Detection method of trans-1,3-dichloropropene in E-1-chloro-6, 6-dimethyl-2-heptenyl-4-yne
A technology of dichloropropene and a detection method is applied in the field of medicine and achieves the effects of good specificity, improved medication safety and simple operation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0040] 1 Instruments and materials
[0041] 1.1 Instrument: TRACE1300 Gas Chromatograph (Thermo Fisher);
[0042] 1.2 Reagent: methanol (chromatographic grade).
[0043] 2 Methods and results
[0044] 2.1 Chromatographic conditions
[0045] Chromatographic column: 6% cyanopropylphenyl 94% dimethylpolysiloxane column DB-624 (30m × 0.53mm, 3.0μm); column temperature: keep at 80°C for 2min, heat up to 10°C / min Keep at 220°C for 10min; inlet temperature: 250°C; carrier gas: high-purity nitrogen; flow rate: 10ml / min; split ratio: 5:1; injection volume: 1μl;
[0046] Detector: electron capture detector (ECD); detector temperature: 300°C.
[0047] 2.2 Solution preparation
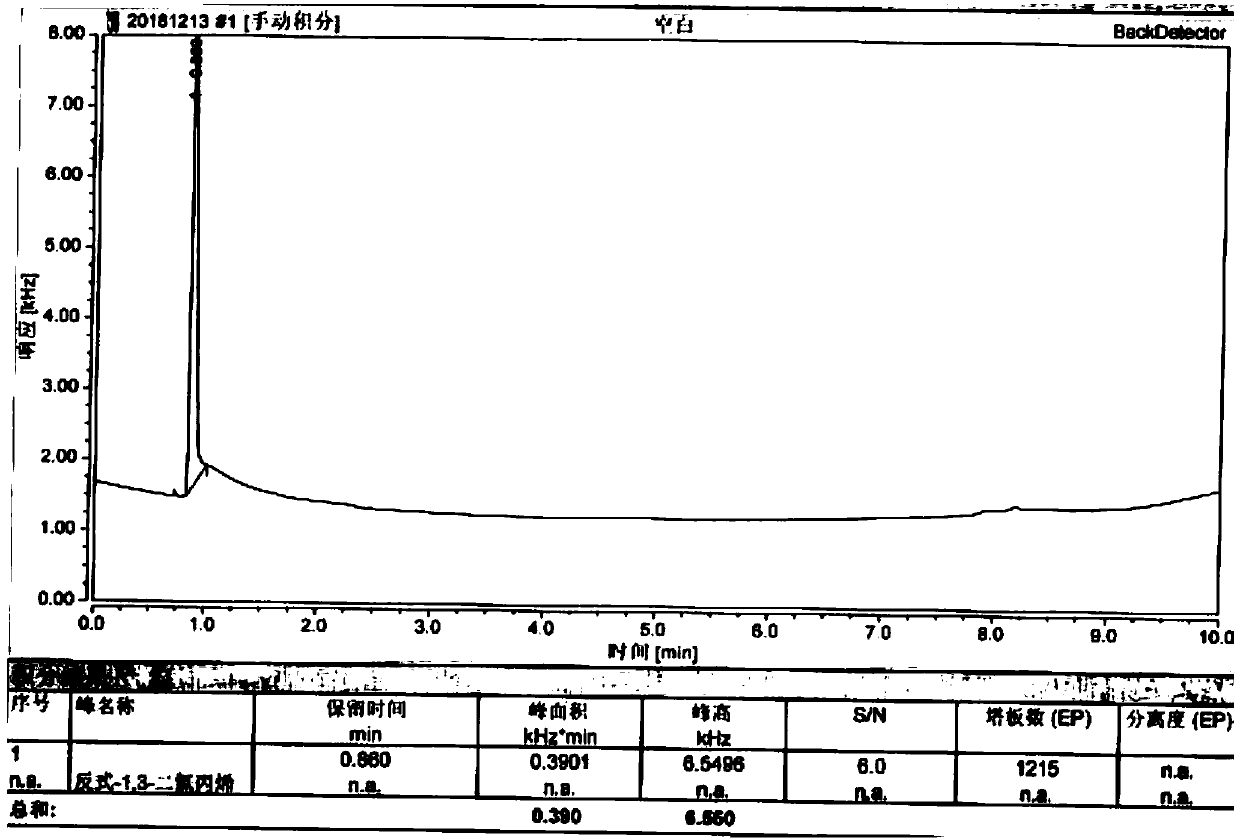
[0048] Blank solution: methanol.
