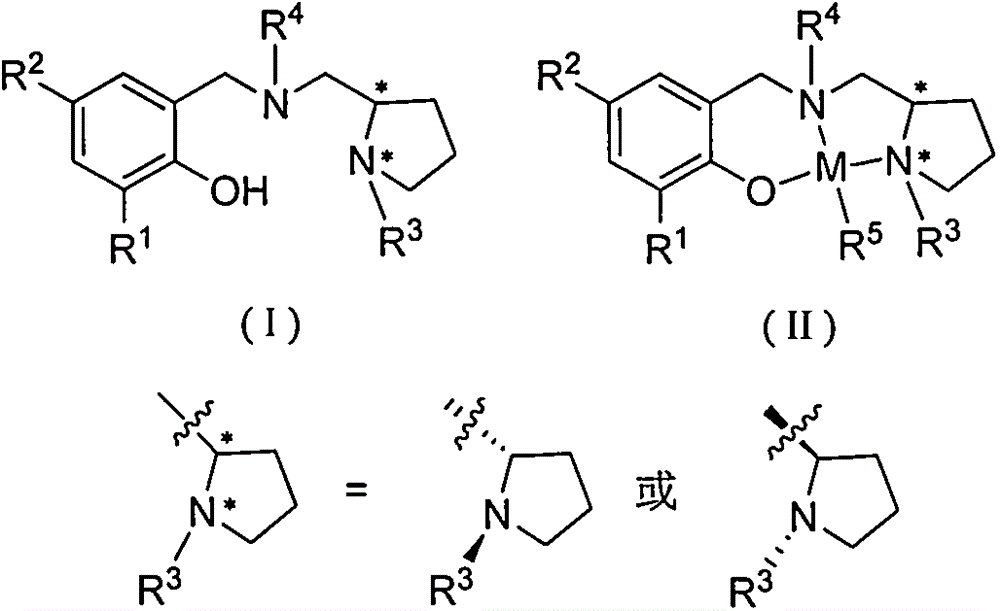
Chiral amino phenoxyzinc and magnesium compounds and their preparation method and application
A technology of amino phenoxyzinc and magnesium compounds, applied in the field of magnesium compounds and chiral amino phenoxyzinc, can solve the problems of low selectivity, high heterotypic selectivity and the like
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0046] Synthesis of Ligand L1
[0047] Add 1.22 g of benzaldehyde, 30 mL of absolute ethanol, and 1.28 g of (S)-1-ethyl-2-aminomethyltetrahydropyrrole into a 100 mL eggplant-shaped bottle, and heat to reflux for 24 hours. Add 0.76 g of sodium borohydride, stir for 3 hours, pour into water, extract the organic phase with dichloromethane, dry over anhydrous magnesium sulfate, and remove the solvent to obtain a light yellow viscous liquid. Add 30 mL of absolute ethanol, 0.6 g of paraformaldehyde, and 1.63 g of 2,4-dichlorophenol, and heat to reflux for 12 hours. The crude product was separated by column chromatography on silica gel to obtain red liquid L1 (2.32 g, 59.0%).
[0048]
[0049] 1 HNMR (400MHz, CDCl 3 ): δ7.22(m, 6H), 6.83(d, 4 J=2.4Hz), 3.84(d, 2 J=14.0Hz, 1H), 3.65(d, 2 J=13.0Hz, 1H), 3.53(d, 2 J=14.0Hz, 1H), 3.39(d, 2 J=13.0Hz, 1H), 3.06(m, 1H), 2.80-2.70(m, 1H), 2.60-2.50(m, 2H), 2.60-2.50(m, 1H), 2.21-2.08(m, 2H) , 2.01-1.94(m, 1H), 1.71-1.63(m, 2H), 1...
Embodiment 2
[0051] Synthesis of Ligand L2
[0052] Add 1.22 g of benzaldehyde, 30 mL of absolute ethanol, and 1.28 g of (S)-1-ethyl-2-aminomethyltetrahydropyrrole into a 100 mL eggplant-shaped bottle, and heat to reflux for 24 hours. Add 0.76 g of sodium borohydride, stir for 3 hours, pour into water, extract the organic phase with dichloromethane, dry over anhydrous magnesium sulfate, and remove the solvent to obtain a light yellow viscous liquid. Add 30 mL of absolute ethanol, 0.6 g of paraformaldehyde, and 2.06 g of 2,4-di-tert-butylphenol, and heat to reflux for 12 hours. The crude product was separated by column chromatography on silica gel to obtain a colorless transparent liquid L2 (1.95 g, 44.7%).
[0053]
[0054] 1 HNMR (400MHz, CDCl 3 ): δ10.55(s, 1H), 7.35-7.23(m, 5H), 7.18(s, 1H), 6.85(s, 1H), 3.97(d, J=13.4Hz, 1H), 3.75(d, J=13.0Hz, 1H), 3.51(d, 2 J=13.4Hz, 1H), 3.37(d, 2J=13.0Hz, 1H), 3.09-3.02(m, 1H), 2.71-2.63(m, 1H), 2.50-2.38(m, 3H), 2.11-2.01(m, 1H), 2.03-1.88...
Embodiment 3
[0056] Synthesis of Ligand L3
[0057] Add 1.22 g of benzaldehyde, 30 mL of absolute ethanol, and 1.28 g of (S)-1-ethyl-2-aminomethyltetrahydropyrrole into a 100 mL eggplant-shaped bottle, and heat to reflux for 24 hours. Add 0.76 g of sodium borohydride, stir for 3 hours, cool to room temperature, pour into water, extract the organic phase with dichloromethane, dry over anhydrous magnesium sulfate, and remove the solvent to obtain a light yellow viscous liquid. Add 30 mL of absolute ethanol, 0.6 g of paraformaldehyde, and 3.30 g of 2,4-dicumylphenol, and heat to reflux for 12 hours. The crude product was separated by column chromatography on silica gel to obtain light brown liquid L3 (3.41 g, 60.9%).
[0058]
[0059] 1 HNMR (400MHz, CDCl 3 ): δ7.16(d, J=12.6Hz, 17H), 6.66(d, J=1.8Hz, 1H), 3.80(d, 2 J=13.6Hz, 1H), 3.46(d, 2 J=13.8Hz, 1H), 3.33(d, 2 J=13.6Hz, 1H), 3.13(d, 2 J=12.8Hz), 2.97-2.86(m, 1H), 2.55(dq, J=14.7, 7.3Hz, 1H), 2.28(dt, J=15.7, 7.9Hz, 3H), 2.01-1....
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight distribution | aaaaa | aaaaa |
| molecular weight distribution | aaaaa | aaaaa |
| molecular weight distribution | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com