Method for preparing scandium fluoride nanocrystal and regulating crystalline phase at low temperature and normal pressure
A low temperature and atmospheric pressure, scandium fluoride technology, applied in chemical instruments and methods, nanotechnology, nanotechnology, etc., can solve problems such as difficult phase transitions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0018] 0.4mmol RECl 3 (ScCl 3 78mol%, YbCl 3 20mol%, ErCl 3 2mol%), 10mL oleic acid and 10mL octadecene were added to a three-necked flask, stirred with a magnet, and protected by nitrogen gas, heated to 160°C, reacted for 30min, cooled to room temperature, added 1.2mmol NH 4 F, slowly raise the temperature to 160°C, stir at a constant temperature for 30 minutes, then heat to 300°C, and react at a constant temperature for 1.5 hours. After the reaction is completed, add excess ethanol, centrifuge, and wash three times to obtain pure cubic scandium fluoride nanocrystals.
Embodiment 2
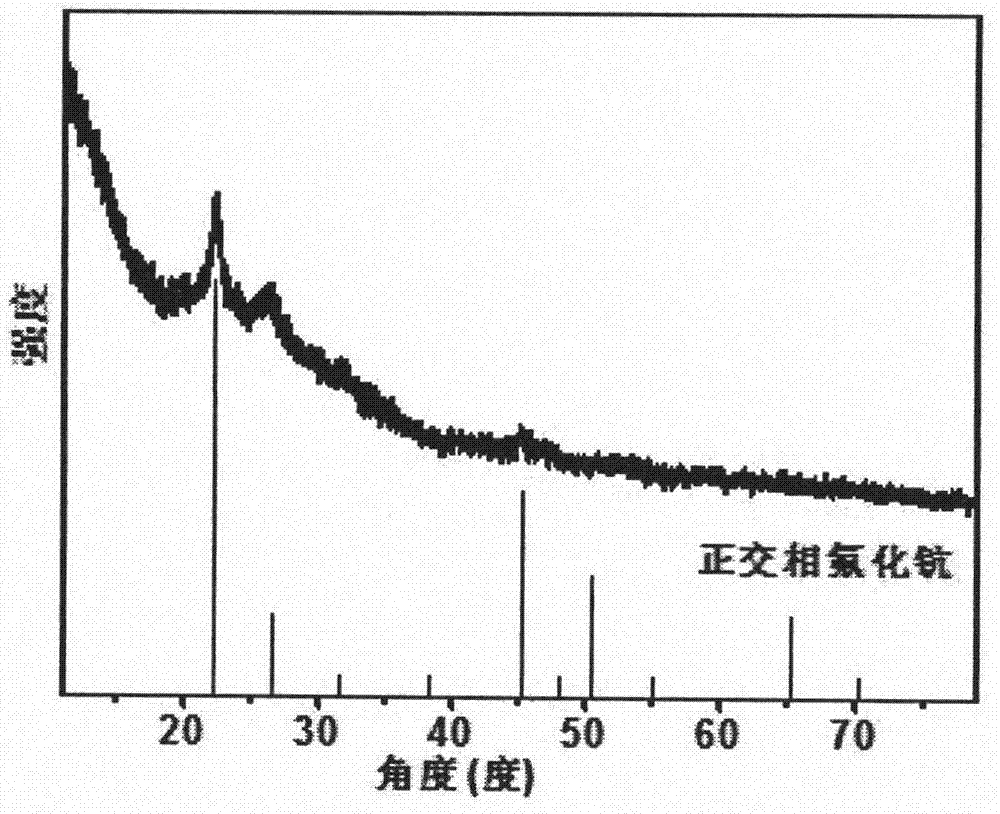
[0020] 0.4mmol RECl 3 (ScCl 3 78mol%, YbCl 3 20mol%, ErCl 3 2mol%), 10mL oleic acid and 10mL octadecene were added to a three-necked flask, stirred with a magnet, and protected by nitrogen gas, heated to 160°C, reacted for 30min, cooled to room temperature, added 1.2mmol NH 4 F. Slowly raise the temperature to 160°C, stir at constant temperature for 30min, and keep it at 160°C for 1.5h after the ammonium fluoride is completely dissolved. After the reaction is completed, add excess ethanol, centrifuge, and wash three times to obtain pure orthogonal scandium nanocrystals.
Embodiment 3
[0022] 0.4mmol RECl 3 (ScCl 3 78mol%, YbCl 3 20mol%, ErCl 3 2mol%), 10mL oleic acid and 10mL octadecene were added to a three-necked flask, stirred with a magnet, and protected by nitrogen gas, heated to 160°C, reacted for 30min, cooled to room temperature, added 1.2mmol NH 4 F, slowly raise the temperature to 160°C, stir for 30 minutes, and keep it at 200°C for 1.5 hours after the ammonium fluoride is completely dissolved. After the reaction is completed, add excess ethanol, centrifuge, and wash three times to obtain pure orthogonal phase Scandium nanocrystals.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com