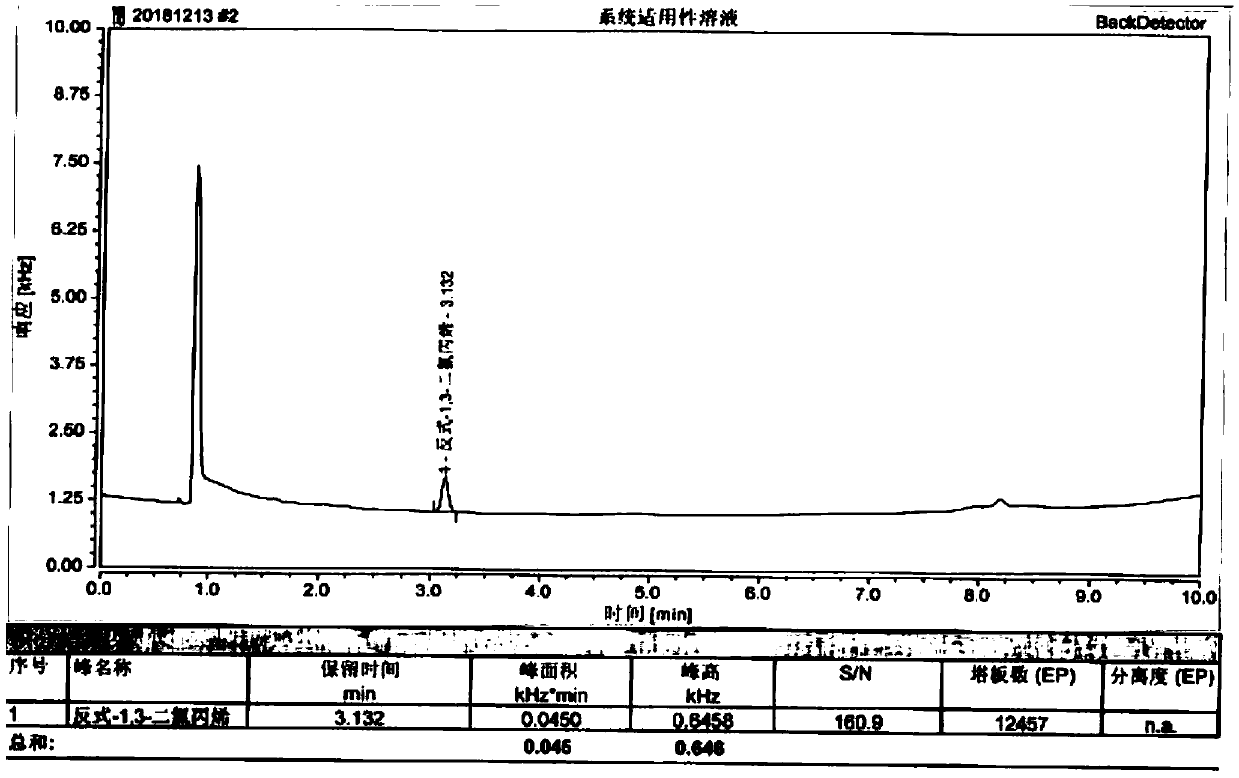
[0049] Control solution: Weigh 30.5mg of trans-1,3-dichloropropene reference substance, put it in a 100ml measuring bottle, add methanol to dilute to the mark, shake well; accurately measure 1ml, put it in a 100ml measuring bottle, add methanol to dilute to the mark , shake ...
