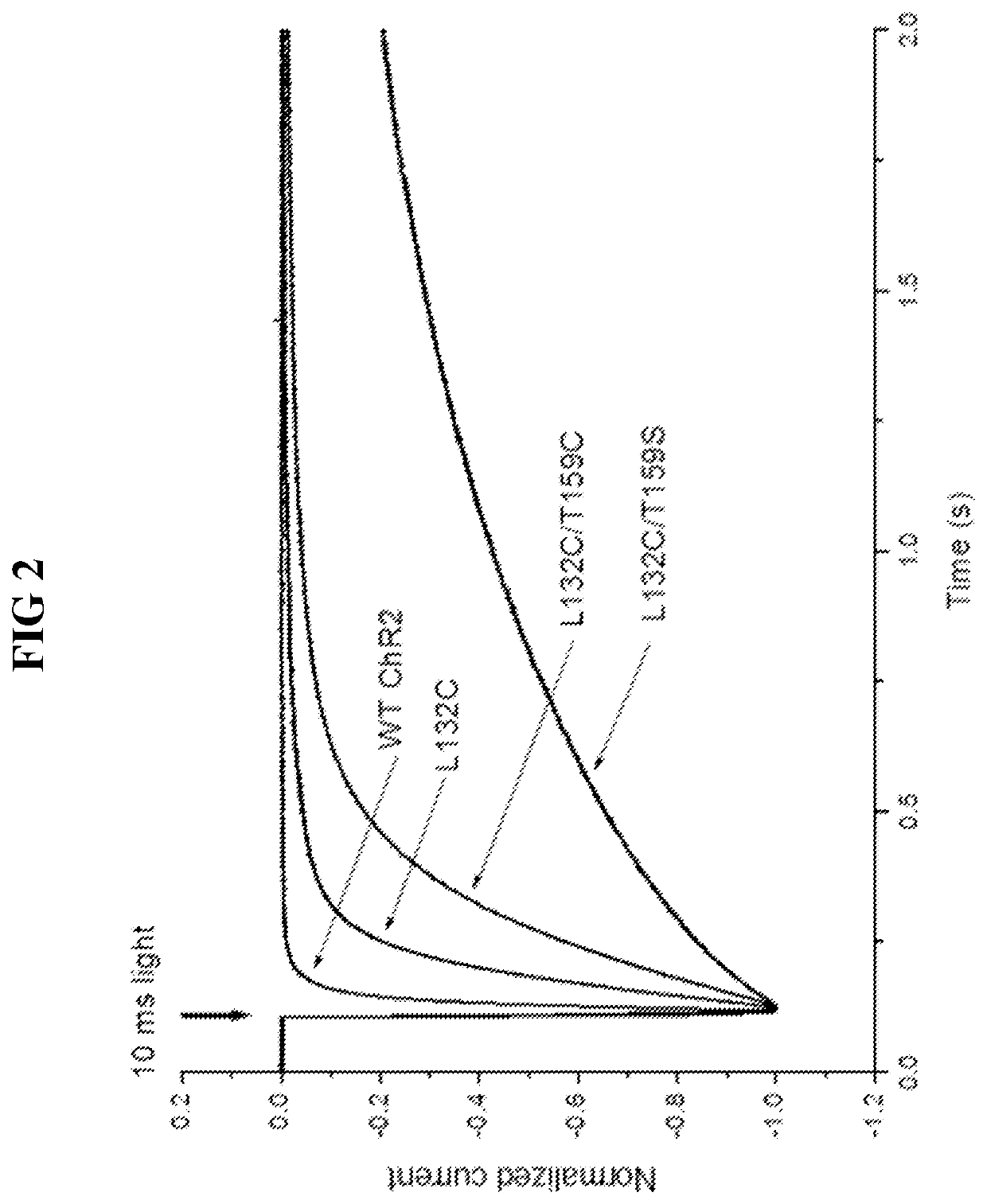
[0114]The functional properties of each mutant Chop2, or a combination thereof, were first examined in HEK cells. Chop2 constructs were delivered to HEK cells by adenoviral infection, for example. Upon expression of the WT or mutant Chop2, functional WT and mutant ChR2 channels were formed. Measurements of the light sensitivity and other properties of the ChR2 channels were assessed as described herein. The light stimuli (photons / cm2·s at 460 nm) were generated by a xenon arc lamp and attenuated by neutral density filters: ND4.0 (2.8×1014), ND3.0 (1.4×1015), ND2.5 (4.8×1015); ND2.0 (1.6×1016), ND1.0 (1.3×1017), ND0 (1.2×1018). Light evoked currents were measured from wild-type ChR2, T159C, L132C, L132C / T159C, and L132C / T159S. Patch clamp recordings were performed using methods known in the art.
[0115]Representative recordings from this experiment comparing light sensitivity between the Chop2 constructs demonstrated that mutations at L132 alone or in...
example 3
cular Administration and Analysis of Mutant Chop2 Constructs
[0117]AAV2 virus vectors carrying mutant Chop2-GFP constructs driven by CAG promoter were made and injected intravitreally into the eyes of C57BL / 6J adult mice. Adult mice were anesthetized by IP injection of ketamine (100 mg / kg) and xylazine (10 mg / kg). Under a dissecting microscope, an incision was made by scissors through the eyelid to expose the sclera. A small perforation was made in the sclera region posterior to the lens with a needle and viral vector suspension of 0.8-1.5 μl at the concentration of approximately 1011 genomic particles / ml was injected into intravitreal space through the hole with a Hamilton syringe with a 32-gauge blunt-ended needle. For each animal, usually only one eye was injected with viral vectors carrying a Chop2 construct, and the other eye was uninjected or injected with control viral vectors carrying GFP alone. Upon expression of the WT or mutant Chop2 of the present invention, functional WT...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com