Embodiment 2
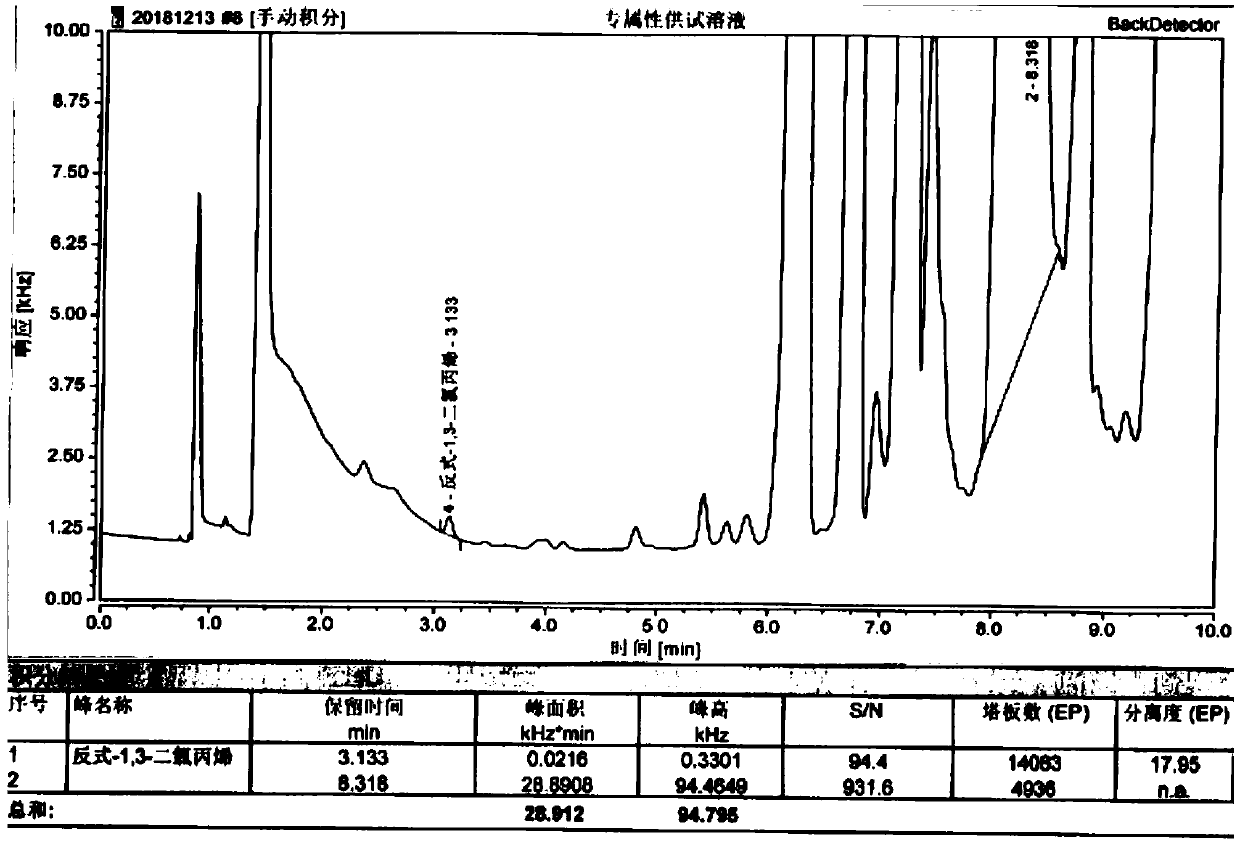
[0088] Embodiment 2: the detection of actual sample
[0089] Get three batches of (E)-1-chloro-6,6-dimethyl-2-heptene-4-yne samples, prepare contrast solution and need testing solution according to 2.1, 2.2 and 2.3 in example 1 and measure according to law, The result is as follows:
[0090] Table 7 Sample Test Results
[0091] batch number
[0092] .
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com